Recent from talks
Nothing was collected or created yet.
Necrotizing gingivitis
View on Wikipedia| Necrotizing gingivitis | |
|---|---|
| Other names | NG, Trench mouth |
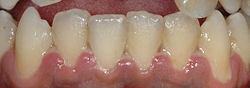 | |
| A fairly mild presentation of necrotizing gingivitis at the typical site on the gums of the anterior mandibular teeth. | |
| Specialty | Periodontology |
| Symptoms | pain, bad breath, fever, bleeding gums |
| Complications | Progression |
| Causes | bacterial infection by Prevotella intermedia, Treponema, Selenomonas, Fusobacterium spp, Spirochetes and compromised host immune response |
| Risk factors | HIV/AIDS, malnutrition, psychological stress, sleep deprivation, inadequate oral hygiene, pre-existing gingivitis, history of necrotizing periodontal disease, tobacco and alcohol use, youth, white ethnicity, orthodontics |
| Diagnostic method | Based on clinical findings (necrosis, ulcer of interdental papilla; gingival bleeding, pain, pseudomembrane formation, halitosis) |
| Differential diagnosis | herpetic gingivostomatitis, mucous membrane pemphigoid, pemphigus vulgaris, toothbrush abrasion |
| Prevention | oral hygiene |
| Treatment | debridement (dental), metronidazole |
Necrotizing gingivitis (NG) is a common, non-contagious infection of the gums with sudden onset. The main features are painful, bleeding gums, and ulceration of interdental papillae (the sections of gum between adjacent teeth). This disease, along with necrotizing periodontitis (NP) and necrotizing stomatitis, is classified as a necrotizing periodontal disease, one of the three general types of gum disease caused by inflammation of the gums (periodontitis).[1]
The often severe gum pain that characterizes NG distinguishes it from the more common gingivitis or chronic periodontitis which is rarely painful. If NG is improperly treated or neglected, it may become chronic and/or recurrent. The causative organisms are mostly anaerobic bacteria, particularly Fusobacteriota and spirochete species.
Predisposing factors include poor oral hygiene, smoking, poor nutrition, psychological stress, and a weakened immune system. When the attachments of the teeth to the bone are involved, the term NP is used. Treatment of NG is by removal of dead gum tissue and antibiotics (usually metronidazole) in the acute phase, and improving oral hygiene to prevent recurrence. Although the condition has a rapid onset and is debilitating, it usually resolves quickly and does no serious harm. The informal name trench mouth arose during World War I as many soldiers developed the disease, probably because of the poor conditions and extreme psychological stress.
Signs and symptoms
[edit]In the early stages some affected people may complain of a feeling of tightness around the teeth.[2] Three signs/symptoms must be present to diagnose this condition:[2]
- Severe gum pain.[3]
- Profuse gum bleeding that requires little or no provocation.[2]
- Interdental papillae are ulcerated with dead tissue.[3] The papillary necrosis of NG has been described as "punched out".[2]
Other signs and symptoms may be present, but not always.[2]
- Foul breath.
- Bad taste (metallic taste).[3]
Malaise, fever and/or cervical lymph node enlargement are rare (unlike the typical features of herpetic stomatitis).[3] Pain is fairly well localized to the affected areas.[3] Systemic reactions may be more pronounced in children.[2] Cancrum oris (noma) is a very rare complication, usually in debilitated children.[3] Similar features but with more intense pain may be seen in necrotizing periodontitis in HIV/AIDS.[3]
Causes
[edit]Necrotizing periodontal diseases are caused by a mixed bacterial infection that includes anaerobes such as P. intermedia[1] and Fusobacterium as well as spirochetes, such as Treponema.[3]
Necrotizing gingivitis may also be associated with diseases in which the immune system is compromised, including HIV/AIDS.[1] Necrotizing gingivitis is an opportunistic infection that occurs on a background of impaired local or systemic host defenses. The predisposing factors for necrotizing gingivitis are smoking, psychological stress, malnutrition, and immunosuppression. Regarding malnutrition, the fact that vitamin C deficiency is associated with gum disease, loosening of teeth, and poor wound healing shows how poor nutrition can predispose to oral disease; in such a circumstance, immune defenses against bacterial attack are most likely lower than usual. It is thus plausible that poor rations and supply line interruptions in World War I contributed to the incidence of trench mouth.
