Recent from talks
Nothing was collected or created yet.
4-Hydroxybenzoic acid
View on Wikipedia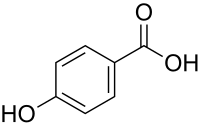 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
4-Hydroxybenzoic acid | |
| Other names
p-Hydroxybenzoic acid
para-Hydroxybenzoic acid PHBA 4-hydroxybenzoate | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.002.550 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C7H6O3 | |
| Molar mass | 138.122 g·mol−1 |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 1.46 g/cm3 |
| Melting point | 214.5 °C (418.1 °F; 487.6 K) |
| Boiling point | N/A, decomposes[1] |
| 0.5 g/100 mL | |
| Solubility |
|
| log P | 1.58 |
| Acidity (pKa) | 4.54 |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Irritant |
| NFPA 704 (fire diamond) | |
| 250 °C (482 °F; 523 K) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
2200 mg/kg (oral, mouse) |
| Safety data sheet (SDS) | HMDB |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
4-Hydroxybenzoic acid, also known as p-hydroxybenzoic acid (PHBA), is a monohydroxybenzoic acid, a phenolic derivative of benzoic acid. It is a white crystalline solid that is slightly soluble in water and chloroform but more soluble in polar organic solvents such as alcohols and acetone. 4-Hydroxybenzoic acid is primarily known as the basis for the preparation of its esters, known as parabens, which are used as preservatives in cosmetics and some ophthalmic solutions. It is isomeric with 2-hydroxybenzoic acid, known as salicylic acid, a precursor to aspirin, and with 3-hydroxybenzoic acid.
Natural occurrences
[edit]It is found in plants of the genus Vitex such as V. agnus-castus or V. negundo, and in Hypericum perforatum (St John's wort). It is also found in Spongiochloris spongiosa, a freshwater green alga.
The compound is also found in Ganoderma lucidum, a medicinal mushroom with the longest record of use.
Cryptanaerobacter phenolicus is a bacterium species that produces benzoate from phenol via 4-hydroxybenzoate.[2]
Occurrences in food
[edit]4-Hydroxybenzoic acid can be found naturally in coconut.[3] It is one of the main catechins metabolites found in humans after consumption of green tea infusions.[4] It is also found in wine,[5] in vanilla, in Macrotyloma uniflorum (horse gram), carob[6] and in Phyllanthus acidus (Otaheite gooseberry).
Açaí oil, obtained from the fruit of the açaí palm (Euterpe oleracea), is rich in p-hydroxybenzoic acid (892±52 mg/kg).[7] It is also found in cloudy olive oil[citation needed] and in the edible mushroom Russula virescens (green-cracking russula).[citation needed]
Related compounds
[edit]p-Hydroxybenzoic acid glucoside can be found in mycorrhizal and non-mycorrhizal roots of Norway spruces (Picea abies).[8]
Violdelphin is an anthocyanin, a type of plant pigments, found in blue flowers and incorporating two p-hydroxybenzoic acid residues, one rutinoside and two glucosides associated with a delphinidin.
Agnuside is the ester of aucubin and p-hydroxybenzoic acid.[9]
Biosynthesis
[edit]Chorismate lyase is an enzyme that transforms chorismate into 4-hydroxybenzoate and pyruvate. This enzyme catalyses the first step in ubiquinone biosynthesis in Escherichia coli and other Gram-negative bacteria.
Benzoate 4-monooxygenase is an enzyme that utilizes benzoate, NADPH, H+ and O2 to produce 4-hydroxybenzoate, NADP+ and H2O. This enzyme can be found in Aspergillus niger.
4-Hydroxybenzoate also arises from tyrosine.[10]
Metabolism
[edit]As an intermediate
[edit]The enzyme 4-methoxybenzoate monooxygenase (O-demethylating) transforms 4-methoxybenzoate, an electron acceptor AH2 and O2 into 4-hydroxybenzoate, formaldehyde, the reduction product A and H2O. This enzyme participates in 2,4-dichlorobenzoate degradation in Pseudomonas putida.
The enzyme 4-hydroxybenzaldehyde dehydrogenase uses 4-hydroxybenzaldehyde, NAD+ and H2O to produce 4-hydroxybenzoate, NADH and H+. This enzyme participates in toluene and xylene degradation in bacteria such as Pseudomonas mendocina. It is also found in carrots (Daucus carota).
The enzyme that 2,4'-dihydroxyacetophenone dioxygenase transforms 2,4'-dihydroxyacetophenone and O2 into 4-hydroxybenzoate and formate. This enzyme participates in bisphenol A degradation. It can be found in Alcaligenes species.
The enzyme 4-chlorobenzoate dehalogenase uses 4-chlorobenzoate and H2O to produce 4-hydroxybenzoate and chloride. It can be found in Pseudomonas species.
The enzyme 4-hydroxybenzoyl-CoA thioesterase utilizes 4-hydroxybenzoyl-CoA and H2O to produce 4-hydroxybenzoate and CoA. This enzyme participates in 2,4-dichlorobenzoate degradation. It can be found in Pseudomonas species.
The enzyme 4-hydroxybenzoate polyprenyltransferase uses a polyprenyl diphosphate and 4-hydroxybenzoate to produce diphosphate and 4-hydroxy-3-polyprenylbenzoate. This enzyme participates in ubiquinone biosynthesis.
