Recent from talks
Nothing was collected or created yet.
Infrared
View on Wikipedia
Infrared (IR; sometimes called infrared light) is electromagnetic radiation (EMR) with wavelengths longer than that of visible light but shorter than microwaves. The infrared spectral band begins with the waves that are just longer than those of red light (the longest waves in the visible spectrum), so IR is invisible to the human eye. IR is generally (according to ISO, CIE) understood to include wavelengths from around 780 nm (380 THz) to 1 mm (300 GHz).[1][2] IR is commonly divided between longer-wavelength thermal IR, emitted from terrestrial sources, and shorter-wavelength IR or near-IR, part of the solar spectrum.[3] Longer IR wavelengths (30–100 μm) are sometimes included as part of the terahertz radiation band.[4] Almost all black-body radiation from objects near room temperature is in the IR band. As a form of EMR, IR carries energy and momentum, exerts radiation pressure, and has properties corresponding to both those of a wave and of a particle, the photon.[5]
It was long known that fires emit invisible heat; in 1681 the pioneering experimenter Edme Mariotte showed that glass, though transparent to sunlight, obstructed radiant heat.[6][7] In 1800 the astronomer Sir William Herschel discovered that infrared radiation is a type of invisible radiation in the spectrum lower in energy than red light, by means of its effect on a thermometer.[8] Slightly more than half of the energy from the Sun was eventually found, through Herschel's studies, to arrive on Earth in the form of infrared. The balance between absorbed and emitted infrared radiation has an important effect on Earth's climate.
Infrared radiation is emitted or absorbed by molecules when changing rotational-vibrational movements. It excites vibrational modes in a molecule through a change in the dipole moment, making it a useful frequency range for study of these energy states for molecules of the proper symmetry. Infrared spectroscopy examines absorption and transmission of photons in the infrared range.[9]
Infrared radiation is used in industrial, scientific, military, commercial, and medical applications. Night-vision devices using active near-infrared illumination allow people or animals to be observed without the observer being detected. Infrared astronomy uses sensor-equipped telescopes to penetrate dusty regions of space such as molecular clouds, to detect objects such as planets, and to view highly red-shifted objects from the early days of the universe.[10] Infrared thermal-imaging cameras are used to detect heat loss in insulated systems, to observe changing blood flow in the skin, to assist firefighting, and to detect the overheating of electrical components.[11] Military and civilian applications include target acquisition, surveillance, night vision, homing, and tracking. Humans at normal body temperature radiate chiefly at wavelengths around 10 μm. Non-military uses include thermal efficiency analysis, environmental monitoring, industrial facility inspections, detection of grow-ops, remote temperature sensing, short-range wireless communication, spectroscopy, and weather forecasting.
Definition and relationship to the electromagnetic spectrum
[edit]There is no universally accepted definition of the range of infrared radiation. Typically, it is taken to extend from the nominal red edge of the visible spectrum at 780 nm to 1 mm. This range of wavelengths corresponds to a frequency range of approximately 430 THz down to 300 GHz. Beyond infrared is the microwave portion of the electromagnetic spectrum. Increasingly, terahertz radiation is counted as part of the microwave band, not infrared, moving the band edge of infrared to 0.1 mm (3 THz).
| Name | Wavelength | Frequency (Hz) | Photon energy (eV) |
|---|---|---|---|
| Gamma ray | less than 10 pm | more than 30 EHz | more than 124 keV |
| X-ray | 10 pm – 10 nm | 30 PHz – 30 EHz | 124 keV – 124 eV |
| Ultraviolet | 10 nm – 400 nm | 750 THz – 30 PHz | 124 eV – 3.3 eV |
| Visible | 400 nm – 700 nm | 430 THz – 750 THz | 3.3 eV – 1.7 eV |
| Infrared | 700 nm – 1 mm | 300 GHz – 430 THz | 1.7 eV – 1.24 meV |
| Microwave | 1 mm – 1 meter | 300 MHz – 300 GHz | 1.24 meV – 1.24 μeV |
| Radio | 1 meter and more | 300 MHz and below | 1.24 μeV and below |
Nature
[edit]Sunlight, at an effective temperature of 5,780 K (5,510 °C, 9,940 °F), is composed of near-thermal-spectrum radiation that is slightly more than half infrared. At zenith, sunlight provides an irradiance of just over 1 kW per square meter at sea level. Of this energy, 527 W is infrared radiation, 445 W is visible light, and 32 W is ultraviolet radiation.[13] Nearly all the infrared radiation in sunlight is near infrared, shorter than 4 μm.
On the surface of Earth, at far lower temperatures than the surface of the Sun, some thermal radiation consists of infrared in the mid-infrared region, much longer than in sunlight. Black-body, or thermal, radiation is continuous: it radiates at all wavelengths. Of these natural thermal radiation processes, only lightning and natural fires are hot enough to produce much visible energy, and fires produce far more infrared than visible-light energy.[14]
Regions
[edit]In general, objects emit infrared radiation across a spectrum of wavelengths, but sometimes only a limited region of the spectrum is of interest because sensors usually collect radiation only within a specific bandwidth. Thermal infrared radiation also has a maximum emission wavelength, which is inversely proportional to the absolute temperature of object, in accordance with Wien's displacement law. The infrared band is often subdivided into smaller sections, although how the IR spectrum is thereby divided varies between different areas in which IR is employed.
Visible limit
[edit]Infrared radiation is generally considered to begin with wavelengths longer than visible by the human eye. There is no hard wavelength limit to what is visible, as the eye's sensitivity decreases rapidly but smoothly, for wavelengths exceeding about 700 nm. Therefore wavelengths just longer than that can be seen if they are sufficiently bright, though they may still be classified as infrared according to usual definitions. Light from a near-IR laser may thus appear dim red and can present a hazard since it may actually carry a large amount of energy. Even IR at wavelengths up to 1,050 nm from pulsed lasers can be seen by humans under certain conditions.[15][16][17]
Commonly used subdivision scheme
[edit]A commonly used subdivision scheme is:[18][19][20]
| Division name | Abbreviation | Wavelength | Frequency | Photon energy | Temperature[i] | Characteristics |
|---|---|---|---|---|---|---|
| Near-infrared | NIR, IR-A DIN | 0.75–1.4 μm | 214–400 THz | 886–1,653 meV | 3,864–2,070 K (3,591–1,797 °C) |
Goes up to the wavelength of the first water absorption band, and commonly used in fiber optic telecommunication because of low attenuation losses in the SiO2 glass (silica) medium. Image intensifiers are sensitive to this area of the spectrum; examples include night vision devices such as night vision goggles. Near-infrared spectroscopy is another common application. |
| Short-wavelength infrared | SWIR, IR-B DIN | 1.4–3 μm | 100–214 THz | 413–886 meV | 2,070–966 K (1,797–693 °C) |
Water absorption increases significantly at 1,450 nm. The 1,530 to 1,560 nm range is the dominant spectral region for long-distance telecommunications (see transmission windows). |
| Mid-wavelength infrared | MWIR, IR-C DIN; MidIR.[22] Also called intermediate infrared (IIR) | 3–8 μm | 37–100 THz | 155–413 meV | 966–362 K (693–89 °C) |
In guided missile technology the 3–5 μm portion of this band is the atmospheric window in which the seekers of passive IR 'heat seeking' missiles are designed to work, homing on to the infrared signature of the target aircraft, typically the jet engine exhaust plume. This region is also known as thermal infrared. |
| Long-wavelength infrared | LWIR, IR-C DIN | 8–15 μm | 20–37 THz | 83–155 meV | 362–193 K (89 – −80 °C) |
The "thermal imaging" region, in which sensors can obtain a completely passive image of objects only slightly higher in temperature than room temperature – for example, the human body – based on thermal emissions only and requiring no illumination such as the sun or moon or an infrared illuminator. This region is also called the "thermal infrared". |
| Far-infrared | FIR | 15–1,000 μm | 0.3–20 THz | 1.2–83 meV | 193–3 K (−80.15 – −270.15 °C) |
(see also far-infrared laser and far-infrared) |
NIR and SWIR together is sometimes called "reflected infrared", whereas MWIR and LWIR is sometimes referred to as "thermal infrared".
CIE division scheme
[edit]The International Commission on Illumination (CIE) recommended the division of infrared radiation into the following three bands:[23][24]
| Abbreviation | Wavelength | Frequency |
|---|---|---|
| IR-A | 780–1400 nm | 215–384 THz |
| IR-B | 1400–3000 nm | 100–215 THz |
| IR-C | 3–1000 μm | 0.3–100 THz |
ISO 20473 scheme
[edit]ISO 20473 specifies the following scheme:[25]
| Designation | Abbreviation | Wavelength |
|---|---|---|
| Near-infrared | NIR | 0.78–3 μm |
| Mid-infrared | MIR | 3–50 μm |
| Far-infrared | FIR | 50–1,000 μm |
Astronomy division scheme
[edit]Astronomers typically divide the infrared spectrum as follows:[26]
| Designation | Abbreviation | Wavelength |
|---|---|---|
| Near-infrared | NIR | 0.7–2.5 μm |
| Mid-infrared | MIR | 3–25 μm |
| Far-infrared | FIR | above 25 μm |
These divisions are not precise and can vary depending on the publication. The three regions are used for observation of different temperature ranges,[27] and hence different environments in space.
The most common photometric system used in astronomy allocates capital letters to different spectral regions according to filters used; I, J, H, and K cover the near-infrared wavelengths; L, M, N, and Q refer to the mid-infrared region. These letters are commonly understood in reference to atmospheric windows and appear, for instance, in the titles of many papers.
Sensor response division scheme
[edit]
A third scheme divides up the band based on the response of various detectors:[28]
- Near-infrared: from 0.7 to 1.0 μm (from the approximate end of the response of the human eye to that of silicon).
- Short-wave infrared: 1.0 to 3 μm (from the cut-off of silicon to that of the MWIR atmospheric window). InGaAs covers to about 1.8 μm; the less sensitive lead salts cover this region. Cryogenically cooled MCT detectors can cover the region of 1.0–2.5 μm.
- Mid-wave infrared: 3 to 5 μm (defined by the atmospheric window and covered by indium antimonide, InSb and mercury cadmium telluride, HgCdTe, and partially by lead selenide, PbSe).
- Long-wave infrared: 8 to 12, or 7 to 14 μm (this is the atmospheric window covered by HgCdTe and microbolometers).
- Very-long wave infrared (VLWIR) (12 to about 30 μm, covered by doped silicon).
Near-infrared is the region closest in wavelength to the radiation detectable by the human eye. Mid- and far-infrared are progressively further from the visible spectrum. Other definitions follow different physical mechanisms (emission peaks, vs. bands, water absorption) and the newest follow technical reasons (the common silicon detectors are sensitive to about 1,050 nm, while InGaAs's sensitivity starts around 950 nm and ends between 1,700 and 2,600 nm, depending on the specific configuration). No international standards for these specifications are currently available.
The onset of infrared is defined (according to different standards) at various values typically between 700 nm and 800 nm, but the boundary between visible and infrared light is not precisely defined. The human eye is markedly less sensitive to light above 700 nm wavelength, so longer wavelengths make insignificant contributions to scenes illuminated by common light sources. Particularly intense near-IR light (e.g., from lasers, LEDs or bright daylight with the visible light filtered out) can be detected up to approximately 780 nm, and will be perceived as red light. Intense light sources providing wavelengths as long as 1,050 nm can be seen as a dull red glow, causing some difficulty in near-IR illumination of scenes in the dark (usually this practical problem is solved by indirect illumination). Leaves are particularly bright in the near IR, and if all visible light leaks from around an IR-filter are blocked, and the eye is given a moment to adjust to the extremely dim image coming through a visually opaque IR-passing photographic filter, it is possible to see the Wood effect, which consists of IR-glowing foliage.[29]
Telecommunication bands
[edit]In optical communications, the part of the infrared spectrum that is used is divided into seven bands based on availability of light sources, transmitting/absorbing materials (fibers), and detectors:[30]
| Band | Descriptor | Wavelength range |
|---|---|---|
| O band | Original | 1,260–1,360 nm |
| E band | Extended | 1,360–1,460 nm |
| S band | Short wavelength | 1,460–1,530 nm |
| C band | Conventional | 1,530–1,565 nm |
| L band | Long wavelength | 1,565–1,625 nm |
| U band | Ultralong wavelength | 1,625–1,675 nm |
The C-band is the dominant band for long-distance telecommunications networks. The S and L bands are based on less well established technology, and are not as widely deployed.