The following zones of infection have been described (superficial to deep): the bacterial zone, the neutrophil rich zone, the necrotic zone and the spirochetal zone.
Diagnosis
[edit]Diagnosis is usually clinical.[3] Smear for fusospirochaetal bacteria and leukocytes; blood picture occasionally.[3] The important differentiation is with acute leukemia or herpetic stomatitis.[3]
Classification
[edit]Necrotizing gingivitis is part of a spectrum of disease termed necrotizing periodontal diseases. It is the most minor form of this spectrum, with more advanced stages being termed necrotizing periodontitis, necrotizing stomatitis, and the most extreme, cancrum oris.[2]
Necrotizing periodontitis (NP) is where the infection leads to attachment loss, and involves only the gingiva, periodontal ligament and alveolar ligament.[2][1] Progression of the disease into tissue beyond the mucogingival junction characterizes necrotizing stomatitis.
Treatment
[edit]Treatment includes irrigation and debridement of necrotic areas (areas of dead and/or dying gum tissue), oral hygiene instruction and the uses of mouth rinses and pain medication. If there is systemic involvement, then oral antibiotics may be given, such as metronidazole.[3] As these diseases are often associated with systemic medical issues, proper management of the systemic disorders is appropriate.[1]
Prognosis
[edit]Untreated, the infection may lead to rapid destruction of the periodontium and can spread, as necrotizing stomatitis or noma, into neighbouring tissues in the cheeks, lips or the bones of the jaw. As stated, the condition can occur and be especially dangerous in people with weakened immune systems. This progression to noma is possible in malnourished susceptible individuals, with severe disfigurement possible.
Epidemiology
[edit]In developed countries, this disease occurs mostly in young adults. In developing countries, NUG may occur in children of low socioeconomic status, usually occurring with malnutrition (especially inadequate protein intake) and shortly after the onset of viral infections (e.g. measles).[2]
Predisposing factors include smoking, viral respiratory infections and immune defects, such as in HIV/AIDS. Uncommon, except in lower socioeconomic classes, this typically affects adolescents and young adults, especially in institutions, armed forces, etc., or people with HIV/AIDS.[3] The disease has occurred in epidemic-like patterns, but it is not contagious.[2]
History
[edit]Necrotizing gingivitis has been observed for centuries. Xenophon observes sore mouth and foul smelling breath in Greek soldiers in the 4th century BC. Hunter describes the clinical features of necrotizing gingivitis in 1778, differentiating it from scurvy (avitaminosis C) and chronic periodontitis. Jean Hyacinthe Vincent, a French physician working at the Paris Pasteur Institute describes a fusospirochetal infection of the pharynx and palatine tonsils, causing "ulcero-membranous pharyngitis and tonsillitis",[4] which later became known as Vincent's angina. Later in 1904, Vincent describes the same pathogenic organisms in "ulceronecrotic gingivitis". Vincent's angina is sometimes confused with NUG, however the former is tonsillitis and pharyngitis, and the latter involves the gums, and usually the two conditions occur in isolation from each other.
The term trench mouth evolved because the disease was observed in front line soldiers during World War I, thought to be a result at least partly because of extreme psychologic stress they were exposed to.[2] The same condition was appearing in civilians during periods of bombing raids, who were away from the front line, and who had relatively good diets during wartime due to rationing, so it is assumed that psychologic stress was the significant causative factor. It has also been associated with high tobacco use in the army.
