Recent from talks
Nothing was collected or created yet.
Hypertension
View on Wikipedia
| Hypertension | |
|---|---|
| Other names | Arterial hypertension, high blood pressure |
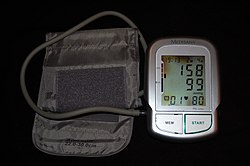 | |
| Automated arm blood pressure meter showing arterial hypertension (shown by a systolic blood pressure 158 mmHg and diastolic blood pressure 99 mmHg, with a heart rate of 80 beats per minute) | |
| Specialty | Cardiology, Nephrology |
| Symptoms | None[1] |
| Complications | Coronary artery disease, stroke, heart failure, peripheral arterial disease, vision loss, chronic kidney disease, dementia[2][3][4] |
| Causes | Usually lifestyle and genetic factors[5] |
| Risk factors | Lack of sleep, excess salt, excess body weight, smoking, alcohol[1][5] |
| Diagnostic method | Resting blood pressure in adults ≥ 140/90 mmHg[6] |
| Treatment | Lifestyle changes, medications[7] |
| Frequency | 33% (all adults), 16% (diagnosed) (globally, 2019)[8][9] |
| Deaths | 10.4 million; 19% of deaths (globally, 2019)[8] |
| Part of a series on |
| Human body weight |
|---|
Hypertension, also known as high blood pressure, is a long-term medical condition in which the blood pressure in the arteries is persistently elevated.[10] High blood pressure usually does not cause symptoms itself.[1] It is, however, a major risk factor for stroke, coronary artery disease, heart failure, atrial fibrillation, peripheral arterial disease, vision loss, chronic kidney disease, and dementia.[2][3][4][11] Hypertension is a major cause of premature death worldwide.[6]
High blood pressure is classified as primary (essential) hypertension or secondary hypertension.[5] About 90–95% of cases are primary, defined as high blood pressure due to non-specific lifestyle and genetic factors.[5] Lifestyle factors that increase the risk include excess salt in the diet, excess body weight, smoking, physical inactivity and alcohol use.[1][5] The remaining 5–10% of cases are categorized as secondary hypertension, defined as high blood pressure due to a clearly identifiable cause, such as chronic kidney disease, narrowing of the kidney arteries, an endocrine disorder, or the use of birth control pills.[5]
Blood pressure is classified by two measurements, the systolic (first number) and diastolic (second number) pressures.[1] For most adults, normal blood pressure at rest is within the range of 100–140 millimeters mercury (mmHg) systolic and 60–90 mmHg diastolic.[12][13] For most adults, high blood pressure is present if the resting blood pressure is persistently at or above 130/80 or 140/90 mmHg.[5][12][13] Different numbers apply to children.[14] Ambulatory blood pressure monitoring over a 24-hour period appears more accurate than office-based blood pressure measurement.[5][10]
Lifestyle changes and medications can lower blood pressure and decrease the risk of health complications.[7] Lifestyle changes include weight loss, physical exercise, decreased salt intake, reducing alcohol intake, and a healthy diet.[5] If lifestyle changes are not sufficient, blood pressure medications are used.[7] Up to three medications taken concurrently can control blood pressure in 90% of people.[5] The treatment of moderately high arterial blood pressure (defined as >160/100 mmHg) with medications is associated with an improved life expectancy.[15] The effect of treatment of blood pressure between 130/80 mmHg and 160/100 mmHg is less clear, with some reviews finding benefit[12][16][17][18] and others finding unclear benefit.[19][20] High blood pressure affects 33% of the population globally.[8] About half of all people with high blood pressure do not know that they have it.[8] In 2019, high blood pressure was believed to have been a factor in 19% of all deaths (10.4 million globally).[8]
Signs and symptoms
[edit]Hypertension is rarely accompanied by symptoms.[1] Half of all people with hypertension are unaware that they have it.[8] Hypertension is usually identified as part of health screening or when seeking healthcare for an unrelated problem.
Some people with high blood pressure report headaches, as well as lightheadedness, vertigo, tinnitus (buzzing or hissing in the ears), altered vision or fainting episodes.[21] These symptoms, however, might be related to associated anxiety rather than the high blood pressure itself.[22]
Long-standing untreated hypertension can cause organ damage with signs such as changes in the optic fundus seen by ophthalmoscopy.[23] The severity of hypertensive retinopathy correlates roughly with the duration or the severity of the hypertension.[21] Other hypertension-caused organ damage include chronic kidney disease and thickening of the heart muscle.[8]
Secondary hypertension
[edit]Secondary hypertension is hypertension due to an identifiable cause and may result in certain specific additional signs and symptoms. For example, as well as causing high blood pressure, Cushing's syndrome frequently causes truncal obesity,[24] glucose intolerance, moon face, a hump of fat behind the neck and shoulders (referred to as a buffalo hump), and purple abdominal stretch marks.[25] Hyperthyroidism frequently causes weight loss with increased appetite, fast heart rate, bulging eyes, and tremor. Renal artery stenosis may be associated with a localized abdominal bruit to the left or right of the midline, or in both locations. Coarctation of the aorta frequently causes a decreased blood pressure in the lower extremities relative to the arms, or delayed or absent femoral arterial pulses. Pheochromocytoma may cause abrupt episodes of hypertension accompanied by headache, palpitations, pale appearance, and excessive sweating.[25]
Hypertensive crisis
[edit]Severely elevated blood pressure (equal to or greater than a systolic pressure of 180 mmHg or a diastolic pressure of 120 mmHg) is referred to as a hypertensive crisis.[26] Hypertensive crisis is categorized as either hypertensive urgency or hypertensive emergency, according to the absence or presence of end-organ damage, respectively.[27][28]
In hypertensive urgency, there is no evidence of end-organ damage resulting from the elevated blood pressure. In these cases, oral medications are used to lower blood pressure over 24 to 48 hours gradually.[29]
In a hypertensive emergency, there is evidence of direct damage to one or more organs.[30][31] The most affected organs include the brain, kidney, heart, and lungs, producing symptoms that may include confusion, drowsiness, chest pain, and breathlessness.[29] In a hypertensive emergency, the blood pressure must be reduced more rapidly to stop ongoing organ damage;[29] however, there is a lack of randomized controlled trial evidence for this approach.[31]
Pregnancy
[edit]Hypertension occurs in approximately 8–10% of pregnancies.[25] Two blood pressure measurements six hours apart of greater than 140/90 mmHg are diagnostic of hypertension in pregnancy.[32] High blood pressure in pregnancy can be classified as pre-existing hypertension, gestational hypertension, or pre-eclampsia.[33] Women who have chronic hypertension before their pregnancy are at increased risk of complications such as premature birth, low birthweight or stillbirth.[34] Women who have high blood pressure and had complications in their pregnancy have three times the risk of developing cardiovascular disease compared to women with normal blood pressure who had no complications in pregnancy.[35][36]
Pre-eclampsia is a serious condition in the second half of pregnancy characterised by increased blood pressure and the presence of protein in the urine.[25] It occurs in about 5% of pregnancies and is responsible for approximately 16% of all maternal deaths globally.[25] Pre-eclampsia also doubles the risk of death of the baby around the time of birth.[25] Usually, there are no symptoms in pre-eclampsia, and it is detected by routine screening. When symptoms of pre-eclampsia occur, the most common are headache, visual disturbance (often "flashing lights"), vomiting, pain over the stomach, and swelling. Pre-eclampsia can occasionally progress to a life-threatening condition called eclampsia, which is a hypertensive emergency and has several serious complications including vision loss, brain swelling, seizures, kidney failure, pulmonary edema, and disseminated intravascular coagulation (a blood clotting disorder).[25][37]
In contrast, gestational hypertension is defined as new-onset hypertension in the second half of pregnancy without protein in the urine.[33]
Exercise during pregnancy is generally safe and may improve outcomes in hypertension during pregnancy,[38] and may prevent gestational hypertension and related complications.[39][40][41]
Children
[edit]Failure to thrive, seizures, irritability, lack of energy, and difficulty in breathing[42] can be associated with hypertension in newborns and young infants. In older infants and children, hypertension can cause headache, unexplained irritability, fatigue, failure to thrive, blurred vision, nosebleeds, and facial paralysis.[42][43]
Causes
[edit]Primary hypertension
[edit]Primary (also termed essential) hypertension results from a complex interaction of genes and environmental factors. More than 2000 common genetic variants with small effects on blood pressure have been identified in association with high blood pressure,[44] as well as some rare genetic variants with large effects on blood pressure.[45] There is also evidence that DNA methylation at multiple nearby CpG sites may link some sequence variation to blood pressure, possibly via effects on vascular or renal function.[46]
Blood pressure rises with aging in societies with a western diet and lifestyle,[47] and the risk of becoming hypertensive in later life is substantial in most such societies.[48] Several environmental or lifestyle factors influence blood pressure. Reducing dietary salt intake lowers blood pressure;[49] as does weight loss,[50] exercise training,[51] vegetarian diets,[52] increased dietary potassium intake[53] and high dietary calcium supplementation.[54] Increasing alcohol intake is associated with higher blood pressure,[55] but the possible roles of other factors such as caffeine consumption,[56] and vitamin D deficiency[57] are less clear. Average blood pressure is higher in the winter than in the summer.[58]
Depression is associated with hypertension[59] and loneliness is also a risk factor.[60] Periodontal disease is also associated with high blood pressure.[61] Arsenic exposure through drinking water is associated with elevated blood pressure.[62] Air pollution is associated with hypertension.[63] Whether these associations are causal is unknown. Gout and elevated blood uric acid are associated with hypertension[64] and evidence from genetic (Mendelian Randomization) studies and clinical trials indicate this relationship is likely to be causal.[65] Insulin resistance, which is common in obesity and is a component of syndrome X (or metabolic syndrome), can cause hyperuricemia and gout[66] and is also associated with elevated blood pressure.[67]
Events in early life, such as low birth weight, maternal smoking, and lack of breastfeeding may be risk factors for adult essential hypertension, although the strength of the relationships is weak and the mechanisms linking these exposures to adult hypertension remain unclear.[68]
Secondary hypertension
[edit]Secondary hypertension results from an identifiable cause. Kidney disease is the most common secondary cause of hypertension.[25] Hypertension can also be caused by endocrine conditions, such as Cushing's syndrome, hyperthyroidism, hypothyroidism, acromegaly, Conn's syndrome or hyperaldosteronism, renal artery stenosis (from atherosclerosis or fibromuscular dysplasia), hyperparathyroidism, and pheochromocytoma.[25][69] Other causes of secondary hypertension include obesity, sleep apnea, pregnancy, coarctation of the aorta, excessive eating of liquorice, excessive drinking of alcohol, certain prescription medicines, herbal remedies, and stimulants such as cocaine and methamphetamine.[25][70]
A 2018 review found that any alcohol increased blood pressure in males, while over one or two drinks increased the risk in females.[71]
Pathophysiology
[edit]

In most people with established essential hypertension, increased resistance to blood flow (total peripheral resistance) accounts for the high pressure while cardiac output remains normal.[72] There is evidence that some younger people with prehypertension or 'borderline hypertension' have high cardiac output, an elevated heart rate and normal peripheral resistance, termed hyperkinetic borderline hypertension.[73] These individuals may develop the typical features of established essential hypertension in later life as their cardiac output falls and peripheral resistance rises with age.[73] Whether this pattern is typical of all people who ultimately develop hypertension is disputed.[74] The increased peripheral resistance in established hypertension is mainly attributable to structural narrowing of small arteries and arterioles,[75] although a reduction in the number or density of capillaries may also contribute.[76]
It is unclear whether or not vasoconstriction of arteriolar blood vessels plays a role in hypertension.[77] Hypertension is also associated with decreased peripheral venous compliance,[78] which may increase venous return, increase cardiac preload and, ultimately, cause diastolic dysfunction. For patients having hypertension, higher heart rate variability (HRV) is a risk factor for atrial fibrillation.[79]
Pulse pressure (the difference between systolic and diastolic blood pressure) is frequently increased in older people with hypertension.[80] This can mean that systolic pressure is abnormally high, but diastolic pressure may be normal or low, a condition termed isolated systolic hypertension.[81] The high pulse pressure in elderly people with hypertension or isolated systolic hypertension is explained by increased arterial stiffness, which typically accompanies aging and may be exacerbated by high blood pressure.[82]
Many mechanisms have been proposed to account for the rise in peripheral resistance in hypertension. Most evidence implicates either disturbances in the kidneys' salt and water handling (particularly abnormalities in the intrarenal renin–angiotensin system)[83] or abnormalities of the sympathetic nervous system.[84] These mechanisms are not mutually exclusive, and both likely contribute to some extent in most cases of essential hypertension. It has also been suggested that endothelial dysfunction and vascular inflammation may also contribute to increased peripheral resistance and vascular damage in hypertension.[85][86] Interleukin 17 has garnered interest for its role in increasing the production of several other immune system chemical signals thought to be involved in hypertension such as tumor necrosis factor alpha, interleukin 1, interleukin 6, and interleukin 8.[87]
Excessive sodium or insufficient potassium in the diet leads to excessive intracellular sodium, which contracts vascular smooth muscle, restricting blood flow and so increases blood pressure.[88][89] Non-modulating essential hypertension is a form of salt-sensitive hypertension, where sodium intake does not modulate either adrenal or renal vascular responses to angiotensin II.[90] They make up 25% of the hypertensive population.[91]
Diagnosis
[edit]Hypertension is diagnosed based on persistently high resting blood pressure. Elevated blood pressure measurements on at least two separate occasions are required for a diagnosis of hypertension.[12][13][6]
Measurement technique
[edit]For an accurate diagnosis of hypertension to be made, proper blood pressure measurement technique must be used.[92] Improper measurement of blood pressure is common and can change the blood pressure reading by up to 10 mmHg, which can lead to misdiagnosis and misclassification of hypertension.[92] The correct blood pressure measurement technique involves several steps. Proper blood pressure measurement requires the person whose blood pressure is being measured to sit quietly for at least five minutes, which is then followed by the application of a properly fitted blood pressure cuff to a bare upper arm.[92] The person should be seated with their back supported, feet flat on the floor, and with their legs uncrossed.[92] The person whose blood pressure is being measured should avoid talking or moving during this process.[92] The arm being measured should be supported on a flat surface at the level of the heart.[92] Blood pressure measurement should be done in a quiet room so the medical professional checking the blood pressure can hear the Korotkoff sounds while listening to the brachial artery with a stethoscope for accurate blood pressure measurements.[92][93] The blood pressure cuff should be deflated slowly (2–3 mmHg per second) while listening for the Korotkoff sounds.[93] The bladder should be emptied before a person's blood pressure is measured since this can increase blood pressure by up to 15/10 mmHg.[92] Multiple blood pressure readings (at least two) spaced 1–2 minutes apart should be obtained to ensure accuracy.[93] Ambulatory blood pressure monitoring over 12 to 24 hours is the most accurate method to confirm the diagnosis.[94] An exception to this is those with very high blood pressure readings, especially when there is poor organ function.[95]
With the availability of 24-hour ambulatory blood pressure monitors and home blood pressure machines, the importance of not wrongly diagnosing those who have white coat hypertension has led to a change in protocols. In the United Kingdom, the current best practice is to follow up a single raised clinic reading with ambulatory measurement, or, less ideally, with home blood pressure monitoring over 7 days.[95] The United States Preventive Services Task Force also recommends getting measurements outside of the healthcare environment.[94] Pseudohypertension in the elderly or noncompressibility artery syndrome may also require consideration. This condition is believed to be due to calcification of the arteries, resulting in abnormally high blood pressure readings with a blood pressure cuff, while intra-arterial measurements of blood pressure are normal.[96] Orthostatic hypertension is when blood pressure increases upon standing.[97]
Other investigations
[edit]Once the diagnosis of hypertension has been made, further testing may be performed to find secondary hypertension, identify comorbidities such as diabetes, identify hypertension-caused organ damage such as chronic kidney disease or thickening of the heart muscle, and for cardiovascular disease risk stratification.[98]
Secondary hypertension is more common in preadolescent children, with most cases caused by kidney disease. Primary or essential hypertension is more common in adolescents and adults and has multiple risk factors, including obesity and a family history of hypertension.[99]
Initial assessment upon diagnosis of hypertension should include a complete history and physical examination. The World Health Organization suggests the following initial tests: serum electrolytes, serum creatinine, lipid panel, HbA1c or fasting glucose, urine dipstick and electrocardiogram (ECG/EKG).[98] Serum creatinine is measured to assess for the presence of kidney disease, which can be either the cause or the result of hypertension.[30] eGFR can also provide a baseline measurement of kidney function that can be used to monitor for side effects of certain anti-hypertensive drugs on kidney function. Testing of urine samples for protein is used as a secondary indicator of kidney disease. Lipid panel and glucose tests are done to identify comorbidities such as diabetes and hyperlipidemia and for cardiovascular risk stratification. Electrocardiogram testing is done to check for evidence that the heart is under strain from high blood pressure, such as thickening of the heart muscle or whether the heart has experienced a prior minor disturbance, such as a silent heart attack.
