Recent from talks
Nothing was collected or created yet.
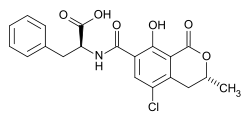
Ochratoxin A
View on Wikipedia
 | |
 | |
| Names | |
|---|---|
| IUPAC name
N-[(3R)-5-Chloro-8-hydroxy-3-methyl-1-oxo-3,4-dihydro-1H-2-benzopyran-7-carbonyl]-L-phenylalanine
| |
| Systematic IUPAC name
(2S)-2-[(3R)-5-Chloro-8-hydroxy-3-methyl-1-oxo-3,4-dihydro-1H-2-benzopyran-7-carboxamido]-3-phenylpropanoic acid | |
| Other names
(R)-N- [(5-Chloro- 3,4-dihydro- 8-hydroxy- 3-methyl- 1-oxo- 1H-2-benzopyran-7-yl) -carbonyl]- L- phenylalanine
(−)-N- [(5-Chloro- 8-hydroxy- 3-methyl- 1-oxo- 7-isochromanyl) carbonyl]- 3-phenylalanine | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.005.586 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
| UN number | 2811 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C20H18ClNO6 | |
| Molar mass | 403.813 |
| Melting point | 169 °C (336 °F; 442 K) |
| Hazards | |
| GHS labelling:[1] | |
  
| |
| Danger | |
| H300, H351, H413 | |
| P203, P260, P264, P264+P265, P270, P271, P273, P280, P284, P301+P316, P304+P340, P305+P351+P338, P316, P318, P320, P321, P330, P337+P317, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Ochratoxin A—a toxin produced by different Aspergillus and Penicillium species—is one of the most-abundant food-contaminating mycotoxins.[1] It is also a frequent contaminant of water-damaged houses and of heating ducts.[2][3] Human exposure can occur through consumption of contaminated food products, particularly contaminated grain and pork products, as well as coffee, wine grapes, and dried grapes.[4][5][6] The toxin has been found in the tissues and organs of animals, including human blood and breast milk.[7] Ochratoxin A, like most toxic substances, has large species- and sex-specific toxicological differences.[5]
Impact on human and animal health
[edit]Carcinogenicity
[edit]Ochratoxin A is potentially carcinogenic to humans (Group 2B), and has been shown to be weakly mutagenic, possibly by induction of oxidative DNA damage.[8]
The evidence in experimental animals is sufficient to indicate carcinogenicity of ochratoxin A. It was tested for carcinogenicity by oral administration in mice and rats. It slightly increased the incidence of hepatocellular carcinomas in mice of each sex.[9] and produced renal adenomas and carcinomas in male mice and in rats (carcinomas in 46% of males and 5% of females).[10] In humans, very little histology data is available, so a relationship between ochratoxin A and renal cell carcinoma has not been found. However, the incidence of transitional cell (urothelial) urinary cancers seems abnormally high in Balkan endemic nephropathy patients, especially for the upper urinary tract.[11] The molecular mechanism of ochratoxin A carcinogenicity has been under debate due to conflicting literature, however this mycotoxin has been proposed to play a major role in reducing antioxidant defenses.[12]
Neurotoxicity
[edit]Ochratoxin A has a strong affinity for the brain, especially the cerebellum (Purkinje cells), ventral mesencephalon, and hippocampal structures.[13] The affinity for the hippocampus could be relevant to the pathogenesis of Alzheimer's disease, and subchronic administration to rodents induces hippocampal neurodegeneration. Ochratoxin causes acute depletion of striatal dopamine, which constitutes the bed of Parkinson's disease, but it did not cause cell death in any of brain regions examined.[14] Teams from Zheijiang Univ. and Kiel Univ. hold that ochratoxin may contribute to Alzheimer's and to Parkinson's diseases. Nonetheless, their study was performed in vitro and may not extrapolate to humans.[15] The developing brain is very susceptible to ochratoxin, hence the need for caution during pregnancy.[16]
Immunosuppression and immunotoxicity
[edit]Ochratoxin A can cause immunosuppression and immunotoxicity in animals. The toxin's immunosuppressant activity in animals may include depressed antibody responses, reduced size of immune organs (such as the thymus, spleen, and lymph nodes), changes in immune cell number and function, and altered cytokine production. Immunotoxicity probably results from cell death following apoptosis and necrosis, in combination with slow replacement of affected immune cells due to inhibition of protein synthesis.[1]
Potential link to nephropathies
[edit]Balkan endemic nephropathy (BEN), a slowly progressive renal disease, appeared in the middle of the 20th century, highly localized around the Danube, but only hitting certain households. Patients over the years develop kidney failure that requires dialysis or transplantation. The initial symptoms are those of a tubulointerstitial nephritis of the sort met with after toxic aggressions to the proximal convoluted tubules. Such proximal tubule nephropathies can be induced by aluminium (e.g. in antiperspirants), antibiotics (vancomycin, aminosides), tenofovir (for AIDS), and cisplatin[citation needed]. Their symptoms are well known to nephrologists: glycosuria without hyperglycemia, microalbuminuria, poor urine concentration capacity, impaired urine acidification, and yet long-lasting normal creatinine clearance.[17] In BEN, renal biopsy shows acellular interstitial fibrosis, tubular atrophy, and karyomegaly in proximal convoluted tubules.[18] A number of descriptive studies have suggested a correlation between exposure to ochratoxin A and BEN, and have found a correlation between its geographical distribution and a high incidence of, and mortality from, urothelial urinary tract tumours.[19] However, insufficient information is currently available to conclusively link ochratoxin A to BEN.[20] The toxin may require synergistic interactions with predisposing genotypes or other environmental toxicants to induce this nephropathy.[21] Ochratoxin possibly is not the cause of this nephropathy, and many authors are in favor of aristolochic acid, that is contained in a plant: birthwort (Aristolochia clematitis). Nevertheless, although many of the pieces of scientific evidence are lacking and/or need serious re-evaluation, it remains that ochratoxin, in pigs, demonstrates direct correlation between exposure and onset and progression of nephropathy.[22] This porcine nephropathy[23] bears typical signs of toxicity to proximal tubules: loss of ability to concentrate urine, glycosuria, and histological proximal tubule degeneration.
Other nephropathies, although not responding to the "classical" definition of BEN, may be linked to ochratoxin. Thus, this could in certain circumstances be the case for focal segmental glomerulosclerosis after inhalational exposure: such a glomerulopathy with noteworthy proteinuria has been described[24] in patients with very high urinary ochratoxin levels (around 10 times levels that can be met with in "normal" subjects, i.e. around 10 ppb or 10 ng/ml).
Food animal industry impact
[edit]Ochratoxin-contaminated feed has its major economic impact on the poultry industry. Chickens, turkeys, and ducklings are susceptible to this toxin. Clinical signs of avian ochratoxicosis generally involve reduction in weight gains, poor feed conversion, reduced egg production, and poor egg shell quality.[25] Economic losses occur also in swine farms, linked to nephropathy and costs for the disposal of carcasses.