Many other historical names for this condition (and Vincent's angina) have occurred, including: "acute membranous gingivitis", "fusospirillary gingivitis", " fusospirillosis", "fusospirochetal gingivitis", "phagedenic gingivitis", "Vincent stomatitis", "Vincent gingivitis", and "Vincent infection".[5]
In the late 1980s-early 1990s, it was originally thought that some necrotizing periodontal diseases seen in severely affected AIDS patients were strictly a sequela of HIV, and it was even called HIV-associated periodontitis.[6] It is now understood that its association with HIV/AIDS was due to the immunocompromised status of such patients; it also occurs with higher prevalence in association with other diseases in which the immune system is compromised.[1]
The 1999 American Academy of Periodontology Classification termed the condition "necrotizing ulcerative periodontitis". The "ulcerative" descriptor was removed from the name, because ulceration is considered to be secondary to the necrosis.[1]
See also
[edit]References
[edit]- ^ a b c d e f g Herrera, D., Retamal-Valdes, B., Alonso, B., Feres, M. (June 2018). "Acute periodontal lesions (periodontal abscesses and necrotizing periodontal diseases) and endo-periodontal lesions: Dd56II Joint EFP-AAP Workshop". Journal of Periodontology. 89: S85 – S102. doi:10.1002/JPER.16-0642. ISSN 0022-3492.
- ^ a b c d e f g h i j k Lindhe, Jan; Lang, Niklaus P.; Karring, Thorkild, eds. (2008). Clinical periodontology and implant dentistry (5th ed.). Oxford: Blackwell Munksgaard. pp. 413, 459. ISBN 9781405160995.
- ^ a b c d e f g h i j k l m Scully, Crispian (2008). Oral and maxillofacial medicine : the basis of diagnosis and treatment (2nd ed.). Edinburgh: Churchill Livingstone. pp. 101, 347. ISBN 9780443068188.
- ^ Taylor, FE; McKinstry, WH (1917). "The Relation of Peri-dental Gingivitis to Vincent's Angina". Proceedings of the Royal Society of Medicine. 10 (Laryngol Sect): 43–8. PMC 2017821. PMID 19979715.
- ^ "Definition of Vincent angina". Medterms.com. 2001-09-13. Archived from the original on 2011-06-05. Retrieved 2010-02-13.
- ^ NYS Department of Health AIDS Institute. "Clinical Manifestations and Management of HIV-Related Periodontal Disease". Oral Health Care for People with HIV Infection: HIV Clinical Guidelines. p. 31.
External links
[edit]Necrotizing gingivitis
View on GrokipediaClinical Presentation
Signs and Symptoms
Necrotizing gingivitis, also known as acute necrotizing ulcerative gingivitis (ANUG), presents with primary symptoms of severe gingival pain, spontaneous bleeding, and foul breath (halitosis).[6][1][7] The pain is often intense and localized to the affected areas, interfering with eating, speaking, and oral hygiene practices, while bleeding occurs readily without provocation and contributes to the patient's discomfort.[1][4] Halitosis arises from the necrotic tissue and is typically described as overwhelmingly fetid.[6][1] Characteristic local signs include interdental gingival necrosis manifesting as punched-out craters at the tips of the interdental papillae, often covered by a grayish pseudomembrane composed of necrotic debris and fibrin.[1][8][9] The gingival margins exhibit erythema, edema, and sloughing, with the necrotic areas appearing ulcerated and sharply demarcated from surrounding healthy tissue.[1][10] In moderate to severe cases, systemic signs such as fever, malaise, and regional lymphadenopathy may accompany the local manifestations.[1][6][8] These symptoms reflect the inflammatory response and can vary in intensity, with lymphadenopathy typically involving submandibular nodes.[3][11] The condition has a rapid onset, often affecting the anterior teeth in the maxillary and mandibular regions, with lesions starting at the interdental papillae and potentially extending to adjacent oral mucosa in severe presentations.[1][12][13] This progression can lead to blunting of the papillae and increased salivation as secondary features.[6][1] It is commonly associated with poor oral hygiene, which exacerbates the acute episode.[14]Complications
Untreated or recurrent necrotizing gingivitis can result in significant local tissue destruction, including gingival recession due to progressive loss of attachment and extensive soft tissue necrosis.[3] This rapid deterioration often leads to tooth loss as a direct consequence of the aggressive periodontal destruction.[3] Furthermore, the condition frequently extends beyond the gingiva, progressing to necrotizing ulcerative periodontitis (NUP), characterized by deeper involvement of periodontal tissues and alveolar bone loss.[1] Systemic complications arise particularly in vulnerable populations, such as immunocompromised individuals, where the infection may cause secondary bacteremia through dissemination of oral pathogens.