Recent from talks
Nothing was collected or created yet.

Male (symbol: ♂) is the sex of an organism that produces the gamete (sex cell) known as sperm, which fuses with the larger female gamete,[1][2][3] or ovum, in the process of fertilisation. A male organism cannot reproduce sexually without access to at least one ovum from a female, but some organisms can reproduce both sexually and asexually.[4] Most male mammals, including male humans, have a Y chromosome,[5][6] which codes for the production of larger amounts of testosterone to develop male reproductive organs.
In humans, the word male can also be used to refer to gender, in the social sense of gender role or gender identity.[7][8]
Overview
[edit]The existence of separate sexes has evolved independently at different times and in different lineages, an example of convergent evolution.[9][10] The repeated pattern is sexual reproduction in isogamous species with two or more mating types with gametes of identical form and behavior (but different at the molecular level) to anisogamous species with gametes of male and female types to oogamous species in which the female gamete is very much larger than the male and has no ability to move. There is a good argument that this pattern was driven by the physical constraints on the mechanisms by which two gametes get together as required for sexual reproduction.[11][page needed]. But in some species males can reproduce by themselves asexually, for example via androgenesis.[12][13]
Accordingly, sex is defined across species by the type of gametes produced (i.e.: spermatozoa vs. ova) and differences between males and females in one lineage are not always predictive of differences in another.[10][14][15]
Male/female dimorphism between organisms or reproductive organs of different sexes is not limited to animals; male gametes are produced by chytrids, diatoms and land plants, among others. In land plants, female and male designate not only the female and male gamete-producing organisms and structures but also the structures of the sporophytes that give rise to male and female plants.[citation needed]
Evolution
[edit]The evolution of anisogamy led to the evolution of male and female function.[16] Before the evolution of anisogamy, mating types in a species were isogamous: the same size and both could move, catalogued only as "+" or "-" types.[17]: 216 In anisogamy, the mating type is called a gamete. The male gamete is smaller than the female gamete, and usually mobile.[18] Anisogamy remains poorly understood, as there is no fossil record of its emergence. Numerous theories exist as to why anisogamy emerged. Many share a common thread, in that larger female gametes are more likely to survive, and that smaller male gametes are more likely to find other gametes because they can travel faster. Current models often fail to account for why isogamy remains in a few species.[19] Anisogamy appears to have evolved multiple times from isogamy; for example, female Volvocales (a type of green algae) evolved from the plus mating type.[19][17]: 222 Although sexual evolution emerged at least 1.2 billion years ago, the lack of anisogamous fossil records make it hard to pinpoint when males evolved.[20] One theory suggests male evolved from the dominant mating type (called mating type minus).[21]
Symbol, etymology, and usage
[edit]Symbol
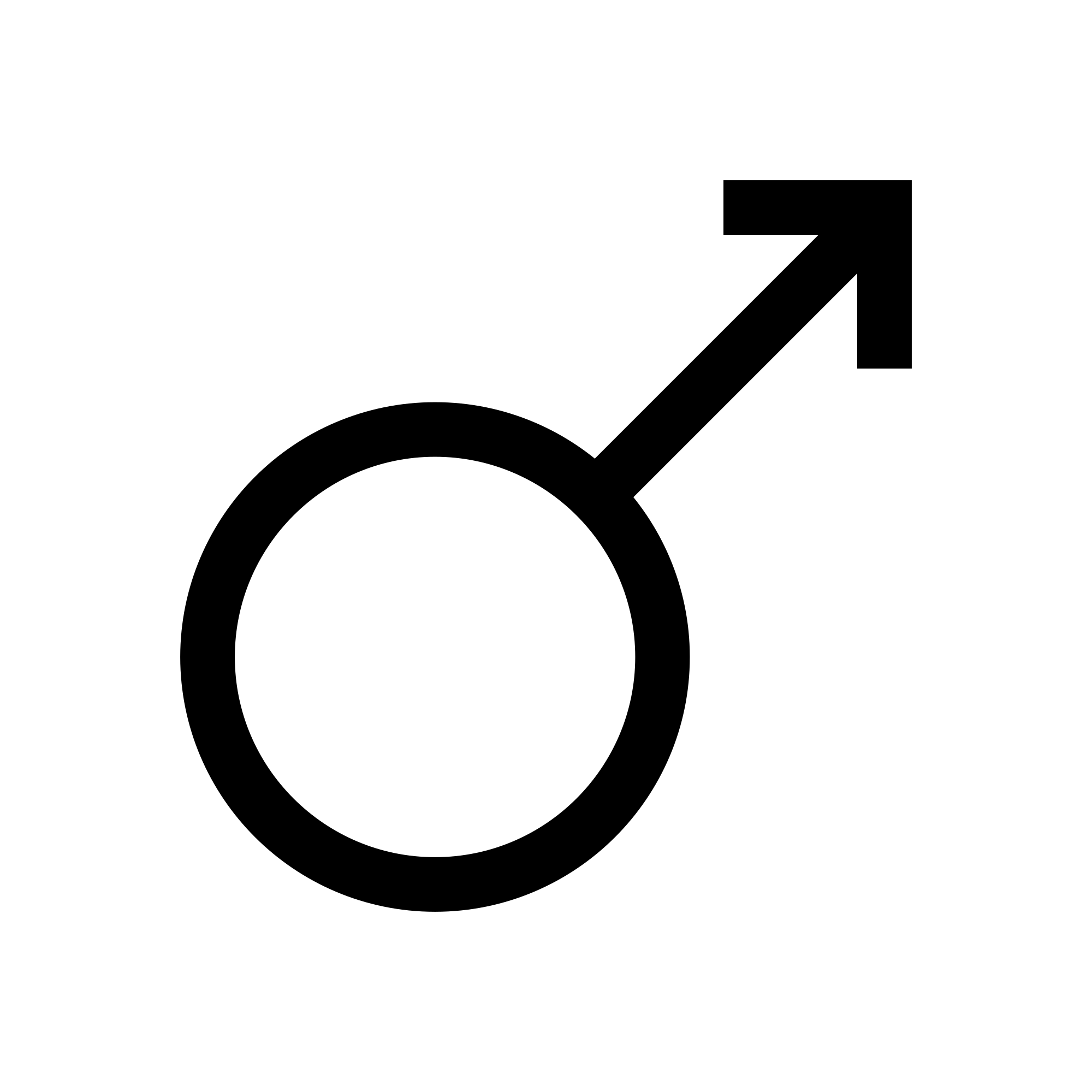
[edit]A common symbol used to represent the male sex is the Mars symbol ♂, a circle with an arrow pointing northeast. The Unicode code-point is:
- U+2642 ♂ MALE SIGN (♂)
The symbol is identical to the planetary symbol of Mars. It was first used to denote sex by Carl Linnaeus in 1751. The symbol is sometimes seen as a stylized representation of the shield and spear of the Roman god Mars. According to William T. Stearn, however, this derivation is "fanciful" and all the historical evidence favours "the conclusion of the French classical scholar Claude de Saumaise (Salmasius, 1588–1683)" that it is derived from θρ, the contraction of a Greek name for the planet Mars, which is Thouros.[22]
Etymology
[edit]Borrowed from Old French masle, from Latin masculus ("masculine, male, worthy of a man"), diminutive of mās ("male person or animal, male").[23]
Usage
[edit]In humans, the word male can be used in the context of gender, such as for gender role or gender identity of a man or boy.[7] For example, according to Merriam-Webster, "male" can refer to "having a gender identity that is the opposite of female".[24] According to the Cambridge Dictionary, "male" can mean "belonging or relating to men".[25]
Male can also refer to a shape of connectors.[26][27]
Sex determination
[edit]
The sex of a particular organism may be determined by a number of factors. These may be genetic or environmental, or may naturally change during the course of an organism's life. Although most species have only two sexes (either male or female),[9][10][2] hermaphroditic animals, such as worms, have both male and female reproductive organs.[28] Species that are divided into females and males are classified as gonochoric in animals, as dioecious in seed plants[2] and as dioicous in cryptogams.[29]: 82 Males can coexist with hermaphrodites, a sexual system called androdioecy. They can also coexist with females and hermaphrodites, a sexual system called trioecy.[30]
Not all species share a common sex-determination system. In most animals, including humans, sex is determined genetically; however, species such as Cymothoa exigua change sex depending on the number of females present in the vicinity.[31]
Genetic determination
[edit]Most mammals, including humans, are genetically determined as such by the XY sex-determination system where males have XY (as opposed to XX in females) sex chromosomes. It is also possible in a variety of species, including humans, to be XX male or have other karyotypes. During reproduction, a male can give either an X sperm or a Y sperm, while a female can only give an X egg. A Y sperm and an X egg produce a male, while an X sperm and an X egg produce a female.[32]
The part of the Y-chromosome which is responsible for maleness is the sex-determining region of the Y-chromosome, the SRY.[33] The SRY activates Sox9, which forms feedforward loops with FGF9 and PGD2 in the gonads, allowing the levels of these genes to stay high enough in order to cause male development;[34] for example, Fgf9 is responsible for development of the spermatic cords and the multiplication of Sertoli cells, both of which are crucial to male sexual development.[35]
The ZW sex-determination system, where males have ZZ (as opposed to ZW in females) sex chromosomes, may be found in birds and some insects (mostly butterflies and moths) and other organisms. Members of the insect order Hymenoptera, such as ants and bees, are often determined by haplodiploidy,[16] where most males are haploid and females and some sterile males are diploid. However, fertile diploid males may still appear in some species, such as Cataglyphis cursor.[36]
Environmental determination
[edit]In some species of reptiles, such as alligators, sex is determined by the temperature at which the egg is incubated. Other species, such as some snails, practice sex change: adults start out male, then become female.[37] In tropical clown fish, the dominant individual in a group becomes female while the other ones are male.[38]
Secondary sex characteristics
[edit]Male animals have evolved to use secondary sex characteristics as a way of displaying traits that signify their fitness. Sexual selection is believed to be the driving force behind the development of these characteristics. Differences in physical size and the ability to fulfill the requirements of sexual selection have contributed significantly to the outcome of secondary sex characteristics in each species.[39]
In many species, males differ from females in more ways than just the production of sperm. For example, in some insects and fish, the male is smaller than the female. In seed plants, the sporophyte sex organ of a single organism includes both the male and female parts.