Toxicity does not seem to constitute a problem in cattle, as the rumen harbors protozoa that hydrolyze OTA.[26] However, contamination of milk is a possibility.[citation needed]
Dietary guidelines
[edit]| Source | Median in μg/kg of food |
Median in ng/kg of food |
Weight in kg |
Diet 1 | Diet 1+ |
|---|---|---|---|---|---|
| Liquorice extract | 26.30 | 26,300 | |||
| Ginger | 5.50 | 5,500 | 0.005 | 27.50 | |
| Nutmeg | 2.27 | 2,265 | 0.005 | 11.33 | |
| Paprika | 1.32 | 1,315 | 0.005 | 6.58 | |
| Pig liver | 1.10 | 1,100 | |||
| Ginseng | 1.10 | 1,100 | |||
| Raisins dry | 0.95 | 950 | 0.1 | 95.00 | |
| Pig kidney | 0.80 | 800 | 0.2 | 160 | |
| Liquorice confectionery | 0.17 | 170 | |||
| Coffee | 0.13 | 125 | 0.3 | 37.50 | |
| Cereals | 0.09 | 87.5 | 0.5 | 43.75 | |
| Peanuts | 0.08 | 79 | 0.2 | 15.80 | |
| Wine | 0.05 | 50 | 0.5 | 25 | |
| Pulses | 0.05 | 49.5 | 0.5 | 24.75 | |
| Beer | 0.05 | 49 | |||
| Salami | 0.05 | 49 | 0.3 | 14.70 | |
| Total in ng | 286.11 | 461.91 |
EFSA established in 2006 the "tolerable weekly intake" (TWI) of ochratoxin A (on advice of the Scientific Panel on Contaminants in the Food Chain) at 120 ng/kg.,[27] equivalent to a tolerable daily intake (TDI) of 14 ng/kg. Other organizations have established even lower limits for intake of ochratoxin A, based on the consumption habits of the population.[28] For USA, the FDA considers a TDI of 5 ng/kg. In the US, mean body weight for men is 86 kg, and for women 74 kg.[citation needed] Hence, the TDI for men is 430 ng and for women is 370 ng. In the joined table "weight in kg" is the weight eaten per day of each of the listed foodstuffs. Diet 1, with small quantities of ginger, nutmeg, and paprika, a good serving of dry raisins, a reasonable amount of coffee, cereals, wine, pulses, and salami, amounts to a safe diet (as for ochratoxin, at least), with 286 ng per day. However, it would be easy to go into excessive levels (Diet 1+), just by eating 200 g of pig kidney and 200 g of peanuts, which would lead to a total of nearly 462 ng of ochratoxin. This shows how delicate a safe diet can be.
| Gender | Weight in kg |
Tolerable OTA in ng |
|---|---|---|
| male | 86 | 430 |
| female | 74 | 370 |
Although ochratoxin A is not held as of today as responsible for renal cell carcinoma (RCC), the most frequent renal cancer, it is frequently written that dietary pattern might decrease or increase the risk of RCC. A Uruguayan case-control study [29] correlates intake of meat with occurrence of RCC. A very large prospective cohort in Sweden [30] explores correlations between RCC occurrence, diets rich in vegetables and poultry (so-called "healthy diets"), and diets rich in meat (especially processed meat: salami, black pudding). The thesis defended is that more fruit and vegetables might have a protective role. Fruit (except raisins and dried fruit) are very poor in ochratoxin, and processed meat can be rich in ochratoxin.
Dermal exposure
[edit]Ochratoxin A can permeate through the human skin.[31] Although no significant health risk is expected after dermal contact in agricultural or residential environments, skin exposure to ochratoxin A should nevertheless be limited.
Genetic resistance
[edit]In 1975, Woolf et al.[32] proposed that the inherited disorder phenylketonuria protects against ochratoxin A poisoning through the production of high levels of phenylalanine. Ochratoxin is a competitive inhibitor of phenylalanine in the phenylalanyl-tRNA-synthetase-catalyzed reaction thus preventing protein synthesis, which can be reversed by introducing phenylalanine, which is in excess in PKU individuals.[33]
See also
[edit]References
[edit]- ^ a b Al-Anati L, Petzinger E (2006). "Immunotoxic activity of ochratoxin A". J. Vet. Pharmacol. Ther. 29 (2): 79–90. doi:10.1111/j.1365-2885.2006.00718.x. PMID 16515661.
- ^ Polizzi V, et al. (2009). "Fungi, mycotoxins and volatile organic compounds in mouldy interiors from water-damaged buildings". Journal of Environmental Monitoring. 11 (10): 1849–1858. doi:10.1039/b906856b. PMID 19809708.
- ^ Richard JL, at al. (1999). "The occurrence of ochratoxin A in dust collected from a problem household". Mycopathologia. 146 (2): 99–103. Bibcode:1999Mycop.146...99R. doi:10.1023/A:1007056627296. PMID 10822509. S2CID 31557794.
- ^ Pfohl-Leszkowicz A, Manderville RA (2007). "Ochratoxin A: An overview on toxicity and carcinogenicity in animals and humans". Mol Nutr Food Res. 51 (1): 61–99. doi:10.1002/mnfr.200600137. PMID 17195275.
- ^ a b O'Brien E, Dietrich DR (2005). "Ochratoxin A: the continuing enigma". Crit. Rev. Toxicol. 35 (1): 33–60. Bibcode:2005CRvTx..35...33O. doi:10.1080/10408440590905948. PMID 15742902. S2CID 15849038.
- ^ Blesa J, et al. (2006). "Factors affecting the presence of ochratoxin A in wines". Critical Reviews in Food Science and Nutrition. 46 (6): 473–8. doi:10.1080/10408390500215803. PMID 16864140. S2CID 45392770.
- ^ Clark HA, Snedeker SM (2006). "Ochratoxin A: its cancer risk and potential for exposure". Journal of Toxicology and Environmental Health Part B: Critical Reviews. 9 (3): 265–96. Bibcode:2006JTEHB...9..265C. doi:10.1080/15287390500195570. PMID 16621780. S2CID 41761053.
- ^ Palma N, et al. (2007). "Ochratoxin A-Induced Mutagenesis in Mammalian Cells Is Consistent with the Production of Oxidative Stress". Chemical Research in Toxicology. 20 (7): 1031–1037. doi:10.1021/tx700027j. PMC 2367102. PMID 17567156.
- ^ Bendele AM, et al. (1985). "Ochratoxin A Carcinogenesis in the (C57BL/6J X C3H) F1mouse". J Natl Cancer Inst. 75 (4): 733–42. PMID 3862905.
- ^ Gary A. Boorman. "Toxicology and Carcinogenesis studies of Ochratoxin A in F344/N rats". National Toxicology Program, May 1989, NTP TR 358.
