Recent from talks
Nothing was collected or created yet.
Arthropod bites and stings
View on Wikipedia| Arthropod bites and stings | |
|---|---|
| Other names | Bug bite |
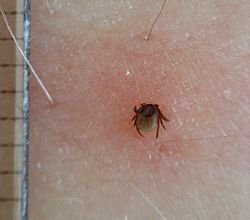 | |
| Tick bite | |
| Symptoms | Swelling, itching, pain |
| Complications | Anaphylaxis, envenomation, disease transmission |
Many species of arthropods (insects, arachnids, millipedes and centipedes) can bite or sting human beings. These bites and stings generally occur as a defense mechanism or during normal arthropod feeding. While most cases cause self-limited irritation, medically relevant complications include envenomation, allergic reactions, and transmission of vector-borne diseases.[1]
Signs and symptoms
[edit]
Most arthropod bites and stings cause self-limited redness, itchiness and/or pain around the site. Less commonly (around 10% of Hymenoptera sting reactions), a large local reaction occurs when the area of swelling is greater than 10 centimetres (4 in). Rarely (1-3% of Hymenoptera sting reactions), systemic reactions can affect multiple organs and pose a medical emergency, as in the case of anaphylactic shock.[2][3]
Defensive and predatory bites and stings
[edit]Many arthropods bite or sting in order to immobilize their prey or deter potential predators as a defense mechanism. Stings containing venom are more likely to be painful. Less frequently, venomous spider bites are also associated with morbidity and mortality in humans.
Most arthropod stings involve Hymenoptera (ants, wasps, and bees). While the majority of Hymenoptera stings are locally painful, their associated venom rarely cause toxic reactions unless victims receive many stings at once. The low mortality (around 60 deaths per year in the US out of unreported millions of stings nationwide) associated with Hymenoptera is mostly due to anaphylaxis from venom hypersensitivity.[4]
Most scorpion stings also cause self-limited pain or paresthesias. Only certain species (from family Buthidae) inject neurotoxic venom, responsible for most morbidity and mortality. Severe toxic reactions can occur resulting in progressive hemodynamic instability, neuromuscular dysfunction, cardiogenic shock, pulmonary edema, multi-organ failure, and death. Although robust epidemiological data is unavailable, global estimates of scorpion stings exceed 1.2 million resulting in more than 3000 deaths annually.[5]
Spider bites most often cause minor symptoms and resolve without intervention. Medically significant spider bites involve substantial envenomation from only certain species such as widow spiders and recluse spiders. Symptoms of latrodectism (from widow spiders) may include pain at the bite or involve the chest and abdomen, sweating, muscle cramps and vomiting among others. By comparison, loxoscelism (from recluse spiders) can present with local necrosis of the surrounding skin and widespread breakdown of red blood cells. Headaches, vomiting and a mild fever may also occur.[6]
Feeding bites
[edit]Feeding bites have characteristic patterns and symptoms that reflect feeding habits of the offending pest and the chemistry of its saliva. Feeding bites are less likely to be felt at the time of the bite, although there are some exceptions. Since feeding requires longer attachment to prey than envenomation, feeding bites are more often associated with vector transmission of disease.[7]
| Pest | Preferred body part | Felt at time of bite | Reaction |
|---|---|---|---|
| Mosquitoes | exposed appendages | usually not | Low raised welt, itches for several hours. |
| Midges and no-see-ums | exposed appendages | usually | Itches for several hours. |
| Fleas | prefer ankles and bare feet | usually | May make red itchy welt; several days. Later bites are less severe. |
| Biting flies | any exposed skin | painful and immediate | Painful welt, several hours. |
| Bed bugs | appendages, neck, exposed skin | usually not | Low red itchy welts, usually several together resembling rash, slow to develop and can last weeks. |
| Hair Lice | pubic area or scalp | usually not | Infested area intensely itchy, with red welts at bite sites. See pediculosis. |
| Larval ticks | Anywhere on body, but prefer covered skin, crevices. | Usually not; may be scratched off before they are seen. | Intensely itchy red welts lasting over a week. |
| Adult ticks | covered skin, crevices, entire body | usually not | Itchy welt, several days. |
| Mites | mainly on the trunk and extremities | usually not | Intensely itchy welts and papules that may last for days. See acariasis. |
As vectors of disease
[edit]In addition to stings and bites causing discomfort in of themselves, bites can also spread secondary infections if the arthropod is carrying a virus, bacteria, or parasite.[8] The World Health Organization (WHO) estimates that 17% of all infectious diseases worldwide were transmitted by arthropod vectors, resulting in over 700,000 deaths annually.[9] The table below lists common arthropod vectors and their associated diseases. The figure below represents endemic areas of common vector-borne diseases.