In mammals, including humans, males are typically larger than females. This is often attributed to the need for male mammals to be physically stronger and more competitive in order to win mating opportunities. In humans specifically, males have more body hair and muscle mass than females.[40][page needed][41][page needed]
Birds often exhibit colorful plumage that attracts females.[42][page needed] This is true for many species of birds where the male displays more vibrant colors than the female, making them more noticeable to potential mates. These characteristics have evolved over time as a result of sexual selection, as males who exhibited these traits were more successful in attracting mates and passing on their genes.
See also
[edit]References
[edit]- ^ Lehtonen, Jussi; Parker, Geoff A. (1 December 2014). "Gamete competition, gamete limitation, and the evolution of the two sexes". Molecular Human Reproduction. 20 (12): 1161–1168. doi:10.1093/molehr/gau068. ISSN 1360-9947. PMID 25323972.
- ^ a b c Fusco, Giuseppe; Minelli, Alessandro (10 October 2019). The Biology of Reproduction. Cambridge University Press. pp. 111–113. ISBN 978-1-108-49985-9.
- ^ Hine, Robert; Martin, Elizabeth (2015). A Dictionary of Biology. Oxford University Press. p. 354. ISBN 978-0-19-871437-8.
- ^ Lively, Curtis M. (1 March 2010). "A Review of Red Queen Models for the Persistence of Obligate Sexual Reproduction". Journal of Heredity. 101 (suppl_1): S13 – S20. doi:10.1093/jhered/esq010. ISSN 0022-1503. PMID 20421322.
- ^ Reference, Genetics Home. "Y chromosome". Genetics Home Reference. Retrieved 22 July 2020.
- ^ "Y Chromosome". Genome.gov. Retrieved 7 September 2020.
- ^ a b Palazzani, Laura; Bailes, Victoria; Fella, Marina (2012). Gender in Philosophy and Law. SpringerBriefs in law. Dordrecht : Springer. p. v. ISBN 978-94-007-4991-7.
'gender' means human gender, male/female gender
{{cite book}}: CS1 maint: publisher location (link) (eBook) - ^ "Definition of MALE". www.merriam-webster.com. 4 February 2025. Retrieved 10 February 2025.
- ^ a b Berrill, N.J. "Sex". Encyclopedia Britannica. Retrieved 22 July 2020.
- ^ a b c Klymkowsky, Michael W.; Melanie M., Cooper (4 June 2016). "4.9: Sexual dimorphism". Biology LibreTexts. Retrieved 22 July 2020.
- ^ Dusenbery, David B. (2009). Living at Micro Scale. Cambridge, Massachusetts: Harvard University Press. Chapter 20. ISBN 978-0-674-03116-6..
- ^ Hedtke, Shannon M.; Stanger-Hall, Kathrin; Baker, Robert J.; Hillis, David M. (May 2008). "All-Male Asexuality: Origin and Maintenance of Androgenesis in the Asian Clam Corbicula". Evolution. 62 (5): 1119–1136. doi:10.1111/j.1558-5646.2008.00344.x. PMID 18266987.
- ^ Schwander, Tanja; Oldroyd, Benjamin P (28 September 2008). "Androgenesis: where males hijack eggs to clone themselves". Philosophical Transactions of the Royal Society B: Biological Sciences. 371 (1706). doi:10.1098/rstb.2015.0534. PMC 5031619. PMID 27619698.
- ^ Wilcox, Christie (23 April 2020). "Why Sex? Biologists Find New Explanations". Quanta Magazine. Retrieved 22 July 2020.
- ^ Lehtonen, Jussi (2017), "Gamete Size", in Shackelford, Todd K.; Weekes-Shackelford, Viviana A. (eds.), Encyclopedia of Evolutionary Psychological Science, Cham: Springer International Publishing, pp. 1–4, doi:10.1007/978-3-319-16999-6_3063-1, ISBN 978-3-319-16999-6
- ^ a b Bachtrog D, Mank JE, Peichel CL, Kirkpatrick M, Otto SP, Ashman TL, Hahn MW, Kitano J, Mayrose I, Ming R, Perrin N, Ross L, Valenzuela N, Vamosi JC (July 2014). "Sex determination: why so many ways of doing it?". PLOS Biology. 12 (7) e1001899. doi:10.1371/journal.pbio.1001899. PMC 4077654. PMID 24983465.
- ^ a b Sawada, Hitoshi; Inoue, Naokazu; Iwano, Megumi (7 February 2014). Sexual Reproduction in Animals and Plants. Springer. ISBN 978-4-431-54589-7.
- ^ Kumar R, Meena M, Swapnil P (2019). "Anisogamy". In Vonk J, Shackelford T (eds.). Anisogamy. Encyclopedia of Animal Cognition and Behavior. Cham: Springer International Publishing. pp. 1–5. doi:10.1007/978-3-319-47829-6_340-1. ISBN 978-3-319-47829-6.
- ^ a b Togashi, Tatsuya; Cox, Paul Alan (14 April 2011). The Evolution of Anisogamy: A Fundamental Phenomenon Underlying Sexual Selection. Cambridge University Press. pp. 1–15. ISBN 978-1-139-50082-1.
- ^ Butterfield, Nicholas J. (2000). "Bangiomorpha pubescens n. gen., n. sp.: implications for the evolution of sex, multicellularity, and the Mesoproterozoic/Neoproterozoic radiation of eukaryotes". Paleobiology. 26 (3): 386. Bibcode:2000Pbio...26..386B. doi:10.1666/0094-8373(2000)026<0386:BPNGNS>2.0.CO;2. S2CID 36648568. Retrieved 12 April 2021.
- ^ Togashi, Tatsuya; Bartelt, John L.; Yoshimura, Jin; Tainaka, Kei-ichi; Cox, Paul Alan (21 August 2012). "Evolutionary trajectories explain the diversified evolution of isogamy and anisogamy in marine green algae". Proceedings of the National Academy of Sciences of the United States of America. 109 (34): 13692–13697. Bibcode:2012PNAS..10913692T. doi:10.1073/pnas.1203495109. ISSN 0027-8424. PMC 3427103. PMID 22869736.
- ^ Stearn, William T. (1962). "The Origin of the Male and Female Symbols of Biology". Taxon. 11 (4): 109–113. doi:10.2307/1217734. JSTOR 1217734.
- ^ "male | Etymology, origin and meaning of male by etymonline". www.etymonline.com. Etymonline. Retrieved 23 July 2023.
- ^ "Definition of MALE". Merriam-Webster. Retrieved 22 March 2023.
- ^ "male". Cambridge Dictionary.
- ^ J. Richard Johnson (1962). How to Build Electronic Equipment. New York: Rider. p. 167.
To minimize confusion, the connector portions with projecting prongs are referred to as the 'male' portion, and the sockets as the 'female' portion.
- ^ Richard Ferncase (2013). Film and Video Lighting Terms and Concepts. Hoboken Taylor and Francis. p. 96. ISBN 978-0-240-80157-5.
female[:] Refers to a socket type connector, which must receive a male connector
- ^ "hermaphroditism | Definition, Types, & Effects". Encyclopedia Britannica. Retrieved 22 July 2020.
- ^ Buck WR; Goffinet, B (August 2000). "Morphology and classification of mosses". In Shaw AJ & Goffinet B (ed.). Bryophyte Biology. New York: Cambridge University Press. ISBN 978-0-521-66794-4.
- ^ Leonard, Janet L. (21 May 2019). Transitions Between Sexual Systems: Understanding the Mechanisms of, and Pathways Between, Dioecy, Hermaphroditism and Other Sexual Systems. Springer. pp. 1–3. ISBN 978-3-319-94139-4.
- ^ Ruiz-L, A.; Madrid-V, J. (6 March 1992). "Studies on the biology of the parasitic isopod Cymothoa exigua Schioedte and Meinert, 1884 and it's relationship with the snapper Lutjanus peru (Pisces: Lutjanidae) Nichols and Murphy, 1922, from commercial catch in Michoacan". Ciencias Marinas. 18 (1): 19–34. doi:10.7773/cm.v18i1.885. ISSN 2395-9053.
- ^ "43.1C: Sex Determination". Biology LibreTexts. 17 July 2018. Retrieved 22 July 2020.
- ^ Reference, Genetics Home. "SRY gene". Genetics Home Reference. Retrieved 22 July 2020.
- ^ Moniot, Brigitte; Declosmenil, Faustine; Barrionuevo, Francisco; Scherer, Gerd; Aritake, Kosuke; Malki, Safia; Marzi, Laetitia; Cohen-Solal, Ann; Georg, Ina; Klattig, Jürgen; Englert, Christoph; Kim, Yuna; Capel, Blanche; Eguchi, Naomi; Urade, Yoshihiro; Boizet-Bonhoure, Brigitte; Poulat, Francis (2009). "The PGD2 pathway, independently of FGF9, amplifies SOX9 activity in Sertoli cells during male sexual differentiation". Development. 136 (11): 1813–1821. doi:10.1242/dev.032631. PMC 4075598. PMID 19429785.
- ^ Kim, Y.; Kobayashi, A.; Sekido, R.; Dinapoli, L.; Brennan, J.; Chaboissier, M. C.; Poulat, F.; Behringer, R. R.; Lovell-Badge, R.; Capel, B. (2006). "Fgf9 and Wnt4 Act as Antagonistic Signals to Regulate Mammalian Sex Determination". PLOS Biology. 4 (6) e187. doi:10.1371/journal.pbio.0040187. PMC 1463023. PMID 16700629.
