Recent from talks
Nothing was collected or created yet.
Haloalkane
View on Wikipedia
The haloalkanes (also known as halogenoalkanes or alkyl halides) are alkanes containing one or more halogen substituents of hydrogen atom.[1] They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalkanes are widely used commercially. They are used as flame retardants, fire extinguishants, refrigerants, propellants, solvents, and pharmaceuticals. Subsequent to the widespread use in commerce, many halocarbons have also been shown to be serious pollutants and toxins. For example, the chlorofluorocarbons have been shown to lead to ozone depletion. Methyl bromide is a controversial fumigant. Only haloalkanes that contain chlorine, bromine, and iodine are a threat to the ozone layer, but fluorinated volatile haloalkanes in theory may have activity as greenhouse gases. Methyl iodide, a naturally occurring substance, however, does not have ozone-depleting properties and the United States Environmental Protection Agency has designated the compound a non-ozone layer depleter. For more information, see Halomethane. Haloalkane or alkyl halides are the compounds which have the general formula "RX" where R is an alkyl or substituted alkyl group and X is a halogen (F, Cl, Br, I).
Haloalkanes have been known for centuries. Chloroethane was produced in the 15th century. The systematic synthesis of such compounds developed in the 19th century in step with the development of organic chemistry and the understanding of the structure of alkanes. Methods were developed for the selective formation of C-halogen bonds. Especially versatile methods included the addition of halogens to alkenes, hydrohalogenation of alkenes, and the conversion of alcohols to alkyl halides. These methods are so reliable and so easily implemented that haloalkanes became cheaply available for use in industrial chemistry because the halide could be further replaced by other functional groups.
While many haloalkanes are human-produced, substantial amounts are biogenic.
Classes
[edit]From the structural perspective, haloalkanes can be classified according to the connectivity of the carbon atom to which the halogen is attached. In primary (1°) haloalkanes, the carbon that carries the halogen atom is only attached to one other alkyl group. An example is chloroethane (CH
3CH
2Cl). In secondary (2°) haloalkanes, the carbon that carries the halogen atom has two C–C bonds. In tertiary (3°) haloalkanes, the carbon that carries the halogen atom has three C–C bonds.[citation needed]
Haloalkanes can also be classified according to the type of halogen on group 17 responding to a specific halogenoalkane. Haloalkanes containing carbon bonded to fluorine, chlorine, bromine, and iodine results in organofluorine, organochlorine, organobromine and organoiodine compounds, respectively. Compounds containing more than one kind of halogen are also possible. Several classes of widely used haloalkanes are classified in this way chlorofluorocarbons (CFCs), hydrochlorofluorocarbons (HCFCs) and hydrofluorocarbons (HFCs). These abbreviations are particularly common in discussions of the environmental impact of haloalkanes.[citation needed]
Properties
[edit]Haloalkanes generally resemble the parent alkanes in being colorless, relatively odorless, and hydrophobic. The melting and boiling points of chloro-, bromo-, and iodoalkanes are higher than the analogous alkanes, scaling with the atomic weight and number of halides. This effect is due to the increased strength of the intermolecular forces—from London dispersion to dipole-dipole interaction because of the increased polarizability. Thus tetraiodomethane (CI
4) is a solid whereas tetrachloromethane (CCl
4) is a liquid. Many fluoroalkanes, however, go against this trend and have lower melting and boiling points than their nonfluorinated analogues due to the decreased polarizability of fluorine. For example, methane (CH
4) has a melting point of −182.5°C whereas tetrafluoromethane (CF
4) has a melting point of −183.6°C.[citation needed]
As they contain fewer C–H bonds, haloalkanes are less flammable than alkanes, and some are used in fire extinguishers. Haloalkanes are better solvents than the corresponding alkanes because of their increased polarity. Haloalkanes containing halogens other than fluorine are more reactive than the parent alkanes—it is this reactivity that is the basis of most controversies. Many are alkylating agents, with primary haloalkanes and those containing heavier halogens being the most active (fluoroalkanes do not act as alkylating agents under normal conditions). The ozone-depleting abilities of the CFCs arises from the photolability of the C–Cl bond.[citation needed]
Natural occurrence
[edit]An estimated 4,100,000,000 kg of chloromethane are produced annually by natural sources.[2] The oceans are estimated to release 1 to 2 million tons of bromomethane annually.[3]
Nomenclature
[edit]IUPAC
[edit]The formal naming of haloalkanes should follow IUPAC nomenclature, which put the halogen as a prefix to the alkane. For example, ethane with bromine becomes bromoethane, methane with four chlorine groups becomes tetrachloromethane. However, many of these compounds have already an established trivial name, which is endorsed by the IUPAC nomenclature, for example chloroform (trichloromethane) and methylene chloride (dichloromethane). But nowadays, IUPAC nomenclature is used. To reduce confusion this article follows the systematic naming scheme throughout.
Production
[edit]Haloalkanes can be produced from virtually all organic precursors. From the perspective of industry, the most important ones are alkanes and alkenes.
