Recent from talks
Nothing was collected or created yet.
Scrotum
View on Wikipedia
In most terrestrial mammals, the scrotum (pl.: scrotums or scrota; possibly from Latin scortum, meaning "hide" or "skin")[1][2] or scrotal sac is a part of the external male genitalia located at the base of the penis. It consists of a sac of skin containing the external spermatic fascia, testicles, epididymides, and vasa deferentia. The scrotum will usually tighten when exposed to cold temperatures.
The scrotum is homologous to the labia majora in females.
Structure
[edit]
In regard to humans, the scrotum is a suspended two-chambered sac of skin and muscular tissue containing the testicles and the lower part of the spermatic cords. It is located behind the penis and above the perineum. The perineal raphe is a small, vertical ridge of skin that expands from the anus and runs through the middle of the scrotum front to back. The scrotum is also a distention of the perineum and carries some abdominal tissues into its cavity including the testicular artery, testicular vein, and pampiniform plexus.
Nerve supply
[edit]| Nerve | Surface[3] |
|---|---|
| Genital branch of genitofemoral nerve | anterolateral |
| Anterior scrotal nerves (from ilioinguinal nerve) | anterior |
| Posterior scrotal nerves (from perineal nerve) | posterior |
| Perineal branches of posterior femoral cutaneous nerve | inferior |
Blood supply
[edit]| Blood vessels[4] | |
|---|---|
| Anterior scrotal artery | originates from the deep external pudendal artery[5] |
| Posterior scrotal artery | |
| Testicular artery |
Skin and glands
[edit]The skin on the scrotum is more highly pigmented in comparison to the rest of the body. The septum is a connective tissue membrane dividing the scrotum into two cavities.[6]
Lymphatic system
[edit]The scrotal lymph initially drains into the superficial inguinal lymph nodes, this then drains into the deep inguinal lymph nodes. The deep inguinal lymph nodes channel into the common iliac, which ultimately releases lymph into the cisterna chyli.
| Lymphatic vessels[7] | |
|---|---|
| Superficial inguinal lymph nodes | |
| Popliteal lymph nodes |
Asymmetry
[edit]One testis is typically lower than the other, which is believed to function to avoid compression in the event of impact; in humans, the left testis is typically lower than the right.[8] An alternative view is that testis descent asymmetry evolved to enable more effective cooling of the testicles.[9]
Internal structure
[edit]
Additional tissues and organs reside inside the scrotum and are described in more detail in the following articles:
Development
[edit]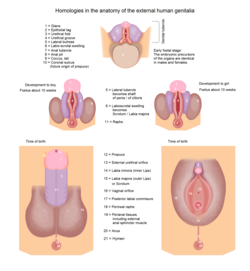
During the fifth week after fertilization, the genital ridge grows behind the peritoneal membrane. By the sixth week, string-like tissues called primary sex cords form within the enlarging genital ridge. Externally, a swelling called the genital tubercule appears over the cloacal membrane.
Testosterone secretion starts during week eight, reaches peak levels during week 13 and eventually declines to very low levels by the end of the second trimester. The testosterone causes the masculinization of the labioscrotal folds into the scrotum. The scrotal raphe is formed when the embryonic, urethral groove closes by week 12.[10]
Scrotal growth and puberty
[edit]Though the testes and scrotum form early in embryonic life, sexual maturation begins upon entering puberty. The increased secretion of testosterone causes the darkening of the skin and development of pubic hair on the scrotum.[11]
Function
[edit]The scrotum regulates the temperature of the testicles and maintains it at 35 degrees Celsius (95 degrees Fahrenheit), i.e. two or three degrees below the body temperature of 37 degrees Celsius (99 degrees Fahrenheit). Higher temperatures affect spermatogenesis.[12] Temperature control is accomplished by the smooth muscles of the scrotum moving the testicles either closer to or further away from the abdomen dependent upon the ambient temperature. This is accomplished by the cremaster muscle in the abdomen and the dartos fascia (muscular tissue under the skin that makes the scrotum appear wrinkly).[11]
During sexual arousal, the scrotum will also tighten and thicken in the course of penile erection.[13]
Having the scrotum and testicles situated outside the abdominal cavity may provide additional advantages. The external scrotum is not affected by abdominal pressure. This may prevent the emptying of the testes before the sperm were matured sufficiently for fertilization.[12] Another advantage is it protects the testes from jolts and compressions associated with an active lifestyle. The scrotum may provide some friction during intercourse, helping to enhance the activity.[14] The scrotum is also considered to be an erogenous zone.[15]
Society and culture
[edit]Common slang terms for the scrotum are ballsack, nutsack, and teabag.
Some men will get a piercing on the skin of the scrotum, any of which is called a hafada (e.g., scrotal ladder). Side-to-side or front-to-back piercings that pass through the scrotum are known as transscrotal piercings.