The enzyme 4-hydroxybenzoate geranyltransferase utilizes geranyl diphosphate and 4-hydroxybenzoate to produce 3-geranyl-4-hydroxybenzoate and diphosphate. Biosynthetically, alkannin is produced in plants from the intermediates 4-hydroxybenzoic acid and geranyl pyrophosphate. This enzyme is involved in shikonin biosynthesis. It can be found in Lithospermum erythrorhizon.
The enzyme 3-hydroxybenzoate—CoA ligase uses ATP, 3-hydroxybenzoate and CoA to produce AMP, diphosphate and 3-hydroxybenzoyl-CoA. The enzyme works equally well with 4-hydroxybenzoate. It can be found in Thauera aromatica.
Biodegradation
[edit]The enzyme 4-hydroxybenzoate 1-hydroxylase transforms 4-hydroxybenzoate, NAD(P)H, 2 H+ and O2 into hydroquinone, NAD(P)+, H2O and CO2. This enzyme participates in 2,4-dichlorobenzoate degradation. It can be found in Candida parapsilosis.
The enzyme 4-hydroxybenzoate 3-monooxygenase transforms 4-hydroxybenzoate, NADPH, H+ and O2 into protocatechuate, NADP+ and H2O. This enzyme participates in benzoate degradation via hydroxylation and 2,4-dichlorobenzoate degradation. It can be found in Pseudomonas putida and Pseudomonas fluorescens.
The enzyme 4-hydroxybenzoate 3-monooxygenase (NAD(P)H) utilizes 4-hydroxybenzoate, NADH, NADPH, H+ and O2 to produce 3,4-dihydroxybenzoate (protocatechuic acid), NAD+, NADP+ and H2O. This enzyme participates in benzoate degradation via hydroxylation and 2,4-dichlorobenzoate degradation. It can be found in Corynebacterium cyclohexanicum and in Pseudomonas sp.
The enzyme 4-hydroxybenzoate decarboxylase uses 4-hydroxybenzoate to produce phenol and CO2. This enzyme participates in benzoate degradation via coenzyme A (CoA) ligation. It can be found in Klebsiella aerogenes (Aerobacter aerogenes).
The enzyme 4-hydroxybenzoate—CoA ligase transforms ATP, 4-hydroxybenzoate and CoA to produce AMP, diphosphate and 4-hydroxybenzoyl-CoA. This enzyme participates in benzoate degradation via CoA ligation. It can be found in Rhodopseudomonas palustris.
Coniochaeta hoffmannii is a plant pathogen that commonly inhabits fertile soil. It is known to metabolize aromatic compounds of low molecular weight, such as p-hydroxybenzoic acid.
Glycosylation
[edit]The enzyme 4-hydroxybenzoate 4-O-beta-D-glucosyltransferase transforms UDP-glucose and 4-hydroxybenzoate into UDP and 4-(beta-D-glucosyloxy)benzoate. It can be found in the pollen of Pinus densiflora.
Chemistry
[edit]The Hammett equation describes a linear free-energy relationship relating reaction rates and equilibrium constants for many reactions involving benzoic acid derivatives with meta- and para-substituents.
Chemical production
[edit]4-Hydroxybenzoic acid is produced commercially from potassium phenoxide and carbon dioxide in the Kolbe-Schmitt reaction.[11] It can also be produced in the laboratory by heating potassium salicylate with potassium carbonate to 240 °C, followed by treating with acid.[12]
Chemical reactions
[edit]4-Hydroxybenzoic acid has about one tenth the acidity of benzoic acid, having an acid dissociation constant Ka = 3.3×10−5 M at 19 °C.[citation needed] Its acid dissociation follows this equation:
- HOC6H4CO2H ⇌ HOC6H4CO−2 + H+
Chemical use
[edit]Vectran is a manufactured fiber, spun from a liquid crystal polymer. Chemically it is an aromatic polyester produced by the polycondensation of 4-hydroxybenzoic acid and 6-hydroxynaphthalene-2-carboxylic acid. The fiber has been shown to exhibit strong radiation shielding used by Bigelow Aerospace and produced by StemRad.[13]
4,4′-Dihydroxybenzophenone is generally prepared by the rearrangement of p-hydroxyphenylbenzoate. Alternatively, p-hydroxybenzoic acid can be converted to p-acetoxybenzoyl chloride. This acid chloride reacts with phenol to give, after deacetylation, 4,4′-dihydroxybenzophenone.
Examples of drugs made from PHBA include nifuroxazide, orthocaine, ormeloxifene and proxymetacaine.
Bioactivity and safety
[edit]4-Hydroxybenzoic acid is a popular antioxidant in part because it is rather nontoxic. The LD50 is 2200 mg/kg in mice (oral).[14]
4-Hydroxybenzoic acid has weak estrogenic activity both in vitro and in vivo,[15] and stimulates the growth of human breast cancer cell lines.[16][17] It is a common metabolite of paraben esters, such as methylparaben.[15][16][17] The compound is a relatively weak estrogen, but can produce uterotrophy with sufficient doses to an equivalent extent relative to estradiol, which is unusual for a weakly estrogenic compound and indicates that it may be a full agonist of the estrogen receptor with relatively low binding affinity for the receptor.[16][18][19] It is about 0.2% to 1% as potent as an estrogen as estradiol.[18]
Research
[edit]4-Hydroxybenzoic acid has been used as a precursor to co-enzyme Q10 as an experimental treatment for mitochondrial encephalopathy caused by an inherited deficiency in 4-hydroxyphenylpyruvate dioxygenase-like protein.[20]
See also
[edit]References
[edit]- ^ "4-Hydroxybenzoic acid" (PDF). International Programme on Chemical Safety (IPCS). Archived from the original (PDF) on 24 September 2015. Retrieved 10 January 2015.
