Octasulfur
View on Wikipedia
| |||
 | |||
 | |||
| Names | |||
|---|---|---|---|
| Systematic IUPAC name | |||
| Other names
Octasulfur
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| 2973 | |||
| MeSH | Cyclooctasulfur | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| S8 | |||
| Molar mass | 256.48 g·mol−1 | ||
| Appearance | Vivid, yellow, translucent crystals | ||
| Density | 2.07 g/cm3 | ||
| Melting point | 119 °C; 246 °F; 392 K | ||
| Boiling point | 444.6 °C; 832.4 °F; 717.8 K | ||
| log P | 6.117 | ||
| Thermochemistry | |||
Std molar
entropy (S⦵298) |
32 J·mol−1·K−1[3] | ||
Std enthalpy of
formation (ΔfH⦵298) |
0 kJ/mol[3] | ||
| Related compounds | |||
Related compounds
|
Hexathiane | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Octasulfur is an inorganic substance with the chemical formula S8. It is an odourless and tasteless yellow solid, and is a major industrial chemical. It is the most common allotrope of sulfur and occurs widely in nature.[4]
Nomenclature
[edit]The name octasulfur is the most commonly used for this chemical. It is systematically named cyclo-octasulfur (which is the preferred IUPAC name) and cyclooctasulfane. It is also the final member of the thiocane heterocylic series, where every carbon atom is substituted with a sulfur atom, thus this sulfur allotrope is systematically named octathiocane as well.
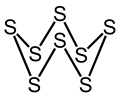
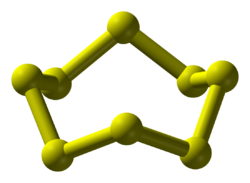
Structure
[edit]The chemical consists of rings of 8 sulfur atoms. It adopts a crown conformation with D4d point group symmetry. The S–S bond lengths are equal, at about 2.05 Å. Octasulfur crystallizes in three distinct polymorphs: rhombohedral, and two monoclinic forms, of which only two are stable at standard conditions. The rhombohedral crystal form is the accepted standard state. The remaining polymorph is only stable between 96 and 115 °C at 100 kPa. Octasulfur forms several allotropes: α-sulfur, β-sulfur, γ-sulfur, and λ-sulfur.
λ-Sulfur is the liquid form of octasulfur, from which γ-sulfur can be crystallised by quenching. If λ-sulfur is crystallised slowly, it will revert to β-sulfur. Since it must have been heated over 115 °C, neither crystallised β-sulfur or γ-sulfur will be pure. The only known method of obtaining pure γ-sulfur is by crystallising from solution.
Octasulfur easily forms large crystals, which are typically yellow and are somewhat translucent.
Production and reactions
[edit]It is the main (99%) component of elemental sulfur, which is recovered from volcanic sources and is a major product of the Claus process, associated with petroleum refineries. In space, sulfur can be formed by energetic processing of solid hydrogen sulfide,[5] which explains the recent detection of octasulfur in the return samples from the carbonaceous asteroid Ryugu.[6]
See also
[edit]References
[edit]- ^ International Union of Pure and Applied Chemistry (2005). Nomenclature of Inorganic Chemistry (IUPAC Recommendations 2005). Cambridge (UK): RSC–IUPAC. ISBN 0-85404-438-8. p. 49. Electronic version.
- ^ "cyclooctasulfur (CHEBI:29385)". Chemical Entities of Biological Interest. UK: European Bioinformatics Institute. Main.
- ^ a b Zumdahl, Steven S. (2009). Chemical Principles 6th Ed. Houghton Mifflin Company. p. A23. ISBN 978-0-618-94690-7.
- ^ Steudel, R., "Homocyclic Sulfur Molecules", Topics Curr. Chem. 1982, 102, 149.
- ^ Herath, Ashanie; Mcanally, Mason; Turner, Andrew M.; Wang, Jia; Marks, Joshua H.; Foretenberry, Ryan C.; Garcia-Alvarez, Jorge C.; Gozem, Samer; Kaiser, Ralf I. (1 July 2025). "Missing interstellar sulfur in inventories of polysulfanes and molecular octasulfur crowns". Nature Communications. doi:10.1038/s41467-025-61259-2. PMC 12215914.
- ^ Takano, Yoshinori (10 July 2024). "Primordial aqueous alteration recorded in water-soluble organic molecules from the carbonaceous asteroid (162173) Ryugu". Nature Communications. doi:10.1038/s41467-024-49237-6. PMC 11237059.
