Recent from talks
Nothing was collected or created yet.
Tissue engineering
View on Wikipedia

Tissue engineering is a biomedical engineering discipline that uses a combination of cells, engineering, materials methods, and suitable biochemical and physicochemical factors to restore, maintain, improve, or replace different types of biological tissues. Tissue engineering often involves the use of cells placed on tissue scaffolds in the formation of new viable tissue for a medical purpose, but is not limited to applications involving cells and tissue scaffolds. While it was once categorized as a sub-field of biomaterials, having grown in scope and importance, it can be considered as a field of its own.[1]
While most definitions[whose?] of tissue engineering cover a broad range of applications, in practice, the term is closely associated with applications that repair or replace portions of or whole tissues (i.e. organs, bone, cartilage,[2] blood vessels, bladder, skin, muscle etc.). Often, the tissues involved require certain mechanical and structural properties for proper functioning. The term has also been applied to efforts to perform specific biochemical functions using cells within an artificially created support system (e.g. an artificial pancreas, or a bio artificial liver). The term regenerative medicine is often used synonymously with tissue engineering, although those involved in regenerative medicine place more emphasis on the use of stem cells or progenitor cells to produce tissues.
Overview
[edit]
A commonly applied definition of tissue engineering, as stated by Langer[3] and Vacanti,[4] is "an interdisciplinary field that applies the principles of engineering and life sciences toward the development of biological substitutes that restore, maintain, or improve [Biological tissue] function or a whole organ".[5] In addition, Langer and Vacanti also state that there are three main types of tissue engineering: cells, tissue-inducing substances, and a cells + matrix approach (often referred to as a scaffold). Tissue engineering has also been defined as "understanding the principles of tissue growth, and applying this to produce functional replacement tissue for clinical use".[6] A further description goes on to say that an "underlying supposition of tissue engineering is that the employment of natural biology of the system will allow for greater success in developing therapeutic strategies aimed at the replacement, repair, maintenance, or enhancement of tissue function".[6]
Developments in the multidisciplinary field of tissue engineering have yielded a novel set of tissue replacement parts and implementation strategies. Scientific advances in biomaterials, stem cells, growth and differentiation factors, and biomimetic environments have created unique opportunities to fabricate or improve existing tissues in the laboratory from combinations of engineered extracellular matrices ("scaffolds"), cells, and biologically active molecules. Among the major challenges now facing tissue engineering is the need for more complex functionality, biomechanical stability, and vascularization in laboratory-grown tissues destined for transplantation.[7]
Etymology
[edit]The historical origin of the term is unclear as the definition of the word has changed throughout the past few decades. The term first appeared in a 1984 publication that described the organization of an endothelium-like membrane on the surface of a long-implanted, synthetic ophthalmic prosthesis.[8]
The first modern use of the term as recognized today was in 1985 by the researcher, physiologist and bioengineer Yuan-Cheng Fung of the Engineering Research Center. He proposed the joining of the terms tissue (in reference to the fundamental relationship between cells and organs) and engineering (in reference to the field of modification of said tissues). The term was officially adopted in 1987.[8]
History
[edit]Ancient era (pre-17th century)
[edit]An understanding of the inner workings of human tissues may date back further than most would expect. As early as the Neolithic period, sutures were being used to close wounds and aid in healing. Later on, societies such as ancient Egypt developed better materials for sewing up wounds such as linen sutures. Around 2500 BC in ancient India, skin grafts were developed by cutting skin from the buttock and suturing it to wound sites in the ear, nose, or lips. Ancient Egyptians often would graft skin from corpses onto living humans and even attempted to use honey as a type of antibiotic and grease as a protective barrier to prevent infection. In the 1st and 2nd centuries AD, Gallo-Romans developed wrought iron implants and dental implants could be found in ancient Mayans.
Enlightenment (17th century–19th century)
[edit]While these ancient societies had developed techniques that were way ahead of their time, they still lacked a mechanistic understanding of how the body was reacting to these procedures. This mechanistic approach came along in tandem with the development of the empirical method of science pioneered by René Descartes. Sir Isaac Newton began to describe the body as a "physiochemical machine" and postured that disease was a breakdown in the machine.
In the 17th century, Robert Hooke discovered the cell and a letter from Benedict de Spinoza brought forward the idea of the homeostasis between the dynamic processes in the body. Hydra experiments performed by Abraham Trembley in the 18th century began to delve into the regenerative capabilities of cells. During the 19th century, a better understanding of how different metals reacted with the body led to the development of better sutures and a shift towards screw and plate implants in bone fixation. Further, it was first hypothesized in the mid-1800s that cell-environment interactions and cell proliferation were vital for tissue regeneration.
Modern era (20th and 21st centuries)
[edit]As time progresses and technology advances, there is a constant need for change in the approach researchers take in their studies. Tissue engineering has continued to evolve over centuries. In the beginning people used to look at and use samples directly from human or animal cadavers. Now, tissue engineers have the ability to remake many of the tissues in the body through the use of modern techniques such as microfabrication and three-dimensional bioprinting in conjunction with native tissue cells/stem cells. These advances have allowed researchers to generate new tissues in a much more efficient manner. For example, these techniques allow for more personalization which allow for better biocompatibility, decreased immune response, cellular integration, and longevity. There is no doubt that these techniques will continue to evolve, as we have continued to see microfabrication and bioprinting evolve over the past decade.
In 1960, Wichterle and Lim were the first to publish experiments on hydrogels for biomedical applications by using them in contact lens construction. Work on the field developed slowly over the next two decades, but later found traction when hydrogels were repurposed for drug delivery. In 1984, Charles Hull developed bioprinting by converting a Hewlett-Packard inkjet printer into a device capable of depositing cells in 2-D. Three dimensional (3-D) printing is a type of additive manufacturing which has since found various applications in medical engineering, due to its high precision and efficiency. With biologist James Thompson's development of first human stem cell lines in 1998 followed by transplantation of first laboratory-grown internal organs in 1999 and creation of the first bioprinter in 2003 by the University of Missouri when they printed spheroids without the need of scaffolds, 3-D bioprinting became more conventionally used in medical field than ever before. So far, scientists have been able to print mini organoids and organs-on-chips that have rendered practical insights into the functions of a human body. Pharmaceutical companies are using these models to test drugs before moving on to animal studies. However, a fully functional and structurally similar organ has not been printed yet. A team at University of Utah has reportedly printed ears and successfully transplanted those onto children born with defects that left their ears partially developed.
Today hydrogels are considered the preferred choice of bio-inks for 3-D bioprinting since they mimic cells' natural ECM while also containing strong mechanical properties capable of sustaining 3-D structures. Furthermore, hydrogels in conjunction with 3-D bioprinting allow researchers to produce different scaffolds which can be used to form new tissues or organs. 3-D printed tissues still face many challenges such as adding vasculature. Meanwhile, 3-D printing parts of tissues definitely will improve our understanding of the human body, thus accelerating both basic and clinical research.
Examples
[edit]
As defined by Langer and Vacanti,[5] examples of tissue engineering fall into one or more of three categories: "just cells," "cells and scaffold," or "tissue-inducing factors."
- In vitro meat: Edible artificial animal muscle tissue cultured in vitro.[citation needed]
- Bioartificial liver device, "Temporary Liver", Extracorporeal Liver Assist Device (ELAD): The human hepatocyte cell line (C3A line) in a hollow fiber bioreactor can mimic the hepatic function of the liver for acute instances of liver failure. A fully capable ELAD would temporarily function as an individual's liver, thus avoiding transplantation and allowing regeneration of their own liver.[citation needed]
- Artificial pancreas: Research involves using islet cells to regulate the body's blood sugar, particularly in cases of diabetes . Biochemical factors may be used to cause human pluripotent stem cells to differentiate (turn into) cells that function similarly to beta cells, which are in an islet cell in charge of producing insulin.[citation needed]
- Artificial bladders: Anthony Atala[9] (Wake Forest University) has successfully implanted artificial bladders, constructed of cultured cells seeded onto a bladder-shaped scaffold, into seven out of approximately 20 human test subjects as part of a long-term experiment.[10]
- Cartilage: lab-grown cartilage, cultured in vitro on a scaffold, was successfully used as an autologous transplant to repair patients' knees.[11]
- Scaffold-free cartilage: Cartilage generated without the use of exogenous scaffold material. In this methodology, all material in the construct is cellular produced directly by the cells.[12]
- Bioartificial heart: Doris Taylor's lab constructed a biocompatible rat heart by re-cellularising a de-cellularised rat heart. This scaffold and cells were placed in a bioreactor, where it matured to become a partially or fully transplantable organ.[13] the work was called a "landmark". The lab first stripped the cells away from a rat heart (a process called "decellularization") and then injected rat stem cells into the decellularized rat heart.[14]
- Tissue-engineered blood vessels:[15] Blood vessels that have been grown in a lab and can be used to repair damaged blood vessels without eliciting an immune response. Tissue engineered blood vessels have been developed by many different approaches. They could be implanted as pre-seeded cellularized blood vessels,[16] as acellular vascular grafts made with decellularized vessels or synthetic vascular grafts.[17]
- Artificial skin constructed from human skin cells embedded in a hydrogel, such as in the case of bio-printed constructs for battlefield burn repairs.[18]
- Artificial bone marrow: Bone marrow cultured in vitro to be transplanted serves as a "just cells" approach to tissue engineering.[19]
- Tissue engineered bone: A structural matrix can be composed of metals such as titanium, polymers of varying degradation rates, or certain types of ceramics.[20] Materials are often chosen to recruit osteoblasts to aid in reforming the bone and returning biological function.[21] Various types of cells can be added directly into the matrix to expedite the process.[20]
- Laboratory-grown penis: Decellularized scaffolds of rabbit penises were recellularised with smooth muscle and endothelial cells. The organ was then transplanted to live rabbits and functioned comparably to the native organ, suggesting potential as treatment for genital trauma.[22]
- Oral mucosa tissue engineering uses a cells and scaffold approach to replicate the 3 dimensional structure and function of oral mucosa.[citation needed]
Cells as building blocks
[edit]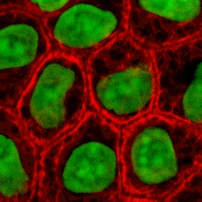
Cells are one of the main components for the success of tissue engineering approaches. Tissue engineering uses cells as strategies for creation/replacement of new tissue. Examples include fibroblasts used for skin repair or renewal,[23] chondrocytes used for cartilage repair (MACI–FDA approved product), and hepatocytes used in liver support systems
Cells can be used alone or with support matrices for tissue engineering applications. An adequate environment for promoting cell growth, differentiation, and integration with the existing tissue is a critical factor for cell-based building blocks.[24] Manipulation of any of these cell processes create alternative avenues for the development of new tissue (e.g., cell reprogramming - somatic cells, vascularization).[citation needed]
Isolation
[edit]Techniques for cell isolation depend on the cell source. Centrifugation and apheresis are techniques used for extracting cells from biofluids (e.g., blood). Whereas digestion processes, typically using enzymes to remove the extracellular matrix (ECM), are required prior to centrifugation or apheresis techniques to extract cells from tissues/organs. Trypsin and collagenase are the most common enzymes used for tissue digestion. While trypsin is temperature dependent, collagenase is less sensitive to changes in temperature.[citation needed]
Cell sources
[edit]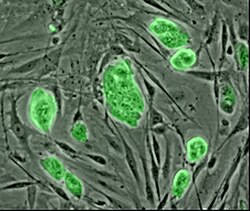
Primary cells are those directly isolated from host tissue. These cells provide an ex-vivo model of cell behavior without any genetic, epigenetic, or developmental changes; making them a closer replication of in-vivo conditions than cells derived from other methods.[25] This constraint however, can also make studying them difficult. These are mature cells, often terminally differentiated, meaning that for many cell types proliferation is difficult or impossible. Additionally, the microenvironments these cells exist in are highly specialized, often making replication of these conditions difficult.[26]
Secondary cells A portion of cells from a primary culture is moved to a new repository/vessel to continue being cultured. Medium from the primary culture is removed, the cells that are desired to be transferred are obtained, and then cultured in a new vessel with fresh growth medium.[27] A secondary cell culture is useful in order to ensure that cells have both the room and nutrients that they require to grow. Secondary cultures are most notably used in any scenario in which a larger quantity of cells than can be found in the primary culture is desired. Secondary cells share the constraints of primary cells (see above) but have an added risk of contamination when transferring to a new vessel.[citation needed]
Genetic classifications of cells
[edit]Autologous: The donor and the recipient of the cells are the same individual. Cells are harvested, cultured or stored, and then reintroduced to the host. As a result of the host's own cells being reintroduced, an antigenic response is not elicited. The body's immune system recognizes these re-implanted cells as its own, and does not target them for attack. Autologous cell dependence on host cell health and donor site morbidity may be deterrents to their use. Adipose-derived and bone marrow-derived mesenchymal stem cells are commonly autologous in nature, and can be used in a myriad of ways, from helping repair skeletal tissue to replenishing beta cells in diabetic patients.[28][29][30][31]
Allogenic: Cells are obtained from the body of a donor of the same species as the recipient. While there are some ethical constraints to the use of human cells for in vitro studies (i.e. human brain tissue chimera development[32]), the employment of dermal fibroblasts from human foreskin demonstrates an immunologically safe and thus a viable choice for allogenic tissue engineering of the skin.
Xenogenic: These cells are derived isolated cells from alternate species from the recipient. A notable example of xenogeneic tissue utilization is cardiovascular implant construction via animal cells. Chimeric human-animal farming raises ethical concerns around the potential for improved consciousness from implanting human organs in animals.[33]
Syngeneic or isogenic: These cells describe those borne from identical genetic code. This imparts an immunologic benefit similar to autologous cell lines (see above).[34] Autologous cells can be considered syngenic, but the classification also extends to non-autologously derived cells such as those from an identical twin, from genetically identical (cloned) research models, or induced stem cells (iSC)[35] as related to the donor.
Stem cells
[edit]Stem cells are undifferentiated cells with the ability to divide in culture and give rise to different forms of specialized cells. Stem cells are divided into "adult" and "embryonic" stem cells according to their source. While there is still a large ethical debate related to the use of embryonic stem cells, it is thought that another alternative source – induced pluripotent stem cells – may be useful for the repair of diseased or damaged tissues, or may be used to grow new organs.
Totipotent cells are stem cells which can divide into further stem cells or differentiate into any cell type in the body, including extra-embryonic tissue.
Pluripotent cells are stem cells which can differentiate into any cell type in the body except extra-embryonic tissue. induced pluripotent stem cells (iPSCs) are subclass of pluripotent stem cells resembling embryonic stem cells (ESCs) that have been derived from adult differentiated cells. iPSCs are created by altering the expression of transcriptional factors in adult cells until they become like embryonic stem cells.
Multipotent stem cells can be differentiated into any cell within the same class, such as blood or bone. A common example of multipotent cells is Mesenchymal stem cells (MSCs).
Scaffolds
[edit]Scaffolds are materials that have been engineered to cause desirable cellular interactions to contribute to the formation of new functional tissues for medical purposes. Cells are often 'seeded' into these structures capable of supporting three-dimensional tissue formation. Scaffolds mimic the extracellular matrix of the native tissue, recapitulating the in vivo milieu and allowing cells to influence their own microenvironments. They usually serve at least one of the following purposes: allowing cell attachment and migration, delivering and retaining cells and biochemical factors, enabling diffusion of vital cell nutrients and expressed products, and exerting certain mechanical and biological influences to modify the behaviour of the cell phase.[citation needed]
In 2009, an interdisciplinary team led by the thoracic surgeon Thorsten Walles implanted the first bioartificial transplant that provides an innate vascular network for post-transplant graft supply successfully into a patient awaiting tracheal reconstruction.[36]
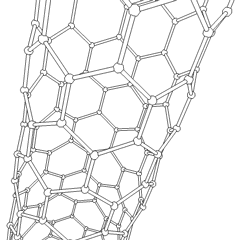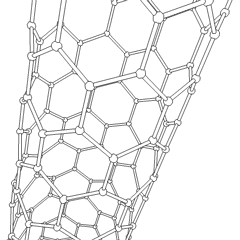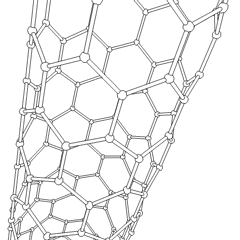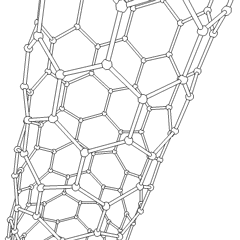
To achieve the goal of tissue reconstruction, scaffolds must meet some specific requirements. High porosity and adequate pore size are necessary to facilitate cell seeding and diffusion throughout the whole structure of both cells and nutrients. Biodegradability is often an essential factor since scaffolds should preferably be absorbed by the surrounding tissues without the necessity of surgical removal. The rate at which degradation occurs has to coincide as much as possible with the rate of tissue formation: this means that while cells are fabricating their own natural matrix structure around themselves, the scaffold is able to provide structural integrity within the body and eventually it will break down leaving the newly formed tissue which will take over the mechanical load. Injectability is also important for clinical uses. Recent research on organ printing is showing how crucial a good control of the 3D environment is to ensure reproducibility of experiments and offer better results.[citation needed]
Materials
[edit]Material selection is an essential aspect of producing a scaffold. The materials utilized can be natural or synthetic and can be biodegradable or non-biodegradable. Additionally, they must be biocompatible, meaning that they do not cause any adverse effects to cells.[38] Silicone, for example, is a synthetic, non-biodegradable material commonly used as a drug delivery material,[39][40] while gelatin is a biodegradable, natural material commonly used in cell-culture scaffolds[41][42][43]
The material needed for each application is different, and dependent on the desired mechanical properties of the material. Tissue engineering of long bone defects for example, will require a rigid scaffold with a compressive strength similar to that of cortical bone (100-150 MPa), which is much higher compared to a scaffold for skin regeneration.[44][45]
There are a few versatile synthetic materials used for many different scaffold applications. One of these commonly used materials is polylactic acid (PLA), a synthetic polymer. PLA – polylactic acid. This is a polyester which degrades within the human body to form lactic acid, a naturally occurring chemical which is easily removed from the body. Similar materials are polyglycolic acid (PGA) and polycaprolactone (PCL): their degradation mechanism is similar to that of PLA, but PCL degrades slower and PGA degrades faster.[citation needed] PLA is commonly combined with PGA to create poly-lactic-co-glycolic acid (PLGA). This is especially useful because the degradation of PLGA can be tailored by altering the weight percentages of PLA and PGA: More PLA – slower degradation, more PGA – faster degradation. This tunability, along with its biocompatibility, makes it an extremely useful material for scaffold creation.[46]
Scaffolds may also be constructed from natural materials: in particular different derivatives of the extracellular matrix have been studied to evaluate their ability to support cell growth. Protein based materials – such as collagen, or fibrin, and polysaccharidic materials- like chitosan[47] or glycosaminoglycans (GAGs), have all proved suitable in terms of cell compatibility. Among GAGs, hyaluronic acid, possibly in combination with cross linking agents (e.g. glutaraldehyde, water-soluble carbodiimide, etc.), is one of the possible choices as scaffold material. Due to the covalent attachment of thiol groups to these polymers, they can crosslink via disulfide bond formation.[48] The use of thiolated polymers (thiomers) as scaffold material for tissue engineering was initially introduced at the 4th Central European Symposium on Pharmaceutical Technology in Vienna 2001.[49] As thiomers are biocompatible, exhibit cellular mimicking properties and efficiently support proliferation and differentiation of various cell types, they are extensively used as scaffolds for tissue engineering.[50][51][52] Furthermore thiomers such as thiolated hyaluronic acid[53] and thiolated chitosan[54] were shown to exhibit wound healing properties and are subject of numerous clinical trials.[55] Additionally, a fragment of an extracellular matrix protein, such as the RGD peptide, can be coupled to a non-bioactive material to promote cell attachment.[56] Another form of scaffold is decellularized tissue. This is a process where chemicals are used to extracts cells from tissues, leaving just the extracellular matrix. This has the benefit of a fully formed matrix specific to the desired tissue type. However, the decellurised scaffold may present immune problems with future introduced cells.[citation needed]
Synthesis
[edit]This section needs additional citations for verification. (August 2024) |


A number of different methods have been described in the literature for preparing porous structures to be employed as tissue engineering scaffolds. Each of these techniques presents its own advantages, but none are free of drawbacks.
