Recent from talks
Nothing was collected or created yet.
Anosmia
View on Wikipedia| Anosmia | |
|---|---|
| Other names | Loss of smell, smell blindness,[1] odor blindness |
 | |
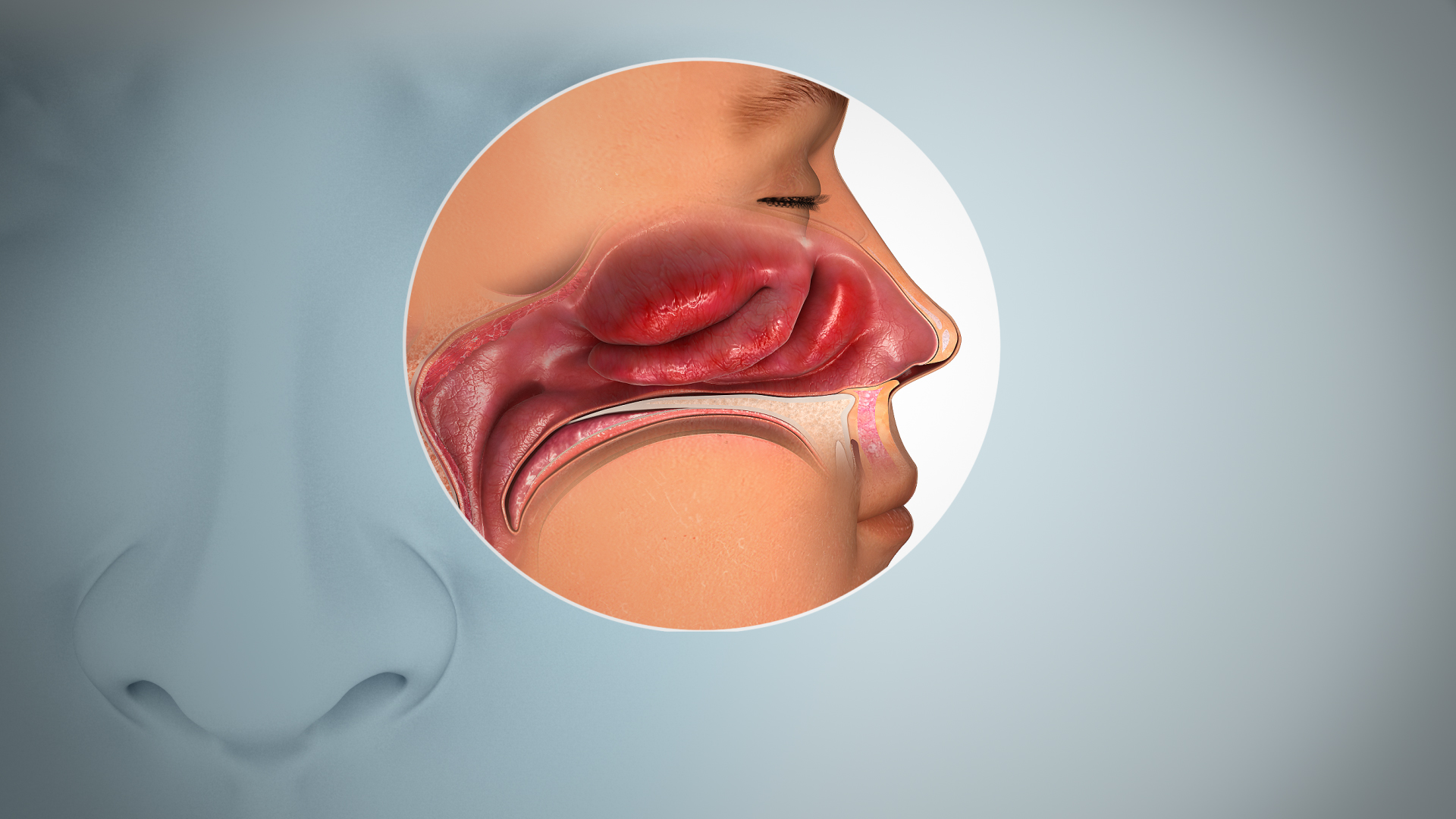
| Inflamed nasal mucosa causing anosmia | |
| Pronunciation | |
| Specialty | Otorhinolaryngology |
| Types | Partial, total[2] |
Anosmia, also known as smell blindness, is the lack of ability to detect one or more smells.[1][2] Anosmia may be temporary or permanent.[3] It differs from hyposmia, which is a decreased sensitivity to some or all smells.[2]
Anosmia can be categorized into acquired anosmia and congenital anosmia. Acquired anosmia develops later in life due to various causes, such as upper respiratory infections, head trauma, or neurodegenerative diseases.[4] In contrast, congenital anosmia is present from birth and is typically caused by genetic factors or developmental abnormalities of the olfactory system.[5] While acquired anosmia may have potential treatments depending on the underlying cause, such as medications or surgery, congenital anosmia currently has no known cure, and management focuses on safety precautions and coping strategies.[6]
Anosmia can be due to a number of factors, including inflammation of the nasal mucosa, blockage of nasal passages, or destruction of temporal lobular tissue.[7] Anosmia stemming from sinus inflammation is due to chronic mucosal changes in the lining of the paranasal sinus and in the middle and superior turbinates.[8][9]
When anosmia is caused by inflammatory changes in the nasal passageways, it is treated simply by reducing inflammation.[10][11] It can be caused by chronic meningitis and neurosyphilis that would increase intracranial pressure over a long period of time,[12] and, in some cases, by ciliopathy,[13] including ciliopathy due to primary ciliary dyskinesia.[14]
The term derives from the Neo-Latin anosmia, based on Ancient Greek ἀν- (an-) + ὀσμή (osmḗ 'smell'; another related term, hyperosmia, refers to an increased ability to smell). Some people may be anosmic for one particular odor, a condition known as "specific anosmia". The absence of the sense of smell from birth is known as congenital anosmia.[15]
In the United States, 3% of people aged over 40 are affected by anosmia.[3]
Anosmia is a common symptom of COVID-19 and can persist as long COVID.[16]
Definition
[edit]Anosmia is the inability to smell.[1] It may be partial or total, and can be specific to certain smells.[2] Reduced sensitivity to some or all smells is hyposmia.[2]
Signs and symptoms
[edit]Anosmia can have a number of harmful effects.[17] People with sudden onset anosmia may find food less appetizing, though congenital anosmics rarely complain about this, and none report a loss in weight. Loss of smell can also be dangerous because it hinders the detection of gas leaks, fire, and spoiled food. Misconceptions of anosmia as trivial can make it more difficult for a patient to receive the same types of medical aid as someone who has lost other senses, such as hearing or sight.[citation needed]
Many experience one sided loss of smell, often as a result of minor head trauma. This type of anosmia is normally only detected if both of the nostrils are tested separately. Using this method of testing each nostril separately will often show a reduced or even completely absent sense of smell in either one nostril or both, something which is often not revealed if both nostrils are simultaneously tested.[18]
