Dimetindene
View on Wikipediafrom Wikipedia
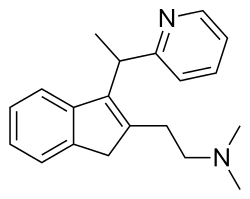 | |
| Clinical data | |
|---|---|
| Trade names | Fenistil |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral, topical |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.024.622 |
| Chemical and physical data | |
| Formula | C20H24N2 |
| Molar mass | 292.426 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| (verify) | |
Dimetindene, also sold under the brand name Fenistil, is an antihistamine/anticholinergic. It is a first generation[1] selective H1 antagonist.[2] Dimetindene is an atypical first generation H1 antagonist as it only minimally[3] passes across the blood–brain barrier.
Dimetindene is also an M2 receptor antagonist.[4]
It was patented in 1958 and came into medical use in 1960.[5]
Medical use
[edit]Dimetindene is used orally and topically as an antipruritic.[6] It is used topically to treat skin irritations, such as insect bites. Dimetindene is also administered orally to treat allergies, such as hay fever.
It is commonly formulated as its maleic acid salt, dimethindene maleate.
Names
[edit]It is sold under the brand name Fenistil among others.
References
[edit]- ^ Fromer LM, Ortiz GR, Dowdee AM (September 2008). "Assessment of Patient Attitudes About Mometasone Furoate Nasal Spray: The Ease-of-Use Patient Survey". The World Allergy Organization Journal. 1 (9): 156–159. doi:10.1186/1939-4551-1-9-145. PMC 3651039. PMID 23282579.
- ^ "Dimetindene". Drugs.com.
- ^ Noguchi S, Inukai T, Kuno T, Tanaka C (June 1992). "The suppression of olfactory bulbectomy-induced muricide by antidepressants and antihistamines via histamine H1 receptor blocking". Physiology & Behavior. 51 (6): 1123–1127. doi:10.1016/0031-9384(92)90297-f. PMID 1353628. S2CID 29562845.
- ^ Matsumoto Y, Miyazato M, Furuta A, Torimoto K, Hirao Y, Chancellor MB, Yoshimura N (April 2010). "Differential roles of M2 and M3 muscarinic receptor subtypes in modulation of bladder afferent activity in rats". Urology. 75 (4): 862–867. doi:10.1016/j.urology.2009.12.013. PMC 2871158. PMID 20156651.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 547. ISBN 9783527607495.
- ^ Wexler L (June 1962). "A clinical evaluation of dimethindene". Current Therapeutic Research, Clinical and Experimental. 4: 306–309. PMID 14006402.
Dimetindene
View on Grokipediafrom Grokipedia
