Recent from talks
Nothing was collected or created yet.
Fire coral
View on WikipediaThis article needs additional citations for verification. (August 2025) |
| Fire coral | |
|---|---|

| |
| Millepora dichotoma | |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Cnidaria |
| Class: | Hydrozoa |
| Order: | Anthoathecata |
| Suborder: | Capitata |
| Family: | Milleporidae Fleming, 1828 |
| Genus: | Millepora Linnaeus, 1758 |
| Diversity | |
| 15 species | |
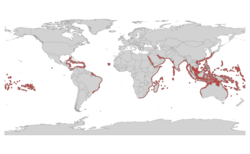
| |
| Fire coral range | |
| Synonyms | |
|
(Family)
(Genus)
| |
Fire corals (Millepora) are a genus of colonial marine organisms that exhibit physical characteristics similar to that of coral. The name coral is somewhat misleading, as fire corals are not true corals but are instead more closely related to Hydra and other hydrozoans, making them hydrocorals. They make up the only genus in the monotypic family Milleporidae.
Anatomy and reproduction
[edit]While most fire corals are yellow or orange, they can also be found in shades of brown, green, and even blue, providing a vibrant display underwater.[1]
Fire coral has several common growth forms; these include branching, plate, and encrusting. Branching fire coral adopts a calcareous structure which branches off into rounded, finger-like tips. Plate-growing fire coral forms a shape similar to that of fellow cnidarian lettuce corals - erect, thin sheets, which group together to form a colony. In encrusting fire coral, growth takes place on the surface structure of calcareous coral or gorgonian structures.[2]
The gonophores in the family Milleporidae arise from the coenosarc (the hollow living tubes of the upright branching individuals of a colony), within chambers embedded entirely in the coenosteum (the calcareous mass forming the skeleton of a compound coral).
Reproduction in fire corals is more complex than in other reef-building corals. The polyp of fire coral releases a medusa that releases its eggs in the water stream. Then another male medusa fertilizes the eggs with its sperm, which then produces a planula.[3] A planula then floats in the water until it finds a reef it is able to attach to and grow back into a polyp, settling on a hard surface. Then the cycle repeats.[4]
Habitat and predators
[edit]Fire corals are found on reefs in tropical and subtropical waters, such as the Indian Ocean, Pacific Ocean, and Atlantic Ocean and the Caribbean Sea.[5][6] They are found in shallow reefs where the most amount of sunlight is able to reach them, allowing for a higher rate of photosynthesis of the algae that lives in their tissues. Fire corals thrive in an environment with a high, strong current, and warm water.[3] They are found in almost all places in the world, except for cold coastal regions. They are also abundant on upper reef slopes and in lagoons, and occur down to 40 meters (131 ft) deep.
Fire corals' predatory threats are mainly from fire worms, certain nudibranchs, and filefish.[7] They are predators to the algae that lives within them, and zooplankton/phytoplankton.[3]
Biology and behaviors
[edit]The polyps of fire corals are near microscopic size and are mostly embedded in the skeleton and connected by a network of minute canals.[8] All that is visible on the smooth surface are pores of two sizes: gastropores and dactylopores. In fact, Millepora means ‘thousand pores’. Dactylozooids have long fine hairs that protrude from the skeleton. The hairs possess clusters of stinging cells and capture prey, which is then engulfed by gastrozooids, or feeding polyps, situated within the gastropores. As well as capturing prey, fire corals gain nutrients via their special symbiotic relationship with algae known as zooxanthellae. The zooxanthellae live inside the tissues of the coral, and provide the coral with food, which they produce through photosynthesis, so require sunlight. In return, the coral provides the algae with protection and access to sunlight. The hollow tubes in fire coral can also be used to store oxygen to offset any organism that bumps into it.[3]
Stings, symptoms, and treatments
[edit]
Upon contact, an intense pain can be felt, lasting from two days to two weeks. Occasional relapses of post-treatment inflammation are common. Prominent side effects can include skin irritation, stinging or burning pain, erythema (skin redness), fever, and/or urticarian (hives) lesions. These side effects are due to venom released from the nematocyte, as venom is part of the defense mechanism of the fire coral. Despite its mild to moderate potential for pain, the venom is nonlethal to humans. The very small nematocysts on fire corals contain tentacles, protruding from numerous surface pores (similar to jellyfish stingers). In addition, fire corals have a sharp, calcified external skeleton that can scrape the skin.[medical citation needed]
The following treatments are suggestions; always seek a medical professional first.
- Rinse with seawater. Freshwater will cause the cnidae to release more venom, which will increase pain, so stay clear of freshwater.
- Apply vinegar or isopropyl alcohol. This helps to deactivate the venom.
- Heat can also help to deactivate the venom.
- Remove any parts of the fire coral; tweezers and tape work very well.
- Keep the infected area still because movement can cause the venom to spread.
- Apply hydrocortisone cream two to three times daily as needed for itching. Stop immediately if any signs of infection appear.
