Recent from talks
Nothing was collected or created yet.
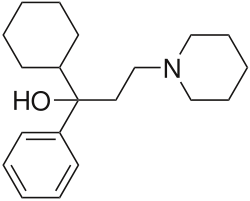
Trihexyphenidyl
View on Wikipedia
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Artane, Parkin,[citation needed] Pacitane, Hexymer |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682160 |
| License data | |
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Elimination half-life | 3.3-4.1 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.005.105 |
| Chemical and physical data | |
| Formula | C20H31NO |
| Molar mass | 301.474 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Trihexyphenidyl (THP, benzhexol, trihex, marketed as Artane and others) is an antispasmodic drug used to treat stiffness, tremors, spasms, and poor muscle control. It is an agent of the antimuscarinic class and is often used in management of Parkinson's disease. It was approved by the FDA for the treatment of Parkinson's in the US in 1949.[2][3]
Trihexyphenidyl is a therapeutic alternative on the World Health Organization's List of Essential Medicines.[4]
Medical uses
[edit]Trihexyphenidyl is used for the symptomatic treatment of Parkinson's disease in mono and combination therapy.[5]
Trihexyphenidyl has also been prescribed for essential tremor and akathisia.[6][7]
In pediatrics, it has been used for children with dystonia due to cerebral palsy,[8][9][10] and to control drooling.[11]
In organophosphate poisoning, trihexyphenidyl is a more effective antidote than atropine to counteract the cholinergic crisis, seizures, and neuropathology.[12]
Contraindications
[edit]Contraindications include according to the Therapeutic Goods Administration Australia from 2022:[13]
- Hypersensitivity to trihexyphenidyl
- Narrow angle glaucoma
- Ileus (disruption of the normal propulsive ability of the intestine)
- Caution: People with obstructive diseases of the urogenital tract, people with a known history of seizures and those with potentially dangerous tachycardia
Adverse effects
[edit]Dose-dependent side effects are frequent, but typically lessen over time as the body adapts to the medication. All of the following symptoms considered, Artane has been shown to dramatically and consistently improve neurologic defects in people aged 16–86 over the course of five years.[14] People who are older or who have psychiatric conditions may become confused or develop delirium. Side effects include but are not limited to:[15]
- Central nervous system: drowsiness, vertigo, headache, and dizziness are frequent. With high doses nervousness, agitation, anxiety, delirium, and confusion are noted. Trihexyphenidyl may be abused due to a short acting mood-elevating and euphoric effect. The normal sleep architecture may be altered (REM sleep depression). Trihexyphenidyl may lower the seizure-threshold.
- Peripheral side effects: dry mouth, impaired sweating, abdominal discomfort, nausea, and constipation are frequent. Tachycardia or heart palpitations (fast heart rate) may be noted. Allergic reactions are rare, but may occur. Many of these peripheral symptoms, especially considering an acute increase in anxiety with various physical complaints, as well as evidence of orthostatic hypotension and tachycardia are indicative of withdrawal, especially in people with psychiatric conditions [16]
- Eyes: trihexyphenidyl causes mydriasis with or without photophobia. It may precipitate narrow angle glaucoma or cause blurred vision.
- Tolerance may develop during therapy which requires dose adjustments.
- Striated musculature and weight gain.
Overdose
[edit]This section needs more reliable medical references for verification or relies too heavily on primary sources. (May 2017) |
Trihexyphenidyl and other antiparkinsonian drugs are known to be substances of abuse. This is true both in abusers of other substances and in chronic schizophrenics, the latter being infrequent abusers of other drugs.[17]
Overdose mimics an atropine intoxication with dryness of mucous membranes, red face, bowel and bladder paralysis, and hyperthermia in high doses. Central nervous system consequences are agitation, confusion, and hallucinations. An untreated overdose may be fatal, particularly in children. Premortal signs are respiratory depression, arrhythmia and cardiac arrest.
A case report of 24-hour long arrhythmia was treated with verapamil.[18]
Excessive myoclonus can be complicated by rhabdomyolysis; in one case risk was increased due to concomitant use of risperidone.[19]
Interactions
[edit]This section needs more reliable medical references for verification or relies too heavily on primary sources. (May 2017) |
- Other anticholinergic drugs (e.g. spasmolytics, antihistamines, TCAs) : Side effects of trihexyphenidyl may be increased.