Heat
[edit]
Infrared radiation is popularly known as "heat radiation",[31] but light and electromagnetic waves of any frequency will heat surfaces that absorb them. Infrared light from the Sun accounts for 49%[32] of the heating of Earth, with the rest being caused by visible light that is absorbed then re-radiated at longer wavelengths. Visible light or ultraviolet-emitting lasers can char paper and incandescently hot objects emit visible radiation. Objects at room temperature will emit radiation concentrated mostly in the 8 to 25 μm band, but this is not distinct from the emission of visible light by incandescent objects and ultraviolet by even hotter objects (see black body and Wien's displacement law).[33]
Heat is energy in transit that flows due to a temperature difference. Unlike heat transmitted by thermal conduction or thermal convection, thermal radiation can propagate through a vacuum. Thermal radiation is characterized by a particular spectrum of many wavelengths that are associated with emission from an object, due to the vibration of its molecules at a given temperature. Thermal radiation can be emitted from objects at any wavelength, and at very high temperatures such radiation is associated with spectra far above the infrared, extending into visible, ultraviolet, and even X-ray regions (e.g. the solar corona). Thus, the popular association of infrared radiation with thermal radiation is only a coincidence based on typical (comparatively low) temperatures often found near the surface of planet Earth.
The concept of emissivity is important in understanding the infrared emissions of objects. This is a property of a surface that describes how its thermal emissions deviate from the ideal of a black body. To further explain, two objects at the same physical temperature may not show the same infrared image if they have differing emissivity. For example, for any pre-set emissivity value, objects with higher emissivity will appear hotter, and those with a lower emissivity will appear cooler (assuming, as is often the case, that the surrounding environment is cooler than the objects being viewed). When an object has less than perfect emissivity, it obtains properties of reflectivity and/or transparency, and so the temperature of the surrounding environment is partially reflected by and/or transmitted through the object. If the object were in a hotter environment, then a lower emissivity object at the same temperature would likely appear to be hotter than a more emissive one. For that reason, incorrect selection of emissivity and not accounting for environmental temperatures will give inaccurate results when using infrared cameras and pyrometers.
Applications
[edit]Night vision
[edit]
Infrared is used in night vision equipment when there is insufficient visible light to see.[34] Night vision devices operate through a process involving the conversion of ambient light photons into electrons that are then amplified by a chemical and electrical process and then converted back into visible light.[34] Infrared light sources can be used to augment the available ambient light for conversion by night vision devices, increasing in-the-dark visibility without actually using a visible light source.[34][1]
The use of infrared light and night vision devices should not be confused with thermal imaging, which creates images based on differences in surface temperature by detecting infrared radiation (heat) that emanates from objects and their surrounding environment.[35][8]
Thermography
[edit]
Infrared radiation can be used to remotely determine the temperature of objects (if the emissivity is known). This is termed thermography, or in the case of very hot objects in the NIR or visible it is termed pyrometry. Thermography (thermal imaging) is mainly used in military and industrial applications but the technology is reaching the public market in the form of infrared cameras on cars due to greatly reduced production costs.
Thermographic cameras detect radiation in the infrared range of the electromagnetic spectrum (roughly 9,000–14,000 nm or 9–14 μm) and produce images of that radiation. Since infrared radiation is emitted by all objects based on their temperatures, according to the black-body radiation law, thermography makes it possible to "see" one's environment with or without visible illumination. The amount of radiation emitted by an object increases with temperature, therefore thermography allows one to see variations in temperature (hence the name).
Hyperspectral imaging
[edit]

A hyperspectral image is a "picture" containing continuous spectrum through a wide spectral range at each pixel. Hyperspectral imaging is gaining importance in the field of applied spectroscopy particularly with NIR, SWIR, MWIR, and LWIR spectral regions. Typical applications include biological, mineralogical, defence, and industrial measurements.
Thermal infrared hyperspectral imaging can be similarly performed using a thermographic camera, with the fundamental difference that each pixel contains a full LWIR spectrum. Consequently, chemical identification of the object can be performed without a need for an external light source such as the Sun or the Moon. Such cameras are typically applied for geological measurements, outdoor surveillance and UAV applications.[37]
Other imaging
[edit]
In infrared photography, infrared filters are used to capture the near-infrared spectrum. Digital cameras often use infrared blockers. Cheaper digital cameras and camera phones have less effective filters and can view intense near-infrared, appearing as a bright purple-white color. This is especially pronounced when taking pictures of subjects near IR-bright areas (such as near a lamp), where the resulting infrared interference can wash out the image. There is also a technique called 'T-ray' imaging, which is imaging using far-infrared or terahertz radiation. Lack of bright sources can make terahertz photography more challenging than most other infrared imaging techniques. Recently T-ray imaging has been of considerable interest due to a number of new developments such as terahertz time-domain spectroscopy.
Tracking
[edit]
Infrared tracking, also known as infrared homing, refers to a passive missile guidance system, which uses the emission from a target of electromagnetic radiation in the infrared part of the spectrum to track it. Missiles that use infrared seeking are often referred to as "heat-seekers" since infrared (IR) is just below the visible spectrum of light in frequency and is radiated strongly by hot bodies. Many objects such as people, vehicle engines, and aircraft generate and retain heat, and as such, are especially visible in the infrared wavelengths of light compared to objects in the background.[38]
Heating
[edit]
Infrared radiation can be used as a deliberate heating source. For example, it is used in infrared saunas to heat the occupants. It may also be used in other heating applications, such as to remove ice from the wings of aircraft (de-icing).[39]
Infrared heating is also becoming more popular in industrial manufacturing processes, e.g. curing of coatings, forming of plastics, annealing, plastic welding, and print drying. In these applications, infrared heaters replace convection ovens and contact heating.
Cooling
[edit]A variety of technologies or proposed technologies take advantage of infrared emissions to cool buildings or other systems. The LWIR (8–15 μm) region is especially useful since some radiation at these wavelengths can escape into space through the atmosphere's infrared window. This is how passive daytime radiative cooling (PDRC) surfaces are able to achieve sub-ambient cooling temperatures under direct solar intensity, enhancing terrestrial heat flow to outer space with zero energy consumption or pollution.[40][41] PDRC surfaces maximize shortwave solar reflectance to lessen heat gain while maintaining strong longwave infrared (LWIR) thermal radiation heat transfer.[42][43] When imagined on a worldwide scale, this cooling method has been proposed as a way to slow and even reverse global warming, with some estimates proposing a global surface area coverage of 1-2% to balance global heat fluxes.[44][45]
Communications
[edit]IR data transmission is also employed in short-range communication among computer peripherals and personal digital assistants. These devices usually conform to standards published by IrDA, the Infrared Data Association. Remote controls and IrDA devices use infrared light-emitting diodes (LEDs) to emit infrared radiation that may be concentrated by a lens into a beam that the user aims at the detector. The beam is modulated, i.e. switched on and off, according to a code which the receiver interprets. Usually very near-IR is used (below 800 nm) for practical reasons. This wavelength is efficiently detected by inexpensive silicon photodiodes, which the receiver uses to convert the detected radiation to an electric current. That electrical signal is passed through a high-pass filter which retains the rapid pulsations due to the IR transmitter but filters out slowly changing infrared radiation from ambient light. Infrared communications are useful for indoor use in areas of high population density. IR does not penetrate walls and so does not interfere with other devices in adjoining rooms. Infrared is the most common way for remote controls to command appliances. Infrared remote control protocols like RC-5, SIRC, are used to communicate with infrared.
Free-space optical communication using infrared lasers can be a relatively inexpensive way to install a communications link in an urban area operating at up to 4 gigabit/s, compared to the cost of burying fiber optic cable, except for the radiation damage. "Since the eye cannot detect IR, blinking or closing the eyes to help prevent or reduce damage may not happen."[46]
Infrared lasers are used to provide the light for optical fiber communications systems. Wavelengths around 1,330 nm (least dispersion) or 1,550 nm (best transmission) are the best choices for standard silica fibers.
IR data transmission of audio versions of printed signs is being researched as an aid for visually impaired people through the Remote infrared audible signage project. Transmitting IR data from one device to another is sometimes referred to as beaming.
IR is sometimes used for assistive audio as an alternative to an audio induction loop.
Spectroscopy
[edit]Infrared vibrational spectroscopy (see also near-infrared spectroscopy) is a technique that can be used to identify molecules by analysis of their constituent bonds. Each chemical bond in a molecule vibrates at a frequency characteristic of that bond. A group of atoms in a molecule (e.g., CH2) may have multiple modes of oscillation caused by the stretching and bending motions of the group as a whole. If an oscillation leads to a change in dipole in the molecule then it will absorb a photon that has the same frequency. The vibrational frequencies of most molecules correspond to the frequencies of infrared light. Typically, the technique is used to study organic compounds using light radiation from the mid-infrared, 4,000–400 cm−1. A spectrum of all the frequencies of absorption in a sample is recorded. This can be used to gain information about the sample composition in terms of chemical groups present and also its purity (for example, a wet sample will show a broad O-H absorption around 3200 cm−1). The unit for expressing radiation in this application, cm−1, is the spectroscopic wavenumber. It is the frequency divided by the speed of light in vacuum.
Thin film metrology
[edit]In the semiconductor industry, infrared light can be used to characterize materials such as thin films and periodic trench structures. By measuring the reflectance of light from the surface of a semiconductor wafer, the index of refraction (n) and the extinction Coefficient (k) can be determined via the Forouhi–Bloomer dispersion equations. The reflectance from the infrared light can also be used to determine the critical dimension, depth, and sidewall angle of high aspect ratio trench structures.
Meteorology
[edit]
Weather satellites equipped with scanning radiometers produce thermal or infrared images, which can then enable a trained analyst to determine cloud heights and types, to calculate land and surface water temperatures, and to locate ocean surface features. The scanning is typically in the range 10.3–12.5 μm (IR4 and IR5 channels).
Clouds with high and cold tops, such as cyclones or cumulonimbus clouds, are often displayed as red or black, lower warmer clouds such as stratus or stratocumulus are displayed as blue or grey, with intermediate clouds shaded accordingly. Hot land surfaces are shown as dark-grey or black. One disadvantage of infrared imagery is that low clouds such as stratus or fog can have a temperature similar to the surrounding land or sea surface and do not show up. However, using the difference in brightness of the IR4 channel (10.3–11.5 μm) and the near-infrared channel (1.58–1.64 μm), low clouds can be distinguished, producing a fog satellite picture. The main advantage of infrared is that images can be produced at night, allowing a continuous sequence of weather to be studied.
These infrared pictures can depict ocean eddies or vortices and map currents such as the Gulf Stream, which are valuable to the shipping industry. Fishermen and farmers are interested in knowing land and water temperatures to protect their crops against frost or increase their catch from the sea. Even El Niño phenomena can be spotted. Using color-digitized techniques, the gray-shaded thermal images can be converted to color for easier identification of desired information.
The main water vapour channel at 6.40 to 7.08 μm can be imaged by some weather satellites and shows the amount of moisture in the atmosphere.
Climatology
[edit]
In the field of climatology, atmospheric infrared radiation is monitored to detect trends in the energy exchange between the Earth and the atmosphere. These trends provide information on long-term changes in Earth's climate. It is one of the primary parameters studied in research into global warming, together with solar radiation.
A pyrgeometer is utilized in this field of research to perform continuous outdoor measurements. This is a broadband infrared radiometer with sensitivity for infrared radiation between approximately 4.5 μm and 50 μm.
