Recent from talks
Nothing was collected or created yet.
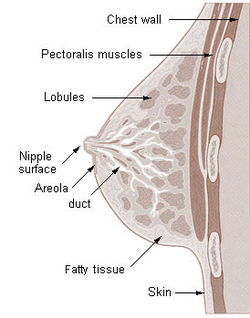
Mammary gland
View on Wikipedia
| Mammary gland | |
|---|---|
 Cross-section of the human mammary gland: | |
| Details | |
| Precursor | Mesoderm (blood vessels and connective tissue) Ectoderm[3] (cellular elements) |
| Artery | Internal thoracic artery Lateral thoracic artery[1] |
| Vein | Internal thoracic vein Axillary vein[1] |
| Nerve | Supraclavicular nerves Intercostal nerves[2] (lateral and medial branches) |
| Lymph | Pectoral axillary lymph nodes[1] |
| Identifiers | |
| TA98 | A16.0.02.006 |
| TA2 | 7099 |
| FMA | 60088 |
| Anatomical terminology | |
A mammary gland is an exocrine gland that produces milk in humans and other mammals. Mammals get their name from the Latin word mamma, "breast". The mammary glands are arranged in organs such as the breasts in primates (for example, humans and chimpanzees), the udder in ruminants (for example, cows, goats, sheep, and deer), and the dugs of other animals (for example, dogs and cats) to feed young offspring. Lactorrhea, the occasional production of milk by the glands, can occur in any mammal, but in most mammals, lactation, the production of enough milk for nursing, occurs only in phenotypic females who have gestated in recent months or years. It is directed by hormonal guidance from sex steroids. In a few mammalian species, male lactation can occur. With humans, male lactation can occur only under specific circumstances.
Mammals are divided into 3 groups: monotremes, metatherians, and eutherians. In the case of monotremes, their mammary glands are modified sebaceous glands and without nipples. Concerning most metatherians and eutherians, only females have functional mammary glands, with the exception of some bat species. Their mammary glands can be termed as breasts or udders. In the case of breasts, each mammary gland has its own nipple (e.g., human mammary glands). In the case of udders, pairs of mammary glands comprise a single mass, with more than one nipple (or teat) hanging from it. For instance, cows and buffalo udders have two pairs of mammary glands and four teats, whereas sheep and goat udders have one pair of mammary glands with two teats protruding from the udder. Each mammary gland produces milk for a single teat and is evolutionarily derived from modified sweat glands.
Structure
[edit]The basic components of a mature mammary gland are the alveoli (hollow cavities, a few millimeters large), which are lined with milk-secreting cuboidal cells and surrounded by myoepithelial cells. These alveoli join to form groups known as lobules. Each lobule has a lactiferous duct that drains into openings in the nipple. The myoepithelial cells contract under the stimulation of oxytocin, excreting the milk secreted by alveolar units into the lobule lumen toward the nipple. As the infant begins to suck, the oxytocin-mediated "let down reflex" ensues, and the mother's milk is secreted—not sucked—from the gland into the infant's mouth.[4]
All the milk-secreting tissue leading to a single lactiferous duct is collectively called a "simple mammary gland"; in a "complex mammary gland", all the simple mammary glands serve one nipple. Humans normally have two complex mammary glands, one in each breast, and each complex mammary gland consists of 10–20 simple glands. The opening of each simple gland on the surface of the nipple is called a "pore."[5] The presence of more than two nipples is known as polythelia and the presence of more than two complex mammary glands as polymastia.
