Recent from talks
Nothing was collected or created yet.
Pediatrics
View on Wikipedia
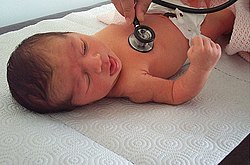 A pediatrician examines a neonate. | |
| Focus | Infants, Children, Adolescents, and Young Adults |
|---|---|
| Subdivisions | Paediatric cardiology, neonatology, critical care, pediatric oncology, hospital medicine, primary care, others (see below) |
| Significant diseases | Congenital diseases, Infectious diseases, Childhood cancer, Mental disorders |
| Significant tests | World Health Organization Child Growth Standards |
| Specialist | Pediatrician |
| Glossary | Glossary of medicine |
Pediatrics (American English), also spelled paediatrics (British English), also known as underage medicine is the branch of medicine that involves the medical care of infants, children, adolescents, and young adults. Within the Commonwealth, pediatrics cover patients until the age of 18, except in India where the paediatric age is 12.[1][2] The American Academy of Pediatrics recommends people seek pediatric care through the age of 21, but some pediatric subspecialists continue to care for adults up to 25.[3][4] Worldwide age limits of pediatrics have been trending upward year after year.[5] A medical doctor who specializes in this area is known as a pediatrician, or paediatrician. The word pediatrics and its cognates mean "healer of children", derived from the two Greek words: παῖς (pais "child") and ἰατρός (iatros "doctor, healer"). Pediatricians work in clinics, research centers, universities, general hospitals and children's hospitals, including those who practice pediatric subspecialties (e.g. neonatology requires resources available in a NICU).
History
[edit]
The earliest mentions of child-specific medical problems appear in the Hippocratic Corpus, published in the fifth century B.C., and the famous Sacred Disease. These publications discussed topics such as childhood epilepsy and premature births. From the first to fourth centuries A.D., Greek philosophers and physicians Celsus, Soranus of Ephesus, Aretaeus, Galen, and Oribasius, also discussed specific illnesses affecting children in their works, such as rashes, epilepsy, and meningitis.[6] Already Hippocrates, Aristotle, Celsus, Soranus, and Galen[7] understood the differences in growing and maturing organisms that necessitated different treatment: Ex toto non sic pueri ut viri curari debent ("In general, boys should not be treated in the same way as men").[8] Some of the oldest traces of pediatrics can be discovered in Ancient India where children's doctors were called kumara bhrtya.[7]
Even though some pediatric works existed during this time, they were scarce and rarely published due to a lack of knowledge in pediatric medicine. Sushruta Samhita, an ayurvedic text composed during the sixth century BCE, contains the text about pediatrics.[9] Another ayurvedic text from this period is Kashyapa Samhita.[10][11] A second century AD manuscript by the Greek physician and gynecologist Soranus of Ephesus dealt with neonatal pediatrics.[12] Byzantine physicians Oribasius, Aëtius of Amida, Alexander Trallianus, and Paulus Aegineta contributed to the field.[7] The Byzantines also built brephotrophia (crêches).[7] Islamic Golden Age writers served as a bridge for Greco-Roman and Byzantine medicine and added ideas of their own, especially Haly Abbas, Yahya Serapion, Abulcasis, Avicenna, and Averroes. The Persian philosopher and physician al-Razi (865–925), sometimes called the father of pediatrics, published a monograph on pediatrics titled Diseases in Children.[13][14] Also among the first books about pediatrics was Libellus [Opusculum] de aegritudinibus et remediis infantium 1472 ("Little Book on Children Diseases and Treatment"), by the Italian pediatrician Paolo Bagellardo.[15][6] In sequence came Bartholomäus Metlinger's Ein Regiment der Jungerkinder 1473, Cornelius Roelans (1450–1525) no title Buchlein, or Latin compendium, 1483, and Heinrich von Louffenburg (1391–1460) Versehung des Leibs written in 1429 (published 1491), together form the Pediatric Incunabula, four great medical treatises on children's physiology and pathology.[7]
While more information about childhood diseases became available, there was little evidence that children received the same kind of medical care that adults did.[16] It was during the seventeenth and eighteenth centuries that medical experts started offering specialized care for children.[6] The Swedish physician Nils Rosén von Rosenstein (1706–1773) is considered to be the founder of modern pediatrics as a medical specialty,[17][18] while his work The diseases of children, and their remedies (1764) is considered to be "the first modern textbook on the subject".[19] However, it was not until the nineteenth century that medical professionals acknowledged pediatrics as a separate field of medicine. The first pediatric-specific publications appeared between the 1790s and the 1920s.[20]
Etymology
[edit]The term pediatrics was first introduced in English in 1859 by Abraham Jacobi. In 1860, he became "the first dedicated professor of pediatrics in the world."[21] Jacobi is known as the father of American pediatrics because of his many contributions to the field.[22][23] He received his medical training in Germany and later practiced in New York City.[24]
The first generally accepted pediatric hospital is the Hôpital des Enfants Malades (French: Hospital for Sick Children), which opened in Paris in June 1802 on the site of a previous orphanage.[25] From its beginning, this famous hospital accepted patients up to the age of fifteen years,[26] and it continues to this day as the pediatric division of the Necker-Enfants Malades Hospital, created in 1920 by merging with the nearby Necker Hospital, founded in 1778.[27]
In other European countries, the Charité (a hospital founded in 1710) in Berlin established a separate Pediatric Pavilion in 1830, followed by similar institutions at Saint Petersburg in 1834, and at Vienna and Breslau (now Wrocław), both in 1837. In 1852 Britain's first pediatric hospital, the Hospital for Sick Children, Great Ormond Street was founded by Charles West.[25] The first Children's hospital in Scotland opened in 1860 in Edinburgh.[28] In the US, the first similar institutions were the Children's Hospital of Philadelphia, which opened in 1855, and then Boston Children's Hospital (1869).[29] Subspecialties in pediatrics were created at the Harriet Lane Home at Johns Hopkins by Edwards A. Park.[30]
Differences between adult and pediatric medicine
[edit]The body size differences are paralleled by maturation changes. The smaller body of an infant or neonate is substantially different physiologically from that of an adult. Congenital defects, genetic variance, and developmental issues are of greater concern to pediatricians than they often are to adult physicians. A common adage is that children are not simply "little adults". The clinician must take into account the immature physiology of the infant or child when considering symptoms, prescribing medications, and diagnosing illnesses.[31]
Pediatric physiology directly impacts the pharmacokinetic properties of drugs that enter the body. The absorption, distribution, metabolism, and elimination of medications differ between developing children and grown adults.[31][32][33] Despite completed studies and reviews, continual research is needed to better understand how these factors should affect the decisions of healthcare providers when prescribing and administering medications to the pediatric population.[31]
Absorption
[edit]Many drug absorption differences between pediatric and adult populations revolve around the stomach. Neonates and young infants have increased stomach pH due to decreased acid secretion, thereby creating a more basic environment for drugs that are taken by mouth.[32][31][33] Acid is essential to degrading certain oral drugs before systemic absorption. Therefore, the absorption of these drugs in children is greater than in adults due to decreased breakdown and increased preservation in a less acidic gastric space.[32]
Children also have an extended rate of gastric emptying, which slows the rate of drug absorption.[32][33]
Drug absorption also depends on specific enzymes that come in contact with the oral drug as it travels through the body. Supply of these enzymes increase as children continue to develop their gastrointestinal tract.[32][33] Pediatric patients have underdeveloped proteins, which leads to decreased metabolism and increased serum concentrations of specific drugs. However, prodrugs experience the opposite effect because enzymes are necessary for allowing their active form to enter systemic circulation.[32]
Distribution
[edit]Percentage of total body water and extracellular fluid volume both decrease as children grow and develop with time. Pediatric patients thus have a larger volume of distribution than adults, which directly affects the dosing of hydrophilic drugs such as beta-lactam antibiotics like ampicillin.[32] Thus, these drugs are administered at greater weight-based doses or with adjusted dosing intervals in children to account for this key difference in body composition.[32][31]
Infants and neonates also have fewer plasma proteins. Thus, highly protein-bound drugs have fewer opportunities for protein binding, leading to increased distribution.[31]
Metabolism
[edit]Drug metabolism primarily occurs via enzymes in the liver and can vary according to which specific enzymes are affected in a specific stage of development.[32] Phase I and Phase II enzymes have different rates of maturation and development, depending on their specific mechanism of action (i.e. oxidation, hydrolysis, acetylation, methylation, etc.). Enzyme capacity, clearance, and half-life are all factors that contribute to metabolism differences between children and adults.[32][33] Drug metabolism can even differ within the pediatric population, separating neonates and infants from young children.[31]
Elimination
[edit]Drug elimination is primarily facilitated via the liver and kidneys.[32] In infants and young children, the larger relative size of their kidneys leads to increased renal clearance of medications that are eliminated through urine.[33] In preterm neonates and infants, their kidneys are slower to mature and thus are unable to clear as much drug as fully developed kidneys. This can cause unwanted drug build-up, which is why it is important to consider lower doses and greater dosing intervals for this population.[31][32] Diseases that negatively affect kidney function can also have the same effect and thus warrant similar considerations.[32]
Pediatric autonomy in healthcare
[edit]A major difference between the practice of pediatric and adult medicine is that children, in most jurisdictions and with certain exceptions, cannot make decisions for themselves. The issues of guardianship, privacy, legal responsibility, and informed consent must always be considered in every pediatric procedure. Pediatricians often have to treat the parents and sometimes, the family, rather than just the child. Adolescents are in their own legal class, having rights to their own health care decisions in certain circumstances. The concept of legal consent combined with the non-legal consent (assent) of the child when considering treatment options, especially in the face of conditions with poor prognosis or complicated and painful procedures/surgeries, means the pediatrician must take into account the desires of many people, in addition to those of the patient.[citation needed]
History of pediatric autonomy
[edit]The term autonomy is traceable to ethical theory and law, where it states that autonomous individuals can make decisions based on their own logic.[34] Hippocrates was the first to use the term in a medical setting. He created a code of ethics for doctors called the Hippocratic Oath that highlighted the importance of putting patients' interests first, making autonomy for patients a top priority in health care.[35]
In ancient times, society did not view pediatric medicine as essential or scientific.[36] Experts considered professional medicine unsuitable for treating children. Children also had no rights. Fathers regarded their children as property, so their children's health decisions were entrusted to them.[6] As a result, mothers, midwives, "wise women", and general practitioners treated the children instead of doctors.[36] Since mothers could not rely on professional medicine to take care of their children, they developed their own methods, such as using alkaline soda ash to remove the vernix at birth and treating teething pain with opium or wine. The absence of proper pediatric care, rights, and laws in health care to prioritize children's health led to many of their deaths. Ancient Greeks and Romans sometimes even killed healthy female babies and infants with deformities since they had no adequate medical treatment and no laws prohibiting infanticide.[6]
In the twentieth century, medical experts began to put more emphasis on children's rights. In 1989, in the United Nations Rights of the Child Convention, medical experts developed the Best Interest Standard of Child to prioritize children's rights and best interests. This event marked the onset of pediatric autonomy. In 1995, the American Academy of Pediatrics (AAP) finally acknowledged the Best Interest Standard of a Child as an ethical principle for pediatric decision-making, and it is still being used today.[35]
Parental authority and current medical issues
[edit]The majority of the time, parents have the authority to decide what happens to their child. Philosopher John Locke argued that it is the responsibility of parents to raise their children and that God gave them this authority. In modern society, Jeffrey Blustein, modern philosopher and author of the book Parents and Children: The Ethics of Family, argues that parental authority is granted because the child requires parents to satisfy their needs. He believes that parental autonomy is more about parents providing good care for their children and treating them with respect than parents having rights.[37] The researcher Kyriakos Martakis, MD, MSc, explains that research shows parental influence negatively affects children's ability to form autonomy. However, involving children in the decision-making process allows children to develop their cognitive skills and create their own opinions and, thus, decisions about their health. Parental authority affects the degree of autonomy the child patient has. As a result, in Argentina, the new National Civil and Commercial Code has enacted various changes to the healthcare system to encourage children and adolescents to develop autonomy. It has become more crucial to let children take accountability for their own health decisions.[38]
In most cases, the pediatrician, parent, and child work as a team to make the best possible medical decision. The pediatrician has the right to intervene for the child's welfare and seek advice from an ethics committee. However, in recent studies, authors have denied that complete autonomy is present in pediatric healthcare. The same moral standards should apply to children as they do to adults. In support of this idea is the concept of paternalism, which negates autonomy when it is in the patient's interests. This concept aims to keep the child's best interests in mind regarding autonomy. Pediatricians can interact with patients and help them make decisions that will benefit them, thus enhancing their autonomy. However, radical theories that question a child's moral worth continue to be debated today.[38] Authors often question whether the treatment and equality of a child and an adult should be the same. Author Tamar Schapiro notes that children need nurturing and cannot exercise the same level of authority as adults.[39] Hence, continuing the discussion on whether children are capable of making important health decisions until this day.