- ^ Basic-Jukic N, et al. (2007). "Renal transplantation in patients with Balkan endemic nephropathy". Transplant Proc. 39 (5): 1432–1435. doi:10.1016/j.transproceed.2006.11.019. PMID 17580155.
- ^ C. Cavin, T. Delatour, M. Marin-Kuan, D. Holzhauser, L. Higgins, C. Bezencon (2007). "REduction in antioxidant defences may contribute to ochratoxin A toxicity and carcinogenicity". Toxicological Sciences. 96 (1): 30–39. doi:10.1093/toxsci/kfl169. PMID 17110534.
- ^ Belmadani A, et al. (1999). "Selective toxicity of ochratoxin A in primary cultures from different brain regions". Arch Toxicol. 73 (2): 108–114. Bibcode:1999ArTox..73..108B. doi:10.1007/s002040050594. PMID 10350191. S2CID 19714271.
- ^ Sava V, et al. (2006). "Acute neurotoxic effects of the fungal metabolite ochratoxin A". Neurotoxicology. 27 (1): 82–92. Bibcode:2006NeuTx..27...82S. doi:10.1016/j.neuro.2005.07.004. PMID 16140385.
- ^ Xiangnan Zhang, et al. (2009). "Ochratoxin A induces apoptosis in neuronal cells". Genes Nutr. 4 (1): 41–48. doi:10.1007/s12263-008-0109-y. PMC 2654052. PMID 19148691.
- ^ Kunio Doi, Koji Uetsuka (2011). "Mechanisms of Mycotoxin-Induced Neurotoxicity through Oxidative Stress-Associated Pathways". International Journal of Molecular Sciences. 12 (8): 5213–5327. Bibcode:2011IJMSc..12.5213D. doi:10.3390/ijms12085213. PMC 3179161. PMID 21954354.
- ^ Aleckovic M, et al. (2010). "Glomerular filtration rate in examined population of Bosnian Posavina – region of Balkan Endemic Nephropathy". Bosn J Basic Med Sci. 10 (3–4): 256–61. doi:10.17305/bjbms.2010.2652. PMC 5627717. PMID 20433435.
- ^ Djukanovic L, et al. (2010). "Investigation of Balkan endemic nephropathy in Serbia: how to proceed?". Srp Arh Celok Lek. 138 (Suppl. 1): S68–72. doi:10.2298/SARH1004256D. PMID 20499513.
- ^ Castegnaro M, et al. (2006). "Balkan endemic nephropathy: role of ochratoxins A through biomarkers" (PDF). Mol Nutr Food Res. 50 (6): 519–29. doi:10.1002/mnfr.200500182. PMID 16715544.
- ^ Long DT, Voice TC (2007). "Role of exposure analysis in solving the mystery of Balkan endemic nephropathy". Croat. Med. J. 48 (3): 300–11. PMC 2080532. PMID 17589972.
- ^ Abouzied MM, Horvath AD, Podlesny PM, et al. (2002). "Ochratoxin A concentrations in food and feed from a region with Balkan Endemic Nephropathy". Food Additives and Contaminants. 19 (8): 755–64. Bibcode:2002FACon..19..755A. doi:10.1080/02652030210145036. PMID 12227939. S2CID 44838516.
- ^ Fink-Gremmels J. (29 June – 1 July 2005). "Conclusions from the workshops on Ochratoxin A in Food: recent developments and significance". Organized by ILSI Europe in Baden (Austria).
- ^ Krogh P, et al. (1976). "Experimental porcine nephropathy: changes of renal function and structure peroraly induced by crystalline ochratoxin A". Acta Pathol Microbiol Scand A. 84 (5): 429–34. PMID 970130.
- ^ Hope JH, Hope BE (2012). "A Review of the Diagnosis and Treatment of Ochratoxin A Inhalational Exposure Associated with Human Illness and Kidney Disease including Focal Segmental Glomerulosclerosis". Journal of Environmental and Public Health. 2012: 1–10. doi:10.1155/2012/835059. PMC 3255309. PMID 22253638. Article ID 835059.
- ^ Niemiec J, Borzemska W (1994). "The effect of Ochratoxin A on egg quality development of embryos and the level of toxin in egg and tissue of hens and chicks". Journal of Animal and Feed Sciences. 3 (4): 309–316. doi:10.22358/jafs/69844/1994.
- ^ Battacone G. Nudda A. Pulina G. (2010). "Effects of Ochratoxin A on Livestock Production". Toxins. 2 (7): 1796–1824. doi:10.3390/toxins2071796. PMC 3153269. PMID 22069661.
- ^ Scientific Panel on Contaminants in the Food Chain (2006). "Opinion of the Scientific Panel on Contaminants in the Food Chain on a request from the Commission related to Ochratoxin A in Food, Question N° EFSA-Q-2005-154, Adopted on 4 April 2006". The EFSA Journal. 365: 1–56.
- ^ Codex Alimentarius Commission (1999). "Codex Committee on Food Additives and Contaminants, Position paper on ochratoxin A, Thirty-first Session, The Hague, The Netherlands, 22-26 March 1999" (PDF). Joint FAO/WHO Food Standards Programme: 1–9. Archived from the original (PDF) on 2017-05-18.
- ^ De Stefani, et al. (1998). "Meat intake, mate drinking and renal cell cancer in Uruguay: a case-control study". Br J Cancer. 78 (9): 1239–1243. doi:10.1038/bjc.1998.661. PMC 2062999. PMID 9820187.
- ^ Rashidkhani B, et al. (2005). "Major dietary patterns and risk of Renal Cell Carcinoma in a prospective cohort of Swedish women". J. Nutr. 135 (7): 1757–1762. doi:10.1093/jn/135.7.1757. PMID 15987861.
- ^ Boonen J, Malysheva SV, Taevernier L, Diana Di Mavungu J, De Saeger S, De Spiegeleer B (2012). "Human skin penetration of selected model mycotoxins". Toxicology. 301 (1–3): 21–32. Bibcode:2012Toxgy.301...21B. doi:10.1016/j.tox.2012.06.012. PMID 22749975.
- ^ Woolf LI (1975). "Phenylketonuria as a balanced polymorphism: the nature of the heterozygote advantage". Annals of Human Genetics. 38 (4): 461–469. doi:10.1111/j.1469-1809.1975.tb00635.x. PMID 1190737. S2CID 970755.
- ^ Withrock IC, Anderson SJ, Jefferson MA, McCormack GR, Mlynarczyk GS, Nakama A, Lange JK, Berg CA, Acharya S, Stock ML, Lind MS, Luna K, Kondru NC, Manne S, Patel BB, de la Rosa BM, Huang KP, Sharma S, Hu HZ, Kanuri SH, Carlson SA (2015). "Genetic diseases conferring resistance to infectious diseases". Genes & Diseases. 2 (3): 247–254. doi:10.1016/j.gendis.2015.02.008. PMC 6150079. PMID 30258868.
