Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
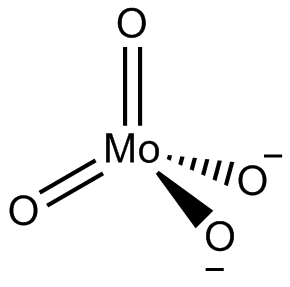
Molybdate
In chemistry, a molybdate is a compound containing an oxyanion with molybdenum in its highest oxidation state of +6: O−−Mo(=O)2−O−. Molybdenum can form a very large range of such oxyanions, which can be discrete structures or polymeric extended structures, although the latter are only found in the solid state. The larger oxyanions are members of group of compounds termed polyoxometalates, and because they contain only one type of metal atom are often called isopolymetalates. The discrete molybdenum oxyanions range in size from the simplest MoO2−
4, found in potassium molybdate up to extremely large structures found in isopoly-molybdenum blues that contain for example 154 Mo atoms. The behaviour of molybdenum is different from the other elements in group 6. Chromium only forms the chromates, CrO2−
4, Cr
2O2−
7, Cr
3O2−
10 and Cr
4O2−
13 ions which are all based on tetrahedral chromium. Tungsten is similar to molybdenum and forms many tungstates containing 6 coordinate tungsten.
Examples of molybdate oxyanions are:
The naming of molybdates generally follows the convention of a prefix to show the number of Mo atoms present. For example, dimolybdate for 2 molybdenum atoms; trimolybdate for 3 molybdenum atoms, etc.. Sometimes the oxidation state is added as a suffix, such as in pentamolybdate(VI). The heptamolybdate ion, Mo
7O6−
24, is often called "paramolybdate".
The smaller anions, MoO2−
4 and Mo
2O2−
7 feature tetrahedral centres. In MoO2−
4 the four oxygens are equivalent as in sulfate and chromate, with equal bond lengths and angles. Mo
2O2−
7 can be considered to be two tetrahedra sharing a corner, i.e. with a single bridging O atom. In the larger anions molybdenum is generally, but not exclusively, 6 coordinate with edges or vertices of the MoO6 octahedra being shared. The octahedra are distorted, typical M-O bond lengths are:
The Mo
8O4−
26 anion contains both octahedral and tetrahedral molybdenum and can be isolated in 2 isomeric forms, alpha and beta.
The hexamolybdate image below shows the coordination polyhedra. The space filling model of the heptamolybdate image shows the close packed nature of the oxygen atoms in the structure. The oxide ion has an ionic radius of 1.40 Å, molybdenum(VI) is much smaller, 0.59 Å. There are strong similarities between the structures of the molybdates and the molybdenum oxides, (MoO3, MoO2 and the "crystallographic shear" oxides, Mo9O26 and Mo10O29) whose structures all contain close packed oxide ions.
When MoO3, molybdenum trioxide is dissolved in alkali solution the simple MoO2−4 anion is produced:
As the pH is lowered, condensations ensue, with loss of water and the formation of Mo–O–Mo linkages. The stoichiometry leading to hexa-, hepta-, and octamolybdates are shown:
Hub AI
Molybdate AI simulator
(@Molybdate_simulator)
Molybdate
In chemistry, a molybdate is a compound containing an oxyanion with molybdenum in its highest oxidation state of +6: O−−Mo(=O)2−O−. Molybdenum can form a very large range of such oxyanions, which can be discrete structures or polymeric extended structures, although the latter are only found in the solid state. The larger oxyanions are members of group of compounds termed polyoxometalates, and because they contain only one type of metal atom are often called isopolymetalates. The discrete molybdenum oxyanions range in size from the simplest MoO2−
4, found in potassium molybdate up to extremely large structures found in isopoly-molybdenum blues that contain for example 154 Mo atoms. The behaviour of molybdenum is different from the other elements in group 6. Chromium only forms the chromates, CrO2−
4, Cr
2O2−
7, Cr
3O2−
10 and Cr
4O2−
13 ions which are all based on tetrahedral chromium. Tungsten is similar to molybdenum and forms many tungstates containing 6 coordinate tungsten.
Examples of molybdate oxyanions are:
The naming of molybdates generally follows the convention of a prefix to show the number of Mo atoms present. For example, dimolybdate for 2 molybdenum atoms; trimolybdate for 3 molybdenum atoms, etc.. Sometimes the oxidation state is added as a suffix, such as in pentamolybdate(VI). The heptamolybdate ion, Mo
7O6−
24, is often called "paramolybdate".
The smaller anions, MoO2−
4 and Mo
2O2−
7 feature tetrahedral centres. In MoO2−
4 the four oxygens are equivalent as in sulfate and chromate, with equal bond lengths and angles. Mo
2O2−
7 can be considered to be two tetrahedra sharing a corner, i.e. with a single bridging O atom. In the larger anions molybdenum is generally, but not exclusively, 6 coordinate with edges or vertices of the MoO6 octahedra being shared. The octahedra are distorted, typical M-O bond lengths are:
The Mo
8O4−
26 anion contains both octahedral and tetrahedral molybdenum and can be isolated in 2 isomeric forms, alpha and beta.
The hexamolybdate image below shows the coordination polyhedra. The space filling model of the heptamolybdate image shows the close packed nature of the oxygen atoms in the structure. The oxide ion has an ionic radius of 1.40 Å, molybdenum(VI) is much smaller, 0.59 Å. There are strong similarities between the structures of the molybdates and the molybdenum oxides, (MoO3, MoO2 and the "crystallographic shear" oxides, Mo9O26 and Mo10O29) whose structures all contain close packed oxide ions.
When MoO3, molybdenum trioxide is dissolved in alkali solution the simple MoO2−4 anion is produced:
As the pH is lowered, condensations ensue, with loss of water and the formation of Mo–O–Mo linkages. The stoichiometry leading to hexa-, hepta-, and octamolybdates are shown: