Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
Food allergy
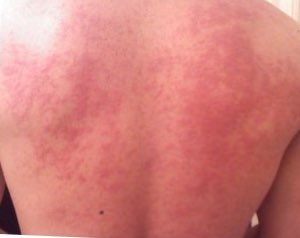
A food allergy is an abnormal immune response to food. The symptoms of the allergic reaction may range from mild to severe. They may include itchiness, swelling of the tongue, vomiting, diarrhea, hives, trouble breathing, or low blood pressure. This typically occurs within minutes to several hours of exposure. When the symptoms are severe, it is known as anaphylaxis. A food intolerance and food poisoning are separate conditions, not due to an immune response.
Common foods involved include cow's milk, peanuts, eggs, shellfish, fish, tree nuts, soy, wheat, and sesame. The common allergies vary depending on the country. Risk factors include a family history of allergies, vitamin D deficiency, obesity, and high levels of cleanliness. Allergies occur when immunoglobulin E (IgE), part of the body's immune system, binds to food molecules. A protein in the food is usually the problem. This triggers the release of inflammatory chemicals such as histamine. Diagnosis is usually based on a medical history, elimination diet, skin prick test, blood tests for food-specific IgE antibodies, or oral food challenge.
Management involves avoiding the food in question and having a plan if exposure occurs. This plan may include giving adrenaline (epinephrine) and wearing medical alert jewelry. Early childhood exposure to potential allergens may be protective against later development of a food allergy. The benefits of allergen immunotherapy for treating food allergies are not proven, thus not recommended as of 2015[update]. Some types of food allergies among children resolve with age, including those to milk, eggs, and soy; while others such as to nuts and shellfish typically do not.
In the developed world, about 4% to 8% of people have at least one food allergy. They are more common in children than adults and appear to be increasing in frequency. Male children appear to be more commonly affected than females. Some allergies more commonly develop early in life, while others typically develop in later life. In developed countries, more people believe they have food allergies when they actually do not have them.
Food allergy symptoms occur within minutes to hours after exposure and may include:
In some cases, however, onset of symptoms may be delayed for hours.
Symptoms can vary. The amount of food needed to trigger a reaction also varies.
Serious danger regarding allergies can begin when the respiratory tract or blood circulation is affected. The former can be indicated through wheezing and cyanosis. Poor blood circulation leads to a weak pulse, pale skin and fainting.
Hub AI
Food allergy AI simulator
(@Food allergy_simulator)
Food allergy
A food allergy is an abnormal immune response to food. The symptoms of the allergic reaction may range from mild to severe. They may include itchiness, swelling of the tongue, vomiting, diarrhea, hives, trouble breathing, or low blood pressure. This typically occurs within minutes to several hours of exposure. When the symptoms are severe, it is known as anaphylaxis. A food intolerance and food poisoning are separate conditions, not due to an immune response.
Common foods involved include cow's milk, peanuts, eggs, shellfish, fish, tree nuts, soy, wheat, and sesame. The common allergies vary depending on the country. Risk factors include a family history of allergies, vitamin D deficiency, obesity, and high levels of cleanliness. Allergies occur when immunoglobulin E (IgE), part of the body's immune system, binds to food molecules. A protein in the food is usually the problem. This triggers the release of inflammatory chemicals such as histamine. Diagnosis is usually based on a medical history, elimination diet, skin prick test, blood tests for food-specific IgE antibodies, or oral food challenge.
Management involves avoiding the food in question and having a plan if exposure occurs. This plan may include giving adrenaline (epinephrine) and wearing medical alert jewelry. Early childhood exposure to potential allergens may be protective against later development of a food allergy. The benefits of allergen immunotherapy for treating food allergies are not proven, thus not recommended as of 2015[update]. Some types of food allergies among children resolve with age, including those to milk, eggs, and soy; while others such as to nuts and shellfish typically do not.
In the developed world, about 4% to 8% of people have at least one food allergy. They are more common in children than adults and appear to be increasing in frequency. Male children appear to be more commonly affected than females. Some allergies more commonly develop early in life, while others typically develop in later life. In developed countries, more people believe they have food allergies when they actually do not have them.
Food allergy symptoms occur within minutes to hours after exposure and may include:
In some cases, however, onset of symptoms may be delayed for hours.
Symptoms can vary. The amount of food needed to trigger a reaction also varies.
Serious danger regarding allergies can begin when the respiratory tract or blood circulation is affected. The former can be indicated through wheezing and cyanosis. Poor blood circulation leads to a weak pulse, pale skin and fainting.