| Talc | |
|---|---|
 | |
| General | |
| Category | Phyllosilicate minerals, pyrophyllite-talc group |
| Formula | Mg3Si4O10(OH)2 |
| IMA symbol | Tlc[1] |
| Strunz classification | 9.EC.05 |
| Crystal system | Monoclinic or triclinic[2] |
| Crystal class | Either prismatic (2m) or pinacoidal (1)[3] |
| Space group | C2/c or C1 |
| Unit cell | a = 5.291 Å, b = 9.173 Å c = 5.290 Å; α = 98.68° β = 119.90°, γ = 90.09°; Z = 2 or a = 5.287 Å, b = 9.158 Å c = 18.95 [Å], β = 99.3°; Z = 4[3] |
| Identification | |
| Color | Light to dark green, brown, white, grey, colorless |
| Crystal habit | Foliated to fibrous masses, rare as platey to pyramidal crystals |
| Cleavage | Perfect on {001} basal cleavage |
| Fracture | Flat surfaces (not cleavage), fracture in an uneven pattern |
| Tenacity | Sectile |
| Mohs scale hardness | 1 (defining mineral) |
| Luster | Waxy or pearly |
| Streak | White jot to pearl black |
| Diaphaneity | Translucent |
| Specific gravity | 2.58–2.83 |
| Optical properties | Biaxial (−) |
| Refractive index | nα = 1.538 – 1.550 nβ = 1.589 – 1.594 nγ = 1.589 – 1.600 |
| Birefringence | δ = 0.051 |
| Pleochroism | Weak in dark varieties |
| Ultraviolet fluorescence | Short UV: orange yellow; long UV: yellow |
| References | [3][4][5] |
Talc, or talcum, is a clay mineral composed of hydrated magnesium silicate, with the chemical formula Mg3Si4O10(OH)2. Talc in powdered form, often combined with corn starch, is used as baby powder. This mineral is used as a thickening agent and lubricant. It is an ingredient in ceramics, paints, and roofing material. It is a main ingredient in many cosmetics.[6] It occurs as foliated to fibrous masses, and in an exceptionally rare crystal form. It has a perfect basal cleavage and an uneven flat fracture, and it is foliated with a two-dimensional platy form.
The Mohs scale of mineral hardness, based on scratch hardness comparison, defines value 1 as the hardness of talc, the softest mineral. When scraped on a streak plate, talc produces a white streak, though this indicator is of little importance, because most silicate minerals produce a white streak. Talc is translucent to opaque, with colors ranging from whitish grey to green with a vitreous and pearly luster. Talc is not soluble in water, and is slightly soluble in dilute mineral acids.[7]
Soapstone is a metamorphic rock composed predominantly of talc.
Etymology
[edit]The word talc derives from Persian: تالک tālk. In ancient times, the word was used for various related minerals, including talc, mica, and selenite.[8]
Formation
[edit]
Talc dominantly forms from the metamorphism of magnesian minerals such as serpentine, pyroxene, amphibole, and olivine, in the presence of carbon dioxide and water. This is known as "talc carbonation" or "steatization" and produces a suite of rocks known as talc carbonates.[citation needed]
Talc is primarily formed by hydration and carbonation by this reaction:
- + → + +
Talc can also be formed via a reaction between dolomite and silica, which is typical of skarnification of dolomites by silica-flooding in contact metamorphic aureoles:
- + + → + +
Talc can also be formed from magnesium chlorite and quartz in blueschist and eclogite metamorphism by the following metamorphic reaction:
Talc is also found as a diagenetic mineral in sedimentary rocks where it can form from the transformation of metastable hydrated magnesium-clay precursors such as kerolite, sepiolite, or stevensite that can precipitate from marine and lake water in certain conditions.[9]
In this reaction, the ratio of talc and kyanite depends on aluminium content, with more aluminous rocks favoring production of kyanite. This is typically associated with high-pressure, low-temperature minerals such as phengite, garnet, and glaucophane within the lower blueschist facies. Such rocks are typically white, friable, and fibrous, and are known as whiteschist.[citation needed]
Talc is a trioctahedral layered mineral; its structure is similar to pyrophyllite, but with magnesium in the octahedral sites of the composite layers.[2] The crystal structure of talc is described as TOT, meaning that it is composed of parallel TOT layers weakly bonded to each other by weak van der Waals forces. The TOT layers in turn consist of two tetrahedral sheets (T) strongly bonded to the two faces of a single trioctahedral sheet (O). It is the weak bonding between TOT layers that gives talc its perfect basal cleavage and softness.[10]
The tetrahedral sheets consist of silica tetrahedra, which are silicon ions surrounded by four oxygen ions. The tetrahedra each share three of their four oxygen ions with neighboring tetrahedra to produce a hexagonal sheet. The remaining oxygen ion (the apical oxygen ion) is available to bond with the trioctahedral sheet.[11]
The trioctahedral sheet has the structure of a sheet of the mineral brucite. Apical oxygens take the place of some of the hydroxyl ions that would be present in a brucite sheet, bonding the tetrahedral sheets tightly to the trioctahedral sheet.[12]
Tetrahedral sheets have a negative charge, since their bulk composition is Si4O4−10. The trioctahedral sheet has an equal positive charge, since its bulk composition is Mg3(OH)4+2 The combined TOT layer thus is electrically neutral.[13]
Because the hexagons in the T and O sheets are slightly different in size, the sheets are slightly distorted when they bond into a TOT layer. This breaks the hexagonal symmetry and reduces it to monoclinic or triclinic symmetry.[14] However, the original hexahedral symmetry is discernible in the pseudotrigonal character of talc crystals.[3]
-
View of tetrahedral sheet structure of talc. The apical oxygen ions are tinted pink.