| Vector | Pathogen class | Disease | Annual disease burden* |
|---|---|---|---|
| Mosquitoes
(Culicidae) |
Arboviruses (Togavirus, Flavivirus, Bunyavirus)
Nematode (Wuchereria bancrofti) |
Chikugunya, Zika, Yellow fever, Dengue, West Nile, California encephalitis, Japanese encephalitis, Equine encephalitis, Rift Valley fever | >300 million |
| Black flies
(Simuliidae) |
Nematode (Onchocerca volvulus) | River blindness | >10 million |
| Assassin bug
(Reduviidae) |
Protozoa (Trypanosoma cruzi) | Chagas disease | >6 million |
| Sand fly
(Phlebotominae) |
Protozoa (Leishmania) | Cutaneous and visceral leishmaniasis | >3 million |
| Ticks
(Ixodidae) |
Arboviruses (Bunyavirus, Flavirus)
Protozoa (Babesia) |
Heartland virus, Tick-borne encephalitis, Crimean-Congo hemorrhagic fever
|
>500,000 |
| Tsetse flies
(Glossinidae) |
Protozoa (Trypanosoma gambiense, T. rhodesesiense) | African sleeping sickness | >10,000 |
| Biting flies
(Tabanidae) |
Nematode (Loa Loa) | African eyeworm | NA |
| Fleas
(Siphonaptera, Pulicidae) |
Bacteria (Yersinia pestis, Bartonella henselae) | Plague, Cat scratch fever | NA |
| Lice
(Phthiraptera, Pediculidae) |
Bacteria (Borrelia recurrentis, Rickettsia prowazekii, Bartonella quintana) | Lice-borne relapsing fever, endemic typhus, Trench fever | NA |
*Estimated global number of cases annually according to WHO in 2017.[9] If a vector transmits multiple diseases, aggregate case numbers are listed. Rough estimates are only meant to provide a sense of scale. Unknown disease burden is listed as NA for not available.

Diagnosis
[edit]
Most arthropod bites and stings do not require a specific diagnosis since they typically improve with supportive management alone. Certain bites and stings present with characteristic appearances and distributions. In general, however, dermoscopic findings of bitten or stung skin rarely aid in diagnosis.[11] Rather, patient history (recent travel to endemic areas, outdoor activities, and other risk factors) primarily guides the diagnostic approach, which can raise clinical suspicion for more serious complications like vector-borne diseases.
Microscopic appearance
[edit]Skin biopsies are not indicated for bites or stings, since the histomorphologic appearance is non-specific. Bites and stings as well as other conditions (e.g. drug reactions, urticarial reactions, and early bullous pemphigoid) can cause microscopic changes such as a wedge-shaped superficial dermal perivascular infiltrate consisting of abundant lymphocytes and scattered eosinophils, as shown in the adjacent figure:[12]
Prevention
[edit]Prevention strategies against arthropod bites and stings comprise measures for personal protection, travel advisories, public health and environmental concerns.
Personal protection
[edit]Travelers should seek to minimize outdoor activity during peak activity times and avoid high risk areas such as regions with known outbreaks or epidemics. Standing water and dense vegetation also commonly attract arthropods. Clothes covering most exposed skin can also provide a measure of physical protection, which may be augmented when the fabric is treated with pesticides such as Permethrin. Topical repellants such as N,N-diethyl-m-toluamide (DEET) is supported by a large body of evidence.[7]
Vaccines may also help prevent vector-borne diseases for eligible patients. For example, Japanese encephalitis, Yellow fever, and Dengue fever have FDA-approved vaccines available. Since they are relatively new vaccines, however, they are not standard of care as of 2023. Additionally, patients traveling to Malaria endemic regions are routinely prescribed Malaria chemoprophylaxis.[13]
Patients with a history of venom hypersensitivity may benefit from venom immunotherapy (VIT). Patients eligibile for VIT include those with a prior anaphylactic reaction to a venomous sting and who have IgE to venom allergens. VIT can help prevent future severe systemic reactions in select patients.[2]
Global health
[edit]International organizations such as WHO aim to reduce disease burdens of neglected tropical diseases, many of which are vector borne.[14] Such campaigns must incorporate multipronged approaches to consider global inequality, access to resources, and climate change.[citation needed]
Management
[edit]Most arthropod bites and stings require only supportive care. However, complications such as envenomation and severe allergic reactions can present as medical emergencies.
Supportive care
[edit]Local reactions to bites and stings are treated symptomatically. If a stinger is still embedded, manual removal can reduce further irritation. Washing the affected area with soap and water can help reduce risk of contamination. Oral antihistamines, calamine lotion, topical corticosteroids and cold compresses are common over the counter remedies to reduce itchiness and local inflammation. In more severe cases, such as large local reactions, systemic glucocorticoids are sometimes prescribed, although limited evidence supports their effectiveness. There are limited data to support one treatment over another.[15]
Medical emergencies
[edit]Systemic reactions from venom hypersensitivity can rapidly progress to a medical emergency. The mainstay of anaphylactic shock management is intramuscularly injected epinephrine. The patient should be stabilized and transferred to an intensive care unit.[2]
Toxic reactions to envenomation are similarly managed with medical stabilization and symptomatic treatment. Tetanus prophylaxis should be up to date but antibiotics are typically unnecessary unless a bacterial superinfection is suspected. Antivenom drugs have been created for certain species such as Centruroides scorpion stings, but these drugs are not yet widely available and so typically reserved for severe systemic toxicity.[15]
Several vector-borne diseases can present emergently.
Treatment of vector-borne diseases
[edit]After confirmation of diagnosis, antimicrobials are prescribed according to standard of care.
Biting and stinging arthropods
[edit]
A bite is defined as coming from the mouthparts of the arthropod. The bite consists of both the bite wound and the saliva. The saliva of the arthropod may contain anticoagulants, as in insects and arachnids which feed from blood. Feeding bites may also contain anaesthetic, to prevent the bite from being felt. Feeding bites may also contain digestive enzymes, as in spiders; spider bites have primarily evolved to paralyse and then digest prey. A sting comes from the abdomen; in most insects (which are all largely hymenopterans), the stinger is a modified ovipositor,[16] which protrudes from the abdomen.
