Acetal
View on Wikipedia
In organic chemistry, an acetal is a functional group with the connectivity R2C(OR')2. Here, the R groups can be organic fragments (a carbon atom, with arbitrary other atoms attached to that) or hydrogen, while the R' groups must be organic fragments not hydrogen. The two R' groups can be equivalent to each other (a "symmetric acetal") or not (a "mixed acetal"). Acetals are formed from and convertible to aldehydes or ketones and have the same oxidation state at the central carbon, but have substantially different chemical stability and reactivity as compared to the analogous carbonyl compounds. The central carbon atom has four bonds to it, and is therefore saturated and has tetrahedral geometry.
The term ketal is sometimes used to identify structures associated with ketones (both R groups organic fragments rather than hydrogen) rather than aldehydes and, historically, the term acetal was used specifically for the aldehyde-related cases (having at least one hydrogen in place of an R on the central carbon).[1] The IUPAC originally deprecated the usage of the word ketal altogether, but has since reversed its decision.[citation needed] However, in contrast to historical usage, ketals are now a subset of acetals, a term that now encompasses both aldehyde- and ketone-derived structures.[citation needed]
If one of the R groups has an oxygen as the first atom (that is, there are more than two oxygens single-bonded to the central carbon), the functional group is instead an orthoester. In contrast to variations of R, both R' groups are organic fragments. If one R' is a hydrogen, the functional group is instead a hemiacetal, while if both are H, the functional group is a ketone hydrate or aldehyde hydrate.
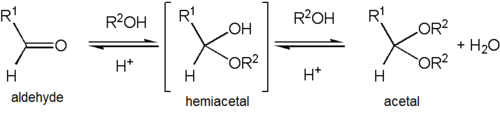
Formation of an acetal occurs when the hydroxyl group of a hemiacetal becomes protonated and is lost as water. The carbocation that is produced is then rapidly attacked by a molecule of alcohol. Loss of the proton from the attached alcohol gives the acetal.
Acetals are stable compared to hemiacetals but their formation is a reversible equilibrium as with esters. As a reaction to create an acetal proceeds, water must be removed from the reaction mixture, for example, with a Dean–Stark apparatus, lest it hydrolyse the product back to the hemiacetal. The formation of acetals reduces the total number of molecules present (carbonyl + 2 alcohol → acetal + water) and therefore is generally not favourable with regards to entropy. One situation where it is not entropically unfavourable is when a single diol molecule is used rather than two separate alcohol molecules (carbonyl + diol → acetal + water).
Acetalization and ketalization
[edit]Acetalization and ketalization are the organic reactions that involve the formation of an acetal (or ketals) from aldehydes and ketones, respectively. These conversions are acid catalysed. They eliminate water. Since each step is often a rapid equilibrium, the reaction must be driven by removal of water. Methods for removing water include azeotropic distillation and trapping water with desiccants like aluminium oxide and molecular sieves. Steps assumed to be involved: protonation of the carbonyl oxygen, addition of the alcohol to the protonated carbonyl, protonolysis of the resulting hemiacetal or hemiketal, and addition of the second alcohol. These steps are illustrated with an aldehyde RCH=O and the alcohol R'OH:
- RCH=O + H+ ⇌ RCH=OH+
- RCH=OH+ + R'OH ⇌ RCH(OH)(OR') + H+
- RCH(OH)(OR') + H+ ⇌ RC+H(OR') + H2O
- RC+H(OR') + R'OH ⇌ RCH(OR')2 + H+
Another way to avoid the entropic cost is to perform the synthesis by acetal exchange (transacetalization), using a pre-existing acetal-type reagent as the OR'-group donor rather than simple addition of alcohols themselves. One type of reagent used for this method is an orthoester. In this case, water produced along with the acetal product is destroyed when it hydrolyses residual orthoester molecules, and this side reaction also produces more alcohol to be used in the main reaction.
Examples
[edit]Sugars
[edit]Since many sugars are polyhydroxy aldehydes and ketones, sugars are a rich source of acetals and ketals. Most glycosidic bonds in carbohydrates and other polysaccharides are acetal linkages.[2] Cellulose is a ubiquitous example of a polyacetal.
Benzylidene acetal and acetonide as protecting groups used in research of modified sugars.
Chiral derivatives
[edit]Acetals also find application as chiral auxiliaries. Indeed acetals of chiral glycols like, e.g. derivatives of tartaric acid can be asymmetrically opened with high selectivity. This enables the construction of new chiral centers.[3]

Formaldehyde and acetaldehyde
[edit]Formaldehyde forms a rich collection of acetals. This tendency reflects the fact that low molecular weight aldehydes are prone to self-condensation such that the C=O bond is replaced by an acetal. The acetal formed from formaldehyde (two hydrogens attached to the central carbon) is sometimes called a formal[4] or the methylenedioxy group. The acetal formed from acetone is sometimes called an acetonide. Formaldehyde forms paraldehyde and 1,3,5-trioxane. Polyoxymethylene (POM) plastic, also known as "acetal" or "polyacetal", is a polyacetal (and a polyether), and a polymer of formaldehyde. Acetaldehyde converts to metaldehyde.
