Bile (from Latin bilis), also known as gall, is a yellow-green fluid produced by the liver of most vertebrates that aids the digestion of lipids in the small intestine. In humans, bile is primarily composed of water, is produced continuously by the liver, and is stored and concentrated in the gallbladder. After a human eats, this stored bile is discharged into the first section of the small intestine, known as the duodenum.[1]
Composition
[edit]In the human liver, bile is composed of 97–98% water, 0.7% bile salts, 0.2% bilirubin, 0.51% fats (cholesterol, fatty acids, and lecithin), and 200 meq/L inorganic salts.[2][3] The two main pigments of bile are bilirubin, which is orange-yellow, and its oxidised form biliverdin, which is green.[4] About 400 to 800 milliliters (14 to 27 U.S. fluid ounces) of bile is produced per day in adult human beings.[5]
Function
[edit]
Bile or gall acts to some extent as a surfactant, helping to emulsify the lipids in food. Bile salt anions are hydrophilic on one side and hydrophobic on the other side; consequently, they tend to aggregate around droplets of lipids (triglycerides and phospholipids) to form micelles, with the hydrophobic sides towards the fat and hydrophilic sides facing outwards.[6] The hydrophilic sides are negatively charged, and this charge prevents fat droplets coated with bile from re-aggregating into larger fat particles. Ordinarily, the micelles in the duodenum have a diameter around 1–50 μm in humans.[7]
The dispersion of food fat into micelles provides a greatly increased surface area for the action of the enzyme pancreatic lipase, which digests the triglycerides, and is able to reach the fatty core through gaps between the bile salts.[8] A triglyceride is broken down into two fatty acids and a monoglyceride, which are absorbed by the villi on the intestine walls. After being transferred across the intestinal membrane, the fatty acids reform into triglycerides (re-esterified), before being absorbed into the lymphatic system through lacteals. Without bile salts, most of the lipids in food would be excreted in feces, undigested.[9]
Since bile increases the absorption of fats, it is an important part of the absorption of the fat-soluble substances,[10] such as the vitamins A, D, E, and K.[11]
Besides its digestive function, bile serves also as the route of excretion for bilirubin, a byproduct of red blood cells recycled by the liver. Bilirubin derives from hemoglobin by glucuronidation.
Bile tends to be alkaline on average. The pH of common duct bile (7.50 to 8.05) is higher than that of the corresponding gallbladder bile (6.80 to 7.65). Bile in the gallbladder becomes more acidic the longer a person goes without eating, though resting slows this fall in pH.[12] As an alkali, it also has the function of neutralizing excess stomach acid before it enters the duodenum, the first section of the small intestine. Bile salts also act as bactericides, destroying many of the microbes that may be present in the food.[13]
Clinical significance
[edit]In the absence of bile, fats become indigestible and are instead excreted in feces, a condition called steatorrhea. Feces lack their characteristic brown color and instead are white or gray, and greasy.[14] Steatorrhea can lead to deficiencies in essential fatty acids and fat-soluble vitamins.[15] In addition, past the small intestine (which is normally responsible for absorbing fat from food) the gastrointestinal tract and gut flora are not adapted to processing fats, leading to problems in the large intestine.[16]
The cholesterol contained in bile will occasionally accrete into lumps in the gallbladder, forming gallstones. Cholesterol gallstones are generally treated through surgical removal of the gallbladder. However, they can sometimes be dissolved by increasing the concentration of certain naturally occurring bile acids, such as chenodeoxycholic acid and ursodeoxycholic acid.[17][18]
On an empty stomach – after repeated vomiting, for example – a person's vomit may be green or dark yellow, and very bitter. The bitter and greenish component may be bile or normal digestive juices originating in the stomach.[19] Bile may be forced into the stomach secondary due to a weakened valve (pylorus), the presence of certain drugs including alcohol, or powerful muscular contractions and duodenal spasms. This is known as biliary reflux.[20]
Obstruction
[edit]Biliary obstruction refers to a condition when bile ducts which deliver bile from the gallbladder or liver to the duodenum become obstructed. The blockage of bile might cause a buildup of bilirubin in the bloodstream which can result in jaundice. There are several potential causes for biliary obstruction including gallstones, cancer,[21] trauma, choledochal cysts, or other benign causes of bile duct narrowing.[22] The most common cause of bile duct obstruction is when gallstone(s) are dislodged from the gallbladder into the cystic duct or common bile duct resulting in a blockage. A blockage of the gallbladder or cystic duct may cause cholecystitis. If the blockage is beyond the confluence of the pancreatic duct, this may cause gallstone pancreatitis. In some instances of biliary obstruction, the bile may become infected by bacteria resulting in ascending cholangitis.