External links
[edit] Media related to Ochratoxin A at Wikimedia Commons
Media related to Ochratoxin A at Wikimedia Commons- COMMISSION REGULATION (EU) 2023/915 of 25 April 2023 on maximum levels for certain contaminants in food and repealing Regulation (EC) No 1881/2006. See section 1.2 Annex I.
Ochratoxin A
View on GrokipediaChemical Properties
Molecular Structure
Ochratoxin A (OTA) is characterized by the molecular formula and a molecular weight of 403.81 g/mol.[8] The molecule is a hybrid structure consisting of a -phenyl--L-phenylalanine moiety linked via an amide bond to a 3,4-dihydro-3-methyl-8-hydroxy-6-methoxy-isocoumarin unit, with a chlorine atom substituted at the para position of the phenyl ring.[8] This arrangement includes key functional groups such as a lactone ring in the isocoumarin portion, a carboxylic acid at the phenylalanine terminus, and the amide linkage that connects the two domains.[8] The full IUPAC name reflects this complexity: (2S)-2-[[(3R)-5-chloro-8-hydroxy-3-methyl-1-oxo-3,4-dihydroisochromene-7-carbonyl]amino]-3-phenylpropanoic acid.[8] OTA features two chiral centers, with the (3R) configuration at the 3-position of the dihydroisocoumarin ring and the (2S) configuration in the L-phenylalanine-derived portion.[8] The L-phenylalanine stereochemistry is essential for OTA's biological potency, as diastereomers and enantiomers with modified configurations at this site demonstrate significantly diminished cytotoxicity in cellular assays. Among related ochratoxins, ochratoxin B serves as the non-chlorinated analog of OTA, differing only by the absence of the chlorine substituent and exhibiting lower toxicity due to this structural variation.[9] In contrast, ochratoxin C is the ethyl ester form of OTA, where the carboxylic acid group is esterified, enhancing lipophilicity but retaining core structural similarities to the parent compound.[10]Physical and Chemical Characteristics
Ochratoxin A appears as a white to off-white crystalline powder.[4][8] Its key physical properties include a melting point of 169°C.[8][11] The compound exhibits low solubility in water, approximately 0.00042 mg/mL (0.42 mg/L) at 25°C, but shows higher solubility in polar organic solvents such as methanol (up to 10 mg/mL), ethanol, acetone, and chloroform.[4][12][13] This solubility profile influences its extraction and detection in contaminated matrices. Ochratoxin A demonstrates chemical stability under acidic and neutral conditions, with high stability during heat treatments like autoclaving at 121°C, showing negligible degradation even after prolonged exposure (e.g., 3 hours).[14] It degrades more readily under alkaline conditions, with up to 50% removal observed, and at high temperatures exceeding 200°C, where partial decomposition occurs.[15][16] The compound is relatively stable to photodegradation in solid form but sensitive to UV light and direct sunlight in solution, leading to hydrolysis of its lactone ring.[8] The pKa values of ochratoxin A are approximately 4.4 for the carboxylic acid group and 7.1 for the phenolic hydroxyl group, which affect its ionization state and bioavailability in different pH environments.[17][18] These values stem from its molecular structure, featuring an isocoumarin derivative linked to L-phenylalanine.[13]Biosynthesis and Producing Organisms
Fungal Producers
Ochratoxin A (OTA) was first isolated in 1965 as a toxic metabolite produced by the fungus Aspergillus ochraceus from cultures grown on sterile maize meal during investigations in South Africa.[19] This discovery, reported by van der Merwe and colleagues, identified OTA as a secondary metabolite responsible for nephrotoxic effects observed in animal models exposed to fungal cultures.[20] The primary fungal producers of OTA belong to the genera Aspergillus and Penicillium, with approximately 20 species confirmed as capable of biosynthesis across these groups.[21] Key Aspergillus species include A. ochraceus, A. carbonarius, A. niger, and A. westerdijkiae, while prominent Penicillium species are P. verrucosum and P. nordicum.[22] These fungi are ubiquitous soil saprophytes and opportunistic pathogens that colonize plant materials post-harvest.[1] Aspergillus species predominate in tropical and subtropical regions, thriving on commodities like coffee, grapes, and cereals in warmer climates, whereas Penicillium species are more prevalent in temperate and cooler areas, often contaminating stored grains such as barley and wheat.[23] For instance, A. carbonarius is a major OTA producer in Mediterranean vineyards, while P. verrucosum is the dominant source in European cereal stores.[24] OTA-producing strains generally favor warm, humid environments with water activity levels above 0.88 and temperatures between 15–30°C, though optimal conditions vary by species.[25]Biosynthetic Pathway
Ochratoxin A (OTA) biosynthesis in producing fungi proceeds via a hybrid polyketide-nonribosomal peptide pathway, initiating with the polyketide synthase (PKS)-mediated assembly of the isocoumarin moiety from acetyl-CoA and malonyl-CoA units, followed by oxidative modifications and amide bond formation with L-phenylalanine. The PKS enzyme catalyzes the iterative condensation to form a dihydroisocoumarin intermediate, specifically 7-methylmellein, which is then oxidized to 7-hydroxymellein (OTβ) by a cytochrome P450 monooxygenase. Subsequently, a nonribosomal peptide synthetase (NRPS) facilitates the amide coupling of OTβ with L-phenylalanine to yield ochratoxin B (OTB), which undergoes chlorination at the para position of the phenyl ring to produce OTA. This pathway is conserved across OTA-producing species, reflecting a modular enzymatic architecture that integrates polyketide chain elongation with peptide ligation.[26] Key enzymes in the pathway include the multifunctional PKS (OtaA), which incorporates domains such as ketosynthase (KS) for carbon-carbon bond formation, acyltransferase (AT) for malonyl-CoA loading, and dehydratase for intermediate processing, culminating in the release of the polyketide product via a thioesterase domain. The NRPS enzyme (OtaB) contains adenylation, peptidyl carrier protein, and condensation domains to selectively activate and couple L-phenylalanine to OTβ, with its thioesterase domain enabling product release as OTB. Chlorination is mediated by a flavin-dependent halogenase (OtaD), which installs the chlorine atom on the phenylalanine-derived moiety post-coupling, while the P450 enzyme (OtaC) performs the essential hydroxylation step. These enzymes operate in a coordinated manner, with the coupling preceding chlorination, as evidenced by gene disruption studies.[26] The OTA biosynthetic pathway is encoded by a compact gene cluster comprising four core structural genes—otaA (PKS), otaB (NRPS-thioesterase), otaC (cytochrome P450), and otaD (halogenase)—along with regulatory elements, identified in both Aspergillus and Penicillium species such as A. ochraceus, A. carbonarius, and P. nordicum. This orthologous cluster spans approximately 7-10 kb and is flanked by transporter and pathway-specific regulator genes, with otaA serving as the hallmark PKS gene for cluster delineation. Comparative genomics has revealed high sequence conservation (>80% identity) among these genes across genera, though Penicillium clusters often include additional tailoring enzymes. Gene knockout experiments in model strains have confirmed the essential roles of each component in OTA production.[26][27] Regulation of the OTA biosynthetic cluster is primarily governed by environmental cues and transcriptional activators within the producing fungi. The bZIP transcription factor OtaR1 positively regulates the otaA-otaD core genes, while OtaR2 fine-tunes expression of otaA, otaC, and otaD under stress conditions. External factors such as pH (optimal at 5.0-6.5, mediated by the PacC pH-response regulator), temperature (peaking at 25-30°C), and nutrient availability (enhanced by lactose as a carbon source and organic nitrogen, repressed by ammonium via CreA and AreA mediators) modulate cluster expression through signaling pathways like HOG for osmolarity. These influences integrate with global secondary metabolism regulators to control OTA output during fungal growth.[26][23]Occurrence in Food and Environment