From alkanes
[edit]Alkanes react with halogens by free radical halogenation. In this reaction a hydrogen atom is removed from the alkane, then replaced by a halogen atom by reaction with a diatomic halogen molecule. Free radical halogenation typically produces a mixture of compounds mono- or multihalogenated at various positions.[citation needed]
From alkenes and alkynes
[edit]In hydrohalogenation, an alkene reacts with a dry hydrogen halide (HX) electrophile like hydrogen chloride (HCl) or hydrogen bromide (HBr) to form a mono-haloalkane. The double bond of the alkene is replaced by two new bonds, one with the halogen and one with the hydrogen atom of the hydrohalic acid. Markovnikov's rule states that under normal conditions, hydrogen is attached to the unsaturated carbon with the most hydrogen substituents. The rule is violated when neighboring functional groups polarize the multiple bond, or in certain additions of hydrogen bromide (addition in the presence of peroxides and the Wohl-Ziegler reaction) which occur by a free-radical mechanism.[citation needed]
Alkenes also react with halogens (X2) to form haloalkanes with two neighboring halogen atoms in a halogen addition reaction. Alkynes react similarly, forming the tetrahalo compounds. This is sometimes known as "decolorizing" the halogen, since the reagent X2 is colored and the product is usually colorless and odorless.[citation needed]
From alcohols
[edit]Alcohol can be converted to haloalkanes. Direct reaction with a hydrohalic acid rarely gives a pure product, instead generating ethers. However, some exceptions are known: ionic liquids suppress the formation or promote the cleavage of ethers,[4] hydrochloric acid converts tertiary alcohols to chloroalkanes, and primary and secondary alcohols convert similarly in the presence of a Lewis acid activator, such as zinc chloride. The latter is exploited in the Lucas test.[citation needed]
In the laboratory, more active deoxygenating and halogenating agents combine with base to effect the conversion. In the "Darzens halogenation", thionyl chloride (SOCl
2) with pyridine converts less reactive alcohols to chlorides. Both phosphorus pentachloride (PCl
5) and phosphorus trichloride (PCl
3) function similarly, and alcohols convert to bromoalkanes under hydrobromic acid or phosphorus tribromide (PBr3). The heavier halogens do not require preformed reagents: A catalytic amount of PBr
3 may be used for the transformation using phosphorus and bromine; PBr
3 is formed in situ.[5] Iodoalkanes may similarly be prepared using red phosphorus and iodine (equivalent to phosphorus triiodide).[citation needed]
One family of named reactions relies on the deoxygenating effect of triphenylphosphine. In the Appel reaction, the reagent is tetrahalomethane and triphenylphosphine; the co-products are haloform and triphenylphosphine oxide. In the Mitsunobu reaction, the reagents are any nucleophile, triphenylphosphine, and a diazodicarboxylate; the coproducts are triphenylphosphine oxide and a hydrazodiamide.[citation needed]
From carboxylic acids
[edit]Two methods for the synthesis of haloalkanes from carboxylic acids are Hunsdiecker reaction and Kochi reaction.[citation needed]
Biosynthesis
[edit]Many chloro- and bromoalkanes are formed naturally. The principal pathways involve the enzymes chloroperoxidase and bromoperoxidase.[citation needed]
From amines by Sandmeyer's Method
[edit]Primary aromatic amines yield diazonium ions in a solution of sodium nitrite. Upon heating this solution with copper(I) chloride, the diazonium group is replaced by -Cl. This is a comparatively easy method to make aryl halides as the gaseous product can be separated easily from aryl halide.[citation needed]
When an iodide is to be made, copper chloride is not needed. Addition of potassium iodide with gentle shaking produces the haloalkane.[citation needed]
Reactions
[edit]Haloalkanes are reactive towards nucleophiles. They are polar molecules: the carbon to which the halogen is attached is slightly electropositive where the halogen is slightly electronegative. This results in an electron deficient (electrophilic) carbon which, inevitably, attracts nucleophiles.[citation needed]
Substitution
[edit]Substitution reactions involve the replacement of the halogen with another molecule—thus leaving saturated hydrocarbons, as well as the halogenated product. Haloalkanes behave as the R+ synthon, and readily react with nucleophiles.[citation needed]
Hydrolysis, a reaction in which water breaks a bond, is a good example of the nucleophilic nature of haloalkanes. The polar bond attracts a hydroxide ion, OH− (NaOH(aq) being a common source of this ion). This OH− is a nucleophile with a clearly negative charge, as it has excess electrons it donates them to the carbon, which results in a covalent bond between the two. Thus C–X is broken by heterolytic fission resulting in a halide ion, X−. As can be seen, the OH is now attached to the alkyl group, creating an alcohol. (Hydrolysis of bromoethane, for example, yields ethanol). Reactions with ammonia give primary amines.[citation needed]
Chloro- and bromoalkanes are readily substituted by iodide in the Finkelstein reaction. The iodoalkanes produced easily undergo further reaction. Sodium iodide is used as a catalyst.[citation needed]
Haloalkanes react with ionic nucleophiles (e.g. cyanide, thiocyanate, azide); the halogen is replaced by the respective group. This is of great synthetic utility: chloroalkanes are often inexpensively available. For example, after undergoing substitution reactions, cyanoalkanes may be hydrolyzed to carboxylic acids, or reduced to primary amines using lithium aluminium hydride. Azoalkanes may be reduced to primary amines by Staudinger reduction or lithium aluminium hydride. Amines may also be prepared from alkyl halides in amine alkylation, Gabriel synthesis and Delepine reaction, by undergoing nucleophilic substitution with potassium phthalimide or hexamine respectively, followed by hydrolysis.[citation needed]
In the presence of a base, haloalkanes alkylate alcohols, amines, and thiols to obtain ethers, N-substituted amines, and thioethers respectively. They are substituted by Grignard reagent to give magnesium salts and an extended alkyl compound.[citation needed]
Elimination
[edit]In dehydrohalogenation reactions, the halogen and an adjacent proton are removed from halocarbons, thus forming an alkene. For example, with bromoethane and sodium hydroxide (NaOH) in ethanol, the hydroxide ion HO− abstracts a hydrogen atom. A Bromide ion is then lost, resulting in ethene, H2O and NaBr. Thus, haloalkanes can be converted to alkenes. Similarly, dihaloalkanes can be converted to alkynes.[citation needed]
In related reactions, 1,2-dibromocompounds are debrominated by zinc dust to give alkenes and geminal dihalides can react with strong bases to give carbenes.[citation needed]
Other
[edit]Haloalkanes undergo free-radical reactions with elemental magnesium to give alkyl-magnesium compound: Grignard reagent. Haloalkanes also react with lithium metal to give organolithium compounds. Both Grignard reagents and organolithium compounds behave as the R− synthon. Alkali metals such as sodium and lithium are able to cause haloalkanes to couple in Wurtz reaction, giving symmetrical alkanes. Haloalkanes, especially iodoalkanes, also undergo oxidative addition reactions to give organometallic compounds.[citation needed]
Applications
[edit]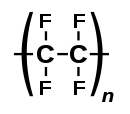
Chlorinated or fluorinated alkenes undergo polymerization. Important halogenated polymers include polyvinyl chloride (PVC), and polytetrafluoroethene (PTFE, or teflon).[citation needed]
- Alkyl fluorides
- An estimated one fifth of pharmaceuticals contain fluorine, including several of the top drugs. Most of these compounds are alkyl fluorides.[6]
- Alkyl chlorides
- Some low molecular weight chlorinated hydrocarbons such as chloroform, dichloromethane, dichloroethene, and trichloroethane are useful solvents. Several million tons of chlorinated methanes are produced annually. Chloromethane is a precursor to chlorosilanes and silicones. Chlorodifluoromethane (CHClF2) is used to make teflon.[7]
- Alkyl bromides
- Large scale applications of alkyl bromides exploit their toxicity, which also limits their usefulness. Methyl bromide is also an effective fumigant, but its production and use are controversial.[citation needed]
- Alkyl iodides
- No large scale applications are known for alkyl iodides. Methyl iodide is a popular methylating agent in organic synthesis.[citation needed]
- Chlorofluorocarbons
- Chlorofluorocarbons were used almost universally as refrigerants and propellants due to their relatively low toxicity and high heat of vaporization. Starting in the 1980s, as their contribution to ozone depletion became known, their use was increasingly restricted, and they have now largely been replaced by HFCs.