- Quinidine : Increased anticholinergic action (particular on AV conduction).
- Antipsychotics : Long term use of trihexyphenidyl may mask or increase the risk of tardive dyskinesia.
- Pethidine (meperidine) : Central effects and side effects of pethidine may be increased.
- Metoclopramide : Action of metoclopramide is decreased.
- Alcohol : Risk of serious intoxication.
Pharmacology
[edit]Pharmacodynamics
[edit]Trihexyphenidyl is an anticholinergic.[20] It is specifically an antimuscarinic and acts as a non-selective antagonist of all five muscarinic acetylcholine receptors.[20] However, its antagonistic activity is much stronger at the muscarinic acetylcholine M1 and M4 receptors, and it can be described as selective for these receptors.[20][21]
The exact mechanism of action in parkinsonian syndromes is not precisely understood, but it is known that trihexyphenidyl blocks efferent impulses in parasympathetically innervated structures like smooth muscles (spasmolytic activity), salivary glands, and eyes (mydriasis). In higher doses direct central inhibition of cerebral motor centers may contribute. In very high doses central toxicity as seen in atropine overdose is noted.
It possibly also binds to dopamine receptors.[22]
Pharmacokinetics
[edit]Trihexyphenidyl is rapidly absorbed from the gastrointestinal tract. The onset of action is within 1 hour after oral dosing. The peak activity is noted after 2 to 3 hours.[23] The duration of action of one single dose is 6 to 12 hours in a dose dependent manner. It is excreted in the urine, probably as unchanged drug. More precise data in animals and humans have so far not been determined.[24][25]
History
[edit]Trihexyphenidyl has been clinically relevant in trials pertaining to Parkinson's disease since 1949.[26]
In the US, the Food and Drug Association approved Artane, or its generic form Trihexyphenidyl HCL, in June 2003, for the clinical use of all types of parkinsonism.[27]
Society and culture
[edit]Recreational use
[edit]The neurologist Oliver Sacks reported using the drug recreationally in the 1960s.[28] He recalled taking "a large dose" knowing full well the drug was intended for people with Parkinson's. More recounts of Dr. Sacks' experiences — including experimentation with mescaline, psilocybin, LSD, and probably DMT[29] — have been compared in his book Hallucinations.
During the 1970s, trihexyphenidyl (trade name Parkan) was the most popular recreationally used prescription drug in Hungary.[30]
In a 2008 news report, trihexyphenidyl was seen to be used for recreational purposes among Iraqi soldiers and police, among other prescription drugs. The report states that the drugs were taken to relieve combat stress reaction.[31] Although that may be the case for some, others used Artane as a substitute or more intense version of LSD. This was especially prevalent in the 1960s, according to a report in "The New Yorker". Similarly to those in Iraqi forces, some of the appeal was that the individual may retain partial control while under the influence.[32] It is still diverted from its primary use, in combination with other drugs, on the Réunion island (France).[33]
Chemistry
[edit]Trihexyphenidyl can be synthesized in two ways, one linear and one convergent synthesis.
In the first way, the initial 2-(1-piperidino)propiophenone is synthesized in turn by the aminomethylation of acetophenone using paraformaldehyde and piperidine in a Mannich reaction. In the second step the 2-(1-piperidino)propiophenone is reacted with cyclohexylmagnesium bromide in a Grignard reaction.[34]

Stereochemistry
[edit]Trihexyphenidyl has a chiral center and two enantiomers. Medications are racemates.[35]
| Enantiomers | |
|---|---|
 CAS number: 40520-25-0 |
 CAS number: 40520-24-9 |
References
[edit]- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- ^ "TGA eBS - Product and Consumer Medicine Information Licence". www.ebs.tga.gov.au. Retrieved 2020-08-20.
- ^ "New Drug Application Approval Notice" (PDF). U.S. Food and Drug Administration. 2003-05-25. Retrieved 2020-08-20.