External links
[edit] Media related to Octasulfur at Wikimedia Commons
Media related to Octasulfur at Wikimedia Commons
Octasulfur
View on GrokipediaNomenclature and Structure
Nomenclature
Octasulfur, denoted as S₈, is the most stable and prevalent molecular form of elemental sulfur under standard conditions. It is commonly referred to by the name "octasulfur," which directly indicates its composition of eight sulfur atoms, or "crown sulfur," alluding to the puckered ring arrangement of its atoms.[3][4] The systematic IUPAC name for this compound is octathiocane, a term derived from the heterocyclic nomenclature where "thiocane" denotes an eight-membered ring containing sulfur, extended here to an all-sulfur cycle. Alternative designations include cyclooctasulfane and cyclo-octasulfur, emphasizing its cyclic nature as the final member in the series of polysulfane rings.[5] Historically, elemental sulfur, including its octasulfur allotrope, has been known since prehistoric times under names like "brimstone," from Old English "brennen stān" meaning "burning stone," due to its ready ignition. In the 17th to 19th centuries, a purified, finely powdered form obtained by sublimation was termed "flowers of sulfur" (or "flores sulphuris" in Latin), which primarily comprises S₈ molecules and was used in early chemical and medicinal applications. The shift to modern systematic nomenclature, such as octasulfur and its IUPAC equivalents, emerged in the early 20th century alongside advancements in structural chemistry and the establishment of IUPAC guidelines for inorganic compounds.[6][7]Molecular Structure
Octasulfur, with the molecular formula S, consists of eight sulfur atoms arranged in a cyclic structure. This molecule adopts a puckered crown conformation, characterized by D point group symmetry, where the ring is non-planar to minimize steric repulsion between lone pairs on adjacent atoms.[8][9] In this arrangement, the S–S bond lengths average 2.05 Å. The intraring bond angles are consistently around 107.8°, reflecting the tetrahedral-like geometry influenced by the valence shell electron pair repulsion. These structural parameters have been precisely determined through single-crystal X-ray diffraction studies at low temperatures, confirming the uniformity of the ring in isolated or solution phases.[10][11] The bonding in octasulfur involves homonuclear single σ-bonds between sulfur atoms, formed by overlap of p-orbitals, with each sulfur atom retaining two lone pairs in its valence shell. This configuration aligns with sulfur's ground-state electron arrangement ([Ne] 3s 3p), where two electrons per atom are shared in bonds, leaving four as non-bonding pairs that occupy hybrid orbitals. The absence of π-bonding contributes to the molecule's reactivity and stability.[12] Compared to smaller cyclic sulfur allotropes such as S and S, the S ring exhibits greater thermodynamic stability, as evidenced by its higher second-order energy difference and lower fragmentation energy, making it the predominant form under ambient conditions. While S (D symmetry) and S can be synthesized and isolated, they are less favored due to increased ring strain and higher energy states relative to the optimally sized eight-membered crown.[13]Polymorphs and Allotropes
Octasulfur exhibits three primary crystalline polymorphs, each consisting of packed S8 rings in distinct arrangements. The α-polymorph, orthorhombic with space group Fddd, is the most stable at room temperature and features 128 sulfur atoms per unit cell, corresponding to 16 S8 molecules arranged in a body-centered lattice.[14] Its unit cell parameters are approximately a = 10.44 Å, b = 12.85 Å, and c = 24.37 Å, resulting in a density of 2.07 g/cm³. The β-polymorph, monoclinic with space group P21/c, forms at higher temperatures and packs 48 sulfur atoms (6 S8 molecules) per unit cell in a less dense configuration, with parameters a ≈ 9.43 Å, b ≈ 11.70 Å, c ≈ 10.07 Å, and β ≈ 107.9°.[15] The γ-polymorph, also monoclinic (P21/c), achieves the highest density among these forms at 2.19 g/cm³ through tighter S8 ring packing, with unit cell parameters a ≈ 9.89 Å, b ≈ 10.82 Å, c ≈ 9.75 Å, and β ≈ 100.9°; it is often considered a standard reference state due to its preparation from precipitated sulfur. In all polymorphs, the S8 rings adopt a crown-like conformation, with intermolecular interactions dominated by van der Waals forces between rings. These octasulfur polymorphs relate to broader sulfur allotropy, where λ-sulfur (also known as fibrous or φ-sulfur) represents a polymeric form with helical catena chains rather than rings, formed by rapid quenching of molten sulfur.