The sting consists of an insertion wound, and venom. The venom is evolved to cause pain to a predator, paralyse a prey item, or both. Because insect stingers evolved from ovipositors, in most hymenopterans only the female can sting. However, there are a few orders of wasp where the male has evolved a "pseudo sting" - the male genitalia has evolved two sharp protrusions which can deliver an insertion wound. However, they do not contain venom, so they are not considered a true sting.[17] In ants that bite instead of sting, such as the Formicinae, the bite causes the wound, but during the bite the abdomen bends forward to spray formic acid into the wound, causing additional pain. In arachnids that sting (all largely scorpions), the stinger is not a modified ovipositor, but instead a metasoma that bears a telson.[18] (Scorpions lack an ovipositor entirely and give birth to live young.)
Insects
[edit]
Diptera (True flies)
[edit]- Black flies (Simuliidae)
- Horse-flies (Tabanidae)
- Deer flies/Yellow flies (Chrysops)
- Tsetse flies (Glossinidae)
- Stable flies (Muscidae)
- Biting midges or No see-ums (Ceratopogonidae)
- Mosquitos (Culicidae)
- Botflies (as larvae, Oestridae)
- Sandflies (Phlebotomidae)
- Blow-flies (as larvae, Calliphoridae)
- Screw-worm flies (as larvae, Calliphoridae)
- Hippoboscidae (Keds)

Hymenoptera (ants, bees and wasps)
[edit]- Ants
- Bull ants (sting)
- Fire ants (both bite and sting)
- Bullet ants (sting)
- Bees
- Honeybees (sting)
- Stingless bees (bite)
- Bumblebees (sting)
- Wasps (sting)
- Hornets (sting)
- Yellow Jackets (sting)
- Paper wasps (sting)

Siphonaptera (Fleas)
[edit]- Fleas (bite)
- Human flea Pulex irritans
- Chigoe flea Tunga penetrans

Phthiraptera (Lice)
[edit]Other insects
[edit]- Assassin bug/Kissing bug
- Bedbugs
- Conenose bug
Arachnids
[edit]
Spiders
[edit]Mites
[edit]- Red Poultry Mite
- Spiny rat mite
- House mouse mite
- Northern fowl mite
- Tropical fowl mite
- Mange mite
- Scabies
- Ticks
Scorpions
[edit]- All species sting
Myriapoda
[edit]References
[edit]- ^ Powers J, McDowell RH (2022). "Insect Bites". StatPearls. Treasure Island (FL): StatPearls Publishing. PMID 30725920. Retrieved 2023-02-09.
- ^ a b c Golden DB, Demain J, Freeman T, Graft D, Tankersley M, Tracy J, et al. (January 2017). "Stinging insect hypersensitivity: A practice parameter update 2016". Annals of Allergy, Asthma & Immunology. 118 (1): 28–54. doi:10.1016/j.anai.2016.10.031. PMID 28007086.
- ^ Goddard J (2002). Physician's guide to arthropods of medical importance. Boca Raton: CRC Press. pp. 14. ISBN 978-0-8493-1387-5.
- ^ Reed H, Landolt PJ (January 2019). "Chapter 22 - Ants, Wasps, and Bees (Hymenoptera)". In Mullen GR, Durden LA (eds.). Medical and Veterinary Entomology (Third ed.). Academic Press. pp. 459–488. doi:10.1016/b978-0-12-814043-7.00022-4. ISBN 978-0-12-814043-7. S2CID 239396551.
- ^ Chippaux JP, Goyffon M (August 2008). "Epidemiology of scorpionism: a global appraisal". Acta Tropica. 107 (2): 71–79. doi:10.1016/j.actatropica.2008.05.021. PMID 18579104.
- ^ Braitberg G, Segal L (November 2009). "Spider bites - Assessment and management". Australian Family Physician. 38 (11): 862–867. PMID 19893831.
- ^ a b Herness J, Snyder MJ, Newman RS. Arthropod bites and stings. American family physician. 2022 Aug;106(2):137-47.
- ^ Schorderet-Weber S, Noack S, Selzer PM, Kaminsky R (April 2017). "Blocking transmission of vector-borne diseases". International Journal for Parasitology. Drugs and Drug Resistance. 7 (1): 90–109. doi:10.1016/j.ijpddr.2017.01.004. PMC 5302141. PMID 28189117.
- ^ a b Global vector control response 2017–2030. Geneva: World Health Organization; 2017. License: CC BY-NC-SA 3.0 IGO.
- ^ Marselle, Melissa R.; Stadler, Jutta; Korn, Horst; Irvine, Katherine N.; Bonn, Aletta, eds. (2019). Biodiversity and Health in the Face of Climate Change. doi:10.1007/978-3-030-02318-8. ISBN 978-3-030-02317-1. S2CID 199491437.
- ^ Chauhan P, Jindal R, Errichetti E (October 2022). "Dermoscopy of skin parasitoses, bites and stings: a systematic review of the literature". Journal of the European Academy of Dermatology and Venereology. 36 (10): 1722–1734. doi:10.1111/jdv.18352. PMID 35735046. S2CID 249955855.
- ^ Alsaad KO, Ghazarian D (December 2005). "My approach to superficial inflammatory dermatoses". Journal of Clinical Pathology. 58 (12): 1233–1241. doi:10.1136/jcp.2005.027151. PMC 1770784. PMID 16311340.
- ^ Murray HW (August 2020). "The Pretravel Consultation: Recent Updates". The American Journal of Medicine. 133 (8): 916–923.e2. doi:10.1016/j.amjmed.2020.02.005. PMID 32179056. S2CID 212741346.
- ^ Global report on neglected tropical diseases 2023. Geneva: World Health Organization; 2023. Licence: CC BY-NC-SA 3.0 IGO.
- ^ a b Erickson TB, Cheema N (May 2017). "Arthropod Envenomation in North America". Emergency Medicine Clinics of North America. Wilderness and Environmental Medicine. 35 (2): 355–375. doi:10.1016/j.emc.2017.01.001. PMID 28411932.