Modern advancements
[edit]According to the Subcommittee of Clinical Ethics of the Argentinean Pediatric Society (SAP), children can understand moral feelings at all ages and can make reasonable decisions based on those feelings. Therefore, children and teens are deemed capable of making their own health decisions when they reach the age of 13. Recently, studies made on the decision-making of children have challenged that age to be 12.[38]
Technology has made several modern advancements that contribute to the future development of child autonomy, for example, unsolicited findings (U.F.s) of pediatric exome sequencing. They are findings based on pediatric exome sequencing that explain in greater detail the intellectual disability of a child and predict to what extent it will affect the child in the future. Genetic and intellectual disorders in children make them incapable of making moral decisions, so people look down upon this kind of testing because the child's future autonomy is at risk. It is still in question whether parents should request these types of testing for their children. Medical experts argue that it could endanger the autonomous rights the child will possess in the future. However, the parents contend that genetic testing would benefit the welfare of their children since it would allow them to make better health care decisions.[40] Exome sequencing for children and the decision to grant parents the right to request them is a medically ethical issue that many still debate today.
Education requirements
[edit]The examples and perspective in this section deal primarily with United States and do not represent a worldwide view of the subject. (September 2019) |
Aspiring medical students will need 4 years of undergraduate courses at a college or university, which will get them a BS, BA or other bachelor's degree. After completing college, future pediatricians will need to attend 4 years of medical school (MD/DO/MBBS) and later do 3 more years of residency training, the first year of which is called "internship." After completing the 3 years of residency, physicians are eligible to become certified in pediatrics by passing a rigorous test that deals with medical conditions related to young children.[citation needed]
In high school, future pediatricians are required to take basic science classes such as biology, chemistry, physics, algebra, geometry, and calculus. It is also advisable to learn a foreign language (preferably Spanish in the United States) and be involved in high school organizations and extracurricular activities. After high school, college students simply need to fulfill the basic science course requirements that most medical schools recommend and will need to prepare to take the MCAT (Medical College Admission Test) in their junior or early senior year in college. Once attending medical school, student courses will focus on basic medical sciences like human anatomy, physiology, chemistry, etc., for the first three years, the second year of which is when medical students start to get hands-on experience with actual patients.[41]
Training of pediatricians
[edit] | |
| Occupation | |
|---|---|
| Names |
|
Occupation type | Specialty |
Activity sectors | Medicine |
| Description | |
Education required | |
Fields of employment | Hospitals, Clinics |
The training of pediatricians varies considerably across the world. Depending on jurisdiction and university, a medical degree course may be either undergraduate-entry or graduate-entry. The former commonly takes five or six years and has been usual in the Commonwealth. Entrants to graduate-entry courses (as in the US), usually lasting four or five years, have previously completed a three- or four-year university degree, commonly but by no means always in sciences. Medical graduates hold a degree specific to the country and university in and from which they graduated. This degree qualifies that medical practitioner to become licensed or registered under the laws of that particular country, and sometimes of several countries, subject to requirements for "internship" or "conditional registration".
Pediatricians must undertake further training in their chosen field. This may take from four to eleven or more years depending on jurisdiction and the degree of specialization.
In the United States, a medical school graduate wishing to specialize in pediatrics must undergo a three-year residency composed of outpatient, inpatient, and critical care rotations. Subspecialties within pediatrics require further training in the form of 3-year fellowships. Subspecialties include critical care, gastroenterology, neurology, infectious disease, hematology/oncology, rheumatology, pulmonology, child abuse, emergency medicine, endocrinology, neonatology, and others.[42]
In most jurisdictions, entry-level degrees are common to all branches of the medical profession, but in some jurisdictions, specialization in pediatrics may begin before completion of this degree. In some jurisdictions, pediatric training is begun immediately following the completion of entry-level training. In other jurisdictions, junior medical doctors must undertake generalist (unstreamed) training for a number of years before commencing pediatric (or any other) specialization. Specialist training is often largely under the control of 'pediatric organizations (see below) rather than universities and depends on the jurisdiction.
Subspecialties
[edit]Subspecialties of pediatrics include:
(not an exhaustive list)
- Addiction medicine (multidisciplinary)
- Adolescent medicine
- Child abuse pediatrics
- Clinical genetics
- Clinical informatics
- Developmental-behavioral pediatrics
- Headache medicine
- Hospital medicine
- Medical toxicology
- Metabolic medicine
- Neonatology/Perinatology
- Pain medicine (multidisciplinary)
- Palliative care (multidisciplinary)
- Pediatric allergy and immunology
- Pediatric cardiology
- Pediatric cardiac critical care
- Pediatric critical care
- Neurocritical care
- Pediatric cardiac critical care
- Pediatric emergency medicine
- Pediatric endocrinology
- Pediatric gastroenterology
- Pediatric hematology
- Pediatric infectious disease
- Pediatric nephrology
- Pediatric oncology
- Pediatric neuro-oncology
- Pediatric pulmonology
- Primary care
- Pediatric rheumatology
- Sleep medicine (multidisciplinary)
- Social pediatrics
- Sports medicine
Other specialties that care for children
[edit](not an exhaustive list)
- Child neurology
- Addiction medicine (multidisciplinary)
- Brain injury medicine
- Clinical neurophysiology
- Epilepsy
- Headache medicine
- Neurocritical care
- Neuroimmunology
- Neuromuscular medicine
- Pain medicine (multidisciplinary)
- Palliative care (multidisciplinary)
- Pediatric neuro-oncology
- Sleep medicine (multidisciplinary)
- Child and adolescent psychiatry, subspecialty of psychiatry
- Neurodevelopmental disabilities
- Pediatric anesthesiology, subspecialty of anesthesiology
- Pediatric dentistry, subspecialty of dentistry
- Pediatric dermatology, subspecialty of dermatology
- Pediatric gynecology
- Pediatric neurosurgery, subspecialty of neurosurgery
- Pediatric ophthalmology, subspecialty of ophthalmology
- Pediatric orthopedic surgery, subspecialty of orthopedic surgery
- Pediatric otolaryngology, subspecialty of otolaryngology
- Pediatric plastic surgery, subspecialty of plastic surgery
- Pediatric radiology, subspecialty of radiology
- Pediatric rehabilitation medicine, subspecialty of physical medicine and rehabilitation
- Pediatric surgery, subspecialty of general surgery
- Pediatric urology, subspecialty of urology
See also
[edit]- American Academy of Pediatrics
- American Osteopathic Board of Pediatrics
- Center on Media and Child Health (CMCH)
- Children's hospital
- List of pediatric organizations
- List of pediatrics journals
- Medical specialty
- Pediatric Oncall
- Pain in babies
- Royal College of Paediatrics and Child Health
- Pediatric environmental health
References
[edit]- ^ "Paediatrics" (PDF). nhs.uk. Archived (PDF) from the original on 13 July 2020. Retrieved 2 July 2020.
- ^ doctor.ndtv.com. "What is the paediatric age in India?". Doctor.ndtv.com. Archived from the original on 18 July 2025. Retrieved 21 October 2025.
- ^ "Choosing a Pediatrician for Your New Baby (for Parents) - Nemours KidsHealth". kidshealth.org. Archived from the original on 14 July 2020. Retrieved 13 July 2020.
- ^ "Age limits of pediatrics". Pediatrics. 81 (5): 736. May 1988. doi:10.1542/peds.81.5.736. PMID 3357740. S2CID 245164191. Archived from the original on 19 April 2017. Retrieved 18 April 2017.
- ^ Sawyer, Susan M.; McNeil, Robyn; Francis, Kate L.; Matskarofski, Juliet Z.; Patton, George C.; Bhutta, Zulfiqar A.; Esangbedo, Dorothy O.; Klein, Jonathan D. (1 November 2019). "The age of paediatrics". The Lancet Child & Adolescent Health. 3 (11): 822–830. doi:10.1016/S2352-4642(19)30266-4. ISSN 2352-4642. PMID 31542355. S2CID 202732818.
- ^ a b c d e Duffin, Jacalyn (2010). History of Medicine, Second Edition: A Scandalously Short Introduction. University of Toronto Press.
- ^ a b c d e Colón, A. R.; Colón, P. A. (January 1999). Nurturing children: a history of pediatrics. Greenwood Press. ISBN 978-0-313-31080-5. Retrieved 20 October 2012.
- ^ Celsus, De Medicina, Book 3, Chapter 7, § 1.
- ^ John G. Raffensperger. Children's Surgery: A Worldwide History. McFarland. p. 21.
- ^ David Levinson; Karen Christensen. Encyclopedia of modern Asia. Vol. 4. Charles Scribner's Sons. p. 116.
- ^ Desai, A.B. Textbook Of Paediatrics. Orient blackswan. p. 1.
- ^ Dunn, P. M. (1995). "Soranus of Ephesus (Circa AD 98-138) and perinatal care in Roman times". Archives of Disease in Childhood. Fetal and Neonatal Edition. 73 (1): F51 – F52. doi:10.1136/fn.73.1.f51. PMC 2528358. PMID 7552600.
- ^ Elgood, Cyril (2010). A Medical History of Persia and The Eastern Caliphate (1st ed.). London: Cambridge. pp. 202–203. ISBN 978-1-108-01588-2.
By writing a monograph on 'Diseases in Children' he may also be looked upon as the father of paediatrics.
- ^ U.S. National Library of Medicine, "Islamic Culture and the Medical Arts, Al-Razi, the Clinician" [1] Archived 5 January 2018 at the Wayback Machine
- ^ "Achar S Textbook Of Pediatrics (Third Edition)". A. B. Desai (ed.) (1989). p.1. ISBN 81-250-0440-8
- ^ Stern, Alexandra Minna; Markel, Howard (2002). Formative Years: Children's Health in the United States, 1880-2000. University of Michigan Press. pp. 23–24. doi:10.3998/mpub.17065. ISBN 978-0-472-02503-9. Archived from the original on 30 November 2021. Retrieved 30 November 2021.
- ^ Lock, Stephen; John M. Last; George Dunea (2001). The Oxford illustrated companion to medicine. Oxford University Press US. p. 173. ISBN 978-0-19-262950-0. Retrieved 9 July 2010.
Rosen von Rosenstein.
- ^ Roberts, Michael (2003). The Age of Liberty: Sweden 1719–1772. Cambridge University Press. p. 216. ISBN 978-0-521-52707-1. Retrieved 9 July 2010.
- ^ Dallas, John. "Classics of Child Care". Royal College of Physicians of Edinburgh. Archived from the original on 27 July 2011. Retrieved 9 July 2010.
- ^ Duffin, Jacalyn (29 May 2010). History of Medicine, Second Edition: A Scandalously Short Introduction. University of Toronto Press.
- ^ Stern, Alexandra Minna; Markel, Howard (2002). Formative Years: Children's Health in the United States, 1880-2000. University of Michigan Press. pp. 23–24. doi:10.3998/mpub.17065. ISBN 978-0-472-02503-9. Archived from the original on 30 November 2021. Retrieved 30 November 2021.
