Anopsia
View on Wikipediafrom Wikipedia
| Anopsia | |
|---|---|
| Other names | Anopia |
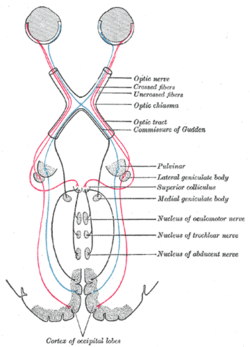 | |
| Scheme showing central connections of the optic nerves and optic tracts. (Lesions at different locations relate to different types of anopsias.) | |
| Specialty | Neurology |
An anopsia, or anopia, (from Ancient Greek ἀν- (an-) 'without' and ὄψις (opsis) 'sight') is a defect in the visual field. If the defect is only partial, then the portion of the field with the defect can be used to isolate the underlying cause. It is particularly used to describe the lack of sight in one eye.[1]
Variants
[edit]Types of partial anopsia include:
References
[edit]- ^ Stanaszek, Walter F.; Stanaszek, Mary J.; Holt, Robert J.; Strauss, Steven (2020-06-29). Understanding Medical Terms: A Guide for Pharmacy Practice (2 ed.). Routledge. doi:10.4324/9780429273032. ISBN 978-0-429-27303-2.
External links
[edit]Anopsia
View on Grokipediafrom Grokipedia
