Recent from talks
Nothing was collected or created yet.
Pulmonology
View on Wikipedia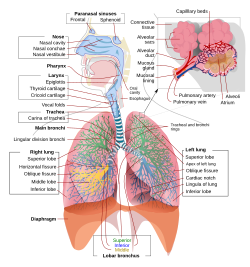 Schematic view of the human respiratory system with their parts and functions. | |
| System | Respiratory |
|---|---|
| Significant diseases | Asthma, Lung cancer, Tuberculosis, Occupational lung disease |
| Significant tests | Bronchoscopy, Sputum studies, Arterial blood gases |
| Specialist | Respiratory Physician, Pulmonologist |
Pulmonology (/ˌpʌlməˈnɒlədʒi/, /ˌpʊlməˈnɒlədʒi/, from Latin pulmō, -ōnis "lung" and the Greek suffix -λογία -logía "study of"), pneumology (/nʊˈmɒlədʒi, njʊ-/, built on Greek πνεύμων pneúmōn "lung") or pneumonology[1] (/nʊmənˈɒlədʒi, njʊ-/) is a medical specialty that deals with diseases involving the respiratory tract.[2] It is also known as respirology, respiratory medicine, or chest medicine in some countries and areas.
Pulmonology is considered a branch of internal medicine, and is related to intensive care medicine. Pulmonology often involves managing patients who need life support and mechanical ventilation. Pulmonologists are specially trained in diseases and conditions of the chest, particularly pneumonia, asthma, tuberculosis, emphysema, and complicated chest infections.[3]
Pulmonology/respirology departments work especially closely with certain other specialties: cardiothoracic surgery departments and cardiology departments.
Journals of pulmonology
[edit]History of pulmonology
[edit]One of the first major discoveries relevant to the field of pulmonology was the discovery of pulmonary circulation. Originally, it was thought that blood reaching the right side of the heart passed through small 'pores' in the septum into the left side to be oxygenated, as theorized by Galen; however, the discovery of pulmonary circulation disproves this theory, which had previously been accepted since the 2nd century. Thirteenth-century anatomist and physiologist Ibn Al-Nafis accurately theorized that there was no 'direct' passage between the two sides (ventricles) of the heart. He believed that the blood must have passed through the pulmonary artery, through the lungs, and back into the heart to be pumped around the body. This is believed by many to be the first scientific description of pulmonary circulation.[4]
Although pulmonary medicine only began to evolve as a medical specialty in the 1950s, William Welch and William Osler founded the 'parent' organization of the American Thoracic Society,[5] the National Association for the Study and Prevention of Tuberculosis.[6] The care, treatment, and study of tuberculosis of the lung is recognised as a discipline in its own right, phthisiology.[7] When the specialty did begin to evolve, several discoveries were being made linking the respiratory system and the measurement of arterial blood gases, attracting more and more physicians and researchers to the developing field.[8]
Pulmonology and its relevance in other medical fields
[edit]Surgery of the respiratory tract is generally performed by specialists in cardiothoracic surgery[9] (or thoracic surgery),[10] though minor procedures may be performed by pulmonologists. Pulmonology is closely related to critical care medicine[11] when dealing with patients who require mechanical ventilation. As a result, many pulmonologists are certified to practice critical care medicine in addition to pulmonary medicine. There are fellowship programs that allow physicians to become board certified in pulmonary and critical care medicine simultaneously. Interventional pulmonology is a relatively new field within pulmonary medicine[12] that deals with the use of procedures such as bronchoscopy[13] and pleuroscopy to treat several pulmonary diseases.[14] Interventional pulmonology is increasingly recognized as a specific medical specialty.[15]
Diagnosis
[edit]The pulmonologist begins the diagnostic process with a general review focusing on:
- hereditary diseases affecting the lungs (cystic fibrosis,[16] alpha 1-antitrypsin deficiency)
- exposure to toxicants (tobacco smoke,[17] asbestos, exhaust fumes, coal mining fumes, e-cigarette aerosol[18])
- exposure to infectious agents (certain types of birds, malt processing)
- an autoimmune diathesis that might predispose to certain conditions (pulmonary fibrosis, pulmonary hypertension)
Physical diagnostics[19] are as important as in other fields of medicine.
