Recent from talks
Nothing was collected or created yet.
Perinatal mortality
View on Wikipedia| Perinatal mortality | |
|---|---|
| Other names | Perinatal death |
 | |
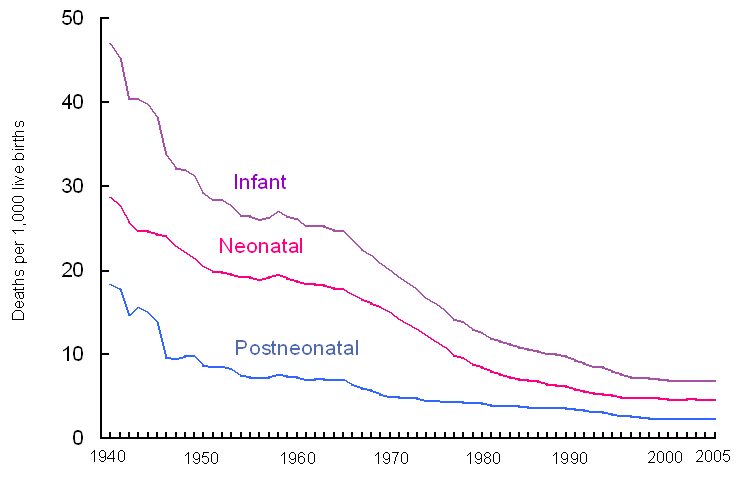
| Infant, neonatal, and postneonatal mortality rates: United States, 1940–2005 | |
| Specialty | Public health |
Perinatal mortality (PNM) is the death of a fetus or neonate and is the basis to calculate the perinatal mortality rate.[1] Perinatal means "relating to the period starting a few weeks before birth and including the birth and a few weeks after birth."[2]
Variations in the precise definition of the perinatal mortality exist, specifically concerning the issue of inclusion or exclusion of early fetal and late neonatal fatalities. The World Health Organization defines perinatal mortality as the "number of stillbirths and deaths in the first week of life per 1,000 total births, the perinatal period commences at 22 completed weeks (154 days) of gestation,[3] and ends seven completed days after birth",[4] but other definitions have been used.[5]
The UK figure is about 8 per 1,000 and varies markedly by social class with the highest rates seen in Asian women. Globally, an estimated 2.6 million neonates died in 2013 before the first month of age down from 4.5 million in 1990.[6]
Causes
[edit]Preterm birth is the most common cause of perinatal mortality, causing almost 30 percent of neonatal deaths.[7] Infant respiratory distress syndrome, in turn, is the leading cause of death in preterm infants, affecting about 1% of newborn infants.[8] Birth defects cause about 21 percent of neonatal death.[7]
Some major causes of perinatal mortality rate is:
- Maternal diseases
- Pelvic diseases; endometriosis, ovarian tumor
- Anatomical defects; Uterine, Cervical anomalies
- Endocrine imbalance
- Blood incompatibilities
- Malnutrition
- Toxemias of pregnancy
- APH
- Congenital defects
- Advanced maternal age
Fetal mortality
[edit]Fetal mortality refers to stillbirths or fetal death.[9] It encompasses any death of a fetus after 20 weeks of gestation or 500 gm. In some definitions of the PNM early fetal mortality (week 20–27 gestation) is not included, and the PNM may only include late fetal death and neonatal death. Fetal death can also be divided into death prior to labor, antenatal (antepartum) death, and death during labor, intranatal (intrapartum) death.
Neonatal mortality
[edit]Neonatal mortality refers to death of a live-born baby within the first 28 days of life. Early neonatal mortality refers to the death of a live-born baby within the first seven days of life, while late neonatal mortality refers to death after 7 days until before 28 days. Some definitions of the PNM include only the early neonatal mortality. Neonatal mortality is affected by the quality of in-hospital care for the neonate. Neonatal mortality and postneonatal mortality (covering the remaining 11 months of the first year of life) are reflected in the infant mortality rate.
Perinatal mortality rate
[edit]| Top ten countries with the highest perinatal mortality rates – 2012[10][11][12] | |||||
|---|---|---|---|---|---|
| Rank | Country | PNMR | Rank | Country | PNMR |
| 1 | 40.7 | 6 | 29.0 | ||
| 2 | 32.7 | 7 | 28.9 | ||
| 3 | 30.8 | 8 | 28.3 | ||
| 4 | 29.7 | 9 | 27.5 | ||
| 5 | 29.4 | 10 | 27.4 | ||
| As per 2014 "Save the Children" report for intrapartum stillbirths and neonatal deaths on first day of birth (per 1,000 total births) | |||||
The PNMR refers to the number of perinatal deaths per 1,000 total births. It is usually reported on an annual basis.[13] It is a major marker to assess the quality of health care delivery. Comparisons between different rates may be hampered by varying definitions, registration bias, and differences in the underlying risks of the populations.
PNMRs vary widely and may be below 10 for certain developed countries and more than 10 times higher in developing countries.[14] The WHO has not published contemporary data.
Effects of neonatal nutrition on neonatal mortality
[edit]Probiotic supplementation of preterm and low birthweight babies during their first month of life can reduce the risk of blood infections, bowel sickness and death in low- and middle-income settings. However, supplementing with Vitamin A does not reduce the risk of death and increases the risk of bulging fontanelle, which may cause brain damage.[15]
See also
[edit]References
[edit]- ^ "Perinatal mortality rate (PMR) — MEASURE Evaluation". www.cpc.unc.edu. 9 September 2020.
- ^ Concise medical dictionary. Jonathan Law, E. A. Martin (10th ed.). Oxford. 2020. ISBN 978-0-19-187376-8. OCLC 1142355883.
{{cite book}}: CS1 maint: location missing publisher (link) CS1 maint: others (link) - ^ "UpToDate".
- ^ "WHO – Maternal and perinatal health". www.who.int. Archived from the original on December 3, 2013.
- ^ Richardus JH, Graafmans WC, Verloove-Vanhorick SP, Mackenbach JP (January 1998). "The perinatal mortality rate as an indicator of quality of care in international comparisons". Medical Care. 36 (1): 54–66. doi:10.1097/00005650-199801000-00007. PMID 9431331.
- ^ "Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013". Lancet. 385 (9963): 117–71. 17 December 2014. doi:10.1016/S0140-6736(14)61682-2. PMC 4340604. PMID 25530442.