[3] Severe pain associated with the necrotic lesions can induce avoidance of eating and drinking, contributing to dehydration and weight loss.[3] In advanced cases, these systemic effects may exacerbate underlying conditions, leading to fever, malaise, and further nutritional deficits.[3] Chronic or recurrent episodes of necrotizing gingivitis heighten the risk of transitioning to persistent forms like NUP, involving irreversible bone resorption and chronic inflammation.[1] This progression underscores the need for vigilant monitoring to prevent long-term periodontal damage.[3] Rare but severe outcomes include involvement of adjacent structures, potentially resulting in osteomyelitis with alveolar bone exposure and sequestration of bone fragments, or progression to noma (cancrum oris), a gangrenous infection primarily affecting malnourished children that destroys oral and facial tissues.[3] Extension of the infection can also lead to life-threatening conditions such as Ludwig's angina, a rapidly spreading cellulitis of the submandibular space.[15]Etiology
Microbial Causes
Necrotizing gingivitis (NG), also known as acute necrotizing ulcerative gingivitis, is primarily caused by a polymicrobial infection involving anaerobic bacteria that form biofilms in the gingival crevices. The key pathogens include fusobacteria, particularly Fusobacterium nucleatum, and spirochetes such as Treponema denticola and other Treponema species, which together constitute the classic fusospirochetal complex responsible for initiating tissue destruction.[1][16] These anaerobes thrive in the oxygen-deprived environment of disrupted gingival flora, leading to an overgrowth that disrupts the microbial balance and promotes invasion of the gingival papillae. F. nucleatum acts as a bridge bacterium in biofilms, facilitating adhesion and coaggregation of other pathogens like spirochetes, which enhances the synergistic production of virulence factors such as lipopolysaccharides and proteases that cause necrosis.[17][16] Recent microbiome studies using 16S rRNA sequencing have expanded this understanding, revealing a more diverse profile in NG lesions, with elevated levels of Bacteroidetes phylum members including Porphyromonas gingivalis, Prevotella intermedia, and Tannerella forsythia, alongside increased spirochetes and fusobacteria compared to chronic gingivitis. These analyses indicate that NG biofilms exhibit dysbiosis with Proteobacteria and Firmicutes also playing roles in disease progression, differing significantly from healthy oral microbiomes.[18][19] Viral co-factors, particularly HIV, contribute to pathogenesis by altering the oral microbiome, increasing susceptibility to NG through immune dysregulation that favors opportunistic anaerobes and spirochetes in subgingival biofilms. In HIV-positive individuals, necrotizing lesions show enriched Borrelia-like organisms and gram-negative anaerobes, exacerbating tissue invasion.[20][21]Risk Factors
Necrotizing gingivitis, also known as acute necrotizing ulcerative gingivitis, is predisposed by various behavioral factors that compromise oral health and immune response. Smoking is a prominent risk factor, with studies indicating that current smokers have approximately 3-4 times higher odds of developing the condition compared to nonsmokers.[22] Poor oral hygiene allows bacterial plaque accumulation, facilitating gingival inflammation and necrosis.[1] Psychological stress, such as that experienced during examinations or wartime conditions, further exacerbates vulnerability by impairing immune function and sleep quality.[1] Nutritional deficiencies and systemic conditions significantly heighten susceptibility. Malnutrition, particularly deficiencies in vitamins like C and B-complex, weakens tissue repair and immune defenses, making the gingiva more prone to destructive infections.[1] Immunosuppressive states, including HIV infection and leukemia, diminish the body's ability to control oral pathogens, leading to higher incidence rates.[1] Inadequate sleep, often linked to stress or lifestyle factors, similarly disrupts immune regulation and oral healing processes.[1] Demographically, the condition shows a higher prevalence among young adults aged 18-30 years, a period often marked by lifestyle changes, academic pressures, and initiation of tobacco use.[1] Individuals from low-socioeconomic backgrounds face elevated risks due to limited access to dental care and nutritious diets, compounding other vulnerabilities.[1] Environmental factors in developing regions, such as overcrowding and poor sanitation, promote the spread of predisposing infections and hinder hygiene practices, particularly in malnourished populations.[1] Emerging research from the 2020s highlights potential links to pandemic-related stressors from COVID-19, which may amplify psychological and immune burdens, and vaping, associated with increased odds of gingival lesions akin to those in necrotizing gingivitis. These non-infectious elements collectively enable microbial overgrowth in the gingival sulcus.Diagnosis and Classification