-
View of trioctahedral sheet of talc. Yellow spheres are hydroxyl; blue are magnesium. Apical oxygen binding sites are white.
-
Talc crystal viewed along the [100] axis, looking along the layers of the crystal
Occurrence
[edit]This section needs additional citations for verification. (October 2025) |
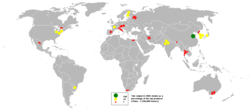

Talc is a common metamorphic mineral in metamorphic belts that contain ultramafic rocks, such as soapstone (a high-talc rock), and within whiteschist and blueschist metamorphic terranes. Prime examples of whiteschists include the Franciscan Metamorphic Belt of the western United States, the western European Alps especially in Italy, certain areas of the Musgrave Block, and some collisional orogens such as the Himalayas, which stretch along Pakistan, India, Nepal, and Bhutan.
Talc carbonate ultramafics are typical of many areas of the Archaean cratons, notably the komatiite belts of the Yilgarn craton in Western Australia. Talc-carbonate ultramafics are also known from the Lachlan Fold Belt, eastern Australia, from Brazil, the Guiana Shield, and from the ophiolite belts of Turkey, Oman, and the Middle East.
China is the key world talc and steatite-producing country with an output of about 2.2 million tonnes (2016), which accounts for 30% of total global output. The other major producers are Brazil (12%), India (11%), the U.S. (9%), France (6%), Finland (4%), Italy, Russia, Canada, and Austria (2%, each).[15]
Notable economic talc occurrences include the Mount Seabrook talc mine, Western Australia, formed upon a polydeformed, layered ultramafic intrusion. The France-based Luzenac Group is the world's largest supplier of mined talc. Its largest talc mine at Trimouns near Luzenac in southern France produces 400,000 tonnes of talc per year.
Conflict mineral
[edit]Extraction in disputed areas of Nangarhar province, Afghanistan, has led the international monitoring group Global Witness to declare talc a conflict resource, as the profits are used to fund armed confrontation between the Taliban and Islamic State.[16]
Uses
[edit]This section needs additional citations for verification. (October 2025) |


Talc is used in many industries, including paper making, plastic, paint and coatings (e.g. for metal casting molds), rubber, food, electric cable, pharmaceuticals, cosmetics, and ceramics. A coarse grayish-green high-talc rock is soapstone or steatite, used for stoves, sinks, electrical switchboards, etc. It is often used for surfaces of laboratory table tops and electrical switchboards because of its resistance to heat, electricity, and acids.
In finely ground form, talc finds use as a cosmetic (talcum powder), as a lubricant, and as a filler in paper manufacture. It is used to coat the insides of inner tubes and rubber gloves during manufacture to keep the surfaces from sticking. Talcum powder, with heavy refinement, has been used in baby powder, an astringent powder used to prevent diaper rash (nappy rash). The American Academy of Pediatrics recommends that parents avoid using baby powder because it poses a risk of respiratory problems, including breathing trouble and serious lung damage if inhaled. The small size of the particles makes it difficult to keep them out of the air while applying the powder. Zinc oxide-based ointments are a much safer alternative.[17]
Soapstone (massive talc) is often used as a marker for welding or metalworking.[18][19]
Talc is also used as food additive or in pharmaceutical products as a glidant. In medicine, talc is used as a pleurodesis agent to prevent recurrent pleural effusion or pneumothorax. In the European Union, the additive number is E553b. Talc may be used in the processing of white rice as a buffing agent in the polishing stage.
Due to its low shear strength, talc is one of the oldest known solid lubricants. It also has some limited use as a friction-reducing additive in lubricating oils.[20]
Talc is widely used in the ceramics industry in both bodies and glazes. In low-fire art-ware bodies, it imparts whiteness and increases thermal expansion to resist crazing. In stonewares, small percentages of talc are used to flux the body and therefore improve strength and vitrification. It is a source of MgO flux in high-temperature glazes (to control melting temperature). It is also employed as a matting agent in earthenware glazes and can be used to produce magnesia mattes at high temperatures.
