Acesulfame potassium
View on Wikipedia
 | |
 | |
| Names | |
|---|---|
| IUPAC name
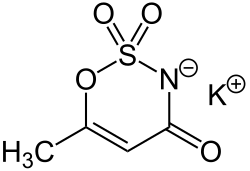
Potassium 6-methyl-2,2-dioxo-2H-1,2λ6,3-oxathiazin-4-olate
| |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.054.269 |
| EC Number |
|
| E number | E950 (glazing agents, ...) |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H4KNO4S | |
| Molar mass | 201.242 |
| Appearance | white crystalline powder |
| Density | 1.81 g/cm3 |
| Melting point | 225 °C (437 °F; 498 K) |
| 270 g/L at 20 °C | |
| Hazards | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Acesulfame potassium (UK: /æsɪˈsʌlfeɪm/,[1] US: /ˌeɪsiːˈsʌlfeɪm/ AY-see-SUL-faym[2] or /ˌæsəˈsʌlfeɪm/[1]), also known as acesulfame K or Ace K, is a synthetic calorie-free sugar substitute (artificial sweetener) often marketed under the trade names Sunett and Sweet One. In the European Union, it is known under the E number (additive code) E950.[3] It was discovered accidentally in 1967 by German chemist Karl Clauss at Hoechst AG (now Nutrinova).[4] Acesulfame potassium is the potassium salt of 6-methyl-1,2,3-oxathiazine-4(3H)-one 2,2-dioxide. It is a white crystalline powder with molecular formula C
4H
4KNO
4S and a molecular weight of 201.24 g/mol.[5]
Properties
[edit]Acesulfame K is 200 times sweeter than sucrose (common sugar), as sweet as aspartame, about two-thirds as sweet as saccharin, and one-third as sweet as sucralose. Like saccharin, it has a slightly bitter aftertaste, especially at high concentrations. Kraft Foods patented the use of sodium ferulate to mask acesulfame's aftertaste.[6] Acesulfame K is often blended with other sweeteners (usually sucralose or aspartame). These blends are reputed to give a more sucrose-like taste whereby each sweetener masks the other's aftertaste, or exhibits a synergistic effect by which the blend is sweeter than its components.[7] Acesulfame potassium has a smaller particle size than sucrose, allowing for its mixtures with other sweeteners to be more uniform.[8]
Unlike aspartame, acesulfame K is stable under heat, even under moderately acidic or basic conditions, allowing it to be used as a food additive in baking, or in products that require a long shelf life. Although acesulfame potassium has a stable shelf life, it can eventually degrade to acetoacetamide, which is toxic in high doses.[9] In carbonated drinks, it is almost always used in conjunction with another sweetener, such as aspartame or sucralose. It is also used as a sweetener in protein shakes and pharmaceutical products,[10] especially chewable and liquid medications, where it can make the active ingredients more palatable. The acceptable daily intake of acesulfame potassium is listed as 15 mg/kg/day.[11]
Acesulfame potassium is widely used in the human diet and excreted by the kidneys. It thus has been used by researchers as a marker to estimate to what degree swimming pools are contaminated by urine.[12]
Other names for acesulfame K are potassium acesulfamate, potassium salt of 6-methyl-1,2,3-oxothiazin-4(3H)-one-2,3-dioxide, and potassium 6-methyl-1,2,3-oxathiazine-4(3H)-one-3-ate-2,2-dioxide.
Effect on body weight
[edit]Acesulfame potassium provides a sweet taste with no caloric value. There is no high-quality evidence that using acesulfame potassium as a sweetener affects body weight or body mass index (BMI).[13][14][15]
Discovery
[edit]Acesulfame potassium was developed after the accidental discovery of a similar compound (5,6-dimethyl-1,2,3-oxathiazin-4(3H)-one 2,2-dioxide) in 1967 by Karl Clauss and Harald Jensen at Hoechst AG.[16][17] After accidentally dipping his fingers into the chemicals with which he was working, Clauss licked them to pick up a piece of paper.[18] Clauss is the inventor listed on a United States patent issued in 1975 to the assignee Hoechst Aktiengesellschaft for one process of manufacturing acesulfame potassium.[19] Subsequent research showed a number of compounds with the same basic ring structure had different levels of sweetness. 6-methyl-1,2,3-oxathiazine-4(3H)-one 2,2-dioxide had particularly favourable taste characteristics and was relatively easy to synthesize, so it was singled out for further research, and received its generic name (acesulfame-K) from the World Health Organization in 1978.[16] Acesulfame potassium first received approval for table top use in the United States in 1988.[11]
Safety
[edit]The United States Food and Drug Administration (FDA) approved its general use as a safe food additive in 1988,[20] and maintained that safety assessment as of 2025.[21]
In a 2000 scientific review, the European Food Safety Authority determined that acesulfame K is safe in typical consumption amounts, and does not increase the risk of diseases.[22]
Other sources
[edit]- von Rymon Lipinski GW (2000). "Sweeteners". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a26_023. ISBN 978-3-527-30385-4.
References
[edit]- ^ a b "acesulfame". Oxford English Dictionary. OED. Retrieved 24 July 2022.
- ^ "acesulfame–K". Merriam-Webster. Archived from the original on 10 March 2017. Retrieved 31 January 2017.
