Recent from talks
Nothing was collected or created yet.
| Burn | |
|---|---|
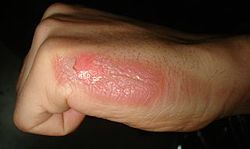 | |
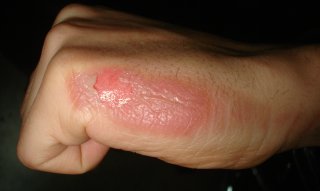
| Second-degree burn of the hand | |
| Specialty | Dermatology, critical care medicine, plastic surgery[1] |
| Symptoms | First degree: Red without blisters[2] Second degree: Blisters and pain[2] Third degree: Area stiff and not painful[2] Fourth degree: Bone and tendon loss[3] |
| Complications | Infection[4] |
| Duration | Days to weeks[2] |
| Types | First degree, second degree, third degree,[2] fourth degree[3] |
| Causes | Heat, cold, electricity, chemicals, friction, radiation[5] |
| Risk factors | Open cooking fires, unsafe cooking stoves, smoking, alcoholism, dangerous work environment[6] |
| Treatment | Depends on the severity[2] |
| Medication | Pain medication, intravenous fluids, tetanus toxoid[2] |
| Frequency | 67 million (2015)[7] |
| Deaths | 176,000 (2015)[8] |
A burn is an injury to skin, or other tissues, caused by heat, electricity, chemicals, friction, or ionizing radiation (such as sunburn, caused by ultraviolet radiation).[5][9] Most burns are due to heat from hot fluids (called scalding), solids, or fire.[10] Burns occur mainly in the home or the workplace. In the home, risks are associated with domestic kitchens, including stoves, flames, and hot liquids.[6] In the workplace, risks are associated with fire and chemical and electric burns.[6] Alcoholism and smoking are other risk factors.[6] Burns can also occur as a result of self-harm or violence between people (assault).[6]
Burns that affect only the superficial skin layers are known as superficial or first-degree burns.[2][11] They appear red without blisters, and pain typically lasts around three days.[2][11] When the injury extends into some of the underlying skin layer, it is a partial-thickness or second-degree burn.[2] Blisters are frequently present and they are often very painful.[2] Healing can require up to eight weeks and scarring may occur.[2] In a full-thickness or third-degree burn, the injury extends to all layers of the skin.[2] Often there is no pain and the burnt area is stiff.[2] Healing typically does not occur on its own.[2] A fourth-degree burn additionally involves injury to deeper tissues, such as muscle, tendons, or bone.[2] The burn is often black and frequently leads to loss of the burned part.[2][12]
Burns are generally preventable.[6] Treatment depends on the severity of the burn.[2] Superficial burns may be managed with little more than simple pain medication, while major burns may require prolonged treatment in specialized burn centers.[2] Cooling with tap water may help pain and decrease damage; however, prolonged cooling may result in low body temperature.[2][11] Partial-thickness burns may require cleaning with soap and water, followed by dressings.[2] It is not clear how to manage blisters, but it is probably reasonable to leave them intact if small and drain them if large.[2] Full-thickness burns usually require surgical treatments, such as skin grafting.[2] Extensive burns often require large amounts of intravenous fluid, due to capillary fluid leakage and tissue swelling.[11] The most common complications of burns involve infection.[4] Tetanus toxoid should be given if not up to date.[2]
In 2015, fire and heat resulted in 67 million injuries.[7] This resulted in about 2.9 million hospitalizations and 176,000 deaths.[8][13] Among women in much of the world, burns are most commonly related to the use of open cooking fires or unsafe cook stoves.[6] Among men, they are more likely a result of unsafe workplace conditions.[6] Most deaths due to burns occur in the developing world, particularly in Southeast Asia.[6] While large burns can be fatal, treatments developed since 1960 have improved outcomes, especially in children and young adults.[14] In the United States, approximately 96% of those admitted to a burn center survive their injuries.[15] The long-term outcome is related to the size of burn and the age of the person affected.[2]
Signs and symptoms
[edit]The characteristics of a burn depend upon its depth. Superficial burns cause pain lasting two or three days, followed by peeling of the skin over the next few days.[11][16] Individuals with more severe burns may indicate discomfort or complain of feeling pressure rather than pain. Full-thickness burns may be entirely insensitive to light touch or puncture.[16] While superficial burns are typically red in color, severe burns may be pink, white or black.[16] Burns around the mouth or singed hair inside the nose may indicate that burns to the airways have occurred, but these findings are not definitive.[17] More worrisome signs include: shortness of breath, hoarseness, and stridor or wheezing.[17] Itchiness is common during the healing process, occurring in up to 90% of adults and nearly all children.[18] Numbness or tingling may persist for a prolonged period of time after an electrical injury.[19] Burns may also produce emotional and psychological distress.[20]
| Type[2] | Layers involved | Appearance | Texture | Sensation | Healing time | Prognosis and complications | Example |
|---|---|---|---|---|---|---|---|
| Superficial (first-degree) | Epidermis[11] | Red without blisters[2] | Dry | Painful[2] | 5–10 days[2][21] | Heals well.[2] | 
|
| Superficial partial thickness (second-degree) | Extends into superficial (papillary) dermis[2] | Redness with clear blister.[2] Blanches with pressure.[2] | Moist[2] | Very painful[2] | 2–3 weeks[2][16] | Local infection (cellulitis) but no scarring typically[16] | |
| Deep partial thickness (second-degree) | Extends into deep (reticular) dermis[2] | Yellow or white. Less blanching. May be blistering.[2] | Fairly dry[16] | Pressure and discomfort[16] | 3–8 weeks[2] | Scarring, contractures (may require excision and skin grafting)[16] | 
|
| Full thickness (third-degree) | Extends through entire dermis[2] | Stiff and white/brown.[2] No blanching.[16] | Leathery[2] | Painless[2] | Prolonged (months) and unfinished/incomplete[2] | Scarring, contractures, amputation (early excision recommended)[16] | 
|
| Fourth-degree | Extends through entire skin, and into underlying fat, muscle and bone[2] | Black; charred with eschar | Dry | Painless | Does not heal; Requires excision[2] | Amputation, significant functional impairment and, in some cases, death.[2] | 
|
Cause
[edit]Burns are caused by a variety of external sources classified as thermal (heat-related), chemical, electrical, and radiation.[22] In the United States, the most common causes of burns are: fire or flame (44%), scalds (33%), hot objects (9%), electricity (4%), and chemicals (3%).[23] Most (69%) burn injuries occur at home or at work (9%),[15] and most are accidental, with 2% due to assault by another, and 1–2% resulting from a suicide attempt.[20] These sources can cause inhalation injury to the airway and/or lungs, occurring in about 6%.[4]
Burn injuries occur more commonly among the poor.[20] Smoking and alcoholism are other risk factors.[10] Fire-related burns are generally more common in colder climates.[20] Specific risk factors in the developing world include cooking with open fires or on the floor[5] as well as developmental disabilities in children and chronic diseases in adults.[24]
Thermal
[edit]
In the United States, fire and hot liquids are the most common causes of burns.[4] Of house fires that result in death, smoking causes 25% and heating devices cause 22%.[5] Almost half of injuries are due to efforts to fight a fire.[5] Scalding is caused by hot liquids or gases and most commonly occurs from exposure to hot drinks, high temperature tap water in baths or showers, hot cooking oil, or steam.[25] Scald injuries are most common in children under the age of five[2] and, in the United States and Australia, this population makes up about two-thirds of all burns.[4] Contact with hot objects is the cause of about 20–30% of burns in children.[4] Generally, scalds are first- or second-degree burns, but third-degree burns may also result, especially with prolonged contact.[26] Fireworks are a common cause of burns during holiday seasons in many countries.[27] This is a particular risk for adolescent males.[28] In the United States, for non-fatal burn injuries to children, white males under the age of 6 comprise most cases.[29] Thermal burns from grabbing/touching and spilling/splashing were the most common type of burn and mechanism, while the bodily areas most impacted were hands and fingers followed by head/neck.[29]
Chemical
[edit]Chemical burns can be caused by over 25,000 substances,[2] most of which are either a strong base (55%) or a strong acid (26%).[30] Most chemical burn deaths are secondary to ingestion.[2] Common agents include: sulfuric acid as found in toilet cleaners, sodium hypochlorite as found in bleach, and halogenated hydrocarbons as found in paint remover, among others.[2] Hydrofluoric acid can cause particularly deep burns that may not become symptomatic until some time after exposure.[31] Formic acid may cause the breakdown of significant numbers of red blood cells.[17]
Electrical
[edit]Electrical burns or injuries are classified as high voltage (greater than or equal to 1000 volts), low voltage (less than 1000 volts), or as flash burns secondary to an electric arc.[2] The most common causes of electrical burns in children are electrical cords (60%) followed by electrical outlets (14%).[4][32] Lightning may also result in electrical burns.[33] Risk factors for being struck include involvement in outdoor activities such as mountain climbing, golf and field sports, and working outside.[19] Mortality from a lightning strike is about 10%.[19]
While electrical injuries primarily result in burns, they may also cause fractures or dislocations secondary to blunt force trauma or muscle contractions.[19] In high voltage injuries, most damage may occur internally and thus the extent of the injury cannot be judged by examination of the skin alone.[19] Contact with either low voltage or high voltage may produce cardiac arrhythmias or cardiac arrest.[19]
Radiation
[edit]Radiation burns may be caused by protracted exposure to ultraviolet light (such as from the sun, tanning booths or arc welding) or from ionizing radiation (such as from radiation therapy, X-rays or radioactive fallout).[34] Sun exposure is the most common cause of radiation burns and the most common cause of superficial burns overall.[35] There is significant variation in how easily people sunburn based on their skin type.[36] Skin effects from ionizing radiation depend on the amount of exposure to the area, with hair loss seen after 3 Gy, redness seen after 10 Gy, wet skin peeling after 20 Gy, and necrosis after 30 Gy.[37] Redness, if it occurs, may not appear until some time after exposure.[37] Radiation burns are treated the same as other burns.[37] Microwave burns occur via thermal heating caused by the microwaves.[38] While exposures as short as two seconds may cause injury, overall this is an uncommon occurrence.[38]
Non-accidental
[edit]In those hospitalized from scalds or fire burns, 3–10% are from assault.[39] Reasons include: child abuse, personal disputes, spousal abuse, elder abuse, and business disputes.[39] An immersion injury or immersion scald may indicate child abuse.[26] It is created when an extremity, or sometimes the buttocks are held under the surface of hot water.[26] It typically produces a sharp upper border and is often symmetrical,[26] known as "sock burns", "glove burns", or "zebra stripes" - where folds have prevented certain areas from burning.[40] Deliberate cigarette burns most often found on the face, or the back of the hands and feet.[40] Other high-risk signs of potential abuse include: circumferential burns, the absence of splash marks, a burn of uniform depth, and association with other signs of neglect or abuse.[41]
Bride burning, a form of domestic violence, occurs in some cultures, such as India where women have been burned in revenge for what the husband or his family consider an inadequate dowry.[42][43] In Pakistan, acid burns represent 13% of intentional burns, and are frequently related to domestic violence.[41] Self-immolation (setting oneself on fire) is also used as a form of protest in various parts of the world.[20]
Pathophysiology
[edit]
At temperatures greater than 44 °C (111 °F), proteins begin losing their three-dimensional shape and start breaking down.[44] This results in cell and tissue damage.[2] Many of the direct health effects of a burn are caused by failure of the skin to perform its normal functions, which include: protection from bacteria, skin sensation, body temperature regulation, and prevention of evaporation of the body's water. Disruption of these functions can lead to infection, loss of skin sensation, hypothermia, and hypovolemic shock via dehydration (i.e. water in the body evaporated away).[2] Disruption of cell membranes causes cells to lose potassium to the spaces outside the cell and to take up water and sodium.[2]
In large burns (over 30% of the total body surface area), there is a significant inflammatory response.[45] This results in increased leakage of fluid from the capillaries,[17] and subsequent tissue edema.[2] This causes overall blood volume loss, with the remaining blood suffering significant plasma loss, making the blood more concentrated.[2] Poor blood flow to organs like the kidneys and gastrointestinal tract may result in kidney failure and stomach ulcers.[46]
Increased levels of catecholamines and cortisol can cause a hypermetabolic state that can last for years.[45] This is associated with increased cardiac output, metabolism, a fast heart rate, and poor immune function.[45]
Diagnosis
[edit]Burns can be classified by depth, mechanism of injury, extent, and associated injuries. The most commonly used classification is based on the depth of injury. The depth of a burn is usually determined via examination, although a biopsy may also be used.[2] It may be difficult to accurately determine the depth of a burn on a single examination and repeated examinations over a few days may be necessary.[17] In those who have a headache or are dizzy and have a fire-related burn, carbon monoxide poisoning should be considered.[47] Cyanide poisoning should also be considered.[17]
Size
[edit]
The size of a burn is measured as a percentage of total body surface area (TBSA) affected by partial thickness or full thickness burns.[2] First-degree burns that are only red in color and are not blistering are not included in this estimation.[2] Most burns (70%) involve less than 10% of the TBSA. Unit of measuring burns is VSD as 10% TBSA is equal to 1VSD.[4]
There are a number of methods to determine the TBSA, including the Wallace rule of nines, Lund and Browder chart, and estimations based on a person's palm size.[11] The rule of nines is easy to remember but only accurate in people over 16 years of age.[11] More accurate estimates can be made using Lund and Browder charts, which take into account the different proportions of body parts in adults and children.[11] The size of a person's handprint (including the palm and fingers) is approximately 1% of their TBSA.[11]
Severity
[edit]| Minor | Moderate | Major |
|---|---|---|
| Adult <10% TBSA | Adult 10–20% TBSA | Adult >20% TBSA |
| Young or old < 5% TBSA | Young or old 5–10% TBSA | Young or old >10% TBSA |
| <2% full thickness burn | 2–5% full thickness burn | >5% full thickness burn |
| High voltage injury | High voltage burn | |
| Possible inhalation injury | Known inhalation injury | |
| Circumferential burn | Significant burn to face, joints, hands, or feet | |
| Other health problems | Associated injuries |
To determine the need for referral to a specialized burn unit, the American Burn Association devised a classification system. Under this system, burns can be classified as major, moderate, and minor. This is assessed based on a number of factors, including total body surface area affected, the involvement of specific anatomical zones, the age of the person, and associated injuries.[47] Minor burns can typically be managed at home, moderate burns are often managed in a hospital, and major burns are managed by a burn center.[47] Severe burn injury represents one of the most devastating forms of trauma.[48] Despite improvements in burn care, patients can be left to suffer for as many as three years post-injury.[49]
Prevention
[edit]Historically, about half of all burns were deemed preventable.[5] Burn prevention programs have significantly decreased rates of serious burns.[44] Preventive measures include: limiting hot water temperatures, smoke alarms, sprinkler systems, proper construction of buildings, and fire-resistant clothing.[5] Experts recommend setting water heaters below 48.8 °C (119.8 °F).[4] Other measures to prevent scalds include using a thermometer to measure bath water temperatures, and splash guards on stoves.[44] While the effect of the regulation of fireworks is unclear, there is tentative evidence of benefit[50] with recommendations including the limitation of the sale of fireworks to children.[4]
Management
