Recent from talks
Nothing was collected or created yet.
Viral vector
View on Wikipedia

A viral vector is a modified virus designed to deliver genetic material into cells. This process can be performed inside an organism or in cell culture. Viral vectors have widespread applications in basic research, agriculture, and medicine.
Viruses have evolved specialized molecular mechanisms to transport their genomes into infected hosts, a process termed transduction. This capability has been exploited for use as viral vectors, which may integrate their genetic cargo—the transgene—into the host genome, although non-integrative vectors are also commonly used. In addition to agriculture and laboratory research, viral vectors are widely applied in gene therapy: as of 2022, all approved gene therapies were viral vector-based. Further, compared to traditional vaccines, the intracellular antigen expression enabled by viral vector vaccines offers more robust immune activation.
Many types of viruses have been developed into viral vector platforms, ranging from retroviruses to cytomegaloviruses. Different viral vector classes vary widely in strengths and limitations, suiting some to specific applications. For instance, relatively non-immunogenic and integrative vectors like lentiviral vectors are commonly employed for gene therapy. Chimeric viral vectors—such as hybrid vectors with qualities of both bacteriophages and eukaryotic viruses—have also been developed.
Viral vectors were first created in 1972 by Paul Berg. Further development was temporarily halted by a recombinant DNA research moratorium following the Asilomar Conference and stringent National Institutes of Health regulations. Once lifted, the 1980s saw both the first recombinant viral vector gene therapy and the first viral vector vaccine. Although the 1990s saw significant advances in viral vectors, clinical trials had a number of setbacks, culminating in Jesse Gelsinger's death. However, in the 21st century, viral vectors experienced a resurgence and have been globally approved for the treatment of various diseases. They have been administered to billions of patients, notably during the COVID-19 pandemic.
Characteristics
[edit]
Viruses, infectious agents composed of a protein coat that encloses a genome, are the most numerous biological entities on Earth.[1][2] As they cannot replicate independently, they must infect cells and hijack the host's replication machinery in order to produce copies of themselves.[2] Viruses do this by inserting their genome—which can be DNA or RNA, either single-stranded or double-stranded—into the host.[3] Some viruses may integrate their genome directly into that of the host in the form of a provirus.[4]
This ability to transfer foreign genetic material has been exploited by genetic engineers to create viral vectors, which can transduce the desired transgene into a target cell.[2] Viral vectors consists of three components:[5][6]
- A protein capsid and sometimes an envelope that encapsidates the genetic payload. This determines the range of cell types that the vector infects, termed its tropism.
- A genetic payload: the transgene that results in the desired effect when expressed.
- A "regulatory cassette" that controls transgene expression, whether integrated into a host chromosome or as an episome. The cassette comprises an enhancer, a promoter, and auxiliary elements.
Applications
[edit]
Basic research
[edit]Viral vectors are routinely used in a basic research setting and can introduce genes encoding, for instance, complementary DNA, short hairpin RNA, or CRISPR/Cas9 systems for gene editing.[8] Viral vectors are employed for cellular reprogramming, like inducing pluripotent stem cells or differentiating adult somatic cells into different cell types.[9] Researchers also use viral vectors to create transgenic mice and rats for experiments.[10] Viral vectors can be used for in vivo imaging via the introduction of a reporter gene. Further, transduction of stem cells can permit the tracing of cell lineage during development.[9]
Gene therapy
[edit]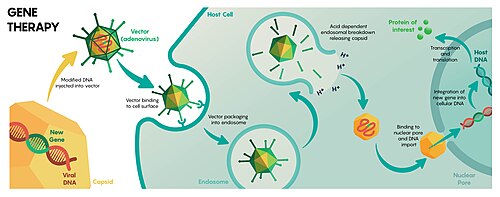
Gene therapy seeks to modulate or otherwise affect gene expression via the introduction of a therapeutic transgene. Gene therapy by viral vectors can be performed by in vivo delivery by directly administering the vector to the patient, or ex vivo by extracting cells from the patient, transducing them, and then reintroducing the modified cells into the patient.[11] Viral vector gene therapies may also be used for plants, tentatively enhancing crop performance or promoting sustainable production.[12]
There are four broad categories of gene therapy: gene replacement, gene silencing, gene addition, or gene editing.[11][13] Relative to other non-integrative gene therapy approaches, transgenes introduced by viral vectors offer multi-year long expression.[14]
Vaccines
[edit]
For use as vaccine platforms, viral vectors can be engineered to carry a specific antigen associated with an infectious disease or a tumor antigen.[15][16] Conventional vaccines are not suitable for protection against some pathogens due to unique immune evasion strategies and differences in pathogenesis.[17] Viral vector-based vaccines, for instance, could eventually offer immunity against HIV-1 and malaria.[18]
While traditional subunit vaccines elicit a humoral response,[19] viral vectors allow for intracellular antigen expression that activates MHC pathways via both direct and crosspresentation pathways. This induces a robust adaptive immune response.[20][21] Viral vector vaccines also have intrinsic adjuvant properties via innate immune system activation and the expression of pathogen-associated molecular patterns, negating the need for any additional adjuvant.[22][15] In addition to a more robust immune response in comparison to other vaccine types, viral vectors offer efficient gene transduction and can target specific cell types.[19] Pre-existing immunity to the virus used as the vector, however, can be a significant issue.[18]
Prior to 2020, viral vector vaccines were widely administered but confined to veterinary medicine.[22] In the global response to the COVID-19 pandemic, viral vector vaccines played a fundamental role and were administered to billions of people, particularly in low and middle-income nations.[23]
Types
[edit]Retroviruses
[edit]Retroviruses—enveloped RNA viruses—are popular viral vector platforms due to their ability to integrate genetic material into the host genome.[2] Retroviral vectors comprise two general classes: gamma retroviral and lentiviral vectors. The fundamental difference between the two are that gamma retroviral vectors can only infect dividing cells, while lentiviral vectors can infect both dividing and resting cells.[24] Notably, retroviral genomes are composed of single-stranded RNA and must be converted to proviral double-stranded DNA, a process known as reverse transcription—before it is integrated into the host genome via viral proteins like integrase.[25]
The most commonly used gammaretroviral vector is a modified Moloney murine leukemia virus (MMLV), able to transduce various mammalian cell types. MMLV vectors have been associated with some cases of carcinogenesis.[26] Gammaretroviral vectors have been successfully applied to ex vivo hematopoietic stem cell to treat multiple genetic diseases.[27]
Lentiviral vectors
[edit]
Most lentiviral vectors are derived from human immunodeficiency virus type 1 (HIV-1), although modified simian immunodeficiency virus (SIV), the feline immunodeficiency virus (FIV), and the equine infectious anaemia virus (EIAV) have also been utilized.[24] As all functional genes are removed or otherwise mutated, the vectors are not cytopathic and can be engineered to be non-integrative.[28]
Lentiviral vectors are able to carry up to 10 kb of foreign genetic material, although 3-4 kb was reported as optimal as of 2023.[24][28] Relative to other viral vectors, lentiviral vectors possess the greatest transduction capacity, due to the formation of a three-stranded "DNA flap" during retro-transcription of the single-strand lentiviral RNA to DNA within the host.[28]
Although largely non-inflammatory,[29] lentiviral vectors can induce robust adaptive immune responses by memory-type cytotoxic T cells and T helper cells.[30] This is largely due to lentiviral vectors' high tropism for dendritic cells, which activate T cells.[30] However, they can infect all types of antigen-presenting cells.[31] Moreover, as they are the only retroviral vectors able to efficiently transduce both dividing and non-dividing cells, make them the most promising vaccine platforms.[31] They have also been trialed as vaccines against cancer.[32]
Lentiviral vectors have been used as in vivo therapies, such as directly treating genetic diseases like haemophilia B and for ex vivo treatments like immune cell modification in CAR T cell therapy.[24] In 2017, the US Food and Drug Administration (FDA) approved tisagenlecleucel, a lentiviral vector, for acute lymphoblastic leukaemia.[33]
Adenoviruses
[edit]
Adenoviruses are double-stranded DNA viruses belonging to the family Adenoviridae.[34][35] Their relatively large genomes, of approximately 30-45 kb, make them ideal candidates for genetic delivery;[34] newer adenoviral vectors can carry up to 37 kb of foreign genetic material.[36] Adenoviral vectors display high transduction efficiency and transgene expression, and can infect both dividing and non-dividing cells.[37]
The adenoviral capsid, an icosahedron, features a fibre "knob" at each of its 12 vertices. These fibre proteins mediate cell entry—greatly affecting efficacy and contribute to its broad tropism—notably via coxsackie–adenovirus receptors (CARs).[34][37] Adenoviral vectors can induce robust innate and adaptive immune responses.[38] Its strong immunogenicity is particularly due to the transduction of dendritic cells (DC), upregulating the expression of both MHC I and II molecules and activating the DCs.[39] They have a strong adjuvant effect, as they display several pathogen-associated molecular patterns.[38] One disadvantage is that pre-existing immunity to adenovirus serotypes is common, reducing efficacy.[37][40] The use of chimpanzee adenoviruses may circumvent this issue.[41]
While the activation of both innate and adaptive immune responses is an obstacle for many therapeutic applications, it makes adenenoviral vectors an ideal vaccine platform.[35] The global response to the COVID-19 pandemic saw the development and use of multiple adenoviral vector vaccines, including Sputnik V, the Oxford–AstraZeneca vaccine, and the Janssen vaccine.[42]
Adeno-associated viruses
[edit]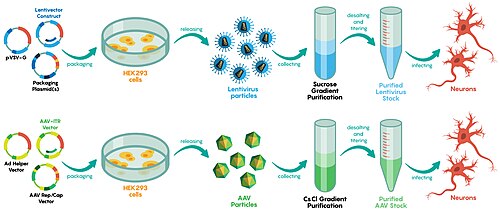
Adeno-associated viruses (AAVs) are relatively small single-stranded DNA viruses belonging to Parvoviridae and, like lentiviral vectors, AAVs can infect both dividing and non-dividing cells.[43] AAVs, however, require the presence of a "helper virus" such as an adenovirus or herpes simplex virus to replicate within the host, although it can do so independently if cellular stress is induced or the helper virus genes are carried by the vector.[44]
AAVs insert themselves into a specific site in the host genome, particularly AAVS1 on chromosome 19 in humans. However, recombinant AAVs have been designed that do not integrate. These are instead stored as episomes that, in non-dividing cells, can last for years.[45] One disadvantage is that they are not able to carry large amounts of foreign genetic materials. Furthermore, the need to express the complementary strand for its single-stranded genome may delay transgene expression.[45]
As of 2020, 11 different AAV serotypes—differing by capsid structure and consequently by tropism—had been identified.[43] The tropism of adeno-associated viral vectors can be tailored by creating recombinant versions from multiple serotypes, termed pseudotyping.[43] Due to their ability to infect and induce longlasting effects within nondividing cells, AAVs are commonly used in basic neuroscience research.[46] Following the approval of the AAV Alipogene tiparvovec in Europe in 2012,[47] in 2017, the FDA approved the first AAV-based in vivo gene therapy—voretigene neparvovec—which treated RPE65-associated Leber congenital amaurosis.[33] As of 2020, 230 clinical trials using AAV-based treatments were either underway or had been completed.[47]
Vaccinia
[edit]
Vaccinia virus, a poxvirus, is another promising candidate for viral vector development.[48] Its use as the smallpox vaccine—first reported by Edward Jenner in 1798—led to the eradication of smallpox and demonstrated vaccinia as safe and effective in humans.[49][48] Moreover, manufacturing procedures developed to mass-produce smallpox vaccine stockpiles may expedite vaccinia viral vector production.[50]