Astronomy
[edit]
Astronomers observe objects in the infrared portion of the electromagnetic spectrum using optical components, including mirrors, lenses and solid state digital detectors. For this reason it is classified as part of optical astronomy. To form an image, the components of an infrared telescope need to be carefully shielded from heat sources, and the detectors are chilled using liquid helium.
The sensitivity of Earth-based infrared telescopes is significantly limited by water vapor in the atmosphere, which absorbs a portion of the infrared radiation arriving from space outside of selected atmospheric windows. This limitation can be partially alleviated by placing the telescope observatory at a high altitude, or by carrying the telescope aloft with a balloon or an aircraft. Space telescopes do not suffer from this handicap, and so outer space is considered the ideal location for infrared astronomy.
The infrared portion of the spectrum has several useful benefits for astronomers. Cold, dark molecular clouds of gas and dust in our galaxy will glow with radiated heat as they are irradiated by imbedded stars. Infrared can also be used to detect protostars before they begin to emit visible light. Stars emit a smaller portion of their energy in the infrared spectrum, so nearby cool objects such as planets can be more readily detected. (In the visible light spectrum, the glare from the star will drown out the reflected light from a planet.)
Infrared light is also useful for observing the cores of active galaxies, which are often cloaked in gas and dust. Distant galaxies with a high redshift will have the peak portion of their spectrum shifted toward longer wavelengths, so they are more readily observed in the infrared.[10]
Cleaning
[edit]Infrared cleaning is a technique used by some motion picture film scanners, film scanners and flatbed scanners to reduce or remove the effect of dust and scratches upon the finished scan. It works by collecting an additional infrared channel from the scan at the same position and resolution as the three visible color channels (red, green, and blue). The infrared channel, in combination with the other channels, is used to detect the location of scratches and dust. Once located, those defects can be corrected by scaling or replaced by inpainting.[47]
Art conservation and analysis
[edit]

Infrared reflectography[48] can be applied to paintings to reveal underlying layers in a non-destructive manner, in particular the artist's underdrawing or outline drawn as a guide. Art conservators use the technique to examine how the visible layers of paint differ from the underdrawing or layers in between (such alterations are called pentimenti when made by the original artist). This is very useful information in deciding whether a painting is the prime version by the original artist or a copy, and whether it has been altered by over-enthusiastic restoration work. In general, the more pentimenti, the more likely a painting is to be the prime version. It also gives useful insights into working practices.[49] Reflectography often reveals the artist's use of carbon black, which shows up well in reflectograms, as long as it has not also been used in the ground underlying the whole painting. Infrared reflectography can be realized by modified commercial digital cameras in the NIR spectral region or by dedicated instruments in the SWIR spectral region.[50] The recent extension of reflectography into the MWIR spectral region[51][52] has proved capable of detecting subtle differences in surface materials.
Finally, NIR reflectography can be performed with good results using smartphone cameras .[53]
Recent progress in the design of infrared-sensitive cameras makes it possible to discover and depict not only underpaintings and pentimenti, but entire paintings that were later overpainted by the artist.[54] Notable examples are Picasso's Woman Ironing and Blue Room, where in both cases a portrait of a man has been made visible under the painting as it is known today.
Similar uses of infrared are made by conservators and scientists on various types of objects, especially very old written documents such as the Dead Sea Scrolls, the Roman works in the Villa of the Papyri, and the Silk Road texts found in the Dunhuang Caves.[55] Carbon black used in ink can show up extremely well.
Biological systems
[edit]
The pit viper has a pair of infrared sensory pits on its head. There is uncertainty regarding the exact thermal sensitivity of this biological infrared detection system.[56][57]
Other organisms that have thermoreceptive organs are pythons (family Pythonidae), some boas (family Boidae), the Common Vampire Bat (Desmodus rotundus), a variety of jewel beetles (Melanophila acuminata),[58] darkly pigmented butterflies (Pachliopta aristolochiae and Troides rhadamantus plateni), and possibly blood-sucking bugs (Triatoma infestans).[59] By detecting the heat that their prey emits, crotaline and boid snakes identify and capture their prey using their IR-sensitive pit organs. Comparably, IR-sensitive pits on the Common Vampire Bat (Desmodus rotundus) aid in the identification of blood-rich regions on its warm-blooded victim. The jewel beetle, Melanophila acuminata, locates forest fires via infrared pit organs, where on recently burnt trees, they deposit their eggs. Thermoreceptors on the wings and antennae of butterflies with dark pigmentation, such Pachliopta aristolochiae and Troides rhadamantus plateni, shield them from heat damage as they sunbathe in the sun. Additionally, it's hypothesised that thermoreceptors let bloodsucking bugs (Triatoma infestans) locate their warm-blooded victims by sensing their body heat.[59]
Some fungi like Venturia inaequalis require near-infrared light for ejection.[60]
Although near-infrared vision (780–1,000 nm) has long been deemed impossible due to noise in visual pigments,[61] sensation of near-infrared light was reported in the common carp and in three cichlid species.[61][62][63][64][65] Fish use NIR to capture prey[61] and for phototactic swimming orientation.[65] NIR sensation in fish may be relevant under poor lighting conditions during twilight[61] and in turbid surface waters.[65]
Photobiomodulation
[edit]Near-infrared light, or photobiomodulation, is used for treatment of chemotherapy-induced oral ulceration as well as wound healing. There is some work relating to anti-herpes virus treatment.[66] Research projects include work on central nervous system healing effects via cytochrome c oxidase upregulation and other possible mechanisms.[67]
Health hazards
[edit]Strong infrared radiation in certain industry high-heat settings may be hazardous to the eyes, resulting in damage or blindness to the user. Since the radiation is invisible, special IR-proof goggles must be worn in such places.[68]
Scientific history
[edit]The discovery of infrared radiation is ascribed to William Herschel, the astronomer, in the early 19th century. Herschel published his results in 1800 before the Royal Society of London. Herschel used a prism to refract light from the sun and detected the infrared, beyond the red part of the spectrum, through an increase in the temperature recorded on a thermometer. He was surprised at the result and called them "Calorific Rays".[69][70] The term "infrared" did not appear until late 19th century.[71] The Latin prefix infra- means below, as it is light below red on the spectrum.[72] An earlier experiment in 1790 by Marc-Auguste Pictet demonstrated the reflection and focusing of radiant heat via mirrors in the absence of visible light.[73]
Other important dates include:[28]

- 1830: Leopoldo Nobili made the first thermopile IR detector.[74]
- 1840: John Herschel produces the first thermal image, called a thermogram.[75]
- 1860: Gustav Kirchhoff formulated the blackbody theorem .[76]
- 1873: Willoughby Smith discovered the photoconductivity of selenium.[77]
- 1878: Samuel Pierpont Langley invents the first bolometer, a device which is able to measure small temperature fluctuations, and thus the power of far infrared sources.[78]
- 1879: Stefan–Boltzmann law formulated empirically that the power radiated by a blackbody is proportional to T4.[79]
- 1880s and 1890s: Lord Rayleigh and Wilhelm Wien solved part of the blackbody equation, but both solutions diverged in parts of the electromagnetic spectrum. This problem was called the "ultraviolet catastrophe and infrared catastrophe".[80]
- 1892: Willem Henri Julius published infrared spectra of 20 organic compounds measured with a bolometer in units of angular displacement.[81]
- 1901: Max Planck published the blackbody equation and theorem. He solved the problem by quantizing the allowable energy transitions.[82]
- 1905: Albert Einstein developed the theory of the photoelectric effect.[83]
- 1905–1908: William Coblentz published infrared spectra in units of wavelength (micrometers) for several chemical compounds in Investigations of Infra-Red Spectra.[84][85][86]
- 1917: Theodore Case developed the thallous sulfide detector, which helped produce the first infrared search and track device able to detect aircraft at a range of one mile (1.6 km).
- 1935: Lead salts – early missile guidance in World War II.
- 1938: Yeou Ta predicted that the pyroelectric effect could be used to detect infrared radiation.[87]
- 1945: The Zielgerät 1229 "Vampir" infrared weapon system was introduced as the first portable infrared device for military applications.
- 1952: Heinrich Welker grew synthetic InSb crystals.
- 1950s and 1960s: Nomenclature and radiometric units defined by Fred Nicodemenus, G. J. Zissis and R. Clark; Robert Clark Jones defined D*.
- 1958: W. D. Lawson (Royal Radar Establishment in Malvern) discovered IR detection properties of Mercury cadmium telluride (HgCdTe).[88]
- 1958: Falcon and Sidewinder missiles were developed using infrared technology.
- 1960s: Paul Kruse and his colleagues at Honeywell Research Center demonstrate the use of HgCdTe as an effective compound for infrared detection.[88]
- 1962: J. Cooper demonstrated pyroelectric detection.[89]
- 1964: W. G. Evans discovered infrared thermoreceptors in a pyrophile beetle.[58]
- 1965: First IR handbook; first commercial imagers (Barnes, Agema (now part of FLIR Systems Inc.)); Richard Hudson's landmark text; F4 TRAM FLIR by Hughes; phenomenology pioneered by Fred Simmons and A. T. Stair; U.S. Army's night vision lab formed (now Night Vision and Electronic Sensors Directorate (NVESD)), and Rachets develops detection, recognition and identification modeling there.
- 1970: Willard Boyle and George E. Smith proposed CCD at Bell Labs for picture phone.
- 1973: Common module program started by NVESD.[90]
- 1978: Infrared imaging astronomy came of age, observatories planned, IRTF on Mauna Kea opened; 32 × 32 and 64 × 64 arrays produced using InSb, HgCdTe and other materials.
- 2013: On 14 February, researchers developed a neural implant that gives rats the ability to sense infrared light, which for the first time provides living creatures with new abilities, instead of simply replacing or augmenting existing abilities.[91]
See also
[edit]Notes
[edit]- ^ Temperatures of black bodies for which spectral peaks fall at the given wavelengths, according to the wavelength form of Wien's displacement law.[21]
References
[edit]- ^ a b Vatansever, Fatma; Hamblin, Michael R. (2012-01-01). "Far infrared radiation (FIR): Its biological effects and medical applications". Photonics & Lasers in Medicine. 1 (4): 255–266. doi:10.1515/plm-2012-0034. ISSN 2193-0643. PMC 3699878. PMID 23833705.
- ^ "Definition of INFRARED". www.merriam-webster.com. 2024-09-18. Archived from the original on 2024-09-22. Retrieved 2024-09-20.
- ^ "IPCC AR4 SYR Appendix Glossary" (PDF). Archived from the original (PDF) on 2018-11-17. Retrieved 2008-12-14.
- ^ Rogalski, Antoni (2019). Infrared and terahertz detectors (3rd ed.). Boca Raton, FL: CRC Press. p. 929. ISBN 978-1-315-27133-0.
- ^ "Infrared radiation | Definition, Wavelengths, & Facts | Britannica". www.britannica.com. 2024-09-18. Retrieved 2024-09-20.
- ^ Calel, Raphael (19 February 2014). "The Founding Fathers v. The Climate Change Skeptics". The Public Domain Review. Archived from the original on 11 October 2019. Retrieved 16 September 2019.
- ^ Fleming, James R. (17 March 2008). "Climate Change and Anthropogenic Greenhouse Warming: A Selection of Key Articles, 1824–1995, with Interpretive Essays". National Science Digital Library Project Archive PALE:ClassicArticles. Archived from the original on 29 September 2019. Retrieved 1 February 2022. Article 1: General remarks on the temperature of the earth and outer space Archived 2023-06-08 at the Wayback Machine.
- ^ a b Michael Rowan-Robinson (2013). Night Vision: Exploring the Infrared Universe. Cambridge University Press. p. 23. ISBN 1107024765.
- ^ Reusch, William (1999). "Infrared Spectroscopy". Michigan State University. Archived from the original on 2007-10-27. Retrieved 2006-10-27.
- ^ a b "IR Astronomy: Overview". NASA Infrared Astronomy and Processing Center. Archived from the original on 2006-12-08. Retrieved 2006-10-30.