- ^ Doums, Claudie (2013). "Fertile diploid males in the ant Cataglyphis cursor: a potential cost of thelytoky?". Behavioral Ecology and Sociobiology. 67 (12): 1983–1993. Bibcode:2013BEcoS..67.1983D. doi:10.1007/s00265-013-1606-6. hdl:10261/88167. S2CID 18141328. Retrieved 2 October 2021.
- ^ Cahill, Abigail E.; Juman, Alia Rehana; Pellman-Isaacs, Aaron; Bruno, William T. (December 2015). "Physical and Chemical Interactions with Conspecifics Mediate Sex Change in a Protandrous Gastropod Crepidula fornicata". The Biological Bulletin. 229 (3): 276–281. doi:10.1086/bblv229n3p276. ISSN 0006-3185. PMID 26695826. S2CID 22783998.
- ^ Bull, J. J. (March 1980). "Sex Determination in Reptiles". The Quarterly Review of Biology. 55 (1): 3–21. doi:10.1086/411613. ISSN 0033-5770. S2CID 85177125.
- ^ Campbell B (2017). Human Evolution: An Introduction to Man's Adaptations. Routledge. pp. 392–393. ISBN 978-1-351-51441-5.
- ^ Ellis, Lee; Hershberger, Scott; Field, Evelyn; Wersinger, Scott; Pellis, Sergio; Geary, David; Palmer, Craig; Hoyenga, Katherine; Hetsroni, Amir (13 May 2013). Sex Differences: Summarizing More than a Century of Scientific Research. Psychology Press. ISBN 978-1-136-87493-2.
- ^ Richards, Julia E.; Hawley, R. Scott (12 December 2010). The Human Genome. Academic Press. ISBN 978-0-08-091865-5.
- ^ switze, International Conference on Comparative Physiology 1992 Crans; Bassau, Short & (4 August 1994). The Differences Between the Sexes. Cambridge University Press. ISBN 978-0-521-44878-9.
{{cite book}}: CS1 maint: numeric names: authors list (link)
Further reading
[edit]- Wedgwood, Hensleigh (1855). "On False Etymologies". Transactions of the Philological Society (6): 68.
Male is the biological sex in gonochoristic species that produces the smaller, motile gametes known as spermatozoa, in contrast to the female sex which produces larger, immotile gametes or ova.[1][2] This dimorphism, termed anisogamy, evolved from isogamous ancestors through disruptive selection favoring gametes optimized for quantity and mobility in males versus size and provisioning in females, establishing the two sexes across eukaryotes.[3][4] In mammals, including humans, males are characterized by the XY sex chromosome system, where the Y chromosome's SRY gene initiates testis development and male phenotypic differentiation during embryogenesis.[5][6] Males typically exhibit higher reproductive variance due to anisogamy's constraints, with greater potential for multiple fertilizations but also higher rates of reproductive failure compared to females.[7] While rare intersex conditions exist, biological sex remains binary at the gametic level, with developmental disorders not altering the foundational reproductive classification.[8][9]
Definition and Terminology
Biological Definition
In biology, the male sex is defined as the reproductive category of organism that produces small, mobile gametes, typically termed sperm or spermatozoa, which are specialized for motility and fusion with larger female gametes (ova or eggs) during fertilization. This distinction arises from anisogamy, the evolutionary divergence in gamete size and investment, where males contribute fewer resources per gamete but produce them in greater numbers to enhance fertilization probability.[10][11] In sexually reproducing species, including humans and other mammals, this gametic criterion serves as the foundational determinant of sex, independent of secondary traits like morphology or chromosomes, as it directly pertains to reproductive role.[12][13] Anisogamy evolved from isogamous ancestors through disruptive selection favoring gametes optimized for either quantity (small, cheap male gametes) or quality (large, nutrient-rich female gametes), leading to the binary sexes observed in gonochoristic species—those with separate male and female individuals.[14] In male organisms, spermatogenesis occurs in gonads (testes in vertebrates), yielding haploid sperm via meiosis, with adaptations like flagella for propulsion and minimal cytoplasm to prioritize genetic delivery over provisioning.[15] This definition holds across taxa exhibiting anisogamy, from insects to mammals, underscoring that biological maleness is not merely phenotypic but causally tied to gamete production for sexual reproduction.[16] Exceptions, such as hermaphroditism, involve dual gamete types but do not negate the male role's association with small gametes in dioecious systems.[10]Etymology and Historical Usage
The English adjective and noun "male" entered the language in the late 14th century, borrowed from Old French masle (modern French mâle), which derives from Latin masculus, a diminutive of mās denoting a male person or animal.[17] In Latin, masculus connoted something masculine, manly, or worthy of a man, emphasizing physical and behavioral traits associated with the sex producing smaller gametes in reproductive contexts.[18] This etymological root underscores a distinction from "female," which originates separately from Latin femella, a diminutive of fēmina ("woman"), refuting folk etymologies linking the pair through superficial phonetic similarity.[19] Historically, "male" in English has denoted the sex or gender characterized by XY chromosomes in humans, production of spermatozoa, or analogous roles in other species, consistently applied to humans, animals, and plants since its adoption.[17] By the 15th century, it appeared in texts distinguishing male from female in biological and anatomical descriptions, such as in Chaucer's works referencing male figures or traits, evolving into precise scientific terminology by the 17th century amid empirical studies of reproduction—e.g., William Harvey's 1651 De Generatione Animalium implicitly aligning with male-female dimorphism in gamete production, though predating modern genetic framing.[17] Unlike the Old English wer (specific to adult males, obsolete by the 13th century) or the gender-neutral mann (human being), "male" provided a Latinate specificity for sexual differentiation, retaining this denotation without significant semantic shift into modern usage.[20]Symbolic Representation
The male sex is conventionally represented by the glyph ♂, consisting of a circle surmounted by an arrow pointing northeast at approximately 45 degrees. This symbol, also known as the Mars sign, originated in ancient Greco-Roman astrology as the emblem of the planet Mars, the celestial body dedicated to the god of war whose attributes—strength, aggression, and virility—aligned with perceptions of masculinity.[21] Scholarly analysis traces ♂ to a contraction or ligature from ancient Greek script abbreviating the name of Mars (Ares), possibly derived from the initial and final letters of Θούρος (Thouros), an epithet for the deity, with the circle representing the planet and the arrow stylizing the crossbar of rho (ρ).[22] Earlier interpretations linked it directly to Mars's shield and spear in Roman iconography, emphasizing martial prowess as a metaphor for male generative and protective roles. In alchemical traditions, ♂ further symbolized iron, the metal linked to weapons and male potency due to its hardness and utility in forging tools of war and labor.[21] By the 18th century, Swedish botanist Carl Linnaeus incorporated ♂ into scientific nomenclature to denote staminate (male) plants in works such as Mantissa Plantarum (1767), extending its use from astronomical to biological contexts for distinguishing sexes in organisms. This adoption facilitated its standardization in medicine, genetics, and taxonomy, where ♂ consistently marks male individuals, gametes, or traits, as seen in pedigree charts and anatomical diagrams.[21] Beyond the glyph, male symbolism encompasses phallic motifs across cultures, representing fertility and generative power; for instance, ancient Egyptian obelisks or Mesopotamian pillars evoked the erect penis as a sign of male creative force, though these lack the universality of ♂ in modern scientific discourse. In contemporary usage, ♂ appears in public signage, such as on restrooms or product labeling, reinforcing its role as an intuitive, cross-cultural indicator of biological maleness.[22]Genetic and Developmental Foundations
Chromosomal and Genetic Determination
In humans, biological sex is chromosomally determined by the XX/XY system, where males possess a 46,XY karyotype consisting of one X chromosome inherited from the mother and one Y chromosome from the father.[23] The presence of the Y chromosome, rather than its size or gene count, is the primary genetic signal for maleness, as the Y is significantly smaller than the X and carries fewer genes overall, with most Y-linked genes focused on male-specific functions like spermatogenesis and sex determination.[23][24] The key determinant on the Y chromosome is the SRY (sex-determining region Y) gene, located on its short arm (Yp11.3), which encodes a 204-amino-acid protein functioning as a transcription factor with a high-mobility-group (HMG) box domain.[25][26] This protein binds to specific DNA sequences in the supporting cells of the developing gonad, inducing structural changes that distort DNA architecture and activate downstream genes, thereby triggering the differentiation of Sertoli cells and subsequent testis formation from the bipotential gonad around embryonic weeks 6–7.[26][27] Without SRY expression, the default developmental pathway leads to ovarian formation, underscoring SRY's causal role as the mammalian master switch for male gonadal fate.[24][27] Genetic evidence confirms SRY's necessity and sufficiency: mutations or deletions in SRY result in 46,XY complete gonadal dysgenesis (Swyer syndrome), where individuals develop female external genitalia and internal structures despite the XY karyotype, with no functional testes.[25] Conversely, translocation of SRY to an X chromosome produces 46,XX males (Sry-positive XX testicular disorder of sex development), who develop testes and male phenotypes, as demonstrated in transgenic mouse models where XX embryos engineered with SRY undergo male differentiation.[25][28] These rare cases, occurring in approximately 1 in 20,000–25,000 births for Sry-positive XX males, highlight that SRY overrides chromosomal sex in directing maleness, independent of X chromosome dosage.[25] Beyond SRY, the Y chromosome harbors ancillary genes like those in the AZF (azoospermia factor) regions that support spermatogenesis but do not initiate sex determination; their loss causes infertility without altering core male gonadal identity.[24] In evolutionary terms, SRY likely arose via duplication and modification of an X-linked SOX3 gene precursor in therian mammals, enabling Y-linked male determination conserved across placental mammals and marsupials.[29] While downstream pathways involve autosomal and X-linked modifiers (e.g., SOX9 upregulation), SRY remains the initiating trigger, with disruptions in its transient expression window (peaking at embryonic day 10.5 in mice, analogous to human week 6) preventing male commitment.[27][30]Embryonic and Hormonal Development