Vaccinia possesses a large DNA genome and can consequently carry up to 40 kb of foreign DNA.[49][51][52][51] Further, vaccinia are unlikely to integrate into the host genome, decreasing the chance of carcinogenesis.[51] Attenuated strains—replicating and non-replicating—have been developed.[49] Although widely characterized due to its use against smallpox, as of 2019 the function of 50 percent of the vaccinia genome was unknown. This may lead to unpredictable effects.[52]
As a vaccine platform, vaccinia vectors display highly effective transgene expression and create a robust immune response.[50] The virus fast-acting: its life cycle produces mature progeny vaccinia within 6 hours, and has three viral spread mechanisms.[52] Vaccinia also has an adjuvant effect, activating a strong innate response via toll-like receptors.[50] A significant disadvantage that can reduce its efficacy, however, is pre-existing immunity against vaccinia in those who received the smallpox vaccine.[50]
Herpesviruses
[edit]
Of the nine herpesviruses that infect humans, herpes simplex virus 1 (HSV-1) is the most well characterized and most commonly used as a viral vector.[53] HSV-1 offers several advantages: it has broad tropism and can deliver therapeutics via specialized expression systems.[54] Moreover, HSV-1 can cross the blood brain barrier if medically-disrupted, enabling it to target neurological diseases. Also, HSV-1 does not integrate into the host genome and can carry large amounts of foreign DNA. The former feature prevents harmful mutagenesis, as can occur with retroviral and adeno-associated vectors. Replication-deficient strains have been developed.[55]
In 2015, talimogene laherparepvec—an HSV-1 vector that triggers an anti-tumor immune response—was approved by the FDA to treat melanoma.[56] As of 2020, HSV-1 vectors have been experimentally applied against sarcomas and cancers of the brain, colon, prostate, and skin.[57]
Cytomegalovirus (CMV), a herpesvirus, has also been developed for use as a viral vector.[58] CMV can infect most cell types and can thus proliferate throughout the body. Although a CMV-based vaccine provided significant immunity against SIV—closely related to HIV—in macaques, development of CMV as a reliable vector was reported to still be in early stages as of 2020.[59][60]
Plant viruses
[edit]Plant viruses are also engineered viral vectors for use in agriculture, horticulture, and biologic production.[61] These vectors have been employed for a range of applications, from increasing the aesthetic quality of ornamental plants to pest biocontrol, rapid expression of recombinant proteins and peptides, and to accelerate crop breeding.[62] The use of engineered plant viruses has been proposed to enhance crop performance and promote sustainable production.[12]
Replicating virus-based vectors are typically used.[63] RNA viruses used for monocots include wheat streak mosaic virus and barley stripe mosaic virus and, for dicots, tobacco rattle virus. Single-stranded DNA viruses like geminiviruses have also been utilized.[63] Viral vectors can be administered to plants via several pathways termed "agro-inoculation", including via rubbing, a biolistic delivery system, agrospray, agroinjection, and even via insect vectors.[64][62] However, Agrobacterium-mediated delivery of viral vectors—in which bacteria are transformed with plasmid DNA encoding the viral vector construct—is the most common approach.[65]
Bacteriophages
[edit]Chimeric vectors combining both bacteriophages and eukaryotic viruses have been developed and are capable of infecting eukaryotic cells.[66][67] Unlike eukaryotic virus-based vectors, such bacteriophage vectors have no innate tropism for eukaryotic cells, allowing them to be engineered to be highly specific for cancer cells.[68]
Bacteriophage vectors are also commonly used in molecular biology.[69] For instance, bacteriophage vectors are used in phage-assisted continuous evolution, promoting rapid mutagenesis of bacteria.[70] Although limited to mycobacteriophages and some phages of gram-negative bacteria, bacteriophages can be used for direct cloning.[71]
Manufacture
[edit]
Viral vector manufacturing methods often vary by vector, although most utilize an adherent or suspension-based system with mammalian cells.[72] For viral vector production on a smaller, laboratory setting, static cell culture systems like Petri dishes are typically used.[73]
Those techniques used in the laboratory are difficult to scale, requiring different approaches on an industrial scale.[72] Large single-use disposable culture systems and bioreactors are commonly used by manufacturers.[72] Vessels such as those with gas permeable surfaces are used to maximize cell culture density and solution transducing units.[72] Depending on the vessel, viruses can be directly isolated from the supernatant or isolated via chemical lysis of the cultured cells or microfluidization.[74] In 2017, The New York Times reported a manufacturing backlog of inactivated viruses, delaying some gene therapy trials by years.[75]
History
[edit]In 1972, Stanford University biochemist Paul Berg developed the first viral vector, incorporating DNA from the lambda phage into the polyomavirus SV40 to infect kidney cells maintained in culture.[76][77][78] The implications of this achievement troubled scientists like Robert Pollack, who convinced Berg not to transduce DNA from SV40 into E. coli via a bacteriophage vector. They feared that introducing the purportedly cancer-causing genes of SV40 would create carcinogenic bacterial strains.[79][80] These concerns and others in the emerging field of recombinant DNA led to the Asilomar Conference of 1975, where attendees agreed to a voluntary moratorium on cloning DNA.[81]
In 1977, the National Institutes of Health (NIH) issued formal guidelines confining viral DNA cloning to rigid BSL-4 conditions, practically preventing such research. However, the NIH loosened these rules in 1979, permitting Bernard Moss to develop a viral vector utilizing vaccinia.[81] In 1982, Moss reported the first use of a viral vector for transient gene expression.[18] The following year, Moss used the vaccinia vector to express a hepatitis B antigen, creating the first viral vector vaccine.[22]
Every realm of medicine has its defining moment, often with a human face attached. Polio had Jonas Salk. In vitro fertilization had Louise Brown, the world's first test-tube baby. Transplant surgery had Barney Clark, the Seattle dentist with the artificial heart. AIDS had Magic Johnson. Now gene therapy has Jesse Gelsinger.
Although a failed gene therapy attempt utilizing wild-type Shope papilloma virus had been made as early as 1972, Martin Cline attempted the first gene therapy utilizing recombinant DNA in 1980. It proved unsuccessful.[83][11] In the 1990s, as genetic diseases were further characterized and viral vector technology improved, there was overoptimism about the capabilities the technology. Many clinical trials proved failures.[84] There were some successes, such as the first effective gene therapy for severe combined immunodeficiency (SCID); it employed a retroviral vector.[11]
However, during a 1999 clinical trial at the University of Pennsylvania, Jesse Gelsinger died from a fatal reaction to an adenoviral vector-based gene therapy.[82][84] It was the first death related to any form of gene therapy.[85] Consequently, the FDA suspended all gene therapy trials at the University of Pennsylvania and investigated 60 others across the US.[85] An anonymous editorial in Nature Medicine noted that it represented a "loss of innocence" for viral vectors.[84] Shortly thereafter, the field's reputation was further damaged when 5 children treated with a SCID gene therapy developed leukemia due to an issue with the retroviral vector.[84][note 1]
Viral vectors experienced a resurgence when they were successfully employed for ex vivo hematopoietic gene delivery in clinical settings.[86] In 2003, China approved the first gene therapy for clinical use: Gendicine, an adenoviral vector encoding p53.[87][88] In 2012, the European Union issued its first approval of a gene therapy, an adeno-associated viral vector.[89] During the COVID-19 pandemic, viral vector vaccines were used to an unprecedented extent: administered to billions of people.[90][22] As of 2022, all approved gene therapies were viral vector-based and over 1000 viral vector clinical trials targeting cancer were underway.[86]
In popular culture
[edit]
In film, viral vectors are often portrayed as unintentionally causing a pandemic and civilizational catastrophe.[91] The 2007 film I Am Legend depicts a cancer-targeting viral vector as unleashing a zombie apocalypse.[92][93] Similarly, a viral vector therapy for Alzheimer's disease in Rise of the Planet of the Apes (2011) becomes a deadly pathogen and causes an ape uprising. Other films featuring viral vectors include The Bourne Legacy (2012) and Resident Evil: The Final Chapter (2016).[94] An advanced form of viral vector vaccine is a critical story element in Jurassic World Dominion (2022), in which it is used to cure a character's genetic disorder and later to stop a man-made ecological crisis.
Notes and references
[edit]Notes
[edit]Citations
[edit]- ^ Pasin, Menzel & Daròs 2019, p. 1010.
- ^ a b c d Labbé, Vessillier & Rafiq 2021, p. 1.
- ^ Kayser et al. 2005, pp. 377–378.
- ^ Barth & Aylward 2024, p. 1.
- ^ Bulcha et al. 2021, pp. 1–2.
- ^ Nomaguchi et al. 2012, p. 1.
- ^ Moen et al. 2012, p. 2.
- ^ Lanigan, Kopera & Saunders 2020, pp. 1, 7.
- ^ a b Sakuma, Barry & Ikeda 2012, p. 612.
- ^ Lanigan, Kopera & Saunders 2020, p. 1.
- ^ a b c d Bulcha et al. 2021, p. 1.
- ^ a b Pasin et al. 2024, p. 1.
- ^ Li et al. 2023, p. 2.
- ^ Sasmita 2019, p. 29.
- ^ a b Wang et al. 2023, p. 1.
- ^ Larocca & Schlom 2011, p. 1.
- ^ Elkashif et al. 2021, p. 1.
- ^ a b c Ura, Okuda & Shimada 2014, p. 625.
- ^ a b Ura, Okuda & Shimada 2014, p. 624.
- ^ McCann et al. 2022, p. 2.
- ^ Ura, Okuda & Shimada 2014, p. 624-625.
- ^ a b c d McCann et al. 2022, p. 1.
- ^ McCann et al. 2022, pp. 1, 6–7.
- ^ a b c d Labbé, Vessillier & Rafiq 2021, p. 2.
- ^ Milone & O'Doherty 2018, pp. 1530–1531.
- ^ Gruntman & Flotte 2018, pp. 1734.
- ^ Gruntman & Flotte 2018, pp. 1733.
- ^ a b c Nemirov et al. 2023, p. 1.
- ^ Nemirov et al. 2023, pp. 1, 4.
- ^ a b Nemirov et al. 2023, pp. 1–2.
- ^ a b Nemirov et al. 2023, p. 4.
- ^ Nemirov et al. 2023, p. 7.
- ^ a b Li & Samulski 2020, p. 255.
- ^ a b c Elkashif et al. 2021, p. 2.
- ^ a b Farhad et al. 2022, p. 2.
- ^ Nemirov et al. 2023, pp. 3–4.
- ^ a b c Ura, Okuda & Shimada 2014, p. 628.
- ^ a b Elkashif et al. 2021, p. 3.
- ^ Elkashif et al. 2021, pp. 3–4.
- ^ Elkashif et al. 2021, p. 8.
- ^ Ewer et al. 2017, p. 3020.
- ^ Elkashif et al. 2021, pp. 10, 11.
- ^ a b c Haggerty et al. 2019, p. 69.
- ^ Haggerty et al. 2019, pp. 69–70.
- ^ a b Haggerty et al. 2019, p. 70.
- ^ Haggerty et al. 2019, pp. 71–74, 78.
- ^ a b Haggerty et al. 2019, p. 75.
- ^ a b Zhang et al. 2021, p. 1578.
- ^ a b c Ura, Okuda & Shimada 2014, p. 626.
- ^ a b c d Ura, Okuda & Shimada 2014, p. 627.
- ^ a b c Kaynarcalidan, Mascaraque & Drexler 2021, p. 1.
- ^ a b c Guo et al. 2019, p. 4.
- ^ Mody et al. 2020, p. 1.
- ^ Mody et al. 2020, pp. 3–4.
- ^ Mody et al. 2020, p. 4.
- ^ Khushalani et al. 2023, p. 1.
- ^ Hromic-Jahjefendic & Lundstrom 2020, p. 631.
- ^ Ura, Okuda & Shimada 2014, p. 631.
- ^ Sasso et al. 2020, p. 10.
- ^ Schaefer et al. 2005, p. 1446.
- ^ Abrahamian, Hammond & Hammond 2020, pp. 513–515.
- ^ a b Pasin, Menzel & Daròs 2019, pp. 1010–1011.
- ^ a b Zaidi & Mansoor 2017, p. 1.
- ^ Abrahamian, Hammond & Hammond 2020, pp. 520–523.
- ^ Abrahamian, Hammond & Hammond 2020, pp. 522–528.
- ^ Petrov, Dymova & Richter 2022, p. 9.
- ^ Pranjol & Hajitou 2015, p. 269.
- ^ Petrov, Dymova & Richter 2022, p. 1.
- ^ Elois et al. 2023, p. 1.
- ^ Abril et al. 2022, p. 11.
- ^ Abril et al. 2022, p. 12.
- ^ a b c d van der Loo & Wright 2016, p. 44.
- ^ Merten et al. 2014, p. 184.
- ^ van der Loo & Wright 2016, p. 45.
- ^ Kolata 2017.
- ^ Travieso et al. 2022, p. 1.
- ^ Lukiw 2023, p. 1.
- ^ Jackson, Symons & Berg 1972, pp. 2904–2909.
- ^ Carmen 1985, pp. 61–62.
- ^ Lukiw 2023, p. 2.
- ^ a b Moss 2013, p. 4220.
- ^ a b Stolberg 1999.
- ^ Wirth, Parker & Ylä-Herttuala 2013, p. 164.
- ^ a b c d e Sheridan 2011, p. 121.
- ^ a b Sibbald 2001, p. 1612.
- ^ a b Bezeljak 2022, pp. 2, 10.
- ^ Wirth, Parker & Ylä-Herttuala 2013, p. 165.
- ^ Bezeljak 2022, p. 23.
- ^ Wirth, Parker & Ylä-Herttuala 2013, pp. 166–167.
- ^ Bezeljak 2022, p. 2.
- ^ Sánchez-Angulo 2023, pp. 1, 16.
- ^ Reuters 2020.
- ^ Feldman & Clayton 2022, pp. 2, 5.
- ^ Sánchez-Angulo 2023, p. 16.
Works cited
[edit]Journal articles
[edit]- Abrahamian P, Hammond RW, Hammond J (2020). "Plant Virus–Derived Vectors: Applications in Agricultural and Medical Biotechnology". Annual Review of Virology. 7 (1): 513–535. doi:10.1146/annurev-virology-010720-054958. PMID 32520661.
- Abril AG, Carrera M, Notario V, Sanchez-Perez A, Villa TG (2022). "The Use of Bacteriophages in Biotechnology and Recent Insights into Proteomics". Antibiotics. 11 (5): 653. doi:10.3390/antibiotics11050653. PMC 9137636. PMID 35625297.