Losing an established and sentimental smell memory (e.g. the smell of grass, of the grandparents' attic, of a particular book, of loved ones, or of oneself) has been known to cause feelings of depression.[19][better source needed]
Loss of the ability to smell may lead to the loss of libido, but this usually does not apply to those with olfactory dysfunction at birth.[19][20]
Often people who have loss of smell at birth report that they pretended to be able to smell as children because they thought that smelling was something that older/mature people could do, or did not understand the concept of smelling but did not want to appear different from others. When children get older, they often realize and report to their parents that they do not actually possess a sense of smell, often to the surprise of their parents.[citation needed]
Causes
[edit]A temporary loss of smell can be caused by a blocked nose or infection. In contrast, a permanent loss of smell may be caused by death of olfactory receptor neurons in the nose or by brain injury in which there is damage to the olfactory nerve or damage to brain areas that process smell (see olfactory system). The lack of the sense of smell at birth, usually due to genetic factors, is referred to as congenital anosmia. Family members of the patient with congenital anosmia are often found with similar histories; this suggests that the anosmia may follow an autosomal dominant pattern.[21] Anosmia may very occasionally be an early sign of a degenerative brain disease such as Parkinson's disease and Alzheimer's disease.[22]
Another specific cause of permanent loss could be from damage to olfactory receptor neurons because of use of certain types of nasal spray; i.e., those that cause vasoconstriction of the nasal microcirculation. To avoid such damage and the subsequent risk of loss of smell, vasoconstricting nasal sprays should be used only when absolutely necessary and then for only a short amount of time. Non-vasoconstricting sprays, such as those used to treat allergy-related congestion, are safe to use for prescribed periods of time.[23] Anosmia can also be caused by nasal polyps. These polyps are found in people with allergies, histories of sinusitis, and family history. Individuals with cystic fibrosis often develop nasal polyps.[citation needed]
Amiodarone is a drug used in the treatment of arrhythmias of the heart. A clinical study demonstrated that the use of this drug induced anosmia in some patients. Although rare, there was a case in which a 66-year-old male was treated with amiodarone for ventricular tachycardia. After the use of the drug he began experiencing olfactory disturbance, however after decreasing the dosage of amiodarone, the severity of the anosmia decreased accordingly, suggesting a relationship between use of amiodarone to the development of anosmia.[24]
COVID-19-related anosmia
[edit]Chemosensory disturbances, including loss of smell or taste, are the predominant neurological symptom of COVID-19.[25][26] As many as 80% of COVID-19 patients exhibit some change in chemesthesis, including smell. Loss of smell has also been found to be more predictive of COVID-19 than all other symptoms, including fever, cough, or fatigue, based on a survey of 2 million participants in the UK and US.[27] Google searches for "smell", "loss of smell", "anosmia", and other similar terms increased since the early months of the pandemic, and strongly correlated with increases in daily cases and deaths.[28] Research into the mechanisms underlying these symptoms is currently ongoing.[29][30]