Society and culture
[edit]In medical theories prevalent in the West from classical antiquity to the Middle Ages, the body's health depended on the equilibrium of four "humors", or vital fluids, two of which related to bile: blood, phlegm, "yellow bile" (choler), and "black bile". These "humors" are believed to have their roots in the appearance of a blood sedimentation test made in open air, which exhibits a dark clot at the bottom ("black bile"), a layer of unclotted erythrocytes ("blood"), a layer of white blood cells ("phlegm") and a layer of clear yellow serum ("yellow bile").[23]
Excesses of black bile and yellow bile were thought to produce depression and aggression, respectively, and the Greek names for them gave rise to the English words cholera (from Greek χολή kholē, "bile") and melancholia. In the former of those senses, the same theories explain the derivation of the English word bilious from bile, the meaning of gall in English as "exasperation" or "impudence", and the Latin word cholera, derived from the Greek kholé, which was passed along into some Romance languages as words connoting anger, such as colère (French) and cólera (Spanish).[24]
Soap
[edit]Soap can be mixed with bile from mammals, such as ox gall. This mixture, called bile soap[25] or gall soap, can be applied to textiles a few hours before washing as a traditional and effective method for removing various kinds of tough stains.[26]
Food
[edit]Pinapaitan is a dish in Philippine cuisine that uses bile as flavoring.[27] Other areas where bile is commonly used as a cooking ingredient include Laos and northern parts of Thailand.
During the Boshin War, Satsuma soldiers of the early Imperial Japanese Army reportedly ate human livers boiled in bile.[28] The practice of eating a slain enemy's liver, known as hiemontori (冷え物取り), was a tradition of the Satsuma people.
Bears
[edit]In regions where bile products are a popular ingredient in traditional medicine, the use of bears in bile-farming has been widespread. This practice has been condemned by activists, and some pharmaceutical companies have developed synthetic (non-ursine) alternatives.[29]
Principal acids
[edit]See also
[edit]References
[edit]- ^ Puestow, Charles B. (1931-12-01). "The Discharge of Bile into the Duodenum". Archives of Surgery. 23 (6): 1013–1029. doi:10.1001/archsurg.1931.01160120127008. ISSN 0272-5533.
- ^ Barrett, Kim E.; Barman, Susan M.; Boitano, Scott; Brooks, Heddwen L. (2012). Ganong's Review of Medical Physiology (24th ed.). New York: McGraw-Hill Medical. p. 512. ISBN 978-0-07-178003-2.
- ^ Guyton and Hall (2011). Textbook of Medical Physiology. U.S.: Saunders Elsevier. p. 784. ISBN 978-1-4160-4574-8.
- ^ "Bile pigments - Oxford Reference". www.oxfordreference.com. Retrieved 2020-01-20.
- ^ "Secretion of Bile and the Role of Bile Acids In Digestion". www.vivo.colostate.edu. Retrieved 2017-03-31.
- ^ A. Potter, Patrica (2013). Fundamentals of Nursing, 8th edition. Elsevier, Inc. p. 1000. ISBN 978-0-323-07933-4.
