Recent from talks
Nothing was collected or created yet.
Sucrose
View on WikipediaIt has been suggested that white sugar be merged into this article. (Discuss) Proposed since October 2025. |
 | |
 | |
| Names | |
|---|---|
| IUPAC name
β-D-Fructofuranosyl α-D-glucopyranoside
| |
| Systematic IUPAC name
(2R,3R,4S,5S,6R)-2-{[(2S,3S,4S,5R)-3,4-Dihydroxy-2,5-bis(hydroxymethyl)oxolan-2-yl]oxy}-6-(hydroxymethyl)oxane-3,4,5-triol | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.000.304 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties[1] | |
| C 12H 22O 11 | |
| Molar mass | 342.30 g/mol |
| Appearance | Colourless crystals or white powder |
| Density | 1.587 g/cm3 (0.0573 lb/cu in), solid |
| Melting point | None; decomposes at 186 °C (367 °F; 459 K) |
| 2.01 g/mL (20 °C (68 °F)) | |
| log P | −3.76 |
| Structure | |
| Monoclinic | |
| P21 | |
| Thermochemistry | |
Std enthalpy of
formation (ΔfH⦵298) |
−2,226.1 kJ/mol (−532.1 kcal/mol)[2] |
Std enthalpy of
combustion (ΔcH⦵298) |
1,349.6 kcal/mol (5,647 kJ/mol)[3] (Higher heating value) |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
29700 mg/kg (oral, rat)[5] |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 15 mg/m3 (total) TWA 5 mg/m3 (resp)[4] |
REL (Recommended)
|
TWA 10 mg/m3 (total) TWA 5 mg/m3 (resp)[4] |
IDLH (Immediate danger)
|
N.D.[4] |
| Safety data sheet (SDS) | ICSC 1507 |
| Related compounds | |
Related compounds
|
Lactose Maltose |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Sucrose, a disaccharide, is a sugar composed of glucose and fructose subunits. It is produced naturally in plants and is the main constituent of white sugar. It has the molecular formula C
12H
22O
11.
For human consumption, sucrose is extracted and refined from either sugarcane or sugar beet. Sugar mills – typically located in tropical regions near where sugarcane is grown – crush the cane and produce raw sugar which is shipped to other factories for refining into pure sucrose. Sugar beet factories are located in temperate climates where the beet is grown, and process the beets directly into refined sugar. The sugar-refining process involves washing the raw sugar crystals before dissolving them into a sugar syrup which is filtered and then passed over carbon to remove any residual colour. The sugar syrup is then concentrated by boiling under a vacuum and crystallized as the final purification process to produce crystals of pure sucrose that are clear, odorless, and sweet.
Sugar is often an added ingredient in food production and recipes. About 185 million tonnes of sugar were produced worldwide in 2017.
Etymology
[edit]The word sucrose was coined in 1857, by the English chemist William Miller[6] from the French sucre ("sugar") and the generic chemical suffix for sugars -ose. The abbreviated term Suc is often used for sucrose in scientific literature.
The name saccharose was coined in 1860 by the French chemist Marcellin Berthelot.[7] Saccharose is an obsolete name for sugars in general, especially sucrose.
Physical and chemical properties
[edit]Structure
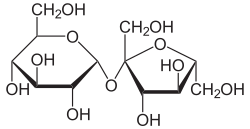
[edit]Sucrose's IUPAC name is O-α-D-glucopyranosyl-(1→2)-β-D-fructofuranoside. In this disaccharide, glucose and fructose are linked via a glycosidic linkage, i.e. an ether bond between C1 on the glucosyl subunit and C2 on the fructosyl unit. Glucose exists predominantly as a mixture of α and β "pyranose" anomers, but sucrose has only the α form. Fructose exists as a mixture of five tautomers but sucrose has only the β-D-fructofuranose form. Unlike most disaccharides, the glycosidic bond in sucrose is formed between the reducing ends of both glucose and fructose, and not between the reducing end of one and the non-reducing end of the other. This linkage inhibits further bonding to other saccharide units, and prevents sucrose from spontaneously reacting with cellular and circulatory macromolecules in the manner that glucose and other reducing sugars do. Since sucrose contains no anomeric hydroxyl groups, it is classified as a non-reducing sugar.[citation needed]
Sucrose crystallizes in the monoclinic space group P21 with room-temperature lattice parameters a = 1.08631 nm, b = 0.87044 nm, c = 0.77624 nm, β = 102.938°.[8][9]
Thermal and oxidative degradation
[edit]Sucrose does not melt at high temperatures. Instead, it decomposes at 186 °C (367 °F) to form caramel. Like other carbohydrates, it combusts to carbon dioxide and water by the simplified equation:
- C12H22O11 + 12 O2 → 12 CO2 + 11 H2O
Mixing sucrose with the oxidizer potassium nitrate produces the fuel known as rocket candy that is used to propel amateur rocket motors.[10]
C12H22O11 + 6 KNO3 → 9 CO + 3 N2 + 11 H2O + 3 K2CO3
This reaction is somewhat simplified though. Some of the carbon does get fully oxidized to carbon dioxide, and other reactions, such as the water-gas shift reaction also take place. A more accurate theoretical equation is:
C12H22O11 + 6.288 KNO3 → 3.796 CO2 + 5.205 CO + 7.794 H2O + 3.065 H2 + 3.143 N2 + 2.988 K2CO3 + 0.274 KOH[11]
Sucrose burns with chloric acid, formed by the reaction of hydrochloric acid and potassium chlorate:
8 HClO3 + C12H22O11 → 11 H2O + 12 CO2 + 8 HCl
Sucrose can be dehydrated with concentrated sulfuric acid to form a black, carbon-rich solid, as indicated in the following idealized equation:
H2SO4 (catalyst) + C12H22O11 → 12 C + 11 H2O + heat (and some H2O + SO3 as a result of the heat).
The formula for sucrose's decomposition can be represented as a two-step reaction: the first simplified reaction is dehydration of sucrose to pure carbon and water, and then carbon is oxidised to CO2 by O2 from air.