Maintaining the correct polarized morphology of the lactiferous duct tree requires another essential component – mammary epithelial cells extracellular matrix (ECM) which, together with adipocytes, fibroblast, inflammatory cells, and others, constitute mammary stroma.[6] Mammary epithelial ECM mainly contains myoepithelial basement membrane and the connective tissue. They not only help to support mammary basic structure, but also serve as a communicating bridge between mammary epithelia and their local and global environment throughout this organ's development.[7][8]
Histology
[edit]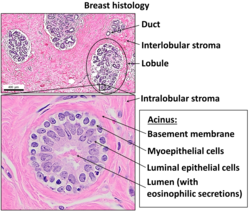

A mammary gland is a specific type of apocrine gland specialized for manufacture of colostrum (first milk) when giving birth. Mammary glands can be identified as apocrine because they exhibit striking "decapitation" secretion. Many sources assert that mammary glands are modified sweat glands.[9][10][11]
Development
[edit]Mammary glands develop during different growth cycles. They exist in both sexes during the embryonic stage, forming only a rudimentary duct tree at birth. In this stage, mammary gland development depends on systemic (and maternal) hormones,[6] but is also under the (local) regulation of paracrine communication between neighboring epithelial and mesenchymal cells by parathyroid hormone-related protein (PTHrP).[12] This locally secreted factor gives rise to a series of outside-in and inside-out positive feedback between these two types of cells, so that mammary bud epithelial cells can proliferate and sprout down into the mesenchymal layer until they reach the fat pad to begin the first round of branching.[6] At the same time, the embryonic mesenchymal cells around the epithelial bud receive secreting factors activated by PTHrP, such as BMP4. These mesenchymal cells can transform into a dense, mammary-specific mesenchyme, which later develop into connective tissue with fibrous threads, forming blood vessels and the lymph system.[13] A basement membrane, mainly containing laminin and collagen, formed afterward by differentiated myoepithelial cells, keeps the polarity of this primary duct tree. These components of the extracellular matrix are strong determinants of duct morphogenesis.[14]
Biochemistry
[edit]Estrogen and growth hormone (GH) are essential for the ductal component of mammary gland development, and act synergistically to mediate it.[15][16][17][18][19] Neither estrogen nor GH are capable of inducing ductal development without the other.[16][17][18][19] The role of GH in ductal development has been found to be mostly mediated by its induction of the secretion of insulin-like growth factor 1 (IGF-1), which occurs both systemically (mainly originating from the liver) and locally in the mammary fat pad through activation of the growth hormone receptor (GHR).[16][17][18][19][20] However, GH itself also acts independently of IGF-1 to stimulate ductal development by upregulating estrogen receptor (ER) expression in mammary gland tissue, which is a downstream effect of mammary gland GHR activation.[19] In any case, unlike IGF-1, GH itself is not essential for mammary gland development, and IGF-1 in conjunction with estrogen can induce normal mammary gland development without the presence of GH.[19] In addition to IGF-1, other paracrine growth factors such as epidermal growth factor (EGF), transforming growth factor beta (TGF-β),[21] amphiregulin,[22] fibroblast growth factor (FGF), and hepatocyte growth factor (HGF)[23] are involved in breast development as mediators downstream to sex hormones and GH/IGF-1.[24][25][26]
During embryonic development, IGF-1 levels are low, and gradually increase from birth to puberty.[27] At puberty, the levels of GH and IGF-1 reach their highest levels in life and estrogen begins to be secreted in high amounts in females, which is when ductal development mostly takes place.[27] Under the influence of estrogen, stromal and fat tissue surrounding the ductal system in the mammary glands also grows.[28] After puberty, GH and IGF-1 levels progressively decrease, which limits further development until pregnancy, if it occurs.[27] During pregnancy, progesterone and prolactin are essential for mediating lobuloalveolar development in estrogen-primed mammary gland tissue, which occurs in preparation of lactation and nursing.[15][29]
Androgens such as testosterone inhibit estrogen-mediated mammary gland development (e.g., by reducing local ER expression) through activation of androgen receptors expressed in mammary gland tissue,[29][30] and in conjunction with relatively low estrogen levels, are the cause of the lack of developed mammary glands in males.[31]
Timeline
[edit]Before birth
[edit]Mammary gland development is characterized by the unique process by which the epithelium invades the stroma. The development of the mammary gland occurs mainly after birth. During puberty, tubule formation is coupled with branching morphogenesis which establishes the basic arboreal network of ducts emanating from the nipple.[32]
Developmentally, mammary gland epithelium is constantly produced and maintained by rare epithelial cells, dubbed as mammary progenitors which are ultimately thought to be derived from tissue-resident stem cells.[33]
Embryonic mammary gland development can be divided into a series of specific stages. Initially, the formation of the milk lines that run between the fore and hind limbs bilaterally on each side of the midline occurs around embryonic day 10.5 (E10.5). The second stage occurs at E11.5 when placode formation begins along the mammary milk line. This will eventually give rise to the nipple. Lastly, the third stage occurs at E12.5 and involves the invagination of cells within the placode into the mesenchyme, leading to a mammary anlage (biology).[34]
The primitive (stem) cells are detected in embryo and their numbers increase steadily during development[35]
Growth
[edit]Postnatally, the mammary ducts elongate into the mammary fat pad. Then, starting around four weeks of age, mammary ductal growth increases significantly with the ducts invading towards the lymph node. Terminal end buds, the highly proliferative structures found at the tips of the invading ducts, expand and increase greatly during this stage. This developmental period is characterized by the emergence of the terminal end buds and lasts until an age of about 7–8 weeks.
By the pubertal stage, the mammary ducts have invaded to the end of the mammary fat pad. At this point, the terminal end buds become less proliferative and decrease in size. Side branches form from the primary ducts and begin to fill the mammary fat pad. Ductal development decreases with the arrival of sexual maturity and undergoes estrous cycles (proestrus, estrus, metestrus, and diestrus). As a result of estrous cycling, the mammary gland undergoes dynamic changes where cells proliferate and then regress in an ordered fashion.[36]
Pregnancy
[edit]During pregnancy, the ductal systems undergo rapid proliferation and form alveolar structures within the branches to be used for milk production. After delivery, lactation occurs within the mammary gland; lactation involves the secretion of milk by the luminal cells in the alveoli. Contraction of the myoepithelial cells surrounding the alveoli will cause the milk to be ejected through the ducts and into the nipple for the nursing infant. Upon weaning of the infant, lactation stops and the mammary gland turns in on itself, a process called involution. This process involves the controlled collapse of mammary epithelial cells where cells begin apoptosis in a controlled manner, reverting the mammary gland back to a pubertal state.