- ^ a b March of Dimes / Neonatal Death Archived 2014-10-24 at the Wayback Machine Retrieved on November 10, 2014
- ^ Rodriguez RJ, Martin RJ, and Fanaroff, AA. Respiratory distress syndrome and its management. Fanaroff and Martin (eds.) Neonatal-perinatal medicine: Diseases of the fetus and infant; 7th ed. (2002):1001–1011. St. Louis: Mosby.
- ^ "NVSS – Fetal Deaths". www.cdc.gov. 8 November 2017.
- ^ "Ending Newborn Deaths" (PDF).
- ^ "Million babies die a year – charity – IOL".
- ^ "Nigeria, Pakistan, India lead the world in infant deaths – PM NEWS Nigeria". 25 February 2014.
- ^ "Miscarriage Risk by Week and What Affects Your Risks of Miscarriage". 23 August 2015.
- ^ "Centre for International Health".
- ^ Imdad, Aamer; Rehman, Faseeha; Davis, Evans; Ranjit, Deepika; Surin, Gamael S. S.; Attia, Suzanna L.; Lawler, Sarah; Smith, Abigail A.; Bhutta, Zulfiqar A. (2021). "Effects of neonatal nutrition interventions on neonatal mortality and child health and development outcomes: A systematic review". Campbell Systematic Reviews. 17 (1) e1141. doi:10.1002/cl2.1141. ISSN 1891-1803. PMC 8356300. PMID 37133295. S2CID 240864559.
External links
[edit]Perinatal mortality
View on GrokipediaPerinatal mortality encompasses stillbirths, defined as fetal deaths occurring after 24 weeks of gestation, and early neonatal deaths within the first seven days of life, typically measured as the number of such events per 1,000 total births (live births plus stillbirths).00169-0/fulltext)[1] This metric serves as a fundamental indicator of the quality and accessibility of maternal, fetal, and newborn care systems, highlighting vulnerabilities in pregnancy outcomes from late gestation through the immediate postnatal period.[2] The primary causes of perinatal mortality, grounded in empirical observations from clinical and epidemiological studies, include preterm birth complications, intrapartum asphyxia, and infections, which account for the majority of cases across diverse settings.[3][4] These factors often stem from causal chains involving maternal health conditions, inadequate antenatal monitoring, and limited intrapartum interventions, with prematurity alone implicated in up to 48% of early neonatal deaths in some cohorts.[5] Globally, rates exhibit stark disparities, remaining low in high-income nations—such as approximately 8.4 per 1,000 in the United States in 2023—while elevated in low-resource regions due to systemic gaps in healthcare infrastructure and socioeconomic determinants like poverty and malnutrition.[6][7] Trends indicate gradual declines attributable to advancements in obstetric practices and neonatal resuscitation, yet progress has stagnated in certain areas, underscoring the need for targeted interventions addressing root biological and environmental risks rather than superficial correlates.[8] In resource-constrained environments, underreporting and inconsistent definitions further complicate accurate assessment, emphasizing the importance of standardized, data-driven surveillance for causal analysis and policy formulation.00125-0/fulltext)
Definition and Classification
Core Definitions
Perinatal mortality is defined as the aggregate of stillbirths (fetal deaths occurring after a viability threshold) and early neonatal deaths (live-born infants dying within the first week of life). This metric captures mortality risks clustered around the birth process, reflecting vulnerabilities in late pregnancy, delivery, and immediate postnatal adaptation.[9][10] A stillbirth constitutes the intrauterine death of a fetus at or after 22 completed weeks of gestation (or equivalent fetal weight of approximately 500 grams), prior to complete expulsion or extraction from the mother, irrespective of the duration of pregnancy once viability is reached. Early neonatal death is the demise of a live-born neonate from birth through the first six completed days of life. The perinatal period relevant to these definitions spans from 22 completed weeks of gestation to seven completed days postpartum, encompassing antepartum, intrapartum, and early postnatal phases.[11][12][13] The perinatal mortality rate is computed as the sum of stillbirths and early neonatal deaths divided by the total number of births (live births plus stillbirths of the defined gestational threshold), multiplied by 1,000, yielding deaths per 1,000 births. This denominator accounts for the exposed population at risk, though exact gestational cutoffs for inclusion (e.g., 20 versus 28 weeks) can vary by jurisdiction or reporting standard.[14][1][15]International Variations and Standards
The World Health Organization (WHO) defines perinatal mortality as the aggregate of stillbirths—fetal deaths occurring after 22 completed weeks of gestation or with a birth weight of at least 500 grams—and early neonatal deaths within the first seven days of life, typically expressed as a rate per 1,000 total births (live births plus stillbirths).[11] This standard aims to facilitate global monitoring and comparison, though WHO acknowledges flexibility in using gestational age thresholds of 22 weeks or birth weight criteria when precise data are unavailable.[11] For cause-of-death attribution, WHO endorses the International Classification of Diseases to Perinatal Mortality (ICD-PM), a simplified tool developed in 2015 to standardize coding of perinatal deaths across resource-limited settings, categorizing causes into fetal, maternal, and placental factors while emphasizing avoidable contributors like intrapartum complications.[16] International standards have evolved with the adoption of ICD-11 in 2022, which provides updated guidance for reporting stillbirths and perinatal deaths, recommending gestational age thresholds of at least 22 weeks for stillbirths and integrating perinatal mortality into broader maternal and child health surveillance frameworks to improve data quality and comparability.[12] The International Federation of Gynecology and Obstetrics (FIGO) aligns with this by advocating perinatal mortality rates calculated from stillbirths at or after 28 weeks plus neonatal deaths from day 0 to 6 for births at or after 28 weeks, divided by total such births and multiplied by 1,000, to support clinical audits and policy interventions.[12] These standards prioritize empirical registration of all fetal and neonatal losses meeting criteria, irrespective of birth weight or viability perceptions, to capture causal pathways like asphyxia or infection without underreporting due to cultural or administrative biases.[17] Despite these efforts, definitional variations persist across countries, undermining direct comparisons; for instance, many high-income nations like those in Europe use a 24-week threshold for stillbirth registration, while others, including parts of the United States under CDC Definition I, apply 28 weeks or more, excluding earlier losses and yielding artificially lower rates.[18] In contrast, broader definitions incorporating 20-week fetal deaths (as in some U.S. contexts under Definition III or certain low-resource settings) inflate rates by including spontaneous abortions misclassified as perinatal events, while pragmatic registration practices—such as mandatory reporting only for live births or institutionally delivered cases—further distort data in regions with high home births or incomplete civil registries.