- ^ "Current EU approved additives and their E Numbers". UK: Food Standards Agency. 2012-03-14. Archived from the original on 2013-08-21. Retrieved 2012-08-07.
- ^ Clauss, K., Jensen, H. (1973). "Oxathiazinone Dioxides - A New Group of Sweetening Agents". Angewandte Chemie International Edition. 12 (11): 869–876. doi:10.1002/anie.197308691.
- ^ Ager, D. J., Pantaleone, D. P., Henderson, S. A., Katritzky, A. R., Prakash, I., Walters, D. E. (1998). "Commercial, Synthetic Nonnutritive Sweeteners" (PDF). Angewandte Chemie International Edition. 37 (13–14): 1802–1817. doi:10.1002/(SICI)1521-3773(19980803)37:13/14<1802::AID-ANIE1802>3.0.CO;2-9. Archived from the original (PDF) on 2008-09-10.
- ^ United States Patent 5,336,513 (expired in 2006)
- ^ Deis RC (November 2006). "Customizing Sweetness Profiles" (PDF). Food Product Design. Archived from the original (PDF) on 11 August 2014. Retrieved 16 May 2018.
- ^ Mullarney, M.; Hancock, B.; Carlson, G.; Ladipo, D.; Langdon, B. "The powder flow and compact mechanical properties of sucrose and three high-intensity sweeteners used in chewable tablets". Int. J. Pharm. 2003, 257, 227–236.
- ^ Findikli, Z.; Zeynep, F.; Sifa, T. Determination of the effects of some artificial sweeteners on human peripheral lymphocytes using the comet assay. Journal of toxicology and environmental health sciences 2014, 6, 147–153.
- ^ "Home – WHO – Prequalification of Medicines Programme". Retrieved 2 March 2017.
- ^ a b Whitehouse, C.; Boullata, J.; McCauley, L. "The potential toxicity of artificial sweeteners". AAOHN J. 2008, 56, 251–259, quiz 260.
- ^ Erika Engelhaupt (March 1, 2017). "Just How Much Pee Is In That Pool?". NPR. Archived from the original on March 1, 2017. Retrieved March 2, 2017.
- ^ Miller PE, Perez V (September 2014). "Low-calorie sweeteners and body weight and composition: a meta-analysis of randomized controlled trials and prospective cohort studies". The American Journal of Clinical Nutrition. 100 (3): 765–777. doi:10.3945/ajcn.113.082826. PMC 4135487. PMID 24944060.
- ^ Azad MB, Abou-Setta AM, Chauhan BF, Rabbani R, Lys J, Copstein L, Mann A, Jeyaraman MM, Reid AE, Fiander M, MacKay DS, McGavock J, Wicklow B, Zarychanski R (July 2017). "Nonnutritive sweeteners and cardiometabolic health: a systematic review and meta-analysis of randomized controlled trials and prospective cohort studies". CMAJ. 189 (28): E929–E939. doi:10.1503/cmaj.161390. PMC 5515645. PMID 28716847.
- ^ Rogers PJ, Hogenkamp PS, de Graaf C, Higgs S, Lluch A, Ness AR, Penfold C, Perry R, Putz P, Yeomans MR, Mela DJ (September 2015). "Does low-energy sweetener consumption affect energy intake and body weight? A systematic review, including meta-analyses, of the evidence from human and animal studies". International Journal of Obesity. 40 (3): 381–94. doi:10.1038/ijo.2015.177. PMC 4786736. PMID 26365102.
- ^ a b O'Brien-Nabors, L. (2001). Alternative Sweeteners. New York, NY: Marcel Dekker. p. 13. ISBN 978-0-8247-0437-7.
- ^ Williams, R. J., Goldberg, I. (1991). Biotechnology and Food Ingredients. New York: Van Nostrand Reinhold. ISBN 978-0-442-00272-5.
- ^ Newton, D. E. (2007). Food Chemistry (New Chemistry). New York: Infobase Publishing. p. 69. ISBN 978-0-8160-5277-6. Archived from the original on 2016-03-05. Retrieved 2017-09-08.
- ^ US 3917589, Clauss, K., "Process for the manufacture of 6-methyl-3,4-dihydro-1,2,3-oxathiazine-4-one-2,2-dioxide", issued 1975
- ^ Kroger, M., Meister, K., Kava, R. (2006). "Low-Calorie Sweeteners and Other Sugar Substitutes: A Review of the Safety Issues". Comprehensive Reviews in Food Science and Food Safety. 5 (2): 35–47. doi:10.1111/j.1541-4337.2006.tb00081.x.
- ^ "Aspartame and Other Sweeteners in Food". US Food and Drug Administration. 27 February 2025. Retrieved 13 November 2025.
- ^ Scientific Committee on Food (2000). "Opinion - Re-evaluation of acesulfame K with reference to the previous SCF opinion of 1991" (PDF). SCF/CS/ADD/EDUL/194 final. EU Commission. Archived from the original (PDF) on 2008-09-10. Retrieved 2007-10-04.
External links
[edit]- Joint FAO/WHO Expert Committee on Food Additives evaluation monograph of Acesulfame Potassium Archived 2011-09-16 at the Wayback Machine
- FDA approval of Acesulfame Potassium
- FDA approval of Acesulfame Potassium as a General Purpose Sweetener in Food
- Elmhurst College, Illinois Virtual ChemBook Acesulfame K
- Discovery News Sweeteners Linger in Groundwater

