Recent from talks
Nothing was collected or created yet.
Neisseria gonorrhoeae
View on Wikipedia
| Neisseria gonorrhoeae | |
|---|---|
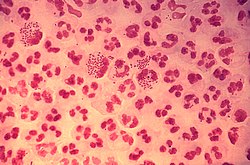
| |
| Gram-stain of gonococcal urethritis. Note distribution in neutrophils and presence of both intracellular and extracellular bacteria. (CDC) | |
| Scientific classification | |
| Domain: | Bacteria |
| Kingdom: | Pseudomonadati |
| Phylum: | Pseudomonadota |
| Class: | Betaproteobacteria |
| Order: | Neisseriales |
| Family: | Neisseriaceae |
| Genus: | Neisseria |
| Species: | N. gonorrhoeae
|
| Binomial name | |
| Neisseria gonorrhoeae | |
| Synonyms | |
Neisseria gonorrhoeae, also known as gonococcus (singular) or gonococci (plural), is a species of Gram-negative diplococci bacteria first isolated by Albert Neisser in 1879.[3] An obligate human pathogen, it primarily colonizes the mucosal lining of the urogenital tract; however, it is also capable of adhering to the mucosa of the nose,[4] pharynx, rectum, and conjunctiva.[5] It causes the sexually transmitted genitourinary infection gonorrhea[6] as well as other forms of gonococcal disease including disseminated gonococcemia, septic arthritis, and gonococcal ophthalmia neonatorum.
N. gonorrhoeae is oxidase positive and a microaerophile that is capable of surviving phagocytosis and growing inside neutrophils.[6] Culturing it requires carbon dioxide supplementation and enriched agar (chocolate agar) with various antibiotics (Thayer–Martin). It exhibits antigenic variation through genetic recombination of its pili and surface proteins that interact with the immune system.[3]
Sexual transmission is through vaginal, anal, or oral sex.[7] Sexual transmission may be prevented through the use of barrier protection.[8] Perinatal transmission may occur during childbirth, though it is preventable through antibiotic treatment of the mother before birth and application of antibiotic eye gel on the eyes of the newborn.[8] Gonococcal infections do not result in protective immunity; therefore, individuals may be infected multiple times. Reinfection is possible due to N. gonorrhoeae's ability to evade the immune system by varying its surface proteins.[9]
Asymptomatic infection is common in both males and females.[8][10] Untreated infection may spread to the rest of the body (disseminated gonorrhea infection), especially the joints (septic arthritis). Untreated infection in women may cause pelvic inflammatory disease and possible infertility due to the resulting scarring.[11] Gonorrhoea is diagnosed through cultures, Gram staining, or nucleic acid tests (i.e. polymerase chain reaction) of urine samples, urethral swabs, or cervical swabs.[12][13] Chlamydia co-testing and testing for other STIs is recommended due to high rates of co-infection.[14]
Antibiotic resistance in N. gonorrhoeae is a growing public health concern, especially given its propensity to develop resistance easily.[15] This ability of N. gonorrhoeae to rapidly adapt to novel antimicrobial treatments has been seen several times since the 1930s, making numerous treatment plans obsolete. Some strains have exhibited resistance to the current ceftriaxone treatments.[16]
Microbiology
[edit]
Neisseria species are fastidious, Gram-negative cocci (though some species are rod-shaped and occur in pairs or short chains) that require nutrient supplementation to grow in laboratory cultures.[18] They are facultative intracellular pathogens, meaning they can persist and colonize within host cells but can also multiply outside the host cellular environment.[19][20][21] They typically appear in pairs (diplococci), resembling the shape of coffee beans.[21] Members of this genus do not form endospores and are nonmotile, except for pathogenic species, which are capable of moving using twitching motility;[22] most are also obligate aerobes.[23][24] Of the 17 species that colonize humans, only two are pathogenic: N. gonorrhoeae, which causes gonorrhea, and N. meningitidis, a leading cause of bacterial meningitis.[25][26]
Culture and identification
[edit]
N. gonorrhoeae can be isolated on Thayer–Martin agar (or VPN) agar in an atmosphere enriched with 3-7% carbon dioxide.[12] Thayer–Martin agar is a chocolate agar plate (heated blood agar) containing nutrients and antimicrobials (vancomycin, colistin, nystatin, and trimethoprim). This agar preparation facilitates the growth of Neisseria species while inhibiting the growth of contaminating bacteria and fungi. Martin Lewis and New York City agar are other types of selective chocolate agar commonly used for Neisseria growth.[12] N. gonorrhoeae is oxidase positive (possessing cytochrome c oxidase) and catalase positive (able to convert hydrogen peroxide to oxygen).[12] When incubated with the carbohydrates lactose, maltose, sucrose, and glucose, N. gonorrhoeae will oxidize only the glucose.[12]
Metabolism
[edit]Carbon
[edit]Unlike other Neisseria species that can also metabolize maltose, N. gonorrhoeae is capable of using only glucose, pyruvate, and lactate as central carbon sources, and glucose is catabolized via both the Entner-Doudoroff (ED) and pentose phosphate (PP) pathways, and the ED pathway is the primary oxidative method.[19][27] Use of these pathways is necessary as N. gonorrhoeae is incapable of glucose catabolism via the Embden-Meyerhof-Parnas (EMP) pathway due its lack of the phosphofructokinase (PFK) gene; however, the fructose 1,6-bisphosphatase enzyme is present to allow for gluconeogenesis to occur.[19]
Glucose is first metabolized through the ED pathway to produce pyruvate and glyceraldehyde 3-phosphate, the latter of which can then further metabolized by enzymes of the EMP pathway to yield another molecule of pyruvate.[28] The resultant pyruvate molecules are then converted into acetyl-CoA, which can then be incorporated as a substrate for the citric acid cycle (CAC) to yield high-energy electron carriers that will be used by the electron transport chain (ETC) for ATP production; however, the CAC is largely used for generating biosynthetic precursors rather than for catabolic purposes.[19][29] This is due in part to inhibited expression of several CAC enzymes in the presence of glucose, pyruvate, or lactate. These enzymes, namely citrate synthase, aconitase, and isocitrate dehydrogenase, are needed for the incorporation of acetate. Instead, a partial CAC has been observed, where α-ketoglutarate is formed by glutamate dehydrogenase or transamination of oxaloacetate and glutamate by aspartate aminotransferase (yielding aspartate and α-ketoglutarate).[27][29] The CAC then continues from there to yield oxaloacetate, which is an important precursor molecule for several biosynthetic pathways.[29] Another differentiating aspect of the gonococcal CAC is the lack of malate dehydrogenase, which is instead replaced by a membrane-bound malate:quinone-oxidoreductase that operates independently of NAD+ by directly transferring electrons to ubiquinone.[19]
Conversely, acetyl-CoA that does not enter the CAC but enters the phosphotransacetylase-acetate kinase (PTA-AckA) pathway, where it can be converted into acetate by phosphorylation (to form acetyl phosphate and release coenzyme A) and dephosphorylation to form ATP.[30] While this acetate can enter the CAC for further oxidation, this does not occur so long as other carbon sources such as glucose or lactate are present, in which case it is excreted from the cell or incorporated for lipid synthesis.[28][31] N. gonorrhoeae lack the glyoxylate shunt, preventing them from using acetate to form CAC intermediates to replenish the cycle.[28][27]
A significant portion of the glyceraldehyde 3-phosphate formed in gonococci is recycled via the gluconeogenic pathway to reform glucose 6-phosphate, as well as the intermediate fructose 6-phosphate. Both of these can then be used for pentose synthesis in the PP pathway via the oxidative and non-oxidative pathways, respectively, for subsequent nucleotide formation as well as energy production.[27]
N. gonorrhoeae, like other pathogenic members of the genus Neisseria, are capnophiles, meaning they require higher-than-normal concentrations of carbon dioxide (CO2) to grow, either in the form of CO2 or bicarbonate (HCO3−) depending on the bacterial strain. This requirement must be met exogenously during the lag and stationary growth phases, though it appears to be met through high metabolic CO2 productions in the exponential phase. Assimilation of this CO2 in Neisseria species is done by carbonic anhydrase and phosphoenolpyruvate enzymes in the periplasmic space and the cytoplasm, respectively.[27]
Lactate catabolism is also of particular importance for gonococci, both for pathogenicity and for growth.[19] External lactate is transported in to the cell via lactate permease (LctP).[19] The N. gonorrhoeae genome encodes for three lactate dehydrogenase (LDH) enzymes for that allow for metabolism of both L-lactate and D-lactate: a cytoplasmic NAD+-dependent D-lactate dehydrogenase (LdhA), which is responsible for and two membrane-bound LDHs, one specific to L-lactate (LldD) and the other specific to D-lactate (LdhD).[19][32] The membrane-bound LDHs have been determined to be flavoprotein-containing respiratory enzymes that directly oxidize lactate to reduce ubiquinone. While these enzymes do not directly pump protons (H+ ions) into the periplasmic space, it is proposed that the reduction of ubiquinone by these enzymes is capable of feeding into the larger ETC.[32]
Electron transport chain and oxidative phosphorylation
[edit]As an obligate human pathogen and a facultative anaerobic capnophile, Neisseria gonorrhoeae typically colonizes mucosal surfaces in microaerobic environments, such as those in the genitourinary tract.[19] Growth in areas where oxygen concentrations are limited requires a terminal oxidase with a high affinity for oxygen; in gonococci, oxygen reduction is performed by a ccb3 -type cytochrome oxidase. In addition to aerobic respiration, gonococci can also perform anaerobic respiration via the reduction of nitrite (NO2) to nitric oxide (NO) as well as reduction of NO to nitrous oxide (N2O).[19][33]
Several enzymes contribute electrons to the intramembranous ubiquinone pool, the first step in the ETC. These include the membrane-bound LDHs (LldD and LdhD), NADH:ubiquinone oxidoreductase (aka NADH dehydrogenase; Nuo complex I), Na+-translocating NADH dehydrogenase (Nqr), succinate dehydrogenase (SDH), and the membrane-bound NAD+-independent malate:quinone-oxidoreductase (MqR).[19]
Following the initial transfer of electrons to ubiquinone, proposed schematics for the organization of the gonococcal ETC suggest the electrons can be further passed down the chain by reduction of the cytochrome bc1 complex or can be directly transferred to NO as a terminal electron acceptor by NO reductase (NorB).[19][34] In the case of the former, electrons can then be passed from the bc1 complex along two alternative pathways via the reduction of either cytochrome c4 or c5. Both of these cytochromes transfer electrons to the terminal cytochrome ccb3 oxidase for the reduction of O2 to form H2O under aerobic conditions.[19][33]
Gonococci also reduce NO2 via an inducible outer membrane-attached copper-containing nitrite reductase (AniA, a member of the NirK protein family) under anaerobic conditions, though this process has also been noted in microaerobic conditions as a means of supplementing growth.[34] This leads to the formation of NO that is subsequently reduced to N2O in a partial denitrification pathway.[19][33][34] The ccb3 oxidase of N. gonorrhoeae, dissimilarly to other members of the Neisseria genus, is a tri-heme protein that can transfer electrons not only to O2 (conserved across Neisseria species) but also to AniA for NO2 reduction. This is in addition to the typical process of receiving electrons transferred from cytochrome c5.[34][35]
The general purpose of the ETC is the formation of the electrochemical gradient of hydrogen ions (H+ or protons), resulting from concentration differences across the plasma membrane, needed to power ATP production in a process known as oxidative phosphorylation.[36] In gonococci, movement of protons into the periplasmic space is accomplished by the Nuo complex I, the cytochrome bc1 complex, and cytochrome ccb3.[19][34][37] Subsequently, ATP synthesis is performed by the F1F0 ATP synthase, a two-part protein complex present in gonococci as well as numerous other species across phylogenetic domains.[38] This complex couples proton translocation back into the cytoplasm along its gradient with mechanical rotation to generate ATP.[39]
Iron
[edit]The general purpose of the ETC is the formation of the electrochemical gradient of hydrogen ions (H+ or protons), resulting from concentration differences across the plasma membrane, needed to power ATP production in a process known as oxidative phosphorylation.[36] In gonococci, movement of protons into the periplasmic space is accomplished by the Nuo complex I, the cytochrome bc1 complex, and cytochrome ccb3.[19][34][37] Subsequently, ATP synthesis is performed by the F1F0 ATP synthase, a two-part protein complex present in gonococci as well as numerous other species across phylogenetic domains.[38] This complex couples proton translocation back into the cytoplasm along its gradient with mechanical rotation to generate ATP.[39]
To acquire the necessary iron, gonococci produce TonB-dependent transporters (TDTs) on the surface of their outer membrane that can directly extract iron, along with other metals, from their respective carrier proteins. Some of these include transferrin binding proteins A (TbpA) and B (TbpB), lactoferrin-binding proteins A (LbpA) and B (LbpB), and hemoglobin/hemoglobin-haptoglobin binding proteins HpuB and HpuA.[19][40] In addition to these proteins, gonococci are also capable of using siderophores, or compounds that are capable of chelating iron in the environment, that are produced by other bacteria; however, gonococcal cells are incapable of synthesizing siderophores themselves. These xenosiderophores are taken up by the TDT FetA through the outer membrane and then brought into the cell by the fetBCDEF transporter system.[19][40]
Along with the sequestration defence that can be further upregulated by host inflammation, humans also produce siderocalins that can chelate siderophores as a further method of inhibiting pathogenic bacterial growth. These are sometimes ineffective against N. gonorrhoeae, which can colonize intracellularly, particularly in phagocytic cells such as macrophages and neutrophils. Increases in host intracellular iron also downregulate some of the intracellular pathogen-killing mechanisms; coincidentally, pathogenic Neisseria can alter several host cell mechanisms that ultimately allow the pathogen to take most of the available iron from the host immune cell.[40]
Surface molecules
[edit]On its surface, N. gonorrhoeae bears hair-like pili, surface proteins with various functions, and sugars called lipooligosaccharide. The pili mediate adherence, movement, and DNA exchange. The opacity-associated (Opa) proteins interact with the immune system, as do the porins. Lipooligosaccharide is an endotoxin that provokes an immune response. All of these are antigenic and exhibit antigenic variation. The pili, Opa proteins, porins, and even the lipooligosaccharide have mechanisms to inhibit the immune response, making asymptomatic infection possible.[41]