[19] These discrepancies, often unadjusted in national statistics, can bias international rankings; a 2017 analysis highlighted how differing inclusion of multiple births, congenital anomalies, or termination-of-pregnancy outcomes leads to perinatal mortality rate variances of up to 50% between ostensibly comparable high-income countries.[18] Efforts to mitigate variations include WHO's Perinatal Death Surveillance and Response initiatives, which promote standardized audits in low- and middle-income countries to link causes to interventions, revealing systemic underreporting where cultural stigma or weak vital registration systems exclude community-level deaths.[20] Peer-reviewed evaluations emphasize that true comparability requires harmonized gestational age cutoffs (ideally 22-28 weeks) and total-birth denominators, as partial live-birth denominators overestimate survival in nations with selective reporting.[21] Ongoing challenges, such as inconsistent application of ICD-PM globally—with adoption limited to about 20 countries as of 2022—underscore the need for causal realism in interpreting rates, prioritizing data from robust registries over survey-based estimates prone to recall bias.[22]Epidemiology
Global Burden and Rates
The global burden of perinatal mortality, encompassing stillbirths (fetal deaths at or after 28 weeks of gestation) and early neonatal deaths (within the first 7 days of life), results in millions of losses annually, predominantly in low- and middle-income countries. In 2023, UNICEF estimated 1.9 million stillbirths worldwide, equivalent to one every 17 seconds and a rate of 14.3 stillbirths per 1,000 total births (live births plus stillbirths).[23] This figure reflects minimal progress, with the stillbirth rate declining only 5.9% since 2000, far slower than reductions in under-5 mortality. Neonatal deaths added 2.3 million cases in 2023, at a rate of 17 per 1,000 live births, accounting for roughly two-thirds of all under-5 deaths and with early neonatal deaths comprising the majority (approximately 75%) due to complications like preterm birth and infections.[24] [25] Combining these components yields an approximate global perinatal mortality rate of 30-32 per 1,000 births, though precise aggregation varies by source owing to differences in early neonatal data capture and denominator definitions (total births versus live births).00125-0/fulltext) Disparities are stark across regions, driven by inequities in healthcare access, with over 80% of perinatal deaths concentrated in sub-Saharan Africa and South Asia. In sub-Saharan Africa, neonatal mortality rates exceed 25 per 1,000 live births, while stillbirth rates surpass 20 per 1,000 total births in some areas; South Asia follows closely with rates around 20-25 for both components. High-income regions, by contrast, report perinatal rates below 5 per 1,000, highlighting the role of systemic factors like poverty, conflict, and inadequate intrapartum care in perpetuating high burdens. These estimates, derived from vital registration, household surveys, and modeling by bodies like the UN Inter-agency Group for Child Mortality Estimation, underscore underreporting in resource-poor settings, where data quality remains a challenge.[26] [23]| Region | Stillbirth Rate (per 1,000 total births, ~2023) | Neonatal Mortality Rate (per 1,000 live births, 2023) |
|---|---|---|
| Sub-Saharan Africa | >20 | 25+ |
| South Asia | 15-20 | 20-25 |
| Global Average | 14.3 | 17 |
| High-Income Countries | <5 | <3 |
Historical and Recent Trends
Perinatal mortality rates have declined substantially in high-income countries over the past century, driven by improvements in antenatal care, obstetric interventions, and neonatal resuscitation techniques. In the United States, for instance, rates fell by approximately 30% from 1990 to 2011, stabilizing thereafter before a modest 4% reduction from 2017 to 2019.[10] By 2021, the U.S. perinatal mortality rate stood at 5.54 per 1,000 live births plus fetal deaths (using a definition including fetal deaths at 20 weeks or more gestation and early neonatal deaths), though broader definitions incorporating earlier gestations yield higher figures around 8 per 1,000.[28] [6] Similar trajectories occurred in other developed nations, where rates dropped from over 40 per 1,000 births in the mid-20th century to single digits by the late 20th century, reflecting causal factors like widespread access to cesarean sections and infection control.[29] Globally, progress has been uneven and slower, with perinatal mortality remaining elevated in low- and middle-income countries due to persistent gaps in healthcare infrastructure and emergency obstetric services. Stillbirth rates, a key component of perinatal mortality, decreased from an estimated 24 per 1,000 total births in 2000 to 13.9 per 1,000 in 2021 (for gestations of 28 weeks or later), equating to about 1.9 million stillbirths annually.[30] [23] Early neonatal mortality, the other primary element, paralleled neonatal trends, declining from roughly 25 per 1,000 live births in 1990 to 17 per 1,000 by 2023, though annual reductions have accelerated post-2000 in most regions.[31] Overall global perinatal rates, combining these, hovered around 30–40 per 1,000 births in the early 2000s and have seen limited further decline, with over 90% of the burden concentrated in resource-limited settings where intrapartum complications predominate.[32] [33] Recent trends indicate stagnation or reversal in some areas amid disruptions like the COVID-19 pandemic, which temporarily elevated rates in high-income countries including the U.S., where perinatal mortality rose nonsignificantly to 8.36 per 1,000 in 2023 from 8.27 in 2022.[6] In developing regions, stillbirth rates have shown minimal improvement since 2012, with annual declines near zero, underscoring failures in scaling interventions like improved monitoring and timely delivery.[34] Despite these challenges, targeted efforts in select low-income countries have yielded reductions, highlighting the potential for causal interventions in high-burden contexts.[35]Geographic and Demographic Variations