- Inspection of the hands for signs of cyanosis or clubbing, chest wall, and respiratory rate.
- Palpation of the cervical lymph nodes, trachea and chest wall movement.
- Percussion of the lung fields for dullness or hyper-resonance.
- Auscultation (with a stethoscope) of the lung fields for diminished or unusual breath sounds.
- Rales or rhonchi heard over lung fields with a stethoscope.
As many heart diseases can give pulmonary signs,[20] a thorough cardiac investigation is usually included.
Procedures
[edit]Clinical procedures
[edit]Pulmonary clinical procedures include the following pulmonary tests and procedures:[21][22]
- Medical laboratory investigation of blood (blood tests). Sometimes arterial blood gas tests are also required.
- Spirometry the determination of maximum airflow at a given lung volume as measured by breathing into a dedicated machine; this is the key test to diagnose airflow obstruction.
- Pulmonary function testing including spirometry, as above, plus response to bronchodilators, lung volumes, and diffusion capacity, the latter being a measure of lung oxygen absorptive area
- Bronchoscopy[23] with bronchoalveolar lavage (BAL), endobronchial and transbronchial biopsy and epithelial brushing
- Chest X-rays[24]
- CT scan[25]
- Scintigraphy and other methods of nuclear medicine
- Positron emission tomography[26] (especially in lung cancer)
- Polysomnography (sleep studies[27]) commonly used for the diagnosis of sleep apnea
Surgical procedures
[edit]Major surgical procedures on the heart and lungs are performed by a thoracic surgeon.[28] Pulmonologists often perform specialized procedures to get samples from the inside of the chest or inside of the lung. They use radiographic techniques to view vasculature of the lungs and heart to assist with diagnosis.
Treatment and therapeutics
[edit]Medication is the most important treatment of most diseases of pulmonology, either by inhalation (bronchodilators and steroids) or in oral form (antibiotics, leukotriene antagonists). A common example being the usage of inhalers in the treatment of inflammatory lung conditions such as asthma or chronic obstructive pulmonary disease. Oxygen therapy[29] is often necessary in severe respiratory disease (emphysema and pulmonary fibrosis). When this is insufficient, the patient might require mechanical ventilation.
Pulmonary rehabilitation[30] has been defined as a multidimensional continuum of services directed to persons with pulmonary disease and their families, usually by an interdisciplinary team of specialists, with the goal of achieving and maintaining the individual's maximum level of independence and functioning in the community. Pulmonary rehabilitation[31] is intended to educate the patient, the family, and improve the overall quality of life and prognosis for the patient. Interventions can include exercise, education, emotional support, oxygen, noninvasive mechanical ventilation, optimization of airway secretion clearance, promoting compliance with medical care to reduce numbers of exacerbations and hospitalizations, and returning to work and/or a more active and emotionally satisfying life. These goals are appropriate for any patients with diminished respiratory reserve whether due to obstructive or intrinsic pulmonary diseases (oxygenation impairment) or neuromuscular weakness (ventilatory impairment). A pulmonary rehabilitation team[32] may include a rehabilitation physician, a pulmonary medicine specialist, physician assistant and allied health professionals including a rehabilitation nurse, a respiratory therapist, a speech-language pathologist, a physical therapist, an occupational therapist, a psychologist, and a social worker among others. Additionally, breathing games are used to motivate children to perform pulmonary rehabilitation.
Education and training
[edit]Pulmonologist
[edit] Physician performing a bronchoscopy. | |
| Occupation | |
|---|---|
| Names |
|
Occupation type | Specialty |
Activity sectors | Medicine |
| Description | |
Education required |
|
Fields of employment | Hospitals, clinics |
In the United States, pulmonologists are physicians who, after receiving a medical degree (MD or DO), complete residency training in internal medicine, followed by at least two additional years of subspeciality fellowship training in pulmonology.[33] After satisfactorily completing a fellowship in pulmonary medicine, the physician is permitted to take the board certification examination in pulmonary medicine. After passing this exam, the physician is then board certified as a pulmonologist. Most pulmonologists complete three years of combined subspecialty fellowship training in pulmonary medicine and critical care medicine.
