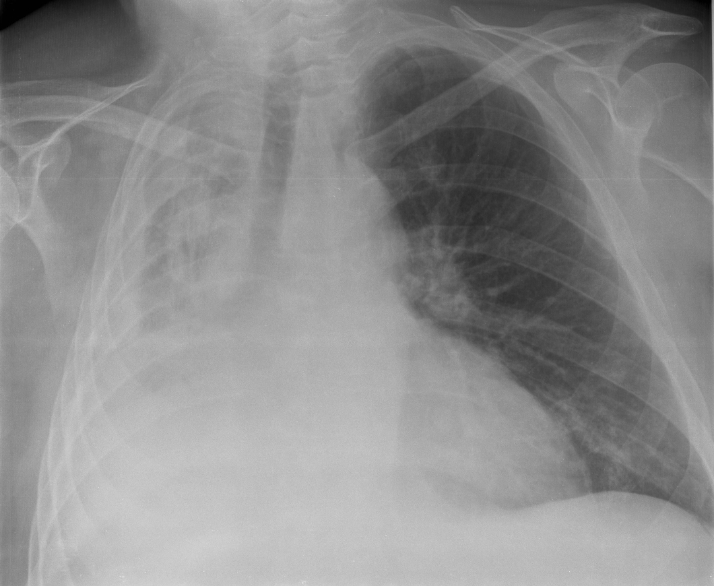
Atelectasis
View on WikipediaThis article needs additional citations for verification. (August 2021) |
| Atelectasis | |
|---|---|
| Other names | Collapsed lung[1] |
 | |
| Atelectasis of a person's right lung | |
| Pronunciation | |
| Specialty | Pulmonology |
Atelectasis is the partial collapse or closure of a lung resulting in reduced or absence in gas exchange. It is usually unilateral, affecting part or all of one lung.[2] It is a condition where the alveoli are deflated down to little or no volume, as distinct from pulmonary consolidation, in which they are filled with liquid. It is often referred to informally as a collapsed lung, although more accurately it usually involves only a partial collapse, and that ambiguous term is also informally used for a fully collapsed lung caused by a pneumothorax.[1]
It is a very common finding in chest X-rays and other radiological studies, and may be caused by normal exhalation or by various medical conditions. Although frequently described as a collapse of lung tissue, atelectasis is not synonymous with a pneumothorax, which is a more specific condition that can cause atelectasis. Acute atelectasis may occur as a post-operative complication or as a result of surfactant deficiency. In premature babies, this leads to infant respiratory distress syndrome.
The term uses combining forms of atel- + ectasis, from Greek: ἀτελής, "incomplete" + Greek: ἔκτασις, "extension".[3]
Signs and symptoms
[edit]
May have no signs and symptoms or they may include:[4]
- cough, but not prominent;
- chest pain (not common);
- breathing difficulty (fast and shallow);
- low oxygen saturation;
- pleural effusion (transudate type);
- cyanosis (late sign);
- increased heart rate.
It is a common misconception and pure speculation that atelectasis cause postoperative fever.[5] This claim has been perpetuated in medical textbooks as recently as 2021.[6] Review articles published in 2011 and 2019 summarizing the available evidence on the association between atelectasis and post-operative fever concluded that there is no clinical evidence supporting this speculation.[7][8] A recent article outlined the history of this myth and the true causes of postoperative fever.[9]
Causes
[edit]The most common cause is post-surgical atelectasis, characterized by splinting, i.e. restricted breathing after abdominal surgery. Atelectasis develops in 75–90% of people undergoing general anesthesia for a surgical procedure.[10]
Another common cause is pulmonary tuberculosis. Smokers and the elderly are also at an increased risk. Outside of this context, atelectasis implies some blockage of a bronchiole or bronchus, which can be within the airway (foreign body, mucus plug), from the wall (tumor, usually squamous cell carcinoma) or compressing from the outside (tumor, lymph node, tubercle). Another cause is poor surfactant spreading during inspiration, causing the surface tension to be at its highest which tends to collapse smaller alveoli. Atelectasis may also occur during suction, as along with sputum, air is withdrawn from the lungs. There are several types of atelectasis according to their underlying mechanisms or the distribution of alveolar collapse; resorption, compression, microatelectasis and contraction atelectasis. Relaxation atelectasis (also called passive atelectasis) is when a pleural effusion or a pneumothorax disrupts the contact between the parietal and visceral pleurae.[11]
Risk factors associated with increased likelihood of the development of atelectasis include: the type of surgery (thoracic and cardiopulmonary surgeries), the use of muscle relaxants, obesity, high oxygen, the lower lung segments.