Scrotoplasty is a sex reassignment surgery that creates a scrotum for trans men using tissue from the labia majora, or a plastic surgery that repairs or reconstructs the scrotum.
Cock and ball torture is a kink that may involve bringing pain to the scrotum. Beyond kink, a person (especially a man) may hit someone in the testicles as a gendered cultural practice known as sack tapping. This phenomenon is complex and contains many (often conflicting) meanings: it is used to both strengthen inclusive bonds and reinforce exclusive hierarchies, it is both humorous and violent, and both juvenile and present in male-dominated social spheres beyond those of adolescence.[16]
Other animals
[edit]A scrotum is present in all boreoeutherian land mammals except hippopotamuses, rhinoceroses, hedgehogs, moles, pangolins, tapirs, and numerous families of bats and rodents.[17] The anus is separated from the scrotum by the perineum in these mammals. The testicles remain in the body cavity in all other vertebrates, including cloacal animals.[18]
Unlike placentals, some male marsupials have a scrotum that is anterior to the penis,[19][20][21][22] which is not homologous to the scrotum of placentals,[23] although there are several marsupial species without an external scrotum.[24]
The scrotum is also absent in marine mammals, such as whales, dolphins, and seals,[25] as well as in lineages of other land mammals, such as the afrotherians (elephants, aardvarks, etc.), xenarthrans (armadillos, anteaters, and sloths),[26][17] and monotremes.[27][28]
Clinical significance
[edit]Diseases and conditions
[edit]The scrotum and its contents can develop many diseases and can incur injuries. These include:
- Candidiasis (yeast infection)
- Sebaceous cyst
- Epidermal cyst
- Hydrocele testis
- Hematocele
- Molluscum contagiosum
- Spermatocele
- Paget's disease of the scrotum[29]
- Varicocele - enlargement of the pampiniform venous complex
- Inguinal hernia
- Epididymo-orchitis
- Testicular torsion
- Pruritus scroti - irritation of the scrotum (itchiness)
- Genital warts - sexually transmitted infection
- Testicular cancer
- Dermatitis
- Undescended testes (also known as cryptorchidism)
- Chyloderma - swollen scrotum caused by a lymphatic obstruction
- Mumps
- Scabies
- Herpes - sexually transmitted infection
- Pubic lice
- Chancroid (Haemophilus ducreyi) - sexually transmitted infection
- Chlamydia (Chlamydia trachomatis) - sexually transmitted infection
- Gonorrhea (Neisseria gonorrhoeae) - sexually transmitted infection
- Granuloma inguinale or (Klebsiella granulomatis)
- Syphilis (Treponema pallidum) - sexually transmitted infection
- Scrotal eczema
- Polyorchidism - the presence of three or more testicles
- Scrotal psoriasis disease
- Riboflavin deficiency
- Chimney sweeps' carcinoma (scrotal cancer)
See also
[edit]- Retroperitoneal lymph node dissection
- Scrotal infusion, a temporary form of body modification
- Testicular self-examination
Bibliography
[edit]- Books
 This article incorporates text in the public domain from page 1237 of the 20th edition of Gray's Anatomy (1918)
This article incorporates text in the public domain from page 1237 of the 20th edition of Gray's Anatomy (1918)- Van De Graaff, Kent M.; Fox, Stuart Ira (1989). Concepts of Human Anatomy and Physiology. Dubuque, Iowa: William C. Brown Publishers. ISBN 978-0697056757.
- Elson, Lawrence; Kapit, Wynn (1977). The Anatomy Coloring Book. New York: Harper & Row. ISBN 978-0064539142.
- "Gross Anatomy Image". Medical Gross Anatomy Atlas Images. University of Michigan Medical School. 1997. Retrieved 2015-02-23.
- Berkow, Robert (1977). The Merck Manual of Medical Information; Home Edition. Whitehouse Station, New Jersey: Merck Research Laboratories. ISBN 978-0911910872.
References
[edit]- ^ van Driel, Mels (2010). Manhood: The Rise and Fall of the Penis. Reaktion Books. p. 11. ISBN 978-1-86189-708-4. Retrieved October 14, 2023.
- ^ Spiegl, Fritz (1996). Fritz Spiegl's Sick Notes: An Alphabetical Browsing-Book of Derivatives, Abbreviations, Mnemonics and Slang for Amusement and Edification of Medics, Nurses, Patients and Hypochondriacs. Taylor & Francis. p. 142. ISBN 978-1-85070-627-4. Retrieved October 14, 2023.
- ^ Moore, Keith; Anne Agur (2007). Essential Clinical Anatomy, Third Edition. Lippincott Williams & Wilkins. p. 132. ISBN 978-0-7817-6274-8.
- ^ Elson & Kapit 1977.
