Recent from talks
Nothing was collected or created yet.
Saiga antelope
View on Wikipedia
| Saiga antelope Temporal range:
| |
|---|---|

| |
| A male at the Stepnoi Nature Sanctuary of Astrakhan Oblast, Russia | |

| |
| A female at the Askania-Nova Biosphere Reserve of Kakhovka Raion, Ukraine | |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Chordata |
| Class: | Mammalia |
| Order: | Artiodactyla |
| Family: | Bovidae |
| Subfamily: | Antilopinae |
| Tribe: | Saigini |
| Genus: | Saiga Gray, 1843 |
| Species: | S. tatarica
|
| Binomial name | |
| Saiga tatarica (Linnaeus, 1766)
| |
| Subspecies | |
| |
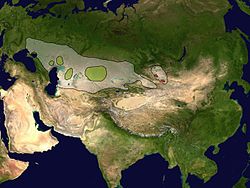
| |
| Reconstructed range (white) and current distribution of the two subspecies Saiga tatarica tatarica (green) and S. t. mongolica (red). | |
| Synonyms[2] | |
|
List
| |
The saiga antelope (/ˈsaɪɡə/, Saiga tatarica), or saiga, is a species of antelope which during antiquity inhabited a vast area of the Eurasian steppe, spanning the foothills of the Carpathian Mountains in the northwest and Caucasus in the southwest into Mongolia in the northeast and Dzungaria in the southeast. During the Pleistocene, it ranged across the mammoth steppe from the British Isles to Beringia. Today, the dominant subspecies (S. t. tatarica) only occurs in Kalmykia and Astrakhan Oblast of Russia and in the Ural, Ustyurt and Betpak-Dala regions of Kazakhstan. A portion of the Ustyurt population migrates south to Uzbekistan and occasionally to Turkmenistan in winter. It is regionally extinct in Romania, Ukraine, Moldova, China and southwestern Mongolia. The Mongolian subspecies (S. t. mongolica) occurs only in western Mongolia.[3][4]
Taxonomy and phylogeny
[edit]The scientific name Capra tatarica was coined by Carl Linnaeus in 1766 in the 12th edition of Systema Naturae.[5] It was reclassified as Saiga tatarica and is the sole living member of the genus Saiga.[6] Two subspecies are recognised:[6][7][1]
- S. t. tatarica (Linnaeus, 1766): also known as the Russian saiga, it is only to be found today in central Asia.
- S. t. mongolica Bannikov, 1946: also known as the Mongolian saiga, it is sometimes treated as an independent species, or as subspecies of the Pleistocene Saiga borealis;[1] it is confined to Mongolia.
In 1945, American paleontologist George Gaylord Simpson classified both in the tribe Saigini under the same subfamily, Caprinae. Subsequent authors were not certain about the relationship between the two, until phylogenetic studies in the 1990s revealed that though morphologically similar, the Tibetan antelope is closer to the Caprinae while the saiga is closer to the Antilopinae.[8]
In a revision of the phylogeny of the tribe Antilopini on the basis of nuclear and mitochondrial data in 2013, Eva Verena Bärmann (of the University of Cambridge) and colleagues showed that the saiga is sister to the clade formed by the springbok (Antidorcas marsupialis) and the gerenuk (Litocranius walleri).[9] The study noted that the saiga and the springbok could be considerably different from the rest of the antilopines; a 2007 phylogenetic study suggested that the two form a clade sister to the gerenuk.[10] The cladogram below is based on the 2013 study.[9]
Evolution
[edit]
Fossils of saiga, concentrated mainly in central and northern Eurasia, date to as early as the late Pleistocene (nearly 0.1 Mya).[11] Several species of extinct Saiga from the Pleistocene of Eurasia and Alaska have been named, including S. borealis,[12] S. prisca, S. binagadensis and S. ricei, although more recent studies suggest that these prehistoric representatives were merely geographical variants of the extant species that was formerly much more widespread.[13] Fossils excavated from the Buran Kaya III site (Crimea) date back to the transition from Pleistocene to Holocene.[14] The morphology of saiga does not seem to have changed significantly since prehistoric times.[2]
Before the Holocene, the saiga ranged across the mammoth steppe from as far west as modern-day England and France to as far east as northern Siberia, Alaska, and probably Canada.[15] The antelope gradually entered the Urals, though it did not colonise southern Europe. A 2010 study revealed that a steep decline has occurred in the genetic variability of the saiga since the late Pleistocene-Holocene, probably due to a population bottleneck.[16]
Characteristics
[edit]