Opa proteins
[edit]Phase-variable opacity-associated (Opa) adhesin proteins are used by N. gonorrhoeae as part of evading the immune response in a host cell. At least 12 Opa proteins are known, and the many variations of surface proteins make recognizing N. gonorrhoeae and mounting a defense by immune cells more difficult.[42] Opa proteins are in the outer membrane and facilitate a response when the bacteria interacts with a variety of host cells. These proteins bind to various epithelial cells, and allow N. gonorrhoeae to increase the length of infection as well as increase the amount of invasion into other host cells.[43]
Type IV pili
[edit]
Dynamic polymeric protein filaments called type IV pili allow N. gonorrhoeae to do many bacterial processes, including adhesion to surfaces, transformation competence, twitching motility, and immune response evasions.[19] To enter the host the bacteria uses the pili to adhere to and penetrate mucosal surfaces. The pili are a pivotal virulence factor for N. gonorrhoeae; without them, the bacterium is unable to promote colonization.[44] For motility, individual bacteria use their pili in a manner that resembles a grappling hook: first, they are extended from the cell surface and attach to a substrate. Subsequent pilus retraction drags the cell forward. The resulting movement is referred to as twitching motility. N. gonorrhoeae can pull 100,000 times its own weight,[45] and the pili used to do so are amongst the strongest biological motors known to date, exerting one nanonewton.[45] The PilF and PilT ATPase proteins are responsible for powering the extension and retraction of the type IV pilus, respectively. The adhesive functions of the gonococcal pilus play a role in microcolony aggregation and biofilm formation. These pili are also used to avoid immune responses from the cell they are invading by having their type IV pili antigenically vary. The main pilus filament is replaced by variable DNA sequences very frequently.[19] By doing this process rapidly, they can create a diversity of pili on their surface and evade the host cell's immune response.[44]
Lipooligosaccharide
[edit]Lipooligosaccharide is a low-weight version of lipopolysaccharide present on the surfaces of most other Gram-negative bacteria. It is a sugar (saccharide) side chain attached to lipid A (thus "lipo-") in the outer membrane coating the cell wall of the bacteria. The root "oligo" refers to the fact that it is a few sugars shorter than the typical lipopolysaccharide.[6] As an endotoxin, it provokes inflammation. The shedding of lipooligosaccharide by the bacteria is sometimes responsible for issues associated with pelvic inflammatory disease.[6] Although it functions primarily as an endotoxin, lipooligosaccharide may disguise itself with host sialic acid and block initiation of the complement cascade.[6]
Antigenic variation
[edit]N. gonorrhoeae evades the immune system through a process called antigenic variation.[46] This process allows N. gonorrhoeae to recombine its genes and alter the antigenic determinants that adorn its surface,[6] such as the Type IV pili.[47] Simply stated, the chemical composition of molecules are changed due to changes at the genetic level.[9] N. gonorrhoeae is able to vary the composition of its pili and lipooligosaccharide. Of these, the pili exhibit the most antigenic variation due to chromosomal rearrangement.[11][6] The pilS gene is an example of this ability to rearrange, as its combination with the pilE gene is estimated to produce over 100 variants of the PilE protein.[9] These changes allow for adjustment to local environmental differences at the site of infection, evasion of recognition by targeted antibodies, and inhibit the formation of an effective vaccine.[9]
In addition to gene rearrangement, it is also naturally competent, meaning it can acquire extracellular DNA from the environment via its type IV pilus, specifically proteins PilQ and PilT.[48] These processes allow N. gonorrhoeae to acquire and spread new genes, disguise itself with different surface proteins, and prevent the development of immunological memory – an ability which has contributed to antibiotic resistance and impeded vaccine development.[49]
Phase variation
[edit]Phase variation is similar to antigenic variation, but instead of changes at the genetic level altering the composition of molecules, these genetic changes result in the activation or deactivation of a gene.[9] Phase variation most often arises from a frameshift in the expressed gene.[9] The Opa proteins of N. gonorrhoeae rely strictly on phase variation.[9] Every time the bacteria replicate, they may switch multiple Opa proteins on or off through slipped-strand mispairing. That is, the bacteria introduce frameshift mutations that bring genes in or out of frame. The result is that different Opa genes are translated every time.[6] Pili are varied by antigenic variation, but also phase variation.[9] Frameshifts occur in both the pilE and pilC genes, effectively turning off the expression of pili in situations when they are not needed, such as during intracellular colonization as opposed to extracellular mucosal cell surface adhesion.[9]
Survival of gonococci
[edit]After gonococci invade and transcytose the host epithelial cells, they land in the submucosa, where neutrophils promptly consume them.[6] The pili and Opa proteins on the surface may interfere with phagocytosis,[11] but most gonococci end up in neutrophils. The exudates from infected individuals contain many neutrophils with ingested gonococci. Neutrophils release an oxidative burst of reactive oxygen species in their phagosomes to kill the gonococci.[50] However, a significant fraction of the gonococci can resist killing through the action of their catalase,[6] which breaks down reactive oxygen species and can reproduce within the neutrophil phagosomes.[51]
The bacterial RecA protein, which mediates repair of DNA damage, plays a crucial role in gonococcal survival.[52] N. gonorrhoeae may replace DNA damaged in neutrophil phagosomes with DNA from neighboring gonococci.[53] The process in which recipient gonococci integrate DNA from neighboring gonococci into their genome is called transformation.[54]

Genome
[edit]The genomes of several strains of N. gonorrhoeae have been sequenced. Most of them are about 2.1 Mb in size and encode 2,100 to 2,600 proteins (although most seem to be in the lower range).[55] For instance, strain NCCP11945 consists of one circular chromosome (2,232,025 bp) encoding 2,662 predicted open reading frames (ORFs) and one plasmid (4,153 bp) encoding 12 predicted ORFs. The estimated coding density over the entire genome is 87%, and the average G+C content is 52.4%, values that are similar to those of strain FA1090. The NCCP11945 genome encodes 54 tRNAs and four copies of 16S-23S-5S rRNA operons.[56]
Horizontal gene transfer
[edit]Horizontal gene transfer, also termed lateral gene transfer, is the sharing of genetic information amongst living organisms.[57] This transmission of information is a driving force of antibiotic resistance in N. gonorrhoeae.[58][59] Studies have identified that N. gonorrhoeae has obtained methods of antimicrobial resistance by way of horizontal gene transfer from other Neisseria species including N. lactamica, N. macacae, and N. mucosa. [60]
Transformation in N. gonorrhoeae is performed by the type IV pilus, where the DNA is bound and brought into the cell, followed by processing and homologous recombination.[61]
Found in some genomes of Neisseria gonorrhoeae, the gonococcal genetic island (GGI), a genomic island (GI) specific to gonococci, has been identified as a mobile genetic element that is horizontally acquired.[62][63] GGI is involved with antimicrobial resistance, transmission of genetic information, and iron acquisition.[62] The genes within the gonococcal genetic island encode for the infamous type IV secretion system (T4SS), which is responsible for DNA secretion and is essential for biofilm formation.[62][64]
In 2011, researchers at Northwestern University found evidence of a human DNA fragment in a N. gonorrhoeae genome, the first example of horizontal gene transfer from humans to a bacterial pathogen.[65][66]
Disease
[edit]Symptoms
[edit]Symptoms of infection with N. gonorrhoeae differ depending on the site of infection and many infections are asymptomatic independent of sex.[67][41][7] Depending on the route of transmission, N. gonorrhoeae may cause infection of the throat (pharyngitis) or infection of the anus/rectum (proctitis).[68][11]
Disseminated gonococcal infections can occur when N. gonorrhoeae enters the bloodstream, often spreading to the joints and causing a rash (dermatitis-arthritis syndrome).[68] Dermatitis-arthritis syndrome results in joint pain (arthritis), tendon inflammation (tenosynovitis), and painless non-pruritic (non-itchy) dermatitis.[11] Disseminated infection and pelvic inflammatory disease in women tend to begin after menses due to reflux during menses, facilitating spread.[68] In rare cases, disseminated infection may cause infection of the meninges of the brain and spinal cord (meningitis) or infection of the heart valves (endocarditis).[68][69]
Male
[edit]In symptomatic men, the primary symptom of genitourinary infection is urethritis – burning with urination (dysuria), increased urge to urinate, and a pus-like (purulent) discharge from the penis. The discharge may be foul-smelling.[68] If untreated, scarring of the urethra may result in difficulty urinating. Infection may spread from the urethra in the penis to nearby structures, including the testicles (epididymitis/orchitis), or to the prostate (prostatitis).[68][11][70]
Female
[edit]
In symptomatic women, the primary symptoms of genitourinary infection are increased vaginal discharge, burning with urination (dysuria), increased urge to urinate, pain with intercourse, or menstrual abnormalities. Pelvic inflammatory disease results if N. gonorrhoeae ascends into the pelvic peritoneum (via the cervix, endometrium, and fallopian tubes). The resulting inflammation and scarring of the fallopian tubes can lead to infertility and an increased risk of ectopic pregnancy.[68] Pelvic inflammatory disease develops in 10 to 20% of the females infected with N. gonorrhoeae.[68]
Neonates (perinatal infection)
[edit]In perinatal infection, the primary manifestation is infection of the eye (neonatal conjunctivitis or ophthalmia neonatorum) when the newborn is exposed to N. gonorrhoeae in the birth canal. The eye infection can lead to corneal scarring or perforation, ultimately resulting in blindness. If the newborn is exposed during birth, conjunctivitis occurs within 2–5 days after birth and is severe.[68][69] Gonococcal ophthalmia neonatorum, once common in newborns, is prevented by the application of erythromycin (antibiotic) gel to the eyes of babies at birth as a public health measure. Silver nitrate is no longer used in the United States.[69][68]
Transmission
[edit]N. gonorrhoeae is most often transmitted through vaginal, oral, or anal sex; nonsexual transmission is unlikely in adult infection.[7] It can also be transmitted to a newborn during passage through the birth canal if the mother has an untreated genitourinary infection. Given the high rate of asymptomatic infection, it is recommended that pregnant women be tested for gonococcal infection before birth.[7] Communal baths, shared towels or fabrics, rectal thermometers, and improper hand hygiene by caregivers have been identified as potential means of transmission in pediatric settings.[71]
Traditionally, the bacterium was thought to move attached to spermatozoa, but this hypothesis did not explain female-to-male transmission of the disease. A recent study suggests that rather than "surf" on wiggling sperm, N. gonorrhoeae bacteria use pili to anchor onto proteins in the sperm and move through coital fluid.[72]
Infection
[edit]Successful transmission is followed by adherence to the epithelial cells found at the infected mucosal site by the bacterium's type IV pili. The pili's ability to attach and subsequently retract pulls N. gonorrhoeae towards the epithelial membrane at the surface of the mucosal cell.[15] Post attachment, N. gonorrhoeae replicates its genome and divides to form microcolonies.[15] Gonococcal infection is sometimes aided by the membrane cofactor protein, CD46, as it has been known to act as a receptor for gonococcal pilus.[41] Additionally, interaction with pili has been shown to cause cytoskeletal rearrangement of the host cell, further demonstrating that gonococcal pili engagement disrupts the response of the host cell and increases the likelihood of successful infection.[41] During growth and colonization, N. gonorrhoeae stimulates the release of pro-inflammatory cytokines and chemokines from host immune cells that result in the recruitment of neutrophils to the area.[9] These phagocytic cells typically take in foreign pathogens and destroy them; however, N. gonorrhoeae's ability to manipulate the host cell response allows the pathogen to survive within these immune cells and evade elimination.[9]
Laboratory diagnosis
[edit]The primary detection methods for Neisseria gonorrhoeae are nucleic acid amplification tests, which are the most sensitive techniques available.[73][74] Other methods of detection include microscopy and culture.[73]
Prevention
[edit]Transmission is reduced by using latex barriers (e.g., condoms or dental dams) during sex and by limiting sexual partners.[8] Condoms and dental dams should be used during oral and anal sex as well. Spermicides, vaginal foams, and douches are not effective methods for transmission prevention.[6]
Vaccine
[edit]A vaccine against N. gonorrhoeae is becoming more necessary due to the growing incidence of cases, increasing antimicrobial resistance, and its impact on reproductive health.[75] There are problems that have hampered vaccine development including: the absence of immunity post-infection, exclusively human hosts, and antigenic and phase variation of potential vaccine targets.[76] Currently, there are several N. gonorrhoeae vaccines in development, including an outer membrane vesicle vaccine.[76] This includes the NGoXIM, the native OMV, and Bexsero/4CMenB vaccine candidates, which are all in the late clinical stages of development.[77] The creation of a vaccine for N. gonorrhoeae has several potential public health impacts. In one estimate, a vaccine for the heterosexual population given before sexual activity occurs showed that the prevalence of N. gonorrhoeae could be reduced by up to 90% after 20 years.[76] In 2023, the FDA gave fast-track designation to a gonorrhoea vaccine candidate from GSK[78] . NHS England has also launched the world's first vaccination program against gonorrhea, using a vaccine against Neisseria meningitidis which was found to be partially effective against N. gonorrhoeae [79][80] .
