Recent from talks
Nothing was collected or created yet.
Xylene
View on WikipediaIn organic chemistry, xylene or xylol (from Greek ξύλον (xylon) 'wood';[1][2] IUPAC name: dimethylbenzene) is any of three organic compounds with the formula (CH3)2C6H4. They are derived from the substitution of two hydrogen atoms with methyl groups in a benzene ring; which hydrogens are substituted determines which of three structural isomers results. It is a colorless, flammable, slightly greasy liquid of great industrial value.[3]
The mixture is referred to as both xylene and, more precisely, xylenes. Mixed xylenes refers to a mixture of the xylenes plus ethylbenzene. The four compounds have identical molecular formulas C8H10. Typically the four compounds are produced together by various catalytic reforming and pyrolysis methods.[4]
Occurrence and production
[edit]Xylenes are an important petrochemical produced by catalytic reforming and also by coal carbonisation in the manufacture of coke fuel. They also occur in crude oil in concentrations of about 0.5–1%, depending on the source. Small quantities occur in gasoline and aircraft fuels.
Xylenes are produced mainly as part of the BTX aromatics (benzene, toluene, and xylenes) extracted from the product of catalytic reforming known as reformate.
Several million tons are produced annually.[3] In 2011, a global consortium began construction of one of the world's largest xylene plants in Singapore.[5]
History
[edit]Xylene was first isolated and named in 1850 by the French chemist Auguste Cahours (1813–1891), having been discovered as a constituent of wood tar.[6]
Industrial production
[edit]Xylenes are produced by the methylation of toluene and benzene.[3][7] Commercial or laboratory-grade xylene produced usually contains about 40–65% of m-xylene and up to 20% each of o-xylene, p-xylene and ethylbenzene.[8][9][10] The ratio of isomers can be shifted to favor the highly valued p-xylene via the patented UOP-Isomar process[11] or by transalkylation of xylene with itself or trimethylbenzene. These conversions are catalyzed by zeolites.[3]
ZSM-5 is used to facilitate some isomerization reactions leading to mass production of modern plastics.
Properties
[edit]The physical properties of the isomers of xylene differ slightly. The melting point ranges from −47.87 °C (−54.17 °F) (m-xylene) to 13.26 °C (55.87 °F) (p-xylene)—as usual, the para isomer's melting point is much higher because it packs more readily in the crystal structure. The boiling point for each isomer is around 140 °C (284 °F). The density of each isomer is around 0.87 g/mL (7.3 lb/US gal; 8.7 lb/imp gal) and thus is less dense than water. The odor of xylene is detectable at concentrations as low as 0.08 to 3.7 ppm (parts of xylene per million parts of air) and can be tasted in water at 0.53 to 1.8 ppm.[9]
| Xylene isomers | ||||
|---|---|---|---|---|
| General | ||||
| Common name | Xylenes (mixture) |
o-Xylene | m-Xylene | p-Xylene |
| Systematic name | Dimethylbenzene | 1,2-Dimethylbenzene | 1,3-Dimethylbenzene | 1,4-Dimethylbenzene |
| Other names | Xylol | o-Xylol; Orthoxylene |
m-Xylol; Metaxylene |
p-Xylol; Paraxylene |
| Molecular formula | C8H10 | |||
| SMILES | Cc1c(C)cccc1 | Cc1cc(C)ccc1 | Cc1ccc(C)cc1 | |
| Molar mass | 106.16 g/mol | |||
| Appearance | Clear, colorless liquid | |||
| CAS number | [1330-20-7] | [95-47-6] | [108-38-3] | [106-42-3] |
| Properties | ||||
| Density and phase | 0.864 g/mL, liquid | 0.88 g/mL, liquid | 0.86 g/mL, liquid | 0.86 g/mL, liquid |