Classification in adults
[edit]The circumstances of measurement can influence blood pressure measurements.[100] Guidelines use different thresholds for office (also known as clinic), home (when the patient measures their blood pressure at home), and ambulatory blood pressure (using an automated device over 24 hours).[100]
| Categories | Systolic blood pressure, mmHg | and/or | Diastolic blood pressure, mmHg | ||||
|---|---|---|---|---|---|---|---|
| Method | Office | Home | 24h ambulatory | Office | Home | 24h ambulatory | |
| American College of Cardiology/American Heart Association (2017)[101] | |||||||
| Normal | <120 | <120 | <115 | and | <80 | <80 | <75 |
| Elevated | 120–129 | 120–129 | 115–124 | and | <80 | <80 | <75 |
| Hypertension, stage 1 | 130–139 | 130–134 | 125–129 | or | 80–89 | 80–84 | 75–79 |
| Hypertension, stage 2 | ≥140 | ≥135 | ≥130 | or | ≥90 | ≥85 | ≥80 |
| European Society of Cardiology (2024)[100] | |||||||
| Non-elevated | <120 | <120 | <115 | and | <70 | <70 | <65 |
| Elevated | 120–139 | 120–135 | 115–129 | and | 70–89 | 70–85 | 65–79 |
| Hypertension | ≥140 | ≥135 | ≥130 | or | ≥90 | ≥85 | ≥80 |
| European Society of Hypertension/International Society of Hypertension (2023)[13] | |||||||
| Optimal | <120 | — | — | and | <80 | — | — |
| Normal | 120–129 | — | — | and/or | 80–84 | — | — |
| High normal | 130–139 | — | — | and/or | 85–89 | — | — |
| Hypertension, grade 1 | 140–159 | ≥135 | ≥130 | and/or | 90–99 | ≥85 | ≥80 |
| Hypertension, grade 2 | 160–179 | — | — | and/or | 100–109 | — | — |
| Hypertension, grade 3 | ≥180 | — | — | and/or | ≥110 | — | — |
Children
[edit]Hypertension occurs in around 0.2 to 3% of newborns; however, blood pressure is not measured routinely in healthy newborns.[43] Hypertension is more common in high risk newborns. A variety of factors, such as gestational age, postconceptional age, and birth weight need to be taken into account when deciding if blood pressure is normal in a newborn.[43]
Hypertension, defined as elevated blood pressure over several visits, affects 1% to 5% of children and adolescents and is associated with long-term risks of ill-health.[102] Blood pressure rises with age in childhood, and, in children, hypertension is defined as an average systolic or diastolic blood pressure on three or more occasions equal or higher than the 95th percentile appropriate for the sex, age, and height of the child. High blood pressure must be confirmed on repeated visits, however, before characterizing a child as having hypertension.[102] In adolescents, it has been proposed that hypertension is diagnosed and classified using the same criteria as in adults.[102]
Prevention
[edit]Much of the disease burden of high blood pressure is experienced by people who are not labeled as hypertensive.[103] Consequently, population strategies are required to reduce the consequences of high blood pressure and reduce the need for antihypertensive medications. Lifestyle changes are recommended to lower blood pressure.
Recommended lifestyle changes for the prevention of hypertension include:
- maintain normal body weight for adults (e.g. body mass index below 25 kg/m2)[13]
- reduce dietary sodium intake to <100 mmol/day (<6 g of salt (sodium chloride) or <2.4 g of sodium per day)[13]
- engage in regular aerobic physical activity with moderate intensity (minimum 150 minutes per week)[13]
- limit alcohol consumption,[13] max 1 drink for women and 2 for men per day[6]
- consume a diet rich in whole grains, fruits, and vegetables,[13] such as the DASH diet[13]
- not smoking[13]
- stress reduction and management,[13] e.g. by meditation and yoga[13]
Effective lifestyle modification may lower blood pressure as much as an individual antihypertensive medication. Combinations of two or more lifestyle modifications can achieve even better results.[103] There is considerable evidence that reducing dietary salt intake lowers blood pressure, but whether this translates into a reduction in mortality and cardiovascular disease remains uncertain.[104] Estimated sodium intake ≥6 g/day and <3 g/day are both associated with high risk of death or major cardiovascular disease, but the association between high sodium intake and adverse outcomes is only observed in people with hypertension.[105] Consequently, in the absence of results from randomized controlled trials, the wisdom of reducing levels of dietary sodium intake below 3 g/day has been questioned.[104] ESC guidelines mention periodontitis is associated with poor cardiovascular health status.[106]
The value of routine screening for hypertension is debated.[107][108][109] In 2004, the National High Blood Pressure Education Program recommended that children aged 3 years and older have blood pressure measurement at least once at every health care visit[102] and the National Heart, Lung, and Blood Institute and American Academy of Pediatrics made a similar recommendation.[110] However, the American Academy of Family Physicians[111] supports the view of the U.S. Preventive Services Task Force that the available evidence is insufficient to determine the balance of benefits and harms of screening for hypertension in children and adolescents who do not have symptoms.[112][113] The US Preventive Services Task Force recommends screening adults 18 years or older for hypertension with office blood pressure measurement.[109][114]
Management
[edit]According to one review published in 2003, reduction of the blood pressure by 5 mmHg can decrease the risk of stroke by 34%, of ischemic heart disease by 21%, and reduce the likelihood of dementia, heart failure, and mortality from cardiovascular disease.[115]
Target blood pressure
[edit]Various expert groups have produced guidelines regarding how low the blood pressure target should be when a person is treated for hypertension. These groups recommend a target below the range of 140–160 / 90–100 mmHg for the general population.[13][14][116][117] Cochrane reviews recommend similar targets for subgroups such as people with diabetes[118] and people with prior cardiovascular disease.[119] Additionally, Cochrane reviews have found that for older individuals with moderate to high cardiovascular risk, the benefits of trying to achieve a lower-than-standard blood pressure target (at or below 140/90 mmHg) are outweighed by the risk associated with the intervention.[120] These findings may not be applicable to other populations.[120]
Many expert groups recommend a slightly higher target of 150/90 mmHg for those somewhere between 60 and 80 years of age.[116][117][121] The JNC 8 and American College of Physicians recommend the target of 150/90 mmHg for those over 60 years of age,[14][121] but some experts within these groups disagree with this recommendation.[122] Some expert groups have also recommended slightly lower targets in those with diabetes[123] or chronic kidney disease,[124] but others recommend the same target as the general population.[14][118] The issue of what is the best target and whether targets should differ for high-risk individuals is unresolved,[125] although some experts propose more intensive blood pressure lowering than advocated in some guidelines.[126]
The 2025 American Heart Association guidelines recommend medication for all adults with average blood pressure 140/90 mmHg or higher.[127] It also recommends medication for adults with blood pressure 130/80 mmHg or higher with a 10-year risk of cardiovascular disease 7,5% or more, and in those with blood pressure 130/80 mmHg or higher with a 10-year cardiovascular risk of less than 7,5% where blood pressure remains above 130/80 mmHg after 3–6 months of lifestyle modifications.[127]
Lifestyle modifications
[edit]The first line of treatment for hypertension is lifestyle changes, including dietary changes, physical activity, and weight loss. Though these have all been recommended in scientific advisories,[128] a Cochrane systematic review found no evidence (due to lack of data) for effects of weight loss diets on death, long-term complications or adverse events in persons with hypertension.[129] The review did find a decrease in body weight and blood pressure.[129] Their potential effectiveness is similar to and at times exceeds a single medication.[13] If hypertension is high enough to justify immediate use of medications, lifestyle changes are still recommended in conjunction with medication.
Dietary changes shown to reduce blood pressure include diets with low sodium,[130][131] the DASH diet (Dietary Approaches to Stop Hypertension),[132] which was the best against 11 other diet in an umbrella review,[133] and plant-based diets.[134] A 2024 clinical guideline recommended an increase dietary fiber intake,[135] with a minimum of 28g/day for women and 38g/day for men diagnosed with hypertension.[136]
Increasing dietary potassium has a potential benefit for lowering the risk of hypertension.[137][138] The 2015 Dietary Guidelines Advisory Committee (DGAC) stated that potassium is one of the shortfall nutrients which is under-consumed in the United States.[139] However, people who take certain antihypertensive medications (such as ACE-inhibitors or ARBs) should not take potassium supplements or potassium-enriched salts due to the risk of high levels of potassium.[140]
Physical exercise regimens which are shown to reduce blood pressure include isometric resistance exercise, aerobic exercise, resistance exercise, and device-guided breathing.[141]
A 2020 Cochrane review examined the impact of walking on blood pressure and heart rate in adults. The review found that walking likely reduces systolic blood pressure, with consistent effects across different age groups and both sexes. There was also some evidence that walking may lower diastolic blood pressure and heart rate. Overall, the certainty of evidence ranged from moderate to low, depending on the outcome and subgroup. Walking appears to be a safe, accessible, and potentially effective strategy for supporting cardiovascular health.[142]
Stress reduction techniques such as biofeedback or transcendental meditation may be considered as an add-on to other treatments to reduce hypertension, but do not have evidence for preventing cardiovascular disease on their own.[141][143][144] Self-monitoring and appointment reminders might support the use of other strategies to improve blood pressure control, but need further evaluation.[145]
Medications
[edit]Several classes of medications, collectively referred to as antihypertensive medications, are available for treating hypertension.
First-line medications for hypertension include thiazide-diuretics, calcium channel blockers, angiotensin converting enzyme inhibitors (ACE inhibitors), and angiotensin receptor blockers (ARBs).[146][14] These medications may be used alone or in combination (ACE inhibitors and ARBs are not recommended for use together); the latter option may serve to minimize counter-regulatory mechanisms that act to restore blood pressure values to pre-treatment levels,[14][147] although the evidence for first-line combination therapy is not strong enough.[148] Most people require more than one medication to control their hypertension.[128] Medications for blood pressure control should be implemented by a stepped care approach when target levels are not reached.[145] Withdrawal of such medications in the elderly can be considered by healthcare professionals, because there is no strong evidence of an effect on mortality, myocardial infarction, or stroke.[149][needs update]
Previously, beta-blockers such as atenolol were thought to have similar beneficial effects when used as first-line therapy for hypertension. However, a Cochrane review that included 13 trials found that the effects of beta-blockers are inferior to those of other antihypertensive medications in preventing cardiovascular disease.[150]
The prescription of antihypertensive medication for children with hypertension has limited evidence. There is limited evidence that compares it with a placebo and shows a modest effect on blood pressure in the short term. Administration of a higher dose did not reduce blood pressure further.[151]
Resistant hypertension
[edit]Resistant hypertension is defined as high blood pressure that remains above a target level, despite being prescribed three or more antihypertensive drugs simultaneously with different mechanisms of action.[152][13][153] Failing to take prescribed medications as directed is an important cause of resistant hypertension.[154] To confirm true resistant hypertension, the drug treatment regimen should consist of optimal or best tolerated doses. Inadequate blood pressure control should be verified by out-of-office measurement methods, such as home blood pressure monitoring or 24-hour ambulatory blood pressure monitoring, to exclude white-coat effect. Adherence to therapy should be confirmed and secondary causes of hypertension excluded. If these factors are not addressed, the terms pseudoresistant or apparent resistant hypertension are proposed.[13]
Some common secondary causes of resistant hypertension include obstructive sleep apnea, primary aldosteronism and renal artery stenosis, and some rare secondary causes are pheochromocytoma and coarctation of the aorta.[155] As many as one in five people with resistant hypertension have primary aldosteronism, which is a treatable and sometimes curable condition.[156] Resistant hypertension may also result from chronically high activity of the autonomic nervous system, an effect known as neurogenic hypertension.[157] Electrical therapies that stimulate the baroreflex are being studied as an option for lowering blood pressure in people in this situation.[158]
Refractory hypertension is described by one source as elevated blood pressure unmitigated by five or more concurrent antihypertensive agents of different classes.[159] People with refractory hypertension typically have increased sympathetic nervous system activity, and are at high risk for more severe cardiovascular diseases and all-cause mortality.[159][160]
Epidemiology
[edit]

| no data <110 110–220 220–330 330–440 440–550 550–660 | 660–770 770–880 880–990 990–1100 1100–1600 >1600 |
Adults
[edit]In 2024, one in three or 33% of the world population were estimated to have hypertension.[6] Of all people with hypertension, almost half (about 44%) do not know that they have hypertension.[6] In 1990, about 650 million people had a diagnosis of hypertension, which increased to 1.4 billion by 2024 mostly due a rise of the number of older adults in low- and middle-income countries.[6]
Hypertension is slightly more frequent in men.[9] In people aged under 50 years, more men than women have hypertension,[9] and in ages above 50 years the prevalence of hypertension is the same in men and women.[9] In ages above 65 years, more women than men have hypertension.[13] Hypertension becomes more common with age.[5] Hypertension is common in high, medium, and low-income countries.[2] It is more common in people of low socioeconomic status.[163] Hypertension is around twice as common in diabetics.[164]
In 2019, rates of diagnosed hypertension were highest in Africa (30% for both sexes), and lowest in the Americas (18% for both sexes).[9] Rates also vary markedly within regions with country-level rates as low as 22.8% (men) and 18.4% (women) in Peru and as high as 61.6% (men) and 50.9% (women) in Paraguay.[9]
In 1995, it was estimated that 24% of the United States population had hypertension or were taking antihypertensive medication.[165] By 2004 this had increased to 29%[166][167] and further to 32% (76 million US adults) by 2017.[12] In 2017, with the American guidelines' change in definition for hypertension, 46% of people in the United States are affected.[12] Some data shows African-American adults in the United States have among the highest rates of hypertension in the world at 44%.[168] However, other research argues there has been a "myopic perspective" on American data and notes that other groups, particularly Russians and Eastern Europeans, have markedly higher rates of hypertension than Black Americans.[169] Differences in hypertension rates are multifactorial and under study.[170]
Children
[edit]Rates of high blood pressure in children and adolescents have increased in the last 20 years in the United States.[171] Childhood hypertension, particularly in pre-adolescents, is more often secondary to an underlying disorder than in adults. Kidney disease is the most common secondary cause of hypertension in children and adolescents. Nevertheless, primary or essential hypertension accounts for most cases.[172]
Prognosis
[edit]
Hypertension is the most important preventable risk factor for premature death worldwide.[173] It increases the risk of ischemic heart disease,[174] stroke,[25] peripheral vascular disease,[175] and other cardiovascular diseases, including heart failure, aortic aneurysms, diffuse atherosclerosis, chronic kidney disease, atrial fibrillation, cancers, leukemia and pulmonary embolism.[11][25] Hypertension is also a risk factor for cognitive impairment and dementia.[25] Other complications include hypertensive retinopathy and hypertensive nephropathy.[30]
History
[edit]
Measurement
[edit]Modern understanding of the cardiovascular system began with the work of physician William Harvey (1578–1657), who described the circulation of blood in his book "De motu cordis". The English clergyman Stephen Hales made the first published measurement of blood pressure in 1733.[176][177] However, hypertension as a clinical entity came into its own with the invention of the cuff-based sphygmomanometer by Scipione Riva-Rocci in 1896.[178] This allowed easy measurement of systolic pressure in the clinic. In 1905, Nikolai Korotkoff improved the technique by describing the Korotkoff sounds that are heard when the artery is auscultated with a stethoscope while the sphygmomanometer cuff is deflated.[177] This permitted systolic and diastolic pressure to be measured.
Identification
[edit]Symptoms similar to those of patients with a hypertensive crisis are discussed in medieval Persian medical texts in the chapter of "fullness disease".[179] The symptoms include headache, heaviness in the head, sluggish movements, general redness and warm to touch feel of the body, prominent, distended and tense vessels, a fullness of the pulse, distension of the skin, coloured and dense urine, loss of appetite, weak eyesight, impairment of thinking, yawning, drowsiness, vascular rupture, and hemorrhagic stroke.[180] Fullness disease was presumed to be due to an excessive amount of blood within the blood vessels.
Descriptions of hypertension as a disease came, among others, from Thomas Young in 1808 and especially Richard Bright in 1836.[176] The first report of elevated blood pressure in a person without evidence of kidney disease was made by Frederick Akbar Mahomed (1849–1884).[181]
Until the 1990s, systolic hypertension was defined as systolic blood pressure of 160 mmHg or greater.[182] In 1993, the WHO/ISH guidelines defined 140 mmHg as the threshold for hypertension.[183]
Treatment
[edit]Historically, the treatment for what was called the "hard pulse disease" consisted of reducing the quantity of blood by bloodletting or the application of leeches.[176] This was advocated by The Yellow Emperor of China, Cornelius Celsus, Galen, and Hippocrates.[176] The therapeutic approach for the treatment of hard pulse disease included lifestyle changes (staying away from anger and sexual intercourse) and dietary program for patients (avoiding the consumption of wine, meat, and pastries, reducing the volume of food in a meal, maintaining a low-energy diet and the dietary usage of spinach and vinegar).
In the 19th and 20th centuries, before effective pharmacological treatment for hypertension became possible, three treatment modalities were used, all with numerous side effects: strict sodium restriction (for example the rice diet[176]), sympathectomy (surgical ablation of parts of the sympathetic nervous system), and pyrogen therapy (injection of substances that caused a fever, indirectly reducing blood pressure).[176][184]
The first chemical for hypertension, sodium thiocyanate, was used in 1900 but had many side effects and was unpopular.[176] Several other agents were developed after the Second World War, the most popular and reasonably effective of which were tetramethylammonium chloride, hexamethonium, hydralazine, and reserpine (derived from the medicinal plant Rauvolfia serpentina). None of these were well tolerated.[185][186] A major breakthrough was achieved with the discovery of the first well-tolerated orally available agents. The first was chlorothiazide, the first thiazide diuretic and developed from the antibiotic sulfanilamide, which became available in 1958.[176][187] Subsequently, beta blockers, calcium channel blockers, angiotensin converting enzyme (ACE) inhibitors, angiotensin receptor blockers, and renin inhibitors were developed as antihypertensive agents.[184]
Society and culture
[edit]Awareness
[edit]
The World Health Organization has identified hypertension (high blood pressure) as the leading cause of cardiovascular mortality.[188] The World Hypertension League (WHL), an umbrella organization of 85 national hypertension societies and leagues, recognized that more than 50% of the hypertensive population worldwide are unaware of their condition.[188] To address this problem, the WHL initiated a global awareness campaign on hypertension in 2005 and dedicated 17 May of each year as World Hypertension Day.[189]
Economics
[edit]High blood pressure is the most common chronic medical problem prompting visits to primary health care providers in the US. The American Heart Association estimated the direct and indirect costs of high blood pressure in 2010 as $76.6 billion.[168] In the US, 80% of people with hypertension are aware of their condition, 71% take some antihypertensive medication, but only 48% of people aware that they have hypertension adequately control it.[168] Adequate management of hypertension can be hampered by inadequacies in the diagnosis, treatment, or control of high blood pressure.[190] Health care providers face many obstacles to achieving blood pressure control, including resistance to taking multiple medications to reach blood pressure goals. People also face the challenges of adhering to medical schedules and making lifestyle changes. Nonetheless, the achievement of blood pressure goals is possible, and most importantly, lowering blood pressure significantly reduces the risk of death due to heart disease and stroke, the development of other debilitating conditions, and the cost associated with advanced medical care.[191][192] The cost of medications discourages many patients from continuing treatment.[193]
Other animals
[edit]Hypertension in cats is indicated by a systolic blood pressure greater than 150 mmHg, with amlodipine the usual first-line treatment. A cat with a systolic blood pressure above 170 mmHg is considered hypertensive. If a cat has other problems, such as kidney disease or retina detachment, then a blood pressure below 160 mmHg may also need to be monitored.[194]
Normal blood pressure in dogs can differ substantially between breeds, but hypertension is often diagnosed if systolic blood pressure is above 160 mmHg, particularly if this is associated with target organ damage.[195] Inhibitors of the renin-angiotensin system and calcium channel blockers are often used to treat hypertension in dogs, although other drugs may be indicated for specific conditions causing high blood pressure.[195]
See also
[edit]References
[edit]- ^ a b c d e f "About High Blood Pressure". Centers for Disease Control and Prevention (CDC). 15 May 2024. Archived from the original on 20 May 2024. Retrieved 22 May 2024.