ISO standard for quality (ISO 3262)
| Type | Talc content min. wt% | Loss on ignition at 1000 °C, wt % | Solubility in HCl, max. wt % |
|---|---|---|---|
| A | 95 | 4–6.5 | 5 |
| B | 90 | 4–9 | 10 |
| C | 70 | 4–18 | 30 |
| D | 50 | 4–27 | 30 |
Patents are pending on the use of magnesium silicate as a cement substitute. Its production requirements are less energy-intensive than ordinary Portland cement (at a heating requirement of around 650 °C for talc compared to 1,500 °C for limestone to produce Portland cement), while it absorbs far more carbon dioxide as it hardens. This results in a negative carbon footprint overall, as the cement substitute removes 0.6 tonnes of CO2 per tonne used. This contrasts with a positive carbon footprint of 0.4 tonnes per tonne of conventional cement.[21]
Talc is sometimes used as an adulterant to illegal heroin, to expand volume and weight and thereby increase its street value. With intravenous use, it may lead to pulmonary talcosis, a granulomatous inflammation in the lungs.
Sterile talc powder
[edit]Sterile talc powder (NDC 63256-200-05) is a sclerosing agent used in the procedure of pleurodesis. This can be helpful as a cancer treatment to prevent pleural effusions (an abnormal collection of fluid in the space between the lungs and the thoracic wall). It is inserted into the space via a chest tube, causing it to close up, so fluid cannot collect there. The product can be sterilized by dry heat, ethylene oxide, or gamma irradiation.[22]
Safety
[edit]Suspicions have been raised that talc use contributes to certain types of disease, mainly cancers of the ovaries and lungs. According to the IARC, talc containing asbestos is classified as a group 1 agent (carcinogenic to humans), talc use in the perineum is classified as group 2B (possibly carcinogenic to humans), and talc not containing asbestos is classified as group 2A (probably carcinogenic to humans).[23] Reviews by Cancer Research UK and the American Cancer Society conclude that some studies have found a link, but other studies have not.[24][25]
The studies discuss pulmonary issues,[26] lung cancer,[27][28] and ovarian cancer.[29] One of these, published in 1993, was a US National Toxicology Program report, which found that cosmetic grade talc containing no asbestos-like fibres was correlated with tumor formation in rats forced to inhale talc for 6 hours a day, five days a week over at least 113 weeks.[27] A 1971 paper found particles of talc embedded in 75% of the ovarian tumors studied.[30] In 2018, Health Canada issued a warning against inhaling talcum powder or women's using it perineally.[31]
In contrast, however, research published in 1995 and 2000 concluded that, although it was plausible that talc could cause ovarian cancer, no conclusive evidence had been shown.[32][33] Further, a 2008 European Journal of Cancer Prevention review of ovarian cancer and talc use studies pointed out that, although many of them examined the duration, frequency, and accumulation of hygienic talc use, few found a positive association among these factors and some found a negative one: "It may be argued that the overall null findings associated with talc-dusted diaphragms and condom use is more convincing evidence for a lack of a carcinogenic effect, especially given the lack of an established correlation between perineal dusting frequency and ovarian tissue talc concentrations and the lack of a consistent dose-response relationship with ovarian cancer risk." Instead, the authors credited powdered talc with "a high degree of safety."[34]
Similarly, in a 2014 article published in a leading cancer journal, the Journal of the National Cancer Institute, researchers reported the results of a survey of 61,576 postmenopausal women, more than half of whom had used talc powder perineally. The researchers compared the subjects' reports of their own talc use with their reports of having had ovarian cancer diagnosed by their doctors, and found, regardless of subjects' age and tubal ligation status, "Ever use of perineal powder ... was not associated with risk of ovarian cancer compared with never use," nor was any greater individual cancer risk associated with longer use of talc powder. On this basis, the article concluded, "perineal powder use does not appear to influence ovarian cancer risk."[35] The Cosmetic Ingredient Review Expert Panel concluded in 2015 that talc, in the concentrations currently used in cosmetics, is safe.[36]
In July 2024, the International Agency for Research on Cancer listed talc as "probably" carcinogenic for humans. The study is based on limited evidence it could cause ovarian cancer in humans.[37][better source needed]
Industrial grade
[edit]In the United States, the Occupational Safety and Health Administration and National Institute for Occupational Safety and Health have set occupational exposure limits to respirable talc dusts at 2 mg/m3 over an eight-hour workday. At levels of 1000 mg/m3, inhalation of talc is considered immediately dangerous to life and health.[38]
Food grade
[edit]The United States Food and Drug Administration considers talc (magnesium silicate) generally recognized as safe (GRAS) to use as an anticaking agent in table salt in concentrations smaller than 2%.[39]
Association with asbestos
[edit]One particular issue with commercial use of talc is its frequent co-location in underground deposits with asbestos ore. Asbestos is a general term for different types of fibrous silicate minerals, desirable in construction for their heat resistant properties.[40] There are six varieties of asbestos; the most common variety in manufacturing, white asbestos, is in the serpentine family.[41] Serpentine minerals are sheet silicates; although not in the serpentine family, talc is also a sheet silicate, with two sheets connected by magnesium cations. The frequent co-location of talc deposits with asbestos may result in contamination of mined talc with white asbestos, which poses serious health risks when dispersed into the air and inhaled. Stringent quality control since 1976, including separating cosmetic- and food-grade talc from that destined for industrial use, has largely eliminated this issue, but it remains a potential hazard requiring mitigation in the mining and processing of talc.[42] A 2010 US FDA survey failed to find asbestos in a variety of talc-containing products.[43] A 2018 Reuters investigation asserted that pharmaceuticals company Johnson & Johnson knew for decades that there was asbestos in its baby powder,[44] and in 2020 the company stopped selling its baby powder in the US and Canada.[45] There were calls for Johnson & Johnson's largest shareholders to force the company to end global sales of baby powder, and hire an independent firm to conduct a racial justice audit as it had been marketed to African American and overweight women.[46] On August 11, 2022, the company announced it would stop making talc-based powder by 2023 and replace it with cornstarch-based powders. The company said the talc-based powder is safe to use and does not contain asbestos.[47]
Litigation
[edit]In 2006 the International Agency for Research on Cancer classified talcum powder as a possible human carcinogen if used in the female genital area. Despite this, no federal agency in the US acted to remove talcum powder from the market or add warnings.[48]
In February 2016, as the result of a lawsuit against Johnson & Johnson (J&J), a St. Louis jury awarded $72 million to the family of an Alabama woman who died from ovarian cancer. The family claimed that the use of talcum powder was responsible for her cancer.