Many countries list anosmia as an official COVID-19 symptom, and some have developed "smell tests" as potential screening tools.[31][32]
In 2020, the Global Consortium for Chemosensory Research, a collaborative research organization of international smell and taste researchers, formed to investigate loss of smell and related chemosensory symptoms.[33]
Decision-making in COVID-19 patients
[edit]Studies have indicated that patients who presented with anosmia during the acute phase of COVID-19 are more likely to develop changes in decision-making, exhibiting more impulsive responses, which are associated with functional and structural brain changes.[34]
Possible causes
[edit]This section needs more reliable medical references for verification or relies too heavily on primary sources. (October 2025) |
- Upper respiratory tract infection (such as sinusitis, the common cold)[35]
- COVID-19[36][37]
- Nasal polyps[38]
- Idiopathic hypogonadotropic hypogonadism
- Hypothyroidism
- Head trauma, damage to the ethmoid bone[39]
- Dementia with Lewy bodies
- Tumors of the frontal lobe
- Antibiotics
- Fibromyalgia
- Multiple sclerosis
- Exposure to hydrogen sulfide (H
2S) by paralysis of the olfactory nerve.[40] - Hypoglycaemia
- Diabetes
- Asthma or allergy
- Hayfever
- Chronic obstructive pulmonary disease (COPD)
- Long term alcoholism
- Cushing's syndrome
- Exposure to a chemical that burns the inside of the nose
- Stroke
- Epilepsy
- Radiation therapy to the head and neck
- Liver or kidney disease
- Parkinson's disease[41]
- Alzheimer's disease[42]
- Toxins (especially acrylates, methacrylates[43] and cadmium[44][45])
- Old age[46]
- Kallmann syndrome
- Primary ciliary dyskinesia
- Post-perfusion syndrome
- Laryngectomy with permanent tracheostomy
- Esthesioneuroblastoma is an exceedingly rare cancerous tumor that originates in or near the olfactory nerve. Symptoms are anosmia (loss of sense of smell) often accompanied by chronic sinusitis.[47]
- Intranasal drug use
- Aspirin-exacerbated respiratory disease, also known as Samter's triad
- Foster Kennedy syndrome
- Cadmium poisoning
- Smoking
- Neurotropic virus[48]
- Schizophrenia[49]
- Pernicious anemia
- Zinc deficiency
- Bell's Palsy or nerve paralysis and damage
- Idiopathic intracranial hypertension
- Suprasellar meningioma
- Refsum's disease
- Adrenergic agonists or withdrawal from alpha blockers (vasoconstriction)
- Sarcoidosis[50]
- Zinc-based intranasal cold products, including remedies labelled as "homeopathic"[51]
- Chronic atrophic rhinitis
- Paget's disease of bone[52]
- Cerebral aneurysm[53]
- Granulomatosis with polyangiitis
- Primary amoebic meningoencephalitis
- Myasthenia gravis[54]
- Snakebite[55]
- Idiopathic anosmia (cause cannot be determined)[3]
Diagnosis
[edit]Diagnosis begins with a detailed history, including possible related injuries, such as upper respiratory infections or head injury. The examination may involve nasal endoscopy for obstructive factors such as polyps or swelling.[7] A nervous system examination is performed to see if the cranial nerves are affected.[7] On occasion, after head traumas, there are people who have unilateral anosmia. The sense of smell should be tested individually in each nostril.[18]
Many cases of congenital anosmia remain unreported and undiagnosed. Since the disorder is present from birth the individual may have little or no understanding of the sense of smell, hence is unaware of the deficit.[56] It may also lead to reduction of appetite.[57]
Treatment