Again, these are just suggestions; always seek a medical professional first.[8][medical citation needed]
Threats and conservation
[edit]Fire corals face the many threats impacting coral reefs globally, including poor land management practices releasing more sediment, nutrients, and pollutants into the oceans and stressing the fragile reef ecosystem. Overfishing has ‘knock-on’ effects that result in the increase of macroalgae that can outcompete and smother corals, and fishing using destructive methods physically devastates the reef. A further potential threat is the increase of coral bleaching events, as a result of global climate change.[9]
Coral bleaching is also a major threat to all types of coral. Coral bleaching is when the coral expels the zooxanthella that they feed on, which causes them to turn white, hence "bleaching." Corals can not live long in this state, yet if environmental conditions return to normal, then the zooxanthella can return and the coral will return healthy again.[4]
Most fire coral species have brittle skeletons that can easily be broken, for example, during storms, or by divers when diving for leisure, or when collecting fish for the aquarium trade. For instance, the yellowtail damselfish (Chrysiptera parasema) tends to dwell close to the branching fire coral colonies, and retreats into its branches when threatened. In Brazil, fire coral colonies are extensively damaged when harvesting the yellowtail damselfish, as the corals are often deliberately smashed and fishes hiding amongst the branches are ‘shaken out’ into plastic bags.[10]
Fire corals are listed on Appendix II of the Convention on International Trade in Endangered Species (CITES).[11]
Species
[edit]
Sixteen species of Millepora are currently recognised:[12]
- Millepora alcicornis Linnaeus, 1758
- Millepora boschmai de Weerdt & Glynn, 1991
- Millepora braziliensis Verrill, 1868
- Millepora complanata Lamarck, 1816
- Millepora dichotoma (Forsskål, 1775)
- Millepora exaesa (Forsskål, 1775)
- Millepora foveolata Crossland, 1952
- Millepora intricata Milne-Edwards & Haime, 1860
- Millepora laboreli Amaral, 2008
- Millepora latifolia Boschma, 1948
- Millepora nitida Verrill, 1868
- Millepora nodulosa Nemenzo, 1984
- Millepora platyphylla Hemprich & Ehrenberg, 1834
- Millepora squarrosa Lamarck, 1816
- Millepora striata Duchassaing & Michelotti, 1864
- Millepora tenera Boschma, 1948
Further reading
[edit]- W. H. de Weerdt & P. W. Glynn (1991). "A new and presumably now extinct species of Millepora (Hydrozoa) in the eastern Pacific" (PDF). Zoologische Mededelingen. 65 (20): 267–276.
- Idaz Greenberg (1986). Guide to Corals & Fishes of Florida, the Bahamas and the Caribbean. Seahawk Press. p. 60. ISBN 978-0-913008-08-9.
References
[edit]- ^ Cornwell, Mureil (March 6, 2024). Smith, Sherman (ed.). "19 Fire Coral Facts". Facts.net. Retrieved April 27, 2024.
- ^ "The Fire Corals". Aquarium Net. October 1996. Archived from the original on 2008-02-06. Retrieved 2007-07-03.
- ^ a b c d "Fire Coral". Lamar University. April 27, 2024. Retrieved April 27, 2024.
- ^ a b "What is Coral Bleaching and Why Should You Care?". Coral Reef Alliance. March 17, 2023. Retrieved April 27, 2024.
- ^ Kropp, M.L. Parsley B. C. Burnett, Lee Omer. (2018). Millepora species(Fire Coral) Sting: A Case Reportand Review of Recommended Management'. Vol. 29. Issue. 4. Wilderness and Environmental Medicine.
- ^ Veron, J.E.N. (2000) Corals of the World. Vol. 3. Australian Institute of Marine Sciences, Townsville, Australia.
- ^ "Fire Coral". Reef Smart Guides. April 27, 2024.
- ^ a b Endrizzi, Mike (April 27, 2024). "Fire Coral". Retrieved April 27, 2024.
- ^ Wilkinson, C. (2004) Status of Coral Reefs around the Word. Australian Institute of Marine Science, Townsville, Australia.
- ^ Gasparini, J.L., Floeter, S.R., Ferreira, C.E.L. and Sazima, I. (2005) Marine ornamental trade in Brazil. Biodiversity and Conservation, 14: 2883 – 2899.
- ^ CITES: Appendices I, II and III Archived November 16, 2008, at the Wayback Machine Retrieved 2011-08-24.
- ^ "WoRMS - World Register of Marine Species - Millepora Linnaeus, 1758". www.marinespecies.org. Retrieved 2018-02-17.