C12H22O11 + heat → 12 C + 11 H2O
12 C + 12 O2 → 12 CO2
| T (°C) | S (g/dL) |
|---|---|
| 50 | 259 |
| 55 | 273 |
| 60 | 289 |
| 65 | 306 |
| 70 | 325 |
| 75 | 346 |
| 80 | 369 |
| 85 | 394 |
| 90 | 420 |
Hydrolysis
[edit]Hydrolysis breaks the glycosidic bond converting sucrose into glucose and fructose. Hydrolysis is, however, so slow that solutions of sucrose can sit for years with negligible change. If the enzyme sucrase is added, however, the reaction will proceed rapidly.[12] Hydrolysis can also be accelerated with acids, such as cream of tartar or lemon juice, both weak acids. Likewise, gastric acidity converts sucrose to glucose and fructose during digestion, the bond between them being an acetal bond which can be broken by an acid.[citation needed]
Given (higher) heats of combustion of 1349.6 kcal/mol for sucrose, 673.0 for glucose, and 675.6 for fructose,[13] hydrolysis releases about 1.0 kcal (4.2 kJ) per mole of sucrose, or about 3 small calories per gram of product.
Synthesis and biosynthesis of sucrose
[edit]The biosynthesis of sucrose proceeds via the precursors UDP-glucose and fructose 6-phosphate, catalyzed by the enzyme sucrose-6-phosphate synthase. The energy for the reaction is gained by the cleavage of uridine diphosphate (UDP). Sucrose is formed by plants, algae and cyanobacteria but not by other organisms. Sucrose is the end product of photosynthesis and is found naturally in many food plants along with the monosaccharide fructose. In many fruits, such as pineapple and apricot, sucrose is the main sugar. In others, such as grapes and pears, fructose is the main sugar.[citation needed]
Chemical synthesis
[edit]After numerous unsuccessful attempts by others, Raymond Lemieux and George Huber succeeded in synthesizing sucrose from acetylated glucose and fructose in 1953.[14]
Measurement
[edit]The purity of sucrose is measured by polarimetry, i.e., the rotation of plane-polarized light by a sugar solution. The specific rotation at 20 °C (68 °F) using yellow "sodium-D" light (589 nm) is +66.47°. Commercial samples of sugar are assayed using this parameter. Sucrose does not deteriorate at ambient conditions.
The sugar industry uses degrees Brix (symbol °Bx), introduced by Adolf Brix, as units of measurement of the mass ratio of dissolved substance to water in a liquid. A 25 °Bx sucrose solution has 25 grams of sucrose per 100 grams of liquid; or, to put it another way, 25 grams of sucrose sugar and 75 grams of water exist in the 100 grams of solution. A 25 °Bx solution therefore has a concentration of 25 mass % sucrose.
The Brix degrees are measured using an infrared sensor. This measurement does not equate to Brix degrees from a density or refractive index measurement, because it will specifically measure dissolved sugar concentration instead of all dissolved solids. When using a refractometer, one should report the result as "refractometric dried substance" (RDS). One might speak of a liquid as having 20 °Bx RDS. This refers to a measure of percent by weight of total dried solids and, although not technically the same as Brix degrees determined through an infrared method, renders an accurate measurement of sucrose content, since sucrose in fact forms the majority of dried solids. The advent of in-line infrared Brix measurement sensors has made measuring the amount of dissolved sugar in products economical using a direct measurement.
Sources
[edit]In nature, sucrose is present in many plants, and in particular their roots, fruits and nectars, because it serves as a way to store energy, primarily from photosynthesis.[15][16] Many mammals, birds, insects and bacteria accumulate and feed on the sucrose in plants and for some it is their main food source. Although honeybees consume sucrose, the honey they produce consists primarily of fructose and glucose, with only trace amounts of sucrose.[17]
As fruits ripen, their sucrose content usually rises sharply, but some fruits contain almost no sucrose at all. This includes grapes, cherries, blueberries, blackberries, figs, pomegranates, tomatoes, avocados, lemons and limes. In grapes, for instance, during ripening the sucrose molecules are hydrolyzed (separated) into glucose and fructose.[citation needed]
Sucrose is a naturally occurring sugar, but with the advent of industrialization, it has been increasingly refined and consumed in all kinds of processed foods.[citation needed]
Production
[edit]Table sugar (sucrose) comes from plant sources. Two important sugar crops predominate: sugarcane (Saccharum spp.) and sugar beets (Beta vulgaris), in which sugar can account for 12% to 20% of the plant's dry weight. The plant material is separated to isolate the sucrose-rich portions. Purification of the sucrose exploits the good solubility of sucrose in water. After this aqueous extraction, a variety of tools and techniques allow further purification and production of solid forms suited for the markets.
Culinary sugars
[edit]
Mill white
[edit]Mill white, also called plantation white, crystal sugar or superior sugar is produced from raw sugar. It is exposed to sulfur dioxide during the production to reduce the concentration of color compounds and helps prevent further color development during the crystallization process. Although common to sugarcane-growing areas, this product does not store or ship well. After a few weeks, its impurities tend to promote discoloration and clumping; therefore this type of sugar is generally limited to local consumption.[18]
Blanco directo
[edit]Blanco directo, a white sugar common in India and other south Asian countries, is produced by precipitating many impurities out of cane juice using phosphoric acid and calcium hydroxide, similar to the carbonatation technique used in beet sugar refining. Blanco directo is purer than mill white sugar, but less pure than white refined sugar.
White refined
[edit]White refined is the most common form of sugar in North America and Europe. Refined sugar is made by dissolving and purifying raw sugar using phosphoric acid similar to the method used for blanco directo, a carbonatation process involving calcium hydroxide and carbon dioxide, or by various filtration strategies. It is then further purified by filtration through a bed of activated carbon or bone char. Beet sugar refineries produce refined white sugar directly without an intermediate raw stage.[clarification needed]
White refined sugar is typically sold as granulated sugar, which has been dried to prevent clumping and comes in various crystal sizes for home and industrial use:

- Coarse-grain, such as sanding sugar (also called "pearl sugar", "decorating sugar", nibbed sugar or sugar nibs) is a coarse grain sugar used to add sparkle and flavor atop baked goods and candies. Its large reflective crystals will not dissolve when subjected to heat.
