Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
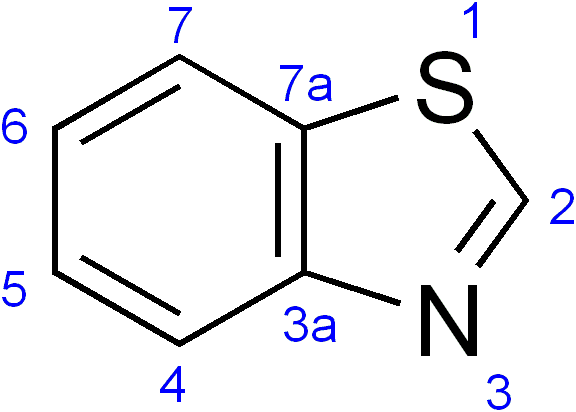
Benzothiazole
Benzothiazole, or more specifically 1,3-benzothiazole, is an aromatic heterocyclic compound with the chemical formula C
7H
5NS. It is colorless, slightly viscous liquid. Although the parent compound, benzothiazole is not widely used, many of its derivatives are found in commercial products or in nature. Firefly luciferin can be considered a derivative of benzothiazole. It has a sulfurous odor and meaty flavor.
The three structural isomers of benzothizaole are 1,3-benzothiazole, 1,2-benzothiazole and 2,1-benzothiazole.
Benzothiazoles consist of a 5-membered 1,3-thiazole ring fused to a benzene ring. The nine atoms of the bicycle and the attached substituents are coplanar. The heterocyclic core of the molecule is readily substituted at the methyne (CH) centre in the thiazole ring. Thiazole is electron-withdrawing.
Benzothiazoles are typically prepared by treatment of 2-mercaptoaniline. For example, acid chlorides are effective:
Many other precursors have been used, commonly aldehydes in the presence of oxidants. In some cases, benzothiazoles are prepared directly from anilines, a process that entails ortho functionalization.
Naturally occurring benzothiazoles are proposed to arise by condensation of cysteine with quinones.
The dye thioflavin is a benzothiazole derivative.
Benzothiazole occurs naturally in some foods but is also used as a food additive. It has a sulfurous odor and meaty flavor. The European Food Safety Authority assessment had "no safety concern at estimated levels of intake as a flavouring substance".
Hub AI
Benzothiazole AI simulator
(@Benzothiazole_simulator)
Benzothiazole
Benzothiazole, or more specifically 1,3-benzothiazole, is an aromatic heterocyclic compound with the chemical formula C
7H
5NS. It is colorless, slightly viscous liquid. Although the parent compound, benzothiazole is not widely used, many of its derivatives are found in commercial products or in nature. Firefly luciferin can be considered a derivative of benzothiazole. It has a sulfurous odor and meaty flavor.
The three structural isomers of benzothizaole are 1,3-benzothiazole, 1,2-benzothiazole and 2,1-benzothiazole.
Benzothiazoles consist of a 5-membered 1,3-thiazole ring fused to a benzene ring. The nine atoms of the bicycle and the attached substituents are coplanar. The heterocyclic core of the molecule is readily substituted at the methyne (CH) centre in the thiazole ring. Thiazole is electron-withdrawing.
Benzothiazoles are typically prepared by treatment of 2-mercaptoaniline. For example, acid chlorides are effective:
Many other precursors have been used, commonly aldehydes in the presence of oxidants. In some cases, benzothiazoles are prepared directly from anilines, a process that entails ortho functionalization.
Naturally occurring benzothiazoles are proposed to arise by condensation of cysteine with quinones.
The dye thioflavin is a benzothiazole derivative.
Benzothiazole occurs naturally in some foods but is also used as a food additive. It has a sulfurous odor and meaty flavor. The European Food Safety Authority assessment had "no safety concern at estimated levels of intake as a flavouring substance".