Factors also associated with the development of atelectasis include: age, presence of chronic obstructive pulmonary disease or asthma, and type of anesthetic.
In the early 1950s, in UK aviation medicine, the condition acceleration atelectasis was given the name "Hunter lung" due to its prevalence in pilots of the transonic fighter jet, the Hawker Hunter, which used a 100% oxygen supply.[12][13]
Diagnosis
[edit]
Clinically significant atelectasis is generally visible on chest X-ray; findings can include lung opacification and/or loss of lung volume. Post-surgical atelectasis will be bibasal in pattern. Chest CT or bronchoscopy may be necessary if the cause of atelectasis is not clinically apparent. Direct signs of atelectasis include displacement of interlobar fissures and mobile structures within the thorax, overinflation of the unaffected ipsilateral lobe or contralateral lung, and opacification of the collapsed lobe. In addition to clinically significant findings on chest X-rays, patients may present with indirect signs and symptoms such as elevation of the diaphragm, shifting of the trachea, heart and mediastinum; displacement of the hilus and shifting granulomas.[14]
Classification
[edit]
Atelectasis is broadly categorized into obstructive (resorptive) and non-obstructive types. It may be further classified as an acute or chronic condition. In acute atelectasis, the lung has recently collapsed and is primarily notable only for airlessness. In chronic atelectasis, the affected area is often characterized by a complex mixture of airlessness, infection, widening of the bronchi (bronchiectasis), destruction, and scarring (fibrosis).
Obstructive (absorptive/resorptive) atelectasis
[edit]This type is defined by blockage of the airway with air trapping and subsequent absorption of air distal to the obstruction.[15] The resulting absorption of air distal to the obstruction results in collapse of the alveoli. It is most commonly due to intrathoracic tumors, aspiration of a foreign body, or mucus plugs.[16] Children are notably more susceptible to atelectasis due to poorly developed collateral airways, which protect against alveolar collapse by maintaining inflation.[15] The Earth's atmosphere is mainly composed of 78% nitrogen and 21% oxygen (+ 1% argon and traces of other gases). Since oxygen is exchanged at the alveoli-capillary membrane, nitrogen is a major component for the alveoli's state of inflation. If a large volume of nitrogen in the lungs is replaced with oxygen, the oxygen may subsequently be absorbed into the blood, reducing the volume of the alveoli, resulting in a form of alveolar collapse known as absorption atelectasis.[17]
Adhesive atelectasis
[edit]This type of atelectasis is due to lack or dysfunction of surfactant, which normally functions to reduce alveolar surface tension. The increased surface tension in the alveoli then results in alveolar instability and collapse.[18] It is most commonly seen in infant respiratory distress syndrome and acute respiratory distress syndrome (ARDS).[19]
Compressive atelectasis
[edit]This type occurs when the extra-alveolar pressure overcomes the intra-alveolar pressure, which results in collapse of the lung tissue.[20] While the cause may vary, it is classically associated with the accumulation of blood, fluid, or air within the pleural cavity. These accumulations result in an increase in extra-alveolar pressure which leads to a pressure gradient favoring collapse of the alveoli. This is a frequent occurrence with pleural effusions secondary to congestive heart failure (CHF). Leakage of air into the pleural cavity (pneumothorax) may also lead to compressive atelectasis.[21]
Relaxation atelectasis
This type of atelectasis occurs when there is loss of contact of the lung to the chest wall. It classically occurs as a result of a pleural effusion or pneumothorax. While relaxation and compressive atelectasis share a lot in common, compressive atelectasis tends to be more focal or localized.[18]
Replacement atelectasis
This type of atelectasis occurs when alveoli of an entire lobe of the lung are filled by a tumor, typically bronchioloalveolar carcinoma.[22] The filling of the alveoli results in a loss of lung volume.[20]
Cicatrization (contraction) atelectasis