[edit]Resuscitation begins with the assessment and stabilization of the person's airway, breathing and circulation.[11] If inhalation injury is suspected, early intubation may be required.[17] This is followed by care of the burn wound itself. People with extensive burns may be wrapped in clean sheets until they arrive at a hospital.[17] As burn wounds are prone to infection, a tetanus booster shot should be given if an individual has not been immunized within the last five years.[51] In the United States, 95% of burns that present to the emergency department are treated and discharged; 5% require hospital admission.[20] With major burns, early feeding is important.[45] Protein intake should also be increased, and trace elements and vitamins are often required.[52] Hyperbaric oxygenation may be useful in addition to traditional treatments.[53]
Intravenous fluids
[edit]In those with poor tissue perfusion, boluses of isotonic crystalloid solution should be given.[11] In children with more than 10–20% TBSA (Total Body Surface Area) burns, and adults with more than 15% TBSA burns, formal fluid resuscitation and monitoring should follow.[11][54][55] This should be begun pre-hospital if possible in those with burns greater than 25% TBSA.[54] The Parkland formula can help determine the volume of intravenous fluids required over the first 24 hours. The formula is based on the affected individual's TBSA and weight. Half of the fluid is administered over the first 8 hours, and the remainder over the following 16 hours. The time is calculated from when the burn occurred, and not from the time that fluid resuscitation began. Children require additional maintenance fluid that includes glucose.[17] Additionally, those with inhalation injuries require more fluid.[56] While inadequate fluid resuscitation may cause problems, over-resuscitation can also be detrimental.[57] The formulas are only a guide, with infusions ideally tailored to a urinary output of >30 mL/h in adults or >1mL/kg in children and mean arterial pressure greater than 60 mmHg.[17]
While lactated Ringer's solution is often used, there is no evidence that it is superior to normal saline.[11] Crystalloid fluids appear just as good as colloid fluids, and as colloids are more expensive they are not recommended.[58][59] Blood transfusions are rarely required.[2] They are typically only recommended when the hemoglobin level falls below 60-80 g/L (6-8 g/dL)[60] due to the associated risk of complications.[17] Intravenous catheters may be placed through burned skin if needed or intraosseous infusions may be used.[17]
Wound care
[edit]Early cooling (within 30 minutes of the burn) reduces burn depth and pain, but care must be taken as over-cooling can result in hypothermia.[2][11] It should be performed with cool water 10–25 °C (50.0–77.0 °F) and not ice water as the latter can cause further injury.[11][44] Chemical burns may require extensive irrigation.[2] Cleaning with soap and water, removal of dead tissue, and application of dressings are important aspects of wound care. If intact blisters are present, it is not clear what should be done with them. Some tentative evidence supports leaving them intact. Second-degree burns should be re-evaluated after two days.[44]
In the management of first and second-degree burns, little quality evidence exists to determine which dressing type to use.[61] It is reasonable to manage first-degree burns without dressings.[44] While topical antibiotics are often recommended, there is little evidence to support their use.[62][63] Silver sulfadiazine (a type of antibiotic) is not recommended as it potentially prolongs healing time.[61][64] There is insufficient evidence to support the use of dressings containing silver[65] or negative-pressure wound therapy.[66] Silver sulfadiazine does not appear to differ from silver containing foam dressings with respect to healing.[67]
Medications
[edit]Burns can be very painful and a number of different options may be used for pain management. These include simple analgesics (such as ibuprofen and acetaminophen) and opioids such as morphine. Benzodiazepines may be used in addition to analgesics to help with anxiety.[44] During the healing process, antihistamines, massage, or transcutaneous nerve stimulation may be used to aid with itching.[18] Antihistamines, however, are only effective for this purpose in 20% of people.[68] There is tentative evidence supporting the use of gabapentin[18] and its use may be reasonable in those who do not improve with antihistamines.[69][70] Intravenous lidocaine requires more study before it can be recommended for pain.[71]
Intravenous antibiotics are recommended before surgery for those with extensive burns (>60% TBSA).[72] As of 2008[update], guidelines do not recommend their general use due to concerns regarding antibiotic resistance[62] and the increased risk of fungal infections.[17] Tentative evidence, however, shows that they may improve survival rates in those with large and severe burns.[62] Erythropoietin has not been found effective to prevent or treat anemia in burn cases.[17] In burns caused by hydrofluoric acid, calcium gluconate is a specific antidote and may be used intravenously and/or topically.[31] Recombinant human growth hormone (rhGH) in those with burns that involve more than 40% of their body appears to speed healing without affecting the risk of death.[73] The use of steroids is of unclear evidence.[74]
Allogeneic cultured keratinocytes and dermal fibroblasts in murine collagen (Stratagraft) was approved for medical use in the United States in June 2021.[75]
Surgery
[edit]Wounds requiring surgical closure with skin grafts or flaps (typically anything more than a small full thickness burn) should be dealt with as early as possible.[76] Circumferential burns of the limbs or chest may need urgent surgical release of the skin, known as an escharotomy.[77] This is done to treat or prevent problems with distal circulation, or ventilation.[77] It is uncertain if it is useful for neck or digit burns.[77] Fasciotomies may be required for electrical burns.[77]
Skin grafts can involve temporary skin substitutes, derived from animal (human donor or pig) skin or synthesized. They are used to cover the wound as a dressing, preventing infection and fluid loss, but will eventually need to be removed. Alternatively, human skin can be treated to be left on permanently without rejection.[78]
There is no evidence that the use of copper sulphate to visualise phosphorus particles for removal can help with wound healing due to phosphorus burns. Meanwhile, absorption of copper sulphate into the blood circulation can be harmful.[79]
Alternative medicine
[edit]Honey has been used since ancient times to aid wound healing and may be beneficial in first- and second-degree burns.[80] There is moderate evidence that honey helps heal partial thickness burns.[81][82] The evidence for aloe vera is of poor quality.[83] While it might be beneficial in reducing pain,[21] and a review from 2007 found tentative evidence of improved healing times,[84] a subsequent review from 2012 did not find improved healing over silver sulfadiazine.[83] A 2014 review found only three randomized controlled trials for the use of plants for burns, two for aloe vera and one for oatmeal.[85] The number of randomized control trials for aloe vera had increased to nine by 2024, when a review found a significant improvement in healing time but not pain relief.[86]
There is little evidence that vitamin E helps with keloids or scarring.[87] Butter is not recommended.[88] In low income countries, burns are treated up to one-third of the time with traditional medicine, which may include applications of eggs, mud, leaves or cow dung.[24] Surgical management is limited in some cases due to insufficient financial resources and availability.[24] There are a number of other methods that may be used in addition to medications to reduce procedural pain and anxiety including virtual reality therapy, hypnosis, and behavioral approaches such as distraction techniques.[69]
Patient support
[edit]Burn patients require support and care – both physiological and psychological. Respiratory failure, sepsis, and multi-organ system failure are common in hospitalized burn patients. To prevent hypothermia and maintain normal body temperature, burn patients with over 20% of burn injuries should be kept in an environment with the temperature at or above 30 degree Celsius.[89][better source needed]
Metabolism in burn patients proceeds at a higher than normal speed due to the whole-body process and rapid fatty acid substrate cycles, which can be countered with an adequate supply of energy, nutrients, and antioxidants. Enteral feeding a day after resuscitation is required to reduce risk of infection, recovery time, non-infectious complications, hospital stay, long-term damage, and mortality. Controlling blood glucose levels can have an impact on liver function and survival.
Risk of thromboembolism is high and acute respiratory distress syndrome (ARDS) that does not resolve with maximal ventilator use is also a common complication. Scars are long-term after-effects of a burn injury. Psychological support is required to cope with the aftermath of a fire accident, while to prevent scars and long-term damage to the skin and other body structures consulting with burn specialists, preventing infections, consuming nutritious foods, early and aggressive rehabilitation, and using compressive clothing are recommended.
Prognosis
[edit]| TBSA | Mortality |
|---|---|
| 0–9% | 0.6% |
| 10–19% | 2.9% |
| 20–29% | 8.6% |
| 30–39% | 16% |
| 40–49% | 25% |
| 50–59% | 37% |
| 60–69% | 43% |
| 70–79% | 57% |
| 80–89% | 73% |
| 90–100% | 85% |
| Inhalation | 23% |
The prognosis is worse in those with larger burns, those who are older, and females.[2] The presence of a smoke inhalation injury, other significant injuries such as long bone fractures, and serious co-morbidities (e.g. heart disease, diabetes, psychiatric illness, and suicidal intent) also influence prognosis.[2] On average, of those admitted to burn centers in the United States, 4% die,[4] with the outcome for individuals dependent on the extent of the burn injury. For example, admittees with burn areas less than 10% TBSA had a mortality rate of less than 1%, while admittees with over 90% TBSA had a mortality rate of 85%.[90] In Afghanistan, people with more than 60% TBSA burns rarely survive.[4] The Baux score has historically been used to determine prognosis of major burns. However, with improved care, it is no longer very accurate.[17] The score is determined by adding the size of the burn (% TBSA) to the age of the person and taking that to be more or less equal to the risk of death.[17] Burns in 2013 resulted in 1.2 million years lived with disability and 12.3 million disability adjusted life years.[13]
Complications
[edit]A number of complications may occur, with infections being the most common.[4] In order of frequency, potential complications include: pneumonia, cellulitis, urinary tract infections and respiratory failure.[4] Risk factors for infection include: burns of more than 30% TBSA, full-thickness burns, extremes of age (young or old), or burns involving the legs or perineum.[91] Pneumonia occurs particularly commonly in those with inhalation injuries.[17]
Anemia secondary to full thickness burns of greater than 10% TBSA is common.[11] Electrical burns may lead to compartment syndrome or rhabdomyolysis due to muscle breakdown.[17] Blood clotting in the veins of the legs is estimated to occur in 6 to 25% of people.[17] The hypermetabolic state that may persist for years after a major burn can result in a decrease in bone density and a loss of muscle mass.[45] Keloids may form subsequent to a burn, particularly in those who are young and dark skinned.[87] Following a burn, children may have significant psychological trauma and experience post-traumatic stress disorder.[92] Scarring may also result in a disturbance in body image.[92] To treat hypertrophic scars (raised, tense, stiff and itchy scars) and limit their effect on physical function and everyday activities, silicone sheeting and compression garments are recommended.[93][94][95] In the developing world, significant burns may result in social isolation, extreme poverty and child abandonment.[20]
Epidemiology
[edit]
| No data < 50 50–100 100–150 150–200 200–250 250–300 | 300–350 350–400 400–450 450–500 500–600 > 600 |
In 2015 fire and heat resulted in 67 million injuries.[7] This resulted in about 2.9 million hospitalizations and 238,000 dying.[13] This is down from 300,000 deaths in 1990.[97] This makes it the fourth leading cause of injuries after motor vehicle collisions, falls, and violence.[20] About 90% of burns occur in the developing world.[20] This has been attributed partly to overcrowding and an unsafe cooking situation.[20] Overall, nearly 60% of fatal burns occur in Southeast Asia with a rate of 11.6 per 100,000.[4] The number of fatal burns has decreased from 280,000 in 1990 to 176,000 in 2015.[98][8]
In the developed world, adult males have twice the mortality as females from burns. This is most probably due to their higher risk occupations and greater risk-taking activities. In many countries in the developing world, however, females have twice the risk of males. This is often related to accidents in the kitchen or domestic violence.[20] In children, deaths from burns occur at more than ten times the rate in the developing than the developed world.[20] Overall, in children it is one of the top fifteen leading causes of death.[5] From the 1980s to 2004, many countries have seen both a decrease in the rates of fatal burns and in burns generally.[20]
Developed countries
[edit]An estimated 500,000 burn injuries receive medical treatment yearly in the United States.[44] They resulted in about 3,300 deaths in 2008.[5] Most burns (70%) and deaths from burns occur in males.[2][15] The highest incidence of fire burns occurs in those 18–35 years old, while the highest incidence of scalds occurs in children less than five years old and adults over 65.[2] Electrical burns result in about 1,000 deaths per year.[99] Lightning results in the death of about 60 people a year.[19] In Europe, intentional burns occur most commonly in middle aged men.[39]
Developing countries
[edit]In India, about 700,000 to 800,000 people per year sustain significant burns, though very few are looked after in specialist burn units.[100] The highest rates occur in women 16–35 years of age.[100] Part of this high rate is related to unsafe kitchens and loose-fitting clothing typical to India.[100] It is estimated that one-third of all burns in India are due to clothing catching fire from open flames.[101] Intentional burns are also a common cause and occur at high rates in young women, secondary to domestic violence and self-harm.[20][39]
History
[edit]
Cave paintings from more than 3,500 years ago document burns and their management.[14] The earliest Egyptian records on treating burns describes dressings prepared with milk from mothers of baby boys,[102] and the 1500 BCE Edwin Smith Papyrus describes treatments using honey and the salve of resin.[14] Many other treatments have been used over the ages, including the use of tea leaves by the Chinese documented to 600 BCE, pig fat and vinegar by Hippocrates documented to 400 BCE, and wine and myrrh by Celsus documented to the 1st century CE.[14] French barber-surgeon Ambroise Paré was the first to describe different degrees of burns in the 1500s.[103] Guillaume Dupuytren expanded these degrees into six different severities in 1832.[14][104]
The first hospital to treat burns opened in 1843 in London, England, and the development of modern burn care began in the late 1800s and early 1900s.[14][103] During World War I, Henry D. Dakin and Alexis Carrel developed standards for the cleaning and disinfecting of burns and wounds using sodium hypochlorite solutions, which significantly reduced mortality.[14] In the 1940s, the importance of early excision and skin grafting was acknowledged, and around the same time, fluid resuscitation and formulas to guide it were developed.[14] In the 1970s, researchers demonstrated the significance of the hypermetabolic state that follows large burns.[14]
The "Evans formula", described in 1952, was the first burn resuscitation formula based on body weight and surface area (BSA) damaged. The first 24 hours of treatment entails 1ml/kg/% BSA of crystalloids plus 1 ml/kg/% BSA colloids plus 2000ml glucose in water, and in the next 24 hours, crystalloids at 0.5 ml/kg/% BSA, colloids at 0.5 ml/kg/% BSA, and the same amount of glucose in water.[105][106]
References
[edit]- ^ "Burns - British Association of Plastic Reconstructive and Aesthetic Surgeons". BAPRAS.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac ad ae af ag ah ai aj ak al am an ao ap aq ar as at au av aw ax ay az ba bb bc bd be bf bg bh bi bj bk bl bm bn bo Tintinalli, Judith E. (2010). Emergency Medicine: A Comprehensive Study Guide (Emergency Medicine (Tintinalli)). New York: McGraw-Hill Companies. pp. 1374–1386. ISBN 978-0-07-148480-0.