- ^ Gwaltney-Brant SM, Dunayer E, Youssef H (2018). "Terrestrial Zootoxins". In Gupta RC (ed.). Veterinary Toxicology. Elsevier. pp. 781–801. doi:10.1016/b978-0-12-811410-0.00058-1. ISBN 978-0-12-811410-0.
- ^ Sugiura S, Tsujii M (December 2022). "Male wasp genitalia as an anti-predator defense". Current Biology. 32 (24): R1336 – R1337. Bibcode:2022CBio...32R1336S. doi:10.1016/j.cub.2022.11.030. PMID 36538881. S2CID 254874084.
- ^ van der Meijden A, Kleinteich T (April 2017). "A biomechanical view on stinger diversity in scorpions". Journal of Anatomy. 230 (4): 497–509. doi:10.1111/joa.12582. PMC 5345679. PMID 28028798.
External links
[edit]Arthropod bites and stings
View on GrokipediaBiology and Mechanisms
Defensive and Predatory Bites and Stings
Arthropod bites and stings function primarily in predation to immobilize prey and in defense to deter predators, with venoms comprising complex mixtures of peptides, enzymes, and neurotoxins adapted to these roles.[5] Defensive actions typically target larger threats, inducing pain via nociceptor stimulation to discourage attacks, while predatory mechanisms focus on smaller arthropod prey, causing rapid paralysis through ion channel disruption.[6] Many venoms exhibit dual functionality, but evolutionary pressures often lead to specialization, such as enhanced insecticidal effects in predatory species versus heightened pain induction in defensive ones.[7] In aculeate Hymenoptera, including bees, wasps, and ants, stings serve predominantly defensive purposes through a modified ovipositor that injects venom subcutaneously.[8] Honeybee stings, for example, deliver melittin and phospholipase A2, which cause membrane lysis, hemolysis, and intense nociceptive signaling, often accompanied by barbed stinger autotomy to prolong venom release post-detachment.[9] Wasp and ant venoms similarly emphasize alarm pheromones and cytotoxic peptides to rally colony defense and inflict localized damage, deterring vertebrates without necessarily paralyzing them.[10] Predatory bites predominate in arachnids like spiders, which use hollow cheliceral fangs to inject venom directly into prey tissues.[11] Spider venoms contain diverse neurotoxins, such as voltage-gated ion channel modulators, that selectively paralyze insects by altering synaptic transmission, enabling efficient predation on arthropods up to the spider's size.[12] Scorpions employ a telson stinger for both predation and defense, injecting alkaloid toxins and peptides that block sodium or potassium channels in insect nerves for immobilization, though defensive stings against mammals may prioritize pain over lethality.[13] Certain predatory ants, such as trap-jaw species, also sting prey to subdue it before dismemberment.[14] These mechanisms highlight causal adaptations: defensive venoms evolve under pressure from vertebrate predators to maximize deterrence with minimal energy cost, while predatory ones optimize for prey-specific efficacy, often trading off against non-target effects.[15] Empirical studies confirm venom potency varies by target, with insect LD50 values far lower than mammalian ones in predatory species.[7]Feeding and Blood-Seeking Bites
Hematophagous arthropods perform blood-seeking bites to obtain vertebrate blood as a nutrient source, a behavior that has evolved independently at least nine times in insects alone and additional times in arachnids.[16] These organisms locate hosts using sensory cues such as carbon dioxide, body heat, and odors, then employ specialized piercing-sucking mouthparts to penetrate skin and extract blood.[17] The feeding process disrupts hemostasis through salivary secretions containing bioactive molecules, including anticoagulants, platelet aggregation inhibitors, and vasodilators, which prevent clotting and promote blood flow.[18] [19] In blood-feeding insects like mosquitoes (order Diptera), females insert a proboscis formed by six stylets—two pairs of maxillary blades for cutting tissue, a labrum forming the food canal, and guiding structures—into capillaries.[20] Cibarial and pharyngeal pumps in the head facilitate blood ingestion, while saliva anesthetizes the site and inhibits coagulation.[21] Fleas (Siphonaptera) utilize lacinial stylets to cannulate capillaries directly, injecting saliva that digests host cells and anticoagulates blood for rapid engorgement.[22] Sucking lice (Anoplura) pierce skin with a proboscis-like haustellum, everting it to form a feeding tube that draws blood, aided by similar antihemostatic saliva.[23] Among arachnids, hard ticks (Ixodida) use toothed chelicerae to incise skin and a hypostome with recurved barbs for anchorage, secreting cement-like saliva to seal the wound and create a blood pool by rupturing capillaries and injecting vasodilators.[24] [25] Feeding duration varies, with insects often completing meals in minutes and ticks attaching for days to weeks, during which saliva modulates host immune responses to sustain intake.[26] This prolonged attachment increases vector potential for pathogen transmission.[27]Clinical Manifestations
Local Reactions
Local reactions to arthropod bites and stings generally involve localized cutaneous inflammation characterized by pain, erythema, edema, and pruritus, resulting from direct tissue trauma, enzymatic venom components, or allergenic salivary proteins that activate mast cells and cytokine release.[1] These responses are typically self-limited, resolving within hours to a week without intervention, though severity varies by species and host sensitivity.[3] In hymenoptera stings (e.g., bees, wasps, ants), immediate intense pain accompanies a wheal-and-flare reaction, with central edema often measuring less than 5 cm in diameter, surrounded by transient erythema; the stinger may remain embedded in apid stings, exacerbating local tissue damage.