A prominent feature of the saiga is the pair of closely spaced, bloated nostrils directed downward. Other facial features include the proboscis, dark markings on the cheeks and the 7–12 cm (2.8–4.7 in) long ears. Its head-and-body length is typically between 100 and 140 cm (39 and 55 in) with a 6–12 cm (2.4–4.7 in) short tail, and it stands 61–81 cm (24–32 in) at the shoulder. It weighs 26–69 kg (57–152 lb).[2][17]
The coat shows seasonal changes. In summer, the coat appears yellow to red, fading toward the flanks. The Mongolian saiga can develop a sandy colour. The coat develops a pale, grayish-brown colour in winter, with a hint of brown on the belly and the neck. The ventral parts are generally white. The hair is 18–30 mm (0.71–1.18 in) long in summer and can grow up to 40–70 mm (1.6–2.8 in) in winter. This forms a 12 to 15 cm (4.7 to 5.9 in) long mane on the neck. Two distinct moults occur, one in spring from April to May and another in autumn from late September or early October to late November or early December.[2][7]
Only males have horns that are thick and slightly translucent, wax-coloured and show 12 to 20 pronounced rings. With a base diameter of 25–33 mm (0.98–1.30 in), the horns of the Russian saiga measure 28–38 cm (11–15 in) in length; the horns of the Mongolian saiga reach a maximum length of 22 cm (8.7 in).[2][7]
Ecology and behaviour
[edit]Saigas form very large herds that graze in semideserts, steppes, grasslands, and possibly open woodlands, eating several species of plants, including some that are poisonous to other animals. They can cover long distances and swim across rivers, but they avoid steep or rugged areas. The mating season starts in November, when stags fight for the acceptance of females. The winner leads a herd of five to ten, occasionally up to 50 females.[2] In springtime, mothers come together in mass to give birth.[18]


Saigas, like the Mongolian gazelles, are known for their extensive migrations across the steppes that allow them to escape natural calamities.[19] Saigas are highly vulnerable to wolves. Juveniles are targeted by foxes, steppe eagles, golden eagles and ravens.[2]
Distribution and habitat
[edit]In the mid-2010s, the populations declined enormously – as much as 95% in 15 years.[20] This led the saiga to be classified as critically endangered on the IUCN Red List. In more recent years, the saiga has experienced massive regrowth. As of 2022, there is an estimated number of 1.38 million saiga surviving in Kazakhstan, per an April aerial count.[21] As of December 2023, the global saiga antelope population is estimated to number 922,600–988,500 mature individuals.[1]
In May 2010, an estimated 12,000 of the 26,000 saiga population in the Ural region of Kazakhstan were found dead. Although the deaths are currently being ascribed to pasteurellosis, an infectious disease that strikes the lungs and intestines, the underlying trigger remains to be identified.[22] In May 2015, what may be the same disease broke out in three northern regions of the country.[23] As of 28 May 2015, more than 120,000 saigas had been confirmed dead in the Betpak-Dala population in central Kazakhstan, representing more than a third of the global population.[24] By April 2016, the saigas appeared to be making a comeback, with an increase of population from 31,000 to 36,000 in the Betpak-Dala area.[25] In April 2021 a survey in Kazakhstan found that the saiga population had risen from an estimated 334,000 to 842,000. The population increase was partially attributed to the government crackdown on poaching and the establishment of conservation areas.[26] UK charity RSPB reported in 2022 that, partly due to their conservation efforts, as well as the designation of the Bokey Orda-Ashiozek protected area by the Kazakhstan government, the population had now risen to a peak of 1.32 million.[27]
Former range
[edit]The saiga was not present in Europe during the Eemian.[28] During the last glacial period, it ranged from the British Isles through Central Asia and the Bering Strait into Alaska and Canada's Yukon and Northwest Territories. By the classical age, they were apparently considered a characteristic animal of Scythia, judging from the historian Strabo's description of an animal called the kolos that was "between the deer and ram in size" and was wrongly believed to drink through its nose.[29]