Treatment
[edit]Currently, the CDC recommends a single dose of the injectable cephalosporin, ceftriaxone, as the first line of defense against gonococcal infections.[81] Individuals weighing less than 150 kg are typically prescribed a ceftriaxone concentration of 500 mg, while individuals who weigh over 150 kg are typically prescribed a dose of 1 g. Although ceftriaxone is not the only cephalosporin that has been effective at treating gonorrhoeae, it is the most advantageous.[75] In the event of a cephalosporin allergy, the CDC recommends a dual treatment of gentamicin and azithromycin. Each drug should be administered as a single dose, with the gentamicin entering intramuscularly at a concentration of 240 mg, along with 2 g of azithromycin taken orally.[75] If an individual is not allergic to cephalosporins but ceftriaxone is unavailable, an alternative treatment is a single dose of 800 mg cefixime consumed orally.[75] In all of these cases, combination therapy and co-treatment for chlamydia is recommended, as simultaneous infections are common.[82]
Antibiotic resistance
[edit]Antibiotic resistance in gonorrhea was first identified in the 1940s. Gonorrhea was treated with penicillin, but doses had to be progressively increased to remain effective. By the 1970s, penicillin-and tetracycline-resistant gonorrhea emerged in the Pacific Basin. These resistant strains then spread to Hawaii, California, the rest of the United States, Australia and Europe. Fluoroquinolones were the next line of defense, but soon resistance to this antibiotic emerged, as well. Since 2007, standard treatment has been third-generation cephalosporins, such as ceftriaxone, which are considered to be our "last line of defense".[83][84] Recently, a high-level ceftriaxone-resistant strain of gonorrhea called H041 was discovered in Japan. Lab tests found it to be resistant to high concentrations of ceftriaxone, as well as most of the other antibiotics tested. Within N. gonorrhoeae, genes exist that confer resistance to every single antibiotic used to cure gonorrhea, but thus far, they do not coexist within a single gonococcus. However, because of N. gonorrhoeae's high affinity for horizontal gene transfer, antibiotic-resistant gonorrhea is seen as an emerging public health threat.[84]
Before 2007, fluoroquinolones were a common treatment recommendation for gonorrhoeae. The CDC stopped suggesting these systemic bacterial agents once a resistant strain of N. gonorrhoeae emerged in the United States. The removal of fluoroquinolones as a potential treatment left cephalosporins as the only viable antimicrobial option for gonorrhea treatment. Wary of further gonococcal resistance, the CDC's recommendations shifted in 2010 to a dual therapy strategy—cephalosporin with either azithromycin or doxycycline. Despite these efforts, resistant N. gonorrhoeae had been reported in five continents by 2011, further limiting treatment options and recommendations. Antimicrobial resistance is not universal and N. gonorrhoeae strains in the United States continue to respond to a combination regimen of ceftriaxone and azithromycin.[85]
Serum resistance
[edit]As a Gram negative bacterium, N. gonorrhoeae requires defense mechanisms to protect itself against the complement system (or complement cascade), whose components are found with human serum.[41] There are three different pathways that activate this system however, they all result in the activation of complement protein 3 (C3).[86] A cleaved portion of this protein, C3b, is deposited on pathogenic surfaces and results in opsonization as well as the downstream activation of the membrane attack complex.[86] N. gonorrhoeae has several mechanisms to avoid this action.[15] As a whole, these mechanisms are referred to as serum resistance.[15]
History
[edit]Name origin
[edit]Neisseria gonorrhoeae is named for Albert Neisser, who isolated it as the causative agent of the disease gonorrhea in 1878.[15][3] Galen (130 AD) coined the term "gonorrhea" from the Greek gonos which means "seed" and rhoe which means "flow".[87][9] Thus, gonorrhea means "flow of seed", a description referring to the white penile discharge, assumed to be semen, seen in male infection.[15]
Discovery
[edit]In 1878, Albert Neisser isolated and visualized N. gonorrhoeae diplococci in samples of pus from 35 men and women with the classic symptoms of genitourinary infection with gonorrhea – two of whom also had infections of the eyes.[9] In 1882, Leistikow and Loeffler were able to grow the organism in culture.[15] Then in 1883, Max Bockhart proved conclusively that the bacterium isolated by Albert Neisser was the causative agent of the disease known as gonorrhea by inoculating the penis of a healthy man with the bacteria.[9] The man developed the classic symptoms of gonorrhea days after, satisfying the last of Koch's postulates. Until this point, researchers debated whether syphilis and gonorrhea were manifestations of the same disease or two distinct entities.[88][9] One such 18th-century researcher, John Hunter, tried to settle the debate in 1767[9] by inoculating a man with pus taken from a patient with gonorrhea. He erroneously concluded that syphilis and gonorrhea were indeed the same disease when the man developed the copper-colored rash that is classic for syphilis.[86][88] Although many sources repeat that Hunter inoculated himself,[86][15] others have argued that it was another man.[89] After Hunter's experiment other scientists sought to disprove his conclusions by inoculating other male physicians, medical students,[15] and incarcerated men with gonorrheal pus, who all developed the burning and discharge of gonorrhea. One researcher, Ricord, took the initiative to perform 667 inoculations of gonorrheal pus on patients of a mental hospital, with zero cases of syphilis.[9][15] Notably, the advent of penicillin in the 1940s made effective treatments for gonorrhea available.[90]
See also
[edit]References
[edit]- ^ Euzéby JP, Parte AC. "Genus Neisseria". List of Prokaryotic Names with Standing in Nomenclature (LPSN). Retrieved 7 July 2017.
- ^ Neisser A (1879). "Ueber eine der Gonorrhoe eigentümliche Micrococusform" [About a micrococus form peculiar to gonorrhea]. Centralblatt für die medizinischen Wissenschaften (in German). 17 (28): 497–500.
- ^ a b c O'Donnell JA, Gelone SP (2009). "Bacterial Cause of PID: Gonorrhoeae". Pelvic Inflammatory Disease. Infobase Publishing. ISBN 978-1-4381-0159-0.
- ^ Inaba S, Aizawa Y, Kataoka S, Saitoh A (December 2023). "Purulent nasal discharge due to gonococcal nasopharyngitis in a neonate". Journal of Infection and Chemotherapy. 29 (12): 1164–1166. doi:10.1016/j.jiac.2023.08.005. PMID 37558089.
- ^ Mahapure K, Singh A (August 2023). "A Review of Recent Advances in Our Understanding of Neisseria gonorrhoeae". Cureus. 15 (8) e43464. doi:10.7759/cureus.43464. PMC 10498933. PMID 37711920.
- ^ a b c d e f g h i j k Ryan KJ, Ray CG, eds. (2004). Sherris Medical Microbiology (4th ed.). McGraw Hill. ISBN 978-0-8385-8529-0.[page needed]
- ^ a b c d "Detailed STD Facts - Gonorrhea". www.cdc.gov. 26 September 2017. Retrieved 7 December 2017.
- ^ a b c d "2015 Sexually Transmitted Diseases Treatment Guidelines". CDC. Centers for Disease Control and Prevention, U.S. Department of Health & Human Services. Archived from the original on 22 December 2015.
- ^ a b c d e f g h i j k l m n o p q Hill SA, Masters TL, Wachter J (September 2016). "Gonorrhea - an evolving disease of the new millennium". Microbial Cell. 3 (9): 371–389. doi:10.15698/mic2016.09.524. PMC 5354566. PMID 28357376.
- ^ "Final Recommendation Statement: Chlamydia and Gonorrhea: Screening". U.S. Preventive Services Task Force. Retrieved 7 December 2017.
- ^ a b c d e f Levinson W (2014). Review of medical microbiology and immunology. McGraw-Hill Education. ISBN 978-0-07-181811-7. OCLC 871305336.[page needed]
- ^ a b c d e Ng LK, Martin IE (January 2005). "The laboratory diagnosis of Neisseria gonorrhoeae". The Canadian Journal of Infectious Diseases & Medical Microbiology. 16 (1): 15–25. doi:10.1155/2005/323082. PMC 2095009. PMID 18159523.
- ^ "Gonococcal Infections - 2015 STD Treatment Guidelines". 4 January 2018.
- ^ MacKenzie CR, Henrich B, eds. (2012). Diagnosis of sexually transmitted diseases: methods and protocols. Humana Press. ISBN 978-1-61779-936-5. OCLC 781681739.
- ^ a b c d e f g h i j k Quillin SJ, Seifert HS (April 2018). "Neisseria gonorrhoeae host adaptation and pathogenesis". Nature Reviews. Microbiology. 16 (4): 226–240. doi:10.1038/nrmicro.2017.169. PMC 6329377. PMID 29430011.
- ^ Costa-Lourenço AP, Barros Dos Santos KT, Moreira BM, Fracalanzza SE, Bonelli RR (1 October 2017). "Antimicrobial resistance in Neisseria gonorrhoeae: history, molecular mechanisms and epidemiological aspects of an emerging global threat". Brazilian Journal of Microbiology. 48 (4): 617–628. doi:10.1016/j.bjm.2017.06.001. PMC 5628311. PMID 28754299.
- ^ Sell J, Nasir M, Courchesne C (May 2021). "Urethritis: Rapid Evidence Review". American Family Physician. 103 (9): 553–558. PMID 33929174.
- ^ Van Putten J, Tønjum T (2010). "Neisseria". Infectious Diseases. pp. 1676–1689. doi:10.1016/b978-0-323-04579-7.00168-4. ISBN 978-0-323-04579-7.
- ^ a b c d e f g h i j k l m n o p q r s t Green LR, Cole J, Parga EF, Shaw JG (2022). Neisseria gonorrhoeae physiology and pathogenesis. Advances in Microbial Physiology. Vol. 80. pp. 35–83. doi:10.1016/bs.ampbs.2022.01.002. ISBN 978-0-323-98869-8. PMID 35489793.
- ^ Silva MT (2012). "Classical labeling of bacterial pathogens according to their lifestyle in the host: inconsistencies and alternatives". Frontiers in Microbiology. 3: 71. doi:10.3389/fmicb.2012.00071. PMC 3289908. PMID 22393329.
- ^ a b Yeshanew AG, Geremew RA (17 July 2018). "Neisseria Gonorrhoae and their antimicrobial susceptibility patterns among symptomatic patients from Gondar town, north West Ethiopia". Antimicrobial Resistance and Infection Control. 7 (1) 85. doi:10.1186/s13756-018-0376-3. PMC 6050735. PMID 30026943.
- ^ Tønjum T, Van Putten J (2017). "Neisseria". Infectious Diseases. pp. 1553–1564.e1. doi:10.1016/B978-0-7020-6285-8.00179-9. ISBN 978-0-7020-6285-8.
Endospores and exotoxins are not found and flagella are absent. Some Neisseria spp., including N. gonorrhoeae and N. meningitidis, may show surface-bound twitching motility due to pilus retraction.
- ^ Gray-Owen SD, Dehio C, Rudel T, Naumann M, Meyer TF (2001). "Neisseria". Principles of Bacterial Pathogenesis. pp. 559–618. doi:10.1016/b978-012304220-0/50013-3. ISBN 978-0-12-304220-0.
- ^ Eriksson J, Eriksson OS, Maudsdotter L, Palm O, Engman J, Sarkissian T, et al. (April 2015). "Characterization of motility and piliation in pathogenic Neisseria". BMC Microbiology. 15 (1) 92. doi:10.1186/s12866-015-0424-6. PMC 4449605. PMID 25925502.
- ^ Ladhani SN, Lucidarme J, Parikh SR, Campbell H, Borrow R, Ramsay ME (June 2020). "Meningococcal disease and sexual transmission: urogenital and anorectal infections and invasive disease due to Neisseria meningitidis". Lancet. 395 (10240): 1865–1877. doi:10.1016/S0140-6736(20)30913-2. PMID 32534649. S2CID 219701418.
- ^ Groisman EA, ed. (2001). Principles of bacterial pathogenesis. San Diego, Calif: Academic Press. ISBN 978-0-12-304220-0.
- ^ a b c d e Morse SA, Cacciapuoti AF, Lysko PG (1 January 1980). "Physiology of Neisseria gonorrhoeae". In Rose AH, Morris JG (eds.). Advances in Microbial Physiology Volume 20. Vol. 20. Academic Press. pp. 251–320. doi:10.1016/s0065-2911(08)60209-x. ISBN 978-0-12-027720-9. PMID 43667.
- ^ a b c Morse SA, Stein S, Hines J (November 1974). "Glucose metabolism in Neisseria gonorrhoeae". Journal of Bacteriology. 120 (2): 702–714. doi:10.1128/jb.120.2.702-714.1974. PMC 245830. PMID 4156358.
- ^ a b c Hebeler BH, Morse SA (October 1976). "Physiology and metabolism of pathogenic neisseria: tricarboxylic acid cycle activity in Neisseria gonorrhoeae". Journal of Bacteriology. 128 (1): 192–201. doi:10.1128/jb.128.1.192-201.1976. PMC 232843. PMID 824268.
