Recent from talks
Nothing was collected or created yet.
Aurelia (cnidarian)
View on Wikipedia
| Aurelia | |
|---|---|

| |
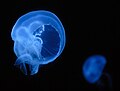
| Adult Aurelia medusa in the Red Sea | |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Cnidaria |
| Class: | Scyphozoa |
| Order: | Semaeostomeae |
| Family: | Ulmaridae |
| Genus: | Aurelia Lamarck, 1816 |
| Synonyms[1] | |
| |
Aurelia is a genus of jellyfish that are commonly called moon jellies, which are in the class Scyphozoa. There are currently 25 accepted species and many that are still not formally described.[2][3][4]
The genus was first described in 1816 by Jean-Baptiste Lamarck in his book Histoire Naturelle des Animaux sans Vertèbres (Natural History of Invertebrates).[5] It has been suggested that Aurelia is the best-studied group of gelatinous zooplankton, with Aurelia aurita the best-studied species in the genus; two other species, Aurelia labiata and Aurelia limbata were also traditionally investigated throughout the 20th century.[6] In the early 2000s, studies that considered genetic data showed that diversity in Aurelia was higher than expected based solely on morphology,[7][8] so one cannot confidently attribute the results from most of the previous studies to the species named. More recently, studies have highlighted the morphological variability[3] (including the potential for phenotypic plasticity[9][10]) in this genus, emphasizing the difficulty of identifying cryptic species.[11]
Species of Aurelia can be found in the Atlantic, Arctic, Pacific and Indian Oceans, and seem to be more common in temperate regions, such as in the waters off northern China, Japan, Korea, New Zealand, the northeastern and northwestern coasts of the United States, and those of northern Europe.[3]
Moon jellies differ from many jellyfish in that they lack long, potent stinging tentacles. Instead they have hundreds of short, fine tentacles that line the bell margin. The sting has a mild effect on humans, with most having little or no reaction.[12]
Aurelia undergoes alternation of generations, whereby the sexually-reproducing pelagic medusa stage is either male or female, and the benthic polyp stage reproduces asexually. Meanwhile, life cycle reversal, in which polyps are formed directly from juvenile and sexually mature medusae or their fragments, was also observed in Aurelia coerulea (= Aurelia sp. 1).[13][not in body]
Description
[edit]
The similar appearances of moon jellyfish species is what has made them so hard to identify. They tend to have a variety of different sizes, however, they typically range 5–38 cm (2.0–15.0 in) in diameter with an average of 18 cm (7.1 in) wide and 8 cm (3.1 in) in height.[14][15] The polyps of these jellyfish can grow to 1.6 cm (0.63 in) tall and their ephyrae have an average diameter of 0.4 cm (0.16 in).[16]
The basic body plan of Aurelia consists of several parts. The animal lacks respiratory, excretory, and circulatory systems. The adult medusa of Aurelia, with a transparent look, has an umbrella margin membrane and tentacles that are attached to the bottom.[17] It has four bright gonads that are under the stomach.[17] The medusae are either male or female (gonochory).[14]
The adult medusae are typically translucent,[16] but the color of their gut can change based on what they eat; for example, if they eat certain crustaceans, they can have a pink or lavender tint to them; if they were to eat brine shrimp, the tint would be more of an orange color.[18] Their polyps usually have around 16 tentacles (although Aurelia insularia has 27–33 tentacles)[3][19] which mostly help with feeding.[16]
Physiology
[edit]
Aurelia does not have respiratory parts such as gills, lungs, or trachea; it respires by diffusing oxygen from water through the thin membrane covering its body. Within the gastrovascular cavity, low oxygenated water can be expelled and high oxygenated water can come in by ciliated action, thus increasing the diffusion of oxygen through the cell.[20] The large surface area membrane to volume ratio helps Aurelia diffuse more oxygen and nutrients into the cells.