- ^ "Broadribb's Introductory Pediatric Nursing". Nancy T. Hatfield (2007). p.4. ISBN 0-7817-7706-2
- ^ "Jacobi Medical Center - General Information". Archived from the original on 18 April 2006. Retrieved 6 April 2006.
- ^ Kutzsche, Stefan (8 April 2021). "Abraham Jacobi (1830–1919) and his transition from political to medical activist". Acta Paediatrica. 110 (8): 2303–2305. doi:10.1111/apa.15887. ISSN 0803-5253. PMID 33963612. S2CID 233998658. Archived from the original on 7 May 2023. Retrieved 7 May 2023.
- ^ a b Ballbriga, Angel (1991). "One century of pediatrics in Europe (section: development of pediatric hospitals in Europe)". In Nichols, Burford L.; et al. (eds.). History of Paediatrics 1850–1950. Nestlé Nutrition Workshop Series. Vol. 22. New York: Raven Press. pp. 6–8. ISBN 0-88167-695-0.
- ^ official history site (in French) of nineteenth century paediatric hospitals in Paris
- ^ "Introducing the Necker-Enfants Malades Hospital". Hôpital des Necker-Enfants Malades. 25 September 2015.
- ^ Young, D.G. (August 1999). "The Mason Brown Lecture: Scots and paediatric surgery". Journal of the Royal College of Surgeons Edinburgh. 44 (4): 211–5. PMID 10453141. Archived from the original on 14 July 2014.
- ^ Pearson, Howard A. (1991). "Pediatrics in the United States". In Nichols, Burford L.; et al. (eds.). History of Paediatrics 1850–1950. Nestlé Nutrition Workshop Series. Vol. 22. New York: Raven Press. pp. 55–63. ISBN 0-88167-695-0.
- ^ "Commentaries: Edwards A Park". Pediatrics. 44 (6). American Academy of Pediatrics: 897–901. 1969. doi:10.1542/peds.44.6.897. PMID 4903838. S2CID 43298798.
- ^ a b c d e f g h O'Hara, Kate (2016). "Paediatric pharmacokinetics and drug doses". Australian Prescriber. 39 (6): 208–210. doi:10.18773/austprescr.2016.071. ISSN 0312-8008. PMC 5155058. PMID 27990048.
- ^ a b c d e f g h i j k l m Wagner, Jonathan; Abdel-Rahman, Susan M. (2013). "Pediatric pharmacokinetics". Pediatrics in Review. 34 (6): 258–269. doi:10.1542/pir.34-6-258. ISSN 1526-3347. PMID 23729775.
- ^ a b c d e f Batchelor, Hannah Katharine; Marriott, John Francis (2015). "Paediatric pharmacokinetics: key considerations". British Journal of Clinical Pharmacology. 79 (3): 395–404. doi:10.1111/bcp.12267. ISSN 1365-2125. PMC 4345950. PMID 25855821.
- ^ Katz, Aviva L.; Webb, Sally A.; COMMITTEE ON BIOETHICS; Macauley, Robert C.; Mercurio, Mark R.; Moon, Margaret R.; Okun, Alexander L.; Opel, Douglas J.; Statter, Mindy B. (1 August 2016). "Informed Consent in Decision-Making in Pediatric Practice". Pediatrics. 138 (2): e20161485. doi:10.1542/peds.2016-1485. ISSN 0031-4005. PMID 27456510. S2CID 7951515.
- ^ a b Mazur, Kate A.; Berg, Stacey L., eds. (2020). Ethical Issues in Pediatric Hematology/Oncology. pp. 13–21. doi:10.1007/978-3-030-22684-8. ISBN 978-3-030-22683-1. S2CID 208302429.
- ^ a b Stern, Alexandra Minna; Markel, Howard (2002). Formative Years: Children's Health in the United States, 1880-2000. University of Michigan Press. pp. 23–24. doi:10.3998/mpub.17065. ISBN 978-0-472-02503-9. Archived from the original on 30 November 2021. Retrieved 30 November 2021.
- ^ Friedman, Lainie Ross (2004). Children, families, and health care decision making. Clarendon Press. ISBN 0-19-925154-1. OCLC 756393117.
- ^ a b c Martakis, K.; Schröder-Bäck, P.; Brand, H. (1 June 2018). "Developing child autonomy in pediatric healthcare: towards an ethical model". Archivos Argentinos de Pediatria. 116 (3): e401 – e408. doi:10.5546/aap.2018.eng.e401. ISSN 0325-0075. PMID 29756714. S2CID 46889502.
- ^ Schapiro, Tamar (1 July 1999). "What Is a Child?". Ethics. 109 (4): 715–738. doi:10.1086/233943. ISSN 0014-1704. S2CID 170129444. Archived from the original on 30 November 2021. Retrieved 30 November 2021.
- ^ Dondorp, W.; Bolt, I.; Tibben, A.; De Wert, G.; Van Summeren, M. (1 September 2021). "'We Should View Him as an Individual': The Role of the Child's Future Autonomy in Shared Decision-Making About Unsolicited Findings in Pediatric Exome Sequencing". Health Care Analysis. 29 (3): 249–261. doi:10.1007/s10728-020-00425-7. ISSN 1573-3394. PMID 33389383. S2CID 230112761.
- ^ "What Education Is Required to Be a Pediatrician?". Archived from the original on 7 June 2017. Retrieved 14 June 2017.
- ^ "CoPS". www.pedsubs.org. Archived from the original on 18 September 2013. Retrieved 14 August 2015.
Further reading
[edit]- BMC Pediatrics - open access
- Clinical Pediatrics
- Developmental Review - partial open access
- JAMA Pediatrics
- The Journal of Pediatrics - partial open access
External links
[edit]Pediatrics
View on GrokipediaDefinition and Scope
Core Principles and Age Demographics
Pediatrics operates on the principle that children's health requires a developmental perspective, accounting for rapid physiological changes, immature organ systems, and evolving cognitive and emotional capacities that differ markedly from adults, thereby demanding tailored diagnostic and therapeutic strategies focused on growth promotion and risk mitigation. Central tenets include preventive health measures, such as routine immunizations and screening for developmental delays, alongside anticipatory guidance for families to address foreseeable challenges in child-rearing. The American Academy of Pediatrics (AAP) underscores the attainment of optimal physical, mental, and social well-being through integrated care models that prioritize early intervention to avert long-term morbidity.[11][12] Family-centered and team-based approaches form foundational elements, involving collaboration among pediatricians, families, educators, and community resources to foster holistic outcomes, with pediatricians positioned as coordinators attuned to the child's continuum of needs from infancy onward. This framework rejects rigid silos between physical and psychosocial domains, emphasizing evidence-based practices like well-child surveillance to detect deviations from normative trajectories promptly.[12][13] Pediatric age demographics extend from periconceptional counseling through young adulthood, without arbitrary upper limits, particularly for individuals with chronic conditions necessitating prolonged specialized oversight; the AAP delineates care up to age 21 for adolescents, with extensions as clinically indicated. Standard classifications segment this span into developmental cohorts to guide age-appropriate interventions:| Age Group | Approximate Range |
|---|---|
| Neonate | Birth to 28 days |
| Infant | 1 to 12 months |
| Toddler/Early Childhood | 1 to 5 years |
| Middle Childhood/School-Age | 6 to 12 years |
| Adolescent | 13 to 21 years |
Distinctions from Adult Medicine
Pediatrics differs from adult medicine primarily due to the dynamic physiological immaturity and developmental changes in children, which alter disease susceptibility, presentation, and response to interventions compared to adults. Children's organ systems, such as the liver, kidneys, and central nervous system, are underdeveloped at birth and mature progressively, leading to variations in homeostasis, immune function, and metabolic processes; for instance, neonates exhibit higher total body water content (70-80% vs. 50-60% in adults) and immature glomerular filtration rates, affecting fluid balance and drug handling.[17][18] These differences result in distinct disease profiles, with children more prone to congenital anomalies, infectious etiologies, and rapid progression of conditions like respiratory distress, whereas adults face higher rates of degenerative and chronic diseases.[18][19] Pharmacokinetic and pharmacodynamic factors further necessitate specialized pediatric approaches, as drug absorption, distribution, metabolism, and elimination vary markedly by age. In neonates and infants, hepatic enzyme systems like cytochrome P450 are immature, prolonging drug half-lives (e.g., 3-9 times longer for certain medications compared to adults), while children aged 2-10 years often require 50% higher doses per body weight due to accelerated clearance and distribution volumes.[20][17] Adolescents may approximate adult dosing, but overall, pediatric formulations are limited, with over half of children receiving off-label prescriptions annually, underscoring the risks of extrapolating adult data.[21][22] These variations demand age-specific dosing algorithms and monitoring to avoid toxicity or inefficacy, distinct from the standardized adult regimens.[23] Clinical management in pediatrics emphasizes preventive strategies, growth surveillance, and family-centered care, contrasting with the autonomy-focused, symptom-driven model of adult medicine. Pediatric encounters integrate parental involvement in decision-making and consent, reflecting minors' legal status, while prioritizing milestones like vaccination schedules and developmental screenings over adult-centric endpoints like comorbidity management.[24] Disease presentations often lack classic adult symptoms—e.g., abdominal pain in pediatric appendicitis may be nonspecific—requiring heightened reliance on history, observation, and ancillary tests.[19] These systemic divergences justify separate residency training pathways, as internal medicine programs do not equip providers for pediatric-specific competencies like rapid physiological decompensation or ethical nuances in assent.[25][26]Historical Development
Etymology and Origins
The term pediatrics originates from the Ancient Greek words pais (παῖς), denoting "child," and iatros (ἰατρός), meaning "physician" or "healer," thus signifying "healer of children."[27] The adjectival form pediatric, referring to medical care or diseases of children, entered English in 1849 via a Latinized adaptation of the Greek stem paid- (from pais) combined with the suffix -iatric, which relates to medical treatment.[27] The nominal form pediatrics, designating the specialized branch of medicine, appeared by 1884, building on the earlier adjective to describe systematic study and treatment of childhood conditions.[28] Preceding English adoption, the term surfaced in German as pädiatrik in the early 19th century, reflecting neoclassical coinage in Continental European medical literature before wider dissemination.[29] Although informal care for children's ailments dates to ancient civilizations—such as Egyptian papyri from circa 1550 BCE documenting pediatric remedies and Hippocratic texts from the 5th century BCE addressing infant diseases—pediatrics emerged as a distinct medical specialty in the mid-19th century amid rising child mortality rates and advances in hospital-based care.[5] The first dedicated pediatric hospitals appeared in the early 1800s, including the Hôpital des Enfants-Malades in Paris (established 1802) and similar institutions in Italy and Germany, which separated child patients from adults to reduce infection risks and enable focused observation.[30] In the United States, Abraham Jacobi delivered the inaugural systematic lectures on childhood diseases at New York University in 1860, pioneering clinical approaches like physical diagnosis and establishing pediatrics as a professional domain independent of general practice.[6] This formalization coincided with epidemiological shifts, including urbanization and industrialization, which highlighted unique pediatric vulnerabilities such as nutritional deficiencies and infectious outbreaks, prompting specialized training and societies like the American Pediatric Society (founded 1888).[31] Early pediatric texts, such as Jacobi's Diseases of Children (1868), emphasized empirical observation over speculative adult extrapolations, laying groundwork for evidence-based differentiation.[5] By the late 19th century, the specialty's delineation owed less to theoretical innovation than to pragmatic responses to verifiable child-specific pathologies, including diphtheria and rickets, documented through autopsy data and vital statistics from emerging public health records.[32]Pre-Modern Practices
In ancient Egypt, child healthcare involved empirical treatments such as herbal poultices, honey-based remedies for infections, and incantations invoking deities like Sekhmet for healing, with evidence from Ebers Papyrus (c. 1550 BCE) describing remedies for childhood ailments like worms and burns.[33] Healers, often priests or midwives, performed basic surgeries using bronze tools and addressed malnutrition through dietary advice, though high infant mortality from diarrhea and respiratory issues persisted due to limited sanitation.[34] Classical Greek and Roman medicine recognized distinct vulnerabilities in children but lacked specialization. Hippocrates (c. 460–370 BCE) in his Aphorisms noted that children were prone to rapid disease progression due to immature humoral balance, recommending gentle purgatives and environmental adjustments over invasive methods.[35] Soranus of Ephesus (c. 98–138 CE), in On Midwifery and the Diseases of Women, provided detailed pediatric guidance, advocating proper swaddling to align limbs, exclusive breastfeeding for the first months, and avoiding overfeeding to prevent convulsions; he emphasized hygiene and rejected superstitious practices like amulets in favor of observation-based care.[35] Roman practices, influenced by Greeks, included wet-nursing for elite infants and rudimentary vaccinations via variolation against smallpox, though child mortality remained high from epidemics and poor weaning foods.[36] During the Islamic Golden Age, scholars advanced pediatric knowledge through translation and experimentation. Rhazes (Al-Razi, 865–925 CE) in Treatise on the Smallpox and Measles differentiated childhood infectious diseases by symptoms and prognosis, recommending isolation, cool cloths, and barley water for fever management, while stressing age-adjusted dosing to avoid toxicity in small bodies.[35] Avicenna (Ibn Sina, 980–1037 CE) in The Canon of Medicine classified pediatric conditions under humoral theory, advising maternal breastfeeding and gentle laxatives for infants, with empirical observations on teething fevers and rickets precursors.[37] In medieval Europe, child medicine blended Galenic humors with folk remedies, viewing children as incomplete adults requiring milder interventions. Physicians like Saliceto ([13th century](/page/13th century)) prescribed diluted herbal teas and baths for digestive issues, while family caregivers used poultices of onions or mustard for coughs; surgical texts rarely addressed pediatrics, focusing instead on adult anatomy.[38] Midwives handled most infant care, including cauterization for umbilical infections, but evidence from skeletal remains indicates frequent untreated fractures and nutritional deficiencies like scurvy in weaned children.[39] Church influence promoted charity for foundlings in hospices, yet systemic neglect contributed to mortality rates exceeding 50% before age five, with no standardized protocols.[39] By the 17th and 18th centuries, proto-pediatric texts emerged amid rising child mortality awareness from urbanization and wars. Daniel Whistler (1645) described rickets as a British disease linked to urban poor diets, advocating cod liver oil precursors; Nils Rosén von Rosenstein's Instructions for the Preservation of Children's Health (1764) systematized care, urging fresh air, inoculation against smallpox, and antispasmodic remedies for whooping cough, influencing hospital wards for children in London and Paris.[37] Treatments emphasized regimen—diet, exercise, and hygiene—over drugs, with mercury and opium used cautiously for convulsions, though iatrogenic harm from purging was common; foundling hospitals like London's (1741) provided institutional care but reported 80-90% mortality due to overcrowding and infection.[40] These practices reflected empirical gains but remained ad hoc, without formal certification or separation from general medicine.[41]19th and 20th Century Formalization