Unusual acetals
[edit]Phenylsulfonylethylidene (PSE) acetal is an example of arylsulfonyl acetal possessing atypical properties, like resistance to acid hydrolysis which leads to selective introduction and removal of the protective group.[5]
Flavors and fragrances
[edit]1,1-Diethoxyethane (acetaldehyde diethyl acetal), sometimes called simply "acetal", is an important flavouring compound in distilled beverages.[6] Two ketals of ethyl acetoacetate are used in commercial fragrances.[7] Fructone (CH3C(O2C2H4)CH2CO2C2H5), an ethylene glycol ketal, and fraistone (CH3C(O2C2H3CH3)CH2CO2C2H5), a propylene glycol ketal, are commercial fragrances.
Related compounds
[edit]Used in a more general sense, the term X,Y-acetal also refers to any functional group that consists of a carbon bearing two heteroatoms X and Y. For example, N,O-acetal refers to compounds of type R1R2C(OR)(NR'2) (R,R' ≠ H) also known as a hemiaminal ether or aminal, a.k.a. aminoacetal.
S,S-acetal refers to compounds of type R1R2C(SR)(SR') (R,R' ≠ H), also known as thioacetal and thioketals.
See also
[edit]References
[edit]- ^ IUPAC, Compendium of Chemical Terminology, 5th ed. (the "Gold Book") (2025). Online version: (2006–) "ketals". doi:10.1351/goldbook.K03376
- ^ IUPAC, Compendium of Chemical Terminology, 5th ed. (the "Gold Book") (2025). Online version: (2006–) "glycosides". doi:10.1351/goldbook.G02661
- ^ Kocieński, Philip J. (1994). Protecting Groups (1st ed.). New York: Georg Thieme Verlag. pp. 164–167. ISBN 3131370017.
- ^ Morrison, Robert T. and Boyd, Robert N., "Organic Chemistry (6th ed)". p683. Prentice-Hall Inc (1992).
- ^ Chéry, Florence; Rollin, Patrick; De Lucchi, Ottorino; Cossu, Sergio (2000). "Phenylsulfonylethylidene (PSE) acetals as atypical carbohydrate-protective groups". Tetrahedron Letters. 41 (14): 2357–2360. doi:10.1016/s0040-4039(00)00199-4. ISSN 0040-4039.
- ^ Maarse, Henk (1991-03-29). Volatile Compounds in Foods and Beverages. CRC Press. ISBN 978-0-8247-8390-7.
- ^ Panten, Johannes; Surburg, Horst (2016). "Flavors and Fragrances, 3. Aromatic and Heterocyclic Compounds". Ullmann's Encyclopedia of Industrial Chemistry. pp. 1–45. doi:10.1002/14356007.t11_t02. ISBN 978-3-527-30673-2.
Acetal
View on GrokipediaChemical Structure and Nomenclature
General Structure
Acetals are geminal diether functional groups derived from aldehydes or ketones, characterized by the general molecular formula $ R_2C(OR')_2 $, where the $ R $ groups are hydrogen, alkyl, or aryl substituents from the original carbonyl compound, and the $ R' $ groups are alkyl or aryl groups from the alcohols used in their derivation.[6] This structure arises from the addition of two alcohol molecules to the carbonyl carbon, with loss of water, resulting in a carbon atom bonded to two oxygen atoms via single bonds.[6] The key structural feature of acetals is the central carbon atom, known as the acetal carbon, which has two alkoxy groups ($ -OR' $) attached geminally—meaning both are linked to the same carbon—effectively supplanting the double-bonded oxygen of the parent carbonyl.[6] This contrasts sharply with the planar $ C=O $ unit in aldehydes and ketones, where the carbonyl carbon is $ sp^2 $ hybridized. In acetals, the central carbon adopts $ sp^3 $ hybridization, leading to a tetrahedral arrangement with bond angles close to 109.5°.[7][8] Acetals exist in both acyclic and cyclic forms, depending on whether the alkoxy groups originate from separate alcohol molecules or from a single diol that forms a ring. Acyclic acetals feature open-chain structures, such as 1,1-dimethoxyethane ($ \ce{CH3CH(OCH3)2} $), which is the dimethyl acetal derived from acetaldehyde and methanol.[9] Cyclic acetals, by contrast, incorporate the two oxygen atoms into a ring system, often five- or six-membered for stability, as seen in 1,3-dioxolanes formed from aldehydes and ethylene glycol.[6] These structural variations influence their stability and utility but maintain the core gem-diether motif.Naming Conventions