- Granulated, familiar as table sugar, with a grain size about 0.5 mm across.[19] "Sugar cubes" are lumps for convenient consumption produced by mixing granulated sugar with sugar syrup.
- Caster (0.35 mm),[19] a very fine sugar in Britain and other Commonwealth countries, so-named because the grains are small enough to fit through a sugar caster which is a small vessel with a perforated top, from which to sprinkle sugar at table.[20] Commonly used in baking and mixed drinks, it is sold as "superfine" sugar in the United States. Because of its fineness, it dissolves faster than regular white sugar and is especially useful in meringues and cold liquids. Caster sugar can be prepared at home by grinding granulated sugar for a couple of minutes in a mortar or food processor.
- Powdered, 10X sugar, confectioner's sugar (0.060 mm), or icing sugar (0.024 mm), produced by grinding sugar to a fine powder. The manufacturer may add a small amount of anticaking agent to prevent clumping — either corn starch (1% to 3%) or tri-calcium phosphate.

Brown sugar comes either from the late stages of cane sugar refining, when sugar forms fine crystals with significant molasses content, or from coating white refined sugar with a cane molasses syrup (blackstrap molasses). Brown sugar's color and taste become stronger with increasing molasses content, as do its moisture-retaining properties. Brown sugars also tend to harden if exposed to the atmosphere, although proper handling can reverse this.
Consumption
[edit]Refined sugar was a luxury before the 18th century. It became widely popular in the 18th century, then graduated to becoming a necessary food in the 19th century. This evolution of taste and demand for sugar as an essential food ingredient unleashed major economic and social changes.[21] Eventually, table sugar became sufficiently cheap and common enough to influence standard cuisine and flavored drinks.
Sucrose forms a major element in confectionery and desserts. Cooks use it for sweetening. It can also act as a food preservative when used in sufficient concentrations, and thus is an important ingredient in the production of fruit preserves. Sucrose is important to the structure of many foods, including biscuits and cookies, cakes and pies, candy, and ice cream and sorbets. It is a common ingredient in many processed and so-called "junk foods".
Nutritional information
[edit]| Nutritional value per 100 g (3.5 oz) | |||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Energy | 1,620 kJ (390 kcal) | ||||||||||||||||||||||||||||
100 g | |||||||||||||||||||||||||||||
0 g | |||||||||||||||||||||||||||||
0 g | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| †Percentages estimated using US recommendations for adults,[22] except for potassium, which is estimated based on expert recommendation from the National Academies.[23] | |||||||||||||||||||||||||||||
Fully refined sugar is 99.9% sucrose, thus providing only carbohydrate as dietary nutrient and 390 kilocalories per 100 g serving (table).[24] There are no micronutrients of significance in fully refined sugar (table).[24]
Metabolism of sucrose
[edit]
In humans and other mammals, sucrose is broken down into its constituent monosaccharides, glucose and fructose, by sucrase or isomaltase glycoside hydrolases, which are located in the membrane of the microvilli lining the duodenum.[25][26] The resulting glucose and fructose molecules are then rapidly absorbed into the bloodstream. In bacteria and some animals, sucrose is digested by the enzyme invertase. Sucrose is an easily assimilated macronutrient that provides a quick source of energy, provoking a rapid rise in blood glucose upon ingestion. Sucrose, as a pure carbohydrate, has an energy content of 3.94 calories per gram (or 17 kilojoules per gram).
If consumed excessively, sucrose may contribute to the development of metabolic syndrome, including increased risk for type 2 diabetes, insulin resistance, weight gain and obesity in adults and children.[27][28]
Tooth decay
[edit]Tooth decay (dental caries) has become a pronounced health hazard associated with the consumption of sugars, especially sucrose. Oral bacteria such as Streptococcus mutans live in dental plaque and metabolize any free sugars (not just sucrose, but also glucose, lactose, fructose, and cooked starches)[29] into lactic acid. The resultant lactic acid lowers the pH of the tooth's surface, stripping it of minerals in the process known as tooth decay.[30][31]
All 6-carbon sugars and disaccharides based on 6-carbon sugars can be converted by dental plaque bacteria into acid that demineralizes teeth, but sucrose may be uniquely useful to Streptococcus sanguinis (formerly Streptococcus sanguis) and Streptococcus mutans.[32][33] Sucrose is the only dietary sugar that can be converted to sticky glucans (dextran-like polysaccharides) by extracellular enzymes.[34] These glucans allow the bacteria to adhere to the tooth surface and to build up thick layers of plaque. The anaerobic conditions deep in the plaque encourage the formation of acids, which leads to carious lesions. Thus, sucrose could enable S. mutans, S. sanguinis and many other species of bacteria to adhere strongly and resist natural removal, e.g. by flow of saliva, although they are easily removed by brushing. The glucans and levans (fructose polysaccharides) produced by the plaque bacteria also act as a reserve food supply for the bacteria. Such a special role of sucrose in the formation of tooth decay is much more significant in light of the almost universal use of sucrose as the most desirable sweetening agent. Widespread replacement of sucrose by high-fructose corn syrup (HFCS) has not diminished the danger from sucrose. If smaller amounts of sucrose are present in the diet, they will still be sufficient for the development of thick, anaerobic plaque and plaque bacteria will metabolise other sugars in the diet,[33] such as the glucose and fructose in HFCS.