Environmental considerations
[edit]Nature produces massive amounts of chloromethane and bromomethane. Most concern focuses on anthropogenic sources, which are potential toxins, even carcinogens. Similarly, great interest has been shown in remediation of man made halocarbons such as those produced on large scale, such as dry cleaning fluids. Volatile halocarbons degrade photochemically because the carbon-halogen bond can be labile. Some microorganisms dehalogenate halocarbons. While this behavior is intriguing, the rates of remediation are generally very slow.[8]
Safety
[edit]As alkylating agents, haloalkanes are potential carcinogens. The more reactive members of this large class of compounds generally pose greater risk, e.g. carbon tetrachloride.[9]
See also
[edit]References
[edit]- ^ Patai, Saul, ed. (1973). The chemistry of the carbon-halogen bond. PATai's Chemistry of Functional Groups. Saul Patai. London: John Wiley & Sons. doi:10.1002/9780470771280. ISBN 978-0-470-77128-0. OCLC 520990502.
- ^ Gribble, Gordon (2023). Kinghorn, A. Douglas; Falk, Heinz; Gibbons, Simon; Asakawa, Yoshinori; Liu, Ji-Kai; Dirsch, Verena M. Cham (eds.). Naturally Occurring Organohalogen Compounds. Progress in the Chemistry of Organic Natural Products. Switzerland: Springer Nature. ISBN 978-3-031-26629-4.
- ^ Gordon W. Gribble (1998). "Naturally Occurring Organohalogen Compounds". Acc. Chem. Res. 31 (3): 141–152. doi:10.1021/ar9701777.
- ^ "Ionic liquids". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. p. 557. doi:10.1002/14356007.l14_l01. ISBN 978-3-527-30673-2.
- ^ "Haloalkanes & Chloroform: Class 12 Organic Chemistry Notes". NEB Notes: SEE, Class 11, Class 12 Notes Exercise Questions. Retrieved 2023-01-07.
- ^ Ann M. Thayer "Fabulous Fluorine" Chemical and Engineering News, June 5, 2006, Volume 84, pp. 15-24. http://pubs.acs.org/cen/coverstory/84/8423cover1.html
- ^ Rossberg, Manfred; Lendle, Wilhelm; Pfleiderer, Gerhard; Tögel, Adolf; Dreher, Eberhard-Ludwig; Langer, Ernst; Rassaerts, Heinz; Kleinschmidt, Peter; Strack, Heinz; Cook, Richard; Beck, Uwe; Lipper, Karl-August; Torkelson, Theodore R.; Löser, Eckhard; Beutel, Klaus K.; Mann, Trevor (2006). "Chlorinated Hydrocarbons". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a06_233.pub2. ISBN 3527306730.
- ^ Fetzner, S.; Lingens, F. (1994). "Bacterial dehalogenases: Biochemistry, genetics, and biotechnological applications". Microbiological Reviews. 58 (4): 641–685. doi:10.1128/mmbr.58.4.641-685.1994. PMC 372986. PMID 7854251.
- ^ Weber, Lutz W. D.; Boll, Meinrad; Stampfl, Andreas (2003). "Hepatotoxicity and Mechanism of Action of Haloalkanes: Carbon Tetrachloride as a Toxicological Model". Critical Reviews in Toxicology. 33 (2): 105–136. doi:10.1080/713611034. PMID 12708612. S2CID 435416.
External links
[edit] Media related to Haloalkanes at Wikimedia Commons
Media related to Haloalkanes at Wikimedia Commons
Haloalkane
View on GrokipediaClassification
Monohaloalkanes and Polyhaloalkanes
Monohaloalkanes are haloalkanes featuring a single halogen atom attached to an sp³-hybridized carbon in an aliphatic chain, with the general formula RX where R represents an alkyl group and X denotes a halogen such as chlorine, bromine, fluorine, or iodine.[8] Representative examples include chloromethane (CH₃Cl) and 1-bromopropane (CH₃CH₂CH₂Br).Complete_and_Semesters_I_and_II/Map%3A_Organic_Chemistry(Wade)/03%3A_Functional_Groups_and_Nomenclature/3.05%3A_Haloalkane_-_Classification_and_Nomenclature) In contrast, polyhaloalkanes incorporate two or more halogen atoms on the alkane framework, encompassing dihaloalkanes like 1,2-dichloroethane (ClCH₂CH₂Cl), trihaloalkanes such as chloroform (CHCl₃), and perhaloalkanes including carbon tetrachloride (CCl₄).[8] These may feature geminal halogens (on the same carbon), vicinal halogens (on adjacent carbons), or positions further apart, altering the overall molecular symmetry and polarity.[8] The structural distinction arises primarily from the number of halogen substituents, which modulates electron density via the inductive effect; halogens, being electronegative, withdraw electrons through sigma bonds (-I effect). In monohaloalkanes, this withdrawal is localized to one site, whereas in polyhaloalkanes, the cumulative -I effect from multiple halogens significantly depletes electron density across the carbon framework, enhancing polarity and influencing intermolecular forces.[9] This electron deficiency in polyhaloalkanes weakens adjacent C-H bonds relative to monohaloalkanes, as evidenced by progressively lower C-H bond dissociation energies moving from CH₃Cl to CHCl₃.[10] Consequently, polyhaloalkanes often display greater stability against oxidation but heightened susceptibility to nucleophilic attack at remaining hydrogens due to reduced bond strengths.[11] Haloalkanes, both mono- and poly-, are defined by their aliphatic C-X bonds involving sp³-hybridized carbons, distinguishing them from haloarenes where the halogen attaches to an sp²-hybridized aromatic carbon, imparting resonance stabilization and shorter, stronger C-X bonds.[9] [12] This aliphatic nature results in more polarized yet labile C-X bonds in haloalkanes, with bond dissociation energies typically ranging from 285 kJ/mol for C-I to 443 kJ/mol for C-F, varying modestly with halogen count but primarily dictated by the halogen identity rather than multiplicity in monohalo cases.[10] Polyhaloalkanes thus exhibit empirical reactivity enhancements from inductive reinforcement, such as easier deprotonation, without the delocalization seen in aromatic systems.[9]Primary, Secondary, and Tertiary Haloalkanes
Haloalkanes are classified as primary, secondary, or tertiary according to the number of alkyl groups attached to the carbon atom bearing the halogen substituent. In primary haloalkanes, the halogen is bonded to a carbon atom that is itself attached to only one other carbon atom, as in the structure R-CH₂-X, where R is an alkyl group or hydrogen and X is the halogen.[13] [14] Secondary haloalkanes feature the halogen on a carbon attached to two other carbon atoms (R₂CH-X), while tertiary haloalkanes have the halogen on a carbon bonded to three other carbon atoms (R₃C-X).[15] [2] This substitution level determines the degree of steric crowding around the reactive carbon and the extent of electron donation from adjacent alkyl groups via inductive effects. For instance, 1-chloropropane (CH₃CH₂CH₂Cl) exemplifies a primary haloalkane, with the chlorine attached to a terminal carbon bearing two hydrogens and one alkyl chain.[14] In contrast, tert-butyl chloride ( (CH₃)₃CCl ), also known as 2-chloro-2-methylpropane, is a tertiary haloalkane, where the chlorine is linked to a central carbon surrounded by three methyl groups.[2] [16] These structural differences causally influence reactivity patterns, particularly in pathways involving carbocation intermediates, where tertiary haloalkanes exhibit higher rates due to enhanced carbocation stability from greater alkyl substitution.[17] The reactivity order for unimolecular nucleophilic substitution (SN1) follows tertiary > secondary > primary, stemming from the relative stabilities of the carbocations formed: tertiary carbocations benefit from three alkyl groups providing inductive electron donation and hyperconjugation, which delocalizes positive charge and lowers the activation energy of the rate-determining ionization step by approximately 10-20 kcal/mol compared to primary analogs, based on empirical solvolysis rate data.[18] [17] Primary carbocations, lacking such stabilization, are highly unstable and rarely form under typical conditions. Conversely, steric bulk in tertiary haloalkanes impedes backside attack in bimolecular (SN2) pathways, favoring carbocation-dependent routes through increased transition state hindrance.[19] This interplay of inductive stabilization and steric effects underscores why substitution classification predicts mechanistic preferences and relative rates across haloalkane types.[18]Historical Development