In May 2016, a South Dakota woman was awarded $55 million as the result of another lawsuit against J&J.[49] The woman had used Johnson & Johnson's Baby Powder for more than 35 years before being diagnosed with ovarian cancer in 2011.[50]
In October 2016, a St. Louis jury awarded $70.1 million to a Californian woman with ovarian cancer who had used Johnson's Baby Powder for 45 years.[51]
In August 2017, a Los Angeles jury awarded $417 million to a Californian woman, Eva Echeverria, who developed ovarian cancer as a "proximate result of the unreasonably dangerous and defective nature of talcum powder", her lawsuit against Johnson & Johnson stated.[52] On 20 October 2017, Los Angeles Superior Court judge Maren Nelson dismissed the verdict. The judge stated that Echeverria proved there is "an ongoing debate in the scientific and medical community about whether talc more probably than not causes ovarian cancer and thus (gives) rise to a duty to warn", but not enough to sustain the jury's imposition of liability against Johnson & Johnson stated, and concluded that Echeverria did not adequately establish that talc causes ovarian cancer.[53][54]
In July 2018, a court in St. Louis awarded a $4.7bn claim ($4.14bn in punitive damages and $550m in compensatory damages) against J&J to 22 claimant women, concluding that the company had suppressed evidence of asbestos in its products for more than four decades.[55]
At least 1,200 to 2,000 other talcum powder-related lawsuits were pending as of 2016[update].[51][56]
In 2020 J&J stopped sales of its talcum-based baby powder, which it had been selling for 130 years. J&J created a subsidiary responsible for the claims in an effort to resolve the lawsuits in bankruptcy court. In 2023 J&J proposed a nearly $9bn settlement with 50,000 claimants saying the claims were "specious" but it wanted to move on from the issue, but judges blocked the plans, ruling that the subsidiary was not in financial distress and could not use the bankruptcy system to resolve the lawsuits.[57]
In July 2023 J&J sued researchers who linked talc to cancer, alleging they used junk science to disparage the company's products, while defendants say the lawsuits are meant to silence scientists.[58]
See also
[edit]- Pyrophyllite – Phyllosilicate mineral in the pyrophyllite-talc group
- Serpentinite – Rock formed by transformation of olivine
- Sillimanite – Nesosilicate mineral
References
[edit]- ^ Warr, L. N. (2021). "IMA–CNMNC approved mineral symbols". Mineralogical Magazine. 85 (3): 291–320. Bibcode:2021MinM...85..291W. doi:10.1180/mgm.2021.43. S2CID 235729616.
- ^ a b W. A. Deer; R. A. Howie; J. Zussman, eds. (1992). An Introduction to the Rock-Forming Minerals (2nd ed.). Prentice Hall. ISBN 0-582-30094-0.
- ^ a b c d Anthony, John W.; Bideaux, Richard A.; Bladh, Kenneth W.; Nichols, Monte C., eds. (1995). "Talc" (PDF). Handbook of Mineralogy. Vol. II (Silica, Silicates). Chantilly, Virginia: Mineralogical Society of America. ISBN 0-9622097-1-6.
- ^ "Talc". Mindat.org.
- ^ Talc Mineral Data, WebMineral.com
- ^ "Talc". Minerals Education Coalition.
- ^ Profiles of Drug Substances, Excipients and Related Methodology, Volume 36 ISBN 978-0-123-87667-6 p. 283
- ^ Harper, Douglas. "talc". Online Etymology Dictionary.
- ^ Strauss, Justin V.; MacDonald, Francis A.; Halverson, Galen P.; Tosca, Nicholas J.; Schrag, Daniel P.; Knoll, Andrew H. (2015). "Stratigraphic evolution of the Neoproterozoic Callison Lake Formation: Linking the break-up of Rodinia to the Islay carbon isotope excursion". American Journal of Science. 315 (10). American Journal of Science (AJS): 881–944. Bibcode:2015AmJS..315..881S. doi:10.2475/10.2015.01. ISSN 0002-9599. S2CID 130671089.
- ^ Nesse, William D. (2000). Introduction to mineralogy. New York: Oxford University Press. p. 238. ISBN 978-0-19-510691-6.