| Solubility in water | Practically insoluble | |||
| Soluble in non-polar solvents such as aromatic hydrocarbons | ||||
| Melting point | −47.4 °C (−53.3 °F; 226 K) | −25 °C (−13 °F; 248 K) | −48 °C (−54 °F; 225 K) | 13 °C (55 °F; 286 K) |
| Boiling point | 138.5 °C (281.3 °F; 412 K) | 144 °C (291 °F; 417 K) | 139 °C (282 °F; 412 K) | 138 °C (280 °F; 411 K) |
| Viscosity | 0.812 cP at 20 °C (68 °F) | 0.62 cP at 20 °C (68 °F) | 0.34 cP at 30 °C (86 °F) | |
| Hazards | ||||
| SDS | Xylenes | o-Xylene Archived 2020-11-06 at the Wayback Machine | m-Xylene Archived 2020-11-06 at the Wayback Machine | p-Xylene Archived 2020-11-06 at the Wayback Machine |
| EU pictograms |   
| |||
| NFPA 704 | ||||
| Flash point | 30 °C (86 °F) | 17 °C (63 °F) | 25 °C (77 °F) | 25 °C (77 °F) |
| H & P phrases | H225, H226, H304, H312, H315, H319, H332, H335, H412 P210, P233, P240, P241, P242, P243, P261, P264, P271, P273, P280, P301+P310, P302+P352, P303+P361+P353, P304+P312, P304+P340, P305+P351+P338, P312, P321, P322, P331, P332+P313, P337+P313, P362, P363, P370+P378, P403+P233, P403+P235, P405, P501 | |||
| RTECS number | ZE2450000 | ZE2275000 | ZE2625000 | |
| Related compounds | ||||
| Related aromatic hydrocarbons |
Toluene, mesitylene, benzene, ethylbenzene | |||
| Related compounds | Xylenols – types of phenols | |||
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Infobox disclaimer and references | ||||
Xylenes form azeotropes with water and a variety of alcohols. The azeotrope with water consists of 60% xylenes and boils at 94.5 °C.[3] As with many alkylbenzene compounds, xylenes form complexes with various halocarbons.[12] The complexes of different isomers often have dramatically different properties from each other.[13]
Applications
[edit]Terephthalic acid and related derivatives
[edit]p-Xylene is the principal precursor to terephthalic acid and dimethyl terephthalate, both monomers used in the production of polyethylene terephthalate (PET) plastic bottles and polyester clothing. 98% of p-xylene production, and half of all xylenes produced is consumed in this manner.[10][14] o-Xylene is an important precursor to phthalic anhydride. The demand for isophthalic acid is relatively modest, so m-xylene is rarely sought (and hence the utility of its conversion to the o- and p-isomers).
Solvent applications and industrial purposes
[edit]Xylenes are used as a solvent in printing, rubber, and leather industries. It is a common component of ink, rubber, and adhesives.[15] In thinning paints and varnishes, it can be substituted for toluene where slower drying is desired, and thus is used by conservators of art objects in solubility testing.[16] Similarly it is a cleaning agent, e.g., for steel, silicon wafers, and integrated circuits. In dentistry, xylene can be used to dissolve gutta percha, a material used for endodontics (root-canal treatments). In the petroleum industry, xylene is also a frequent component of paraffin solvents, used when the tubing becomes clogged with paraffin wax.
Laboratory use
[edit]Xylene is used in the laboratory to make baths with dry ice to cool reaction vessels,[17] and as a solvent to remove synthetic immersion oil from the microscope objective in light microscopy.[18] In histology, xylene is the most widely used clearing agent.[19] Xylene is used to remove paraffin from dried microscope slides prior to staining. After staining, microscope slides are put in xylene prior to mounting with a coverslip.