Early Observations and Isolation
Chloroethane, the earliest documented haloalkane, was synthesized in the 15th century through alchemical reactions of ethanol with hydrochloric acid, yielding a volatile, colorless liquid observed for its pungent odor and rapid evaporation.[20] This compound, termed spiritus salis dulci or "sweet spirit of salt," emerged from empirical distillations aimed at purifying mineral acids, where alchemists noted its formation as a byproduct distinct from the parent alcohol and acid due to its lower boiling point and flammability.[21] The synthesis is attributed to the pseudonymous alchemist Basil Valentine, active around 1440, who described the reaction in treatises on antimonial preparations, emphasizing reproducible heating and condensation steps that isolated the haloalkane without theoretical understanding of its carbon-chlorine bond.[20] Empirical evidence from these processes included the compound's solubility in alcohol, its reaction with metals to produce hydrogen chloride gas, and its use in rudimentary medicinal applications for its anesthetic-like effects on skin contact, though these observations predated any causal analysis of substitution mechanisms.[21] In the 17th century, Johann Rudolf Glauber refined the preparation by reacting ethanol with zinc chloride, achieving higher yields and confirming the substance's consistency across batches through distillation yields of approximately 20-30% under controlled heating.[20] These isolations relied on basic apparatus like alembics and retorts, with alchemists documenting the haloalkane's density (around 0.91 g/cm³) and refractive properties as hallmarks distinguishing it from ethers or acids, grounding early recognition in sensory and volumetric data rather than molecular models.[21] Such discoveries highlighted haloalkanes' stability relative to alkanes while foreshadowing their reactivity, as inadvertent exposures produced corrosive fumes verifiable by litmus-like color changes in nearby materials.Systematic Synthesis and Key Discoveries
The systematic synthesis of haloalkanes progressed in the 19th century through direct free radical halogenation of alkanes, where chlorine or bromine substitutes hydrogen atoms under ultraviolet light or thermal conditions, yielding monohaloalkanes such as chloromethane (CH₃Cl) from methane. This method, characterized by initiation via homolytic cleavage of the halogen molecule, propagation through hydrogen abstraction and halogen addition, and termination steps, provided a versatile route for preparing simple alkyl halides from abundant hydrocarbons.[22] A pivotal advancement occurred in 1900 when Victor Grignard discovered that alkyl halides react with magnesium turnings in anhydrous diethyl ether to form Grignard reagents (RMgX), highly reactive organometallics that enable nucleophilic addition to carbonyl compounds, facilitating carbon-carbon bond formation and expanding haloalkane utility in synthetic sequences. This breakthrough, detailed in Grignard's doctoral work under Philippe Barbier, earned him the 1912 Nobel Prize in Chemistry and marked a shift toward controlled reactivity beyond substitution.[23][24] Industrial-scale production emerged in the early 20th century, exemplified by Hoechst's implementation of gas-phase chlorination of methane in 1923, which generated chloromethanes (CH₃Cl, CH₂Cl₂, CHCl₃, CCl₄) via controlled radical processes for use as solvents and intermediates. This transition from laboratory methods to continuous high-yield processes, often at elevated temperatures (400–500°C), addressed selectivity challenges through optimized ratios and conditions, supporting burgeoning chemical manufacturing.[25]Physical Properties
Molecular Structure and Bonding
Haloalkanes contain a covalent carbon-halogen (C-X) bond, where the carbon atom bonded to the halogen is sp³ hybridized, adopting a tetrahedral geometry with bond angles of approximately 109.5° around that carbon, consistent with VSEPR theory for AX₃E systems when considering the alkyl group as an effective substituent./Alkyl_Halides/Properties_of_Alkyl_Halides/Structure_of_Alkyl_Halides) The C-X bond exhibits characteristics influenced by the halogen's atomic size and electronegativity; bond lengths increase down group 17 due to expanding atomic radii, as seen in methyl halides: C-F ≈ 138 pm, C-Cl ≈ 178 pm, C-Br ≈ 193 pm, C-I ≈ 215 pm.[26] Bond dissociation energies decrease correspondingly, from C-F at 485 kJ/mol to C-I at 213 kJ/mol, arising from diminished s-p orbital overlap efficiency with larger, more diffuse halogen p-orbitals despite the smaller size of fluorine's orbitals leading to stronger bonds via better overlap and higher electronegativity-driven ionic contributions.[26] The polarity of the C-X bond stems from electronegativity differences (Pauling scale: C 2.55, F 3.98, Cl 3.16, Br 2.96, I 2.66), rendering the bond polar covalent with partial positive charge (δ⁺) on carbon and partial negative (δ⁻) on halogen, most extreme for C-F due to fluorine's highest electronegativity.[27] This manifests in dipole moments for methyl halides, peaking at 1.87 D for CH₃Cl owing to optimal charge separation balancing bond length and polarity, followed by CH₃F (1.85 D), CH₃Br (1.81 D), and CH₃I (1.62 D).[28] Infrared spectroscopy confirms these structural features through C-X stretching frequencies, which decrease with heavier halogens due to reduced force constants from longer, weaker bonds: C-F at 1000–1400 cm⁻¹, C-Cl at 600–850 cm⁻¹, C-Br at 500–700 cm⁻¹, and C-I below 500 cm⁻¹, often requiring far-IR for detection.[29] Quantum mechanical calculations, such as Hartree-Fock or DFT methods, reproduce these trends by accounting for electronegativity-induced charge transfer and hybridization effects, validating empirical data without invoking reactivity pathways.[30]Thermodynamic Properties