Contaminated Commodities
Ochratoxin A (OTA) contamination is prevalent in cereal grains such as wheat, barley, oats, and corn, particularly in stored products where levels can reach up to 100 µg/kg under suboptimal conditions. Surveys have detected OTA in 15-70% of cereal samples, with concentrations often ranging from 0.1 to 5 µg/kg in processed forms, though exceedances of the European Union maximum limit of 5 µg/kg occur in unprocessed grains like barley and wheat (3 µg/kg for processed cereal products). This contamination primarily arises from fungal growth during storage, affecting both human food and animal feed supplies. Note that EU maximum levels were updated in 2022, lowering limits for several products including processed cereals (to 3 µg/kg). In beverages, OTA frequently contaminates wine derived from grapes, especially in European regions where surveys indicate presence in 20-85% of samples at levels of 0.09-1.5 µg/L, with up to 20% exceeding the EU limit of 2 µg/L in certain types of wines. Coffee, particularly green beans infected by Aspergillus species, shows OTA levels from 0.4 to 50 µg/kg, reducing slightly upon roasting but remaining a notable source (EU limit for roasted coffee: 3 µg/kg for beans/ground, 5 µg/kg for instant, as updated in 2022). Beer, sourced from contaminated barley malt, contains OTA in 45% of samples at 0.04-0.35 µg/L, transferred during brewing processes. Animal products, including pork and poultry, exhibit OTA carryover from contaminated feed, with highest concentrations in organs like kidneys and livers. In pork, OTA levels range from 0.1 to 0.8 µg/kg in muscle but up to 23 µg/kg in kidneys, while poultry kidneys show 0.9-23 µg/kg, posing risks in meat supply chains. This bioaccumulation occurs in species like pigs and chickens fed OTA-tainted grains. Other commodities such as dried fruits, spices, and cocoa are also affected, with OTA levels in dried fruits reaching up to 10 µg/kg (though the current EU limit is 2 µg/kg, updated in 2022), spices like paprika and pepper reaching 15 µg/kg (EU limit: 15 µg/kg), and cocoa beans showing variable contamination during processing and storage. Environmentally, OTA persists in soil and airborne dust, linked to fungal spores from agricultural residues. Globally, OTA incidence is higher in developing regions due to inadequate storage practices, leading to elevated levels in staples like cereals and contributing to greater dietary exposure compared to regulated areas in Europe and North America.Factors Influencing Production
The production of ochratoxin A (OTA) by toxigenic fungi such as species of Aspergillus and Penicillium is modulated by a range of environmental and biological conditions that influence fungal growth and secondary metabolite biosynthesis.[22] These factors determine the onset and extent of OTA synthesis, often peaking under suboptimal conditions for fungal proliferation but favorable for toxin accumulation.[28] Abiotic conditions play a central role in OTA production. Temperature optima vary by species, with Aspergillus carbonarius and A. niger favoring 25–30°C, while A. ochraceus produces maximally at 20–30°C.[28][22] Water activity (a_w) is critical, with OTA synthesis occurring between 0.87–0.99 a_w but optimizing at 0.95–0.99 for most producers, such as P. verrucosum on wheat grain where levels drop sharply below 0.90 a_w.[29][28] Relative humidity exceeding 80% further promotes fungal sporulation and toxin elaboration by maintaining high a_w in substrates.[22] Acidic pH environments, typically 4–6, enhance production, with peaks for A. carbonarius and A. niger at pH 5.0–6.5 and for P. citrinum at pH 5.[22][28] Substrate composition significantly affects OTA yields, as nutrient availability directs metabolic pathways toward secondary metabolites. Carbon sources like arabinose, sucrose, and glucose at 6% concentration stimulate biosynthesis in A. niger, with arabinose yielding up to 21.93 µg/g dry hyphae, whereas nitrogen-rich peptone inhibits it.[30] Nutrient-dense media such as grains (e.g., corn, rice) or fruit-based substrates (e.g., grapes) support higher OTA levels compared to simpler media, due to enhanced fungal biomass and polyketide synthase activity.[29][28] Optimal pH in these substrates often shifts during growth as glucose catabolism acidifies the medium, further favoring OTA accumulation.[29] Biotic interactions and stress responses also upregulate OTA production. Competition among microbial communities can alter toxin levels, with certain indigenous fungi reducing OTA output from P. verrucosum and P. nordicum through resource competition or antagonism.[31] Oxidative stress, marked by elevated superoxide anions and hydrogen peroxide alongside reduced catalase and peroxidase activity, triggers OTA synthesis via gene upregulation (e.g., pks, nrps) in A. niger.[30] Similarly, osmotic stress from high NaCl concentrations activates the high osmolarity glycerol (HOG) pathway in P. nordicum, increasing OTA as an adaptive response.[22] Post-harvest conditions exacerbate OTA risks by creating conducive microenvironments for fungal proliferation. Inadequate ventilation and moisture control in storage facilities elevate humidity and a_w, promoting mold growth and toxin production in crops like cereals.[22] Climate change contributes by expanding warmer, humid regions suitable for Aspergillus species, thereby increasing OTA incidence in temperate zones shifting toward subtropical climates.[29][28]Toxicity and Health Effects
Mechanisms of Toxicity