Postmenopausal
[edit]During postmenopause, due to much lower levels of estrogen, and due to lower levels of GH and IGF-1, which decrease with age, mammary gland tissue atrophies and the mammary glands become smaller.
Physiology
[edit]Hormonal control
[edit]Lactiferous duct development occurs in females in response to circulating hormones. First development is frequently seen during pre- and postnatal stages, and later during puberty. Estrogen promotes branching differentiation,[37] whereas in males testosterone inhibits it. A mature duct tree reaching the limit of the fat pad of the mammary gland comes into being by bifurcation of duct terminal end buds (TEB), secondary branches sprouting from primary ducts[7][38] and proper duct lumen formation. These processes are tightly modulated by components of mammary epithelial ECM interacting with systemic hormones and local secreting factors. However, for each mechanism the epithelial cells' "niche" can be delicately unique with different membrane receptor profiles and basement membrane thickness from specific branching area to area, so as to regulate cell growth or differentiation sub-locally.[39] Important players include beta-1 integrin, epidermal growth factor receptor (EGFR), laminin-1/5, collagen-IV, matrix metalloproteinase (MMPs), heparan sulfate proteoglycans, and others. Elevated circulating level of growth hormone and estrogen get to multipotent cap cells on TEB tips through a thin, leaky layer of basement membrane. These hormones promote specific gene expression. Hence cap cells can differentiate into myoepithelial and luminal (duct) epithelial cells, and the increased amount of activated MMPs can degrade surrounding ECM helping duct buds to reach further in the fat pads.[40][41] On the other hand, basement membrane along the mature mammary ducts is thicker, with strong adhesion to epithelial cells via binding to integrin and non-integrin receptors. When side branches develop, it is a much more "pushing-forward" working process including extending through myoepithelial cells, degrading basement membrane and then invading into a periductal layer of fibrous stromal tissue.[7] Degraded basement membrane fragments (laminin-5) roles to lead the way of mammary epithelial cells migration.[42] Whereas, laminin-1 interacts with non-integrin receptor dystroglycan negatively regulates this side branching process in case of cancer.[43] These complex "Yin-yang" balancing crosstalks between mammary ECM and epithelial cells "instruct" healthy mammary gland development until adult.
There is preliminary evidence that soybean intake mildly stimulates the breast glands in pre- and postmenopausal women.[44]
Pregnancy
[edit]Secretory alveoli develop mainly in pregnancy, when rising levels of prolactin, estrogen, and progesterone cause further branching, together with an increase in adipose tissue and a richer blood flow. In gestation, serum progesterone remains at a stably high concentration so signaling through its receptor is continuously activated. As one of the transcribed genes, Wnts secreted from mammary epithelial cells act paracrinely to induce more neighboring cells' branching.[45][46] When the lactiferous duct tree is almost ready, "leaves" alveoli are differentiated from luminal epithelial cells and added at the end of each branch. In late pregnancy and for the first few days after giving birth, colostrum is secreted. Milk secretion (lactation) begins a few days later due to reduction in circulating progesterone and the presence of another important hormone prolactin, which mediates further alveologenesis, milk protein production, and regulates osmotic balance and tight junction function. Laminin and collagen in myoepithelial basement membrane interacting with beta-1 integrin on epithelial surface again, is essential in this process.[47][48] Their binding ensures correct placement of prolactin receptors on the basal lateral side of alveoli cells and directional secretion of milk into lactiferous ducts.[47][48] Suckling of the baby causes release of the hormone oxytocin, which stimulates contraction of the myoepithelial cells. In this combined control from ECM and systemic hormones, milk secretion can be reciprocally amplified so as to provide enough nutrition for the baby.
Weaning
[edit]During weaning, decreased prolactin, missing mechanical stimulation (baby suckling), and changes in osmotic balance caused by milk stasis and leaking of tight junctions cause cessation of milk production. It is the (passive) process of a child or animal ceasing to be dependent on the mother for nourishment. In some species there is complete or partial involution of alveolar structures after weaning, in humans there is only partial involution and the level of involution in humans appears to be highly individual. The glands in the breast do secrete fluid also in nonlactating women.[49] In some other species (such as cows), all alveoli and secretory duct structures collapse by programmed cell death (apoptosis) and autophagy for lack of growth promoting factors either from the ECM or circulating hormones.[50][51] At the same time, apoptosis of blood capillary endothelial cells speeds up the regression of lactation ductal beds. Shrinkage of the mammary duct tree and ECM remodeling by various proteinase is under the control of somatostatin and other growth inhibiting hormones and local factors.[52] This major structural change leads loose fat tissue to fill the empty space afterward. But a functional lactiferous duct tree can be formed again when a female is pregnant again.