- ^ Ingram-Smith C, Martin SR, Smith KS (June 2006). "Acetate kinase: not just a bacterial enzyme". Trends in Microbiology. 14 (6): 249–253. doi:10.1016/j.tim.2006.04.001. PMID 16678422.
- ^ Leighton MP, Kelly DJ, Williamson MP, Shaw JG (June 2001). "An NMR and enzyme study of the carbon metabolism of Neisseria meningitidis". Microbiology. 147 (Pt 6): 1473–1482. doi:10.1099/00221287-147-6-1473. PMID 11390678.
- ^ a b Atack JM, Ibranovic I, Ong CL, Djoko KY, Chen NH, Vanden Hoven R, et al. (October 2014). "A role for lactate dehydrogenases in the survival of Neisseria gonorrhoeae in human polymorphonuclear leukocytes and cervical epithelial cells". The Journal of Infectious Diseases. 210 (8): 1311–1318. doi:10.1093/infdis/jiu230. PMC 4215069. PMID 24737798.
- ^ a b c Li Y, Hopper A, Overton T, Squire DJ, Cole J, Tovell N (May 2010). "Organization of the electron transfer chain to oxygen in the obligate human pathogen Neisseria gonorrhoeae: roles for cytochromes c4 and c5, but not cytochrome c2, in oxygen reduction". Journal of Bacteriology. 192 (9): 2395–2406. doi:10.1128/JB.00002-10. PMC 2863483. PMID 20154126.
- ^ a b c d e f Aspholm M, Aas FE, Harrison OB, Quinn D, Vik A, Viburiene R, et al. (August 2010). "Structural alterations in a component of cytochrome c oxidase and molecular evolution of pathogenic Neisseria in humans". PLOS Pathogens. 6 (8) e1001055. doi:10.1371/journal.ppat.1001055. PMC 2924362. PMID 20808844.
- ^ Hopper A, Tovell N, Cole J (December 2009). "A physiologically significant role in nitrite reduction of the CcoP subunit of the cytochrome oxidase cbb 3 from Neisseria gonorrhoeae". FEMS Microbiology Letters. 301 (2): 232–240. doi:10.1111/j.1574-6968.2009.01824.x. PMID 19889029.
- ^ a b Anraku Y (June 1988). "Bacterial electron transport chains". Annual Review of Biochemistry. 57 (1): 101–132. doi:10.1146/annurev.bi.57.070188.000533. PMID 3052268.
- ^ a b Trumpower BL (June 1990). "Cytochrome bc1 complexes of microorganisms". Microbiological Reviews. 54 (2): 101–129. doi:10.1128/mr.54.2.101-129.1990. PMC 372766. PMID 2163487.
- ^ a b "UniProt". www.uniprot.org. Retrieved 18 November 2024.
- ^ a b Capaldi RA, Aggeler R (March 2002). "Mechanism of the F(1)F(0)-type ATP synthase, a biological rotary motor". Trends in Biochemical Sciences. 27 (3): 154–160. doi:10.1016/s0968-0004(01)02051-5. PMID 11893513.
- ^ a b c Stoudenmire JL, Greenawalt AN, Cornelissen CN (15 September 2022). "Stealthy microbes: How Neisseria gonorrhoeae hijacks bulwarked iron during infection". Frontiers in Cellular and Infection Microbiology. 12 1017348. doi:10.3389/fcimb.2022.1017348. PMC 9519893. PMID 36189345.
- ^ a b c d e Edwards JL, Apicella MA (October 2004). "The molecular mechanisms used by Neisseria gonorrhoeae to initiate infection differ between men and women". Clinical Microbiology Reviews. 17 (4): 965–81, table of contents. doi:10.1128/CMR.17.4.965-981.2004. PMC 523569. PMID 15489357.
- ^ "STI Awareness: Gonorrhea". Planned Parenthood Advocates of Arizona. 11 April 2011. Archived from the original on 3 November 2012. Retrieved 31 August 2011.
- ^ Sadarangani M, Pollard AJ, Gray-Owen SD (May 2011). "Opa proteins and CEACAMs: pathways of immune engagement for pathogenic Neisseria". FEMS Microbiology Reviews. 35 (3): 498–514. doi:10.1111/j.1574-6976.2010.00260.x. PMID 21204865.
- ^ a b Hu LI, Yin S, Ozer EA, Sewell L, Rehman S, Garnett JA, et al. (October 2020). Justice S (ed.). "Discovery of a New Neisseria gonorrhoeae Type IV Pilus Assembly Factor, TfpC". mBio. 11 (5). doi:10.1128/mBio.02528-20. PMC 7593972. PMID 33109763.
- ^ a b Merz AJ, So M, Sheetz MP (September 2000). "Pilus retraction powers bacterial twitching motility". Nature. 407 (6800): 98–102. Bibcode:2000Natur.407...98M. doi:10.1038/35024105. PMID 10993081.
- ^ Stern A, Brown M, Nickel P, Meyer TF (October 1986). "Opacity genes in Neisseria gonorrhoeae: control of phase and antigenic variation". Cell. 47 (1): 61–71. doi:10.1016/0092-8674(86)90366-1. PMID 3093085. S2CID 21366517.
- ^ Cahoon LA, Seifert HS (September 2011). "Focusing homologous recombination: pilin antigenic variation in the pathogenic Neisseria". Molecular Microbiology. 81 (5): 1136–1143. doi:10.1111/j.1365-2958.2011.07773.x. PMC 3181079. PMID 21812841.
- ^ Obergfell KP, Seifert HS (February 2015). "Mobile DNA in the pathogenic Neisseria". Microbiology Spectrum. 3 (3). doi:10.1128/microbiolspec.MDNA3-0015-2014. PMC 4389775. PMID 25866700.
- ^ Aas FE, Wolfgang M, Frye S, Dunham S, Løvold C, Koomey M (November 2002). "Competence for natural transformation in Neisseria gonorrhoeae: components of DNA binding and uptake linked to type IV pilus expression". Molecular Microbiology. 46 (3): 749–760. doi:10.1046/j.1365-2958.2002.03193.x. PMID 12410832. S2CID 21854666.
- ^ Simons MP, Nauseef WM, Apicella MA (April 2005). "Interactions of Neisseria gonorrhoeae with adherent polymorphonuclear leukocytes". Infection and Immunity. 73 (4): 1971–1977. doi:10.1128/iai.73.4.1971-1977.2005. PMC 1087443. PMID 15784537.
- ^ Escobar A, Rodas PI, Acuña-Castillo C (2018). "Macrophage-Neisseria gonorrhoeae Interactions: A Better Understanding of Pathogen Mechanisms of Immunomodulation". Frontiers in Immunology. 9: 3044. doi:10.3389/fimmu.2018.03044. PMC 6309159. PMID 30627130.
- ^ Stohl EA, Seifert HS (November 2006). "Neisseria gonorrhoeae DNA recombination and repair enzymes protect against oxidative damage caused by hydrogen peroxide". Journal of Bacteriology. 188 (21): 7645–7651. doi:10.1128/JB.00801-06. PMC 1636252. PMID 16936020.
- ^ Michod RE, Bernstein H, Nedelcu AM (May 2008). "Adaptive value of sex in microbial pathogens". Infection, Genetics and Evolution. 8 (3): 267–285. Bibcode:2008InfGE...8..267M. doi:10.1016/j.meegid.2008.01.002. PMID 18295550.
- ^ Blokesch M (November 2016). "Natural competence for transformation". Current Biology. 26 (21): R1126 – R1130. Bibcode:2016CBio...26R1126B. doi:10.1016/j.cub.2016.08.058. PMID 27825443.
- ^ "Neisseria gonorrhoeae genome statistics". Broad Institute. Retrieved 8 April 2017.
- ^ Chung GT, Yoo JS, Oh HB, Lee YS, Cha SH, Kim SJ, et al. (September 2008). "Complete genome sequence of Neisseria gonorrhoeae NCCP11945". Journal of Bacteriology. 190 (17): 6035–6036. doi:10.1128/JB.00566-08. PMC 2519540. PMID 18586945.
- ^ Burmeister AR (July 2015). "Horizontal Gene Transfer". Evolution, Medicine, and Public Health. 2015 (1): 193–194. doi:10.1093/emph/eov018. PMC 4536854. PMID 26224621.
- ^ Unemo M, Shafer WM (August 2011). "Antibiotic resistance in Neisseria gonorrhoeae: origin, evolution, and lessons learned for the future". Annals of the New York Academy of Sciences. 1230 (1): E19 – E28. Bibcode:2011NYASA1230E..19U. doi:10.1111/j.1749-6632.2011.06215.x. PMC 4510988. PMID 22239555.
- ^ Spencer-Smith R, Roberts S, Gurung N, Snyder LA (August 2016). "DNA uptake sequences in Neisseria gonorrhoeae as intrinsic transcriptional terminators and markers of horizontal gene transfer". Microbial Genomics. 2 (8) e000069. doi:10.1099/mgen.0.000069. PMC 5320588. PMID 28348864.
- ^ Manoharan-Basil SS, González N, Laumen JG, Kenyon C (17 March 2022). "Horizontal Gene Transfer of Fluoroquinolone Resistance-Conferring Genes From Commensal Neisseria to Neisseria gonorrhoeae: A Global Phylogenetic Analysis of 20,047 Isolates". Frontiers in Microbiology. 13 793612. doi:10.3389/fmicb.2022.793612. PMC 8973304. PMID 35369513.
- ^ Hamilton HL, Dillard JP (January 2006). "Natural transformation of Neisseria gonorrhoeae: from DNA donation to homologous recombination". Molecular Microbiology. 59 (2): 376–385. doi:10.1111/j.1365-2958.2005.04964.x. PMID 16390436.
- ^ a b c Cehovin A, Lewis SB (August 2017). "Mobile genetic elements in Neisseria gonorrhoeae: movement for change". Pathogens and Disease. 75 (6). doi:10.1093/femspd/ftx071. PMID 28645177.
- ^ Kravtsov D, Gryadunov D, Shaskolskiy B (June 2023). "Gonococcal Genetic Island in the Global Neisseria gonorrhoeae Population: A Model of Genetic Diversity and Association with Resistance to Antimicrobials". Microorganisms. 11 (6): 1547. doi:10.3390/microorganisms11061547. PMC 10301925. PMID 37375049.
- ^ Zweig M, Schork S, Koerdt A, Siewering K, Sternberg C, Thormann K, et al. (April 2014). "Secreted single-stranded DNA is involved in the initial phase of biofilm formation by Neisseria gonorrhoeae" (PDF). Environmental Microbiology. 16 (4): 1040–1052. Bibcode:2014EnvMi..16.1040Z. doi:10.1111/1462-2920.12291. PMID 24119133.
- ^ Anderson MT, Seifert HS (May 2011). "Neisseria gonorrhoeae and humans perform an evolutionary LINE dance". Mobile Genetic Elements. 1 (1): 85–87. doi:10.4161/mge.1.1.15868. PMC 3190277. PMID 22016852.
- ^ Anderson MT, Seifert HS (2011). "Opportunity and means: horizontal gene transfer from the human host to a bacterial pathogen". mBio. 2 (1): e00005 – e00011. doi:10.1128/mBio.00005-11. PMC 3042738. PMID 21325040.
- ^ Detels R, Green AM, Klausner JD, Katzenstein D, Gaydos C, Handsfield H, et al. (June 2011). "The incidence and correlates of symptomatic and asymptomatic Chlamydia trachomatis and Neisseria gonorrhoeae infections in selected populations in five countries". Sexually Transmitted Diseases. 38 (6): 503–509. doi:10.1097/OLQ.0b013e318206c288. PMC 3408314. PMID 22256336.
- ^ a b c d e f g h i j Ryan KJ, Ray CG, eds. (2004). Sherris Medical Microbiology (4th ed.). McGraw Hill. ISBN 978-0-8385-8529-0.[page needed]
- ^ a b c "Gonococcal Infections - 2015 STD Treatment Guidelines". www.cdc.gov. Retrieved 7 December 2017.
- ^ "Gonorrhea (the clap) Symptoms". std-gov.org. 2 April 2015.
- ^ Goodyear-Smith F (November 2007). "What is the evidence for non-sexual transmission of gonorrhoea in children after the neonatal period? A systematic review". Journal of Forensic and Legal Medicine. 14 (8): 489–502. doi:10.1016/j.jflm.2007.04.001. PMID 17961874.
- ^ Anderson MT, Dewenter L, Maier B, Seifert HS (March 2014). "Seminal plasma initiates a Neisseria gonorrhoeae transmission state". mBio. 5 (2): e01004 – e01013. doi:10.1128/mBio.01004-13. PMC 3958800. PMID 24595372.
- ^ a b Meyer T, Buder S (January 2020). "The Laboratory Diagnosis of Neisseria gonorrhoeae: Current Testing and Future Demands". Pathogens. 9 (2): 91. doi:10.3390/pathogens9020091. PMC 7169389. PMID 32024032.
- ^ Cosentino LA, Campbell T, Jett A, Macio I, Zamborsky T, Cranston RD, et al. (June 2012). "Use of nucleic acid amplification testing for diagnosis of anorectal sexually transmitted infections". Journal of Clinical Microbiology. 50 (6): 2005–2008. doi:10.1128/JCM.00185-12. PMC 3372150. PMID 22493338.
- ^ a b c d "Gonococcal Infections Among Adolescents and Adults - STI Treatment Guidelines". www.cdc.gov. 5 December 2022. Retrieved 19 November 2024.