- ^ Juteau, P.; Côté, V.; Duckett, M.-F.; Beaudet, R.; Lépine, F.; Villemur, R.; Bisaillon, J.-G. (January 2005). "Cryptanaerobacter phenolicus gen. nov., sp. nov., an anaerobe that transforms phenol into benzoate via 4-hydroxybenzoate". International Journal of Systematic and Evolutionary Microbiology. 55 (1): 245–250. doi:10.1099/ijs.0.02914-0. PMID 15653882.
- ^ Dey, G.; Chakraborty, M.; Mitra, A. (April 2005). "Profiling C6–C3 and C6–C1 phenolic metabolites in Cocos nucifera". Journal of Plant Physiology. 162 (4): 375–381. doi:10.1016/j.jplph.2004.08.006. PMID 15900879.
- ^ Pietta, P. G.; Simonetti, P.; Gardana, C.; Brusamolino, A.; Morazzoni, P.; Bombardelli, E. (1998). "Catechin metabolites after intake of green tea infusions". BioFactors. 8 (1–2): 111–118. doi:10.1002/biof.5520080119. PMID 9699018. S2CID 37684286.
- ^ Tian, R.-R.; Pan, Q.-H.; Zhan, J.-C.; Li, J.-M.; Wan, S.-B.; Zhang, Q.-H.; Huang, W.-D. (2009). "Comparison of phenolic acids and flavan-3-ols during wine fermentation of grapes with different harvest times". Molecules. 14 (2): 827–838. doi:10.3390/molecules14020827. PMC 6253884. PMID 19255542.
- ^ Goulas, V.; Stylos, E.; Chatziathanasiadou, M. V.; Mavromoustakos, T.; Tzakos, A. G. (2016). "Functional Components of Carob Fruit: Linking the Chemical and Biological Space". International Journal of Molecular Sciences. 17 (11): 1875. doi:10.3390/ijms17111875. PMC 5133875. PMID 27834921.
- ^ Pacheco Palencia, L. A.; Mertens-Talcott, S.; Talcott, S. T. (June 2008). "Chemical composition, antioxidant properties, and thermal stability of a phytochemical enriched oil from Açaí (Euterpe oleracea Mart.)". Journal of Agricultural and Food Chemistry. 56 (12): 4631–4636. doi:10.1021/jf800161u. PMID 18522407.
- ^ Münzenberger, B.; Heilemann, J.; Strack, D.; Kottke, I.; Oberwinkler, F. (1990). "Phenolics of mycorrhizas and non-mycorrhizal roots of Norway spruce". Planta. 182 (1): 142–148. doi:10.1007/BF00239996. PMID 24197010. S2CID 43504838.
- ^ Hoberg, E.; Meier, B.; Sticher, O. (September 2000). "An analytical high performance liquid chromatographic method for the determination of agnuside and p-hydroxybenzoic acid contents in Agni-casti fructose". Phytochemical Analysis. 11 (5): 327–329. Bibcode:2000PChAn..11..327H. doi:10.1002/1099-1565(200009/10)11:5<327::AID-PCA523>3.0.CO;2-0.
- ^ Acosta, Manuel Jesús; Vazquez Fonseca, Luis; Desbats, Maria Andrea; Cerqua, Cristina; Zordan, Roberta; Trevisson, Eva; Salviati, Leonardo (2016). "Coenzyme Q biosynthesis in health and disease". Biochimica et Biophysica Acta (BBA) - Bioenergetics. 1857 (8): 1079–1085. doi:10.1016/j.bbabio.2016.03.036. PMID 27060254.
- ^ Edwin Ritzer and Rudolf Sundermann "Hydroxycarboxylic Acids, Aromatic" in Ullmann's Encyclopedia of Industrial Chemistry 2002, Wiley-VCH, Weinheim. doi:10.1002/14356007.a13_519
- ^ Buehler, C. A.; Cate, W. E. (1943). "p-Hydroxybenzoic acid". Organic Syntheses; Collected Volumes, vol. 2, p. 341.
- ^ Charles Fishman, Dan Winters (2016-04-11). "This Expandable Structure Could Become the Future of Living in Space". Smithsonian Magazine. Retrieved 2020-12-07.
- ^ Lewis, R. J., ed. (1996). Sax's Dangerous Properties of Industrial Materials. Vol. 1–3 (9th ed.). New York, NY: Van Nostrand Reinhold. p. 2897.
- ^ a b Khetan, S. K. (23 May 2014). Endocrine Disruptors in the Environment. Wiley. p. 109. ISBN 978-1-118-89115-5.