Considerable evidence shows the importance of the antelope to Andronovo culture settlements. Illustrations of saiga antelopes can be found among the cave paintings that were dated back to seventh to fifth century BC. Moreover, saiga bones were found among the remains of other wild animals near the human settlements.[30]
The fragmented information shows an abundance of saigas on the territory of modern Kazakhstan in the 14th-16th centuries. The migratory routes ranged throughout the country's area, especially the region between the Volga and Ural Rivers was heavily populated.[31] The population's size remained high until the second half of the 19th century, when excessive horn export began. The high price and demand for horns drove radical hunting. The number of animals decreased in all regions and the migratory routes shifted southward.[32] Populations in Ukraine were driven to extirpation in the 18th century.[1]
After a rapid decline, they were nearly completely exterminated in the 1920s, but they were able to recover. By 1950, two million of them were found in the steppes of the USSR. Their population fell drastically following the collapse of the USSR due to uncontrolled hunting and demand for horns in Chinese medicine. At one point, some conservation groups, such as the World Wildlife Fund, encouraged the hunting of this species, as its horn was presented as an alternative to that of a rhinoceros.[33]
Mongolian saiga
[edit]The Mongolian saiga (S. t. mongolica) is found in a small area in western Mongolia around the Sharga and Mankhan Nature Reserves.[34]
Threats
[edit]

The horn of the saiga antelope is used in traditional Chinese medicine and can sell for as much as US$150. Demand for the horns drives poaching and smuggling, which has wiped out the population in China, where the saiga antelope is a class I protected species.[35]
In June 2014, Chinese customs at the Kazakh border uncovered 66 cases containing 2,351 saiga antelope horns, estimated to be worth over Y70.5 million (US$11 million).[36] In June 2015, E. J. Milner-Gulland (chair of Saiga Conservation Alliance) said: "Antipoaching needs to be a top priority for the Russian and Kazakh governments."[18]
Hunting
[edit]Saigas have been a target of hunting since prehistoric ages, when hunting was an essential means to acquire food. Saigas' horns, meat, and skin have commercial value and are exported from Kazakhstan.
Saiga horn, known as Cornu Antelopis, is one of the main ingredients in traditional Chinese medicine that is used as an extract or powder additive to the elixirs, ointments, and drinks. Saiga horn's value is equal to rhinoceros horn, whose trade was banned in 1993. Cornu Antelopis is thought to be a cheaper substitute of rare rhino horn in most TCM recipes.[37]
In the period from 1955 to 1989, over 87 thousand tonnes of meat were collected in Kazakhstan by killing more than five million saiga.[38] In 2011, Kazakhstan reaffirmed a ban on hunting saiga and extended this ban until 2021.[39] Hunting of the species was allowed in Kazakhstan in 2025 due to concerns over damage to crops and rapid population growth.[40]
Saiga meat is compared to lamb, considered to be nutritious and delicious. Numerous recipes for cooking the antelope's meat can be found.[41] Both meat and byproducts are sold in the country and outside of it. About 45–80 dm2 of skin can be harvested from one individual depending on its age and sex.[32]
Physical barriers
[edit]Agricultural advancement and human settlements have been shrinking habitat areas of the saigas since the 20th century.[32] Occupants limited saiga's passage to water resources and the winter and summer habitats. The ever-changing face of steppe requires saigas to search for new routes to their habitual lands. Currently, saiga populations' migratory routes pass five countries and different human-made constructions, such as railways, trenches, mining sites, and pipelines.[32] These physical barriers limit movement of the antelopes. Cases of saiga herds being trapped within fenced areas and starving to death have been reported.[42]
Climatic variability
[edit]Saigas are dependent on weather and affected by climate fluctuations to a great extent due to their migratory nature.[43] Harsh winters with strong winds or high snow coverage prevent them from feeding on the underlying grass. Population size usually dramatically decreases after severe cold months.[32] Recent trends in climate change have increased the aridity of the steppe region, leading an estimated 14% or more of available pastureland to be considered degraded and useless.[44] Concurrently, small steppe rivers dry faster, limiting water resources to large lakes and rivers, which are usually populated by human settlements; high temperatures in the steppe region lead to springtime floods, in which saiga calves can drown.[32]