- ^ World Health Organization (2025). The selection and use of essential medicines, 2025: WHO Model List of Essential Medicines, 24th list. Geneva: World Health Organization. hdl:10665/382243.
- ^ Harris MK, Shneyder N, Borazanci A, Korniychuk E, Kelley RE, Minagar A (March 2009). "Movement disorders". The Medical Clinics of North America. Common Neurologic Disorders. 93 (2): 371–88, viii. doi:10.1016/j.mcna.2008.09.002. PMID 19272514.
- ^ Inada T (December 2017). "[Drug-Induced Akathisia]". Brain and Nerve = Shinkei Kenkyu No Shinpo. 69 (12): 1417–1424. doi:10.11477/mf.1416200927. PMID 29282345.
- ^ Duma SR, Fung VS (April 2019). "Drug-induced movement disorders". Australian Prescriber. 42 (2): 56–61. doi:10.18773/austprescr.2019.014. PMC 6478951. PMID 31048939.
- ^ Ben-Pazi H (July 2011). "Trihexyphenidyl improves motor function in children with dystonic cerebral palsy: a retrospective analysis". Journal of Child Neurology. 26 (7): 810–816. doi:10.1177/0883073810392582. PMID 21498790.
- ^ Sanger TD, Bastian A, Brunstrom J, Damiano D, Delgado M, Dure L, et al. (May 2007). "Prospective open-label clinical trial of trihexyphenidyl in children with secondary dystonia due to cerebral palsy". Journal of Child Neurology. 22 (5): 530–7. doi:10.1177/0883073807302601. PMID 17690057. S2CID 73087776.
- ^ Tarnopolsky M, Alshahoumi R (November 2015). ""Complex I Deficiency". In Saneto R, Parikh S, Cohen BH (eds.). Mitochondrial Case Studies: Underlying Mechanisms and Diagnosis. Academic Press. pp. 257–64. ISBN 978-0-12-801149-2.
- ^ Reid SM, Westbury C, Guzys AT, Reddihough DS (March 2020). "Anticholinergic medications for reducing drooling in children with developmental disability". Developmental Medicine and Child Neurology. 62 (3): 346–353. doi:10.1111/dmcn.14350. hdl:11343/286392. PMID 31495925.
- ^ Aracava Y, Albuquerque EX, Pereira EF (November 2023). "(R,S)-trihexyphenidyl, acting via a muscarinic receptor-independent mechanism, inhibits hippocampal glutamatergic and GABAergic synaptic transmissions: Potential relevance for treatment of organophosphorus intoxication". Neuropharmacology. 239 109684. doi:10.1016/j.neuropharm.2023.109684. PMC 10590273. PMID 37549771.
- ^ "TGA eBS - Product and Consumer Medicine Information Licence". Therapeutic Goods Administration. Australian Government. July 2022. Retrieved 2024-02-12.
- ^ Doshay LJ, Constable K, Zier A (April 1954). "Five year follow-up of treatment with trihexyphenidyl (artane); outcome in four hundred eleven cases of paralysis agitans". Journal of the American Medical Association. 154 (16): 1334–6. doi:10.1001/jama.1954.02940500014005. PMID 13151847.
- ^ "Trihexyphenidyl". Web MD. First Databank Inc. nd.
- ^ "Trihexyphenidyl". Toxnet. Archived from the original on June 29, 2019.
- ^ Chiappini S, Mosca A, Miuli A, Semeraro FM, Mancusi G, Santovito MC, et al. (February 2022). "Misuse of Anticholinergic Medications: A Systematic Review". Biomedicines. 10 (2): 355. doi:10.3390/biomedicines10020355. PMC 8962297. PMID 35203563.
- ^ Liao WB, Bullard MJ, Kuo CT, Hsiao CT, Chu PH, Chiang CW (November 1996). "Anticholinergic overdose induced torsade de pointes successfully treated with verapamil". Japanese Heart Journal. 37 (6): 925–931. doi:10.1536/ihj.37.925. PMID 9057687.
- ^ Hsu YC, Yeh YW (December 2014). "Multidrug overdose-induced myoclonus complicated by rhabdomyolysis: possible role and mechanism of muscle toxicity of risperidone". Journal of Clinical Pharmacy and Therapeutics. 39 (6): 698–700. doi:10.1111/jcpt.12205. PMID 25203795.