- ^ a b c Lackland DT, Weber MA (May 2015). "Global burden of cardiovascular disease and stroke: hypertension at the core". The Canadian Journal of Cardiology. 31 (5): 569–571. doi:10.1016/j.cjca.2015.01.009. PMID 25795106.
- ^ a b Mendis S, Puska P, Norrving B (2011). Global atlas on cardiovascular disease prevention and control (PDF) (1st ed.). Geneva: World Health Organization in collaboration with the World Heart Federation and the World Stroke Organization. p. 38. ISBN 978-92-4-156437-3. Archived from the original (PDF) on 17 August 2014.
- ^ a b Hernandorena I, Duron E, Vidal JS, Hanon O (July 2017). "Treatment options and considerations for hypertensive patients to prevent dementia". Expert Opinion on Pharmacotherapy (Review). 18 (10): 989–1000. doi:10.1080/14656566.2017.1333599. PMID 28532183. S2CID 46601689.
- ^ a b c d e f g h i j k Poulter NR, Prabhakaran D, Caulfield M (August 2015). "Hypertension". Lancet. 386 (9995): 801–812. doi:10.1016/s0140-6736(14)61468-9. PMID 25832858. S2CID 208792897.
- ^ a b c d e f g "Hypertension". World Health Organization (WHO). 25 September 2025. Retrieved 1 October 2025.
- ^ a b c "How Is High Blood Pressure Treated?". National Heart, Lung, and Blood Institute. 10 September 2015. Archived from the original on 6 April 2016. Retrieved 6 March 2016.
- ^ a b c d e f g Global report on hypertension: the race against a silent killer. Geneva: World Health Organization (WHO). 19 March 2023. ISBN 978-92-4-008106-2.
- ^ a b c d e f Ezzati M, Zhou B, Carrillo-Larco RM, Danaei G, Riley LM, Paciorek CJ, et al. (NCD Risk Factor Collaboration) (11 September 2021). "Worldwide trends in hypertension prevalence and progress in treatment and control from 1990 to 2019: a pooled analysis of 1201 population-representative studies with 104 million participants". The Lancet. 398 (10304): 957–980. doi:10.1016/S0140-6736(21)01330-1. ISSN 0140-6736. PMC 8446938. PMID 34450083. S2CID 237286310.
- ^ a b Naish J, Court DS (2014). Medical sciences (2 ed.). Elsevier Health Sciences. p. 562. ISBN 978-0-7020-5249-1.
- ^ a b Lau DH, Nattel S, Kalman JM, Sanders P (August 2017). "Modifiable Risk Factors and Atrial Fibrillation". Circulation (Review). 136 (6): 583–596. doi:10.1161/CIRCULATIONAHA.116.023163. PMID 28784826.
- ^ a b c d e f Whelton PK, Carey RM, Aronow WS, Casey DE, Collins KJ, Dennison Himmelfarb C, DePalma SM, Gidding S, Jamerson KA, Jones DW, MacLaughlin EJ, Muntner P, Ovbiagele B, Smith SC, Spencer CC, Stafford RS, Taler SJ, Thomas RJ, Williams KA, Williamson JD, Wright JT (June 2018). "2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines". Hypertension. 71 (6): e13 – e115. doi:10.1161/HYP.0000000000000065. PMID 29133356.
- ^ a b c d e f g h i j k l m n o p q r Mancia G, Kreutz R, Brunström M, Burnier M, Grassi G, Januszewicz A, et al. (1 December 2023). "2023 ESH Guidelines for the management of arterial hypertension: The Task Force for the management of arterial hypertension of the European Society of Hypertension: Endorsed by the International Society of Hypertension (ISH) and the European Renal Association (ERA)". Journal of Hypertension. 41 (12): 1874–2071. doi:10.1097/HJH.0000000000003480. hdl:11379/603005. ISSN 1473-5598. PMID 37345492.
- ^ a b c d e f James PA, Oparil S, Carter BL, Cushman WC, Dennison-Himmelfarb C, Handler J, Lackland DT, LeFevre ML, MacKenzie TD, Ogedegbe O, Smith SC, Svetkey LP, Taler SJ, Townsend RR, Wright JT, Narva AS, Ortiz E (February 2014). "2014 evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8)". JAMA. 311 (5): 507–520. doi:10.1001/jama.2013.284427. PMID 24352797.
- ^ Musini VM, Tejani AM, Bassett K, Puil L, Wright JM (June 2019). "Pharmacotherapy for hypertension in adults 60 years or older". The Cochrane Database of Systematic Reviews. 6 (6) CD000028. doi:10.1002/14651858.CD000028.pub3. PMC 6550717. PMID 31167038.
- ^ Sundström J, Arima H, Jackson R, Turnbull F, Rahimi K, Chalmers J, Woodward M, Neal B (February 2015). "Effects of blood pressure reduction in mild hypertension: a systematic review and meta-analysis". Annals of Internal Medicine. 162 (3): 184–191. doi:10.7326/M14-0773. PMID 25531552. S2CID 46553658.
- ^ Xie X, Atkins E, Lv J, Bennett A, Neal B, Ninomiya T, Woodward M, MacMahon S, Turnbull F, Hillis GS, Chalmers J, Mant J, Salam A, Rahimi K, Perkovic V, Rodgers A (January 2016). "Effects of intensive blood pressure lowering on cardiovascular and renal outcomes: updated systematic review and meta-analysis". Lancet. 387 (10017): 435–443. doi:10.1016/S0140-6736(15)00805-3. PMID 26559744. S2CID 36805676.
- ^ Falk JM, Froentjes L, Kirkwood JE, Heran BS, Kolber MR, Allan GM, Korownyk CS, Garrison SR (17 December 2024). "Higher blood pressure targets for hypertension in older adults". The Cochrane Database of Systematic Reviews. 2024 (12) CD011575. doi:10.1002/14651858.CD011575.pub3. ISSN 1469-493X. PMC 11650777. PMID 39688187.
- ^ Diao D, Wright JM, Cundiff DK, Gueyffier F (August 2012). "Pharmacotherapy for mild hypertension". The Cochrane Database of Systematic Reviews. 8 (8) CD006742. doi:10.1002/14651858.CD006742.pub2. PMC 8985074. PMID 22895954. S2CID 42363250.
- ^ Musini VM, Gueyffier F, Puil L, Salzwedel DM, Wright JM (August 2017). "Pharmacotherapy for hypertension in adults aged 18 to 59 years". The Cochrane Database of Systematic Reviews. 2017 (8) CD008276. doi:10.1002/14651858.CD008276.pub2. PMC 6483466. PMID 28813123.
- ^ a b Fisher ND, Williams GH (2005). "Hypertensive vascular disease". In Kasper DL, Braunwald E, Fauci AS, et al. (eds.). Harrison's Principles of Internal Medicine (16th ed.). New York: McGraw-Hill. pp. 1463–1481. ISBN 978-0-07-139140-5.
- ^ Marshall IJ, Wolfe CD, McKevitt C (July 2012). "Lay perspectives on hypertension and drug adherence: systematic review of qualitative research". The BMJ. 345 e3953. doi:10.1136/bmj.e3953. PMC 3392078. PMID 22777025.
- ^ Wong TY, Wong T, Mitchell P (February 2007). "The eye in hypertension". Lancet. 369 (9559): 425–435. doi:10.1016/S0140-6736(07)60198-6. PMID 17276782. S2CID 28579025.
- ^ "Truncal obesity (Concept Id: C4551560) – MedGen – NCBI". ncbi.nlm.nih.gov. Retrieved 24 April 2022.
- ^ a b c d e f g h i j k l m O'Brien E, Beevers DG, Lip GY (2007). ABC of hypertension. London: BMJ Books. ISBN 978-1-4051-3061-5.
- ^ "High Blood Pressure – Understanding the Silent Killer". Center for Drug Evaluation and Research. U.S. Food and Drug Administration. 21 January 2021.
- ^ Rodriguez MA, Kumar SK, De Caro M (1 April 2010). "Hypertensive crisis". Cardiology in Review. 18 (2): 102–107. doi:10.1097/CRD.0b013e3181c307b7. PMID 20160537. S2CID 34137590.
- ^ "Hypertensive Crisis". heart.org. Archived from the original on 25 July 2015. Retrieved 25 July 2015.
- ^ a b c Marik PE, Varon J (June 2007). "Hypertensive crises: challenges and management". Chest. 131 (6): 1949–1962. doi:10.1378/chest.06-2490. PMID 17565029. Archived from the original on 4 December 2012.
- ^ a b c Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL, Jones DW, Materson BJ, Oparil S, Wright JT, Roccella EJ, et al. (Joint National Committee on Prevention, National High Blood Pressure Education Program Coordinating Committee) (December 2003). "Seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure". Hypertension. 42 (6): 1206–1252. doi:10.1161/01.HYP.0000107251.49515.c2. PMID 14656957.
- ^ a b Perez MI, Musini VM (January 2008). "Pharmacological interventions for hypertensive emergencies". The Cochrane Database of Systematic Reviews. 2008 (1) CD003653. doi:10.1002/14651858.CD003653.pub3. PMC 6991936. PMID 18254026.
- ^ Harrison's principles of internal medicine (18th ed.). New York: McGraw-Hill. 2011. pp. 55–61. ISBN 978-0-07-174889-6.
- ^ a b Garovic VD, Dechend R, Easterling T, Karumanchi SA, McMurtry Baird S, Magee LA, Rana S, Vermunt JV, August P (February 2022). "Hypertension in Pregnancy: Diagnosis, Blood Pressure Goals, and Pharmacotherapy: A Scientific Statement From the American Heart Association". Hypertension. 79 (2): e21 – e41. doi:10.1161/HYP.0000000000000208. ISSN 1524-4563. PMC 9031058. PMID 34905954.
- ^ Al Khalaf SY, O'Reilly ÉJ, Barrett PM, B Leite DF, Pawley LC, McCarthy FP, Khashan AS (May 2021). "Impact of Chronic Hypertension and Antihypertensive Treatment on Adverse Perinatal Outcomes: Systematic Review and Meta-Analysis". Journal of the American Heart Association. 10 (9) e018494. doi:10.1161/JAHA.120.018494. PMC 8200761. PMID 33870708.
- ^ "Pregnancy complications increase the risk of heart attacks and stroke in women with high blood pressure". NIHR Evidence. 21 November 2023. doi:10.3310/nihrevidence_60660. S2CID 265356623.
- ^ Al Khalaf S, Chappell LC, Khashan AS, McCarthy FP, O'Reilly ÉJ (July 2023). "Association Between Chronic Hypertension and the Risk of 12 Cardiovascular Diseases Among Parous Women: The Role of Adverse Pregnancy Outcomes". Hypertension. 80 (7): 1427–1438. doi:10.1161/HYPERTENSIONAHA.122.20628. PMID 37170819.
- ^ Gibson P (30 July 2009). "Hypertension and Pregnancy". eMedicine Obstetrics and Gynecology. Medscape. Archived from the original on 24 July 2009. Retrieved 16 June 2009.
- ^ Ribeiro MM, Andrade A, Nunes I (27 January 2022). "Physical exercise in pregnancy: benefits, risks and prescription". Journal of Perinatal Medicine. 50 (1): 4–17. doi:10.1515/jpm-2021-0315. ISSN 1619-3997. PMID 34478617.
- ^ Martínez-Vizcaíno V, Sanabria-Martínez G, Fernández-Rodríguez R, Cavero-Redondo I, Pascual-Morena C, Álvarez-Bueno C, Martínez-Hortelano JA (February 2023). "Exercise during pregnancy for preventing gestational diabetes mellitus and hypertensive disorders: An umbrella review of randomised controlled trials and an updated meta-analysis". BJOG: An International Journal of Obstetrics and Gynaecology. 130 (3): 264–275. doi:10.1111/1471-0528.17304. ISSN 1471-0528. PMC 10092296. PMID 36156844.
- ^ Liu YH, Zhang YS, Chen JY, Wang ZJ, Liu YX, Li JQ, Xu XJ, Xie NJ, Lye S (May 2023). "Comparative effectiveness of prophylactic strategies for preeclampsia: a network meta-analysis of randomized controlled trials". American Journal of Obstetrics and Gynecology. 228 (5): 535–546. doi:10.1016/j.ajog.2022.10.014. ISSN 1097-6868. PMID 36283479.
- ^ Zhang J, Wang HP, Wang XX (July 2023). "Effects of aerobic exercise performed during pregnancy on hypertension and gestational diabetes: a systematic review and meta-analysis". The Journal of Sports Medicine and Physical Fitness. 63 (7): 852–863. doi:10.23736/S0022-4707.23.14578-6. ISSN 1827-1928. PMID 37067246.
- ^ a b Rodriguez-Cruz E, Ettinger LM (6 April 2010). "Hypertension". eMedicine Pediatrics: Cardiac Disease and Critical Care Medicine. Medscape. Archived from the original on 15 August 2009. Retrieved 16 June 2009.
- ^ a b c Dionne JM, Abitbol CL, Flynn JT (January 2012). "Hypertension in infancy: diagnosis, management and outcome". Pediatric Nephrology. 27 (1): 17–32. doi:10.1007/s00467-010-1755-z. PMID 21258818. S2CID 10698052.
- ^ Keaton JM, Kamali Z, Xie T, Vaez A, Williams A, Goleva SB, Ani A, Evangelou E, Hellwege JN, Yengo L, Young WJ, Traylor M, Giri A, Zheng Z, Zeng J (May 2024). "Genome-wide analysis in over 1 million individuals of European ancestry yields improved polygenic risk scores for blood pressure traits". Nature Genetics. 56 (5): 778–791. doi:10.1038/s41588-024-01714-w. ISSN 1061-4036. PMC 11096100. PMID 38689001.
- ^ Lifton RP, Gharavi AG, Geller DS (February 2001). "Molecular mechanisms of human hypertension". Cell. 104 (4): 545–556. doi:10.1016/S0092-8674(01)00241-0. PMID 11239411.
- ^ Kato N, Loh M, Takeuchi F, Verweij N, Wang X, Zhang W, et al. (November 2015). "Trans-ancestry genome-wide association study identifies 12 genetic loci influencing blood pressure and implicates a role for DNA methylation". Nature Genetics. 47 (11): 1282–1293. doi:10.1038/ng.3405. PMC 4719169. PMID 26390057.
- ^ Carrera-Bastos P, Fontes-Villalba M, O'Keefe JH, Lindeberg S, Cordain L (9 March 2011). "The western diet and lifestyle and diseases of civilization". Research Reports in Clinical Cardiology. 2: 15–35. doi:10.2147/RRCC.S16919. S2CID 3231706. Retrieved 9 February 2021.
- ^ Vasan RS, Beiser A, Seshadri S, Larson MG, Kannel WB, D'Agostino RB, Levy D (February 2002). "Residual lifetime risk for developing hypertension in middle-aged women and men: The Framingham Heart Study". JAMA. 287 (8): 1003–1010. doi:10.1001/jama.287.8.1003. PMID 11866648.
- ^ Filippini T, Malavolti M, Whelton PK, Naska A, Orsini N, Vinceti M (20 April 2021). "Blood Pressure Effects of Sodium Reduction: Dose–Response Meta-Analysis of Experimental Studies". Circulation. 143 (16): 1542–1567. doi:10.1161/CIRCULATIONAHA.120.050371. ISSN 0009-7322. PMC 8055199. PMID 33586450.
- ^ Hall ME, Cohen JB, Ard JD, Egan BM, Hall JE, Lavie CJ, Ma J, Ndumele CE, Schauer PR, Shimbo D (November 2021). "Weight-Loss Strategies for Prevention and Treatment of Hypertension: A Scientific Statement From the American Heart Association". Hypertension. 78 (5): e38 – e50. doi:10.1161/HYP.0000000000000202. ISSN 0194-911X. PMID 34538096.
- ^ Edwards JJ, Deenmamode AH, Griffiths M, Arnold O, Cooper NJ, Wiles JD, O'Driscoll JM (October 2023). "Exercise training and resting blood pressure: a large-scale pairwise and network meta-analysis of randomised controlled trials". British Journal of Sports Medicine. 57 (20): 1317–1326. doi:10.1136/bjsports-2022-106503. ISSN 1473-0480. PMID 37491419.
- ^ Lee KW, Loh HC, Ching SM, Devaraj NK, Hoo FK (29 May 2020). "Effects of Vegetarian Diets on Blood Pressure Lowering: A Systematic Review with Meta-Analysis and Trial Sequential Analysis". Nutrients. 12 (6): 1604. doi:10.3390/nu12061604. PMC 7352826. PMID 32486102.
- ^ Sriperumbuduri S, Welling P, Ruzicka M, Hundemer GL, Hiremath S (29 September 2023). "Potassium and Hypertension: A State-of-the-Art Review". American Journal of Hypertension. 37 (2): 91–100. doi:10.1093/ajh/hpad094. ISSN 0895-7061. PMID 37772757.