[edit]Though anosmia caused by brain damage cannot be treated, anosmia caused by inflammatory changes in the mucosa may be treated with glucocorticoids. Reduction of inflammation through the use of oral glucocorticoids such as prednisone, followed by long term topical glucocorticoid nasal spray, would easily and safely treat the anosmia. A prednisone regimen is adjusted based on the degree of the thickness of mucosa, the discharge of oedema and the presence or absence of nasal polyps.[10] However, the treatment is not permanent and may have to be repeated after a short while.[10] Together with medication, pressure of the upper area of the nose must be mitigated through aeration and drainage.[58]
Anosmia caused by a nasal polyp may be treated by steroidal treatment or removal of the polyp.[59]
Although very early in development, gene therapy has restored a sense of smell in mice with congenital anosmia when caused by ciliopathy. In this case, a genetic condition had affected cilia in their bodies which normally enabled them to detect air-borne chemicals, and an adenovirus was used to implant a working version of the IFT88 gene into defective cells in the nose, which restored the cilia and allowed a sense of smell.[60][61]
Epidemiology
[edit]In the United States, 3% of people aged over 40 are affected by anosmia.[3]
In 2012, smell was assessed in persons aged 40 years and older with rates of anosmia/severe hyposmia of 0.3% at age 40–49 rising to 14.1% at age 80+. Rates of hyposmia were much higher: 3.7% at age 40–49 and 25.9% at 80+.[62]
Famous people with anosmia
[edit]This section needs expansion. You can help by adding to it. (January 2024) |
- Kathy Clugston, British radio presenter[63][64]
- Ben Cohen of Ben and Jerry's[65][66]
- Perrie Edwards, singer of Little Mix[67]
- Lorenzo de' Medici, 15th-century ruler of Florence[68]
- Bill Pullman, American actor[69]
- Jason Sudeikis, American actor[70]
- Simon Tatham, British computer programmer[71]
- William Wordsworth, poet[72][73]
See also
[edit]- Ageusia, the loss of the sense of taste
- Anosmia Awareness Day
- Congenital anosmia
- Olfactory fatigue, the habituation of the sense of smell to a particular odor or odors
- Parosmia
- Phantosmia
- Zicam, a medicine that caused some users to permanently lose their sense of smell
References
[edit]- ^ a b c Coon D, Mitterer J (2014). "4. Sensation and perception". Introduction to Psychology: Gateways to Mind and Behavior. Boston: Cengage Learning. p. 136. ISBN 978-1-305-09187-0. LCCN 2014942026.
- ^ a b c d e Jones N (2010). "2. Making sense of symptoms". In Jones N (ed.). Practical Rhinology. CRC Press. pp. 24–25. ISBN 978-1-4441-0861-3.
- ^ a b c d Li X, Lui F (6 July 2020). "Anosmia". StatPearls. StatPearls Publishing. PMID 29489163. Retrieved 1 December 2020.
- ^ Li, X.; Lui, F. (2021). "Anosmia - StatPearls - NCBI Bookshelf". NCBI. PMID 29489163. Retrieved 2024-07-06.
- ^ "Congenital Anosmia". Fifth Sense. Retrieved 2024-07-06.
- ^ "Loss of smell (anosmia) Causes". Mayo Clinic. Retrieved 2024-07-06.
- ^ a b c Huynh PP, Ishii LE, Ishii M (July 2020). "What Is Anosmia?". JAMA. 324 (2): 206. doi:10.1001/jama.2020.10966. PMID 32556300. S2CID 219916772.
- ^ Helliwell T (June 2010). "Inflammatory diseases of the nasal cavities and paranasal sinuses". Diagnostic Histopathology. 16 (6): 255–264. doi:10.1016/j.mpdhp.2010.03.008. PMC 7172334. PMID 32336992.
- ^ Lin YT, Yeh TH (2022). "Studies on Clinical Features, Mechanisms, and Management of Olfactory Dysfunction Secondary to Chronic Rhinosinusitis". Frontiers in Allergy. 3 835151. doi:10.3389/falgy.2022.835151. PMC 8974686. PMID 35386650.