Nanofiber self-assembly
[edit]Molecular self-assembly is one of the few methods for creating biomaterials with properties similar in scale and chemistry to that of the natural in vivo extracellular matrix (ECM), a crucial step toward tissue engineering of complex tissues.[57] Moreover, these hydrogel scaffolds have shown superiority in in vivo toxicology and biocompatibility compared to traditional macro-scaffolds and animal-derived materials.
Textile technologies
[edit]These techniques include all the approaches that have been successfully employed for the preparation of non-woven meshes of different polymers. In particular, non-woven polyglycolide structures have been tested for tissue engineering applications: such fibrous structures have been found useful to grow different types of cells. The principal drawbacks are related to the difficulties in obtaining high porosity and regular pore size.
Solvent casting and particulate leaching
[edit]Solvent casting and particulate leaching (SCPL) allows for the preparation of structures with regular porosity, but with limited thickness. First, the polymer is dissolved into a suitable organic solvent (e.g. polylactic acid could be dissolved into dichloromethane), then the solution is cast into a mold filled with porogen particles. Such porogen can be an inorganic salt like sodium chloride, crystals of saccharose, gelatin spheres or paraffin spheres. The size of the porogen particles will affect the size of the scaffold pores, while the polymer to porogen ratio is directly correlated to the amount of porosity of the final structure. After the polymer solution has been cast the solvent is allowed to fully evaporate, then the composite structure in the mold is immersed in a bath of a liquid suitable for dissolving the porogen: water in the case of sodium chloride, saccharose and gelatin or an aliphatic solvent like hexane for use with paraffin. Once the porogen has been fully dissolved, a porous structure is obtained. Other than the small thickness range that can be obtained, another drawback of SCPL lies in its use of organic solvents which must be fully removed to avoid any possible damage to the cells seeded on the scaffold.
Gas foaming
[edit]To overcome the need to use organic solvents and solid porogens, a technique using gas as a porogen has been developed. First, disc-shaped structures made of the desired polymer are prepared by means of compression molding using a heated mold. The discs are then placed in a chamber where they are exposed to high pressure CO2 for several days. The pressure inside the chamber is gradually restored to atmospheric levels. During this procedure the pores are formed by the carbon dioxide molecules that abandon the polymer, resulting in a sponge-like structure. The main problems resulting from such a technique are caused by the excessive heat used during compression molding (which prohibits the incorporation of any temperature labile material into the polymer matrix) and by the fact that the pores do not form an interconnected structure.
Emulsification freeze-drying
[edit]This technique does not require the use of a solid porogen like SCPL. First, a synthetic polymer is dissolved into a suitable solvent (e.g. polylactic acid in dichloromethane) then water is added to the polymeric solution and the two liquids are mixed in order to obtain an emulsion. Before the two phases can separate, the emulsion is cast into a mold and quickly frozen by means of immersion into liquid nitrogen. The frozen emulsion is subsequently freeze-dried to remove the dispersed water and the solvent, thus leaving a solidified, porous polymeric structure. While emulsification and freeze-drying allow for a faster preparation when compared to SCPL (since it does not require a time-consuming leaching step), it still requires the use of solvents. Moreover, pore size is relatively small and porosity is often irregular. Freeze-drying by itself is also a commonly employed technique for the fabrication of scaffolds. In particular, it is used to prepare collagen sponges: collagen is dissolved into acidic solutions of acetic acid or hydrochloric acid that are cast into a mold, frozen with liquid nitrogen and then lyophilized.
Thermally induced phase separation
[edit]Similar to the previous technique, the TIPS phase separation procedure requires the use of a solvent with a low melting point that is easy to sublime. For example, dioxane could be used to dissolve polylactic acid, then phase separation is induced through the addition of a small quantity of water: a polymer-rich and a polymer-poor phase are formed. Following cooling below the solvent melting point and some days of vacuum-drying to sublime the solvent, a porous scaffold is obtained. Liquid-liquid phase separation presents the same drawbacks of emulsification/freeze-drying.[58]
Electrospinning
[edit]Electrospinning is a highly versatile technique that can be used to produce continuous fibers ranging in diameter from a few microns to a few nanometers. In a typical electrospinning set-up, the desired scaffold material is dissolved within a solvent and placed within a syringe. This solution is fed through a needle and a high voltage is applied to the tip and to a conductive collection surface. The buildup of electrostatic forces within the solution causes it to eject a thin fibrous stream towards the oppositely charged or grounded collection surface. During this process the solvent evaporates, leaving solid fibers leaving a highly porous network. This technique is highly tunable, with variation to solvent, voltage, working distance (distance from the needle to collection surface), flow rate of solution, solute concentration, and collection surface. This allows for precise control of fiber morphology.
On a commercial level however, due to scalability reasons, there are 40 or sometimes 96 needles involved operating at once. The bottle-necks in such set-ups are: 1) Maintaining the aforementioned variables uniformly for all of the needles and 2) formation of "beads" in single fibers that we as engineers, want to be of a uniform diameter. By modifying variables such as the distance to collector, magnitude of applied voltage, or solution flow rate – researchers can dramatically change the overall scaffold architecture.
Historically, research on electrospun fibrous scaffolds dates back to at least the late 1980s when Simon showed that electrospinning could be used to produce nano- and submicron-scale fibrous scaffolds from polymer solutions specifically intended for use as in vitro cell and tissue substrates. This early use of electrospun lattices for cell culture and tissue engineering showed that various cell types would adhere to and proliferate upon polycarbonate fibers. It was noted that as opposed to the flattened morphology typically seen in 2D culture, cells grown on the electrospun fibers exhibited a more rounded 3-dimensional morphology generally observed of tissues in vivo.[59]
CAD/CAM technologies
[edit]Because most of the above techniques are limited when it comes to the control of porosity and pore size, computer assisted design and manufacturing techniques have been introduced to tissue engineering. First, a three-dimensional structure is designed using CAD software. The porosity can be tailored using algorithms within the software.[60] The scaffold is then realized by using ink-jet printing of polymer powders or through Fused Deposition Modeling of a polymer melt.[61]
A 2011 study by El-Ayoubi et al. investigated "3D-plotting technique to produce (biocompatible and biodegradable) poly-L-Lactide macroporous scaffolds with two different pore sizes" via solid free-form fabrication (SSF) with computer-aided-design (CAD), to explore therapeutic articular cartilage replacement as an "alternative to conventional tissue repair".[62] The study found the smaller the pore size paired with mechanical stress in a bioreactor (to induce in vivo-like conditions), the higher the cell viability in potential therapeutic functionality via decreasing recovery time and increasing transplant effectiveness.[62]
Laser-assisted bioprinting
[edit]In a 2012 study,[63] Koch et al. focused on whether Laser-assisted BioPrinting (LaBP) can be used to build multicellular 3D patterns in natural matrix, and whether the generated constructs are functioning and forming tissue. LaBP arranges small volumes of living cell suspensions in set high-resolution patterns.[63] The investigation was successful, the researchers foresee that "generated tissue constructs might be used for in vivo testing by implanting them into animal models" (14). As of this study, only human skin tissue has been synthesized, though researchers project that "by integrating further cell types (e.g. melanocytes, Schwann cells, hair follicle cells) into the printed cell construct, the behavior of these cells in a 3D in vitro microenvironment similar to their natural one can be analyzed", which is useful for drug discovery and toxicology studies.[63]
Self-assembled recombinant spider silk nanomembranes
[edit]Gustafsson et al.[64] demonstrated free‐standing, bioactive membranes of cm-sized area, but only 250 nm thin, that were formed by self‐assembly of spider silk at the interface of an aqueous solution. The membranes uniquely combine nanoscale thickness, biodegradability, ultrahigh strain and strength, permeability to proteins and promote rapid cell adherence and proliferation. They demonstrated growing a coherent layer of keratinocytes. These spider silk nanomembranes have also been used to create a static in-vitro model of a blood vessel.[65]
Tissue engineering in situ
[edit]In situ tissue regeneration is defined as the implantation of biomaterials (alone or in combination with cells and/or biomolecules) into the tissue defect, using the surrounding microenvironment of the organism as a natural bioreactor.[66] This approach has found application in bone regeneration,[67] allowing the formation of cell-seeded constructs directly in the operating room.[68]
Assembly methods
[edit]A persistent problem within tissue engineering is mass transport limitations. Engineered tissues generally lack an initial blood supply, thus making it difficult for any implanted cells to obtain sufficient oxygen and nutrients to survive, or function properly.[citation needed]
Self-assembly
[edit]Self-assembly methods have been shown to be promising methods for tissue engineering. Self-assembly methods have the advantage of allowing tissues to develop their own extracellular matrix, resulting in tissue that better recapitulates biochemical and biomechanical properties of native tissue. Self-assembling engineered articular cartilage was introduced by Jerry Hu and Kyriacos A. Athanasiou in 2006[69] and applications of the process have resulted in engineered cartilage approaching the strength of native tissue.[70] Self-assembly is a prime technology to get cells grown in a lab to assemble into three-dimensional shapes. To break down tissues into cells, researchers first have to dissolve the extracellular matrix that normally binds them together. Once cells are isolated, they must form the complex structures that make up our natural tissues.[citation needed]
Liquid-based template assembly
[edit]The air-liquid surface established by Faraday waves is explored as a template to assemble biological entities for bottom-up tissue engineering. This liquid-based template can be dynamically reconfigured in a few seconds, and the assembly on the template can be achieved in a scalable and parallel manner. Assembly of microscale hydrogels, cells, neuron-seeded micro-carrier beads, cell spheroids into various symmetrical and periodic structures was demonstrated with good cell viability. Formation of 3-D neural network was achieved after 14-day tissue culture.[71]
Additive manufacturing
[edit]It might be possible to print organs, or possibly entire organisms using additive manufacturing techniques. A recent innovative method of construction uses an ink-jet mechanism to print precise layers of cells in a matrix of thermo-reversible gel. Endothelial cells, the cells that line blood vessels, have been printed in a set of stacked rings. When incubated, these fused into a tube.[61][72] This technique has been referred to as "bioprinting" within the field as it involves the printing of biological components in a structure resembling the organ of focus.[citation needed]
The field of three-dimensional and highly accurate models of biological systems is pioneered by multiple projects and technologies including a rapid method for creating tissues and even whole organs involve a 3-D printer that can bio-print the scaffolding and cells layer by layer into a working tissue sample or organ. The device is presented in a TED talk by Dr. Anthony Atala, M.D. the Director of the Wake Forest Institute for Regenerative Medicine, and the W.H. Boyce Professor and Chair of the Department of Urology at Wake Forest University, in which a kidney is printed on stage during the seminar and then presented to the crowd.[73][74][75] It is anticipated that this technology will enable the production of livers in the future for transplantation and theoretically for toxicology and other biological studies as well.[citation needed]
In 2015 Multi-Photon Processing (MPP) was employed for in vivo experiments by engineering artificial cartilage constructs. An ex vivo histological examination showed that certain pore geometry and the pre-growing of chondrocytes (Cho) prior to implantation significantly improves the performance of the created 3-D scaffolds. The achieved biocompatibility was comparable to the commercially available collagen membranes. The successful outcome of this study supports the idea that hexagonal-pore-shaped hybrid organic-inorganic micro-structured scaffolds in combination with Cho seeding may be successfully implemented for cartilage tissue engineering.[76]
Recently, tissue engineering has advanced with a focus on vascularization. Using Two-Photon Polymerization-based additive manufacturing, synthetic 3D microvessel networks are created from tubular hydrogel structures. These networks can perfuse tissues several cubic millimeters in size, enabling long-term viability and cell growth in vitro. This innovation marks a significant step forward in tissue engineering, facilitating the development of complex human tissue models.[77]
Scaffolding
[edit]In 2013, using a 3-D scaffolding of Matrigel in various configurations, substantial pancreatic organoids was produced in vitro. Clusters of small numbers of cells proliferated into 40,000 cells within one week. The clusters transform into cells that make either digestive enzymes or hormones like insulin, self-organizing into branched pancreatic organoids that resemble the pancreas.[78]
The cells are sensitive to the environment, such as gel stiffness and contact with other cells. Individual cells do not thrive; a minimum of four proximate cells was required for subsequent organoid development. Modifications to the medium composition produced either hollow spheres mainly composed of pancreatic progenitors, or complex organoids that spontaneously undergo pancreatic morphogenesis and differentiation. Maintenance and expansion of pancreatic progenitors require active Notch and FGF signaling, recapitulating in vivo niche signaling interactions.[78]
The organoids were seen as potentially offering mini-organs for drug testing and for spare insulin-producing cells.[78]
Aside from Matrigel 3-D scaffolds, other collagen gel systems have been developed. Collagen/hyaluronic acid scaffolds have been used for modeling the mammary gland In Vitro while co-coculturing epithelial and adipocyte cells. The HyStem kit is another 3-D platform containing ECM components and hyaluronic acid that has been used for cancer research. Additionally, hydrogel constituents can be chemically modified to assist in crosslinking and enhance their mechanical properties.[citation needed]
Tissue culture
[edit]In many cases, creation of functional tissues and biological structures in vitro requires extensive culturing to promote survival, growth and inducement of functionality. In general, the basic requirements of cells must be maintained in culture, which include oxygen, pH, humidity, temperature, nutrients and osmotic pressure maintenance.[citation needed]
Tissue engineered cultures also present additional problems in maintaining culture conditions. In standard cell culture, diffusion is often the sole means of nutrient and metabolite transport. However, as a culture becomes larger and more complex, such as the case with engineered organs and whole tissues, other mechanisms must be employed to maintain the culture, such as the creation of capillary networks within the tissue.[citation needed]

Another issue with tissue culture is introducing the proper factors or stimuli required to induce functionality. In many cases, simple maintenance culture is not sufficient. Growth factors, hormones, specific metabolites or nutrients, chemical and physical stimuli are sometimes required. For example, certain cells respond to changes in oxygen tension as part of their normal development, such as chondrocytes, which must adapt to low oxygen conditions or hypoxia during skeletal development. Others, such as endothelial cells, respond to shear stress from fluid flow, which is encountered in blood vessels. Mechanical stimuli, such as pressure pulses seem to be beneficial to all kind of cardiovascular tissue such as heart valves, blood vessels or pericardium.[citation needed]
Bioreactors
[edit]In tissue engineering, a bioreactor is a device that attempts to simulate a physiological environment in order to promote cell or tissue growth in vitro. A physiological environment can consist of many different parameters such as temperature, pressure, oxygen or carbon dioxide concentration, or osmolality of fluid environment, and it can extend to all kinds of biological, chemical or mechanical stimuli. Therefore, there are systems that may include the application of forces such as electromagnetic forces, mechanical pressures, or fluid pressures to the tissue. These systems can be two- or three-dimensional setups. Bioreactors can be used in both academic and industry applications. General-use and application-specific bioreactors are also commercially available, which may provide static chemical stimulation or a combination of chemical and mechanical stimulation.[citation needed]
Cell proliferation and differentiation are largely influenced by mechanical[79] and biochemical[80] cues in the surrounding extracellular matrix environment. Bioreactors are typically developed to replicate the specific physiological environment of the tissue being grown (e.g., flex and fluid shearing for heart tissue growth).[81] This can allow specialized cell lines to thrive in cultures replicating their native environments, but it also makes bioreactors attractive tools for culturing stem cells. A successful stem-cell-based bioreactor is effective at expanding stem cells with uniform properties and/or promoting controlled, reproducible differentiation into selected mature cell types.[82]
There are a variety of bioreactors designed for 3D cell cultures. There are small plastic cylindrical chambers, as well as glass chambers, with regulated internal humidity and moisture specifically engineered for the purpose of growing cells in three dimensions.[83] The bioreactor uses bioactive synthetic materials such as polyethylene terephthalate membranes to surround the spheroid cells in an environment that maintains high levels of nutrients.[84][85] They are easy to open and close, so that cell spheroids can be removed for testing, yet the chamber is able to maintain 100% humidity throughout.[86] This humidity is important to achieve maximum cell growth and function. The bioreactor chamber is part of a larger device that rotates to ensure equal cell growth in each direction across three dimensions.[86]
QuinXell Technologies now under Quintech Life Sciences from Singapore has developed a bioreactor known as the TisXell Biaxial Bioreactor which is specially designed for the purpose of tissue engineering. It is the first bioreactor in the world to have a spherical glass chamber with biaxial rotation; specifically to mimic the rotation of the fetus in the womb; which provides a conducive environment for the growth of tissues.[87]
Multiple forms of mechanical stimulation have also been combined into a single bioreactor. Using gene expression analysis, one academic study found that applying a combination of cyclic strain and ultrasound stimulation to pre-osteoblast cells in a bioreactor accelerated matrix maturation and differentiation.[88] The technology of this combined stimulation bioreactor could be used to grow bone cells more quickly and effectively in future clinical stem cell therapies.[89]
MC2 Biotek has also developed a bioreactor known as ProtoTissue[83] that uses gas exchange to maintain high oxygen levels within the cell chamber; improving upon previous bioreactors, since the higher oxygen levels help the cell grow and undergo normal cell respiration.[90]
Active areas of research on bioreactors includes increasing production scale and refining the physiological environment, both of which could improve the efficiency and efficacy of bioreactors in research or clinical use. Bioreactors are currently used to study, among other things, cell and tissue level therapies, cell and tissue response to specific physiological environment changes, and development of disease and injury.[citation needed]
Long fiber generation
[edit]In 2013, a group from the University of Tokyo developed cell laden fibers up to a meter in length and on the order of 100 μm in size.[91] These fibers were created using a microfluidic device that forms a double coaxial laminar flow. Each 'layer' of the microfluidic device (cells seeded in ECM, a hydrogel sheath, and finally a calcium chloride solution). The seeded cells culture within the hydrogel sheath for several days, and then the sheath is removed with viable cell fibers. Various cell types were inserted into the ECM core, including myocytes, endothelial cells, nerve cell fibers, and epithelial cell fibers. This group then showed that these fibers can be woven together to fabricate tissues or organs in a mechanism similar to textile weaving. Fibrous morphologies are advantageous in that they provide an alternative to traditional scaffold design, and many organs (such as muscle) are composed of fibrous cells.