In human embryos, sexual differentiation begins with the bipotential gonad, which remains undifferentiated until approximately the 6th week of gestation, when no morphological sex differences are observable.[31] The presence of the Y chromosome triggers expression of the SRY gene around 6-7 weeks post-fertilization, initiating Sertoli cell differentiation and testis formation from the gonadal ridge.[31] [32] This genetic cascade upregulates genes like SOX9, committing the gonad to testicular development and suppressing ovarian pathways.[33] Sertoli cells in the developing testes secrete anti-Müllerian hormone (AMH) starting around 7-8 weeks, which binds to receptors on Müllerian duct cells, inducing apoptosis and regression of structures that would otherwise form the uterus and fallopian tubes in females.[31] [34] Concurrently, interstitial Leydig cells emerge and produce testosterone by 8 weeks, peaking at 11-18 weeks to promote survival and differentiation of Wolffian ducts into the epididymis, vas deferens, and seminal vesicles.[32] [33] Testosterone diffuses locally to stabilize these ducts via androgen receptor activation, a process absent in XX embryos lacking sufficient androgens.[31] External genitalia masculinization requires conversion of testosterone to dihydrotestosterone (DHT) by 5α-reductase type 2 enzyme, expressed in the urogenital sinus and genital tubercle from 9-12 weeks onward.[35] DHT drives elongation of the phallus into the penis and fusion of urethral folds, while the genital swellings form the scrotum; this occurs between 9-14 weeks, with full differentiation by 14 weeks if androgen levels and receptor function are intact.[31] [33] Fetal testosterone levels, derived primarily from Leydig cell synthesis rather than maternal sources, reach adult male concentrations by mid-gestation, underscoring the testis's autonomous role in driving male phenotypic commitment.[32] Testicular descent begins around 25 weeks, facilitated by regression of the gubernaculum under androgen influence, with completion by 35 weeks; failure here can result from insufficient AMH or testosterone signaling.[32] Postnatally, a transient surge in gonadotropins and testosterone occurs in the first few months (minipuberty), supporting further genital maturation, though primary embryonic programming establishes the male trajectory.[31] Disruptions in these hormonal axes, such as SRY mutations or enzyme deficiencies, lead to atypical development, highlighting the causal specificity of Y-linked and androgen-dependent mechanisms.[33]Disorders of Sex Development
Disorders of sex development (DSD) encompass congenital conditions in which chromosomal, gonadal, or anatomical sex is atypical, particularly in 46,XY individuals where male gonadal development or androgen action fails to produce typical male phenotypes.[36] These arise from disruptions in genetic regulation, gonadal differentiation, hormone synthesis, or receptor function during embryogenesis, leading to undervirilization such as micropenis, hypospadias, or cryptorchidism.[37] Incidence of 46,XY DSD varies by etiology but collectively affects approximately 1 in 20,000 to 1 in 100,000 male births, though underdiagnosis occurs due to subtle presentations.[38] Diagnosis typically involves karyotyping, hormone assays, imaging, and genetic sequencing to identify specific defects, with multidisciplinary management focusing on fertility preservation, hormone replacement, and surgical correction where indicated.[39] Sex chromosome DSDs, such as Klinefelter syndrome (47,XXY), result from nondisjunction during meiosis, yielding an extra X chromosome in genetic males.[40] Prevalence stands at 1 in 500 to 1 in 1,000 newborn males, with symptoms including small testes (hypogonadism), azoospermia, elevated gonadotropins, gynecomastia, and increased risk of metabolic disorders like type 2 diabetes.[41] [42] Testosterone levels are often low post-puberty, necessitating replacement therapy to mitigate osteoporosis and improve muscle mass, though fertility requires assisted reproduction via testicular sperm extraction in select cases.[43] In 46,XY DSD due to androgen action defects, androgen insensitivity syndrome (AIS) stems from mutations in the AR gene on the X chromosome, impairing testosterone and dihydrotestosterone signaling.[44] Complete AIS (CAIS) presents with female external genitalia, intra-abdominal testes, and absent uterus, typically diagnosed at puberty via primary amenorrhea; partial AIS (PAIS) yields ambiguous genitalia with varying virilization; mild AIS (MAIS) manifests as infertility or gynecomastia in phenotypic males.[45] Gonadectomy is recommended in CAIS post-puberty to reduce gonadal malignancy risk (up to 15-20%), followed by estrogen replacement. Defects in androgen synthesis or metabolism, such as 5-alpha-reductase type 2 deficiency (5ARD2), involve SRD5A2 gene mutations preventing conversion of testosterone to dihydrotestosterone, essential for external genital masculinization.[46] Affected 46,XY infants exhibit perineoscrotal hypospadias, pseudovagina, and clitoromegaly at birth, but often virilize at puberty with phallic growth and male gender identity due to rising testosterone.[47] Autosomal recessive inheritance predominates, with higher prevalence in consanguineous populations; management includes watchful waiting for spontaneous masculinization or reconstructive surgery, avoiding early interventions that contradict pubertal outcomes.[48] Gonadal dysgenesis in 46,XY individuals, often from SRY gene deletions or SF1/NR5A1 mutations, leads to streak gonads, female or ambiguous genitalia, and streak gonad syndrome with high malignancy risk (30% by age 30), necessitating prophylactic gonadectomy.[38] Persistent müllerian duct syndrome, due to AMH or AMHR2 defects, causes male external genitalia with internal female structures like uterus, increasing hernia and infertility risks.[49] Empirical data underscore that while DSDs disrupt typical dimorphism, genetic sex remains XY, with outcomes influenced by androgen exposure timing rather than social constructs.[37] Long-term studies emphasize early genetic diagnosis to guide sex assignment based on potential for male functionality, challenging earlier practices of routine female reassignment.[50]Physical and Physiological Traits
Primary Sex Organs
The primary sex organs in human males are the testes, paired gonads that serve exocrine functions in producing spermatozoa and endocrine functions in secreting androgens such as testosterone.[51] These organs develop from the genital ridge in the posterior abdominal wall during embryogenesis and descend into the scrotum via the inguinal canal, typically completing descent by the first year of life to ensure a lower ambient temperature conducive to sperm production.[51] The scrotal positioning maintains intratesticular temperature at 34–35°C, approximately 2–3°C below core body temperature, as higher temperatures impair spermatogenesis by disrupting meiosis and increasing germ cell apoptosis.[51] Each testis is an ovoid structure encased in a fibrous tunica albuginea, measuring roughly 4–5 cm in length, 2–3 cm in width, and 3 cm in anteroposterior diameter in adults, with a total mass of about 10–15 grams per testis.[52] [53] Internally, the testis comprises approximately 250–300 seminiferous tubules, coiled structures that constitute 80–90% of the organ's volume and are the site of spermatogenesis.[54] These tubules are lined with multilayered germ cells progressing from spermatogonia at the basal lamina through meiosis to form spermatids, which differentiate into spermatozoa under the support of Sertoli cells; Sertoli cells also secrete inhibin B to regulate follicle-stimulating hormone (FSH) and form tight junctions creating the blood-testis barrier to protect developing gametes from immune surveillance.[51] [54] Interstitial spaces between tubules contain Leydig cells, which comprise 10–20% of testicular volume and synthesize testosterone in response to luteinizing hormone (LH) from the pituitary, with peak production rates supporting systemic levels of 300–1000 ng/dL in adult males.[51] Spermatogenesis, initiated at puberty under FSH and testosterone influence, cycles continuously over approximately 74 days per cohort of germ cells, yielding 100–200 million spermatozoa daily across both testes, though only a fraction achieve maturity after epididymal transit.[54] [55] Normal ejaculate parameters include sperm concentration exceeding 15 million per milliliter, with over 40% motility, reflecting the testes' high-output capacity evolved for reproductive success despite high attrition rates in gamete viability.[51] Disruptions such as cryptorchidism, where testes fail to descend, elevate risks of infertility and germ cell tumors due to sustained higher temperatures.[51]Secondary Sexual Characteristics
Secondary sexual characteristics in males develop during puberty as a result of increased androgen production, primarily testosterone from the Leydig cells in the testes, stimulated by luteinizing hormone (LH) from the anterior pituitary.[56] This process typically begins between ages 9 and 14, with a mean onset around 11-12 years, following initial gonadarche marked by testicular enlargement to a volume exceeding 4 mL.[57] Testosterone concentrations rise progressively, reaching adult levels by late puberty (Tanner stage 5, around ages 15-17), driving dimorphic traits that enhance male-typical morphology without direct involvement in gamete production.[58] Recent cohort studies indicate a secular trend toward earlier onset, with mean ages for initial pubic hair (Tanner stage 2) at 10.4 years in Black boys and 11.2 years in White boys, compared to 12-13 years in mid-20th-century data, potentially linked to factors like improved nutrition or endocrine disruptors, though causation remains unestablished.[59] Key androgen-mediated changes include growth of terminal hair on the face, chest, back, and pubic regions, mediated by dihydrotestosterone (DHT), a potent metabolite of testosterone acting on androgen receptors in hair follicles.[56] Pubic hair emerges as coarse and curly, spreading in an inverse triangle pattern by Tanner stage 4 (ages 13-14), while axillary hair follows approximately two years after adrenarche.[56] Facial hair development begins around age 13-15, with beard growth accelerating in late adolescence due to localized DHT sensitivity.[58] Skeletal and muscular adaptations contribute to sexual dimorphism, with testosterone promoting linear bone growth via epiphyseal plate stimulation and increased muscle protein synthesis through androgen receptor upregulation, resulting in greater upper-body mass (about 40% more lean muscle than females by adulthood).[60] Shoulder breadth increases disproportionately to hip width, yielding a higher shoulder-to-hip ratio (average 1.4:1 in males versus 0.8:1 in females), driven by estrogen aromatized from testosterone influencing pelvic closure but less so in males due to lower conversion efficiency.[56] Peak height velocity occurs at Tanner stage 3-4 (ages 13-14), averaging 9-10 cm/year, followed by epiphyseal fusion under combined androgen-estrogen effects.[57] Laryngeal enlargement, manifesting as voice deepening and thyroid cartilage prominence (Adam's apple), results from testosterone-induced hypertrophy of vocal cord and cartilage tissues, with fundamental frequency dropping from ~250 Hz prepubertally to ~120 Hz in adults, typically between ages 12-15.[58] Other traits include sebaceous gland hyperactivity leading to acne (peaking at ages 14-17) and reduced subcutaneous fat, shifting distribution away from hips toward abdomen, though visceral fat accumulation in adulthood correlates inversely with testosterone levels.[61] These features vary by genetics, ethnicity, and environment; for instance, East Asian males often exhibit later facial hair onset despite comparable testosterone rises, attributable to androgen receptor polymorphisms.[59] Hypogonadism delays or impairs these developments, reversible with testosterone replacement inducing full secondary characteristic maturation in deficient males.[62]Hormonal and Metabolic Profiles