- ^ a b c Pugazhendhi, D.; Pope, G. S.; Darbre, P. D. (2005). "Oestrogenic activity of p-hydroxybenzoic acid (common metabolite of paraben esters) and methylparaben in human breast cancer cell lines". Journal of Applied Toxicology. 25 (4): 301–309. doi:10.1002/jat.1066. PMID 16021681. S2CID 12342018.
- ^ a b Gabriel, J. (April 2013). Holistic Beauty from the Inside Out: Your Complete Guide to Natural Health, Nutrition, and Skincare. Seven Stories Press. p. 31. ISBN 978-1-60980-462-6.
- ^ a b Lemini, C.; Silva, G.; Timossi, C.; Luque, D.; Valverde, A.; González Martínez, M.; Hernández, A.; Rubio Póo, C.; Chávez Lara, B.; Valenzuela, F. (1997). "Estrogenic effects of p-hydroxybenzoic acid in CD1 mice". Environmental Research. 75 (2): 130–134. Bibcode:1997ER.....75..130L. doi:10.1006/enrs.1997.3782. PMID 9417843.
- ^ OECD (November 2004). OECD Guidelines for the Testing of Chemicals / OECD Series on Testing and Assessment Detailed Background Review of the Uterotrophic Bioassay. OECD Publishing. p. 183. ISBN 978-92-64-07885-7.
- ^ Shi, G; Miller, C; Kuno, S; Rey Hipolito, AG; El Nagar, S; Riboldi, GM; Korn, M; Tran, WC; Wang, Z; Ficaro, L; Lin, T; Spillier, Q; Gamallo-Lana, B; Jones, DR; Snuderl, M; Song, SC; Mar, AC; Joyner, AL; Sillitoe, RV; Banh, RS; Pacold, ME. "Coenzyme Q headgroup intermediates can ameliorate a mitochondrial encephalopathy". Nature. doi:10.1038/s41586-025-09246-x. PMID 40634618.
External links
[edit]4-Hydroxybenzoic acid
View on Grokipedia4-Hydroxybenzoic acid is a monohydroxy derivative of benzoic acid characterized by a hydroxy group at the 4-position of the benzene ring, with the molecular formula C₇H₆O₃ and a molecular weight of 138.12 g/mol.[1] It appears as a white, crystalline solid with a melting point of 214.5 °C and low solubility in water (approximately 0.5 g/100 mL at room temperature).[1] The compound's structure enables it to participate in hydrogen bonding, contributing to its physical properties and reactivity as both a weak acid (pKa ≈ 4.54 for the carboxylic group) and a phenolic moiety.[1] In industrial applications, 4-hydroxybenzoic acid serves primarily as the precursor for paraben esters, which function as effective antimicrobial preservatives in cosmetics, pharmaceuticals, and food products due to their broad-spectrum activity against bacteria and fungi.[1] It is also employed in the synthesis of liquid crystal polymers and polyesters, enhancing material properties such as thermal stability and rigidity in applications ranging from electronics to textiles.[1] Additionally, the compound acts as an intermediate for producing dyes, fungicides, and antioxidants, leveraging its aromatic framework for further derivatization.[2] Biochemically, 4-hydroxybenzoic acid occurs as a natural metabolite in microbial catabolic pathways, where it is degraded via protocatechuate or gentisate routes, and has demonstrated antioxidant, anti-inflammatory, and antimicrobial effects in various studies, though its direct physiological roles in higher organisms remain limited.[3] Synthetic production often involves microbial fermentation from renewable feedstocks like glucose, offering sustainable alternatives to traditional chemical synthesis methods.[4]
Chemical Properties
Molecular Structure and Physical Characteristics
4-Hydroxybenzoic acid possesses the molecular formula C₇H₆O₃ and a structure derived from benzoic acid, featuring a hydroxyl group attached to the benzene ring at the para position relative to the carboxylic acid substituent. The molecule's planarity arises from the sp²-hybridized carbon atoms in the aromatic ring, with bond angles approximately 120°, and it exhibits polarity due to the dipole moments of the electron-withdrawing -COOH and hydrogen-bond donor/acceptor -OH groups, facilitating intermolecular hydrogen bonding in the solid state. [5] As a white crystalline solid with a molar mass of 138.12 g/mol, it has a density of 1.46 g/cm³. [6] Its melting point ranges from 214 to 217 °C, while the boiling point is approximately 334 °C at standard pressure, though decomposition may occur prior to boiling.[6] [3] The compound demonstrates limited solubility in water, approximately 5 g/L at 20 °C, attributable to hydrogen bonding with water molecules despite the hydrophobic aromatic core.[6] It possesses two ionizable protons: the carboxylic acid with pKₐ 4.54 and the phenolic hydroxyl with pKₐ approximately 9.4, influencing its acidity and behavior in aqueous environments. [7]| Property | Value |
|---|---|
| Molecular formula | C₇H₆O₃ |
| Molar mass | 138.12 g/mol |
| Appearance | White crystalline solid |
| Melting point | 214–217 °C |
| Boiling point | ~334 °C |
| Water solubility (20 °C) | ~5 g/L |
| pKₐ (carboxylic) | 4.54 |
| pKₐ (phenolic) | ~9.4 |
Reactivity and Derivatives