- ^ Chilton, Alexander (2013-10-07). "The Working Principle and Key Applications of Infrared Sensors". AZoSensors. Archived from the original on 2020-07-11. Retrieved 2020-07-11.
- ^ Haynes, William M., ed. (2011). CRC Handbook of Chemistry and Physics (92nd ed.). CRC Press. p. 10.233. ISBN 978-1-4398-5511-9.
- ^ "Reference Solar Spectral Irradiance: Air Mass 1.5". Archived from the original on 2019-05-12. Retrieved 2009-11-12.
- ^ "Blackbody Radiation | Astronomy 801: Planets, Stars, Galaxies, and the Universe". Archived from the original on 2019-05-01. Retrieved 2019-02-12.
- ^ Sliney, David H.; Wangemann, Robert T.; Franks, James K.; Wolbarsht, Myron L. (1976). "Visual sensitivity of the eye to infrared laser radiation". Journal of the Optical Society of America. 66 (4): 339–341. Bibcode:1976JOSA...66..339S. doi:10.1364/JOSA.66.000339. PMID 1262982.
The foveal sensitivity to several near-infrared laser wavelengths was measured. It was found that the eye could respond to radiation at wavelengths at least as far as 1064 nm. A continuous 1064 nm laser source appeared red, but a 1060 nm pulsed laser source appeared green, which suggests the presence of second harmonic generation in the retina.
- ^ Lynch, David K.; Livingston, William Charles (2001). Color and Light in Nature (2nd ed.). Cambridge, UK: Cambridge University Press. p. 231. ISBN 978-0-521-77504-5. Archived from the original on 29 May 2024. Retrieved 12 October 2013.
Limits of the eye's overall range of sensitivity extends from about 310 to 1,050 nanometers
- ^ Saidman, Jean (15 May 1933). "Sur la visibilité de l'ultraviolet jusqu'à la longueur d'onde 3130" [The visibility of the ultraviolet to the wave length of 3130]. Comptes rendus de l'Académie des sciences (in French). 196: 1537–9. Archived from the original on 24 October 2013. Retrieved 3 July 2014.
- ^ Byrnes, James (2009). Unexploded Ordnance Detection and Mitigation. Springer. pp. 21–22. Bibcode:2009uodm.book.....B. ISBN 978-1-4020-9252-7.
- ^ Paschotta, Dr Rüdiger (13 May 2008). "Infrared Light". RP Photonics Encyclopedia. RP Photonics. doi:10.61835/gvw. Archived from the original on 1 August 2021. Retrieved 20 July 2021.
- ^ "Definition of NEAR-INFRARED". www.merriam-webster.com. 2024-09-16. Retrieved 2024-10-02.
- ^ "Peaks of Blackbody Radiation Intensity". Archived from the original on 18 March 2011. Retrieved 27 July 2016.
- ^ "Photoacoustic technique 'hears' the sound of dangerous chemical agents". R&D Magazine. August 14, 2012. rdmag.com. Archived from the original on September 22, 2024. Retrieved September 8, 2012.
- ^ Henderson, Roy. "Wavelength considerations". Instituts für Umform- und Hochleistungs. Archived from the original on 2007-10-28. Retrieved 2007-10-18.
- ^ CIE (International Commission on Illumination). "infrared radiation IR radiation IRR". 17-21-004. Archived from the original on 22 September 2024. Retrieved 18 October 2022.
- ^ ISO 20473:2007 – Optics and photonics – Spectral bands.
- ^ "Near, Mid and Far-Infrared". NASA IPAC. Archived from the original on 2012-05-29. Retrieved 2007-04-04.
- ^ "Near, Mid and Far-Infrared". www.icc.dur.ac.uk. Archived from the original on 2024-03-28. Retrieved 2024-03-28.
- ^ a b Miller, Principles of Infrared Technology (Van Nostrand Reinhold, 1992), and Miller and Friedman, Photonic Rules of Thumb, 2004. ISBN 978-0-442-01210-6[page needed]
- ^ Griffin, Donald R.; Hubbard, Ruth; Wald, George (1947). "The Sensitivity of the Human Eye to Infra-Red Radiation". Journal of the Optical Society of America. 37 (7): 546–553. Bibcode:1947JOSA...37..546G. doi:10.1364/JOSA.37.000546. PMID 20256359.
- ^ Ramaswami, Rajiv (May 2002). "Optical Fiber Communication: From Transmission to Networking". IEEE Communications Magazine. 40 (5): 138–147. Bibcode:2002IComM..40e.138R. doi:10.1109/MCOM.2002.1006983. S2CID 29838317.
- ^ "Infrared Radiation". Infrared Radiation. Van Nostrand's Scientific Encyclopedia. John Wiley & Sons, Inc. 2007. doi:10.1002/0471743984.vse4181.pub2. ISBN 978-0-471-74398-9.
- ^ "Introduction to Solar Energy". Passive Solar Heating & Cooling Manual. Rodale Press, Inc. 1980. Archived from the original (DOC) on 2009-03-18. Retrieved 2007-08-12.
- ^ McCreary, Jeremy (October 30, 2004). "Infrared (IR) basics for digital photographers-capturing the unseen (Sidebar: Black Body Radiation)". Digital Photography For What It's Worth. Archived from the original on 2008-12-18. Retrieved 2006-11-07.
- ^ a b c "How Night Vision Works". American Technologies Network Corporation. Archived from the original on 2015-08-24. Retrieved 2007-08-12.
- ^ Bryant, Lynn (2007-06-11). "How does thermal imaging work? A closer look at what is behind this remarkable technology". Archived from the original on 2007-07-28. Retrieved 2007-08-12.
- ^ Holma, H., (May 2011), Thermische Hyperspektralbildgebung im langwelligen Infrarot Archived 2011-07-26 at the Wayback Machine, Photonik
- ^ Frost&Sullivan, Technical Insights, Aerospace&Defence (Feb 2011): World First Thermal Hyperspectral Camera for Unmanned Aerial Vehicles Archived 2012-03-10 at the Wayback Machine.
- ^ Mahulikar, S.P.; Sonawane, H.R.; Rao, G.A. (2007). "Infrared signature studies of aerospace vehicles" (PDF). Progress in Aerospace Sciences. 43 (7–8): 218–245. Bibcode:2007PrAeS..43..218M. CiteSeerX 10.1.1.456.9135. doi:10.1016/j.paerosci.2007.06.002. Archived (PDF) from the original on 2021-03-04. Retrieved 2013-04-12.
- ^ White, Richard P. (2000) "Infrared deicing system for aircraft" U.S. patent 6,092,765
- ^ Chen, Meijie; Pang, Dan; Chen, Xingyu; Yan, Hongjie; Yang, Yuan (2022). "Passive daytime radiative cooling: Fundamentals, material designs, and applications". EcoMat. 4 (1) e12153. doi:10.1002/eom2.12153. S2CID 240331557.
Passive daytime radiative cooling (PDRC) dissipates terrestrial heat to the extremely cold outer space without using any energy input or producing pollution. It has the potential to simultaneously alleviate the two major problems of energy crisis and global warming.
- ^ Munday, Jeremy (2019). "Tackling Climate Change through Radiative Cooling". Joule. 3 (9): 2057–2060. Bibcode:2019Joule...3.2057M. doi:10.1016/j.joule.2019.07.010. S2CID 201590290.
By covering the Earth with a small fraction of thermally emitting materials, the heat flow away from the Earth can be increased, and the net radiative flux can be reduced to zero (or even made negative), thus stabilizing (or cooling) the Earth.
- ^ Wang, Tong; Wu, Yi; Shi, Lan; Hu, Xinhua; Chen, Min; Wu, Limin (2021). "A structural polymer for highly efficient all-day passive radiative cooling". Nature Communications. 12 (365): 365. doi:10.1038/s41467-020-20646-7. PMC 7809060. PMID 33446648.
Accordingly, designing and fabricating efficient PDRC with sufficiently high solar reflectance (𝜌¯solar) (λ ~ 0.3–2.5 μm) to minimize solar heat gain and simultaneously strong LWIR thermal emittance (ε¯LWIR) to maximize radiative heat loss is highly desirable. When the incoming radiative heat from the Sun is balanced by the outgoing radiative heat emission, the temperature of the Earth can reach its steady state.
- ^ Zevenhovena, Ron; Fält, Martin (June 2018). "Radiative cooling through the atmospheric window: A third, less intrusive geoengineering approach" (PDF). Energy. 152: 27. Bibcode:2018Ene...152...27Z. doi:10.1016/j.energy.2018.03.084. Retrieved 2022-10-13 – via Elsevier Science Direct.
- ^ Munday, Jeremy (2019). "Tackling Climate Change through Radiative Cooling". Joule. 3 (9): 2057–2060. Bibcode:2019Joule...3.2057M. doi:10.1016/j.joule.2019.07.010. S2CID 201590290.
If only 1%–2% of the Earth's surface were instead made to radiate at this rate rather than its current average value, the total heat fluxes into and away from the entire Earth would be balanced and warming would cease.
- ^ Zevenhovena, Ron; Fält, Martin (June 2018). "Radiative cooling through the atmospheric window: A third, less intrusive geoengineering approach" (PDF). Energy. 152: 27–33. Bibcode:2018Ene...152...27Z. doi:10.1016/j.energy.2018.03.084. Archived (PDF) from the original on 2024-10-03. Retrieved 2022-10-13 – via Elsevier Science Direct.
With 100 W/m2 as a demonstrated passive cooling effect, a surface coverage of 0.3% would then be needed, or 1% of Earth's land mass surface. If half of it would be installed in urban, built areas which cover roughly 3% of the Earth's land mass, a 17% coverage would be needed there, with the remainder being installed in rural areas.
- ^ "Dangers of Overexposure to ultraviolet, infrared and high-energy visible light". ishn.com – Industrial Safety & Health News. 2013-01-03. Archived from the original on 2016-08-16. Retrieved 2017-04-26.
- ^ Digital ICE. kodak.com
- ^ "IR Reflectography for Non-destructive Analysis of Underdrawings in Art Objects". Sensors Unlimited, Inc. Archived from the original on 2008-12-08. Retrieved 2009-02-20.
- ^ "The Mass of Saint Gregory: Examining a Painting Using Infrared Reflectography". The Cleveland Museum of Art. Archived from the original on 2009-01-13. Retrieved 2009-02-20.
- ^ Ambrosini, D.; Daffara, C; Di Biase, R.; Paoletti, D.; Pezzati, L.; Bellucci, R.; Bettini, F. (13 November 2009). "Integrated reflectography and thermography for wooden painting diagnostics" (PDF). Journal of Cultural Heritage. 11 (2): 196–204. doi:10.1016/j.culher.2009.05.001.
- ^ Daffara, C.; Ambrosini, D.; Pezzati, L.; Paoletti, D. (18 June 2012). "Thermal-quasi-reflectography: a new imaging tool in art conservation". Optics Express. 20 (13): 14746–14753. Bibcode:2012OExpr..2014746D. doi:10.1364/OE.20.014746. PMID 22714535.
- ^ Daffara, C.; Parisotto, S.; Mariotti, P. I.; Ambrosini, D. (18 November 2021). "Dual mode imaging in mid infrared with thermal signal reconstruction for innovative diagnostics of the "Monocromo" by Leonardo da Vinci". Scientific Reports. 11 22482. Bibcode:2021NatSR..1122482D. doi:10.1038/s41598-021-01837-8. PMC 8602283. PMID 34795373.
- ^ Daffara, C.; Ambrosini, D. (14 August 2024). "Smartphone-based diagnostics with coherent and infrared imaging for cultural heritage". Journal of Physics: Photonics. 6 (4): 045006. Bibcode:2024JPhP....6d5006D. doi:10.1088/2515-7647/ad6abc.
- ^ Infrared reflectography in analysis of paintings Archived 2015-12-22 at the Wayback Machine at ColourLex.
- ^ "International Dunhuang Project An Introduction to digital infrared photography and its application within IDP". Idp.bl.uk. Archived from the original on 2008-12-02. Retrieved 2011-11-08.