Diagnostic Methods
Diagnosis of necrotizing gingivitis is primarily clinical, relying on characteristic features including rapid onset of gingival pain, interdental ulceration and necrosis of the papillae, spontaneous bleeding, and pseudomembrane formation, as outlined in the 2018 World Workshop classification of periodontal diseases.[23] The condition is confirmed when at least three of these signs are present: necrosis or ulceration of the interdental papilla, bleeding on minimal provocation, and acute pain or discomfort, often accompanied by fetid odor from spirochetal and fusobacterial overgrowth.[24] No specific CDC case definition exists for necrotizing gingivitis, but it aligns with surveillance criteria for acute periodontal infections emphasizing clinical attachment loss limited to the soft tissues without radiographic bone involvement.[25] Clinical examination begins with visual inspection to identify punched-out crater-like lesions on the interdental papillae, covered by a grayish pseudomembrane that can be wiped away to reveal bleeding ulcerated tissue.[1] Periodontal probing assesses the depth of these craters, typically revealing minimal attachment loss confined to the marginal gingiva, distinguishing it from deeper periodontal destruction.[6] Assessment of foul odor, often described as fetor oris, is a key sensory indicator of the anaerobic bacterial involvement.[3] Adjunctive tools are used sparingly to support the diagnosis and exclude complications. Radiographs, such as periapical views, help rule out underlying bone loss, confirming that necrosis is limited to soft tissues in uncomplicated cases.[3] Biopsy is rarely indicated but, if performed in atypical presentations, reveals histopathologic findings of coagulative necrosis, vascular thrombi, and dense neutrophilic infiltration with spirochetes visible on dark-field microscopy.[26] Emerging technologies are enhancing diagnostic precision, particularly in resource-limited settings. AI-assisted imaging systems, such as those integrating intraoral scans and cone-beam computed tomography, enable automated detection of gingival bone depth and early necrotic changes with high accuracy, as demonstrated in 2025 studies.[27] Point-of-care tests for inflammatory markers, such as saliva-based assays for active matrix metalloproteinase-8 (aMMP-8), provide rapid assessment of periodontal inflammation, aiding confirmation of necrotizing processes with results in under 5 minutes.[28] To exclude mimics, a brief differential checklist includes evaluating for viral infections like acute herpetic gingivostomatitis (vesicular lesions without interdental specificity), bacterial causes such as streptococcal gingivitis (diffuse erythema without necrosis), and systemic conditions like agranulocytosis (widespread oral ulceration with neutropenia on blood work).[1] If systemic symptoms or atypical features are present, laboratory tests for immune status or viral serology may be warranted to differentiate from these entities.[3]Classification
Necrotizing gingivitis is classified as a distinct entity within the broader category of necrotizing periodontal diseases, as outlined in the 1999 classification system developed by the American Academy of Periodontology (AAP) during the International Workshop for a Classification of Periodontal Diseases and Conditions.[29] In this framework, it is termed necrotizing ulcerative gingivitis (NUG), characterized by necrosis limited to the gingival tissues without involvement of the periodontal ligament or alveolar bone, distinguishing it from more advanced forms like necrotizing ulcerative periodontitis (NUP).[1] This classification emphasized NUG's acute, destructive nature in the context of impaired host response, often linked to stress or poor oral hygiene.[30] The condition is further categorized by extent into localized and generalized forms, where localized NUG affects isolated interdental papillae, typically in a few sites, while generalized involvement spans multiple areas of the gingiva.[9] Progression occurs in untreated cases, advancing from NUG to NUP, marked by rapid loss of periodontal attachment and bone exposure, and potentially to necrotizing stomatitis if necrosis extends beyond the gingiva.[7] These stages represent a continuum of the same pathological process driven by polymicrobial infection and host susceptibility.