- ^ Cormick G, Ciapponi A, Cafferata ML, Cormick MS, Belizán JM (11 January 2022). Cochrane Hypertension Group (ed.). "Calcium supplementation for prevention of primary hypertension". Cochrane Database of Systematic Reviews. 2022 (1) CD010037. doi:10.1002/14651858.CD010037.pub4. PMC 8748265. PMID 35014026.
- ^ Di Federico S, Filippini T, Whelton PK, Cecchini M, Iamandii I, Boriani G, Vinceti M (October 2023). "Alcohol Intake and Blood Pressure Levels: A Dose-Response Meta-Analysis of Nonexperimental Cohort Studies". Hypertension. 80 (10): 1961–1969. doi:10.1161/HYPERTENSIONAHA.123.21224. ISSN 0194-911X. PMC 10510850. PMID 37522179.
- ^ Mesas AE, Leon-Muñoz LM, Rodriguez-Artalejo F, Lopez-Garcia E (October 2011). "The effect of coffee on blood pressure and cardiovascular disease in hypertensive individuals: a systematic review and meta-analysis". The American Journal of Clinical Nutrition. 94 (4): 1113–1126. doi:10.3945/ajcn.111.016667. PMID 21880846.
- ^ Vaidya A, Forman JP (November 2010). "Vitamin D and hypertension: current evidence and future directions". Hypertension. 56 (5): 774–779. doi:10.1161/HYPERTENSIONAHA.109.140160. PMID 20937970.
- ^ Narita K, Hoshide S, Kario K (November 2021). "Seasonal variation in blood pressure: current evidence and recommendations for hypertension management". Hypertension Research. 44 (11): 1363–1372. doi:10.1038/s41440-021-00732-z. ISSN 1348-4214. PMID 34489592.
- ^ Meng L, Chen D, Yang Y, Zheng Y, Hui R (May 2012). "Depression increases the risk of hypertension incidence: a meta-analysis of prospective cohort studies". Journal of Hypertension. 30 (5): 842–851. doi:10.1097/hjh.0b013e32835080b7. PMID 22343537. S2CID 32187480.
- ^ Hawkley LC, Cacioppo JT (October 2010). "Loneliness matters: a theoretical and empirical review of consequences and mechanisms". Annals of Behavioral Medicine. 40 (2): 218–227. doi:10.1007/s12160-010-9210-8. PMC 3874845. PMID 20652462.
- ^ Muñoz Aguilera E, Suvan J, Buti J, Czesnikiewicz-Guzik M, Barbosa Ribeiro A, Orlandi M, et al. (January 2020). Lembo G (ed.). "Periodontitis is associated with hypertension: a systematic review and meta-analysis". Cardiovascular Research. 116 (1): 28–39. doi:10.1093/cvr/cvz201. PMID 31549149.
- ^ Abhyankar LN, Jones MR, Guallar E, Navas-Acien A (April 2012). "Arsenic exposure and hypertension: a systematic review". Environmental Health Perspectives. 120 (4): 494–500. Bibcode:2012EnvHP.120..494A. doi:10.1289/ehp.1103988. PMC 3339454. PMID 22138666.
- ^ Yang BY, Qian Z, Howard SW, Vaughn MG, Fan SJ, Liu KK, Dong GH (April 2018). "Global association between ambient air pollution and blood pressure: A systematic review and meta-analysis". Environmental Pollution. 235: 576–588. Bibcode:2018EPoll.235..576Y. doi:10.1016/j.envpol.2018.01.001. PMID 29331891.
- ^ Sandoval-Plata G, Nakafero G, Chakravorty M, Morgan K, Abhishek A (1 July 2021). "Association between serum urate, gout and comorbidities: a case–control study using data from the UK Biobank". Rheumatology. 60 (7): 3243–3251. doi:10.1093/rheumatology/keaa773. ISSN 1462-0324. PMID 33313843.
- ^ Gill D, Cameron AC, Burgess S, Li X, Doherty DJ, Karhunen V, Abdul-Rahim AH, Taylor-Rowan M, Zuber V, Tsao PS, Klarin D, VA Million Veteran Program, Evangelou E, Elliott P, Damrauer SM (February 2021). "Urate, Blood Pressure, and Cardiovascular Disease: Evidence From Mendelian Randomization and Meta-Analysis of Clinical Trials". Hypertension. 77 (2): 383–392. doi:10.1161/HYPERTENSIONAHA.120.16547. ISSN 0194-911X. PMC 7803439. PMID 33356394.
- ^ McCormick N, O'Connor MJ, Yokose C, Merriman TR, Mount DB, Leong A, Choi HK (November 2021). "Assessing the Causal Relationships Between Insulin Resistance and Hyperuricemia and Gout Using Bidirectional Mendelian Randomization". Arthritis & Rheumatology. 73 (11): 2096–2104. doi:10.1002/art.41779. ISSN 2326-5205. PMC 8568618. PMID 33982892.
- ^ Quesada O, Claggett B, Rodriguez F, Cai J, Moncrieft AE, Garcia K, Del Rios Rivera M, Hanna DB, Daviglus ML, Talavera GA, Bairey Merz CN, Solomon SD, Cheng S, Bello NA (September 2021). "Associations of Insulin Resistance With Systolic and Diastolic Blood Pressure: A Study From the HCHS/SOL". Hypertension. 78 (3): 716–725. doi:10.1161/HYPERTENSIONAHA.120.16905. ISSN 0194-911X. PMC 8650976. PMID 34379440.
- ^ Lawlor DA, Smith GD (May 2005). "Early life determinants of adult blood pressure". Current Opinion in Nephrology and Hypertension. 14 (3): 259–264. doi:10.1097/01.mnh.0000165893.13620.2b. PMID 15821420. S2CID 10646150.
- ^ Dluhy RG, Williams GH (1998). "Endocrine hypertension". In Wilson JD, Foster DW, Kronenberg HM (eds.). Williams textbook of endocrinology (9th ed.). Philadelphia; Montreal: W.B. Saunders. pp. 729–749. ISBN 978-0-7216-6152-0.
- ^ Grossman E, Messerli FH (January 2012). "Drug-induced hypertension: an unappreciated cause of secondary hypertension". The American Journal of Medicine. 125 (1): 14–22. doi:10.1016/j.amjmed.2011.05.024. PMID 22195528.
- ^ Roerecke M, Tobe SW, Kaczorowski J, Bacon SL, Vafaei A, Hasan OS, Krishnan RJ, Raifu AO, Rehm J (June 2018). "Sex-Specific Associations Between Alcohol Consumption and Incidence of Hypertension: A Systematic Review and Meta-Analysis of Cohort Studies". Journal of the American Heart Association. 7 (13) e008202. doi:10.1161/JAHA.117.008202. PMC 6064910. PMID 29950485.
- ^ Conway J (April 1984). "Hemodynamic aspects of essential hypertension in humans". Physiological Reviews. 64 (2): 617–660. doi:10.1152/physrev.1984.64.2.617. PMID 6369352.
- ^ a b Palatini P, Julius S (June 2009). "The role of cardiac autonomic function in hypertension and cardiovascular disease". Current Hypertension Reports. 11 (3): 199–205. doi:10.1007/s11906-009-0035-4. PMID 19442329. S2CID 11320300.
- ^ Andersson OK, Lingman M, Himmelmann A, Sivertsson R, Widgren BR (2004). "Prediction of future hypertension by casual blood pressure or invasive hemodynamics? A 30-year follow-up study". Blood Pressure. 13 (6): 350–354. doi:10.1080/08037050410004819. PMID 15771219. S2CID 28992820.
- ^ Folkow B (April 1982). "Physiological aspects of primary hypertension". Physiological Reviews. 62 (2): 347–504. doi:10.1152/physrev.1982.62.2.347. PMID 6461865.
- ^ Struijker Boudier HA, le Noble JL, Messing MW, Huijberts MS, le Noble FA, van Essen H (December 1992). "The microcirculation and hypertension". Journal of Hypertension Supplement. 10 (7): S147–156. doi:10.1097/00004872-199212000-00016. PMID 1291649.
- ^ Schiffrin EL (February 1992). "Reactivity of small blood vessels in hypertension: relation with structural changes. State of the art lecture". Hypertension. 19 (2 Suppl): II1-9. doi:10.1161/01.HYP.19.2_Suppl.II1-a. PMID 1735561.
- ^ Safar ME, London GM (August 1987). "Arterial and venous compliance in sustained essential hypertension". Hypertension. 10 (2): 133–139. doi:10.1161/01.HYP.10.2.133. PMID 3301662.
- ^ Kim SH, Lim KR, Chun KJ (2022). "Higher heart rate variability as a predictor of atrial fibrillation in patients with hypertensione". Scientific Reports. 12 (1) 3702. Bibcode:2022NatSR..12.3702K. doi:10.1038/s41598-022-07783-3. PMC 8904557. PMID 35260686.
- ^ Steppan J, Barodka V, Berkowitz DE, Nyhan D (2 August 2011). "Vascular stiffness and increased pulse pressure in the aging cardiovascular system". Cardiology Research and Practice. 2011 263585. doi:10.4061/2011/263585. PMC 3154449. PMID 21845218.
- ^ Chobanian AV (August 2007). "Clinical practice. Isolated systolic hypertension in the elderly". The New England Journal of Medicine. 357 (8): 789–796. doi:10.1056/NEJMcp071137. PMID 17715411. S2CID 42515260.
- ^ Zieman SJ, Melenovsky V, Kass DA (May 2005). "Mechanisms, pathophysiology, and therapy of arterial stiffness". Arteriosclerosis, Thrombosis, and Vascular Biology. 25 (5): 932–943. doi:10.1161/01.ATV.0000160548.78317.29. PMID 15731494.
- ^ Navar LG (December 2010). "Counterpoint: Activation of the intrarenal renin-angiotensin system is the dominant contributor to systemic hypertension". Journal of Applied Physiology. 109 (6): 1998–2000, discussion 2015. doi:10.1152/japplphysiol.00182.2010a. PMC 3006411. PMID 21148349.
- ^ Esler M, Lambert E, Schlaich M (December 2010). "Point: Chronic activation of the sympathetic nervous system is the dominant contributor to systemic hypertension". Journal of Applied Physiology. 109 (6): 1996–1998, discussion 2016. doi:10.1152/japplphysiol.00182.2010. PMID 20185633. S2CID 7685157.
- ^ Versari D, Daghini E, Virdis A, Ghiadoni L, Taddei S (June 2009). "Endothelium-dependent contractions and endothelial dysfunction in human hypertension". British Journal of Pharmacology. 157 (4): 527–536. doi:10.1111/j.1476-5381.2009.00240.x. PMC 2707964. PMID 19630832.
- ^ Marchesi C, Paradis P, Schiffrin EL (July 2008). "Role of the renin-angiotensin system in vascular inflammation". Trends in Pharmacological Sciences. 29 (7): 367–374. doi:10.1016/j.tips.2008.05.003. PMID 18579222.
- ^ Gooch JL, Sharma AC (September 2014). "Targeting the immune system to treat hypertension: where are we?". Current Opinion in Nephrology and Hypertension. 23 (5): 473–479. doi:10.1097/MNH.0000000000000052. PMID 25036747. S2CID 13383731.
- ^ Adrogué HJ, Madias NE (May 2007). "Sodium and potassium in the pathogenesis of hypertension". The New England Journal of Medicine. 356 (19): 1966–1978. doi:10.1056/NEJMra064486. PMID 17494929. S2CID 22345731.
- ^ Perez V, Chang ET (November 2014). "Sodium-to-potassium ratio and blood pressure, hypertension, and related factors". Advances in Nutrition. 5 (6): 712–741. doi:10.3945/an.114.006783. PMC 4224208. PMID 25398734.
- ^ Williams GH, Hollenberg NK (November 1985). "Non-modulating essential hypertension: a subset particularly responsive to converting enzyme inhibitors". Journal of Hypertension Supplement. 3 (2): S81 – S87. PMID 3003304.
- ^ Harrison TR, Jameson JL, Fauci AS, Kasper DL, Hauser SL, Longo DL, Loscalzo J, eds. (2018). Harrison's Principles of Internal Medicine (20th ed.). McGraw-Hill Education. p. 1896. ISBN 978-1-259-64404-7.
When plasma renin activity (PRA) is plotted against 24-h sodium excretion, ~10–15% of hypertensive patients have high PRA and 25% have low PRA. High-renin patients may have a vasoconstrictor form of hypertension, whereas low-renin patients may have volume-dependent hypertension.
- ^ a b c d e f g h Viera AJ (July 2017). "Screening for Hypertension and Lowering Blood Pressure for Prevention of Cardiovascular Disease Events". The Medical Clinics of North America (Review). 101 (4): 701–712. doi:10.1016/j.mcna.2017.03.003. PMID 28577621.
- ^ a b c Vischer AS, Burkard T (2017). "Principles of Blood Pressure Measurement – Current Techniques, Office vs Ambulatory Blood Pressure Measurement". Hypertension: From basic research to clinical practice (Review). Advances in Experimental Medicine and Biology. Vol. 956. pp. 85–96. doi:10.1007/5584_2016_49. ISBN 978-3-319-44250-1. PMID 27417699.
- ^ a b Siu AL (November 2015). "Screening for high blood pressure in adults: U.S. Preventive Services Task Force recommendation statement". Annals of Internal Medicine. 163 (10): 778–786. doi:10.7326/m15-2223. PMID 26458123.
- ^ a b National Clinical Guidance Centre (August 2011). "7 Diagnosis of Hypertension, 7.5 Link from evidence to recommendations" (PDF). Hypertension (NICE CG 127). National Institute for Health and Clinical Excellence. p. 102. Archived from the original (PDF) on 23 July 2013. Retrieved 22 December 2011.
- ^ Franklin SS, Wilkinson IB, McEniery CM (February 2012). "Unusual hypertensive phenotypes: what is their significance?". Hypertension. 59 (2): 173–178. doi:10.1161/HYPERTENSIONAHA.111.182956. PMID 22184330.
- ^ Kario K (June 2009). "Orthostatic hypertension: a measure of blood pressure variation for predicting cardiovascular risk". Circulation Journal. 73 (6): 1002–1007. doi:10.1253/circj.cj-09-0286. PMID 19430163.
- ^ a b Guideline for the pharmacological treatment of hypertension in adults (PDF). World Health Organization (WHO). 2021. ISBN 9789240033993. Retrieved 26 May 2024.
- ^ Luma GB, Spiotta RT (May 2006). "Hypertension in children and adolescents". American Family Physician. 73 (9): 1558–1568. PMID 16719248.
- ^ a b c McEvoy JW, McCarthy CP, Bruno RM, Brouwers S, Canavan MD, et al. (30 August 2024). "2024 ESC Guidelines for the management of elevated blood pressure and hypertension: Developed by the task force on the management of elevated blood pressure and hypertension of the European Society of Cardiology (ESC) and endorsed by the European Society of Endocrinology (ESE) and the European Stroke Organisation (ESO)". European Heart Journal. 45 (38): 3912–4018. doi:10.1093/eurheartj/ehae178. ISSN 0195-668X. PMID 39210715.
- ^ Whelton PK, Carey RM, Mancia G, Kreutz R, Bundy JD, Williams B (14 September 2022). "Harmonization of the American College of Cardiology/American Heart Association and European Society of Cardiology/European Society of Hypertension Blood Pressure/Hypertension Guidelines". European Heart Journal. 43 (35): 3302–3311. doi:10.1093/eurheartj/ehac432. ISSN 0195-668X. PMC 9470378. PMID 36100239.
- ^ a b c d National High Blood Pressure Education Program Working Group on High Blood Pressure in Children Adolescents (August 2004). "The fourth report on the diagnosis, evaluation, and treatment of high blood pressure in children and adolescents". Pediatrics. 114 (2 Suppl 4th Report): 555–576. doi:10.1542/peds.114.2.S2.555 (inactive 3 July 2025). hdl:2027/uc1.c095473177. PMID 15286277.
{{cite journal}}: CS1 maint: DOI inactive as of July 2025 (link) - ^ a b Williams B, Poulter NR, Brown MJ, Davis M, McInnes GT, Potter JF, Sever PS, McG Thom S (March 2004). "Guidelines for management of hypertension: report of the fourth working party of the British Hypertension Society, 2004-BHS IV". Journal of Human Hypertension. 18 (3): 139–185. doi:10.1038/sj.jhh.1001683. PMID 14973512.
- ^ a b "Evidence-based policy for salt reduction is needed". Lancet. 388 (10043): 438. July 2016. doi:10.1016/S0140-6736(16)31205-3. PMID 27507743. S2CID 205982690.
- ^ Mente A, O'Donnell M, Rangarajan S, Dagenais G, Lear S, McQueen M, Diaz R, Avezum A, Lopez-Jaramillo P, Lanas F, Li W, Lu Y, Yi S, Rensheng L, Iqbal R, Mony P, Yusuf R, Yusoff K, Szuba A, Oguz A, Rosengren A, Bahonar A, Yusufali A, Schutte AE, Chifamba J, Mann JF, Anand SS, Teo K, Yusuf S (July 2016). "Associations of urinary sodium excretion with cardiovascular events in individuals with and without hypertension: a pooled analysis of data from four studies". Lancet. 388 (10043): 464–475. doi:10.1016/S0140-6736(16)30467-6. hdl:10379/16625. PMID 27216139. S2CID 44581906.
The results showed that cardiovascular disease and death are increased with low sodium intake (compared with moderate intake) irrespective of hypertension status, whereas there is a higher risk of cardiovascular disease and death only in individuals with hypertension consuming more than 6 g of sodium per day (representing only 10% of the population studied)
- ^ Perk J, De Backer G, Gohlke H, Graham I, Reiner Z, Verschuren M, Albus C, Benlian P, Boysen G, Cifkova R, Deaton C, Ebrahim S, Fisher M, Germano G, Hobbs R, Hoes A, Karadeniz S, Mezzani A, Prescott E, Ryden L, Scherer M, Syvänne M, Scholte op Reimer WJ, Vrints C, Wood D, Zamorano JL, Zannad F (July 2012). "European Guidelines on cardiovascular disease prevention in clinical practice (version 2012). The Fifth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of nine societies and by invited experts)". European Heart Journal. 33 (13): 1635–1701. doi:10.1093/eurheartj/ehs092. PMID 22555213.