- ^ antthigh at The Anatomy Lesson by Wesley Norman (Georgetown University)
- ^ "Scrotum". Encyclopædia Britannica. Retrieved 2015-02-24.
- ^ "VIII. The Lymphatic System. 5. The Lymphatics of the Lower Extremity. Gray, Henry. 1918. Anatomy of the Human Body". Retrieved 2015-02-24.
- ^ Bogaert, Anthony F. (1997). "Genital asymmetry in men" (PDF). Human Reproduction. 12 (1): 68–72. doi:10.1093/humrep/12.1.68. PMID 9043905. Archived from the original (PDF) on 2015-05-28. Retrieved 2015-06-29.
- ^ Gallup, Gordon G.; Finn, Mary M.; Sammis, Becky (2009). "On the Origin of Descended Scrotal Testicles: The Activation Hypothesis". Evolutionary Psychology. 7 (4): 147470490900700. doi:10.1177/147470490900700402.
- ^ Van De Graaff & Fox 1989, pp. 927–931.
- ^ a b Van De Graaff & Fox 1989, p. 935.
- ^ a b Van De Graaff & Fox 1989, p. 936.
- ^ Jequier, Anne M. (2008). Male Infertility: A Guide for the Clinician. Wiley. p. 180. ISBN 978-0-47069-526-5.
- ^ Jones, Richard (2013). Human Reproductive Biology. Academic Press. p. 74. ISBN 9780123821850.
The rear-entry position of mating may allow the scrotum to stimulate the clitoris and, in this way, may produce an orgasm ...
- ^ Redmon, George L. (2002). Sensual for Life. Kensington Publishing Corporation. p. 176. ISBN 978-0-75820-138-6.
- ^ Smith, C. Brian (2018-07-31). "The Quick, Painful and 'Totally Not Gay' Cultural History of Sack Whacking". MEL Magazine. Retrieved 2025-06-03.
- ^ a b Lovegrove, B. G. (2014). "Cool sperm: Why some placental mammals have a scrotum". Journal of Evolutionary Biology. 27 (5): 801–814. doi:10.1111/jeb.12373. PMID 24735476. S2CID 24332311.
- ^ "Science : Bumpy lifestyle led to external testes - 17 August 1996 - New Scientist". New Scientist. Retrieved 2007-11-06.
- ^ Hugh Tyndale-Biscoe; Marilyn Renfree (30 January 1987). Reproductive Physiology of Marsupials. Cambridge University Press. ISBN 978-0-521-33792-2.
- ^ Libbie Henrietta Hyman (15 September 1992). Hyman's Comparative Vertebrate Anatomy. University of Chicago Press. pp. 583–. ISBN 978-0-226-87013-7.
- ^ Menna Jones; Chris R. Dickman; Michael Archer (2003). Predators with Pouches: The Biology of Carnivorous Marsupials. Csiro Publishing. ISBN 978-0-643-06634-2.
- ^ Norman Saunders; Lyn Hinds (1997). Marsupial Biology: Recent Research, New Perspectives. UNSW Press. ISBN 978-0-86840-311-3.
- ^ Patricia J. Armati; Chris R. Dickman; Ian D. Hume (17 August 2006). Marsupials. Cambridge University Press. ISBN 978-1-139-45742-2.
- ^ C. Hugh Tyndale-Biscoe (2005). Life of Marsupials. Csiro Publishing. ISBN 978-0-643-06257-3.
- ^ William F. Perrin; Bernd Würsig; J.G.M. Thewissen (26 February 2009). Encyclopedia of Marine Mammals. Academic Press. ISBN 978-0-08-091993-5.
- ^ "Scrotum". National Institutes of Health. Retrieved 6 January 2011.
- ^ Griffiths, Mervyn (2012-12-02). The Biology of the Monotremes. Elsevier. ISBN 978-0-323-15331-7.
- ^ Hayssen, Virginia; Orr, Teri J. (2017-10-27). Reproduction in Mammals: The Female Perspective. JHU Press. ISBN 978-1-4214-2315-9.
- ^ "Paget's disease of the scrotum Symptoms, Diagnosis, Treatments and Causes". RightDiagnosis.com. Retrieved 2015-02-24.