The formalization of pediatrics as a distinct medical specialty in the 19th century was marked by the establishment of dedicated children's hospitals and the recognition of unique pediatric pathologies separate from adult medicine. The first hospital exclusively for children, Hôpital Necker-Enfants Malades, opened in Paris in 1802, providing specialized inpatient care for pediatric patients and influencing subsequent European developments.[42] In Britain, Great Ormond Street Hospital, founded in 1852, became the first such institution in the English-speaking world, focusing on diseases prevalent in childhood and advancing clinical separation from general hospitals.[43] These facilities addressed high infant and child mortality rates, driven by infectious diseases and nutritional deficiencies, which empirical observations showed required tailored interventions not suited to adult wards.[6] In the United States, Abraham Jacobi, recognized as the founder of American pediatrics, advanced the field through systematic lectures on childhood diseases beginning in 1860 at New York University and the establishment of the first free pediatric clinic at Bellevue Hospital in 1874.[6] [44] Jacobi's work emphasized evidence-based diagnostics, such as the use of the stethoscope for infant auscultation, and advocated for public health measures like pasteurization to combat diarrheal diseases, which caused significant child mortality.[45] The American Pediatric Society, formed in 1888 under Jacobi's influence, provided a platform for research dissemination and professional standards, solidifying pediatrics' academic foundation.[46] Early 20th-century formalization involved institutionalizing training and certification to ensure competency in child-specific care. The American Academy of Pediatrics was established in 1930 to promote education, research, and preventive strategies amid declining infectious disease rates due to sanitation improvements.[47] The American Board of Pediatrics, created in 1933, introduced certification processes requiring residency training focused on growth, development, and age-dependent pharmacokinetics, distinguishing pediatric practice from general medicine.[48] Subspecialties, such as pediatric cardiology and endocrinology, began emerging in the 1930s through dedicated clinics in medical schools, enabling targeted expertise in congenital and developmental disorders.[48] These milestones reflected causal insights into children's physiological immaturity, including immature liver metabolism and immune responses, necessitating specialized protocols verified through clinical data.[31]Post-1945 Expansion and Specialization
Following World War II, pediatrics expanded rapidly due to breakthroughs in antibiotics, vaccines, and public health measures that drastically reduced infectious disease mortality among children. In the United States, the infant mortality rate declined from about 30 deaths per 1,000 live births in 1950 to 7.2 by 1997, reflecting over a 75% drop in that period alone, largely from penicillin's widespread adoption and sulfonamide drugs.[49] [50] This shift allowed pediatric care to move beyond acute survival toward managing chronic conditions, developmental disorders, and rare diseases, fostering institutional growth in children's hospitals and dedicated pediatric departments.[51] Subspecialization accelerated as general pediatric practice gave way to focused expertise, with academic centers prioritizing fellowship-trained specialists over generalists by the mid-20th century. The American Board of Pediatrics (ABP), established in 1933, began certifying subspecialties in 1961 with pediatric cardiology, marking the formal recognition of advanced training pathways typically requiring 2-3 years beyond general residency.[48] [52] Subsequent certifications followed for pediatric hematology-oncology, nephrology, and neonatal-perinatal medicine, with the latter's first examination offered in 1975 after the emergence of neonatal intensive care units (NICUs) in the 1960s equipped with ventilators and incubators for premature infants.[48] [53] Pediatric critical care formalized as a subspecialty from late-1950s innovations in mechanical ventilation and monitoring, while pediatric surgery established structured three-year training programs post-war under leaders like Robert E. Gross.[54] [55] By the 1970s, over a dozen pediatric subspecialties existed, including endocrinology, gastroenterology, and infectious diseases, supported by federal research funding and professional organizations like the American Academy of Pediatrics, which grew its membership amid rising demand for specialized care. This era saw pediatric workforce expansion, with U.S. pediatricians increasing from fewer than 5,000 in 1940 to over 20,000 by 1970, enabling multidisciplinary teams in areas like oncology and neurology.[6] Globally, similar trends emerged, with the World Health Organization promoting child health initiatives that integrated specialized interventions into primary care frameworks.[30]Biological and Pharmacological Differences
Growth, Maturation, and Organ System Variations
Growth in children occurs in distinct phases characterized by varying velocities influenced by genetic, nutritional, and hormonal factors. During infancy, linear growth is rapid, with length increasing by about 25 cm in the first year, while weight typically triples from birth weight by 12 months.[56] In early childhood up to age 4 years, growth velocity averages 5-6 cm per year in height and 2.5 kg per year in weight, with girls initially growing slightly faster than boys.[57] From ages 2 to puberty, annual height gains stabilize at 5-8 cm and weight at 2-3 kg, reflecting steady somatic development.[58] Pubertal growth features a peak height velocity of approximately 9.5 cm/year in boys and 8.3 cm/year in girls, contributing an average total pubertal height gain of 31 cm in boys and 29 cm in girls, driven by sex steroid surges.[59] Maturation encompasses progressive organ system refinement and secondary sexual development, extending brain structural changes into early adulthood. Puberty onset, marked by gonadarche around ages 8-13 in girls and 9-14 in boys, triggers not only the growth spurt but also gonadal maturation and skeletal remodeling, with peak velocities preceding menarche by 6-12 months in females.[60] Brain development involves rapid synaptogenesis in infancy followed by pruning and myelination, with the prefrontal cortex maturing last during adolescence, enhancing executive functions like impulse control; by age 3, brain volume reaches 80% of adult size, and by age 5-6, 90-95%.[61] This protracted neural maturation, influenced by sex hormones, underlies differences in cognitive processing compared to adults, where children recruit more diffuse brain regions for tasks.[62] Organ system variations stem from anatomical proportions, higher metabolic demands, and incomplete physiological maturity relative to adults. The central nervous system features a disproportionately large head and brain in infants, with higher plasticity but vulnerability to injury due to thinner skulls and incomplete myelination.[63] Cardiovascularly, children maintain higher heart rates (neonates 120-160 bpm vs. adult 60-100 bpm) and stroke volumes to meet elevated oxygen demands from greater body surface area-to-mass ratios and metabolic rates up to twice that of adults.[64] Respiratory systems exhibit higher baseline rates (infants 30-60 breaths/min), smaller airways prone to obstruction, and more compliant rib cages leading to reliance on diaphragmatic breathing, increasing risks in distress.[65] Renal function matures gradually, with neonatal glomerular filtration rates at 30-50% of adult levels, rising to adult capacity by 2 years, affecting fluid-electrolyte balance and drug clearance.[66] Hepatic enzyme systems, particularly cytochrome P450, remain underdeveloped in neonates and infants, prolonging drug half-lives compared to adults.[67] The immune system operates differently rather than being inherently deficient, with newborns relying on alternative T-cell activation pathways for efficient responses to certain pathogens, though with reduced adaptive memory initially.[68][69] These variations necessitate age-specific pediatric approaches, as adult physiology assumptions can lead to suboptimal outcomes.[70]Pharmacokinetic and Pharmacodynamic Factors
In pediatric populations, pharmacokinetic processes—encompassing drug absorption, distribution, metabolism, and elimination—differ markedly from those in adults due to ongoing physiological maturation, particularly in neonates and infants. Neonates exhibit higher gastric pH (around 6-8 compared to 1-3 in adults), reduced gastric emptying, and immature intestinal motility, leading to slower and more variable oral absorption of drugs.[71] These factors necessitate formulation adjustments, such as liquid preparations, to enhance bioavailability, as evidenced by studies showing up to 50% lower absorption rates for certain acidic drugs in newborns.[20] Distribution volumes are altered by age-specific body composition: newborns have 70-80% total body water (versus 50-60% in adults) and lower adipose tissue (10-15% body weight versus 20-30%), resulting in larger volumes of distribution for hydrophilic drugs like aminoglycosides, often requiring higher loading doses.[72] Plasma protein binding is reduced in infants due to lower albumin levels (20-30 g/L versus 35-50 g/L in adults), increasing free drug fractions and potential toxicity for highly bound agents like phenytoin.[20] By adolescence, these parameters approach adult norms, but transitional changes from infancy to childhood demand age-stratified dosing. Hepatic metabolism matures unevenly; phase I cytochrome P450 (CYP) activity, such as CYP3A4, is 30-50% of adult levels at birth, surging to exceed adult capacity by ages 2-5 years before stabilizing.[23] Glucuronidation pathways, critical for drugs like morphine, remain immature until 2-3 years, prolonging half-lives in young children and necessitating dose reductions—e.g., neonatal morphine clearance is 50% lower than in adults.[72] This ontogeny explains why children aged 2-10 often require 50% higher mg/kg doses than adults for CYP-metabolized drugs to achieve equivalent exposure.[17] Renal elimination, responsible for 20-30% of drug clearance in pediatrics, is limited at birth with glomerular filtration rates (GFR) at 30-40% of adult values (around 30 mL/min/1.73 m²), maturing to adult levels (100-120 mL/min/1.73 m²) by 1-2 years.[23] Tubular secretion and reabsorption are also underdeveloped, prolonging half-lives for renally excreted drugs like gentamicin, where neonatal dosing intervals extend to 36-48 hours versus 8-12 hours in adults.[73] In preterm infants, GFR can be as low as 10-20 mL/min/1.73 m², amplifying risks of accumulation and nephrotoxicity.[74] Pharmacodynamic responses in children generally mirror adults for most drugs, with efficacy tied to plasma concentrations, but developmental variations in receptor density and signaling can alter sensitivity—e.g., increased benzodiazepine responsiveness in neonates due to immature GABA receptors.[75] True ontogenic PD differences are less common than PK-driven effects, though stage-specific responses occur, such as heightened chemotherapy toxicity in rapidly dividing pediatric cells.[20] Therapeutic drug monitoring and population-specific trials are essential, as adult-derived PD indices often overestimate pediatric efficacy, contributing to off-label prescribing in 70-80% of inpatient cases.[76] These factors underscore the need for extrapolation models incorporating maturation functions, as validated in FDA pediatric guidelines.[77]Professional Education and Training
Prerequisites and Medical Education Pathway