Glycemic index
[edit]Sucrose is a disaccharide made up of 50% glucose and 50% fructose and has a glycemic index of 65.[35] Sucrose is digested rapidly,[36][37] but has a relatively low glycemic index due to its content of fructose, which has a minimal effect on blood glucose.[36]
As with other sugars, sucrose is digested into its components via the enzyme sucrase to glucose (blood sugar). The glucose component is transported into the blood where it serves immediate metabolic demands, or is converted and reserved in the liver as glycogen.[37]
Gout
[edit]The occurrence of gout is connected with an excess production of uric acid. A diet rich in sucrose may lead to gout as it raises the level of insulin, which prevents excretion of uric acid from the body. As the concentration of uric acid in the body increases, so does the concentration of uric acid in the joint liquid and beyond a critical concentration, the uric acid begins to precipitate into crystals. Researchers have implicated sugary drinks high in fructose in a surge in cases of gout.[38]
Sucrose intolerance
[edit]UN dietary recommendation
[edit]In 2015, the World Health Organization published a new guideline on sugars intake for adults and children, as a result of an extensive review of the available scientific evidence by a multidisciplinary group of experts. The guideline recommends that both adults and children ensure their intake of free sugars (monosaccharides and disaccharides added to foods and beverages by the manufacturer, cook or consumer, and sugars naturally present in honey, syrups, fruit juices and fruit juice concentrates) is less than 10% of total energy intake. A level below 5% of total energy intake brings additional health benefits, especially with regards to dental caries.[39]
Religious concerns
[edit]The sugar refining industry often uses bone char (calcinated animal bones) for decolorizing.[40][41] About 25% of sugar produced in the U.S. is processed using bone char as a filter, the remainder being processed with activated carbon. As bone char does not seem to remain in finished sugar, Jewish religious leaders consider sugar filtered through it to be pareve, meaning that it is neither meat nor dairy and may be used with either type of food. However, the bone char must source to a kosher animal (e.g. cow, sheep) for the sugar to be kosher.[41]
References
[edit]- ^ Sucrose, International Chemical Safety Card 1507, Geneva: International Programme on Chemical Safety, November 2003
- ^ Haynes, W. M.; Lide, David R.; Bruno, Thomas J., eds. (2014–2015). CRC Handbook of Chemistry and Physics (95th ed.). CRC Press. pp. 5–40. ISBN 978-1482208672.
- ^ CRC Handbook of Chemistry and Physics, 49th edition, 1968–1969, p. D-188.
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0574". National Institute for Occupational Safety and Health (NIOSH).
- ^ "57-50-1 - CZMRCDWAGMRECN-UGDNZRGBSA-N - Sucrose [JAN:NF]". ChemIDplus. Archived from the original on 2014-08-12. Retrieved 2014-08-10.
- ^ William Allen Miller, Elements of Chemistry: Theoretical and Practical, Part III. Organic Chemistry (London, England: John W. Parker and son, 1857), pages 52 and 54 Archived 2017-04-24 at the Wayback Machine.
- ^ Marcellin Berthelot, Chimie organique fondée sur la synthèse (Paris, France: Mallet-Bachelier, 1860), pp. 254–55 Archived 2014-06-27 at the Wayback Machine.
- ^ Beevers, C. A.; McDonald, T. R. R.; Robertson, J. H.; Stern, F. (1952). "The crystal structure of sucrose". Acta Crystallogr. 5 (5): 689–90. Bibcode:1952AcCry...5..689B. doi:10.1107/S0365110X52001908.
- ^ Hynes, R. C.; Le Page, Y. (1991). "Sucrose, a convenient test crystal for absolute structures". Journal of Applied Crystallography. 24 (4): 352. Bibcode:1991JApCr..24..352H. doi:10.1107/S0021889891002492.
- ^ Nakka, Richard. "Potassium Nitrate/Sucrose Propellant (KNSU)". Experimental Rocketry. Archived from the original on 2015-10-26. Retrieved 2015-11-19.
- ^ Nakka, Richard. "KN-Sucrose (KNSU) Propellant Chemistry and Performance Characteristics". Experimental Rocketry. Archived from the original on 2014-11-18. Retrieved 2014-08-21.
- ^ "Sucrase" Archived 2010-04-25 at the Wayback Machine, Encyclopædia Britannica Online
- ^ All three from CRC Handbook of Chemistry and Physics, 49th edition, 1968-1969, pp. D-184-189.
- ^ Lemieux, R. U.; Huber, G. (1953). "A chemical synthesis of sucrose". J. Am. Chem. Soc. 75 (16): 4118. Bibcode:1953JAChS..75.4118L. doi:10.1021/ja01112a545.
- ^ John E. Lunn (December 2008). "Sucrose Metabolism". Encyclopedia of Life Sciences. John Wiley & Sons Ltd. doi:10.1002/9780470015902.a0021259. ISBN 978-0470016176.
- ^ "Foods highest in Sucrose". SelfNutritiondata. Condé Nast. Archived from the original on 2015-07-19.
- ^ Douglas M. Considine (1982). Considine, Douglas M; Considine, Glenn D (eds.). Foods and Food Production Encyclopedia (1 ed.). Van Nostrand Reinhold Company Inc. p. 956. doi:10.1007/978-1-4684-8511-0. ISBN 978-1-4684-8513-4.
- ^ Steindl, Roderick (2005). Hogarth, DM (ed.). Syrup Clarification for Plantation White Sugar to meet New Quality Standards (PDF). Guatemala, Guatemala City: Proceedings of the XXV Congress of International Society of Sugar Cane Technologists. pp. 106–16. Archived (PDF) from the original on 2013-08-10.
- ^ a b Sugar Crystal Challenge Archived 2013-05-08 at the Wayback Machine. IEEE
- ^ "castor, n.2." OED Online. Oxford University Press, June 2017. Web. 25 July 2017. It says castor is a misspelling that is now the preferred spelling.
- ^ Mintz, Sidney (1986). Sweetness and Power: The Place of Sugar in Modern History. Penguin. ISBN 978-0-14-009233-2.
- ^ United States Food and Drug Administration (2024). "Daily Value on the Nutrition and Supplement Facts Labels". FDA. Archived from the original on 2024-03-27. Retrieved 2024-03-28.
- ^ "TABLE 4-7 Comparison of Potassium Adequate Intakes Established in This Report to Potassium Adequate Intakes Established in the 2005 DRI Report". p. 120. In: Stallings, Virginia A.; Harrison, Meghan; Oria, Maria, eds. (2019). "Potassium: Dietary Reference Intakes for Adequacy". Dietary Reference Intakes for Sodium and Potassium. pp. 101–124. doi:10.17226/25353. ISBN 978-0-309-48834-1. PMID 30844154. NCBI NBK545428.