External links
[edit] Media related to Scrotums at Wikimedia Commons
Media related to Scrotums at Wikimedia Commons
Scrotum
View on GrokipediaAnatomy
External Layers and Skin
The scrotum's outermost layer consists of thin skin that is darker and more pigmented than the surrounding perineal skin, featuring prominent rugae—irregular folds that increase surface area and facilitate contraction.[1] This skin comprises stratified squamous keratinized epithelium, dermis with collagen and elastic fibers, hair follicles, sebaceous glands, and sweat glands, but lacks a distinct subcutaneous adipose layer, allowing close adherence to underlying structures.[5] The absence of fat contributes to the skin's mobility and thin profile, typically measuring 0.5 to 1 mm in thickness.[6] Immediately beneath the skin lies the dartos fascia, a superficial fibromuscular layer unique to the scrotum and penis, composed of loose areolar connective tissue blended with smooth muscle fibers forming the dartos muscle.[7] This layer is continuous with the superficial fascia of the anterior abdominal wall and perineum, extending without interruption across the midline via a thin septum that divides the scrotal compartments.[1] The dartos fascia contains no fat but is richly vascularized and innervated, enabling involuntary contractions that alter scrotal tautness.[6] A visible median raphe runs along the scrotum's ventral surface, representing the remnant of embryonic fusion of the urethral folds and genital swellings, often appearing as a faint ridge or line in the skin.[8] This structure marks the bilateral symmetry of the scrotum, with each lateral half derived from distinct developmental precursors.[1] In histological sections, the interface between skin and dartos fascia shows intimate adherence, with smooth muscle bundles oriented longitudinally and obliquely to support wrinkling.[5]Internal Compartments and Supports
The scrotum is partitioned into two distinct compartments by a midline scrotal septum, a fibromuscular structure that extends from the perineal raphe and separates the left and right hemiscrota, with each compartment housing one testis and associated epididymis.[1] This septum, formed by fusion of the embryonic labioscrotal swellings, consists of subcutaneous tissue reinforced by dartos muscle fibers and provides structural integrity to prevent inter-compartmental shifting while allowing independent thermoregulation.[1] The average scrotal wall thickness, including contributions to the septum, measures approximately 8 mm.[1] Internally, each compartment is lined by the tunica vaginalis, a closed serous sac derived from the peritoneal vaginal process, comprising a parietal layer adhering to the inner scrotal wall and a visceral layer directly coating the anterior and lateral surfaces of the testis and epididymis.[1][9] This double-layered structure creates a potential serous cavity (cavum vaginale) filled with a small volume of fluid, which facilitates smooth gliding of the testis within the compartment and cushions against mechanical stress, while the posterior aspect remains uncovered to accommodate attachments.[9] Beneath the visceral layer lies the tunica albuginea of the testis, a dense fibrous capsule that anchors the organ but functions primarily as testicular support rather than scrotal compartmentalization.[9] The testes within these compartments are suspended and supported superiorly by the spermatic cord, a cord-like structure traversing the inguinal canal and containing the vas deferens, testicular artery and veins, lymphatics, and nerves, enveloped by internal, cremasteric, and external spermatic fasciae.[9] The cremaster muscle, integrated into the middle fascial layer, provides dynamic contractile support to elevate the testis toward the body in response to stimuli like cold or touch.[1] Inferiorly, a fibrous scrotal ligament (remnant of the gubernaculum) anchors the testis to the scrotal floor, preventing excessive pendular motion and maintaining positional stability.[9] These supports collectively ensure secure positioning while permitting the necessary mobility for physiological functions.[9]Vascular, Neural, and Lymphatic Supply
The arterial supply to the scrotum derives from branches of both the external and internal pudendal arteries, forming a rich network of arteriovenous anastomoses and subcutaneous plexuses that support its thermoregulatory and protective functions.[1] The anterior scrotal arteries, originating from the external pudendal artery (a femoral artery branch), provide blood to the anterior and lateral scrotal skin.[8] The posterior scrotal arteries arise from the perineal artery, a branch of the internal pudendal artery, supplying the posterior aspect and ensuring extensive interconnections with anterior vessels for robust perfusion.[10] [1] Venous drainage parallels the arterial supply, with anterior scrotal veins emptying into the external pudendal vein and posterior scrotal veins into the internal pudendal vein, ultimately contributing to the saphenous and internal iliac venous systems, respectively.[8] This dual drainage facilitates efficient return of deoxygenated blood while aiding in heat dissipation through the pampiniform plexus connections proximal to the testes.[1] Sensory innervation of the scrotum is segmental and multifocal, primarily from lumbar and sacral sources. The anterior and anterolateral surfaces receive fibers from the ilioinguinal nerve (L1) and the genital branch of the genitofemoral nerve (L1-L2), providing dermatomal sensation to the upper and lateral regions.[8] [6] The posterior scrotum is innervated by the perineal branches of the pudendal nerve (S2-S4) and the posterior femoral cutaneous nerve (S2-S3), completing circumferential coverage and enabling reflexive cremasteric contraction via sympathetic influences.[8] [3] Lymphatic drainage from the scrotal skin and wall follows its superficial ectodermal origin, directing vessels ipsilaterally to the superficial inguinal lymph nodes without crossing the median raphe septum.[1] [10] This pathway contrasts with testicular lymphatics, which ascend to para-aortic nodes, underscoring the scrotum's distinct drainage pattern that aligns with lower abdominal wall embryology and supports localized immune surveillance.[1]Asymmetry and Variations