- ^ Dickinson, Eric; Leser, Martin E. (2007-10-31). Food Colloids: Self-Assembly and Material Science. Royal Society of Chemistry. p. 22. ISBN 978-1-84755-769-8.
- ^ Lowe, Mark E. (2002-12-01). "The triglyceride lipases of the pancreas". Journal of Lipid Research. 43 (12): 2007–2016. doi:10.1194/jlr.R200012-JLR200. ISSN 0022-2275. PMID 12454260.
- ^ Starr, Cecie (2007-09-20). Biology: Concepts and Applications. Cengage Learning. p. 650. ISBN 978-0-495-11981-4.
- ^ "Secretion of Bile and the Role of Bile Acids In Digestion". www.vivo.colostate.edu. Retrieved 2016-06-05.
- ^ "Secretion of Bile and the Role of Bile Acids In Digestion". www.vivo.colostate.edu. Retrieved 2018-04-09.
- ^ Sutor, D. June (1976). "Diurnal Variations in the pH of Pathological Gallbladder Bile". Gut. 17 (12): 971–974. doi:10.1136/gut.17.12.971. PMC 1411240. PMID 14056.
- ^ Merritt, M. E.; Donaldson, J. R. (2009-09-17). "Effect of bile salts on the DNA and membrane integrity of enteric bacteria". Journal of Medical Microbiology. 58 (12): 1533–1541. doi:10.1099/jmm.0.014092-0. ISSN 0022-2615. PMID 19762477.
- ^ Barabote RD, Tamang DG, Abeywardena SN, et al. (2006). "Extra domains in secondary transport carriers and channel proteins". Biochim. Biophys. Acta. 1758 (10): 1557–79. doi:10.1016/j.bbamem.2006.06.018. PMID 16905115.
- ^ Azer, Samy A.; Sankararaman, Senthilkumar (2019), "Steatorrhea", StatPearls, StatPearls Publishing, PMID 31082099, retrieved 2020-01-20
- ^ Gorbach, Sherwood L. (1971-06-01). "Intestinal Microflora". Gastroenterology. 60 (6): 1110–1129. doi:10.1016/S0016-5085(71)80039-2. ISSN 0016-5085. PMID 4933894.
- ^ Bell, G. D. (1980-01-01), Dukes, M. N. G. (ed.), Drugs used in the management of gallstones, Side Effects of Drugs Annual, vol. 4, Elsevier, pp. 258–263, doi:10.1016/S0378-6080(80)80042-0, ISBN 9780444901309, retrieved 2020-01-20
- ^ Guarino, Michele Pier Luc a; Cocca, Silvia; Altomare, Annamaria; Emerenziani, Sara; Cicala, Michele (2013-08-21). "Ursodeoxycholic acid therapy in gallbladder disease, a story not yet completed". World Journal of Gastroenterology. 19 (31): 5029–5034. doi:10.3748/wjg.v19.i31.5029. ISSN 1007-9327. PMC 3746374. PMID 23964136.
- ^ Choices, NHS. "Nausea and vomiting in adults - NHS Choices". www.nhs.uk. Retrieved 2016-06-05.
- ^ Iacobuzio-Donahue, Christine A.; Montgomery, Elizabeth A. (2011-06-06). Gastrointestinal and Liver Pathology E-Book: A Volume in the Series: Foundations in Diagnostic Pathology. Elsevier Health Sciences. p. 71. ISBN 978-1-4557-1193-2.
- ^ Boulay, Brian R; Birg, Aleksandr (2016-06-15). "Malignant biliary obstruction: From palliation to treatment". World Journal of Gastrointestinal Oncology. 8 (6): 498–508. doi:10.4251/wjgo.v8.i6.498. ISSN 1948-5204. PMC 4909451. PMID 27326319.