- ^ a b "Nutrition Facts for sugars, granulated [sucrose] per 100 g (USDA National Nutrient Database, SR-21)". Conde Nast. 2014. Archived from the original on 7 March 2015. Retrieved 6 March 2015.
- ^ Gray GM (1971). "Intestinal digestion and maldigestion of dietary carbohydrate". Annual Review of Medicine. 22: 391–404. doi:10.1146/annurev.me.22.020171.002135. PMID 4944426.
- ^ Kaneko J.J. (2008) "Carbohydrate metabolism and its diseases" Archived 2014-09-22 at the Wayback Machine, p. 46 in Kaneko J.J., Harvey J.W., Bruss M.L. (eds.) Clinical Biochemistry of Domestic Animals, San Diego, CA: Academic Press, ISBN 012370491X.
- ^ Malik, V. S.; Popkin, B. M.; Bray, G. A.; Despres, J.-P.; Willett, W. C.; Hu, F. B. (2010). "Sugar-sweetened beverages and risk of metabolic syndrome and type 2 diabetes: A meta-analysis". Diabetes Care. 33 (11): 2477–83. doi:10.2337/dc10-1079. PMC 2963518. PMID 20693348.
- ^ Malik, Vasanti S.; Pan, An; Willett, Walter C.; Hu, Frank B. (2013-10-01). "Sugar-sweetened beverages and weight gain in children and adults: a systematic review and meta-analysis". The American Journal of Clinical Nutrition. 98 (4): 1084–1102. doi:10.3945/ajcn.113.058362. ISSN 0002-9165. PMC 3778861. PMID 23966427. Archived from the original on 2018-01-09. Retrieved 2018-12-21.
- ^ "What causes tooth decay?". Animated-teeth.com. Archived from the original on 2010-02-09. Retrieved 2010-05-05.
- ^ Tooth Decay Archived 2014-10-25 at the Wayback Machine. Elmhurst.edu. Retrieved on 2011-11-18.
- ^ What causes tooth decay? Archived 2010-02-09 at the Wayback Machine. Animated-teeth.com. Retrieved on 2011-11-18.
- ^ Tanzer, JM (August 1979). "Essential dependence of smooth surface caries on, and augmentation of fissure caries by, sucrose and Streptococcus mutans infection". Infection and Immunity. 25 (2): 526–31. doi:10.1128/IAI.25.2.526-531.1979. PMC 443577. PMID 489122.
- ^ a b Darlington, W. (August 1979). Metabolism of sucrose by Stepococcus sanguis 804 (NCTC 10904) and its relevance to the oral environment (Ph.D Thesis). University of Glasgow.
- ^ Ryan KJ, Ray CG, eds. (2004). Sherris Medical Microbiology (4th ed.). McGraw Hill. ISBN 978-0-8385-8529-0.
- ^ Wolever, Thomas M. S. (2006). The Glycaemic Index: A Physiological Classification of Dietary Carbohydrate. CABI. p. 64. ISBN 9781845930523. Archived from the original on 2017-12-16.
- ^ a b Wolever, Thomas M. S. (2006). The Glycaemic Index: A Physiological Classification of Dietary Carbohydrate. CABI. p. 65. ISBN 9781845930523. Archived from the original on 2017-12-16.
- ^ a b Food and Nutrition Board, Institute of Medicine of the US National Academies (2005). Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein, and amino acids (macronutrients). National Academies Press. p. 323. doi:10.17226/10490. ISBN 978-0-309-08525-0. Archived from the original on 2015-07-15.
- ^ Magidenko, Leonid (2007-07-30). "Nutrients for Gout – good and bad". ABCVitaminsLife.com. Archived from the original on 2009-02-23. Retrieved 2010-05-05.
- ^ See Guideline: Sugars intake for adults and children. Geneva: World Health Organization; 2015 Archived 2015-08-17 at the Wayback Machine
- ^ The Great Sugar Debate: Is it Vegan? Archived 2009-09-19 at the Wayback Machine. Vegfamily.com. Retrieved on 2011-11-18.
- ^ a b Yacoubou, MS, Jeanne (2007). "Is Your Sugar Vegan? An Update on Sugar Processing Practices" (PDF). Vegetarian Journal. 26 (4). Baltimore, MD: The Vegetarian Resource Group: 16–20. Archived (PDF) from the original on 2008-04-09. Retrieved 2007-04-04.
Further reading
[edit]- Yudkin, J.; Edelman, J.; Hough, L. (1973). Sugar: Chemical, Biological and Nutritional Aspects of Sucrose. Butterworth. ISBN 978-0-408-70172-3.
