Recent from talks
Nothing was collected or created yet.
Tin(IV) oxide
View on Wikipedia
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Tin (IV) Oxide
| |
| Other names
Stannic oxide, Tin(IV) oxide, Flowers of tin,[1] Cassiterite
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.038.311 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| O2Sn | |
| Molar mass | 150.708 g·mol−1 |
| Appearance | Yellowish or light grey powder[2] |
| Odor | Odorless |
| Density | 6.95 g/cm3 (20 °C)[3] 6.85 g/cm3 (24 °C)[4] |
| Melting point | 1,630 °C (2,970 °F; 1,900 K)[3][4] |
| Boiling point | 1,800–1,900 °C (3,270–3,450 °F; 2,070–2,170 K) Sublimes[3] |
| Insoluble[4] | |
| Solubility | Soluble in hot concentrated alkalis,[4] concentrated acids Insoluble in alcohol[3] |
| −4.1·10−5 cm3/mol[4] | |
Refractive index (nD)
|
2.006[5] |
| Structure | |
| Rutile tetragonal, tP6[6] | |
| P42/mnm, No. 136[6] | |
| 4/m 2/m 2/m[6] | |
a = 4.737 Å, c = 3.185 Å[6] α = 90°, β = 90°, γ = 90°
| |
| Octahedral (Sn4+) Trigonal planar (O2−) | |
| Thermochemistry | |
Heat capacity (C)
|
52.6 J/mol·K[4] |
Std molar
entropy (S⦵298) |
49.04 J/mol·K[4][7] |
Std enthalpy of
formation (ΔfH⦵298) |
−577.63 kJ/mol[4][7] |
Gibbs free energy (ΔfG⦵)
|
−515.8 kJ/mol[4] |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
> 20 g/kg (rats, oral)[8] |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
none[2] |
REL (Recommended)
|
TWA 2 mg/m3[2] |
IDLH (Immediate danger)
|
N.D.[2] |
| Safety data sheet (SDS) | ICSC 0954 |
| Related compounds | |
| Tin(II) oxide | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Tin(IV) oxide, also known as stannic oxide, is the inorganic compound with the formula SnO2. The mineral form of SnO2 is called cassiterite, and this is the main ore of tin.[9] With many other names, this oxide of tin is an important material in tin chemistry. It is a colourless, diamagnetic, amphoteric solid.
Structure
[edit]
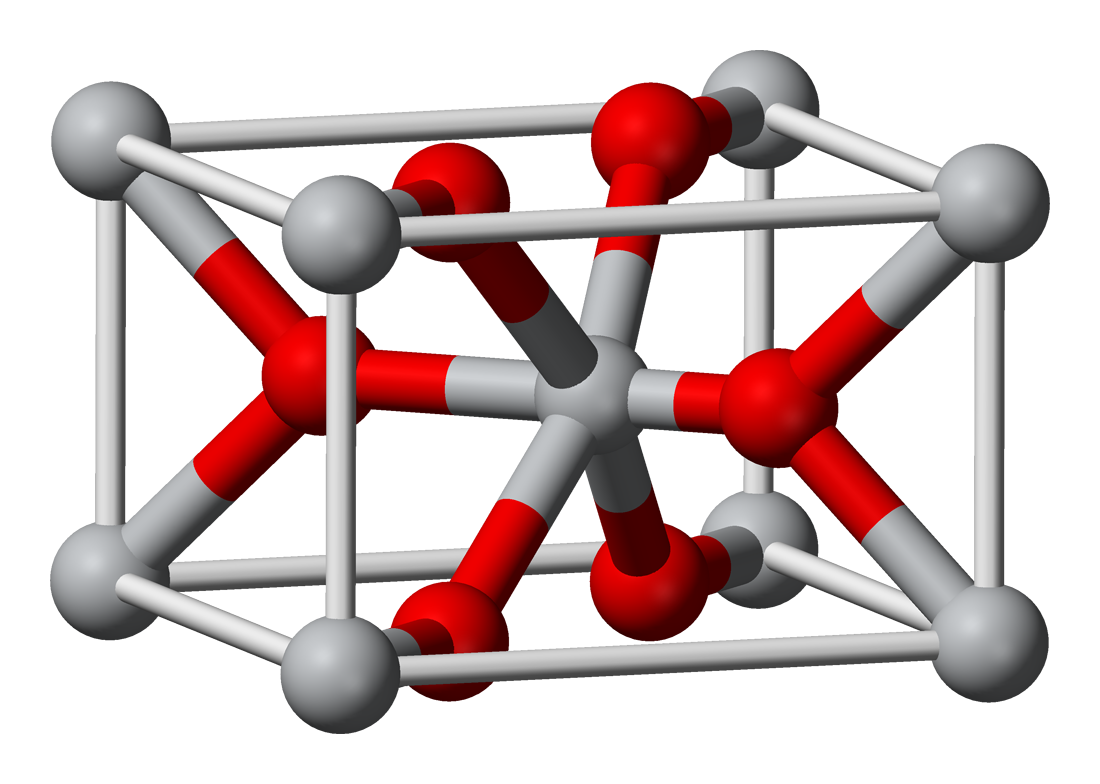
Tin(IV) oxide crystallises with the rutile structure. As such the tin atoms are six coordinate and the oxygen atoms three coordinate.[9] SnO2 is usually regarded as an oxygen-deficient n-type semiconductor.[10]
Hydrous forms of SnO2 have been described as stannic acid. Such materials appear to be hydrated particles of SnO2 where the composition reflects the particle size.[11]
Preparation
[edit]Tin(IV) oxide occurs naturally. Synthetic tin(IV) oxide is produced by burning tin metal in air.[11] Annual production is in the range of 10 kilotons.[11] SnO2 is reduced industrially to the metal with carbon in a reverberatory furnace at 1200–1300 °C.[12]
Reactions