- ^ a b Singer A (June 2007). "Management of local burn wounds in the ED". The American Journal of Emergency Medicine. 25 (6): 666–671. doi:10.1016/j.ajem.2006.12.008. PMID 17606093.
- ^ a b c d e f g h i j k l m n o Herndon D, ed. (2012). "Chapter 3: Epidemiological, Demographic, and Outcome Characteristics of Burn Injury". Total burn care (4th ed.). Edinburgh: Saunders. p. 23. ISBN 978-1-4377-2786-9.
- ^ a b c d e f g h i Herndon D, ed. (2012). "Chapter 4: Prevention of Burn Injuries". Total burn care (4th ed.). Edinburgh: Saunders. p. 46. ISBN 978-1-4377-2786-9.
- ^ a b c d e f g h i "Burns". World Health Organization. September 2016. Archived from the original on 21 July 2017. Retrieved 1 August 2017.
- ^ a b c Vos T, Allen C, Arora M, Barber RM, Bhutta ZA, Brown A, et al. (October 2016). "Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015". The Lancet. 388 (10053): 1545–1602. doi:10.1016/S0140-6736(16)31678-6. PMC 5055577. PMID 27733282.
- ^ a b c Wang H, Naghavi M, Allen C, Barber R, Bhutta Z, Carter A, et al. (October 2016). "Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980-2015: a systematic analysis for the Global Burden of Disease Study 2015". The Lancet. 388 (10053): 1459–1544. doi:10.1016/S0140-6736(16)31012-1. PMC 5388903. PMID 27733281.
- ^ Moore K (2014). Clinically Oriented Anatomy (7th ed.). Lippincott Williams & Wilkins. p. 45. ISBN 978-1-4511-1945-9.
- ^ a b "Burns Fact sheet N°365". WHO. April 2014. Archived from the original on 10 November 2015. Retrieved 3 March 2016.
- ^ a b c d e f g h i j k l m n o p q Granger J (January 2009). "An Evidence-Based Approach to Pediatric Burns". Pediatric Emergency Medicine Practice. 6 (1). Archived from the original on 17 October 2013.
- ^ Ferri FF (2012). Ferri's netter patient advisor (2nd ed.). Philadelphia, PA: Saunders. p. 235. ISBN 978-1-4557-2826-8. Archived from the original on 21 December 2016.
- ^ a b c Haagsma JA, Graetz N, Bolliger I, Naghavi M, Higashi H, Mullany EC, et al. (February 2016). "The global burden of injury: incidence, mortality, disability-adjusted life years and time trends from the Global Burden of Disease study 2013". Injury Prevention. 22 (1): 3–18. doi:10.1136/injuryprev-2015-041616. PMC 4752630. PMID 26635210.
- ^ a b c d e f g h i Herndon D, ed. (2012). "Chapter 1: A Brief History of Acute Burn Care Management". Total burn care (4th ed.). Edinburgh: Saunders. p. 1. ISBN 978-1-4377-2786-9.[permanent dead link]
- ^ a b c "Burn Incidence and Treatment in the United States: 2012 Fact Sheet". American Burn Association. 2012. Archived from the original on 21 February 2013. Retrieved 20 April 2013.
- ^ a b c d e f g h i j Herndon D, ed. (2012). "Chapter 10: Evaluation of the burn wound: management decisions". Total burn care (4th ed.). Edinburgh: Saunders. p. 127. ISBN 978-1-4377-2786-9.
- ^ a b c d e f g h i j k l m n o p q r s Brunicardi C (2010). "Chapter 8: Burns". Schwartz's principles of surgery (9th ed.). New York: McGraw-Hill, Medical Pub. Division. ISBN 978-0-07-154769-7.
- ^ a b c Goutos I, Dziewulski P, Richardson PM (March–April 2009). "Pruritus in burns: review article". Journal of Burn Care & Research. 30 (2): 221–8. doi:10.1097/BCR.0b013e318198a2fa. PMID 19165110. S2CID 3679902.
- ^ a b c d e f g Marx J (2010). "Chapter 140: Electrical and Lightning Injuries". Rosen's emergency medicine: concepts and clinical practice (7th ed.). Philadelphia: Mosby/Elsevier. ISBN 978-0-323-05472-0.
- ^ a b c d e f g h i j k l m n Peck MD (November 2011). "Epidemiology of burns throughout the world. Part I: Distribution and risk factors". Burns. 37 (7): 1087–100. doi:10.1016/j.burns.2011.06.005. PMID 21802856.
- ^ a b Lloyd EC, Rodgers BC, Michener M, Williams MS (January 2012). "Outpatient burns: prevention and care". American Family Physician. 85 (1): 25–32. PMID 22230304.
- ^ Rosdahl CB, Kowalski MT (2008). Textbook of basic nursing (9th ed.). Philadelphia: Lippincott Williams & Wilkins. p. 1109. ISBN 978-0-7817-6521-3. Archived from the original on 12 May 2016.
- ^ National Burn Repository Pg. i
- ^ a b c Forjuoh SN (August 2006). "Burns in low- and middle-income countries: a review of available literature on descriptive epidemiology, risk factors, treatment, and prevention". Burns. 32 (5): 529–37. doi:10.1016/j.burns.2006.04.002. PMID 16777340.
- ^ Eisen S, Murphy C (2009). Murphy C, Gardiner M, Eisen S (eds.). Training in paediatrics: the essential curriculum. Oxford: Oxford University Press. p. 36. ISBN 978-0-19-922773-0. Archived from the original on 25 April 2016.
- ^ a b c d Maguire S, Moynihan S, Mann M, Potokar T, Kemp AM (December 2008). "A systematic review of the features that indicate intentional scalds in children". Burns. 34 (8): 1072–81. doi:10.1016/j.burns.2008.02.011. PMID 18538478.
- ^ Peden M (2008). World report on child injury prevention. Geneva, Switzerland: World Health Organization. p. 86. ISBN 978-92-4-156357-4. Archived from the original on 24 April 2016.
- ^ World Health Organization. "World report on child injury prevention" (PDF). Archived (PDF) from the original on 31 May 2013.
- ^ a b Mitchell M, Kistamgari S, Chounthirath T, McKenzie LB, Smith GA (January 2020). "Children Younger Than 18 Years Treated for Nonfatal Burns in US Emergency Departments". Clinical Pediatrics. 59 (1): 34–44. doi:10.1177/0009922819884568. PMID 31672059. S2CID 207816299.
- ^ Hardwicke J, Hunter T, Staruch R, Moiemen N (May 2012). "Chemical burns--an historical comparison and review of the literature". Burns. 38 (3): 383–7. doi:10.1016/j.burns.2011.09.014. PMID 22037150.
- ^ a b Makarovsky I, Markel G, Dushnitsky T, Eisenkraft A (May 2008). "Hydrogen fluoride--the protoplasmic poison". The Israel Medical Association Journal. 10 (5): 381–5. PMID 18605366.
- ^ Nasoori A, Hoomand R (2017). "Maggot debridement therapy for an electrical burn injury with instructions for the use of Lucilia sericata larvae". Journal of Wound Care. 26 (12): 734–741. doi:10.12968/jowc.2017.26.12.734. PMID 29244970.
- ^ Edlich RF, Farinholt HM, Winters KL, Britt LD, Long WB (2005). "Modern concepts of treatment and prevention of lightning injuries". Journal of Long-Term Effects of Medical Implants. 15 (2): 185–96. doi:10.1615/jlongtermeffmedimplants.v15.i2.60. PMID 15777170.
- ^ Prahlow J (2010). Forensic pathology for police, death investigators, and forensic scientists. Totowa, N.J.: Humana. p. 485. ISBN 978-1-59745-404-9. Archived from the original on 20 May 2016.
- ^ Kearns RD, Cairns CB, Holmes JH, Rich PB, Cairns BA (January 2013). "Thermal burn care: a review of best practices. What should prehospital providers do for these patients?". EMS World. 42 (1): 43–51. PMID 23393776.
- ^ Balk SJ (March 2011). "Ultraviolet radiation: a hazard to children and adolescents". Pediatrics. 127 (3): e791-817. doi:10.1542/peds.2010-3502. PMID 21357345.
- ^ a b c Marx J (2010). "Chapter 144: Radiation Injuries". Rosen's emergency medicine: concepts and clinical practice (7th ed.). Philadelphia: Mosby/Elsevier. ISBN 978-0-323-05472-0.
- ^ a b Krieger J (2001). Clinical environmental health and toxic exposures (2nd ed.). Philadelphia, Pa. [u.a.]: Lippincott Williams & Wilkins. p. 205. ISBN 978-0-683-08027-8. Archived from the original on 5 May 2016.
- ^ a b c d Peck MD (August 2012). "Epidemiology of burns throughout the World. Part II: intentional burns in adults". Burns. 38 (5): 630–7. doi:10.1016/j.burns.2011.12.028. PMID 22325849.
- ^ a b Gondim RM, Muñoz DR, Petri V (June 2011). "Child abuse: skin markers and differential diagnosis". Anais Brasileiros de Dermatologia. 86 (3): 527–36. doi:10.1590/S0365-05962011000300015. PMID 21738970.
- ^ a b Herndon D, ed. (2012). "Chapter 61: Intential burn injuries". Total burn care (4th ed.). Edinburgh: Saunders. pp. 689–698. ISBN 978-1-4377-2786-9.
- ^ Jutla RK, Heimbach D (March–April 2004). "Love burns: An essay about bride burning in India". The Journal of Burn Care & Rehabilitation. 25 (2): 165–70. doi:10.1097/01.bcr.0000111929.70876.1f. PMID 15091143.
- ^ Peden M (2008). World report on child injury prevention. Geneva, Switzerland: World Health Organization. p. 82. ISBN 978-92-4-156357-4. Archived from the original on 17 June 2016.
- ^ a b c d e f g h Marx J (2010). "Chapter 60: Thermal Burns". Rosen's emergency medicine: concepts and clinical practice (7th ed.). Philadelphia: Mosby/Elsevier. ISBN 978-0-323-05472-0.
- ^ a b c d e Rojas Y, Finnerty CC, Radhakrishnan RS, Herndon DN (December 2012). "Burns: an update on current pharmacotherapy". Expert Opinion on Pharmacotherapy. 13 (17): 2485–94. doi:10.1517/14656566.2012.738195. PMC 3576016. PMID 23121414.
- ^ Hannon R (2010). Porth pathophysiology: concepts of altered health states (1st Canadian ed.). Philadelphia, PA: Wolters Kluwer Health/Lippincott Williams & Wilkins. p. 1516. ISBN 978-1-60547-781-7. Archived from the original on 1 May 2016.
- ^ a b c d Mahadevan SV, Garmel GM, eds. (2012). An introduction to clinical emergency medicine (2nd ed.). Cambridge: Cambridge University Press. pp. 216–219. ISBN 978-0-521-74776-9. Archived from the original on 20 May 2016.
- ^ Barayan D, Vinaik R, Auger C, Knuth CM, Abdullahi A, Jeschke MG. Inhibition of Lipolysis With Acipimox Attenuates Postburn White Adipose Tissue Browning and Hepatic Fat Infiltration. Shock. 2020;53(2):137-145. doi:10.1097/SHK.0000000000001439, 10.1097/SHK.0000000000001439
- ^ Jeschke MG, Gauglitz GG, Kulp GA, Finnerty CC, Williams FN, Kraft R, Suman OE, Mlcak RP, Herndon DN: Long-term persistence of the pathophysi-ologic response to severe burn injury.PLoS One6:E21245, 2011.
- ^ Jeschke M (2012). Handbook of Burns Volume 1: Acute Burn Care. Springer. p. 46. ISBN 978-3-7091-0348-7. Archived from the original on 17 May 2016.
- ^ Klingensmith M, ed. (2007). The Washington manual of surgery (5th ed.). Philadelphia, Pa.: Lippincott Williams & Wilkins. p. 422. ISBN 978-0-7817-7447-5. Archived from the original on 20 May 2016.
- ^ Rousseau AF, Losser MR, Ichai C, Berger MM (August 2013). "ESPEN endorsed recommendations: nutritional therapy in major burns". Clinical Nutrition. 32 (4): 497–502. doi:10.1016/j.clnu.2013.02.012. PMID 23582468.
- ^ Cianci P, Slade JB, Sato RM, Faulkner J (January–February 2013). "Adjunctive hyperbaric oxygen therapy in the treatment of thermal burns". Undersea & Hyperbaric Medicine. 40 (1): 89–108. PMID 23397872.