[edit]This type occurs when there is contraction of the lung tissue due to the presence of scar tissue. The local or generalized fibrotic changes in the lung or pleura decrease expansion of the lung and increase elastic recoil during expiration.[21][20] Causes include granulomatous disease (e.g., sarcoidosis), necrotizing pneumonia and radiation pneumonitis.[23]
Special cases
[edit]Right middle lobe syndrome
[edit]In right middle lobe syndrome, the middle lobe of the right lung contracts due to pressure on the bronchus. This many be secondary to an enlarged lymph node or occasionally a tumor.[24] The blocked, contracted lung may develop pneumonia that fails to resolve completely and leads to chronic inflammation, scarring, and bronchiectasis.[20] Right middle lobe syndrome may occasionally occur in the absence of obvious obstruction. It is hypothesized that the etiology of non-obstructive right middle lobe syndrome is transient hypoventilation secondary to chronic or acute inflammation.[24]
Rounded atelectasis
[edit]Rounded atelectasis (folded lung or Blesovsky syndrome[25]) is often mistaken for lung cancer on imaging. The most common current theory for rounded atelectasis is that local pleural irritation leads to thickening and shrinkage of the pleura which causes the adjacent lung to shrink with it.[26] The outer portion of the lung slowly collapses as a result of scarring and shrinkage of the membrane layers covering the lungs (pleura), which would show as visceral pleural thickening and entrapment of lung tissue. This produces a rounded appearance on X-ray that doctors may mistake for a tumor. Rounded atelectasis is usually a complication of asbestos-induced disease of the pleura, but it may also result from other types of chronic scarring and thickening of the pleura.[26]
Treatment
[edit]Treatment is directed at correcting the underlying cause. In atelectasis manifestations that result from the mucus plugging of the airways as seen in patients with cystic fibrosis and pneumonia, mucolytic agents such as acetylcysteine (NAC) is used. This nebulized treatment works by reducing mucous viscosity and elasticity by breaking disulfide bonds in mucoproteins within the mucus complex, thus facilitating mucus clearance.[22] Post-surgical atelectasis is treated by physiotherapy, focusing on deep breathing and encouraging coughing. An incentive spirometer is often used as part of the breathing exercises. Walking is also highly encouraged to improve lung inflation. People with chest deformities or neurologic conditions that cause shallow breathing for long periods may benefit from mechanical devices that assist their breathing.[15]
The primary treatment for acute massive atelectasis is correction of the underlying cause. A blockage that cannot be removed by coughing or by suctioning the airways often can be removed by bronchoscopy. Antibiotics are given for an infection. Chronic atelectasis is often treated with antibiotics because infection is almost inevitable. In certain cases, the affected part of the lung may be surgically removed when recurring or chronic infections become disabling or bleeding is significant. If a tumor is blocking the airway, relieving the obstruction by surgery, radiation therapy, chemotherapy, or laser therapy may prevent atelectasis from progressing and recurrent obstructive pneumonia from developing.[20]
See also
[edit]- Alveolar capillary dysplasia, a very rare type of diffuse congenital disorder of the lung
- Flat-chested kitten syndrome or FCKS: atelectasis in neo-natal kittens
- Tympanic membrane atelectasis: Retraction of the ear drum into the middle ear can also be referred to as atelectasis.
- William Pasteur, pioneer pulmonologist
References
[edit]- ^ a b Orenstein, David M. (2004). Cystic Fibrosis: A Guide for Patient and Family. Lippincott Williams & Wilkins. p. 62. ISBN 9780781741521.
- ^ Wedding, Mary Ellen; Gylys, Barbara A. (2005). Medical Terminology Systems: A Body Systems Approach: A Body Systems Approach. Philadelphia, Pa: F. A. Davis Company. ISBN 0-8036-1289-3.[page needed]
- ^ Haubrich, William S. (2003). Medical Meanings: A Glossary of Word Origins (2nd ed.). Philadelphia: ACP Press. p. 25. ISBN 978-1-930513-49-5.
- ^ "Atelectasis". MayoClinic. Retrieved 20 February 2017.