In human anatomy, the scrotum typically displays asymmetry in testicular positioning, with the left testis suspended lower than the right in most males due to the longer left spermatic cord.[11] This configuration prevents mutual compression of the testes and facilitates thermoregulation by allowing slight temperature differentials between sides. Volume asymmetry is also common, with the right testis averaging larger; in a study of 345 healthy adolescent boys aged 11–16, 58.84% had a smaller left testis, with a mean volume difference of 0.3 mL favoring the right.[12] Such differences persist into adulthood, though they are generally within normal limits unless exceeding 20% discrepancy, which may warrant evaluation.[12] Anatomical variations in scrotal structure include congenital anomalies arising from disruptions in embryonic fusion or migration of the labioscrotal folds. A bifid scrotum features a midline cleft from incomplete fusion, often associated with hypospadias or androgen deficiencies during the 9th–12th gestational weeks.[1] Accessory scrotum presents as an extra, non-testicular perineal skin fold, resulting from aberrant swelling migration and lacking functional compartments.[1] Ectopic scrotum occurs when the sac develops in atypical positions, such as perineal or inguinal sites, due to gubernaculum defects.[1] Penoscrotal transposition, where the penis emerges cephalad to the scrotum, represents a spectrum of fusion failures and may be partial or complete.[1] These variants are rare, with prevalence data limited, but they highlight the scrotum's developmental plasticity beyond symmetric norms.[1]Embryology and Development
Fetal Origin and Descent
The scrotum originates from the paired labioscrotal swellings, embryonic structures that appear in the fourth week of gestation lateral to the genital tubercle.[1] These swellings arise as mesenchymal proliferations covered by surface ectoderm, initially undifferentiated and homologous to the future labia majora in females.[1] In male embryos, driven by dihydrotestosterone (DHT) converted from testosterone secreted by differentiating testes, the labioscrotal swellings undergo hypertrophy, migrate caudally and medially from weeks 9 to 11, and fuse along the ventral midline by week 12 to form the scrotal sac and its characteristic raphe.[13][1] This fusion delineates the scrotum's bilateral compartments, each destined to house a testis, and establishes its external pouch-like configuration suspended below the perineum.[1] Testicular descent into the scrotum occurs later in a biphasic process, independent of but coordinated with scrotal formation. The testes develop from genital ridges originating in the intermediate mesoderm of the urogenital ridge around week 6, with primordial germ cells migrating to colonize these ridges by week 5.[14] The first transabdominal phase, from weeks 8 to 15 post-fertilization, involves caudal migration of the testis from the posterior abdominal wall to the internal inguinal ring, primarily mediated by enlargement of the gubernaculum—a gelatinous mesenchymal cord anchoring the gonad—and regression of the cranial suspensory ligament, under the influence of insulin-like factor 3 (INSL3) from Leydig cells.[14] The second inguinoscrotal phase, commencing around week 25 and completing by week 35, is androgen-dependent and features androgen-stimulated outgrowth and subsequent regression of the gubernaculum, facilitating passage of the testis through the inguinal canal into the scrotum, accompanied by evagination of the processus vaginalis—a peritoneal outpouching that forms the tunica vaginalis.[14] By term (approximately week 40), over 95% of testes reside in the scrotum, with the processus vaginalis obliterating proximally to seal the pathway and minimize hernia risk.[14]Postnatal Growth and Pubertal Maturation
Following birth, the scrotum undergoes initial growth during the minipubertal phase, a postnatal surge in gonadotropins (luteinizing hormone and follicle-stimulating hormone) occurring roughly from 1 week to 6 months of age, which elevates testosterone levels and promotes testicular enlargement from approximately 0.5 mL to 1.5 mL in volume, with the scrotal sac expanding accordingly to accommodate this.[15] [16] Testicular volume then stabilizes or slightly decreases through late infancy and childhood, reflecting a quiescent period of low gonadal activity, during which the scrotum maintains its prepubertal dimensions with minimal changes in skin texture or pendency.[16] [17] Pubertal maturation of the scrotum begins with Tanner stage 2, typically between ages 9 and 14 years, marked by the first visible enlargement of the testes (exceeding 3 mL in volume) and scrotum, driven by rising hypothalamic-pituitary-gonadal axis activity that increases intratesticular testosterone production, leading to seminiferous tubule expansion and germ cell proliferation.[18] [19] The scrotal skin reddens, thins, and develops increased rugosity (wrinkling) and pigmentation, enhancing surface area for thermoregulation as the testes grow rapidly—reaching adult volumes of 15-25 mL by Tanner stage 5—while the dartos fascia and cremaster muscle contribute to greater contractility and descent.[20] [18] This androgen-dependent process correlates with overall male secondary sexual characteristics, though individual variation exists due to genetic and environmental factors influencing hormone sensitivity.[21] [18] Longitudinal studies indicate that scrotal and testicular growth accelerates most between Tanner stages 2 and 4, with annual volume increases of up to 50% in some cohorts, plateauing post-stage 4 as spermatogenesis matures; disruptions like congenital hypogonadotropic hypogonadism can delay these changes, underscoring the causal role of endogenous androgens.[18] [22]Physiology