[3] Mosquito and flea bites produce delayed pruritic papules or urticarial wheals, peaking 24-48 hours post-bite due to IgE-mediated hypersensitivity to saliva, sometimes forming linear clusters from multiple assaults. Bite marks from dengue-transmitting mosquitoes (Aedes aegypti or Aedes albopictus) are generally indistinguishable from those of other common mosquitoes, typically appearing as red, itchy, raised bumps or welts on the skin. There is no reliable visual difference in the bite mark itself to determine if it came from a dengue-carrying mosquito. Identification relies on symptoms of dengue fever (if developed), time of day of the bite (Aedes bite during daytime), or laboratory testing, not the appearance of the bite.[4][28] Tick bites often elicit minimal initial reaction but can develop into persistent erythematous nodules or eschars in sensitized individuals, with hypersensitivity manifesting as itchy swellings under 5 cm within 48 hours of attachment.[29][30] Spider bites typically cause prompt puncture-site pain and pallor, evolving to indurated erythema and mild swelling in most cases, though loxoscelid species may induce delayed ischemic necrosis in susceptible hosts via sphingomyelinase D toxin, forming ulcers up to several centimeters over 3-7 days.[31][32] Local reactions may be intensified when multiple bites or stings occur in proximity, leading to greater swelling and warmth due to additive inflammatory effects.[3] Secondary excoriation from scratching can lead to bacterial superinfection. Signs of secondary infection include increasing redness, swelling, warmth, pain or tenderness, pus drainage, red streaks extending from the site, fever, or chills. Such infections may present as pustules or cellulitis, necessitating topical or oral antibiotics in 5-10% of uncomplicated cases.[3][33] A red, swollen bite persisting for 15 days or longer without improvement is atypical and may indicate secondary bacterial infection (e.g., cellulitis with worsening redness, swelling, warmth, and pain that may spread) or an exaggerated local allergic reaction (e.g., skeeter syndrome from mosquito bites, causing large areas of swelling, redness, and itching lasting longer than usual). Other possibilities include impetigo or rare complications. Medical evaluation is recommended if symptoms persist beyond a week, worsen, or show signs of spreading or infection.[34][35] Persistent or exaggerated local responses, such as bullous, vasculitic, or granulomatous lesions, arise in chronic exposures or immunocompromised states, potentially mimicking infections or autoimmune dermatoses; biopsy may reveal eosinophilic infiltrates or dermal edema confirming arthropod etiology.[4] Management prioritizes symptom relief with cool compresses, antihistamines, and corticosteroids for moderate cases, avoiding unproven therapies like intralesional injections absent necrosis.[3]Systemic and Allergic Responses
Systemic responses to arthropod bites and stings encompass both IgE-mediated allergic reactions, such as anaphylaxis, and non-allergic toxic effects from venom or saliva components that affect multiple organ systems beyond the site of envenomation.[1] Allergic reactions typically manifest rapidly, within minutes to hours, involving mast cell degranulation triggered by specific venom allergens, leading to histamine release and potential cardiovascular collapse.[36] Non-allergic systemic effects arise from pharmacologically active venom peptides or enzymes, causing autonomic dysregulation, neuromuscular excitation, or cytokine storms, with symptom onset varying by species and dose.[3] Anaphylaxis, the most severe allergic response, occurs in approximately 0.5-3% of individuals following Hymenoptera stings (bees, wasps, ants), with adults at higher risk than children (3% vs. 1%).[37] Symptoms include generalized urticaria, angioedema, bronchospasm, laryngeal edema, hypotension, and gastrointestinal distress, potentially progressing to shock or respiratory arrest if untreated.[38] Patients with prior systemic reactions face a 50-60% recurrence risk upon re-stinging, necessitating venom immunotherapy for sensitization confirmed by skin testing or serum IgE levels.[3] Imported fire ant stings (Solenopsis invicta) similarly provoke anaphylaxis in sensitized individuals, with venom alkaloids acting as haptens to elicit IgE responses.[39] Non-allergic systemic envenomation predominates in arachnid incidents. Latrodectism from black widow spider (Latrodectus spp.) bites involves α-latrotoxin, inducing calcium-dependent neurotransmitter release, resulting in severe muscle cramps, rigidity, diaphoresis, hypertension, and priapism in males, with symptoms peaking 2-4 hours post-bite and lasting days.[40] Scorpion stings by medically significant species (e.g., Tityus, Androctonus) release neurotoxins that overstimulate sodium channels, causing sympathetic and parasympathetic surges: tachycardia, pulmonary edema, pancreatitis, and neuromuscular excitability, with pediatric mortality up to 10% in endemic regions without antivenom.[41] Multiple Hymenoptera stings (>50) can overwhelm via venom dose, yielding hemolysis, rhabdomyolysis, and acute kidney injury independent of allergy.[36] Rarely, arthropod exposures trigger delayed systemic hypersensitivity, such as alpha-gal syndrome from lone star tick (Amblyomma americanum) bites, where salivary IgE cross-reacts with mammalian oligosaccharides, causing anaphylaxis to red meat 3-6 hours post-ingestion.[3] Overall, systemic reactions demand prompt epinephrine for anaphylaxis and supportive care or antivenom for toxidromes, with fatality rates under 0.1% in developed settings due to access to interventions.[42]Epidemiology
Global and Regional Prevalence