Precursor to other compounds
[edit]In one large-scale application, para-xylene is converted to terephthalic acid. The major application of ortho-xylene is as a precursor to phthalate esters, used as plasticizer. Meta-xylene is converted to isophthalic acid derivatives, which are components of alkyd resins.[3]
Chemical properties
[edit]Generally, two kinds of reactions occur with xylenes: those involving the methyl groups and those involving the ring C–H bonds. Being benzylic and hence weakened, the C–H bonds of the methyl groups are susceptible to free-radical reactions, including halogenation to the corresponding xylene dichlorides (bis(chloromethyl)benzenes), while mono-bromination yields xylyl bromide, a tear gas agent. Oxidation and ammoxidation also target the methyl groups, affording dicarboxylic acids and the dinitriles. Electrophiles attack the aromatic ring, leading to chloro- and nitroxylenes.[3]
Health and safety
[edit]Xylene is flammable but of modest acute toxicity, with LD50 ranges from 200 to 5000 mg/kg for animals. Oral LD50 for rats is 4300 mg/kg. The principal mechanism of detoxification is oxidation to methylbenzoic acid and hydroxylation to hydroxylene.[3]
The main effect of inhaling xylene vapor is depression of the central nervous system (CNS), with symptoms such as headache, dizziness, nausea and vomiting. At an exposure of 100 ppm, one may experience nausea or a headache. At an exposure between 200 and 500 ppm, symptoms can include feeling "high", dizziness, weakness, irritability, vomiting, and slowed reaction time.[20][21]
The side effects of exposure to low concentrations of xylene (< 200 ppm) are reversible and do not cause permanent damage. Long-term exposure may lead to headaches, irritability, depression, insomnia, agitation, extreme tiredness, tremors, hearing loss, impaired concentration and short-term memory loss.[22][clarification needed] A condition called chronic solvent-induced encephalopathy, commonly known as "organic-solvent syndrome" has been associated with xylene exposure. There is very little information available that isolates xylene from other solvent exposures in the examination of these effects.[20]
Hearing disorders have been also linked to xylene exposure, both from studies with experimental animals,[23][24] as well as clinical studies.[25][26][27]
Xylene is also a skin irritant and strips the skin of its oils, making it more permeable to other chemicals. The use of impervious gloves and masks, along with respirators where appropriate, is recommended to avoid occupational health issues from xylene exposure.[20]
Xylenes are metabolized to methylhippuric acids.[28][29] The presence of methylhippuric acid can be used as a biomarker to determine exposure to xylene.[29][30]
See also
[edit]- Alkylbenzene – Family of organic compounds
- C2-Benzenes
- Hydrodealkylation – Chemical reaction
- Transalkylation – Chemical reaction which transfers an alkyl group between molecules
- Xylene cyanol – Dye used as an electrophoretic color marker
References
[edit]- ^ ξύλον. Liddell, Henry George; Scott, Robert; A Greek–English Lexicon at the Perseus Project.
- ^ Harper, Douglas. "xylene". Online Etymology Dictionary.
- ^ a b c d e f g h Fabri, Jörg; Graeser, Ulrich; Simo, Thomas A. (2000). "Xylenes". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a28_433. ISBN 978-3527306732.
- ^ Cannella, William J. (2000). "Xylenes and Ethylbenzene". Kirk-Othmer Encyclopedia of Chemical Technology. doi:10.1002/0471238961.2425120503011414.a01. ISBN 0471238961.
- ^ Tremblay, Jean-François (2011). "Making Aromatics in Singapore". Chemical & Engineering News Archive. 89 (38): 18–19. doi:10.1021/cen-v089n038.p018.
- ^ Cahours, Auguste (1850) "Recherches sur les huiles légéres obtenues dans la distillation du bois" (Investigations of light oils obtained by the distillation of wood), Compte rendus, 30 : 319-323; see especially p. 321. From p. 321: "Je le désignerai sous le nomme xylène." (I will designate it by the name of xylene.) Note: Cahours' empirical formula for xylene is incorrect because chemists at that time used the wrong atomic mass for carbon (6 instead of 12).
- ^ Martindale, David C. and Kuchar, Paul J., Production of xylenes from light aliphatic hydrocarbons via dehydrocyclodimerization and methylation, United States Patent No. 5,043,502, 1991-8-27. Accessed 2012-4-28.
- ^ "Xylene (Mixed Isomers), Air Toxic Hazard Summary". United States Environmental Protection Agency. Archived from the original on February 8, 2015. Retrieved 8 February 2015.
- ^ a b Kandyala, Reena; Raghavendra, Sumanth Phani C.; Rajasekharan, Saraswathi T. (2010). "Xylene: An overview of its health hazards and preventive measures". J Oral Maxillofac Pathol. 14 (1): 1–5. doi:10.4103/0973-029X.64299. PMC 2996004. PMID 21180450.
- ^ a b Xylene (Archived August 11, 2011, at the Wayback Machine), Swedish Chemicals Agency, apps.kemi.se, 2010. Accessed 2012-4-28.
- ^ "Capturing Opportunities for Para-xylene Production". UOP, A Honeywell Company. Retrieved 8 February 2015.
- ^ Clark J. E.; Luthy, R. V. (1955). "Separation of Xylenes". Ind. Eng. Chem. 47 (2): 250–253. doi:10.1021/ie50542a028.
- ^ Stevenson, Cheryl D.; McElheny, Daniel J.; Kage, David E.; Ciszewski, James T.; Reiter, Richard C. (1998). "Separation of Closely Boiling Isomers and Identically Boiling Isotopomers via Electron-Transfer-Assisted Extraction". Analytical Chemistry. 70 (18): 3880. doi:10.1021/ac980221b.