[16] Catena-sulfur encompasses various chain-like structures, including long linear polymers that emerge above the λ-transition temperature of approximately 159°C in the melt, contrasting with the cyclic S8 dominance in the solid polymorphs. Octasulfur polymorphs remain the prevalent solid forms up to 95.5°C, beyond which β predominates until melting. Phase transitions among these polymorphs are temperature-dependent. The α-to-β conversion occurs reversibly at 95.5°C, allowing β to revert to α upon cooling below this threshold.[17] In contrast, the γ-polymorph undergoes an irreversible transition to α upon heating or prolonged exposure at room temperature.[18] Recent analyses of samples from the Ryugu asteroid, returned by the Hayabusa2 mission, have detected S8 octasulfur alongside other sulfur species, providing evidence of extraterrestrial occurrence in carbonaceous materials and insights into solar system sulfur distribution as of 2024.[19]Physical and Thermodynamic Properties
Physical Characteristics
Octasulfur, in its stable α-form, manifests as a vivid yellow, translucent crystalline solid composed of orthorhombic crystals. This characteristic appearance arises from the packing of S₈ rings in a puckered crown conformation, giving it a distinctive bright hue under ambient conditions.[2] The material exhibits a density of 2.07 g/cm³ at 20°C, reflecting its compact molecular structure. Its melting point is 112.8°C for the α-form, transitioning to a viscous yellow liquid, while the boiling point reaches 444.6°C under standard pressure.[20] These thermal transitions are influenced by polymorphic variations, though the α-form predominates at room temperature.[21] Octasulfur demonstrates negligible solubility in water, rendering it hydrophobic and unreactive in aqueous environments. In contrast, it dissolves readily in nonpolar organic solvents, such as carbon disulfide (39 g/100 mL at 25°C), benzene (approximately 1.2 g/100 mL at 25°C), and toluene (approximately 2 g/100 mL at 25°C).[22] The compound possesses a faint odor, often described as mild or nearly odorless in pure form, and exhibits low volatility due to its minimal vapor pressure at room temperature.[2] Optically, octasulfur in the α-form has a refractive index of 1.957, contributing to its translucency and high light dispersion in crystalline samples.[20]Thermodynamic Data
Octasulfur, in its standard rhombic form (α-S₈), serves as the reference state for sulfur's thermodynamic properties, with the standard enthalpy of formation ΔH_f° and Gibbs free energy of formation ΔG_f° both defined as 0 kJ/mol at 298 K and 1 bar, underscoring its thermodynamic stability as the elemental standard.[23] The standard molar entropy S° is 32.1 J/mol·K at 298 K, reflecting the ordered molecular structure in the solid phase.[23] The molar heat capacity at constant pressure C_p for the solid is 22.74 J/mol·K at 298 K, indicating moderate thermal response typical of covalent network solids.[24] Phase transitions of octasulfur involve characteristic energy changes that govern its behavior under varying conditions. The enthalpy of fusion is 1.727 kJ/mol at the melting point of 112.8°C, representing the energy required to disrupt the solid lattice into the liquid phase.[25] The enthalpy of vaporization is approximately 45 kJ/mol at the normal boiling point of 444.6°C, highlighting the significant intermolecular forces in the liquid that must be overcome for gas formation.[26] The phase diagram of octasulfur features a triple point at approximately 115.2°C and 0.00026 MPa (26 Pa), where the monoclinic solid, liquid, and vapor phases coexist in equilibrium; this point marks the lower limit of the liquid-vapor boundary for the β-form. Due to the α-to-β transition at 95.5 °C, the rhombic (α) form does not directly coexist with liquid and vapor at this temperature.[27] The critical point occurs at 1041°C and 20.7 MPa, beyond which the distinction between liquid and vapor phases vanishes, defining the upper limit of the two-phase region. These parameters illustrate the energetic constraints on octasulfur's phase stability, with brief links to polymorph transitions such as rhombic to monoclinic observed in the solid region.[28]| Property | Value | Conditions | Source |
|---|---|---|---|
| Standard enthalpy of formation (ΔH_f°) | 0 kJ/mol | 298 K, solid | Standard Thermodynamic Values PDF |
| Standard Gibbs free energy of formation (ΔG_f°) | 0 kJ/mol | 298 K, solid | Standard Thermodynamic Values PDF |
| Standard entropy (S°) | 32.1 J/mol·K | 298 K, solid | Standard Thermodynamic Values PDF |
| Molar heat capacity (C_p) | 22.74 J/mol·K | 298 K, solid | Heat Capacity Table |
| Enthalpy of fusion (ΔH_fus) | 1.727 kJ/mol | 112.8°C | WebElements Sulfur Thermochemistry |
| Enthalpy of vaporization (ΔH_vap) | 45 kJ/mol | 444.6°C | WebElements Sulfur Thermochemistry |
| Triple point | 115.2°C, 0.00026 MPa | Monoclinic solid-liquid-vapor | Phase Diagram Explanation |
| Critical point | 1041°C, 20.7 MPa | Liquid-vapor | Periodic Table Data |