Bioartificial organs
[edit]An artificial organ is an engineered device that can be extra corporeal or implanted to support impaired or failing organ systems.[92] Bioartificial organs are typically created with the intent to restore critical biological functions like in the replacement of diseased hearts and lungs, or provide drastic quality of life improvements like in the use of engineered skin on burn victims.[92] While some examples of bioartificial organs are still in the research stage of development due to the limitations involved with creating functional organs, others are currently being used in clinical settings experimentally and commercially.[93]
Lung
[edit]Extracorporeal membrane oxygenation (ECMO) machines, otherwise known as heart and lung machines, are an adaptation of cardiopulmonary bypass techniques that provide heart and lung support.[94] It is used primarily to support the lungs for a prolonged but still temporary timeframe (1–30 days) and allow for recovery from reversible diseases.[94] Robert Bartlett is known as the father of ECMO and performed the first treatment of a newborn using an ECMO machine in 1975.[95]
Skin
Tissue-engineered skin is a type of bioartificial organ that is often used to treat burns, diabetic foot ulcers, or other large wounds that cannot heal well on their own. Artificial skin can be made from autografts, allografts, and xenografts. Autografted skin comes from a patient's own skin, which allows the dermis to have a faster healing rate, and the donor site can be re-harvested a few times. Allograft skin often comes from cadaver skin and is mostly used to treat burn victims. Lastly, xenografted skin comes from animals and provides a temporary healing structure for the skin. They assist in dermal regeneration, but cannot become part of the host skin.[23] Tissue-engineered skin is now available in commercial products. Integra, originally used to only treat burns, consists of a collagen matrix and chondroitin sulfate that can be used as a skin replacement. The chondroitin sulfate functions as a component of proteoglycans, which helps to form the extracellular matrix.[96] Integra can be repopulated and revascularized while maintaining its dermal collagen architecture, making it a bioartificial organ[97] Dermagraft, another commercial-made tissue-engineered skin product, is made out of living fibroblasts. These fibroblasts proliferate and produce growth factors, collagen, and ECM proteins, that help build granulation tissue.[98]
Heart
[edit]Since the number of patients awaiting a heart transplant is continuously increasing over time, and the number of patients on the waiting list surpasses the organ availability,[99] artificial organs used as replacement therapy for terminal heart failure would help alleviate this difficulty. Artificial hearts are usually used to bridge the heart transplantation or can be applied as replacement therapy for terminal heart malfunction.[100] The total artificial heart (TAH), first introduced by Dr. Vladimir P. Demikhov in 1937,[101] emerged as an ideal alternative. Since then it has been developed and improved as a mechanical pump that provides long-term circulatory support and replaces diseased or damaged heart ventricles that cannot properly pump the blood, restoring thus the pulmonary and systemic flow.[102] Some of the current TAHs include AbioCor, an FDA-approved device that comprises two artificial ventricles and their valves, and does not require subcutaneous connections, and is indicated for patients with biventricular heart failure. In 2010 SynCardia released the portable freedom driver that allows patients to have a portable device without being confined to the hospital.[103]
Kidney
[edit]While kidney transplants are possible, renal failure is more often treated using an artificial kidney.[104] The first artificial kidneys and the majority of those currently in use are extracorporeal, such as with hemodialysis, which filters blood directly, or peritoneal dialysis, which filters via a fluid in the abdomen.[104][105] In order to contribute to the biological functions of a kidney such as producing metabolic factors or hormones, some artificial kidneys incorporate renal cells.[104][105] There has been progress in the way of making these devices smaller and more transportable, or even implantable . One challenge still to be faced in these smaller devices is countering the limited volume and therefore limited filtering capabilities.[104]
Bioscaffolds have also been introduced to provide a framework upon which normal kidney tissue can be regenerated. These scaffolds encompass natural scaffolds (e.g., decellularized kidneys,[106] collagen hydrogel,[107][108] or silk fibroin[109]), synthetic scaffolds (e.g., poly[lactic-co-glycolic acid][110][111] or other polymers), or a combination of two or more natural and synthetic scaffolds. These scaffolds can be implanted into the body either without cell treatment or after a period of stem cell seeding and incubation. In vitro and In vivo studies are being conducted to compare and optimize the type of scaffold and to assess whether cell seeding prior to implantation adds to the viability, regeneration and effective function of the kidneys. A recent systematic review and meta-analysis compared the results of published animal studies and identified that improved outcomes are reported with the use of hybrid (mixed) scaffolds and cell seeding;[112] however, the meta-analysis of these results were not in agreement with the evaluation of descriptive results from the review. Therefore, further studies involving larger animals and novel scaffolds, and more transparent reproduction of previous studies are advisable.
Biomimetics
[edit]This section's tone or style may not reflect the encyclopedic tone used on Wikipedia. (December 2019) |
Biomimetics is a field that aims to produce materials and systems that replicate those present in nature.[113] In the context of tissue engineering, this is a common approach used by engineers to create materials for these applications that are comparable to native tissues in terms of their structure, properties, and biocompatibility. Material properties are largely dependent on physical, structural, and chemical characteristics of that material. Subsequently, a biomimetic approach to system design will become significant in material integration, and a sufficient understanding of biological processes and interactions will be necessary. Replication of biological systems and processes may also be used in the synthesis of bio-inspired materials to achieve conditions that produce the desired biological material. Therefore, if a material is synthesized having the same characteristics of biological tissues both structurally and chemically, then ideally the synthesized material will have similar properties. This technique has an extensive history originating from the idea of using natural phenomenon as design inspiration for solutions to human problems. Many modern advancements in technology have been inspired by nature and natural systems, including aircraft, automobiles, architecture, and even industrial systems. Advancements in nanotechnology initiated the application of this technique to micro- and nano-scale problems, including tissue engineering. This technique has been used to develop synthetic bone tissues, vascular technologies, scaffolding materials and integration techniques, and functionalized nanoparticles.[113]
Constructing neural networks in soft material
[edit]In 2018, scientists at Brandeis University reported their research on soft material embedded with chemical networks which can mimic the smooth and coordinated behavior of neural tissue. This research was funded by the U.S. Army Research Laboratory.[114] The researchers presented an experimental system of neural networks, theoretically modeled as reaction-diffusion systems. Within the networks was an array of patterned reactors, each performing the Belousov-Zhabotinsky (BZ) reaction. These reactors could function on a nanoliter scale.[115]
The researchers state that the inspiration for their project was the movement of the blue ribbon eel. The eel's movements are controlled by electrical impulses determined by a class of neural networks called the central pattern generator. Central Pattern Generators function within the autonomic nervous system to control bodily functions such as respiration, movement, and peristalsis.[116]
Qualities of the reactor that were designed were the network topology, boundary conditions, initial conditions, reactor volume, coupling strength, and the synaptic polarity of the reactor (whether its behavior is inhibitory or excitatory).[116] A BZ emulsion system with a solid elastomer polydimethylsiloxane (PDMS) was designed. Both light and bromine permeable PDMS have been reported as viable methods to create a pacemaker for neural networks.[115]
Market
[edit]The history of the tissue engineering market can be divided into three major parts. The time before the crash of the biotech market in the early 2000s, the crash and the time afterward.
Beginning
[edit]Most early progress in tissue engineering research was done in the US. This is due to less strict regulations regarding stem cell research and more available funding than in other countries. This leads to the creation of academic startups many of them coming from Harvard or MIT. Examples are BioHybrid Technologies whose founder, Bill Chick, went to Harvard Medical School and focused on the creation of artificial pancreas. Another example would be Organogenesis Inc. whose founder went to MIT and worked on skin engineering products. Other companies with links to the MIT are TEI Biosciences, Therics and Guilford Pharmaceuticals.[8] The renewed interest in biotechnologies in the 1980s leads to many private investors investing in these new technologies even though the business models of these early startups were often not very clear and did not present a path to long term profitability.[117] Government sponsors were more restrained in their funding as tissue engineering was considered a high-risk investment.[8]
In the UK the market got off to a slower start even though the regulations on stem cell research were not strict as well. This is mainly due to more investors being less willing to invest in these new technologies which were considered to be high-risk investments.[117] Another problem faced by British companies was getting the NHS to pay for their products. This especially because the NHS runs a cost-effectiveness analysis on all supported products. Novel technologies often do not do well in this respect.[117]
In Japan, the regulatory situation was quite different. First cell cultivation was only allowed in a hospital setting and second academic scientists employed by state-owned universities were not allowed outside employment until 1998. Moreover, the Japanese authorities took longer to approve new drugs and treatments than there US and European counterparts.[117]
For these reasons in the early days of the Japanese market, the focus was mainly on getting products that were already approved elsewhere in Japan and selling them. Contrary to the US market the early actors in Japan were mainly big firms or sub-companies of such big firms, such as J-TEC, Menicon and Terumo, and not small startups.[117] After regulatory changes in 2014, which allowed cell cultivation outside of a hospital setting, the speed of research in Japan increased and Japanese companies also started to develop their own products.[117]
Crash
[edit]Soon after the big boom, the first problems started to appear. There were problems getting products approved by the FDA and if they got approved there were often difficulties in getting insurance providers to pay for the products and getting it accepted by health care providers.[117][118]
For example, organogenesis ran into problems marketing its product and integrating its product in the health system. This partially due to the difficulties of handling living cells and the increased difficulties faced by physicians in using these products over conventional methods.[117]
Another example would be Advanced Tissue Sciences Dermagraft skin product which could not create a high enough demand without reimbursements from insurance providers. Reasons for this were $4000 price-tag and the circumstance that Additionally Advanced Tissue Sciences struggled to get their product known by physicians.[117]
The above examples demonstrate how companies struggled to make profit. This, in turn, lead investors to lose patience and stopping further funding. In consequence, several Tissue Engineering companies such as Organogenesis and Advanced Tissue Sciences filed for bankruptcy in the early 2000s. At this time, these were the only ones having commercial skin products on the market.[118]
Reemergence
[edit]The technologies of the bankrupt or struggling companies were often bought by other companies which continued the development under more conservative business models.[118] Examples of companies who sold their products after folding were Curis[118] and Intercytex.[117]
Many of the companies abandoned their long-term goals of developing fully functional organs in favor of products and technologies that could turn a profit in the short run.[117] Examples of these kinds of products are products in the cosmetic and testing industry.
In other cases such as in the case of Advanced Tissue Sciences, the founders started new companies.[117]
In the 2010s the regulatory framework also started to facilitate faster time to market especially in the US as new centres and pathways were created by the FDA specifically aimed at products coming from living cells such as the Center for Biologics Evaluation and Research.[117]
The first tissue engineering products started to get commercially profitable in the 2010s.[118]
Regulation
[edit]In Europe, regulation is currently split into three areas of regulation: medical devices, medicinal products, and biologics. Tissue engineering products are often of hybrid nature, as they are often composed of cells and a supporting structure. While some products can be approved as medicinal products, others need to gain approval as medical devices.[119] Derksen explains in her thesis that tissue engineering researchers are sometimes confronted with regulation that does not fit the characteristics of tissue engineering.[120]
New regulatory regimes have been observed in Europe that tackle these issues.[121] An explanation for the difficulties in finding regulatory consensus in this matter is given by a survey conducted in the UK.[119] The authors attribute these problems to the close relatedness and overlap with other technologies such as xenotransplantation. It can therefore not be handled separately by regulatory bodies.[119] Regulation is further complicated by the ethical controversies associated with this and related fields of research (e.g. stem cells controversy, ethics of organ transplantation). The same survey as mentioned above[119] shows on the example of autologous cartilage transplantation that a specific technology can be regarded as 'pure' or 'polluted' by the same social actor.
Two regulatory movements are most relevant to tissue engineering in the European Union. These are Directive 2004/23/EC on standards of quality and safety for the sourcing and processing of human tissues[122] which was adopted by the European Parliament in 2004 and a proposed Human Tissue-Engineered Products regulation. The latter was developed under the auspices of the European Commission DG Enterprise and presented in Brussels in 2004.[123]
See also
[edit]- Biomedical engineering
- Biological engineering
- Biomolecular engineering
- Biochemical engineering
- Cell engineering
- Chemical engineering
- ECM Biomaterial
- In vivo bioreactor
- Induced stem cells
- Molecular processor
- Molecular self-assembly
- Muscle tissue engineering
- National Institutes of Health
- National Science Foundation
- Quality control in tissue engineering
- Regeneration in humans
- Soft tissues
- Thiomers
- Tissue Engineering and Regenerative Medicine International Society
- Tissue engineering of heart valves
- Xenotransplantation
Notes
[edit]- ^ Kim, Yu Seon; Smoak, Mollie M.; Melchiorri, Anthony J.; Mikos, Antonios G. (1 January 2019). "An Overview of the Tissue Engineering Market in the United States from 2011 to 2018". Tissue Engineering. Part A. 25 (1–2): 1–8. doi:10.1089/ten.tea.2018.0138. ISSN 1937-3341. PMC 6352506. PMID 30027831.
- ^ Whitney GA, Jayaraman K, Dennis JE, Mansour JM (February 2017). "Scaffold-free cartilage subjected to frictional shear stress demonstrates damage by cracking and surface peeling". Journal of Tissue Engineering and Regenerative Medicine. 11 (2): 412–24. doi:10.1002/term.1925. PMC 4641823. PMID 24965503.
- ^ "Langer Lab – MIT Department of Chemical Engineering". langerlab.mit.edu. Retrieved 13 March 2023.
- ^ "The Laboratory for Tissue Engineering and Organ Fabrication". Massachusetts General Hospital, Boston, MA. Archived from the original on 1 December 2016. Retrieved 31 October 2009.
- ^ a b Langer R, Vacanti JP (May 1993). "Tissue engineering". Science. 260 (5110): 920–26. Bibcode:1993Sci...260..920L. doi:10.1126/science.8493529. PMID 8493529.
- ^ a b MacArthur BD, Oreffo RO (January 2005). "Bridging the gap". Nature. 433 (7021): 19. Bibcode:2005Natur.433...19M. doi:10.1038/433019a. PMID 15635390. S2CID 2683429.
- ^ Thomas D, Singh D (July 2019). "Novel techniques of engineering 3D vasculature tissue for surgical procedures". American Journal of Surgery. 218 (1): 235–236. doi:10.1016/j.amjsurg.2018.06.004. PMID 29929908. S2CID 49350846.
- ^ a b c d Viola J, Lal B, Grad O (14 October 2003). The Emergence of Tissue Engineering as a Research Field (PDF) (Report). Abt Associates Inc. "Alternate webpage". Retrieved 28 April 2006 – via nsf.gov.
- ^ "Anthony Atala, MD". Wake Forest Baptist Health.
- ^ "Doctors grow organs from patients' own cells". CNN. 3 April 2006.
- ^ Simonite T (5 July 2006). "Lab-grown cartilage fixes damaged knees". New Scientist.
- ^ Whitney GA, Mera H, Weidenbecher M, Awadallah A, Mansour JM, Dennis JE (August 2012). "Methods for producing scaffold-free engineered cartilage sheets from auricular and articular chondrocyte cell sources and attachment to porous tantalum". BioResearch Open Access. 1 (4): 157–65. doi:10.1089/biores.2012.0231. PMC 3559237. PMID 23514898.
- ^ Ott HC, Matthiesen TS, Goh SK, Black LD, Kren SM, Netoff TI, Taylor DA (February 2008). "Perfusion-decellularized matrix: using nature's platform to engineer a bioartificial heart". Nature Medicine. 14 (2): 213–21. doi:10.1038/nm1684. PMID 18193059. S2CID 12765933.
- ^ Altman LK (13 January 2008). "Researchers Create New Rat Heart in Lab". The New York Times.
- ^ Zilla P, Greisler H (1999). Tissue Engineering of Vascular Prosthetic Grafts. R.G. Landes Company. ISBN 978-1-57059-549-3.[page needed]
- ^ Naegeli KM, Kural MH, Li Y, Wang J, Hugentobler EA, Niklason LE (June 2022). "Bioengineering Human Tissues and the Future of Vascular Replacement". Circulation Research. 131 (1): 109–126. doi:10.1161/CIRCRESAHA.121.319984. PMC 9213087. PMID 35737757.
- ^ Chang WG, Niklason LE (27 March 2017). "A short discourse on vascular tissue engineering". npj Regenerative Medicine. 2 (1) 7: 1–8. doi:10.1038/s41536-017-0011-6. PMC 5649630. PMID 29057097.
- ^ "Tissue Engineering". microfab.com.
- ^ "Bone in a bottle: Attempts to create artificial bone marrow have failed until now". The Economist. 7 January 2009.
- ^ a b Amini AR, Laurencin CT, Nukavarapu SP (2012). "Bone tissue engineering: recent advances and challenges". Critical Reviews in Biomedical Engineering. 40 (5): 363–408. doi:10.1615/critrevbiomedeng.v40.i5.10. PMC 3766369. PMID 23339648.
- ^ Thomas D (16 April 2021). "3D printing cross-linkable calcium phosphate biocomposites for biocompatible surgical implantation". Bioprinting. 22 e00141. doi:10.1016/j.bprint.2021.e00141. ISSN 2405-8866. S2CID 234851322.
- ^ Choi CQ (9 November 2009). "Artificial Penis Tissue Proves Promising in Lab Tests". Live Science.
- ^ a b Vig K, Chaudhari A, Tripathi S, Dixit S, Sahu R, Pillai S, et al. (April 2017). "Advances in Skin Regeneration Using Tissue Engineering". International Journal of Molecular Sciences. 18 (4): 789. doi:10.3390/ijms18040789. PMC 5412373. PMID 28387714.
- ^ Heath CA (January 2000). "Cells for tissue engineering". Trends in Biotechnology. 18 (1): 17–19. doi:10.1016/S0167-7799(99)01396-7. PMID 10631775.
- ^ Welser, Jennifer, et al. (November 2015). "Primary Cells Versus Cell Lines". ScienCell Research Laboratories.
- ^ Buttery LD, Bishop AE (2005). "Introduction to tissue engineering". Biomaterials, Artificial Organs and Tissue Engineering. Vol. 279. pp. 193–200. doi:10.1533/9781845690861.4.193. ISBN 978-1-85573-737-2.
- ^ Malik, Parth, et al. (January 2023). "Mammalian Cell Culture Types and Guidelines of Their Maintenance". Practical Approach to Mammalian Cell and Organ Culture. Springer Nature. pp. 233–259. doi:10.1007/978-981-19-1731-8_6-2. ISBN 978-981-19-1730-1.
- ^ Parekkadan B, Milwid JM (August 2010). "Mesenchymal stem cells as therapeutics". Annual Review of Biomedical Engineering. 12: 87–117. doi:10.1146/annurev-bioeng-070909-105309. PMC 3759519. PMID 20415588.
- ^ Domínguez-Bendala J, Lanzoni G, Inverardi L, Ricordi C (January 2012). "Concise review: mesenchymal stem cells for diabetes". Stem Cells Translational Medicine. 1 (1): 59–63. doi:10.5966/sctm.2011-0017. PMC 3727686. PMID 23197641.
- ^ Bara JJ, Richards RG, Alini M, Stoddart MJ (July 2014). "Concise review: Bone marrow-derived mesenchymal stem cells change phenotype following in vitro culture: implications for basic research and the clinic". Stem Cells. 32 (7): 1713–23. doi:10.1002/stem.1649. PMID 24449458. S2CID 30744973.
- ^ Minteer D, Marra KG, Rubin JP (2013). "Adipose-derived mesenchymal stem cells: biology and potential applications". Advances in Biochemical Engineering/Biotechnology. 129: 59–71. doi:10.1007/10_2012_146. ISBN 978-3-642-35670-4. PMID 22825719.
- ^ Farahany NA, Greely HT, Hyman S, Koch C, Grady C, Pașca SP, et al. (April 2018). "The ethics of experimenting with human brain tissue". Nature. 556 (7702): 429–32. Bibcode:2018Natur.556..429F. doi:10.1038/d41586-018-04813-x. PMC 6010307. PMID 29691509.
- ^ Bourret R, Martinez E, Vialla F, Giquel C, Thonnat-Marin A, De Vos J (June 2016). "Human-animal chimeras: ethical issues about farming chimeric animals bearing human organs". Stem Cell Research & Therapy. 7 (1) 87. doi:10.1186/s13287-016-0345-9. PMC 4928294. PMID 27356872.
- ^ Murphy K (2016). Janeway's Immunobiology. Norton, W.W. & Company, Inc. ISBN 978-0-8153-4505-3.[page needed]
- ^ Grobarczyk B, Franco B, Hanon K, Malgrange B (October 2015). "Generation of Isogenic Human iPS Cell Line Precisely Corrected by Genome Editing Using the CRISPR/Cas9 System". Stem Cell Reviews and Reports. 11 (5): 774–87. doi:10.1007/s12015-015-9600-1. PMID 26059412. S2CID 18897400.
- ^ Mertsching H, Schanz J, Steger V, Schandar M, Schenk M, Hansmann J, et al. (July 2009). "Generation and transplantation of an autologous vascularized bioartificial human tissue". Transplantation. 88 (2): 203–210. doi:10.1097/TP.0b013e3181ac15e1. PMID 19623015. S2CID 46083673.
- ^ Newman P, Minett A, Ellis-Behnke R, Zreiqat H (November 2013). "Carbon nanotubes: their potential and pitfalls for bone tissue regeneration and engineering". Nanomedicine. 9 (8): 1139–1158. doi:10.1016/j.nano.2013.06.001. PMID 23770067.
- ^ Patrick CW, Mikos AG, McIntire LV (1998). Frontiers in tissue engineering (1st ed.). Oxford, UK: Pergamon. ISBN 978-0-08-042689-1. OCLC 162130841.