- ^ Chiolero A, Bovet P, Paradis G (March 2013). "Screening for elevated blood pressure in children and adolescents: a critical appraisal". JAMA Pediatrics. 167 (3): 266–273. doi:10.1001/jamapediatrics.2013.438. PMID 23303490.
- ^ Daniels SR, Gidding SS (March 2013). "Blood pressure screening in children and adolescents: is the glass half empty or more than half full?". JAMA Pediatrics. 167 (3): 302–304. doi:10.1001/jamapediatrics.2013.439. PMID 23303514.
- ^ a b Schmidt BM, Durao S, Toews I, Bavuma CM, Hohlfeld A, Nury E, et al. (Cochrane Hypertension Group) (May 2020). "Screening strategies for hypertension". The Cochrane Database of Systematic Reviews. 2020 (5) CD013212. doi:10.1002/14651858.CD013212.pub2. PMC 7203601. PMID 32378196.
- ^ "Expert panel on integrated guidelines for cardiovascular health and risk reduction in children and adolescents: summary report". Pediatrics. 128 (Suppl 5): S213 – S256. December 2011. doi:10.1542/peds.2009-2107C. PMC 4536582. PMID 22084329.
- ^ "Hypertension – Clinical Preventive Service Recommendation". AAFP. Archived from the original on 1 November 2014. Retrieved 13 October 2013.
- ^ Moyer VA (November 2013). "Screening for primary hypertension in children and adolescents: U.S. Preventive Services Task Force recommendation statement". Annals of Internal Medicine. 159 (9): 613–619. doi:10.7326/0003-4819-159-9-201311050-00725. PMID 24097285. S2CID 20193715.
- ^ "Document | United States Preventive Services Taskforce". uspreventiveservicestaskforce.org. Archived from the original on 22 May 2020. Retrieved 22 April 2020.
- ^ Krist AH, Davidson KW, Mangione CM, Cabana M, Caughey AB, Davis EM, et al. (April 2021). "Screening for Hypertension in Adults: US Preventive Services Task Force Reaffirmation Recommendation Statement". JAMA. 325 (16): 1650–1656. doi:10.1001/jama.2021.4987. PMID 33904861. S2CID 233409679.
- ^ Law M, Wald N, Morris J (2003). "Lowering blood pressure to prevent myocardial infarction and stroke: a new preventive strategy". Health Technology Assessment. 7 (31): 1–94. doi:10.3310/hta7310. PMID 14604498.
- ^ a b Daskalopoulou SS, Rabi DM, Zarnke KB, Dasgupta K, Nerenberg K, Cloutier L, et al. (May 2015). "The 2015 Canadian Hypertension Education Program recommendations for blood pressure measurement, diagnosis, assessment of risk, prevention, and treatment of hypertension". The Canadian Journal of Cardiology. 31 (5): 549–568. doi:10.1016/j.cjca.2015.02.016. PMID 25936483.
- ^ a b "Hypertension: Recommendations, Guidance and guidelines". NICE. Archived from the original on 3 October 2006. Retrieved 4 August 2015.
- ^ a b Arguedas JA, Leiva V, Wright JM (October 2013). "Blood pressure targets for hypertension in people with diabetes mellitus". The Cochrane Database of Systematic Reviews (10) CD008277. doi:10.1002/14651858.cd008277.pub2. PMC 11365096. PMID 24170669.
- ^ Saiz LC, Gorricho J, Garjón J, Celaya MC, Erviti J, Leache L (November 2022). "Blood pressure targets for the treatment of people with hypertension and cardiovascular disease". The Cochrane Database of Systematic Reviews. 2022 (11) CD010315. doi:10.1002/14651858.CD010315.pub5. PMC 9673465. PMID 36398903.
- ^ a b Arguedas JA, Leiva V, Wright JM (December 2020). "Blood pressure targets in adults with hypertension". The Cochrane Database of Systematic Reviews. 2020 (12) CD004349. doi:10.1002/14651858.CD004349.pub3. PMC 8094587. PMID 33332584.
- ^ a b Qaseem A, Wilt TJ, Rich R, Humphrey LL, Frost J, Forciea MA (March 2017). "Pharmacologic Treatment of Hypertension in Adults Aged 60 Years or Older to Higher Versus Lower Blood Pressure Targets: A Clinical Practice Guideline From the American College of Physicians and the American Academy of Family Physicians". Annals of Internal Medicine. 166 (6): 430–437. doi:10.7326/M16-1785. PMID 28135725.
- ^ Wright JT, Fine LJ, Lackland DT, Ogedegbe G, Dennison Himmelfarb CR (April 2014). "Evidence supporting a systolic blood pressure goal of less than 150 mm Hg in patients aged 60 years or older: the minority view". Annals of Internal Medicine. 160 (7): 499–503. doi:10.7326/m13-2981. PMID 24424788.
- ^ Passarella P, Kiseleva TA, Valeeva FV, Gosmanov AR (1 August 2018). "Hypertension Management in Diabetes: 2018 Update". Diabetes Spectrum. 31 (3): 218–224. doi:10.2337/ds17-0085. ISSN 1040-9165. PMC 6092891. PMID 30140137.
- ^ Cheung AK, Chang TI, Cushman WC, Furth SL, Hou FF, Ix JH, Knoll GA, Muntner P, Pecoits-Filho R, Sarnak MJ, Tobe SW, Tomson CR, Mann JF (March 2021). "KDIGO 2021 Clinical Practice Guideline for the Management of Blood Pressure in Chronic Kidney Disease". Kidney International. 99 (3): S1 – S87. doi:10.1016/j.kint.2020.11.003. PMID 33637192.
- ^ Brunström M, Carlberg B (January 2016). "Lower blood pressure targets: to whom do they apply?". Lancet. 387 (10017): 405–406. doi:10.1016/S0140-6736(15)00816-8. PMID 26559745. S2CID 44282689.
- ^ Xie X, Atkins E, Lv J, Rodgers A (June 2016). "Intensive blood pressure lowering – Authors' reply". Lancet. 387 (10035): 2291. doi:10.1016/S0140-6736(16)30366-X. PMID 27302266.
- ^ a b Jones DW, Ferdinand KC, Taler SJ, Johnson HM, Shimbo D, Abdalla M, et al. (16 September 2025). "2025 AHA/ACC/AANP/AAPA/ABC/ACCP/ACPM/AGS/AMA/ASPC/NMA/PCNA/SGIM guideline for the prevention, detection, evaluation and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines". Circulation. 152 (11): e114 – e218. doi:10.1161/CIR.0000000000001356. PMID 40811497.
- ^ a b Go AS, Bauman MA, Coleman King SM, Fonarow GC, Lawrence W, Williams KA, Sanchez E (April 2014). "An effective approach to high blood pressure control: a science advisory from the American Heart Association, the American College of Cardiology, and the Centers for Disease Control and Prevention". Hypertension. 63 (4): 878–885. doi:10.1161/HYP.0000000000000003. PMC 10280688. PMID 24243703.
- ^ a b Semlitsch T, Krenn C, Jeitler K, Berghold A, Horvath K, Siebenhofer A (February 2021). "Long-term effects of weight-reducing diets in people with hypertension". The Cochrane Database of Systematic Reviews. 2021 (2) CD008274. doi:10.1002/14651858.CD008274.pub4. PMC 8093137. PMID 33555049.
- ^ He FJ, Li J, Macgregor GA (April 2013). "Effect of longer-term modest salt reduction on blood pressure". The Cochrane Database of Systematic Reviews (Systematic Review & Meta-Analysis). 30 (4) CD004937. doi:10.1002/14651858.CD004937.pub2. PMC 11537250. PMID 23633321. S2CID 23522004.
- ^ Huang L, Trieu K, Yoshimura S, Neal B, Woodward M, Campbell NR, et al. (February 2020). "Effect of dose and duration of reduction in dietary sodium on blood pressure levels: systematic review and meta-analysis of randomised trials". The BMJ. 368: m315. doi:10.1136/bmj.m315. PMC 7190039. PMID 32094151.
- ^ Sacks FM, Svetkey LP, Vollmer WM, Appel LJ, Bray GA, Harsha D, et al. (January 2001). "Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) diet. DASH-Sodium Collaborative Research Group". The New England Journal of Medicine. 344 (1): 3–10. doi:10.1056/NEJM200101043440101. PMID 11136953.
- ^ Sukhato K, Akksilp K, Dellow A, Vathesatogkit P, Anothaisintawee T (December 2020). "Efficacy of different dietary patterns on lowering of blood pressure level: an umbrella review". The American Journal of Clinical Nutrition. 112 (6): 1584–1598. doi:10.1093/ajcn/nqaa252. PMID 33022695.
- ^ Joshi S, Ettinger L, Liebman SE (2020). "Plant-Based Diets and Hypertension". American Journal of Lifestyle Medicine. 14 (4): 397–405. doi:10.1177/1559827619875411. PMC 7692016. PMID 33281520.
- ^ Charchar FJ, Prestes PR, Mills C (2024). "Lifestyle management of hypertension: International Society of Hypertension position paper endorsed by the World Hypertension League and European Society of Hypertension". Journal of Hypertension. 42 (1): 23–49. doi:10.1097/HJH.0000000000003563. PMC 10713007. PMID 37712135.
- ^ Jama, HA, Snelson M, Schutte AE (2024). "Recommendations for the Use of Dietary Fiber to Improve Blood Pressure Control". Hypertension. 81 (7): 1450–1459. doi:10.1161/HYPERTENSIONAHA.123.22575. PMID 38586958.
- ^ Aburto NJ, Hanson S, Gutierrez H, Hooper L, Elliott P, Cappuccio FP (April 2013). "Effect of increased potassium intake on cardiovascular risk factors and disease: systematic review and meta-analyses". The BMJ. 346 f1378. doi:10.1136/bmj.f1378. PMC 4816263. PMID 23558164.
- ^ Stone MS, Martyn L, Weaver CM (July 2016). "Potassium Intake, Bioavailability, Hypertension, and Glucose Control". Nutrients. 8 (7): 444. doi:10.3390/nu8070444. PMC 4963920. PMID 27455317.
- ^ "Scientific Report of the 2015 Dietary Guidelines Advisory Committee". Archived from the original on 26 April 2017. Retrieved 26 April 2017.
- ^ Raebel MA (June 2012). "Hyperkalemia associated with use of angiotensin-converting enzyme inhibitors and angiotensin receptor blockers". Cardiovascular Therapeutics. 30 (3): e156–166. doi:10.1111/j.1755-5922.2010.00258.x. PMID 21883995.
- ^ a b Brook RD, Appel LJ, Rubenfire M, Ogedegbe G, Bisognano JD, Elliott WJ, Fuchs FD, Hughes JW, Lackland DT, Staffileno BA, Townsend RR, Rajagopalan S (June 2013). "Beyond medications and diet: alternative approaches to lowering blood pressure: a scientific statement from the american heart association". Hypertension. 61 (6): 1360–1383. doi:10.1161/HYP.0b013e318293645f. PMID 23608661.
- ^ Lou Y, Ye G, Liu W, Lv Z, Jia Y, Li C, Zhang Y (February 2021). "Walking for hypertension". The Cochrane Database of Systematic Reviews (Systematic Review & Meta-Analysis). 2013 (3): CD004937. doi:10.1002/14651858.CD008823.pub2. PMC 11537250. PMID 23633321.
- ^ Nagele E, Jeitler K, Horvath K, Semlitsch T, Posch N, Herrmann KH, Grouven U, Hermanns T, Hemkens LG, Siebenhofer A (October 2014). "Clinical effectiveness of stress-reduction techniques in patients with hypertension: systematic review and meta-analysis". Journal of Hypertension. 32 (10): 1936–1944, discussion 1944. doi:10.1097/HJH.0000000000000298. PMID 25084308. S2CID 20098894.
- ^ Dickinson HO, Campbell F, Beyer FR, Nicolson DJ, Cook JV, Ford GA, Mason JM (January 2008). "Relaxation therapies for the management of primary hypertension in adults". The Cochrane Database of Systematic Reviews (1) CD004935. doi:10.1002/14651858.CD004935.pub2. PMID 18254065.
- ^ a b Glynn LG, Murphy AW, Smith SM, Schroeder K, Fahey T (March 2010). "Interventions used to improve control of blood pressure in patients with hypertension" (PDF). The Cochrane Database of Systematic Reviews (3) CD005182. doi:10.1002/14651858.cd005182.pub4. hdl:10344/9179. PMID 20238338. Archived (PDF) from the original on 12 April 2019. Retrieved 11 February 2019.
- ^ Wright JM, Musini VM, Gill R (April 2018). "First-line drugs for hypertension". The Cochrane Database of Systematic Reviews. 2018 (4) CD001841. doi:10.1002/14651858.CD001841.pub3. PMC 6513559. PMID 29667175.
- ^ Chen JM, Heran BS, Wright JM (October 2009). "Blood pressure lowering efficacy of diuretics as second-line therapy for primary hypertension". The Cochrane Database of Systematic Reviews. 2010 (4) CD007187. doi:10.1002/14651858.CD007187.pub2. PMC 12410055. PMID 19821398. S2CID 73993182.
- ^ Garjón J, Saiz LC, Azparren A, Gaminde I, Ariz MJ, Erviti J, et al. (Cochrane Hypertension Group) (February 2020). "First-line combination therapy versus first-line monotherapy for primary hypertension". The Cochrane Database of Systematic Reviews. 2 (2) CD010316. doi:10.1002/14651858.CD010316.pub3. PMC 7002970. PMID 32026465.
- ^ Reeve E, Jordan V, Thompson W, Sawan M, Todd A, Gammie TM, et al. (Cochrane Hypertension Group) (June 2020). "Withdrawal of antihypertensive drugs in older people". The Cochrane Database of Systematic Reviews. 2020 (6) CD012572. doi:10.1002/14651858.CD012572.pub2. PMC 7387859. PMID 32519776.
- ^ Wiysonge CS, Bradley HA, Volmink J, Mayosi BM, Opie LH (January 2017). "Beta-blockers for hypertension". The Cochrane Database of Systematic Reviews. 1 (1) CD002003. doi:10.1002/14651858.CD002003.pub5. PMC 5369873. PMID 28107561.
- ^ Chaturvedi S, Lipszyc DH, Licht C, Craig JC, Parekh R, et al. (Cochrane Hypertension Group) (February 2014). "Pharmacological interventions for hypertension in children". The Cochrane Database of Systematic Reviews (2) CD008117. doi:10.1002/14651858.CD008117.pub2. PMC 11056235. PMID 24488616.
- ^ Giacona JM, Kositanurit W, Vongpatanasin W (April 2024). "Management of Resistant Hypertension-An Update". JAMA Intern Med. 184 (4): 433–434. doi:10.1001/jamainternmed.2023.8555. PMID 38372970.
- ^ McEvoy JW, McCarthy CP, Bruno RM, Brouwers S, Canavan MD, Ceconi C, Christodorescu RM, Daskalopoulou SS, Ferro CJ, Gerdts E, Hanssen H, Harris J, Lauder L, McManus RJ, Molloy GJ (7 October 2024). "2024 ESC Guidelines for the management of elevated blood pressure and hypertension". European Heart Journal. 45 (38): 3912–4018. doi:10.1093/eurheartj/ehae178. ISSN 0195-668X.
- ^ Santschi V, Chiolero A, Burnier M (November 2009). "Electronic monitors of drug adherence: tools to make rational therapeutic decisions". Journal of Hypertension. 27 (11): 2294–2295, author reply 2295. doi:10.1097/hjh.0b013e328332a501. PMID 20724871.
- ^ Sarwar MS, Islam MS, Al Baker SM, Hasnat A (May 2013). "Resistant hypertension: underlying causes and treatment". Drug Research. 63 (5): 217–223. doi:10.1055/s-0033-1337930. PMID 23526242. S2CID 8247941.
- ^ Young WF (February 2019). "Diagnosis and treatment of primary aldosteronism: practical clinical perspectives". Journal of Internal Medicine. 285 (2): 126–148. doi:10.1111/joim.12831. PMID 30255616. S2CID 52824356.
- ^ Zubcevic J, Waki H, Raizada MK, Paton JF (June 2011). "Autonomic-immune-vascular interaction: an emerging concept for neurogenic hypertension". Hypertension. 57 (6): 1026–1033. doi:10.1161/HYPERTENSIONAHA.111.169748. PMC 3105900. PMID 21536990.
- ^ Wallbach M, Koziolek MJ (September 2018). "Baroreceptors in the carotid and hypertension-systematic review and meta-analysis of the effects of baroreflex activation therapy on blood pressure". Nephrology, Dialysis, Transplantation. 33 (9): 1485–1493. doi:10.1093/ndt/gfx279. PMID 29136223.
- ^ a b Acelajado MC, Hughes ZH, Oparil S, Calhoun DA (March 2019). "Treatment of resistant and refractory hypertension". Circulation Research. 124 (7): 1061–1070. doi:10.1161/CIRCRESAHA.118.312156. PMC 6469348. PMID 30920924.
- ^ Dudenbostel T, Siddiqui M, Oparil S, Calhoun DA (June 2016). "Refractory hypertension: A novel phenotype of antihypertensive treatment failure". Hypertension. 67 (6): 1085–1092. doi:10.1161/HYPERTENSIONAHA.116.06587. PMC 5425297. PMID 27091893.
- ^ "Blood Pressure". World Health Organization. Archived from the original on 18 April 2017. Retrieved 22 April 2017.
- ^ "WHO Disease and injury country estimates". World Health Organization. 2009. Archived from the original on 11 November 2009. Retrieved 11 November 2009.
- ^ Leng B, Jin Y, Li G, Chen L, Jin N (February 2015). "Socioeconomic status and hypertension: a meta-analysis". Journal of Hypertension. 33 (2): 221–229. doi:10.1097/HJH.0000000000000428. ISSN 0263-6352. PMID 25479029.