- Barth ZK, Aylward FO (2024). "March of the proviruses". Proceedings of the National Academy of Sciences of the United States of America. 121 (14) e2402541121. Bibcode:2024PNAS..12102541B. doi:10.1073/pnas.2402541121. PMC 10998573. PMID 38527209.
- Bezeljak U (2022). "Cancer gene therapy goes viral: viral vector platforms come of age". Radiology and Oncology. 56 (1): 1–13. doi:10.2478/raon-2022-0002. PMC 8884858. PMID 35148469.
- Bulcha JT, Wang Y, Ma H, Tai PW, Gao G (2021). "Viral vector platforms within the gene therapy landscape". Signal Transduction and Targeted Therapy. 6 (1): 53. doi:10.1038/s41392-021-00487-6. PMC 7868676. PMID 33558455.
- Elkashif A, Alhashimi M, Sayedahmed EE, Sambhara S, Mittal SK (2021). "Adenoviral vector-based platforms for developing effective vaccines to combat respiratory viral infections". Clinical and Translational Immunology. 10 (10) e1345. doi:10.1002/cti2.1345. PMC 8510854. PMID 34667600.
- Elois MA, Silva R, Pilati GV, Rodriguez-Lazaro D, Fongaro G (2023). "Bacteriophages as Biotechnological Tools". Viruses. 15 (2): 268–284. doi:10.3390/v15020349. PMC 9963553. PMID 36851563.
- Ewer K, Sebastian S, Spencer AJ, Gilbert S, Hill AV, Lambe T (2017). "Chimpanzee adenoviral vectors as vaccines for outbreak pathogens". Human Vaccines & Immunotherapeutics. 13 (12): 3020–3032. doi:10.1080/21645515.2017.1383575. PMC 5718829. PMID 29083948.
- Farhad T, Neves K, Arbuthnot P, Maepa MB (2022). "Adenoviral Vectors: Potential as Anti-HBV Vaccines and Therapeutics". Genes. 13 (11): 1941. doi:10.3390/genes13111941. PMC 9689594. PMID 36360178.
- Feldman ZB, Clayton J (2022). "Genetics and Ethics in the "I am Legend" Corpus". Journal of Literature and Science. 14 (1–2): 94–107. PMC 9764423. PMID 36545402.
- Glorioso JC, Cohen JB, Goins WF, Hall B, Jackson JW, Kohanbash G, et al. (2020). "Oncolytic HSV Vectors and Anti-Tumor Immunity". Current Issues in Molecular Biology. 41: 381–468. PMID 32938804.
- Gruntman AM, Flotte TR (2018). "The rapidly evolving state of gene therapy". The FASEB Journal. 32 (4): 1733–1740. doi:10.1096/fj.201700982R. PMID 31282760.
- Guo ZS, Lu B, Guo Z, Giehl E, Feist M, Dai E, et al. (2019). "Vaccinia virus-mediated cancer immunotherapy: cancer vaccines and oncolytics". Journal of Immunotherapy of Cancer. 9 (7): 6. doi:10.1186/s40425-018-0495-7. PMC 6325819. PMID 30626434.
- Haggerty CL, Grecco GG, Reeves KC, Atwood B (2019). "Adeno-Associated Viral Vectors in Neuroscience Research". Molecular Therapy - Methods and Clinical Development. 17: 69–82. doi:10.1016/j.omtm.2019.11.012. PMC 6931098. PMID 31890742.
- Hromic-Jahjefendic A, Lundstrom K (2020). "Viral Vector-Based Melanoma Gene Therapy". Biomedicines. 8 (60): 60. doi:10.3390/biomedicines8030060. PMC 7148454. PMID 32187995.
- Jackson DA, Symons RH, Berg P (1972). "Biochemical method for inserting new genetic information into DNA of Simian Virus 40: circular SV40 DNA molecules containing lambda phage genes and the galactose operon of Escherichia coli". Proceedings of the National Academy of Sciences of the United States of America. 69 (10): 2904–2909. Bibcode:1972PNAS...69.2904J. doi:10.1073/pnas.69.10.2904. PMC 389671. PMID 4342968.
- Kaynarcalidan O, Mascaraque SM, Drexler I (2021). "Vaccinia Virus: From Crude Smallpox Vaccines to Elaborate Viral Vector Vaccine Design". Biomedicines. 9 (11): 1780. doi:10.3390/biomedicines9121780. PMC 8698642. PMID 34944596.
- Khushalani NI, Harrington KJ, Melcher A, Bommareddy PK, Zamarin D (2023). "Breaking the barriers in cancer care: The next generation of herpes simplex virus-based oncolytic immunotherapies for cancer treatment". Molecular Therapy Oncolytics. 31 100729. doi:10.1016/j.omto.2023.100729. PMC 10570124. PMID 37841530.
- Labbé RP, Vessillier S, Rafiq QA (2021). "Lentiviral Vectors for T Cell Engineering: Clinical Applications, Bioprocessing and Future Perspectives". Viruses. 13 (152): 1528. doi:10.3390/v13081528. PMC 8402758. PMID 34452392.
- Lanigan TM, Kopera HC, Saunders TL (2020). "Principles of Genetic Engineering". Genes. 11 (3): 603–618. doi:10.3390/genes11030291. PMC 7140808. PMID 32164255.
- Larocca C, Schlom J (2011). "Viral vector-based therapeutic cancer vaccines". The Cancer Journal. 17 (5): 359–371. doi:10.1097/PPO.0b013e3182325e63. PMC 3207353. PMID 21952287.
- Li C, Samulski RJ (2020). "Engineering adeno-associated virus vectors for gene therapy". Nature Reviews Genetics. 21 (4): 255–272. doi:10.1038/s41576-019-0205-4. PMID 32042148.
- Li X, Le Y, Zhang Z, Nian X, Liu B, Yang X (2023). "Viral Vector-Based Gene Therapy". International Journal of Molecular Sciences. 24 (9): 7736. doi:10.3390/ijms24097736. PMC 10177981. PMID 37175441.
- Lukiw WJ (2023). "Commentary: A tribute to Dr. Paul Berg (1926-2023) American biochemist, Nobel Laureate and discoverer of recombinant DNA technology, vaccine and genetic engineering". Frontiers in Cell and Developmental Biology. 11 1210530. doi:10.3389/fcell.2023.1210530. PMC 10233203. PMID 37274735.
- McCann N, O'Connor D, Lambe T, Pollard AJ (2022). "Viral vector vaccines". Current Opinion in Immunology. 77. doi:10.1016/j.coi.2022.102210. PMC 9612401. PMID 35643023.
- Merten O, Schweizer M, Chahal P, Kamen AA (2014). "Manufacturing of viral vectors for gene therapy: part I. Upstream processing". Pharmaceutical Bioprocessing. 2 (2): 183–203. doi:10.4155/pbp.14.16.
- Milone MC, O'Doherty U (2018). "Clinical use of lentiviral vectors". Leukemia. 32 (7): 1529–1541. doi:10.1038/s41375-018-0106-0. PMC 6035154. PMID 29654266.
- Mody PH, Pathak S, Hanson LK, Spencer JV (2020). "Herpes Simplex Virus: A Versatile Tool for Insights Into Evolution, Gene Delivery, and Tumor Immunotherapy". Virology. 11 1178122X20913274. doi:10.1177/1178122X20913274. PMC 8142529. PMID 34093008.
- Moen I, Jevne C, Wang J, Kalland K, Chekenya M, Akslen LA, et al. (2012). "Gene expression in tumor cells and stroma in dsRed 4T1 tumors in eGFP-expressing mice with and without enhanced oxygenation". BMC Cancer. 12 (21): 21. doi:10.1186/1471-2407-12-21. PMC 3274430. PMID 22251838.
- Moss B (2013). "Reflections on the early development of poxvirus vectors". Vaccine. 31 (39): 4220–4222. doi:10.1016/j.vaccine.2013.03.042. PMC 3755097. PMID 23583893.
- Nemirov K, Bourgine M, Anna F, Wei Y, Charneau P, Majlessi L (2023). "Lentiviral Vectors as a Vaccine Platform against Infectious Diseases". Pharmaceutics. 15 (3): 846. doi:10.3390/pharmaceutics15030846. PMC 10053212. PMID 36986707.
- Nomaguchi M, Fujita M, Miyazaki Y, Adachi A (2012). "Viral Tropism". Frontiers in Microbiology. 3 (281): 281. doi:10.3389/fmicb.2012.00281. PMC 3411105. PMID 22876241.
- Pasin F, Menzel W, Daròs JA (2019). "Harnessed viruses in the age of metagenomics and synthetic biology: an update on infectious clone assembly and biotechnologies of plant viruses". Plant Biotechnology Journal. 17 (6): 1010–1026. doi:10.1111/pbi.13084. PMC 6523588. PMID 30677208.
- Pasin F, Uranga M, Charudattan R, Kwon CT (2024). "Engineering good viruses to improve crop performance". Nature Reviews Bioengineering. 2 (7): 532–534. doi:10.1038/s44222-024-00197-y. hdl:10261/394453.
- Petrov G, Dymova M, Richter V (2022). "Bacteriophage-Mediated Cancer Gene Therapy". International Journal of Molecular Sciences. 23 14245. doi:10.3390/ijms232214245. PMC 9697857. PMID 36430720.
- Pranjol ZI, Hajitou A (2015). "Bacteriophage-Mediated Cancer Gene Therapy". Viruses. 7 (1): 268–284. doi:10.3390/v7010268. PMC 4306838. PMID 25606974.
- Sakuma T, Barry MA, Ikeda Y (2012). "Lentiviral vectors: basic to translational". Biochemical Journal. 443 (3): 603–618. doi:10.1042/BJ20120146. PMID 22507128.
- Sasmita AO (2019). "Current viral-mediated gene transfer research for treatment of Alzheimer's disease". Biotechnology & Genetic Engineering Reviews. 35 (1): 26–45. doi:10.1080/02648725.2018.1523521. PMID 30317930.
- Sánchez-Angulo M (2023). "Microbial pathogens in the movies". FEMS Microbiology Letters. 370. doi:10.1093/femsle/fnad129. PMC 10754150. PMID 38059853.
- Sasso E, D'Alise AM, Zambrano N, Scarselli E, Folgori A, Nicosia A (2020). "New viral vectors for infectious diseases and cancer". Seminars in Immunology. 50. doi:10.1016/j.smim.2020.101430. PMID 33262065.
- Schaefer A, Robbins KE, Nzilambi EN, Louis ME, Quinn TC, Folks TM, et al. (2005). "Divergent HIV and Simian Immunodeficiency Virus Surveillance, Zaire". Emerging Infectious Diseases. 11 (9): 1446–1448. doi:10.3201/eid1109.050179. PMC 3310624. PMID 16229778.
- Sibbald B (2001). "Death but one unintended consequence of gene-therapy trial". CMAJ. 164 (11): 1612. PMC 81135. PMID 11402803.
- Sheridan C (2011). "Gene therapy finds its niche". Nature Biotechnology. 29 (2): 121–128. doi:10.1038/nbt.1769. PMID 21301435.
- Smith GL, Mackett M, Moss B (1983). "Infectious vaccinia virus recombinants that express hepatitis B virus surface antigen". Nature. 302 (5908): 490–495. Bibcode:1983Natur.302..490S. doi:10.1038/302490a0. PMID 6835382.
- Travieso T, Li J, Mahesh S, Mello JD, Blasi M (2022). "The use of viral vectors in vaccine development". npj Vaccines. 7 (1): 75. doi:10.1038/s41541-022-00503-y. PMC 9253346. PMID 35787629.
- Ura T, Okuda K, Shimada M (2014). "Developments in Viral Vector-Based Vaccines". Vaccines. 2 (3): 624–641. doi:10.3390/vaccines2030624. PMC 4494222. PMID 26344749.
- van der Loo J, Wright JF (2016). "Progress and challenges in viral vector manufacturing". Human Molecular Genetics. 25 (R1): R42-52. doi:10.1093/hmg/ddv451. PMC 4802372. PMID 26519140.
- Wang S, Liang B, Wang W, Li L, Feng N, Zhao Y, et al. (2023). "Viral vectored vaccines: design, development, preventive and therapeutic applications in human diseases". Signal Transduction and Targeted Therapy. 8 (1): 149. doi:10.1038/s41392-023-01408-5. PMC 10081433. PMID 37029123.
- Wirth T, Parker N, Ylä-Herttuala S (2013). "History of gene therapy". Gene. 525 (2): 162–169. doi:10.1016/j.gene.2013.03.137. PMID 23618815.
- Zaidi SS, Mansoor S (2017). "Viral Vectors for Plant Genome Engineering". Frontiers in Plant Science. 8: 539. doi:10.3389/fpls.2017.00539. PMC 5386974. PMID 28443125.
- Zhang Z, Dong L, Zhao C, Zheng P, Zhang X, Xu J (2021). "Vaccinia virus-based vector against infectious diseases and tumors". Human Vaccines & Immunotherapeutics. 17 (6): 1578–1585. doi:10.1080/21645515.2020.1840887. PMC 8115763. PMID 33606578.
News articles
[edit]- "Fact check: A vaccine did not turn characters in the movie 'I Am Legend' into zombies". Reuters. December 18, 2020. Retrieved 27 April 2024.