[3] Differentiation from related necrotizing conditions is based on tissue involvement: necrotizing gingivitis is confined to soft gingival tissues with ulceration and pseudomembrane formation, whereas necrotizing periodontitis includes attachment loss and bone necrosis, and necrotizing stomatitis involves extension to adjacent oral mucosa, leading to broader tissue destruction.[3] This taxonomic distinction aids in prognostic assessment and management planning.[7] The 2017 AAP/European Federation of Periodontology (EFP) classification updated this framework by consolidating necrotizing periodontal diseases under a single category, including necrotizing gingivitis, necrotizing periodontitis, and necrotizing stomatitis, with emphasis on their association with systemic immune deficiencies such as HIV infection.[31] HIV-associated forms are integrated as variants of these diseases, often presenting with increased severity and higher recurrence rates due to persistent immunosuppression, particularly when CD4 counts are low.[32] Recurrence patterns in HIV patients show no direct correlation with CD4 levels but are influenced by ongoing viral control and oral hygiene.[33] Subtypes are delineated as primary (idiopathic) occurring in otherwise healthy individuals under acute stressors, and secondary forms linked to underlying conditions. These secondary cases highlight the role of iatrogenic factors in disease manifestation.Treatment
Initial Management
The initial management of necrotizing gingivitis prioritizes gentle mechanical removal of necrotic tissue and debris to halt disease progression and alleviate acute symptoms, typically performed by a dental professional over several visits to minimize discomfort. This involves careful supragingival debridement using ultrasonic scalers or hand instruments to remove necrotic tissue, plaques, and calculi without aggressive probing into sensitive areas, often under local anesthesia if needed to manage pain during the procedure.[1][6] Patients receive detailed oral hygiene instructions to support debridement, including gentle wiping of teeth with a soft toothbrush or washcloth initially to avoid further irritation, followed by twice-daily rinses with diluted hydrogen peroxide (mixed half-and-half with water for a 1.5% solution) or 0.12% chlorhexidine gluconate to remove debris and provide antimicrobial effects. These rinses, used hourly with saline or peroxide in the first few days post-debridement, help reduce bacterial load and promote healing without requiring vigorous brushing.[6][2] Basic pain control focuses on non-invasive measures alongside over-the-counter analgesics such as ibuprofen to manage discomfort, combined with dietary modifications like consuming soft, nutritious foods while avoiding hot, spicy, or irritating items that could worsen inflammation. Supportive care emphasizes adequate hydration, rest, and overall nutrition to address systemic effects like fever or malaise associated with the condition's underlying necrosis.[6][2]Pharmacological Therapy
The pharmacological therapy for necrotizing gingivitis primarily targets the anaerobic bacterial infection while minimizing the risk of antimicrobial resistance through short, targeted courses. First-line treatment involves systemic antibiotics such as metronidazole, administered at 250 mg three times daily for 5 to 7 days, which effectively addresses the predominant anaerobic pathogens like Fusobacterium and Prevotella species.[1][6] For patients with metronidazole allergy or intolerance, alternatives include amoxicillin at 500 mg every 8 hours for the same duration. In pediatric or immunocompromised patients, dosing should be adjusted per guidelines, and antifungal agents may be added if superinfection (e.g., candidiasis) develops post-antibiotics.[1][34][3] Adjunctive medications support infection control and symptom relief without replacing mechanical debridement. Chlorhexidine gluconate 0.12% mouthwash, used as a twice-daily rinse for up to 30 days, reduces plaque accumulation and bacterial load in the gingival crevices.[1][3] Nonsteroidal anti-inflammatory drugs (NSAIDs), such as ibuprofen at 400-600 mg three times daily as needed, provide analgesia and help manage associated gingival inflammation and pain.[35][36] Treatment duration is limited to 5-7 days for antibiotics, or until 72 hours after symptom resolution, to promote antibiotic stewardship and prevent resistance development, as emphasized in 2020s dental guidelines.[6][37] Follow-up evaluations within 3-7 days assess clinical resolution, including reduced pain, ulceration healing, and absence of systemic signs like fever.[1] In special cases with systemic involvement or immunocompromise (e.g., HIV patients), broader-spectrum regimens such as amoxicillin 500 mg plus metronidazole 250 mg every 8 hours for 7 days may be used, alongside monitoring for opportunistic infections.[34][3] Preliminary evidence from case reports, such as the use of Lactobacillus reuteri lozenges dissolved slowly in the mouth daily for 4 weeks in a refractory orthodontic case, suggests potential as an adjunct for microbiome restoration post-antibiotic therapy to reduce gingival inflammation, but further clinical trials are needed to establish efficacy comparable to standard treatments.[38][39] This approach aligns with antibiotic stewardship by potentially shortening systemic antibiotic needs and aiding long-term periodontal health.[37][40]Prognosis and Prevention