Clinical significance
[edit]Tumorigenesis in mammary glands can be induced biochemically by abnormal expression level of circulating hormones or local ECM components,[53] or from a mechanical change in the tension of mammary stroma.[54] Under either of the two circumstances, mammary epithelial cells would grow out of control and eventually result in cancer. Almost all instances of breast cancer originate in the lobules or ducts of the mammary glands.
Other mammals
[edit]General
[edit]The breasts of female humans vary from most other mammals that tend to have less conspicuous mammary glands. The number and positioning of mammary glands varies widely in different mammals. The protruding teats and accompanying glands can be located anywhere along the two milk lines. In general most mammals develop mammary glands in pairs along these lines, with a number approximating the number of young typically birthed at a time. The number of teats varies from 2 (in most primates) to 18 (in pigs). The Virginia opossum has 13, one of the few mammals with an odd number.[55][56] The following table lists the number and position of teats and glands found in a range of mammals:
| Species[57] | Anterior (thoracic) |
Intermediate (abdominal) |
Posterior (inguinal) |
Total |
|---|---|---|---|---|
| Goat, sheep, horse guinea pig |
0 | 0 | 2 | 2 |
| Cattle | 0 | 0 | 4 | 4 |
| Cat | 2 | 2 | 4 | 8 |
| Dog[58] | 4 | 2 | 2 or 4 | 8 or 10 |
| Mouse | 6 | 0 | 4 | 10 |
| Rat | 6 | 2 | 4 | 12 |
| Pig | 6 | 6 | 6 | 18 |
| Proboscideans, primates | 2 | 0 | 0 | 2 |
| Virginia opossum[55][56] | 0 | 0 | 13 | 13 |
| Southern red-sided opossum[59] | 0 | 0 | 25 to 27 | 25 to 27 |
Male mammals typically have rudimentary mammary glands and nipples, with a few exceptions: male mice do not have nipples,[60] male marsupials do not have mammary glands,[61] and male horses lack nipples.[62] The male dayak fruit bat has lactating mammary glands.[63] Male lactation occurs infrequently in some species.[64]
Mammary glands are true protein factories,[65] and several labs have constructed transgenic animals, mainly goats and cows, to produce proteins for pharmaceutical use.[66] Complex glycoproteins such as monoclonal antibodies or antithrombin cannot be produced by genetically engineered bacteria, and the production in live mammals is much cheaper than the use of mammalian cell cultures.
Evolution
[edit]There are many theories on how mammary glands evolved. For example, it is thought that the mammary gland is a transformed sweat gland, more closely related to apocrine sweat glands.[67] Because mammary glands do not fossilize well, supporting such theories with fossil evidence is difficult. Many of the current theories are based on comparisons between lines of living mammals—monotremes, marsupials, and eutherians. One theory proposes that mammary glands evolved from glands that were used to keep the eggs of early mammals moist[68][69] and free from infection[70][71] (monotremes still lay eggs). Other theories suggest that early secretions were used directly by hatched young,[72] or that the secretions were used by young to help them orient to their mothers.[73]
Lactation is thought to have developed long before the evolution of the mammary gland and mammals; see evolution of lactation.
Additional images
[edit]-
Cross section of the breast of a human female
See also
[edit]References
[edit]- ^ a b c Macéa, José Rafael; Fregnani, José Humberto Tavares Guerreiro (1 December 2006). "Anatomy of the Thoracic Wall, Axilla and Breast" (PDF). International Journal of Morphology. 24 (4). doi:10.4067/S0717-95022006000500030.
- ^ Lawrence, Ruth A.; Lawrence, Robert M. (30 September 2010). Breastfeeding: A Guide for the Medical Profession (7th ed.). Maryland Heights, Maryland: Mosby/Elsevier. p. 54. ISBN 978-1-4377-3590-1.
- ^ Gray, Henry (1918). Anatomy of the Human Body.
- ^ Newton, Michael; Newton, Niles Rumely (December 1948). "The let-down reflex in human lactation". The Journal of Pediatrics. 33 (6): 698–704. doi:10.1016/S0022-3476(48)80075-2. PMID 18101061.
- ^ Zucca-Matthes, Gustavo; Urban, Cícero; Vallejo, André (February 2016). "Anatomy of the nipple and breast ducts". Gland Surgery. 5 (1): 32–36. doi:10.3978/j.issn.2227-684X.2015.05.10. ISSN 2227-684X. PMC 4716863. PMID 26855906.
- ^ a b c Watson, C. J.; Khaled, W. T. (2008). "Mammary development in the embryo and adult: A journey of morphogenesis and commitment". Development. 135 (6): 995–1003. doi:10.1242/dev.005439. PMID 18296651. S2CID 9089976.
- ^ a b c Wiseman, B. S.; Werb, Z. (2002). "Stromal Effects on Mammary Gland Development and Breast Cancer". Science. 296 (5570): 1046–1049. Bibcode:2002Sci...296.1046W. doi:10.1126/science.1067431. PMC 2788989. PMID 12004111.