- ^ a b Enoch S, Roshan A, Shah M (April 2009). "Emergency and early management of burns and scalds". BMJ. 338 b1037. doi:10.1136/bmj.b1037. PMID 19357185. S2CID 40561988.
- ^ Hettiaratchy S, Papini R (July 2004). "Initial management of a major burn: II--assessment and resuscitation". BMJ. 329 (7457): 101–3. doi:10.1136/bmj.329.7457.101. PMC 449823. PMID 15242917.
- ^ Jeschke M (2012). Handbook of Burns Volume 1: Acute Burn Care. Springer. p. 77. ISBN 978-3-7091-0348-7. Archived from the original on 19 May 2016.
- ^ Endorf FW, Ahrenholz D (December 2011). "Burn management". Current Opinion in Critical Care. 17 (6): 601–5. doi:10.1097/MCC.0b013e32834c563f. PMID 21986459. S2CID 5525939.
- ^ Lewis SR, Pritchard MW, Evans DJ, Butler AR, Alderson P, Smith AF, Roberts I (August 2018). "Colloids versus crystalloids for fluid resuscitation in critically ill people". The Cochrane Database of Systematic Reviews. 8 (8) CD000567. doi:10.1002/14651858.CD000567.pub7. PMC 6513027. PMID 30073665.
- ^ Eljaiek R, Heylbroeck C, Dubois MJ (February 2017). "Albumin administration for fluid resuscitation in burn patients: A systematic review and meta-analysis". Burns. 43 (1): 17–24. doi:10.1016/j.burns.2016.08.001. PMID 27613476.
- ^ Curinga G, Jain A, Feldman M, Prosciak M, Phillips B, Milner S (August 2011). "Red blood cell transfusion following burn". Burns. 37 (5): 742–52. doi:10.1016/j.burns.2011.01.016. PMID 21367529.
- ^ a b Wasiak J, Cleland H, Campbell F, Spinks A (March 2013). "Dressings for superficial and partial thickness burns". The Cochrane Database of Systematic Reviews. 2013 (3) CD002106. doi:10.1002/14651858.CD002106.pub4. hdl:10072/58266. PMC 7065523. PMID 23543513.
- ^ a b c Avni T, Levcovich A, Ad-El DD, Leibovici L, Paul M (February 2010). "Prophylactic antibiotics for burns patients: systematic review and meta-analysis". BMJ. 340: c241. doi:10.1136/bmj.c241. PMC 2822136. PMID 20156911.
- ^ Hoogewerf CJ, Hop MJ, Nieuwenhuis MK, Oen IM, Middelkoop E, Van Baar ME (July 2020). "Topical treatment for facial burns". The Cochrane Database of Systematic Reviews. 2020 (7) CD008058. doi:10.1002/14651858.cd008058.pub3. PMC 7390507. PMID 32725896.
- ^ Barajas-Nava LA, López-Alcalde J, Roqué i Figuls M, Solà I, Bonfill Cosp X (June 2013). "Antibiotic prophylaxis for preventing burn wound infection". The Cochrane Database of Systematic Reviews (6) CD008738. doi:10.1002/14651858.CD008738.pub2. PMC 11303740. PMID 23740764.
- ^ Storm-Versloot MN, Vos CG, Ubbink DT, Vermeulen H (March 2010). Storm-Versloot MN (ed.). "Topical silver for preventing wound infection". The Cochrane Database of Systematic Reviews (3) CD006478. doi:10.1002/14651858.CD006478.pub2. PMID 20238345.
- ^ Dumville JC, Munson C, Christie J (December 2014). "Negative pressure wound therapy for partial-thickness burns". The Cochrane Database of Systematic Reviews. 2014 (12) CD006215. doi:10.1002/14651858.CD006215.pub4. PMC 7389115. PMID 25500895.
- ^ Chaganti P, Gordon I, Chao JH, Zehtabchi S (June 2019). "A systematic review of foam dressings for partial thickness burns". The American Journal of Emergency Medicine. 37 (6): 1184–1190. doi:10.1016/j.ajem.2019.04.014. PMID 31000315. S2CID 121615225.
- ^ Zachariah JR, Rao AL, Prabha R, Gupta AK, Paul MK, Lamba S (August 2012). "Post burn pruritus--a review of current treatment options". Burns. 38 (5): 621–9. doi:10.1016/j.burns.2011.12.003. PMID 22244605.
- ^ a b Herndon D, ed. (2012). "Chapter 64: Management of pain and other discomforts in burned patients". Total burn care (4th ed.). Edinburgh: Saunders. p. 726. ISBN 978-1-4377-2786-9.
- ^ Sinha S, Gabriel VA, Arora R, Shin W, Scott J, Bharadia S, Verly M, Rahmani W, Nickerson DA, Fraulin F, Chatterjee P, Ahuja R, Biernaskie J (June 2024). "Interventions for postburn pruritus". Cochrane Database of Systematic Reviews. 2024 (6) CD013468. doi:10.1002/14651858.CD013468.pub2. PMC 11152192. PMID 38837237.
- ^ Wasiak J, Mahar PD, McGuinness SK, Spinks A, Danilla S, Cleland H, Tan HB (October 2014). "Intravenous lidocaine for the treatment of background or procedural burn pain". The Cochrane Database of Systematic Reviews. 10 (10) CD005622. doi:10.1002/14651858.CD005622.pub4. PMC 6508369. PMID 25321859.
- ^ Herndon D, ed. (2012). "Chapter 31: Etiology and prevention of multisystem organ failure". Total burn care (4th ed.). Edinburgh: Saunders. p. 664. ISBN 978-1-4377-2786-9.
- ^ Breederveld RS, Tuinebreijer WE (September 2014). "Recombinant human growth hormone for treating burns and donor sites". The Cochrane Database of Systematic Reviews. 2014 (9) CD008990. doi:10.1002/14651858.CD008990.pub3. PMC 7119450. PMID 25222766.
- ^ Snell JA, Loh NH, Mahambrey T, Shokrollahi K (October 2013). "Clinical review: the critical care management of the burn patient". Critical Care. 17 (5): 241. doi:10.1186/cc12706. PMC 4057496. PMID 24093225.
- ^ "FDA Approves StrataGraft for the Treatment of Adults with Thermal Burns". U.S. Food and Drug Administration (FDA) (Press release). 15 June 2021. Archived from the original on 15 June 2021. Retrieved 20 April 2023.
- ^ Jeschke M (2012). Handbook of Burns Volume 1: Acute Burn Care. Springer. p. 266. ISBN 978-3-7091-0348-7. Archived from the original on 10 May 2016.
- ^ a b c d Orgill DP, Piccolo N (September–October 2009). "Escharotomy and decompressive therapies in burns". Journal of Burn Care & Research. 30 (5): 759–68. doi:10.1097/BCR.0b013e3181b47cd3. PMID 19692906.
- ^ "General data about burns". Burn Centre Care. Archived from the original on 18 October 2018. Retrieved 24 June 2019.
- ^ Barqouni L, Abu Shaaban N, Elessi K, et al. (Cochrane Wounds Group) (June 2014). "Interventions for treating phosphorus burns". The Cochrane Database of Systematic Reviews. 2014 (6) CD008805. doi:10.1002/14651858.CD008805.pub3. PMC 7173745. PMID 24896368.
- ^ Wijesinghe M, Weatherall M, Perrin K, Beasley R (May 2009). "Honey in the treatment of burns: a systematic review and meta-analysis of its efficacy". The New Zealand Medical Journal. 122 (1295): 47–60. PMID 19648986.
- ^ Norman G, Christie J, Liu Z, Westby MJ, Jefferies JM, Hudson T, et al. (July 2017). "Antiseptics for burns". The Cochrane Database of Systematic Reviews. 7 (7) CD011821. doi:10.1002/14651858.cd011821.pub2. PMC 6483239. PMID 28700086.
- ^ Jull AB, Cullum N, Dumville JC, Westby MJ, Deshpande S, Walker N (March 2015). "Honey as a topical treatment for wounds". The Cochrane Database of Systematic Reviews. 3 (3) CD005083. doi:10.1002/14651858.CD005083.pub4. PMC 9719456. PMID 25742878.
- ^ a b Dat AD, Poon F, Pham KB, Doust J (February 2012). "Aloe vera for treating acute and chronic wounds". The Cochrane Database of Systematic Reviews. 2012 (2) CD008762. doi:10.1002/14651858.CD008762.pub2. PMC 9943919. PMID 22336851.[permanent dead link]
- ^ Maenthaisong R, Chaiyakunapruk N, Niruntraporn S, Kongkaew C (September 2007). "The efficacy of aloe vera used for burn wound healing: a systematic review". Burns. 33 (6): 713–8. doi:10.1016/j.burns.2006.10.384. PMID 17499928.
- ^ Bahramsoltani R, Farzaei MH, Rahimi R (September 2014). "Medicinal plants and their natural components as future drugs for the treatment of burn wounds: an integrative review". Archives of Dermatological Research. 306 (7): 601–17. doi:10.1007/s00403-014-1474-6. PMID 24895176. S2CID 23859340.
- ^ Huang YN, Chen KC, Wang JH, Lin YK (2024). "Effects of Aloe vera on Burn Injuries: A Systematic Review and Meta-Analysis of Randomized Controlled Trials". Journal of Burn Care & Research. 45 (6): 1536–1545. doi:10.1093/jbcr/irae061. PMID 38605441.
- ^ a b Juckett G, Hartman-Adams H (August 2009). "Management of keloids and hypertrophic scars". American Family Physician. 80 (3): 253–60. PMID 19621835.
- ^ Turkington C, Dover JS, Cox B (2007). The encyclopedia of skin and skin disorders (3rd ed.). New York, NY: Facts on File. p. 64. ISBN 978-0-8160-7509-6. Archived from the original on 18 May 2016.
- ^ "Medically Sound: Treating and Caring for Burn, Electricity, and Radiation Victims". Medically Sound. 26 September 2020. Retrieved 1 November 2020.
- ^ a b National Burn Repository, Pg. 10
- ^ King C, Henretig FM, King BR, Loiselle J, Ruddy RM, Wiley II JF, eds. (2008). Textbook of pediatric emergency procedures (2nd ed.). Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins. p. 1077. ISBN 978-0-7817-5386-9. Archived from the original on 22 May 2016.
- ^ a b Roberts MC (2009). Handbook of pediatric psychology (4th ed.). New York: Guilford. p. 421. ISBN 978-1-60918-175-8. Archived from the original on 30 April 2016.
- ^ "ACI Statewide Burn Injury Service. Physiotherapy and Occupational Therapy Clinical Practice Guidelines" (PDF). 5 March 2018. Archived from the original (PDF) on 19 April 2017. Retrieved 18 February 2023.
- ^ Monstrey S, Middelkoop E, Vranckx JJ, Bassetto F, Ziegler UE, Meaume S, Téot L (August 2014). "Updated Scar Management Practical Guidelines: Non-invasive and invasive measures". Journal of Plastic, Reconstructive & Aesthetic Surgery. 67 (8): 1017–1025. doi:10.1016/j.bjps.2014.04.011. hdl:11577/2834337. PMID 24888226.
- ^ Meaume S, Le Pillouer-Prost A, Richert B, Roseeuw D, Vadoud J (July 2014). "Management of scars: updated practical guidelines and use of silicones". European Journal of Dermatology. 24 (4): 435–443. doi:10.1684/ejd.2014.2356. ISSN 1167-1122. PMID 25141160. S2CID 25937084.
- ^ "WHO Disease and injury country estimates". World Health Organization. 2009. Archived from the original on 11 November 2009. Retrieved 11 November 2009.
- ^ GBD 2013 Mortality and Causes of Death Collaborators (January 2015). "Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013". The Lancet. 385 (9963): 117–71. doi:10.1016/s0140-6736(14)61682-2. PMC 4340604. PMID 25530442.
- ^ Lozano R, Naghavi M, Foreman K, Lim S, Shibuya K, Aboyans V, et al. (December 2012). "Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010". The Lancet. 380 (9859): 2095–128. doi:10.1016/S0140-6736(12)61728-0. hdl:10536/DRO/DU:30050819. PMC 10790329. PMID 23245604. S2CID 1541253.
- ^ Edlich RF, Farinholt HM, Winters KL, Britt LD, Long WB (2005). "Modern concepts of treatment and prevention of electrical burns". Journal of Long-Term Effects of Medical Implants. 15 (5): 511–32. doi:10.1615/jlongtermeffmedimplants.v15.i5.50. PMID 16218900.
- ^ a b c Ahuja RB, Bhattacharya S (August 2004). "Burns in the developing world and burn disasters". BMJ. 329 (7463): 447–9. doi:10.1136/bmj.329.7463.447. PMC 514214. PMID 15321905.
- ^ Gupta (2003). Textbook of Surgery. Jaypee Brothers Publishers. p. 42. ISBN 978-81-7179-965-7. Archived from the original on 27 April 2016.
- ^ Pećanac M, Janjić Z, Komarcević A, Pajić M, Dobanovacki D, Misković SS (2013). "Burns treatment in ancient times". Medicinski Pregled. 66 (5–6): 263–7. doi:10.1016/s0264-410x(02)00603-5. PMID 23888738.
- ^ a b Song D (5 September 2012). Plastic surgery (3rd ed.). Edinburgh: Saunders. p. 393.e1. ISBN 978-1-4557-1055-3. Archived from the original on 2 May 2016.
- ^ Wylock P (2010). The life and times of Guillaume Dupuytren, 1777–1835. Brussels: Brussels University Press. p. 60. ISBN 978-90-5487-572-7. Archived from the original on 16 May 2016.
- ^ Hutcher N, Haynes BW Jr (June 1972). "The Evans Formula Revisited". Journal of Trauma and Acute Care Surgery. 12 (6): 453–8. doi:10.1097/00005373-197206000-00001. PMID 5033490.
- ^ Regan A, Hotwagner DT (2023). "Burn Fluid Management". StatPearls. StatPearls Publishing. PMID 30480960. Retrieved 31 October 2023.