To enter the medical education pathway leading to a career in pediatrics, candidates must first complete an undergraduate bachelor's degree, typically spanning four years, while fulfilling pre-medical prerequisites that prepare them for the scientific and analytical demands of medical training. These prerequisites, which vary slightly by medical school but follow standard guidelines, generally include two semesters each of biology (with lab), general chemistry (with lab), organic chemistry (with lab), and physics (with lab), plus one semester of biochemistry, college-level mathematics (such as calculus or statistics), English or writing-intensive courses, and behavioral sciences like psychology and sociology.[78][79] No specific undergraduate major is required, though science-related fields predominate among successful applicants, allowing flexibility for interdisciplinary interests.[80] Competitive undergraduate performance is essential, with average GPAs for 2024 U.S. medical school matriculants at 3.77 overall and 3.71 in biology, chemistry, physics, and mathematics (BCPM) courses.[81] Admission to medical school hinges on a holistic review, including the Medical College Admission Test (MCAT), a standardized exam evaluating mastery of natural, behavioral, and social sciences, as well as critical analysis and reasoning skills. The average MCAT score for 2024 matriculants was approximately 511.9, with scores below 500 correlating to significantly lower acceptance rates.[82] Additional factors include letters of recommendation, a personal statement articulating motivation—ideally demonstrating early interest in child health through shadowing physicians or volunteering with pediatric populations—and evidence of extracurricular involvement, such as research or community service, to evidence attributes like empathy, resilience, and commitment to service.[80][83] International medical graduates must attend schools recognized by bodies like the World Federation for Medical Education to later qualify for U.S. residency, though domestic LCME- or COCA-accredited programs are standard for MD or DO degrees.[84] Medical school itself requires four years of rigorous training, divided into preclinical and clinical phases, accredited by the Liaison Committee on Medical Education (LCME) for allopathic (MD) programs or the Commission on Osteopathic College Accreditation (COCA) for osteopathic (DO) programs—both pathways equally eligible for pediatric specialization. The first two years emphasize didactic coursework in biomedical sciences (e.g., gross anatomy, histology, microbiology, immunology, and pharmacology) and foundational clinical skills like patient interviewing and basic procedures, building causal understanding of disease mechanisms relevant to pediatric vulnerabilities, such as developmental immunology.[84] The latter two years shift to clinical clerkships, where students rotate through specialties under supervision; pediatrics is a required core clerkship, usually 4 to 8 weeks in duration during the third year, encompassing inpatient care (e.g., newborn nurseries, general pediatrics wards), outpatient clinics, and subspecialty exposures like neonatology or adolescent medicine to develop competencies in age-specific diagnostics, family-centered communication, and preventive counseling.[85][86] Students aspiring to pediatrics often pursue additional electives or away rotations to strengthen residency applications, accumulating empirical exposure to conditions like congenital anomalies or infectious diseases prevalent in children.[87] Upon completing medical school and passing the United States Medical Licensing Examination (USMLE) Step 1 and Step 2 (or equivalent COMLEX for DOs), graduates receive their degree and qualify to apply for pediatric residency via the National Resident Matching Program, marking the transition to postgraduate training focused on child-specific practice.[88] This pathway, totaling about eight years from undergraduate entry to medical school graduation, demands sustained academic rigor and practical preparation to address pediatrics' emphasis on longitudinal growth monitoring and family dynamics over acute adult interventions.[89]Residency, Fellowships, and Certification Processes
Pediatric residency training in the United States typically spans three years following completion of medical school, with programs accredited by the Accreditation Council for Graduate Medical Education (ACGME).[90] Trainees progress through postgraduate year 1 (PGY-1), PGY-2, and PGY-3 levels, each requiring 12 months of full-time clinical experience in an ACGME-approved program.[84] Core rotations emphasize general inpatient and outpatient pediatrics, newborn care, emergency medicine, subspecialty exposures, and community pediatrics, with updated ACGME requirements effective July 2025 mandating at least 40 weeks of inpatient training, including a minimum of 16 weeks in general pediatrics or pediatric hospital medicine.[91] Successful completion qualifies graduates for initial certification in general pediatrics by the American Board of Pediatrics (ABP).[92] Fellowships provide advanced subspecialty training beyond residency, generally lasting three years for most disciplines such as pediatric cardiology, endocrinology, or neonatology, though durations vary (e.g., two years for some like sports medicine).[93] ABP eligibility for subspecialty certification requires prior ABP certification or active pursuit in general pediatrics, completion of an ACGME-accredited fellowship, active medical licensure, and demonstration of scholarly activity, such as original research or quality improvement projects.[94] Training emphasizes clinical expertise, research, and teaching, with fast-tracking options available for highly qualified candidates allowing combined residency-fellowship pathways under specific waivers.[95] Fellowships are competitive, often selected via the National Resident Matching Program, and prepare physicians for roles in academic medicine, specialized clinical practice, or research.[96] Certification processes are overseen by the ABP, which administers initial exams for general pediatrics and subspecialties. For general pediatrics, candidates must complete residency, hold an unrestricted license, and pass a computer-based certifying examination consisting of approximately 330-350 multiple-choice questions over seven hours, offered annually.[97] Subspecialty certification follows fellowship completion and involves a similar secure exam format tailored to the discipline, with admission requiring verification of training and scholarly output.[93] Initial certifications are time-limited, necessitating Maintenance of Certification (MOC) every three to ten years, involving lifelong learning, assessments, and practice improvement activities, though the focus here is on entry-level processes.[98] These standards ensure competency in child health care, with ABP emphasizing evidence-based training amid evolving pediatric needs.[99]Ongoing Professional Development
Ongoing professional development for pediatricians primarily occurs through the Maintenance of Certification (MOC) program administered by the American Board of Pediatrics (ABP), which mandates a structured, cyclical process every five years to ensure lifelong learning, self-assessment, and clinical improvement among board-certified practitioners.[100] This framework, adopted across ABMS member boards, emphasizes continuous engagement with evolving pediatric knowledge rather than one-time certification, addressing rapid advancements in child health from neonatal care to adolescent medicine.[101] The MOC process comprises four interdependent components: Part 1 requires maintenance of a valid, unrestricted medical license to uphold professional standing; Part 2 involves completing lifelong learning activities, such as approved continuing medical education (CME) credits and self-assessment modules tailored to pediatrics; Part 3 assesses cognitive expertise via secure exams administered every 10 years; and Part 4 focuses on practice improvement through quality improvement projects and performance assessments.[102] [101] Compliance with these elements is tracked online via the ABP's MOCA-Peds platform, allowing pediatricians to earn credits for activities like journal-based learning or multimedia modules on topics such as infectious diseases or developmental disorders.[100] CME requirements integrate with MOC, with board-certified pediatricians needing to accumulate sufficient Category 1 credits—often 75 to 100 hours per three-year cycle, depending on state licensure mandates—to fulfill Part 2 obligations, many of which overlap with general medical licensing renewals that vary by jurisdiction (e.g., 40 hours biennially in Florida, including pediatric-specific content).[103] [104] The American Academy of Pediatrics (AAP) endorses and facilitates this by offering accredited resources through PediaLink, including online courses, webinars, and annual conferences like the AAP National Conference, which provide credits for updates on evidence-based guidelines in areas such as vaccination protocols and chronic disease management.[105] [106] Beyond formal MOC, pediatricians engage in voluntary professional development via AAP-supported initiatives, peer-reviewed journals like Pediatrics, and interdisciplinary workshops to address emerging challenges, such as genomic advancements or mental health integration in primary care, fostering adaptation to demographic shifts like rising childhood obesity rates documented at 19.7% in U.S. youth aged 2-19 as of 2020 CDC data.[107] Failure to maintain MOC can result in loss of certification status, underscoring its role in verifying competence amid critiques that such programs impose administrative burdens without proportionally enhancing patient outcomes, though ABP data indicate high participation rates exceeding 90% among eligible diplomates.[100][102]Clinical Practices and Interventions
Diagnostic and Therapeutic Approaches
Diagnostic approaches in pediatrics prioritize a comprehensive history obtained primarily from caregivers, emphasizing prenatal and perinatal events, developmental milestones, immunization status, and social determinants of health, as children often cannot articulate symptoms reliably.[108] The physical examination adapts to the child's age and temperament, typically proceeding from least to most invasive maneuvers—such as observing general appearance, vital signs, and anthropometric measurements (weight, length/height, head circumference, and body mass index plotted on age- and sex-specific growth charts from the World Health Organization or Centers for Disease Control and Prevention)—before proceeding to auscultation, palpation, and orifices to minimize distress and ensure cooperation.[109] For infants, examinations often occur on the table with full exposure while keeping the child warm; toddlers may be examined on a parent's lap, integrating play to build rapport.[108] Ancillary diagnostics, including laboratory tests, imaging, and specialized evaluations like genetic testing for developmental delays, follow evidence-based guidelines from bodies such as the American Academy of Pediatrics (AAP), with protocols minimizing risks like radiation exposure in children due to their greater lifetime cancer susceptibility.[110] Diagnostic error rates in pediatrics range from 10-20% in ambulatory settings, often stemming from atypical presentations, communication barriers, and cognitive biases, underscoring the need for systematic checklists and multidisciplinary input.[111] Therapeutic interventions in pediatrics account for ontogenic changes in physiology, where immature organ systems—such as reduced glomerular filtration rates in neonates and variable hepatic metabolism—affect drug absorption, distribution, metabolism, and excretion compared to adults, necessitating weight- or body surface area-based dosing and pediatric-specific formulations like elixirs or chewables to enhance palatability and adherence.[112] Evidence-based treatments adhere to AAP clinical practice guidelines, which classify recommendations by evidence strength; for instance, acute otitis media management may involve observation without antibiotics for non-severe cases in children over 2 years to curb antimicrobial resistance, while severe infections warrant prompt pharmacotherapy.[113] Non-pharmacologic approaches, including behavioral therapies like cognitive-behavioral interventions for chronic pain or attention-deficit/hyperactivity disorder, demonstrate moderate to strong efficacy in reducing symptoms and disability when tailored to developmental stages, often involving family education to foster long-term compliance.[114] Sedation for procedures follows AAP-monitored guidelines prioritizing safety, with goals of maintaining airway patency and minimizing respiratory depression, using agents like midazolam or propofol titrated by age and weight.[115] Overall, pediatric therapeutics emphasize holistic, family-centered care, weighing immediate symptom relief against potential impacts on growth, neurodevelopment, and future health, with only 10.6% of AAP recommendations deriving from high-quality evidence like randomized controlled trials, highlighting ongoing needs for pediatric-specific research.[116]Preventive Medicine and Public Health Measures