Mass epizootic mortality
[edit]1980 to 2015 events
[edit]For ungulates, mass mortalities are not uncommon. In the 1980s, several saiga die-offs occurred, and between 2010 and 2014, one occurred every year. The deaths could be linked to calving aggregation, which is when they are most vulnerable.[18] More recent research involving a mass die-off in 2015 indicates warmer weather and attendant humidity led bacteria common in saiga antelopes to move into the bloodstream and cause hemorrhagic septicemia.[45]
2015–2016 epizootic
[edit]In May 2015, uncommonly large numbers of saigas began to die from a mysterious epizootic illness suspected to be pasteurellosis.[18][46] Herd fatality is 100% once infected, with an estimated 40% of the species' total population already dead.[47] More than 120,000 carcasses had been found by late May 2015, while the estimated total population was only 250,000.[48]
Biologist Murat Nurushev suggested that the cause might be acute ruminal tympany, whose symptoms (bloating, mouth foaming, and diarrhea) had been observed in dead saiga antelopes.[49] According to Nurushev, this disease occurred as a result of foraging on a large amount of easily fermenting plants (alfalfa, clover, sainfoins, and mixed wet, green grass).[49] In May 2015, the United Nations agency which is involved in saiga conservation efforts issued a statement that the mass die-off had ended.[citation needed] By June 2015, no definitive cause for the epizootic had been found.[50]
At a scientific meeting in November 2015 in Tashkent, Uzbekistan, Dr. Richard A. Kock (of the Royal Veterinary College in London) reported that his colleagues and he had narrowed down the possible culprits. Climate change and stormy spring weather, they said, may have transformed harmless bacteria, carried by the saigas, into lethal pathogens.[51]
Pasteurella multocida, a bacterium, was determined to be the cause of death. The bacterium occurs in the antelopes and is normally harmless; the reason for the change in behavior of the bacterium is unknown.[52]
Now, scientists and researchers believe the unusually warm and wet uncontrolled environmental variables caused the bacterium to enter the bloodstream and become septic. Hemorrhagic septicemia is the likely cause of the most recent deaths[53] The change of the bacteria may be attributed to "the response of opportunistic microbes to changing environmental conditions".[54]
The Betpak-Dala saiga population in central Kazakhstan, which saw the most deaths, increased from 31,000 after the epidemic to 36,000 by April 2016.[52]
In late 2016, a large loss of the population happened in Mongolia. The etiology was confirmed to be goat plague in early 2017.[55]
Conservation
[edit]Under the auspices of the Convention on the Conservation of Migratory Species of Wild Animals, the Saiga Antelope Memorandum of Understanding was concluded and came into effect on 24 September 2006.[56]
In captivity
[edit]Currently, only the Almaty Zoo and Askania-Nova keep saigas.[57]
References
[edit]- ^ a b c d e f IUCN SSC Antelope Specialist Group (2023). "Saiga tatarica". IUCN Red List of Threatened Species. 2023 e.T19832A233712210. Retrieved 11 December 2023.
- ^ a b c d e f g Sokolov, V.E. (1974). "Saiga tatarica" (PDF). Mammalian Species (38): 1–4. doi:10.2307/3503906. JSTOR 3503906. Archived from the original (PDF) on 4 March 2016.
- ^ Neronov, V. M.; Arylova, N. Yu.; Dubinin, M. Yu.; Karimova, T. Yu.; Lushchekina, A. A. (2013). "Current state and prospects of preserving saiga antelope in Northwest Pre-Caspian region". Arid Ecosystems. 3 (2): 57–64. Bibcode:2013ArEco...3...57N. doi:10.1134/S2079096113020078. S2CID 18451816.
- ^ "Saiga/mongolian Saiga (Saiga tatarica)". Evolutionarily Distinct and Globally Endangered. Zoological Society of London. Archived from the original on 30 May 2008. Retrieved 19 December 2012.
- ^ Linnaeus, C. (1766). "Capra tatarica". Systema Naturae: per Regna tria Natura, secundum Classes, Ordines, Genera, Species, cum Characteribus, Differentiis, Synonymis, Locis. Vol. t.1, pt. 1 (Regnum animale) (1766) (12th ed.). Impensis direct. Laurentii Salvii. p. 97.