Food travels through the muscular manubrium while the radial canals help disperse the food.[17] There is a middle layer of mesoglea, a gastrodervascular cavity with a gastrodermis, and an epidermis.[21] There is a nerve net that is responsible for contractions in swimming muscles and feeding responses.[14]
They are able to sense light and dark and up and down due to rhopalia around the bell margin.[15]
Venom
[edit]Testing on frogs determined that A. aurita has a proteinaceous venom that causes muscle twitching by inducing the irreversible depolarization of the muscle membrane that is believed to be caused by an increase in the membrane's permeability to sodium ions.[22]
Species
[edit]This section is missing information about locales/geographical distribution for each species. (February 2026) |
The following species of Aurelia is accepted by the World Register of Marine Species:[1]
- Aurelia aurita (Linnaeus, 1758)
- Aurelia ayla Lawley, Gamero-Mora, Maronna, Chiaverano, Stampar, Hopcroft, Collins & Morandini, 2021
- Aurelia cebimarensis Lawley, Gamero-Mora, Maronna, Chiaverano, Stampar, Hopcroft, Collins & Morandini, 2021
- Aurelia clausa Lesson, 1830
- Aurelia coerulea von Lendenfeld, 1884
- Aurelia colpota Brandt, 1835
- Aurelia columbia Lawley, Gamero-Mora, Maronna, Chiaverano, Stampar, Hopcroft, Collins & Morandini, 2021
- Aurelia dubia Vanhöffen, 1888
- Aurelia hyalina Brandt, 1835
- Aurelia insularia Lawley, Gamero-Mora, Maronna, Chiaverano, Stampar, Hopcroft, Collins & Morandini, 2021
- Aurelia labiata Chamisso & Eysenhardt, 1821
- Aurelia limbata Brandt, 1835
- Aurelia malayensis Lawley, Gamero-Mora, Maronna, Chiaverano, Stampar, Hopcroft, Collins & Morandini, 2021
- Aurelia maldivensis Bigelow, 1904
- Aurelia marginalis Agassiz, 1862
- Aurelia mianzani Lawley, Gamero-Mora, Maronna, Chiaverano, Stampar, Hopcroft, Collins & Morandini, 2021
- Aurelia miyakei Lawley, Gamero-Mora, Maronna, Chiaverano, Stampar, Hopcroft, Collins & Morandini, 2021
- Aurelia montyi Lawley, Gamero-Mora, Maronna, Chiaverano, Stampar, Hopcroft, Collins & Morandini, 2021
- Aurelia mozambica Brown & Gibbons, 2021
- Aurelia persea (Forsskål, 1775)
- Aurelia profunda Frolova, Mammone & Miglietta, 2025
- Aurelia pseudosolida Garić & Batistić, 2022
- Aurelia rara Lawley, Gamero-Mora, Maronna, Chiaverano, Stampar, Hopcroft, Collins & Morandini, 2021
- Aurelia relicta Scorrano, Aglieri, Boero, Dawson & Piraino, 2016
- Aurelia smithsoniana Lawley, Gamero-Mora, Maronna, Chiaverano, Stampar, Hopcroft, Collins & Morandini, 2021
- Aurelia solida Browne, 1905
- Aurelia vitiana Agassiz & Mayer, 1899
-
Aurelia sp. from the Monterey Bay Aquarium
-
A damaged Aurelia sp. individual
-
An adult Aurelia aurita
-
On the beach
-
Aurelia aurita washed up on the beach, Jūrmala
-
Aurelia aurita at Ozeaneum Stralsund
Distribution and habitat
[edit]Aurelia species inhabit worldwide habitats.[23]It is found in the North, Black, Baltic and Caspian Seas, Northeast Atlantic, Greenland, northeastern USA and Canada, Northwest Pacific and South America.[3][24][25] In general, Aurelia is an inshore genus that can be found in estuaries and harbors.[17]
Aurelia live in ocean water temperatures ranging from 6–31 °C (43–88 °F); with optimum temperatures of 9–19 °C (48–66 °F). It prefers temperate seas with consistent currents. It has been found in waters with salinity as low as 6 parts per thousand.[26]
The relation between summer hypoxia and moon jellyfish distribution is prominent during the summer months of July and August where temperatures are high and dissolved oxygen (DO) is low. Of the three environmental conditions tested, bottom DO has the most significant effect on moon jellyfish abundance. Moon jellyfish abundance is the highest when bottom dissolved oxygen concentration is lower than 2.0 mg L−1.[27] Moon jellyfish show a strong tolerance to low DO conditions, which is why their population is still relatively high during the summer. Generally, hypoxia causes species to move from the oxygen depleted zone, but this is not the case for the moon jellyfish. Furthermore, bell contract rate, which indicates moon jellyfish feeding activity, remains constant despite lower DO concentrations than normal.[27] Other major fish predators that are also present in these coastal waters do not seem to show the same high tolerance to low DO concentrations that the moon jellyfish exhibit. The feeding and predatory performance of these fish significantly decreases when DO concentrations are so low. This allows for less competition between the moon jellyfish and other fish predators for zooplankton. Low DO concentrations in coastal waters such as Tokyo Bay in Japan and the Seto Inland Sea prove to be advantageous for the moon jellyfish in terms of feeding, growth, and survival.