Preventive medicine in pediatrics centers on evidence-based interventions delivered during well-child visits to promote health, detect issues early, and avert diseases and injuries. The American Academy of Pediatrics (AAP), through its Bright Futures initiative, outlines periodicity schedules recommending health supervision visits at birth, 3-5 days, 1 month, 2 months, 4 months, 6 months, 9 months, 12 months, 15 months, 18 months, 2 years, and annually from 3 to 21 years, incorporating anticipatory guidance on nutrition, behavior, and safety.[117] These visits facilitate universal screenings for developmental milestones, maternal depression, substance exposure, and social determinants of health, with targeted tools like the Ages and Stages Questionnaire for early identification of delays.[118] Immunization schedules represent a primary public health tool, targeting diseases such as measles, pertussis, polio, and Haemophilus influenzae type b. The U.S. Centers for Disease Control and Prevention (CDC) routine childhood vaccinations for children born between 1994 and 2023 averted approximately 508 million cases of illness, 32 million hospitalizations, and 1.1 million deaths, yielding a net societal benefit of $540 billion in direct costs and $2.8 trillion in societal costs.[119] Compliance with the schedule, which includes vaccines like DTaP, MMR, and varicella by age 2, correlates with reduced outbreak incidence, as evidenced by post-licensure surveillance data showing herd immunity thresholds met in high-uptake populations.[120] Screenings for vision, hearing, anemia, lead exposure, and dyslipidemia occur at specified ages, with AAP guidelines endorsing universal lipid screening between 9-11 and 17-21 years to identify familial hypercholesterolemia early.[121] Injury prevention counseling, such as promoting rear-facing car seats until at least 2 years and booster seats until 4 feet 9 inches tall, has demonstrated effectiveness; proper restraint use reduces infant death risk by 71% and serious injury by 70% in crashes.[122] AAP's The Injury Prevention Program (TIPP), implemented in primary care, increased safety practices like smoke detector use and poison control access, reducing injury rates by up to 25% in participating cohorts.[123] Public health measures extend to nutrition and environmental interventions, including fluoride varnish applications starting at eruption of first tooth to prevent caries, which affect 23% of U.S. children aged 2-5, and promotion of exclusive breastfeeding for 6 months to lower obesity and infection risks.[124] Obesity screening via BMI plotting at every visit addresses rising prevalence, with counseling on diet and activity yielding modest sustained weight reductions in randomized trials.[125] Community-level efforts, such as school-based vaccination mandates and lead abatement programs, further amplify individual preventive strategies by mitigating population risks.[121]Recent Technological and Therapeutic Advances
In gene therapy, a landmark advancement occurred in May 2025 when an infant with a rare, incurable genetic disorder became the first patient successfully treated with personalized CRISPR-based gene editing, delivered via a customized adeno-associated virus vector, resulting in sustained therapeutic effects without severe adverse events.[126] This built on prior approvals, such as the FDA's June 2023 authorization of ELEVIDYS for ambulatory children aged 4-5 with Duchenne muscular dystrophy, marking the first gene therapy for that condition via single intravenous infusion.[127] Clinical trials have also shown long-term benefits in pediatric cerebral adrenoleukodystrophy, with a 2024 study of 32 patients demonstrating that most remained free of major disabilities six years post-therapy.[128] Advances in AAV-mediated replacement therapy continue for monogenetic pediatric diseases, with preclinical and early-phase trials emphasizing improved vector efficiency and reduced immunogenicity.[129] Chimeric antigen receptor (CAR) T-cell therapy has expanded for refractory pediatric B-cell acute lymphoblastic leukemia (ALL), achieving remission rates of 70-90% in children and young adults, as reported in multiple trials since the FDA's 2017 approval of the first such therapy.[130] A 2025 phase 2 trial (PLAT-02/03) using CD19-targeted CAR T-cells yielded leukemia-free survival exceeding 90% at one year in low-burden disease groups.[131] Ongoing research addresses challenges like cytokine release syndrome and neurotoxicity, with pediatric-specific protocols improving tolerability, while trials explore applications in solid tumors and autoimmune conditions.[132] [133] Artificial intelligence (AI) integration has enhanced pediatric diagnostics and treatment personalization, with machine learning algorithms achieving high accuracy in identifying conditions like fractures on X-rays and rare diseases from genomic data.[134] [135] In 2025, initiatives like Mount Sinai's Center for AI in Children's Health focused on AI-driven imaging analysis and predictive modeling to optimize outcomes in youth.[136] AI tools also support precision medicine by analyzing patient data for targeted therapies, though experts emphasize needs for trustworthy, pediatric-specific validation to mitigate biases.[137] [138] Telemedicine adoption surged post-COVID-19, with pediatric subspecialty visits increasing dramatically during lockdowns and sustaining higher utilization thereafter, often matching or exceeding in-person efficacy for consultations and follow-ups.[139] [140] By 2025, platforms enabled remote monitoring and expert consultations, reducing barriers in rural or underserved areas, though disparities persist in access across demographics.[141] Innovations in AI-enhanced telehealth further predict clinical courses and automate documentation, alleviating clinician burden.[142] Pediatric MRI protocols advanced in 2025 through AI-optimized imaging, enabling faster scans and linking brain development to genetic-environmental factors in large cohorts, surpassing traditional diagnostics.[143] These developments collectively prioritize empirical efficacy, with trials underscoring durable remissions and reduced invasiveness, though long-term safety data remain under scrutiny in pediatric populations.[144]Subspecialties
Core Subspecialties in Pediatrics
The core subspecialties in pediatrics involve advanced fellowship training beyond general pediatrics residency, focusing on specific organ systems, high-acuity conditions, or developmental stages in children, with certification offered by the American Board of Pediatrics (ABP) following rigorous examinations and maintenance of general pediatrics certification.[94] As of 2025, the ABP recognizes 17 primary pediatric subspecialties, each typically requiring 3 years of accredited fellowship training, enabling physicians to manage complex cases such as congenital disorders, chronic illnesses, and acute emergencies that demand specialized expertise unavailable in primary care settings.[145] [93] These fields emerged from the recognition that pediatric physiology differs markedly from adult medicine, necessitating targeted interventions based on growth, developmental vulnerabilities, and disease etiologies unique to youth.[146] Prominent core subspecialties address systemic or organ-specific pathologies, with neonatology handling over 400,000 preterm births annually in the U.S. alone, where survival rates for infants under 1,000 grams have improved to approximately 70% due to advances in ventilatory support and surfactant therapy.[147]- Neonatal-Perinatal Medicine: Focuses on the medical care of newborns, especially premature or ill infants, addressing issues like respiratory failure, sepsis, and intraventricular hemorrhage through intensive care unit management.[147]
- Pediatric Cardiology: Manages congenital and acquired heart diseases, including structural defects like ventricular septal defects affecting 2-4 per 1,000 live births, utilizing echocardiography, catheterizations, and surgical collaborations.[147] [148]
- Pediatric Endocrinology: Treats hormonal imbalances such as type 1 diabetes, which has an incidence of 15-20 per 100,000 children annually, involving insulin regimens, growth hormone therapy, and thyroid disorder management.[147]
- Pediatric Gastroenterology: Diagnoses and manages digestive tract disorders like inflammatory bowel disease and celiac disease, with procedures including endoscopies for conditions impacting up to 1% of children.[147]
- Pediatric Hematology-Oncology: Addresses blood disorders and malignancies, such as acute lymphoblastic leukemia comprising 75% of childhood cancers with 5-year survival rates exceeding 90% due to multi-agent chemotherapy protocols.[147]
- Pediatric Infectious Diseases: Specializes in complex infections like multidrug-resistant tuberculosis or congenital infections, guiding antimicrobial stewardship amid rising antimicrobial resistance rates reported at 20-50% in pediatric pathogens.[147]
- Pediatric Nephrology: Manages kidney diseases including chronic kidney disease stage 5 in children at a prevalence of 13 per million, involving dialysis, transplants, and electrolyte homeostasis.[147]
- Pediatric Pulmonology: Treats respiratory conditions such as cystic fibrosis affecting 1 in 3,500 births, employing pulmonary function tests, inhaled therapies, and ventilator support.[147]
- Pediatric Rheumatology: Handles autoimmune and inflammatory disorders like juvenile idiopathic arthritis impacting 16-150 per 100,000 children, using immunomodulators and biologics to control joint damage and systemic inflammation.[147]
Emerging and Interdisciplinary Subspecialties
Pediatric hospital medicine represents a key emerging subspecialty, emphasizing comprehensive inpatient care for children from birth through adolescence, including coordination of diagnostics, treatments, and family-centered discharge planning. The field originated from the broader hospitalist movement in the late 1990s, with the first dedicated fellowships established in 2003 and formal recognition by the American Board of Pediatrics (ABP) in 2016 to meet rising demands for specialized hospital-based expertise amid increasing pediatric admissions.[151][152] The inaugural ABP certification examination occurred in 2020, following a practice pathway that concluded in 2024, with fellowship training typically lasting two to three years and focusing on quality improvement, sedation, and procedures.[153][154] This subspecialty intersects with core areas like critical care and infectious diseases, enabling multidisciplinary teams to reduce readmissions, which affected 20-30% of pediatric inpatients in studies prior to its formalization.[155] Clinical informatics in pediatrics constitutes an interdisciplinary subspecialty bridging medicine, data science, and technology to enhance electronic health records, decision support systems, and population health analytics tailored to pediatric populations. Recognized by the American Board of Medical Specialties in 2011, it requires pediatricians to complete two-year ACGME-accredited fellowships emphasizing applied informatics, such as optimizing algorithms for pediatric dosing and predictive modeling for rare diseases.[156][157] Programs at institutions like Children's Hospital of Philadelphia integrate rotations in software development and regulatory compliance, addressing challenges like age-specific data privacy under HIPAA and the integration of genomic data into clinical workflows.[158] This field has expanded in the 2020s due to electronic health record adoption rates exceeding 90% in U.S. pediatric hospitals, enabling real-time surveillance of outbreaks and personalized medicine applications.[159] Pediatric hospice and palliative medicine, an interdisciplinary domain combining pediatrics with psychology, ethics, and social services, focuses on symptom management and quality-of-life support for children with life-limiting conditions, such as advanced cancers or genetic disorders. Certified through ABP pathways since the early 2010s, it involves one-year fellowships post-pediatrics residency, with practitioners managing teams that include chaplains and therapists to address holistic needs, including bereavement care for families.[157] Enrollment in U.S. programs grew by over 50% from 2015 to 2020, driven by evidence that early palliative integration reduces end-of-life hospitalizations by 30-40% in eligible cohorts.[160] Similarly, pediatric movement disorders neurology, a nascent subspecialty within child neurology, applies interdisciplinary insights from genetics, pharmacology, and rehabilitation to diagnose and treat conditions like dystonia and tics, with dedicated fellowships emerging post-2020 and intersecting with developmental pediatrics for early interventions.[161] These areas reflect pediatrics' adaptation to complex, technology-driven care models, though workforce projections indicate potential shortages in informatics and palliative roles by 2040 absent increased training slots.[162]Ethical, Legal, and Autonomy Issues
Balancing Parental Authority and Child Best Interests
In pediatric medicine, parents or legal guardians hold primary authority to consent to or refuse treatments on behalf of their minor children, with the presumption that they act in the child's best interests based on their intimate knowledge of the family context and values. This authority stems from the legal recognition of parental autonomy in child-rearing, tempered by the state's parens patriae doctrine, which empowers courts to intervene when parental decisions pose substantial risk of serious harm or death to the child.[163][164] Courts apply a "best interests" standard, weighing factors such as the child's prognosis, treatment efficacy, potential burdens, and parental reasoning, but intervention requires clear evidence that refusal constitutes medical neglect rather than mere disagreement with providers.[165][166] Physicians encountering parental refusal of recommended care, such as for life-threatening conditions like leukemia or bacterial meningitis, must first attempt education and negotiation to align decisions with evidence-based outcomes, documenting discussions thoroughly to demonstrate informed refusal. The American Academy of Pediatrics (AAP) advises escalating to ethics consultations or child protective services only when imminent harm is evident, as in cases of withheld antibiotics for sepsis, where delay can result in mortality rates exceeding 20% without intervention. For non-emergent refusals, such as vaccinations, the AAP endorses continued care with monitoring rather than immediate dismissal, recognizing that most parental hesitancy arises from misinformation rather than intent to harm, though legal mandates in 45 U.S. states allow school-entry requirements with limited exemptions.[167][168][169] Judicial overrides occur infrequently but decisively in acute scenarios, as in Jehovah's Witnesses' refusals of blood transfusions, where U.S. courts have ordered treatment in over 90% of reported cases since the 1960s, citing survival rates near 100% with transfusion versus high fatality without. High-profile disputes, such as the 2017 Charlie Gard case in the UK, illustrate tensions: parents sought experimental nucleoside therapy for their infant with mitochondrial disease, but courts ruled against it after expert testimony deemed it futile, prioritizing avoidance of prolonged suffering over unproven interventions; the child died shortly after life support withdrawal. Empirical data on outcomes remain sparse due to rarity, but reviews of state interventions for medical neglect indicate improved short-term survival in intervened cases (e.g., 85-95% for curable infections), though long-term family disruption and parental alienation can occur without post-intervention support.[170][171][172] Critics argue that expansive "best interests" applications risk eroding parental rights, particularly when cultural or religious beliefs underpin refusals, as empirical thresholds for "significant harm" vary by jurisdiction—U.S. courts often require near-certainty of death, while some European systems emphasize quality-of-life metrics. Studies of neglect interventions show that multidisciplinary approaches, including family therapy, yield better adherence and child welfare than adversarial court actions alone, with recurrence rates dropping 40-60% when education addresses root causes like distrust in medical systems. Ultimately, balancing preserves parental primacy except where causal evidence demonstrates irreversible detriment, aligning with first-principles prioritization of the child's survival and development over ideological impositions.[173][172][174]Consent, Assent, and Decision-Making Frameworks