4-Hydroxybenzoic acid undergoes electrophilic aromatic substitution reactions, directed primarily by the strongly activating hydroxyl group at the para position relative to the carboxylic acid, favoring ortho positions (3 and 5) on the benzene ring despite the deactivating, meta-directing influence of the -COOH group.[11][12] Halogenation, such as bromination, occurs under these conditions, with the hydroxyl directing electrophile addition.[12] The carboxylic acid functionality readily undergoes esterification with alcohols in the presence of acid catalysts, yielding alkyl esters known as parabens, such as methylparaben (methyl 4-hydroxybenzoate), ethylparaben, propylparaben, and butylparaben, which exhibit antimicrobial properties.[13][6] These reactions typically proceed via Fischer esterification, where the acid is activated by protonation, facilitating nucleophilic attack by the alcohol.[13] Decarboxylation of 4-hydroxybenzoic acid produces phenol, often requiring catalytic conditions such as palladium complexes or high temperatures; for instance, electron-rich Pd catalysts enable selective decarboxylation of hydroxybenzoic acids to substituted phenols.[14][15] This process involves beta-keto acid-like tautomerism facilitated by the ortho relationship in the ionized form, though para substitution alters the kinetics compared to the ortho isomer salicylic acid.[14] Under oxidative conditions, such as Fenton's reagent (Fe²⁺/H₂O₂), 4-hydroxybenzoic acid generates quinone-like byproducts that participate in redox cycling, enhancing iron reduction and radical propagation, though direct quinone formation requires strong oxidants or enzymatic catalysis like tyrosinase.[16][17] The compound exhibits hydrolytic stability across pH 4–9 at 25°C, resisting decomposition in neutral to mildly acidic or basic aqueous environments.[18]| Common Paraben Derivatives | Formula | Application Note |
|---|---|---|
| Methylparaben | C₈H₈O₃ | Preservative in cosmetics[13] |
| Ethylparaben | C₉H₁₀O₃ | Antimicrobial agent[13] |
| Propylparaben | C₁₀H₁₂O₃ | Used in pharmaceuticals[13] |
| Butylparaben | C₁₁H₁₄O₃ | Food and cosmetic preservative[13] |
Natural Occurrence
In Plants and Foods
4-Hydroxybenzoic acid occurs naturally in numerous plant-based foods, where it acts as a phenolic compound aiding in antioxidant protection and defense against environmental stresses such as pathogens and UV radiation. Concentrations vary by source and form, often present as free acid, glycosides, or esters; for instance, in berries like blackberries, total hydroxybenzoic acids range from 8 to 27 mg per 100 g fresh weight, with 4-hydroxybenzoic acid contributing notably after hydrolysis.[19] In spices from the Apiaceae family, such as anise, p-hydroxybenzoic acid-O-glucoside levels attain 730–1080 mg/kg fresh weight, representing a substantial reservoir of the compound.[20] Specific examples include green tea leaves (Camellia sinensis), containing approximately 6.6 mg/kg of 4-hydroxybenzoic acid, and grapes (Vitis vinifera), where it is identified in the berry's solid components, contributing to the phenolic profile transferred to wine.[21] [22] Traces appear in honey and red wines, though at lower levels compared to berries and spices; in red wines, total hydroxybenzoic acids may reach up to 218 mg/L, with 4-hydroxybenzoic acid as a component.[23] Unlike synthetic variants employed industrially, naturally occurring forms in plants are predominantly conjugated, influencing bioavailability upon consumption.[20] Estimated dietary intake of hydroxybenzoic acids, encompassing 4-hydroxybenzoic acid from these sources, averages around 11 mg per day in populations with typical European diets, varying with fruit, vegetable, and spice consumption.[24]Biosynthesis
Pathways in Organisms
In bacteria such as Escherichia coli and other proteobacteria, 4-hydroxybenzoic acid is biosynthesized from chorismate, an intermediate of the shikimate pathway, via the enzyme chorismate pyruvate-lyase (encoded by ubiC), which catalyzes the elimination of pyruvate to form 4-hydroxybenzoic acid as part of ubiquinone (coenzyme Q) biosynthesis.[25][26] This reaction proceeds without coenzyme A involvement, distinguishing it from certain eukaryotic routes, and is regulated by feedback mechanisms tied to cellular quinone demands.[27] In the phytopathogen Xanthomonas campestris, genes encoding enzymes for 4-hydroxybenzoic acid synthesis are clustered with those for its transport and utilization, forming a coordinated operon-like structure that supports both endogenous production and scavenging from the environment, as identified in genomic analyses from 2015.[28] Similar genetic organization occurs in other gamma-proteobacteria like Lysobacter enzymogenes, where 4-hydroxybenzoic acid links shikimate flux to secondary metabolite pathways, such as antifungal heat-stable antifungals, via diffusible signaling.[29] Plants employ a CoA-dependent pathway for 4-hydroxybenzoic acid production, diverging from the bacterial lyase mechanism; chorismate is first converted to isochorismate or routed through tyrosine-derived intermediates like 4-hydroxyphenylpyruvate within the shikimate network, followed by ligation to coenzyme A and subsequent decarboxylation or oxidation steps to yield the free acid for ubiquinone or phenolic precursor roles.[30] Anthranilate-derived routes have been proposed in some species but lack widespread enzymatic confirmation, contrasting with the direct lyase in microbes.[31] Across species, pathway efficiency varies due to enzyme kinetics and precursor availability; for instance, Pseudomonas species primarily utilize the ubiC-like lyase but can incorporate tyrosine catabolism under nutrient stress, enhancing flux.[30] In biotechnological applications, engineering microbes like Corynebacterium glutamicum by overexpressing shikimate pathway genes (aro cluster) and a resistant ubiC variant achieves titers exceeding 10 g/L from glucose, demonstrating scalable variations not native to wild-type organisms.[32][27]Industrial Production