General and cited references
[edit]- National Burn Repository 2012 Report (PDF). Dataset Version 8.0. Chicago: American Burn Association. 2012. Archived from the original (PDF) on 3 March 2016. Retrieved 20 April 2013.
External links
[edit]- WHO fact sheet on burns
- Parkland Formula
- "Burns". MedlinePlus. U.S. National Library of Medicine.
Definition and Classification
Definition
A burn is tissue damage to the skin or other organic tissues caused by exposure to excessive heat, chemicals, electricity, radiation, or friction.[1][8][10] Such injuries disrupt cellular integrity through coagulation necrosis when thermal energy exceeds tissue tolerance, typically above 44°C (111°F) for skin, leading to localized destruction that varies by agent and exposure duration.[9][11] Burns primarily affect the epidermis and dermis but can extend to subcutaneous fat, muscle, or bone in severe cases, with depth determined by factors like temperature, contact time, and tissue conductivity.[3][2] Unlike abrasions or lacerations, burns involve denaturation of proteins and disruption of vascular supply, often triggering an inflammatory response and potential systemic effects such as fluid shifts or infection risk even in localized injuries.[12] The term encompasses thermal scalds from hot liquids (e.g., water above 60°C causing injury in seconds), contact burns from solids, and non-thermal variants like chemical corrosion from acids or alkalis.[8][11]Classification by Depth
Burns are classified by depth according to the layers of skin and underlying tissues affected, traditionally using first-, second-, third-, and fourth-degree categories, though modern terminology often employs superficial, partial-thickness (superficial and deep), full-thickness, and deeper extensions.[3][13] This classification determines healing potential, pain, appearance, and required interventions, with shallower burns typically healing spontaneously while deeper ones necessitate surgical excision and grafting.[4] Assessment involves evaluating color, blanching, sensation, and moisture, as initial clinical judgment can overestimate depth due to evolving injury.[3] First-degree burns, also termed superficial burns, involve only the epidermis, the outermost skin layer, causing erythema, mild edema, and pain without blistering or tissue destruction.[3][6] These injuries heal within 3 to 6 days via epithelial regeneration, leaving no scarring but possible hyperpigmentation.[14] Common examples include mild sunburns, where ultraviolet radiation damages keratinocytes without deeper penetration.[8] Second-degree burns penetrate the epidermis and varying depths of the dermis, subdivided into superficial partial-thickness (affecting upper dermis) and deep partial-thickness (reaching deeper dermis).[13] Superficial partial-thickness burns appear moist, red or mottled, form blisters, blanch to pressure, and exhibit intense pain due to preserved nerve endings; they heal in 7 to 21 days with minimal scarring if infection is avoided.[3] Deep partial-thickness burns are drier, paler or white, with fixed staining, reduced sensation, and slower healing often requiring debridement or grafting to prevent contractures.[15] Third-degree burns, or full-thickness burns, destroy the entire epidermis and dermis, appearing leathery, dry, white, brown, or charred, with thrombosed vessels preventing blanching and insensate areas from nerve destruction.[3][6] These do not heal spontaneously, forming eschar that must be excised surgically, followed by skin grafting, as adnexal structures for re-epithelialization are obliterated.[9] Fourth-degree burns extend beyond full-thickness into subcutaneous fat, muscle, tendon, or bone, often resulting from high-voltage electricity, prolonged flame exposure, or chemicals, presenting as blackened or carbonized tissue with extensive necrosis.[4] Such injuries carry high morbidity, frequently necessitating amputation, fasciotomy, or reconstruction, and involve systemic complications like compartment syndrome.[3] Accurate depth classification guides prognosis, with deeper burns correlating to poorer outcomes and higher risks of infection or functional loss.[13]Classification by Extent
Classification of burns by extent quantifies the percentage of total body surface area (TBSA) affected by partial-thickness (second-degree) or deeper burns, excluding superficial (first-degree) burns which do not require the same resuscitative measures.[3] This assessment guides fluid resuscitation, determines the need for transfer to specialized burn centers, and informs prognosis, as burns exceeding 20% TBSA in adults are considered major and associated with higher mortality risks without intensive care.[16] [17] Thresholds for severity are lower in children and elderly patients, where even 10-15% TBSA involvement can necessitate aggressive intervention due to reduced physiological reserves.[18] The Rule of Nines, also known as the Wallace Rule, provides a rapid estimation method for adults by dividing the body into regions approximating 9% or 18% of TBSA:| Body Region | TBSA Percentage |
|---|---|
| Head and neck | 9% |
| Each upper extremity | 9% |
| Each lower extremity | 18% |
| Anterior trunk | 18% |
| Posterior trunk | 18% |
| Perineum/genitalia | 1% |
Special Classifications
Special classifications of burns encompass injuries that, regardless of depth or extent, require unique management due to their location, associated complications, or impact on underlying structures. These include burns to critical anatomic sites, circumferential burns, inhalation injuries, and burns in vulnerable populations, as outlined by referral criteria from the American Burn Association (ABA). Such classifications prioritize prognostic implications and the need for specialized intervention, often mandating transfer to a verified burn center.[27] Burns involving special anatomic regions—such as the face, eyes, ears, neck, hands, feet, genitalia, perineum, or major joints—demand heightened attention even if limited in size, due to risks of functional impairment, scarring, and infection. For instance, facial burns threaten airway patency and aesthetics, while hand burns can compromise dexterity through contractures; perineal involvement elevates sepsis risk from bacterial colonization. The ABA recommends burn center referral for any second- or third-degree burns in these areas, irrespective of total body surface area (TBSA) affected.[27][28] Circumferential burns, encircling a limb or the trunk, pose acute threats from compartment syndrome due to eschar-induced constriction of underlying tissues and vasculature. In extremities, this can lead to ischemia and necrosis, necessitating urgent escharotomy or fasciotomy to restore perfusion; torso involvement may impair ventilation by restricting chest expansion. Diagnosis relies on clinical assessment of distal pulses, capillary refill, and sensation, with prophylactic intervention advised in deep burns exceeding 360 degrees around a limb.[29][30] Inhalation injuries represent a distinct category affecting the upper and lower airways, often complicating cutaneous burns from enclosed-space fires. They arise from thermal damage, smoke toxins (e.g., carbon monoxide, cyanide), or particulate aspiration, manifesting as stridor, carbonaceous sputum, or bronchospasm; diagnosis involves bronchoscopy and carboxyhemoglobin levels. Mortality doubles with coexisting skin burns, driven by pulmonary edema and acute respiratory distress syndrome.[28][31] Burns in special populations, including children under 10 years, adults over 50, or those with comorbidities (e.g., diabetes, cardiopulmonary disease), warrant classification as high-risk due to altered physiology and healing capacity. Pediatric burns >10% TBSA or elderly burns >10-20% TBSA elevate fluid requirements and infection susceptibility; preexisting conditions prolong recovery and amplify mortality. The ABA criteria emphasize transfer for these cases to mitigate systemic complications like hypovolemic shock or organ failure.[27][28]Causes
Thermal Causes
Thermal burns result from exposure to excessive heat sources that raise skin and tissue temperatures, leading to protein denaturation and cell death. These injuries account for approximately 86% of all burn cases requiring medical attention. Common mechanisms include direct contact with flames, hot liquids, steam, or solid objects, with heat transfer occurring via conduction, convection, or radiation.[11][32] Flame burns, often from open fires or ignited clothing, represent a major cause of severe thermal injuries, comprising about 37% of such cases. These typically involve prolonged exposure to temperatures exceeding 44°C (111°F), resulting in deeper tissue damage due to sustained heat application. In the United States, fire-related injuries numbered around 398,000 in 2021, frequently linked to residential fires or accidents involving flammable materials.[32][33] Scald burns arise from hot liquids or steam, accounting for roughly 24% of severe burns and predominating in pediatric cases, particularly among children under 2 years. Sources include boiling water, cooking oils, or beverages, with immersion or splash mechanisms causing rapid heat transfer to the skin. Steam scalds can penetrate deeper than equivalent liquid temperatures due to latent heat release. Globally, scalds contribute significantly to burn morbidity in low- and middle-income countries.[32][1][6] Contact burns occur from brief or prolonged touch with hot solids, such as irons, ovens, or vehicle exhausts, leading to localized injuries proportional to contact time and temperature. These comprise a substantial portion of outpatient-treated burns, with over 252,000 hot object-related cases reported in the US in 2021. Friction can exacerbate contact burns, as seen in road rash combined with hot surface exposure.[33][11]Chemical Causes
Chemical burns occur when living tissue is exposed to corrosive or reactive substances that cause damage through chemical reactions, distinct from thermal burns by continuing to injure tissue until the agent is neutralized or removed.[34] These injuries typically involve acids or alkalis, but can also result from other agents like hydrofluoric acid or phenols.[35] In the United States, chemical burns account for approximately 3-10% of burn center admissions, often occurring in occupational settings such as manufacturing or construction, or domestically via household cleaners.[34] Acids, defined as proton donors (H⁺), induce coagulation necrosis by denaturing proteins and dehydrating cells, forming a leathery eschar that partially self-limits penetration depth.[35] Common acid culprits include sulfuric acid (from car batteries), hydrochloric acid (muriatic acid in pool cleaners), and nitric acid (used in metal processing), which produce immediate pain and visible coagulation upon contact.[35] For instance, battery acid exposures frequently cause localized burns to extremities during vehicle maintenance.[34] Alkalis, or bases, act as proton acceptors (OH⁻ or other anions), causing liquefaction necrosis through saponification of fats and solubilization of proteins, resulting in deeper tissue invasion without eschar formation.[35] [36] Typical alkalis include sodium hydroxide (lye in drain or oven cleaners) and ammonia (in fertilizers or cleaners), which penetrate more readily and may delay symptoms, exacerbating damage.[37] Cement, containing calcium oxide, causes alkaline burns via hydration to form calcium hydroxide, common in construction-related injuries.[38] Specialized chemicals like hydrofluoric acid (HF) combine acid and fluoride ion effects, with the latter binding calcium and magnesium in tissues, leading to profound systemic toxicity beyond local necrosis; exposures often occur in glass etching or semiconductor industries.[38] Phenol (carbolic acid), used in disinfectants or plastics, denatures proteins and lipids while being lipid-soluble for deep absorption, causing progressive numbness followed by necrosis.[35] Organic compounds such as petroleum distillates or formic acid (in some industrial processes) can also induce burns via solvent action or exothermic reactions.[34] Intentional chemical assaults, involving acids like sulfuric, have risen in some regions, contributing to severe mutilating injuries.[39]Electrical Causes
Electrical burns arise when electric current passes through the body, converting electrical energy into heat via Joule heating (I²R losses), where tissue resistance generates localized temperatures exceeding 1000°C, leading to coagulation necrosis and vascular thrombosis.[40] The extent of damage depends on current type (alternating vs. direct), voltage, duration of exposure, current path, and individual factors like skin moisture and thickness.[41] Unlike thermal burns, electrical injuries often produce minimal external charring but profound internal destruction along the current's trajectory, affecting muscles, nerves, and bones disproportionately.[40] High-voltage injuries, defined as greater than 1000 volts, predominate in occupational settings such as construction or utility work, involving power lines or industrial equipment, and account for the majority of severe cases due to deeper penetration and higher energy transfer.[42] [43] Low-voltage injuries, typically under 1000 volts from household sources like outlets or appliances, more commonly occur in domestic accidents, especially among children, and usually cause superficial contact burns without extensive deep-tissue involvement unless prolonged.[44] In the United States, electrical injuries represent about 5% of burn center admissions and contribute to roughly 1000 deaths annually, with high-voltage cases linked to higher amputation rates and complications like compartment syndrome.[45] Approximately 4400 electrical injuries are reported yearly, though underreporting may occur in milder cases.[43] Characteristic features include entry wounds—small, crater-like lesions with central pallor from vaporized tissue—and exit wounds, often larger and explosive due to concentrated current dispersal, typically on the lower limbs or buttocks.[41] Arc or flash burns result from external arcing without body conduction, causing superficial cutaneous injuries from radiant heat or ignited clothing, while true conduction burns follow the path of least resistance through vascular structures, sparing skin but devastating deeper tissues.[46] Pathophysiological mechanisms extend beyond thermal effects, including electroporation disrupting cell membranes, neuromuscular tetany causing falls or fractures, and systemic disruptions like arrhythmias from myocardial conduction interference.[47] [48]Radiation Causes
Radiation burns result from exposure to high-energy electromagnetic radiation that damages skin cells without direct thermal effects, primarily ultraviolet (UV) or ionizing radiation such as X-rays and gamma rays.[49] These injuries differ from thermal burns by involving photochemical or ionization mechanisms leading to DNA damage, cell death, and inflammation.[50] The most prevalent radiation burns are sunburns caused by UV radiation, particularly UVB wavelengths (280-315 nm), which penetrate the epidermis and induce direct DNA lesions like cyclobutane pyrimidine dimers in keratinocytes.[51] This damage triggers apoptosis of affected cells and release of pro-inflammatory cytokines, resulting in erythema, edema, and pain typically appearing 4-6 hours post-exposure and peaking at 24-36 hours.[51] Sources include solar UV, with overexposure linked to factors like skin type, altitude, and time of day; artificial sources such as tanning beds amplify risk, with UVA (315-400 nm) contributing to deeper penetration and oxidative stress via reactive oxygen species.[51][52] Ionizing radiation burns arise from high doses (>2 Gy) of X-rays or gamma rays, often in radiotherapy or accidental exposures like nuclear incidents, causing basal cell layer destruction, vascular endothelial damage, and subsequent erythema or desquamation.[53][54] In radiation therapy for cancer, nearly all patients develop some degree of radiation dermatitis, graded from mild erythema at 10-20 Gy to severe ulceration beyond 50 Gy cumulative dose.[55] Acute high-dose exposures (>20 Gy localized) can produce prodromal erythema within hours, followed by main-phase burns resembling second- or third-degree thermal injuries, complicated by delayed necrosis weeks later due to progressive vascular sclerosis.[56][53] Such burns from events like the Chernobyl disaster in 1986 demonstrated dose-dependent severity, with beta-particle contamination exacerbating cutaneous injury.[50]Other Causes