- Kolata G (27 November 2017). "Gene Therapy Hits a Peculiar Roadblock: A Virus Shortage". The New York Times. Archived from the original on 25 April 2023. Retrieved 20 May 2024.
- Stolberg SG (28 November 1999). "The Biotech Death of Jesse Gelsinger". The New York Times Magazine. Archived from the original on 25 October 2012. Retrieved 29 April 2024.
Books and protocols
[edit]- Carmen I (1985). Cloning and the Constitution: An Inquiry into Governmental Policymaking and Genetic Experimentation. University of Wisconsin Press. ISBN 978-0-299-10340-8.
- Kayser FH, Bienz KA, Eckert J, Zinkernagel RM (2005). Medical Microbiology (10 ed.). Thieme. ISBN 1-58890-245-5.
- Warnock JN, Daigre C, Al-Rubeai M (2011). "Introduction to Viral Vectors". In Manfredsson FP, Benskey MJ (eds.). Viral Vectors for Gene Therapy: Methods and Protocols. Springer. pp. 1–25. ISBN 978-1-4939-9064-1.
Viral vector
View on GrokipediaDefinition and Characteristics
Core Definition and Mechanism
A viral vector is a genetically modified virus engineered to deliver exogenous genetic material into target cells, exploiting the virus's natural ability to infect cells and introduce nucleic acids without causing productive infection or disease in the host.[13] This modification typically involves removing viral genes responsible for replication and pathogenesis, replacing portions of the viral genome with a therapeutic transgene under control of appropriate promoters.[6] Viral vectors are classified based on their nucleic acid type (DNA or RNA) and integration capability, but all rely on the virus's evolved machinery for efficient cellular entry and gene transfer.[14] The engineering process begins with selecting a parental virus, such as adenovirus or lentivirus, whose genome is cloned into a plasmid backbone for manipulation.[11] Essential viral structural and replication genes are deleted or separated into helper constructs to prevent vector replication outside controlled production systems, while the transgene cassette—containing the gene of interest, regulatory elements, and polyadenylation signals—is inserted.[1] Packaging cell lines or transient transfection systems provide the missing viral components, enabling assembly of non-replicative vector particles at titers often exceeding 10^12 viral genomes per milliliter for clinical-scale production.[15] This results in high-titer, purified vectors capable of transducing specific cell types based on the parental virus's tropism.[16] In terms of mechanism, the vector initiates infection by binding host cell receptors, such as coxsackievirus and adenovirus receptor (CAR) for adenoviral vectors, triggering endocytosis or fusion for internalization.[17] Endosomal escape follows, mediated by viral proteins that disrupt the endosomal membrane, allowing the capsid to release its genome into the cytoplasm.[2] For DNA vectors, the genetic payload traffics to the nucleus via nuclear localization signals or passive diffusion through nuclear pores during cell division; RNA vectors like retroviral types reverse transcribe to DNA for nuclear import.[18] Once in the nucleus, the transgene may persist episomally, as in adeno-associated virus (AAV) vectors, enabling long-term expression in non-dividing cells, or integrate into the host genome via viral integrase for stable inheritance in dividing cells.[17] Expression occurs through host machinery transcribing the transgene into mRNA, which is exported and translated into functional protein, with efficacy depending on vector dose, target tissue accessibility, and immune evasion.[19]Physical and Biological Properties
![Icosahedral_Adenoviruses.jpg][float-right] Viral vectors possess distinct physical properties that influence their production, storage, and delivery efficiency. Most derive from enveloped or non-enveloped viruses with diameters typically ranging from 20 nm to 150 nm; for example, adenoviral vectors feature non-enveloped icosahedral capsids of 80-100 nm, composed of 252 capsomers, while lentiviral vectors are enveloped retroviral particles approximately 100-120 nm in size.[20][21] These structures confer varying stability profiles, with non-enveloped vectors like adenoviruses exhibiting greater resilience to environmental stresses but sensitivity to pH and temperature fluctuations, necessitating formulation with buffers and excipients to prevent capsid disassembly or aggregation.[22] Enveloped vectors, such as lentiviruses, are more labile due to lipid bilayers prone to disruption by detergents or freeze-thaw cycles, though cryoprotectants enhance long-term viability.[23] Biologically, viral vectors are replication-incompetent through genetic modifications that excise essential replication genes, ensuring safety by limiting propagation beyond initial transduction while preserving entry and gene expression mechanisms.[24] They exhibit engineered tropism via capsid or envelope proteins targeting specific cellular receptors, such as coxsackievirus-adenovirus receptor (CAR) for adenoviruses or CD4/CCR5 for lentiviruses, enabling broad or tissue-specific delivery.[25][26] Genome capacities vary: adenoviral vectors accommodate up to 36 kb of DNA as episomal elements, lentivirals integrate ~8-10 kb into host chromatin for stable expression in dividing and non-dividing cells, and adeno-associated vectors (AAVs) carry ~4.7 kb ssDNA persisting extrachromosomally. Immunogenicity arises from viral proteins triggering innate responses via pattern recognition receptors and adaptive immunity, particularly pronounced in adenoviral vectors due to pre-existing antibodies in 40-90% of populations, potentially reducing efficacy in repeat dosing but bolstering vaccine-induced T-cell responses.[20][2][27] These properties underpin their utility, though off-target effects and insertional mutagenesis risks, mitigated by self-inactivating designs, demand rigorous preclinical assessment.[28]Types of Viral Vectors
Retroviral and Lentiviral Vectors
Retroviral vectors are derived from retroviruses, such as murine leukemia virus (MLV), and consist of a modified single-stranded RNA genome that is reverse-transcribed into double-stranded DNA upon entry into host cells, followed by integration into the host genome via viral integrase.[29] This integration enables stable, long-term expression of the therapeutic transgene, theoretically requiring only a single administration for persistent effects.[30] They offer a packaging capacity of approximately 8-9 kb, suitable for many therapeutic genes, and have been foundational in early gene therapy efforts due to efficient transduction in dividing cells.[31] However, retroviral vectors are limited to transducing actively dividing cells, as their pre-integration complex cannot efficiently cross the intact nuclear membrane in non-dividing cells, restricting applications in tissues like neurons or quiescent hematopoietic stem cells.[32] A major drawback is the risk of insertional mutagenesis, where random integration near proto-oncogenes can activate them, leading to malignancies; this was evidenced in clinical trials for X-linked severe combined immunodeficiency (SCID-X1), where two patients developed T-cell leukemia due to LMO2 proto-oncogene activation.[29] To mitigate replication-competent virus production, vectors employ split-genome packaging systems separating gag-pol, env, and vector components across multiple plasmids.[33] Lentiviral vectors, a subclass of retroviral vectors primarily based on HIV-1, address key limitations by enabling transduction of both dividing and non-dividing cells through active nuclear import mediated by viral accessory proteins like matrix and Vpr.[34] They maintain a slightly larger packaging capacity of around 9 kb and are often pseudotyped with vesicular stomatitis virus glycoprotein (VSV-G) for enhanced stability, broad tropism, and concentrated production yields.[2] Unlike simple retroviruses, lentiviruses integrate preferentially into active transcription units, potentially reducing but not eliminating genotoxicity risks.[32] Safety has evolved across generations: first-generation systems retained significant HIV sequences including accessory genes, increasing recombination risks; second-generation removed vif, vpr, vpu, and nef; third-generation further split packaging into four or more plasmids, eliminating tat and incorporating self-inactivating (SIN) long terminal repeats (LTRs) that abolish promoter activity post-integration, minimizing insertional activation.[35] [36] These designs reduce the probability of generating replication-competent lentivirus (RCL) to below detectable levels in preclinical assays, enabling over 100 clinical trials by 2018 with no RCL detections.[37] Third-generation lentiviral vectors have thus become preferred for hematopoietic stem cell gene therapy, as in approved treatments for beta-thalassemia and cerebral adrenoleukodystrophy, balancing efficacy with reduced oncogenic potential compared to earlier retroviral systems.[35]Adenoviral Vectors
Adenoviral vectors are engineered derivatives of adenoviruses, a family of non-enveloped, icosahedral viruses characterized by a linear double-stranded DNA genome of approximately 36 kilobases.[20] These vectors typically employ human adenovirus serotype 5 (Ad5) or chimpanzee-derived adenoviruses like ChAdOx1 to evade pre-existing human immunity.[38] The viral capsid, composed of 252 capsomeres including hexons, pentons, and fiber proteins, facilitates broad cell tropism via receptor interactions such as coxsackievirus-adenovirus receptor (CAR) and integrins.[39] To render them replication-deficient, essential early genes like E1 (encoding proteins for viral DNA replication) and often E3 (modulating host immune responses) are deleted, with therapeutic transgenes inserted in their place, allowing packaging of up to 8 kb of foreign DNA in first-generation vectors or over 30 kb in helper-dependent "gutless" variants lacking all viral coding sequences.[40] Upon transduction, the vector genome persists episomally in the nucleus, enabling high-level but transient transgene expression without genomic integration, which reduces risks of insertional mutagenesis compared to integrating vectors.[41] Production occurs at high titers (10^12-10^13 viral particles per liter) in complementing cell lines like HEK293, which supply the deleted E1 functions.[42] Adenoviral vectors excel in delivering robust transgene expression across dividing and non-dividing cells due to efficient nuclear entry and strong promoters, making them suitable for applications requiring potent immune stimulation.[43] Their advantages include large cloning capacity, ease of manufacturing, and inherent adjuvanticity, which promotes both humoral and cellular immunity—key for vaccine platforms.[44] However, drawbacks predominate in long-term gene therapy: strong innate and adaptive immune responses, including cytotoxic T-lymphocyte activation against transduced cells and vector capsids, limit durability of expression to weeks.[45] Pre-existing neutralizing antibodies, prevalent in 40-90% of populations for common serotypes like Ad5, reduce efficacy and necessitate rarer serotypes or shielding strategies.[46] Additionally, potential for liver tropism and cytokine storms at high doses has constrained systemic use.[25] In vaccine development, adenoviral vectors have proven effective, as seen in authorized products like the AstraZeneca-Oxford ChAdOx1-S (encoding SARS-CoV-2 spike protein), Johnson & Johnson Ad26.COV2.S, and Russia's Sputnik V (heterologous Ad5/Ad26 prime-boost), which elicited protective immunity against COVID-19 in billions of doses administered globally by 2023.[47] The replication-competent Ad26.ZEBOV/MVA-BN-Filo (Ervebo) vaccine, approved in 2019, demonstrated 97.5% efficacy against Ebola virus disease in a 2019-2020 trial.[48] For gene therapy, applications are niche, primarily oncolytic vectors like China's approved H101 (replication-conditional, targeting p53-deficient tumors) for head and neck cancer, though first-generation non-replicating vectors faced setbacks, such as the 1999 ornithine transcarbamylase trial fatality from inflammatory response.[49] Ongoing trials explore gutless vectors for hereditary diseases, leveraging reduced immunogenicity for safer, prolonged expression.[50]Adeno-Associated Viral Vectors
Adeno-associated viral (AAV) vectors derive from adeno-associated virus, a non-enveloped, single-stranded DNA dependovirus in the Parvoviridae family with a genome of approximately 4.7 kilobases flanked by inverted terminal repeats (ITRs).[17] The wild-type virus requires co-infection with a helper virus, such as adenovirus or herpesvirus, for replication, but recombinant AAV (rAAV) vectors used in applications are engineered to be replication-incompetent by removing viral rep and cap genes, which are supplied in trans via helper plasmids or viruses during production.[17] These vectors package transgenes up to about 4.7 kb efficiently, though capacities up to 6 kb are possible with reduced yields and increased genome instability.[51] AAV exhibits low pathogenicity in humans, with no associated diseases, making it suitable for therapeutic delivery.[52] Over 170 AAV serotypes have been identified, primarily from primate tissues, with capsid variations dictating tissue tropism and transduction efficiency.[53] AAV2, the first serotype isolated in 1965 as a contaminant in human adenovirus preparations, transduces dividing and non-dividing cells but primarily targets muscle, liver, and retina; it was used in the earliest clinical trials due to its established receptor interactions.[53] AAV9 demonstrates broad tropism, including efficient central nervous system transduction via blood-brain barrier crossing in neonates, while AAV8 favors hepatocytes for liver-directed therapies.[54] Capsid engineering, such as through directed evolution, has yielded variants like AAV-PHP.eB for enhanced neuronal targeting in rodents, though translation to humans remains limited by species-specific differences.[54] In transduction, rAAV enters cells via receptor-mediated endocytosis, traffics to the nucleus, and converts its single-stranded genome to double-stranded DNA, which persists episomally in non-dividing cells for years, enabling stable transgene expression without integration into the host genome in most cases.[17] Integration occurs rarely at AAVS1 on chromosome 19, posing minimal oncogenic risk compared to retroviral vectors.[55] However, innate immune activation via Toll-like receptor 9 and pattern recognition of vector components can trigger inflammation, while adaptive responses, including pre-existing neutralizing antibodies (NAbs) prevalent in 30-80% of adults depending on serotype exposure history, reduce efficacy and preclude redosing.[56][57] AAV vectors dominate gene therapy applications due to their safety profile and durability, with eight FDA approvals as of 2023, including Luxturna (voretigene neparvovec, AAV2 delivering RPE65 for Leber congenital amaurosis, approved December 19, 2017) and Zolgensma (onasemnogene abeparvovec, AAV9 delivering SMN1 for spinal muscular atrophy type 1, approved May 24, 2019).[58][59] Limitations include transgene size constraints, which exclude larger genes like dystrophin for Duchenne muscular dystrophy, and manufacturing scalability issues, as high-titer production requires transient transfection of HEK293 cells with yields of 10^14-10^15 vector genomes per batch.[52] Ongoing challenges involve capsid optimization to evade NAbs and dual-vector strategies for oversized payloads.[53]Other Vectors (Herpesviral, Poxviral, and Non-Mammalian)