Haloalkanes display boiling points that increase with molecular mass, primarily due to enhanced London dispersion forces from greater molecular polarizability and surface area. For a fixed alkyl group, replacing a lighter halogen with a heavier one elevates the boiling point, as seen in the methyl halide series: fluoromethane (−78.4 °C), chloromethane (−23.7 °C), bromomethane (3.6 °C), and iodomethane (42.4 °C).[31] Within homologous series, extending the carbon chain also raises boiling points; for instance, chloromethane boils at −23.7 °C, while 1-chloroethane boils at 12.3 °C and 1-chloropropane at 46.6 °C.| Methyl Halide | Boiling Point (°C) | Melting Point (°C) |
|---|---|---|
| CH₃F | −78.4 | −141.8 |
| CH₃Cl | −23.7 | −97.6 |
| CH₃Br | 3.6 | −93.7 |
| CH₃I | 42.4 | −66.0 |
Spectroscopic Characteristics
In nuclear magnetic resonance (NMR) spectroscopy, haloalkanes display deshielded signals for nuclei adjacent to the halogen due to its electronegativity. For ¹H NMR, alpha protons (on the carbon directly attached to halogen) typically resonate at 3.0–4.5 ppm; for instance, the -CH₂Cl protons in chloromethane appear at 3.05 ppm, while those in bromoethane are around 3.4 ppm, with shifts decreasing slightly from chlorine to iodine owing to reduced inductive withdrawal.[35] In ¹³C NMR, the ipso carbon (bearing the halogen) exhibits shifts of 0–35 ppm for CH₃-X, 0–45 ppm for -CH₂-X, and up to 55 ppm for tertiary C-X, varying by halogen: fluorocarbons at 70–110 ppm, chlorocarbons at 25–50 ppm, bromocarbons at 10–40 ppm, and iodocarbons near 0–20 ppm.[36] Infrared (IR) spectroscopy identifies haloalkanes via C-X stretching bands in the fingerprint region (below 1500 cm⁻¹), where frequencies inversely correlate with halogen mass: C-F at 1000–1360 cm⁻¹ (strong), C-Cl at 600–840 cm⁻¹ (medium), C-Br at 500–700 cm⁻¹ (medium), and C-I at 450–570 cm⁻¹ (weak).[37] These bands are often broad and may overlap with alkane C-H deformations, but their presence alongside absent O-H or C=O stretches confirms haloalkane functionality; heavier halogens yield lower-energy vibrations due to reduced bond force constants. Mass spectrometry of haloalkanes features a often weak molecular ion (M⁺) due to facile fragmentation, with common losses of halogen radical (M – X•) or alkyl group via alpha-cleavage, yielding acylium-like ions or halocarbenium species (e.g., R⁺ or X⁺). Chlorine and bromine isotopes produce diagnostic peak pairs: ³⁵Cl/³⁷Cl ratio ~3:1 at Δm/z=2, and ⁷⁹Br/⁸¹Br ~1:1, aiding elemental confirmation; primary haloalkanes favor inductive cleavage over rearrangement./Instrumentation_and_Analysis/Mass_Spectrometry/Mass_Spec/Mass_Spectrometry_-_Fragmentation_Patterns)[38] These patterns distinguish haloalkanes from hydrocarbons, where such halogen-specific ions and isotope clusters are absent.Chemical Properties
Reactivity Trends by Halogen
The reactivity of haloalkanes in nucleophilic substitution reactions decreases in the order RI > RBr > RCl > RF, reflecting the ease with which the halide acts as a leaving group.[39] This trend holds across both SN1 and SN2 mechanisms, though it is most pronounced in SN2 reactions where bond breaking is rate-determining. Alkyl fluorides are notably unreactive, often requiring harsher conditions or alternative pathways for substitution, while alkyl iodides react readily under mild conditions.[40] The primary factor driving this order is the decreasing carbon-halogen bond dissociation energy (BDE) from fluorine to iodine, which lowers the activation energy for C-X bond cleavage:| Halogen | C-X BDE (kJ/mol) |
|---|---|
| F | 473 |
| Cl | 347 |
| Br | 293 |
| I | 238 |
Influence of Alkyl Group Structure
The structure of the alkyl group profoundly affects haloalkane reactivity in nucleophilic substitution, primarily through stabilization of intermediates and steric modulation of transition states. In unimolecular substitution (SN1), increased branching—such as in tertiary alkyl halides—enhances rates by forming more stable carbocations, where adjacent alkyl groups provide electron donation via hyperconjugation from C-H sigma orbitals overlapping the empty p-orbital and, to a lesser extent, inductive effects. Relative solvolysis rates in aqueous ethanol at 50°C illustrate this: tertiary chlorides react approximately 10^5 times faster than primary ones, reflecting the number of stabilizing alkyl substituents (zero for methyl, one for primary, two for secondary, three for tertiary). Recent computational analyses confirm hyperconjugation as the dominant factor, with traditional views of strong inductive donation from alkyl groups overstated; alkyls exhibit sigma-withdrawing character in isolated inductive assessments, yet net stabilization occurs in carbocations.[44][45][46] In bimolecular substitution (SN2), alkyl branching imposes steric hindrance, reducing reactivity by impeding backside nucleophilic attack. Primary alkyl halides exhibit the highest SN2 rates, with each additional branching level (secondary to tertiary) decreasing rates by factors of 10^2 to 10^4 in prototypical reactions like iodide displacement in acetone. Neopentyl halides, featuring beta-branching, show particularly sluggish kinetics due to conformational crowding that enforces a compact transition state geometry incompatible with nucleophile approach. Quantitative steric assessment via A-values in monosubstituted cyclohexanes underscores the modest bulk of halogens themselves (e.g., 0.43 kcal/mol for Cl, 0.38 kcal/mol for Br, 0.47 kcal/mol for I), contrasting sharply with larger alkyl groups like methyl (1.70 kcal/mol) or isopropyl (2.15 kcal/mol), which exacerbate hindrance in branched haloalkanes.[19][47]Natural Occurrence and Biosynthesis
Environmental Sources
Oceans serve as a primary natural reservoir for methyl halides, with emissions driven by biological production in marine environments. Marine phytoplankton, algae, and bacteria generate compounds such as chloromethane (CH₃Cl), bromomethane (CH₃Br), and iodomethane (CH₃I) through methylation processes, leading to sea-to-air fluxes observed in field measurements. For instance, surveys in the western Pacific Ocean between 2°N and 24°N documented elevated seawater concentrations of these halides, correlating with nutrient levels and contributing to atmospheric burdens via volatilization.[48] Global oceanic emissions of CH₃Cl are estimated at approximately 655 Gg yr⁻¹ based on empirical models incorporating sea surface temperature and concentration data.[49] For CH₃Br, oceans represent a net source overall, though regional sinks exist; measurements indicate supersaturation in surface waters, with the Southern Ocean potentially contributing substantially to the atmospheric flux due to high biological activity.[50] Coastal and wetland ecosystems, including salt marshes, provide additional verified reservoirs, where vegetation and microbial activity release CH₃Cl and CH₃Br. Empirical flux measurements from diverse vegetation zones in coastal salt marshes confirm emissions across tidal and upland areas, with rates varying by plant species and salinity but consistently positive to the atmosphere.