Friction burns arise from the rapid shearing force of skin against a rough surface, such as pavement, carpet, or machinery, generating localized heat and ablating superficial tissue layers. These injuries, often termed abrasions or road rash, combine mechanical disruption with frictional heating, typically resulting in partial-thickness damage that exposes dermis and risks secondary infection from embedded debris. In clinical practice, friction burns constitute a significant portion of trauma cases, particularly in traffic accidents where speeds exceed 30 km/h, as higher velocities increase kinetic energy transfer and tissue depth affected.[57][58][59] Cold-induced injuries, known as frostbite, occur when tissues freeze due to exposure to temperatures below 0°C, often accelerated by wind chill or conductive cooling from wet clothing, forming ice crystals that disrupt cell membranes and vasculature. Unlike heat-based burns, frostbite involves vasoconstriction followed by reperfusion injury upon thawing, leading to thrombosis and necrosis; it is graded by depth, with first-degree limited to epidermal pallor and second-degree progressing to subepidermal blisters containing clear fluid. Incidence peaks in outdoor occupations or recreational activities in subzero conditions, with global estimates from military data indicating rates up to 10% in extreme polar expeditions without protective gear.[1][60][14]Intentional Causes
Intentional burns arise from deliberate acts, encompassing self-inflicted injuries and those imposed by others through assault, abuse, or violence. These differ from accidental burns in etiology, often linked to psychiatric conditions, interpersonal conflict, or socioeconomic stressors, and typically exhibit distinct patterns such as immersion scalds, contact burns from heated objects, or flame exposure confined to accessible body areas.[61] Outcomes are generally more severe, with higher mortality rates, prolonged hospital stays, and increased complication risks compared to unintentional burns.[62] Self-inflicted burns, comprising approximately 49% of intentional cases in U.S. burn center data from over 54,000 patients, frequently stem from suicide attempts or non-suicidal self-harm associated with mental health disorders like depression or psychosis.[63] These injuries represent 0.67% to 9% of total burn admissions in developed countries, with perpetrators more often male (up to 85.7% in some cohorts) and involving larger total body surface area (mean 29.6% in recent analyses).[64] [65] Incidence rates are low, such as 0.22 per 100,000 adults in Finland, but long-term mortality remains elevated due to recurrent self-harm and comorbidities.[66] Assault-related burns account for the remaining 51% of intentional injuries in similar U.S. datasets, including spousal abuse, elder mistreatment, and attacks by acquaintances or strangers.[63] In adult populations, 4% of burn admissions (148 out of 3,678 cases) result from interpersonal assault, often involving scalding liquids or open flames during domestic disputes.[67] Prevalence varies regionally, reaching 17.4% of burns in South African studies, with males 1.5 times more likely to be victims.[68] These injuries correlate with substance abuse, prior trauma histories, and urban settings, yielding fewer hospital-free days and higher resource demands.[69] In pediatric cases, non-accidental burns from abuse constitute 1% to 25% of childhood burn injuries, with estimates up to 20% in specialized reviews and 9.6% in targeted cohorts.[70] [71] [72] Common mechanisms include forced immersion in hot water (producing glove-and-stocking distribution) or contact with irons, cigarettes, or radiators, often affecting the lower extremities or perineal areas inconsistent with accidental patterns. Risk factors include prior child protective services involvement, low household income, and caregiver substance use or neglect histories.[73] Distinguishing these requires multidisciplinary assessment, as delayed presentation or inconsistent histories heighten suspicion.[74] Intentional burns overall predominate in low- and middle-income countries, where cultural factors like dowry-related violence exacerbate assault rates among young women.[61]Pathophysiology
Local Tissue Damage
Burn injuries induce local tissue damage primarily through direct thermal, chemical, electrical, or radiation energy transfer, resulting in protein denaturation and coagulative necrosis of affected cells.[9] In thermal burns, heat disrupts cell membranes, causes immediate thrombosis of microvasculature, and leads to irreversible cell death in the central zone of injury.[15] The epidermis, with its high water and low protein content, resists damage more than the protein-rich dermis, where collagen denaturation predominates.[9] This necrosis extends variably based on exposure factors such as temperature above 44°C, duration, and tissue conductivity, with temperatures exceeding 70°C causing near-instantaneous full-thickness destruction.[75] The local injury manifests in three concentric zones: the central zone of coagulation features avascular, necrotic tissue with fixed thrombosed vessels and no viable cells, rendering it unsalvageable.[11] Surrounding it, the zone of stasis exhibits reduced perfusion from edema, inflammation, and microvascular damage, where tissue viability depends on timely intervention to prevent progression to necrosis via oxygen free radicals and cytokine release.[76] [11] The outermost zone of hyperemia involves transient vasodilation and inflammatory hyperperfusion, typically resolving without permanent damage unless infection or hypoperfusion converts adjacent zones.[11] Vascular permeability increases rapidly post-injury due to endothelial damage and mediator release (e.g., histamine, prostaglandins), causing interstitial edema that blisters in partial-thickness burns and contributes to compartment syndrome in deeper injuries involving muscle or bone.[9] In full-thickness burns, destruction extends beyond dermis to subcutaneous fat, fascia, or deeper structures, impairing sensory nerves and resulting in insensate, leathery eschar.[75] Electrical burns uniquely produce progressive necrosis along current paths due to arcing and deep tissue heating, often exceeding surface appearance.[11] Chemical burns involve ongoing tissue dissolution until neutralization, with corrosives like acids causing protein coagulation and bases saponification of lipids.[9] Depth of damage correlates with clinical burn degree: first-degree limited to epidermis with erythema and no necrosis; second-degree involving partial dermis with blistering and viable appendages; third-degree full dermal destruction; and fourth-degree charring of underlying muscle, tendon, or bone.[9] Local immune suppression occurs via keratinocyte damage and reduced antimicrobial peptide production, heightening infection risk despite initial hyperinflammation.[77] Healing potential diminishes with depth, as adnexal structures for re-epithelialization are lost beyond superficial partial-thickness injuries.[75]Systemic Inflammatory Response
Severe burn injuries, typically involving more than 20-30% total body surface area (TBSA), initiate a local inflammatory cascade at the wound site that rapidly escalates to systemic involvement due to the release of damage-associated molecular patterns (DAMPs) from necrotic tissue, such as high-mobility group box 1 (HMGB1) and heat shock proteins.[78][79] These DAMPs, along with endogenous cytokines like interleukin-1 (IL-1), IL-6, and tumor necrosis factor-alpha (TNF-α), activate innate immune cells including macrophages and neutrophils, triggering a "cytokine storm" that amplifies inflammation beyond the burn zone.[79][80] This response is evolutionarily designed to combat infection and promote repair but becomes dysregulated in major burns, leading to systemic inflammatory response syndrome (SIRS).[81][82] SIRS manifests clinically through criteria including body temperature above 38°C or below 36°C, heart rate exceeding 90 beats per minute, respiratory rate over 20 breaths per minute or hyperventilation with PaCO2 below 32 mmHg, and abnormal white blood cell count (greater than 12,000/mm³, less than 4,000/mm³, or more than 10% immature forms).[81] In burn patients, this is driven by widespread endothelial activation and damage, causing increased vascular permeability, fluid leakage into tissues, and profound edema not limited to the burn area.[78] Complement activation and coagulopathy further exacerbate the process, with plasmin and other proteases contributing to remote organ injury via microvascular thrombosis and ischemia.[83] Persistent elevation of pro-inflammatory mediators persists for weeks, correlating with burn size; for instance, studies show IL-6 levels peaking within hours and remaining high up to 14 days post-injury in patients with >40% TBSA burns.[84][85] The systemic effects extend to immunosuppression and hypermetabolism, where unchecked inflammation shifts T-cell populations toward exhaustion and releases immature neutrophils, increasing susceptibility to sepsis while inducing a catabolic state with protein breakdown and organ stress.[85][86] In severe cases, this progresses to multiple organ dysfunction syndrome (MODS), with pulmonary complications like acute respiratory distress syndrome (ARDS) occurring in up to 50% of patients with burns over 40% TBSA due to neutrophil-mediated lung injury.[78] Cardiac output initially increases compensatorily but can fail under cytokine-induced myocardial depression, while hepatic and renal dysfunction arises from hypoperfusion and direct inflammatory insult.[87] This biphasic response—initial hyperinflammation followed by immune paralysis—underscores the need for targeted modulation, as evidenced by reduced cytokine levels with early wound excision in clinical trials.[88]Hypermetabolic and Immunological Changes
Severe burn injuries trigger a hypermetabolic state characterized by elevated resting energy expenditure, reaching 130-180% of predicted values and peaking around two weeks post-injury, with onset typically within five days and persistence up to two to three years depending on burn extent.[87][89] This response is driven primarily by central nervous system-mediated elevations in catecholamines, cortisol (increasing 5-7 fold in urinary excretion), and glucagon, alongside inflammatory cytokines, leading to insulin resistance and hyperglycemia with blood glucose levels of 170-180 mg/dl.[87] Cellular mechanisms include endoplasmic reticulum stress, unfolded protein response activation, and inflammasome signaling in tissues such as muscle and adipose, exacerbating catabolic processes.[89] Metabolically, hypermetabolism manifests as accelerated lipolysis with 2-4 fold rises in free fatty acids and triglycerides, hepatic triglyceride accumulation (3-5 fold increase), and profound muscle protein breakdown exceeding synthesis, resulting in net negative protein balance and lean body mass losses of 10-25% within weeks to months.[87][89] These alterations contribute to systemic consequences including bone mineral density reduction (up to 3-5%), cardiac output elevation (to 160% of predicted), and heightened vulnerability to infections (average 2.5 episodes per patient), with unresolved catabolism linked to prolonged morbidity and mortality rates around 8% in severe cases.[87] Concomitant immunological changes involve a biphasic response: an acute systemic inflammatory response syndrome (SIRS) driven by damage-associated molecular patterns activating pattern recognition receptors, prompting neutrophil and macrophage influx with surges in pro-inflammatory cytokines such as IL-1, IL-6, and TNF-α peaking at 24-48 hours.[79] This transitions to compensatory anti-inflammatory response syndrome (CARS) by days 3-7, marked by immunosuppression through T-cell apoptosis, lymphocyte depletion, a Th1-to-Th2 shift suppressing cellular immunity, and elevations in regulatory T cells, myeloid-derived suppressor cells, and anti-inflammatory cytokines like IL-10 and TGF-β.[79][90] The resulting immune dysregulation, including monocyte dysfunction and intestinal barrier compromise facilitating bacterial translocation, substantially elevates sepsis risk, with IL-10 levels correlating to burn size and prognosis.[90] In chronic phases exceeding two weeks, unresolved responses evolve into persistent inflammation, immunosuppression, and catabolism syndrome (PIICS), featuring sustained myeloid-derived suppressor cell activity, T-cell exhaustion, and protracted muscle wasting alongside ongoing infection susceptibility, potentially lasting up to 36 months.[79] These immunological shifts intersect with hypermetabolism, amplifying catabolic demands and organ dysfunction through intertwined inflammatory and hormonal pathways.[87]Clinical Presentation and Diagnosis
Signs and Symptoms
Burns exhibit local signs and symptoms primarily determined by the depth of tissue injury, classified into degrees based on affected skin layers, while systemic manifestations arise in extensive injuries involving greater than 20% total body surface area (TBSA). Local changes include alterations in skin color, texture, sensation, and integrity, with pain intensity varying by nerve involvement. Systemic effects, such as hypovolemic shock from fluid loss, may present with tachycardia, hypotension, and altered mental status in severe cases.[15][8] First-degree burns, confined to the epidermis, cause erythema, mild edema, and intense pain without blistering or skin breakage; the affected area appears red, dry, and blanches under pressure, with symptoms peaking within 48-72 hours before peeling occurs, typically resolving in 3-6 days without scarring.[91][5][92] Second-degree burns, extending into the dermis, are subdivided into superficial partial-thickness (moist, red or pink, blistered, blanching, and severely painful) and deep partial-thickness (paler, drier, with thicker blisters or eschar, reduced sensation, and slower healing potential); common features include fluid-filled blisters prone to rupture, significant swelling, and weeping from damaged capillaries, with pain exacerbated by air exposure or touch.[8][3][92] These often heal in 1-3 weeks for superficial types but may scar or convert to deeper injury if infected.[14] Blisters in second-degree burns typically contain clear fluid and are surrounded by red, swollen skin, with the affected area being painful; normal healing proceeds gradually over days to weeks. Signs of infection in a burn blister or wound include increased or worsening pain, spreading redness, swelling, or warmth around the wound, pus, foul-smelling discharge or oozing, fever, red streaks extending from the wound, and lack of improvement or worsening symptoms. Immediate medical attention should be sought if any of these signs develop to prevent serious complications such as cellulitis or sepsis.[8][93][14] Third-degree burns involve full-thickness dermal destruction, presenting as dry, leathery, or waxy skin that is white, gray, or charred, with thrombosed vessels visible beneath; due to nerve endings destruction, the area is insensate to pain, though surrounding partial-thickness zones may remain painful, and no blanching occurs.[5][28] Fourth-degree burns extend beyond skin into subcutaneous tissue, muscle, or bone, appearing blackened or eschar-covered with profound necrosis and absent sensation, often accompanied by compartment syndrome signs like tense swelling and vascular compromise.[3] In large or circumferential burns, additional local signs include restricted movement from eschar formation and potential airway compromise from facial or inhalational injury, evidenced by singed nasal hairs, carbonaceous sputum, or stridor. Systemic symptoms in major burns include early hyperdynamic circulation with fever, tachycardia, and oliguria from fluid shifts, progressing to shock characterized by cool clammy skin, rapid shallow breathing, and confusion if resuscitation is inadequate.[15][2]Diagnostic Assessment