[edit]The reaction from tin(IV) oxide with hot carbon monoxide is practiced on a large scale as this carbothermal reduction is used to obtain tin metal from its ores:
- SnO2 + 2 CO → Sn + 2 CO2
Some other reactions relevant to purifying tin from its ores are:[13]
- SnO2 + MgCl2 + CO → SnCl2 + MgO +CO2
- 4 SnO2 + 6 FeCl2 → 2 SnCl2 + 2 SnCl4 + 2 Fe3O4
SnO2 converts to the monoxide at 1500 °C:[13]
- 2 SnO2 → 2 SnO + O2
SnO2 is insoluble in water. It dissolves in sulfuric acid and in molten sodium hydroxide. It is not amphoteric. Like rutile, it is not attacked by solutions of acid or base.
Dissolution of SnO2 in sulfuric acid gives the sulfate:[11]
- SnO2 + 2 H2SO4 → Sn(SO4)2 + 2 H2O
The latter compound can add additional hydrogen sulfate ligands to give hexahydrogensulfatostannic acid.[14]
SnO2 dissolves in molten alkali to give "stannates," with the nominal formula Na2SnO3.[11] Dissolving the solidified SnO2/NaOH melt in water gives Na2[Sn(OH)6], "preparing salt," which is used in the dye industry.[11]
Uses
[edit]In conjunction with vanadium oxide, it is used as a catalyst for the oxidation of aromatic compounds in the synthesis of carboxylic acids and acid anhydrides.[9]
Ceramic glazes
[edit]SnO2 is used as pigment in the manufacture of glasses, enamels and ceramic glazes. Thousands of tons of SnO2 are produced annually for this application. Pure SnO2 gives a milky white colour; other colours are achieved when mixed with other metallic oxides e.g. V2O5 yellow; Cr2O3 pink; and Sb2O5 grey blue.[11][15] This use probably led to the discovery of the pigment lead-tin-yellow, which was produced using tin(IV) oxide as a compound.[16] The use of tin(IV) oxide has been particularly common in glazes for earthenware, sanitaryware and wall tiles; see the articles tin-glazing and Tin-glazed pottery. Tin oxide remains in suspension in vitreous matrix of the fired glazes, and, with its high refractive index being sufficiently different from the matrix, light is scattered, and hence increases the opacity of the glaze. The degree of dissolution increases with the firing temperature, and hence the extent of opacity diminishes.[17] Although dependent on the other constituents the solubility of tin oxide in glaze melts is generally low. Its solubility is increased by Na2O, K2O and B2O3, and reduced by CaO, BaO, ZnO, Al2O3, and to a limited extent PbO.[18]
Glass coatings
[edit]SnO2 coatings are valued as transparent conducting oxides (TCOs). Like other TCOs, SnO2 has significant electrical conductivity but is transparent, an unusual combination of properties. Windows coated with SnO2 also reflect infrared radiation, which is relevant to temperature control for smart windows.[19] Coatings can be applied using chemical vapor deposition, vapour deposition techniques that employ SnCl4[9] or organotin trihalides[20] e.g. butyltin trichloride as the volatile agent. This technique is used to coat glass bottles with a thin (<0.1 μm) layer of SnO2, which helps to adhere a subsequent, protective polymer coating such as polyethylene to the glass.[9]