- ^ Shanbhogue, Alampady Krishna Prasad; Tirumani, Sree Harsha; Prasad, Srinivasa R.; Fasih, Najla; McInnes, Matthew (2011-08-01). "Benign Biliary Strictures: A Current Comprehensive Clinical and Imaging Review". American Journal of Roentgenology. 197 (2): W295 – W306. doi:10.2214/AJR.10.6002. ISSN 0361-803X. PMID 21785056.
- ^ Johansson, Ingvar; Lynøe, Niels (2008). Medicine & Philosophy: A Twenty-First Century Introduction. Walter de Gruyter. p. 27. ISBN 9783110321364. Retrieved 2015-04-23.
If blood is poured into a glass jar, a process of coagulation and sedimentation starts. It ends with four clearly distinct layers: a red region, a yellowish one, a black one, and a white one (Figure 4, left) ... The lowest part of the same column consists of sediment that is too dense to permit light to pass through. Therefore, this part of the column looks black and might be referred to as the 'black bile'. On the top of the column there is a white layer, which we today classify as fibrin; it might correspond to Galen's 'phlegm'. The remaining part is a rather clear but somewhat yellowish fluid that surrounds the coagulated column in the middle. It might be called 'yellow bile', but today we recognize it as blood serum.
- ^ Boddice, Rob (2017). Pain: A Very Short Introduction. Oxford University Press. p. 10. ISBN 978-0-19-873856-5.
- ^ Newton, W. (1837). "The invention of certain improvements in the manufacture of soap, which will be particularly applicable to the felting of woollen cloths". The London Journal of Arts and Sciences; and Repertory of Patent Inventions. IX: 289. Retrieved 2007-02-08.
- ^ Martin, Geoffrey (1951). The Modern Soap and Detergent Industry: The manufacture of special soaps and detergent compositions. Technical Press. p. 15.
- ^ "Pinapaitan - Ang Sarap". Ang Sarap (A Tagalog word for "It's Delicious"). 2013-08-13. Retrieved 2016-06-05.
- ^ 牧原 Makihara, 憲夫 Norio (December 26, 2008). 文明国をめざして. Japan: 小学館 Shōgakukan. ISBN 4096221139.
- ^ Hance, J. (2015). "Is the end of 'house of horror' bear bile factories in sight?". The Guardian.
Further reading
[edit]- Bowen, R. (2001-11-23). "Secretion of Bile and the Role of Bile Acids In Digestion". Colorado State Hypertextbook article on Bile. Archived from the original on 29 May 2007. Retrieved 2007-07-17.
- Krejčí, Z; Hanuš L.; Podstatová H.; Reifová E (1983). "A contribution to the problems of the pathogenesis and microbial etiology of cholelithiasis". Acta Universitatis Palackianae Olomucensis Facultatis Medicae. 104: 279–286. PMID 6222611.
- Maton, Anthea; Jean Hopkins; Charles William McLaughlin; Susan Johnson; Maryanna Quon Warner; David LaHart; Jill D. Wright (1993). Human Biology and Health. Englewood Cliffs, New Jersey: Prentice Hall. ISBN 0-13-981176-1.
- Seleem HM, Nada AS, Naguib MA, Abdelmaksoud OR, El-Gazzarah AR (2021). Serum immunoglobulin G4 in patients with nonmalignant common bile duct stricture. Menoufia Med J; 34:1275-83.