In pediatric practice, informed consent for medical treatment and research involving minors is typically obtained from parents or legal guardians, as children under the age of majority—generally 18 in the United States—lack the legal capacity to provide binding consent.[175] This requirement stems from common law principles and federal regulations, such as 45 CFR 46 for research, which mandate parental permission to protect vulnerable populations while allowing necessary interventions.[176] The American Academy of Pediatrics (AAP) emphasizes that parental permission must be informed, voluntary, and based on a clear explanation of risks, benefits, and alternatives, aligning with ethical obligations to respect family authority in promoting the child's welfare.[177] Child assent complements parental permission by involving the minor in decision-making to the extent of their developmental capacity, fostering autonomy without overriding parental rights. The AAP recommends seeking assent from children aged 7 years and older who can comprehend basic information, with mandatory assent for research participants aged 7 to 18 under federal guidelines unless waived for minimal risk studies.[163] Assent involves age-appropriate discussions of procedures, potential harms, and the right to withdraw, documented in medical records to ensure the process respects the child's emerging reasoning abilities.[177] This practice, updated in AAP policy as of 2016, acknowledges cognitive maturation but prioritizes protection from undue influence or coercion.[163] Exceptions to standard parental consent arise under doctrines like the mature minor rule, recognized in most U.S. states, which permits adolescents—typically 14 to 17 years old—who demonstrate sufficient understanding and maturity to consent independently to certain treatments, such as routine care or emergencies.[178] Emancipated minors, defined by state statutes as those married, in the military, or living independently with financial self-sufficiency, hold full consent authority equivalent to adults.[179] These frameworks vary by jurisdiction; for instance, some states statutorily limit mature minor application to non-controversial interventions, reflecting judicial balancing of autonomy against parental oversight.[180] Decision-making frameworks prioritize the child's best interests standard, where parents act as surrogates evaluating medical options based on evidence of health outcomes, potential harms, and quality-of-life factors, rather than solely familial or ideological preferences.[181] Courts intervene when parental refusals endanger the child, as in cases of life-saving treatments like chemotherapy or transfusions, applying substituted judgment or strict scrutiny to override decisions not aligned with objective medical evidence.[181] Internationally, similar principles appear in the UK's Gillick competence test, allowing children under 16 to consent if they grasp the treatment's implications, though parental involvement remains ethically encouraged.[182] These standards ensure decisions are evidence-driven, with empirical data on prognosis guiding resolutions over subjective assessments.[163]End-of-Life and Resource Allocation Dilemmas
In pediatric medicine, end-of-life decisions often involve withholding or withdrawing life-sustaining treatments for infants and children with irreversible conditions, such as severe congenital anomalies or progressive neurodegenerative diseases, guided by principles of beneficence and non-maleficence.[183] These decisions typically require multidisciplinary input from physicians, ethicists, palliative care specialists, and parents, emphasizing shared decision-making to align with the child's best interests, though empirical data indicate that conflicts arise in approximately 20-30% of cases in neonatal and pediatric intensive care units (ICUs).[184] For instance, in a 2021 review of pediatric oncology end-of-life care, bioethical frameworks stressed evaluating treatment futility based on evidence of net harm, such as prolonged suffering without realistic prognosis for meaningful recovery, rather than solely parental preferences.[185] High-profile cases illustrate tensions between parental autonomy and institutional judgments. In the 2017 Charlie Gard case, a UK infant with mitochondrial DNA depletion syndrome faced court intervention when Great Ormond Street Hospital deemed further treatment futile after seizures caused irreversible brain damage; despite parental efforts to pursue experimental nucleoside therapy abroad, the European Court of Human Rights upheld withdrawal, citing lack of evidence for benefit and potential harm, leading to Gard's death on July 28, 2017, without the intervention.[171] This outcome underscored the "best interests" standard in UK law, which prioritizes objective medical prognosis over parental rights when treatments offer no realistic chance of improvement, though critics argued it undervalued experimental options supported by some international experts.[186] Similar dynamics appeared in the 2018 Alfie Evans case, where Liverpool courts authorized treatment cessation for a child with an undiagnosed mitochondrial disorder, rejecting parental transfer requests; Evans died shortly after ventilator withdrawal, highlighting how judicial overrides can resolve disputes but may erode trust in systems perceived as biased toward resource conservation.[187] Resource allocation dilemmas intensify these challenges, particularly in neonatal ICUs where preterm infants at the limits of viability (e.g., 22-24 weeks gestation) compete for ventilators, incubators, and staff amid finite capacities. Ethical guidelines advocate frameworks assessing likelihood of survival to discharge and potential for intact neurological function, rejecting simplistic age- or disability-based rationing; a 2023 analysis of periviable care emphasized professional responsibility to avoid reductionist rights claims, instead weighing empirical outcomes like 50-70% survival rates for 23-week infants with significant morbidity risks.[188] Moral distress among providers is prevalent, with surveys reporting it stems from periviable resuscitations and end-of-life withdrawals, as decisions balancing one infant's intensive needs against others' access can lead to burnout rates exceeding 40% in NICU staff.[189] Pandemic scenarios exacerbate scarcity, as seen during COVID-19 when U.S. triage protocols varied regionally; some penalized infants with disabilities by excluding them from ventilator priority based on baseline function, creating inequities despite recommendations for objective metrics like Sequential Organ Failure Assessment scores adapted for pediatrics.[190] A 2020 review of ICU allocation stressed maximizing overall lives saved through short-term prognosis, but pediatric-specific adaptations often prioritized children under utilitarian models, though implementation flaws risked discriminating against those with comorbidities.[191] Post-mortem resource considerations include organ donation after circulatory death (DCD), which has expanded; from 2017 to 2023, 76 of 288 U.S. pediatric deaths resulted in organ donation, with DCD yielding viable hearts, livers, and kidneys despite lower initial yields compared to brain-death donation, supported by 1-year graft survival rates of 82-100% in select cohorts.[192][193] These practices require precise timing post-declaration of death to minimize warm ischemia, balancing ethical imperatives for donation against family grief.[194] Overall, dilemmas persist due to subjective quality-of-life projections, with evidence favoring transparent, evidence-based protocols to mitigate biases in prognosis assessment.[195]Major Controversies and Empirical Debates
Vaccine Efficacy, Safety, and Policy Mandates
Routine childhood vaccines, such as those for measles, mumps, rubella (MMR), diphtheria, tetanus, acellular pertussis (DTaP), and polio, demonstrate high efficacy in preventing targeted diseases among pediatric populations. A two-dose MMR regimen achieves 96% effectiveness against measles, 88% against mumps, and near-complete protection against rubella in clinical and observational studies.[196][197] Similarly, DTaP vaccines reduce pertussis incidence by over 80% after three doses, though efficacy wanes over time, necessitating boosters.[198] Polio vaccines, including inactivated polio vaccine (IPV), confer protection exceeding 80% after routine dosing starting at two months of age.[199] These figures derive from randomized trials and cohort studies, accounting for both direct protection and reductions in disease transmission; however, breakthrough infections occur, particularly for pertussis and mumps, due to factors like vaccine-induced immunity duration and pathogen evolution.[197] Overall, U.S. routine immunization has averted an estimated 508 million illnesses and 1 million deaths among children born 1994–2023, with incidence reductions ranging from 17% for influenza to 100% for diseases like smallpox.[119] Safety profiles indicate that most adverse events from these vaccines are mild and transient, including localized pain, fever, or rash, affecting a minority of recipients.[200] Serious events are rare, with anaphylaxis occurring at rates below 1 per million doses across vaccines.[201] Specific risks include febrile seizures following MMR or MMRV, elevated 1–2 weeks post-vaccination in toddlers, though not associated with long-term neurological harm.[202] The Vaccine Adverse Event Reporting System (VAERS) captures potential signals, such as intussusception after early rotavirus vaccines, leading to formulation changes, but its passive nature limits causality assessment due to underreporting of mild events and overreporting without verification.[203][204] Systematic reviews confirm no causal links to autism or widespread chronic conditions, despite public concerns amplified by anecdotal reports; however, temporal associations with emergency visits post-multi-vaccine doses at 12–18 months warrant ongoing surveillance.[205][206] Benefit-risk analyses emphasize net population-level gains, as vaccine-preventable diseases historically caused higher morbidity, including encephalitis from measles (1 in 1,000 cases).[197] Policy mandates for childhood vaccines, primarily enforced via school entry requirements in the U.S. and similar systems globally, aim to achieve herd immunity thresholds—95% for measles, lower for others like 85–90% for mumps—to curb outbreaks.[207][208] These policies correlate with higher coverage, reducing exemption clusters and disease incidence; for instance, states with stricter mandates show fewer pertussis cases linked to unvaccinated children.[209][210] Exemptions—medical, religious, or philosophical—have risen to 3% nationally by 2022–2023, contributing to localized outbreaks, as seen in 2019 U.S. measles resurgences despite overall high vaccination.[211] Recent policy shifts, including eased non-medical exemptions in some states post-COVID, highlight tensions: mandates boost uptake but face criticism for overriding parental autonomy and potentially eroding trust when rare harms occur without recourse.[212][213] Empirical data show mandates effective for population control yet insufficient alone against waning immunity or importation, underscoring needs for voluntary education over coercion; public support remains strong (79% for routine requirements), driven by perceived efficacy, though hesitancy persists amid socioeconomic and informational barriers.[214][215] In pediatrics, where children lack decisional capacity, mandates balance communal protection against individual risks, with ethical debates centering on proportionality given low baseline disease rates in vaccinated eras.[216]Gender Dysphoria Treatments and Long-Term Outcomes in Minors