- ^ Nesse 2000, p. 235.
- ^ Nesse 2000, pp. 235–237.
- ^ Nesse 2000, p. 238.
- ^ Nesse 2000, pp. 239, 242.
- ^ Sergeeva, Anna (18 July 2018). "China, Brazil, the U.S. and India Remain the Major Consumers on the Global Talc Market". IndexBox.
- ^ "Talc: the everyday mineral funding Afghan insurgents". Global Witness. Archived from the original on 24 May 2018. Retrieved 24 May 2018.
- ^ "Is it safe to use baby powder on my baby?". Babycenter.com (2017-05-01). Retrieved on 2017-05-06.
- ^ "Why Do Welders Use Soapstone?". Welders Manual. 28 March 2021. Retrieved 7 March 2022.
- ^ Young, Pierre (10 June 2020). "What Is Soapstone Used for in Welding?". Welding Headquarters. Retrieved 7 March 2022.
- ^ Rudenko, Pavlo; Bandyopadhyay, Amit (2013). "Talc as friction-reducing additive to lubricating oil". Applied Surface Science. 276: 383–389. Bibcode:2013ApSS..276..383R. doi:10.1016/j.apsusc.2013.03.102.
- ^ Jha, Alok (31 December 2008) Revealed: The cement that eats carbon dioxide, The Guardian
- ^ American Thoracic Society (November 2000). "Management of Malignant Pleural Effusions". Am J Respir Crit Care Med. 162 (5): 1987–2001. doi:10.1164/ajrccm.162.5.ats8-00. PMID 11069845.
- ^ List of Classifications Archived 10 June 2017 at the Wayback Machine, International Agency for Research on Cancer
- ^ Talcum powder and cancer Archived 15 August 2012 at the Wayback Machine, Cancerresearch.uk
- ^ Talcum Powder and Cancer Archived 20 November 2016 at the Wayback Machine, American Cancer Society
- ^ Hollinger, MA (1990). "Pulmonary toxicity of inhaled and intravenous talc". Toxicology Letters. 52 (2): 121–7, discussion 117–9. doi:10.1016/0378-4274(90)90145-C. PMID 2198684.
- ^ a b National Toxicology, Program (1993). "NTP Toxicology and Carcinogenesis Studies of Talc (Non-Asbestiform) in Rats and Mice (Inhalation Studies)". National Toxicology Program Technical Report Series. 421: 1–287. PMID 12616290.
- ^ "Worker Health Study Summaries – Talc Miners & Millers". The National Institute for Occupational Safety and Health (NIOSH). 1995. Archived from the original on 7 July 2022. Retrieved 31 August 2022.
- ^
Harlow, Cramer, Bell; et al. (1992). "Perineal exposure to talc and ovarian cancer risk". Obstetrics and Gynecology. 80 (1): 19–26. PMID 1603491.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Henderson WJ, Joslin CA, Turnbull AC, Griffiths K (1971). "Talc and carcinoma of the ovary and cervix". J Obstet Gynaecol Br Commonw. 78 (3): 266–272. doi:10.1111/j.1471-0528.1971.tb00267.x. PMID 5558843. S2CID 32300387.
- ^ "Talc – Potential Risk of Lung Effects and Ovarian Cancer". 19 November 2018.
- ^ Harlow, BL; Hartge, PA (April 1995). "A review of perineal talc exposure and risk of ovarian cancer". Regulatory Toxicology and Pharmacology. 21 (2): 254–60. doi:10.1006/rtph.1995.1039. PMID 7644715.
- ^ Gertig, D. M.; Hunter, D. J.; Cramer, D. W.; Colditz, G. A.; Speizer, F. E.; Willett, W. C.; Hankinson, S. E. (2 February 2000). "Prospective Study of Talc Use and Ovarian Cancer" (PDF). JNCI Journal of the National Cancer Institute. 92 (3): 249–252. doi:10.1093/jnci/92.3.249. PMID 10655442.
- ^ Muscat, Joshua E.; Huncharek, Michael S. (April 2008). "Perineal Talc Use and Ovarian Cancer: A Critical Review". European Journal of Cancer Prevention. 17 (2): 139–146. doi:10.1097/CEJ.0b013e32811080ef. PMC 3621109. PMID 18287871.
- ^ Houghton, Serena C.; Reeves, Katherine W.; Hankinson, Susan E.; Crawford, Lori; Lane, Dorothy; Wactawski-Wende, Jean; Thomson, Cynthia A.; Ockene, Judith K. (11 September 2014). "Perineal Powder Use and Risk of Ovarian Cancer". JNCI Journal of the National Cancer Institute. 106 (9) dju208. doi:10.1093/jnci/dju208. PMC 4200058. PMID 25214560.
- ^ Gruber, James (November–December 2019). "Do Cosmetic Consumers Really Know What Ingredients They Are Using? An Inquiry into the Search for the 'Truth'". Skeptical Inquirer. 43 (6): 54.
- ^ "WHO agency says talc is 'probably' cancer-causing". Philippine Daily Inquirer. 6 July 2024. Retrieved 7 July 2024.
- ^ "NIOSH Pocket Guide to Chemical Hazards". Centers for Disease Control and Prevention. 2011.