Diagnosis of burn injuries begins with a systematic evaluation following the Advanced Trauma Life Support (ATLS) protocol, prioritizing airway, breathing, circulation, disability, and exposure to identify life-threatening issues alongside burn-specific assessments.[94] A detailed patient history is essential, including the mechanism and timing of injury, duration of exposure, ambient temperature, first-aid measures applied, associated trauma, pre-existing medical conditions, medications, allergies, and tetanus immunization status, as these inform prognosis and management needs.[12] Physical examination requires full exposure of the patient while preventing hypothermia, encompassing a head-to-toe survey to map all burn sites, evaluate depth, extent, and circumferential involvement that may compromise circulation or respiration.[12] Burn depth is clinically classified based on tissue appearance, sensation, and capillary response, though initial assessments may evolve over 48 hours as edema develops and superficial injuries demarcate.[3] First-degree burns affect only the epidermis, presenting as erythematous, dry, and painful without blisters or edema.[3] Superficial partial-thickness (second-degree) burns involve the upper dermis, appearing moist, blistered, red or mottled, with brisk capillary refill and severe pain to pinprick.[3] Deep partial-thickness burns extend into the deeper dermis, showing white or pale red hues, sluggish refill, reduced sensation, and potential for re-epithelialization if managed promptly.[3] Full-thickness (third-degree) burns destroy the entire dermis, exhibiting a leathery, dry texture in white, brown, or charred tones, with insensate areas due to nerve destruction and requiring surgical excision.[3] Fourth-degree burns penetrate underlying tissues like muscle or bone, often with eschar and systemic implications.[27] Extent is quantified as percentage of total body surface area (%TBSA) affected by partial- and full-thickness burns (excluding first-degree), using the Rule of Nines for adults—assigning 9% or multiples to body regions—or the more precise Lund-Browder chart for children, which adjusts for age-related proportions.[27] Accurate %TBSA guides fluid resuscitation, transfer decisions, and mortality risk, with burns exceeding 20-25% in adults or 10-15% in children warranting specialized care.[95] Circumferential burns necessitate vigilance for compartment syndrome, assessed via serial exams or Doppler for distal pulses.[12] Inhalation injury suspicion arises from history of smoke exposure in enclosed spaces, facial or oropharyngeal burns, carbonaceous sputum, singed nasal hairs, or voice changes, prompting evaluation for airway edema or carbon monoxide/cyanide poisoning.[12] Diagnostic adjuncts include arterial blood gas for carboxyhemoglobin levels (>10% indicative in non-smokers), fiberoptic bronchoscopy to visualize mucosal damage, and baseline chest radiography, though the latter has low sensitivity early on.[12] Laboratory tests such as complete blood count, electrolytes, renal function, and lactate support resuscitation monitoring but are not diagnostic for the burn itself.[96] Rarely, biopsy confirms indeterminate depth, but clinical judgment by experienced providers remains the standard, with accuracy improving to 80-90% post-48 hours.[95]Severity Evaluation
Severity evaluation of burns integrates assessment of depth, extent, location, patient-specific factors, and associated injuries to guide prognosis, treatment, and referral decisions. Depth determines tissue involvement and healing potential, while extent, measured as total body surface area (TBSA) affected by partial- and full-thickness burns, correlates with mortality risk and resource needs; superficial burns are excluded from TBSA calculations as they heal without scarring.[3][4] Location matters due to functional implications—burns to the face, hands, feet, genitalia, perineum, or major joints elevate severity owing to risks of contracture, sensory_loss, or airway compromise.[27] Patient age extremes (under 10 or over 50 years), inhalation injury, electrical or chemical mechanisms, and comorbidities like diabetes or immunosuppression further amplify severity by increasing complication rates.[97] Burn depth is classified based on clinical appearance, blanching response, pain to pinprick, and sensation, with final determination often refined at 24-48 hours post-injury as edema evolves. Superficial burns (first-degree) affect only the epidermis, presenting as erythematous, dry, painful skin that blanches and heals in 3-6 days without scarring. Superficial partial-thickness burns (second-degree, superficial) involve the upper dermis, appearing moist, blistered, and hypersensitive with capillary refill, healing in 1-3 weeks via re-epithelialization. Deep partial-thickness burns (second-degree, deep) extend to deeper dermis, showing white or mottled red hues, reduced sensation, and sluggish blanching, often requiring weeks to months for healing and risking hypertrophic scars or grafting. Full-thickness burns (third-degree) destroy the entire dermis, manifesting as leathery, insensate, charred or waxy tissue without blanching, necessitating surgical excision and grafting due to absent regenerative elements. Fourth-degree burns extend to subcutaneous fat, muscle, or bone, typically from high-voltage electricity or prolonged exposure, with profound functional loss and high amputation risk.[3][4][98] Extent is quantified via diagrams or methods adjusted for age and accuracy. The Rule of Nines divides the body into segments worth 9% or multiples of 9% TBSA (e.g., head 9%, each arm 9%, front torso 18%), suitable for adults but overestimating in children; the Lund-Browder chart provides age-specific proportions for pediatrics, improving precision. The palmar method estimates 0.8-1% TBSA per patient's hand (palm plus fingers), useful for scattered burns. TBSA exceeding 20-30% in adults or 10-15% in extremes of age signals major burn status, with mortality models like the Baux score (age + TBSA) or revised Baux (incorporating inhalation) predicting outcomes empirically.[19][97][23] The American Burn Association outlines referral criteria to specialized centers for optimal outcomes, recommending transfer for partial-thickness burns ≥10% TBSA, any full-thickness burns, or those involving critical areas regardless of size. Additional indications include burns in patients under 10 or over 50 years with ≥5% partial-thickness or any full-thickness involvement, suspected inhalation injury (e.g., singed nasal hairs, carbonaceous sputum), chemical/electrical burns, or circumferential burns risking compartment syndrome. These criteria stem from data showing reduced mortality and morbidity at verified burn centers handling high volumes annually.[27][97][99]Management
Initial and Prehospital Care
Initial and prehospital care for burn injuries prioritizes halting the burning process, stabilizing vital functions, and facilitating rapid transport to appropriate facilities to minimize secondary damage from hypovolemia, hypothermia, and infection. Scene safety must be ensured before approaching the patient, followed by removal from the heat, chemical, or electrical source; for flame burns, victims should be instructed to stop, drop, and roll or have flames extinguished with water while lying with the burning side uppermost to prevent flame spread upward.[100] Clothing and jewelry near the burn should be gently removed if not adherent, as swelling can cause constriction.[101] For thermal burns presenting within the first few minutes post-injury, the affected area should be irrigated with tepid (15-25°C, not ice-cold) running water for 10-20 minutes to dissipate heat and reduce tissue damage depth, though prolonged cooling risks hypothermia and is avoided in large burns exceeding 20% total body surface area (TBSA); burn blisters should generally not be popped or intentionally ruptured, as this increases the risk of infection and the intact blister serves as a natural protective barrier. Do not pop blisters yourself. If a blister is large, painful, or requires drainage, it should be managed by a healthcare professional (such as a doctor or in a hospital setting) using sterile techniques, not by a pharmacist; pharmacists can provide advice on dressings or over-the-counter treatments but do not perform procedures like draining blisters.[101] [102] [103] [104] [105] After cooling, gently pat the burned area dry without rubbing to avoid further tissue damage; for burns on the hand, cover loosely with a sterile bandage or clean cloth (e.g., aluminum foil if nothing else available), elevate the hand to reduce swelling, and take an over-the-counter pain reliever such as ibuprofen or paracetamol per package instructions.[104] [102] For minor (first-degree) burns characterized by redness and pain without blisters or deeper tissue involvement, when specific medicated burn ointments are unavailable, the following evidence-based home care steps apply after initial cooling: apply pure aloe vera gel or petroleum jelly (e.g., Vaseline) to moisturize and soothe the area; cover loosely with a clean, non-stick bandage or gauze to protect the skin; take over-the-counter pain relievers such as ibuprofen or acetaminophen if needed for pain. Avoid butter, ice, toothpaste, oils, egg whites, or other unproven remedies, as they can worsen the injury, trap heat, or cause infection. These home measures are appropriate only for minor burns; seek immediate medical help for severe burns (including those with blisters, deep skin damage, large areas, or involving the face, hands, feet, genitals, or major joints), signs of infection (increased pain, redness, swelling, pus), or persistent pain.[106] [107] [108] [104] Chemical burns require immediate copius irrigation with water for at least 20 minutes after identifying and removing the agent if safe, while electrical burns necessitate ensuring the power source is off before contact to prevent rescuer injury.[28] Ointments, butter, or adhesive materials should not be applied, as they trap heat and promote infection; instead, burns are covered loosely with a clean, dry sheet, sterile non-stick dressing such as gauze or plastic wrap to protect from contamination while allowing air circulation.[102] A primary survey follows advanced trauma life support principles, assessing airway patency (with early intubation considered for suspected inhalation injury indicated by facial burns, singed nasal hairs, or carbonaceous sputum), breathing (supplemental oxygen via non-rebreather mask at 10-15 L/min for carbon monoxide risk), and circulation (monitoring for shock in burns >15-20% TBSA).[28] [96] Intravenous access is established with two large-bore lines in unburned areas, initiating isotonic crystalloid fluids (e.g., lactated Ringer's) at maintenance rates or per Parkland formula estimates (4 mL/kg/%TBSA in first 24 hours, half in first 8 hours) for major burns to counteract capillary leak and hypovolemic shock.[101] Analgesia with opioids like morphine (2-5 mg IV titrated) is administered for pain, and tetanus prophylaxis updated if needed.[100] Patients are kept normothermic using blankets or warm environments post-cooling, as heat loss through damaged skin exacerbates metabolic demands.[101] Secondary assessment estimates burn extent using the rule of nines or Lund-Browder chart for TBSA, evaluating depth (superficial, partial, full-thickness), and screening for associated trauma or comorbidities; medical attention should be sought promptly for burns covering an area larger than the palm of the hand, those with large blisters, white or charred skin, signs of infection, or in children and elderly individuals.[96] Triage directs patients with burns >10% TBSA, involving critical areas (face, hands, feet, genitals, joints), third-degree burns, inhalation injury, or comorbidities to verified burn centers via direct transport or helicopter if feasible, bypassing non-specialized facilities to optimize outcomes.[27] For critically injured burn patients arriving by medevac, receiving facilities prepare by assembling a full multidisciplinary team with personal protective equipment (PPE), activating the massive transfusion protocol, readying burn sheets and escharotomy kits, and placing the operating room on standby; the team assesses and manages the patient according to the exposure type (chemical, thermal, or electrical) while estimating fluid needs via the Parkland formula (4 mL × %TBSA × kg body weight over 24 hours, half in the first 8 hours).[96] Prehospital providers avoid debridement or escharotomy, deferring to hospital teams, and consult medical control for protocol deviations in resource-limited settings.[28] Evidence from EMS protocols underscores that timely adherence to these steps reduces mortality, with delays in fluid resuscitation correlating to higher complication rates in severe cases.[109]Fluid Resuscitation
Fluid resuscitation in burn management aims to counteract the profound hypovolemia resulting from increased vascular permeability and evaporative losses, which peak within the first 24-48 hours post-injury and can lead to shock if unaddressed.[110] In major burns, capillary leak causes plasma to shift into interstitial spaces, reducing intravascular volume by up to 30-50% in severe cases, necessitating aggressive replacement to maintain organ perfusion.[111] Resuscitation is indicated for adults with burns exceeding 20% total body surface area (TBSA) and children with burns over 10-15% TBSA, with earlier initiation improving outcomes by preventing renal failure and multi-organ dysfunction.[110][111] The Parkland formula, developed empirically in the 1960s based on observational data from over 300 patients, calculates initial crystalloid requirements as 4 mL of lactated Ringer's (LR) solution per kilogram of body weight per percentage of TBSA burned, administered over the first 24 hours from the time of injury; half is given in the initial 8 hours, and the remainder over the subsequent 16 hours.[112] This yields, for example, 8 liters for a 70 kg adult with 30% TBSA burn (4 × 70 × 30 = 8,400 mL total, adjusted for rounding).[112] Lactated Ringer's is preferred over normal saline due to lower hyperchloremic acidosis risk and better electrolyte matching to lost plasma.[110] Hypotonic solutions are generally avoided in the fluid resuscitation of burn patients because they can exacerbate edema by promoting excess fluid shift into the interstitial space and may provide unnecessary free water, potentially leading to hyponatremia or inadequate resuscitation. Isotonic crystalloids, particularly Lactated Ringer's solution, are the recommended standard for initial resuscitation to restore intravascular volume and maintain tissue perfusion without worsening edema.[110] However, the formula serves as a starting point rather than a rigid target, as prospective randomized trials validating its superiority over other methods are lacking, though retrospective analyses confirm it prevents under-resuscitation without inferiority to modern goal-directed approaches.[113] Recent American Burn Association (ABA) guidelines, informed by meta-analyses showing associations between excessive volumes and complications like abdominal compartment syndrome, recommend initiating at a lower rate of 2 mL/kg/%TBSA to minimize over-resuscitation, with titration based on clinical endpoints.[114] Adjustments are guided by urine output (target 0.5-1.0 mL/kg/hour in adults, 1.0-1.5 mL/kg/hour in children), mean arterial pressure above 65 mmHg, and normalization of base deficit or lactate levels, using invasive monitoring like arterial lines or central venous pressure in severe cases.[110][114] "Fluid creep"—unintended volume escalation from boluses or ongoing losses—occurs in up to 80% of cases and correlates with increased mortality, prolonged ventilation, and renal replacement therapy needs, prompting protocols emphasizing restrictive strategies after initial stabilization.[115][116] Colloids such as albumin are generally deferred until after 8-24 hours or if crystalloids alone fail to achieve endpoints, as early use does not reduce total volumes and may exacerbate edema in the permeable phase; hypertonic saline shows promise in select trials for reducing crystalloid needs by 20-30% but lacks broad endorsement due to inconsistent outcomes.[116] Inhalation injury may necessitate 30-50% additional fluids due to airway edema, though some protocols advocate caution to avoid overload.[117] Overall, successful resuscitation hinges on frequent reassessment, as static formulas overlook individual variability in burn depth, age, and comorbidities.[114]Wound Management