Arthropod bites and stings impose a substantial global health burden, though comprehensive prevalence data remain limited due to widespread underreporting of minor or self-resolving cases. Medically significant envenomations from scorpions, spiders, and hymenopterans (bees, wasps, hornets) predominate in estimates, with scorpion stings alone affecting over 1.2 million people annually and causing more than 3,000 deaths, primarily through neurotoxic effects in untreated cases.[43] Hymenopteran stings contribute to systemic reactions worldwide, including anaphylaxis, though exact global incidence is elusive; in the United States, such stings prompt thousands of poison control consultations yearly and result in an average of 72 fatalities annually from 2011 to 2021.[44] Tick bites, often prolonged for blood-feeding, affect millions but are infrequently quantified beyond associated diseases like Lyme borreliosis, which implies high exposure rates in endemic areas.[1] Prevalence varies markedly by arthropod type and geography, driven by climatic factors, habitat proximity, and human activity. In tropical and subtropical regions, scorpionism predominates, with 2.5 billion people at risk and annual stings reaching 1.5 million, concentrated in Africa, the Middle East, Latin America, and South Asia where arid environments favor species like Tityus and Androctonus.[45] Brazil reports over 145,000 arthropod envenomations yearly, with scorpions accounting for more than 60% of the 145 associated deaths, highest in southeastern and northeastern states due to urban expansion into scorpion habitats.[46] In contrast, temperate zones see elevated hymenopteran and tick exposures; Europe records disproportionate sting fatalities in western (42.8%) and eastern (31.9%) regions over 1994–2016, linked to foraging behaviors of Vespula wasps and Apis bees.[47] Urban-rural divides further modulate regional risks, with rural agricultural workers facing higher rates from ground-dwelling arthropods like scorpions and spiders, while urban areas in developing nations report surges in indoor infestations. In sub-Saharan Africa and Southeast Asia, mosquito bites occur ubiquitously—billions daily—but escalate to clinical concern in vector hotspots, correlating with 1.5% of hospital admissions for insect exposures in studied tropical settings like Zimbabwe.[2] Developed regions like the United States experience up to 1 million emergency visits annually for bites and stings, predominantly from ticks, spiders, and hymenopterans, with underreporting masking true population-level incidence.[48] These patterns underscore environmental determinism in prevalence, with warming climates potentially expanding ranges of vectors like ticks into higher latitudes.[1]Trends and Risk Factors
The incidence of arthropod bites and stings has shown variable trends globally, with notable increases in vector-related exposures such as tick and mosquito bites attributed to climate warming, which extends arthropod active seasons and expands habitats northward. For instance, in the United States, reported Lyme disease cases—primarily transmitted via tick bites—rose from approximately 9,000 in 1992 to over 476,000 annually by recent estimates, correlating with warmer temperatures facilitating tick range expansion into previously cooler regions. Similarly, mosquito-borne diseases like West Nile virus and Eastern equine encephalitis have seen heightened transmission risks due to prolonged warm and wet seasons, with experts projecting further rises as global temperatures increase.[49][50] Hymenopteran stings (e.g., bees, wasps) exhibit more stable patterns in mortality, averaging 72 deaths per year in the U.S. from 2011–2021, though emergency department visits for arthropod envenomations remain common, with rates around 19.3 incidents per 10,000 person-years in active-duty military personnel from 2014–2023. Google Trends data from 2004–2021 indicate rising public interest in tick bites, insect bites, and bed bug infestations, potentially reflecting increased encounters amid urbanization and travel, while pubic lice queries declined. Overall, vector-borne diseases linked to arthropod bites account for over 700,000 deaths annually worldwide, underscoring a persistent but uneven epidemiological burden.[44][51][52] Key risk factors for arthropod bites and stings include environmental exposure, such as residing or working in endemic areas with high arthropod densities, particularly during peak seasons (e.g., summer for wasps and hornets). Outdoor occupations like farming, forestry, and landscaping elevate incidence, as do recreational activities in natural settings; travelers to tropical or subtropical regions face amplified risks from unfamiliar vectors. Individual factors encompass a history of atopy or prior allergic reactions, which heighten susceptibility to severe responses, alongside advanced age and conditions like clonal mast cell disorders that predispose to systemic envenomation effects. Behavioral elements, such as inadequate use of repellents or protective clothing, further compound risks, especially in children and immunocompromised individuals who may experience amplified local or systemic reactions.[53][4][54]Taxonomy of Culprit Arthropods
Insects
Insects implicated in human bites and stings primarily belong to the orders Hymenoptera, Diptera, Siphonaptera, and Phthiraptera, with Hymenoptera responsible for venomous stings and the others mainly for blood-feeding bites that provoke local dermal reactions or transmit pathogens.[1] These interactions often result from defensive behaviors in Hymenoptera or obligatory hematophagy in the parasitic orders, leading to clinical effects ranging from transient irritation to severe anaphylaxis or vector-borne diseases.[8] Order Hymenoptera encompasses the stinging insects, including the superfamilies Apoidea (bees and stinging wasps) and Vespoidea (yellowjackets, hornets, and ants). Key families are Apidae (honeybees and bumblebees), Vespidae (wasps and hornets), and Formicidae (fire ants and other stinging ants), which deploy modified ovipositors as stingers to inject alkaline or acidic venoms containing peptides, amines, and enzymes that induce pain, edema, and potential systemic toxicity.[8] Hymenopteran stings account for the majority of insect-related envenomations requiring medical attention, with anaphylaxis occurring in up to 3% of the population upon re-exposure due to IgE-mediated hypersensitivity to venom allergens like phospholipase A2 and hyaluronidase.