Thermoregulation Mechanism
The scrotum regulates testicular temperature to approximately 34–35 °C, or 2–3 °C below the human core body temperature of 37 °C, as spermatogenesis requires this lower thermal range for proper meiotic progression and sperm maturation; elevations as small as 1.5–2 °C above baseline impair sperm production and morphology.[23][24] This thermoregulation integrates passive anatomical features with active physiological responses to environmental heat or cold, preventing both overheating—which disrupts germ cell DNA integrity and apoptosis rates—and excessive cooling, which could halt enzymatic processes in sperm development.[25] Empirical measurements confirm that intrascrotal temperatures exceeding 36 °C correlate with reduced semen quality in controlled heat exposure studies.[26] Passive heat dissipation relies on the scrotum's external positioning and cutaneous properties: its thin, pigmented, relatively hairless skin facilitates radiative and convective heat loss, while superficial wrinkling increases surface area for evaporation when relaxed.[27] The pampiniform plexus, a venous network enveloping the testicular artery, functions as a countercurrent heat exchanger; cooler venous blood returning from the testis absorbs heat from warmer arterial inflow, thereby precooled blood reaches the gonad, maintaining a thermal gradient independent of ambient fluctuations.[23] This vascular arrangement reduces arterial blood temperature by up to 2 °C before testicular entry, as demonstrated in anatomical dissections and thermal imaging.[28] Active regulation involves muscular adjustments modulated by thermal sensors in the scrotal skin and testes. The tunica dartos, a layer of smooth muscle fibers within the scrotal subcutaneous tissue, contracts under cold stimuli (via androgen-sensitive alpha-adrenergic pathways), wrinkling the skin to trap insulating air layers, decrease surface area, and vasoconstrict cutaneous vessels, thereby minimizing conductive and convective heat loss.[29] In heat, dartos relaxation smooths the scrotum, dilates vessels for increased blood flow to the skin (promoting dissipation), and enhances sweat gland activity—though human scrotal sweating is modest compared to other regions.[30] Concurrently, the cremaster muscle, a skeletal extension of the internal oblique, elevates the testes toward the abdominal wall during cold exposure (sympathetically mediated via genitofemoral nerve), reducing the distance for conductive warming; it relaxes in warmth to descend the testes, maximizing exposure to cooler ambient air.[31] These responses operate reflexively, with cremaster contraction detectable within seconds of temperature drops below 33 °C in experimental settings.[32] Disruptions to these mechanisms, such as varicocele-induced venous reflux or obesity-related insulation, elevate baseline scrotal temperatures by 0.5–1 °C, linking to oligospermia in cohort studies; conversely, compensatory hypercontractility in cold-adapted individuals preserves spermatogenic efficiency.[27][33] Overall, this multifaceted system ensures thermal homeostasis through coupled vasomotor, myogenic, and positional controls, substantiated by direct thermometry in humans showing intrascrotal stability across diurnal and seasonal variations.[34]Protective and Contractile Functions
The scrotum provides mechanical protection to the testes through its multilayered structure, including a tough, rugose skin layer reinforced by underlying smooth muscle, which shields the delicate testicular tissue from external trauma.[1] This protective role is enhanced by the scrotum's ability to contract, drawing the testes closer to the body wall during conditions of potential injury or environmental stress, thereby minimizing exposure to impacts or temperature extremes that could impair spermatogenesis.[6][35] The primary contractile functions are mediated by the tunica dartos and cremaster muscle. The tunica dartos, a layer of smooth muscle fibers embedded within the scrotal fascia and intimately adherent to the skin, contracts in response to sympathetic innervation, particularly during cold exposure, causing the scrotal skin to wrinkle and reduce its surface area.[36][37] This contraction limits heat loss by decreasing exposed skin and cutaneous blood flow, maintaining testicular temperature approximately 2–3 °C below core body temperature (around 34–35 °C) to safeguard sperm production, as spermatogenesis is highly sensitive to overheating.[30][29] In warmer conditions, dartos relaxation promotes heat dissipation through increased surface area and vasodilation.[38] The cremaster muscle, a paired striated muscle originating from the internal oblique and transversus abdominis, envelops each spermatic cord and testis, enabling active elevation of the testes via the cremasteric reflex.[39] This reflex, elicited by stimuli such as cold, light touch to the inner thigh, or emotional arousal, contracts the muscle to retract the testes superiorly toward the inguinal canal, protecting them from hypothermia or mechanical injury while further aiding thermoregulation.[35][40] Studies indicate cremaster contraction increases in frequency during colder seasons, correlating with reduced testicular vulnerability, though excessive reflex activity may contribute to conditions like testicular torsion.[35] Together, these mechanisms ensure the scrotum's dual role in immediate physical safeguarding and dynamic physiological adjustment, with empirical observations in mammals confirming their conservation across species for reproductive viability.[41][42]Evolutionary Biology