Biology
[edit]
The diet of Aurelia is similar to that of other jellyfish. They primarily feed on zooplankton.[15] Occasionally, they are also seen feeding on gelatinous zooplankton such as hydromedusae and ctenophores.[26]
The food is caught with its nematocyst-laden tentacles, tied with mucus, brought to the gastrovascular cavity, and passed into the cavity by ciliated action. There, digestive enzymes from serous cells break down the food. Little is known about the requirements for particular vitamins and minerals, but due to the presence of some digestive enzymes, we can deduce in general that Aurelia can process carbohydrates, proteins, and lipids.[14]
During July and August, it is observed that moon jellyfish aggregations of 250 individuals consumed an estimated 100% of the mesozooplankton biomass in the Seto Inland Sea.[28] They may prey on or compete with commercially important fish and their larvae, as well as cause several issues for trawling boats when large aggregations occur,[29] as they may clog and damage fishing nets as well as force fisherman to relocate.[30]
A 2020 study found that Aurelia's body system is not significantly affected by artificial materials like microbeads, which can be found in cosmetic and personal care products. Aurelia aurita was able to recognize that microbeads were not food so there was not any physiological or histological harm.[31]

Aurelia have high proportions of polyunsaturated fatty acids compared to other prey types and are a source of vital nutrients for predators.[32] Aurelia are known to be eaten by a wide variety of predators, including the ocean sunfish (Mola mola), the leatherback sea turtle (Dermochelys coriacea), the scyphomedusa Phacellophora camtschatica,[33][34] and a very large hydromedusa (Aequorea victoria).[14] In 2016, it was reported from the Red Sea that Aurelia were seasonally preyed upon by two herbivorous fish.[35] Moon jellies are also fed upon by sea birds, which may be more interested in the amphipods and other small arthropods that frequent the bells of Aurelia, but in any case, birds do some substantial amount of damage to these jellyfish that often are found just at the surface of bays. Aurelia species have been suggested to have high mortality during the ephyra stage, which potentially affects the population size of the later medusa stage. While the main cause remains unknown, it is believed that they are consumed by one of three potential predatory filter-feeding sessile organisms: mussels, ascidians, and barnacles.[36]
Some metazoan parasites attack Aurelia, as well as most other species of jellyfish.[14]
Reproduction
[edit]
The medusa stage of the jellyfish reproduce sexually. The males release strings of sperm and the females ingest them.[18] Once the ciliated larvae develop from the egg, they settle on or near the sea floor and develop into benthic polyps. The polyps then reproduce asexually and bud into ephyrae which later turn into medusae.