Ochratoxin A (OTA) is rapidly absorbed from the gastrointestinal tract following oral exposure, with bioavailability ranging from 40% to 80% in various species, and is distributed primarily to the kidneys and liver due to its affinity for plasma proteins such as albumin.[4] In the kidneys, OTA undergoes active uptake via organic anion transporters (OATs), including OAT1, OAT3, and OAT4, which facilitate its secretion and reabsorption in proximal tubules, contributing to its accumulation.[32] The pharmacokinetics of OTA exhibit linear characteristics over typical exposure ranges, with a notably long elimination half-life of approximately 35 days in humans, attributed to extensive protein binding that slows clearance.[18] The primary mechanism of OTA toxicity involves inhibition of protein synthesis through competitive binding to phenylalanyl-tRNA synthetase (PheRS), an enzyme essential for attaching phenylalanine to its cognate tRNA.[33] This inhibition arises from OTA's structural resemblance to phenylalanine, as OTA consists of a dihydroisocoumarin moiety linked via an amide bond to L-β-phenylalanine, allowing it to mimic the amino acid substrate and disrupt translation elongation at the ribosomal level.[34] Studies in bacterial and mammalian systems have confirmed that OTA reduces PheRS activity in a dose-dependent manner, leading to impaired cellular protein production without affecting other aminoacyl-tRNA synthetases significantly.[33] OTA also induces oxidative stress by generating reactive oxygen species (ROS), which trigger lipid peroxidation and oxidative DNA damage in affected cells.[35] This process is mediated in part by the depletion of cellular antioxidants, such as glutathione, and inhibition of Nrf2 signaling pathways, exacerbating the imbalance between oxidant production and detoxification.[35] In vitro models demonstrate that OTA exposure elevates markers of oxidative damage, including malondialdehyde and 8-oxoguanine, underscoring ROS as a key contributor to its cytotoxic effects.[36] Bioactivation of OTA occurs primarily through cytochrome P450 enzymes (e.g., CYP1A2 and CYP3A4), which metabolize it into reactive intermediates such as quinone derivatives capable of forming adducts with cellular macromolecules.[37] Additionally, OTA directly inhibits mitochondrial function by disrupting electron transport chain complexes, particularly at low concentrations, leading to reduced ATP production and uncoupling of oxidative phosphorylation.[38] The acute oral LD50 of OTA in rats is approximately 20-22 mg/kg body weight, reflecting its potency in eliciting these biochemical disruptions.[39]Nephrotoxicity and Related Diseases
Ochratoxin A (OTA) primarily targets the kidneys, where it accumulates in proximal tubule epithelial cells due to active transport via organic anion transporters, leading to degeneration characterized by oxidative stress, DNA damage, and apoptosis through pathways such as PTEN/AKT and ERK1/2.[40] This degeneration manifests as cellular necrosis and impaired proximal tubule function, including reduced secretory capacity for organic anions and endocytic uptake of proteins like albumin.[41] Chronic exposure further promotes renal fibrosis via epithelial-to-mesenchymal transition (EMT), with upregulation of transforming growth factor-β (TGF-β), α-smooth muscle actin (α-SMA), and tissue inhibitor of metalloproteinase-1 (TIMP-1), resulting in increased collagen deposition (up to 244% in renal cortex) and extracellular matrix accumulation.[42][40] Additionally, OTA reduces glomerular filtration rate (GFR) by altering renal hemodynamics, such as increasing efferent arteriolar resistance and decreasing renal blood flow, which collectively impair overall kidney function.[41][40] OTA has been associated with human nephropathies, particularly Balkan endemic nephropathy (BEN), a chronic tubulointerstitial disease prevalent in rural areas along the Danube River tributaries in the Balkans, and chronic interstitial nephropathy in other regions.[43] In BEN patients, serum OTA levels have been reported as elevated, with medians around 1.15 ng/mL (range 0.4–3.9 ng/mL), higher than in controls. Urinary OTA levels in endemic areas are generally low, with means around 0.007 ng/mL (range up to 0.86 ng/mL).[2][44] In serum of Polish patients with chronic renal failure on dialysis, including those with interstitial nephropathy, OTA was detected in 95% with mean 52.7 ng/mL (maximum 62.8 ng/mL).[45] However, causality remains debated, as recent reviews emphasize a multifactorial etiology for BEN, with aristolochic acid from Aristolochia plants identified as the primary cause, while OTA may act as a contributing or synergistic factor rather than the sole agent.[43][46] In animal models, the pig kidney serves as a sensitive indicator of OTA nephrotoxicity, mirroring human pathology in conditions like Danish porcine nephropathy.[41] Pigs exposed to OTA exhibit histopathological changes including proximal tubular necrosis, interstitial fibrosis, and reduced urine concentrating ability, with effects observable at chronic low doses that impair postproximal nephron function and electrolyte handling.[41] These findings underscore the organ's vulnerability, as OTA concentrations in pig kidneys can reach levels 6.25 times higher than in controls after intoxication.[47] Epidemiological data link OTA exposure to renal diseases in regions with high cereal contamination, such as the Balkans, where grains like maize and wheat often exceed OTA thresholds due to Aspergillus and Penicillium growth under humid conditions.[43] Studies show correlations between dietary OTA intake from contaminated cereals and elevated biomarkers in affected populations, but establishing direct causality is complicated by confounding factors like genetic susceptibility and co-exposures, as highlighted in 2021 reviews questioning OTA's primacy in BEN etiology.[46][48]Carcinogenicity
Ochratoxin A (OTA) was classified by the International Agency for Research on Cancer (IARC) in 1993 as a Group 2B carcinogen, indicating it is possibly carcinogenic to humans, based primarily on sufficient evidence of carcinogenicity in experimental animals. As of 2024, this classification remains unchanged, with the EFSA's 2020 opinion reaffirming genotoxic concerns without revising tolerable intake due to data limitations.[49][4][50] This classification stems from studies demonstrating OTA's ability to induce renal tumors in rodents following oral administration. In animal models, OTA consistently promotes renal carcinogenesis, particularly in rats, where dietary exposure leads to the development of renal cell adenomas and carcinomas. For instance, in male Fischer 344 rats administered OTA by gavage at doses of 21, 70, or 210 μg/kg body weight for two years, renal tumor incidence increased dose-dependently, reaching up to 72% (adenomas and carcinomas combined) at the highest dose of 210 μg/kg.[51][52] Similar findings in other rat studies report tumor incidences ranging from 20% to 80% at dietary concentrations of 5 mg/kg, highlighting OTA's potent nephrocarcinogenic effects in males, who are more susceptible than females.[53] The proposed mechanism involves oxidative metabolism of OTA to its hydroquinone derivative (OTHQ), which autoxidizes to a quinone (OTQ) intermediate capable of forming DNA adducts, such as the OTB-deoxyguanosine adduct, thereby contributing to genotoxic damage.[54] Regarding genotoxicity, OTA exhibits indirect mutagenic potential, testing negative in the Ames assay without metabolic activation but positive when rat liver microsomes or transition metals (e.g., Fe(III)) are present, indicating bioactivation is required for DNA interaction.[54] In humans, epidemiological evidence links OTA exposure to increased risk of urinary tract tumors, particularly in regions affected by Balkan endemic nephropathy (BEN), where OTA levels in blood were detectable in 26.7% of patients with BEN or urinary tract tumors versus 12.1% in healthy endemic controls.[55] Systematic reviews of such studies report unadjusted odds ratios of approximately 2-3 for associations between OTA exposure and BEN or upper urinary tract cancers, though results are often not statistically significant due to confounding factors like co-exposures.[2]Other Toxic Effects