- ^ Mavros, Michael N.; Velmahos, George C.; Falagas, Matthew E. (2011-08-01). "Atelectasis as a Cause of Postoperative Fever: Where Is the Clinical Evidence?". Chest. 140 (2): 418–424. doi:10.1378/chest.11-0127. ISSN 0012-3692. PMID 21527508.
- ^ Townsend, Courtney M., ed. (2021). Sabiston Textbook of Surgery: The Biological Basis of Modern Surgical Practice. St. Louis: Elsevier. p. 247. ISBN 978-0-323-64063-3.
Atelectasis is the most common cause of postoperative fever in the early postoperative period.
- ^ Mavros, Michael N.; Velmahos, George C.; Falagas, Matthew E. (August 2011). "Atelectasis as a Cause of Postoperative Fever". Chest. 140 (2): 418–424. doi:10.1378/chest.11-0127. PMID 21527508.
- ^ Crompton, Joseph G.; Crompton, Peter D.; Matzinger, Polly (2019-03-06). "Does Atelectasis Cause Fever After Surgery? Putting a Damper on Dogma". JAMA Surgery. 154 (5). American Medical Association (AMA): 375–376. doi:10.1001/jamasurg.2018.5645. ISSN 2168-6254. PMID 30840058.
- ^ Stein, Hadassah; Denning, John; Ahmed, Huma; Bruno, Michael A.; Gosselin, Marc; Scott, Jinel; Waite, Stephen (2025-01-01). "Debunking a mythology: Atelectasis is not a cause of postoperative fever". Clinical Imaging. 117 110358. doi:10.1016/j.clinimag.2024.110358. ISSN 0899-7071. PMID 39566396.
- ^ Lumb, Andrew B (2017). Nunn's Applied Respiratory Physiology (8th ed.). Elsevier. p. 289. ISBN 9780702062940.
- ^ Tarun Madappa (November 28, 2017). "Atelectasis". Medscape. Retrieved 2018-02-02.
- ^ Air Vice-Marshal John Ernsting (2008). "The RAF institute of aviation medicine 1945-1994 contributions to aviation and flight safety" (PDF). Royal Air Force Historical Society Journal (43): 18–53. ISSN 1361-4231.
- ^ Lt Col Rob "Mongo" Monberg. "Review of acceleration atelectasis: An old problem in new settings" (PDF). IAMFSP. Archived from the original on April 20, 2018.
- ^ Woodring, John H., and James C. Reed. "Types and mechanisms of pulmonary atelectasis." Journal of thoracic imaging 11.2 (1996): 92-108.
- ^ a b c Grott, Kelly; Chauhan, Shaylika; Sanghavi, Devang K.; Dunlap, Julie D. (2025), "Atelectasis", StatPearls, Treasure Island (FL): StatPearls Publishing, PMID 31424900, retrieved 2025-01-08
- ^ Ray, Komal; Bodenham, Andrew; Paramasivam, Elankumaran (2014-10-01). "Pulmonary atelectasis in anaesthesia and critical care". Continuing Education in Anaesthesia Critical Care & Pain. 14 (5): 236–245. doi:10.1093/bjaceaccp/mkt064. ISSN 1743-1816.
- ^ White, Gary C. (2002). Basic Clinical Lab Competencies for Respiratory Care, 4th ed. Delmar Cengage Learning. p. 230. ISBN 978-0-7668-2532-1.
- ^ a b Stark, Paul (March 3, 2025). Muller, Nestor L; Finlay, Geraldine (eds.). "Atelectasis: Types and pathogenesis in adults". uptodate.com. Retrieved March 23, 2025.
- ^ Kaplan medical pathology lecture notes book (2019). p.118
- ^ a b c d e Grott, Kelly; Chauhan, Shaylika; Sanghavi, Devang K.; Dunlap, Julie D.; Lee, Shelley (2025), "Atelectasis (Nursing)", StatPearls, Treasure Island (FL): StatPearls Publishing, PMID 34283499, retrieved 2025-03-23
- ^ a b Robbins (2013). Basic Pathology. Elsevier. p. 460. ISBN 978-1-4377-1781-5.
- ^ a b Grott, K.; Chauhan, S.; Dunlap, J. D. (2021). "Atelectasis". StatPearls. StatPearls. PMID 31424900.