- ^ Jones, B.S.; Lynn, W.F.; Stone, M.O. (2001). "Thermal Modeling of Snake Infrared Reception: Evidence for Limited Detection Range". Journal of Theoretical Biology. 209 (2): 201–211. Bibcode:2001JThBi.209..201J. doi:10.1006/jtbi.2000.2256. PMID 11401462. Archived from the original on 2020-03-17. Retrieved 2019-09-06.
- ^ Gorbunov, V.; Fuchigami, N.; Stone, M.; Grace, M.; Tsukruk, V. V. (2002). "Biological Thermal Detection: Micromechanical and Microthermal Properties of Biological Infrared Receptors". Biomacromolecules. 3 (1): 106–115. doi:10.1021/bm015591f. PMID 11866562. S2CID 21737304.
- ^ a b Evans, W.G. (1966). "Infrared receptors in Melanophila acuminata De Geer". Nature. 202 (4928): 211. Bibcode:1964Natur.202..211E. doi:10.1038/202211a0. PMID 14156319. S2CID 2553265.
- ^ a b Campbell, Angela L.; Naik, Rajesh R.; Sowards, Laura; Stone, Morley O. (2002). "Biological infrared imaging and sensing". Micrometre. 33 (2): 211–225. doi:10.1016/S0968-4328(01)00010-5. PMID 11567889. Archived from the original on 2020-03-17. Retrieved 2019-06-13.
- ^ Brook, P. J. (26 April 1969). "Stimulation of Ascospore Release in Venturia inaequalis by Far Red Light". Nature. 222 (5191): 390–392. Bibcode:1969Natur.222..390B. doi:10.1038/222390a0. ISSN 0028-0836. S2CID 4293713.
- ^ a b c d Meuthen, Denis; Rick, Ingolf P.; Thünken, Timo; Baldauf, Sebastian A. (2012). "Visual prey detection by near-infrared cues in a fish". Naturwissenschaften. 99 (12): 1063–6. Bibcode:2012NW.....99.1063M. doi:10.1007/s00114-012-0980-7. PMID 23086394. S2CID 4512517.
- ^ Endo, M.; Kobayashi R.; Ariga, K.; Yoshizaki, G.; Takeuchi, T. (2002). "Postural control in tilapia under microgravity and the near infrared irradiated conditions". Nippon Suisan Gakkaishi. 68 (6): 887–892. doi:10.2331/suisan.68.887.
- ^ Kobayashi R.; Endo, M.; Yoshizaki, G.; Takeuchi, T. (2002). "Sensitivity of tilapia to infrared light measured using a rotating striped drum differs between two strains". Nippon Suisan Gakkaishi. 68 (5): 646–651. doi:10.2331/suisan.68.646.
- ^ Matsumoto, Taro; Kawamura, Gunzo (2005). "The eyes of the common carp and Nile tilapia are sensitive to near-infrared". Fisheries Science. 71 (2): 350–355. Bibcode:2005FisSc..71..350M. doi:10.1111/j.1444-2906.2005.00971.x. S2CID 24556470.
- ^ a b c Shcherbakov, Denis; Knörzer, Alexandra; Hilbig, Reinhard; Haas, Ulrich; Blum, Martin (2012). "Near-infrared orientation of Mozambique tilapia Oreochromis mossambicus". Zoology. 115 (4): 233–238. Bibcode:2012Zool..115..233S. doi:10.1016/j.zool.2012.01.005. PMID 22770589.
- ^ Hargate, G (2006). "A randomised double-blind study comparing the effect of 1072-nm light against placebo for the treatment of herpes labialis". Clinical and Experimental Dermatology. 31 (5): 638–41. doi:10.1111/j.1365-2230.2006.02191.x. PMID 16780494. S2CID 26977101.
- ^ Desmet KD, Paz DA, Corry JJ, Eells JT, Wong-Riley MT, Henry MM, Buchmann EV, Connelly MP, Dovi JV, Liang HL, Henshel DS, Yeager RL, Millsap DS, Lim J, Gould LJ, Das R, Jett M, Hodgson BD, Margolis D, Whelan HT (May 2006). "Clinical and experimental applications of NIR-LED photobiomodulation". Photomedicine and Laser Surgery. 24 (2): 121–8. doi:10.1089/pho.2006.24.121. PMID 16706690. S2CID 22442409. Archived from the original on 2020-03-16. Retrieved 2019-06-13.
- ^ Rosso, Monona l (2001). The Artist's Complete Health and Safety Guide. Allworth Press. pp. 33–. ISBN 978-1-58115-204-3.
- ^ Herschel, William (1800). "Experiments on the refrangibility of the invisible rays of the Sun". Philosophical Transactions of the Royal Society of London. 90: 284–292. doi:10.1098/rstl.1800.0015. JSTOR 107057. Archived from the original on 2021-02-04. Retrieved 2018-04-11.
- ^ "Herschel Discovers Infrared Light". Coolcosmos.ipac.caltech.edu. Archived from the original on 2012-02-25. Retrieved 2011-11-08.
- ^ In 1867, French physicist Edmond Becquerel coined the term infra-rouge (infra-red):
- Becquerel, Edmond (1867). La Lumiere: Ses causes et ses effets [Light: Its causes and effects] (in French). Paris, France: Didot Frères, Fils et Cie. pp. 141–145. Archived from the original on 2024-09-22. Retrieved 2018-04-15.
- de Saint-Florent (10 April 1874). "Photography in natural colours". The Photographic News. 18: 175–176. Archived from the original on 5 February 2021. Retrieved 15 April 2018. From p. 176: "As to the infra-red rays, they may be absorbed by means of a weak solution of sulphate of copper, ..."
- Rosenberg, Gary (2012). "Letter to the Editors: Infrared dating". American Scientist. 100 (5): 355. Archived from the original on 2018-04-15. Retrieved 2018-04-15.
- ^ Harper, Douglas. "infrared". Online Etymology Dictionary. Retrieved 2025-04-07.
- ^ Chang, Hasok (2007). Inventing temperature: measurement and scientific progress. Oxford studies in philosophy of science (1. issued as paperback ed.). Oxford: Oxford University Press. pp. 166–167. ISBN 978-0-19-533738-9.
- ^ See:
- Nobili, Leopoldo (1830). "Description d'un thermo-multiplicateur ou thermoscope électrique" [Description of a thermo-multiplier or electric thermoscope]. Bibliothèque Universelle (in French). 44: 225–234. Archived from the original on 2021-02-24. Retrieved 2018-04-12.
- Nobili; Melloni (1831). "Recherches sur plusieurs phénomènes calorifiques entreprises au moyen du thermo-multiplicateur" [Investigations of several heat phenomena undertaken via a thermo-multiplier]. Annales de Chimie et de Physique. 2nd series (in French). 48: 198–218. Archived from the original on 2021-02-05. Retrieved 2018-04-12.
- Vollmer, Michael; Möllmann, Klaus-Peter (2010). Infrared Thermal Imaging: Fundamentals, Research and Applications (2nd ed.). Berlin, Germany: Wiley-VCH. pp. 1–67. ISBN 978-3-527-69329-0. Archived from the original on 2024-09-22. Retrieved 2018-04-12.
- ^ Herschel, John F. W. (1840). "On chemical action of rays of solar spectrum on preparation of silver and other substances both metallic and nonmetallic and on some photographic processes". Philosophical Transactions of the Royal Society of London. 130: 1–59. Bibcode:1840RSPT..130....1H. doi:10.1098/rstl.1840.0002. S2CID 98119765. Archived from the original on 2021-02-05. Retrieved 2018-04-09. The term "thermograph" is coined on p. 51: " ... I have discovered a process by which the calorific rays in the solar spectrum are made to leave their impress on a surface properly prepared for the purpose, so as to form what may be called a thermograph of the spectrum, ... ".
- ^ See:
- Kirchhoff (1859). "Ueber den Zusammenhang von Emission und Absorption von Licht und Warme" [On the relation between emission and absorption of light and heat]. Monatsberichte der Königlich-Preussischen Akademie der Wissenschaften zu Berlin (Monthly Reports of the Royal Prussian Academy of Philosophy in Berlin) (in German): 783–787. Archived from the original on 2020-09-25. Retrieved 2018-04-10.
- Kirchhoff, G. (1860). "Ueber das Verhältnis zwischen dem Emissionsvermögen und dem Absorptionsvermögen der Körper für Wärme und Licht" [On the relation between bodies' emission capacity and absorption capacity for heat and light]. Annalen der Physik und Chemie (in German). 109 (2): 275–301. Bibcode:1860AnP...185..275K. doi:10.1002/andp.18601850205. Retrieved 2018-04-10.
- English translation: Kirchhoff, G. (1860). "On the relation between the radiating and absorbing powers of different bodies for light and heat". Philosophical Magazine. 4th series. 20: 1–21. Archived from the original on 2021-02-05. Retrieved 2018-04-11.
- ^ See:
- Smith, Willoughby (1873). "The action of light on selenium". Journal of the Society of Telegraph Engineers. 2 (4): 31–33. doi:10.1049/jste-1.1873.0023. Archived from the original on 2024-10-03. Retrieved 2018-04-09.
- Smith, Willoughby (20 February 1873). "Effect of light on selenium during the passage of an electric current". Nature. 7 (173): 303. Bibcode:1873Natur...7R.303.. doi:10.1038/007303e0. Archived from the original on 3 January 2021. Retrieved 9 April 2018.
- ^ See:
- Langley, S. P. (1880). "The bolometer". Proceedings of the American Metrological Society. 2: 184–190. Archived from the original on 2021-02-05. Retrieved 2018-04-09.
- Langley, S. P. (1881). "The bolometer and radiant energy". Proceedings of the American Academy of Arts and Sciences. 16: 342–358. doi:10.2307/25138616. JSTOR 25138616. Archived from the original on 2021-02-05. Retrieved 2018-04-09.
- ^ Stefan, J. (1879). "Über die Beziehung zwischen der Wärmestrahlung und der Temperatur" [On the relation between heat radiation and temperature]. Sitzungsberichte der Kaiserlichen Akademie der Wissenschaften [Wien]: Mathematisch-naturwissenschaftlichen Classe (Proceedings of the Imperial Academy of Philosophy [in Vienna]: Mathematical-scientific Class) (in German). 79: 391–428. Archived from the original on 2019-04-02. Retrieved 2018-04-11.
- ^ See:
- Wien, Willy (1896). "Ueber die Energieverteilung im Emissionsspektrum eines schwarzen Körpers" [On the energy distribution in the emission spectrum of a black body]. Annalen der Physik und Chemie. 3rd series (in German). 58: 662–669. Archived from the original on 2021-02-24. Retrieved 2018-04-10.
- English translation: Wien, Willy (1897). "On the division of energy in the emission-spectrum of a black body". Philosophical Magazine. 5th series. 43 (262): 214–220. doi:10.1080/14786449708620983. Archived from the original on 2021-02-05. Retrieved 2018-04-10.
- ^ Julius, Willem Henri (1892). Bolometrisch onderzoek van absorptiespectra (in Dutch). J. Müller. Archived from the original on 2024-09-22. Retrieved 2020-10-18.
- ^ See:
- Planck, M. (1900). "Ueber eine Verbesserung der Wien'schen Spectralgleichung" [On an improvement of Wien's spectral equation]. Verhandlungen der Deutschen Physikalischen Gesellschaft (in German). 2: 202–204. Archived from the original on 2024-09-22. Retrieved 2018-04-10.
- Planck, M. (1900). "Zur Theorie des Gesetzes der Energieverteilung im Normalspectrum" [On the theory of the law of energy distribution in the normal spectrum]. Verhandlungen der Deutschen Physikalischen Gesellschaft (in German). 2: 237–245. Archived from the original on 2021-02-25. Retrieved 2018-04-10.
- Planck, Max (1901). "Ueber das Gesetz der Energieverteilung im Normalspectrum" [On the law of energy distribution in the normal spectrum]. Annalen der Physik. 4th series (in German). 4 (3): 553–563. Bibcode:1901AnP...309..553P. doi:10.1002/andp.19013090310. Archived from the original on 2021-02-06. Retrieved 2018-04-10.