- ^ a b c Lakstygal AM, Kolesnikova TO, Khatsko SL, Zabegalov KN, Volgin AD, Demin KA, et al. (May 2019). "DARK Classics in Chemical Neuroscience: Atropine, Scopolamine, and Other Anticholinergic Deliriant Hallucinogens". ACS Chem Neurosci. 10 (5): 2144–2159. doi:10.1021/acschemneuro.8b00615. PMID 30566832.
- ^ Giachetti A, Giraldo E, Ladinsky H, Montagna E (September 1986). "Binding and functional profiles of the selective M1 muscarinic receptor antagonists trihexyphenidyl and dicyclomine". Br J Pharmacol. 89 (1): 83–90. doi:10.1111/j.1476-5381.1986.tb11123.x. PMC 1917044. PMID 2432979.
- ^ Berke JD, Hyman SE (March 2000). "Addiction, dopamine, and the molecular mechanisms of memory". Neuron. 25 (3): 515–32. doi:10.1016/S0896-6273(00)81056-9. PMID 10774721. S2CID 14766533.
- ^ "Trihexyphenidyl Hydrochloride". Drugs.com.
- ^ Watson Laboratories Inc. trihexyphenidyl hydrochloride tablets, USP. prescribing information. Corona, CA; 2005 May.
- ^ McEvoy GK, ed. (2006). "Trihexyphenidyl". AHFS drug information. Bethesda, MD: American Society of Health-System Pharmacists. p. 1256.
- ^ Doshay LJ, Constable K (August 1949). "Artane therapy for parkinsonism; a preliminary study of results in 117 cases". Journal of the American Medical Association. 140 (17): 1317–22. doi:10.1001/jama.1949.02900520003002. PMID 18137284.
- ^ Katz R, Feeney J, Ressler T, David P. "Approval Package for Application No. 6-773/36" (PDF). U.S. Food and Drug Association. Archived from the original (PDF) on February 11, 2017. Retrieved 8 May 2017.
- ^ Smith K (2012-10-30), "Oliver Sacks shares his hallucinations", The Guardian, Guardian
- ^ Sacks O (2012). "Chapter 6". Hallucinations. Random House Inc.
- ^ Sándor B, József R, Zsófia TE (19 November 2011). "Kábítószerek a szocializmusban" [Drug use in socialist Hungary]. Múlt-kor.
- ^ Al-Husaini M, Goode E (2008-12-20). "Abuse of Prescription Drugs Rises Among Stressed Iraqi Soldiers". The New York Times.
- ^ Sacks O (20 August 2012). "Altered States". The New Yorker. Condé Nast. Retrieved 7 May 2017.
- ^ Guyon J, Maillot A, Bastard S, Weisse F, Daveluy A, Mété D (February 2025). "Psychoactive cocktail consumption on Reunion Island: A case report". Journal of Analytical Toxicology. 49 (5): 369–373. doi:10.1093/jat/bkaf009. PMID 39953779.
- ^ Weiss MJ, O'Donoghue MD (September 1957). "Synthesis of Certain 3-Hydroxy-3-phenylpropylsulfonium Salts. Sulfonium Analogs of Artane (Trihexyphenidyl) and Pathilon (Tridihexethyl Iodide)". Journal of the American Chemical Society. 79 (17): 4771–6. Bibcode:1957JAChS..79.4771W. doi:10.1021/ja01574a048.
- ^ Rote Liste Service GmbH (Hrsg.) (2017). Rote Liste 2017 – Arzneimittelverzeichnis für Deutschland (einschließlich EU-Zulassungen und bestimmter Medizinprodukte). Vol. 57. Frankfurt/Main: Rote Liste Service GmbH. p. 224. ISBN 978-3-946057-10-9.
![]() This article incorporates public domain material from Toxnet:Trihexyphenidyl. United States Department of Health and Human Services. Retrieved 8 May 2017.
This article incorporates public domain material from Toxnet:Trihexyphenidyl. United States Department of Health and Human Services. Retrieved 8 May 2017.