- ^ Sheikh, Zishan; Weerakkody, Yuranga. "Lung atelectasis". Radiopaedia. Retrieved 20 February 2017.
- ^ a b Shaikh, Umair; Heller, Daniel (2025), "Right Middle Lobe Syndrome", StatPearls, Treasure Island (FL): StatPearls Publishing, PMID 31613437, retrieved 2025-03-24
- ^ Payne, C. R; Jaques, P; Kerr, I. H (1980). "Lung folding simulating peripheral pulmonary neoplasm (Blesovsky's syndrome)". Thorax. 35 (12): 936–940. doi:10.1136/thx.35.12.936. PMC 471419. PMID 7268670.
- ^ a b Serafin, Zbigniew (2014). "Rounded atelectasis of the lung: A pictorial review". Polish Journal of Radiology. 79: 203–209. doi:10.12659/PJR.889983. ISSN 0137-7183. PMC 4102604. PMID 25045412.
External links
[edit]Atelectasis
View on GrokipediaOverview and Pathophysiology
Definition
Atelectasis is defined as the partial or complete collapse of lung tissue, leading to reduced or absent gas exchange in the affected alveoli. This condition occurs when the alveoli, the tiny air sacs at the end of the respiratory tree responsible for oxygen and carbon dioxide exchange, deflate and fail to participate in ventilation.[1][3] The term "atelectasis" originates from Greek roots: "atelēs," meaning incomplete or imperfect, and "ektasis," meaning expansion or stretching, thereby describing the incomplete expansion of lung tissue.[1] It was first introduced into medical literature in the early 19th century by the German physician Eduard Jörg in 1832.[8] Maintaining alveolar patency relies on pulmonary surfactant, a lipoprotein complex secreted by type II alveolar cells that lowers surface tension at the air-liquid interface, preventing collapse during exhalation.[9] Atelectasis is distinct from pneumothorax, which involves air accumulation in the pleural space exerting external pressure on the lung, and from consolidation, such as in pneumonia, where alveoli fill with fluid, blood, or pus while preserving overall lung volume.[2][10]Mechanisms of Lung Collapse
Atelectasis involves several primary mechanisms that lead to lung collapse, categorized as resorptive, compressive, and adhesive processes. Resorptive atelectasis occurs when airway obstruction prevents fresh air from entering the alveoli, resulting in the gradual absorption of trapped alveolar gas into the bloodstream; oxygen is absorbed more rapidly than nitrogen, creating a subatmospheric pressure that causes alveolar collapse.[1] Compressive atelectasis arises from extrinsic forces that reduce lung volume, such as pleural effusions, pneumothorax, or abdominal distension, which mechanically displace lung tissue and promote alveolar emptying without direct airway blockage.[1] Adhesive atelectasis, in contrast, stems from alveolar instability due to insufficient pulmonary surfactant, leading to increased surface tension and spontaneous collapse of alveoli, particularly in conditions impairing surfactant production or function.[11] Biophysically, alveolar stability is governed by Laplace's law, which describes the transmural pressure required to maintain an alveolus open:where is the pressure difference across the alveolar wall, is surface tension, and is the alveolar radius. Smaller alveoli (lower ) experience higher collapsing pressures unless is reduced; pulmonary surfactant lowers dynamically during expiration, preventing smaller alveoli from emptying into larger ones and averting widespread collapse.[12] In adhesive atelectasis, surfactant deficiency elevates , destabilizing alveoli according to this principle and facilitating collapse even without obstruction or compression.[11] The resulting non-ventilation impairs gas exchange through intrapulmonary shunting, where deoxygenated blood perfuses collapsed regions without participating in oxygenation, leading to systemic hypoxemia that is often refractory to supplemental oxygen.[13] Development of atelectasis progresses in stages: initially, gas resorption or compression causes partial alveolar deflation and volume loss; this advances to complete lobar or segmental collapse over hours to days as hypoxic pulmonary vasoconstriction partially redirects blood flow but fails to fully compensate; re-expansion remains possible if the underlying mechanism is promptly reversed, though prolonged collapse risks fibrosis and permanent impairment.[14]