Herpesviral vectors, predominantly based on herpes simplex virus type 1 (HSV-1), exploit the virus's natural neurotropism and large genome to deliver transgenes, particularly for central nervous system targeting.[60] These vectors are engineered as replication-defective forms, where essential viral genes are deleted to prevent replication while retaining the ability to infect and express foreign genes in non-dividing cells.[61] HSV-1 vectors support insert sizes exceeding 100 kb, far surpassing many other systems, enabling delivery of large therapeutic cassettes such as whole genomic loci or multiple genes.[60] Applications include gene therapy for neurodegenerative diseases like Parkinson's, where HSV vectors have demonstrated efficient transduction of neurons in preclinical models, and oncolytic variants for tumor lysis in cancers such as melanoma, achieving objective response rates of up to 24.6% in checkpoint-refractory cases.[62] Limitations include potential immunogenicity and cytotoxicity from residual viral proteins, though next-generation designs minimize these by eliminating all viral gene expression.[61] Poxviral vectors, derived from viruses like vaccinia, modified vaccinia Ankara (MVA), and fowlpox, are DNA-based systems valued for their cytoplasmic replication, which avoids genomic integration risks, and capacity for inserts up to 25 kb.[63] These vectors induce robust cellular and humoral immune responses, making them suitable for vaccine platforms; for instance, recombinant vaccinia expressing antigens has been used since 1982 for immunization studies.[64] MVA, attenuated to prevent replication in mammalian cells, enhances safety and is employed in prime-boost regimens to amplify heterologous antigen presentation, as seen in HIV and Ebola vaccine trials.[65] In cancer immunotherapy, poxvirus vectors deliver tumor-associated antigens alongside cytokines, showing prolonged survival in preclinical models when combined with checkpoint inhibitors.[66] Drawbacks include pre-existing immunity from historical smallpox vaccination, which can reduce efficacy, though avian poxviruses like fowlpox mitigate this via lack of cross-reactivity.[67] Non-mammalian viral vectors, such as baculoviruses from insect hosts, provide a safe alternative for mammalian gene delivery since they do not replicate in vertebrate cells, eliminating risks of uncontrolled spread.[68] Autographa californica multiple nucleopolyhedrovirus (AcMNPV), the most studied, transduces a broad range of mammalian cell types via glycoproteins like GP64 binding to vertebrate receptors, achieving transient expression suitable for vaccine production or short-term therapy.[69] With insert capacities over 100 kb, baculovirus vectors have been applied in preclinical gene therapy for liver diseases and cancer, displaying low immunogenicity and toxicity compared to mammalian viruses.[70] Challenges include inefficient nuclear entry leading to episomal persistence rather than integration, limiting long-term expression, though pseudotyping with vesicular stomatitis virus G protein improves transduction efficiency.[71] These vectors also facilitate scalable manufacturing in insect cells, supporting their use in personalized medicine approaches.[70]Applications
Basic and Preclinical Research
Viral vectors enable precise genetic manipulation in cellular and animal models, facilitating investigations into gene function, protein interactions, and signaling pathways central to basic research. Lentiviral vectors, derived from HIV-1, integrate transgenes into the host genome for stable, heritable expression in proliferating cells, commonly used to generate knockout or overexpression lines via shRNA or cDNA delivery, respectively.[72] Adeno-associated viral (AAV) vectors, conversely, persist episomally in non-dividing cells like neurons, supporting long-term expression without insertional risks, as evidenced by their application in over 170 neuroscience studies by 2019 for optogenetics and circuit mapping.[54] In neuroscience, AAV serotypes such as AAV9 achieve widespread transduction across brain regions in rodents and primates, revealing synaptic plasticity mechanisms and basal ganglia connectivity complexities unattainable with chemical tracers alone.[73] Lentiviral vectors complement this by enabling Cre-lox recombination in stem cell-derived models, dissecting developmental pathways with efficiencies exceeding 90% in human iPSCs.[72] These tools have advanced understanding of disease mechanisms, such as in Parkinson's models where AAV-delivered alpha-synuclein aggregates recapitulate pathology.[54] Preclinical research employs viral vectors to assess therapeutic candidates in vivo, evaluating efficacy, biodistribution, and immunogenicity in disease-specific animal models before human trials. AAV vectors, for example, have demonstrated sustained transgene expression up to 10 years in non-human primates, guiding hemophilia B dosing at 2×10^12 vg/kg for factor IX restoration achieving 10-30% normal levels.[52] Lentiviral vectors in muscular dystrophy models correct dystrophin expression in mdx mice, improving muscle function by 50-70% and informing scalable production needs.[6] Such studies highlight vector tropism variations, with AAV8 optimizing liver targeting in metabolic disorder models, while underscoring challenges like pre-existing immunity affecting 30-50% of adults.[11] These findings from rodent, canine, and primate models validate causal links between gene delivery and phenotypic correction, prioritizing vectors with minimal off-target effects for clinical progression.[74]Gene Therapy
Viral vectors serve as primary delivery vehicles in gene therapy, transporting therapeutic transgenes into patient cells to correct genetic deficiencies or express functional proteins. Adeno-associated virus (AAV) vectors predominate for in vivo applications due to their low immunogenicity, ability to transduce non-dividing cells, and capacity for long-term episomal gene expression, while lentiviral vectors excel in ex vivo modification of hematopoietic stem cells for stable integration.[2][19] By 2023, over 20 FDA-approved gene therapies utilized viral vectors, targeting monogenic disorders such as spinal muscular atrophy (SMA), hemophilia, and inherited blindness.[58][75] Early clinical trials in the 1990s employed retroviral vectors for ex vivo gene correction in adenosine deaminase (ADA)-deficient severe combined immunodeficiency (SCID), achieving immune reconstitution in some patients but revealing insertional mutagenesis risks. Subsequent advancements shifted toward AAV vectors; Luxturna (voretigene neparvovec), approved by the FDA in December 2017, uses AAV2 to deliver the RPE65 gene to retinal cells, restoring vision in patients with Leber congenital amaurosis caused by biallelic RPE65 mutations, with clinical trials demonstrating improved multi-luminance mobility testing scores.[76][77] For SMA, Zolgensma (onasemnogene abeparvovec), an AAV9-based therapy approved in May 2019, delivers the SMN1 gene via intravenous infusion, enabling survival and motor milestone achievement in infants with SMA type 1, as evidenced by phase 3 trials showing 100% event-free survival at 14 months versus 26% in controls.[78] Lentiviral vectors have succeeded in ex vivo therapies for blood disorders; Zynteglo (betibeglogene autotemcel), approved in August 2022, integrates a functional β-globin gene into autologous hematopoietic stem cells for transfusion-dependent β-thalassemia, with pivotal trials reporting transfusion independence in 31 of 42 patients after a median 3.8 years. Hemgenix (etranacogene dezaparvovec), an AAV5 vector approved in November 2022 for hemophilia B, expresses factor IX, reducing annualized bleeding rates by 54% in phase 3 studies compared to prior prophylaxis. These outcomes underscore viral vectors' efficacy in achieving durable phenotypic correction, though scalability and vector dosing limitations persist.[24][79] Ongoing trials expand applications to central nervous system disorders, leveraging AAV's neurotropism; for instance, AAV9 vectors target neurons for metachromatic leukodystrophy, with preclinical data showing widespread CNS transduction. Despite historical setbacks like the 1999 adenovirus-related death in an ornithine transcarbamylase deficiency trial, refined vector designs have minimized acute toxicities, enabling broader adoption. As of 2024, viral vector gene therapies treat over a dozen rare diseases, with cumulative evidence from thousands of patients affirming their transformative potential when transgene expression aligns with disease pathology.[52][80]Vaccine Development
Viral vector vaccines are engineered by inserting genetic sequences encoding pathogen antigens into replication-deficient viruses, enabling host cells to produce the antigens and trigger both humoral and cellular immune responses.[81] This approach leverages the virus's natural ability to infect cells and stimulate immunity while minimizing disease risk through genetic modifications that prevent replication.[82] Development begins with vector selection, such as adenoviruses or poxviruses, followed by transgene insertion, preclinical efficacy testing in animal models, and phased clinical trials assessing safety, immunogenicity, and protection.[27] The foundational milestone occurred in 1982 when researchers inserted the hepatitis B surface antigen gene into vaccinia virus, demonstrating proof-of-concept for antigen expression in vivo.[14] Subsequent progress focused on adenoviral vectors, with early trials in the 1990s for HIV and malaria, though pre-existing immunity to common serotypes like Ad5 posed challenges, leading to strategies like using rare or chimpanzee-derived adenoviruses.[27] The Ervebo Ebola vaccine, utilizing a vesicular stomatitis virus vector expressing Ebola glycoprotein, marked the first licensed viral vector vaccine in 2019, approved by the European Medicines Agency on November 1, 2019, after demonstrating 97.5% efficacy in a 2014-2016 outbreak ring vaccination trial involving 3,000 participants.[83] The COVID-19 pandemic accelerated adenoviral vector vaccine deployment, with the AstraZeneca-Oxford vaccine (ChAdOx1 nCoV-19), based on a chimpanzee adenovirus expressing SARS-CoV-2 spike protein, receiving emergency authorization in the UK on December 30, 2020, following Phase III trials showing 70.4% efficacy against symptomatic infection in over 23,000 participants.[47] Similarly, the Janssen vaccine (Ad26.COV2.S), using human adenovirus type 26, was authorized by the FDA on February 27, 2021, with 66% efficacy against moderate to severe disease in a trial of approximately 44,000 individuals.[47] Russia's Sputnik V, combining Ad26 and Ad5 vectors in a prime-boost regimen, reported 91.6% efficacy in a 2021 Lancet-published trial of 19,866 participants.[84] Advantages include potent T-cell responses mimicking natural infection, enabling protection against diverse pathogens like viruses requiring cellular immunity, unlike subunit vaccines.[81][85] However, drawbacks encompass vector-specific immunity reducing efficacy in repeat dosing and complex manufacturing requiring biosafety level 2 facilities.[86][87] Ongoing innovations involve capsid modifications to evade immunity and heterologous prime-boost schedules to enhance responses, as seen in Ebola and COVID regimens.[40]
Oncolytic and Other Therapeutic Uses
Oncolytic virotherapy employs genetically modified viral vectors designed to selectively replicate within and destroy cancer cells while sparing healthy tissue, often eliciting an antitumor immune response. These vectors exploit molecular defects common in malignancies, such as impaired interferon signaling or dysregulated cell cycle control, enabling preferential tumor tropism. Attenuation mutations reduce pathogenicity in normal cells, while transgenes like granulocyte-macrophage colony-stimulating factor (GM-CSF) enhance immunogenicity by promoting dendritic cell activation and T-cell infiltration.[88][89] Talimogene laherparepvec (T-VEC), an oncolytic herpes simplex virus type 1 (HSV-1) vector, exemplifies this approach. It features deletions in ICP34.5 genes to limit replication to cells with defective protein kinase R (PKR) pathways, prevalent in cancers, and insertion of the human GM-CSF gene to boost local immunity. Administered intratumorally, T-VEC lyses injected lesions, releases tumor antigens, and induces abscopal effects on distant metastases via systemic T-cell responses. The phase III OPTiM trial (NCT00763608), involving 436 patients with advanced melanoma, demonstrated a 26.4% durable response rate (≥6 months) for T-VEC versus 2.1% for GM-CSF alone, leading to FDA approval on October 27, 2015, for unresectable cutaneous, subcutaneous, or nodal melanoma post-surgery. European Medicines Agency approval followed in December 2015. Real-world data from over 1,000 patients confirm a favorable safety profile, with flu-like symptoms and injection-site reactions as primary adverse events, though efficacy diminishes in visceral disease.[90][91][92] Other oncolytic vectors include adenoviral platforms like H101 (recombinant adenovirus type 5), approved by China's State Food and Drug Administration in November 2005 for head and neck squamous cell carcinoma refractory to chemotherapy, following a phase III trial showing 78.8% response rate in combination with cisplatin/5-fluorouracil versus 39.6% for chemotherapy alone. Pexastimogene devacirepvec (JX-594, modified vaccinia virus) expresses GM-CSF and thymidine kinase for selective replication in EGFR/RAS pathway-dysregulated tumors; phase II trials reported median survival of 14.1 months in hepatocellular carcinoma patients versus 6.7 months on placebo, though phase III results remain pending. Reovirus (Reolysin) and Newcastle disease virus have advanced to phase II/III trials, often combined with checkpoint inhibitors like pembrolizumab, yielding objective response rates up to 36% in refractory solid tumors. As of 2023, over 100 oncolytic virus trials are registered on ClinicalTrials.gov, predominantly phase I/II, highlighting immune evasion challenges and the need for combinatorial strategies.[93][88][94] Beyond oncolysis, viral vectors enable suicide gene therapies, where delivered transgenes convert non-toxic prodrugs into cytotoxins, amplifying cell death in targeted tissues. For instance, retroviral or adenoviral vectors encoding herpes simplex virus thymidine kinase (HSV-TK) facilitate ganciclovir activation, restricting replication-competent viruses to proliferative cells like tumors or vascular lesions. Clinical applications include glioblastoma treatment, with phase III data showing prolonged survival when combined with radiotherapy. In cardiovascular therapy, adenovirus-mediated HSV-TK/ganciclovir has inhibited intimal hyperplasia in vein grafts, reducing restenosis rates by 50-70% in preclinical models translated to early human trials. These approaches, distinct from stable transgene expression in gene therapy, leverage transient viral replication for localized cytotoxicity, though immunogenicity limits repeat dosing.[95][96]Production and Manufacturing