- ^ a b Grubb, P. (2005). "Species Saiga tatarica". In Wilson, D.E.; Reeder, D.M (eds.). Mammal Species of the World: A Taxonomic and Geographic Reference (3rd ed.). Johns Hopkins University Press. p. 688. ISBN 978-0-8018-8221-0. OCLC 62265494.
- ^ a b c Groves, C.; Grubb, P. (2011). Ungulate Taxonomy. Baltimore, USA: Johns Hopkins University Press. p. 157. ISBN 978-1-4214-0093-8.
- ^ Schaller, G.B. (1998). Wildlife of the Tibetan steppe. Chicago, USA: University of Chicago Press. pp. 254–5. ISBN 978-0-226-73652-5.
- ^ a b Bärmann, E.V.; Rössner, G.E.; Wörheide, G. (2013). "A revised phylogeny of Antilopini (Bovidae, Artiodactyla) using combined mitochondrial and nuclear genes". Molecular Phylogenetics and Evolution. 67 (2): 484–93. Bibcode:2013MolPE..67..484B. doi:10.1016/j.ympev.2013.02.015. PMID 23485920.
- ^ Marcot, J.D. (2007). "Molecular phylogeny of terrestrial artiodactyls". In Prothero, D.R.; Foss, S.E. (eds.). The Evolution of Artiodactyls (Illustrated ed.). Baltimore, USA: Johns Hopkins University Press. pp. 4–18. ISBN 978-0-8018-8735-2.
- ^ Currant, A.P. (1987). "Late Pleistocene Saiga antelope Saiga tatarica on Mendip" (PDF). UBSS Proceedings. 18 (1): 74–80.
- ^ Baryshnikov, G.; Tikhonov, A. (1994). "Notes on skulls of Pleistocene Saiga of Northern Eurasia". Historical Biology. 8 (1–4): 209–34. Bibcode:1994HBio....8..209B. doi:10.1080/10292389409380478.
- ^ Ratajczak, U. (2016). "Quaternary skulls of the saiga antelope from Eastern Europe and Siberia: Saiga borealis versus Saiga tatarica — One species or two?" (PDF). Quaternary International. 420 (28): 329–347. Bibcode:2016QuInt.420..329R. doi:10.1016/j.quaint.2015.09.040. S2CID 131699605.
- ^ Lanoë, F.B.; Péan, S.; Yanevich, A. (2015). "Saiga antelope hunting in Crimea at the Pleistocene–Holocene transition: the site of Buran-Kaya III Layer 4". Journal of Archaeological Science. 54: 270–8. Bibcode:2015JArSc..54..270L. doi:10.1016/j.jas.2014.12.012.
- ^ Jürgensen, Jonathan; Drucker, Dorothée G.; Stuart, Anthony J.; Schneider, Matthias; Buuveibaatar, Bayarbaatar; Bocherens, Hervé (March 2017). "Diet and habitat of the saiga antelope during the late Quaternary using stable carbon and nitrogen isotope ratios". Quaternary Science Reviews. 160: 150–161. Bibcode:2017QSRv..160..150J. doi:10.1016/j.quascirev.2017.01.022.
- ^ Campos, P.F.; Kristensen, T.; Orlando, L.; Sher, A.; Kholodova, M.V.; Gotherstrom, A.; Hofreiter, M.; Drucker, D.G.; Kosintsev, P.; Tikhonov, A.; Baryshnikov, G.F.; Willerslev, E.; Gilbert, M.T.P. (2010). "Ancient DNA sequences point to a large loss of mitochondrial genetic diversity in the saiga antelope (Saiga tatarica) since the Pleistocene" (PDF). Molecular Ecology. 19 (22): 4863–75. Bibcode:2010MolEc..19.4863C. doi:10.1111/j.1365-294X.2010.04826.x. PMID 20874761. S2CID 205363165.
- ^ Burton, M.; Burton, R. (2002). International Wildlife Encyclopedia (Third ed.). New York: Marshall Cavendish. pp. 2217–2218. ISBN 978-0-7614-7282-7.
- ^ a b c d Nicholls, Henry (12 June 2015). "134,000 saiga antelope dead in two weeks. What is the probable cause?". The Guardian.
- ^ Owen-Smith, N. (2008). "The comparative population dynamics of browsing and grazing ungulates". In Gordon, I.J.; Prins, H.H.T. (eds.). The ecology of browsing and grazing. Berlin, Germany: Springer. p. 163. ISBN 978-3-540-72422-3.