Primary Hypotheses for Testicular Descent
The thermoregulatory hypothesis posits that testicular descent evolved primarily to enable spermatogenesis at temperatures 2–3°C below core body temperature, as mammalian sperm production is highly sensitive to heat stress, which impairs meiosis and increases DNA damage in germ cells.[43] Experimental studies in rodents and primates demonstrate that elevating scrotal temperature to abdominal levels (e.g., via surgical relocation or external heating) rapidly induces germ cell apoptosis and azoospermia, reversible upon cooling, supporting the causal role of temperature in fertility optimization.[23] This mechanism is facilitated by the scrotum's vascular anatomy, including the pampiniform plexus for countercurrent heat exchange, which dissipates heat from arterial blood to cooler venous return, maintaining intra-scrotal temperatures around 33–35°C in humans despite ambient fluctuations.[44] Comparative data across therian mammals (marsupials and placentals) show that descent is ancestral, coinciding with the evolution of endothermy, while basal mammals like monotremes retain intra-abdominal testes compatible with lower metabolic rates and ectothermy.[45][46] An alternative, the heat dissipation hypothesis, extends thermoregulation by proposing that spermatogenesis itself generates substantial metabolic heat as a byproduct of energy-intensive processes like ATP hydrolysis in flagellar assembly, necessitating externalization to prevent thermal buildup and oxidative damage within the abdomen.[23] This view aligns with observations in large mammals, where internal testes (e.g., in elephants) correlate with reduced spermatogenic rates or compensatory cooling via specialized vascular networks, though descent predominates in mobile, endothermic lineages.[47] Empirical support includes in vitro assays showing elevated testicular temperatures during active gametogenesis, but this hypothesis remains secondary to sensitivity-driven cooling, as baseline spermatogenic heat output alone does not explain the universal descent in scrotal mammals absent heat stress experiments.[48] Less supported hypotheses include mechanical protection, such as the "galloping hypothesis," which suggests descent shields testes from abdominal jolts during locomotion in cursorial ancestors, with the scrotum's elasticity absorbing impacts.[49] However, this lacks robust evidence, as many non-galloping mammals exhibit descent, and abdominal testes in whales endure hydrodynamic stresses without impairment.[50] Phylogenetic reconstructions indicate thermoregulation as the parsimonious driver, with descent evolving once in the therian stem around 160–180 million years ago, predating specialized gaits.[45] Ongoing genomic analyses of gubernaculum genes reinforce developmental conservation tied to thermal needs over locomotor adaptations.[51]Empirical Evidence and Comparative Data
Empirical studies demonstrate that spermatogenesis in mammals is highly sensitive to temperature, with optimal function occurring at 2–4°C below core body temperature, typically around 33–35°C for humans and similar species. Experimental elevations to 37–39°C induce germ cell apoptosis, oxidative stress, DNA fragmentation, and reduced sperm motility and morphology, leading to a 14% decline in spermatogenic output per 1°C rise in testicular temperature.[52][53] In controlled heat stress models, such as in birds and rodents, acute exposure to supraoptimal temperatures decreases viable sperm counts by up to 50% within days, underscoring the selective pressure for thermoregulatory adaptations like testicular descent.[54][48] Phylogenetic analyses of mammalian orders reveal that descended scrotal testes characterize approximately 80% of species, including most eutherians and marsupials, with genetic markers indicating this trait originated in the common therian ancestor around 160–180 million years ago, prior to full endothermy.[45] Molecular vestiges, such as conserved insulin-like factor 3 (Insl3) genes essential for gubernacular descent, persist in afrotherian mammals like elephants and hyraxes, which secondarily internalized testes, suggesting ancestral descent followed by reversal in lineages adapting to aquatic or high-body-mass lifestyles.[55] Comparative dissections show that internal-testis mammals, such as whales and elephants (about 10% of placentals), compensate via vascular countercurrent exchangers that cool testes by 2–5°C despite abdominal positioning, maintaining spermatogenic efficiency comparable to scrotal species under normothermic conditions.[56] Cross-species data highlight variability: marsupials exhibit scrotal testes even in neonates, predating descent in placentals, while rodents like moles retain undescended testes with elevated apoptotic thresholds to ambient fluctuations.[57] Fossil-calibrated models link scrotum evolution to Cenozoic pulses in basal metabolic rate and body temperature, with descended testes correlating to higher endothermic demands in boreoeutherian clades, where internal retention would elevate failure rates in gamete production by 20–30% based on thermal sensitivity assays.[58] These patterns refute protection-only hypotheses, as vulnerability to predation does not consistently predict descent across arboreal versus terrestrial mammals, but align with thermoregulatory causality evidenced by convergent cooling in non-scrotal outliers.[59]Ongoing Debates and Alternative Theories