Synthetic Methods
The principal industrial synthesis of 4-hydroxybenzoic acid employs the Kolbe-Schmitt carboxylation, involving the reaction of potassium phenoxide with carbon dioxide at elevated temperatures (typically 150–200 °C) and pressures (up to 100 atm), followed by acidification to yield the product alongside the ortho isomer (salicylic acid), which is separated via fractional crystallization or distillation.[33][34] This method, commercialized since the 1870s, achieves overall yields of approximately 70–90% after purification, leveraging inexpensive phenol and CO2 feedstocks for economic viability on multi-ton scales.[35][36] Alternative chemical routes include the oxidation of p-cresol using molecular oxygen or air in the presence of cobalt-based catalysts (e.g., CoCl2 or Co3O4) under alkaline conditions, which can proceed stepwise via p-hydroxybenzaldehyde to the carboxylic acid, though selectivity and catalyst recovery pose challenges for large-scale adoption.[37][38] Another approach entails hydrolysis of esters such as methyl 4-hydroxybenzoate, sourced from electrophilic aromatic substitution on phenol followed by esterification and saponification, offering high purity (>99%) but higher costs due to multi-step processing and byproduct management.[39] Commercial products from these chemical methods routinely meet purity standards exceeding 99%, with scalability favored by the Kolbe-Schmitt process owing to its direct carboxylation and established infrastructure.[40] Emerging bio-based alternatives utilize metabolic engineering of microorganisms, such as Pseudomonas taiwanensis VLB120, to ferment renewable feedstocks like glucose, xylose, or glycerol into 4-hydroxybenzoic acid via shikimate pathway overexpression and directed evolution, attaining titers up to 10 g/L and molar yields around 20–30% in fed-batch processes as reported in 2021 studies.[41][27] These microbial routes prioritize sustainability by avoiding petrochemical inputs but lag in economic competitiveness due to lower productivity (e.g., 0.1–0.5 g/L/h), higher purification demands, and unproven industrial scalability relative to chemical synthesis.[42]Metabolism and Biodegradation
In Human and Animal Systems
In mammals, 4-hydroxybenzoic acid is rapidly absorbed following oral administration, with predicted human intestinal absorption exceeding 98% based on in silico models validated against experimental data.[43] Distribution is primarily to plasma and tissues, facilitated by its moderate lipophilicity (log Kow ≈1.6), though it does not readily cross the blood-brain barrier.[43] In hepatic tissues, it undergoes phase II conjugation, predominantly to glucuronides and sulfates, which enhance water solubility for elimination; this process mirrors the metabolism of related phenolic acids.[44] Excretion occurs mainly via urine, with over 70% of an oral dose recovered as conjugated metabolites within 24 hours in rodent models, indicating efficient clearance. The plasma half-life is short, typically 1–2 hours for phenolic acids like 4-hydroxybenzoic acid in humans, reflecting rapid biotransformation and renal elimination.[45] A 2019 intervention study in humans showed significantly elevated urinary levels of 4-hydroxybenzoic acid following consumption of an organic diet (>80% organic products for 4 days), with excretion increasing up to 4-fold, attributed to higher dietary phenolic precursors from plant sources.[46] In animal systems, pharmacokinetics align with human patterns, featuring quick absorption and conjugation; for instance, in mice, oral dosing yields an LD50 of 2200 mg/kg, consistent with low systemic retention due to metabolic efficiency.[1] The compound serves as a minor intermediate in gut microbial catabolism of tyrosine or lignin-derived aromatics in ruminants, where ruminal bacteria convert complex phenolics to 4-hydroxybenzoic acid before host absorption and further processing.[47] No bioaccumulation occurs in mammals, owing to its polarity, short half-life, and conversion to polar conjugates that preclude tissue partitioning.[18]Microbial and Environmental Degradation
Bacteria such as Pseudomonas and Acinetobacter species degrade 4-hydroxybenzoic acid primarily through the protocatechuate pathway, initiating with hydroxylation to protocatechuate via 4-hydroxybenzoate 3-hydroxylase, followed by extradiol ring cleavage by protocatechuate 3,4-dioxygenase, and subsequent funneling into central metabolism for complete mineralization to CO₂ and H₂O.[48][49] This process enables aerobic soil isolates like Acinetobacter johnsonii FZ-5 to utilize 4-hydroxybenzoic acid as a sole carbon source, achieving substantial breakdown under both aerobic and anaerobic conditions.[48] Specific strains demonstrate high efficiency; Herbaspirillum aquaticum KLS-1, isolated from tailing soil, degrades p-hydroxybenzoic acid (synonymous with 4-hydroxybenzoic acid) via protocatechuate ortho-cleavage, with optimal rates at pH 6.0–8.0, 30–35 °C, and 180 rpm shaking, supporting energy acquisition and removing over 90% within days under favorable conditions.[50][51] Similarly, Pseudarthrobacter phenanthrenivorans Sphe3 employs versatile catabolic routes for 4-hydroxybenzoic acid, including meta- and ortho-cleavage variants, allowing growth on it as the sole carbon source and >90% degradation in short-term cultures as detailed in 2024 analyses of growth profiles and metabolites.