External links
[edit]Fire coral
View on GrokipediaTaxonomy and classification
Phylogenetic position and distinction from true corals
Fire corals of the genus Millepora occupy a distinct phylogenetic position within the phylum Cnidaria, specifically in the class Hydrozoa under the subclass Hydroidolina and order Anthoathecata (formerly Capitata), family Milleporidae.[6][5] This places them in the subphylum Medusozoa, which includes organisms capable of producing both polyp and medusa stages in their life cycles, contrasting with the exclusively polypoid Anthozoa.[7] Molecular studies, including transcriptome analyses of species like Millepora alcicornis and Millepora complanata, confirm their deep divergence from anthozoans, with Milleporidae forming a monophyletic group within hydrozoans that diverged approximately 500 million years ago during the Cambrian period.[8][9] In contrast to true corals, which belong to the class Anthozoa and order Scleractinia, fire corals lack the defining anthozoan traits such as bilateral symmetry in polyps and septate corallites.[10] Scleractinian corals produce rigid skeletons via aragonite deposition within individual corallites housing solitary or colonial polyps with six-fold radial symmetry and tentacles in multiples of six.[11] Fire corals, as hydrozoans, exhibit a coenosteum—a continuous calcareous skeleton perforated by scattered gastropores (for feeding gastrozooids) and dactylopores (for defensive dactylozooids), without discrete corallites or septa.[12][13] Their polyps are dimorphic and microscopic, integrated into a shared living tissue (coenosarc) that lacks the muscular retractor systems typical of anthozoan polyps.[7]| Feature | Fire Corals (Millepora, Hydrozoa) | True Corals (Scleractinia, Anthozoa) |
|---|---|---|
| Class | Hydrozoa | Anthozoa |
| Polyp Organization | Dimorphic (gastrozooids, dactylozooids); microscopic, in pores | Monomorphic or dimorphic; larger, in corallites |
| Skeleton Structure | Porous coenosteum, no septa | Septate corallites with aragonite walls |
| Symmetry | Variable, often irregular | Six-fold radial |
| Life Cycle Stages | Polyp and medusa (medusae rare) | Polyp only |
Etymology and historical classification
The common name "fire coral" derives from the intense, burning sensation caused by contact with the organism's nematocysts, which deliver a painful sting comparable to a burn.[7] This vernacular term reflects empirical observations of its defensive mechanism rather than any thermal property, as the colonies lack heat-generating capabilities.[3] The genus name Millepora, established by Carl Linnaeus in Systema Naturae (10th edition, 1758), originates from Latin roots: mille meaning "thousand" and pora (from Greek poros, "pore"), alluding to the multitude of small pores visible on the calcareous skeleton, known as the coenosteum.[16] These pores include gastropores for gastrozooids and dactylopores for dactylozooids, distinguishing the structure from that of true corals.[7] Historically, Millepora species were misclassified among stony corals (class Anthozoa) due to their rigid, branching calcareous skeletons and reef-building morphology, which superficially resembled scleractinians.[3] Linnaeus initially placed the genus within Madrepora, a polyphyletic group encompassing various calcifying cnidarians.[16] By the early 19th century, advancements in microscopy revealed distinct polyp dimorphism—feeding gastrozooids and defensive dactylozooids—prompting reclassification as hydrozoans (class Hydrozoa, order Anthoathecata).[7] The family Milleporidae was formally defined by John Fleming in 1828, solidifying their separation from anthozoans based on reproductive and skeletal evidence.[17] This distinction, confirmed through comparative anatomy, underscores that fire corals are more closely related to jellyfish than to scleractinian corals, despite ecological convergence in tropical reefs.[3]Accepted species
The genus Millepora contains 14 accepted species according to the World Register of Marine Species (WoRMS), a peer-reviewed database of marine taxonomy.[18] These species exhibit varying morphologies and distributions, primarily in tropical and subtropical waters, though taxonomic revisions continue due to phenotypic plasticity and limited molecular data, with some sources recognizing 16–19 species.[14] [8] The accepted species are:- Millepora alcicornis Linnaeus, 1758 (widespread in the Atlantic)[18]
- Millepora boschmai de Weerdt & Glynn, 1991 (Indo-Pacific)[18]
- Millepora complanata Lamarck, 1816 (Atlantic and Indo-Pacific)[18]
- Millepora dichotoma Forsskål, 1775 (Red Sea and Indo-Pacific)[18]
- Millepora exaesa Forsskål, 1775 (Indo-Pacific, including type locality in the Red Sea)[18]
- Millepora foveolata d'Orbigny, 1828 (Caribbean)[18]
- Millepora intricata Milne Edwards & Haime, 1851 (Indo-Pacific)[18]
- Millepora latifolia Boschma, 1948 (Indo-Pacific)[18]
- Millepora murrayi Ridley, 1884 (Indo-Pacific)[18]
- Millepora nitida Verrill, 1868 (Caribbean)[18]
- Millepora platyphylla Hemprich & Ehrenberg, 1834 (Indo-Pacific)[18]
- Millepora squarrosa Lamarck, 1816 (Atlantic)[18]
- Millepora striata Duchassaing & Michelotti, 1864 (Caribbean)[18]
- Millepora tenera Boschma, 1948 (Indo-Pacific)[18]