Perinatal mortality rates exhibit stark geographic disparities, with low- and lower-middle-income countries bearing the heaviest burden. In high-income countries, rates typically range from 3 to 6 per 1,000 births, reflecting advanced healthcare access, prenatal screening, and obstetric interventions. In contrast, many low-income countries report rates exceeding 40 per 1,000 births, driven by limited antenatal care, infectious diseases, and malnutrition. Sub-Saharan Africa and South Asia account for approximately three-quarters of global stillbirths, which constitute a major component of perinatal deaths; in 2023, sub-Saharan Africa alone contributed nearly 45% of the world's 1.9 million stillbirths occurring at or after 28 weeks' gestation.[23][36] Neonatal mortality, the other key element, follows a similar pattern: regional rates in sub-Saharan Africa reached about 27 per 1,000 live births in recent estimates, compared to 4-5 in high-income regions.[24] Country-level data underscore these regional trends. Nations such as Pakistan, Nigeria, and the Democratic Republic of the Congo consistently rank among the highest, with combined stillbirth and early neonatal rates often surpassing 50 per 1,000 births due to factors like poor infrastructure and high maternal anemia prevalence. For instance, Pakistan's neonatal mortality rate stood at around 42 per 1,000 live births in 2021 data, augmented by stillbirth rates over 20 per 1,000. In Europe and North America, rates are markedly lower; the United States reported a perinatal mortality rate of approximately 5.6 per 1,000 in 2021, though this exceeds peers like Japan or Finland at under 3 per 1,000. These variations persist despite global declines, with progress slowest in fragile states affected by conflict or weak governance.[37][10] Demographic factors further modulate perinatal risks within and across regions. Maternal age shows a U-shaped association: rates are elevated among adolescents under 20 (e.g., 7 per 1,000 in U.S. data for 2021) and women over 35 (rising to 6-8 per 1,000), linked to physiological vulnerabilities like preterm labor or chromosomal anomalies, independent of socioeconomic confounders.[10] By race and ethnicity, non-Hispanic Black women in the United States experience roughly twice the perinatal mortality risk of White women (e.g., infant mortality components at 10-12 per 1,000 vs. 4-5), even after adjusting for education and income, pointing to unmeasured factors such as differential access to quality care or underlying health disparities. Similar patterns emerge globally: African-origin women in Europe face 1.7-fold higher fetal mortality odds compared to native populations.[38][39] Socioeconomic status exhibits an inverse gradient, with higher household income correlating to 20-30% lower perinatal risks in cohort studies, attributable to better nutrition, timely interventions, and environmental exposures. In low-resource settings, rural residence amplifies these effects, often doubling rates relative to urban areas due to distance from facilities. Ethnic minorities in high-income countries, such as South Asian groups, show elevated risks (1.8-fold for stillbirths), potentially tied to consanguinity or cultural delays in seeking care, though data emphasize the interplay of biology and environment over singular causes.[40][39]| Region/Group | Approximate PNMR (per 1,000 births) | Key Source |
|---|---|---|
| High-income countries | 3-6 | CDC/WHO estimates[10] |
| Sub-Saharan Africa | 40-60 | UNICEF stillbirth/neonatal data[23][24] |
| South Asia | 30-50 | Regional aggregates[41] |
| U.S. Black ethnicity | 8-10 (adjusted components) | KFF/CDC[38] |
| Maternal age <20 or >35 | 6-8 | U.S. vital statistics[10] |
Etiology and Pathophysiology
Fetal and Placental Abnormalities
Fetal abnormalities, encompassing congenital malformations and genetic anomalies, represent a significant etiology of perinatal mortality, particularly through mechanisms incompatible with extrauterine life or leading to intrauterine demise. Structural defects such as neural tube defects (e.g., anencephaly and spina bifida), congenital heart anomalies, and chromosomal aberrations (e.g., trisomies 13, 18, and 21) are among the most lethal, often resulting in stillbirth or early neonatal death due to respiratory failure, cardiac insufficiency, or multiorgan dysfunction.[42] In population-based studies, congenital anomalies account for approximately 17-33% of perinatal deaths, with lethal malformations identified in up to 51% of cases within affected cohorts.[43] [44] These anomalies arise primarily from disruptions in embryogenesis, influenced by genetic factors, teratogens, or multifactorial inheritance, culminating in fetal demise when vital organ systems fail to sustain viability.[45] Placental abnormalities contribute to perinatal mortality via impaired maternofetal exchange, leading to chronic hypoxia, acidosis, and growth restriction. Placental insufficiency, characterized by inadequate vascular remodeling of uterine spiral arteries, reduces oxygen and nutrient delivery, often manifesting as fetal growth restriction (FGR) and culminating in stillbirth from hypoxic-ischemic injury.[46] Pathological findings such as infarction, abruption, or villous immaturity are prevalent in 11-84% of stillbirth cases, with uteroplacental vascular lesions directly implicated in fetal death through acute or subacute decompensation.[47] [48] In detailed classifications, placental pathology accounts for about 23% of stillbirths, often coexisting with fetal anomalies or maternal conditions that exacerbate perfusion deficits.[44] The interplay between fetal and placental pathologies amplifies risk; for instance, fetal malformations may secondarily impair placental function through altered vascular demands, while primary placental defects can induce fetal stress responses leading to demise. Autopsy and histopathological evaluations remain essential for attribution, revealing that up to 65% of unexplained stillbirths harbor subtle placental lesions upon scrutiny.[49] Early detection via ultrasonography or biomarkers can mitigate some outcomes, though many lethal anomalies evade antenatal diagnosis.[50]Maternal Physiological and Pathological Factors
Maternal advanced age, typically defined as 35 years or older, is associated with increased perinatal mortality risk, with meta-analyses indicating an odds ratio of 1.58 (95% CI: 1.25-2.00) for women aged 35 and above compared to younger counterparts.[51] This risk escalates further for ages 40 and older, with odds ratios reaching 2.57 (95% CI: 1.57-4.22) for antenatal deaths, attributed to higher incidences of chromosomal anomalies, placental insufficiency, and preterm delivery.[52] Maternal obesity, measured by pre-pregnancy body mass index (BMI) ≥30 kg/m², correlates with elevated infant mortality, showing a pooled odds ratio of 1.42 (95% CI: 1.25-1.62) versus normal BMI, with risks amplifying incrementally per 5-unit BMI increase due to complications like macrosomia, gestational diabetes, and preeclampsia.[53] Grand multiparity (≥5 previous births) also heightens vulnerability, as evidenced in cohort studies where it predicts perinatal loss through mechanisms including uterine atony and exhaustion of reproductive reserves, though exact odds vary by population.[54] Pathological conditions such as hypertensive disorders, particularly preeclampsia, substantially contribute to perinatal mortality, accounting for 1 in 10 to 1 in 4 such deaths globally via placental hypoperfusion, intrauterine growth restriction, and preterm birth.[55] Preeclampsia elevates fetal death risk markedly in preterm gestations, with relative risks exceeding 10-fold in affected cases before 34 weeks, underscoring its role in abruptio placentae and eclampsia.[56] Gestational diabetes mellitus shows inconsistent direct links to stillbirth or neonatal death after glycemic control, with some analyses reporting no significant increase (relative risk 0.75 for diet-controlled cases), though pregestational diabetes doubles severe neonatal morbidity and mortality risks through hyperglycemia-induced fetal macrosomia and metabolic derangements.[57][58] Maternal infections, including HIV and malaria, independently and synergistically amplify perinatal mortality; dual infection raises risks of maternal anemia, low birth weight, and early infant death, with odds ratios for perinatal loss up to 3-4 times higher in co-infected pregnancies due to placental sequestration and immune suppression.[59] In high-burden regions, malaria alone associates with perinatal mortality rates 2-5 times baseline via fetal parasitemia and inflammation, while HIV exacerbates vertical transmission and prematurity.[60] Other pathologies like severe anemia (hemoglobin <7 g/dL) and underlying cardiac disease further compound risks by impairing oxygen delivery to the fetus, though data emphasize treatable causes predominate in low-resource settings.[61] These factors interact with socioeconomic determinants, but causal pathways center on uteroplacental insufficiency and systemic inflammation, as confirmed in histopathological reviews.[62]Intrapartum and Delivery-Related Mechanisms