Its primary components include conjugated bile salts derived from cholesterol, phospholipids such as lecithin, cholesterol itself, electrolytes, and conjugated bilirubin, which together enable bile's detergent-like action on lipids and its role in excreting metabolic waste products like excess cholesterol and heme breakdown remnants.[1][2]
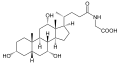
Bile acids, the active agents in bile salts, are synthesized in the liver from cholesterol via cytochrome P450 enzymes, primarily as cholic acid and chenodeoxycholic acid, which are then conjugated with glycine or taurine to enhance solubility and antimicrobial properties before secretion.[3][1]
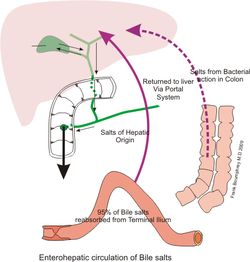
Through enterohepatic circulation, about 95% of bile acids are reabsorbed in the ileum, returned to the liver via the portal vein, and recycled multiple times per day, conserving cholesterol and minimizing the liver's synthetic burden while also modulating gut microbiota and influencing metabolic signaling pathways.[1][3]
Anatomy and Physiology
Production in the Liver
Bile is synthesized and secreted continuously by hepatocytes, the primary functional cells of the liver, at a rate of approximately 500–800 milliliters per day in human adults.[1] This production forms the initial hepatocyte-derived component of bile, which constitutes the majority of total bile flow, estimated at around 620 milliliters daily, with roughly half being bile salt-dependent and the other half independent.[2] Hepatocytes take up bile acids and other precursors from sinusoidal blood via basolateral membrane transporters, process them intracellularly, and actively secrete the resulting bile components into the apical canalicular domain.[1] The core of bile production involves the hepatic synthesis of primary bile acids from cholesterol through a multi-step enzymatic pathway requiring at least 14 reactions, primarily occurring in the endoplasmic reticulum and peroxisomes of hepatocytes.[3] The rate-limiting step is catalyzed by the enzyme cholesterol 7α-hydroxylase (CYP7A1), which initiates the conversion of cholesterol to 7α-hydroxycholesterol.[4] The two principal primary bile acids produced are cholic acid, comprising about 45–50% of the total, and chenodeoxycholic acid.[5] These unconjugated bile acids are then conjugated in hepatocytes with glycine or taurine—predominantly glycine in humans—to form water-soluble bile salts, enhancing their detergent properties and preventing passive reabsorption.[3] Conjugation occurs via bile acid-CoA:amino acid N-acyltransferase (BAAT), increasing solubility at physiological pH.[6] Secretion into bile canaliculi, the narrow intercellular spaces between adjacent hepatocytes sealed by tight junctions, is driven by ATP-dependent transporters on the canalicular membrane. Conjugated bile salts are exported primarily via the bile salt export pump (BSEP, encoded by ABCB11), creating an osmotic gradient that draws water and solutes into the canaliculi through aquaporin channels and other conduits.[7] This vectorial transport generates bile salt-dependent flow, while independent flow arises from the secretion of bicarbonate and other organic anions via transporters like multidrug resistance-associated protein 2 (MRP2).[8] The canalicular lumen expands in response to bile acid load through actin cytoskeleton remodeling, ensuring efficient bile formation without cellular damage under normal conditions.[9] Additional bile constituents, including phospholipids via ABCB4 (MDR3) and cholesterol via ABCG5/G8, are incorporated during this process to form micelles and prevent membrane injury from bile acids.[10] Disruptions in these transporters, as seen in genetic defects like progressive familial intrahepatic cholestasis, impair production and lead to cholestasis.[11]Storage and Gallbladder Role