[51] These terrestrial biotic sources complement oceanic inputs, though their global contribution remains smaller, on the order of tens to hundreds of Gg yr⁻¹ for CH₃Cl from wetlands and similar habitats.[52] Abiotic processes like natural biomass burning from wildfires also liberate haloalkanes, primarily through incomplete combustion of vegetation. Global modeling of biomass burning events, such as those in 2003–2020, attributes significant CH₃Cl emissions (part of non-anthropogenic totals exceeding 4700 Gg yr⁻¹ when excluding controlled fires) to pyrogenic release, with CH₃Br fluxes similarly elevated during peak fire seasons.[53] Volcanic activity contributes minor amounts via diffuse degassing from flanks and craters, where trace organohalogens including CH₃Br have been detected in emissions, potentially addressing budget discrepancies but with fluxes orders of magnitude below oceanic or biomass sources.[54] Overall, these natural reservoirs sustain baseline atmospheric levels of short-chain haloalkanes, with empirical budgets highlighting oceans as the dominant contributor for methyl bromides and chlorides.[52]Biological Production Pathways
Haloperoxidases, enzymes prevalent in marine algae and certain microbes, catalyze the biological formation of haloalkanes through the oxidation of halide ions (Cl⁻, Br⁻, or I⁻) using hydrogen peroxide (H₂O₂) as the oxidant. These vanadium- or heme-dependent enzymes generate hypohalous acids (HOX) as reactive intermediates, which electrophilically abstract hydrogen from C-H bonds in substrates, yielding C-X bonds and producing haloalkanes or more complex organohalides.[55] This pathway is site-selective in some cases, influenced by substrate proximity to the enzyme active site, and predominates in organisms accumulating high halide concentrations from seawater.[56] In algae such as red seaweeds (e.g., Asparagopsis armata), haloperoxidases facilitate bromoalkane synthesis, including bromoform (CHBr₃), which functions in chemical defense by deterring herbivores and inhibiting bacterial pathogens. Bromoform production escalates under environmental stressors like oxidative conditions, correlating with enhanced resistance to grazing and microbial fouling, as evidenced by greater bioactivity against stress-induced bacterial isolates compared to non-producers.[57][58] Such compounds likely evolved as adaptive responses to predation pressure in halide-rich marine niches, prioritizing bromination due to seawater's bromide abundance over chlorination.[59] Beyond haloperoxidases, cyanobacteria employ flavin-dependent halogenases for haloalkane incorporation into natural products like cylindrocyclophanes, chlorinated cyclic peptides with alkyl chloride moieties. The enzyme CylC in the cylindrocyclophane pathway executes regioselective chlorination of amino acid precursors, enabling dimerization and cyclization.[60] A 2024 workflow exploiting halide anion depletion—mimicking substrate limitation to trigger shunt products—uncovered novel alkyl halide-derived cylindrocyclophane variants from cyanobacterial cultures, confirming halogenase dependency and accelerating identification of underrepresented chlorinated metabolites.[61] This approach highlights enzymatic versatility in prokaryotes, where halogenation supports structural diversity for potential ecological advantages, though direct defense roles remain under investigation.[62]Nomenclature
IUPAC Systematic Naming
In IUPAC systematic nomenclature, haloalkanes are named using substitutive nomenclature, treating halogen atoms as substituents on the parent alkane chain. The name consists of the prefix for the halogen (fluoro-, chloro-, bromo-, or iodo-) followed by the name of the alkane corresponding to the longest continuous carbon chain, with locants specifying the positions of the substituents to ensure the lowest possible numbers. For unbranched chains with a single halogen at the end, no locant is needed, as in chloromethane (CH₃Cl) or bromoethane (CH₃CH₂Br); however, for internal positions or branched structures, locants are required, such as 2-chloropropane for CH₃CHClCH₃.[15][63] For compounds containing multiple identical halogen substituents, numerical prefixes such as di-, tri-, or tetra- are employed, with locants cited in ascending order and the chain numbered to minimize the set of locants; for example, 1,2-dibromoethane for BrCH₂CH₂Br or 2,2-dichloropropane for (CH₃)₂CCl₂. When different halogens are present, their prefixes are listed in alphabetical order (ignoring multiplicative prefixes), and the carbon chain is numbered to assign the lowest possible locants to the substituents collectively, starting from the end that yields the lowest number at the first point of difference. An illustration is 1-bromo-2-chloroethane for BrCH₂CH₂Cl, where "bromo" precedes "chloro" alphabetically, and locants 1 and 2 are preferred over 2 and 1 for the combined set.[64][65] Halogen substituents hold low priority in the order of functional group precedence, functioning solely as prefixes when a higher-priority characteristic group (such as -OH in alcohols or -COOH in carboxylic acids) is present in the molecule, which then determines the parent chain and suffix. For instance, in ClCH₂CH₂OH, the compound is named 2-chloroethanol, with the hydroxy group receiving the suffix "-ol" and the chloro- as a prefix, numbered to prioritize the principal function. This ensures consistent naming across organic compounds by subordinating halogens to more senior functional groups.[66][15]Historical and Common Naming Conventions
Prior to systematic IUPAC nomenclature, haloalkanes were named using a straightforward convention that appended the halogen name (ending in "-ide") to the alkyl group derived from the parent alkane, such as ethyl bromide for the compound with the formula CH₃CH₂Br.[65] This approach, rooted in 19th-century organic chemistry practices, emphasized the alkyl substituent and halogen identity for quick recognition in early synthetic and descriptive work.[66] Polyhaloalkanes followed similar descriptive patterns, often highlighting the central carbon and halogen count, as in carbon tetrachloride (CCl₄), first prepared in 1839 by Henri Victor Regnault via chlorination of chloroform.[67] Chloroform itself, synthesized in 1831 by Justus von Liebig, exemplified this by directly denoting its trichloromethane structure through common usage.[68] Industrial applications led to proprietary names like Freon, a DuPont trademark introduced in 1930 for chlorofluorocarbons such as dichlorodifluoromethane (designated Freon-12), prioritizing commercial brevity over structural detail in refrigeration contexts.[69] These historical names endured in technical literature and patents well into the 20th century for their simplicity in denoting familiar compounds, even as the International Union of Pure and Applied Chemistry, founded in 1919, advanced substitutive nomenclature to handle increasingly complex molecules systematically.[70] By the mid-20th century, IUPAC recommendations favored alkane-parent names with halo-prefixes for precision, yet common terms like methylene chloride (for dichloromethane) and vinyl chloride persisted in applied fields for practical reference, reflecting a balance between tradition and standardization.[65][71]Synthesis