[8] Fire ants (Solenopsis invicta) deliver multiple stings forming sterile pustules via alkaloid venom, contributing significantly to morbidity in endemic regions like the southeastern United States, where over 50% of households report encounters.[1] Order Diptera, the true flies, includes hematophagous species with piercing-sucking mouthparts that lacerate skin and inject saliva to facilitate blood meals, often eliciting pruritic wheals from anticoagulants and vasodilators.[1] Dominant families are Culicidae (mosquitoes, such as Aedes and Anopheles species), which transmit malaria, dengue, and Zika via salivary pathogens, and Ceratopogonidae (biting midges or no-see-ums), notorious for intense itching in clusters due to small size (1-3 mm) and swarming behavior in coastal or wetland areas.[55] Tabanidae (horseflies) cause larger, more painful bites from robust mouthparts, occasionally leading to secondary infections or allergic responses, while Simuliidae (blackflies) provoke edematous reactions in riverine environments.[1] Order Siphonaptera comprises fleas, small (1-4 mm), wingless ectoparasites with laterally compressed bodies and powerful hind legs for jumping up to 30 cm. Primary human pests include Pulex irritans (human flea) and Ctenocephalides felis (cat flea), which bite preferentially at ankles and waistlines, injecting saliva that causes erythematous papules and intense pruritus, sometimes with central vesicles.[1] Fleas serve as vectors for Yersinia pestis (plague) and Bartonella henselae (cat-scratch disease), with historical pandemics like the Black Death killing an estimated 75-200 million people in Eurasia during the 14th century.[1] Order Phthiraptera consists of lice, obligate parasites divided into Anoplura (sucking lice) and Mallophaga (biting lice, less relevant to humans). Pediculus humanus (body louse) and Pthirus pubis (pubic louse) use stylet-like mouthparts to pierce skin for blood, resulting in maculopapular rashes and secondary excoriations from scratching, with body lice transmitting Rickettsia prowazekii (epidemic typhus).[1] Infestations thrive in crowded, unhygienic conditions, as evidenced by increased prevalence during World War I and II outbreaks affecting millions of soldiers.[1] Other orders like Lepidoptera (caterpillars with urticating hairs) and Coleoptera (blister beetles secreting cantharidin) occasionally cause dermatitis or vesication but are not primary biters or stingers in humans.[56]Arachnids
Arachnids responsible for human bites and stings belong to the class Arachnida within phylum Arthropoda, distinguished by eight legs in adults and lacking antennae. The primary orders implicated include Araneae (spiders), Scorpiones (scorpions), and elements of Acari (ticks and mites), where interactions with humans typically involve defensive envenomation or parasitic feeding.[57][58] Spiders (order Araneae) deliver bites via hollow chelicerae that inject venom, with medical significance limited to fewer than 100 of approximately 50,000 species worldwide. Theridiidae family includes Latrodectus genus (widow spiders), whose neurotoxic venom causes latrodectism characterized by muscle cramps and autonomic effects; species like Latrodectus mactans (black widow) in North America and Latrodectus tredecimguttatus in the Mediterranean are notable. Sicariidae family harbors Loxosceles genus (recluse spiders), producing cytotoxins leading to necrotic arachnidism, as seen in Loxosceles reclusa bites in the United States. Other genera like Phoneutria (Ctenidae family) in South America cause priapism and systemic symptoms due to potent neurotoxins. Most spider bites result in minor local reactions without confirmed species identification, emphasizing taxonomic precision for assessing risk.[59][60][61] Scorpions (order Scorpiones) sting using a telson-mounted aculeus, with venom comprising neurotoxins affecting ion channels; of over 2,200 species, about 50 pose significant threat, predominantly in Buthidae family. Leiurus quinquestriatus (deathstalker) in North Africa and Middle East exemplifies high toxicity, causing severe pain, cardiovascular instability, and occasional fatalities, particularly in children. Androctonus and Tityus genera in regions like North Africa and South America similarly yield potent venoms leading to scorpionism, with annual global estimates exceeding 1 million envenomations and thousands of deaths concentrated in arid tropics. Non-Buthidae like Hemiscorpius lepturus induce hemolysis and renal failure via unique cytotoxins.[62][63][64] Within subclass Acari, ticks (superorder Parasitiformes, order Ixodida) are obligate hematophagous ectoparasites that attach via hypostome barbs for prolonged feeding, often transmitting pathogens like Borrelia burgdorferi in Ixodidae family hard ticks (e.g., Ixodes scapularis). Argasidae soft ticks bite nocturnally, associated with relapsing fever via Borrelia species. Mites, particularly Trombiculidae family chiggers (larval stage of trombiculid mites), inject salivary enzymes causing intense pruritus and papular dermatitis through superficial skin digestion, without deep penetration or disease vectoring in most cases. These arachnids underscore Acari's role in both direct irritation and vector-borne transmission, with over 900 tick species globally.[65][66][67]Myriapods