- ^ Pavlovich, A. L.; Manivannan, S.; Nelson, C. M. (2010). "Adipose Stroma Induces Branching Morphogenesis of Engineered Epithelial Tubules". Tissue Engineering Part A. 16 (12): 3719–3726. doi:10.1089/ten.TEA.2009.0836. PMC 2991209. PMID 20649458.
- ^ Moore, Keith L.; Dalley, Arthur F.; Agur, Anne M. R. (2018). Clinically oriented anatomy (Eighth ed.). Philadelphia Baltimore New York London Buenos Aires Hong Kong Sydney Tokyo: Wolters Kluwer. p. 318. ISBN 978-1-4963-4721-3.
- ^ Ackerman (2005) ch.1 Apocrine Units Archived 21 April 2011 at the Wayback Machine
- ^ Krstic, Radivoj V. (18 March 2004). Human Microscopic Anatomy: An Atlas for Students of Medicine and Biology. Springer. p. 466. ISBN 978-3-540-53666-6.
- ^ Wysolmerski, J. J.; Philbrick, W. M.; Dunbar, M. E.; Lanske, B.; Kronenberg, H.; Broadus, A. E. (1998). "Rescue of the parathyroid hormone-related protein knockout mouse demonstrates that parathyroid hormone-related protein is essential for mammary gland development". Development. 125 (7): 1285–1294. doi:10.1242/dev.125.7.1285. PMID 9477327.
- ^ Hens, J. R.; Wysolmerski, J. J. (2005). "Key stages of mammary gland development: Molecular mechanisms involved in the formation of the embryonic mammary gland". Breast Cancer Research. 7 (5): 220–224. doi:10.1186/bcr1306. PMC 1242158. PMID 16168142.
- ^ Montévil, Maël; Speroni, Lucia; Sonnenschein, Carlos; Soto, Ana M. (1 October 2016). "Modeling mammary organogenesis from biological first principles: Cells and their physical constraints". Progress in Biophysics and Molecular Biology. From the Century of the Genome to the Century of the Organism: New Theoretical Approaches. 122 (1): 58–69. doi:10.1016/j.pbiomolbio.2016.08.004. PMC 5563449. PMID 27544910.
- ^ a b Brisken; Malley (2 December 2010). "Hormone Action in the Mammary Gland". Cold Spring Harbor Perspectives in Biology. 2 (12) a003178. doi:10.1101/cshperspect.a003178. PMC 2982168. PMID 20739412.
- ^ a b c Kleinberg DL (1998). "Role of IGF-I in normal mammary development". Breast Cancer Res. Treat. 47 (3): 201–8. doi:10.1023/a:1005998832636. PMID 9516076. S2CID 30440069.
- ^ a b c Kleinberg DL (1997). "Early mammary development: growth hormone and IGF-1". J Mammary Gland Biol Neoplasia. 2 (1): 49–57. doi:10.1023/A:1026373513521. PMID 10887519. S2CID 41667675.
- ^ a b c Ruan W, Kleinberg DL (1999). "Insulin-like growth factor I is essential for terminal end bud formation and ductal morphogenesis during mammary development". Endocrinology. 140 (11): 5075–81. doi:10.1210/endo.140.11.7095. PMID 10537134.
- ^ a b c d e Kleinberg DL, Feldman M, Ruan W (2000). "IGF-I: an essential factor in terminal end bud formation and ductal morphogenesis". J Mammary Gland Biol Neoplasia. 5 (1): 7–17. doi:10.1023/A:1009507030633. PMID 10791764. S2CID 25656770.
- ^ Kleinberg DL, Ruan W (2008). "IGF-I, GH, and sex steroid effects in normal mammary gland development". J Mammary Gland Biol Neoplasia. 13 (4): 353–60. doi:10.1007/s10911-008-9103-7. PMID 19034633. S2CID 24786346.
- ^ Serra R, Crowley MR (2005). "Mouse models of transforming growth factor beta impact in breast development and cancer". Endocr. Relat. Cancer. 12 (4): 749–60. doi:10.1677/erc.1.00936. PMID 16322320.
- ^ LaMarca HL, Rosen JM (2007). "Estrogen regulation of mammary gland development and breast cancer: amphiregulin takes center stage". Breast Cancer Res. 9 (4): 304. doi:10.1186/bcr1740. PMC 2206713. PMID 17659070.
- ^ El-Attar HA, Sheta MI (2011). "Hepatocyte growth factor profile with breast cancer". Indian J Pathol Microbiol. 54 (3): 509–13. doi:10.4103/0377-4929.85083. PMID 21934211.
- ^ Coad, Jane; Dunstall, Melvyn (2011). Anatomy and Physiology for Midwives. Elsevier Health Sciences. pp. 413–. ISBN 978-0-7020-3489-3.