Males exhibit distinctly higher circulating levels of androgens, particularly testosterone, compared to females, with adult male serum testosterone typically ranging from 300 to 1000 ng/dL, produced primarily by the testes at rates approximately 30 times greater than ovarian production in females.[63][60] This hormonal profile drives the development and maintenance of male secondary sexual characteristics, including increased muscle mass and reduced subcutaneous fat deposition.[64] Testosterone levels peak during early adulthood, declining gradually with age at about 1-2% per year after age 30, independent of comorbidities in some populations.[65] Estrogen levels in males, derived mainly from peripheral aromatization of testosterone, remain low at 10-40 pg/mL, contrasting sharply with female ranges that fluctuate widely due to ovarian cycles.[66] Gonadotropins such as luteinizing hormone (LH) and follicle-stimulating hormone (FSH) regulate testicular function, with LH stimulating Leydig cell testosterone production and FSH supporting spermatogenesis; disruptions in these axes, as in hypogonadism, lead to subnormal androgen levels and associated metabolic shifts.[67] Metabolically, male hormonal profiles contribute to greater skeletal muscle mass—averaging 33 kg versus 21 kg in females of similar body mass—and higher basal metabolic rates, partly attributable to androgen-mediated enhancements in lean tissue and lipid oxidation efficiency.[68][69] Androgens inhibit visceral adiposity accumulation and bolster insulin sensitivity, reducing risks of metabolic syndrome when levels are adequate; conversely, androgen deficiency correlates with central obesity, dyslipidemia, and impaired glucose tolerance.[70][71] While protein turnover rates in muscle are comparable between sexes when normalized to lean mass, males demonstrate superior anabolic responses to resistance stimuli, sustaining higher energy expenditure.[72][73] These differences underscore testosterone's role in partitioning energy toward musculoskeletal growth over fat storage, though environmental factors like obesity can suppress endogenous production via feedback inhibition.[74]Reproductive Biology
Spermatogenesis and Gamete Production
Spermatogenesis is the biological process by which diploid germ cells in the male testes develop into mature haploid spermatozoa, the male gametes essential for fertilization.[54] This process occurs continuously after puberty within the seminiferous tubules of the testes, where germ cells interact closely with Sertoli cells for structural and nutritional support.[55] Unlike oogenesis in females, which is finite and begins prenatally, male gamete production is ongoing and replenishes daily throughout reproductive life, reflecting the evolutionary emphasis on quantity over individual gamete quality in males.[75] The process begins with spermatogonia, diploid stem cells located at the basal compartment of the seminiferous epithelium, which undergo mitotic divisions to maintain a stem cell pool and produce type A spermatogonia that commit to differentiation.[76] These differentiate into type B spermatogonia, which then form primary spermatocytes that enter meiosis I, yielding secondary spermatocytes. Meiosis II follows rapidly, producing haploid round spermatids. Spermiogenesis then transforms these spermatids into elongated spermatozoa through nuclear condensation, acrosome formation, flagellum development, and cytoplasmic reduction, without further cell division.[54] The resulting spermatozoa are released into the tubular lumen via spermiation, facilitated by Sertoli cells, and subsequently mature further in the epididymis.[77] Hormonal regulation is critical, with luteinizing hormone (LH) from the pituitary stimulating Leydig cells in the testicular interstitium to secrete testosterone, which acts locally at high concentrations to sustain meiosis and spermiogenesis in a paracrine manner.[78] Follicle-stimulating hormone (FSH) targets Sertoli cells, promoting their proliferation during puberty and secretion of factors like androgen-binding protein and inhibin B, which support germ cell survival and provide negative feedback to modulate FSH release.[79] Testosterone and FSH synergize, as testosterone alone maintains spermatogenesis in adults but requires FSH initiation, ensuring quantitative sperm output without compromising germ cell quality.[80] In humans, the full spermatogenic cycle spans approximately 64 to 74 days, encompassing about 4.5 epithelial cycles in the seminiferous tubules, with meiosis and spermiogenesis alone taking around 24 days.[81] Each testis produces roughly 100 to 200 million spermatozoa daily, equating to about 1,500 per second per testicle, though only a fraction achieve motility and fertility post-ejaculation.[82] This high-volume production compensates for substantial attrition rates during transit and selection, underscoring the process's efficiency in generating gametes capable of genetic recombination via meiosis, which introduces variability essential for evolutionary adaptation.[83] Disruptions, such as hormonal imbalances or genetic factors, can reduce output, highlighting the system's sensitivity to environmental and physiological cues.[84]Fertility Dynamics and Paternal Investment
Male fertility persists from puberty through advanced age, in contrast to the finite reproductive window in females delimited by menopause, typically occurring around age 50. Spermatogenesis continues lifelong, enabling potential reproduction into the eighth or ninth decade, though with diminishing efficacy due to age-related declines in sperm parameters such as concentration, motility, and morphology. A systematic review indicates that advanced paternal age correlates with reduced semen volume, sperm count, and DNA integrity, alongside increased risks of miscarriage and offspring genetic disorders.[85][86][87] Global trends reveal a decline in sperm concentration and total sperm count, particularly in North America, Europe, Australia, and Asia, with meta-analyses estimating a more than 50% reduction from the 1970s to recent decades, accelerating post-2000. This temporal decrease, observed across over 185 studies involving millions of men, associates with environmental, lifestyle, and endocrine-disrupting factors, though some analyses of unselected populations report stability in recent years. Male-factor infertility contributes to approximately one-third of human infertility cases, often stemming from low sperm production, abnormal function, or delivery blockages.[88][89][90][91] Key modifiable factors impairing male fertility include obesity, which elevates estrogen and reduces testosterone, thereby lowering sperm quality; tobacco use, which damages sperm DNA; excessive alcohol consumption; and exposure to pollutants like pesticides and plastics. Advancing age exacerbates these via oxidative stress and hormonal shifts, with men over 40 showing heightened infertility risks independent of female partner age. The World Health Organization identifies these lifestyle and environmental elements as primary contributors to subfertility, affecting 11-13% of men aged 15-49.[92][93][94][95] Paternal investment in humans, informed by evolutionary parental investment theory, reflects the asymmetry of gamete costs: males produce abundant, low-cost sperm, fostering strategies favoring quantity over quality in reproduction, while females commit substantial resources to gestation and lactation. This disparity predicts lower obligatory male investment post-conception, with empirical data showing fathers allocating less direct care—typically 20-30% of parental time—compared to mothers, though cultural and economic contexts modulate involvement. In monogamous pair-bonds, male provisioning enhances offspring survival, yet paternity uncertainty and mating opportunities often reduce sustained investment, as evidenced in cross-cultural fertility patterns where male effort correlates with resource availability rather than biological imperatives alone.[96][97][98]Recent Advances and Challenges in Male Reproduction
Global meta-analyses have reported a decline in sperm concentration by approximately 1.4% per year from 1973 to 2011 in Western countries, with similar trends observed in other regions up to 2020, though methodological inconsistencies and geographic variations complicate interpretations. Recent U.S.-based studies from 2025, analyzing semen samples from over 1,000 men without known fertility issues, indicate stable sperm counts over the past two decades, suggesting that reported declines may primarily affect subfertile populations rather than the general male cohort.[90] Advancing paternal age correlates with reduced sperm quality, including lower concentration, motility, and increased DNA fragmentation index (DFI), with significant declines noted after age 40 in large cohort analyses.[99] Environmental and lifestyle factors exacerbate these challenges, including exposure to endocrine-disrupting chemicals like phthalates in plastics, which impair Leydig cell function and testosterone production; chronic stress elevating cortisol and suppressing spermatogenesis; and obesity-linked metabolic disruptions reducing semen volume and motility.[100] Urban pollution and sedentary behaviors further contribute to oxidative stress in semen, detectable via biomarkers like malondialdehyde levels, which correlate with asthenozoospermia in 20-30% of infertile cases.[101] Non-obstructive azoospermia (NOA), affecting 10-15% of infertile men, remains a persistent hurdle, often linked to genetic factors like Y-chromosome microdeletions, with retrieval rates via micro-TESE hovering at 40-50% despite refinements.[102] Advances in diagnostics include AI-assisted semen analysis achieving 95% accuracy in motility assessment, surpassing manual methods, and shear wave elastography for non-invasive testicular fibrosis evaluation, improving prediction of sperm retrieval success in NOA by 20-30%.[101] Robotic-assisted microsurgery for varicocele repair has elevated success rates to 70-80% for semen parameter improvement, minimizing complications compared to open techniques.[101] In vitro fertilization with intracytoplasmic sperm injection (ICSI) now yields 50-60% live birth rates per cycle for male-factor infertility, bolstered by preimplantation genetic testing to mitigate risks from fragmented sperm DNA.[103] Regenerative approaches show promise, with mesenchymal stem cell (MSC) therapies enhancing spermatogenic microenvironment in preclinical models, potentially restoring sperm production in NOA via paracrine effects on Sertoli cells.[104] In vitro spermatogenesis (IVS) using 3D bioprinted scaffolds has progressed to generating functional haploid gametes from human induced pluripotent stem cells (iPSCs), addressing ethical barriers to embryo-derived methods.[105] Mitochondrial research elucidates energy deficits in spermatogenesis, with targeted antioxidants improving motility by 15-20% in oxidative stress cases.[106] Male contraception innovations include the ADAM hydrogel, a vas-occlusive agent demonstrating safety in first-in-human trials as of April 2025, offering reversible occlusion without surgery.[107] The non-hormonal pill YCT-529, blocking retinoic acid receptors to halt spermiogenesis, completed phase I safety testing in July 2025 with no serious adverse events in 20 participants.[108] Hormonal options, such as Nestorone-testosterone transdermal gels, achieve 95-99% efficacy in suppressing spermatogenesis in phase II trials, though side effects like acne limit adoption.[109] Standardization efforts, including international core outcome sets for infertility trials, aim to reduce heterogeneity and enhance comparability across studies.[110]Evolutionary Origins
Anisogamy and the Emergence of Males