- ^ "Code of Federal Regulations". U.S. Food and Drug Administration. 2009.
- ^ "Asbestos". Minerals Education Coalition.
- ^ Plummer, Charles C.; Carlson, Diane H.; Hammersley, Lisa (22 January 2018). Physical Geology (Sixteenth ed.). McGraw-Hill Education. ISBN 978-1-260-09165-6.
- ^ "Is talcum powder asbestos?". The Straight Dope. 16 February 1990. Retrieved 9 March 2019.
- ^ "Talc Ingredients". U.S. Food and Drug Administration. 2010.
- ^ "J&J knew for decades that asbestos lurked in its Baby Powder". Reuters. Retrieved 15 December 2018.
- ^ "Johnson & Johnson stops selling baby powder in US". BBC News. 20 May 2020. Retrieved 20 May 2020.
- ^ Perkins, Tom (27 April 2022). "Johnson & Johnson shareholders called on to end talc baby powder sales". The Guardian.
- ^ "J&J to replace talc-based powder with cornstarch after cancer lawsuits". BBC News. 12 August 2022. Retrieved 12 August 2022.
- ^ Rabin, Roni Caryn (22 August 2017). "$417 Million Awarded in Suit Tying Johnson's Baby Powder to Cancer". The New York Times. Retrieved 13 July 2018.
- ^ McLean, Rob (3 May 2016). "Johnson & Johnson just lost another talcum powder cancer lawsuit". CNNMoney. Retrieved 18 August 2016.
- ^ "Professor Hanington's Speaking of Science: Does talc cause cancer?". Elko Daily Free Press. 15 October 2016. Retrieved 9 March 2021.
- ^ a b Does baby powder cause cancer? Another jury thinks so, awarding $70 million to a California woman. LA Times (2016-10-28). Retrieved on 2017-05-06.
- ^ Jury awards $417M in lawsuit linking talcum powder to cancer. The Chronicle Herald (21 August 2017)
- ^ Bellon, Tina (21 October 2017). "California judge tosses $417 million talc cancer verdict against..." Reuters. Retrieved 13 July 2018.
- ^ Frankel, Alison (24 October 2017). "Dismissal of $417 million verdict v. J&J is disaster for talc..." Reuters. Retrieved 13 July 2018.
- ^ Butler, Sarah (13 July 2018). "Johnson & Johnson ordered to pay $4.7bn in talc powder claim". The Guardian. Retrieved 13 July 2018.
- ^ Woman wins $55M verdict against Johnson & Johnson in cancer suit. NY Daily News (3 May 2016)
- ^ "J&J strikes US states deal over baby powder claims". BBC News. 23 January 2024. Retrieved 14 April 2024.
- ^ Knauth, Dietrich (13 July 2023). "Johnson & Johnson sues researchers who linked talc to cancer". Reuters. Retrieved 17 July 2023.
Chemical and Physical Properties
Composition and Structure
Talc is a hydrous magnesium silicate mineral with the ideal chemical formula Mg₃Si₄O₁₀(OH)₂.[10][11] This composition consists of 63.6% SiO₂, 31.9% MgO (as Mg), and 4.8% H₂O by weight in pure form.[12] Talc exhibits a trioctahedral structure within the phyllosilicate (sheet silicate) group, featuring alternating layers of tetrahedral silica sheets and octahedral magnesium hydroxide sheets.[13] The tetrahedral sheets comprise SiO₄ units linked in a hexagonal pattern, while the central octahedral layer has magnesium ions octahedrally coordinated by oxygen and hydroxyl groups.[14] The individual 2:1 layers in talc are stacked and held together primarily by weak van der Waals forces rather than strong ionic or covalent bonds, resulting in perfect basal cleavage and a platy habit.[14] Substitutions within the lattice, such as Fe²⁺ or Al³⁺ replacing Mg²⁺ in octahedral sites or Al³⁺ substituting for Si⁴⁺ in tetrahedral sites, introduce minor compositional variations that can alter color from white to gray or green.[15] Associated impurities in natural deposits often include carbonates like calcite or dolomite, silicates such as chlorite or serpentine, and quartz, which affect overall purity and are deposit-specific.[12][16] Talc is distinguished from similar phyllosilicates by its magnesium-dominated trioctahedral occupancy; pyrophyllite, for instance, is dioctahedral with Al₂Si₄O₁₀(OH)₂, lacking magnesium in the octahedral sheet.[17] Chlorite, another sheet silicate, incorporates an additional interlayer of brucite-like (Mg,Fe)(OH)₂ sheets between the 2:1 talc-like layers, yielding a formula approximating (Mg,Fe,Al)₆(AlSi₃)O₁₀(OH)₈.[17] These structural and compositional differences underpin distinct mineral behaviors, though impure deposits may require analytical methods like X-ray diffraction for accurate identification.[10]Physical Characteristics and Mohs Scale
Talc exhibits a Mohs hardness of 1, defining it as the softest mineral and the standard reference for the lowest point on the scale.[18] This exceptional softness arises from its layered silicate structure, featuring weak interlayer bonds that allow easy deformation and scratching by a fingernail.[19] The mineral displays a characteristic greasy feel when handled, resulting from the sliding of its fine, platy particles against the skin.[18] In terms of luster, talc shows a pearly to greasy appearance, often translucent in thin sheets.[20] It possesses perfect cleavage along the {001} basal plane, enabling it to split into flexible, thin laminae without brittle fracture.[19] Color variations typically range from white to pale green or grayish hues, influenced by minor impurities, with a white streak.[11] The density of talc falls between 2.7 and 2.8 g/cm³, reflecting its relatively low mass due to the predominance of lightweight elements in its composition.[11] These physical traits, including the platy crystal habit and interlayer weakness, confer poor thermal and electrical conductivity, positioning talc as an effective insulator in bulk form.[21] Such properties underpin its industrial value as a lubricant and filler, where minimal friction and reinforcement without added hardness are desired.[18]Etymology and History