- ^ Stewart SA, Domínguez-Robles J, Donnelly RF, Larrañeta E (December 2018). "Implantable Polymeric Drug Delivery Devices: Classification, Manufacture, Materials, and Clinical Applications". Polymers. 10 (12): 1379. doi:10.3390/polym10121379. PMC 6401754. PMID 30961303.
- ^ Kajihara M, Sugie T, Maeda H, Sano A, Fujioka K, Urabe Y, et al. (January 2003). "Novel drug delivery device using silicone: controlled release of insoluble drugs or two kinds of water-soluble drugs". Chemical & Pharmaceutical Bulletin. 51 (1): 15–19. doi:10.1248/cpb.51.15. PMID 12520121.
- ^ Afewerki S, Sheikhi A, Kannan S, Ahadian S, Khademhosseini A (January 2019). "Gelatin-polysaccharide composite scaffolds for 3D cell culture and tissue engineering: Towards natural therapeutics". Bioengineering & Translational Medicine. 4 (1): 96–115. doi:10.1002/btm2.10124. PMC 6336672. PMID 30680322.
- ^ Martin CA, Radhakrishnan S, Nagarajan S, Muthukoori S, Dueñas JM, Ribelles JL, et al. (2019). "An innovative bioresorbable gelatin based 3D scaffold that maintains the stemness of adipose tissue derived stem cells and the plasticity of differentiated neurons". RSC Advances. 9 (25): 14452–64. Bibcode:2019RSCAd...914452M. doi:10.1039/C8RA09688K. ISSN 2046-2069. PMC 9064131. PMID 35519343.
- ^ Takagi Y, Tanaka S, Tomita S, Akiyama S, Maki Y, Yamamoto T, Uehara M, Dobashi T (2017). "Preparation of gelatin scaffold and fibroblast cell culture". Journal of Biorheology. 31 (1): 2–5. doi:10.17106/jbr.31.2. ISSN 1867-0466.
- ^ Roohani-Esfahani SI, Newman P, Zreiqat H (January 2016). "Design and Fabrication of 3D printed Scaffolds with a Mechanical Strength Comparable to Cortical Bone to Repair Large Bone Defects". Scientific Reports. 6 (1) 19468. Bibcode:2016NatSR...619468R. doi:10.1038/srep19468. PMC 4726111. PMID 26782020.
- ^ Nokoorani YD, Shamloo A, Bahadoran M, Moravvej H (August 2021). "Fabrication and characterization of scaffolds containing different amounts of allantoin for skin tissue engineering". Scientific Reports. 11 (1) 16164. Bibcode:2021NatSR..1116164N. doi:10.1038/s41598-021-95763-4. PMC 8352935. PMID 34373593.
- ^ Gentile P, Chiono V, Carmagnola I, Hatton PV (February 2014). "An overview of poly(lactic-co-glycolic) acid (PLGA)-based biomaterials for bone tissue engineering". International Journal of Molecular Sciences. 15 (3): 3640–59. doi:10.3390/ijms15033640. PMC 3975359. PMID 24590126.
- ^ Park JH, Schwartz Z, Olivares-Navarrete R, Boyan BD, Tannenbaum R (May 2011). "Enhancement of surface wettability via the modification of microtextured titanium implant surfaces with polyelectrolytes". Langmuir. 27 (10): 5976–85. doi:10.1021/la2000415. PMC 4287413. PMID 21513319.
- ^ Leichner C, Jelkmann M, Bernkop-Schnürch A (2019). "Thiolated polymers: Bioinspired polymers utilizing one of the most important bridging structures in nature". Advanced Drug Delivery Reviews. 151–152: 191–221. doi:10.1016/j.addr.2019.04.007. PMID 31028759. S2CID 135464452.
- ^ Kast CE, Frick W, Losert U, Bernkop-Schnürch A (April 2003). "Chitosan-thioglycolic acid conjugate: a new scaffold material for tissue engineering?". International Journal of Pharmaceutics. 256 (1–2): 183–189. doi:10.1016/S0378-5173(03)00076-0. PMID 12695025.
- ^ Bae IH, Jeong BC, Kook MS, Kim SH, Koh JT (2013). "Evaluation of a thiolated chitosan scaffold for local delivery of BMP-2 for osteogenic differentiation and ectopic bone formation". BioMed Research International. 2013 878930. doi:10.1155/2013/878930. PMC 3760211. PMID 24024213.
- ^ Bian S, He M, Sui J, Cai H, Sun Y, Liang J, et al. (April 2016). "The self-crosslinking smart hyaluronic acid hydrogels as injectable three-dimensional scaffolds for cells culture". Colloids and Surfaces B: Biointerfaces. 140: 392–402. doi:10.1016/j.colsurfb.2016.01.008. PMID 26780252.
- ^ Gajendiran M, Rhee JS, Kim K (February 2018). "Recent Developments in Thiolated Polymeric Hydrogels for Tissue Engineering Applications". Tissue Engineering. Part B, Reviews. 24 (1): 66–74. doi:10.1089/ten.TEB.2016.0442. PMID 28726576.
- ^ Bauer C, Jeyakumar V, Niculescu-Morzsa E, Kern D, Nehrer S (December 2017). "Hyaluronan thiomer gel/matrix mediated healing of articular cartilage defects in New Zealand White rabbits-a pilot study". Journal of Experimental Orthopaedics. 4 (1) 14. doi:10.1186/s40634-017-0089-1. PMC 5415448. PMID 28470629.
- ^ Zahir-Jouzdani F, Mahbod M, Soleimani M, Vakhshiteh F, Arefian E, Shahosseini S, et al. (January 2018). "Chitosan and thiolated chitosan: Novel therapeutic approach for preventing corneal haze after chemical injuries". Carbohydrate Polymers. 179: 42–49. doi:10.1016/j.carbpol.2017.09.062. PMID 29111069.
- ^ "Studies in humans clinical trials" (PDF).
- ^ Pomeroy JE, Helfer A, Bursac N (1 September 2020). "Biomaterializing the promise of cardiac tissue engineering". Biotechnology Advances. 42 107353. doi:10.1016/j.biotechadv.2019.02.009. PMC 6702110. PMID 30794878.
- ^ Cassidy JW (November 2014). "Nanotechnology in the Regeneration of Complex Tissues" (PDF). Bone and Tissue Regeneration Insights. 5: 25–35. doi:10.4137/BTRI.S12331. PMC 4471123. PMID 26097381.
- ^ Nam YS, Park TG (October 1999). "Biodegradable polymeric microcellular foams by modified thermally induced phase separation method". Biomaterials. 20 (19): 1783–90. doi:10.1016/S0142-9612(99)00073-3. PMID 10509188.
- ^ Simon EM (1988). "NIH Phase I Final Report: Fibrous Substrates for Cell Culture (R3RR03544A)". ResearchGate. Retrieved 22 May 2017.
- ^ Melchels F, Wiggenhauser PS, Warne D, Barry M, Ong FR, Chong WS, et al. (September 2011). "CAD/CAM-assisted breast reconstruction" (PDF). Biofabrication. 3 (3) 034114. Bibcode:2011BioFa...3c4114M. doi:10.1088/1758-5082/3/3/034114. PMID 21900731. S2CID 206108959.
- ^ a b Elisseeff J, Ma PX (2005). Scaffolding in Tissue Engineering. Boca Raton: CRC. ISBN 978-1-57444-521-3.
- ^ a b Lee GY, Kenny PA, Lee EH, Bissell MJ (April 2007). "Three-dimensional culture models of normal and malignant breast epithelial cells". Nature Methods. 4 (4): 359–65. doi:10.1038/nmeth1015. PMC 2933182. PMID 17396127.
- ^ a b c Lai Y, Asthana A, Kisaalita WS (April 2011). "Biomarkers for simplifying HTS 3D cell culture platforms for drug discovery: the case for cytokines". Drug Discovery Today. 16 (7–8): 293–97. doi:10.1016/j.drudis.2011.01.009. PMID 21277382.
- ^ Gustafsson L, Tasiopoulos CP, Jansson R, Kvick M, Duursma T, Gasser TC, van der Wijngaart W, Hedhammar M (16 August 2020). "Recombinant Spider Silk Forms Tough and Elastic Nanomembranes that are Protein-Permeable and Support Cell Attachment and Growth". Advanced Functional Materials. 30 (40) 2002982. doi:10.1002/adfm.202002982.
- ^ Tasiopoulos CP, Gustafsson L, van der Wijngaart W, Hedhammar M (July 2021). "Fibrillar Nanomembranes of Recombinant Spider Silk Protein Support Cell Co-culture in an In Vitro Blood Vessel Wall Model". ACS Biomaterials Science & Engineering. 7 (7): 3332–3339. doi:10.1021/acsbiomaterials.1c00612. PMC 8290846. PMID 34169711.
- ^ Dias, Juliana R.; Ribeiro, Nilza; Baptista-Silva, Sara; Costa-Pinto, Ana Rita; Alves, Nuno; Oliveira, Ana L. (2020). "In situ Enabling Approaches for Tissue Regeneration: Current Challenges and New Developments". Frontiers in Bioengineering and Biotechnology. 8 85. doi:10.3389/fbioe.2020.00085. ISSN 2296-4185. PMC 7039825. PMID 32133354.
- ^ Malek-Khatabi, A.; Javar, H.A.; Dashtimoghadam, E.; Ansari, S.; Hasani-Sadrabadi, M.M.; Moshaverinia, A. (2020). "In situ bone tissue engineering using gene delivery nanocomplexes". Acta Biomaterialia. 108: 326–336. doi:10.1016/j.actbio.2020.03.008. ISSN 1878-7568. PMID 32160962. S2CID 212679291.
- ^ Krasilnikova, O.A.; Baranovskii, D.S.; Yakimova, A.O.; Arguchinskaya, N.; Kisel, A.; Sosin, D.; Sulina, Y.; Ivanov, S.A.; Shegay, P.V.; Kaprin, A.D.; Klabukov, I.D. (2022). "Intraoperative Creation of Tissue-Engineered Grafts with Minimally Manipulated Cells: New Concept of Bone Tissue Engineering In Situ". Bioengineering. 9 (11): 704. doi:10.3390/bioengineering9110704. ISSN 2306-5354. PMC 9687730. PMID 36421105.
- ^ Hu JC, Athanasiou KA (April 2006). "A self-assembling process in articular cartilage tissue engineering". Tissue Engineering. 12 (4): 969–79. doi:10.1089/ten.2006.12.969. PMID 16674308.
- ^ Lee JK, Huwe LW, Paschos N, Aryaei A, Gegg CA, Hu JC, Athanasiou KA (August 2017). "Tension stimulation drives tissue formation in scaffold-free systems". Nature Materials. 16 (8): 864–73. Bibcode:2017NatMa..16..864L. doi:10.1038/nmat4917. PMC 5532069. PMID 28604717.
- ^ Chen P, Luo Z, Güven S, Tasoglu S, Ganesan AV, Weng A, Demirci U (September 2014). "Microscale assembly directed by liquid-based template". Advanced Materials. 26 (34): 5936–41. Bibcode:2014AdM....26.5936C. doi:10.1002/adma.201402079. PMC 4159433. PMID 24956442.
- ^ Mironov V, Boland T, Trusk T, Forgacs G, Markwald RR (April 2003). "Organ printing: computer-aided jet-based 3D tissue engineering". Trends in Biotechnology. 21 (4): 157–61. doi:10.1016/S0167-7799(03)00033-7. PMID 12679063.
- ^ Alta A (March 2011). "Printing a human kidney". TED. Archived from the original on 26 February 2014. Retrieved 29 November 2013.
- ^ Du Y, Han R, Wen F, Ng San San S, Xia L, Wohland T, et al. (January 2008). "Synthetic sandwich culture of 3D hepatocyte monolayer". Biomaterials. 29 (3): 290–301. doi:10.1016/j.biomaterials.2007.09.016. PMID 17964646.
- ^ Kang HW, Lee SJ, Ko IK, Kengla C, Yoo JJ, Atala A (March 2016). "A 3D bioprinting system to produce human-scale tissue constructs with structural integrity". Nature Biotechnology. 34 (3): 312–19. doi:10.1038/nbt.3413. PMID 26878319. S2CID 9073831.
- ^ Mačiulaitis J, Deveikytė M, Rekštytė S, Bratchikov M, Darinskas A, Šimbelytė A, et al. (March 2015). "Preclinical study of SZ2080 material 3D microstructured scaffolds for cartilage tissue engineering made by femtosecond direct laser writing lithography". Biofabrication. 7 (1) 015015. Bibcode:2015BioFa...7a5015M. doi:10.1088/1758-5090/7/1/015015. PMID 25797444. S2CID 40712319.
- ^ Grebenyuk, Sergei; Abdel Fattah, Abdel Rahman; Kumar, Manoj; Toprakhisar, Burak; Rustandi, Gregorius; Vananroye, Anja; Salmon, Idris; Verfaillie, Catherine; Grillo, Mark; Ranga, Adrian (12 January 2023). "Large-scale perfused tissues via synthetic 3D soft microfluidics". Nature Communications. 14 (1): 193. Bibcode:2023NatCo..14..193G. doi:10.1038/s41467-022-35619-1. ISSN 2041-1723. PMC 9837048. PMID 36635264.
- ^ a b c Greggio C, De Franceschi F, Figueiredo-Larsen M, Gobaa S, Ranga A, Semb H, et al. (November 2013). "Artificial three-dimensional niches deconstruct pancreas development in vitro". Development. 140 (21): 4452–4462. doi:10.1242/dev.096628. PMC 4007719. PMID 24130330.
Lay summary: "New 3D method used to grow miniature pancreas model". Kurzweil. 17 October 2013. - ^ Maul TM, Chew DW, Nieponice A, Vorp DA (December 2011). "Mechanical stimuli differentially control stem cell behavior: morphology, proliferation, and differentiation". Biomechanics and Modeling in Mechanobiology. 10 (6): 939–53. doi:10.1007/s10237-010-0285-8. PMC 3208754. PMID 21253809.
- ^ Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, et al. (April 1999). "Multilineage potential of adult human mesenchymal stem cells". Science. 284 (5411): 143–47. Bibcode:1999Sci...284..143P. doi:10.1126/science.284.5411.143. PMID 10102814.
- ^ Lee EL, von Recum HA (May 2010). "Cell culture platform with mechanical conditioning and nondamaging cellular detachment". Journal of Biomedical Materials Research. Part A. 93 (2): 411–18. doi:10.1002/jbm.a.32754. PMID 20358641.
- ^ King JA, Miller WM (August 2007). "Bioreactor development for stem cell expansion and controlled differentiation". Current Opinion in Chemical Biology. 11 (4): 394–98. doi:10.1016/j.cbpa.2007.05.034. PMC 2038982. PMID 17656148.
- ^ a b "MC2 Biotek – 3D Tissue Culture – The 3D ProtoTissue System™". Archived from the original on 28 May 2012.
- ^ Prestwich GD (January 2008). "Evaluating drug efficacy and toxicology in three dimensions: using synthetic extracellular matrices in drug discovery". Accounts of Chemical Research. 41 (1): 139–48. doi:10.1021/ar7000827. PMID 17655274.
- ^ Friedrich J, Seidel C, Ebner R, Kunz-Schughart LA (2009). "Spheroid-based drug screen: considerations and practical approach". Nature Protocols. 4 (3): 309–24. doi:10.1038/nprot.2008.226. PMID 19214182. S2CID 21783074.
- ^ a b Marx V (April 2013). "Cell culture: a better brew". Nature. 496 (7444): 253–58. Bibcode:2013Natur.496..253M. doi:10.1038/496253a. PMID 23579682. S2CID 4399610.
- ^ Zhang ZY, Teoh SH, Chong WS, Foo TT, Chng YC, Choolani M, Chan J (May 2009). "A biaxial rotating bioreactor for the culture of fetal mesenchymal stem cells for bone tissue engineering". Biomaterials. 30 (14): 2694–704. doi:10.1016/j.biomaterials.2009.01.028. PMID 19223070.
- ^ Kang KS, Lee SJ, Lee HS, Moon W, Cho DW (June 2011). "Effects of combined mechanical stimulation on the proliferation and differentiation of pre-osteoblasts". Experimental & Molecular Medicine. 43 (6): 367–73. doi:10.3858/emm.2011.43.6.040. PMC 3128915. PMID 21532314.
- ^ Rosser J, Thomas DJ (January 2018). "10 – Bioreactor processes for maturation of 3D bioprinted tissue". In Thomas DJ, Jessop ZM, Whitaker IS (eds.). 3D Bioprinting for Reconstructive Surgery. Woodhead Publishing. pp. 191–215. ISBN 978-0-08-101103-4.
- ^ Griffith LG, Swartz MA (March 2006). "Capturing complex 3D tissue physiology in vitro". Nature Reviews. Molecular Cell Biology. 7 (3): 211–24. doi:10.1038/nrm1858. PMID 16496023. S2CID 34783641.
- ^ Onoe H, Okitsu T, Itou A, Kato-Negishi M, Gojo R, Kiriya D, et al. (June 2013). "Metre-long cell-laden microfibres exhibit tissue morphologies and functions". Nature Materials. 12 (6): 584–90. Bibcode:2013NatMa..12..584O. doi:10.1038/nmat3606. PMID 23542870.
- ^ a b Wang X (January 2019). "Bioartificial Organ Manufacturing Technologies". Cell Transplantation. 28 (1): 5–17. doi:10.1177/0963689718809918. PMC 6322143. PMID 30477315.
- ^ Sawa Y, Matsumiya G, Matsuda K, Tatsumi E, Abe T, Fukunaga K, et al. (March 2018). "Journal of Artificial Organs 2017: the year in review: Journal of Artificial Organs Editorial Committee". Journal of Artificial Organs. 21 (1): 1–7. doi:10.1007/s10047-018-1018-5. PMC 7102331. PMID 29426998.
- ^ a b Chauhan S, Subin S (1 September 2011). "Extracorporeal membrane oxygenation, an anesthesiologist's perspective: physiology and principles. Part 1". Annals of Cardiac Anaesthesia. 14 (3): 218–29. doi:10.4103/0971-9784.84030. PMID 21860197.
- ^ "How did ECMO get started?". University of Iowa Hospitals & Clinics. 20 June 2017. Retrieved 4 December 2020.
- ^ "Chondroitin sulfate is a component of Integra® Dermal Regeneration Template". fdocuments.us. Retrieved 5 December 2020.
- ^ "Integra" (PDF). Retrieved 13 March 2023.
- ^ "Dermagraft Human Fibroblast-derived Dermal Substitute". dermagraft.com. Retrieved 5 December 2020.
- ^ Colvin M, Smith JM, Hadley N, Skeans MA, Uccellini K, Lehman R, et al. (February 2019). "OPTN/SRTR 2017 Annual Data Report: Heart". American Journal of Transplantation. 19 (Suppl 2): 323–403. doi:10.1111/ajt.15278. hdl:2027.42/172019. PMID 30811894. S2CID 73510324.
- ^ Smith PA, Cohn WE, Frazier OH (1 January 2018). "Chapter 7 – Total artificial hearts". Mechanical Circulatory and Respiratory Support. Academic Press. pp. 221–44. doi:10.1016/B978-0-12-810491-0.00007-2.
- ^ Khan S, Jehangir W (October 2014). "Evolution of Artificial Hearts: An Overview and History". Cardiology Research. 5 (5): 121–25. doi:10.14740/cr354w. PMC 5358116. PMID 28348709.
- ^ Melton N, Soleimani B, Dowling R (November 2019). "Current Role of the Total Artificial Heart in the Management of Advanced Heart Failure". Current Cardiology Reports. 21 (11) 142. doi:10.1007/s11886-019-1242-5. PMID 31758343. S2CID 208212152.
- ^ Cook JA, Shah KB, Quader MA, Cooke RH, Kasirajan V, Rao KK, et al. (December 2015). "The total artificial heart". Journal of Thoracic Disease. 7 (12): 2172–80. doi:10.3978/j.issn.2072-1439.2015.10.70. PMC 4703693. PMID 26793338.