Vector Design and Construction
Viral vectors are engineered by modifying the genetic backbone of wild-type viruses to eliminate replication capacity and pathogenicity while incorporating a therapeutic transgene. This involves selecting a viral serotype or strain suited to the application's requirements, such as tissue tropism, payload capacity, and persistence— for instance, adeno-associated virus (AAV) for episomal expression with low immunogenicity, lentiviral vectors for genomic integration, or adenoviral vectors for high-capacity transient delivery.[2][19] The core design principle is to retain essential cis-acting elements like inverted terminal repeats (ITRs) in AAV or long terminal repeats (LTRs) in lentiviruses for packaging and transduction, while excising trans-acting genes (e.g., rep/cap in AAV, gag/pol/env in lentiviruses) that drive replication.[2] The transgene cassette is cloned into a transfer plasmid, typically comprising a strong or tissue-specific promoter (e.g., CMV for broad expression or synapsin for neurons), the codon-optimized coding sequence, polyadenylation signals, and optional elements like woodchuck hepatitis virus posttranscriptional regulatory element (WPRE) for enhanced mRNA stability or insulators to prevent silencing.[97] Payload limits dictate design: AAV accommodates ~4.7 kb (or ~2.4 kb for self-complementary variants using mutated ITRs), lentiviruses up to 9-10 kb, and gutless adenoviruses ~36 kb by retaining only ITRs and packaging signals.[2][19] To mitigate immunogenicity, sequences are depleted of CpG motifs, and microRNA binding sites (e.g., miR-142) are inserted to evade innate immune detection in antigen-presenting cells.[2] Construction employs plasmid-based recombination to generate replication-deficient vectors, avoiding direct manipulation of infectious virus. For AAV, triple transfection into HEK293 cells uses the transfer plasmid, a rep/cap plasmid, and an adenoviral helper providing E1/E4/VP proteins; homologous recombination assembles the vector genome.[2] Lentiviral vectors utilize a four-plasmid system: transfer vector with self-inactivating 3' LTR, packaging plasmid (gag/pol), rev-expressing plasmid, and envelope (e.g., VSV-G pseudotype for broad tropism), transfected into HEK293T cells.[2] Adenoviral vectors, particularly helper-dependent "gutless" designs, involve co-transfection of the minimal genome plasmid with a helper adenovirus genome flanked by loxP sites, followed by Cre recombinase excision to separate vector from helper particles.[2][6] Advanced engineering refines targeting and safety: capsid shuffling or directed evolution creates novel serotypes (e.g., AAV2.7m8 for muscle), while fiber knob modifications in adenoviruses (e.g., RGD-4C insertion) enhance receptor binding.[2] Dual-vector strategies split oversized transgenes for AAV, relying on ITR-mediated recombination or inteins for reconstitution.[2] These modular approaches ensure scalability and compliance with good manufacturing practices, though challenges like pre-existing immunity necessitate serotype switching or chimeric designs.[19]Scalable Production Methods
Scalable production of viral vectors primarily employs mammalian or insect cell lines cultured in bioreactors to achieve high titers suitable for clinical and commercial applications. Key methods include transient transfection with helper plasmids or viruses in suspension-adapted cells, such as HEK293 for adenoviral and adeno-associated viral (AAV) vectors, enabling scale-up from shake flasks to stirred-tank bioreactors of 25–200 liters or more.[98][99] Stable producer cell lines, incorporating integrated vector genomes and helper elements, offer reproducibility for large-scale manufacturing but require extensive validation for genomic stability.[2] Insect cell systems using baculovirus expression vectors (BEVS) provide an alternative for AAV serotypes, supporting scalability in volumes up to 1,000 liters while avoiding human cell immunogenicity concerns.[100] For adenoviral vectors, optimized processes in HEK293 suspension cultures utilize perfusion or fed-batch modes in fixed-bed bioreactors like the iCELLis system, yielding up to 10^14 viral particles per batch in GMP settings.[101] These methods incorporate design-of-experiments for media optimization, achieving functional titers exceeding 10^12 infectious units per milliliter post-harvest.[102] Lentiviral vectors follow similar transient triple-plasmid transfection in HEK293-derived suspension lines, with bioreactors enabling titers 10-fold higher than adherent methods through cell retention and intensified feeding strategies.[103] AAV production has advanced with HEK293 triple-transfection protocols scaled to 50-liter bioreactors, delivering vector genome titers of 10^14–10^15 vg/L via in situ cell lysis and downstream-compatible harvests.[104] BEVS in Sf9 cells offers higher volumetric productivity for certain capsids, with recent optimizations reaching 10^15 vg/L in wave or stirred bioreactors, though empty/full capsid ratios demand rigorous analytics.[105] Challenges persist in yield consistency across serotypes, prompting hybrid platforms and continuous manufacturing to meet surging gene therapy demands projected at over 1 million doses annually by 2030.[106]Quality Control and Purification
Purification of viral vectors involves downstream processing to separate the target vector particles from host cell components, media, and production byproducts following harvest from cell cultures. Common methods include tangential flow filtration for initial clarification and concentration, followed by chromatography techniques such as anion-exchange, cation-exchange, or affinity chromatography to capture and polish the vectors based on charge, size, or specific ligands.[107] For adeno-associated virus (AAV) vectors, affinity resins targeting capsid proteins enable high specificity, while density gradient ultracentrifugation, though effective for research-scale purity, is limited by poor scalability and is largely replaced by chromatographic approaches in manufacturing.[108] Lentiviral and adenoviral vectors often require additional steps to remove envelope glycoproteins or non-infectious particles, with ultrafiltration/diafiltration used for buffer exchange and final formulation to achieve concentrations suitable for clinical dosing, typically exceeding 10^12 vector genomes per milliliter.[109] These processes must minimize aggregation and loss of infectivity, with yields varying from 20-50% depending on vector type and scale.[110] Quality control (QC) for viral vectors encompasses assays to verify identity, purity, potency, quantity, and safety, ensuring compliance with regulatory standards such as those outlined by the FDA for chemistry, manufacturing, and controls (CMC) in gene therapy products.[111] Identity confirmation typically involves PCR-based sequencing of the transgene cassette or serotype-specific ELISA to distinguish vector variants, critical for preventing mix-ups in multi-product facilities.[112] Purity assessment quantifies contaminants like host cell proteins (via ELISA, targeting <100 ng per dose), residual DNA (<10 ng per dose), and empty capsids (via analytical ultracentrifugation or capillary electrophoresis, aiming for >50% full capsids in AAV preparations).[113] Quantity is measured by quantitative PCR for genome titers (vg/mL) and infectious titers (TU/mL), with ratios ideally between 10:1 and 100:1 to indicate vector functionality.[112] Potency testing evaluates biological activity through in vitro transduction assays in relevant cell lines, measuring transgene expression via qPCR or reporter fluorescence, while safety checks include sterility (USP <71>), endotoxin levels (<5 EU/mL by LAL assay), and adventitious agent screening via next-generation sequencing or PCR panels for viruses, mycoplasma, and bacteria.[111] [114] For GMP production, these assays are phased, with full characterization required for master viral banks and lot release, addressing risks like immunogenic impurities that could trigger adverse immune responses in patients.[115] Regulatory guidance emphasizes process validation to demonstrate consistency, with analytics evolving to include mass spectrometry for capsid profiling and flow virometry for particle enumeration.[113] Challenges persist in standardizing assays across vector types, as enveloped vectors like lentiviruses may co-purify extracellular vesicles, potentially confounding purity metrics.[107]Risks and Safety Concerns
Immunogenicity and Immune Evasion
Viral vectors provoke both innate and adaptive immune responses that can compromise therapeutic efficacy and safety. Innate immunity detects viral capsid proteins via pattern recognition receptors such as Toll-like receptors (TLRs), triggering cytokine release, inflammation, and rapid vector clearance; for adeno-associated virus (AAV) vectors, TLR9 activation by single-stranded DNA genomes exacerbates this, leading to dose-dependent hepatotoxicity observed in high-dose clinical trials for hemophilia B.[116] Adaptive responses include neutralizing antibodies (NAbs) against capsids, prevalent in 30-80% of humans due to prior natural infections, which block vector transduction and exclude seropositive patients from AAV trials, as seen in Luxturna approvals requiring NAb screening.[57] Cellular immunity, via CD8+ T cells targeting transduced cells, causes transgene loss, with capsid-specific T cells correlating to liver toxicity in trials like those for spinal muscular atrophy using AAV9-Zolgensma.[117] Pre-existing immunity particularly hampers adenoviral vectors, where serotype 5 (Ad5) NAbs in over 90% of adults reduce transgene expression by up to 100-fold in preclinical models and diminish vaccine immunogenicity in HIV trials, prompting shifts to rarer serotypes like Ad26 or Ad35.[118] In lentiviral vectors, pseudotyped with envelopes like VSV-G, humoral responses are lower but T cell-mediated clearance persists, limiting redosing; clinical data from CAR-T therapies show vector-specific immunity reducing persistence.[119] These responses not only curtail efficacy but pose risks, including acute anaphylaxis or delayed hypersensitivity, as in the 1999 ornithine transcarbamylase AAV trial fatality from inflammatory cytokines.[117] To evade immunity, strategies focus on vector engineering and adjunct therapies. Capsid modifications, such as AAV shuffling or rational mutagenesis to alter surface epitopes, reduce NAb binding by 10-100 fold in vitro and enhance transduction in seropositive models; for instance, AAV2 capsid variants evade 70% of human sera NAbs.[120] Rare serotypes (e.g., AAV8, AAV9) or nonhuman primate-derived capsids minimize cross-reactivity, enabling higher liver transduction in trials despite 20-40% seroprevalence.[121] Immune stealth approaches include peptide insertion for shielding or empty/full capsid ratios to decoy antibodies, improving durability in nonhuman primates.[122] Pharmacological evasion employs transient immunosuppression, like corticosteroids or rituximab, to suppress T cell responses during vector delivery, boosting expression 5-10 fold in preclinical hemophilia models but risking infections.[123] For redosing, heterologous vectors (e.g., switching AAV serotypes) or non-viral chimeras mitigate adaptive memory, though clinical translation remains limited by incomplete evasion.[124] Ongoing innovations, such as TLR antagonists or capsid deimmunization via CRISPR editing, aim to balance immunogenicity reduction with transduction efficiency, informed by failures in early trials.[116]Insertional Mutagenesis and Oncogenic Risks
Insertional mutagenesis refers to the integration of viral vector DNA into the host cell genome, which can disrupt normal gene function or activate proto-oncogenes, potentially leading to oncogenic transformation.[125] This risk is most pronounced with integrating vectors such as gamma-retroviral and lentiviral systems, where random insertion sites may preferentially target transcriptionally active regions like promoters or enhancers.[126] In contrast, non-integrating vectors like adeno-associated virus (AAV) primarily persist as episomes, though rare integration events have been observed, particularly at hotspots such as the AAVS1 locus on chromosome 19.[127] Early clinical trials of gamma-retroviral vectors for X-linked severe combined immunodeficiency (SCID-X1) demonstrated this hazard concretely; between 2002 and 2006, five of 20 treated patients developed T-cell acute lymphoblastic leukemia due to vector insertions near the LMO2 proto-oncogene, which aberrantly activated its expression and cooperated with secondary somatic mutations.[128] Insertional activation of oncogenes like LMO2, CCND2, and BCL2 was confirmed through genomic analysis, halting the trial and prompting vector redesigns with self-inactivating (SIN) long terminal repeats to minimize enhancer-driven dysregulation.[129] By 2008, four additional cases of insertional oncogenesis were reported in retroviral gene therapy for SCID, underscoring the vectors' bias toward integrating near hematopoietic genes.[128] Lentiviral vectors, derived from HIV-1, exhibit a safer integration profile, favoring gene bodies over promoters and reducing transformation risk by approximately 10-fold compared to gamma-retroviral vectors in preclinical models.[130] SIN lentiviral designs further mitigate genotoxicity, as evidenced by lower oncogenic potential in comparative studies.[35] However, recent data from lentiviral-based therapies for cerebral adrenoleukodystrophy, such as Skysona (elivaldogene autotemcel), reported seven cases of hematological malignancies as of 2025, prompting reevaluation of long-term risks despite no definitive vector causation in all instances.[131] The European Medicines Agency's 2013 reflection paper documented serious adverse events from insertional mutagenesis in 12 patients across primary immunodeficiency trials, emphasizing vigilant monitoring.[132] For AAV vectors, genotoxicity remains theoretical and low-probability in humans, with mouse studies showing elevated hepatocellular carcinoma incidence only at supraphysiological doses exceeding clinical relevance.[133] Regulatory assessments, including those from 2022 onward, conclude that AAV integration does not clearly elevate tumorigenesis risk in standard applications, though high-dose hepatic trials warrant caution.[134] Mitigation strategies across vector types now include insulator elements, genome-wide insertion site mapping via high-throughput sequencing, and preclinical genotoxicity assays to predict and avert oncogenic hotspots.[135] Despite advances, long-term follow-up in over 20 approved gene therapies as of 2025 reveals no new consensus on zero risk, with calls for enhanced biodistribution and clonal tracking to address residual uncertainties.[136]Off-Target Effects and Long-Term Toxicity