Etymology
The word talc derives from the Arabic ṭalq (طَلْق), originally referring to mica or a similar flaky mineral, due to the shared schistose texture and appearance of early specimens.[22][23] This term traces further to the Persian talk or tālk, possibly denoting a medicament or pure substance, which facilitated its transmission through trade routes.[24][25] By the 16th century, the word entered European languages via Medieval Latin talcum or talcus, as documented in mineralogical texts, where it initially encompassed various lightweight, lamellar minerals before being narrowed to the specific hydrous magnesium silicate Mg₃Si₄O₁₀(OH)₂.[22][23] Georgius Agricola formalized its usage in 1564, distinguishing talc from mica amid growing systematic classification efforts in geology.[26] The English adoption occurred around 1610, aligning with the term's refinement to exclude broader connotations of purity or unrelated phyllosilicates.[23]Historical Discovery and Early Uses
Talc, primarily recognized in its massive form as soapstone, was utilized in prehistoric Europe for carving vessels, tools, and ornaments due to its softness and workability. Archaeological evidence from Scandinavia indicates soapstone quarrying for cooking pots and household items dating back to the Neolithic period, with production continuing through the Viking Age for jewelry and structural elements like stove linings.[27] Similarly, prehistoric sites in North America reveal Native American use of soapstone for bowls, cooking vessels, and shaft straighteners, exploiting its thermal stability to retain heat without cracking.[28] In ancient Mediterranean civilizations, soapstone's properties enabled fine carvings, such as scarab amulets by Egyptians and stamps by Cretans around 2000 BCE, demonstrating early appreciation for its carvability and polishability.[29] These applications underscored talc's empirical value in durable, heat-resistant artifacts suitable for daily and ritual use. The scientific identification of talc advanced in the late 18th century amid the development of crystallography, with René-Just Haüy's 1784 observations on crystal geometry laying groundwork for systematic mineral classification, including talc's recognition as a distinct phyllosilicate by the early 19th century.[30] Early European mining focused on high-quality deposits, such as Norway's prehistoric quarries yielding steatite blocks and Italy's Pinerolo region, exploited since medieval times for pure talc in cosmetics and pigments due to its inertness and fineness.[31] Pre-20th century uses extended to sculpture, where talc-rich soapstone allowed intricate detailing in European and Asian artworks, and to rudimentary ceramics, incorporating ground talc for enhanced whiteness and thermal expansion in glazes and bodies, providing practical durability without modern scaling.[32] These applications highlighted talc's utility in contexts demanding resistance to wear and heat, predating industrial refinement.Geological Formation and Occurrence
Geological Formation Processes
Talc forms predominantly through metamorphic alteration of magnesium-rich protoliths, such as ultramafic rocks, dolomites, and serpentinites, under low-grade conditions involving silica introduction via fluids. Regional metamorphism drives this process by subjecting these rocks to temperatures of approximately 200–400°C and pressures up to 2 kbar, facilitating reactions like dolomite + quartz → talc + calcite + CO₂ in the CaO–MgO–SiO₂–CO₂–H₂O system.[33][34] Field observations in metamorphic belts reveal talc in foliated assemblages with tremolite or chlorite, confirming protolith transformation without melting, while phase equilibria modeling supports stability in greenschist facies.[35][36] Hydrothermal processes contribute significantly, particularly through metasomatic exchange where hot, silica-bearing fluids (often meteoric or magmatic brines) interact with magnesium-enriched hosts like serpentinites. In serpentinized peridotites, silica metasomatism replaces antigorite or forsterite with talc at temperatures below 550°C and elevated CO₂ partial pressures, producing replacement textures observed petrographically.[37][35] Magnesium metasomatism occurs less frequently, as in cases of Mg loss from serpentinite enriching adjacent silica sources, but empirical stable isotope data (e.g., Mg fractionation) trace fluid pathways linking alteration to slab dehydration or seafloor processes.[38] Laboratory hydrothermal experiments replicate these reactions, demonstrating talc nucleation via dissolution-reprecipitation under controlled P-T-fluid conditions.[39] Igneous-related talc formation is uncommon, typically limited to contact metamorphism near intrusions or late-stage hydrothermal veins in pegmatites, where magmatic fluids provide silica but do not dominate global deposits.[40] Validation across pathways relies on empirical proxies like mineral zoning, fluid inclusion thermometry (yielding 350–500°C for some acicular varieties), and experimental petrology, underscoring causal fluid-rock ratios and metasomatic gradients over speculative diffusion models.[41][42]Global Occurrence and Major Deposits