- ^ a b c Williams E, Seib KL, Fairley CK, Pollock GL, Hocking JS, McCarthy JS, et al. (March 2024). Forrest GN (ed.). "Neisseria gonorrhoeae vaccines: a contemporary overview". Clinical Microbiology Reviews. 37 (1): e0009423. doi:10.1128/cmr.00094-23. PMC 10938898. PMID 38226640.
- ^ Clinical trial number NCT05630859 for "Safety and Efficacy of GSK Neisseria Gonorrhoeae GMMA (NgG) Investigational Vaccine When Administered to Healthy Adults 18 to 50 Years of Age." at ClinicalTrials.gov
- ^ "GSK receives US FDA Fast Track designation for investigational vaccine against gonorrhoea" (Press release). GSK. 27 June 2023. Archived from the original on 24 July 2025. Retrieved 24 July 2025.
- ^ Rogers A, Gallagher J (21 May 2025). "World-first gonorrhoea vaccine launched by NHS England as infections soar". BBC News. Archived from the original on 24 July 2025. Retrieved 24 July 2025.
- ^ "Gonorrhoea vaccine". Vaccine Knowledge Project. Oxford Vaccine Group, University of Oxford. 20 June 2025. Archived from the original on 24 July 2025. Retrieved 24 July 2025.
- ^ CDC (16 May 2024). "Drug-Resistant Gonorrhea". Gonorrhea. Retrieved 20 November 2024.
- ^ St Cyr S, Barbee L, Workowski KA, Bachmann LH, Pham C, Schlanger K, et al. (December 2020). "Update to CDC's Treatment Guidelines for Gonococcal Infection, 2020". MMWR. Morbidity and Mortality Weekly Report. 69 (50): 1911–1916. doi:10.15585/mmwr.mm6950a6. PMC 7745960. PMID 33332296.
- ^ "UK doctors advised gonorrhoea has turned drug resistant". BBC News. 10 October 2011.
- ^ a b "STI Awareness: Antibiotic-Resistant Gonorrhea". Planned Parenthood Advocates of Arizona. 6 March 2012. Archived from the original on 5 November 2012. Retrieved 6 March 2012.
- ^ Tuddenham S, Hamill MM, Ghanem KG (January 2022). "Diagnosis and Treatment of Sexually Transmitted Infections: A Review". JAMA. 327 (2): 161–172. doi:10.1001/jama.2021.23487. PMID 35015033.
- ^ a b c d Charles A Janeway J, Travers P, Walport M, Shlomchik MJ (2001). "The complement system and innate immunity". Immunobiology: The Immune System in Health and Disease. 5th edition. Garland Science.
- ^ "gonorrhea | Origin and meaning of gonorrhea by Online Etymology Dictionary". www.etymonline.com. Retrieved 5 December 2017.
- ^ a b Singal A, Grover C (2016). Comprehensive Approach to Infections in Dermatology. JP Medical. p. 470. ISBN 978-93-5152-748-0.
- ^ Gladstein J (July 2005). "Hunter's chancre: did the surgeon give himself syphilis?". Clinical Infectious Diseases. 41 (1): 128, author reply 128-128, author reply 129. doi:10.1086/430834. PMID 15937780.
- ^ Jose PP, Vivekanandan V, Sobhanakumari K (15 October 2020). "Gonorrhea: Historical outlook". Journal of Skin and Sexually Transmitted Diseases. 2 110: 110–114. doi:10.25259/JSSTD_4_2020.
External links
[edit]- Todar K. "Pathogenic Neisseriae: Gonorrhea, Neonatal Ophthalmia and Meningococcal Meningitis". Todar's Online Textbook of Bacteriology. Archived from the original on 16 October 2022. Retrieved 12 October 2008.
- Gonorrhea at eMedicine
- "Neisseria gonorrhoeae". NCBI Taxonomy Browser. 485.
Neisseria gonorrhoeae
View on GrokipediaNeisseria gonorrhoeae is a Gram-negative diplococcus bacterium that functions as an obligate human pathogen, causing gonorrhea, a widespread sexually transmitted infection primarily acquired through vaginal, anal, or oral sexual contact.[1][2][3] The organism infects mucosal surfaces of the urogenital tract, rectum, pharynx, and conjunctiva, manifesting commonly as urethritis in males and cervicitis in females, though infections are frequently asymptomatic, especially in women, facilitating undetected transmission.[4][5] Untreated cases can lead to severe complications including pelvic inflammatory disease, ectopic pregnancy, infertility, and disseminated gonococcal infection with potential for arthritis or meningitis.[1][3] N. gonorrhoeae demonstrates high genetic variability and phase variation, enabling immune evasion and adaptation, while its increasing multidrug resistance—spanning penicillins, fluoroquinolones, and cephalosporins—threatens effective therapy and underscores the need for vigilant surveillance and novel interventions.[6][7][8]
Taxonomy and Phylogeny
Classification and Etymology
Neisseria gonorrhoeae is a Gram-negative bacterium classified within the domain Bacteria, phylum Pseudomonadota, class Betaproteobacteria, order Neisseriales, family Neisseriaceae, and genus Neisseria.00084-8) This placement reflects its phylogenetic position among proteobacteria, a diverse group encompassing both free-living and host-associated bacteria. The genus Neisseria comprises approximately 20 species, including the pathogenic N. meningitidis, which causes meningococcal disease, and commensal species such as N. lactamica and N. flavescens that typically inhabit the human nasopharynx without causing illness.00084-8)[9] Unlike many Neisseria species that serve as commensals or opportunistic pathogens with broader host ranges or asymptomatic colonization potential, N. gonorrhoeae is an obligate human pathogen, restricted to human mucosal surfaces with no known animal reservoirs or environmental persistence.[9][10] This host specificity underscores its evolutionary adaptation to human urogenital and pharyngeal epithelia, distinguishing it from relatives capable of transient or persistent carriage in healthy individuals.[1] The genus name Neisseria derives from Albert Ludwig Sigesmund Neisser, the German physician who isolated and described the gonococcus in purulent discharges from gonorrhea patients in 1879, establishing its etiology through microscopic observation and inoculation experiments.[11][12] The specific epithet gonorrhoeae references gonorrhea, coined by the Roman physician Galen around 130–200 CE from the Greek gonē (γόνη; "seed" or "semen") and rhein (ῥεῖν; "to flow"), based on the erroneous ancient belief that the urethral discharge represented seminal fluid rather than infection.[12] Neisser's work formalized the binomial Neisseria gonorrhoeae, shifting understanding from humoral imbalances to microbial causation.[11]Evolutionary Relationships
Neisseria gonorrhoeae shares a close phylogenetic relationship with Neisseria meningitidis, the two species exhibiting approximately 80-90% nucleotide sequence identity across their core genomes, indicative of recent divergence from a common ancestor.[13] This genetic proximity is underscored by shared housekeeping genes and chromosomal synteny, despite adaptations to distinct host niches: N. gonorrhoeae specializes in urogenital and anorectal mucosal infections, while N. meningitidis primarily colonizes the nasopharynx.[14] Phylogenetic reconstructions based on whole-genome sequences position both pathogens within the human-restricted clade of the Neisseria genus, distinct from environmental relatives.[15] The evolutionary trajectory of N. gonorrhoeae traces back to commensal Neisseria species inhabiting the oropharynx, with genomic evidence suggesting divergence through selection for obligate mucosal parasitism.[16] Multi-locus sequence typing (MLST) analyses of housekeeping genes reveal that pathogenic Neisseria form discrete clades amid a diverse commensal background, with N. gonorrhoeae displaying clonal expansions linked to global dissemination and reduced recombination rates compared to N. meningitidis.[17] This clonality likely arose from transmission bottlenecks in sexual networks, contrasting the panmictic diversity of oropharyngeal commensals.[18] Horizontal gene transfer, particularly homologous recombination with commensal Neisseria, has driven antigenic variation in N. gonorrhoeae, enabling immune evasion and adaptation.[19] Post-2010 genomic surveys document frequent allele exchange at surface loci, with commensals serving as reservoirs for novel sequences that enhance pathogen fitness without conferring full pathogenicity.[20] Such interspecies recombination underscores the dynamic evolution within the genus, where gene flow from non-pathogenic relatives sustains diversity in the specialized N. gonorrhoeae lineage.[21]Biological and Microbiological Features
Morphology and Growth Characteristics
is a Gram-negative diplococcus, typically appearing as pairs of kidney bean-shaped cocci with flattened adjacent sides, measuring 0.6 to 1.0 μm in diameter.[10][22] The bacterium is non-motile and lacks a polysaccharide capsule, distinguishing it from related species like N. meningitidis.[23] Its outer membrane contains lipooligosaccharides (LOS), which contribute to structural integrity and host interactions, rather than the full lipopolysaccharide found in many other Gram-negative bacteria.[24] N. gonorrhoeae exhibits fastidious growth requirements as an obligate aerobe, thriving at temperatures of 35–37°C in an atmosphere enriched with 5% CO₂.[25] Optimal cultivation occurs on enriched media such as chocolate agar or selective Thayer-Martin agar, where it forms small (0.5–2 mm), round, smooth, translucent to grayish-opaque colonies after 24–48 hours of incubation.[26][27] The organism is oxidase-positive and catalase-positive, traits consistent with its classification in the genus Neisseria.[28]Culture and Identification Methods
Neisseria gonorrhoeae requires fastidious culture conditions for isolation, typically using selective media such as modified Thayer-Martin (MTM) or modified New York City (NYC) agar, which incorporate antibiotics like vancomycin, colistin, trimethoprim, and nystatin to inhibit competing flora.[29] Specimens from urogenital, rectal, pharyngeal, or conjunctival sites must be transported in non-nutritive media like Amies or Stuart's to maintain viability, with inoculation onto media followed by incubation at 35–37°C in 3–7% CO₂ for 24–48 hours.[30] Colonies appear as small, grayish-white, translucent, and non-hemolytic, often requiring subculture to chocolate agar for further growth if initial yields are low.[29] Presumptive identification relies on colonial morphology and rapid biochemical tests: Gram staining reveals Gram-negative diplococci, frequently intracellular within polymorphonuclear leukocytes from symptomatic genital sites, while the oxidase test yields a positive dark-purple reaction within 10 seconds.[29] The catalase test, using 3% hydrogen peroxide, produces immediate bubbling, but the superoxol variant with 30% hydrogen peroxide elicits a more vigorous, explosive effervescence characteristic of N. gonorrhoeae, aiding differentiation from commensal Neisseria species.[31] [29] Confirmatory identification employs carbohydrate utilization tests, where isolates acidify glucose (producing acid and no gas) but fail to utilize maltose, sucrose, lactose, or fructose, distinguishing N. gonorrhoeae from N. meningitidis (which ferments both glucose and maltose).[29] These tests, often performed via cystine trypticase agar (CTA) or rapid disk methods, achieve high specificity when combined with prior presumptive results.[30] Although nucleic acid amplification tests (NAATs) have largely supplanted culture for routine detection due to higher sensitivity, especially at extragenital sites, culture remains indispensable for antimicrobial susceptibility testing amid rising resistance, such as to cephalosporins.[30] Limitations include reduced recovery (85–95% sensitivity under optimal conditions) from pharyngeal or rectal specimens, where viability declines rapidly post-collection, necessitating immediate processing and enriched atmospheres.[30] Selective media may inhibit certain strains, prompting parallel use of non-selective chocolate agar in challenging cases.[29]Physiology and Metabolism
Nutrient Acquisition and Utilization
Neisseria gonorrhoeae primarily utilizes glucose as its preferred carbon and energy source, which is catabolized through the Entner-Doudoroff pathway to yield pyruvate and glyceraldehyde-3-phosphate, bypassing the conventional Embden-Meyerhof-Parnas glycolysis route.[32] This pathway is obligatory for the metabolism of gluconate and supports efficient energy generation in the nutrient-limited mucosal environments colonized by the bacterium.[33] While capable of utilizing alternative substrates such as lactate under certain conditions, glucose remains central to its carbon metabolism, reflecting adaptations to host-derived carbohydrates.[34] The bacterium exhibits limited capacity for de novo amino acid biosynthesis, functioning as an auxotroph for several essential amino acids and relying instead on host-derived peptides and free amino acids scavenged from mucosal secretions and inflamed tissues.[5] This dependence underscores its obligate parasitism, with peptide uptake facilitated by specific ABC transporters and peptidases that hydrolyze host proteins into utilizable forms during infection.[34] As a strict aerobe incapable of anaerobic growth, N. gonorrhoeae depends on aerobic respiration for ATP production, employing a branched electron transport chain featuring multiple c-type cytochromes, b-type cytochromes, and non-heme iron centers that terminate at cytochrome o or alternative oxidases.[35] [36] Oxygen serves as the terminal electron acceptor, with the chain's organization enabling efficient proton motive force generation despite variable host oxygen levels.[35] Iron acquisition is critical for survival in iron-restricted host niches, achieved without siderophore production but via TonB-dependent outer membrane transporters that bind and strip ferric iron from host glycoproteins.[37] Specific systems include the transferrin receptor complex (TbpA and TbpB) for serum transferrin and the lactoferrin receptor (LbpA and LbpB) for mucosal lactoferrin, which facilitate iron uptake into the periplasm followed by cytoplasmic transport via periplasmic binding proteins like FbpA.[38] [39] These mechanisms ensure iron homeostasis essential for enzymatic functions in respiration and other metabolic processes.[37]Energy Metabolism and Iron Handling
Neisseria gonorrhoeae relies on aerobic respiration for energy production, employing oxidative phosphorylation where electrons from central carbon metabolism fuel a respiratory chain that generates a proton motive force (PMF) across the cytoplasmic membrane to drive ATP synthesis via ATP synthase.[40] The electron transport chain includes cytochromes b, c, and a terminal cytochrome o oxidase, enabling adaptation to the microaerobic conditions of human mucosal niches.[41] Exposure to host-derived reactive oxygen species (ROS) during infection imposes oxidative stress, to which N. gonorrhoeae responds primarily through manganese (Mn) homeostasis rather than robust superoxide dismutase (SOD) activity. The bacterium expresses a cytoplasmic SodB enzyme with minimal catalytic efficiency, insufficient for significant ROS detoxification; instead, elevated intracellular Mn²⁺ levels act as a non-enzymatic scavenger of superoxide anions, coordinated by regulators like PerR to enhance survival under oxidative conditions.[42][43] Iron is an essential cofactor for respiratory enzymes and other metabolic processes, yet scarce in the host due to sequestration by proteins like transferrin; N. gonorrhoeae overcomes this via a TonB-dependent transport system featuring the outer membrane receptor TbpA and accessory lipoprotein TbpB, which cooperatively bind human apo- or holo-transferrin to facilitate iron extraction and periplasmic release.[44][45] Expression of TbpA/B is tightly regulated by the Fur repressor, which binds iron to silence transcription under replete conditions, ensuring resource allocation only during limitation.[46] Metabolic flexibility supports persistence in nutrient-variable host sites, such as glucose-depleted mucosa, where N. gonorrhoeae shifts from the Entner-Doudoroff pathway for glucose catabolism to utilization of alternative substrates like lactate, maintaining energy yield through adjusted transcriptional profiles and central carbon flux.[40][47] This adaptability links bioenergetics to iron-dependent survival, as iron-handling systems integrate with PMF-driven transport to sustain respiration amid oxidative challenges.[48]Virulence Mechanisms