The young larval stage, a planula, has small ciliated cells and after swimming freely in the plankton for a day or more, settles on an appropriate substrate, where it changes into a special type of polyp called a "scyphistoma", which divides by strobilation into small ephyrae that swim off to grow up as medusae.[37][38] There is an increasing size from starting stage planula to ephyra, from less than 1 mm in the planula stage, up to about 1 cm in ephyra stage, and then to several cm in diameter in the medusa stage.[17]
Aurelia jellyfish naturally die after living and reproducing for several months. It is probably rare for these moon jellies to live more than about six months in the wild, although specimens cared for in public aquarium exhibits typically live several to many years. In the wild, the warm water at the end of summer combines with exhaustive daily reproduction and lower natural levels of food for tissue repair, leaving these jellyfish more susceptible to bacterial and other disease problems that likely lead to the demise of most individuals. Such problems are responsible for the demise of many smaller species of jellyfish.[39] In 1997, it was summarized that seasonal reproduction leaves the gonads open to infection and degradation.[14]
A 2015 study has found that A. aurita are capable of life cycle reversal where individuals grow younger instead of older, akin to the "immortal jellyfish" Turritopsis dohrnii.[40]
See also
[edit]References
[edit]- ^ a b "Aurelia Lamarck, 1816". WoRMS. World Register of Marine Species. Retrieved 2026-02-11.
- ^ Collins, A. G.; Jarms, G.; Morandini, A. C. (September 12, 2021). "World List of Scyphozoa. Aurelia Lamarck, 1816". WoRMS. World Register of Marine Species. Retrieved September 12, 2021.
- ^ a b c d e Lawley, J. W.; Gamero-Mora, E.; Maronna, M. M.; Chiaverano, L. M.; Stampar, S. N.; Hopcroft, R. R.; Collins, A. G.; Morandini, A. C. (2021). "The importance of molecular characters when morphological variability hinders diagnosability: systematics of the moon jellyfish genus Aurelia (Cnidaria: Scyphozoa)". PeerJ. 9:e11954 e11954. doi:10.7717/peerj.11954. PMC 8435205. PMID 34589293.
- ^ Brown, M.; Scorrano, S.; Kuplik, Z.; Kuyper, D.; Ras, V.; Thibault, D.; Engelbrecht, A.; Gibbons, M. J. (2021). "A new macromedusa from the coast of Mozambique: Aurelia mozambica sp. nov. (Scyphozoa: Ulmaridae)". Zootaxa. 4933 (2): 263–276. doi:10.11646/zootaxa.4933.2.5. hdl:10566/6168. PMID 33756798. S2CID 232339936.
- ^ Lamarck, J.-B. M. de. (1816). "Histoire naturelle des animaux sans vertèbres". www.biodiversitylibrary.org. Verdière: Biodiversity Heritage Library. Archived from the original on 2014-05-13. Retrieved 23 June 2021.
- ^ Arai, Mary Needler. A Functional Biology of Scyphozoa. London: Chapman and Hall. pp. 68–206.
- ^ Schroth, Werner; Jarms, Gerhard; Streit, Bruno; Schierwater, Bernd (2 January 2002). "Speciation and phylogeography in the cosmopolitan marine moon jelly, Aurelia sp". BMC Evolutionary Biology. 2: 1. doi:10.1186/1471-2148-2-1. PMC 64640. PMID 11801181.
- ^ Dawson, Michael N (2003). "Macro-morphological variation among cryptic species of the moon jellyfish, Aurelia (Cnidaria: Scyphozoa)". Marine Biology. 143 (2): 369–379. Bibcode:2003MarBi.143..369D. doi:10.1007/s00227-003-1070-3. S2CID 189820003.
- ^ Chiaverano, Luciano M.; Bayha, Keith W.; Graham, William M. (2016-06-22). Colgan, Donald James (ed.). "Local versus Generalized Phenotypes in Two Sympatric Aurelia Species: Understanding Jellyfish Ecology Using Genetics and Morphometrics". PLOS ONE. 11 (6) e0156588. Bibcode:2016PLoSO..1156588C. doi:10.1371/journal.pone.0156588. ISSN 1932-6203. PMC 4917110. PMID 27332545.
- ^ Chiaverano, Lm; Graham, Wm (2017-11-06). "Morphological plasticity in Aurelia polyps, with subsequent effects on asexual fecundity and morphology of young medusae". Marine Ecology Progress Series. 582: 79–92. Bibcode:2017MEPS..582...79C. doi:10.3354/meps12314. ISSN 0171-8630. S2CID 90233141.