Prognosis
With early intervention, necrotizing gingivitis typically exhibits rapid symptom resolution, with ulcer healing often occurring within a few days and substantial improvement within 1-2 weeks following appropriate management.[1] Full recovery is achievable in the majority of cases when treatment addresses both local and systemic risk factors, preventing progression to more severe forms such as necrotizing ulcerative periodontitis.[3] Prompt treatment significantly influences outcomes by significantly reducing the likelihood of recurrence with ongoing maintenance of oral hygiene and risk factor control, whereas delays in care can result in progression to chronic periodontitis in untreated or inadequately managed cases.[3] Long-term prognosis includes a potential for periodontal attachment loss if the condition recurs or advances, necessitating regular monitoring, particularly in high-risk groups such as smokers, malnourished individuals, or those with immunosuppression.[1] Comorbidities like diabetes exacerbate healing challenges and increase recurrence risk due to impaired immune response and glycemic control issues.[41][42] If untreated, however, it may lead to complications such as extensive tissue destruction or secondary infections.[1]Prevention Strategies
Maintaining rigorous oral hygiene is fundamental to preventing necrotizing gingivitis, as it disrupts plaque accumulation and bacterial overgrowth that contribute to the condition. Individuals should brush their teeth twice daily for two minutes using a soft-bristled toothbrush and fluoride toothpaste, floss daily to remove interdental debris, and use antimicrobial mouth rinses when indicated. Professional dental cleanings every six months allow for early detection and removal of calculus, significantly reducing the risk of progression to necrotizing lesions.[43][44] Lifestyle modifications play a critical role in lowering susceptibility to necrotizing gingivitis by addressing modifiable risk factors. Smoking cessation is particularly impactful, as tobacco use impairs immune response and gingival vascularity, increasing disease incidence; quitting has been shown to reduce periodontal disease risk and support tissue healing. Stress management techniques, such as mindfulness practices, help mitigate psychological stressors that exacerbate immune suppression, while a balanced diet rich in vitamins (e.g., C and B-complex) and minerals supports gingival health and reduces inflammation.[45][46][3] In high-risk populations, such as military personnel under extreme stress or individuals with HIV, targeted interventions can prevent outbreaks. Prophylactic use of 0.12% chlorhexidine mouth rinses, typically twice daily for short periods, has been recommended to control bacterial load in these settings, particularly during periods of compromised immunity. Emerging research into vaccines targeting periodontal pathogens shows promise for mucosal immunity enhancement, though clinical trials remain in early stages and focus primarily on broader periodontitis prevention.[47][48][49] At the community level, education campaigns in endemic areas—often regions with poor sanitation and malnutrition—emphasize hygiene promotion and infrastructure improvements to curb transmission. Programs teaching basic oral care and access to clean water have demonstrated reduced incidence of acute necrotizing gingivitis precursors, aligning with broader neglected tropical disease strategies. Following WHO's 2023 recognition of noma as a neglected tropical disease, community prevention efforts in endemic regions have intensified, focusing on nutrition and hygiene to address ANUG precursors.[50][51] Recent studies (as of 2024) highlight the potential of dietary interventions to modulate the oral microbiome, offering a novel preventive avenue. Prebiotic-rich foods, such as those containing inulin or fructooligosaccharides, foster beneficial bacteria that inhibit pathogenic overgrowth associated with necrotizing gingivitis; research on probiotic-prebiotic combinations in periodontal therapy supports their adjunctive role in maintaining microbial balance, though evidence for necrotizing forms is limited.[52][53]Historical and Epidemiological Context