Adhesion and Invasion Factors
initiates infection through type IV pili, which extend from the bacterial surface and mediate initial attachment to host mucosal epithelial cells. These retractable filaments, composed primarily of PilE pilin subunits, enable twitching motility—a form of surface translocation driven by pilus extension, attachment, and retraction cycles—that positions the bacterium for stable adherence. [49] [50] The pilus tip-associated adhesin PilC1 binds specifically to the complement regulatory protein CD46 on human epithelial cells, promoting close bacterial-host contact essential for colonization. [51] [52] Following pilus-mediated docking, opacity-associated (Opa) proteins embedded in the outer membrane drive intimate adhesion and facilitate bacterial invasion into non-phagocytic cells. At least 11 phase-variable opa genes encode Opa variants, with expression toggled by slipped-strand mispairing in pentameric repeat tracts, allowing stochastic on-off switching during infection. [53] Specific Opa variants recognize human carcinoembryonic antigen-related cell adhesion molecule (CEACAM) receptors, such as CEACAM1, CEACAM3, and CEACAM6, on epithelial and immune cells; this interaction induces host actin rearrangements and receptor clustering, culminating in bacterial endocytosis. [54] [55] PorB, the major outer membrane porin of N. gonorrhoeae, supports invasion indirectly by integrating into host mitochondrial membranes post-uptake, where it modulates cellular signaling and delays apoptosis to favor intracellular survival. [56] As a β-barrel trimer forming a channel for passive diffusion of small hydrophilic nutrients (up to ~600 Da), PorB also sustains bacterial viability during host cell transit, though its primary role in adhesion involves voltage-dependent conductance alterations that influence epithelial barrier integrity. [57] [58]Immune Evasion and Variation Strategies
Neisseria gonorrhoeae employs phase variation and antigenic variation to alter its surface structures, primarily type IV pili and opacity-associated (Opa) proteins, enabling evasion of host adaptive immunity. Phase variation in pilin genes occurs through slipped-strand mispairing during replication of guanine tetrads in the pilE locus, leading to reversible on-off switching of pilus expression and antigenic diversity via recombination with silent pilS loci. [59] Similarly, Opa proteins undergo phase variation via pentameric repeat tracts in their leader sequences, resulting in heterogeneous expression across bacterial populations that modulates adhesion, invasion, and recognition by host antibodies. [60] These mechanisms generate microheterogeneity within infections, allowing subpopulations to persist despite targeted immune responses. [61] Lipooligosaccharide (LOS) sialylation represents a key molecular mimicry strategy, where the bacterium incorporates host cytidine monophosphate-N-acetylneuraminic acid (CMP-Neu5Ac) to cap terminal lactosamine residues, mimicking human sialylated glycans and thereby inhibiting complement activation and phagocytosis. [62] Sialylated LOS reduces deposition of C3b and subsequent opsonization, enhancing serum resistance, while also suppressing neutrophil activation independently of complement pathways. [63] This modification is phase-variable and dependent on bacterial sialyltransferase activity, contributing to tissue tropism and disseminated infection potential. [64] The pathogen secretes IgA1 protease, an extracellular enzyme that specifically cleaves the hinge region of human IgA1 antibodies at the Pro227-Thr228 peptide bond, neutralizing mucosal secretory IgA and impairing antibody-mediated aggregation and clearance. [65] Mutants lacking this protease exhibit reduced survival on mucosal surfaces, underscoring its role in initial colonization. [66] Gene structure analysis reveals autoproteolytic processing for secretion, ensuring active enzyme release without host cell internalization. [67] Complement evasion is further augmented by recruitment of host factor H (fH), a negative regulator that accelerates C3b decay and inhibits the membrane attack complex via binding to bacterial surface ligands such as PorB porin and sialylated LOS. [68] This interaction is species-specific to humans, with fH short consensus repeats binding non-sialylated Por1A loop 5 and sialic acid enhancing affinity, thereby conferring resistance to non-immune serum killing. [69] [70] Such recruitment exploits host regulatory proteins, promoting bacterial persistence in complement-rich environments. [64]Survival in Host Environments
Neisseria gonorrhoeae exhibits adaptations enabling intracellular persistence within neutrophils, where it delays phagosome-lysosome fusion and modulates the oxidative burst to avoid killing.[71] In vitro assays with adherent human polymorphonuclear leukocytes (PMNs) demonstrate that over 50% of internalized gonococci remain viable for up to 6 hours post-phagocytosis.[72] This survival facilitates non-lytic egress, allowing bacteria to exit host cells without triggering full lysis and propagate infection.[73] To counter nutritional immunity, N. gonorrhoeae employs the TdfH transporter to pirate zinc from calprotectin (S100A8/A9), a highly expressed neutrophil protein that sequesters metals to starve pathogens.[74] Structural analyses reveal TdfH binds metal-loaded tetrameric calprotectin, enabling zinc acquisition and resistance to this antimicrobial effector, which is abundant in genital secretions.[75] Mutants lacking TdfH show reduced intracellular viability in PMNs, underscoring this mechanism's role in host adaptation.[76] Extracellular persistence involves tolerance to cationic antimicrobial peptides (CAMPs) via the MtrCDE efflux pump, which exports peptides like LL-37 and enhances survival in peptide-rich environments such as the male urethra.[77] [78] Membrane modifications, including lipopolysaccharide sialylation and phosphoethanolamine addition to lipid A, further reduce CAMP binding and permeabilization.[79] These efflux and remodeling systems synergize to confer resilience against innate defenses. Biofilm formation supports persistence in vitro, with gonococci aggregating via extracellular DNA, pili, and matrix components on abiotic surfaces and epithelial cells, increasing antibiotic tolerance.[80] However, its occurrence and significance in vivo remain debated, with limited direct evidence from human infections despite transcriptional profiles suggesting biofilm-like states during cervical colonization.[81] Asymptomatic carriage exemplifies extracellular reservoir formation, disproportionately affecting females where 30–50% of infections lack symptoms compared to 5–10% in males, per clinical data from screened populations.[82] This silent persistence, observed in 1–5% of sexually active female adolescents, sustains transmission without eliciting clearance.[83] Empirical screening studies confirm higher undetected prevalence in women, enabling chronic low-level shedding.[84]Genomics
Genome Structure and Size
The genome of Neisseria gonorrhoeae consists of a single circular chromosome approximately 2.2 million base pairs (Mbp) in length, encoding roughly 2,100 protein-coding genes.[85] [86] The overall GC content is about 52.5%, which is relatively high for a pathogen adapted to the human host.[87] [88] Unlike many bacteria, N. gonorrhoeae lacks a functional CRISPR-Cas system, relying instead on other mechanisms for defense against foreign DNA.[89] The core genome, comprising genes essential for basic cellular functions and present in nearly all strains, is highly conserved and estimated at over 1,600 genes, reflecting the bacterium's obligate parasitic lifestyle.[90] In contrast, the accessory genome varies significantly between isolates and includes mobile elements such as multiple insertion sequences (e.g., families like IS1655 and Correia repeats) that promote genomic rearrangements and plasticity.[91] [92] Pathogenicity islands, such as the Gonococcal Genetic Island (GGI), contribute to this variability by harboring strain-specific genes, though their presence is not universal across populations.[93] The first complete genome sequence was determined for strain FA 1090, isolated in 1983 from a case of disseminated infection, with assembly data released around 2000, revealing the absence of CRISPR arrays and highlighting the role of repetitive elements in phase variation.[86] [94] This reference genome, spanning 2,153,922 bp, has served as a benchmark for subsequent comparative analyses, underscoring the chromosome's compact organization with a coding density near 87%.[95]Genetic Diversity and Horizontal Transfer
Neisseria gonorrhoeae displays substantial genetic diversity, driven by high rates of homologous recombination and horizontal gene transfer (HGT), which facilitate rapid adaptation including the evolution of antimicrobial resistance. Whole-genome analyses reveal frequent recombination events, with studies estimating recombination-to-mutation ratios exceeding 10 in core genomes, indicating recombination dominates over mutation in shaping population structure.[96] [97] This diversity is amplified by interspecies exchanges with commensal Neisseria species, such as N. lactamica, contributing mosaic alleles to loci like porB.[98] Multi-locus sequence typing (MLST) schemes, including traditional seven-gene MLST and expanded NG-STAR, delineate clonal complexes amid this diversity, with certain sequence types (STs) undergoing pandemic expansion. For instance, ST-1901 and related lineages, characterized by mosaic penA alleles, have disseminated globally, associating with cephalosporin resistance.[99] [100] Genomic surveillance from 2016–2023 in regions like Europe and Norway documents clonal expansions of multidrug-resistant strains, alongside occasional lineage eradications linked to treatment shifts.[101] [102] HGT occurs primarily via natural transformation, for which N. gonorrhoeae is constitutively competent due to expression of type IV pilus and DNA uptake machinery, enabling uptake of exogenous DNA at frequencies up to 10^{-3} per donor genome under optimal conditions.[103] This mechanism drives acquisition of resistance determinants, notably mosaic penA alleles encoding altered penicillin-binding protein 2 (PBP2), imported from commensals via recombination tracts of 200–500 bp.[104] [105] Conjugative plasmids, such as the 24.5-MDa type carrying tetM for tetracycline resistance, further mediate HGT of accessory resistance genes, with host-range limited to Neisseria but enabling intra- and inter-strain transfer.[106] [107] These processes underpin "superbug" evolution, as evidenced by genomic data showing persistent circulation of resistance-conferring elements in diverse lineages during the 2020s. Surveillance highlights how recombination integrates chromosomal mutations (e.g., in gyrA or parC) with plasmid-borne traits, fostering multidrug resistance without complete clonality.[108] [109]Pathogenesis and Clinical Manifestations
Transmission Modes
Neisseria gonorrhoeae is transmitted predominantly through sexual contact, with direct mucosal exposure to infected genital secretions during vaginal, anal, or oral intercourse serving as the primary vector. Ejaculation is not required for transmission, which occurs via contact between the penis, vagina, mouth, or anus.[2] [10] Transmission efficiency varies by sex and site; per-act probabilities during vaginal sex range from 50% to 70% from infected males to females and 20% to 50% from females to males.[5] Pharyngeal gonorrhea may also spread via oral sex or, less commonly, deep kissing with significant saliva exchange, though empirical evidence for kissing as a major route remains limited.[110] [111] A substantial proportion of infections—up to 80% in women and 10% in men—are asymptomatic, facilitating undetected shedding and onward transmission within sexual partner networks.[112] [1] This silent carriage, particularly prolonged in females due to milder or absent symptoms, contributes to sustained endemicity, with epidemiological models attributing persistence to high-activity "core groups" exhibiting elevated partner turnover rates and prevalence levels approaching 20% in certain populations.[113] Perinatal transmission represents a non-sexual route, occurring vertically from infected mothers to neonates during passage through the birth canal, with risks manifesting as acute infections 2–5 days postpartum if untreated.[114] [83] Non-sexual adult transmission via fomites, contaminated objects, or indirect contact is exceedingly rare, given the bacterium's fragility outside the host—it dries quickly and survives poorly in environmental conditions—though isolated pediatric cases linked to shared towels or bathing sites have been documented.[10] [115] [116]Symptoms and Sites of Infection
The incubation period for Neisseria gonorrhoeae infection is typically 2–7 days after exposure, though it may range from 1–14 days in some cases.[117] In males, the primary site of infection is the urethra, manifesting as acute gonococcal urethritis with symptoms including purulent or mucopurulent discharge and dysuria; most infected men develop noticeable symptoms within this timeframe.[1] [118] In females, cervicitis represents the most common urogenital manifestation, often asymptomatic in 50–80% of cases, which contributes to undetected transmission; when symptomatic, it presents with increased vaginal discharge, dysuria, vaginal bleeding between periods, or dyspareunia.[119] [117] Pharyngeal and rectal infections occur via oral or anal exposure, respectively, and are frequently asymptomatic across sexes, though pharyngitis may cause sore throat and rectal involvement can lead to discharge, pain, or tenesmus in symptomatic individuals.[10] [120] Disseminated gonococcal infection arises rarely, in approximately 0.5–3% of untreated cases, primarily affecting the joints, skin, or tendons with symptoms such as asymmetric polyarthralgia, tenosynovitis, or petechial/pustular dermatitis.[112] In neonates, infection acquired during passage through an infected birth canal typically localizes to the conjunctivae, resulting in purulent ophthalmia neonatorum within the first week of life.[4]Complications and Long-Term Effects