- ^ Lawley, Jonathan W.; Gamero-Mora, Edgar; Maronna, Maximiliano M.; Chiaverano, Luciano M.; Stampar, Sérgio N.; Hopcroft, Russell R.; Collins, Allen G.; Morandini, André C. (2022-09-29). "Morphology is not always useful for diagnosis, and that's ok: Species hypotheses should not be bound to a class of data. Reply to Brown and Gibbons (S Afr J Sci. 2022;118(9/10), Art. #12590)". South African Journal of Science. 118 (9/10). doi:10.17159/sajs.2022/14495. ISSN 1996-7489. S2CID 252562185.
- ^ "Moon Jelly | Learn With the South Carolina Aquarium". South Carolina Aquarium. Retrieved 2025-08-22.
- ^ He, Jinru; Zheng, Lianming; Zhang, Wenjing; Lin, Yuanshao; Steele, Robert E. (21 December 2015). "Life Cycle Reversal in Aurelia sp.1 (Cnidaria, Scyphozoa)". PLOS ONE. 10 (12) e0145314. Bibcode:2015PLoSO..1045314H. doi:10.1371/journal.pone.0145314. PMC 4687044. PMID 26690755.
- ^ a b c d e f g Arai, M. N. (1997). A Functional Biology of Scyphozoa. London: Chapman and Hall. pp. 68–206. ISBN 978-0-412-45110-2.
- ^ a b c "Moon Jellyfish". www.dwazoo.com. Dallas World Aquarium. 2 December 2013. Retrieved 23 June 2021.
- ^ a b c Fletcher, McKenzie. "Aurelia". Animal Diversity Web. Retrieved 23 June 2021.
- ^ a b c d e f Russell, F. S. (1953). The Medusae of the British Isles II. London: Cambridge University Press. pp. 81–186. Archived from the original on 2016-06-23. Retrieved 2012-07-03.
- ^ a b "Moon jelly". www.montereybayaquarium.org/. Monterey Bay Aquarium. Retrieved 23 June 2021.
- ^ Gambill, Maria; Jarms, Gerhard (2014-12-26). "Can Aurelia (Cnidaria, Scyphozoa) species be differentiated by comparing their scyphistomae and ephyrae?". European Journal of Taxonomy (107). doi:10.5852/ejt.2014.107. ISSN 2118-9773.
- ^ Rees, W. J. (1966). The Cnidaria and Their Evolution. London: Academic Press. pp. 77–104.
- ^ Solomon, E. P.; Berg, L. R.; Martin, W. W. (2002). Biology (6th ed.). London: Brooks/Cole. pp. 602–608. ISBN 978-0-534-39175-1.
- ^ Ponce, Dalia; López-Vera, Estuardo; Aguilar, Manuel B.; Sánchez-Rodríguez, Judith (6 December 2013). "Preliminary Results of the in Vivo and in Vitro Characterization of a Tentacle Venom Fraction from the Jellyfish Aurelia aurita". Toxins. 5 (12): 2420–2433. doi:10.3390/toxins5122420. PMC 3873694. PMID 24322597.
- ^ Schroth, Werner; Jarms, Gerhard; Streit, Bruno; Schierwater, Bernd (2002-01-02). "Speciation and phylogeography in the cosmopolitan marine moon jelly, Aurelia sp". BMC Evolutionary Biology. 2 (1): 1. doi:10.1186/1471-2148-2-1. ISSN 1471-2148. PMC 64640. PMID 11801181.
- ^ Dawson, M. N.; Sen Gupta, A.; England, M. H. (2005). "Coupled biophysical global ocean model and molecular genetic analyses identify multiple introductions of cryptogenic species". Proc. Natl. Acad. Sci. USA. 102 (34): 11968–73. Bibcode:2005PNAS..10211968D. doi:10.1073/pnas.0503811102. PMC 1189321. PMID 16103373.
- ^ Dawson, M. N. (2003). "Macro-morphological variation among cryptic species of the moon jellyfish, Aurelia (Cnidaria: Scyphozoa)". Marine Biology. 143 (2): 369–79. Bibcode:2003MarBi.143..369D. doi:10.1007/s00227-003-1070-3. S2CID 189820003.
- ^ a b Rodriguez, R. J. (February 1996). "Aurelia aurita (Saucer Jelly, Moon Jelly, Common Sea Jelly Jellyfish) Narrative".