- ^ See:
- Einstein, A. (1905). "Über einen die Erzeugung und Verwandlung des Lichtes betreffenden heuristischen Gesichtspunkt" [On heuristic viewpoint concerning the production and transformation of light]. Annalen der Physik. 4th series (in German). 17 (6): 132–148. Bibcode:1905AnP...322..132E. doi:10.1002/andp.19053220607.
- English translation: Arons, A. B.; Peppard, M. B. (1965). "Einstein's proposal of the photon concept—a translation of the Annalen der Physik paper of 1905". American Journal of Physics. 33 (5): 367–374. Bibcode:1965AmJPh..33..367A. doi:10.1119/1.1971542. S2CID 27091754. Available at Wayback Machine.
- ^ Coblentz, William Weber (1905). Investigations of Infra-red Spectra: Part I, II. Carnegie institution of Washington. Archived from the original on 2024-09-22. Retrieved 2020-10-18.
- ^ Coblentz, William Weber (1905). Investigations of Infra-red Spectra: Part III, IV. University of Michigan. Washington, D.C., Carnegie institution of Washington.
- ^ Coblentz, William Weber (August 1905). Investigations of Infra-red Spectra: Part V, VI, VII. University of California Libraries. Washington, D.C. : Carnegie Institution of Washington.
- ^ Waste Energy Harvesting: Mechanical and Thermal Energies. Springer Science & Business Media. 2014. p. 406. ISBN 978-3-642-54634-1. Archived from the original on 2024-09-22. Retrieved 2020-01-07.
- ^ a b Marion B. Reine (2015). "Interview with Paul W. Kruse on the Early History of HgCdTe (1980)" (PDF). Journal of Electronic Materials. 44 (9). doi:10.1007/s11664-015-3737-1. S2CID 95341284. Archived (PDF) from the original on 2020-07-30. Retrieved 2020-01-07.
- ^ J Cooper (1962). "A fast-response pyroelectric thermal detector". Journal of Scientific Instruments. 39 (9): 467–472. Bibcode:1962JScI...39..467C. doi:10.1088/0950-7671/39/9/308.
- ^ "History of Army Night Vision". C5ISR Center. Archived from the original on March 16, 2020. Retrieved 2020-01-07.
- ^ "Implant gives rats sixth sense for infrared light". Wired UK. 14 February 2013. Archived from the original on 17 February 2013. Retrieved 14 February 2013.
External links
[edit]- Infrared: A Historical Perspective Archived 2007-08-07 at the Wayback Machine (Omega Engineering)
- Infrared Data Association Archived 2008-05-22 at the Wayback Machine, a standards organization for infrared data interconnection
- SIRC Protocol Archived 2011-10-13 at the Wayback Machine
- How to build a USB infrared receiver to control PC's remotely Archived 2011-07-19 at the Wayback Machine
- Infrared Waves: detailed explanation of infrared light. (NASA)
- Herschel's original paper from 1800 announcing the discovery of infrared light
- The thermographic's library Archived 2013-06-11 at the Wayback Machine, collection of thermogram
- Infrared reflectography in analysis of paintings Archived 2015-12-22 at the Wayback Machine at ColourLex
- Molly Faries, Techniques and Applications – Analytical Capabilities of Infrared Reflectography: An Art Historian s Perspective Archived 2015-12-22 at the Wayback Machine, in Scientific Examination of Art: Modern Techniques in Conservation and Analysis, Sackler NAS Colloquium, 2005
Infrared
View on GrokipediaFundamentals
Definition and Electromagnetic Spectrum
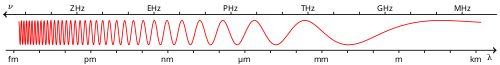
Infrared radiation, often abbreviated as IR, is a form of electromagnetic radiation characterized by wavelengths ranging from approximately 700 nanometers (nm) to 1 millimeter (mm). This corresponds to a frequency range of about 430 terahertz (THz) to 300 gigahertz (GHz).[6] These parameters place infrared just beyond the red end of the visible spectrum, where human eyes perceive light up to around 700 nm.[7] Within the broader electromagnetic spectrum, infrared occupies the band between visible light—with its shorter wavelengths from about 400 nm to 700 nm—and microwaves, which extend from 1 mm to 1 meter in wavelength. This positioning means infrared has longer wavelengths and lower frequencies than visible light but higher frequencies than microwaves, influencing its interactions with matter, such as penetration and absorption properties. Infrared radiation is emitted by all objects above absolute zero due to thermal motion, though this thermal aspect is explored further elsewhere.[6] As with all electromagnetic radiation, infrared demonstrates wave-particle duality, manifesting as propagating waves or discrete packets of energy known as photons. The energy of an individual infrared photon is determined by the formulawhere is Planck's constant ( J·s) and is the photon's frequency. This relationship underscores the quantized nature of infrared energy, with photon energies typically ranging from about 1.24 millielectronvolts (meV) to 1.7 electronvolts (eV) across the infrared band.[9] Infrared wavelengths are typically measured in nanometers (nm) for near-infrared regions or micrometers (μm) for longer wavelengths, with 1 μm equaling 1000 nm. In infrared spectroscopy, the reciprocal of wavelength, known as wavenumber and expressed in inverse centimeters (cm⁻¹), is commonly used; for example, the infrared range spans roughly 14,000 cm⁻¹ to 10 cm⁻¹. This unit facilitates analysis of molecular vibrations, as higher wavenumbers correspond to higher energies.
Physical Properties
Infrared radiation, as a form of electromagnetic waves, propagates through vacuum at the speed of light, m/s.[10] In various media, its speed is reduced according to , where is the refractive index of the medium, which for infrared wavelengths typically ranges from about 1.3 to 4 depending on the material, such as glasses or semiconductors used in optics.[11] This velocity reduction leads to phenomena like refraction when infrared waves pass through interfaces between media, enabling applications in lenses and waveguides.[12] Infrared radiation interacts with matter primarily through absorption, reflection, transmission, and scattering processes.[13] Absorption occurs when infrared photons excite molecular vibrations, converting energy into heat; reflection and transmission depend on the material's surface properties and refractive index contrast; while scattering redirects waves without significant energy loss, often via Rayleigh or Mie mechanisms for small particles.[14] In the Earth's atmosphere, these interactions are pronounced, with strong absorption bands due to water vapor (particularly in the 2.7 μm and 6.3 μm regions) and carbon dioxide (around 4.3 μm and 15 μm), which attenuate infrared transmission over long paths and contribute to the greenhouse effect.[15] Atmospheric aerosols further enhance scattering and absorption, reducing visibility in the infrared spectrum.[14] A key aspect of infrared-matter interaction is penetration depth, which varies by wavelength and material. Near-infrared (0.7–1.4 μm) penetrates biological tissues deeper, up to 10–15 mm in models like bovine tissue, due to lower absorption by water and hemoglobin compared to longer wavelengths.[16] In contrast, far-infrared (above 3 μm) is absorbed more superficially, up to several centimeters (e.g., ~4 cm) in skin, as it strongly excites water molecule vibrations, enabling heating effects in deeper tissues while primarily interacting with surface layers.[17] Infrared radiation exhibits polarization and coherence properties inherent to electromagnetic waves. Polarization describes the orientation of the electric field vector, which can be linear, circular, or elliptical; natural sources like thermal emitters produce unpolarized infrared, but it can be polarized through reflection, scattering, or devices like wire-grid polarizers.[18] Coherence refers to the phase correlation of waves: thermal infrared sources, such as blackbodies, are typically incoherent with short coherence lengths (on the order of micrometers), leading to diffuse emission, whereas infrared lasers generate highly coherent radiation with long coherence lengths (centimeters to meters), enabling interferometry and precise imaging.[19][20]Spectral Regions
Common Subdivision Schemes
The infrared spectrum is commonly subdivided into three primary regions—near-infrared (NIR), mid-infrared (MIR), and far-infrared (FIR)—based on wavelength ranges that align with distinct physical interactions and technological capabilities. The near-infrared region spans approximately 0.7 to 1.4 μm, overlapping with the visible spectrum and enabling applications like fiber optics and night vision due to its proximity to visible light.[21] The mid-infrared region extends from 1.4 to 15 μm, often further divided into short-wavelength infrared (1.4–3 μm) and longer mid-wave (3–15 μm) sub-bands to reflect varying absorption characteristics.[22] The far-infrared region covers 15 to 1000 μm, where thermal emission dominates and wavelengths approach millimeter scales.[21] These divisions arise from key physical and practical considerations, including the excitation of molecular vibrations, which are most prominently observed in the mid-infrared where fundamental stretching and bending modes occur, providing a basis for spectroscopic identification of chemical bonds.[23] Atmospheric transmission windows also influence the boundaries, with relatively clear paths in the near-infrared (around 0.7–1.1 μm) and mid-infrared (3–5 μm and 8–14 μm) allowing for effective ground-based observations and remote sensing, while stronger absorption by water vapor and CO₂ limits transmission elsewhere.[24] Detector sensitivities further justify the scheme, as silicon-based photodiodes excel in the near-infrared, mercury cadmium telluride (MCT) detectors perform optimally in the mid-infrared, and microbolometers or cryogenic sensors are required for the far-infrared due to lower photon energies.[21] In spectroscopic contexts, these wavelength ranges correspond to specific wavenumber intervals, with the mid-infrared commonly expressed as 4000–400 cm⁻¹ to encompass the functional group and fingerprint regions for molecular analysis.[23] Boundaries exhibit overlaps and variations across disciplines; for instance, optics often uses 0.78–3 μm for near- to mid-infrared transitions to align with laser sources, while spectroscopy may extend near-infrared to 2.5 μm to include overtone vibrations.[21] Such inconsistencies stem from historical conventions and application-specific needs, like eye safety standards (e.g., IR-A for near-infrared) versus thermal imaging bands.[22]Specialized Division Schemes
The International Commission on Illumination (CIE) defines infrared radiation as optical radiation with wavelengths longer than the visible spectrum, starting at 780 nm where human visual response diminishes, though sensation may extend slightly beyond for intense sources; it further subdivides IR into IR-A (780 nm to 1.4 μm), IR-B (1.4 to 3 μm), and IR-C (3 μm to 1 mm) to align with physiological effects on the eye.[25] This scheme prioritizes boundaries tied to human perception and biological interaction rather than uniform wavelength intervals. The ISO 20473:2007 standard, developed for optics and photonics applications excluding lighting and telecommunications, divides infrared into short-wave (near-infrared, 0.78–3 μm), mid-wave (3–50 μm), and long-wave (50–1000 μm) regions to standardize spectral band descriptions across technical fields.[17] Unlike broader schemes, this emphasizes practical delimitation for instrument calibration and material testing, with mid-wave encompassing vibrational spectroscopy bands and long-wave covering thermal emissions. In astronomy, infrared is segmented into near-IR (0.8–5 μm), mid-IR (5–30 μm), and far-IR (30–1000 μm) to match observational challenges like atmospheric absorption and telescope capabilities; the far-IR regime is particularly vital for detecting thermal emissions from cosmic dust grains, which absorb ultraviolet-visible starlight and re-emit at these longer wavelengths, revealing interstellar structures such as molecular clouds and protostellar envelopes.[26] Telecommunications employs fine-grained near-IR bands optimized for low-loss propagation in silica fiber optics, including the O-band (1260–1360 nm) for short-haul metropolitan networks and the C-band (1530–1565 nm) for long-haul amplification via erbium-doped fibers, enabling high-capacity data transmission with minimal signal attenuation.[27] Infrared divisions based on sensor response align with detector material sensitivities, such as indium antimonide (InSb) photodiodes effective in the mid-IR (1–5.5 μm) for high-speed, cooled applications like thermal imaging, while mercury cadmium telluride (HgCdTe) extends to long-wave IR (up to 15 μm) and bolometers handle far-IR beyond 20 μm via thermal absorption.[28] These material-driven boundaries facilitate selection for specific detection tasks, prioritizing quantum efficiency over arbitrary wavelength cuts.Thermal Radiation
Infrared as Heat Radiation