- ^ a b c Knight A (August 1988). "Anosmia". Lancet. 2 (8609): 512. doi:10.1016/s0140-6736(88)90160-2. PMID 2900434. S2CID 208793859.(subscription required)
- ^ "Sinusitis Clinical Practice Guideline Quick Reference Tools", Pediatric Clinical Practice Guidelines & Policies, American Academy of Pediatrics, pp. 481–484, 2022-04-11, doi:10.1542/9781610026086-part01-27, ISBN 978-1-61002-608-6
- ^ "Anosmia". The Lancet. 241 (6228): 55. 1943. doi:10.1016/S0140-6736(00)89085-6. S2CID 241387103.
- ^ Uytingco CR, Green WW, Martens JR (2019). "Olfactory Loss and Dysfunction in Ciliopathies: Molecular Mechanisms and Potential Therapies". Current Medicinal Chemistry. 26 (17): 3103–3119. doi:10.2174/0929867325666180105102447. PMC 6034980. PMID 29303074.
- ^ Ul Hassan A, Hassan G, Khan SH, Rasool Z, Abida A (January 2009). "Ciliopathy with special emphasis on kartageners syndrome". International Journal of Health Sciences. 3 (1): 65–69. PMC 3068795. PMID 21475513.
- ^ Boesveldt S, Postma EM, Boak D, Welge-Luessen A, Schöpf V, Mainland JD, et al. (September 2017). "Anosmia-A Clinical Review". Chemical Senses. 42 (7): 513–523. doi:10.1093/chemse/bjx025. PMC 5863566. PMID 28531300.
- ^ "Q&A: COVID-19 and loss of smell, taste". Mayo Clinic Health System. Retrieved 2022-02-23.
- ^ Toller SV (December 1999). "Assessing the impact of anosmia: review of a questionnaire's findings". Chemical Senses. 24 (6): 705–712. doi:10.1093/chemse/24.6.705. PMID 10587505.
- ^ a b Harvey P (February 2006). "Anosmia". Practical Neurology. 6 (1): 65.
- ^ a b Heald C (December 27, 2006). "Sense and scent ability". BBC News. Retrieved April 25, 2010.
- ^ Gudziol V, Wolff-Stephan S, Aschenbrenner K, Joraschky P, Hummel T (July 2009). "Depression resulting from olfactory dysfunction is associated with reduced sexual appetite--a cross-sectional cohort study". The Journal of Sexual Medicine. 6 (7): 1924–1929. doi:10.1111/j.1743-6109.2009.01266.x. PMID 19453919.
- ^ Waguespack RW (1992). "Congenital Anosmia". Archives of Otolaryngology–Head & Neck Surgery. 118 (1): 10. doi:10.1001/archotol.1992.01880010012002.
- ^ Walker, Ian M.; Fullard, Michelle E.; Morley, James F.; Duda, John E. (2021), "Olfaction as an early marker of Parkinson's disease and Alzheimer's disease", Handbook of Clinical Neurology, vol. 182, Elsevier, pp. 317–329, doi:10.1016/b978-0-12-819973-2.00030-7, ISBN 978-0-12-819973-2, PMID 34266602, retrieved 2024-12-26
- ^ Preventing Anosmia from Intranasal Zinc Administration[unreliable medical source?]
- ^ Maruyama T, Yasuda S, Odashiro K, Kaji Y, Harada M (November 2007). "Anosmia induced by amiodarone". The American Journal of Medicine. 120 (11) e9. doi:10.1016/j.amjmed.2006.08.029. PMID 17976411.
- ^ Lechien JR, Chiesa-Estomba CM, De Siati DR, et al. (August 2020). "Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): a multicenter European study". European Archives of Oto-Rhino-Laryngology. 277 (8): 2251–2261. doi:10.1007/s00405-020-05965-1. PMC 7134551. PMID 32253535.
- ^ Xydakis MS, Dehgani-Mobaraki P, Holbrook EH, et al. (September 2020). "Smell and taste dysfunction in patients with COVID-19". The Lancet. Infectious Diseases. 20 (9): 1015–1016. doi:10.1016/S1473-3099(20)30293-0. PMC 7159875. PMID 32304629.
- ^ Menni C, Valdes AM, Freidin MB, et al. (July 2020). "Real-time tracking of self-reported symptoms to predict potential COVID-19". Nature Medicine. 26 (7): 1037–1040. doi:10.1038/s41591-020-0916-2. PMC 7751267. PMID 32393804.
- ^ Walker A, Hopkins C, Surda P (July 2020). "Use of Google Trends to investigate loss-of-smell-related searches during the COVID-19 outbreak". International Forum of Allergy & Rhinology. 10 (7): 839–847. doi:10.1002/alr.22580. PMC 7262261. PMID 32279437.