Thicker layers doped with Sb or F ions are electrically conducting and used in electroluminescent devices and photovoltaics.[9]
Gas sensing
[edit]SnO2 has been evaluated as sensors of combustible gases including carbon monoxide detectors. In these the sensor area is heated to a constant temperature (few hundred °C) and in the presence of a combustible gas the electrical resistivity drops.[21]
Historical uses
[edit]This oxide of tin has been utilized as a mordant in the dyeing process since ancient Egypt.[22] A German by the name of Kuster first introduced its use to London in 1533 and by means of it alone, the color scarlet was produced there.[23]
Tin(IV) oxide for this use is sometimes called as "putty powder"[24] or "jeweler's putty".[1]
Polishing
[edit]Tin(IV) oxide can be used as a polishing powder,[11] sometimes in mixtures also with lead oxide, for polishing glass, jewelry, marble and silver.[1]
Further reading
[edit]- Wang, Chun-Ming; Wang, Jin-Feng; Su, Wen-Bin (2006). "Microstructural Morphology and Electrical Properties of Copper- and Niobium-Doped Tin (IV) oxide Polycrystalline Varistors". Journal of the American Ceramic Society. 89 (8): 2502–2508. doi:10.1111/j.1551-2916.2006.01076.x.[1]
- Dibb A.; Cilense M; Bueno P.R; Maniette Y.; Varela J.A.; Longo E. (2006). "Evaluation of Rare Earth Oxides doping SnO2.(Co0.25,Mn0.75)O-based Varistor System". Materials Research. 9 (3): 339–343. doi:10.1590/S1516-14392006000300015. hdl:11449/30580.
- A. Punnoose; J. Hays; A. Thurber; M. H. Engelhard; R. K. Kukkadapu; C. Wang; V. Shutthanandan & S. Thevuthasan (2005). "Development of high-temperature ferromagnetism in SnO2 and paramagnetism in SnO by Fe doping". Phys. Rev. B. 72 (8) 054402. Bibcode:2005PhRvB..72e4402P. doi:10.1103/PhysRevB.72.054402.
References
[edit]- ^ a b c "Material Name: stannic oxide". Museum of Fine Arts, Boston. February 10, 2007. Archived from the original on November 4, 2012. Retrieved March 29, 2013.
- ^ a b c d NIOSH Pocket Guide to Chemical Hazards. "#0616". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b c d CID 29011 from PubChem
- ^ a b c d e f g h i Lide, David R., ed. (2009). CRC Handbook of Chemistry and Physics (90th ed.). Boca Raton, Florida: CRC Press. ISBN 978-1-4200-9084-0.
- ^ Pradyot, Patnaik (2003). Handbook of Inorganic Chemicals. The McGraw-Hill Companies, Inc. p. 940. ISBN 0-07-049439-8.