Infrared radiation serves as the primary mechanism for thermal emission from objects at room temperature, where the peak wavelength of emission falls within the long-wavelength infrared range around 10 μm, as determined by Wien's displacement law. This law states that the wavelength at which a blackbody emits the maximum intensity of radiation, λ_max, is inversely proportional to its absolute temperature T, given by the formula λ_max T = 2.897 × 10^{-3} m·K. For typical room temperatures of approximately 300 K, this yields λ_max ≈ 9.7 μm, placing the strongest emission in the infrared spectrum and explaining why everyday objects like walls or furniture radiate heat primarily as infrared waves.[29] Kirchhoff's law of thermal radiation further elucidates this process by establishing that, at thermal equilibrium and for a given wavelength, the emissivity ε of a surface—which measures its efficiency in emitting radiation—equals its absorptivity α, the fraction of incident radiation it absorbs. Thus, surfaces that are good absorbers of infrared radiation, such as dark or matte materials, are also efficient emitters of thermal infrared at the same wavelengths, ensuring a balance in radiative heat exchange. This principle underlies why blackened surfaces, which absorb nearly all incident radiation (α ≈ 1), emit strongly when heated, while reflective surfaces like polished metals emit poorly.[30] A practical example is the human body, which at a normal temperature of about 310 K emits peak infrared radiation in the long-wavelength infrared band at approximately 9.3 μm, contributing to sensible heat loss in environments. In daily life, infrared facilitates heat transfer through radiation, such as the warmth felt from sunlight on the skin—where solar infrared accounts for much of the heating effect—or the radiative cooling of a hot cup of coffee to the surrounding air, independent of conduction or convection.[31][32] It is important to distinguish thermal infrared sources, which arise from the random thermal motion of particles in matter and follow blackbody-like spectra dependent on temperature, from non-thermal sources that produce infrared without significant heat generation. Non-thermal examples include infrared lasers or light-emitting diodes (LEDs) used in remote controls, which emit coherent or directed beams at specific wavelengths like 940 nm through electronic excitation rather than thermal processes.[7]Blackbody Emission
A blackbody is defined as an idealized physical body that absorbs all incident electromagnetic radiation, regardless of frequency or angle of incidence, and re-emits energy solely based on its temperature, with no reflection or transmission.[33] This perfect absorber also serves as a perfect emitter, providing a theoretical benchmark for thermal radiation spectra.[34] The spectral radiance of blackbody radiation is described by Planck's law, which quantifies the power emitted per unit area per unit solid angle per unit frequency as a function of wavelength and temperature : where is Planck's constant, is the speed of light, and is Boltzmann's constant.[34] This formula reveals that the emission spectrum peaks at a wavelength that shifts with temperature, with significant infrared contributions dominating at lower temperatures due to the exponential term in the denominator.[35] Wien's displacement law further characterizes this peak shift, stating that the wavelength at which the spectral radiance is maximum satisfies , where is Wien's displacement constant.[35] For terrestrial temperatures around 300 K, falls in the long-wavelength infrared range near 10 m, explaining why blackbody emission from everyday objects is predominantly infrared.[36] The total radiant power emitted by a blackbody is governed by the Stefan-Boltzmann law, , where is the Stefan-Boltzmann constant and is the surface area.[37] At temperatures below 1000 K, the majority of this power is emitted in the infrared spectrum, as shorter wavelengths become negligible.[36]Detection and Measurement
Infrared Detectors
Infrared detectors convert incident infrared radiation into measurable electrical signals, broadly categorized into thermal detectors, which respond to the heat generated by absorbed photons, and quantum detectors, which rely on photon-induced electronic transitions. Thermal detectors operate by sensing temperature changes caused by radiation absorption and can function across all infrared spectral bands without wavelength selectivity, making them suitable for broadband detection. Quantum detectors, in contrast, exhibit wavelength-specific responses based on material bandgaps and typically offer higher sensitivity but often require cooling to minimize thermal noise.[38][39] Thermal detectors include bolometers, which measure radiation through changes in electrical resistance due to heating of a sensitive element, such as a thin film of vanadium oxide or amorphous silicon. The resistance variation is proportional to the temperature rise from absorbed infrared power, enabling room-temperature operation with responsivities on the order of 10^4 to 10^5 V/W in microbolometer arrays. Pyroelectric detectors, another type of thermal device, generate a voltage in response to rapid temperature changes via the pyroelectric effect in materials like lithium tantalate or PZT ceramics, producing charge proportional to the rate of incident radiation flux. These detectors are inherently AC-coupled, requiring modulation of the input signal, and achieve noise equivalent powers (NEPs) as low as 10^{-9} W/√Hz at room temperature. Both bolometer and pyroelectric types are uncooled and versatile for near- to far-infrared detection, though they exhibit slower response times (milliseconds) compared to quantum alternatives.[40][41][42] Quantum detectors, such as photodiodes, operate by absorbing photons to excite charge carriers across a bandgap, with materials like mercury cadmium telluride (HgCdTe) commonly used for mid-infrared detection due to its tunable bandgap (0.1–1.5 eV) via composition control. In photovoltaic mode, the photodiode generates a voltage without external bias, relying on the built-in electric field to separate electron-hole pairs, which suits low-noise, power-limited applications. Photoconductive mode applies reverse bias to increase carrier collection speed and gain, though it introduces higher dark current; quantum efficiency η, defined as quantifies the conversion efficiency, often exceeding 70% for optimized HgCdTe devices in the 3–5 μm band. These detectors provide high speed (nanoseconds) and spectral selectivity but suffer from thermal generation noise at longer wavelengths.[43][44][45] For mid- and far-infrared quantum detection, cryogenic cooling is essential to reduce thermal excitation of carriers, which otherwise overwhelms the signal; typical operating temperatures are 77 K using liquid nitrogen or below 50 K with mechanical coolers for HgCdTe or InSb devices, achieving background-limited performance with dark currents reduced by orders of magnitude. Uncooled operation is feasible for short-wave infrared but degrades detectivity in longer bands due to increased noise. A key figure of merit for comparing detector performance is the specific detectivity D^*, given by where A is the detector area, Δf is the electrical bandwidth, and NEP is the noise equivalent power (the incident power yielding a signal-to-noise ratio of 1 in 1 Hz bandwidth, in W/√Hz); D^* normalizes for size and speed, with values around 10^{10} cm √Hz/W for room-temperature thermal detectors and up to 10^{12} cm √Hz/W for cooled quantum types.[46][47][48]Spectroscopy Techniques
Infrared spectroscopy techniques enable the analysis of materials by measuring their interaction with infrared radiation, primarily through absorption, emission, or reflection of light in the mid-infrared region (approximately 4000–400 cm⁻¹). These methods exploit the fact that molecules absorb IR radiation at frequencies corresponding to their vibrational transitions, producing spectra that serve as unique signatures for chemical identification and quantification. Instrumentation typically involves a broadband IR source, a sample interface, and detectors to record the modulated signal, with data processed to yield absorbance or transmittance spectra.[49] Fourier-transform infrared (FTIR) spectroscopy is the predominant technique, utilizing a Michelson interferometer to achieve high-resolution spectra across a wide wavelength range simultaneously. In the Michelson setup, incoming IR radiation is split by a beam splitter into two paths: one reflects off a fixed mirror, while the other travels to a movable mirror that scans along the optical axis, creating a variable path difference. The recombined beams interfere, producing an interferogram—a time-domain signal that encodes the spectral information—which is then converted to a frequency-domain spectrum via Fourier transform. This approach offers advantages over dispersive methods, including higher signal-to-noise ratios and faster acquisition times. The spectral resolution in FTIR, denoted as Δν (in cm⁻¹), is determined by the maximum mirror displacement L (in cm), following the relation Δν = 1/(2L), where 2L represents the maximum optical path difference. For example, a maximum path difference of 2 cm yields a resolution of 0.5 cm⁻¹, allowing distinction of closely spaced absorption bands.[49][49] Absorption spectroscopy, often performed in transmission mode, quantifies how much IR light is absorbed by a sample as it passes through, providing direct insight into molecular concentrations. The fundamental principle is the Beer-Lambert law, which states that absorbance A is linearly proportional to the analyte concentration c (in mol/L), the path length l (in cm), and the molar absorptivity ε (in L mol⁻¹ cm⁻¹) at a specific wavelength:This law holds for dilute solutions or gases where interactions are minimal, enabling quantitative analysis such as determining protein concentrations in biochemical samples from characteristic amide band intensities. In practice, samples are prepared as thin films, pellets (e.g., KBr matrices for solids), or gas cells to ensure sufficient transmission without saturation. Deviations occur at high concentrations due to non-ideal behaviors, but the law remains foundational for calibration curves in analytical applications.[50][50] Emission and reflection modes extend IR spectroscopy to samples unsuitable for transmission, such as opaque solids or viscous liquids, by measuring emitted or reflected radiation instead of transmitted light. In emission spectroscopy, thermally excited samples (e.g., at elevated temperatures) radiate IR light characteristic of their vibrational modes, useful for studying hot gases or surfaces. Reflection techniques, particularly attenuated total reflectance (ATR), facilitate analysis without extensive preparation by exploiting total internal reflection at a high-refractive-index crystal (e.g., diamond or ZnSe) in contact with the sample. The evanescent wave penetrates ~1–5 μm into the sample, interacting with it and attenuating based on absorption; the reflected beam is then analyzed via FTIR. ATR is ideal for solids and liquids, requiring only a small amount of material pressed against the crystal, and spectra are corrected for the wavelength-dependent penetration depth to mimic transmission data. This mode minimizes artifacts from sample thickness and is widely used for quality control in polymers and pharmaceuticals. A key aspect of spectral interpretation in mid-IR spectroscopy is the functional group region (4000–1500 cm⁻¹), featuring characteristic absorptions from functional groups, such as O-H stretches near 3500 cm⁻¹ or C=O stretches around 1700 cm⁻¹. The fingerprint region (1500–400 cm⁻¹), where complex combinations of stretching and bending vibrations produce unique patterns for molecular identification, allows differentiation of isomers or contaminants by matching against reference libraries. Unlike the higher-wavenumber region, the fingerprint area is more skeletal-specific and provides a holistic "signature" for compound verification, often confirmed by overlaying experimental spectra with standards in database searches.[51]
Applications
Imaging and Sensing
Thermal imaging, also known as thermography, utilizes passive detection of infrared radiation emitted by objects to create visual maps of temperature distributions. This technique captures the thermal emissions in the mid-wave or long-wave infrared spectrum, where objects above absolute zero radiate heat according to their surface temperature, enabling non-contact assessment without external illumination.[52] The spatial resolution of such systems is fundamentally constrained by optical diffraction, approximated by the Abbe limit , where is the wavelength and is the numerical aperture of the imaging optics, limiting the smallest resolvable feature to scales on the order of microns to millimeters depending on the infrared band employed.[53] Night vision systems leverage infrared for low-light visibility, distinguishing between active and passive approaches. Active systems employ near-infrared illumination, such as laser or LED sources around 850–1060 nm, to reflect off targets and enhance visibility in complete darkness, often paired with image intensifiers that amplify the reflected photons for visible output.[54] In contrast, passive thermal night vision detects naturally emitted mid- or long-wave infrared radiation using focal plane arrays (FPAs), such as uncooled microbolometers or cooled photon detectors, to form images based on heat signatures without any active source, providing effective detection in total darkness but with lower resolution compared to active methods.[55] Image intensifiers excel in near-infrared amplification for brighter, higher-contrast images under minimal ambient light, while FPAs dominate thermal imaging for their sensitivity to temperature differentials.[56] Hyperspectral infrared imaging extends beyond standard broadband capture by recording data across numerous contiguous narrow spectral bands, typically tens to hundreds in the short-, mid-, or long-wave infrared regions, forming a three-dimensional spectral cube for detailed analysis. This allows precise material discrimination by exploiting unique spectral reflectance or emissivity signatures, such as distinguishing vegetation types or detecting camouflaged objects through subtle absorption features invisible to panchromatic sensors.[57] Systems often integrate tunable Fabry-Pérot filters or dispersive gratings with focal plane arrays to achieve high spectral resolution, enabling applications in remote material identification with spectral resolutions on the order of 10–50 nm (or equivalent in wavenumbers).[57] In remote sensing, satellite-based infrared imaging supports global earth observation by monitoring thermal and reflective properties across large scales. The Moderate Resolution Imaging Spectroradiometer (MODIS) on NASA's Terra and Aqua satellites exemplifies this, featuring multiple infrared bands such as Band 21 (3.929–3.989 µm) for fire detection, Bands 31–32 (10.780–12.270 µm) for surface temperature mapping, and Bands 20, 27–29 (3.660–8.700 µm) for cloud and atmospheric profiling, providing daily coverage at resolutions from 250 m to 1 km.[58] These bands capture emitted thermal radiation for deriving land surface temperatures and vegetation health indices, with thermal contrast arising from blackbody emission variations.[58]Communications and Heating