- ^ ICIS, Paraxylene-Orthoxylene | Prices, News & Market Analysis, icis.com, 2012. Accessed 2012-04-28.
- ^ Bostik, Safety Data Sheet Blu-Tack (Archived September 11, 2011, at the Wayback Machine), No. 13135, Bostik Corp., June 2007. Accessed 2012-04-28.
- ^ Samet, Wendy, (comp.), Appendix I, Painting Conservation Catalog, American Institute for Conservation of Historic and Artistic Works, conservation-wiki.com, September 1997. Accessed 2012-04-28.
- ^ "Cooling baths". UC Davis Chem Wiki. 2013-10-02. Archived from the original on 2015-02-08. Retrieved 8 February 2015.
- ^ Cargille, John (1985) [1964], "Immersion Oil and the Microscope", New York Microscopical Society Yearbook, archived from the original on 2011-09-11, retrieved 2011-03-10
- ^ Carson, Freida; Hladik, Christa (2009). Histotechnology: A Self-Instructional Text (3 ed.). American Society for Clinical Pathology Press. p. 35. ISBN 9780891895817.
- ^ a b c Kandyala, Reena; Raghavendra, Sumanth Phani C.; Rajasekharan, Saraswathi T. (2010-01-01). "Xylene: An overview of its health hazards and preventive measures". Journal of Oral and Maxillofacial Pathology. 14 (1): 1–5. doi:10.4103/0973-029X.64299. ISSN 0973-029X. PMC 2996004. PMID 21180450.
- ^ "ACUTE TOXICITY SUMMARY: XYLENES" (PDF). Archived from the original (PDF) on October 22, 2015.
- ^ "Xylenes (EHC 190, 1997)".
- ^ Gagnaire, F.; Marignac, B.; Langlais, C.; Bonnet, P. (July 2001). "Ototoxicity in rats exposed to ortho-, meta- and para-xylene vapours for 13 weeks". Pharmacology & Toxicology. 89 (1): 6–14. doi:10.1034/j.1600-0773.2001.d01-129.x. ISSN 0901-9928. PMID 11484912.
- ^ Gagnaire, F.; Marignac, B.; Blachère, V.; Grossmann, S.; Langlais, C. (2007-03-07). "The role of toxicokinetics in xylene-induced ototoxicity in the rat and guinea pig". Toxicology. 231 (2–3): 147–158. Bibcode:2007Toxgy.231..147G. doi:10.1016/j.tox.2006.11.075. ISSN 0300-483X. PMID 17210216.
- ^ Fuente, Adrian; McPherson, Bradley; Cardemil, Felipe (September 2013). "Xylene-induced auditory dysfunction in humans". Ear and Hearing. 34 (5): 651–660. doi:10.1097/AUD.0b013e31828d27d7. hdl:10533/134303. ISSN 1538-4667. PMID 23598724. S2CID 45206975.
- ^ Draper, T. H. J.; Bamiou, D.-E. (April 2009). "Auditory neuropathy in a patient exposed to xylene: case report" (PDF). The Journal of Laryngology & Otology. 123 (4): 462–465. doi:10.1017/S0022215108002399. ISSN 1748-5460. PMID 18439334. Archived (PDF) from the original on 2017-08-09.
- ^ Fuente, Adrian; McPherson, Bradley; Hood, Linda J. (November 2012). "Hearing loss associated with xylene exposure in a laboratory worker". Journal of the American Academy of Audiology. 23 (10): 824–830. doi:10.3766/jaaa.23.10.7. hdl:10533/137495. ISSN 1050-0545. PMID 23169198.
- ^ "HIPPURIC and METHYL HIPPURIC ACIDS in urine" (PDF). NIOSH Manual of Analytical Methods (NMAM) (Fourth ed.).
- ^ a b Inoue, O.; Seiji, K.; Kawai, T.; Watanabe, T.; Jin, C.; Cai, S. X.; Chen, Z.; Qu, Q. S.; Zhang, T.; Ikeda, M. (1993). "Excretion of methylhippuric acids in urine of workers exposed to a xylene mixture: Comparison among three xylene isomers and toluene". International Archives of Occupational and Environmental Health. 64 (7): 533–539. Bibcode:1993IAOEH..64..533I. doi:10.1007/bf00381104. PMID 8482596. S2CID 21534640.