- ^ Petrie JR, Guzik TJ, Touyz RM (May 2018). "Diabetes, Hypertension, and Cardiovascular Disease: Clinical Insights and Vascular Mechanisms". The Canadian Journal of Cardiology. 34 (5): 575–584. doi:10.1016/j.cjca.2017.12.005. PMC 5953551. PMID 29459239.
- ^ Burt VL, Whelton P, Roccella EJ, Brown C, Cutler JA, Higgins M, Horan MJ, Labarthe D (March 1995). "Prevalence of hypertension in the US adult population. Results from the Third National Health and Nutrition Examination Survey, 1988–1991". Hypertension. 25 (3): 305–313. doi:10.1161/01.HYP.25.3.305. PMID 7875754. S2CID 23660820.
- ^ a b Burt VL, Cutler JA, Higgins M, Horan MJ, Labarthe D, Whelton P, Brown C, Roccella EJ (July 1995). "Trends in the prevalence, awareness, treatment, and control of hypertension in the adult US population. Data from the health examination surveys, 1960 to 1991". Hypertension. 26 (1): 60–69. doi:10.1161/01.HYP.26.1.60. PMID 7607734.
- ^ Ostchega Y, Dillon CF, Hughes JP, Carroll M, Yoon S (July 2007). "Trends in hypertension prevalence, awareness, treatment, and control in older U.S. adults: data from the National Health and Nutrition Examination Survey 1988 to 2004". Journal of the American Geriatrics Society. 55 (7): 1056–1065. doi:10.1111/j.1532-5415.2007.01215.x. PMID 17608879. S2CID 27522876.
- ^ a b c Lloyd-Jones D, Adams RJ, Brown TM, Carnethon M, Dai S, De Simone G, et al. (February 2010). "Heart disease and stroke statistics – 2010 update: a report from the American Heart Association". Circulation. 121 (7): e46 – e215. doi:10.1161/CIRCULATIONAHA.109.192667. PMID 20019324.
- ^ Cooper RS, Forrester TE, Plange-Rhule J, Bovet P, Lambert EV, Dugas LR, Cargill KE, Durazo-Arvizu RA, Shoham DA, Tong L, Cao G, Luke A (2015). "Elevated hypertension risk for African-origin populations in biracial societies: Modeling the Epidemiologic Transition Study". Journal of Hypertension. 33 (3): 473–80, discussion 480-1. doi:10.1097/HJH.0000000000000429. PMC 4476314. PMID 25426566.
- ^ Frohlich ED (October 2011). "Epidemiological issues are not simply black and white". Hypertension. 58 (4): 546–547. doi:10.1161/HYPERTENSIONAHA.111.178541. PMID 21911712.
- ^ Falkner B (July 2010). "Hypertension in children and adolescents: epidemiology and natural history". Pediatric Nephrology. 25 (7): 1219–1224. doi:10.1007/s00467-009-1200-3. PMC 2874036. PMID 19421783.
- ^ Luma GB, Spiotta RT (May 2006). "Hypertension in children and adolescents". American Family Physician. 73 (9): 1558–1568. PMID 16719248. Archived from the original on 26 September 2007.
- ^ "Global health risks: mortality and burden of disease attributable to selected major risks" (PDF). World Health Organization. 2009. Archived (PDF) from the original on 14 February 2012. Retrieved 10 February 2012.
- ^ Lewington S, Clarke R, Qizilbash N, Peto R, Collins R (December 2002). "Age-specific relevance of usual blood pressure to vascular mortality: a meta-analysis of individual data for one million adults in 61 prospective studies". Lancet. 360 (9349): 1903–1913. doi:10.1016/S0140-6736(02)11911-8. PMID 12493255. S2CID 54363452.
- ^ Singer DR, Kite A (June 2008). "Management of hypertension in peripheral arterial disease: does the choice of drugs matter?". European Journal of Vascular and Endovascular Surgery. 35 (6): 701–708. doi:10.1016/j.ejvs.2008.01.007. PMID 18375152.
- ^ a b c d e f g h Esunge PM (October 1991). "From blood pressure to hypertension: the history of research". Journal of the Royal Society of Medicine. 84 (10): 621. doi:10.1177/014107689108401019. PMC 1295564. PMID 1744849.
- ^ a b Kotchen TA (October 2011). "Historical trends and milestones in hypertension research: a model of the process of translational research". Hypertension. 58 (4): 522–38. doi:10.1161/HYPERTENSIONAHA.111.177766. PMID 21859967.
- ^ Postel-Vinay N, ed. (1996). A century of arterial hypertension 1896–1996. Chichester: Wiley. p. 213. ISBN 978-0-471-96788-0.
- ^ Heydari M, Dalfardi B, Golzari SE, Habibi H, Zarshenas MM (July 2014). "The medieval origins of the concept of hypertension". Heart Views. 15 (3): 96–98. doi:10.4103/1995-705X.144807. PMC 4268622. PMID 25538828.
- ^ Emtiazy M, Choopani R, Khodadoost M, Tansaz M, Dehghan S, Ghahremani Z (2014). "Avicenna's doctrine about arterial hypertension". Acta medico-historica Adriatica. 12 (1): 157–162. PMID 25310615.
- ^ Swales JD, ed. (1995). Manual of hypertension. Oxford: Blackwell Science. p. xiii. ISBN 978-0-86542-861-4.
- ^ Wilking SV (16 December 1988). "Determinants of Isolated Systolic Hypertension". JAMA: The Journal of the American Medical Association. 260 (23): 3451–3455. doi:10.1001/jama.1988.03410230069030. ISSN 0098-7484. PMID 3210285.
- ^ "1993 guidelines for the management of mild hypertension: memorandum from a WHO/ISH meeting". Bulletin of the World Health Organization. 71 (5): 503–517. 1993. ISSN 0042-9686. PMC 2393474. PMID 8261554.
- ^ a b Dustan HP, Roccella EJ, Garrison HH (September 1996). "Controlling hypertension. A research success story". Archives of Internal Medicine. 156 (17): 1926–1935. doi:10.1001/archinte.156.17.1926. PMID 8823146.
- ^ Lyons HH, Hoobler SW (February 1948). "Experiences with tetraethylammonium chloride in hypertension". Journal of the American Medical Association. 136 (9): 608–613. doi:10.1001/jama.1948.02890260016005. PMID 18899127.
- ^ Bakris GL, Frohlich ED (December 1989). "The evolution of antihypertensive therapy: an overview of four decades of experience". Journal of the American College of Cardiology. 14 (7): 1595–1608. doi:10.1016/0735-1097(89)90002-8. PMID 2685075.
- ^ Novello FC, Sprague JM (1957). "Benzothiadiazine dioxides as novel diuretics". J. Am. Chem. Soc. 79 (8): 2028–2029. Bibcode:1957JAChS..79.2028N. doi:10.1021/ja01565a079.
- ^ a b Chockalingam A (May 2007). "Impact of World Hypertension Day". The Canadian Journal of Cardiology. 23 (7): 517–519. doi:10.1016/S0828-282X(07)70795-X. PMC 2650754. PMID 17534457.
- ^ Chockalingam A (June 2008). "World Hypertension Day and global awareness". The Canadian Journal of Cardiology. 24 (6): 441–444. doi:10.1016/S0828-282X(08)70617-2. PMC 2643187. PMID 18548140.
- ^ Alcocer L, Cueto L (June 2008). "Hypertension, a health economics perspective". Therapeutic Advances in Cardiovascular Disease. 2 (3): 147–155. doi:10.1177/1753944708090572. PMID 19124418. S2CID 31053059.
- ^ Elliott WJ (October 2003). "The economic impact of hypertension". Journal of Clinical Hypertension. 5 (3 Suppl 2): 3–13. doi:10.1111/j.1524-6175.2003.02463.x. PMC 8099256. PMID 12826765. S2CID 26799038.
- ^ Coca A (2008). "Economic benefits of treating high-risk hypertension with angiotensin II receptor antagonists (blockers)". Clinical Drug Investigation. 28 (4): 211–220. doi:10.2165/00044011-200828040-00002. PMID 18345711. S2CID 8294060.
- ^ Global report on hypertension 2025: high stakes – turning evidence into action. Geneva: World Health Organization (WHO). 23 September 2025. ISBN 978-92-4-011556-9.
- ^ Taylor SS, Sparkes AH, Briscoe K, Carter J, Sala SC, Jepson RE, Reynolds BS, Scansen BA (March 2017). "ISFM Consensus Guidelines on the Diagnosis and Management of Hypertension in Cats". Journal of Feline Medicine and Surgery. 19 (3): 288–303. doi:10.1177/1098612X17693500. PMC 11119534. PMID 28245741.
- ^ a b Acierno MJ, Brown S, Coleman AE, Jepson RE, Papich M, Stepien RL, Syme HM (November 2018). "ACVIM consensus statement: Guidelines for the identification, evaluation, and management of systemic hypertension in dogs and cats". Journal of Veterinary Internal Medicine. 32 (6): 1803–1822. doi:10.1111/jvim.15331. PMC 6271319. PMID 30353952.
Further reading
[edit]- 2025 AHA/ACC guideline: Jones DW, Ferdinand KC, Taler SJ, Johnson HM, Shimbo D, Abdalla M, et al. (16 September 2025). "2025 AHA/ACC/AANP/AAPA/ABC/ACCP/ACPM/AGS/AMA/ASPC/NMA/PCNA/SGIM guideline for the prevention, detection, evaluation and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines". Circulation. 152 (11): e114 – e218. doi:10.1161/CIR.0000000000001356. PMID 40811497.
- 2024 ESC guideline: McEvoy JW, McCarthy CP, Bruno RM, Brouwers S, Canavan MD, et al. (30 August 2024). "2024 ESC Guidelines for the management of elevated blood pressure and hypertension: Developed by the task force on the management of elevated blood pressure and hypertension of the European Society of Cardiology (ESC) and endorsed by the European Society of Endocrinology (ESE) and the European Stroke Organisation (ESO)". European Heart Journal. 45 (38): 3912–4018. doi:10.1093/eurheartj/ehae178. ISSN 0195-668X. PMID 39210715.
- 2023 ESH guideline: Mancia G, Kreutz R, Brunström M, Burnier M, Grassi G, Januszewicz A, et al. (1 December 2023). "2023 ESH Guidelines for the management of arterial hypertension The Task Force for the management of arterial hypertension of the European Society of Hypertension: Endorsed by the International Society of Hypertension (ISH) and the European Renal Association (ERA)". Journal of Hypertension. 41 (12): 1874–2071. doi:10.1097/HJH.0000000000003480. hdl:11379/603005. ISSN 1473-5598. PMID 37345492.
- 2022 AAFP guideline: Coles S, Fisher L, Lin KW, Lyon C, Vosooney AA, Bird MD (December 2022). "Blood Pressure Targets in Adults With Hypertension: A Clinical Practice Guideline From the AAFP". American Family Physician. 106 (6): Online. ISSN 1532-0650. PMID 36521481. Key recommendations.
- 2019 NICE guideline: "Hypertension in adults: diagnosis and management". National Institute for Health and Clinical Excellence (NICE). 28 August 2019.
Hypertension
View on GrokipediaSigns and Symptoms
General Manifestations
Hypertension is frequently asymptomatic, earning it the moniker "silent killer" due to its ability to progress undetected while causing progressive damage to blood vessels and organs.[8] Most individuals experience no noticeable signs, even when blood pressure reaches dangerously elevated levels, which underscores the importance of routine screening.[1] Globally, nearly half of people with hypertension remain unaware of their condition, contributing to higher risks of complications like cardiovascular disease.[9] In mild cases, hypertension may occasionally manifest with nonspecific symptoms such as headaches, fatigue, or nosebleeds, though these are uncommon and not reliably indicative of the condition.[1] Headaches, for instance, can arise from vascular strain but are more typical in advanced stages rather than early hypertension.[4] Fatigue has been reported in a substantial proportion of affected individuals, potentially linked to reduced cardiac efficiency or associated comorbidities.[10] These subtle presentations often go unnoticed, delaying diagnosis until routine evaluations reveal elevated blood pressure. Physical examination can uncover indirect signs of hypertension, particularly through targeted assessments of end organs. Fundoscopic examination of the retina may reveal arteriolar narrowing, arteriovenous (AV) nicking, and flame-shaped hemorrhages, reflecting chronic vascular stress in the eyes.[11] Cardiac auscultation might detect signs of left ventricular hypertrophy (LVH), such as an accentuated second heart sound or a displaced apex beat, resulting from the heart's adaptation to increased afterload over time.[12] Additionally, auscultation over the abdomen could identify renal artery bruits, which suggest underlying secondary causes like renal artery stenosis.[13] These manifestations, though subtle, signal early end-organ damage from sustained high pressure: retinal changes indicate microvascular injury akin to broader vascular pathology, LVH represents cardiac remodeling that heightens arrhythmia risk, and renal bruits point to potential renovascular compromise.[11][12] Such findings during routine exams highlight hypertension's insidious progression, where asymptomatic elevation quietly erodes organ function before overt symptoms emerge.[4]Hypertensive Crisis
A hypertensive crisis is defined as a severe elevation in blood pressure, typically with systolic blood pressure exceeding 180 mm Hg or diastolic blood pressure exceeding 120 mm Hg. This condition is categorized into two types: hypertensive emergencies, which involve acute end-organ damage such as hypertensive encephalopathy, stroke, acute heart failure, aortic dissection, or pulmonary edema, and hypertensive urgencies, characterized by severe blood pressure elevation without evidence of acute target organ damage.[14][15][16] Symptoms of a hypertensive crisis often include severe headache, visual disturbances, chest pain, dyspnea, and confusion, signaling potential acute organ involvement. Diagnosis requires prompt evaluation to assess for end-organ damage, including electrocardiogram (ECG) to detect cardiac ischemia or arrhythmias, laboratory tests such as troponin levels for myocardial injury and creatinine for renal function, and imaging like computed tomography (CT) of the head for neurological complications or chest X-ray for pulmonary or cardiac issues.[17][15][18] Initial management of hypertensive emergencies focuses on gradual blood pressure reduction to prevent further organ damage, aiming to lower mean arterial pressure by approximately 25% within the first hour, followed by further cautious normalization; full treatment details are addressed in dedicated management sections. Untreated hypertensive crises carry a high mortality risk, with 1-year survival rates below 21% in historical untreated cases. The incidence of hypertensive crisis affects approximately 1-2% of patients with hypertension annually.[18][19][14]In Pregnancy
Hypertensive disorders of pregnancy encompass a spectrum of conditions characterized by new-onset hypertension after 20 weeks of gestation, including gestational hypertension, preeclampsia, and eclampsia. Gestational hypertension is defined as systolic blood pressure of 140 mm Hg or higher, or diastolic blood pressure of 90 mm Hg or higher, on two occasions at least 4 hours apart, without proteinuria or other features of preeclampsia. Preeclampsia involves the same blood pressure criteria accompanied by proteinuria (typically ≥300 mg in a 24-hour urine collection) or evidence of end-organ dysfunction, such as thrombocytopenia, renal insufficiency, impaired liver function, pulmonary edema, or cerebral/visual symptoms. Eclampsia represents a severe progression of preeclampsia marked by new-onset generalized tonic-clonic seizures not attributable to other causes. These disorders collectively affect 5-10% of pregnancies worldwide, with gestational hypertension comprising the majority and preeclampsia occurring in approximately 3-5%.[20][21] Symptoms of these conditions often overlap with normal pregnancy changes but can signal progression to severe disease. Common manifestations include edema (particularly in the face and hands), sudden weight gain, persistent headache, visual disturbances (such as blurred vision or scotoma), and epigastric or right upper quadrant pain due to hepatic involvement. Proteinuria may be asymptomatic but indicates renal involvement. Maternal risks escalate with preeclampsia and eclampsia, including placental abruption, stroke, disseminated intravascular coagulation, and HELLP syndrome (hemolysis, elevated liver enzymes, and low platelet count), which complicates 10-20% of severe preeclampsia cases. Fetal implications involve intrauterine growth restriction, preterm birth (often iatrogenic to protect maternal health), and increased perinatal mortality, with risks amplified in superimposed preeclampsia on preexisting chronic hypertension.[20][22][23] Diagnosis requires careful timing relative to gestational age and exclusion of chronic hypertension. Blood pressure measurements should use validated devices, with confirmation on separate occasions; ambulatory or home monitoring can aid accuracy but is not always feasible in pregnancy. For preeclampsia, beyond hypertension, assessment includes a spot urine protein-to-creatinine ratio ≥0.3 mg/mg (equivalent to ≥300 mg/24 hours) or, in its absence, laboratory evidence of organ involvement such as serum creatinine >1.1 mg/dL or doubling of baseline, platelet count <100,000/μL, or liver transaminases twice the upper limit of normal. Eclampsia is diagnosed clinically by seizures in the context of preeclampsia, prompting immediate magnesium sulfate administration. The 2024 European Society of Cardiology guidelines emphasize low-dose aspirin (75-150 mg daily) prophylaxis starting at 12 weeks gestation until 36 weeks or delivery for high-risk women (e.g., history of preeclampsia, multiple gestation, or chronic hypertension) to reduce incidence by 10-20%. Delivery remains the definitive treatment, timed based on severity and gestational age.[20][24]In Children and Adolescents
Hypertension in children and adolescents is defined as systolic or diastolic blood pressure at or above the 95th percentile for age, sex, and height on three or more separate occasions, according to guidelines from the American Academy of Pediatrics (AAP) and endorsed by the American Heart Association (AHA).[25] These percentiles account for normal variations in blood pressure related to growth and development, with updated tools available in 2025 for precise calculation via age-, sex-, and height-adjusted models.[26] The global prevalence of sustained hypertension in this population is approximately 3.89%, with rates increasing to over 10% when including elevated blood pressure, particularly among those with obesity or overweight.[27] Unlike in adults, symptoms are often absent or subtle due to the greater elasticity of pediatric blood vessels, which may buffer against immediate end-organ damage.[28] In children and adolescents, hypertension is predominantly secondary, accounting for up to 80-90% of cases, with renal parenchymal disease as the most common underlying cause, followed by cardiac conditions such as coarctation of the aorta.[29] Other renal etiologies include chronic kidney disease and renovascular hypertension, while cardiac issues often involve structural abnormalities leading to increased afterload.[13] Primary hypertension, linked to lifestyle factors like obesity, is rising but remains less frequent than in adults.[25] Most affected youth remain asymptomatic, but when present, manifestations may include headaches, fatigue, and poor sleep quality; in severe or longstanding cases, growth failure can occur due to associated metabolic or renal disturbances.[30] Hypertensive emergencies, though rare, can present with seizures, vomiting, or encephalopathy, particularly in infants who may show irritability or apnea.[31] White coat hypertension, where clinic readings are elevated due to anxiety but ambulatory monitoring is normal, is common, affecting up to 43% of referred children and often requiring confirmation with out-of-office measurements.[32] Physical examination may reveal bounding pulses, suggestive of hyperdynamic circulation in conditions like renal artery stenosis or anemia-associated hypertension, and organomegaly such as hepatosplenomegaly in cases of chronic renal disease.[33] Left ventricular hypertrophy or retinal changes may also indicate target-organ involvement, underscoring the need for early screening in at-risk groups like obese adolescents.[25]Causes
Primary Hypertension