Pediatric pulmonologist
[edit]In the United States, pediatric pulmonologists[34] are physicians who, after receiving a medical degree (MD, DO, MBBS, MBBCh, etc.), complete residency training in pediatrics, followed by at least three additional years of subspeciality fellowship training in pulmonology. Pediatric pulmonologists treat diseases of the airways, lungs, respiratory mechanics and aerodigestive system.
Scientific research
[edit]Pulmonologists are involved in both clinical and basic research of the respiratory system, ranging from the anatomy of the respiratory epithelium to the most effective treatment of pulmonary hypertension. Scientific research also takes place to look for causes and possible treatment in diseases such as pulmonary tuberculosis and lung cancer.
References
[edit]- ^ Ramoutsaki, Ioanna; Ramoutsakis, Ioannis; Bouros, Demosthenes (May 2002). "Pneumonology or Pneumology?". Chest. 121 (5): 1385–1387. doi:10.1378/chest.121.5.1385. PMID 12006412.
- ^ ACP: Pulmonology: Internal Medicine Subspecialty Archived 2015-08-11 at the Wayback Machine. Acponline.org. Retrieved on 2011-09-30.
- ^ Sengupta, Nandini; Sahidullah, Md; Saha, Goutam (August 2016). "Lung sound classification using cepstral-based statistical features". Computers in Biology and Medicine. 75 (1): 118–129. doi:10.1016/j.compbiomed.2016.05.013. PMID 27286184.
- ^ Sharif Kaf A-Ghazal (2002). "The discovery of the pulmonary circulation – who should get the credit: ibn Al-Nafis or William Harvey" (PDF). Journal of the International Society for the History of Islamic Medicine. 2: 46. Archived (PDF) from the original on 2010-06-12. Retrieved 2010-07-17.
- ^ "American Thoracic Society - Overview". www.thoracic.org. Archived from the original on 2019-04-26. Retrieved 2019-01-25.
- ^ Jacobs, Henry Barton (1904-12-03). "National Association for the Study and Prevention of Tuberculosis". Journal of the American Medical Association. XLIII (23): 1712. doi:10.1001/jama.1904.02500230042014. ISSN 0002-9955. Archived from the original on 2021-12-13. Retrieved 2019-01-25.
- ^ Lauzardo, Michael; Ashkin, David (2000-05-01). "Phthisiology at the Dawn of the New Century". Chest. 117 (5): 1455–1473. doi:10.1378/chest.117.5.1455. ISSN 0012-3692. PMID 10807837.
- ^ History of the Division Archived 2017-08-11 at the Wayback Machine. Hopkinsmedicine.org. Retrieved on 2011-09-30.
- ^ "Cardiothoracic Surgery | Essentia Health | MN, WI, ND". www.essentiahealth.org. Archived from the original on 2019-01-26. Retrieved 2019-01-25.
- ^ "Cardiothoracic surgery". Health Careers. 2015-04-07. Archived from the original on 2019-01-26. Retrieved 2019-01-25.
- ^ "Pulmonary Disease | Internal Medicine Subspecialties | ACP". www.acponline.org. Archived from the original on 2015-09-09. Retrieved 2019-01-25.
- ^ "Interventional Pulmonology: Procedures, Uses, and Effects". WebMD. Archived from the original on 2019-01-26. Retrieved 2019-01-25.
- ^ Seijo, Luis M.; Sterman, Daniel H. (2001-03-08). "Interventional Pulmonology". New England Journal of Medicine. 344 (10): 740–749. doi:10.1056/NEJM200103083441007. ISSN 0028-4793. PMID 11236779.
- ^ "Interventional Pulmonology Diagnostic and Therapeutic Procedures". www.pamf.org. Archived from the original on 2019-01-17. Retrieved 2019-01-25.