- ^ a b Shoji, J.; Yamashita, R.; Tanaka, M. (2005). "Effect of low dissolved oxygen concentrations on behavior and predation rates on fish larvae by moon jellyfish Aurelia aurita and by a juvenile piscivore, Spanish mackerel Scomberomorus niphonius". Marine Biology. 147 (4): 863–68. doi:10.1007/s00227-005-1579-8. S2CID 83862921.
- ^ Uye, S.; Fujii, N.; Takeoka, H. (2003). "Unusual aggregations of the scyphomedusa Aurelia aurita in coastal waters along western Shikoku, Japan" (PDF). Plankton Biology and Ecology. 50 (1): 17–21.
- ^ Försterra, Günter; Häussermann, Vreni (2009). "First Record of the Moon Jellyfish, Aurelia for Chile" (PDF). Spixiana. 32 (1): 3–7.
- ^ Conley, Keats R.; Sutherland, Kelly R. (1 June 2015). "Commercial fishers' perceptions of jellyfish interference in the Northern California Current". ICES Journal of Marine Science. 72 (5): 1565–1575. doi:10.1093/icesjms/fsv007.
- ^ Sucharitakul, Phuping (2020). "Limited ingestion, rapid egestion and no detectable impacts of microbeads on the moon jellyfish, Aurelia aurita". Marine Pollution Bulletin. 156 111208. Bibcode:2020MarPB.15611208S. doi:10.1016/j.marpolbul.2020.111208. hdl:10072/393652. PMID 32366368. S2CID 218504266.
- ^ "Jellyfish contain no calories, so why do they still attract predators?". ScienceDaily. Retrieved 2020-06-29.
- ^ Strand, S. W.; Hamner, W. M. (1988). "Predatory behavior of Phacellophora camtschatica and size-selective predation upon Aurelia aurita (Scyphozoa: Cnidaria) in Saanich Inlet, British Columbia". Marine Biology. 99 (3): 409–414. Bibcode:1988MarBi..99..409S. doi:10.1007/BF02112134. S2CID 84652019.
- ^ Towanda, T.; Thuesen, E. V. (2006). "Ectosymbiotic behavior of Cancer gracilis and its trophic relationships with its host Phacellophora camtschatica and the parasitoid Hyperia medusarum". Marine Ecology Progress Series. 315: 221–236. Bibcode:2006MEPS..315..221T. doi:10.3354/meps315221.
- ^ Bos A.R., Cruz-Rivera E. and Sanad A.M. (2016). "Herbivorous fishes Siganus rivulatus (Siganidae) and Zebrasoma desjardinii (Acanthuridae) feed on Ctenophora and Scyphozoa in the Red Sea". Marine Biodiversity. 47: 243–246. doi:10.1007/s12526-016-0454-9. S2CID 24694789.
- ^ Suzuki, Kentaro S.; Kumakura, Emi; Nogata, Yasuyuki (2016). "Incidental consumption of ephyrae of moon jellyfish Aurelia aurita s.l. by three filter-feeding sessile organisms: laboratory experiments". Fisheries Science. 82 (6): 923–930. Bibcode:2016FisSc..82..923S. doi:10.1007/s12562-016-1034-4.
- ^ http://tolweb.org/treehouses/?treehouse_id=3539 Tree of Life – NJ Jellyfish – Aurelia aurita
- ^ Gilbertson, L. (1999). Zoology Laboratory Manual (4th ed.). McGraw-Hill. pp. 9.2 – 9.7. ISBN 978-0-07-229641-9.
- ^ Mills, C. E. (1993). "Natural mortality in NE Pacific coastal hydromedusae: grazing predation, wound healing and senescence". Bulletin of Marine Science. 53 (Proceedings of the Zooplankton Ecology Symposium): 194–203.
- ^ He, J; Zheng, L; Zhang, W; Lin, Y (2015). "Life cycle reversal in Aurelia sp.1 (Cnidaria, Scyphozoa)". PLoS ONE. 10 (12) e0145314. Bibcode:2015PLoSO..1045314H. doi:10.1371/journal.pone.0145314. PMC 4687044. PMID 26690755.