Intrapartum-related perinatal mortality encompasses fetal deaths occurring during labor and delivery, as well as early neonatal deaths attributable to events in this period, primarily driven by acute interruptions in fetal oxygenation and circulatory compromise. Hypoxia accounts for approximately 78% of intrapartum stillbirths, often resulting from placental insufficiency exacerbated by labor dynamics or mechanical obstructions.00163-4/fulltext) These mechanisms differ from antepartum causes by their temporal proximity to delivery, where dynamic factors like uterine contractions amplify underlying vulnerabilities, leading to rapid decompensation in fetal reserve.[63] A primary mechanism is birth asphyxia, characterized by failure to initiate or sustain effective respiration at birth due to intrapartum hypoxia, contributing to 23-29% of global neonatal deaths and a substantial fraction of perinatal losses in resource-limited settings.[64] [65] Asphyxia arises from prolonged or obstructed labor, where cephalopelvic disproportion or fetal malposition impedes descent, causing sustained compression of the umbilical cord or reduced placental blood flow; for instance, cord prolapse or nuchal cord entanglement can precipitate acute ischemia within minutes.[3] Uterine rupture or abruption during active labor further compromises perfusion, with studies identifying these as key precipitants in up to 36% of unexpected intrapartum fetal deaths when excluding malformations.[66] Delivery trauma represents another critical pathway, particularly in operative births or malpresentations, where excessive force during vacuum extraction, forceps application, or breech delivery inflicts direct injury, such as intracranial hemorrhage or brachial plexus damage, elevating early neonatal mortality risk.[25] Shoulder dystocia, occurring in 0.2-3% of vaginal deliveries, exemplifies this by compressing the umbilical cord against the maternal pelvis, leading to hypoxia and potential neurological devastation if unresolved promptly; unresolved cases correlate with 10-20% perinatal loss rates in affected cohorts.[67] In settings with delayed intervention, secondary infection from prolonged membrane rupture or chorioamnionitis during labor compounds these risks, transitioning mechanical insults into inflammatory cascades that impair neonatal adaptation.[68] Overall, these mechanisms underscore the interplay of fetal physiology with labor physiology, where deviations from optimal progression—often measurable via fetal heart rate monitoring—signal impending demise if unmitigated.[69]Risk Factors
Biological and Demographic Risks
Maternal age at delivery is a key demographic risk factor for perinatal mortality, with elevated rates observed at both extremes. Adolescents under 20 years face higher perinatal mortality rates, approximately 6.97 per 1,000 births in the United States in 2021, compared to 4.98 per 1,000 for mothers aged 30–34, primarily due to increased preterm birth and low birth weight.[10] Advanced maternal age over 35 similarly increases risk, with rates rising progressively; for instance, women over 40 experience heightened early neonatal mortality linked to chromosomal abnormalities and placental insufficiency.[70] A maternal age below 29 or above 40 years correlates with relatively higher early neonatal mortality odds, independent of other confounders.[70] Parity influences perinatal outcomes, where both primiparity and high parity (grand multiparity, typically five or more births) elevate risks. Short interpregnancy intervals under two years are associated with increased perinatal mortality, often through mechanisms like maternal nutrient depletion and uterine overdistension.[71] Primiparous women exhibit higher rates due to immature reproductive physiology and complications such as preeclampsia.[72] Racial and ethnic demographics show persistent disparities in perinatal mortality. Non-Hispanic Black infants in the United States face approximately twice the infant mortality risk compared to White infants, with perinatal components driven by higher preterm delivery and congenital anomalies; this disparity holds across maternal age groups.[73] Black women experience a 3.2-fold increased risk of maternal mortality contributing to perinatal loss, even after adjusting for socioeconomic factors, suggesting underlying biological and systemic elements.[74] American Indian/Alaska Native, Hispanic, and Native Hawaiian/Pacific Islander groups also show elevated neonatal death risks, with Hispanic neonates facing up to three times the odds compared to White neonates in high-income settings.[38][75] Ethnic variations in optimal birth weight for survival further indicate biological differences, as term mortality risks differ by maternal ethnicity beyond gestational age alone.[76] Biological factors include fetal sex, with male infants exhibiting higher neonatal mortality rates across settings, attributed to greater vulnerability to prematurity and asphyxia.[77] Multiple gestations inherently double or triple perinatal mortality risks due to preterm labor and growth restriction.[78] Pre-existing maternal conditions such as hypertension and diabetes, which have biological underpinnings, independently predict perinatal death through placental pathology and macrosomia or intrauterine growth restriction.[72] Congenital anomalies account for a significant portion of cases, remaining a leading cause alongside prematurity.[25]Lifestyle and Behavioral Contributors