- ^ "Welcome to the Saiga Conservation Alliance". Saiga Conservation Alliance. Archived from the original on 22 May 2013. Retrieved 19 December 2012.
- ^ "Rare saiga antelope population now over a million in Kazakhstan".
- ^ "Mystery over mass antelope deaths in Kazakhstan". BBC News. 2010. Retrieved 19 December 2012.
- ^ "Saiga Antelope Death Toll in Kazakhstan Reaches 85,000". Radio Free Europe/Radio Liberty. 22 May 2015. Retrieved 25 May 2015.
- ^ "Catastrophic Collapse of Saiga Antelopes in Central Asia". United Nations Environment Programme. 2015. Archived from the original on 4 March 2016. Retrieved 5 August 2015.
- ^ Yin, S. (2016). "Saiga Population Grows After Mysterious Epidemic". The New York Times. Retrieved 19 April 2018.
- ^ Briggs, Helen (2021). "Critically endangered antelope saiga makes comeback". BBC News. Retrieved 4 July 2021.
- ^ "Endangered antelope numbers bounce back from near extinction".
- ^ Nadachowski, A.; Lipecki, G.; Ratajczak, Urszula; Stefaniak, K.; Wojtal, P. (2016). "Dispersal events of the saiga antelope (Saiga tatarica) in Central Europe in response to the climatic fluctuations in MIS 2 and the early part of MIS 1". Quaternary International. 420: 357–362. Bibcode:2016QuInt.420..357N. doi:10.1016/j.quaint.2015.11.068.
- ^ Strabo (25 September 2012). "Book VII, Chapter 4, Paragraph 8". Geography. Retrieved 19 December 2012.
- ^ Маргулан, А.X.; Акишев, К.А.; Кадырбаев, М.К.; Оразбаев, А.М. (1966). Древняя культура Центрального Казахстана. Алма-Ата, КазССР: Наука. pp. 11, 238.
- ^ В. А. Фадеев; А. А. Слудский (1982). Сайгак в Казахстане. Алма-Ата, КазССР: Наука.
- ^ a b c d e f Bekenov, A. B.; Grachev, Iu. A.; Milner-Gulland, E. J. (1998). "The ecology and management of the Saiga antelope in Kazakhstan". Mammal Review. 28 (1): 1–52. Bibcode:1998MamRv..28....1B. doi:10.1046/j.1365-2907.1998.281024.x.
- ^ Ellis, R. (2004). No Turning Back: The Life and Death of Animal Species. New York: Harper Perennial. p. 210. ISBN 978-0-06-055804-8. Retrieved 19 December 2012.
- ^ Mallon, David P.; Kingswood, Steven Charles (2001). Antelopes: Part 4 – North Africa, the Middle East, and Asia: Global Survey and Regional Action Plans. International Union for Conservation of Nature. p. 164. ISBN 978-2-8317-0594-1. Retrieved 19 December 2012.
- ^ van Uhm, D. P. (2016). The Illegal Wildlife Trade: Inside the World of Poachers, Smugglers and Traders (Studies of Organized Crime). New York: Springer.
- ^ 新疆霍尔果斯海关破获一起羚羊角走私案 天山网 (Chinese) 23 June 2014
- ^ "Saiga Antelope". programs.wcs.org. Retrieved 5 May 2016.
- ^ Milner-Gulland, E. J. (1994). "A Population Model for the Management of the Saiga Antelope". Journal of Applied Ecology. 31 (1): 25–39. Bibcode:1994JApEc..31...25M. doi:10.2307/2404596. JSTOR 2404596.
- ^ "Kazakhstan extends Saiga antelope hunting ban until 2021". Silk Road Intelligencer. 2011. Archived from the original on 29 July 2011. Retrieved 19 December 2012.
- ^ "Kazakhstan to allow hunting once endangered antelopes". France 24. 28 May 2025. Retrieved 28 May 2025.
- ^ "ОХОТНИЧЬЯ КУХНЯ. САЙГАК. Рецепты БЛЮД ИЗ САЙГАКА". Supercook. Archived from the original on 21 November 2014. Retrieved 5 May 2016.
- ^ "Казахстан обвинили в дезориентации сайгаков". lenta.ru. Retrieved 5 May 2016.