Primary hypertension, also known as essential hypertension, accounts for 90-95% of all hypertension cases and is defined as persistently elevated blood pressure without an identifiable secondary cause.[34][35] This form arises from complex interactions between genetic predisposition and environmental influences, leading to a gradual increase in blood pressure over many years.[4] Unlike secondary hypertension, which stems from specific underlying conditions, primary hypertension has no single causative factor but rather multifactorial origins that contribute to its idiopathic nature.[35] Key risk factors for primary hypertension include genetic factors, with heritability estimates for blood pressure ranging from 30% to 50%, indicating a substantial inherited component.[36] Advancing age is a major contributor, as arterial stiffness increases and vascular function declines over time.[37] Other modifiable risks encompass obesity, which promotes endothelial dysfunction and inflammation; salt sensitivity, where high dietary sodium intake exacerbates blood pressure elevation in susceptible individuals; sedentary lifestyle, leading to reduced cardiovascular fitness; and excessive alcohol consumption, which can directly raise blood pressure through vascular and neurohormonal effects.[38][39] These factors often interact synergistically to heighten risk.[39] Among the pathogenic mechanisms, increased peripheral vascular resistance plays a central role, driven by structural changes such as arteriolar wall thickening and reduced lumen diameter in resistance vessels.[40] Insulin resistance contributes by impairing vasodilation and promoting sodium retention, while sympathetic nervous system overactivity heightens vasoconstriction and cardiac output.[41][42] These processes are interconnected and detailed further in pathophysiology discussions. Emerging research highlights the polygenic basis of primary hypertension, with no single gene dominating but rather thousands of common variants contributing small effects. Genome-wide association studies (GWAS) since 2020 have identified numerous loci influencing blood pressure traits, enabling the development of polygenic risk scores that predict hypertension susceptibility and cardiovascular outcomes with improving accuracy across diverse populations.[43][44]Secondary Hypertension
Secondary hypertension refers to elevated blood pressure resulting from an identifiable underlying medical condition, accounting for approximately 5% to 10% of all hypertension cases in adults.[45] Unlike primary hypertension, secondary forms often present opportunities for cure or significant improvement through targeted treatment of the root cause, underscoring the importance of screening in select patients.[13] Recent studies, including large cohort analyses, suggest this prevalence may be underestimated, particularly in resistant hypertension where rates can exceed 20%.[46] The most frequent causes fall into renal, endocrine, vascular, sleep-related, and medication-induced categories. Renal disorders, such as chronic kidney disease and renovascular stenosis due to atherosclerotic or fibromuscular dysplasia, contribute to about 2% to 5% of secondary hypertension cases, with renovascular stenosis specifically implicated in roughly 1% to 5% of overall hypertension.[13] Endocrine etiologies include primary aldosteronism (Conn's syndrome), which affects 5% to 14% of hypertensive patients and up to 20-30% of those with resistant hypertension according to 2025 Endocrine Society guidelines and cohort studies; Cushing's syndrome and pheochromocytoma are rarer, each accounting for less than 1%.[46][47] Other notable causes encompass coarctation of the aorta (prevalent in 0.03% to 0.1% of the general population but a key reversible factor in young adults), obstructive sleep apnea (linked to 20% to 40% of resistant cases in obese individuals), and drug-induced hypertension from agents like nonsteroidal anti-inflammatory drugs (NSAIDs), corticosteroids, or oral contraceptives.[13][48] Clinical clues prompting evaluation for secondary hypertension include onset before age 30 or after 55, resistant blood pressure despite three antihypertensive medications, severe hypertension exceeding 180/110 mmHg, and biochemical abnormalities such as hypokalemia (occurring in approximately 20-40% of primary aldosteronism cases), which increases suspicion for primary aldosteronism as it is found in up to 30% of hypertensive patients with hypokalemia.[29][49] Additional red flags are an abdominal bruit indicating renovascular disease or episodic symptoms like headaches and palpitations hinting at pheochromocytoma. Diagnostic screening often begins with targeted tests: the plasma aldosterone-to-renin ratio (ARR) for endocrine causes, where a ratio greater than 20 to 30 with elevated aldosterone prompts confirmatory saline infusion or fludrocortisone suppression; renal artery imaging via duplex ultrasound or CT angiography for renovascular suspicion; polysomnography for sleep apnea; and echocardiography or MRI for coarctation.[13] A thorough medication review is essential for drug-related cases. Addressing the underlying condition can resolve hypertension in many instances, such as surgical correction of coarctation yielding normalization in approximately 50-70% of patients or adrenalectomy for unilateral primary aldosteronism curing hypertension in 30% to 60% of cases and improving control in most others.[13][50] Revascularization for renal artery stenosis or continuous positive airway pressure for sleep apnea similarly enhances blood pressure management, reducing reliance on medications and mitigating cardiovascular risks.[29] Early identification through systematic screening in high-risk groups is thus critical to leverage these reversible pathways.[51]Pathophysiology
Hemodynamic and Vascular Mechanisms
Hypertension arises from alterations in the fundamental hemodynamic determinants of blood pressure, primarily governed by the equation for mean arterial pressure (MAP), which is derived from the steady-state relationship between blood flow and vascular resistance. MAP is calculated as the product of cardiac output (CO) and total peripheral resistance (TPR): This equation stems from the principle that arterial pressure represents the force exerted by blood volume flow (CO) against the resistance offered by the systemic vasculature (TPR), assuming a closed circulatory system in equilibrium. In essential hypertension, the primary driver of elevated MAP is an increase in TPR, often through vasoconstriction of arterioles and reduced arterial compliance, while CO remains normal or near-normal in the early stages.[52][53] Systemic vascular resistance increases due to heightened vascular tone and structural changes in the vessel walls, leading to persistent vasoconstriction that elevates TPR without initially affecting CO. As hypertension progresses, particularly in cases involving volume overload such as obesity-related hypertension, CO may rise to accommodate expanded plasma volume, further contributing to elevated MAP. Endothelial dysfunction plays a central role in these vascular changes, characterized by diminished production of nitric oxide (NO), a key vasodilator, which impairs endothelium-dependent relaxation and promotes vasoconstriction. Concurrently, increased endothelin-1 expression, a potent vasoconstrictor, exacerbates this imbalance, fostering a pro-hypertensive state in the vascular endothelium.[53][54][55][56] Arterial stiffness, another critical vascular mechanism, results from age-related and hypertensive remodeling, including excessive collagen deposition in the arterial media and adventitia, which reduces vessel distensibility and compliance. This stiffening disproportionately affects large elastic arteries like the aorta, leading to a widened pulse pressure—the difference between systolic and diastolic pressures—as systolic pressure rises while diastolic pressure falls due to faster pressure wave propagation and reduced damping. With advancing age, this widening of pulse pressure becomes more pronounced, reflecting progressive large-artery stiffening and amplifying the hemodynamic burden on the cardiovascular system, which can contribute to end-organ damage such as left ventricular hypertrophy.[57][58][59]Neurohormonal and Renal Factors
The renin-angiotensin-aldosterone system (RAAS) plays a central role in the neurohormonal regulation of blood pressure, particularly in hypertension, by modulating vascular tone and fluid balance. Renin, an enzyme secreted by the juxtaglomerular cells of the kidney in response to reduced renal perfusion pressure, low sodium delivery to the distal tubule, or sympathetic stimulation, cleaves angiotensinogen (produced by the liver) to form angiotensin I. Angiotensin-converting enzyme (ACE), primarily in the lungs, then converts angiotensin I to angiotensin II, a potent vasoconstrictor that elevates blood pressure by increasing systemic vascular resistance and promoting aldosterone release from the adrenal cortex. Aldosterone enhances sodium reabsorption in the renal collecting ducts, leading to water retention and expanded plasma volume, which further sustains hypertension. This cascade can be summarized as: low renal perfusion → renin release → angiotensin II formation → vasoconstriction and aldosterone-mediated sodium retention → elevated blood pressure.[60] Overactivity of the sympathetic nervous system contributes to hypertension by augmenting RAAS activation and directly influencing vascular and renal function. Increased sympathetic outflow, often originating from central nervous system dysregulation, stimulates renin release via β1-adrenergic receptors in the kidney and promotes norepinephrine-mediated vasoconstriction, leading to sustained elevations in blood pressure. This overactivity is evident in essential hypertension, where muscle sympathetic nerve activity is heightened, correlating with disease severity and resistance to treatment. Additionally, an imbalance in natriuretic peptides, such as atrial natriuretic peptide (ANP) and B-type natriuretic peptide (BNP), exacerbates this process; these peptides normally counteract RAAS by promoting natriuresis, vasodilation, and inhibition of renin and aldosterone secretion, but their reduced levels or impaired signaling in hypertension diminish this protective effect, allowing unchecked sodium retention and pressure elevation.[61][62][63] Renal mechanisms are integral to neurohormonal hypertension, primarily through alterations in pressure natriuresis and sodium handling. Pressure natriuresis refers to the kidney's ability to increase sodium and water excretion in response to elevated renal perfusion pressure, thereby normalizing blood volume; in hypertension, this curve shifts rightward due to intrinsic renal defects or neurohormonal influences, requiring higher pressures to achieve adequate natriuresis and perpetuating sodium retention. Defects in renal sodium handling, such as impaired proximal tubule reabsorption or distal tubule dysfunction influenced by RAAS overactivation, further impair this process, leading to volume-dependent hypertension. RAAS inhibitors, including ACE inhibitors and angiotensin receptor blockers, effectively target these pathways, reducing blood pressure in a majority of patients by blocking angiotensin II effects and restoring natriuretic balance.[64][65][66] Recent research highlights emerging influences, such as gut microbiome alterations modulating RAAS activity through microbial metabolites that affect angiotensin II signaling, potentially offering new therapeutic avenues.[67][68] Advances in 2025 include small interfering RNA (siRNA) therapies targeting RAAS components, such as zilebesiran, which silences hepatic angiotensinogen expression to suppress the entire cascade at its source, demonstrating sustained blood pressure reductions in clinical trials with quarterly dosing. These therapies address limitations of traditional RAAS inhibitors by providing longer-lasting effects and improved adherence, particularly in resistant hypertension.[69][70]Diagnosis
Blood Pressure Measurement Techniques
Blood pressure measurement in clinical settings primarily relies on office-based techniques using either auscultatory or oscillometric methods to assess hypertension accurately. The auscultatory method involves a mercury or aneroid sphygmomanometer and stethoscope to detect Korotkoff sounds over the brachial artery, requiring trained personnel for precise detection of systolic and diastolic phases, though it is susceptible to observer errors such as rapid cuff deflation exceeding 2-3 mmHg per second.[71] In contrast, the oscillometric method, commonly used in automated devices, detects arterial pulsations through cuff pressure oscillations and is preferred for its reduced operator dependence when the device is validated, making it suitable for both attended and unattended measurements.[72][71] Accurate office measurements necessitate standardized protocols to minimize variability. Proper cuff selection is essential, with the bladder width covering 40% of arm circumference and length encircling 80% to avoid overestimation by up to 20 mmHg from an undersized cuff or underestimation from an oversized one; validated devices are recommended, and resources like the Validate BP list ensure compliance.[72][73] Patients should be seated with back supported, feet flat, and arm at heart level after 5 minutes of rest, avoiding caffeine, exercise, or talking prior to measurement to prevent artifacts.[71] A single reading is insufficient for diagnosis, as it can lead to misclassification; instead, the average of at least two readings, taken 1-2 minutes apart, over two or more visits is required, with improper techniques potentially causing errors of 10-20 mmHg.[72][74] Out-of-office monitoring complements office assessments, particularly ambulatory blood pressure monitoring (ABPM) and home blood pressure monitoring (HBPM), to detect phenomena like white-coat or masked hypertension. ABPM employs a portable oscillometric device for 24-hour automated recordings, typically at 15-30 minute intervals daytime and 30-60 minutes nighttime, providing averages such as daytime ≥130/80 mmHg corresponding to elevated risk, and is a Class I recommendation for confirming diagnoses, including stage 1 hypertension (office 130-139/80-89 mmHg) in the 2025 guidelines.[72] HBPM involves self-measurements with validated upper-arm cuff devices over several days, averaging ≥130/80 mmHg to align with office thresholds, and is emphasized for ongoing management, especially when integrated with telehealth or team-based care to improve control.[72] These methods enhance prognostic accuracy over office readings alone, with ABPM particularly valued for its ability to predict cardiovascular events independently.[71]Additional Diagnostic Tests
Following confirmation of hypertension through standardized blood pressure measurement techniques, additional diagnostic tests are essential to identify underlying causes, assess target organ damage, and evaluate cardiovascular risk. These evaluations help guide treatment decisions and monitor complications such as chronic kidney disease (CKD) and left ventricular hypertrophy (LVH).[2] Laboratory assessments form the cornerstone of this evaluation, including a complete blood count, basic metabolic panel with electrolytes (sodium, potassium, and calcium), thyroid-stimulating hormone (TSH), serum creatinine to calculate estimated glomerular filtration rate (eGFR), fasting glucose or hemoglobin A1c, lipid profile, and urinalysis with urine albumin-to-creatinine ratio (UACR). Abnormal electrolytes may indicate secondary hypertension due to conditions like hyperaldosteronism, while an eGFR below 60 mL/min/1.73 m² signals CKD risk, warranting closer monitoring and specific therapies. The lipid profile and fasting glucose contribute to overall cardiovascular disease (CVD) risk stratification, with tests repeated annually or as needed based on initial results. For suspected secondary causes, the plasma aldosterone-to-renin activity ratio is recommended as a screening tool for primary aldosteronism, particularly in patients with resistant hypertension or hypokalemia. Urinalysis detects proteinuria, and a UACR of 30 mg/g or higher identifies early kidney damage, influencing the choice of renin-angiotensin system inhibitors.[2][72] Cardiac evaluations include a 12-lead electrocardiogram (ECG) to detect LVH, a common marker of prolonged hypertension exposure, and echocardiography to assess left ventricular structure and function. Echocardiography provides detailed insights into systolic and diastolic dysfunction, refining CVD risk estimates beyond basic labs. These tests are performed at diagnosis, especially in patients with symptoms or risk factors for heart disease.[2] Renal imaging, such as ultrasound or Doppler studies, is indicated to evaluate for renal artery stenosis in cases of resistant hypertension or asymmetric kidney sizes. This non-invasive approach helps identify structural abnormalities contributing to secondary hypertension without routine use in all patients.[2] Routine screening for target organ damage integrates these tests, emphasizing eGFR and UACR for renal involvement, ECG and echocardiography for cardiac effects, and fundoscopy for retinopathy when appropriate. Early detection of damage, such as reduced eGFR, informs aggressive management to prevent progression.[2] Integration of the 2025 PREVENT risk calculator enhances CVD assessment by incorporating lab results like lipids and glucose, along with demographics and comorbidities, to estimate 10-year risk; a threshold of 7.5% or higher supports intensified therapy. This tool, updated for broader applicability, is used at diagnosis to personalize prevention strategies.[2][75]Classification in Adults
Hypertension in adults is classified based on office blood pressure measurements, with categories defined by systolic and diastolic thresholds in millimeters of mercury (mmHg). These classifications guide risk assessment and management decisions according to the 2025 American Heart Association (AHA)/American College of Cardiology (ACC) guideline. The categories emphasize the importance of consistent readings obtained through validated techniques to ensure accuracy.[72] The following table outlines the blood pressure categories for adults:| Category | Systolic BP (mmHg) | Diastolic BP (mmHg) |
|---|---|---|
| Normal | <120 | <80 |
| Elevated | 120–129 | <80 |
| Stage 1 Hypertension | 130–139 | or 80–89 |
| Stage 2 Hypertension | ≥140 | or ≥90 |
| Hypertensive Crisis | >180 | and/or >120 |
Classification in Children
The classification of hypertension in children and adolescents relies on age-, sex-, and height-specific percentiles derived from normative data from the 2017 American Academy of Pediatrics (AAP) guideline, rather than fixed absolute thresholds used in adults. This approach accounts for physiological variations during growth. Normal blood pressure is defined as below the 90th percentile. Elevated blood pressure is >=90th percentile but <95th percentile, or 120 to 129/<80 mmHg (whichever is lower) for those >=13 years. Stage 1 hypertension is >=95th percentile but <95th percentile + 12 mmHg, or 130 to 139/80 to 89 mmHg (whichever is lower). Stage 2 hypertension is >=95th percentile + 12 mmHg, or >=140/90 mmHg (whichever is lower). These categories require confirmation on at least three separate occasions to distinguish persistent hypertension from transient elevations.[77] In pediatric populations, hypertension is predominantly secondary, particularly in younger children, with underlying causes such as renal or cardiovascular disorders accounting for 45% to 85% of cases in specialized clinics. This contrasts with the increasing prevalence of primary hypertension linked to obesity in adolescents. Accurate classification is crucial, as untreated pediatric hypertension can lead to long-term cardiovascular risks. Additionally, the white coat effect—where anxiety in clinical settings elevates readings—is more prevalent in children than adults, often affecting up to 52% of referred cases, necessitating ambulatory or home monitoring for confirmation.[78][79] Recent reviews as of 2025 have addressed limitations in traditional norms by discussing obesity-adjusted references, recognizing that standard tables may underestimate hypertension in obese youth due to exclusion of overweight children in normative data. These discussions highlight ongoing knowledge gaps in inclusive normative data, but the core classification remains based on the 2017 AAP guideline.[80]Prevention
Lifestyle Modifications for Prevention