In women, untreated Neisseria gonorrhoeae infection ascends from the lower genital tract to cause pelvic inflammatory disease (PID) in approximately 10-20% of cases, resulting in tubal scarring that impairs ovum transport and leads to infertility in about 15% of affected individuals.[121] This scarring also elevates the risk of ectopic pregnancy by obstructing fallopian tubes and promoting abnormal implantation sites.[2] Chronic pelvic pain arises from persistent inflammation and adhesions in the reproductive organs, with empirical cohort studies linking untreated gonorrhea to long-term sequelae in up to 20% of PID patients.[1] In men, ascending infection causes epididymitis, characterized by inflammation of the epididymis that can progress to abscess formation or testicular damage, contributing to infertility through ductal obstruction or impaired spermatogenesis in severe cases.[122] Untreated epididymo-orchitis may result in chronic discomfort and reduced fertility rates observed in longitudinal studies of infected males.[119] Disseminated gonococcal infection (DGI), occurring in 0.5-3% of untreated cases via hematogenous spread, manifests as septic arthritis, tenosynovitis, or dermatitis-arthritis syndrome, with rare mortality under modern care but potential for severe joint destruction or endocarditis if dissemination persists. Neonatal DGI from perinatal transmission presents as sepsis, meningitis, or arthritis, with bacteremia risking multi-organ failure in vulnerable infants.[123] Mucosal disruption and inflammatory recruitment of CD4+ T cells and dendritic cells during N. gonorrhoeae infection increase HIV acquisition risk by 2- to 5-fold, as evidenced by cohort analyses showing heightened viral entry at inflamed genital sites.[124] Epidemiological data indicate that co-infection facilitates HIV transmission through epithelial barrier compromise and elevated target cell density.[125] Perinatal exposure heightens risks of preterm birth, low birth weight, and neonatal sepsis, with N. gonorrhoeae ascending to amniotic membranes or causing direct bloodstream invasion in newborns.[126] Some observational studies suggest chronic or recurrent carriage may synergize with oncogenic co-infections like HPV to promote cervical carcinogenesis via persistent inflammation, though causal evidence remains associative.[127]Epidemiology
Global Incidence and Prevalence
The World Health Organization (WHO) estimated 82.4 million incident cases of Neisseria gonorrhoeae infection among adults aged 15–49 years globally in 2020, representing approximately 1.0% of this population cohort annually.[119] This figure contributes to the broader estimate of 374 million new infections from four curable sexually transmitted infections (chlamydia, gonorrhoea, syphilis, and trichomoniasis) in the same demographic for that year.[128] Incidence rates vary markedly by region, with the highest burdens reported in the WHO African Region (encompassing sub-Saharan Africa) and the Western Pacific Region, where limited healthcare access and diagnostic capacity exacerbate under-detection.[119] Prevalence metrics from sentinel surveillance systems indicate point prevalences of 0.5–1.5% in general adult populations in high-burden areas, rising to 5–10% among high-risk groups such as female sex workers and men who have sex with men in urban settings across Africa and Asia.[129] However, these data suffer from substantial underreporting, primarily due to the high proportion of asymptomatic infections—estimated at 50–80% in women and 10–50% in men—which evade routine screening outside symptomatic presentations or targeted programs.[130] Socioeconomic disparities further confound estimates, with lower-income regions showing 2–5 times higher adjusted incidence rates linked to factors like population density and healthcare inequities, though causal attribution requires disentangling from behavioral confounders.[131] Post-COVID-19 disruptions in testing and prevention services led to temporary dips in reported cases during 2020–2021, followed by rebounds in 2022–2024 as mobility and sexual activity normalized; for instance, European surveillance documented a 30–50% increase in notifications from 2021 to 2023, signaling a potential global uptick beyond the 2020 baseline.[132] Updated modeling suggests annual incident cases may now exceed 90 million, though comprehensive 2023–2025 global estimates remain pending enhanced surveillance integration.[133]Risk Factors and Transmission Dynamics
Risk factors for Neisseria gonorrhoeae infection primarily involve behavioral patterns that elevate exposure probability, such as multiple concurrent sexual partners and inconsistent condom use during vaginal, anal, or oral intercourse.[134] [135] Studies quantify this through adjusted odds ratios; for instance, individuals reporting more than one partner in the preceding months face significantly higher infection odds compared to monogamous counterparts, independent of other variables.[1] Prior sexually transmitted infection history further amplifies susceptibility via potential mucosal disruption or behavioral persistence.[1] Adolescent and young adult age groups, particularly those aged 15-24, exhibit heightened biological and behavioral vulnerability, with physiological factors like cervical ectopy in females increasing mucosal exposure alongside patterns of partner concurrency and experimentation.[1] [134] Men who have sex with men (MSM) represent a disproportionate risk category due to network density and anatomic preferences favoring pharyngeal and rectal sites, where asymptomatic carriage facilitates onward spread.[135] [136] Transmission dynamics follow core-periphery models, wherein small, high-activity "core" groups—characterized by elevated partner turnover rates—sustain endemic circulation by repeatedly bridging to lower-risk "periphery" populations with infrequent exposures.[137] In a North Carolina analysis, core subgroups (e.g., adolescent females, adult MSM, heterosexual adults with high connectivity) accounted for 68% of cases, underscoring how localized networks drive persistence despite overall population-level reproductive numbers near unity.[137] Among MSM, pharyngeal strains amplify via dense oral-genital-anal circuits, with core hubs exhibiting rapid turnover.[137] Empirical data refute assumptions of negligible oral transmission risk; pharyngeal gonorrhea, often asymptomatic, seeds genital infections through kissing, fellatio, or saliva-mediated contact, with detection in saliva confirming viable bacteria beyond throat tissues.[138] [139] In MSM cohorts, tongue kissing independently elevates oropharyngeal acquisition odds, while rimming contributes to rectal seeding, highlighting underreporting from symptom absence and selective screening.[140] [141] These dynamics persist causally from bacterial adherence via pili and immune evasion, not mitigated by anatomic barriers presumed protective.[138]Recent Trends and Outbreaks
In high-income countries, gonorrhea incidence has continued to rise despite expanded screening and public health interventions. In the European Union/European Economic Area (EU/EEA), 96,969 confirmed cases were reported in 2023, yielding a notification rate of 23.7 per 100,000 population—a 31% increase from 2022 and over 300% since 2014—driven largely by surges among young adults aged 20-24.[132] Similar post-pandemic rebounds occurred after the lifting of COVID-19 restrictions, with gonorrhea diagnoses in England increasing markedly from quarter 3 of 2021 onward, attributed to resumed high-risk sexual behaviors and reduced access to care during lockdowns.[142] In the United States, while provisional 2024 data indicated a 9% overall decline in reported chlamydia, gonorrhea, and syphilis cases compared to 2023, earlier years (2021-2023) saw sustained elevations linked to behavioral shifts post-restrictions.[143] Antibiotic resistance complicates these trends, with extensively drug-resistant (XDR) strains emerging in North America and Europe. In Canada, a ceftriaxone- and cefixime-resistant, high-level azithromycin-resistant N. gonorrhoeae isolate was identified in 2024, marking the first domestically reported XDR case amid 35,956 total infections that year, the second most common bacterial STI.[144] [145] European surveillance similarly documented rising resistance, including to cephalosporins and macrolides, with ciprofloxacin resistance reaching 65.8% in some isolates by 2022.[146] In the US, tetM-mediated tetracycline resistance expanded significantly from 2018 to 2024, with tetM-carrying strains—often multidrug-resistant—proliferating, potentially exacerbated by doxycycline post-exposure prophylaxis (doxyPEP) use in high-risk groups, though causal links require further genomic confirmation.[147] Surveillance gaps in low-resource settings amplify global spread risks, as many developing countries lack robust systems like the WHO Gonococcal Antimicrobial Surveillance Programme (GASP), hindering timely detection of resistant strains.[148] [149] This underreporting, combined with limited access to molecular diagnostics, allows undetected transmission chains to fuel international dissemination, as evidenced by the global proliferation of multidrug-resistant lineages documented in WHO-enhanced surveillance across 87 countries by 2023.[150]Diagnosis
Laboratory Diagnostic Techniques
Gram staining of clinical specimens serves as a rapid presumptive diagnostic method for Neisseria gonorrhoeae, particularly in symptomatic males with urethritis. In urethral exudates, the detection of gram-negative diplococci located intracellularly within polymorphonuclear leukocytes demonstrates sensitivity exceeding 95% and specificity greater than 99%, allowing for immediate empirical treatment initiation in resource-limited settings.[112][151] However, this technique exhibits reduced sensitivity in females (approximately 50%) and for extragenital sites such as pharyngeal or rectal specimens, where false negatives are common due to lower bacterial loads or paucicellular exudates.[119][152] Proper specimen collection, such as first-void urine sediment or direct swabs, is critical to optimize visualization, though Gram staining alone lacks sufficient negative predictive value for ruling out infection in low-prevalence or asymptomatic cases.[153] Culture-based isolation remains the reference standard for confirming viable N. gonorrhoeae and enabling antimicrobial susceptibility testing, essential amid rising resistance patterns. The bacterium, being fastidious and capnophilic, requires inoculation onto enriched selective media like modified Thayer-Martin agar, which incorporates vancomycin, colistin, nystatin, and trimethoprim to inhibit competing flora while supporting gonococcal growth in a 3-5% CO2 atmosphere at 35-37°C.[153][154] Specimens from diverse sites—urethral, cervical, rectal, pharyngeal, or conjunctival—must be collected using appropriate swabs and transported promptly in holding media like CO2 jars or Amies with charcoal to preserve viability, as delays beyond 24 hours or prior antibiotic exposure can diminish recovery rates to below 80%.[155][156] Isolated colonies appear as small, grayish-white, translucent mucoid growths after 24-48 hours, confirmed via oxidase positivity, carbohydrate utilization (glucose fermentation without maltose or sucrose), and morphological consistency under microscopy.[4] ![Colonies of Neisseria gonorrhoeae on agar bacterial culture plates][center] Culture sensitivity approaches 95% for genital specimens from symptomatic individuals when performed optimally but is lower (50-70%) for extragenital or asymptomatic infections due to intermittent shedding and overgrowth by normal microbiota.[157] Integration with susceptibility testing is imperative, as isolates are subcultured onto media like GC agar base for disk diffusion or broth microdilution to assess minimum inhibitory concentrations against agents like ceftriaxone, guiding personalized therapy in confirmed cephalosporin-resistant cases reported globally since 2009.[155] Limitations include the need for specialized laboratory infrastructure, biosafety level 2 containment, and potential for overgrowth if selective antibiotics fail against certain commensals, underscoring the technique's role as complementary to non-culture methods in comprehensive diagnostics.[29]Molecular and Serological Methods
Nucleic acid amplification tests (NAATs) represent the primary molecular methods for detecting Neisseria gonorrhoeae, offering high sensitivity exceeding 95% and specificity greater than 99% in urogenital, rectal, and pharyngeal specimens.[30] These assays typically target conserved genes such as the multicopy opa gene or the pilin gene (pilE), with some dual-target formats incorporating porA pseudogene sequences to enhance specificity and reduce cross-reactivity with commensal Neisseria species.[158] Multiplex NAAT platforms, commonly combining N. gonorrhoeae detection with Chlamydia trachomatis, enable simultaneous screening for co-infections, which occur in up to 20-40% of cases depending on population demographics, facilitating efficient extragenital site testing.[159][160] Serological methods, relying on detection of antibodies against gonococcal antigens like outer membrane proteins, are rarely used for routine diagnosis due to poor sensitivity and specificity stemming from the pathogen's localized mucosal infection, which elicits minimal systemic humoral immunity.[30] Historical enzyme-linked immunosorbent assays (ELISAs) targeting IgG to gonococcal pili or porin proteins have shown variable performance, often failing to distinguish active from past or cross-reactive infections with other Neisseria species.[161] Whole-genome sequencing (WGS) of N. gonorrhoeae isolates, increasingly applied in epidemiological surveillance, supports molecular subtyping via multi-locus sequence typing (MLST) or core-genome analysis but is not a frontline diagnostic tool; it aids in tracking transmission clusters and distinguishing true positives from contaminants.[162] False-positive NAAT results, particularly in pharyngeal samples, can arise from amplification of nucleic acids from commensal Neisseria like N. meningitidis or N. lactamica, necessitating confirmatory testing such as culture or supplementary NAATs against culture-validated specimens to maintain accuracy.[163][164]Treatment
Current Therapeutic Guidelines