Talc deposits are distributed worldwide, occurring primarily in metamorphic terrains within orogenic belts and associated with ultramafic rocks such as serpentinite and dolomite. These include major concentrations in the Appalachian Mountains of the eastern United States, extending from Vermont southward to Alabama, as well as in the Piedmont region.[43] In Europe, significant deposits are found in the Alpine belt, spanning countries like France, Italy, Austria, and Switzerland, where talc lenses form within folded metamorphic sequences.[44] The Ural Mountains in Russia host similar deposits in serpentinite belts, reflecting Paleozoic orogenic activity.[45] In Asia, talc is abundant in ophiolite complexes and metamorphic zones, particularly in China and India. China's Liaoning Province features the Haicheng deposit, one of the largest known talc occurrences, characterized by exceptionally pure, massive talc bodies within altered ultramafics.[46] Indian deposits align with Himalayan ophiolites and associated metamorphic belts, though often intermingled with phyllites. Tectonic settings in these regions, including subduction-related metamorphism, contribute to variations in deposit purity, with vein and massive forms generally yielding higher-grade material compared to schistose varieties embedded in foliated host rocks.[39] Other notable regions encompass Brazil's Minas Gerais state, where deposits occur in Precambrian shields, and Australia's Western Australia, including the Three Springs area with large soapstone-type talc. Globally, talc resources are substantial, with identified reserves estimated to support long-term abundance, as world resources approximate five times the current reserve base according to U.S. Geological Survey assessments.[47] Accessibility is influenced by the structural integrity of host formations, with purer deposits often in less deformed massive lenses versus disseminated schistose occurrences.[48]Mining, Production, and Economics
Mining Methods and Challenges
Talc is predominantly mined using open-pit methods, which are well-suited to its soft, friable deposits typically found near the surface, allowing for efficient extraction without extensive underground operations.[49] Selective mining practices, including careful ore zone delineation and hand sorting, are applied to segregate high-quality talc from associated impurities such as carbonate minerals and asbestos-bearing amphiboles like tremolite, thereby minimizing contamination in the feedstock.[44] Post-extraction, beneficiation begins with primary and secondary crushing to break down the ore, followed by screening to classify particles by size.[50] Flotation processes exploit talc's inherent hydrophobicity to float and separate it from hydrophilic gangue, often enhanced by collectors and frothers for optimal recovery.[51] The concentrate is then dried and subjected to dry grinding or micronization to achieve fine particle sizes, yielding industrial-grade talc with purity levels typically exceeding 95%.[52] Operational challenges include managing respirable dust generated during drilling, crushing, and grinding, which is mitigated through water sprays, enclosed systems, and ventilation to protect workers and reduce airborne emissions.[53] Water consumption arises primarily from flotation circuits and dust suppression, requiring recycling strategies to address scarcity in arid mining regions, though talc's low hardness reduces overall processing energy compared to silicate or metallic ores.[54] Complete avoidance of asbestos remains difficult due to geological intergrowths with tremolite-actinolite series minerals, demanding vigilant deposit characterization and multi-stage purification to meet safety thresholds.[55] Despite these hurdles, talc mining exhibits a relatively low ecological footprint, involving minimal blasting and rapid site rehabilitation potential owing to the absence of acid mine drainage risks.[56]Global Production Statistics and Trade
Global talc production reached approximately 8 million metric tons in 2024, with projections indicating growth to 8.07 million tons in 2025.[57] China dominated output at 1.8 million metric tons in 2023, accounting for roughly 40-50% of the total, followed by India at 1.0 million metric tons and Brazil at 0.85 million metric tons.[58] Other notable producers included the United States, France, and Finland, though their shares were smaller.[59] In the United States, three companies operated five talc mines across three states in 2023, with total sales estimated at 510,000 tons in the subsequent year.[60][47] Key domestic production occurred in Montana and Texas, contributing to an output of around 0.5-1 million tons annually.[47] The global market value stood at approximately USD 2.9 billion in 2024, reflecting demand in sectors such as plastics, paper, and cosmetics.[61] Industry growth has sustained a compound annual growth rate (CAGR) of 3.5-4.35% in recent years, driven primarily by expanding applications in polymer composites and personal care products.[59][57] Trade patterns feature significant exports from Asia—led by China and India—to Europe and North America, with the United States also exporting USD 133 million worth in 2021.[62] Supply chains demonstrated resilience following COVID-19 disruptions, supported by diversified sourcing and stabilized mining operations.[63]| Top Producers (2023, million metric tons) | Output |
|---|---|
| China | 1.8 |
| India | 1.0 |
| Brazil | 0.85 |
| United States | ~0.5 |



![Talc crystal viewed along the [100] axis, looking along the layers of the crystal](http://upload.wikimedia.org/wikipedia/commons/thumb/b/b9/Talc_structure.jpg/120px-Talc_structure.jpg)