- ^ a b c d Baur, W.H. (1956). "Über die Verfeinerung der Kristallstrukturbestimmung einiger Vertreter des Rutiltyps: TiO2, SnO2, GeO2 und MgF2". Acta Crystallographica. 9 (6): 515–520. Bibcode:1956AcCry...9..515B. doi:10.1107/S0365110X56001388.
- ^ a b Stannic oxide in Linstrom, Peter J.; Mallard, William G. (eds.); NIST Chemistry WebBook, NIST Standard Reference Database Number 69, National Institute of Standards and Technology, Gaithersburg (MD) (retrieved July 4, 2014)
- ^ a b "MSDS of Tin(IV) oxide". fishersci.ca. Fisher Scientific. Retrieved July 4, 2014.
- ^ a b c d e f Greenwood, Norman N.; Earnshaw, Alan (1984). Chemistry of the Elements. Oxford: Pergamon Press. pp. 447–48. ISBN 978-0-08-022057-4.
- ^ Solid State Chemistry: An Introduction Lesley Smart, Elaine A. Moore (2005) CRC Press ISBN 0-7487-7516-1
- ^ a b c d e f g h Holleman, Arnold Frederik; Wiberg, Egon (2001), Wiberg, Nils (ed.), Inorganic Chemistry, translated by Eagleson, Mary; Brewer, William, San Diego/Berlin: Academic Press/De Gruyter, ISBN 0-12-352651-5
- ^ Tin: Inorganic chemistry, J L Wardell, Encyclopedia of Inorganic Chemistry ed R. Bruce King, John Wiley & Son Ltd., (1995) ISBN 0-471-93620-0
- ^ a b Graf, Günter G. (2000). "Tin, Tin Alloys, and Tin Compounds". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a27_049. ISBN 978-3-527-30385-4.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1984). Chemistry of the Elements. Oxford: Pergamon Press. p. 844. ISBN 978-0-08-022057-4.
- ^ 'The Glazer's Book' – 2nd edition. A.B.Searle.The Technical Press Limited. London. 1935.
- ^ Hermann Kühn, 1967, "Blei-Zinn-Gelb und seine Verwendung in der Malerei", Farbe und Lack 73: 938-949
- ^ 'A Treatise On Ceramic Industries.' E.Bourry. Fourth edition. Scott, Greenwood & son. London. 1926.
- ^ 'Ceramic Glazes' Third edition. C.W.Parmelee & C.G.Harman. Cahners Books, Boston, Massachusetts. 1973.
- ^ Batzill, Matthias; Diebold, Ulrike (2005). "The surface and materials science of tin oxide". Progress in Surface Science. 79 (2–4): 47–154. Bibcode:2005PrSS...79...47B. doi:10.1016/j.progsurf.2005.09.002.
- ^ US 4130673, Larkin, William A., "Process of applying tin oxide on glass using butyltin trichloride", published December 19, 1978, assigned to M & T Chemicals Inc.
- ^ Joseph Watson The stannic oxide semiconductor gas sensor in The Electrical engineering Handbook 3d Edition; Sensors Nanoscience Biomedical Engineering and Instruments ed R.C Dorf CRC Press Taylor and Francis ISBN 0-8493-7346-8
- ^ Sir Thomas Edward Thorpe History of Chemistry (1909) Vol. 1, pp. 11-12.
- ^ Thomas Mortimer, A General Dictionary of Commerce, Trade, and Manufactures (1810) "Dying or Dyeing"
- ^ Inorganic & Theoretical chemistry, F. Sherwood Taylor, Heineman, 6th Edition (1942)
Further reading
[edit]- "How Pilkington Energy Advantage™ Low-E Glass Works" (PDF). Pilkington Group Limited. July 18, 2005. Retrieved December 2, 2012.[permanent dead link] Technical discussion of how SnO2:F is used in low-emissivity (low-E) windows. The report includes reflectance and transmittance spectra.
- "NIOSH Pocket Guide to Chemical Hazards - Tin(IV) oxide (as Sn)". Centers for Disease Control and Prevention. April 4, 2011. Retrieved November 5, 2013. Information on chemical safety and exposure limits