Maternal smoking during pregnancy substantially elevates the risk of perinatal mortality, with meta-analyses indicating dose-dependent increases in stillbirth (odds ratio approximately 1.2–1.5), neonatal death, and overall perinatal death rates.[79] [80] Prenatal smoking is linked to a 20–30% higher likelihood of stillbirth and a 40% increase in infant mortality, primarily through mechanisms such as placental insufficiency, preterm birth, and intrauterine growth restriction.[81] Obesity in pregnant women, often stemming from sustained lifestyle patterns of caloric excess and sedentary behavior, is associated with heightened perinatal mortality risks. Obese mothers (BMI ≥30 kg/m²) face a 42% increased odds of infant death compared to normal-weight counterparts, with adjusted perinatal death rates up to 55% higher, attributable to complications like gestational diabetes, preeclampsia, and macrosomia leading to birth trauma.[82] [83] Overweight and obese categories show adjusted odds ratios of 1.22 and higher for perinatal mortality in large cohort studies.[84] Prenatal alcohol exposure contributes to stillbirth risk, with heavy consumption linked to elevated odds independent of other factors, and combined use with smoking tripling late-term stillbirth rates (up to 15 per 1,000 births versus 4 per 1,000 in unexposed pregnancies).[85] [86] Illicit drug use, including opioids and cocaine, correlates with increased perinatal mortality through placental abruption, preterm delivery, and neonatal complications, with exposed infants showing up to 82% higher adjusted hazard ratios for early mortality in population data.[87] [88] Maternal nutritional deficiencies, such as inadequate iron, folate, or overall caloric intake, exacerbate perinatal risks by promoting anemia, fetal growth restriction, and preterm birth, though direct causal links to mortality are mediated by these intermediates and strongest in contexts of severe undernutrition.[89] In contrast, moderate physical activity during pregnancy does not increase perinatal mortality and may mitigate risks from other lifestyle factors by reducing gestational complications, underscoring inactivity as a potential indirect contributor when compounded with obesity or poor diet.[90]Socioeconomic and Environmental Influences
Low socioeconomic status (SES), encompassing factors such as household income, parental education, and wealth, consistently correlates with elevated perinatal mortality rates across diverse populations. In a multinational study involving over 100,000 pregnancies, women classified in low SES categories—based on assets, housing quality, and sanitation access—faced significantly higher risks of stillbirth (adjusted odds ratio approximately 1.5-2.0) and perinatal mortality compared to higher SES counterparts, even after adjusting for obstetric factors.[91] Similarly, in the United States, higher household income levels were linked to lower stillbirth rates (e.g., a 10% increase in income associated with 1-2 fewer stillbirths per 1,000 births) and reduced early neonatal deaths, reflecting improved access to prenatal monitoring and interventions.[40] These patterns persist globally, with poverty during pregnancy independently raising risks for preterm birth, intrauterine growth restriction, and subsequent neonatal death through mechanisms like inadequate nutrition and delayed medical care.[92] Maternal and paternal education levels further mediate these disparities, with lower attainment strongly predicting higher neonatal mortality. For instance, in population-based analyses from low-resource settings, neonates born to mothers with no formal education or only primary schooling experienced 1.5- to 2-fold higher mortality rates than those with secondary or higher education, attributable in part to reduced health literacy and utilization of antenatal services.[93] In regions like Tigray, Ethiopia, children from households in the lowest wealth quintiles—proxied by ownership of durable goods and living standards—had perinatal mortality rates up to 50% higher than wealthier groups, underscoring the role of economic constraints in limiting preventive care.[94] Such inequalities often exhibit a gradient effect, where incremental improvements in SES yield proportional declines in risk, though residual disparities remain after accounting for confounders like maternal age and parity.[95] Environmental exposures, including ambient air pollution and extreme heat, independently contribute to perinatal mortality by impairing fetal development and increasing vulnerability during labor and early neonatal periods. Cohort studies have demonstrated that prenatal exposure to fine particulate matter (PM2.5) from air pollution reduces birth weight by 10-20 grams per 10 μg/m³ increment, heightening risks for stillbirth and neonatal death through placental insufficiency and inflammation.[96] In utero exposure to extreme heat—defined as temperatures exceeding local 95th percentiles—has been associated with a 0.1-0.3% increase in neonatal mortality per degree Celsius rise, as evidenced by analyses of over 55 million U.S. births from 1997-2018, likely via dehydration, preterm labor induction, and cardiorespiratory stress on the fetus.[97] Broader chemical contaminants in air, water, and soil, such as heavy metals and endocrine disruptors, further exacerbate these outcomes by disrupting maternal physiology and fetal growth, with effects persisting across income levels but disproportionately burdening low-SES communities with higher exposure profiles.[98] Climate-driven changes amplifying these exposures pose emerging threats, particularly in tropical and urbanizing regions where adaptive infrastructure lags.[99]Measurement and Surveillance
Rate Calculation Methods
The perinatal mortality rate (PNMR) is calculated as the number of perinatal deaths—comprising late fetal deaths (stillbirths) and early neonatal deaths—divided by the number of total births (live births plus stillbirths), multiplied by 1,000 to yield deaths per 1,000 births.[100][101] This formula standardizes reporting for international comparisons, though exact thresholds for inclusion vary. The World Health Organization (WHO) defines stillbirths as fetal deaths at or after 22 completed weeks of gestation (or ≥500 grams birthweight if gestation is unknown), paired with early neonatal deaths occurring within the first 7 days of life.[102] In the United States, the Centers for Disease Control and Prevention (CDC) employs a broader threshold under Definition III, incorporating fetal deaths at 20 weeks of gestation or more alongside infant deaths under 7 days, with the rate computed per 1,000 live births plus qualifying fetal deaths.[10][103] This approach, updated in vital statistics reporting as of 2025, aims to capture more events but can inflate rates relative to stricter gestational cutoffs like 28 weeks used in some European countries or by the Organisation for Economic Co-operation and Development (OECD).[15][101] Alternative weight-based criteria, such as fetal deaths ≥400 grams (used in some national registries like New Zealand's Perinatal and Maternal Mortality Review Committee), provide flexibility where gestational dating is unreliable, but they risk inconsistencies in low-resource settings.[104] Denominator variations further complicate cross-country analysis: while total births is preferred for inclusivity, some jurisdictions report per live births alone, underestimating rates by excluding stillbirths from the base.[105] These definitional differences, rooted in registration laws and data availability, necessitate caution in global benchmarking, as evidenced by studies showing up to 30% rate disparities attributable to methodological heterogeneity rather than true epidemiological variance.[106][105]Data Collection Challenges and Standardization Efforts