- ^ Milner-Gulland; Kholodova; Bekenov; Bukreeva; Grachev; Amgalan; Lushchekina (2001). "Dramatic declines in saiga antelope populations". Oryx. 35 (4): 340–345. doi:10.1046/j.1365-3008.2001.00202.x.
- ^ Saparov, Abdulla (2014). "Soil Resources of the Republic of Kazakhstan: Current Status, Problems and Solutions". In Mueller, L.; Saparov, A.; Lischeid, G. (eds.). Novel Measurement and Assessment Tools for Monitoring and Management of Land and Water Resources in Agricultural Landscapes of Central Asia. Environmental Science and Engineering. Springer International Publishing. pp. 61–73. doi:10.1007/978-3-319-01017-5_2. ISBN 978-3-319-01016-8. S2CID 55229757.
- ^ Yong, Ed (2018). "Why Did Two-Thirds of These Weird Antelope Suddenly Drop Dead?". The Atlantic.
- ^ "Endangered saiga antelope mysteriously dying in vast numbers in Kazakhstan". The Independent. Associated Press. 25 May 2015.
- ^ "Mass deaths hit Kazakhstan's endangered Ice Age antelope species". Reuters. 27 May 2015.
- ^ Taylor, Adam (29 May 2015). "Kazakhstan's econological mystery". The Washington Post. Worldviews.
- ^ a b "Свою версию гибели сайгаков выдвинул академик Нурушев". Informburo.kz (in Russian). 24 May 2015. Retrieved 1 June 2015.
- ^ Saiga mystery disease; 29 May 2015
- ^ Zimmer, C. (2015). "More Than Half of Entire Species of Saigas Gone in Mysterious Die-Off". The New York Times. Retrieved 3 February 2018.
- ^ a b Yin, Steph (2016). "Saiga Population Grows After Mysterious Epidemic". The New York Times. Retrieved 26 June 2016.
- ^ "Why 220,000 saiga antelope died suddenly in Kazakhstan in 2015 - CBC Radio". cbc.ca. Retrieved 19 April 2018.
- ^ Kock, R. A.; Orynbayev, M.; Robinson, S.; Zuther, S.; Singh, N. J.; Beauvais, W.; Morgan, E. R.; Kerimbayev, A.; Khomenko, S.; Martineau, H. M.; Rystaeva, R.; Omarova, Z.; Wolfs, S.; Hawotte, F.; Radoux, J.; Milner-Gulland, E. J. (2018). "Saigas on the brink: Multidisciplinary analysis of the factors influencing mass mortality events". Science Advances. 4 (1) eaao2314. Bibcode:2018SciA....4.2314K. doi:10.1126/sciadv.aao2314. PMC 5777396. PMID 29376120.
- ^ "Peste Des Petits Ruminants – Mongolia (03): (Hovd) Saiga Antelope". ProMED-mail. 2017. Retrieved 9 March 2017.
- ^ "Memorandum of Understanding concerning Conservation, Restoration and Sustainable Use of the Saiga Antelope (Saiga spp)" (PDF). Convention on Migratory Species. 2011. Retrieved 19 December 2012.[permanent dead link]
- ^ "Western saiga (Russian saiga)". Zootierliste. Retrieved 28 November 2021.
Further reading
[edit]- Singh, N.J.; Milner-Gulland, E.J. (2011). "Conserving a moving target: planning protection for a migratory species as its distribution changes" (PDF). Journal of Applied Ecology. 48 (1): 35–46. Bibcode:2011JApEc..48...35S. doi:10.1111/j.1365-2664.2010.01905.x.[permanent dead link]
- Singh, N.J.; Grachev, Iu.A.; Bekenov, A.B.; Milner-Gulland, E.J. (2010). "Tracking greenery in Central Asia: The migration of the saiga antelope". Diversity and Distributions. 16 (4): 663–675. doi:10.1111/j.1472-4642.2010.00671.x. S2CID 83904273.
- Singh, N.J.; Grachev, Iu.A.; Bekenov, A.B.; Milner-Gulland, E.J. (2010). "Saiga antelope calving site selection is increasingly driven by human disturbance". Biological Conservation. 143 (7): 1770–1779. Bibcode:2010BCons.143.1770S. doi:10.1016/j.biocon.2010.04.026.