- ^ a b c d Tasnim F, Deng R, Hu M, Liour S, Li Y, Ni M, et al. (August 2010). "Achievements and challenges in bioartificial kidney development". Fibrogenesis & Tissue Repair. 3 (14) 14. doi:10.1186/1755-1536-3-14. PMC 2925816. PMID 20698955.
- ^ a b Humes HD, Buffington D, Westover AJ, Roy S, Fissell WH (March 2014). "The bioartificial kidney: current status and future promise". Pediatric Nephrology. 29 (3): 343–51. doi:10.1007/s00467-013-2467-y. PMID 23619508. S2CID 19376597.
- ^ Su J, Satchell SC, Shah RN, Wertheim JA (September 2018). "Kidney decellularized extracellular matrix hydrogels: Rheological characterization and human glomerular endothelial cell response to encapsulation". Journal of Biomedical Materials Research. Part A. 106 (9): 2448–2462. doi:10.1002/jbm.a.36439. PMC 6376869. PMID 29664217.
- ^ Lee SJ, Wang HJ, Kim TH, Choi JS, Kulkarni G, Jackson JD, et al. (February 2018). "In Situ Tissue Regeneration of Renal Tissue Induced by Collagen Hydrogel Injection". Stem Cells Translational Medicine. 7 (2): 241–250. doi:10.1002/sctm.16-0361. PMC 5788870. PMID 29380564.
- ^ Wu H, Zhang R, Hu B, He Y, Zhang Y, Cai L, Wang L, Wang G, Hou H, Qiu X (December 2021). "A porous hydrogel scaffold mimicking the extracellular matrix with swim bladder derived collagen for renal tissue regeneration". Chinese Chemical Letters. 32 (12): 3940–3947. doi:10.1016/j.cclet.2021.04.043. S2CID 235570487.
- ^ Mou X, Shah J, Bhattacharya R, Kalejaiye TD, Sun B, Hsu PC, Musah S (April 2022). "A Biomimetic Electrospun Membrane Supports the Differentiation and Maturation of Kidney Epithelium from Human Stem Cells". Bioengineering. 9 (5): 188. doi:10.3390/bioengineering9050188. PMC 9137565. PMID 35621466.
- ^ Lih E, Park KW, Chun SY, Kim H, Kwon TG, Joung YK, Han DK (August 2016). "Biomimetic Porous PLGA Scaffolds Incorporating Decellularized Extracellular Matrix for Kidney Tissue Regeneration". ACS Applied Materials & Interfaces. 8 (33): 21145–21154. doi:10.1021/acsami.6b03771. PMID 27456613.
- ^ Burton TP, Callanan A (June 2018). "A Non-woven Path: Electrospun Poly(lactic acid) Scaffolds for Kidney Tissue Engineering". Tissue Engineering and Regenerative Medicine. 15 (3): 301–310. doi:10.1007/s13770-017-0107-5. PMC 6171675. PMID 30603555.
- ^ Mirmoghtadaei M, Khaboushan AS, Mohammadi B, Sadr M, Farmand H, Hassannejad Z, Kajbafzadeh AM (December 2022). "Kidney tissue engineering in preclinical models of renal failure: a systematic review and meta-analysis". Regenerative Medicine. 17 (12): 941–955. doi:10.2217/rme-2022-0084. PMID 36154467. S2CID 252543376.
- ^ a b Hwang J, Jeong Y, Park JM, Lee KH, Hong JW, Choi J (8 September 2015). "Biomimetics: forecasting the future of science, engineering, and medicine". International Journal of Nanomedicine. 10: 5701–13. doi:10.2147/IJN.S83642. PMC 4572716. PMID 26388692.
- ^ "Scientists mimic neural tissue in Army-funded research that could lead to artificial skin". U.S. Army Research Laboratory. 20 March 2018. Retrieved 22 September 2020.
- ^ a b Litschel T, Norton MM, Tserunyan V, Fraden S (February 2018). "Engineering reaction-diffusion networks with properties of neural tissue". Lab on a Chip. 18 (5): 714–22. arXiv:1711.00444. doi:10.1039/C7LC01187C. PMID 29297916. S2CID 3567908.
- ^ a b "Soft Robots". fradenlab. Retrieved 22 August 2018.
- ^ a b c d e f g h i j k l m Umemura M (3 April 2019). "Challenging the Problem of 'Fit': Advancing the Regenerative Medicine Industries in the United States, Britain and Japan". Business History. 61 (3): 456–480. doi:10.1080/00076791.2018.1476496. ISSN 0007-6791. S2CID 158171857.
- ^ a b c d e Bouchie A (December 2002). "Tissue engineering firms go under". Nature Biotechnology. 20 (12): 1178–79. doi:10.1038/nbt1202-1178. PMID 12454657. S2CID 9682305.
- ^ a b c d Brown N, Faulkner A, Kent J, Michael M (1 February 2006). "Regulating Hybrids: 'Making a Mess' and 'Cleaning Up' in Tissue Engineering and Transpecies Transplantation". Social Theory & Health. 4 (1): 1–24. doi:10.1057/palgrave.sth.8700062. PMC 7099299. PMID 32226319.
- ^ Derksen MH (2008). Engineering flesh: towards professional responsibility for 'lived bodies' in tissue engineering. Technische Universiteit Eindhoven. ISBN 978-90-386-1428-1.[page needed]
- ^ Kent J, Faulkner A, Geesink I, Fitzpatrick D (1 January 2006). "Towards governance of human tissue engineered technologies in Europe: Framing the case for a new regulatory regime". Technological Forecasting and Social Change. Technology and Social Risk. 73 (1): 41–60. doi:10.1016/j.techfore.2005.06.006.
- ^ Directive 2004/23/EC of the European Parliament and of the Council of 31 March 2004 on setting standards of quality and safety for the donation, procurement, testing, processing, preservation, storage and distribution of human tissues and cells, vol. OJ L, 7 April 2004, retrieved 7 May 2020
- ^ Faulkner A, Kent J, Geesink I, FitzPatrick D (November 2006). "Purity and the dangers of regenerative medicine: regulatory innovation of human tissue-engineered technology". Social Science & Medicine. 63 (9): 2277–88. doi:10.1016/j.socscimed.2006.06.006. PMC 7130933. PMID 16905231.
References
[edit]- Davis ME, Motion JP, Narmoneva DA, Takahashi T, Hakuno D, Kamm RD, et al. (February 2005). "Injectable self-assembling peptide nanofibers create intramyocardial microenvironments for endothelial cells". Circulation. 111 (4): 442–50. doi:10.1161/01.CIR.0000153847.47301.80. PMC 2754569. PMID 15687132.
- Derksen MH (2008). Engineering flesh: towards professional responsibility for 'lived bodies' in tissue engineering. Technische Universiteit Eindhoven. ISBN 978-90-386-1428-1.
- Fountain HA (15 September 2012). "First: Organs Tailor-Made With Body's Own Cells". The New York Times.
- Holmes TC, de Lacalle S, Su X, Liu G, Rich A, Zhang S (June 2000). "Extensive neurite outgrowth and active synapse formation on self-assembling peptide scaffolds". Proceedings of the National Academy of Sciences of the United States of America. 97 (12): 6728–6733. Bibcode:2000PNAS...97.6728H. doi:10.1073/pnas.97.12.6728. PMC 18719. PMID 10841570.
- Idrus AA (12 September 2018). "Vital Therapies falls 88%, ditches liver treatment after phase 3 fail". FierceBiotech.
- Ma PX (May 2004). "Scaffolds for tissue fabrication". Materials Today. 7 (5): 30–40. doi:10.1016/s1369-7021(04)00233-0.
- Mikos AG, Temenoff JS (2000). "Formation of highly porous biodegradable scaffolds for tissue engineering" (PDF). Electronic Journal of Biotechnology. 3 (2): 114–19. doi:10.2225/vol3-issue2-fulltext-5.
- Nerem RM (2000). Vacanti J, Lanza RP, Langer RS (eds.). Principles of tissue engineering (2nd ed.). Boston: Academic Press. ISBN 978-0-12-436630-5.
- Semino CE, Kasahara J, Hayashi Y, Zhang S (2004). "Entrapment of migrating hippocampal neural cells in three-dimensional peptide nanofiber scaffold". Tissue Engineering. 10 (3–4): 643–55. doi:10.1089/107632704323061997. PMID 15165480.
- Thompson J, Jones N, Al-Khafaji A, Malik S, Reich D, Munoz S, et al. (March 2018). "Extracorporeal cellular therapy (ELAD) in severe alcoholic hepatitis: A multinational, prospective, controlled, randomized trial". Liver Transplantation. 24 (3): 380–93. doi:10.1002/lt.24986. PMC 5873437. PMID 29171941.
- Vishwakarma A (2014). Stem Cell Biology and Tissue Engineering in Dental Sciences. Elsevier. ISBN 978-0-12-397157-9.
- Viola J, Lal B, Grad O (14 October 2003). The Emergence of Tissue Engineering as a Research Field (PDF) (Report). Abt Associates Inc. "Alternate webpage" – via nsf.gov.
External links
[edit]- Cell-Based Bone Tissue Engineering
- Clinical Tissue Engineering Center State of Ohio Initiative for Tissue Engineering (National Center for Regenerative Medicine)
- Organ Printing Archived 28 August 2008 at the Wayback Machine Multi-site NSF-funded initiative
- LOEX Center Université Laval Initiative for Tissue Engineering
Tissue engineering
View on GrokipediaFundamentals
Definition and Principles
Tissue engineering is an interdisciplinary field that integrates principles from engineering and the life sciences to develop biological substitutes capable of restoring, maintaining, or improving tissue function or replacing whole organs.[1][3] This approach addresses limitations in traditional transplantation by creating functional tissues through controlled cellular processes, often mimicking native extracellular matrix environments to support cell viability and organization.[7] The foundational principles of tissue engineering center on the triad of cells, scaffolds, and bioactive molecules. Cells, sourced from autologous, allogeneic, or stem cell populations, serve as the building blocks, providing the regenerative potential through proliferation, differentiation, and extracellular matrix production.[1] Biocompatible scaffolds, typically polymeric biomaterials, offer structural integrity, facilitate cell adhesion, and enable nutrient diffusion while degrading over time to allow native tissue integration; their design must match the mechanical properties and architecture of the target tissue to prevent stress shielding or failure.[7][8] Bioactive factors, such as growth factors and cytokines, regulate cellular behavior by promoting specific signaling pathways for directed tissue formation.[9] Core engineering strategies emphasize first-principles control of biological systems, including bioreactor conditioning for dynamic mechanical and biochemical stimuli to enhance tissue maturation, and computational modeling to predict scaffold degradation and cell-scaffold interactions.[10] These principles prioritize causal mechanisms like diffusion-limited oxygen supply and shear stress responses, ensuring engineered constructs achieve vascularization and functional equivalence to native tissues before implantation.[11] Success hinges on empirical validation through in vitro and in vivo testing, revealing challenges such as immune rejection and scale-up limitations that demand iterative refinement based on measurable outcomes like cell viability rates exceeding 90% in optimized scaffolds.[12]Etymology and Conceptual Foundations
The term "tissue engineering" was coined in 1985 by Yuan-Cheng Fung, a pioneering bioengineer and physiologist at the University of California, San Diego, during discussions at the Engineering Research Center on biomechanics and bioengineering applications.[13][14] Fung's usage emphasized the application of engineering principles to biological tissues, building on his foundational work in biomechanics, which modeled tissues as mechanical systems subject to stress-strain relationships and fluid dynamics.[13] This etymology reflects a fusion of "tissue," derived from Latin textum meaning "woven fabric" via Old French, denoting organized biological structures, with "engineering," from Latin ingenium for "cleverness" or "device," signifying systematic design and construction.[13] Conceptually, tissue engineering rests on first-principles integration of cell biology, materials science, and biochemical signaling to replicate native tissue architecture and function, addressing the causal limitations of traditional transplantation—such as donor shortages and immune rejection—through de novo construction.[15] In 1993, Robert Langer and Joseph Vacanti formalized it as an interdisciplinary field applying engineering and life sciences principles to develop biological substitutes that restore, maintain, or improve tissue function or a whole organ.[16] This paradigm draws from developmental biology's understanding of morphogenesis, where cells self-organize via gradients of soluble factors and mechanical cues, and from polymer chemistry's ability to fabricate porous scaffolds mimicking the extracellular matrix (ECM), which provides structural support, diffusivity for nutrients, and topographic signals for cell adhesion and migration.[2] Empirical evidence for these foundations emerged from early experiments, such as the 1960s development of dermal substitutes using collagen matrices to promote fibroblast infiltration, demonstrating that engineered constructs could integrate with host vasculature and remodel via endogenous proteases.[17] At its core, the discipline operates on a triadic framework: seeded cells as functional units capable of proliferation and differentiation; biodegradable scaffolds to guide spatial organization and degrade at rates matching tissue regeneration (typically 10-100 μm pore sizes for vascular ingrowth); and bioactive molecules like growth factors (e.g., vascular endothelial growth factor at 10-100 ng/mL concentrations) to orchestrate paracrine signaling and ECM deposition.[2] This causal realism prioritizes empirical validation through bioreactors simulating physiological shear stresses (0.1-10 dyn/cm²) and oxygen gradients, revealing that mismatched mechanics can induce apoptosis or fibrosis, as quantified in hydrogel models where stiffness modulates lineage commitment (e.g., soft gels favor neurogenesis, stiff ones osteogenesis).[15] Unlike speculative biofabrication, these foundations demand quantitative metrics—such as cell viability >80% post-seeding and tensile moduli aligning with native tissues (e.g., 1-10 kPa for cartilage)—to ensure translational viability, underscoring the field's evolution from empirical grafting to predictive modeling.[17]Historical Development
Pre-Modern Origins
The earliest documented attempts at tissue repair resembling modern tissue engineering principles involved rudimentary skin grafting techniques in ancient civilizations. The Ebers Papyrus, an Egyptian medical text dating to approximately 1550 BC, records the use of xenografting—applying skin from animals such as pigs or frogs to human burn wounds—to promote healing, reflecting an empirical recognition of tissue transfer for defect coverage.[18][19] These practices were driven by necessity in treating injuries, though success rates were limited by infection and rejection, absent any understanding of immunology. In ancient India, more sophisticated autologous techniques emerged around 600 BC, as described in the Sushruta Samhita, a foundational surgical treatise attributed to the physician Sushruta. This text details pedicled skin flaps harvested from the forehead or cheek to reconstruct noses amputated as punishment for theft or adultery, involving precise incision, pedicle preservation for vascular supply, and postoperative care to ensure flap viability.[20][21] These methods emphasized tissue viability through maintained blood supply, a causal precursor to contemporary scaffold-free engineering approaches, and were empirically refined over centuries within the Ayurvedic tradition.[22] Such innovations persisted and evolved through classical antiquity and the Renaissance. Roman author Aulus Cornelius Celsus, in his 1st-century AD encyclopedia De Medicina, prescribed skin transplantation from adjacent areas to cover wounds, advocating for thin grafts to minimize contraction and improve integration.[23] By the 16th century, Italian surgeon Gaspare Tagliacozzi systematized arm-based pedicle flaps for facial reconstruction, publishing detailed illustrations and protocols in De Chirurgia Curatorum Veste (1597), which highlighted the importance of tissue matching and immobilization—principles causally linked to reducing ischemia in transfers.[24] These pre-modern efforts laid groundwork for tissue engineering by demonstrating that viable tissue relocation could restore form and function, albeit constrained by high complication rates from sepsis and poor vascular anastomosis.[25]20th-Century Foundations
The foundations of tissue engineering were laid in the mid-to-late 20th century through the convergence of advances in cell biology, biomaterials, and bioengineering, addressing the shortages in organ transplantation and the limitations of synthetic prosthetics. Early conceptual work emphasized the regeneration of functional tissues by combining living cells with supportive scaffolds, drawing from principles of developmental biology and polymer chemistry.[15] This interdisciplinary approach aimed to harness the body's regenerative capacity via engineered constructs rather than mere replacement materials.[26] The term "tissue engineering" was coined in 1985 by bioengineer Yuan-Cheng Fung in a proposal to the National Science Foundation, proposing an engineering framework for manipulating biological tissues to restore function.[27] Building on this, researchers Robert Langer and Joseph Vacanti advanced the field in the late 1980s by developing biodegradable polymer scaffolds, such as polyglycolic acid, to support cell attachment and tissue formation in three dimensions. Their seminal 1993 review in Science formalized the triad of cells, scaffolds, and bioactive signals as the core strategy for tissue regeneration.[28] These scaffolds mimicked the extracellular matrix, enabling controlled degradation and nutrient delivery, which proved essential for overcoming the diffusion limitations of avascular tissue constructs.[26] Early experimental milestones included skin substitutes pioneered in the 1970s and 1980s by Ioannis Yannas and John Burke at Massachusetts Institute of Technology, who created collagen-glycosaminoglycan matrices that promoted dermal regeneration in burn patients.[17] This work culminated in the FDA approval of Integra Artificial Dermis in 1996, marking the first commercial tissue-engineered product derived from these foundational efforts.[29] Parallel developments in cartilage engineering demonstrated the seeding of chondrocytes onto scaffolds, yielding viable constructs implanted in animal models by the early 1990s, validating the approach for load-bearing tissues.[30] The field's institutionalization occurred with the founding of the Tissue Engineering Society in 1994, later renamed the Tissue Engineering and Regenerative Medicine International Society, alongside the launch of the Tissue Engineering journal, fostering collaborative research and standardization.[26] These 20th-century foundations emphasized empirical validation through in vitro and small-animal models, prioritizing biocompatibility and mechanical integrity over speculative organ complexity, which set the stage for subsequent scalability challenges.[28]21st-Century Advancements and Milestones
The generation of induced pluripotent stem cells (iPSCs) by Shinya Yamanaka in 2006 marked a foundational milestone, enabling the reprogramming of somatic cells into a pluripotent state using four transcription factors, thereby providing an ethical, patient-specific source of cells for tissue engineering without reliance on embryonic stem cells.[31] This breakthrough facilitated autologous tissue constructs, reducing immunogenicity risks and accelerating applications in regenerative medicine.[32] In the same year, Anthony Atala's team at Wake Forest University achieved the first clinical implantation of tissue-engineered bladders in seven patients aged 4 to 19 with myelomeningocele, using autologous urothelial and muscle cells seeded onto collagen-polyglycolic acid scaffolds; follow-up studies over 22 to 46 months demonstrated improved bladder capacity and compliance without major complications.[33] Building on this, the team reported in 2014 the successful implantation of engineered vaginas in four adolescent girls with Mayer-Rokitansky-Küster-Hauser syndrome, constructed from autologous cells on biodegradable scaffolds, with functional outcomes including menstruation and sexual activity sustained over years.[34] Advancements in 3D bioprinting emerged prominently in the 2010s, with Wake Forest's 2016 development of the Integrated Tissue and Organ Printing (ITOP) system enabling the fabrication of scalable, vascularized tissues using cell-laden hydrogels and sacrificial inks for channel formation.[34] By 2022, initial human implants of 3D-bioprinted tissues, such as cartilage patches, entered clinical evaluation, demonstrating viability for orthopedic applications amid ongoing challenges in vascularization and scale-up.[35] These efforts have expanded to experimental solid organs, including recellularized livers and penile tissue by 2010, though full clinical translation remains limited by integration and immune barriers.[34] Integration of gene editing, such as CRISPR-Cas9, with tissue scaffolds has further propelled progress since the mid-2010s, enhancing cell functionality for complex tissues like cartilage and neural structures, as evidenced in preclinical models showing improved regeneration.[36] As of 2025, over 20 tissue-engineered technologies have advanced to human trials, primarily for skin, cartilage, and vascular grafts, underscoring a shift toward personalized, off-the-shelf constructs despite persistent hurdles in manufacturing scalability and regulatory approval.[17]Biological Components
Cell Sources and Isolation
Cell sources in tissue engineering are selected based on their proliferative potential, differentiation capacity, and compatibility with the host to minimize immune rejection and ensure functional tissue regeneration. Primary autologous cells, derived directly from the patient's own tissues via biopsy, such as skin fibroblasts or chondrocytes from cartilage, offer the advantage of immunological tolerance but are limited by low initial yields and reduced proliferative ability in diseased states.[1] For example, urothelial cells isolated from a 1 cm² bladder biopsy can be expanded in vitro to cover over 4,000 cm² within eight weeks, demonstrating feasibility for clinical applications like bladder reconstruction.[1] Allogeneic cells, sourced from healthy donors, provide greater availability and standardized quality, enabling off-the-shelf therapies, though they require immunosuppression or HLA matching to mitigate rejection risks.[37] Stem cells, including mesenchymal stromal/stem cells (MSCs) from bone marrow, adipose tissue, or umbilical cord, are favored for their multipotency and self-renewal, allowing differentiation into multiple lineages relevant to target tissues like bone or cartilage.[38] Adipose-derived stem cells (ADSCs), for instance, constitute about 1% of stromal vascular fraction cells and yield approximately 10^5 cells per gram of lipoaspirate after isolation.[38] Isolation begins with tissue procurement, followed by mechanical dissociation through mincing or grinding to disrupt the extracellular matrix, often combined with enzymatic digestion using agents like collagenase type I or II and dispase to liberate individual cells.[1] For MSCs, purification typically involves density gradient centrifugation with Ficoll to enrich mononuclear cells, yielding 0.01-0.001% MSCs from bone marrow aspirates of 20-50 mL, or plastic adherence in culture to select adherent populations over 24-48 hours.[38] Explant culture, where minced tissue fragments allow cell migration onto surfaces, offers a non-enzymatic alternative but results in lower yields compared to enzymatic methods.[38] Post-isolation, cells are qualified per International Society for Cellular Therapy criteria, confirming plastic adherence, expression of surface markers CD73, CD90, and CD105 (>95% positive), absence of hematopoietic markers, and trilineage differentiation potential into osteoblasts, adipocytes, and chondrocytes.[38] Emerging techniques, such as microfluidic inertial separation or magnetic-activated cell sorting, enhance purity and viability by label-free or marker-based (e.g., CD31+ for endothelial progenitors) enrichment, addressing challenges like heterogeneity and potency loss during expansion.[39] These methods support scalability for clinical translation, though variability in donor age, health, and processing conditions impacts reproducibility.[39]Stem Cells and Genetic Classifications
