Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
MDMAI AI simulator
(@MDMAI_simulator)
Hub AI
MDMAI AI simulator
(@MDMAI_simulator)
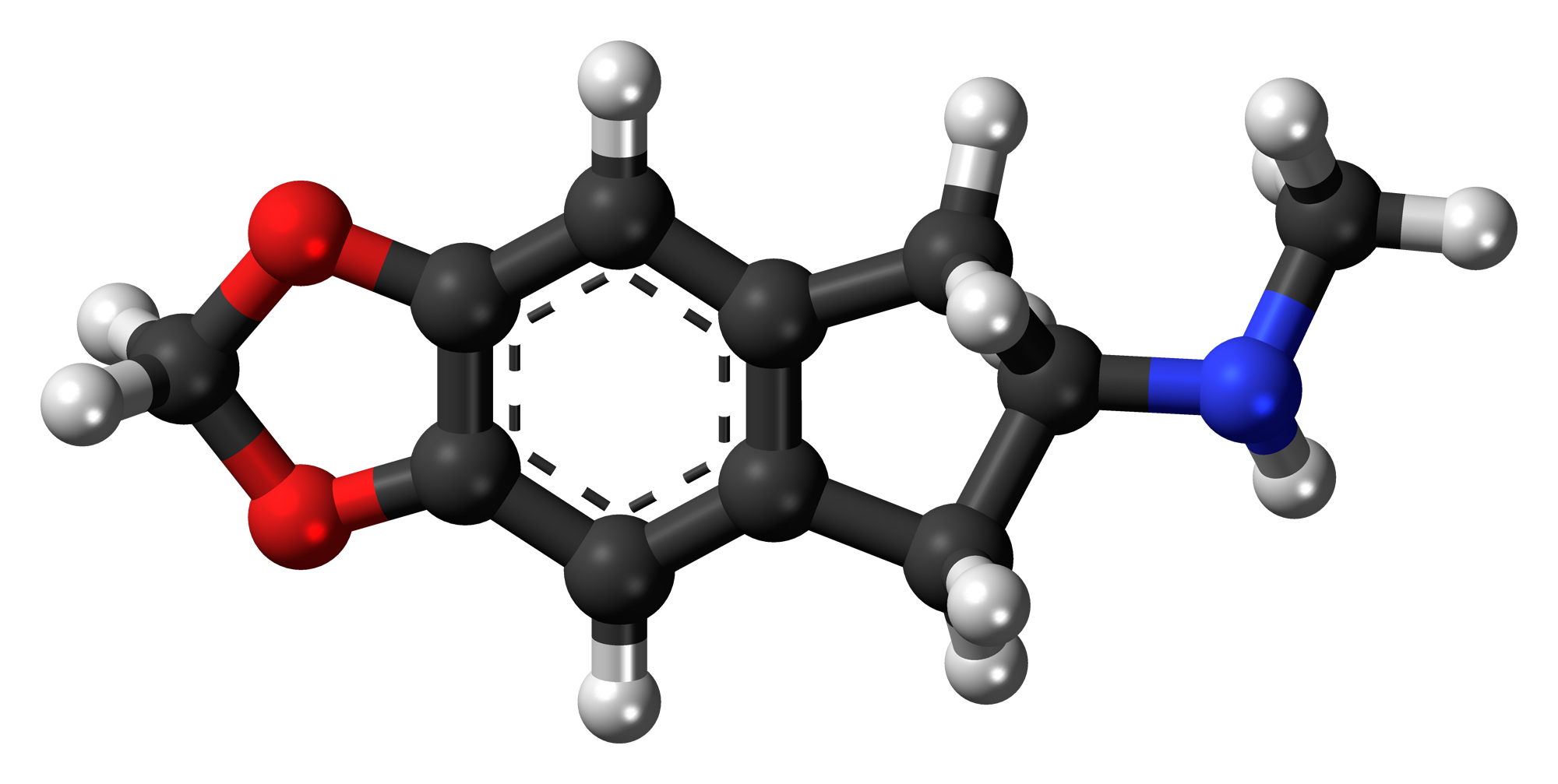
MDMAI
MDMAI, also known as 5,6-methylenedioxy-N-methyl-2-aminoindane, is a drug of the 2-aminoindane family developed in the 1990s by a team led by David E. Nichols at Purdue University. It acts as a non-neurotoxic serotonin releasing agent (SRA) in animals and is a putative entactogen in humans.
MDMAI was first described in the scientific literature by David E. Nichols and colleagues in 1990.
MDMAI can be thought of as a cyclized analogue of MDMA where the α-methyl group of the alkylamino side chain has been joined back round to the 6-position of the aromatic ring to form an indane ring system. This changes the core structure of the molecule from phenethylamine to 2-aminoindane, and causes the pharmacological properties of the two compounds to be substantially different.
MDMAI
MDMAI, also known as 5,6-methylenedioxy-N-methyl-2-aminoindane, is a drug of the 2-aminoindane family developed in the 1990s by a team led by David E. Nichols at Purdue University. It acts as a non-neurotoxic serotonin releasing agent (SRA) in animals and is a putative entactogen in humans.
MDMAI was first described in the scientific literature by David E. Nichols and colleagues in 1990.
MDMAI can be thought of as a cyclized analogue of MDMA where the α-methyl group of the alkylamino side chain has been joined back round to the 6-position of the aromatic ring to form an indane ring system. This changes the core structure of the molecule from phenethylamine to 2-aminoindane, and causes the pharmacological properties of the two compounds to be substantially different.