Lifestyle modifications represent a cornerstone of primary prevention for hypertension, particularly in at-risk populations such as those with prehypertension, obesity, or family history of the condition. These evidence-based strategies target modifiable risk factors to lower blood pressure and reduce the incidence of hypertension without pharmacological intervention. Comprehensive adoption of multiple changes, including dietary adjustments, physical activity, and behavioral shifts, has demonstrated substantial benefits in large-scale clinical trials.[81] The Dietary Approaches to Stop Hypertension (DASH) eating plan is a key dietary intervention emphasizing high intake of fruits, vegetables, whole grains, and low-fat dairy while limiting saturated fats, sweets, and sodium to less than 2,300 mg per day. This pattern promotes nutrient-rich foods that support vascular health and electrolyte balance, leading to systolic blood pressure reductions of approximately 5-11 mmHg in individuals with elevated blood pressure. Clinical trials have shown that even without sodium restriction, the DASH diet lowers blood pressure within weeks compared to a typical American diet.[82][83] Weight management through caloric restriction and balanced nutrition is another critical modification, with evidence indicating that a 5-10% reduction in body weight, particularly when accompanied by reductions in waist circumference or central adiposity, can decrease systolic blood pressure by 5-20 mmHg in overweight adults. This effect is attributed to decreased sympathetic nervous system activity and improved insulin sensitivity, which help prevent the progression to hypertension. Sustained modest weight loss has been linked to a 20-30% lower risk of developing hypertension over several years.[84][85][86] Regular aerobic exercise, recommended at a minimum of 150 minutes per week of moderate-intensity activity such as brisk walking or cycling, enhances cardiovascular fitness and endothelial function, reducing systolic blood pressure by 4-9 mmHg. Guidelines from major health organizations endorse this level of activity for all adults to mitigate hypertension risk, with benefits accruing from consistent participation that also aids in weight control.[87][88] Moderating alcohol consumption is advised, with limits of no more than two standard drinks per day for men and one for women, as excessive intake raises blood pressure through mechanisms like increased cortisol and vascular stiffness. Adhering to these thresholds can prevent alcohol-related elevations in blood pressure and lower hypertension risk by up to 20% in moderate drinkers.[89][90] Smoking cessation is essential, as tobacco use accelerates vascular damage and sympathetic activation, contributing to hypertension development; quitting reduces this risk by improving arterial compliance and decreasing the incidence of hypertension by 25-50% within years of abstinence. Support programs combining counseling and pharmacotherapy facilitate long-term success and yield cardiovascular protective effects.[91][92] Stress management techniques, including mindfulness-based practices, yoga, and progressive muscle relaxation, help counteract chronic stress-induced blood pressure elevations by modulating the autonomic nervous system. Regular implementation of these methods can lower systolic blood pressure by 2-5 mmHg and support overall prevention efforts in high-stress individuals.[93][94] Combined lifestyle modifications amplify preventive outcomes, with trials like the PREMIER study demonstrating that integrated interventions—encompassing DASH diet, weight loss, exercise, and sodium reduction—reduce blood pressure by 6-12 mmHg and lower hypertension incidence by 20-50% over 18-48 months compared to usual care. Similarly, the Trials of Hypertension Prevention II (TOHP II) showed that weight loss combined with sodium restriction decreased hypertension development by 38% during extended follow-up. These multifaceted approaches not only prevent hypertension but also enhance overall cardiovascular health.[81][95] Emerging public health emphases in 2025 highlight precision nutrition, leveraging genomic profiling to tailor dietary recommendations for hypertension prevention, such as personalized sodium or potassium intake based on gene-diet interactions. This approach promises to optimize outcomes in genetically susceptible individuals by addressing variability in metabolic responses to standard lifestyle advice.[96]Pharmacological Prevention in High-Risk Groups
Pharmacological prevention of hypertension targets individuals at elevated risk for cardiovascular disease (CVD), particularly those with high-normal blood pressure (defined as stage 1 hypertension: 130-139 mm Hg systolic or 80-89 mm Hg diastolic) accompanied by comorbidities such as diabetes mellitus or chronic kidney disease (CKD).[72] In these groups, early initiation of low-dose antihypertensive therapy aims to delay or prevent the onset of frank hypertension and mitigate associated organ damage, with evidence supporting a focus on renin-angiotensin system (RAS) inhibitors.[72] The 2025 American Heart Association/American College of Cardiology (AHA/ACC) guideline incorporates the PREVENT (Precision Risk Estimation to inform Treatment) equation to estimate 10-year CVD risk, recommending pharmacologic intervention alongside lifestyle modifications for adults with stage 1 hypertension and a PREVENT risk of ≥7.5%, with a higher threshold of ≥10% for those with diabetes or target organ damage.[72] This threshold identifies those likely to benefit from early drug therapy to avert progression to stage 2 hypertension (≥140/90 mm Hg) and subsequent CVD events.[76] In patients with diabetes and high-normal blood pressure, low-dose angiotensin-converting enzyme (ACE) inhibitors or angiotensin II receptor blockers (ARBs) are preferred due to their dual benefits in blood pressure control and renoprotection.[97] For instance, these agents reduce the progression from microalbuminuria to macroalbuminuria—a precursor to overt nephropathy and hypertension—by approximately 30-50% in normotensive or prehypertensive individuals with type 2 diabetes.[98] Similarly, in those with CKD and albuminuria but without established hypertension, ACE inhibitors or ARBs slow disease progression and lower the risk of end-stage renal disease by about 31%, thereby delaying hypertension development linked to worsening kidney function.[99] The TROPHY trial demonstrated that low-dose candesartan (an ARB) in prehypertensive adults reduced the incidence of new-onset hypertension by 66% during two years of active treatment, with a sustained 16% relative risk reduction over four years including post-treatment follow-up.[100] For broader CVD prevention in high-risk stage 1 hypertension per PREVENT criteria (≥7.5% risk), statins may be initiated concurrently with antihypertensives to address dyslipidemia and further lower overall cardiovascular burden, as elevated cholesterol amplifies hypertension-related risks.[72] This combined approach, integrated with lifestyle measures like dietary sodium reduction, enhances outcomes without increasing adverse events in select populations.[72] Overall, such targeted pharmacologic strategies in vulnerable groups can reduce progression to clinical hypertension and CVD complications by 20-40%, based on risk reductions observed in landmark trials and meta-analyses of RAS blockade.[98][100]Management
Treatment Goals and Guidelines
The 2025 American Heart Association (AHA)/American College of Cardiology (ACC) guideline establishes a universal blood pressure treatment goal of less than 130/80 mm Hg for all adults with hypertension, aiming to reduce cardiovascular disease (CVD) risk through early intervention and consistent monitoring.[72] This target applies broadly, with therapy initiation recommended at 140/90 mm Hg or higher, or at 130/80 mm Hg for those with elevated 10-year CVD risk (≥7.5%) as assessed by the PREVENT equations, which incorporate factors like kidney function and statin use for improved risk stratification.[72] For adults over 65 years, the guideline endorses a systolic target below 130 mm Hg if tolerated, emphasizing individualized assessment to balance benefits against potential harms such as orthostatic hypotension.[72] In patients with chronic kidney disease (CKD), the goal remains less than 130/80 mm Hg (with systolic below 130 mm Hg), prioritizing renoprotective agents while monitoring for adverse kidney events.[72] Similarly, for those with diabetes, the target is less than 130/80 mm Hg (systolic below 130 mm Hg), supported by evidence of improved microvascular and macrovascular outcomes.[72] The 2024 European Society of Cardiology (ESC) guidelines align closely, recommending a blood pressure target of less than 130/80 mm Hg for most hypertensive adults, with a preferred systolic range of 120-129 mm Hg to optimize CVD prevention without excessive risk.[101] This framework incorporates risk stratification tools akin to PREVENT, such as SCORE2, to guide therapeutic decisions in high-risk groups, though it emphasizes office and home monitoring for accurate goal assessment.[101] For elderly patients (including those over 65), the ESC advocates targets below 130/80 mm Hg if feasible, with adjustments for frailty and comorbidities to ensure tolerability.[101] In CKD and diabetes, the guidelines maintain the less than 130/80 mm Hg goal, highlighting the need for personalized approaches to mitigate complications like progression of kidney disease.[101] Achieving tighter blood pressure control, as demonstrated in seminal trials like SPRINT, reduces major CVD events by approximately 25% compared to standard targets, underscoring the value of these guidelines in clinical practice.[102] The World Health Organization's 2023 Global report on hypertension: the race against a silent killer emphasizes that hypertension is often asymptomatic until complications arise, describing it as a "silent killer." The report highlights that effective treatment and improved control rates could prevent millions of premature deaths and major cardiovascular events globally. Specifically, scaling up hypertension treatment to levels observed in high-performing countries could avert approximately 76 million deaths, 120 million strokes, 79 million heart attacks, and 17 million cases of heart failure between 2023 and 2050.[6] These projections underscore the profound impact of adhering to evidence-based management strategies and achieving blood pressure control. Adherence monitoring is integral, with both AHA/ACC and ESC recommending regular follow-up using validated measurement techniques to evaluate progress and adjust therapy as needed.[72][101]Nonpharmacologic Interventions
Nonpharmacologic interventions are essential first-line strategies in the management of hypertension, particularly for patients with mild or stage 1 disease, and serve as adjuncts to pharmacologic therapy in more severe cases. These approaches, including targeted lifestyle modifications, can achieve meaningful blood pressure reductions and improve overall cardiovascular health without the risks associated with medications. According to the 2025 American Heart Association/American College of_Cardiology guidelines, all adults with hypertension should prioritize these interventions to reach treatment goals of less than 130/80 mm Hg.[103] Dietary modifications, especially sodium restriction, form a foundational element. Limiting sodium intake to less than 2,300 mg per day, ideally 1,500 mg, by avoiding processed foods and table salt, can lower systolic blood pressure by 2-5 mm Hg across populations. The Dietary Approaches to Stop Hypertension (DASH) eating plan, which emphasizes 4–5 servings each of fruits and vegetables daily, along with whole grains, lean proteins, nuts, and low-fat dairy while limiting red meat, sweets, and added sugars, produces additive benefits; randomized trials show it reduces systolic blood pressure by 5-6 mm Hg compared to a standard diet, with effects amplified in hypertensive individuals to up to 11 mm Hg when combined with low-sodium intake, and similar benefits for diastolic pressure of 3-5 mm Hg.[104][105] Weight management is particularly impactful for overweight or obese patients, where excess adiposity contributes to hypertension pathogenesis. Intentional weight loss through caloric restriction and behavioral changes yields blood pressure reductions proportional to the degree of loss; meta-analyses indicate approximately 1 mm Hg systolic decrease per kilogram lost, with modest reductions of 5-10 pounds (2-5 kg) if overweight associated with 5-10 mm Hg drops in systolic pressure, particularly when accompanied by decreases in waist circumference or central adiposity.[106][107][86] The 2025 guidelines recommend aiming for a body mass index below 25 kg/m² to optimize these outcomes.[103] Physical activity is another cornerstone, with guidelines endorsing at least 150 minutes per week of moderate-intensity aerobic exercise, such as brisk walking, cycling, or swimming, supplemented by 2–3 days of strength training and isometric exercises like wall sits or planks. Such regimens consistently lower systolic blood pressure by 5-8 mm Hg in hypertensive adults, with comparable diastolic reductions, and benefits persisting long-term through improved endothelial function and reduced sympathetic activity.[87][108] Alcohol moderation addresses a modifiable risk factor, as excessive intake elevates blood pressure dose-dependently. Restricting consumption to no more than one standard drink per day for women and two for men can prevent rises and achieve reductions of 3-4 mm Hg systolic in heavy drinkers who cut back; complete abstinence is advised for those with uncontrolled hypertension.[109][110] Smoking cessation is also recommended, as tobacco use exacerbates vascular damage and elevates blood pressure; quitting can contribute to overall reductions within weeks to months. Stress management techniques, including deep breathing, meditation, and yoga, further support blood pressure control by mitigating sympathetic activation. Vitamin D supplementation has been investigated as a potential nonpharmacologic intervention for hypertension. However, large meta-analyses and randomized controlled trials have shown that vitamin D supplementation, including high doses (e.g., 4000 IU/day or more), does not significantly lower blood pressure in the general population or most hypertensive patients. Some studies indicate possible modest benefits in specific subgroups, such as older adults with obesity or vitamin D deficiency, but high doses provide no additional benefit over standard doses, and overall evidence does not support its use as an antihypertensive therapy.[111][112][113] For patients with persistent hypertension despite optimized lifestyle and multidrug therapy, device-based renal denervation provides a targeted option in select cases. This minimally invasive procedure uses catheter-delivered energy to ablate overactive renal sympathetic nerves, addressing neurogenic contributions to resistant hypertension. The U.S. Food and Drug Administration approved renal denervation systems in 2023, with endorsements in the 2025 hypertension guidelines for adults with uncontrolled blood pressure on three or more medications. Sham-controlled trials, including the SPYRAL HTN series, report durable systolic reductions of 6-9 mm Hg at 12 months and up to 3 years, with low procedural risks.[114][115] Patient education is integral to the success of nonpharmacologic interventions, empowering individuals to adopt and sustain changes through knowledge of hypertension risks and self-monitoring techniques such as home blood pressure tracking. Educational programs improve adherence, enhance self-efficacy, and boost blood pressure control rates by 10-20% in clinical settings; patients should consult healthcare providers to evaluate underlying causes or medication needs. In mild hypertension, these strategies alone can normalize blood pressure and obviate the need for drugs in 20-30% of cases, underscoring their role in personalized management, with noticeable effects often emerging in weeks to months.[116][117]Pharmacologic Therapy
Pharmacologic therapy for hypertension targets key physiological mechanisms to reduce blood pressure and cardiovascular risk. The primary antihypertensive drug classes include angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin II receptor blockers (ARBs), which inhibit the renin-angiotensin-aldosterone system (RAAS) to decrease vasoconstriction and sodium retention; calcium channel blockers (CCBs), particularly long-acting dihydropyridine types, which promote vascular smooth muscle relaxation and vasodilation; thiazide diuretics, such as chlorthalidone and indapamide, which enhance sodium and water excretion to reduce plasma volume; and beta-blockers, which antagonize sympathetic nervous system activity to lower heart rate and cardiac output.[72] According to the 2025 American Heart Association (AHA)/American College of Cardiology (ACC) guidelines, ACEIs, ARBs, CCBs, and thiazide diuretics are recommended as first-line agents (Class of Recommendation [COR] I, Level of Evidence [LOE] A), with selection guided by patient-specific factors such as comorbidities, age, and ethnicity.[72] For stage 1 hypertension (blood pressure 130-139/80-89 mm Hg) in low-risk adults, monotherapy is typically initiated after lifestyle modifications, while stage 2 hypertension (≥140/90 mm Hg), particularly with initial readings >150/100 mm Hg, or stage 1 in high-risk individuals (e.g., with cardiovascular disease, diabetes, chronic kidney disease, or 10-year cardiovascular risk ≥7.5% via the PREVENT equation) warrants initial dual therapy to achieve prompt control. For such stage 2 cases, especially in young patients, dual therapy (particularly ARB + CCB) is recommended over monotherapy, as it achieves better and faster blood pressure control, whereas monotherapy often fails to provide rapid reductions in higher readings.[72] Single-pill combinations (SPCs), such as an RAAS blocker paired with a CCB or thiazide diuretic, are strongly preferred for stage 2 and high-risk cases to improve adherence and efficacy (COR I, LOE A).[72] Therapy must be individualized to optimize outcomes and minimize adverse effects. For example, in Black adults, thiazide diuretics or CCBs are preferred as initial monotherapy due to greater efficacy compared to RAAS inhibitors alone, which show reduced blood pressure-lowering response in this population (COR I, LOE A).[72] Common side effects include dry cough and angioedema with ACEIs (affecting 5-20% and <0.1% of patients, respectively), hyperkalemia with ARBs, hypokalemia and hyponatremia with thiazides, peripheral edema with CCBs, and fatigue or bradycardia with beta-blockers; monitoring and dose adjustments are essential to manage these risks.[72] The 2025 ACC highlights emerging options from recent trials, including aprocitentan, a dual endothelin receptor antagonist that reduces systolic blood pressure by 4-6 mm Hg when added to standard therapy in resistant cases (from the PRECISION trial), and lorundrostat, an aldosterone synthase inhibitor that achieved significant systolic blood pressure reductions versus placebo at 12 weeks in the ADVANCE-HTN trial.[72][118] These agents represent potential adjuncts, though established classes remain the cornerstone of therapy.[72]| Drug Class | Mechanism | First-Line Use | Key Considerations |
|---|---|---|---|
| ACEIs/ARBs | RAAS blockade reducing vasoconstriction and aldosterone | Yes (COR I, LOE A) | Less effective monotherapy in Black patients; monitor renal function and potassium |
| CCBs (dihydropyridine) | Vasodilation via calcium influx inhibition | Yes (COR I, LOE A) | Preferred in Black patients; effective for stroke prevention |
| Thiazide diuretics | Diuresis and natriuresis reducing volume | Yes (COR I, LOE A) | Preferred in Black patients; monitor electrolytes |
| Beta-blockers | Sympathetic inhibition lowering cardiac output | Compelling indications (e.g., heart failure, post-MI) | Not first-line generally; use in younger patients or with tachycardia |