[48] In plant-pathogen contexts, benzoic acid competitively inhibits 4-hydroxybenzoic acid degradation in Xanthomonas campestris, reducing virulence by blocking uptake and catabolism, as observed in cabbage host interactions in 2024 studies.[52] Fungi contribute to degradation, with endophytic Phomopsis liquidambari B3 capable of metabolizing 4-hydroxybenzoic acid through similar aromatic ring-opening mechanisms, and white-rot fungi like those funneling lignin-derived aromatics exhibiting enzyme-mediated conversion to central metabolites.[53][54] 4-Hydroxybenzoic acid exhibits ready biodegradability per OECD 301 guidelines, with 100% degradation in 28 days under aerobic conditions, reflecting complete microbial mineralization.[18] In environmental matrices, aerobic half-lives in soil and aquifer water are short, typically under one week, with 34–70% mineralization reported in sediments within 6 days.[1][55]Applications
Preservative and Antimicrobial Uses
Ester derivatives of 4-hydroxybenzoic acid, known as parabens (e.g., methylparaben, propylparaben), are employed as broad-spectrum preservatives in cosmetics, pharmaceuticals, and certain food products at concentrations typically ranging from 0.1% to 0.4% for individual esters, with mixtures limited to 0.8% to inhibit microbial growth and extend shelf life.[56][57][58] These compounds demonstrate efficacy against Gram-positive bacteria, Gram-negative bacteria such as Escherichia coli (with minimum inhibitory concentrations often 200–1000 ppm at pH 6), and fungi, achieving substantial reductions in microbial load through disruption of cell membrane transport processes and integrity.[59][60][61] The antimicrobial action of parabens operates via partitioning into lipid membranes, altering fluidity and inhibiting energy-dependent transport, which is more pronounced in their undissociated form prevalent at pH 4–8.[60][62] This pH range provides an advantage over alternatives like sorbic acid, which exhibits optimal activity at lower pH (3.0–6.5) and reduced efficacy in neutral or alkaline formulations.[62][63] Parabens were first utilized as preservatives in the 1920s, following demonstrations of their inhibitory effects by researchers like Theodor Sabalitschka, and have since been integral to preventing spoilage in water-based products.[64][65] Additionally, 4-hydroxybenzoic acid itself exhibits antioxidant properties suitable for stabilizing food and beverage formulations against oxidative degradation.[66][67]Other Industrial Applications
4-Hydroxybenzoic acid functions as a chemical intermediate in the manufacture of liquid crystal polymers, where it contributes to the structural properties enabling applications in displays and optical films.[1] It is also incorporated into polyester resins, enhancing thermal stability and mechanical performance in industrial polymers.[1] In the dyes sector, the compound serves as a precursor for diazo pigments and oil-soluble azo pigments, which are employed in printing inks, textiles, and coatings for colorfastness.[68] Thermosensitive dye developers derived from 4-hydroxybenzoic acid find use in thermal paper production for receipts and labels.[69] For agrochemicals, it acts as an intermediate in synthesizing fungicides and organophosphorus insecticides, supporting crop protection formulations.[18] In pharmaceuticals, derivatives are utilized in antiseptics and other drug intermediates, leveraging its phenolic structure for targeted synthesis.[18] Additionally, it contributes to ultraviolet absorbers in coatings and plastics, providing photostability against UV degradation.[68] Other roles include corrosion inhibition in industrial fluids and emulsification in formulations, owing to its solubility and surface-active properties.[1] Global market analyses indicate production supports diverse sectors, with estimated values around USD 80-150 million annually as of recent years, reflecting steady demand for these non-preservative applications.[70]Toxicology and Safety
Biological Activity and Health Effects
4-Hydroxybenzoic acid (4-HBA) demonstrates in vitro antioxidant activity primarily through its phenolic hydroxyl group, which facilitates hydrogen atom donation to neutralize free radicals such as peroxyl species. Studies report its oxygen radical absorbance capacity (ORAC) comparable to other hydroxybenzoic acids, with values reflecting moderate peroxyl radical scavenging efficiency relative to trolox equivalents.[71][72] This bioactivity contributes to reducing oxidative stress in cellular models, though structure-activity analyses indicate lower potency than multi-hydroxylated analogs like protocatechuic acid.[73] In terms of anti-inflammatory and anti-allergic effects, 4-HBA inhibits NLRP3 inflammasome priming and activation, thereby attenuating cytokine release and systemic inflammation in lipopolysaccharide-challenged models. A 2025 murine study on allergic asthma further showed that hydroxybenzoic acid suppresses eosinophil-driven inflammation and interleukin-5 production, highlighting selective modulation of allergic responses without broad immunosuppression. These effects occur at concentrations achievable via dietary or supplemental intake, supporting potential roles in mitigating inflammation-related conditions.