- ^ Shelton, J. F., Shastri, A. J., Fletez-Brant, K., Aslibekyan, S., & Auton, A. (2022). The UGT2A1/UGT2A2 locus is associated with COVID-19-related loss of smell or taste. Nature Genetics, 54(2), 121-124.
- ^ Cooper KW, Brann DH, Farruggia MC, Bhutani S, Pellegrino R, Tsukahara T, et al. (July 2020). "COVID-19 and the Chemical Senses: Supporting Players Take Center Stage". Neuron. 107 (2): 219–233. doi:10.1016/j.neuron.2020.06.032. PMC 7328585. PMID 32640192.
- ^ Iravani B, Arshamian A, Ravia A, Mishor E, Snitz K, Shushan S, et al. (11 May 2020). "Relationship between odor intensity estimates and COVID-19 population prediction in a Swedish sample". medRxiv 10.1101/2020.05.07.20094516.
- ^ Rodriguez S, Cao L, Rickenbacher GT, Benz EG, Magdamo C, Ramirez Gomez LA, et al. (June 2020). "Innate immune signaling in the olfactory epithelium reduces odorant receptor levels: modeling transient smell loss in COVID-19 patients". medRxiv 10.1101/2020.06.14.20131128.
- ^ "Global Consortium for Chemosensory Research". Global Consortium for Chemosensory Research (GCCR). Retrieved 10 July 2020.
- ^ Kausel, L. (2024). "Patients recovering from COVID-19 who presented with anosmia during their acute episode have behavioral, functional, and structural brain alterations". Scientific Reports. 14 (1): 19049. Bibcode:2024NatSR..1419049K. doi:10.1038/s41598-024-69772-y. PMC 11329703. PMID 39152190.
- ^ Doty RL, Mishra A (March 2001). "Olfaction and its alteration by nasal obstruction, rhinitis, and rhinosinusitis". The Laryngoscope. 111 (3): 409–423. doi:10.1097/00005537-200103000-00008. PMC 7165948. PMID 11224769.
- ^ Grant MC, Geoghegan L, Arbyn M, Mohammed Z, McGuinness L, Clarke EL, Wade RG (23 June 2020). "The prevalence of symptoms in 24,410 adults infected by the novel coronavirus (SARS-CoV-2; COVID-19): A systematic review and meta-analysis of 148 studies from 9 countries". PLOS ONE. 15 (6) e0234765. Bibcode:2020PLoSO..1534765G. doi:10.1371/journal.pone.0234765. PMC 7310678. PMID 32574165.
- ^ "Coronavirus Disease 2019 (COVID-19) – Symptoms". Centers for Disease Control and Prevention. 13 May 2020. Retrieved 14 May 2020.
- ^ Ta NH (January 2019). "Will we ever cure nasal polyps?". Annals of the Royal College of Surgeons of England. 101 (1): 35–39. doi:10.1308/rcsann.2018.0149. PMC 6303820. PMID 30286644.
- ^ Doty RL, Yousem DM, Pham LT, Kreshak AA, Geckle R, Lee WW (September 1997). "Olfactory dysfunction in patients with head trauma". Archives of Neurology. 54 (9): 1131–1140. doi:10.1001/archneur.1997.00550210061014. PMID 9311357.
- ^ National Research Council (US) Committee on Emergency and Continuous Exposure Guidance Levels for Selected Submarine Contaminants (2009). Hydrogen Sulfide. National Academies Press (US).
- ^ Doty RL, Deems DA, Stellar S (August 1988). "Olfactory dysfunction in parkinsonism: a general deficit unrelated to neurologic signs, disease stage, or disease duration". Neurology. 38 (8): 1237–1244. doi:10.1212/WNL.38.8.1237. PMID 3399075. S2CID 3009692.
- ^ Murphy C (April 1999). "Loss of olfactory function in dementing disease". Physiology & Behavior. 66 (2): 177–182. doi:10.1016/S0031-9384(98)00262-5. PMID 10336141. S2CID 26110446.
- ^ Schwartz BS, Doty RL, Monroe C, Frye R, Barker S (May 1989). "Olfactory function in chemical workers exposed to acrylate and methacrylate vapors". American Journal of Public Health. 79 (5): 613–618. doi:10.2105/AJPH.79.5.613. PMC 1349504. PMID 2784947.
- ^ Rose CS, Heywood PG, Costanzo RM (June 1992). "Olfactory impairment after chronic occupational cadmium exposure". Journal of Occupational Medicine. 34 (6): 600–605. PMID 1619490.