Treatments for gender dysphoria in minors typically include psychological interventions, such as exploratory therapy to address underlying factors like trauma, autism, or co-occurring mental health conditions, alongside options for medical interventions like puberty blockers (GnRH analogues), cross-sex hormones, and, rarely, surgeries.[217] Systematic reviews commissioned by the UK's Cass Review in 2024 found the evidence base for medical interventions to be of low quality, with no reliable demonstration of benefits for gender dysphoria persistence, mental health, or body image satisfaction.[218] [219] Puberty blockers effectively suppress pubertal development but show little to no improvement in psychosocial functioning, and their long-term impacts remain uncertain due to limited controlled studies.[220] In children with gender dysphoria, desistance rates without medical intervention historically range from 60% to 88%, with persistence into adulthood occurring in a minority, often linked to sexual orientation rather than fixed transgender identity.[221] [222] A 2021 German study of clinic-referred youth reported persistence rates as low as 27% in adolescent females, rising to 50% in young adult males, suggesting many cases resolve naturally or with non-medical support.[222] Social transition in early childhood, however, correlates with higher persistence rates (up to 94% continuing to medical steps), potentially influencing developmental trajectories away from natural resolution.[223] Medical interventions carry documented risks: puberty blockers are associated with decreased bone mineral density (recovering variably post-treatment), potential fertility impairment, and uncertain effects on brain maturation and cognitive function.[224] [225] Cross-sex hormones, often initiated shortly after blockers (93-98% progression in Dutch cohorts followed over 20 years), increase risks of infertility, cardiovascular issues, and elevated cancer incidence in adults, with pediatric data limited by short follow-up periods.[226] Regret and detransition rates are underreported due to high loss to follow-up (e.g., 20-30% in some registries) and brief study durations, but recent surveys indicate 10-30% detransition within years, often citing unresolved mental health or realization of co-morbidities.[227] Long-term outcomes remain poorly evidenced, with a 2023 Swedish systematic review concluding that hormone therapy's effects on psychosocial health cannot be evaluated due to inadequate comparative studies.[228] Similarly, Finnish guidelines from 2020 and updated in 2023 prioritize psychotherapy over hormones for minors, deeming medical transitions experimental given risks outweighing unproven benefits.[229] Following the Cass Review, NHS England in 2024 restricted puberty blockers to clinical trials and emphasized comprehensive assessments, reflecting a shift from affirmative models toward caution informed by evidentiary gaps.[230] Sweden's 2022 national guidelines similarly limit interventions to exceptional cases post-maturity, citing systematic evidence reviews showing no net mental health gains.[231]| Study/Review | Key Finding on Outcomes | Evidence Quality |
|---|---|---|
| Cass Review (2024) | No improvement in GD or mental health from blockers/hormones; recommend research-only use.[217] | Low; few RCTs, methodological flaws. |
| Dutch Protocol Follow-up (2023, n=7200+) | 93-98% progressed to hormones; persistent GD but high co-morbidities; regret data incomplete.[226] | Moderate; long-term but non-randomized. |
| Swedish Systematic Review (2023) | Hormones' psychosocial effects unevaluable; risks like infertility prominent.[228] | Low; lack of controls. |
| NICE Reviews (2021) | Little/no change in body image or functioning from blockers.[219] | Very low; small samples, bias risks. |
Diagnostic Overreach in Behavioral and Developmental Disorders
Diagnostic overreach in behavioral and developmental disorders refers to the expansion of diagnoses such as attention-deficit/hyperactivity disorder (ADHD) and autism spectrum disorder (ASD) beyond what empirical evidence supports as genuine prevalence increases, often driven by broadened diagnostic criteria, subjective assessments, and external pressures including pharmaceutical marketing and demands for school accommodations. In the United States, ADHD diagnoses among children aged 3-17 rose from approximately 6.1% in 1997 to 10.2% by 2016, with 11.4% ever diagnosed by 2022 according to parent-reported national surveys. Similarly, ASD identification increased from 6.7 per 1,000 children in 2000 to 27.6 per 1,000 by 2020, reflecting a more than fourfold rise. These trends have prompted debates, with systematic reviews identifying convincing evidence of overdiagnosis, particularly in milder cases where symptoms overlap with normal developmental variations or environmental stressors.[232][233][234] For ADHD, overdiagnosis manifests in inconsistent application of criteria, with studies documenting higher rates among relatively younger children within the same school grade—suggesting maturation delays are misattributed to pathology—and geographic variations uncorrelated with underlying biology. A scoping review of 334 studies from 1989 to 2018 found consistent increases in diagnoses (45 studies) and pharmacological treatments (83 studies), often involving milder presentations (25 studies), where long-term harms of stimulants, such as growth suppression and cardiovascular risks, may exceed benefits. Overtreatment is evident in rising stimulant prescriptions, with U.S. rates tripling alongside diagnoses, despite limited evidence that medications address root causes like poor sleep or family dynamics in non-severe cases. Critics attribute this to diagnostic subjectivity in the DSM, where behavioral checklists incentivize labeling over comprehensive evaluation, potentially pathologizing traits adaptive in some contexts but disruptive in rigid educational settings.[235] ASD diagnoses have similarly ballooned due to the shift from narrow, severe criteria to a broad spectrum encompassing milder social and sensory quirks, a conceptual expansion pioneered by Lorna Wing in the 1980s and formalized in DSM-IV and DSM-5. This resulted in a 500% U.S. increase over 16 years ending around 2020, with current CDC estimates at 1 in 36 children, though profound cases requiring lifelong support remain stable at about 25% of diagnoses. Allen Frances, chair of the DSM-IV task force, has expressed regret for contributing to lowered thresholds, warning that DSM-5's merger of autism, Asperger's, and pervasive developmental disorder-not otherwise specified into one category risks "massive over-diagnosis" by capturing normal neurodiversity as disorder. The introduction of disruptive mood dysregulation disorder in DSM-5, intended to curb pediatric bipolar overdiagnosis, instead threatens to medicalize common tantrums in irritable children, further inflating rates of what Frances notes have already multiplied 40-fold for childhood bipolar and over 20-fold for autism in two decades.[236][237] Empirical indicators of overreach include discordance between self-reported symptoms and clinician diagnoses, with many children failing to meet full DSM criteria yet receiving labels and interventions, and international prevalence disparities—higher in the U.S. than in countries with stricter assessments—pointing to cultural and systemic influences rather than epidemics. While proponents cite improved awareness and access, counter-evidence from longitudinal data shows that expanded criteria capture cases with better prognoses, diluting resources for severe pathology and exposing low-risk youth to unnecessary therapies or medications with unproven long-term efficacy. This diagnostic inflation, Frances argues, stems from psychiatry's vulnerability to fads and industry pressures, prioritizing categorical labels over dimensional assessments of behavior rooted in context and causality.[235][237] Consequences include overmedicalization, with ADHD stimulant use linked to dependency risks and ASD diagnoses enabling special education but stigmatizing children as inherently defective rather than adaptable. Addressing overreach requires rigorous, multi-informant evaluations emphasizing first-onset severity and functional impairment over checklists, alongside scrutiny of incentives like pharmaceutical trials that favor positive outcomes in broadened populations. Despite academic resistance—potentially influenced by funding ties to expansive models—reverting to narrower criteria, as Frances advocates ignoring DSM-5's most inflationary changes, could realign diagnoses with verifiable pathology.[235][237]Child Abuse Diagnostics and Risks of Misattribution
Child abuse diagnostics in pediatrics primarily involve assessing injuries such as fractures, bruises, burns, and head trauma through clinical history, physical examination, skeletal surveys, and neuroimaging like CT or MRI scans.[238] These methods aim to distinguish inflicted injuries from accidental or medically explained ones, but challenges arise due to overlapping presentations; for instance, long-bone fractures in infants under 12 months carry a 16.7% to 35.2% abuse rate across studies, yet require ruling out conditions like osteogenesis imperfecta or rickets.[239] Diagnostic tools, including clinical decision rules like the Burnaby Criteria for rib fractures or the Pediatric Brain Injury Research Network (pediBIRN) criteria for head trauma, exhibit variability in sensitivity and specificity, with false positive rates influenced by factors such as non-focal subdural hemorrhages or acute encephalopathy.[240] [241] Misattribution risks are elevated in abusive head trauma (AHT), formerly termed shaken baby syndrome (SBS), where the diagnostic triad—subdural hematoma, retinal hemorrhages, and encephalopathy—is not pathognomonic, as it can stem from non-inflicted causes like birth trauma, coagulopathies, or metabolic disorders.[241] A 2023 analysis of pediBIRN data found significant misdiagnosis of non-AHT as AHT, with risks increasing in cases lacking focal injury or with alternative explanations overlooked, potentially leading to erroneous family separations.[242] Peer-reviewed reviews highlight that caregiver histories, when truthful, often align with non-abusive mechanisms, yet presumptive abuse labeling persists due to institutional pressures favoring Type I errors (false positives) over Type II (false negatives) to avoid missing maltreatment.[243] [244] For skeletal injuries, up to 20% of maltreatment-related fractures in children under three years are initially misdiagnosed as non-abusive, but the converse—attributing non-abusive fractures to abuse—occurs when rare genetic conditions like hypophosphatasia or temporary brittle bone disease are not considered, prompting unnecessary child protective interventions.[245] [246] Studies on overturned AHT/SBS convictions, numbering over a dozen in recent analyses, frequently cite diagnostic controversy, including failure to validate shaking biomechanics or exclusion of accidental falls, underscoring how reliance on historical assumptions without biomechanical evidence contributes to errors.[247] While organizations like the American Academy of Pediatrics maintain AHT's validity based on epidemiological patterns, dissenting forensic and biomechanical research questions the specificity of imaging findings, advocating multidisciplinary reviews to mitigate bias toward abuse attribution.[248] [249] Consequences of misattribution include profound familial and legal harms, such as wrongful prosecutions and foster care placements, with empirical data indicating that false positives disrupt attachments without proportional benefits in detection.[250] Balancing diagnostics requires integrating genetic testing, biomechanical modeling, and ophthalmologic expertise, as incomplete workups amplify errors; for example, 27 of 38 non-abused mild head injury cases were flagged high-risk without full evaluation.[251] Mainstream pediatric guidelines, while emphasizing vigilance, have faced critique for underweighting alternative etiologies amid systemic incentives to report, potentially inflating abuse rates beyond true incidence of 15.3 per 100,000 fractures in children under 36 months.[252] Rigorous, evidence-based protocols prioritizing causal mechanisms over presumptive patterns are essential to reduce these risks.[241]Global and Societal Impacts
Disparities in Access and Outcomes Across Regions
In low- and middle-income regions, particularly sub-Saharan Africa and South Asia, pediatric healthcare access remains severely limited compared to high-income areas, resulting in poorer health outcomes for children. The median number of pediatricians per 100,000 children is 0.5 in low-income countries versus 72 in high-income countries, based on surveys from over 80 nations conducted around 2019.[253] This scarcity contributes to inadequate preventive and curative services, with nearly 700 million children under 18 worldwide lacking access to safely managed drinking water as of recent estimates, predominantly in developing regions.[254] Fewer than one in ten children in low-income countries receive child benefits or social protection, heightening risks from infectious diseases, malnutrition, and environmental hazards.[255] Under-five mortality rates underscore these gaps: globally, the rate was 37 deaths per 1,000 live births in 2023, but sub-Saharan Africa reported approximately 74 per 1,000 in 2021 data, more than double the worldwide figure and over ten times higher than in Europe or high-income regions.[256][257] Neonatal deaths, totaling 2.3 million globally in 2023, are concentrated in low-resource settings due to insufficient neonatal intensive care and basic interventions like clean delivery and resuscitation.[258] Malnutrition exacerbates outcomes, with 150.2 million children under five stunted in 2024—rates highest in sub-Saharan Africa (around 30-40% prevalence) and South Asia—linked to poor access to nutritious food, sanitation, and routine health checks.[259][260] Vaccination coverage further highlights regional inequities, with routine childhood immunizations like the third dose of pneumococcal conjugate vaccine reaching 90.2% in Europe but only 30.1% in the WHO South-East Asia Region in 2021.[261] Overall, first-dose diphtheria-tetanus-pertussis vaccine coverage varies from over 90% in high-income regions to below 80% in parts of Africa and Asia, contributing to preventable outbreaks and higher morbidity.[262] These patterns persist despite global progress, as low-income regions face infrastructure deficits, supply chain issues, and workforce shortages that hinder service delivery.[257]| Indicator | High-Income Regions (e.g., Europe) | Low-Income Regions (e.g., Sub-Saharan Africa) |
|---|---|---|
| Under-5 Mortality Rate (per 1,000 live births, ~2021-2023) | ~4-5 | ~74 |
| Pediatricians per 100,000 Children (median) | 72 | 0.5 |
| PCV3 Vaccination Coverage (~2021) | 90% | Varies; lower in Africa/Asia (~50-70% for similar vaccines) |