Anisogamy refers to the dimorphism in gamete size and production strategy between the two sexes, where one sex produces numerous small gametes (spermatozoa or microgametes) and the other produces fewer large gametes (ova or macrogametes). This distinction defines the sexes across most anisogamous species, with males conventionally identified as the producers of the smaller, mobile gametes optimized for quantity and fusion success, while females invest in larger gametes provisioning zygotic development.[111] The emergence of males as a distinct sex traces to this gametic asymmetry, which arose evolutionarily from ancestral isogamous systems where gametes were of uniform size.[112] The prevailing model for anisogamy's origin, proposed by Parker, Baker, and Smith in 1972, posits disruptive selection acting on gamete size in an isogamous population.[111] In this framework, intermediate-sized gametes face fitness disadvantages: smaller gametes achieve higher fertilization rates through increased numbers and motility, exploiting larger gametes for zygote formation, while larger gametes enhance offspring viability by providing more resources, but at the cost of reduced quantity.[113] This selection pressure bifurcates the population into "male" and "female" strategies, with males evolving as the lineage specializing in cheap, high-volume gamete production to maximize mating opportunities.[111] Mathematical simulations confirm that zygote survival probability, which rises nonlinearly with size (due to factors like nutrient reserves and protection against environmental hazards), is essential for stabilizing this dimorphism.[114] Empirical evidence supports this transition in diverse taxa, including volvocine algae where isogamy grades into anisogamy, and early-diverging fungi like Allomyces, demonstrating gamete size dimorphism evolving via similar selective dynamics.[115] In animals, anisogamy's establishment around 1-1.2 billion years ago in eukaryotic lineages marked the origin of males as a sex defined by minimal per-gamete investment, facilitating genetic recombination while shifting provisioning burdens to females.[116] Alternative models, such as those invoking gametic conflict or hermaphroditic intermediates, refine but do not supplant the disruptive selection core, as they still hinge on trade-offs in gamete competition and zygote fitness.[117] Consequently, male emergence fundamentally alters reproductive dynamics, prioritizing fertilization efficiency over parental resources in male evolution.[118]Sexual Selection and Dimorphism
Sexual selection, as articulated by Charles Darwin in The Descent of Man (1871), refers to evolutionary pressures arising from competition for mates and mate choice, which can produce sexual dimorphism—morphological differences between males and females beyond those required for reproduction.[119] In humans, this manifests primarily as greater male size and strength, with adult males averaging 7-15% taller and possessing 50-60% more upper-body muscle mass than females, traits linked to intrasexual competition where males vie for reproductive access.[120] [121] Comparative studies across primates indicate that such dimorphism correlates with polygynous mating systems, where dominant males monopolize multiple females, a pattern evidenced in early hominid fossils showing higher dimorphism (up to 50% body mass difference) that moderated over time as human mating shifted toward relative monogamy.[122] [121] Male-biased dimorphism in humans stems from anisogamy—the disparity in gamete investment—driving males toward riskier strategies, including physical contests that select for robust skeletons, larger canines in ancestral forms, and enhanced testosterone-driven musculature.[123] Empirical data from modern populations reveal consistent preferences in mate choice: women rate taller, more muscular men as more attractive, correlating with perceived dominance and health, while male preferences emphasize female fertility cues like waist-to-hip ratio, though less intensely.[124] Facial dimorphism also varies geographically, with stronger masculine features (e.g., broader jaws) in some populations potentially reflecting local sexual selection intensities, though natural selection for climate adaptation confounds pure sexual signals.[125] Evidence against purely natural selection explanations includes the exaggeration of costly traits like male beard growth and deeper voices, which signal genetic quality but impose metabolic burdens, aligning with handicap principle models where only high-fitness males afford such displays.[124] Fossil records of Australopithecus and early Homo species document a decline in canine dimorphism alongside encephalization, suggesting cultural or provisioning factors mitigated intense male competition, yet persistent body size gaps indicate ongoing sexual selection.[121] Recent analyses confirm that human dimorphism exceeds predictions from solely ecological pressures, supporting sexual selection's role in shaping male morphology for competitive advantage.[120] While debates persist on the balance between monogamy and polygyny in human history—evidenced by genetic data showing modest Y-chromosome bottlenecks—the consensus from cross-species comparisons upholds sexual selection as the primary driver of observed dimorphism.[122][123]Adaptive Strategies in Male Evolution
In evolutionary biology, male adaptive strategies primarily stem from anisogamy, the disparity in gamete size and investment where males produce vast quantities of small, inexpensive sperm, contrasting with females' fewer, resource-intensive eggs. This asymmetry, formalized in Bateman's principles from 1948 fruit fly experiments, demonstrates that male reproductive success exhibits greater variance and benefits disproportionately from multiple matings compared to females, driving selection for tactics that secure access to more partners rather than intensive parental care.[126][127] Empirical measures of Bateman gradients across species, including birds, mammals, and insects, consistently reveal steeper slopes in males, quantifying how additional mates yield compounding fitness gains for them.[128] Intrasexual selection shapes male strategies through direct competition, favoring traits and behaviors that enable dominance over rivals, such as increased body size, weaponry (e.g., antlers in deer or horns in beetles), or aggressive displays. In rodents and primates, genomic evidence indicates accelerated adaptive evolution in male-biased genes linked to postcopulatory competition, like sperm production and morphology, under intense sperm competition.[129] Male-male contests often escalate to physical confrontations, with winners gaining priority access to females, as observed in elephant seals where dominant bulls monopolize harems, achieving up to 80-90% of matings in breeding colonies.[130] This competitive emphasis correlates with higher male mortality risks from fights, underscoring the trade-off where mating effort supplants survival in male life-history allocation.[131] Intersexual selection complements competition by rewarding males who attract female choice through elaborate signals of genetic quality or resources, such as vibrant plumage in birds or provisioning in some insects. Sexual selection theory posits these displays evolve via runaway processes or handicap principles, where costly signals (e.g., peacock tails) reliably indicate viability, as handicap models predict only high-fitness males can afford the energetic or predation burdens.[132] In humans, cross-cultural data from 33 nonindustrial societies show high-status males—via hunting prowess or resource control—secure elevated reproductive success, often through polygynous arrangements, aligning with ancestral environments where status signaled provisioning capacity.[133] Contextual plasticity further refines strategies; for instance, male rodents exposed to female cues pre-mating enhance lifetime reproductive output by adjusting sperm allocation or courtship vigor.[134] These strategies manifest in sex-biased gene expression turnover, with sexual selection accelerating fixation of advantageous male-specific alleles, as seen in Drosophila where polyandry intensifies selection on seminal proteins.[135] While female strategies emphasize choosiness and quality, male tactics prioritize opportunity maximization, explaining pervasive polygyny in mammals (over 90% of species) and the evolutionary primacy of mating variance in males.[136] Challenges like sexual conflict arise when male coercion tactics (e.g., forced copulations in ducks) evolve, but empirical gradients affirm that cooperative signaling often prevails under mutual interests.[137] Overall, male evolution reflects causal pressures from gametic economics, yielding risk-prone, competitive adaptations empirically validated across taxa.Health, Mortality, and Longevity
Leading Causes of Male Mortality
In developed countries, cardiovascular diseases constitute the foremost cause of male mortality, surpassing other categories in absolute numbers. In the United States, heart disease accounted for the highest number of male deaths in 2023, with an age-adjusted death rate of approximately 200 per 100,000 males, reflecting persistent sex disparities driven by factors such as higher prevalence of hypertension and coronary artery disease in men.[138] [139] Malignant neoplasms rank second globally and in the U.S., with males experiencing elevated rates for lung, prostate, and colorectal cancers; ischemic heart disease and stroke together claimed over 16% of male deaths worldwide in recent estimates, though male-specific burdens are amplified by smoking and occupational exposures.[140] [141] Unintentional injuries, encompassing motor vehicle accidents, falls, and poisonings, emerge as a leading cause among younger males, ranking third in the U.S. for 2023 with rates 2-3 times higher than in females due to behavioral patterns including riskier driving and hazardous occupations.[138] [142] Suicide rates further underscore male vulnerabilities, consistently 3-4 times higher than female rates across age groups in high-income nations, contributing significantly to the sex gap in life expectancy through mechanisms like completed attempts via more lethal means.[143] [144] Chronic respiratory diseases and diabetes mellitus follow, with males showing disproportionate impacts from chronic obstructive pulmonary disease linked to historical smoking disparities.[145] Globally, noncommunicable diseases dominate male mortality, accounting for over 70% of deaths in 2021, yet external causes like interpersonal violence and road injuries disproportionately affect males, particularly in low- and middle-income regions where they comprise up to 15% of male deaths under age 70.[140] Liver cirrhosis, often tied to alcohol consumption patterns more prevalent among males, ranks higher in male-specific lists, as evidenced by its position as the ninth leading cause in U.S. males for 2023.[139] These patterns persist despite advances in medical interventions, highlighting causal roles of modifiable risks such as substance use and injury-prone activities, which empirical data link to shorter male longevity across cohorts.[146]Sex-Specific Health Vulnerabilities