The gallbladder functions as the principal reservoir for bile secreted by the liver, storing and concentrating it during fasting periods to prepare for digestive demands. Hepatic bile enters the gallbladder via the cystic duct, where it is held until needed for fat emulsification in the duodenum. This storage allows for the accumulation of bile produced continuously by hepatocytes, preventing continuous low-level leakage into the intestine during non-feeding states.[12][2] Concentration occurs primarily through active absorption of water and inorganic electrolytes by the gallbladder epithelium, reducing bile volume by up to 90% and increasing the concentration of bile salts, bilirubin, and other solutes 5- to 18-fold compared to hepatic bile. This process is mediated by sodium-potassium pumps and chloride channels in the mucosal cells, creating an osmotic gradient that draws water from the bile lumen into the bloodstream. The resulting hyperconcentrated bile enhances its efficiency in micelle formation upon release. Without this concentration, bile would be too dilute to effectively aid lipid digestion, as hepatic bile alone has lower bile acid levels (approximately 0.2-0.5% vs. 5-10% in gallbladder bile).[13][14][15] The human gallbladder has a resting volume of about 25 mL and a maximum capacity of 30-50 mL, sufficient to store the equivalent of several hours' hepatic bile output under normal conditions. Distension of the gallbladder wall during filling triggers neural feedback via vagal afferents, modulating storage dynamics. In the absence of a gallbladder, as after cholecystectomy, bile drips continuously from the liver into the duodenum at a lower concentration, which can impair fat absorption efficiency in some individuals.[16][17] Bile release is triggered postprandially, particularly by fatty meals, which stimulate enteroendocrine I-cells in the duodenum to secrete cholecystokinin (CCK). CCK binds to CCK-A receptors on gallbladder smooth muscle cells, inducing rhythmic contractions that empty up to 80% of stored bile within 30-60 minutes. This ejection propels bile through the cystic duct, common bile duct, and sphincter of Oddi into the duodenum, where CCK also promotes sphincter relaxation to facilitate flow. Parasympathetic innervation via the vagus nerve enhances contractility, while sympathetic input inhibits it during fasting.[18][19][20]Secretion and Enterohepatic Circulation
Bile is secreted continuously by hepatocytes into the canalicular lumen at a rate contributing to the total daily hepatic output of approximately 500–800 mL in adult humans, with the majority (about 80–90%) derived from hepatocyte secretion and the remainder from cholangiocyte contributions along the biliary tree.[1][21] This primary bile, rich in bile acids, cholesterol, phospholipids, and bilirubin, flows through intrahepatic ducts into the gallbladder via the cystic duct during fasting states, where it is concentrated up to 10-fold through active absorption of water and electrolytes by the gallbladder epithelium.[1] Postprandial release is triggered primarily by cholecystokinin (CCK), a hormone secreted by duodenal I-cells in response to luminal fatty acids and amino acids; CCK induces gallbladder smooth muscle contraction and relaxation of the sphincter of Oddi, propelling bile through the common bile duct and ampulla of Vater into the duodenal lumen to mix with chyme.[18][2] Secretin, released in response to duodenal acidification, further modulates secretion by stimulating bicarbonate-rich fluid from cholangiocytes, enhancing bile's buffering capacity.[2] In the intestinal lumen, bile acids facilitate lipid emulsification and micelle formation for efficient absorption of fats and fat-soluble vitamins, after which approximately 95% of bile acids are reabsorbed, predominantly in the terminal ileum via active transport mediated by the apical sodium-dependent bile acid transporter (ASBT).[1][22] The reabsorbed bile acids enter portal venous circulation unbound to albumin, achieving near-complete first-pass uptake (over 90%) by hepatocytes via the Na+-taurocholate cotransporting polypeptide (NTCP) on the sinusoidal membrane.[23] Within hepatocytes, bile acids are reconjugated if necessary and resecreted into the canaliculi via the bile salt export pump (BSEP), reentering the biliary system to perpetuate the cycle.