- Kuhl, A.; Mysterud, A.; Grachev, Iu.A.; Bekenov, A.B.; Ubushaev, B.S.; Lushchekina, A.A.; Milner-Gulland, E.J. (2009). "Monitoring population productivity in the saiga antelope". Animal Conservation. 12 (4): 355–363. Bibcode:2009AnCon..12..355K. doi:10.1111/j.1469-1795.2009.00260.x. S2CID 86008409.
- Kuhl, A.; Balinova, N.; Bykova, E.; Esipov, A.; Arylov, Iu.A.; Lushchekina, A.A.; Milner-Gulland, E.J. (2009). "The role of saiga poaching in rural communities: Linkages between attitudes, socio-economic circumstances and behaviour". Biological Conservation. 142 (7): 1442–1449. Bibcode:2009BCons.142.1442K. doi:10.1016/j.biocon.2009.02.009.
- Kuhl, A.; Mysterud, A.; Erdnenov, G.I.; Lushchekina, A.A.; Grachev, Iu. A.; Bekenov, A.B.; Milner-Gulland, E.J. (2007). "The big spenders of the steppe: sex-specific maternal allocation and twinning in the saiga antelope". Proceedings of the Royal Society B. 274 (1615): 1293–1299. doi:10.1098/rspb.2007.0038. PMC 2176182. PMID 17341456..
- Morgan, E.R.; Medley, G.F.; Torgerson, P.R.; Shaikenov, B. & Milner-Gulland, E.J. (2007). "Parasite transmission in a migratory multiple host system". Ecological Modelling. 200 (3–4): 511–520. Bibcode:2007EcMod.200..511M. doi:10.1016/j.ecolmodel.2006.09.002.
- Kholodova, M.V.; Milner-Gulland, E.J.; Easton, A.J.; Amgalan, L.; Arylov, Iu.; Bekenov, A.; Grachev, Iu.A.; Lushchekina, A.A.; Ryder, O. (2006). "Mitochondrial DNA variation and population structure of the Critically Endangered saiga antelope Saiga tatarica". Oryx. 40: 103–107. doi:10.1017/S0030605306000135.
- Morgan, E.R.; Lundervold, M.; Medley, G.F.; Shaikenov, B.S.; Torgerson, P.R.; Milner-Gulland, E.J. (2006). "Assessing risks of disease transmission between wildlife and livestock: the Saiga antelope as a case study". Biological Conservation. 131 (2): 244–254. Bibcode:2006BCons.131..244M. doi:10.1016/j.biocon.2006.04.012.
- Morgan, E.R.; Shaikenov, B.; Torgerson, P.R.; Medley, G.F.; Milner-Gulland, E.J. (2005). "Helminths of saiga antelopes in Kazakhstan: Implications for conservation and livestock production". Journal of Wildlife Diseases. 41 (1): 149–162. doi:10.7589/0090-3558-41.1.149. PMID 15827221. S2CID 22806238.
- Milner-Gulland, E.J.; Bukreeva, O.M.; Coulson, T.N.; Lushchekina, A.A.; Kholodova, M.V.; Bekenov, A.B.; Grachev, Iu.A. (2003). "Reproductive collapse in saiga antelope harems". Nature. 422 (6928): 135. doi:10.1038/422135a. PMID 12634775. S2CID 4409236.
- Robinson, S.; Milner-Gulland, E.J. (2003). "Political change and factors limiting numbers of wild and domestic ungulates in Kazakhstan". Human Ecology. 31 (1): 87–110. Bibcode:2003HumEc..31...87R. doi:10.1023/A:1022834224257. S2CID 67810286.
- Milner-Gulland, E.J.; Kholodova, M.V.; Bekenov, A.B.; Bukreeva, O.M.; Grachev, Iu.A.; Amgalan, L.; Lushchekina, A.A. (2001). "Dramatic declines in saiga antelope populations". Oryx. 35 (4): 340–345. doi:10.1046/j.1365-3008.2001.00202.x.
External links
[edit]- CMS Saiga Memorandum of Understanding
- Ultimate Ungulate
- WWF species profile: Saiga antelope
- "IFAW Russia – Saiga antelopes are rare creatures". YouTube. 3 July 2012. Archived from the original on 21 December 2021.