The recommended treatment for uncomplicated urogenital, rectal, or pharyngeal gonorrhea caused by Neisseria gonorrhoeae is a single intramuscular dose of ceftriaxone 500 mg for individuals weighing ≤150 kg or 1 g for those >150 kg, reflecting efficacy rates exceeding 95% in clinical trials and surveillance data for susceptible strains.[112] This regimen represents an evolution from prior dual therapy with ceftriaxone and azithromycin, driven by high azithromycin resistance rates (up to 30% in some regions) and preserved ceftriaxone susceptibility, allowing monotherapy to minimize unnecessary antimicrobial exposure while maintaining cure rates.[165] Co-infection with Chlamydia trachomatis should be addressed empirically with doxycycline 100 mg orally twice daily for 7 days if not excluded by testing, as gonorrhea often co-occurs with chlamydia in 20-40% of cases.[112] For cephalosporin-allergic patients, alternatives include gentamicin 240 mg intramuscularly plus azithromycin 2 g orally as a single dose, supported by randomized trials showing 94-100% efficacy but higher gastrointestinal side effects.[7] In pregnancy, ceftriaxone remains first-line, with spectinomycin 2 g intramuscularly as an alternative if cephalosporins are contraindicated, based on pharmacokinetic and safety data avoiding fetal risks from other agents.[112] Test-of-cure via culture or nucleic acid amplification testing is advised 7-14 days post-treatment for pharyngeal infections due to lower efficacy (around 90%) compared to other sites, and for any treatment failure or re-exposure.[166] Patients should abstain from sexual activity for 7 days post-treatment and until symptoms resolve and partners are treated to prevent reinfection, with expedited partner therapy (EPT) recommended where legal, providing prescriptions to recent partners without examination, which reduces reinfection by 50% in controlled studies.[165] All treatments assume local susceptibility patterns; consultation with public health authorities is essential in areas with elevated minimum inhibitory concentrations to ceftriaxone (>0.125 μg/mL).[166]Antibiotic Resistance Mechanisms and Surveillance
Neisseria gonorrhoeae has developed resistance to multiple antibiotic classes through a combination of chromosomal mutations and plasmid acquisition, driven by selective pressures from widespread antibiotic use since the 1980s.[6] [7] Key mechanisms include the plasmid-mediated production of beta-lactamase (TEM-1), which hydrolyzes penicillins and early cephalosporins, rendering these drugs ineffective in over 95% of isolates globally by the 1980s.[6] Chromosomal alterations, such as mutations in the mtrR repressor gene, upregulate the MtrCDE efflux pump, expelling macrolides like azithromycin, tetracyclines, and fluoroquinolones, contributing to multidrug resistance phenotypes.[167] [168] For extended-spectrum cephalosporins like ceftriaxone, resistance primarily arises from mosaic alleles of the penA gene, encoding penicillin-binding protein 2 (PBP2) with reduced affinity for beta-lactams due to recombination with commensal Neisseria species.[169] [170] These mosaic penA variants, such as penA-60.001, combined with efflux overexpression, elevate minimum inhibitory concentrations (MICs), with ceftriaxone-resistant strains emerging as early as 2009 and persisting in clonal lineages.[170] Additional targets include ribosomal mutations (e.g., 23S rRNA) for high-level azithromycin resistance and gyrA/parC mutations for quinolones, often co-occurring to produce extensively drug-resistant (XDR) profiles.[7] [171] Suboptimal dosing and monotherapy regimens have accelerated this adaptation by favoring surviving mutants with fitness-compensatory changes.[6] Global surveillance, coordinated by the World Health Organization's Global Antimicrobial Resistance and Use Surveillance System (GLASS) and Gonococcal Antimicrobial Surveillance Programme (GASP), tracks resistance trends through standardized MIC testing and molecular characterization.[172] 00109-0/fulltext) The 2025 GLASS report documents widespread N. gonorrhoeae resistance to azithromycin (up to 25% in sentinel sites across multiple countries) and rising ceftriaxone MICs, with XDR clones like multilocus sequence type (MLST) ST-16406—harboring penA-60.001, high-level azithromycin resistance, and quinolone resistance—detected in Europe, Canada, and Asia-Pacific regions since 2022.[172] [173] 00486-9/fulltext) These clones, resistant to ceftriaxone, cefixime, azithromycin, and fluoroquinolones, underscore the need for genomic surveillance to detect transnational spread, as evidenced by 4 European cases by 2024 and subsequent detections in 2025.[174] [144] National programs, such as those in the US and UK, complement WHO efforts by monitoring local MIC creep and plasmid profiles.[175]Prevention
Behavioral and Public Health Interventions
Partner notification and contact tracing are established public health strategies for controlling Neisseria gonorrhoeae transmission, involving index patients or providers alerting recent sexual partners for testing and treatment. Clinical trials indicate these methods reduce the index patient's reinfection risk by interrupting transmission chains, with modeling suggesting that enhanced partner notification could decrease gonorrhea cases by up to 45% in targeted populations.[176][177] Effectiveness depends on timely implementation, as provider-referred notification outperforms patient self-referral in detecting new infections among partners.[178] Screening programs targeting high-risk groups, such as sexually active adolescents, men who have sex with men, and individuals with multiple partners, have demonstrated reductions in gonorrhea incidence. Retrospective analyses estimate that routine screening averted approximately 30% of infections that would otherwise have occurred between 2000 and 2015 in the United States, aligning with broader evidence of 20-50% incidence reductions through sustained efforts in vulnerable populations.[179] These interventions prioritize empirical detection in groups with elevated prevalence, though low screening uptake due to barriers like stigma limits overall impact.[180] Promotion of barrier methods like consistent condom use provides partial protection against gonorrhea transmission, with epidemiological studies associating regular use with lower infection risks in both men and women. Latex condoms, when applied correctly and consistently, reduce transmission probabilities for gonorrhea by covering exposed areas, though efficacy varies by site of infection (e.g., higher for penile than pharyngeal exposure) and adherence levels.[181][182] Empirical data underscore that condoms mitigate but do not eliminate risk, as incomplete coverage and breakage contribute to breakthrough infections.[183] Abstinence from sexual activity and mutual monogamy with uninfected partners represent zero-risk behavioral strategies for preventing gonorrhea acquisition, grounded in the pathogen's exclusive transmission via mucosal contact during intercourse. Public health guidance emphasizes these as the only absolute preventives, with evidence showing no transmission in the absence of sexual exposure or when partners are verified negative through prior testing and maintained fidelity.[184] Monogamy's protective effect holds only if both parties abstain from extrapair contact, as undisclosed prior infections can persist asymptomatically.[185] Routine testing alone, without concurrent behavioral modifications, fails to curb gonorrhea's spread due to high reinfection rates from untreated partners or persistent high-risk practices. Up to 20% of treated individuals experience repeat chlamydia or gonorrhea within six months, often attributable to re-exposure rather than treatment failure, highlighting the need for integrated risk reduction counseling.[186][187] Studies confirm that retesting protocols without addressing partner treatment or sexual network dynamics yield limited long-term incidence declines, as chains of transmission persist absent causal interventions like partner management.[188][189]Vaccine Development and Challenges
No licensed vaccine exists for Neisseria gonorrhoeae, despite decades of research efforts hampered by the bacterium's immune evasion strategies.[190] Observational studies have demonstrated partial cross-protection against gonorrhea from outer membrane vesicle (OMV)-based meningococcal serogroup B (MenB) vaccines, such as 4CMenB (Bexsero) and MenB-FHbp (Trumenba), with vaccine effectiveness estimates ranging from 20% to 40% against infection, particularly following full vaccination series.[190][191] This cross-protection arises from shared antigens like factor H binding protein (fHbp) and PorA between N. meningitidis and N. gonorrhoeae, though efficacy varies by strain and population, with stronger effects observed in New Zealand's MeNZB campaign (31% reduction).[192] In response, public health bodies in regions like England and Wales initiated targeted MenB vaccination programs in 2025 for high-risk groups to leverage this incidental benefit amid rising gonorrhea incidence.[193] Promising gonorrhea-specific candidates are advancing in preclinical and early clinical stages. A generalized modules for membrane antigens (GMMA)-based vaccine from GSK, incorporating detergent-free outer membrane vesicles from N. gonorrhoeae, elicited functional antibodies in preclinical models, including bactericidal, opsonophagocytic, and serum-resistant responses against diverse strains as of July 2025.[194] This approach builds on OMV immunogenicity while addressing scalability, with phase 1/2 trials planned for at-risk adults aged 18-50.[195] Complementing this, artificial intelligence-driven reverse vaccinology has identified conserved epitopes on proteins like adhesins and transporters, prioritizing candidates with high immunogenicity and low variability across global isolates for epitope-based subunit vaccines.[196] These computational methods integrate genomic data from over 5,000 strains to predict T- and B-cell epitopes, potentially overcoming historical trial failures like the 1970s pilin vaccine, which faltered due to phase-variable expression.[197] Key challenges persist, rooted in N. gonorrhoeae's antigenic hypervariability and niche biology. Surface structures such as type IV pili, opacity proteins (Opa), and lipooligosaccharide undergo frequent phase and antigenic variation, enabling immune escape and reducing epitope conservation to below 50% in many candidates across isolates.[198] Effective immunity requires robust mucosal IgA and T-cell responses at urogenital sites, yet systemic vaccines often fail to induce site-specific protection, as evidenced by limited correlation between serum bactericidal activity and mucosal clearance in animal models.[199] Human challenge models and non-human primates partially mimic disease but underestimate chronicity and ascension risks, complicating efficacy readouts.[197] Development timelines for viable candidates exceed 10 years, factoring in large-scale trials needed for sexually transmitted infection endpoints amid ethical constraints on placebo use in endemic areas.[195]History
Discovery and Initial Characterization
In 1879, German physician Albert Ludwig Sigesmund Neisser identified gram-negative diplococci in microscopic examinations of purulent discharge from patients with gonorrhea, establishing these bacteria as the causative agent of the disease.[200][201] Neisser's observations built on earlier microscopic work but provided the first definitive link between the diplococci and gonococcal infection through consistent findings in affected tissues. The organism was subsequently named Neisseria gonorrhoeae in his honor, reflecting its paired (diplo-) coccus morphology and association with gonorrhea.[203] Initial characterization emphasized the bacterium's fastidious growth requirements and intracellular presence within polymorphonuclear leukocytes, distinguishing it from contaminants or other flora in clinical samples.[11] By the early 1880s, N. gonorrhoeae was differentiated from Neisseria meningitidis, isolated in 1887 from cerebrospinal fluid in meningitis cases, based on differences in colonial morphology, growth on selective media, and disease specificity—gonococci failing to ferment certain sugars and requiring enriched atmospheres unlike meningococci.[204] These distinctions relied on phenotypic traits rather than genetic analysis, as Koch's postulates were applied to confirm pathogenicity through animal inoculation and culture attempts, though N. gonorrhoeae's obligate human tropism limited full fulfillment.[200] Prior to antibiotics, gonorrhea was widely recognized as a sexually transmitted infection, with symptoms documented since antiquity—Hippocrates described urethral discharge in the 5th century BCE—yet Neisser's work shifted understanding from humoral theories to microbial etiology.[201] Treatments involved irrigants like tannic acid or calomel, but transmission via sexual contact, perinatal routes, and fomites was acknowledged, prompting early public health measures like contact tracing in military contexts.[203] Neisser's inoculation experiments on human subjects to verify causality, including self-experimentation, later drew ethical scrutiny, particularly his unrelated 1890s syphilis serum trials on non-consenting individuals, which catalyzed the 1900 Prussian guidelines mandating consent in medical research.[205]Key Advances in Research and Control
In the 1940s, penicillin emerged as a highly effective treatment for gonorrhea caused by Neisseria gonorrhoeae, dramatically reducing case fatality and morbidity rates compared to prior silver-based therapies.[206] By the 1960s, however, plasmid-mediated and chromosomal resistance mechanisms led to widespread treatment failures, prompting shifts to alternative antibiotics like tetracyclines and later fluoroquinolones.[207] This early evolution highlighted the bacterium's capacity for rapid adaptation via genetic recombination and horizontal gene transfer, setting the stage for ongoing therapeutic challenges.[6] The genomic era, accelerating after the first complete N. gonorrhoeae genome sequences in the early 2000s, revealed extensive strain diversity and resistance determinants, enabling whole-genome sequencing (WGS) for epidemiological tracking.[208] In the 2010s, the World Health Organization issued urgent warnings on multidrug-resistant strains, including a 2012 alert on cephalosporin-resistant "superbug" variants spreading globally, which spurred enhanced surveillance programs like the Global Gonococcal Antimicrobial Surveillance Programme (GASP).[209] By the 2020s, WGS integration into routine surveillance allowed real-time detection of resistance mutations, such as those in penA and mtrR genes, facilitating outbreak containment and informing local treatment adaptations.[210][102] Responding to cephalosporin resistance threats, the U.S. Centers for Disease Control and Prevention recommended dual therapy—ceftriaxone plus azithromycin—in its 2010 guidelines to suppress resistant subpopulations and delay further evolution.[112] This approach, adopted internationally, reduced treatment failure rates initially but faced challenges from azithromycin resistance exceeding 5% in some regions by the mid-2010s.[211] Vaccine development saw resurgence in the 2010s, driven by observational data showing 40-50% cross-protection from meningococcal serogroup B vaccines against gonorrhea due to shared outer membrane proteins like PorA and factor H-binding protein.[212] Phase II trials of these vaccines reported efficacy signals, prompting dedicated gonococcal candidates, including outer membrane vesicle-based formulations entering clinical testing by 2023, with fast-track designations for novel constructs targeting pilin and opacity proteins.[213][214]References
- https://pubmed.ncbi.nlm.nih.gov/16210113/