History
Necrotizing gingivitis, also known as acute necrotizing ulcerative gingivitis (ANUG), has been documented for centuries, with ancient references to painful oral ulcerations in military contexts dating back to the 4th century BCE among Greek soldiers. Its systematic recognition in modern medicine began in the late 19th century, when French army physician Jean Hyacinthe Vincent described the condition as a fusospirochetal infection involving fusobacteria and spirochetes, initially referring to it as "phagedenic gingivitis" due to its rapidly destructive, gangrenous nature.[12] This early work, published around 1896 while Vincent was at the Pasteur Institute in Paris, established the microbial etiology and differentiated it from other ulcerative oral conditions.[54] The disease gained widespread attention during World War I, when outbreaks occurred among soldiers in the trenches, leading to the colloquial term "trench mouth." These epidemics were attributed to predisposing factors such as psychological stress, sleep deprivation, malnutrition, and neglected oral hygiene amid the harsh wartime conditions, leading to widespread cases among troops.[54] The association with military life persisted into World War II, but post-war observations shifted focus to civilian cases, prompting a terminological evolution to ANUG to emphasize its acute, ulcerative, and necrotic features without the wartime connotation.[1] In the 1920s, Vincent and collaborators, including German physician Hugo Plaut who independently described similar fusospirochetal infections, further elucidated the polymicrobial nature of the condition through histological and bacteriological studies, solidifying its infectious profile. By the mid-20th century, ANUG became the preferred clinical descriptor in periodontal literature, reflecting improved understanding of its non-contagious yet opportunistic pathogenesis in immunocompromised or stressed individuals. In 1999, the International Workshop for a Classification of Periodontal Diseases and Conditions, convened by the American Academy of Periodontology, formalized its place within necrotizing periodontal diseases, distinguishing necrotizing ulcerative gingivitis from more severe forms like necrotizing ulcerative periodontitis.[12] Into the 21st century, necrotizing gingivitis has been increasingly highlighted in global health contexts, particularly among vulnerable populations in refugee and displacement crises, where malnutrition, overcrowding, and limited access to care exacerbate risks. Reports from the 2010s onward document cases in migrant and refugee settings, underscoring its persistence as a marker of socioeconomic hardship and the need for integrated oral health interventions in humanitarian responses.Epidemiology
Necrotizing gingivitis (NG), also known as acute necrotizing ulcerative gingivitis, exhibits a low global prevalence estimated at less than 1% in the general population.[3] A recent meta-analysis reports an overall prevalence of 0.04%, with variations based on risk factors such as HIV infection, where it reaches 0.07%, and among drug users at 0.08%.[55] In high-risk groups including severely malnourished children in areas prone to noma, prevalence can be significantly higher, up to 6.8% in affected villages in Niger.[51] Geographically, NG occurs more frequently in developing countries across Africa, Asia, and Latin America, where socioeconomic challenges like poverty and malnutrition contribute to elevated rates; for instance, prevalence is highest in Latin America at 0.08% and Asia at 0.07%.[55] In contrast, it is uncommon in high-income nations, with rates below 0.5%.[56] Demographically, the condition predominantly affects young adults aged 18 to 30 years, with a peak incidence in the 20-29 age group, and shows a slight male predominance in some populations, though gender distribution is often equal.[3] It also exhibits seasonal variations, with higher occurrences during periods of increased stress, such as summer months in certain regions.[57] Over time, NG has declined in developed countries since the post-1950s era, attributed to improvements in oral hygiene and living standards following World War II, when incidence was notably higher among stressed populations like military personnel.[26] Incidence rises during conflicts or pandemics due to associated stressors and immunocompromise.[1] In the 2020s, post-COVID-19 reports indicate an association with increased gingival inflammation and potential rises in NG cases among young adults, linked to viral-induced stress and immune dysregulation, though exact incidence increases remain under study.[58]References
- https://wikem.org/wiki/Acute_necrotizing_ulcerative_gingivitis