Stem cells serve as a primary cellular component in tissue engineering, enabling the regeneration of functional tissues by providing a source of undifferentiated cells capable of self-renewal and differentiation into specialized cell types. In tissue engineering applications, these cells are often seeded onto scaffolds or directed to form organoids, mimicking native tissue architecture and function.[40] Stem cells are classified primarily by their differentiation potential, or potency, which is determined by their genetic and epigenetic profiles. Totipotent stem cells, such as the zygote, possess the broadest potential, capable of developing into all cell types of an organism, including extra-embryonic tissues; however, they are rarely utilized in tissue engineering due to their early embryonic origin and limited accessibility. Pluripotent stem cells, including embryonic stem cells (ESCs) derived from the inner cell mass of blastocysts and induced pluripotent stem cells (iPSCs) reprogrammed from somatic cells, can differentiate into derivatives of all three germ layers—ectoderm, mesoderm, and endoderm—but not extra-embryonic tissues. ESCs, first isolated in humans in 1998, offer high proliferative capacity but raise ethical concerns due to the destruction of viable embryos required for their derivation. iPSCs, generated since 2006 through overexpression of transcription factors like Oct4, Sox2, Klf4, and c-Myc, provide an autologous alternative, circumventing ethical issues while allowing patient-specific genetic matching to reduce immunogenicity.[41][42][32] Multipotent stem cells, also known as adult or tissue-specific stem cells, exhibit more restricted differentiation potential, limited to cell types within a particular lineage or tissue. Examples include mesenchymal stem cells (MSCs) from bone marrow, which can differentiate into osteoblasts, chondrocytes, adipocytes, and myocytes, and hematopoietic stem cells (HSCs) that generate blood cells. These cells are favored in tissue engineering for their lower risk of teratoma formation compared to pluripotent types and their relative ease of isolation from adult tissues, though their potency diminishes with donor age and they face challenges in scalability. Oligopotent and unipotent stem cells further narrow this spectrum, differentiating into a few or single cell types, respectively, such as lymphoid progenitors from HSCs.[40][43][44] Genetic classifications of stem cells emphasize molecular markers and gene expression signatures that underpin their potency and functionality. For instance, pluripotent stem cells express core transcription factors like Nanog, Oct4, and Sox2, which maintain self-renewal and inhibit differentiation; disruptions in these networks, detectable via genetic profiling, can shift cells toward multipotency. MSCs are identified by surface markers such as CD73, CD90, and CD105, with absence of hematopoietic markers like CD45, enabling genetic sorting for purity in engineering applications. In regenerative contexts, genetic engineering techniques, including CRISPR-Cas9 editing, are applied to enhance stem cell therapeutic potential, such as correcting mutations in iPSCs for disease modeling or improving homing signals in MSCs for targeted tissue repair. These modifications must balance efficacy with risks like off-target effects, as evidenced by studies showing stable integration of therapeutic genes without compromising pluripotency.[42][45][46]Biomaterials and Scaffolds
Materials and Properties
Natural polymers, such as collagen and chitosan, are widely used in scaffolds due to their inherent biocompatibility and ability to mimic the extracellular matrix, with collagen featuring RGD sequences that promote cell adhesion and proliferation.[47] Chitosan exhibits biodegradability through enzymatic hydrolysis and antimicrobial properties, supporting applications in wound healing and cartilage regeneration.[12] Alginate, derived from algae, forms hydrogels with high biocompatibility and controlled degradation, achieving up to 100% cell viability in fibroblast cultures when crosslinked.[12] Synthetic polymers like polycaprolactone (PCL) and poly(lactic-co-glycolic acid) (PLGA) provide mechanical robustness and tunable degradation profiles, with PCL scaffolds demonstrating 99.1% porosity and slow hydrolysis over years to accommodate long-term tissue remodeling.[12] PLGA degrades via hydrolysis into lactic and glycolic acids, with rates adjustable from weeks to months based on lactide:glycolide ratios, ensuring compatibility with bone and soft tissue ingrowth while maintaining structural integrity.[12] Polylactic acid (PLA) offers similar biodegradability and FDA approval for biomedical use, with scaffolds exhibiting 86-90% porosity for nutrient diffusion.[12] Essential properties of scaffold materials include biocompatibility to minimize inflammatory responses and support cell viability above 80-100% in vitro; biodegradability synchronized with tissue regeneration to avoid chronic foreign body reactions; mechanical strength matching native tissues, such as compressive moduli of 10-20 GPa for bone-mimicking scaffolds; and interconnected porosity of 100-500 μm to enable vascularization, cell migration, and waste removal.[48][47] Bioactivity, often enhanced in composites like hydroxyapatite (HA)-polymer blends, promotes osteoconduction through ion release that stimulates mineralization.[48] Surface chemistry influences protein adsorption and cell signaling, with hydrophilic modifications improving wettability and adhesion without compromising bulk degradation.[9]| Property | Description | Examples in Materials |
|---|---|---|
| Biocompatibility | Non-toxic interaction supporting cell attachment and minimal immunogenicity | Collagen (RGD-mediated adhesion), PCL (>99% viability)[47][12] |
| Biodegradability | Controlled breakdown via hydrolysis or enzymes, rate-matched to tissue formation | PLGA (weeks-months), alginate (enzymatic)[12] |
| Mechanical Strength | Tailored modulus and tensile/compressive properties for load-bearing | HA composites (17-135 MPa tension for bone), PU elasticity for soft tissues[48][47] |
| Porosity | Interconnected pores for diffusion and infiltration | PCL (99.1%), general range 100-500 μm[12][47] |
Synthesis and Fabrication Techniques
Scaffold fabrication techniques encompass a range of methods to process biomaterials into three-dimensional porous structures that mimic the extracellular matrix, providing mechanical support, nutrient diffusion, and cell adhesion sites for tissue regeneration. These techniques are categorized into conventional approaches, which rely on basic physical or chemical processes, and advanced methods, which offer greater control over architecture and incorporate emerging technologies. Selection of a technique depends on the biomaterial properties, desired porosity (typically 80-95%), pore interconnectivity, and mechanical strength required for specific tissues.[7][49] Conventional techniques include solvent casting combined with particulate leaching, where a polymer such as polycaprolactone (PCL) is dissolved in an organic solvent and mixed with porogens like sodium chloride particles (200-500 μm diameter); after casting and solvent evaporation, porogens are leached out with water to yield scaffolds with controlled porosity up to 90% but limited interconnectivity and potential residual solvent toxicity.[7][49] Freeze-drying, or lyophilization, involves freezing a polymer solution (e.g., collagen or chitosan at -20°C to -80°C), followed by sublimation under vacuum to remove ice crystals, producing anisotropic scaffolds with pore sizes of 15-200 μm and high porosity (30-90%) suitable for soft tissues, though mechanical fragility persists without reinforcement.[50][49] Gas foaming employs inert gases like CO2 under high pressure (up to 800 psi) to nucleate bubbles within a polymer matrix such as PCL, expanding to form closed-pore structures with porosities exceeding 90%, avoiding toxic solvents but often resulting in poor interconnectivity that hinders cell migration.[7][49] Phase separation techniques induce polymer-solvent demixing, either thermally (cooling a homogeneous solution of poly(L-lactic acid) to form nanofibrous pores) or non-solvent induced (adding a poor solvent), yielding scaffolds with high surface area but variable mechanical integrity and limited scalability for load-bearing applications.[7][49] Melt molding heats biomaterials with porogens to 100-200°C, shapes them via compression, and removes porogens, enabling precise geometries for bone scaffolds but risking thermal degradation of bioactive components like growth factors.[7] Advanced fabrication methods enhance precision and biomimicry. Electrospinning applies high voltage (10-30 kV) to eject polymer solutions (e.g., PCL or gelatin at 10-20% concentration) through a needle, drawing nanofibers (100 nm-6 μm diameter) with porosities of 80-95%, mimicking native ECM to promote cell adhesion and proliferation, though scaffold thickness is often limited to <1 mm without collectors.[50][49] Variants like core-shell electrospinning encapsulate drugs for sustained release, while melt electrospinning avoids solvents for thicker 3D structures. Rapid prototyping, including 3D printing and stereolithography, builds scaffolds layer-by-layer from bioinks (e.g., PCL-hydroxyapatite composites) with resolutions of 100-150 μm, allowing patient-specific designs and integration of cells or gradients, but requires compatible biomaterials and can be cost-prohibitive.[50][7] These methods collectively address challenges in achieving hierarchical porosity and vascularization, with ongoing refinements focusing on hybrid approaches for clinical translation.[50][49]Engineering and Assembly Methods
Self-Assembly and Template-Based Approaches
Self-assembly methods in tissue engineering involve scaffold-free techniques where cells aggregate, proliferate, and deposit their own extracellular matrix (ECM) to form organized tissue structures, mimicking natural developmental processes without exogenous supports.[51] These approaches rely on cell-intrinsic mechanisms such as cadherin-mediated adhesion, cytoskeletal remodeling, and paracrine signaling to drive spontaneous organization into multicellular aggregates like spheroids or sheets.[52] Pioneered in the early 2000s, self-assembly has enabled the production of tissues with native-like ECM composition, reducing risks of inflammatory responses from synthetic materials.[53] Key techniques include high-density cell seeding in non-adherent cultures to form spheroids, which can fuse into larger constructs, and cell sheet engineering, where confluent monolayers are harvested intact for stacking into three-dimensional tissues.[54] For instance, micromass cultures of mesenchymal stem cells induce chondrogenesis, yielding cartilage nodules with glycosaminoglycan-rich matrices as visualized by Alcian blue staining after 21 days of culture.[51] Organoids represent advanced self-assembled structures derived from pluripotent stem cells, self-organizing into organ-specific architectures; human intestinal organoids, first reported in 2010 from adult stem cells, exhibit crypt-villus patterning and functional barrier properties.[55] In 2021, self-assembling human heart organoids from induced pluripotent stem cells demonstrated synchronized contractions and vascular networks, recapitulating embryonic heart development over 85 days.[56] Advantages of self-assembly include biocompatibility and scalability for autologous therapies, but challenges persist, such as limited vascularization restricting construct size to under 1 mm due to diffusion constraints, and variable reproducibility from heterogeneous cell responses.[57] Recent innovations incorporate bioreactors to enhance nutrient perfusion and mechanical conditioning, improving maturation; for example, dynamic culture of self-assembled skin equivalents yields stratified epithelia with basement membrane formation comparable to native tissue.[58] Template-based approaches, conversely, employ pre-fabricated scaffolds as structural templates to guide cell attachment, proliferation, and ECM deposition, providing immediate mechanical integrity and topographic cues for tissue morphogenesis.[7] Scaffolds, typically porous networks with pore sizes of 100-500 μm to facilitate cell infiltration and vascular ingrowth, are seeded with cells prior to in vitro conditioning or in vivo implantation.[59] Common materials include biodegradable polymers like poly(lactic-co-glycolic acid) (PLGA) and natural hydrogels such as collagen or fibrin, which degrade over 4-12 weeks to be replaced by host ECM.[60] Fabrication methods for templates encompass electrospinning for nanofibrous mats mimicking ECM fibril dimensions (50-500 nm), freeze-drying to create interconnected pores, and decellularized matrices retaining native bioactivity.[61] For instance, decellularization removes cells from a donor rat forelimb, leaving an ECM scaffold that is seeded with muscle and vascular progenitor cells and cultured in a bioreactor to regenerate functional muscle and blood vessels, contrasting with de novo growth from stem cells without a pre-existing scaffold.[62] In orthopedic applications, hydroxyapatite-based scaffolds templated with mesenchymal stem cells promote osteogenesis, achieving bone mineral densities of 0.5-1 g/cm³ after 8 weeks in osteogenic media.[63] Proangiogenic templates, such as those with embedded vascular endothelial growth factor (VEGF), have facilitated cardiac patch integration, with host vessel ingrowth observed within 7 days post-implantation in rodent models.[64] Hybrid strategies merge self-assembly with templates, such as pre-seeding scaffolds with cell sheets to enhance uniformity and biointegration; this has produced vascularized bone constructs with 20-30% higher compressive strength than scaffold-only methods.[58] Limitations include potential scaffold remnants triggering chronic inflammation if degradation is incomplete, and the need for precise matching of scaffold stiffness (1-100 kPa) to tissue-specific mechanics to avoid aberrant differentiation.[65] Ongoing research focuses on smart templates with stimuli-responsive degradation, improving outcomes in load-bearing tissues like cartilage, where bilayer scaffolds support zonal ECM gradients.[60]Additive Manufacturing and Bioprinting
Additive manufacturing, commonly known as 3D printing, enables the layer-by-layer fabrication of tissue scaffolds with precise control over microstructure, porosity, and geometry, addressing limitations of traditional subtractive methods in replicating complex native tissue architectures. In tissue engineering, techniques such as fused deposition modeling (FDM), selective laser sintering (SLS), and stereolithography (SLA) utilize biomaterials like polycaprolactone (PCL) or poly(lactic-co-glycolic acid) (PLGA) to create scaffolds that support cell adhesion, proliferation, and differentiation. For instance, FDM has produced bone scaffolds with 60-70% porosity and compressive strengths matching trabecular bone (2-12 MPa), promoting osteogenesis in vivo when seeded with mesenchymal stem cells. Bioprinting extends additive manufacturing by incorporating living cells into printable bioinks, allowing direct deposition of cellularized constructs that mimic tissue heterogeneity. Common modalities include extrusion-based bioprinting, which handles high-viscosity hydrogels like alginate or gelatin methacryloyl (GelMA) at resolutions of 100-400 μm, achieving cell viabilities above 85% post-printing; inkjet bioprinting, offering droplet sizes of 50-300 μm for faster deposition but requiring shear-thinning inks to minimize cell damage; and laser-assisted bioprinting, providing sub-20 μm resolution via laser-induced forward transfer without nozzle-induced shear stress. A 2019 study demonstrated extrusion bioprinting of a tri-layered skin construct with keratinocytes, fibroblasts, and endothelial cells, exhibiting vascular-like networks after 21 days in culture. Advancements in multi-nozzle and hybrid systems enable co-printing of cells, growth factors, and supporting matrices, as seen in 2022 reports of bioprinted cardiac patches integrating cardiomyocytes with aligned nanofibers, restoring 20-30% of ejection fraction in rat myocardial infarction models. However, challenges persist, including bioink cytocompatibility—where crosslinking methods like UV exposure can reduce viability to below 70%—and limited z-resolution (often >100 μm), hindering recapitulation of microvascular networks essential for nutrient diffusion beyond 200 μm tissue depths. Recent innovations, such as sacrificial inks for perfusable channels and embedded printing in supportive gels, have produced vascularized liver lobules with bile canaliculi functionality, though scalability for human-scale organs remains constrained by print times exceeding 24 hours for cm-scale constructs. Clinical translation is nascent, with the first FDA-approved bioprinted skin graft for burn wounds reported in 2023, demonstrating re-epithelialization rates comparable to autografts in phase I trials.In Situ Tissue Engineering
In situ tissue engineering involves the direct regeneration of damaged tissues within the body by deploying acellular biomaterials that recruit and activate endogenous stem or progenitor cells at the injury site, enabling their proliferation, differentiation, and extracellular matrix deposition as the scaffold degrades over time.[66] This cell-free paradigm harnesses the host's native microenvironment as a bioreactor, bypassing the complexities of ex vivo cell expansion and transplantation.[67] Key methods center on injectable systems and minimally invasive delivery of scaffolds, such as hydrogels formed via crosslinking (e.g., chitosan/PEG or hyaluronic acid derivatives) that gel in situ to fill irregular defects, or nanofibrous mats produced by electrospinning for enhanced cell adhesion.[66] Growth factors like bone morphogenetic protein-2 (BMP-2) or recombinant human BMP-7 are often incorporated into these matrices to direct osteogenesis or angiogenesis, with release kinetics tuned for sustained signaling.[66] Emerging techniques include in situ 3D bioprinting, where bioinks of gelatin methacryloyl (GelMA) or collagen/nano-hydroxyapatite are extruded or laser-deposited directly into defects, achieving resolutions of 20-100 μm and cell viabilities up to 95% in preclinical models.[67] Biomaterials commonly used include synthetic polymers like poly(lactide-co-glycolide) (PLGA) and polycaprolactone (PCL) for tunable degradation (weeks to months), natural polymers such as collagen or chitosan for bioactivity, and bioceramics like hydroxyapatite for mechanical reinforcement in load-bearing sites.[66] These are often functionalized with chemokines (e.g., SDF-1 for stem cell homing) or anti-inflammatory cues to modulate the immune response and promote constructive remodeling.[68] Advantages over ex vivo approaches include minimized risks of cell senescence during culture, reduced costs from avoiding bioreactors, and superior host integration via natural vascular ingrowth and innervation.[66] Preclinical examples demonstrate efficacy, such as PLGA scaffolds loaded with growth factors restoring bone volume in rabbit calvarial defects by 8 weeks post-implantation, or handheld bioprinting of GelMA hydrogels repairing full-thickness cartilage lesions in sheep knees with neocartilage formation histologically resembling native tissue.[66][67] Challenges persist in achieving precise spatiotemporal control of regeneration, as host factors like age-related declines in stem cell potency (noted in studies of elderly cohorts) can yield inconsistent outcomes, and large defects often suffer from inadequate vascularization leading to necrosis.[66] Scaffold mechanical mismatch with native tissue and potential ectopic mineralization from unbound growth factors further complicate translation, with clinical trials limited primarily to bone void fillers as of 2020.[67] Ongoing research emphasizes hybrid systems combining mechanical cues (e.g., aligned nanofibers) with immunomodulatory biomaterials to enhance reliability across patient variability.[68]Cultivation and Maturation Processes
Bioreactors and Tissue Culture
Bioreactors in tissue engineering are specialized devices that maintain controlled environments for the cultivation of cells, scaffolds, or tissue constructs, facilitating nutrient delivery, waste removal, and the application of biophysical stimuli such as mechanical loading, shear stress, or electrical fields to mimic in vivo conditions and promote tissue maturation.[69] These systems enable dynamic culture, which surpasses static methods by ensuring uniform distribution of oxygen and nutrients, reducing necrosis in larger constructs, and enhancing extracellular matrix (ECM) deposition through controlled environmental cues.[70] Key parameters regulated include pH, temperature, dissolved oxygen levels (typically 20-200% air saturation), and hydrodynamic forces, with designs optimized to minimize shear-induced cell damage while maximizing mass transfer efficiency.[71] Common bioreactor types for tissue culture include stirred-tank reactors, which use impellers for mixing and are suitable for suspension cultures or scaffold-free aggregates; rotating wall vessels (RWVs), which generate low-shear microgravity-like conditions ideal for cartilage or bone constructs; and perfusion bioreactors, which continuously flow media through porous scaffolds to improve nutrient penetration and waste clearance in 3D tissues.[72] Spinner flasks provide moderate agitation for early-stage expansion, while advanced systems like compressive or tensile bioreactors apply cyclic mechanical strains—e.g., 5-15% deformation at 0.1-1 Hz—to stimulate mechanotransduction pathways in load-bearing tissues such as tendons or heart valves.[73] For instance, in cartilage tissue engineering, bioreactors combining hydrostatic pressure (0.1-10 MPa) and dynamic compression have demonstrated up to 2-fold increases in glycosaminoglycan content compared to static controls after 21 days of culture.[71] Tissue culture within bioreactors emphasizes three-dimensional (3D) architectures over traditional two-dimensional monolayers, as 3D setups better replicate native tissue heterogeneity, cell-cell interactions, and gradient formations essential for functionality.[74] Recent advances include perfusion-based systems for organoid maturation, where laminar flow at 0.1-1 mL/min supports vascular-like networks and has enabled the culture of intestinal organoids with improved villus-crypt structures.[75] Additionally, 3D-printed perfusion bioreactors fabricated in 2025 allow customizable chambers for complex geometries, facilitating in situ differentiation of human induced pluripotent stem cells into neural tissues with enhanced viability exceeding 90%.[76] Despite these benefits, challenges persist, including scalability for clinical volumes (e.g., producing grams of tissue versus milligrams in lab settings) and reproducibility across batches due to variability in cell sourcing and sensor precision.[77] Hybrid bioreactors integrating sensors for real-time monitoring of metabolites via optical or electrochemical methods are emerging to address these, with studies reporting improved consistency in ECM production for vascular grafts.[78]Biomimetics and Long-Term Maintenance