Acetals are systematically named according to IUPAC recommendations using substitutive nomenclature, where the compounds are treated as derivatives of the parent hydrocarbon chain with alkoxy or aryloxy substituents. For acyclic acetals derived from aldehydes or ketones, the general structure R-CH(OR')₂ or R₂C(OR')₂ is named as a dialkoxyalkane, with the parent chain selected to include the carbon atom bearing the two oxygen atoms, numbered to give the lowest locants to these substituents. For example, the compound CH₃CH(OCH₂CH₃)₂ is named 1,1-diethoxyethane.[10] The term "acetal" originally referred specifically to derivatives of aldehydes (where one R group is hydrogen), while derivatives of ketones (both R groups alkyl or aryl) were called "ketals," but modern IUPAC nomenclature unifies both under the term "acetal" to reflect their structural and functional similarities.[11] This distinction persists in some educational and older literature but is not used in preferred IUPAC names. Cyclic acetals, formed typically from 1,2- or 1,3-diols, are named as heterocyclic compounds, such as 1,3-dioxolane for the five-membered ring from ethylene glycol or 1,3-dioxane for the six-membered ring from a 1,3-diol such as propane-1,3-diol, with substituents numbered starting from the acetal carbon as position 2.[12] Common names for acetals often derive from the carbonyl precursor and the alcohol used, providing a historical shorthand. The diethyl acetal of acetaldehyde, CH₃CH(OCH₂CH₃)₂, is simply called "acetal," while formaldehyde-derived acetals are termed "formals," such as dimethoxymethane known as methylal. These names are retained in IUPAC for general use but are not preferred for indexing.[13][14] In naming substituted acetals, additional groups are prefixed to the parent name with locants that ensure the acetal functionality receives the lowest possible numbers, following standard rules for chain selection and functional group priority. For chiral acetals, where the central carbon or other centers introduce asymmetry, stereodescriptors such as (R) or (S) are incorporated according to Cahn-Ingold-Prelog rules, as in [(4R)-2,2-dimethyl-1,3-dioxolan-4-yl]methanol for a specific enantiomer.[10][11]Formation and Hydrolysis
Acetalisation Mechanism
The acetalisation mechanism describes the acid-catalyzed reaction of an aldehyde with two equivalents of an alcohol to form an acetal, a process that proceeds through reversible steps under equilibrium conditions.[15][16] The overall reaction is represented as:Ketalisation Mechanism
Ketalisation is the acid-catalyzed reaction of a ketone with two molecules of an alcohol to form a ketal, releasing a molecule of water. The general equation is:Properties
Chemical Reactivity
Acetals exhibit distinctive reactivity primarily governed by their geminal diether structure, which renders them stable under neutral and basic conditions but highly susceptible to acid-catalyzed transformations. The most prominent reaction is acid-catalyzed hydrolysis, which reverses the formation process to yield the parent carbonyl compound and two equivalents of alcohol. This equilibrium reaction is represented as:Physical Characteristics
Acetals exhibit low polarity due to their geminal diether structure, resulting in physical properties akin to those of ethers, such as appearing as volatile liquids or low-melting solids at room temperature.[29] Their boiling points are generally similar to those of ethers with comparable molecular weights, reflecting moderate intermolecular forces without strong hydrogen bonding. For instance, 1,1-diethoxyethane (commonly known as diethyl acetal) boils at 102–104 °C. Solubility characteristics of acetals align with their nonpolar nature, rendering them highly soluble in organic solvents like alcohols, ethers, and hydrocarbons, but sparingly soluble in water. Smaller acetals, however, show greater hydrophilicity; dimethoxymethane (methyl formal), for example, is miscible with water to the extent of approximately 330 g/L at 20 °C. In contrast, larger homologs like 1,1-diethoxyethane exhibit limited water solubility of about 46 g/L. Spectroscopically, acetals lack the characteristic infrared absorption band for the carbonyl stretch observed in their precursor aldehydes or ketones at around 1720 cm⁻¹, confirming the absence of the C=O group. In ¹H NMR spectra, the methine proton attached to the acetal carbon (R-CH(OR')₂) typically appears in the deshielded region of δ 4.3–4.8 ppm, influenced by the adjacent oxygen atoms.[30] Common acetals also display densities close to those of ethers, typically in the range of 0.8–0.9 g/cm³, with viscosities comparable to light organic liquids (around 0.5–1 cP at 20 °C). The following table summarizes key physical data for two representative acetals:| Compound | Boiling Point (°C) | Density (g/cm³ at 20 °C) | Water Solubility (g/L at 20 °C) |
|---|---|---|---|
| Dimethoxymethane | 42 | 0.864 | 330 |
| 1,1-Diethoxyethane | 102–104 | 0.831 | 46 |