Off-target effects in viral vector applications refer to unintended interactions, such as transduction of non-target cells or tissues, leading to ectopic gene expression or genomic alterations outside the intended site. In adenoviral and adeno-associated virus (AAV) vectors, off-target delivery can occur due to systemic administration, resulting in transgene expression in organs like the liver or spleen, which may provoke localized toxicity or immune activation.[137] For gene editing payloads delivered via viral vectors, such as CRISPR-Cas9 in lentiviral systems, off-target cleavage at homologous genomic sites has been documented, potentially causing unintended mutations with frequencies varying by guide RNA design and Cas9 variant, though high-fidelity variants reduce this to below 1% in some assays.[138] These effects are assessed through methods like GUIDE-seq or CIRCLE-seq, which detect double-strand breaks at non-canonical sites.[139] Long-term toxicity concerns primarily stem from insertional mutagenesis in integrating vectors like lentiviral and gammaretroviral systems, where proviral DNA insertion near proto-oncogenes can disrupt tumor suppressors or activate oncogenes, as evidenced by leukemia cases in early severe combined immunodeficiency (SCID) gene therapy trials using gammaretroviral vectors between 2002 and 2006, affecting 5 of 20 patients.[140] Self-inactivating (SIN) lentiviral vectors mitigate this risk by reducing enhancer activity, with preclinical models showing lower genotoxicity compared to gammaretrovirals; clinical follow-up of 783 patients treated with lentiviral-modified T cells over more than 2,200 patient-years reported no vector-related malignancies as of 2024.[35][141] Nonetheless, theoretical risks persist due to preferred integration near transcription start sites, necessitating long-term monitoring protocols that extend up to 15 years post-treatment per FDA guidelines.[142] For non-integrating AAV vectors, long-term risks include rare genomic integration events, estimated at less than 0.1% of transduced cells in vivo, potentially leading to delayed oncogenic transformation, though large-scale clinical data from over 255 trials indicate primarily acute toxicities like hepatotoxicity at doses exceeding 10^13 vg/kg rather than chronic carcinogenesis.[143][123] High-dose AAV administration has been linked to fatal complement-mediated toxicities and myocarditis in recent trials, such as those for cardiac diseases in 2023-2024, highlighting dose-dependent innate immune responses as a causal factor over insertional risks.[144] Persistent episomal AAV genomes can also drive chronic transgene overexpression, contributing to off-target toxicities like retinal degeneration in ocular applications observed in nonhuman primate studies.[145] Overall, while empirical data from approved therapies like Zolgensma (AAV9 for SMA, approved 2019) show durable efficacy with manageable risks up to 5 years post-infusion, genotoxicity assays and extended surveillance remain essential to quantify rare long-term events.[52][123]History and Milestones
Origins and Early Development (1970s–1980s)
The origins of viral vectors trace back to early attempts at leveraging viruses for gene transfer, predating formal recombinant DNA techniques. In 1970, Stanfield Rogers administered Shope papilloma virus to two young sisters with argininemia, a disorder caused by arginase deficiency, based on observations that the virus could induce arginase activity in infected cells, marking the first documented use of a virus as a potential therapeutic gene carrier.[146] However, the treatment failed to produce sustained enzyme activity and drew criticism for lacking ethical oversight and preclinical validation, highlighting early risks of uncontrolled viral delivery.[147] This experiment underscored viruses' natural capacity to introduce genetic material but did not involve genetic engineering.[148] Advancements accelerated with recombinant DNA technology in 1972, when Paul Berg's laboratory constructed the first chimeric DNA molecule by ligating SV40 viral DNA—a simian polyomavirus known for its transforming potential—with lambda phage DNA using EcoRI restriction enzyme and DNA ligase, enabling in vitro propagation of hybrid viral genomes.[149] This SV40-based construct demonstrated the feasibility of engineering viral DNA for foreign gene insertion, laying foundational principles for viral vectors despite concerns over oncogenicity from SV40's tumor-inducing properties.[150] Berg's work, published in Proceedings of the National Academy of Sciences, shifted focus from natural viral infection to deliberate genetic modification, though initial applications remained limited to bacterial and viral propagation rather than mammalian gene delivery.[4] These innovations prompted safety deliberations, culminating in the 1975 Asilomar Conference on Recombinant DNA Molecules, convened by Paul Berg and others at the Asilomar Conference Grounds to assess biohazards, particularly from recombinant viral DNAs that could create novel pathogens or enhance transmissibility.[151] The conference recommended a voluntary moratorium on certain experiments, including cloning tumor virus DNA in prokaryotes or linking viral genomes to non-viral vectors, establishing physical and biological containment guidelines that influenced NIH policies and slowed but did not halt vector development.[152] Attendees emphasized risk proportionality, prohibiting high-risk viral recombinants until safer protocols emerged, reflecting early recognition of insertional mutagenesis and uncontrolled replication as causal threats.[153] By the late 1970s, researchers exploited viral genomes for efficient gene transfer into cultured mammalian cells, with retroviruses gaining attention after Harold Varmus and Michael Bishop's 1970s discoveries that these viruses could stably integrate cellular oncogenes via reverse transcription, inspiring engineered vectors for heritable gene insertion.[154] Initial retroviral constructs, derived from murine leukemia viruses, focused on packaging foreign DNA into viral particles without replication competence, though packaging cell lines were not refined until the early 1980s.[155] Concurrently, adenovirus studies advanced, with early 1980s designs at Cold Spring Harbor Laboratory producing replication-defective vectors by deleting essential early region genes (E1), enabling transient gene expression in non-dividing cells and setting the stage for in vivo applications.[156] These efforts prioritized empirical containment over speculative safety, prioritizing vectors' natural tropism while mitigating pathogenicity through genetic deletions.[157]Initial Clinical Trials and Major Setbacks (1990s–2000s)
The first approved gene therapy clinical trial utilizing a viral vector commenced in 1990, targeting adenosine deaminase (ADA) deficiency, a form of severe combined immunodeficiency (SCID). This ex vivo approach employed a retroviral vector to transduce autologous T cells with the human ADA cDNA, which were then reinfused into the patient, a four-year-old girl named Ashanthi DeSilva. Initial results demonstrated partial restoration of ADA enzyme activity and immune function, though long-term efficacy required ongoing enzyme replacement therapy, marking a proof-of-concept rather than a definitive cure.[158][159] Throughout the 1990s, retroviral vectors dominated early trials for monogenic disorders, with over 100 protocols approved by 1999, focusing on conditions like cystic fibrosis and hemophilia; however, transduction efficiencies remained low, and sustained gene expression proved challenging due to vector instability and host immune responses. Adenoviral vectors emerged in trials by 1993, prized for their high transduction efficiency and capacity for larger transgenes, but preclinical data overlooked potent immunogenicity in humans.[159][79] A pivotal setback occurred on September 17, 1999, when 18-year-old Jesse Gelsinger died during a phase I trial at the University of Pennsylvania testing an E1/E3-deleted adenoviral vector for ornithine transcarbamylase (OTC) deficiency. Gelsinger, who had partial OTC function, received a high-dose (3.8 × 10^13 particles) intraportal infusion, triggering a cytokine storm, disseminated intravascular coagulation, and multi-organ failure attributed to vector-induced innate immune activation rather than the transgene itself. Investigations revealed protocol lapses, including undeclared animal toxicity data and conflicts of interest involving the principal investigator, James Wilson, leading the FDA to suspend the trial, impose institutional holds, and halt many adenoviral programs nationwide. This incident, the first confirmed gene therapy fatality, eroded public trust and prompted stricter oversight, effectively stalling systemic vector delivery approaches for years.[160][161][162] Further complications arose in early 2000s trials for X-linked SCID (SCID-X1) using gamma-retroviral vectors targeting the IL2RG gene. French and British studies reported immune reconstitution in most of 20 treated infants by 2000–2002, with vector-marked T cells persisting long-term, offering apparent cures without conditioning chemotherapy. However, by 2002–2003, five patients developed T-cell acute lymphoblastic leukemia linked to insertional mutagenesis: the strong viral enhancer activated the adjacent LMO2 proto-oncogene, cooperating with secondary mutations. This oncogenic risk, inherent to gamma-retroviruses' preference for integrating near transcription start sites, prompted trial suspensions in 2003 and a field-wide shift away from such vectors, underscoring the need for safer integrase designs and self-inactivating configurations.[128][163][164]Revival, Approvals, and Expansion (2010s–Present)
Following the setbacks of the preceding decades, viral vector technologies experienced a revival in the 2010s driven by engineering advancements that enhanced safety profiles, such as capsid modifications in adeno-associated viruses (AAV) to reduce immunogenicity and improve tissue specificity.[6] These refinements addressed prior issues like insertional mutagenesis and immune responses, enabling progression to clinical success.[2] By the mid-2010s, AAV vectors emerged as the dominant platform for in vivo gene delivery, with lentiviral and retroviral vectors supporting ex vivo applications like CAR-T cell therapies.[165] The European Medicines Agency approved Glybera, an AAV1-based therapy for lipoprotein lipase deficiency, in October 2012 as the first gene therapy product utilizing viral vectors, though it was withdrawn in 2017 due to limited efficacy and high cost.[166] In the United States, the FDA granted approval to Luxturna (voretigene neparvovec), an AAV2 vector for RPE65-mediated retinal dystrophy, on December 19, 2017, marking the first AAV gene therapy for an inherited disease.[58] This was followed by Zolgensma (onasemnogene abeparvovec), an AAV9 vector for spinal muscular atrophy, approved on May 24, 2019, demonstrating durable efficacy in infants. Additional approvals included Hemgenix (etranacogene dezaparvovec), an AAV5 vector for hemophilia B, in November 2022, reflecting growing regulatory confidence.[58] Viral vectors also expanded into vaccine development, with the FDA approving Ervebo (rVSV-ZEBOV), a vesicular stomatitis virus-based vector for Ebola virus disease, on December 20, 2019. The COVID-19 pandemic accelerated adenoviral vector vaccines: Sputnik V (Ad26/Ad5) received emergency approval in Russia on August 11, 2020; AstraZeneca's ChAdOx1-S was authorized in the UK on December 30, 2020; and Johnson & Johnson's Ad26.COV2.S gained FDA emergency use authorization on February 27, 2021.[48] At least six adenoviral vector-based COVID-19 vaccines achieved regulatory approval globally, leveraging prior platforms from Ebola research to enable rapid deployment and billions of doses administered.[48] By 2022, eight viral vector-based gene therapies had received FDA approval, with the pace increasing to four in 2022, seven in 2023, and six in 2024, signaling robust expansion amid a burgeoning clinical pipeline exceeding hundreds of trials.[167][168] This growth underscores viral vectors' versatility across monogenic diseases, oncology, and infectious diseases, though manufacturing scalability remains a bottleneck for widespread adoption.[167]
Regulatory and Ethical Considerations
Biosafety and Risk Assessment Protocols
Biosafety protocols for viral vectors are primarily governed by the NIH Guidelines for Research Involving Recombinant or Synthetic Nucleic Acid Molecules, which classify experiments based on potential risks such as replication competence, host range, and genetic inserts.[169] These guidelines require institutional biosafety committees (IBCs) to conduct site-specific risk assessments, evaluating factors including vector tropism, potential for recombination, and environmental release risks before approving work.[169] The CDC and NIH's Biosafety in Microbiological and Biomedical Laboratories (BMBL, 6th edition, 2020) emphasizes a protocol-driven risk assessment approach, integrating agent hazards, laboratory procedures, and personnel training to determine appropriate biosafety levels (BSL-1 to BSL-4).[170] Risk assessments distinguish between replication-deficient and replication-competent vectors, with the former typically requiring lower containment if they lack virulence factors or oncogenic inserts. For lentiviral vectors, derived from HIV-1, NIH recommends BSL-2 containment due to their Risk Group 2 (RG2) classification, involving work in biosafety cabinets (BSCs), personal protective equipment (PPE) like gloves and lab coats, and decontamination procedures; higher levels apply if pseudotyped for broad tropism or containing hazardous transgenes.[171] Adenoviral vectors, often RG1 or RG2, mandate BSL-2 practices including access restriction, medical surveillance, and spill response protocols, as they can cause respiratory illness despite attenuation.[172] Adeno-associated virus (AAV) vectors may qualify for BSL-1 if replication-incompetent, free of helper virus, and without toxic inserts, but default to BSL-2 for animal or high-titer work to mitigate aerosol transmission risks.[173] Protocols extend to animal studies under Animal Biosafety Levels (ABSL-1 to ABSL-3), with viral shedding monitoring post-administration; for instance, ABSL-2 is required for adenoviral vector use in rodents to prevent horizontal transmission.[174] Waste inactivation via autoclaving or chemical disinfectants, vector storage at -70°C or below, and annual IBC reviews ensure containment integrity.[175] For clinical applications, WHO and FDA-aligned assessments incorporate patient-specific risks like immune responses and long-term genomic integration, mandating Good Manufacturing Practice (GMP) facilities with validated inactivation methods.[11] These measures address empirical evidence of rare but documented incidents, such as inadvertent exposures in early trials, prioritizing causal pathways like vector escape over unsubstantiated fears.[176]Approval Processes and Global Variations