- ^ Rydzewski B, Sułkowski W, Miarzyńska M (1998). "Olfactory disorders induced by cadmium exposure: a clinical study". International Journal of Occupational Medicine and Environmental Health. 11 (3): 235–245. PMID 9844306.
- ^ Doty RL, Shaman P, Applebaum SL, Giberson R, Siksorski L, Rosenberg L (December 1984). "Smell identification ability: changes with age". Science. 226 (4681): 1441–1443. Bibcode:1984Sci...226.1441D. doi:10.1126/science.6505700. PMID 6505700.
- ^ Somenek M (October 30, 2009). Harris JE (ed.). "Esthesioneuroblastoma". eMedicine.
- ^ Seo BS, Lee HJ, Mo JH, Lee CH, Rhee CS, Kim JW (October 2009). "Treatment of postviral olfactory loss with glucocorticoids, Ginkgo biloba, and mometasone nasal spray". Archives of Otolaryngology–Head & Neck Surgery. 135 (10): 1000–1004. doi:10.1001/archoto.2009.141. PMID 19841338.
- Lay summary in: "Study Examines Treatment For Olfactory Loss After Viral Infection". ScienceDaily (Press release). October 21, 2009.
- ^ Rupp CI, Fleischhacker WW, Kemmler G, Kremser C, Bilder RM, Mechtcheriakov S, et al. (May 2005). "Olfactory functions and volumetric measures of orbitofrontal and limbic regions in schizophrenia". Schizophrenia Research. 74 (2–3): 149–161. doi:10.1016/j.schres.2004.07.010. PMID 15721995. S2CID 11026266.
- ^ Kieff DA, Boey H, Schaefer PW, Goodman M, Joseph MP (December 1997). "Isolated neurosarcoidosis presenting as anosmia and visual changes". Otolaryngology–Head and Neck Surgery. 117 (6): S183 – S186. doi:10.1016/S0194-5998(97)70097-4. PMID 9419143.
- ^ Harris G (June 16, 2009). "F.D.A. Warns Against Use of Popular Cold Remedy". New York Times.
- ^ Wheeler TT, Alberts MA, Dolan TA, McGorray SP (December 1995). "Dental, visual, auditory and olfactory complications in Paget's disease of bone". Journal of the American Geriatrics Society. 43 (12): 1384–1391. doi:10.1111/j.1532-5415.1995.tb06618.x. PMID 7490390. S2CID 26893932.
- ^ Eriksen KD, Bøge-Rasmussen T, Kruse-Larsen C (June 1990). "Anosmia following operation for cerebral aneurysms in the anterior circulation". Journal of Neurosurgery. 72 (6): 864–865. doi:10.3171/jns.1990.72.6.0864. PMID 2338570.
- ^ Leon-Sarmiento FE, Bayona EA, Bayona-Prieto J, Osman A, Doty RL (2012). "Profound olfactory dysfunction in myasthenia gravis". PLOS ONE. 7 (10) e45544. Bibcode:2012PLoSO...745544L. doi:10.1371/journal.pone.0045544. PMC 3474814. PMID 23082113.
- ^ Churchman A, O'Leary MA, Buckley NA, Page CB, Tankel A, Gavaghan C, et al. (December 2010). "Clinical effects of red-bellied black snake (Pseudechis porphyriacus) envenoming and correlation with venom concentrations: Australian Snakebite Project (ASP-11)". The Medical Journal of Australia. 193 (11–12): 696–700. doi:10.5694/j.1326-5377.2010.tb04108.x. PMID 21143062. S2CID 15915175.
- ^ Vowles RH, Bleach NR, Rowe-Jones JM (August 1997). "Congenital anosmia". International Journal of Pediatric Otorhinolaryngology. 41 (2): 207–214. doi:10.1016/S0165-5876(97)00075-X. PMID 9306177.
- ^ Sumner D (1971). "Appetite and Anosmia". The Lancet. 297 (7706): 970. doi:10.1016/S0140-6736(71)91470-X.
- ^ Turnley WH (April 1963). "Anosmia". The Laryngoscope. 73 (4): 468–473. doi:10.1288/00005537-196304000-00012. PMID 13994924. S2CID 221921289.
- ^ McClay JE (May 1, 2014). "Nasal Polyps Treatment & Management". Medscape.