Biomimetic strategies in tissue engineering cultivation replicate native physiological environments to enhance maturation, including nanoscale topography and biochemical gradients that direct cell behavior akin to in vivo conditions. Scaffolds engineered with hierarchical structures mimicking the extracellular matrix (ECM) promote cell adhesion, proliferation, and differentiation by providing topographic cues at the 10-100 nm scale, as demonstrated in collagen-based constructs for bone tissue.[79] These approaches utilize materials like decellularized ECM or synthetic polymers functionalized with bioactive motifs, such as RGD peptides, to emulate integrin-binding sites and foster organized tissue assembly.[80] In bioreactor systems, biomimetics incorporates dynamic stimuli like fluid perfusion and mechanical loading to simulate hemodynamic forces and interstitial flows, accelerating ECM remodeling and functional maturation. Perfusion bioreactors delivering pulsatile flow at rates of 0.1-1 mL/min per cm² have been shown to improve nutrient distribution and waste removal in cartilage constructs, yielding tissues with compressive moduli approaching native values of 0.5-1 MPa after 4-6 weeks of culture.[81] Cyclic compressive or tensile strains, applied at 5-10% amplitude and 0.1-1 Hz frequencies, mimic joint or vascular loading, enhancing collagen alignment and mechanical anisotropy in engineered tendons and heart valves.[82] Long-term maintenance of engineered tissues demands scaffolds that support sustained viability and functionality post-implantation, often challenged by inadequate vascularization and mechanical mismatch. Biomimetic designs address this through degradable scaffolds with resorption rates tuned to tissue ingrowth timelines, such as poly(ε-caprolactone) composites degrading over 6-12 months while neocells deposit mature ECM.[83] Incorporation of angiogenic factors like VEGF in gradient-releasing systems promotes neovascularization, extending tissue survival beyond diffusion limits of 100-200 μm, as evidenced in skin substitutes maintaining epidermal integrity for over 3 months in vivo.[84] Persistent issues include phenotypic drift and fibrosis, where biomimetic cues from mineralized collagen scaffolds have sustained osteogenic markers like RUNX2 expression for up to 12 weeks in vitro, outperforming non-biomimetic controls.[85] Hybrid systems integrating self-assembling peptides with natural polymers further enable adaptive remodeling, resisting enzymatic degradation while allowing host cell infiltration, though clinical translation remains limited by variability in patient-specific responses.[86] Ongoing research emphasizes multi-scale modeling to predict long-term stability, with finite element analyses validating biomimetic scaffolds' endurance under physiological loads for orthopedic applications.[87]Applications and Case Studies
Skin and Soft Tissue Repair
Tissue engineering approaches for skin repair focus on developing substitutes that mimic the epidermis, dermis, or full-thickness structure to treat extensive burns, chronic wounds such as diabetic foot ulcers, and surgical defects. Acellular dermal scaffolds, including the Integra Dermal Regeneration Template—a bovine collagen matrix cross-linked with chondroitin-6-sulfate derived from shark cartilage—were first approved by the U.S. Food and Drug Administration (FDA) on March 1, 1996, for reconstructing full-thickness skin loss in life-threatening burns where sufficient autograft is unavailable.[88] This template supports fibroblast infiltration and neovascularization, forming a neodermis that is subsequently covered by a thin epidermal autograft, reducing donor site morbidity by up to 50% in burn patients compared to traditional meshed autografts.[89] Cellular bilayered skin equivalents, such as Apligraf, which comprises human neonatal fibroblasts and keratinocytes cultured on a bovine type I collagen lattice, received FDA approval on May 22, 1998, for treating non-healing venous leg ulcers unresponsive to compression therapy.[90] Subsequent approvals extended its use to diabetic foot ulcers, where randomized trials demonstrated complete wound closure in 63% of Apligraf-treated cases versus 36% with conventional care at 12 weeks.[91] Meta-analyses of cellular versus acellular matrix products for diabetic foot ulcers indicate superior 12-week healing rates (odds ratio 1.48) with cellular therapies, though outcomes depend on ulcer chronicity and patient comorbidities.[92] In soft tissue reconstruction, such as for subcutaneous defects or volumetric augmentation post-trauma, hydrogel-based scaffolds facilitate minimally invasive delivery via injection after shrinkage to match defect geometry, promoting adipose and fibrous tissue ingrowth without open surgery.[93] Natural polymer scaffolds, including collagen and hyaluronic acid composites, exhibit tunable degradation rates (e.g., 4-8 weeks in vivo) and mechanical properties matching native soft tissues (Young's modulus 0.1-1 MPa), enhancing integration in reconstructive applications like breast or facial contouring.[94] Recent integrations of mesenchymal stem cells into these scaffolds have improved vascularization and reduced fibrosis in preclinical models, though clinical translation remains limited by scalability and immunogenicity concerns.[95] Microscopic scaffolds engineered with biochemical cues are enabling muscle cells to regenerate organized, functional tissue, offering a promising path for recovery after severe muscle loss. Despite these advances, challenges persist, including high production costs (e.g., 3,000 per graft) and variable long-term durability, with rejection rates under 5% for allogeneic products but higher in immunocompromised patients.[96]Orthopedic Tissues (Bone and Cartilage)
Tissue engineering strategies for bone regeneration typically employ scaffolds composed of hydroxyapatite (HA), a calcium phosphate ceramic that constitutes approximately 70% of natural bone's mineral content, to provide osteoconductive surfaces for cell attachment and mineralization. These scaffolds are often combined with mesenchymal stem cells (MSCs) derived from bone marrow, which differentiate into osteoblasts under inductive cues like bone morphogenetic protein-2 (BMP-2). In a 2022 study, 3D-printed polylactic acid (PLA)/HA scaffolds with over 20% HA loading exhibited enhanced osteogenic differentiation of MSCs in vitro, with increased alkaline phosphatase activity and calcium deposition compared to PLA alone.[97] Recent progress includes electroconductive gelatin/hyaluronic acid/HA scaffolds that support MSC proliferation and bone-like matrix formation, leveraging electrical stimulation to mimic physiological signaling.[98] Clinical translation remains limited, though HA-MSC constructs have shown promise in preclinical models for critical-sized defects, promoting vascularized bone ingrowth within 4-8 weeks.[99] Cartilage tissue engineering addresses the tissue's avascular nature and poor intrinsic repair by utilizing hydrogels that replicate its high water content (70-80%) and low mechanical stiffness. Natural polysaccharide hydrogels, such as hyaluronic acid or alginate-based, serve as carriers for chondrocytes or MSCs, often incorporating transforming growth factor-β (TGF-β) for chondrogenesis. A 2024 study demonstrated decellularized cartilage matrix hydrogels with TGF-β-loaded microspheres yielding neocartilage with compressive moduli approaching native tissue (0.5-1 MPa) in rabbit osteoarthritis models after 12 weeks of dynamic loading.[100] 3D bioprinting has advanced precision, with GelMA bioinks enabling layered constructs that integrate superficial and deep zone properties, improving defect filling in porcine models.[101] Stem cell approaches, including induced pluripotent stem cell (iPSC)-derived MSCs, repaired full-thickness defects in rabbits by 2020 trials, with histological scores indicating hyaline-like cartilage formation superior to controls.[102] However, long-term durability remains challenged by fibrotic integration and mechanical mismatch. Osteochondral tissue engineering targets the bone-cartilage interface, employing biphasic scaffolds or organoids to regenerate both compartments simultaneously. In 2023, gelatin microcryogel-based osteochondral organoids from human iPSCs formed dual-layered structures with distinct osteogenic (HA mineralization) and chondrogenic (glycosaminoglycan deposition) zones, exhibiting mechanical properties suitable for joint repair in rodent models.[103] These advances facilitate in situ self-assembly, reducing surgical invasiveness, though scalability and host integration persist as barriers, with preclinical outcomes showing 60-80% defect restoration but variable clinical efficacy in human trials.[102]Cardiovascular and Organ-Specific Engineering
Tissue engineering for cardiovascular applications targets the regeneration of myocardium, blood vessels, and heart valves, employing stem cell-derived cardiomyocytes, endothelial cells, and biomaterials to mimic native tissue mechanics and function. Engineered heart tissues (EHTs) constructed from human induced pluripotent stem cell (iPSC)-derived cardiomyocytes have demonstrated contractile properties and electrical coupling in vitro, with maturation enhanced through mechanical conditioning in bioreactors.[104] In preclinical primate models, epicardial engineered heart muscle (EHM) allografts integrated with host vasculature, showing reduced scar formation and improved ejection fraction up to 12 weeks post-implantation.[105] Clinical translation of EHTs remains in early stages, with the BIOVAT trial initiating in 2021 to assess safety of iPSC-derived EHM patches in ischemic heart failure patients, reporting no major adverse events in initial cohorts as of 2023.[106] Separately, the FDA approved Phase I trials for AD-NP1, a nanoparticle-based therapy promoting cardiomyocyte proliferation, in October 2025, following rodent studies showing 20-30% increase in heart tissue regeneration post-injury.[107] For vascular grafts, tissue-engineered vessels (TEVGs) seeded with autologous bone marrow mononuclear cells have achieved patency rates exceeding 80% in pediatric extracardiac conduits over 5 years, remodeling into living conduits with endothelialization and reduced thrombosis.[108] Human trials of acellular bioengineered arteries for peripheral vascular repair reported 90% graft patency at 1 year, with no infections or aneurysms in 2024 evaluations.[109] Heart valve tissue engineering utilizes decellularized matrices or hybrid scaffolds to produce valves with growth potential, addressing calcification issues in synthetic prosthetics; ovine models of bioengineered trileaflet valves exhibited native-like hemodynamics and recellularization after 6 months.[110] Organ-specific engineering extends to whole-heart constructs, where decellularized scaffolds repopulated with iPSC-derived cells restored partial electromechanical function in rat models, though vascularization limits scale beyond millimeters.[111] Advances in heart organoids, such as Stanford's 2025 vascularized models self-assembling endothelial networks, enable drug testing but face scalability challenges for therapeutic implantation.[112] For non-cardiac organs, liver tissue engineering via bioprinted hepatocyte spheroids achieves albumin secretion comparable to native tissue in microfluidic devices, with 2025 reviews highlighting improved zonation mimicking lobular architecture.[113] Kidney proximal tubule engineering using aligned nanofibrous scaffolds supports reabsorption functions in vitro, yet glomerular filtration remains unscaled in preclinical implants as of 2025.[114] These efforts underscore persistent hurdles in innervation and immune compatibility for functional organ replacement.Neural and Other Specialized Tissues
Neural tissue engineering employs scaffolds, stem cells, and bioactive molecules to regenerate damaged peripheral nerves, spinal cord, and central nervous system components. Nerve guidance conduits (NGCs) represent a primary application, with FDA-approved collagen-based devices like NeuraGen facilitating repair of peripheral nerve gaps under 3 cm since 2001 by supporting axonal regrowth and myelination.[115] Third-generation NGCs incorporating electrospun fibers, microchannels, and stem cells such as adipose-derived mesenchymal stem cells have demonstrated enhanced regeneration in rat sciatic nerve defect models, achieving functional recovery over 3 months in 10-mm gaps as of 2024.[115] Three-dimensional bioprinting advances neural applications by fabricating patient-specific constructs, such as fibrin-based lattices with human induced pluripotent stem cell (iPSC)-derived neural aggregates, which promote cell viability and integration in vitro.[116] In vivo, collagen/silk fibroin bioprinted scaffolds implanted in rat traumatic brain injury models improved cognitive and locomotor outcomes in studies from 2022.[116] Similarly, bioprinted hydrogel structures with neural stem cells have supported axon extension in spinal cord injury rat models, though human clinical translation remains limited by integration challenges.[116] Ocular tissue engineering targets specialized structures like the retina and cornea. iPSC-derived retinal organoids and retinal progenitor cells transplanted subretinally or intravitreally have restored photoreceptor function in preclinical models, with swine studies in 2021 showing preserved retinal pigment epithelium morphology and electroretinogram responses post-transplantation.[117] For corneal repair, limbal stem cell therapies have treated epithelial deficiencies since 2010, while biosynthetic keratoprostheses like CorNeat, implanted in humans by 2023, achieved medium-term visual acuity gains without rejection in initial cases.[117] Liver tissue engineering develops functional hepatic constructs via decellularization and recellularization, yielding scaffolds that maintain vascular architecture and support hepatocyte attachment. Recellularized human liver matrices exhibited synthetic function, including albumin production, for 21 days ex vivo, while rat models demonstrated viable grafts post-transplantation with improved survival in acute failure scenarios.[118] Three-dimensional bioprinting of hepatocyte-laden hydrogels has produced liver-like tissues for drug testing, with mouse studies confirming in vivo engraftment of iPSC-derived hepatocytes since 2015, though scalability limits clinical use.[118]Technical Challenges and Limitations
Vascularization and Integration Issues
One of the foremost challenges in tissue engineering is achieving sufficient vascularization, as engineered tissues thicker than approximately 100-200 micrometers exceed the passive diffusion limit for oxygen and nutrients, resulting in hypoxic cores and subsequent cell necrosis.[119] [120] This constraint arises from the reliance on diffusion from peripheral surfaces or host ingrowth, which cannot sustain metabolically active constructs mimicking native organ-scale tissues.[121] Strategies to promote vascularization, such as delivering angiogenic factors like vascular endothelial growth factor (VEGF) or co-culturing parenchymal cells with endothelial cells and pericytes, often yield immature capillary networks prone to regression, leakage, or inadequate perfusion pressure.[122] Advanced techniques including 3D bioprinting perfusable channels or decellularized extracellular matrix scaffolds have shown promise in preclinical models, yet they struggle with hierarchical vessel formation—spanning macrovasculature to capillaries—and long-term patency, frequently limited by thrombosis or endothelial dysfunction.[123] [124] Integration of vascularized constructs with host tissues exacerbates these issues, requiring rapid anastomosis to native vessels to avert ischemia during the critical post-implantation window, typically within hours to days.[125] Immune-mediated inflammation, foreign body responses to scaffolds, and biomechanical mismatches—such as stiffness disparities causing shear stress mismatches—can disrupt vessel connectivity and trigger graft failure, with studies reporting perfusion deficits in up to 70% of larger implants due to incomplete host remodeling.[126] [127] These persistent hurdles, rooted in the complexity of recapitulating native angiogenic cascades and extracellular matrix signaling, continue to impede clinical scalability despite iterative refinements.[128]Scalability and Reproducibility Problems
One major barrier to widespread adoption of tissue-engineered products is the challenge of scaling production from laboratory prototypes to clinical volumes, which demands consistent supply of cells, scaffolds, and bioactive factors while maintaining quality control. Autologous cell sourcing often yields insufficient quantities, particularly from elderly or diseased patients, necessitating prolonged expansion in controlled facilities that are resource-intensive and prone to contamination risks from media components like fetal calf serum.[5] Scaffold fabrication further complicates scalability, as achieving uniform microporosity (typically 100–500 μm) and interconnected structures for nutrient diffusion proves difficult at larger scales, with degradation rates varying widely—e.g., polyglycolic acid (PGA) erodes in about two weeks versus polylactic acid (PLLA) over 3–6 years—leading to unpredictable mechanical integrity.[5] A notable case illustrating these issues is Dermagraft, a fibroblast-seeded dermal substitute approved by the FDA for diabetic foot ulcers, where manufacturer Smith & Nephew encountered insurmountable production hurdles post-approval, including high operational costs and manual processes ill-suited for commercial throughput, ultimately rendering the product unprofitable despite demonstrated efficacy.[129] Transitioning to automated or bioreactor-based systems remains limited by the need for non-destructive in-process monitoring, as destructive testing disrupts batch integrity, and the integration of multiple raw materials (cells, polymers, growth factors) introduces compounding variables that amplify costs and timelines.[129] Reproducibility compounds scalability woes, stemming from the absence of standardized protocols for scaffold morpho-mechanical evaluation and cell-scaffold assembly, resulting in batch-to-batch variability influenced by factors like tissue heterogeneity, anisotropy, and hydration levels.[130] Biomechanical assessments lack consensus on parameters such as sample geometry, clamping, and size, with small cohorts undermining statistical power while larger sampling proves impractical for scarce human-derived materials; no dedicated standards exist for biological scaffolds akin to those for synthetic ones.[130] Cell phenotype instability during expansion—often leading to de-differentiation—and inconsistent neovascularization further erode reliability, as in vitro conditions fail to replicate in vivo stimuli uniformly across replicates.[5] These issues persist as of 2025, with editorial reviews highlighting ongoing gaps in protocol harmonization that impede industrial translation.[130]Biological and Mechanical Mismatches
Biological mismatches in tissue-engineered constructs stem from the inability of scaffolds and cells to fully replicate the dynamic biochemical and cellular signaling environments of native extracellular matrices (ECMs). Native tissues feature hierarchical, bioactive ECMs that provide spatiotemporal cues for cell adhesion, migration, proliferation, and differentiation through specific ligands and growth factors, which synthetic or decellularized scaffolds often lack without extensive functionalization. For instance, synthetic polymers like polycaprolactone exhibit limited inherent bioactivity, necessitating chemical modifications such as RGD peptide grafting to promote cell-ECM interactions, yet these modifications rarely achieve the multifunctionality of natural ECMs, leading to suboptimal cell phenotype maintenance and tissue remodeling.[131] [8] Inadequate replication of these cues can result in dedifferentiation of seeded cells or failure to induce host cell infiltration, as observed in many hydrogel-based constructs where static ligand presentation does not mimic the dynamic remodeling seen in vivo.[7] Immune incompatibility represents a further biological challenge, where non-native scaffold materials trigger chronic foreign body responses, including macrophage activation and fibrosis, rather than regenerative integration. Studies indicate that even biocompatible materials like poly(lactic-co-glycolic acid) elicit persistent inflammation due to degradation byproducts or surface topography mismatches with host tissues, contrasting with the immunotolerance of autologous ECMs. This mismatch is exacerbated in allogeneic or xenogeneic cell sources, where major histocompatibility complex disparities provoke rejection, underscoring the need for autologous cells or advanced immunomodulatory strategies.[132] [7] Mechanical mismatches occur when the stiffness, tensile strength, and viscoelastic properties of engineered constructs deviate from those of native tissues, compromising load-bearing capacity and mechanotransduction signals essential for tissue homeostasis. For bone tissue engineering, native cortical bone exhibits a Young's modulus of 7-30 GPa, while common polymeric scaffolds like collagen or poly(ε-caprolactone) typically range from 0.1-10 MPa, resulting in insufficient mechanical integrity and stress concentration at implant-host interfaces that promotes failure.[133] [134] In articular cartilage, where native compressive moduli approximate 0.5-2 MPa, engineered hydrogels often underperform due to poor fibril reinforcement, leading to premature wear under cyclic loading and altered chondrocyte responses via mismatched pericellular matrix-extracellular matrix interactions.[135] [136] Vascular grafts highlight compliance mismatches, with native arteries displaying dynamic compliance of 10-20% per 100 mmHg pressure change; mismatched synthetic grafts induce turbulent flow and intimal hyperplasia, as evidenced by burst failures in small-diameter constructs lacking sufficient elasticity. These disparities disrupt cellular mechanosensing, impairing ECM deposition and vascular smooth muscle cell alignment, and necessitate graded material designs to approximate native heterogeneity. Overall, such mismatches contribute to high failure rates in load-bearing applications, with computational models revealing that even 20-50% deviations in modulus can halve construct longevity in simulated physiological conditions.[137] [138] [134]Ethical and Societal Controversies