Myriapods, encompassing the classes Chilopoda (centipedes) and Diplopoda (millipedes), infrequently cause human injuries compared to other arthropods, but centipede bites can produce significant envenomation while millipede defensive secretions lead to chemical dermatitis.[68] Centipedes deploy modified first-leg appendages called forcipules to deliver venom, resulting in puncture wounds akin to stings, whereas millipedes lack biting mouthparts and instead exude irritant fluids from lateral repugnatorial glands when threatened.[69][70] Injuries occur primarily in tropical and subtropical regions where larger species abound, often during accidental encounters in soil, leaf litter, or homes.[71] Centipede envenomations manifest with immediate, intense localized pain described as burning or lancinating, accompanied by erythema, edema, and induration at the bite site, which may persist for hours to days.[69] Venom components, including peptides and proteins, target ion channels and neurotransmitters, exacerbating pain via neurotoxic effects; larger species like Scolopendra spp. in tropical areas can induce rarer systemic symptoms such as nausea, vomiting, headache, fever, or localized lymphangitis, though fatalities are exceedingly rare and typically linked to anaphylaxis or secondary infection in vulnerable individuals.[68] In a prospective Australian study of 14 cases, pain was universal and severe in half, with swelling in 43% and no systemic features reported, resolving without sequelae via symptomatic management.[72] Treatment emphasizes analgesia (e.g., opioids for severe pain), wound cleansing, elevation, and tetanus prophylaxis; antihistamines or corticosteroids may mitigate inflammation, but antivenom is unavailable and unnecessary for most cases.[69] Millipede interactions with humans involve topical exposure to benzoquinones and other phenols secreted from ozopores, causing a characteristic chemical burn rather than mechanical injury.[70] Effects include immediate burning sensation, pruritus, vesiculation, and delayed brown hyperpigmentation or "dermonecrosis" that can last weeks to months, with ocular exposure risking conjunctivitis or keratitis if secretions contact eyes.[73][74] Unlike centipede venom, these secretions are not injected but act as vesicants on contact, with severity depending on volume and skin integrity; most resolve with soap-water irrigation, cool compresses, and topical steroids, though blistering may necessitate wound care to prevent secondary bacterial infection.[75] Human cases remain underreported, often self-limiting, but accidental crushing of millipedes on skin—such as in bedding—can produce persistent lesions mimicking thermal burns.[76] No fatalities are documented from millipede secretions alone.[70]Vector Role in Disease Transmission
Key Pathogens and Diseases
Arthropods serve as vectors for numerous pathogens, primarily through bites that facilitate mechanical or biological transmission during blood meals. Mosquitoes, ticks, fleas, lice, and other hematophagous species transmit bacteria, viruses, protozoa, and helminths responsible for diseases affecting millions annually, with over 700,000 deaths from vector-borne illnesses reported each year, predominantly malaria.[53] Biological transmission involves pathogen replication or development within the vector, as seen in malaria parasites undergoing sporogony in mosquito salivary glands, whereas mechanical transmission occurs via contaminated mouthparts, such as fleas spreading plague bacteria.[77] Mosquito-borne pathogens dominate global vector-borne disease burden. Plasmodium falciparum and other Plasmodium species cause malaria, with Anopheles mosquitoes injecting sporozoites during bites; in 2023, malaria resulted in 249 million cases and 608,000 deaths worldwide.[53] Arboviruses like dengue virus (serotypes 1-4), transmitted by Aedes aegypti and Aedes albopictus, lead to severe hemorrhagic fever, with over 5 million cases reported in the Americas alone in 2023.[78] Zika virus, also Aedes-transmitted, causes microcephaly in congenital infections, while West Nile virus, spread by Culex species, induces neuroinvasive disease in up to 1% of cases.[79] Yellow fever virus, vectored by Aedes and Haemagogus mosquitoes, persists in sylvatic cycles with urban outbreaks killing up to 50% of severe cases without vaccination.[53] Tick-borne diseases involve spirochetes, rickettsiae, and viruses acquired during prolonged attachment. Borrelia burgdorferi, transmitted by Ixodes ticks, causes Lyme disease, characterized by erythema migrans rash and potential dissemination to joints and nerves; U.S. cases exceeded 476,000 annually as of 2018 data.[79] Rickettsia rickettsii, vectored by Dermacentor ticks, leads to Rocky Mountain spotted fever with vasculitis and mortality up to 20% if untreated.[30] Anaplasma phagocytophilum and Ehrlichia species cause anaplasmosis and ehrlichiosis via Ixodes and Amblyomma ticks, respectively, presenting as flu-like illness with leukopenia.[30] Tick-borne encephalitis virus, spread by Ixodes in Europe and Asia, results in meningoencephalitis with 1-2% fatality in severe forms.[53] Flea- and louse-borne bacterial diseases highlight mechanical transmission risks. Yersinia pestis, carried by rodent fleas like Xenopsylla cheopis, causes bubonic plague, with human cases reaching 2,000-3,000 yearly globally, often from sylvatic reservoirs.[53] Body lice (Pediculus humanus corporis) transmit Rickettsia prowazekii, etiologic agent of epidemic typhus, which ravaged populations in wars and famines with mortality up to 60% pre-antibiotics.[53] Lice also vector Borrelia recurrentis for louse-borne relapsing fever, featuring cyclic fevers due to antigenic variation.[53] Other notable transmissions include Trypanosoma cruzi via triatomine bug ("kissing bug") bites for Chagas disease, causing chronic cardiomyopathy in 20-30% of infected individuals in endemic Americas, and Leishmania species by sandfly bites leading to cutaneous or visceral leishmaniasis with 700,000-1 million new cases yearly.[53] These pathogens exploit vector physiology, evading innate immunity via mechanisms like salivary immunomodulators that enhance host infectivity.[80]| Arthropod Vector | Key Pathogen(s) | Primary Disease(s) | Global Burden (Recent Estimates) |
|---|---|---|---|
| Mosquitoes (Anopheles, Aedes) | Plasmodium spp., Dengue virus, Zika virus | Malaria, Dengue, Zika | 249M malaria cases (2023)[53] |
| Ticks (Ixodes, Dermacentor) | Borrelia burgdorferi, Rickettsia rickettsii | Lyme disease, Rocky Mountain spotted fever | >476K Lyme cases/year (U.S.)[79] |
| Fleas (Xenopsylla) | Yersinia pestis | Plague | 2K-3K cases/year[53] |
| Lice (Pediculus humanus) | Rickettsia prowazekii | Epidemic typhus | Sporadic outbreaks in conflict zones[53] |