Variations in definitions of perinatal mortality across countries pose significant challenges to data comparability. The World Health Organization (WHO) recommends defining stillbirth as fetal death at or after 28 completed weeks of gestation or weighing 1,000 grams or more, while many high-income countries, including the United States, include fetal deaths from 20 weeks, leading to inflated rates in comparative analyses.[107] [10] Discrepancies in gestational age thresholds for early neonatal deaths and borderline viability cases (e.g., 22-23 weeks) further exacerbate inconsistencies, with reporting practices varying widely; for instance, 18 of 30 European regions showed substantial differences in periviable birth documentation.[108] [109] Underreporting remains prevalent, particularly in low- and middle-income countries, due to incomplete vital registration systems, cultural stigmas around stillbirth disclosure, and resource constraints in humanitarian settings. Household surveys have revealed omission rates and misclassifications in routine facility data, limiting the utility of national statistics for global monitoring.[35] [110] In high-income contexts like the United States, gaps in fetal death certificates, missing maternal demographics, and coding errors contribute to incomplete datasets, hindering trend analysis.[111] To address these issues, the WHO has promoted the International Classification of Diseases, Perinatal Mortality (ICD-PM) extension of ICD-10, providing standardized coding for causes of stillbirths and neonatal deaths to enhance attribution consistency and data interpretation.[112] The transition to ICD-11 includes updated guidance for uniform reporting of perinatal events, emphasizing gestational age and birth weight criteria.[12] Global initiatives, such as Maternal and Perinatal Death Surveillance and Response (MPDSR), encourage facility-based audits and community reporting to identify gaps, though implementation varies by context.[107] In the United States, the National Center for Health Statistics shifted primary perinatal mortality measures in 2021 to align with international standards, incorporating fetal deaths from 20 weeks alongside early neonatal deaths under 7 days.[103] These efforts aim to reduce systematic biases in cross-national comparisons, though persistent local adaptations underscore ongoing needs for harmonized protocols.[109]Prevention and Management Strategies
Antenatal Screening and Interventions
Antenatal screening encompasses routine prenatal evaluations, including ultrasound imaging, maternal serum screening for chromosomal anomalies, and tests for infections, hypertension, and gestational diabetes, aimed at identifying fetal and maternal risks that contribute to perinatal mortality. Systematic reviews of randomized trials indicate that comprehensive antenatal care packages, incorporating evidence-based screenings, can reduce stillbirth and neonatal death rates, with one meta-analysis estimating an 18% relative risk reduction in stillbirth through strategies like infection prevention and nutritional support.[113] However, the impact varies by context; in low- and middle-income settings, adequate antenatal visits—defined as four or more—correlate with a 79% lower likelihood of adverse perinatal outcomes compared to fewer visits, primarily through early detection and management of conditions like anemia and preeclampsia.[114] Ultrasound screening, a cornerstone of antenatal assessment, detects fetal growth restriction, congenital anomalies, and placental issues, but evidence for routine use in low-risk pregnancies is limited regarding perinatal mortality reduction. A 2020 systematic review and meta-analysis of trials involving over 50,000 low-risk women found no significant decrease in perinatal death rates with routine third-trimester ultrasound compared to serial fundal height measurements (risk ratio 0.86, 95% CI 0.28-2.66).[115] Similarly, the 1993 RADIUS trial, involving 15,151 low-risk pregnancies, showed no improvement in perinatal outcomes from screening ultrasound versus selective use based on clinical judgment.[116] In contrast, targeted Doppler ultrasound in high-risk cases, such as suspected growth restriction, identifies fetuses at elevated risk of adverse outcomes, enabling interventions like early delivery, though broad application requires resource-appropriate implementation to avoid overuse without mortality benefits.[117] Infection screening and treatment represent high-impact interventions, particularly in endemic areas. Antenatal testing and penicillin treatment for syphilis prevent up to 82% of associated perinatal deaths, as modeled in Lives Saved Tool analyses incorporating trial data from syphilis-endemic regions.00355-0/abstract) Similarly, screening for malaria and prompt antimalarial therapy in pregnancy reduces stillbirth risk by addressing placental sequestration, with meta-analyses confirming efficacy in sub-Saharan Africa.[113] For gestational diabetes, universal screening via glucose challenge tests followed by metformin or insulin management lowers composite neonatal morbidity and mortality risks, including perinatal death, by mitigating macrosomia and preterm birth. Nutritional and pharmacological interventions triggered by screening further target modifiable risks. Low-dose aspirin (81-150 mg daily) initiated before 16 weeks for women screened at high preeclampsia risk—via history, blood pressure, and biomarkers—reduces preterm birth and stillbirth by 14-20%, per meta-analyses of over 40,000 participants, through improved placental perfusion.[113] Progesterone supplementation, indicated after screening for short cervical length (<25 mm) via transvaginal ultrasound, prevents preterm labor and associated neonatal deaths in singleton pregnancies with prior preterm birth history (relative risk 0.50 for perinatal death).[118] Multiple micronutrient supplementation, recommended following anemia screening, decreases low birth weight and perinatal mortality by 10-12% compared to iron-folic acid alone, based on pooled trial data from diverse settings.00355-0/abstract) Despite these benefits, Cochrane reviews highlight that many antenatal strategies lack robust evidence for direct stillbirth prevention, underscoring the need for context-specific application to avoid ineffective resource allocation.[113]Intrapartum and Perinatal Care Protocols