- ^ Luis M. Seijo & Daniel H. Sterman (2001). "Interventional Pulmonology". N. Engl. J. Med. 344 (10): 740–49. doi:10.1056/NEJM200103083441007. PMID 11236779.
- ^ "Cystic Fibrosis - Pediatrics". MSD Manual Professional Edition. Archived from the original on 2019-01-26. Retrieved 2019-01-25.
- ^ Breland, Alison; Soule, Eric; Lopez, Alexa; Ramôa, Carolina; El-Hellani, Ahmad; Eissenberg, Thomas (2017). "Electronic cigarettes: what are they and what do they do?". Annals of the New York Academy of Sciences. 1394 (1): 5–30. Bibcode:2017NYASA1394....5B. doi:10.1111/nyas.12977. ISSN 0077-8923. PMC 4947026. PMID 26774031.
- ^ Perikleous, Evanthia P.; Steiropoulos, Paschalis; Paraskakis, Emmanouil; Constantinidis, Theodoros C.; Nena, Evangelia (2018). "E-Cigarette Use Among Adolescents: An Overview of the Literature and Future Perspectives". Frontiers in Public Health. 6: 86. doi:10.3389/fpubh.2018.00086. ISSN 2296-2565. PMC 5879739. PMID 29632856.
- ^ "Evaluation of the Pulmonary Patient - Pulmonary Disorders". MSD Manual Professional Edition. Archived from the original on 2019-01-26. Retrieved 2019-01-25.
- ^ "Pulmonary Tests and Procedures | Johns Hopkins Medicine Health Library". www.hopkinsmedicine.org. Archived from the original on 2019-01-26. Retrieved 2019-01-25.
- ^ "Pulmonary Tests and Procedures | Johns Hopkins Medicine Health Library". www.hopkinsmedicine.org. Archived from the original on 2019-01-26. Retrieved 2019-01-25.
- ^ "Pulmonary Diagnostic Tests & Procedures | Essentia Health". www.essentiahealth.org. Archived from the original on 2019-01-26. Retrieved 2019-01-25.
- ^ "Bronchoscopy | Johns Hopkins Medicine Health Library". www.hopkinsmedicine.org. Archived from the original on 2019-01-26. Retrieved 2019-01-25.
- ^ "How Do Chest X-Rays Work?". www.hopkinsmedicine.org. Archived from the original on 2019-01-26. Retrieved 2019-01-25.
- ^ "Computed Tomography (CT or CAT) Scan of the Chest | Johns Hopkins Medicine Health Library". www.hopkinsmedicine.org. Archived from the original on 2019-01-26. Retrieved 2019-01-25.
- ^ "How Does a PET Scan Work?". www.hopkinsmedicine.org. Archived from the original on 2021-12-13. Retrieved 2019-01-25.
- ^ "How to Prepare for a Sleep Study". www.hopkinsmedicine.org. Archived from the original on 2019-01-26. Retrieved 2019-01-25.
- ^ "Cardiothoracic surgery". Health Careers. 2015-04-07. Archived from the original on 2019-01-26. Retrieved 2019-01-25.
- ^ "Oxygen Therapy - Lung and Airway Disorders". MSD Manual Consumer Version. Archived from the original on 2021-12-13. Retrieved 2019-01-25.
- ^ "Pulmonary Rehabilitation | Essentia Health | MN, ND, WI". www.essentiahealth.org. Archived from the original on 2019-01-26. Retrieved 2019-01-25.
- ^ "Overview of Pulmonary Rehabilitation - Lung and Airway Disorders". MSD Manual Consumer Version. Archived from the original on 2019-01-26. Retrieved 2019-01-25.
- ^ "Overview of Pulmonary Rehabilitation - Lung and Airway Disorders". MSD Manual Consumer Version. Archived from the original on 2019-01-26. Retrieved 2019-01-25.
- ^ "Pulmonary Disease | Internal Medicine Subspecialties | ACP". www.acponline.org. Archived from the original on 2015-09-09. Retrieved 2019-01-25.
- ^ "What is a Pediatric Pulmonologist?". HealthyChildren.org. Archived from the original on 2019-01-26. Retrieved 2019-01-25.