External links
[edit]Sucrose
View on GrokipediaNomenclature and Etymology
Origins of the term and chemical designation
The term "sucrose" was coined in 1857 by English chemist William Miller, combining the French word sucre (sugar) with the suffix -ose, conventionally used for carbohydrates in chemical nomenclature.[3][4] The root sucre derives from medieval Latin saccharum, which entered European languages via Arabic sukkar and Greek sakkharon, ultimately tracing to Sanskrit śarkarā, an ancient term for gravel or pebbles that evoked the crystalline granules of refined sugar.[5][6] In scientific contexts, "sucrose" distinguishes the pure disaccharide from broader vernacular terms like "table sugar" or "cane sugar," which historically encompassed impure or mixed sweeteners.[7] The alternative name "saccharose," prevalent in French and some older chemical literature, emphasizes the Latin saccharum root but has largely yielded to "sucrose" in English for consistency with IUPAC conventions.[7] Sucrose's systematic chemical designation, α-D-glucopyranosyl-(1→2)-β-D-fructofuranoside, specifies its glycosidic linkage between an α-D-glucose unit in pyranose form and a β-D-fructose unit in furanose form, as standardized in 19th-century carbohydrate chemistry amid advances in structural elucidation.[8][9] This IUPAC name prioritizes precision over common descriptors, enabling unambiguous reference in empirical and theoretical work.[8]Chemical Structure and Properties
Molecular composition and structural formula
Sucrose possesses the molecular formula C12H22O11, consisting of 12 carbon atoms, 22 hydrogen atoms, and 11 oxygen atoms arranged in a disaccharide structure.[8] It comprises one molecule of α-D-glucose and one molecule of β-D-fructose, linked via a glycosidic bond that connects the anomeric carbon at position 1 (C1) of the glucose unit to the anomeric carbon at position 2 (C2) of the fructose unit.[10] This bond is specifically an α(1→2) glycosidic linkage, with the glucose adopting a pyranose ring conformation and the fructose a furanose ring in the solid state and predominantly in solution. The involvement of both anomeric carbons in the glycosidic bond distinguishes sucrose as a non-reducing disaccharide, incapable of undergoing mutarotation or reducing reactions like those observed with Tollens' or Fehling's reagents, unlike reducing disaccharides such as maltose (α-D-glucose α(1→4) D-glucose) or lactose (β-D-galactose β(1→4) D-glucose), which possess a free anomeric hydroxyl group.[11] This structural feature, confirmed through X-ray crystallography and spectroscopic analyses, ensures the stability of the acetal linkages without equilibrium between open-chain and cyclic forms.[12] The full systematic name is O-α-D-glucopyranosyl-(1→2)-β-D-fructofuranoside, reflecting the stereospecific D-configurations and ring forms inherent to its biosynthesis and isolation from natural sources.[8]Physical characteristics
Sucrose is a white, odorless crystalline solid at room temperature.[8] Its density measures 1.587 g/cm³.[3] The compound does not have a distinct melting point but decomposes at 186 °C.[8] Sucrose exhibits high solubility in water, dissolving at approximately 200 g per 100 mL at 20 °C, while its solubility decreases markedly in alcohols such as ethanol (0.6 g/100 mL) and methanol (1 g/100 mL).[13] It displays optical activity with a specific rotation of +66.5° in water.[14] Sucrose is moderately hygroscopic, readily absorbing atmospheric moisture, though it remains stable under dry conditions.[8] In terms of relative sweetness, sucrose is the reference standard with an index of 1.0; glucose rates at 0.65–0.75 and fructose at 1.05–1.25 relative to sucrose, though fructose can reach up to 1.7 times under certain concentrations and temperatures.[9]Chemical reactivity and degradation
Sucrose, a non-reducing disaccharide, primarily undergoes hydrolysis via cleavage of its α-D-glucopyranosyl-(1→2)-β-D-fructofuranosidic bond, yielding equimolar amounts of D-glucose and D-fructose, collectively termed invert sugar.[15] This reaction proceeds under acidic conditions through a protonation mechanism involving the glycosidic oxygen, facilitating nucleophilic attack by water and subsequent bond fission; the process is pseudo-first-order with respect to sucrose concentration and exhibits strong dependence on pH and temperature, with rate constants increasing exponentially per the Arrhenius equation (activation energy approximately 120-130 kJ/mol in dilute acid). [16] Enzymatic hydrolysis, catalyzed by β-fructofuranosidase (invertase), accelerates the reaction significantly at neutral to mildly acidic pH (optimum around 4.5) and moderate temperatures (optimal 50-60°C for many sources), following Michaelis-Menten kinetics with Km values typically 10-300 mM for sucrose.[17] [18] Thermal degradation of sucrose initiates upon melting (around 186°C) or in aqueous solutions at lower temperatures (above 110-150°C), primarily through caramelization—a pyrolysis process involving dehydration, fragmentation, and polymerization to form colored melanoidins and volatile compounds.[19] Key intermediates include 5-hydroxymethylfurfural (HMF), derived from the fructosyl moiety via triple dehydration, with yields increasing with temperature and time; further oxidative breakdown can produce levulinic acid and formic acid from HMF hydrolysis under acidic or hydrothermal conditions.[20] [21] The reaction kinetics are complex, often modeled as consecutive first-order steps, with HMF formation rates peaking before secondary degradation.[22] Sucrose exhibits resistance to direct microbial fermentation due to its non-reducing nature and the energetic barrier of the glycosidic bond, which prevents facile uptake or metabolism by many yeasts and bacteria lacking extracellular invertase; hydrolysis to monosaccharides is prerequisite for subsequent glycolytic utilization.[23] [24] Chemical synthesis of sucrose remains challenging, stemming from the bond's high free energy (comparable to nucleotide-activated sugars) and regioselective demands of forming the specific 1→2 linkage without side reactions; early total syntheses required multi-step protection and activation strategies, with modern approaches relying on fructofuranosyl donors but still yielding low efficiencies due to competing anomeric configurations.[18] [25] [26]Biosynthesis and Natural Occurrence
Biosynthetic pathways in plants