Ochratoxin A (OTA) exhibits neurotoxic effects beyond its primary renal impacts, primarily through disruption of neurotransmitter systems and induction of oxidative stress in the brain. In rodent models, OTA interferes with glutamate uptake by downregulating transporters such as GLT-1 in astrocytes, leading to excitotoxicity and impaired neurotransmitter homeostasis.[56] This disruption contributes to dopaminergic dysfunction, as evidenced by acute depletion of dopamine and its metabolites in the striatum of exposed mice.[57] Behavioral alterations, including reduced locomotion and motor impairment, have been observed in male Balb/C mice following subchronic oral exposure to OTA at doses of 0.21–0.5 mg/kg, with effects persisting for up to six months post-treatment.[56] The immunotoxic potential of OTA manifests as suppression of key immune responses, increasing vulnerability to pathogens in animal models. OTA inhibits T-cell proliferation by depleting cellular ATP and downregulating signaling pathways such as STAT1 and STAT3 in murine splenocytes at concentrations as low as 10 ng/mL in vitro.[58] This leads to impaired adaptive immunity, including reduced antibody production; for instance, in broiler chicks fed OTA at 0.5–2 mg/kg in the diet, IgY and IgG titers against sheep red blood cells were significantly lowered by day 14 post-immunization.[58] Consequently, exposed animals show heightened susceptibility to infections, such as exacerbated E. coli colonization and Newcastle disease in chickens administered 0.1–1.5 mg/kg OTA in feed, due to diminished NK cell activity and interferon induction.[58] OTA also induces hepatotoxicity characterized by oxidative damage and inflammatory responses in the liver, often marked by elevated serum enzymes. In rats, subacute exposure to OTA via oral gavage elevates levels of alanine aminotransferase (ALT) and aspartate aminotransferase (AST), indicative of hepatocellular injury, with protective agents like silymarin mitigating these increases.[59] Teratogenic effects further highlight OTA's developmental toxicity, particularly in rodents, where single oral doses around 2.75 mg/kg body weight during gestational days 6–7 in Wistar rats cause fetal skeletal malformations, including defects in sternebrae, vertebrae, and ribs, alongside visceral anomalies like renal hypoplasia.[60] These outcomes stem from OTA's inhibition of protein synthesis, a mechanism that broadly underlies its toxic profile.[61] Evidence for endocrine disruption by OTA remains limited but suggests interference with hormone biosynthesis rather than direct receptor binding. In human adrenocortical H295R cells, OTA at 1000 ng/mL upregulates aromatase expression, resulting in elevated estradiol production without agonistic or strong antagonistic effects on steroid receptors.[62] This alteration in steroidogenesis has been linked to reproductive impairments in rats, though further studies are needed to clarify its physiological relevance.[63]Exposure Assessment
Routes of Exposure
The primary route of exposure to ochratoxin A (OTA) for humans and animals is dietary intake, occurring through the consumption of contaminated food and feed such as cereals, coffee, wine, dried fruits, and pork products.[3] This pathway accounts for the majority of overall exposure in the general population, with OTA being rapidly absorbed from the gastrointestinal tract into the bloodstream.[3] Inhalation represents a minor route, primarily relevant in occupational settings such as grain handling, agriculture, or waste management, where workers may inhale dust containing OTA produced by molds like Aspergillus and Penicillium species.[64] Case reports have linked acute inhalation exposure, such as during prolonged granary work, to symptoms including renal failure, though such incidents are rare and recovery is possible with supportive care.[64] Dermal exposure occurs through direct skin contact with moldy materials or contaminated dust, but it is of low significance due to limited absorption and bioavailability.[3] Studies indicate that OTA can penetrate human skin, though no substantial health risks are typically associated with this route in agricultural or domestic settings.[65] Additional exposure pathways include maternal transfer to offspring. OTA crosses the placenta, with detectable levels found in umbilical cord blood, sometimes exceeding maternal concentrations, as observed in studies from regions with high dietary contamination.[66] In lactating individuals, OTA is excreted into breast milk, exposing nursing infants to levels ranging from traces to over 100 ng/L depending on maternal intake.[3] Similar transfer has been documented in animals via milk.[67] Due to its high affinity for serum albumin, OTA exhibits significant bioaccumulation in the body, particularly in the kidneys, leading to chronic low-level exposure even after intermittent intake.[68] In humans, the plasma half-life is approximately 35 days following oral exposure, far longer than in most other species, which contributes to its persistence and potential for cumulative effects.[69]Human and Animal Intake Levels
Human dietary exposure to ochratoxin A (OTA) primarily occurs through contaminated food, with average chronic intake levels in Europe estimated at 0.6–2.1 ng/kg body weight per day for adults and higher for children and infants (0.9–5.1 ng/kg body weight per day). High-percentile (95th) exposures reach 1.2–4.9 ng/kg body weight per day in adults and up to 11.9 ng/kg body weight per day in younger populations, based on occurrence data from cereals, coffee, wine, and other commodities. In high-risk regions, such as areas affected by Balkan endemic nephropathy, intake levels have historically been elevated, reaching up to 15 ng/kg body weight per day due to persistent contamination in staple foods like grains and pork products.[4][4][70] Among animals, pigs exhibit the highest sensitivity to OTA among animals, with the Joint FAO/WHO Expert Committee on Food Additives (JECFA) establishing a provisional tolerable weekly intake (PTWI) of 100 ng/kg body weight for humans, derived from renal effects observed in porcine studies.[71] In contrast, poultry and ruminants like cattle are less susceptible, as OTA undergoes substantial degradation in the rumen—primarily by protozoa and bacteria—to the less toxic ochratoxin α, reducing bioavailability and mitigating nephrotoxic risks. This species-specific metabolism highlights why monogastric animals like pigs face greater exposure challenges compared to herbivores. A 2023 EFSA assessment concluded that risks to animal health from OTA in feed are low for pigs, chickens for fattening, laying hens, and rabbits at observed exposure levels.[72][73][74] Risk assessments for OTA employ the margin of exposure (MOE) approach, particularly for nephrotoxicity, where the benchmark dose lower confidence limit (BMDL10) for non-neoplastic renal effects in pigs is 4.73 μg/kg body weight per day, yielding an MOE of approximately 10,000 relative to typical human exposures of 0.5–1 ng/kg body weight per day, indicating low concern but warranting continued monitoring. Biomonitoring relies on detecting OTA in urine (typically 0.013–110 ng/L) and serum (0.15–18 ng/L), which correlate with dietary intake and provide direct evidence of internal exposure in both humans and animals.[4][75][2] In livestock, OTA contamination leads to notable industry impacts, particularly in swine production, where feed levels of 1 mg/kg can reduce growth rates by 5–12% through impaired feed efficiency and weight gain, alongside increased susceptibility to secondary infections. These effects contribute to broader economic losses in pork production, estimated in the millions annually due to decreased carcass quality, higher veterinary costs, and rejected batches from residue carryover into meat.[76][73]Detection and Analysis