- ^ Hynes, N. E.; Watson, C. J. (2010). "Mammary Gland Growth Factors: Roles in Normal Development and in Cancer". Cold Spring Harbor Perspectives in Biology. 2 (8) a003186. doi:10.1101/cshperspect.a003186. ISSN 1943-0264. PMC 2908768. PMID 20554705.
- ^ Jay R. Harris; Marc E. Lippman; C. Kent Osborne; Monica Morrow (28 March 2012). Diseases of the Breast. Lippincott Williams & Wilkins. pp. 94–. ISBN 978-1-4511-4870-1.
- ^ a b c Chong YM, Subramanian A, Sharma AK, Mokbel K (2007). "The potential clinical applications of insulin-like growth factor-1 ligand in human breast cancer". Anticancer Res. 27 (3B): 1617–24. PMID 17595785.
- ^ Leonard R. Johnson (2003). Essential Medical Physiology. Academic Press. pp. 770–. ISBN 978-0-12-387584-6.
- ^ a b Jernström H, Olsson H (1997). "Breast size in relation to endogenous hormone levels, body constitution, and oral contraceptive use in healthy nulligravid women aged 19–25 years". Am. J. Epidemiol. 145 (7): 571–80. doi:10.1093/oxfordjournals.aje.a009153. PMID 9098173.
- ^ Zhou J, Ng S, Adesanya-Famuiya O, Anderson K, Bondy CA (2000). "Testosterone inhibits estrogen-induced mammary epithelial proliferation and suppresses estrogen receptor expression". FASEB J. 14 (12): 1725–30. doi:10.1096/fj.99-0863com. PMID 10973921. S2CID 17172449.
- ^ Lemaine V, Cayci C, Simmons PS, Petty P (2013). "Gynecomastia in adolescent males". Semin Plast Surg. 27 (1): 56–61. doi:10.1055/s-0033-1347166. PMC 3706045. PMID 24872741.
- ^ Sekhri, KK; Pitelka, DR; Deome, KB (September 1967). "Studies of mouse mammary glands. I. Cytomorphology of the normal mammary gland". J Natl Cancer Inst. 39 (3): 459–90. PMID 6053715.
- ^ Tharmapalan, Pirashaanthy; Mahendralingam, Mathepan; Berman, Hal K; Khokha, Rama (15 July 2019). "Mammary stem cells and progenitors: targeting the roots of breast cancer for prevention". The EMBO Journal. 38 (14) e100852. doi:10.15252/embj.2018100852. ISSN 0261-4189. PMC 6627238. PMID 31267556.
- ^ Hens, JR; Wysolmerski JJ (10 August 2005). "Key stages of mammary gland development: molecular mechanisms involved in the formation of the embryonic mammary gland". Breast Cancer Res. 7 (5): 220–4. doi:10.1186/bcr1306. PMC 1242158. PMID 16168142.
- ^ Makarem, M; Eaves C (April 2013). "Stem Cells and the Developing Mammary Gland". J Mammary Gland Biol Neoplasia. 18 (2): 209–19. doi:10.1007/s10911-013-9284-6. PMC 4161372. PMID 23624881.
- ^ Daniel, CW; Smith, GH (January 1999). "The mammary gland: a model for development". Journal of Mammary Gland Biology and Neoplasia. 4 (1): 3–8. doi:10.1023/A:1018796301609. PMID 10219902. S2CID 36670489.
- ^ Sternlicht, M. D. (2006). "Key stages in mammary gland development: The cues that regulate ductal branching morphogenesis". Breast Cancer Research. 8 (1): 201–203. doi:10.1186/bcr1368. PMC 1413974. PMID 16524451.
- ^ Sternlicht, M. D.; Kouros-Mehr, H.; Lu, P.; Werb, Z. (2006). "Hormonal and local control of mammary branching morphogenesis". Differentiation. 74 (7): 365–381. doi:10.1111/j.1432-0436.2006.00105.x. PMC 2580831. PMID 16916375.
- ^ Fata, J. E.; Werb, Z.; Bissell, M. J. (2003). "Regulation of mammary gland branching morphogenesis by the extracellular matrix and its remodeling enzymes". Breast Cancer Research. 6 (1): 1–11. doi:10.1186/bcr634. PMC 314442. PMID 14680479.
- ^ Wiseman, B. S.; Sternlicht, M. D.; Lund, L. R.; Alexander, C. M.; Mott, J.; Bissell, M. J.; Soloway, P.; Itohara, S.; Werb, Z. (2003). "Site-specific inductive and inhibitory activities of MMP-2 and MMP-3 orchestrate mammary gland branching morphogenesis". The Journal of Cell Biology. 162 (6): 1123–1133. doi:10.1083/jcb.200302090. PMC 2172848. PMID 12975354.
- ^ Koshikawa, N.; Giannelli, G.; Cirulli, V.; Miyazaki, K.; Quaranta, V. (2000). "Role of cell surface metalloprotease MT1-MMP in epithelial cell migration over laminin-5". The Journal of Cell Biology. 148 (3): 615–624. doi:10.1083/jcb.148.3.615. PMC 2174802. PMID 10662785.