Intrapartum care protocols prioritize vigilant monitoring of labor progression and fetal status to avert hypoxia, uterine rupture, and other complications implicated in approximately 50% of stillbirths occurring during labor in low- and middle-income countries. The World Health Organization (WHO) endorses the partograph as a simple tool for plotting cervical dilation, fetal heart rate, and maternal vital signs against time, enabling early detection of abnormal labor patterns that necessitate interventions like oxytocin augmentation or operative delivery; implementation has correlated with decreased prolonged labor durations exceeding 12 hours, a known risk for fetal distress.[119] [8] Fetal heart rate (FHR) surveillance constitutes a cornerstone, with intermittent auscultation recommended for low-risk pregnancies every 15-30 minutes in the first stage of labor and more frequently thereafter, while electronic fetal monitoring (EFM) is advised for high-risk cases to identify category II or III tracings signaling potential acidosis or compromise, prompting actions such as maternal repositioning, oxygen administration, or cesarean section. American College of Obstetricians and Gynecologists (ACOG) guidelines emphasize standardized interpretation of FHR patterns, noting that while continuous EFM reduces neonatal seizures by up to 50% in randomized trials, it does not consistently lower overall perinatal mortality or cerebral palsy rates in low-risk cohorts, underscoring the need for adjunctive clinical judgment over reliance on tracing alone.[120] [121] Aseptic techniques, including hand hygiene and perineal disinfection, form essential components to curb intrapartum infections, which contribute to 10-20% of perinatal deaths globally; WHO protocols advocate for delayed umbilical cord clamping by at least 1 minute in vigorous term newborns to enhance placental transfusion and reduce anemia-related mortality risks. In resource-constrained settings, integrated bundles such as Tanzania's Safer Births initiative—encompassing simulation training for vacuum extraction, newborn resuscitation, and essential newborn care—yielded a 32% reduction in intrapartum stillbirths and a 15% drop in early neonatal mortality in cluster-randomized trials conducted through 2023.[119] [122] Perinatal care protocols extend into the immediate postpartum period, focusing on rapid newborn assessment and resuscitation to address birth asphyxia, responsible for 23% of neonatal deaths worldwide. The American Heart Association's 2025 Neonatal Resuscitation guidelines delineate a stepwise algorithm: initial drying and stimulation within 30 seconds for non-vigorous infants, followed by positive pressure ventilation at 40-60 breaths per minute if heart rate remains below 100 bpm after 30 seconds, with chest compressions initiated for rates under 60 bpm; these measures, when protocolized, have reduced asphyxia-specific mortality by 47% in trained cohorts. Programs like Helping Babies Breathe, emphasizing basic bag-mask ventilation training, demonstrated a 30% decline in intrapartum-related neonatal deaths in Nepalese facilities from 2013 to 2015, with sustained effects in subsequent meta-analyses across low-resource sites.[123] [124] [125] Adherence to these evidence-based steps, rather than unstandardized practices, directly correlates with improved Apgar scores and survival, though gaps in skilled attendance persist, contributing to 2.4 million annual perinatal deaths amenable to such interventions.[126]Neonatal Resuscitation and Support
Neonatal resuscitation encompasses immediate interventions to support vital functions in newborns who fail to establish effective respiration and circulation post-delivery, addressing a primary cause of early neonatal deaths within perinatal mortality. Approximately 10% of term newborns require some form of assistance at birth, with asphyxia-related events contributing to up to 30% of neonatal deaths globally, particularly in low-resource settings where basic measures like bag-and-mask ventilation can avert a substantial portion.[127][128] Standard protocols, such as those from the Neonatal Resuscitation Program (NRP) developed by the American Academy of Pediatrics and American Heart Association, emphasize a stepwise approach starting with anticipation based on risk factors like preterm birth or meconium-stained amniotic fluid.[123] Updated guidelines from 2025 highlight the use of 21% oxygen for initial resuscitation in term infants to minimize oxidative stress while ensuring adequate oxygenation.[129] The initial steps involve rapid assessment within the first minute: drying the infant, providing warmth, positioning the head to open the airway, and stimulating crying through gentle rubbing. If the heart rate remains below 100 beats per minute after 30 seconds, positive pressure ventilation (PPV) via a self-inflating bag and mask at 40-60 breaths per minute is initiated, with chest compressions added if the rate falls below 60 beats per minute using a 3:1 ratio coordinated with ventilations.[123] Advanced support includes endotracheal intubation for ineffective PPV, epinephrine administration for persistent bradycardia, and volume expansion for hypovolemia, guided by tools like pulse oximetry or ECG for heart rate monitoring when available.[123] In very low birth weight infants, adherence to these escalated levels has been linked to reduced mortality and morbidities such as bronchopulmonary dysplasia.[130] Training programs like NRP and Helping Babies Breathe have demonstrated effectiveness in improving outcomes; a meta-analysis of neonatal resuscitation trainings reported a 32% relative reduction in perinatal mortality (RR 0.68, 95% CI 0.52-0.88), though effects on overall neonatal mortality were less consistent due to confounding factors like prematurity.[131] Post-training assessments show significant gains in provider knowledge and skills, with hybrid simulations enhancing retention in resource-limited areas.[132] In low-resource settings, World Health Organization guidelines from 2012 prioritize basic resuscitation—focusing on ventilation without advanced equipment—to target the 6 million annual cases needing intervention, potentially preventing 30% of intrapartum-related deaths where skilled attendants are scarce.[133][128] Supportive care following resuscitation includes thermal stabilization using skin-to-skin contact or radiant warmers, glucose monitoring to prevent hypoglycemia, and transfer to neonatal intensive care for ongoing ventilation or surfactant therapy in preterm cases.[123] Effective implementation requires multidisciplinary teams and regular simulations, as performance declines without reinforcement, underscoring the need for sustained training to translate protocols into mortality reductions.[134] Challenges persist in scaling these interventions globally, where facility births with trained providers remain uneven, but evidence supports their causal role in averting asphyxia-driven perinatal losses when prioritized.[135][136]Disparities and Equity Issues
Racial and Ethnic Disparities
In the United States, perinatal mortality rates exhibit marked racial and ethnic disparities, with non-Hispanic Black mothers consistently experiencing the highest rates. For 2022, the CDC reported a rate of 15.05 perinatal deaths per 1,000 live births plus fetal deaths for non-Hispanic Black mothers, more than twice the 6.70 rate for non-Hispanic White mothers. Non-Hispanic Asian mothers had the lowest rate at 5.71 per 1,000, while Hispanic mothers recorded 7.26, non-Hispanic American Indian or Alaska Native mothers 10.92, and non-Hispanic Native Hawaiian or Other Pacific Islander mothers 13.69 per 1,000. These disparities showed no significant change in 2023, except for a 4% increase among Hispanic mothers to 7.57 per 1,000.[6]| Maternal Race/Ethnicity (Non-Hispanic unless noted) | 2022 Rate (per 1,000) | 2023 Rate (per 1,000) |
|---|---|---|
| Black | 15.05 | 15.04 |
| Native Hawaiian/Other Pacific Islander | 13.69 | 14.09 |
| American Indian/Alaska Native | 10.92 | 10.63 |
| Hispanic | 7.26 | 7.57 |
| White | 6.70 | 6.76 |
| Asian | 5.71 | 6.03 |