Sucrose biosynthesis in plants primarily occurs in the cytosol of photosynthetic leaf mesophyll cells, serving as a mechanism to export excess photosynthate from the chloroplast where triose phosphates are generated via the Calvin-Benson cycle. The committed step involves the condensation of uridine diphosphate glucose (UDP-glucose) and fructose 6-phosphate to form sucrose 6-phosphate, catalyzed by the enzyme sucrose-phosphate synthase (SPS; EC 2.4.1.14). This intermediate is then rapidly dephosphorylated to sucrose by sucrose-phosphate phosphatase (SPP; EC 3.1.3.24), rendering the reaction effectively irreversible under physiological conditions due to the high free energy of hydrolysis.[27][28] SPS activity is tightly regulated to match sucrose production with photosynthetic carbon flux and sink demand, primarily through reversible protein phosphorylation and allosteric modulation. Dephosphorylation activates SPS, mediated by protein phosphatases responsive to light and osmotic signals, while phosphorylation by kinases inhibits it, often triggered by darkness or high sucrose levels. Allosteric activators such as glucose 6-phosphate enhance SPS affinity for substrates, whereas inorganic phosphate acts as an inhibitor, linking enzyme activity to cellular phosphate status and photosynthetic rates.[28][29][30] In sink tissues, sucrose synthase (SuSy; EC 2.4.1.13) facilitates the reversible cleavage of sucrose into UDP-glucose and fructose, providing precursors for starch synthesis, cell wall biosynthesis, and glycolysis while unloading sucrose from phloem during source-to-sink transport. This contrasts with the SPS pathway's dominance in sources, where SuSy contributes minimally to net synthesis but supports transient UDP-glucose recycling. SuSy's orientation toward the plasma membrane in phloem parenchyma aids efficient carbon partitioning, with isoform-specific expression influencing sink strength and overall plant growth.[31][32][33] Recent studies have explored engineering sucrose pathways via heterologous expression of microbial enzymes like sucrose phosphorylase (SPase; EC 2.4.1.7), which catalyzes sucrose formation from glucose 1-phosphate and fructose, to enhance yields in non-plant systems or modify plant metabolism for biotech applications such as glycoside production. Directed evolution and structural analyses of SPase variants have improved thermostability and substrate specificity, with 2024 reports detailing optimized expression in Escherichia coli yielding up to 200 g/L sucrose equivalents, informing potential chloroplast-targeted modifications in crops for increased sink capacity.[34][35]Natural sources and ecological role
Sucrose accumulates in high concentrations in the mature stems of sugarcane (Saccharum officinarum), reaching up to 20% of culm dry weight or 400–700 mM in internodes, enabling efficient carbon storage in this tropical grass.[36] [37] In sugar beet (Beta vulgaris subsp. vulgaris), a biennial root crop, sucrose comprises 16–20% of root fresh weight, primarily in the taproot as a overwintering energy reserve.[38] Lower levels occur transiently in photosynthetic source tissues across many plants, including fruits such as apples (1–3% of total sugars) and vegetables like carrots, as well as in sap from sugar maple (Acer saccharum) and nectar of flowering plants.[1] In plant physiology, sucrose functions as the primary transport carbohydrate, synthesized in source leaves from photosynthetic products via sucrose phosphate synthase and sucrose synthase, then loaded into phloem sieve elements for long-distance translocation to sink organs like roots, stems, and reproductive tissues.[39] [40] This phloem loading, often via apoplastic or symplastic pathways involving sucrose transporters (SUTs), drives mass flow under pressure gradients, supplying carbon skeletons and energy for growth, maintenance, and reproduction while preventing feedback inhibition of photosynthesis.[41] Sucrose also regulates cellular osmosis by maintaining turgor in expanding tissues and acts as a compatible solute during stress, such as drought, where its accumulation stabilizes membranes and proteins without disrupting metabolism.[42] Ecologically, sucrose's role extends to interspecies interactions, as its export into nectar attracts pollinators in angiosperms, enhancing reproductive success, while high stem accumulation in species like sugarcane supports rapid biomass production in competitive tropical environments.[43] Evolutionarily, sucrose as the dominant non-reducing disaccharide for transport is conserved across angiosperms, with ancient SUT gene families predating whole-genome duplications, reflecting selection for efficient photoassimilate partitioning over alternatives like raffinose in some non-angiosperm lineages.[44] In non-accumulator species, sucrose levels remain low to favor hexose metabolism, but its biosynthetic and transport machinery underscores a core adaptation for vascular plant dominance.[45]Industrial Production
Historical development of refining
The extraction and crystallization of sucrose from sugarcane juice originated in ancient India, where rudimentary refining techniques involved pressing the cane to obtain juice, boiling it to concentrate and form crude crystals, a process documented by around 500 BCE and yielding shard-like sugar known as khanda.[46] These methods spread to China by the 7th century CE through technical exchanges, where further boiling and cooling produced block sugar, enhancing yield through iterative purification.[47] Arab advancements in the 8th century built on Indian and Persian knowledge, introducing scaled crystallization in dedicated mills and early purification via filtration and defecation with lime or ash, which produced whiter, more refined sugar disseminated across the Mediterranean and to Sicily and Spain.[48] By the 12th-13th centuries, these techniques reached Europe via trade and conquest, with initial refineries in Venice employing clay fining—coating sugar cones with clay slurry to draw out impurities—and manual separation, yielding luxury white sugar molded into loaves.[49] In 1747, Prussian chemist Andreas Marggraf demonstrated sucrose extraction from beets using alcohol precipitation, isolating crystals chemically identical to cane sugar and enabling diversification beyond tropical sources.[50] Colonial-era refining intensified in the 17th-18th centuries around Caribbean plantations, where muscovado (unrefined brown sugar) underwent affination—washing with syrup—and lime clarification in European ports to produce granulated white sugar via repeated boiling and cooling.[47] The 1840s marked a pivotal shift with the invention of centrifugal separators, which used rapid spinning to efficiently separate massecuite (crystal-molasses mixture) into raw sugar and syrup, first commercialized in Java by 1853 and rapidly adopted globally to boost throughput.[51] Industrial refinement in the late 19th century incorporated chemical clarification methods like carbonatation, where lime-saturated juice reacted with carbon dioxide to form chalk precipitates trapping impurities, followed by filtration, particularly refining beet-derived liquors into high-purity products.[52]Extraction from sugarcane
Sugarcane stalks reach maturity for harvest after 12 to 18 months of growth in tropical and subtropical regions, at which point the sucrose concentration in the stalks peaks at around 12-15% of the fresh weight.[53] Harvesting involves cutting the stalks close to the ground using manual labor or mechanical harvesters, followed by removal of leaves and tops to prepare the cane for transport to processing mills.[53] Brazil and India lead global sugarcane production, accounting for the majority of the world's supply due to favorable climates and large-scale farming.[54] At the mill, the cane is shredded into chips and crushed through a series of rollers, with water added via imbibition to dilute and extract the juice, achieving extraction rates of 95-98% of available sucrose.[53] The resulting mixed juice, containing 10-15% sucrose along with water, fiber, and impurities, undergoes clarification by heating to about 115°C and adding lime (calcium hydroxide) to precipitate non-sugars and neutralize acids.[55] The clarified juice is then evaporated under vacuum to form a thick syrup with approximately 60% solids, which is seeded and cooled to induce crystallization, yielding raw sugar (sucrose crystals) and molasses as the mother liquor.[53] The fibrous residue, bagasse, comprises about 30% of the cane's weight and is primarily burned for cogeneration of steam and electricity in the mill, with excess used for ethanol production or as biofuel.[56] Molasses, rich in residual sugars, serves as a feedstock for ethanol fermentation, animal feed, or rum production.[56] In tropical conditions, optimized cultivation and extraction yield approximately 10 tonnes of sucrose per hectare, reflecting cane productivity of 70-100 tonnes per hectare and recovery efficiencies of 85-90%.[57]Extraction from sugar beet