[1] This enterohepatic circulation recycles the bile acid pool—typically 2–4 grams in adults—4 to 12 times per day, with daily losses of about 5% (0.2–0.6 grams) compensated by de novo hepatic synthesis to maintain steady-state levels, underscoring the system's efficiency in conserving these amphipathic molecules essential for lipid homeostasis.[24][1] Disruptions, such as ileal resection, impair reabsorption and expand the pool through upregulated synthesis, while feedback mechanisms involving farnesoid X receptor (FXR) in ileal enterocytes and hepatocytes suppress further production and promote transporters to regulate pool size.[25][4] The circulation's high fidelity minimizes fecal excretion, but secondary modifications by gut microbiota in the colon generate diverse bile acid species, some of which are passively reabsorbed or contribute to microbial signaling.[4]Biochemistry
Chemical Composition
Bile is an alkaline aqueous solution secreted by hepatocytes, comprising approximately 95% water along with dissolved organic solutes and electrolytes. The major organic components include conjugated bile salts (primarily glycine and taurine conjugates of cholic acid and chenodeoxycholic acid), which constitute about 67% of the non-aqueous organic solids; phospholipids (mainly phosphatidylcholine, 22%); cholesterol (4-5%); conjugated bilirubin (1-2%); and minor amounts of proteins (4.5%), fatty acids (0.5%), glutathione, and other metabolites.[26][1][27] Electrolytes such as sodium (141-165 mEq/L), potassium (2.7-6.7 mEq/L), chloride, calcium (≈4.3 mM in hepatic bile), and bicarbonate (higher than in plasma to maintain alkalinity) comprise the inorganic fraction, rendering bile isosmotic with plasma.[27][28] In hepatic bile, total bile salt concentrations typically range from 3-45 mmol/L, bilirubin from 1-2 mmol/L (or ≈63 μM total bilirubin), phospholipids from 140-810 mg/dL, and cholesterol from 97-320 mg/dL; these values vary with hepatic secretion rates and enterohepatic cycling.[27][28] Gallbladder storage concentrates bile by absorbing water and electrolytes (up to 5-20-fold), elevating bile salt levels to 100-200 mmol/L or higher while preserving the relative proportions of organic solutes, though absolute electrolyte concentrations decrease proportionally.[1][29] The primary bile salts are conjugates of cholic acid (3α,7α,12α-trihydroxy-5β-cholan-24-oic acid) and chenodeoxycholic acid (3α,7α-dihydroxy-5β-cholan-24-oic acid) in a ratio of approximately 2:1 to 1:1, with glycine conjugation predominating (≈75%) over taurine due to the liver's amidation preferences.[30][31] Secondary bile acids, such as deoxycholic acid and lithocholic acid (formed via gut microbial 7α-dehydroxylation), constitute 20-30% of the total bile acid pool in humans after reabsorption but are minor in freshly secreted hepatic bile.[32][33]| Component | Approximate Hepatic Bile Concentration | Notes |
|---|---|---|
| Bile salts | 3-45 mmol/L | Primarily conjugated CA and CDCA; increases with secretion rate.[27] |
| Phospholipids | 140-810 mg/dL | Mainly phosphatidylcholine; stabilizes cholesterol in micelles.[27] |
| Cholesterol | 97-320 mg/dL | Saturated in bile; risk factor for gallstone formation if supersaturated.[27] |
| Bilirubin (total) | 29-63 μM | Conjugated form; pigment responsible for bile color.[28] |
| Sodium | 141-165 mEq/L | Major cation; parallels plasma isosmolarity.[27] |
Biosynthesis Pathways