In the United States, the Food and Drug Administration (FDA) regulates viral vector-based products, including gene therapies and vaccines, as biologics under the Center for Biologics Evaluation and Research (CBER). The approval pathway begins with an Investigational New Drug (IND) application, supported by preclinical data on vector safety, biodistribution, and genotoxicity, followed by phased clinical trials demonstrating efficacy and managing risks like immunogenicity and insertional mutagenesis.[177] Full approval requires a Biologics License Application (BLA) with substantial evidence from controlled trials, chemistry, manufacturing, and controls (CMC) data ensuring vector purity and replication incompetence, and commitments to long-term follow-up for at least 15 years to monitor delayed adverse events.[111][142] The first FDA-approved viral vector gene therapy, voretigene neparvovec (Luxturna, an AAV2 vector for RPE65-mediated retinal dystrophy), received accelerated approval on December 19, 2017, based on phase 3 trial data showing improved vision, though confirmatory studies were required.[178] In the European Union, the European Medicines Agency (EMA) classifies viral vector products as advanced therapy medicinal products (ATMPs), requiring centralized marketing authorization with guidelines emphasizing vector design, non-clinical toxicology in relevant species, and clinical data on durability of transgene expression.[179] Approvals mandate comprehensive quality controls for adventitious agents and genomic integration risks, plus post-authorization safety monitoring via the Pharmacovigilance Risk Assessment Committee. The EMA's first viral vector approval was alipogene tiparvovec (Glybera, an AAV1 vector for lipoprotein lipase deficiency) on October 25, 2012, granted after phase 3 evidence of triglyceride reduction, but it was withdrawn in 2017 due to commercial viability despite conditional efficacy.[180] Recent approvals, such as those for AAV-based therapies, incorporate immunogenicity assessments tailored to vector capsid and seroprevalence.[181] Global variations reflect differing priorities in data rigor, timelines, and manufacturing oversight. China approved the world's first commercial gene therapy, Gendicine (an adenoviral vector expressing p53 for head and neck cancer), on October 16, 2003, via the National Medical Products Administration (NMPA), based on phase 1-3 trials showing tumor response rates up to 64%, though long-term survival data were limited compared to Western standards.[79] In Russia, the Ministry of Health granted full approval to Sputnik V (a human adenoviral vector COVID-19 vaccine) on August 11, 2020, after phase 1-2 interim data from 76 participants demonstrating immunogenicity, preceding phase 3 completion and sparking international scrutiny over expedited processes without emergency use restrictions.[182] Such approaches contrast with FDA and EMA requirements for larger phase 3 cohorts and manufacturing consistency, as evidenced by the World Health Organization's 2021 suspension of Sputnik V's emergency listing review due to Good Manufacturing Practice deficiencies at certain facilities.[183] In regions like India and Brazil, conditional approvals for viral vector vaccines during the COVID-19 pandemic often relied on bridging studies to foreign data, highlighting tensions between rapid access and harmonized safety evaluations under frameworks like the International Council for Harmonisation.[184] These divergences can accelerate deployment in urgent scenarios but raise concerns about equitable risk assessment, with Western agencies prioritizing extended follow-up to address vector-specific toxicities absent in faster tracks.[185]Ethical Debates on Human Application
The development of viral vectors for human gene therapy has prompted debates over informed consent, given the irreversible nature of genetic modifications and the potential for unforeseen adverse effects. In the 1999 clinical trial at the University of Pennsylvania involving an adenovirus vector for ornithine transcarbamylase deficiency, 18-year-old participant Jesse Gelsinger died from a massive immune response leading to multi-organ failure, highlighting failures in risk disclosure: prior animal studies showing deaths from similar doses were not fully conveyed to participants or regulators, and investigators had undisclosed financial ties to the sponsoring company.[186][187] This incident, which prompted a temporary halt to U.S. gene therapy trials by the FDA, underscored ethical lapses in balancing therapeutic promise against empirical risks, as vectors like adenoviruses can trigger severe inflammation despite preclinical data suggesting tolerability at lower doses.[161] Distinctions between somatic and germline applications intensify ethical scrutiny, as viral vectors primarily target non-reproductive cells for therapy but carry risks of off-target integration or unintended germline transmission via viral recombination or cellular uptake. Somatic edits address diseases like spinal muscular atrophy, approved in therapies such as Zolgensma using AAV9 vectors since 2019, yet critics argue that incomplete vector containment—evidenced by rare cases of vector DNA persistence in non-target tissues—raises consent issues for future offspring without their input.[188][189] Germline editing, though largely prohibited internationally due to heritable alterations, evokes concerns over eugenics-like selection and inequality, as empirical data from animal models show viral vectors can achieve germline integration at rates up to 10-20% in some protocols, potentially enabling enhancements beyond therapy.[190] Proponents contend that first-in-human somatic trials, regulated under frameworks like the NIH's Recombinant DNA Advisory Committee, mitigate these via phased risk assessments, but skeptics highlight systemic underreporting of long-term oncogenic risks in peer-reviewed literature.[191] Equity in access forms another core debate, as viral vector therapies command costs exceeding $2 million per treatment—e.g., Luxturna for retinal dystrophy at $850,000 in 2017—limiting availability to affluent populations despite public funding of foundational research.[192] This disparity, compounded by manufacturing scalability challenges for biologics like AAV vectors requiring specialized bioreactors, raises causal questions about whether such technologies exacerbate social divides rather than equitably curing monogenic diseases affecting 1 in 250 births globally.[188] In vaccine contexts, such as adenoviral vectors in AstraZeneca's COVID-19 product authorized in 2020, ethical tensions arose over mandates and global distribution inequities, with lower-income nations receiving only 0.1 doses per capita by mid-2021 versus 20+ in high-income countries, though vector-specific risks like rare thrombosis were empirically low at 1-2 per million doses.[193][83] Early precedents, including the 1975 Asilomar Conference organized by Paul Berg, established voluntary moratoriums on certain recombinant DNA experiments involving viral vectors to avert hypothetical biohazards like oncogenesis, influencing modern biosafety levels (BL1-BL4) that prioritize containment over outright bans.[151] These guidelines, born from first-principles assessment of vector pathogenicity—e.g., SV40 contamination risks in early polio vaccines—emphasized empirical containment efficacy, yet debates persist on whether self-regulation suffices amid incentives for rapid commercialization, as evidenced by post-Asilomar accelerations in vector engineering without proportional ethical oversight reforms.[194] Overall, while viral vectors have enabled successes like curing 90% of severe hemophilia B cases in AAV trials by 2022, ethical frameworks demand rigorous, transparent risk-benefit analyses to avoid prioritizing innovation over verifiable safety.[17]Recent Advances and Future Prospects
Innovations in Vector Engineering
Innovations in viral vector engineering have primarily focused on enhancing transduction efficiency, reducing immunogenicity, and improving tissue specificity, particularly for adeno-associated virus (AAV) capsids. Advances in AAV capsid design integrate rational mutagenesis, directed evolution, and artificial intelligence-driven approaches to generate novel variants with superior targeting capabilities; for instance, machine learning models trained since 2015 have enabled the creation of capsids exhibiting up to tenfold higher transduction in specific tissues while evading pre-existing immunity.[195][196] These engineered capsids, such as those developed for non-human primate models like AAV-ShD, demonstrate enhanced delivery to muscle and central nervous system tissues, addressing limitations in wild-type AAV serotypes.[197] Lentiviral vector engineering has emphasized safety enhancements through multi-generational refinements, including the development of second- and third-generation systems that eliminate accessory genes and incorporate self-inactivating long terminal repeats (LTRs) to minimize insertional mutagenesis risks.[198] Production protocols have been optimized to reduce replication-competent virus formation, with modifications like separated packaging and envelope cassettes achieving near-zero RCV incidence in clinical-grade batches as of 2020.[199] Recent strategies further boost efficiency by leveraging hypoxia-inducible factors for improved transduction in hard-to-transfect cells, yielding up to 5-fold increases in stable gene integration without compromising vector stability.[200] Adenoviral vectors have seen progress in gutless or helper-dependent designs, which remove all viral coding sequences to curtail inflammatory responses and enable larger transgene payloads up to 36 kb.[25] Recombineering techniques, advanced in the 2020s, facilitate rapid construction of serotype chimeras that evade hepatic sequestration by Kupffer cells, improving systemic delivery efficiency by 20-50% in preclinical models.[201] These modifications, combined with tropism-altering fiber protein swaps, have expanded applications in oncolytic therapies and vaccination platforms.[202]Clinical Pipeline and Market Trends
As of the second quarter of 2025, the clinical pipeline for viral vector-based therapies encompasses over 2,154 gene therapy candidates, with adeno-associated virus (AAV) vectors comprising the majority due to their favorable safety profile and tissue tropism for in vivo applications.[203] Lentiviral vectors dominate ex vivo approaches, particularly in oncology for CAR-T cell therapies, while adenoviral vectors persist in vaccine development and certain oncolytic applications. Approximately 250 AAV-specific candidates are advancing, led by over 180 companies targeting indications such as neuromuscular disorders, hemophilia, and inherited blindness.[204] Phase III trials predominate for hemophilia A and B, with ongoing efforts to mitigate immunogenicity and dose-limiting toxicities through capsid engineering.[205] Recent U.S. Food and Drug Administration (FDA) approvals underscore pipeline maturation: Elevidys (delandistrogene moxeparvovec, AAVrh74 for Duchenne muscular dystrophy) received full approval for ambulatory patients and accelerated approval for non-ambulatory ones in 2024; Beqvez (fidanacogene elaparvovec, AAV5 for hemophilia B) was approved in April 2024; and SKYSONA (elivaldogene autotemcel, lentiviral for cerebral adrenoleukodystrophy) marked the second lentiviral vector approval.[206][24][207] These follow earlier AAV successes like Roctavian (valoctocogene roxaparvovec for hemophilia A, 2023) and Hemgenix (etranacogene dezaparvovec for hemophilia B, 2022), reflecting a shift toward durable, one-time treatments despite challenges like vector shedding and hepatic toxicity.[58] Globally, China's approval of BBM-H901 (AAV-based) in April 2025 highlights regulatory divergence, with faster pathways in Asia accelerating pipeline diversity.[208] Market trends indicate robust expansion driven by approval momentum and rising demand for personalized therapies. The viral vector manufacturing sector, valued at $5.5 billion in 2023, is projected to reach $6.3 billion in 2025 and $18.8 billion by 2030, with a compound annual growth rate (CAGR) exceeding 19%.[209][210] North America holds 47% market share, fueled by U.S. biopharma investments, while Asia-Pacific grows fastest at over 22% CAGR through 2030 due to contract manufacturing scale-up.[211] Key drivers include scalability improvements in bioreactors and purification, though bottlenecks in plasmid DNA supply and empty capsid removal constrain output. Development costs remain high, averaging $1-2 billion per therapy, prompting partnerships like Coave Therapeutics' CNS-targeted capsid launch in May 2025.[212] Gene therapy financing dipped in Q2 2025 amid economic pressures, yet trial initiations rose, signaling sustained investor confidence in viral vectors' therapeutic edge over non-viral alternatives.[213]| Vector Type | Key Pipeline Focus | Notable 2023-2025 Approvals | Market Projection (2025 Value) |
|---|---|---|---|
| AAV | In vivo gene delivery (e.g., neuromuscular, ocular) | Elevidys (2024), Beqvez (2024), Roctavian (2023) | Dominant in $6.3B manufacturing segment[209] |
| Lentiviral | Ex vivo (e.g., CAR-T, hematopoietic) | SKYSONA (prior, reinforced pipeline) | Growing in oncology, ~20% CAGR[212] |
| Adenoviral | Vaccines, oncolytics | Limited new; legacy in COVID-era | Niche, <10% share but scalable[211] |
.pdf/page1-232px-How_viral_vector_COVID-19_Vaccines_Work_(English).pdf.jpg)
.pdf/page1-1545px-How_viral_vector_COVID-19_Vaccines_Work_(English).pdf.jpg)