Sugar beets (Beta vulgaris subsp. vulgaris) are harvested from temperate regions during autumn and winter campaigns, typically spanning October to March in Europe, when root sucrose content reaches 15-20% of fresh weight.[58] The roots are mechanically lifted, topped, and transported to processing factories, where they undergo washing to remove adhering soil and debris.[59] At the factory, cleaned beets are sliced into thin, V-shaped strips called cossettes to maximize surface area for extraction.[59] These cossettes are then placed in a multicell countercurrent diffuser, where they contact hot water at 70-75°C for approximately one hour, allowing sucrose to diffuse out of the plant cells into the extraction liquor, yielding raw juice with about 10-15% dissolved solids, primarily sucrose.[60] [61] The diffusion process exploits the concentration gradient across semi-permeable cell membranes, with the resulting pulp (cossette residue) pressed and dried for use as animal feed.[62] Raw juice purification begins with clarification and carbonatation, a process tailored to beet juice's relatively low impurity profile compared to sugarcane juice, featuring fewer colorants and organic acids but notable nitrogenous compounds and invert sugars.[63] Lime milk (calcium hydroxide suspension) is added to raise pH and precipitate impurities, followed by carbon dioxide injection in two sequential stages to form insoluble calcium carbonate complexes that trap non-sugars; these are filtered out, yielding clear thin juice.[64] [65] The thin juice is then evaporated under vacuum to produce thick juice (60-70% solids), which undergoes multiple crystallization steps in vacuum pans to form massecuite, from which sucrose crystals are separated via centrifugation, washed, and dried.[62] In major producing areas such as the European Union (including France and Germany) and Russia, which accounted for significant shares of global output in 2023, extractable sugar yields typically range from 7-12 tons per hectare, influenced by root yield (40-80 tons/ha) and sucrose purity.[66] [67] [68] Beet extraction's adaptation to cooler climates enables reliable production without tropical dependencies, with modern diffusers and ion-exchange refinements recovering up to 90% of sucrose while minimizing molasses losses.[69]Modern production trends and technological advances
Global sugar production, predominantly sucrose from sugarcane and sugar beets, is projected to reach 185-189 million metric tons in the 2025/26 marketing year, reflecting a year-over-year increase of approximately 3-5% driven by expanded acreage and favorable weather in key producers Brazil and India.[70][71] Brazil's output is forecasted at 41.42 million metric tons, supported by recovering yields, while India's contributions bolster overall growth amid rising demand for integrated sugar-ethanol processing.[72] Modern production increasingly integrates biorefineries, where sugarcane mills co-produce sucrose with biofuels like ethanol, enhancing efficiency by utilizing bagasse for cogeneration and diverting feedstock based on market signals.[73] This model, dominant in Brazil, allows flexible allocation between sugar and ethanol, though it introduces volatility as mills prioritize higher-value ethanol during periods of elevated fuel prices or mandates.[74] Technological advances include genetic breeding programs that have elevated sucrose content through selective hybridization and marker-assisted selection, with improved varieties achieving up to 20% sucrose levels in elite lines by enhancing biomass quality and stress tolerance.[75][76] Precision agriculture employs satellite imagery, AI-driven analytics, and variable-rate inputs to optimize planting and irrigation, yielding water savings of 20-30% and productivity gains in sugarcane fields.[77][78] Sustainable practices further reduce water intensity via drip irrigation, recycled process water, and "crop per drop" benchmarks that match top-quartile efficiency, minimizing freshwater demands in water-stressed regions.[79] Challenges persist from climate variability, including droughts and erratic rainfall in Brazil's Center-South region, which have disrupted yields and prompted adaptive breeding for resilience.[80] Ethanol diversion exacerbates supply fluctuations, as mills shift cane to fuel production under policy incentives, potentially constraining sucrose availability despite overall output gains.[74]Applications and Uses
Culinary and food industry applications
Sucrose functions as the predominant sweetener in culinary preparations and food processing, imparting balanced sweetness to beverages, baked goods, jams, and confections.[81] Its sensory profile derives from equal parts glucose and fructose, yielding a clean taste without the fruitier notes of alternatives like high-fructose corn syrup.[82] Beyond sweetness, sucrose acts as a preservative by lowering water activity to inhibit microbial growth, a bulking agent to provide structure and volume in products like ice cream, and a texture enhancer that contributes to tenderness in baked items by competing for water and limiting starch gelatinization.[83][84] In processed foods, it facilitates flavor balance, color development through heating, and fermentation control in doughs and batters.[85] Culinary sucrose appears in refined forms tailored to applications: granulated sugar, with mid-sized crystals and 99.85% sucrose purity after molasses removal, suits general baking and cooking for even dissolution; powdered sugar, mechanically ground to fine particles often with 3% cornstarch anti-caking agent, enables smooth icings and dustings; brown sugar, incorporating 3-7% molasses, delivers moisture retention and caramel notes ideal for cookies and sauces.[86][87] During baking, sucrose promotes crust browning via caramelization above 160°C and partial thermal inversion to glucose and fructose, which as reducing sugars react with amino acids in the Maillard reaction to form melanoidins responsible for golden hues and roasted flavors.[88][89] In confectionery, enzymatic or acid-catalyzed inversion produces invert sugar syrup, a 50:50 glucose-fructose mix that prevents sucrose recrystallization, ensuring creamy textures in fondants, creams, and fruit preserves.[90] Food industry quality control employs the Brix scale (°Bx), defined as grams of sucrose per 100 grams of solution, measured refractometrically to assess dissolved solids in syrups, juices, and soft drinks, with values guiding formulation for consistency—e.g., 12-15°Bx in colas or 65-68°Bx in candies.[91][92] Sucrose's stability under processing conditions often favors it over substitutes for predictable viscosity and flavor retention in high-heat applications.[93]