[74][75] 4-HBA exhibits antimicrobial activity against gram-positive bacteria, including pathogens like Staphylococcus species, with minimum inhibitory concentrations in the range of those for related phenolic acids; this is attributed to membrane disruption and enzyme inhibition. In food preservation contexts, such activity enhances safety by inhibiting microbial growth without requiring esterification to parabens. Human and animal exposure data indicate low acute toxicity, with no observed adverse effects in repeated oral dosing up to 1000 mg/kg/day in rodents, corresponding to substantial margins over typical dietary levels.[76][77] However, topical application can induce mild skin sensitization in susceptible individuals, classified as a weak dermal sensitizer due to potential hapten formation with skin proteins, though absorption is limited and irritation is generally minimal at concentrations below 1%. Dose-response evidence from animal studies confirms safety at high systemic doses but underscores the need for caution in prolonged cutaneous exposure. Overall, while 4-HBA's bioactivities offer benefits for antioxidant defense and microbial control, its health effects are context-dependent, with benefits outweighing risks at low environmental or dietary exposures.[78]Regulatory Status and Risk Assessments
The U.S. Food and Drug Administration (FDA) approves 4-hydroxybenzoic acid for use as a food preservative at concentrations up to 0.1%, reflecting its established safety profile in direct food contact applications.[1] It is also affirmed for use as a flavoring agent or adjuvant under FDA listings for substances added to food, with no requirement for pre-market approval beyond these parameters due to its low toxicity. In the European Union, 4-hydroxybenzoic acid and its salts and esters are regulated as permitted preservatives in cosmetics under Annex V of Regulation (EC) No 1223/2009, with maximum concentrations of 0.4% for a single ester or 0.8% for mixtures of esters, as amended by Commission Regulation (EU) No 1004/2014.[79] These limits are based on toxicological data indicating minimal risk at approved levels, including rapid hydrolysis to the parent acid, which undergoes complete metabolism and excretion without significant accumulation.[80] The Joint FAO/WHO Expert Committee on Food Additives (JECFA) evaluated 4-hydroxybenzoic acid in 2001 and concluded no safety concern at current levels of intake when used as a flavoring agent, supported by acute oral LD50 values exceeding 2,000 mg/kg body weight in rats and mice.[81][82] OECD Screening Information Data Set (SIDS) assessments similarly classify it as having low acute toxicity, with no observed adverse effects in repeated oral dosing studies at or above 1,000 mg/kg body weight in rats (NOAEL ≥1,000 mg/kg), and no evidence of carcinogenicity in rodent models.[82][83] Chronic exposure studies confirm no reproductive or developmental toxicity below 500 mg/kg body weight, aligning with regulatory thresholds that prioritize empirical no-effect levels over speculative risks.[84]Environmental Impact
Fate in the Environment
4-Hydroxybenzoic acid exhibits low volatility, with an extrapolated vapor pressure of 1.9 × 10^{-7} mm Hg at 25 °C, limiting its partitioning into the atmosphere and aerial transport.[1] Its moderate water solubility of approximately 6 g/L at 25 °C facilitates dissolution in aqueous environments, while the low octanol-water partition coefficient (log K_{ow} ≈ 1.5) indicates minimal sorption to organic matter in sediments or soils.[18] [1] In aquatic and soil systems, 4-hydroxybenzoic acid undergoes rapid aerobic biodegradation, achieving complete mineralization within 28 days under standardized conditions (OECD 301C), with half-lives typically under 10 days in adapted environments.[18] [1] This contrasts with more persistent paraben esters, as the free acid form supports faster microbial uptake and transformation, reducing overall environmental persistence.[55] The compound shows no significant bioaccumulation potential due to its low log K_{ow}, with predicted bioconcentration factors below levels of concern.[18] Runoff from industrial or agricultural sites is minimal, as rapid metabolic degradation in receiving waters and soils limits long-range transport.[1]Ecotoxicological Effects
4-Hydroxybenzoic acid demonstrates low acute toxicity to representative aquatic species across trophic levels. The 96-hour LC50 for the fish Oryzias latipes is 92.8 mg/L, while the 14-day LC50 is 66.5 mg/L. For the crustacean Daphnia magna, the 48-hour EC50 for immobilization is 135.7 mg/L. Algal growth inhibition, measured as the 72-hour EC50 for Selenastrum capricornutum, is 68.5 mg/L.[18][82] Chronic exposure yields similarly low effect levels. The no-observed-effect concentration (NOEC) for algal growth is 32.0 mg/L over 72 hours, and for Daphnia magna reproduction, the 21-day NOEC exceeds 100 mg/L.[18] These values, all exceeding typical thresholds for high toxicity (e.g., <10 mg/L), support assessments of minimal risk to aquatic populations at predicted environmental concentrations.[82]| Organism | Endpoint | Value (mg/L) | Duration | Reference |
|---|---|---|---|---|
| Oryzias latipes (fish) | LC50 | 92.8 | 96 hours | [18] |
| Daphnia magna | EC50 | 135.7 | 48 hours | [18] |
| Selenastrum capricornutum (algae) | EC50 | 68.5 | 72 hours | [18] |
| Selenastrum capricornutum (algae) | NOEC | 32.0 | 72 hours | [18] |
| Daphnia magna | NOEC (reproduction) | >100 | 21 days | [18] |