Males demonstrate heightened vulnerability to a range of infectious diseases compared to females, attributable in part to sex differences in immune responses, where females generally mount stronger adaptive immunity via estrogen-mediated effects and the presence of two X chromosomes. [147] [148] For instance, epidemiological data from the COVID-19 pandemic revealed males experiencing approximately 45% higher in-hospital mortality risk, linked to weaker interferon responses and higher viral loads. [149] [150] Similarly, males show greater susceptibility to bacterial infections, such as those caused by Leishmania species leading to visceral leishmaniasis, due to reduced immune efficiency rather than exposure differences alone. [151] [152] Cardiovascular disease manifests earlier and more lethally in males, contributing to divergent long-term mortality trends; historical data indicate men's greater uptake of smoking and biological predispositions, including androgen influences on lipid profiles, exacerbate this risk. [153] Recent genomic analyses link age-related loss of the Y chromosome (LOY) in blood cells to cardiac fibrosis, heart failure, and elevated fatal heart attack incidence, with LOY detected in up to 40% of men over 80 and correlating with shortened lifespan. [154] [155] This mosaicism impairs endothelial function and promotes inflammation, independent of traditional risk factors like hypertension. [156] Certain cancers pose amplified risks for males, with prostate cancer incidence peaking in later decades and Y chromosome genes implicated in fertility-related and oncogenic pathways. [157] LOY further heightens susceptibility to solid tumors, including prostate, bladder, and colorectal cancers, as well as Alzheimer's disease, through mechanisms like unchecked cellular proliferation and immune dysregulation. [158] [159] Males also face disproportionate burden from X-linked recessive disorders, such as hemophilia and Duchenne muscular dystrophy, owing to hemizygosity on the single X chromosome, resulting in full phenotypic expression absent a second allele for compensation. [160] Behavioral factors intertwined with biological drivers, such as testosterone-elevated risk-taking, amplify male vulnerabilities to trauma and occupational hazards, with men comprising over 90% of workplace fatalities in high-risk sectors like construction and mining as of 2023 data. [161] [162] This aligns with empirical patterns of excess male mortality from external causes, including road injuries and violence, persisting across lifespans and regions. [163] [142] While social norms influence health-seeking, underlying physiological sex differences in stress responses and pain tolerance contribute to delayed diagnoses and poorer outcomes in chronic conditions. [164]Factors Influencing Male Lifespan
Globally, males exhibit a shorter average lifespan than females, with life expectancy at birth in 2023 estimated at 70.8 years for males compared to 76.0 years for females, a gap of approximately 5.2 years.[165] In the United States, the disparity stood at 5.3 years in 2023, with males at 75.8 years and females at 81.1 years.[141] This sex difference persists across nearly all populations and historical periods, including during severe famines and epidemics, indicating a robust influence beyond transient environmental conditions.[166] Biological vulnerabilities contribute significantly, as males demonstrate higher mortality rates from most major causes at any given age, potentially rooted in genetic programming evident from conception.[167][168] Genetic and physiological factors underpin much of the male disadvantage. Females possess two X chromosomes, offering genetic redundancy that mitigates the impact of deleterious mutations accumulating over time, whereas the male XY configuration exposes them to greater risk from X-linked harmful variants.[169] Genetic associations with longevity traits appear stronger in females, influencing survival disparities.[170] Hormonally, higher testosterone levels in males correlate inversely with all-cause and cardiovascular mortality in observational data, yet may exacerbate vulnerabilities through promotion of risk-prone physiology or interactions with disease pathways like inflammation and senescence.[171] Females exhibit superior immune responses, reducing susceptibility to infections and certain chronic conditions, though this advantage can manifest as higher rates of autoimmune disorders later in life.[153] These innate differences interact with aging, amplifying male mortality in cardiovascular and respiratory domains.[172] Behavioral patterns further widen the gap, with males disproportionately engaging in high-risk activities. Men historically and currently smoke at higher rates, a factor that shortens male life expectancy by an average of nine years, far exceeding female losses from the habit.[173] Excessive alcohol consumption, more prevalent among males, compounds mortality risks, particularly when combined with smoking, though modest drinking shows minimal net benefit offset by other behaviors.[174] Risk-taking behaviors, including dangerous occupations, reckless driving, and violence, elevate accidental and injury-related deaths, which disproportionately affect males and are preventable.[175] Males also seek preventive healthcare less frequently, delaying interventions for conditions like cancer and heart disease.[174] The recent widening of the longevity gap in some regions stems from rising male-specific preventable deaths, such as from opioids, suicides, and gun violence.[176] Evolutionary and socio-environmental dynamics reinforce these patterns. In resource-scarce or high-infection settings, male senescence accelerates faster, suggesting adaptive trade-offs favoring early reproduction over extended lifespan.[177] Occupational exposures in male-dominated fields, such as mining and construction, contribute to chronic injuries and diseases like pneumoconiosis.[174] While interventions targeting behaviors—such as smoking cessation and safety regulations—have narrowed the gap historically, persistent biological baselines limit convergence, with projections indicating sustained female advantages absent major shifts.[178][172]Psychological and Behavioral Dimensions
Empirical Sex Differences in Cognition
Empirical studies indicate that males and females exhibit similar average levels of general cognitive ability, as measured by the g-factor, with meta-analyses showing no significant overall sex difference in general intelligence.[179] However, consistent differences emerge in specific cognitive domains, with males showing advantages in spatial reasoning and certain quantitative tasks, while females demonstrate superior performance in verbal fluency and episodic memory.[180] [181] These patterns persist across age groups, including into advanced age, where sex differences in memory and executive function remain evident even among octogenarians.[182] Males display greater variability in cognitive performance compared to females, a phenomenon supported by multiple meta-analyses and large-scale studies, resulting in higher proportions of males at both high and low extremes of intelligence distributions.[183] [184] This greater male variability extends beyond IQ to traits like time preferences, risk-taking, and social preferences, potentially contributing to observed sex disparities in fields requiring extreme cognitive ability.[185] [186] In contrast, female performance clusters more toward the mean, which aligns with narrower variance in cognitive test scores across standardized assessments like the Wechsler Intelligence Scale.[187] In spatial cognition, males consistently outperform females, particularly in mental rotation tasks, with meta-analyses confirming a moderate to large effect size that emerges early in schooling and persists in STEM experts.[188] [189] This advantage is linked to sex differences in visual-spatial working memory and may underpin male edges in mechanical reasoning and object visualization.[190] [191] Females, however, show strengths in verbal domains, including phonemic fluency, verbal recall, and recognition, with a stable female advantage observed over decades in meta-analytic syntheses spanning 50 years of data.[192] [181] Earlier meta-analyses reported a small overall female superiority in verbal ability (effect size d = +0.11), though some trends suggest a narrowing gap in recent cohorts.[193] Mathematical cognition reveals minimal mean differences between sexes, with recent neuroimaging and behavioral studies emphasizing similarities in neural processing during early development and overall aptitude.[194] [195] Nonetheless, male advantages appear in cognitive reflection tasks involving numerical reasoning, where meta-analyses report higher male scores, particularly on numerical rather than verbal variants.[196] Greater male variability in math performance also contributes to overrepresentation of males at the upper tail, as evidenced in large-scale assessments.[197] These domain-specific differences are not fully attributable to socialization, as they align with patterns observed in non-human primates and early childhood, suggesting biological underpinnings including prenatal hormone influences.[198] [180]Personality and Behavioral Patterns
Males exhibit distinct patterns in personality traits as measured by the Big Five model, with meta-analyses showing consistent sex differences across large samples and cultures. In Neuroticism, males score lower on average, indicating greater emotional stability, while females score higher, particularly on facets like anxiety and vulnerability.[199] [200] In Agreeableness, males tend to score lower, reflecting reduced tendencies toward compassion and cooperation, a pattern observed in international comparisons.[201] For Extraversion, differences vary by facet: males score higher on assertiveness and excitement-seeking but lower on warmth and gregariousness.[199] These traits show moderate effect sizes (d ≈ 0.2–0.5) and hold after controlling for measurement artifacts, with heritability estimates not moderated by sex.[202] Behaviorally, males display higher rates of physical aggression, as evidenced by meta-analytic reviews of real-world settings, where the sex difference narrows under provocation but remains significant (d ≈ 0.4–0.6).[203] This manifests in adolescent and adult violence, with males committing the majority of homicides and assaults; for instance, U.S. data indicate males account for 81% of arrests for violent crimes.[204] [205] Criminality patterns reinforce this, with males comprising 75–83% of offenders in violent and sexual crimes globally, driven by factors like impulsivity rather than opportunity alone.[206] [207] Risk-taking behaviors further differentiate males, who engage more frequently in dangerous activities across domains like driving, substance use, and financial gambles, per a meta-analysis of 150 studies showing a consistent male advantage (d ≈ 0.13–0.50, larger in real-world than lab settings).[208] Competitiveness aligns similarly, with experimental studies revealing males enter tournaments more readily and perform better under competitive incentives, while females opt out more often despite equal ability; this gap emerges in childhood and persists, influencing career and economic outcomes.[209] [210] These patterns are empirically robust, with effect sizes stable over decades, though cultural variations exist—stronger in individualistic societies for some traits.[211]Evolutionary Underpinnings of Differences