Stem Cell Sourcing and Embryonic Concerns
Human embryonic stem cells (hESCs), derived from the inner cell mass of blastocysts typically obtained from surplus in vitro fertilization embryos, have been a primary source for tissue engineering due to their pluripotency, enabling differentiation into multiple cell types essential for constructing complex tissues. The process of isolating hESCs requires destroying the embryo, which constitutes a human organism at the blastocyst stage capable of further development into a full human being if implanted.[139] [140] This destruction raises profound ethical concerns, as it involves the termination of what proponents of embryo protection argue is nascent human life with inherent moral status from fertilization onward.[141] Critics, including bioethicists, contend that such research prioritizes potential therapeutic benefits over the rights of the embryo, equating the act to a form of early human experimentation historically deemed unethical.[142] In tissue engineering applications, such as generating neural or cardiac tissues, hESCs offer high fidelity to developmental processes but at the cost of these moral objections, which have limited their widespread adoption.[143] In the United States, federal policy has reflected these tensions: President George W. Bush's 2001 executive order restricted National Institutes of Health funding to hESC lines derived before August 9, 2001, to avoid incentivizing new embryo destruction, a stance rooted in respecting the ethical boundaries of human life.[144] President Barack Obama reversed this in 2009 via executive order, expanding funding, though court challenges and subsequent administrations, including restrictions under President Donald Trump in 2019, underscored ongoing debates.[145] [146] Globally, countries like Germany and Austria prohibit hESC derivation involving embryo destruction, while others permit it under strict oversight, highlighting varying attributions of moral weight to the embryo.[147] These policies have pushed tissue engineering toward alternatives, as hESC reliance risks funding instability and public backlash, with surveys indicating majority opposition in some demographics to embryo-destructive research.[148] Induced pluripotent stem cells (iPSCs), reprogrammed from adult somatic cells using factors like Oct4, Sox2, Klf4, and c-Myc—first demonstrated in humans in 2007—provide a non-embryonic alternative, circumventing destruction concerns by generating patient-matched pluripotent cells for autologous tissue engineering.[32] iPSCs enable personalized constructs, reducing immune rejection risks inherent in allogeneic hESC transplants, and have been applied in engineering skin, cartilage, and vascular tissues.[149] However, iPSCs carry distinct risks compared to hESCs: reprogramming can induce genetic instability, epigenetic memory from the donor cell type, and higher tumorigenicity, including teratoma formation post-transplantation due to residual undifferentiated cells or insertional mutagenesis from viral vectors.[150] [151] Studies show iPSCs exhibit more transcriptional differences from hESCs than hESCs do among themselves, potentially affecting differentiation efficiency in tissue scaffolds, though non-integrating methods like mRNA delivery mitigate some oncogenic risks.[152] Adult stem cells, such as mesenchymal stem cells from bone marrow, offer multipotent sourcing without ethical or pluripotency-related hazards but limited differentiation scope, restricting them to orthopedic or soft tissue applications.[153] [154] While iPSCs have largely supplanted hESCs in recent tissue engineering protocols—evidenced by over 1,000 clinical trials involving iPSCs by 2023—their variability in quality control and potential for aberrant differentiation necessitate rigorous validation, as incomplete reprogramming can lead to off-target cell types unsuitable for functional tissues.[155] hESCs may retain advantages in purity and lower mutation rates for certain derivations, but the ethical imperative against embryo destruction, coupled with iPSC advancements, favors the latter for scalable, morally uncontroversial progress in regenerative medicine.[156] Ongoing research addresses iPSC limitations through genome editing and bioreactor optimization, yet the foundational debate persists: utilitarian arguments for hESC benefits must confront the causal reality that embryo sourcing precludes the potential life trajectory of the destroyed entity.[157] [158]Patient Autonomy, Identity, and Equity Debates
Patient autonomy in tissue engineering encompasses challenges in obtaining truly informed consent for experimental therapies, given the uncertainties inherent in novel constructs like bioengineered scaffolds and cell-based implants. Unlike conventional treatments, tissue-engineered products often involve unproven long-term integration with host tissues, raising questions about patients' ability to fully comprehend risks such as immune rejection or unforeseen oncogenic potential.[6] Ethical analyses emphasize that autonomy requires transparent disclosure of these limitations, yet clinical trial designs may pressure vulnerable patients—such as those with end-stage organ failure—into participation without adequate alternatives.[159] This tension underscores first-principles concerns over whether patient choice is meaningfully autonomous when desperation overrides risk evaluation.[160] Debates on identity arise from the potential for tissue-engineered interventions to alter bodily integrity and self-perception, particularly in cases of regenerative implants that replace or augment native anatomy. For instance, engineered organs derived from a patient's own cells may mitigate identity disruptions associated with allogeneic transplants, where recipients grapple with psychological incorporation of foreign donor tissue.[143] However, advanced applications like vascularized tissue constructs or neural interfaces could blur distinctions between natural and artificial body components, prompting philosophical inquiries into whether such modifications fundamentally redefine personal identity or embodiment.[161] Critics argue that while therapeutic intent predominates, elective uses for body modification—such as customized dermal grafts—risk commodifying identity, echoing broader bioethical concerns over enhancement versus repair.[162] Equity issues in tissue engineering highlight stark disparities in access, driven by high development costs and manufacturing complexities that limit scalability to affluent populations or regions with robust healthcare infrastructure. As of 2022, regenerative therapies like autologous cartilage implants remain confined to specialized centers in high-income countries, exacerbating global health divides where low-resource settings face barriers like supply chain vulnerabilities and regulatory gaps.[163] Empirical data from biomedical manufacturing critiques reveal that without targeted interventions, such innovations widen inequalities, as seen in uneven adoption of tissue-engineered skin substitutes post-burn, favoring insured patients over underserved demographics.[164] Proponents advocate for policy reforms to prioritize equitable distribution, yet systemic factors—including intellectual property barriers—persistently hinder broader dissemination.[165]Risk-Benefit Assessments and Overhype Critiques
Tissue engineering holds potential benefits in addressing organ shortages and treating degenerative diseases, such as regenerating bone defects or vascular grafts to improve patient outcomes and reduce reliance on donors.[166] However, risk-benefit assessments reveal significant uncertainties, particularly in clinical translation, where preclinical successes often fail to materialize due to issues like immune rejection, incomplete vascularization, and long-term functionality.[5] For instance, while engineered tissues may offer localized repair, the high costs and procedural complexities can outweigh benefits for non-life-threatening conditions unless superior efficacy is demonstrated over existing therapies.[167] A primary risk involves tumorigenicity, especially with pluripotent stem cell-derived constructs, where residual undifferentiated cells can form teratomas post-implantation, posing a direct threat to patient safety.[168] Studies emphasize that this risk stems from the inherent pluripotency enabling uncontrolled proliferation, necessitating rigorous purification protocols, yet even mesenchymal stem cells raise concerns over potential promotion of existing tumors via trophic effects.[169] [170] Balancing this against benefits requires evaluating incidence rates; preclinical models show variable tumorigenic potential depending on cell source and differentiation efficiency, but human data remain limited, complicating informed consent and trial approvals.[171] Critiques of overhype highlight how optimistic narratives in regenerative medicine literature amplify expectations, often portraying tissue engineering as imminent for whole-organ replacement despite persistent technical barriers.[172] For example, early promises of lab-grown bladders or tracheas in the 2000s generated media buzz, yet clinical outcomes have been inconsistent, with many trials stalling in phase II due to efficacy shortfalls or safety issues, representing less than 5% of advanced therapy medicinal products in ongoing European trials.[173] [174] This discrepancy arises from overreliance on animal models that inadequately predict human integration, fostering a cycle where funding chases hype rather than addressing root challenges like scalability.[175] Skeptics argue that such promotional discourse, prevalent in academic and industry reports, undervalues the incremental nature of progress, as evidenced by decades of research yielding few marketable products beyond simple skin or cartilage substitutes.[166] [176] Realistic assessments suggest that while niche applications like orthopedic scaffolds show cost-effectiveness in specific cases, broad overhype risks eroding public trust when timelines for complex organs extend indefinitely, urging a shift toward evidence-based projections over speculative breakthroughs.[177][178]Regulatory and Safety Frameworks
Current Global Regulations
Tissue-engineered products, encompassing scaffolds combined with cells or bioactive molecules for regenerative purposes, lack a unified global regulatory framework and are instead governed by national or regional agencies focused on safety, efficacy, and manufacturing consistency.[179] These products are typically classified as biologics, combination devices, or advanced therapies, requiring preclinical data, clinical trials, and post-market surveillance to mitigate risks such as immunogenicity, tumorigenesis, and contamination.[180] Regulatory stringency varies, with pathways emphasizing risk-based assessments to expedite innovative therapies while preventing unproven claims, though harmonization efforts through bodies like the International Pharmaceutical Regulators Forum remain limited to information-sharing rather than binding standards.[179] In the United States, the Food and Drug Administration (FDA) regulates tissue-engineered products as human cells, tissues, and cellular and tissue-based products (HCT/Ps) under 21 CFR Part 1271, distinguishing between section 361 products (minimal manipulation and homologous use, exempt from premarket approval but subject to good manufacturing practices and donor screening) and section 351 products (more than minimal manipulation, requiring investigational new drug applications and biologics license applications).[181] The Center for Biologics Evaluation and Research (CBER) issued its 2025 guidance agenda, prioritizing potency assays, viral safety, and materials sourcing for cellular therapies, with recent drafts addressing human- and animal-derived components to ensure identity and purity.[182][183] As of September 2025, FDA approvals for stem cell-derived tissue products remain sparse, with ongoing trials emphasizing vascularized constructs under expedited pathways like regenerative medicine advanced therapy designation.[184] The European Union classifies tissue-engineered medicinal products as a subset of advanced therapy medicinal products (ATMPs) under Regulation (EC) No 1394/2007, mandating centralized authorization via the European Medicines Agency (EMA) following scientific recommendations from the Committee for Advanced Therapies (CAT).[185] This includes requirements for quality control, non-clinical studies, and phased clinical trials, with guidelines updated in 2017 for cell therapy and tissue engineering potency and comparability.[186] By May 2025, 19 ATMPs were authorized EU-wide, though only a fraction involved tissue engineering (e.g., ChondroCelect in 2009 for cartilage repair), reflecting challenges in demonstrating long-term integration and scalability.[187] A June 2025 EMA guideline on clinical-stage ATMPs streamlines development by clarifying expectations for risk mitigation and adaptive trials.[188] Japan's Pharmaceuticals and Medical Devices Agency (PMDA) oversees regenerative medical products under the 2014 Pharmaceuticals and Medical Devices Act (PMD Act), enabling conditional and time-limited approvals for class III (high-risk) products after exploratory trials demonstrating probable benefit, with full approval contingent on confirmatory data within seven years.[189] Risk stratification (classes I-III) dictates oversight, from notification for low-risk autologous therapies to stringent review for allogeneic or genetically modified constructs; amendments effective June 2025 expanded the Act on the Safety of Regenerative Medicine to include in vivo gene therapies integrated with tissue engineering.[190] This framework has accelerated approvals, such as stem cell-based corneal therapies, prioritizing patient access amid evidence gaps.[180] In China, the National Medical Products Administration (NMPA) regulates tissue-engineered products as cell and gene therapy products (CTGTPs) or emerging ATMPs, with tightened controls since 2015 prohibiting unapproved stem cell clinics and requiring three-phase trials for marketing authorization.[191] Draft guidelines released June 10, 2025, define ATMP scope and classification, facilitating clinical trials amid a surge in applications (over 100 ongoing by mid-2025), though dual tracks for "techniques" versus "products" persist, complicating oversight of scaffold-cell hybrids.[192] Parallel requirements under the Drug Administration Law emphasize good manufacturing practices and ethical sourcing, with first stem cell therapy approvals emerging in 2025.[193]| Jurisdiction | Regulatory Body | Key Classification | Approval Features |
|---|---|---|---|
| United States | FDA/CBER | HCT/Ps (361/351) | Risk-based; minimal manipulation exempt from BLA; 2025 guidances on potency and materials.[194] |
| European Union | EMA/CAT | ATMPs (tissue-engineered) | Centralized MA; clinical guidelines effective 2025 for adaptive trials.[185] |
| Japan | PMDA | Regenerative Medical Products | Conditional approval for high-risk; time-limited post-market data (up to 7 years).[195] |
| China | NMPA | CTGTPs/ATMPs | Strict phased trials; 2025 drafts for classification amid trial surge.[192] |
Clinical Translation and Approval Hurdles
The clinical translation of tissue-engineered products (TEPs) is impeded by stringent regulatory requirements that demand rigorous demonstration of safety, efficacy, and manufacturing reproducibility, often spanning over a decade from preclinical stages to approval. In the United States, the FDA classifies many TEPs as biologics or combination products under Section 351 of the Public Health Service Act, necessitating a Biologics License Application (BLA) process that includes phased clinical trials (Phases I-III) to assess pharmacokinetics, immunogenicity, and long-term integration risks such as graft rejection or tumorigenesis.[196] Products ineligible for the less burdensome Section 361 human cells, tissues, and cellular and tissue-based products (HCT/P) pathway—due to significant manipulation or non-homologous use—face extended timelines, with preclinical data requirements emphasizing Good Laboratory Practice (GLP) compliance and large-animal models to predict human outcomes.[196] These pathways contribute to high attrition, as variability in patient-specific factors like immune response complicates endpoint definitions for efficacy, such as functional tissue restoration versus mere engraftment.[166] Approval hurdles are exacerbated by manufacturing challenges that must align with Current Good Manufacturing Practices (cGMP), including scalable production of viable cells or scaffolds without batch-to-batch inconsistencies, which regulators scrutinize for contamination risks or loss of bioactivity during cryopreservation and transport.[197] First-in-human trials often encounter logistical barriers, such as coordinating multidisciplinary teams for real-time cell processing and patient enrollment, leading to delays in trial initiation and data collection; a 2024 study of European first-in-human TEP trials highlighted communication gaps between engineers, clinicians, and regulators as a recurrent issue, prolonging investigational new drug (IND) submissions.[197] Cost estimates for advancing a TEP through FDA approval exceed $100 million, driven by the need for adaptive trial designs to address heterogeneous disease states, with failure rates in regenerative medicine trials reaching approximately 86% due to inadequate powering for rare adverse events or suboptimal surrogate markers.[166] Internationally, parallel challenges arise under frameworks like the European Medicines Agency's (EMA) Advanced Therapy Medicinal Products (ATMP) regulation, which mandates centralized authorization and post-approval pharmacovigilance, yet suffers from inconsistent harmonization across member states, resulting in prolonged review periods averaging 200-300 days.[198] In orthopedic applications, for instance, TEPs for cartilage repair struggle with regulatory demands for biomechanical equivalence to native tissue, where Phase II/III trials reveal mismatches in durability under physiological loads, prompting iterative redesigns and resubmissions.[199] Despite expedited designations like FDA's Regenerative Medicine Advanced Therapy (RMAT), uptake remains limited by evidentiary gaps, as only a fraction of the 834 global clinical trials involving engineered biomaterials (as of 2024) progress beyond Phase II, underscoring the causal link between regulatory stringency—rooted in historical gene therapy setbacks—and cautious approval thresholds.[200] These hurdles collectively constrain TEP commercialization, with fewer than 20 FDA approvals for complex TEPs as of 2023, primarily confined to skin and cartilage substitutes rather than vascular or organ-scale constructs.[201]Commercial and Economic Dimensions
Historical Market Trajectories
The field of tissue engineering emerged as a commercial endeavor in the early 1990s, spurred by foundational research combining scaffolds, cells, and bioactive factors to regenerate tissues. From 1990 to 2000, more than 70 companies worldwide invested over $3.5 billion in the sector, reflecting a compound annual growth rate of 16% amid optimism for applications in skin, cartilage, and vascular grafts.[202] Early efforts centered on allogeneic cell-based products, but high development costs and limited scalability constrained widespread adoption, leading to a wave of consolidations and bankruptcies by the mid-2000s.[3] Commercial milestones accelerated in the late 1990s with regulatory approvals for skin substitutes, such as Organogenesis's Apligraf bilayered skin equivalent, cleared by the FDA in 1998 for treating diabetic foot ulcers and venous leg ulcers, marking one of the first tissue-engineered products to reach market.[203] Subsequent approvals included Integra LifeSciences' dermal regeneration template in 1996 for burn wounds and Advanced Tissue Sciences' Dermagraft in 2001 for diabetic ulcers, though the latter company filed for bankruptcy in 2002 due to reimbursement challenges and manufacturing issues.[204] By the early 2000s, the market remained niche, dominated by orthopedics and wound care segments, with global sales estimated under $1 billion annually, hampered by clinical variability and payer skepticism over long-term efficacy compared to traditional grafts.[205] Market expansion gained traction from 2008 to 2011, when sales of commercial engineered tissue products tripled to $3.5 billion, driven by improved biomaterials and partnerships with established medical device firms like Stryker and Medtronic.[205] Venture funding surged in the 2010s, supporting innovations in 3D bioprinting and stem cell integration, though historical data indicate persistent hurdles: by 2016, the global tissue engineering and regeneration market stood at approximately $13.6 billion, reflecting a CAGR of around 12-15% from prior years but still below projections due to regulatory delays and ethical debates over cell sourcing.[206] Key players like Vericel Corporation commercialized autologous therapies, such as Carticel for cartilage repair approved in 1997, while Japan Tissue Engineering Co. advanced corneal equivalents, underscoring regional variations in adoption—North America led with robust FDA pathways, whereas Europe emphasized combination products under advanced therapy medicinal product regulations.[203]| Period | Key Market Indicators | Notable Developments |
|---|---|---|
| 1990-2000 | >$3.5B investments; 16% CAGR in companies | Formation of 70+ firms; focus on scaffolds and basic constructs[202] |
| 1998-2002 | First FDA approvals (e.g., Apligraf, Dermagraft); < $1B annual sales | Skin/wound products dominate; early bankruptcies highlight scalability risks[204] |
| 2008-2011 | Sales triple to $3.5B | Expansion into orthopedics; industry consolidations[205] |
| 2011-2016 | ~$13.6B global market by 2016; 12-15% CAGR | Rise in stem cell and bioprinting investments; persistent clinical hurdles[206] |