- ^ McIntyre JC, Davis EE, Joiner A, Williams CL, Tsai IC, Jenkins PM, et al. (September 2012). "Gene therapy rescues cilia defects and restores olfactory function in a mammalian ciliopathy model". Nature Medicine. 18 (9): 1423–1428. doi:10.1038/nm.2860. PMC 3645984. PMID 22941275.
- ^ Gallagher J (September 3, 2012). "Gene therapy restores sense of smell in mice". BBC News.
- ^ Hoffman HJ, Rawal S, Li CM, Duffy VB (June 2016). "New chemosensory component in the U.S. National Health and Nutrition Examination Survey (NHANES): first-year results for measured olfactory dysfunction". Reviews in Endocrine & Metabolic Disorders. 17 (2): 221–240. doi:10.1007/s11154-016-9364-1. PMC 5033684. PMID 27287364.
- ^ Parker, Olivia (11 April 2016). "'My partner does "armpit duty"': What it's like to live with no sense of smell". The Telegraph. Retrieved 8 December 2018.
- ^ "The Neglected Sense". BBC Radio 4. Retrieved 8 December 2018.
- ^ DeGregory, Lane (30 November 2023). "For decades, a Florida woman had no sense of smell. Can she get it back?". Tampa Bay Times. Retrieved 2023-12-05.
- ^ Sullivan, Justin (8 Apr 2021). "Ben & Jerry's' Ben Cohen Interview". Delish. Retrieved 6 Jan 2024.
- ^ Izundu, Chi Chi (7 October 2013). "Little Mix's Perrie Edwards: I have no sense of smell - BBC Newsbeat". BBC News. Archived from the original on 8 November 2020. Retrieved 11 May 2020.
- ^ Ross, Janet (1910). Lives of the early Medici as told in their correspondence. Chatto & Windus. p. 144.
- ^ Blair, Iain (6 July 2011). "A Minute With: Bill Pullman about "Torchwood"". Reuters. Retrieved 30 July 2024.
- ^ "Why Does Olivia Wilde's Fiancé Jason Sudeikis Think Her Cooking Always Tastes Good?". People. February 27, 2015. Archived from the original on January 25, 2016. Retrieved April 25, 2016.
My fiancé [Jason Sudeikis] has no sense of smell — he was born without it....
- ^ "The 'Simon Tatham Has No Sense Of Smell' FAQ". www.chiark.greenend.org.uk. Retrieved 2025-08-02.
- ^ Boyson, Rowan Rose (2011). "Wordsworth's Anosmia". La questione Romantica. 3 (2): 63–80.
- ^ Healey, Nicola (24 June 2025). "Losing 'a sense sublime': anosmia and poetry". Wild Court. Retrieved 5 August 2025.
Further reading
[edit]- Blodgett B (2010). Remembering Smell: A Memoir of Losing—and Discovering—the Primal Sense. Houghton Mifflin Harcourt. ISBN 978-0-618-86188-0.
- Birnbaum M (2011). Season to Taste: How I Lost my Sense of Smell and Found my Way. Ecco. ISBN 978-0-06-191531-4.
- DeVere R, Calvert M (2010). Navigating Smell and Taste Disorders. Demos Health. ISBN 978-1-932603-96-5.
- Gilbert A (2008). What the Nose Knows.The Science of Scent in Everyday Life. Crown. ISBN 978-1-4000-8234-6.
- Herz R (2007). The Scent of Desire. Discovering Our Enigmatic Sense of Smell. HarperCollins. ISBN 978-0-06-082538-6.
- Tafalla M (2010). Nunca sabrás a qué huele Bagdad (You will never know the smell of Bagdad) (in Spanish). Autonomous University of Barcelona. ISBN 978-84-490-2611-9. - Novel dealing with congenital anosmia.
- Keller A, Malaspina D (July 2013). "Hidden consequences of olfactory dysfunction: a patient report series". BMC Ear, Nose and Throat Disorders. 13 (1): 8. doi:10.1186/1472-6815-13-8. PMC 3733708. PMID 23875929.
- Tafalla M (September 2013). "A world without the olfactory dimension". Anatomical Record. 296 (9): 1287–1296. doi:10.1002/ar.22734. PMID 23907763.
- Tafalla M (2013). "Anosmic Aesthetics". Estetika: The Central European Journal of Aesthetics. 50 (1/2013). Prag: 53–80. doi:10.33134/eeja.103.
- Wilson DA, Stevenson RJ (2006). Learning to Smell. Olfactory Perception from Neurobiology to Behavior. The Johns Hopkins University Press. ISBN 978-0-8018-8368-2.