Parental investment theory, proposed by Robert Trivers in 1972, posits that anisogamy—the differing sizes and investments in gametes—leads to fundamental sex differences in reproductive strategies, with females committing greater resources to each offspring, fostering choosiness in mate selection, while males prioritize quantity of matings, promoting intrasexual competition and riskier behaviors.[212] This framework explains observed male-biased patterns in aggression and risk-taking, as males historically competed for access to female reproductive potential, with higher variance in male reproductive success incentivizing bold, competitive traits.[213] Empirical data across species, including humans, show males exhibiting greater physical aggression, peaking in young adulthood when mating competition intensifies, a pattern consistent with sexual selection pressures rather than solely cultural influences.[214] In cognition, evolutionary pressures from ancestral divisions of labor underpin sex differences, such as the hunter-gatherer hypothesis, which attributes male advantages in mental rotation and navigation to foraging demands involving large-scale spatial tracking for hunting, while female strengths in object location memory align with gathering tasks requiring precise recall of resource locations.[215] Cross-cultural studies, including data from over 40 countries via large-scale internet surveys, confirm these patterns hold universally, with males outperforming in three-dimensional spatial tasks and females in landmark-based memory, supporting adaptive specialization over socialization alone.[216] Greater male variability in cognitive traits, including intelligence, further aligns with evolutionary models where X-chromosome effects and sexual selection amplify male extremes, leading to more males at both high and low ends of distributions, as evidenced in meta-analyses of test scores showing variance ratios favoring males by 1.1 to 1.2.[184] Personality differences, such as males scoring higher on extraversion facets like assertiveness and sensation-seeking in Big Five inventories, trace to selection for status competition and mate attraction, with meta-analyses revealing effect sizes (d ≈ 0.4-0.6) stable across cultures and predictive of mating success.[217] These traits manifest in behavioral patterns like increased male risk-taking in economic games and real-world domains (e.g., financial investing, extreme sports), where sexual selection favors variance in male strategies to outcompete rivals, contrasting with female risk-aversion tied to higher offspring investment costs.[218] While evolutionary psychology faces criticism for potential overreach, its predictions are bolstered by converging evidence from endocrinology (e.g., testosterone correlations with aggression) and comparative primatology, distinguishing it from less empirically grounded social constructivist accounts prevalent in some academic discourse.[219]Controversies and Societal Implications
Debates on Sex Binary vs. Spectrum Claims
Biological sex in humans is defined by the type of gametes produced: small, mobile sperm by males or large, immobile ova by females, establishing a binary classification rooted in anisogamy across sexually reproducing species including mammals.[220] This definition aligns with evolutionary principles where reproductive roles determine sex, with no observed third gamete type in humans capable of independent fertilization.[220] Proponents of the binary view, such as evolutionary biologists, emphasize that variations in secondary traits like chromosomes or hormones do not alter the fundamental reproductive dichotomy, as even individuals with disorders of sexual development (DSDs) typically retain alignment with one sex or the other in gamete production capacity.[221] Claims that sex constitutes a spectrum often arise from observations of DSDs, chromosomal anomalies (e.g., XXY Klinefelter syndrome or XO Turner syndrome), or intermediate hormonal profiles, arguing these blur the male-female divide.[222] For instance, articles in Scientific American have asserted that biological complexity, including intersex conditions affecting up to 1.7% of births under broad definitions, challenges the binary as overly simplistic.[223] However, rigorous epidemiological reviews adjust this figure downward; Leonard Sax's 2002 analysis of peer-reviewed data estimates true DSD prevalence—cases involving ambiguous genitalia or mismatched gonadal and chromosomal sex—at approximately 0.018%, nearly 100 times lower than inflated estimates that include non-reproductive traits like late-onset congenital adrenal hyperplasia.[224] These DSDs represent developmental disorders rather than evidence of a spectrum, as affected individuals do not produce a novel gamete type but exhibit anomalies within the binary framework, often requiring medical intervention for fertility or health.[221] Critics of spectrum arguments, including publications in BioEssays, contend that conflating sex with phenotypic variability or gender roles introduces non-biological elements, driven partly by ideological pressures in academia and media where empirical reproductive criteria are downplayed.[220] Empirical data from genetics and endocrinology reinforce dimorphism: over 99.98% of humans conform to typical male (XY, testes, sperm) or female (XX, ovaries, ova) patterns, with DSDs not constituting additional sexes but rare exceptions akin to other congenital anomalies like polydactyly, which do not negate the human digit norm of five per hand.[224][221] In mammals, including humans, sex determination cascades from gamete-defining genes like SRY on the Y chromosome, leading to binary outcomes in gonadal development, underscoring causal realism in sex classification over probabilistic spectra.[220] Debates persist in policy contexts, such as sports eligibility or medical research, where binary adherence ensures fairness and precision—e.g., NIH guidelines treat sex as a dimorphic variable for clinical trials—while spectrum advocacy risks overlooking causal health disparities tied to sex-specific biology.[8] Peer-reviewed syntheses affirm the binary as the operative model for human biology, with spectrum claims lacking substantiation from reproductive endpoints and often reflecting interpretive biases rather than data-driven conclusions.[220][221]Sex vs. Gender Distinctions
Biological sex in humans is defined by the type of gametes produced, with males characterized by the production of small, mobile gametes known as sperm, in contrast to females who produce large, immobile gametes or ova.[220] This binary classification is rooted in reproductive function and is typically determined by chromosomal composition, where males possess one X and one Y chromosome, with the SRY gene on the Y chromosome initiating male gonadal development around the 6th week of embryonic life.[23] Rare disorders of sex development (DSDs), affecting approximately 0.018% to 1.7% of births depending on criteria, represent developmental anomalies rather than additional sexes, as they do not produce a third gamete type.[220] The distinction between sex and gender emerged in the mid-20th century, primarily through the work of psychologist John Money in the 1950s, who introduced "gender role" to describe psychosocial behaviors and identities separate from biological sex.[225] Money posited that gender identity is primarily learned through socialization, independent of chromosomal or anatomical sex, influencing early interventions for intersex conditions by advocating surgical alignment with perceived gender.[226] This framework gained traction in social sciences and medical practices, but Money's theories faced empirical refutation, notably in the case of David Reimer, a genetically XY male subjected to gender reassignment after a botched circumcision in 1966 and raised as "Brenda." Despite intensive socialization efforts, Reimer exhibited male-typical behaviors and distress, ultimately rejecting the female identity, undergoing reversal surgery, and dying by suicide in 2004 at age 38.[227] The Reimer outcome, documented in John Colapinto's 2000 book As Nature Made Him, undermined claims of gender malleability, highlighting innate biological influences on identity.[228] Contemporary usage often frames gender as a social construct or spectrum of identities decoupled from sex, a perspective prevalent in fields like gender studies, which exhibit systemic biases toward constructivist interpretations over biological data.[225] However, twin studies and neuroimaging reveal substantial genetic and hormonal contributions to behavioral sex differences, with heritability estimates for traits like aggression and spatial cognition ranging from 30-50%, consistent across cultures and evident prenatally via hormone exposure effects on brain dimorphism.[229] For instance, exposure to prenatal testosterone correlates with male-typical play preferences in both sexes, as shown in longitudinal studies of children with congenital adrenal hyperplasia.[230] These findings indicate that while cultural factors modulate expression, core differences in personality, cognition, and mate preferences stem from sex-linked biology rather than socialization alone, challenging strict sex-gender dichotomies as causal fictions unsupported by causal mechanisms in development.[229] Sources promoting pure social construction, often from ideologically aligned academic outlets, underemphasize such evidence, whereas evolutionary biology and endocrinology prioritize dimorphic reproductive roles as the foundational driver.[220]Empirical Realities of Male Disparities
In the United States, males have a life expectancy of 75.8 years compared to 81.1 years for females as of 2023, resulting in a gender gap of 5.3 years primarily driven by higher male mortality from external causes such as accidents, homicides, and suicides.[231] [232] This disparity persists globally, with similar patterns observed in OECD countries where behavioral risks like smoking and occupational hazards contribute causally to elevated male death rates at younger ages.[233] Suicide rates underscore a stark male vulnerability, with males dying by suicide at approximately four times the rate of females in 2023, equating to an age-adjusted rate for males around 25 per 100,000 versus under 7 for females.[234] [235] This ratio has held steady over two decades, linked empirically to factors including higher male impulsivity, substance abuse prevalence, and lower help-seeking behaviors, rather than mere reporting differences.[236] Males also dominate occupational fatalities, comprising over 90% of the 5,283 work-related deaths recorded in 2023, concentrated in high-risk sectors like construction and transportation where male labor participation exceeds 90%.[237] Educational outcomes reveal boys lagging behind girls from early schooling onward. In OECD PISA 2022 assessments, girls outperformed boys in reading by an average of 27 points across countries, while the male advantage in mathematics averaged only 14 points, contributing to higher female postsecondary enrollment.[238] [239] By 2023, women accounted for 58% of U.S. college enrollees, with 47% of women aged 25-34 holding bachelor's degrees versus 37% of men, patterns attributed to boys' lower average conscientiousness and reading proficiency rather than systemic discrimination.[240] [241] Socioeconomic indicators further highlight male overrepresentation in adverse outcomes. Around 60% of unsheltered homeless individuals in the U.S. are male, with 2023 point-in-time counts showing men comprising 59.6% of the total homeless population, often tied to higher rates of severe mental illness, addiction, and economic displacement in male-dominated low-skill trades.[242] [243] In the criminal justice system, males constitute over 93% of the federal and state prison population as of 2023, with incarceration rates for men roughly 14 times higher than for women, reflecting empirically higher male involvement in violent and property crimes driven by biological and developmental factors like testosterone-linked aggression.[244] [245]| Disparity Area | Male Statistic (2023, U.S.) | Female Comparison | Source |
|---|---|---|---|
| Life Expectancy | 75.8 years | 81.1 years (5.3-year gap) | CDC[231] |
| Suicide Rate | ~4x higher (age-adjusted ~25/100k) | Baseline rate | CDC[234] |
| Occupational Fatalities | >90% of 5,283 deaths | <10% | BLS[237] |
| College Enrollment Share | 42% of undergraduates | 58% | NCES/Pew[241] |
| Homeless Population Share | 59.6%-60% | 39.2%-40% | HUD[243] |
| Prison Population Share | >93% | <7% | BJS/Sentencing Project[245] |
References
- https://en.wiktionary.org/wiki/masculus