- ^ Dogic, D.; Rousselle, P.; Aumailley, M. (1998). "Cell adhesion to laminin 1 or 5 induces isoform-specific clustering of integrins and other focal adhesion components" (PDF). Journal of Cell Science. 111 (6): 793–802. doi:10.1242/jcs.111.6.793. PMID 9472007.
- ^ Muschler, J.; Levy, D.; Boudreau, R.; Henry, M.; Campbell, K.; Bissell, M. J. (2002). "A role for dystroglycan in epithelial polarization: Loss of function in breast tumor cells". Cancer Research. 62 (23): 7102–7109. PMID 12460932.
- ^ Kurzer MS (March 2002). "Hormonal effects of soy in premenopausal women and men". The Journal of Nutrition. 132 (3): 570S – 573S. doi:10.1093/jn/132.3.570S. PMID 11880595. Also cited by Petrakis NL, Barnes S, King EB, Lowenstein J, Wiencke J, Lee MM, Miike R, Kirk M, Coward L (October 1996). "Stimulatory influence of soy protein isolate on breast secretion in pre- and postmenopausal people AFAB". Cancer Epidemiology, Biomarkers & Prevention (review). 5 (10): 785–94. PMID 8896889.
- ^ Robinson, G. W.; Hennighausen, L.; Johnson, P. F. (2000). "Side-branching in the mammary gland: The progesterone-Wnt connection". Genes & Development. 14 (8): 889–894. doi:10.1101/gad.14.8.889. PMID 10783160. S2CID 2319046.
- ^ Brisken, C.; Heineman, A.; Chavarria, T.; Elenbaas, B.; Tan, J.; Dey, S. K.; McMahon, J. A.; McMahon, A. P.; Weinberg, R. A. (2000). "Essential function of Wnt-4 in mammary gland development downstream of progesterone signaling". Genes & Development. 14 (6): 650–654. doi:10.1101/gad.14.6.650. PMC 316462. PMID 10733525.
- ^ a b Streuli, C. H.; Bailey, N.; Bissell, M. J. (1991). "Control of mammary epithelial differentiation: Basement membrane induces tissue-specific gene expression in the absence of cell-cell interaction and morphological polarity". The Journal of Cell Biology. 115 (5): 1383–1395. doi:10.1083/jcb.115.5.1383. PMC 2289247. PMID 1955479.
- ^ a b Streuli, C. H.; Schmidhauser, C.; Bailey, N.; Yurchenco, P.; Skubitz, A. P.; Roskelley, C.; Bissell, M. J. (1995). "Laminin mediates tissue-specific gene expression in mammary epithelia". The Journal of Cell Biology. 129 (3): 591–603. doi:10.1083/jcb.129.3.591. PMC 2120432. PMID 7730398.
- ^ Nicholas L. Petrakis; Lynn Mason; Rose Lee; Barbara Sugimoto; Stella Pawson; Frank Catchpool (1975). "Association of Race, Age, Menopausal Status, and Cerumen Type With Breast Fluid Secretion in Nonlactating Women, as Determined by Nipple Aspiration". Journal of the National Cancer Institute. 54 (4): 829–834. doi:10.1093/jnci/54.4.829. PMID 1168727.
- ^ Zarzynska, J.; Motyl, T. (2008). "Apoptosis and autophagy in involuting bovine mammary gland". Journal of Physiology and Pharmacology. 59 (Suppl 9): 275–288. PMID 19261986.
- ^ Fadok, V. A. (1999). "Clearance: The last and often forgotten stage of apoptosis". Journal of Mammary Gland Biology and Neoplasia. 4 (2): 203–211. doi:10.1023/A:1011384009787. PMID 10426399. S2CID 5926448.
- ^ Motyl, T.; Gajkowska, B.; Zarzyńska, J.; Gajewska, M.; Lamparska-Przybysz, M. (2006). "Apoptosis and autophagy in mammary gland remodeling and breast cancer chemotherapy". Journal of Physiology and Pharmacology. 57 (Suppl 7): 17–32. PMID 17228094.
- ^ Gudjonsson, T.; Rønnov-Jessen, L.; Villadsen, R.; Rank, F.; Bissell, M. J.; Petersen, O. W. (2002). "Normal and tumor-derived myoepithelial cells differ in their ability to interact with luminal breast epithelial cells for polarity and basement membrane deposition". Journal of Cell Science. 115 (Pt 1): 39–50. doi:10.1242/jcs.115.1.39. PMC 2933194. PMID 11801722.
- ^ Provenzano, P. P.; Inman, D. R.; Eliceiri, K. W.; Knittel, J. G.; Yan, L.; Rueden, C. T.; White, J. G.; Keely, P. J. (2008). "Collagen density promotes mammary tumor initiation and progression". BMC Medicine. 6: 11. doi:10.1186/1741-7015-6-11. PMC 2386807. PMID 18442412.