Bile acids are synthesized primarily in hepatocytes from cholesterol through two biosynthetic pathways: the classical (or neutral) pathway and the alternative (or acidic) pathway.[34] The classical pathway accounts for 75-95% of total bile acid production in humans under normal conditions, initiating with the microsomal enzyme cholesterol 7α-hydroxylase (CYP7A1), which performs the rate-limiting 7α-hydroxylation of cholesterol to form 7α-hydroxycholesterol.[4] [35] This step is followed by 10-17 enzymatic reactions, including sterol 12α-hydroxylase (CYP8B1) for cholic acid specificity, 24-hydroxylation, side-chain oxidation by mitochondrial CYP27A1, and cleavage to yield the primary bile acids cholic acid (three hydroxyl groups) and chenodeoxycholic acid (two hydroxyl groups).[36] [35] The alternative pathway, comprising the minor fraction of synthesis, begins extrahepatically or in liver mitochondria with 27-hydroxylation of cholesterol by sterol 27-hydroxylase (CYP27A1), producing 27-hydroxycholesterol, which is then transported to the endoplasmic reticulum for 7α-hydroxylation by oxysterol 7α-hydroxylase (CYP7B1).[35] This route bypasses CYP7A1 and predominates when the classical pathway is suppressed, such as in CYP7A1 deficiencies, yielding primarily chenodeoxycholic acid with less cholic acid due to absent CYP8B1 activity in early steps.[36] [35] Both pathways converge at 3α,7α-dihydroxy-5β-cholestanoic acid intermediates, undergoing peroxisomal β-oxidation for side-chain shortening.[34] Newly synthesized bile acids are conjugated in the endoplasmic reticulum with glycine (forming glycocholic and glycochenodeoxycholic acids) or taurine (taurocholic and taurochenodeoxycholic acids) via bile acid-CoA:amino acid N-acyltransferase (BAAT), enhancing solubility at physiological pH and facilitating secretion into bile canaliculi.[34] Conjugation ratios vary by species and diet, with humans favoring glycine (3:1 over taurine) under standard conditions.[4] Daily hepatic synthesis produces 0.2-0.6 grams of bile acids in adults, tightly regulated by feedback inhibition of CYP7A1 via farnesoid X receptor (FXR)-mediated transcription of small heterodimer partner (SHP), which represses CYP7A1 expression in response to elevated intracellular bile acids.[36] [37] Disruptions in these pathways, such as CYP7A1 polymorphisms, can alter synthesis rates and contribute to cholestatic disorders.[37]Principal Bile Acids and Salts
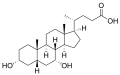
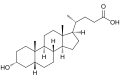
The principal bile acids in humans consist of primary and secondary types, with primary bile acids synthesized directly in the liver from cholesterol and secondary bile acids formed through microbial transformation in the intestine. Primary bile acids include cholic acid (CA), a trihydroxylated steroid with hydroxyl groups at positions 3α, 7α, and 12α, and chenodeoxycholic acid (CDCA), which has hydroxyl groups at 3α and 7α.[3][32] These primary acids constitute the majority of newly synthesized bile acids, with CA and CDCA produced in a typical ratio of approximately 1:1 in human hepatic biosynthesis.[3] Secondary bile acids arise from bacterial 7α-dehydroxylation of primary acids in the gut: deoxycholic acid (DCA) from CA (lacking the 7α-hydroxyl) and lithocholic acid (LCA) from CDCA (monohydroxylated at 3α).[3][4] DCA and LCA form a smaller portion of the total bile acid pool, typically 20-30% combined, as they are reabsorbed via enterohepatic circulation and partially resecreted.[32] These secondary acids contribute to the diversity of the bile acid pool, which recirculates 6-10 times daily, with a total pool size of about 2-4 grams in adults.[3] Bile salts refer to the conjugated forms of these acids, where unconjugated bile acids are amidated in hepatocytes with glycine or taurine (in a ratio favoring glycine, approximately 3:1 in humans) to enhance aqueous solubility and reduce toxicity at physiological pH.[3][38] Principal conjugated bile salts include glycocholic acid and taurocholic acid from CA, glycochenodeoxycholic acid and taurochenodeoxycholic acid from CDCA, and corresponding conjugates of DCA and LCA such as glycodeoxycholic acid.[3] Over 95% of bile acids in secreted bile exist as these ionized salts, facilitating micelle formation for lipid emulsification.[3]| Bile Acid Type | Examples | Origin | Key Features |
|---|---|---|---|
| Primary | Cholic acid (CA), Chenodeoxycholic acid (CDCA) | Hepatic synthesis from cholesterol | Di- or tri-hydroxylated; conjugated before secretion |
| Secondary | Deoxycholic acid (DCA), Lithocholic acid (LCA) | Gut bacterial dehydroxylation | Reduced hydroxylation; contribute to pool diversity |