Free Radical Halogenation of Alkanes
Free radical halogenation of alkanes is a substitution reaction in which a hydrogen atom is replaced by a halogen atom (typically chlorine or bromine) through a chain mechanism involving free radicals, initiated by ultraviolet light or heat.[72][73] This process is exothermic for chlorination and less so for bromination, with the reaction proceeding under conditions that generate halogen radicals without requiring peroxide initiators, unlike anti-Markovnikov additions to alkenes.[74][75] The mechanism consists of three stages: initiation, propagation, and termination. In initiation, homolytic cleavage of the halogen molecule (X₂ → 2X•) produces halogen radicals, requiring energy input such as 242 kJ/mol for Cl–Cl or 193 kJ/mol for Br–Br bonds.[73][76] Propagation involves two steps: abstraction of a hydrogen atom by the halogen radical (X• + R–H → HX + R•), forming an alkyl radical, followed by reaction of the alkyl radical with X₂ (R• + X₂ → R–X + X•), regenerating the halogen radical and yielding the haloalkane.[75][72] Termination occurs when two radicals combine (e.g., R• + R• → R–R or X• + R• → R–X), reducing the radical concentration and halting the chain.[76] These termination steps are less frequent due to low radical concentrations but contribute to minor byproducts.[73] Selectivity in hydrogen abstraction depends on the stability of the resulting alkyl radical and the exothermicity of the step, with tertiary > secondary > primary hydrogens reflecting C–H bond dissociation energies of approximately 410 kJ/mol (3°), 397 kJ/mol (2°), and 423 kJ/mol (1°).[77] For chlorination, the relative reactivities are 1:3.8:5.0 for primary:secondary:tertiary hydrogens, indicating low selectivity due to the high reactivity of Cl• and early transition state.[78][79] Bromination exhibits high selectivity with relative reactivities of 1:82:1600, as Br• abstraction is endothermic, leading to a late transition state that favors the more stable tertiary radical.[78][80] Side products arise primarily from polyhalogenation, as the C–H bond in the mono-haloalkane is weakened (e.g., by 10–20 kJ/mol for chlorination), allowing further substitution, and from non-selective abstraction yielding constitutional isomers.[72][74] To optimize monohaloalkane yields, reactions employ excess alkane (e.g., 10:1 molar ratio to halogen), low halogen concentration, and controlled temperatures (around 25–40°C for chlorination to balance rate and selectivity).[73] Fluorination is too violent and unselective for practical use, while iodination does not propagate effectively due to endothermic steps.[81]Addition Reactions to Unsaturated Hydrocarbons
Haloalkanes can be synthesized through electrophilic addition reactions of hydrogen halides (HX, where X is chlorine, bromine, or iodine) to alkenes, which follow Markovnikov's rule: the hydrogen atom adds to the sp²-hybridized carbon bearing the greater number of hydrogen substituents, while the halide adds to the other carbon, reflecting the formation of the more stable carbocation intermediate.[82][83] This regioselectivity arises because the protonation of the alkene generates a carbocation at the carbon that can better stabilize the positive charge through hyperconjugation and inductive effects from adjacent alkyl groups.[84] For instance, propene (CH₃-CH=CH₂) reacts with HBr to predominantly yield 2-bromopropane (CH₃-CHBr-CH₃), with the secondary carbocation forming preferentially over the primary alternative.[82][85] In the absence of peroxides, the reaction mechanism involves two key steps: initial protonation of the double bond by HX to form the carbocation, followed by rapid nucleophilic attack by the halide anion.[82] This pathway is favored under ionic conditions and applies to HCl, HBr, and HI, though HI additions can be complicated by competing elimination due to its stronger acidity.[83] The reaction typically occurs at room temperature or with mild heating, often in inert solvents, and yields are high for terminal alkenes. Addition of molecular halogens (X₂, where X = Cl or Br) to alkenes also produces haloalkanes, specifically vicinal dihalides, via an anti addition mechanism involving a cyclic halonium ion intermediate that enforces stereospecific trans geometry.[86] The halogen molecule polarizes upon approach to the π-bond, with the electrophilic halogen bridging the carbons, followed by backside attack by the nucleophilic halide.[87] For example, ethene reacts with Br₂ to form 1,2-dibromoethane, a vicinal dibromide used in further synthesis.[86] This method extends to alkynes, where one equivalent of X₂ yields vinyl dihalides, but excess halogen can lead to tetrahalides.[88] An exception to Markovnikov regioselectivity occurs in the addition of HBr to alkenes in the presence of peroxides (ROOR), which initiates a free radical chain mechanism yielding the anti-Markovnikov product, where bromide attaches to the less substituted carbon.[89][90] The mechanism proceeds via peroxide decomposition to alkoxy radicals, bromine radical addition to the alkene forming the more stable carbon radical (at the less substituted carbon due to lower steric hindrance), and subsequent abstraction of hydrogen from HBr. This peroxide effect is unique to HBr, as the H-Br bond dissociation energy (366 kJ/mol) supports efficient radical propagation, unlike HCl or HI.[89] For propene, this yields 1-bromopropane as the major product under radical conditions.[84]Conversion from Alcohols and Other Functional Groups
Haloalkanes can be synthesized from alcohols through nucleophilic substitution reactions in which the hydroxyl group is replaced by a halogen atom, typically under acidic conditions that facilitate the departure of water as a leaving group.[91] For tertiary alcohols, concentrated hydrohalic acids such as HCl or HBr react rapidly via an SN1 mechanism involving carbocation intermediates, often at room temperature.[92] Secondary alcohols follow a similar SN1 pathway but require mild heating, while primary alcohols proceed via SN2 with HX reagents, sometimes assisted by zinc chloride to enhance reactivity, though this risks rearrangement in branched systems.[93] The Lucas reagent, a mixture of concentrated HCl and anhydrous ZnCl₂, is particularly effective for converting secondary and tertiary alcohols to alkyl chlorides, with reaction rates distinguishing alcohol types: tertiary alcohols react immediately at room temperature, secondary within minutes upon warming, and primary only under forcing conditions or not at all without rearrangement.[94] To minimize carbocation-mediated side reactions in primary and secondary alcohols, alternative reagents like thionyl chloride (SOCl₂) for chlorides or phosphorus tribromide (PBr₃) for bromides are preferred, as they operate via SN2 mechanisms with retention or inversion of configuration depending on conditions (e.g., SOCl₂ with pyridine yields retention via an internal return mechanism).