- ^ a b "With the Wild Things – Transcripts". Digitalcollections.fiu.edu. Archived from the original on 23 March 2013. Retrieved 5 April 2013.
- ^ a b Stockard, Mary (2005) Raising Orphaned Baby Opossums. Alabama Wildlife Center.
- ^ Cunningham, Merle; LaTour, Mickey A. & Acker, Duane (2005). Animal Science and Industry. Pearson Prentice Hall. ISBN 978-0-13-046256-5.
- ^ Dog breeds vary in the number of mammary glands: larger breeds tend to have 5 pairs, smaller breeds have 4 pairs.[citation needed]
- ^ P Smith 2008 Red-Sided Short-Tailed Opossum. Fauna Paraguay
- ^ Julie Ann Mayer; John Foley; Damon De La Cruz; Cheng-Ming Chuong; Randall Widelitz (November 2008). "Conversion of the Nipple to Hair-Bearing Epithelia by Lowering Bone Morphogenetic Protein Pathway Activity at the Dermal-Epidermal Interface". Am J Pathol. 173 (5): 1339–48. doi:10.2353/ajpath.2008.070920. PMC 2570124. PMID 18832580.
- ^ Patricia J. Armati; Chris R. Dickman; Ian D. Hume (17 August 2006). Marsupials. Cambridge University Press. ISBN 978-1-139-45742-2.
- ^ Hughes, Katherine (2021). "Development and Pathology of the Equine Mammary Gland". Journal of Mammary Gland Biology and Neoplasia. 26 (2): 121–134. doi:10.1007/s10911-020-09471-2. PMC 8236023. PMID 33280071.
- ^ Francis, C. M.; Anthony, E. L. P.; Brunton, J. A.; Kunz, T. H. (1994). "Lactation in male fruit bats" (PDF). Nature. 367 (6465): 691–692. Bibcode:1994Natur.367..691F. doi:10.1038/367691a0. S2CID 4369716.
- ^ Kunz, T; Hosken, D (2009). "Male lactation: why, why not and is it care?". Trends in Ecology & Evolution. 24 (2): 80–85. doi:10.1016/j.tree.2008.09.009. PMID 19100649.
- ^ Li, Peng; Knabe, Darrell A.; Kim, Sung Woo; Lynch, Christopher J.; Hutson, Susan M.; Wu, Guoyao (1 August 2009). "Lactating Porcine Mammary Tissue Catabolizes Branched-Chain Amino Acids for Glutamine and Aspartate Synthesis". The Journal of Nutrition. 139 (8): 1502–1509. doi:10.3945/jn.109.105957. ISSN 0022-3166. PMC 3151199. PMID 19549750.
- ^ "BBC News – The goats with spider genes and silk in their milk". bbc.co.uk. 17 January 2012. Retrieved 26 April 2012.
- ^ Oftedal, O. T. (2002). "The origin of lactation as a water source for parchment-shelled eggs". Journal of Mammary Gland Biology and Neoplasia. 7 (3): 253–266. doi:10.1023/A:1022848632125. PMID 12751890. S2CID 8319185.
- ^ Lactating on Eggs. Smithsonian National Zoo, 14 July 2003.
- ^ Oftedal, OT (2002). "The mammary gland and its origin during synapsid evolution". Journal of Mammary Gland Biology and Neoplasia. 7 (3): 225–52. doi:10.1023/A:1022896515287. PMID 12751889. S2CID 25806501.
- ^ Breast beginnings. scienceblogs.com
- ^ Vorbach, C.; Capecchi, M. R.; Penninger, J. M. (2006). "Evolution of the mammary gland from the innate immune system?". BioEssays. 28 (6): 606–616. doi:10.1002/bies.20423. PMID 16700061.
- ^ Lefèvre, C. M.; Sharp, J. A.; Nicholas, K. R. (2010). "Evolution of Lactation: Ancient Origin and Extreme Adaptations of the Lactation System". Annual Review of Genomics and Human Genetics. 11: 219–238. doi:10.1146/annurev-genom-082509-141806. PMID 20565255.
- ^ Graves, B. M.; Duvall, D. (1983). "A Role for Aggregation Pheromones in the Evolution of Mammallike Reptile Lactation". The American Naturalist. 122 (6): 835. doi:10.1086/284177. S2CID 84089647.
Bibliography
[edit]- Ackerman, A. Bernard; Almut Böer; Bruce Bennin; Geoffrey J. Gottlieb (2005). Histologic Diagnosis of Inflammatory Skin Diseases An Algorithmic Method Based on Pattern Analysis. Ardor Scribendi. ISBN 978-1-893357-25-9. Archived from the original on 21 April 2011.
- Moore, Keith L. et al. (2010) Clinically Oriented Anatomy 6th Ed
External links
[edit]- Comparative Mammary Gland Anatomy by W. L. Hurley
- On the anatomy of the breast by Sir Astley Paston Cooper (1840). Numerous drawings, in the public domain.