Infrared radiation plays a crucial role in optical communications, particularly in free-space and fiber-optic systems, where near-infrared wavelengths enable high-bandwidth data transmission with minimal atmospheric or material interference. Free-space optical (FSO) communication utilizes near-IR lasers, such as those operating at 1550 nm, to establish wireless links capable of terabit-per-second rates over distances up to several kilometers, offering an alternative to radio-frequency systems in scenarios like satellite-to-ground or urban networking. These systems often employ on-off keying (OOK) modulation for direct detection, where the laser beam is intensity-modulated to encode binary data, achieving bit error rates below 10^{-9} under clear atmospheric conditions with adaptive optics to mitigate turbulence. For instance, coherent FSO links at 1550 nm have demonstrated 100 Gbps transmission over 500–700 m with low-orbit satellite-like tracking rates, leveraging dual-polarization quadrature phase-shift keying alongside OOK for enhanced spectral efficiency.[59][60] Fiber-optic telecommunications predominantly rely on infrared wavelengths around the 1.55 μm window in silica fibers, where attenuation is minimized to approximately 0.2 dB/km due to low Rayleigh scattering and negligible multiphonon absorption, enabling long-haul transmission over thousands of kilometers without amplification. This spectral region, corresponding to the third telecommunication window (C-band, 1530–1565 nm), supports dense wavelength-division multiplexing (DWDM) with up to 80 channels, each carrying 100 Gbps or more, as silica's transparency peaks near this wavelength from intrinsic material properties. Erbium-doped fiber amplifiers further exploit this band for signal regeneration, making 1.55 μm the standard for global internet backbone infrastructure.[61][62] Beyond communications, infrared is integral to thermal applications, including heating and cooling processes that leverage radiative transfer for energy efficiency. Infrared radiative heaters, often using far-IR emitters like ceramic or quartz elements peaking at 2–8 μm, are employed in industrial drying operations such as paint curing on automotive parts or solvent evaporation in textiles, where direct radiation penetrates surfaces to accelerate moisture removal at rates up to 50% faster than convective methods. The efficiency of these heaters, typically 60–90% in converting electrical input to radiant output, depends on the emissivity ε of both the emitter and target material—high-ε surfaces (ε ≈ 0.8–0.9 for oxidized metals or organics) absorb up to 90% of incident energy, optimizing heat transfer while minimizing convection losses in enclosed systems.[63][64] Infrared-based radiative cooling achieves sub-ambient temperatures passively by employing selective emitters that radiate heat through the atmospheric transparency window of 8–13 μm, where Earth's atmosphere is nearly transparent, allowing thermal photons to escape to space at 3 K. These metamaterial or photonic structures, such as multilayer silicon dioxide films, exhibit high emissivity (ε > 0.9) in this band while reflecting >95% of solar radiation, with demonstrated net cooling powers of around 40–100 W/m² daytime and higher nighttime values up to ~120 W/m² under clear skies, achieving temperature drops of 5–10°C below ambient air temperature. This approach, distinct from near-IR telecom uses, targets far-IR for thermal management in buildings and electronics without energy input.[65][66]Scientific and Medical Uses
Infrared radiation plays a pivotal role in astronomy by enabling observations that penetrate interstellar dust clouds, which obscure visible light. The James Webb Space Telescope (JWST) utilizes near-infrared (NIR) and mid-infrared (MIR) instruments, such as the Near-Infrared Camera (NIRCam) and the Mid-Infrared Instrument (MIRI), to capture wavelengths from approximately 0.6 to 28.5 μm. These capabilities allow astronomers to study star-forming regions within dense nebulae, like the Pillars of Creation, where NIR light reveals embedded protostars and warm dust that would otherwise be hidden. As of 2025, JWST observations have revealed detailed exoplanet atmospheres and confirmed galaxies from 300 million years after the Big Bang, leveraging IR to penetrate cosmic dust.[67][68] Furthermore, infrared astronomy facilitates the detection of redshifted light from distant galaxies, where cosmological expansion stretches ultraviolet and visible emissions into longer IR wavelengths. JWST's NIRSpec spectrograph, operating from 0.6 to 5.3 μm, analyzes this light to probe the early universe, including the formation of the first stars and galaxies at redshifts greater than 7, providing insights into cosmic evolution and dark matter. MIRI's medium-resolution spectroscopy (4.9–27.9 μm) complements this by characterizing molecular gas and dust in high-redshift environments.[67][69][68] In medicine, infrared light is harnessed through photobiomodulation (PBM) therapy, which employs red and near-infrared wavelengths (600–1000 nm) to promote wound healing by stimulating cellular processes. The primary mechanism involves absorption by cytochrome c oxidase in mitochondria, leading to increased ATP production, reduced inflammation via lowered pro-inflammatory cytokines like TNF-α, and enhanced tissue repair through fibroblast proliferation and collagen synthesis. Clinical evidence supports its efficacy for chronic wounds, such as diabetic ulcers, with studies showing accelerated healing rates using 810 nm light at energy densities of 0.04–50 J/cm².[70][71] However, high-intensity near-infrared radiation poses significant hazards, particularly retinal damage due to its transmission through the cornea and lens to focus on the retina. In the 400–1400 nm range, including near-IR (780–1400 nm), exposure can cause thermal burns and permanent lesions in the macula, as the eye's optics concentrate energy up to 100,000 times, leading to irreversible tissue destruction without pain warning. Safety guidelines emphasize limiting exposure to prevent such photochemical and thermal effects in ophthalmic and laser applications.[72][73] Environmentally, infrared spectroscopy is essential in climatology for identifying greenhouse gas absorption bands, with carbon dioxide (CO₂) exhibiting strong peaks at 4.3 μm and 15 μm that trap outgoing thermal radiation. These bands, particularly the 15 μm vibration-rotation absorption near the Earth's blackbody emission peak, contribute significantly to the greenhouse effect; for example, in the 4.3 μm band, absorption coefficients increase from about 0.11 m⁻¹ at 100 ppm to 0.44 m⁻¹ at 400 ppm near Earth's surface for 10 km paths. In meteorology, infrared satellite imagery from sensors like those on GOES satellites measures cloud-top temperatures in the 10–12 μm thermal IR window, enabling day-night monitoring of storm intensity and fog detection by distinguishing cold cloud heights from warmer surfaces.[74][75] Biologically, certain snakes, such as pit vipers in the Crotalinae subfamily, possess specialized pit organs that detect mid-infrared radiation (peaking around 10 μm) for thermoreception. These facial pits feature a thin, vascularized membrane that senses temperature differentials as small as 0.003°C from warm-blooded prey, with signals processed via the trigeminal ganglion to integrate IR cues with visual input for accurate targeting up to 66 cm away. This evolutionary adaptation, found in boid and crotaline snakes, relies on ~7000 nerve endings converting radiant heat flux (threshold ~10.75 μW/cm²) into neural impulses.[76]History and Development
Early Discoveries
In 1800, British astronomer William Herschel conducted experiments to determine which colors in the solar spectrum contributed most to heating. By dispersing sunlight through a glass prism and placing mercury-in-glass thermometers in the resulting spectrum, he observed that temperatures were highest beyond the visible red band, where no light was apparent. This indicated the presence of invisible rays responsible for the heating effect, which he termed "invisible rays" and later recognized as part of the solar spectrum extending beyond the visible range. Herschel's findings, detailed in his paper presented to the Royal Society, marked the first identification of what would become known as infrared radiation.[77] During the 1830s, Italian physicist Macedonio Melloni significantly advanced infrared detection through his development of the thermopile, an instrument comprising alternating bars of antimony and bismuth connected in series to amplify thermal signals via the Seebeck effect. This device allowed precise measurement of infrared radiation's intensity and properties, enabling Melloni to detect "heat rays" from sources as distant as a human body 30 feet away. He coined the term "rayons calorifiques" (heat rays) and conducted experiments demonstrating that these rays could be reflected by parabolic mirrors, refracted by rock salt prisms, diffracted, and even polarized, behaviors analogous to visible light. Melloni's work, published in French scientific journals, established infrared as a form of radiant heat with optical characteristics.[78] Early understandings of infrared often framed it within the caloric theory, positing it as a fluid-like substance of heat rather than an electromagnetic phenomenon akin to light. This misconception, rooted in 18th-century views of heat as a material entity, was gradually challenged by Melloni's wave-like demonstrations and Herschel's spectral placement, though full acceptance as electromagnetic waves awaited James Clerk Maxwell's 1865 theory.[79]Modern Advancements
In 1900, Max Planck introduced his quantum hypothesis to resolve the discrepancies in classical theories of blackbody radiation, proposing that energy is emitted in discrete quanta, which accurately described the infrared-dominated spectrum of thermal radiation from blackbodies at various temperatures./01:_The_Dawn_of_the_Quantum_Theory/1.02:_Quantum_Hypothesis_Used_for_Blackbody_Radiation_Law) This foundational concept, building on earlier empirical observations of thermal detectors, laid the groundwork for quantum mechanics and revolutionized the theoretical understanding of infrared emission.[80] During the 1910s to 1940s, the development of lead sulfide (PbS) photoconductive detectors marked a significant technological leap, offering practical sensitivity to near-infrared wavelengths up to about 3 μm.[81] These detectors, refined extensively in the 1940s, were pivotal for World War II applications, enabling early active infrared night vision systems like the German Vampir device and Allied "sniperscopes" for low-light combat operations.[82] The 1960s brought breakthroughs in infrared laser technology, with the first continuous-wave helium-neon (He-Ne) laser operating at 3.39 μm in the mid-infrared range, demonstrated by researchers at Bell Laboratories.[83] This achievement, leveraging interband transitions in neon atoms pumped by helium, enabled stable coherent infrared sources for spectroscopy and alignment applications.[84] By the 1970s, semiconductor diode lasers emerged, with heterostructure designs using materials like GaAs achieving room-temperature continuous-wave operation in the near-infrared around 0.8–0.9 μm, dramatically improving efficiency and portability for telecommunications and sensing.[85] In 1994, the invention of quantum cascade lasers (QCLs) at Bell Laboratories introduced a novel unipolar electron injection mechanism across engineered quantum wells, enabling tunable emission in the mid-infrared (4–12 μm) without relying on bandgap energies.[86] This high-impact innovation, now commercially available, supports compact, high-power sources for trace gas detection and free-space communications.[87] Post-2000 developments in hyperspectral infrared sensors have enhanced remote sensing capabilities, with satellite-based systems like NASA's Hyperion (launched 2000) providing high-resolution spectral imaging across hundreds of infrared bands for Earth observation and material identification.[88] In the 2020s, advances in room-temperature long-wave infrared detectors, such as graphene-based devices for long-wave infrared (8–15 μm) detection, have achieved high detectivity and ultrafast response times, promising applications in thermal imaging and astrophysics without cryogenic cooling.[89]References
- https://science.[nasa](/page/NASA).gov/ems/07_infraredwaves/
- https://science.[nasa](/page/NASA).gov/ems/07_infraredwaves