One ongoing debate concerns the sufficiency of thermoregulation as the sole adaptive driver for testicular descent, given the vulnerability of external testes to physical trauma and predation, which imposes significant evolutionary costs without clear offsetting benefits in all species. While spermatogenesis demonstrably requires temperatures 2–7°C below core body temperature, species such as elephants and whales maintain functional internal testes through vascular countercurrent heat exchange, suggesting alternative physiological adaptations could obviate the need for descent in mammals. Critics argue that the thermoregulation hypothesis fails to account for why descent evolved in most therian mammals but not in monotremes or certain afrotherians, implying phylogenetic contingency or additional selective pressures beyond mere cooling. The activation hypothesis posits that scrotal descent evolved primarily to enable situational cooling of spermatozoa immediately prior to ejaculation, enhancing motility and fertilization success rather than continuous low-temperature storage. Under this view, internal testes suffice for gamete production, but descent into the scrotum—triggered by arousal—provides a targeted thermal gradient that activates sperm hypermotility via mitochondrial responses to transient hypoxia and cooling, as evidenced by in vitro studies showing improved sperm performance at scrotal temperatures. Proponents, drawing from comparative data across primates and artiodactyls, contend this mechanism resolves the paradox of high-energy costs for maintaining an external sac, as activation occurs only during copulation, minimizing baseline risks. However, empirical validation remains limited, with counterevidence from species exhibiting constant descent lacking arousal-linked retraction.[60][56] Alternative theories invoke mechanical or behavioral factors, such as the "galloping hypothesis," which suggests descent prevents testicular damage from inertial forces during high-speed locomotion in early mammals, allowing testes to "float" freely rather than compress against abdominal walls. This idea, rooted in biomechanical modeling of cursorial therapsids, predicts descent correlating with terrestrial galloping gaits, though it struggles to explain scrotal retention in sedentary or aquatic mammals. Sexual selection hypotheses propose the scrotum as a visual or tactile signal of male quality, with size variations correlating to testosterone levels and mating success in polygynous species like red deer, yet phylogenetic reconstructions indicate such display functions likely secondary to thermoregulatory origins.[61] Phylogenetic debates persist on whether scrotal testes represent a single evolutionary innovation in the therian mammal ancestor or multiple convergent events, with molecular clock estimates placing descent around 166–178 million years ago amid rising endothermy. Comparative genomic analyses reveal gubernaculum gene expression shifts as key to positional evolution, but discordance between fossil records and genetic data fuels arguments for reversals in lineages like xenarthrans. These uncertainties underscore the interplay of developmental constraints and selective pressures, with no consensus model integrating all empirical data from extant and extinct taxa.[62]Comparative Anatomy
In Mammals
In therian mammals, the scrotum serves as an external pouch suspending the testes below body temperature to optimize spermatogenesis, typically 2–4°C cooler than core temperature via vascular countercurrent exchange and muscular contraction.[56] Monotremes, the basal mammals, lack a scrotum, retaining testicond testes intra-abdominally near the kidneys.[51] Therians exhibit testicular descent, but scrotal presence varies: most marsupials and placentals possess a scrotum, while select placentals show ascrotal or testicond configurations.[45] Marsupials display an anteriorly positioned scrotum, developing as bulges ahead of the genital tubercle early in embryogenesis, with testes descending into it before birth to protect developing young in the pouch.[58] This pre-penile orientation contrasts with placentals, where the scrotum forms posteriorly to the penis.[56] In placentals, scrotal structure includes rugose, often pigmented skin with the dartos tunic—a smooth muscle layer enabling contraction for thermoregulation and protection—and the cremaster muscle for reflexive elevation.[56] Size and pendulosity vary; for example, large pendulous scrotums occur in ungulates like bulls and red deer for heat dissipation, while tighter apposition aids locomotion in carnivores.[56] Notable absences occur in Afrotheria, such as elephants and manatees with intra-abdominal testicond testes lacking descent or scrotum, relying on body size for cooling.[51] Cetaceans and some sirenians feature incompletely descended ascrotal testes in inguinal or subcutaneous positions without a defined scrotal sac, adapted to aquatic thermoregulation via blubber and water contact.[56] Rodents and primates consistently show fully scrotal testes, with gubernaculum-mediated descent ensuring complete exteriorization.[51] These variations reflect clade-specific evolutionary pressures, with scrotal loss or modification in lineages facing aquatic or high-mass constraints.[45]