- ^ Kira S. (1977). "Measurement by gas chromatography of urinary hippuric acid and methylhippuric acid as indices of toluene and xylene exposure". Occupational and Environmental Medicine. 34 (305–309): 305–309. doi:10.1136/oem.34.4.305. PMC 1008281. PMID 588486.
External links
[edit]- Chisholm, Hugh, ed. (1911). . Encyclopædia Britannica (11th ed.). Cambridge University Press.
- NIOSH Pocket Guide to Chemical Hazards (o-Xylene)
- NIOSH Pocket Guide to Chemical Hazards (m-Xylene)
- NIOSH Pocket Guide to Chemical Hazards (p-Xylene)
- Xylene, Hazard Summary (EPA) (Mixed Isomers)
- The Ear Poisons Archived 2022-11-26 at the Wayback Machine, The Synergist, American Industrial Hygiene Association, November, 2018
Xylene
View on GrokipediaChemical Structure and Properties
Isomers and Nomenclature
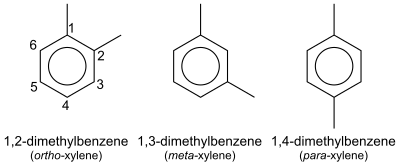
Xylene has the molecular formula C₈H₁₀ and consists of a benzene ring with two methyl groups attached, represented as C₆H₄(CH₃)₂.[10] This structure gives rise to three isomeric forms based on the positions of the methyl substituents on the benzene ring. The isomers differ in the relative placement of the methyl groups, which affects their spatial arrangement and symmetry. The ortho-xylene isomer, also known as 1,2-dimethylbenzene, features the two methyl groups on adjacent carbon atoms (positions 1 and 2) of the benzene ring, resulting in a structure where the substituents are next to each other.[11] Meta-xylene, or 1,3-dimethylbenzene, has the methyl groups separated by one carbon atom (positions 1 and 3), creating a less symmetric arrangement.[11] Para-xylene, designated as 1,4-dimethylbenzene, positions the methyl groups opposite each other (positions 1 and 4) on the ring, leading to a highly symmetric molecule.[11] These positional differences are illustrated in standard chemical notation, where the benzene ring is depicted as a hexagon with methyl groups (-CH₃) attached at the specified carbons. In IUPAC nomenclature, the isomers are systematically named as 1,2-dimethylbenzene (ortho), 1,3-dimethylbenzene (meta), and 1,4-dimethylbenzene (para), reflecting the locant positions of the methyl groups.[11] Common abbreviations include o-xylene, m-xylene, and p-xylene, derived from the Greek prefixes for position: "ortho" (beside), "meta" (beyond), and "para" (opposite).[11] The term "xylene" originates from the Greek word "xylon," meaning wood, as the compound was first isolated from wood tar in 1850 by French chemist Auguste Cahours. Commercially, xylenes are often produced as mixed xylenes, a blend typically containing 40-65% m-xylene, up to 20% o-xylene, 10-20% p-xylene, and ethylbenzene.[4] Separating these isomers from the mixture poses challenges due to their similar boiling points and chemical properties, requiring advanced distillation or adsorption techniques.[10]Physical Properties
Xylenes are colorless, flammable liquids characterized by a sweet, aromatic odor detectable at concentrations as low as 1 ppm.[12] These properties make them volatile and suitable for solvent applications, with flash points ranging from 25°C for m- and p-xylenes to 32°C for o-xylene in pure forms, and approximately 25–27°C for mixed xylene.[13] Vapor pressures at 25°C are around 8–9 mm Hg for the individual isomers and 6.6–8.3 mm Hg for mixed xylene, contributing to their flammability and ease of evaporation.[14] The physical properties of the xylene isomers vary slightly due to differences in their molecular structures, as outlined in the nomenclature section. Key data for the ortho-, meta-, and para-isomers, along with typical mixed xylene, are summarized below:| Property | o-Xylene | m-Xylene | p-Xylene | Mixed Xylene |
|---|---|---|---|---|
| Boiling point (°C) | 144.4 | 139.1 | 138.4 | 137–140 |
| Melting point (°C) | −25.2 | −47.4 | 13.3 | −47 to 6 |
| Density (g/cm³ at 20°C) | 0.880 | 0.864 | 0.861 | 0.87 |
| Refractive index (n²⁰_D) | 1.505 | 1.497 | 1.495 | 1.497 |