Analytical Methods
The detection and quantification of ochratoxin A (OTA) in food and feed samples require robust analytical methods to ensure accuracy, sensitivity, and specificity, given its low regulatory limits and complex matrices such as cereals, coffee, and wine.[50] Sample preparation is a critical initial step, typically involving extraction with methanol-water mixtures (e.g., 80:20 v/v) to solubilize OTA from the matrix, followed by filtration and dilution to minimize interferences.[77] Cleanup is often achieved using immunoaffinity columns (IAC), which selectively bind OTA via antibodies, enabling elution with methanol for purer extracts and reducing matrix effects in subsequent analysis.[78] This approach has been validated for various commodities, including roasted coffee and wheat, where it enhances recovery rates to 80-110%.[79] Instrumental methods dominate confirmatory analysis due to their precision. High-performance liquid chromatography with fluorescence detection (HPLC-FLD) is a standard technique, employing reversed-phase columns and excitation at 333 nm/emission at 460 nm to detect OTA's native fluorescence, achieving limits of detection (LOD) as low as 0.1 µg/kg in matrices like grains and dry-cured meats.[80] For enhanced specificity and multi-mycotoxin screening, liquid chromatography-tandem mass spectrometry (LC-MS/MS) is preferred, using electrospray ionization in negative mode to confirm OTA identity via multiple reaction monitoring, with LODs below 0.1 µg/kg and the ability to quantify over 90 mycotoxins simultaneously in complex samples.[50] These methods comply with international validation protocols, ensuring reproducibility across laboratories.[81] Rapid screening tests provide quick, on-site alternatives for high-throughput monitoring. Enzyme-linked immunosorbent assay (ELISA) kits utilize competitive antibody binding to OTA, offering sensitivity around 0.5 µg/kg in grains and feeds, with results obtainable in under 2 hours and suitable for preliminary assessments before instrumental confirmation.[82] Emerging biosensors, particularly aptamer-based designs, leverage single-stranded DNA or RNA aptamers that specifically recognize OTA, enabling label-free or electrochemical detection with LODs in the ng/mL range; for instance, structure-switching aptamer platforms integrated with lateral flow devices allow instrument-free readout in minutes for food samples.[83] These innovations improve portability and reduce costs compared to traditional immunoassays.[84] Method validation follows AOAC International and ISO standards to guarantee reliability. AOAC Official Method 2000.09, for example, outlines HPLC-FLD procedures for OTA in roasted coffee, specifying recovery, precision, and LOD criteria, while ISO 15141-1 and -2 provide guidelines for LC-based detection in foodstuffs at levels above 0.2-3 µg/kg.[85] Recent advances include portable near-infrared (NIR) spectroscopy, which uses chemometric models like partial least squares to non-destructively screen OTA in wheat, with potential for prediction accuracies over 90% and LODs around 1 µg/kg through integration with machine learning.[86] These spectroscopic tools facilitate real-time monitoring in agricultural settings without sample destruction.[50]| Method | Type | LOD (µg/kg) | Advantages | Limitations | Reference |
|---|---|---|---|---|---|
| HPLC-FLD | Instrumental | 0.1 | High sensitivity, routine use | Requires cleanup, lab-based | [80] |
| LC-MS/MS | Instrumental | <0.1 | Confirmation, multi-analyte | Expensive equipment | [50] |
| ELISA | Rapid screening | 0.5 | Quick, cost-effective | Semi-quantitative, matrix effects | [82] |
| Aptamer-based biosensor | Rapid/emerging | 0.01-0.1 | Portable, specific | Emerging validation | [83] |
| Portable NIR | Emerging | ~1 | Non-destructive, on-site | Calibration needed | [86] |
Regulatory Limits
The European Union has established maximum levels for ochratoxin A (OTA) in various foodstuffs through Commission Regulation (EU) 2023/915, which updated and consolidated previous measures under Regulation (EC) No 1881/2006, effective from May 2023.[87] These levels range from 0.50 µg/kg in baby foods and processed cereal-based foods for infants to 5.0 µg/kg in unprocessed cereal grains, with 3.0 µg/kg for most derived cereal products; for wine and grape juice intended for direct consumption, the limit is 2.0 µg/L.[87] Higher thresholds apply to certain commodities like dried vine fruits (8.0 µg/kg), pistachios for final use (5.0 µg/kg), and spices such as Capsicum spp. (20 µg/kg), reflecting considerations of typical contamination patterns and dietary exposure risks.[87] The Codex Alimentarius Commission recommends maximum levels for OTA aligned with international trade needs, including 5 µg/kg in raw cereal grains as a guideline to protect consumer health while facilitating global commerce.[88] For animal feed, Codex references general principles in standard CXS 193-1995, but specific limits such as 100–250 µg/kg in complete feed (e.g., 100 µg/kg for poultry, 250 µg/kg for pigs) are adopted from regional frameworks like the EU to prevent carryover into food products.[72] In the United States, the Food and Drug Administration (FDA) does not enforce binding regulatory limits for OTA in food or feed but relies on guidance levels and monitoring to ensure safety, evaluating contaminants under the Federal Food, Drug, and Cosmetic Act.[89] The FDA's Mycotoxins in Domestic and Imported Human Foods Compliance Program, updated in September 2024, targets OTA in grains, coffee, dried fruits, and other susceptible commodities through sampling and analysis to support risk assessments and import refusals when levels pose health concerns.[90] The World Health Organization's Joint FAO/WHO Expert Committee on Food Additives (JECFA) established a provisional tolerable weekly intake (PTWI) of 100 ng/kg body weight for OTA in 2007, reaffirmed in subsequent evaluations as a benchmark for dietary exposure. National monitoring efforts, such as the FDA's ongoing surveys, demonstrate high compliance in imported goods, with recent data indicating that OTA detections rarely exceed advisory thresholds in regulated commodities like cereals and coffee.[91] These regulatory frameworks emphasize prevention through analytical detection methods to enforce limits and minimize public health risks from OTA exposure.[90]| Region/Organization | Key Commodity | Maximum Level |
|---|---|---|
| EU (Regulation (EU) 2023/915) | Unprocessed cereals | 5.0 µg/kg |
| EU (Regulation (EU) 2023/915) | Cereal-derived products | 3.0 µg/kg |
| EU (Regulation (EU) 2023/915) | Wine | 2.0 µg/L |
| Codex Alimentarius | Raw cereal grains | 5 µg/kg (guideline) |
| US FDA | Grains (guidance, no binding limit) | Monitored; action if unsafe |
| WHO JECFA | Dietary intake (PTWI) | 100 ng/kg bw/week |