[92] These methods avoid the need for strong acids and provide cleaner conversions, with SOCl₂ producing SO₂ and HCl as byproducts.[91] From carboxylic acids, haloalkanes are obtained via the Hunsdiecker reaction, where silver(I) salts of the acids (RCOOAg) are treated with bromine in carbon tetrachloride, leading to decarboxylative bromination and formation of RBr with one fewer carbon atom than the original acid.[95] This radical process involves homolytic cleavage of Br₂, attack on the carboxylate, and subsequent decarboxylation of an acyl hypobromite intermediate, typically conducted under reflux with yields of 60-80% for unbranched chains, though it is less effective for α-branched acids due to steric hindrance.[95] Unlike the Sandmeyer reaction, which applies to aromatic diazonium salts for aryl halides, the Hunsdiecker method is specific to alkyl chains from aliphatic carboxylic acids and does not involve diazotization.[96]Advanced and Green Synthetic Methods
In the 2020s, electrochemical methods have advanced selective C-H halogenation of alkanes, providing sustainable alternatives to classical free radical processes by operating under mild conditions with electricity as the oxidant, thus reducing reliance on chemical initiators and waste generation. A metal-free protocol reported in 2025 enables regioselective chlorination of terminal C-H bonds in unfunctionalized linear alkanes using chloride salts, achieving up to 80% yield for primary chlorides with minimal over-chlorination, attributed to anodic generation of chlorine radicals that preferentially abstract terminal hydrogens.[97] This approach demonstrates improved atom economy and scalability compared to thermal methods, with empirical yields exceeding 70% for substrates like n-hexane in undivided cells at room temperature.[97] Photocatalytic strategies, utilizing visible light and earth-abundant catalysts, facilitate radical-mediated C-H halogenation of aliphatic chains, emphasizing green metrics such as solvent minimization and operational simplicity. For example, visible-light-driven systems with organic dyes or metal complexes generate halogen radicals for site-selective bromination or chlorination, yielding alkyl bromides from cycloalkanes with selectivities up to 90% at benzylic or allylic positions, as evidenced by hydrogen atom transfer mechanisms that avoid stoichiometric oxidants.[98] These methods, developed post-2010, often proceed in aqueous or solvent-free media, with turnover numbers exceeding 100 for iridium-based photocatalysts in deformylative halogenations of aldehydes to primary alkyl halides, highlighting reduced energy input relative to UV-initiated classical routes.[99] Biocatalytic halogenation employs flavin-dependent halogenases (FDHs) to catalyze regioselective C-X bond formation, drawing from enzymatic mechanisms for precise control in aqueous environments at ambient conditions, thereby aligning with sustainability goals through cofactor recycling and minimal byproduct formation. Engineered FDH variants, such as those derived from PrnA or RebH, have been applied to aliphatic and amino acid substrates, achieving chlorination yields of 50-90% for epsilon positions in lysine derivatives via hypohalite intermediates generated from O2 and halide ions.[100] Recent protein engineering efforts, including directed evolution, expand substrate scope to non-natural alkanes, with immobilized systems demonstrating reusability over multiple cycles and enantioselectivities >95% ee, underscoring their potential for scalable green synthesis despite challenges in cofactor supply.[101]Reactivity and Reactions
Nucleophilic Substitution Mechanisms
Haloalkanes undergo nucleophilic substitution reactions where a nucleophile displaces a halide leaving group, proceeding via either SN1 or SN2 mechanisms depending on substrate structure, nucleophile strength, solvent polarity, and leaving group ability.[102] The SN2 pathway is a concerted, bimolecular process characterized by second-order kinetics, where the rate depends on both the concentrations of the haloalkane and nucleophile, as evidenced by experimental rate laws such as rate = k [RX][Nu⁻].[103] This mechanism involves backside attack of the nucleophile on the carbon atom, resulting in complete inversion of stereochemistry, a phenomenon confirmed through stereochemical studies on chiral secondary alkyl halides like 2-bromobutane reacting with hydroxide in acetone, yielding inverted products with high enantiomeric excess.[104] In contrast, the SN1 mechanism is a stepwise, unimolecular process with first-order kinetics, where the rate-determining step is the dissociation of the haloalkane to form a carbocation intermediate, independent of nucleophile concentration (rate = k [RX]).[105] This leads to racemization at the reaction center due to planar carbocation geometry allowing attack from either face, though partial inversion may occur from ion-pair effects; kinetic studies on tertiary halides like tert-butyl chloride in aqueous ethanol show approximately 50:50 mixtures of enantiomers.[105] SN1 is favored for tertiary haloalkanes and secondary ones under ionizing conditions, as carbocation stability increases with alkyl substitution, supported by relative rate data: methyl < primary << secondary < tertiary, with tertiary rates up to 10^5 times faster than primary in solvolysis reactions.[102] Key factors influencing mechanism selection include substrate type, where primary haloalkanes predominantly follow SN2 due to minimal steric hindrance, while tertiary favor SN1 from enhanced carbocation stabilization.[102] Strong nucleophiles like alkoxides promote SN2 by direct displacement, whereas weak ones like water favor SN1 via carbocation capture.[106] Solvent effects are pronounced: polar protic solvents (e.g., water, alcohols) stabilize the SN1 transition state and carbocation through hydrogen bonding but solvate anions, slowing SN2; polar aprotic solvents (e.g., DMSO, acetone) enhance SN2 rates by up to 10^6-fold for primary halides by leaving nucleophiles unsolvated, while minimally aiding SN1.[106] Leaving group ability correlates with halide basicity, following the order I⁻ > Br⁻ > Cl⁻ > F⁻, as iodide's weaker C–I bond (bond dissociation energy ~234 kJ/mol vs. 485 kJ/mol for C–F) and lower basicity facilitate departure, with rate enhancements of 10^4–10^5 for iodides over fluorides in substitution kinetics.[39]| Factor | SN2 | SN1 |
|---|---|---|
| Kinetics | Second-order: rate = k [RX][Nu] | First-order: rate = k [RX] |
| Stereochemistry | Inversion | Racemization (partial) |
| Substrate Preference | Primary > secondary >> tertiary | Tertiary > secondary >> primary |
| Nucleophile | Strong, good nucleofugicity | Weak, often solvent |
| Solvent | Polar aprotic | Polar protic |
| Leaving Group Effect | Moderate; better groups accelerate both | Pronounced; stabilizes carbocation formation[102][105][106][39] |