Burn wound management prioritizes the removal of necrotic tissue, control of microbial colonization, maintenance of a moist healing environment, and facilitation of re-epithelialization to minimize infection risk and optimize outcomes. Initial wound cleaning involves irrigation with sterile saline or chlorhexidine solution to eliminate debris, blisters, and loose eschar, typically performed within hours of injury to reduce bacterial load.[118] For partial-thickness burns, debridement of ruptured or intact blisters is standard practice to accurately assess depth, prevent fluid accumulation that harbors bacteria, and promote faster healing, with evidence indicating reduced infection rates compared to intact blister management.[119] However, burn blister management remains controversial, with evidence and guidelines varying on whether intact blisters should routinely be debrided. Some protocols, including those from certain burns services, support debridement of intact blisters to allow better wound assessment, dressing contact, and to avoid potential complications such as pressure on underlying tissue or infection foci.[119] In contrast, many authoritative first-aid and clinical guidelines recommend leaving intact blisters undisturbed wherever possible, as the blister serves as a natural protective barrier against infection, and deroofing or aspiration can increase infection risk in some cases.[120][104][121] Patients should not attempt to pop or drain burn blisters themselves, as this increases the risk of infection. If a blister is large, painful, tense, or requires drainage (e.g., to relieve pressure or for functional reasons), it should be managed by a healthcare professional (such as a doctor or in a hospital setting) using sterile techniques. Pharmacists can provide advice on dressings or over-the-counter treatments but do not perform procedures like draining or deroofing blisters. Debridement techniques vary by burn depth and patient condition; mechanical methods, such as wet-to-dry gauze dressings, physically remove devitalized tissue but cause significant pain and potential damage to viable dermis.[122] Enzymatic debridement, using agents like bromelain-based Nexobrid, offers a selective alternative, achieving eschar separation in 4 to 24 hours with lower blood loss and need for anesthesia than traditional surgical excision, particularly effective for indeterminate-depth burns up to 15% total body surface area (TBSA).[123] Autolytic debridement via occlusive dressings promotes natural enzymatic breakdown by endogenous proteases but progresses slowly and risks maceration in exuding wounds.[124] For full-thickness burns, early tangential excision within 48-72 hours followed by temporary coverage reduces sepsis incidence and mortality, as supported by randomized trials showing improved survival rates over conservative eschar management.[125] Topical antimicrobial therapy targets planktonic and biofilm bacteria while preserving host healing processes. Silver sulfadiazine (SSD) cream, introduced in 1968, provides broad-spectrum coverage against gram-positive, gram-negative, and some fungi, but prospective studies report delayed re-epithelialization due to sulfadiazine cytotoxicity to fibroblasts and keratinocytes, transient leukopenia in up to 5% of patients, and frequent dressing changes increasing pain and costs.[123] Mafenide acetate penetrates eschar effectively but inhibits carbonic anhydrase, risking metabolic acidosis, limiting its use to adjunctive roles.[123] Evidence from randomized controlled trials favors alternatives for partial-thickness burns: medical-grade honey dressings reduced healing time to 9-15 days versus 15-21 days with SSD, attributed to hyperosmolar antibacterial effects, hydrogen peroxide generation, and low pH without impairing epithelial migration.[126] Aloe vera cream similarly accelerated second-degree burn closure compared to SSD, with fewer hypertrophic scars in follow-up assessments.[127] Petrolatum gel alone matched or exceeded SSD in superficial partial-thickness burns by supporting moist healing without antibiotics, absent infection in low-risk cases, and lower expense.[128] Dressings are selected based on exudate level, infection risk, and burn depth to minimize adherence and trauma during changes. Nonadherent interfaces like silicone or petrolatum-impregnated gauze prevent eschar disruption in superficial burns, while silver-impregnated options such as nanocrystalline silver (Acticoat) sustain antimicrobial activity for 3-7 days, reducing colonization by 99% in vitro and bacterial burden in clinical use.[129] Hydrofiber or alginate dressings absorb moderate-to-high exudate, with meta-analyses confirming alginate shortens healing times by 1-2 days over traditional gauze in partial-thickness burns due to ion exchange promoting hemostasis and autolysis.[130] For temporary coverage in extensive burns awaiting autografting, biological dressings like porcine xenografts or allografts maintain physiologic moisture and reduce evaporative losses, though they carry rejection risks after 7-10 days.[125] Synthetic matrices, such as Integra, integrate with granulation tissue to support neodermis formation in deep wounds. Wound assessments occur daily or more frequently in hospitalized patients, evaluating for infection indicators including erythema extension, purulent discharge, odor, cellulitis, or quantitative cultures exceeding 10^5 CFU/g tissue, prompting escalation to systemic antibiotics or excision.[131] In outpatient minor burns (<10% TBSA), self-adherent wraps with topical ointments suffice, with tetanus prophylaxis if unimmunized and follow-up to detect delayed complications like contractures.[131] Multidisciplinary protocols emphasize pain control during procedures, using opioids or regional anesthesia, to ensure compliance and reduce stress-induced catabolism.[132]Infection Prevention and Treatment
Burn wounds are highly susceptible to infection due to the loss of the skin's protective barrier, allowing colonization by endogenous flora such as Pseudomonas aeruginosa and exogenous pathogens from the environment or healthcare settings.[133] Prevention begins with meticulous initial wound cleansing using sterile techniques and hydrotherapy to remove debris and necrotic tissue, reducing bacterial load.[134] Early excision of devitalized tissue, ideally within the first few days post-injury for deep partial- and full-thickness burns, significantly lowers infection rates by minimizing the necrotic substrate for microbial proliferation.[125] Topical antimicrobial agents play a central role in prophylaxis for larger burns. Silver sulfadiazine (SSD) has been widely used since the 1960s for its broad-spectrum activity against Gram-positive and Gram-negative bacteria, though randomized trials show limited superiority over simpler dressings in preventing infection and potential delays in re-epithelialization due to cytotoxicity.[135] Alternatives like polyhexanide-based solutions demonstrate reduced healing times and infection rates compared to SSD in partial-thickness burns (p < 0.001), while silver-impregnated dressings such as Acticoat provide sustained bactericidal effects without frequent reapplication.[135] Natural agents, including aloe vera, offer comparable antimicrobial benefits to 1% SSD with faster healing and less pain in second-degree burns, supported by clinical trials.[135] Routine prophylactic systemic antibiotics are discouraged outside the immediate perioperative period for excision or grafting, as they promote multidrug-resistant organisms, with evidence from burn units showing increased resistance in prolonged users.[133][136] Institutional infection control measures are essential, particularly for severe burns exceeding 40% total body surface area (TBSA). Contact precautions, including gloves, gowns, and masks, alongside single-patient rooms or cohort nursing, limit cross-contamination.[133] Surveillance involves routine quantitative wound biopsies or swabs for culture, guiding empirical therapy and tracking local resistance patterns via antibiograms, as recommended by burn societies to detect invasive infections early.[133] Minimizing invasive devices like central lines and ventilators, with prompt removal, further curbs nosocomial risks.[133] Treatment of established infections requires prompt diagnosis through clinical signs such as increased pain, erythema beyond burn margins, purulent discharge, foul odor, or systemic indicators like fever and leukocytosis, confirmed by tissue biopsy showing >10^5 bacteria per gram of tissue indicating invasion.[133] Burn wounds, particularly those involving blisters in partial-thickness burns, require vigilant monitoring for signs of infection. A normal blister in a superficial second-degree burn typically features red, swollen surrounding skin with a clear fluid-filled blister, pain, shiny moist appearance, and gradual healing over days to weeks.[137] Signs suggestive of infection include increased or worsening pain, spreading redness, swelling, or warmth beyond the original burn area, pus, foul-smelling or oozing discharge, fever, red streaks extending from the wound, or lack of improvement or worsening symptoms.[93][121] If these signs are observed, immediate medical attention should be sought to prevent progression to serious complications such as cellulitis or sepsis.[93] Necrotic tissue harboring infection must be surgically debrided, often enzymatically with agents like bromelain-based Nexobrid for selective eschar removal, reducing the need for extensive surgery and associated sepsis risk.[135] Systemic antibiotics are initiated empirically targeting likely pathogens (e.g., piperacillin-tazobactam for Pseudomonas coverage) but adjusted per culture sensitivities, with duration limited to clinical response to avoid resistance; in sepsis, adherence to Surviving Sepsis Campaign protocols adapted for burns emphasizes source control and hemodynamic support.[138][139] For multidrug-resistant cases, common in prolonged hospitalizations, unit-specific guidelines prioritize de-escalation and stewardship, as infections contribute to up to 20% sepsis incidence in adult burn patients with mortality rising with TBSA and age.[136][140]Pain and Supportive Care
Pain in burn injuries, particularly partial-thickness burns, arises from nociceptor activation, inflammation, and procedural interventions like debridement, often described as severe and multifaceted including background, breakthrough, and procedural components.[141] The American Burn Association (ABA) guidelines emphasize routine, protocolized pain assessment multiple times daily using validated patient-reported tools such as the Numerical Rating Scale, with the Critical-Care Pain Observation Tool for non-communicative patients, to guide individualized therapy and minimize undertreatment.[141] Opioids remain the cornerstone for moderate-to-severe acute burn pain, with choices like morphine or fentanyl selected based on pharmacokinetics, burn pathophysiology, and patient factors, administered via continuous infusion or patient-controlled analgesia while continuously titrating to response and minimizing doses through multimodal adjuncts.[141] Non-opioid pharmacotherapies include acetaminophen (up to 4 g/day monitored for hepatotoxicity), nonsteroidal anti-inflammatory drugs (NSAIDs) when renal and gastrointestinal risks permit, gabapentinoids for neuropathic components, low-dose ketamine for procedural analgesia, and alpha-2 agonists like dexmedetomidine for anxiety-associated pain exacerbation.[141] Regional anesthesia techniques, such as nerve blocks, reduce systemic opioid requirements and improve outcomes in select cases like graft harvesting.[141] Nonpharmacologic interventions complement pharmacotherapy, with strong evidence (Level A) supporting cognitive-behavioral therapy, hypnosis, and virtual reality distraction to mitigate procedural pain and anxiety.[141] Supportive measures include wound elevation, splinting to reduce contractures, and environmental controls like temperature regulation to alleviate discomfort. Nutritional support addresses the post-burn hypermetabolic response, characterized by elevated resting energy expenditure up to 1.5-2 times normal, requiring early enteral feeding within 24 hours of injury to preserve gut integrity and attenuate catabolism, targeting 25-40 kcal/kg/day initially adjusted via indirect calorimetry.[142] Protein provision of 1.5-2 g/kg/day, or 25% of total calories, promotes anabolism and wound healing, with supplementation of micronutrients like zinc, copper, and selenium to counter deficiencies from exudative losses.[143] High-carbohydrate (≥60% of calories), moderate-fat formulations minimize proteolysis compared to high-fat diets.[142] Psychological supportive care mitigates prevalent distress, including acute stress disorder in up to 25% of major burn survivors and chronic issues like PTSD affecting 20-30%, through multidisciplinary screening using tools like the Impact of Event Scale and interventions such as cognitive processing therapy or peer support groups.[144] Family involvement enhances coping and adherence, reducing isolation, while early pharmacotherapy for comorbid depression or anxiety (e.g., SSRIs) integrates with pain management to improve overall recovery.[145]Surgical Options
Surgical intervention is primarily indicated for deep partial-thickness, full-thickness, or fourth-degree burns that fail to heal spontaneously within 2-3 weeks, or for circumferential burns causing vascular or respiratory compromise.[122] Deep burns destroy dermal elements, necessitating excision of nonviable tissue to prevent infection, reduce sepsis risk, and enable epithelialization via grafting.[146] Early surgical excision, typically within 7 days post-injury, has been associated with reduced mortality, shorter hospital stays, and lower infection rates in severe burns exceeding 20% total body surface area (TBSA), though it increases intraoperative blood loss requiring transfusion.[147] Delayed excision beyond this window correlates with higher rates of wound infection and prolonged hospitalization.[148] Emergency procedures such as escharotomy are performed for circumferential full-thickness burns constricting underlying structures, particularly on extremities, trunk, or neck, where edema causes compartment syndrome or impaired ventilation.[149] This involves longitudinal incisions through the eschar into subcutaneous fat along tension lines, often bilaterally (e.g., medial and lateral on limbs), to restore distal perfusion and chest excursion; indications include absent distal pulses, pallor, or rising compartment pressures exceeding 30 mmHg.[150] Fasciotomy extends deeper into muscle fascia if compartment syndrome persists post-escharotomy, recommended prophylactically in high-risk cases like electrical burns.[151] These incisions are typically done at bedside under local anesthesia in unstable patients, with subsequent formal debridement in the operating room.[152] Definitive wound closure follows initial debridement via tangential excision, the preferred technique over fascial excision, which involves sequential removal of thin layers of necrotic tissue using dermatomes or scissors until punctate bleeding confirms a viable dermal or subcutaneous bed.[153] This method preserves maximal viable tissue for better functional and cosmetic outcomes compared to deeper fascial excision, which sacrifices more dermis and increases contracture risk.[154] Post-excision, temporary coverage with allografts, xenografts, or dermal substitutes bridges to autografting, especially in large TBSA burns where donor sites are limited.[155] Split-thickness skin autografts (STSG), harvested at 0.008-0.012 inches thick from unburned areas like thighs or back, provide permanent coverage and are meshed (e.g., 1:1.5 to 1:6 expansion ratios) to cover larger defects, promoting drainage and rapid integration via host angiogenesis.[122] Full-thickness grafts are reserved for smaller, visible areas (e.g., hands, face) due to higher metabolic demands and limited donor availability, yielding superior durability but requiring more precise vascularization.[155] Cultured epithelial autografts or bioengineered substitutes serve as adjuncts in extensive burns (>70% TBSA), though they exhibit higher fragility and contraction rates than traditional autografts.[156] Postoperative immobilization and nutrition optimization are critical to graft take rates, which average 80-95% in optimal conditions.[138] Reconstructive options, including local flaps, free tissue transfers, or tissue expansion, address late contractures or exposed structures (e.g., tendons, joints) not amenable to grafting alone, often staged over months to years post-initial healing.[122] In resource-limited settings, barriers to grafting include donor site morbidity and infection control, underscoring the need for multidisciplinary timing based on patient stability rather than rigid protocols.[157]Rehabilitation and Long-term Care
Rehabilitation for burn injuries initiates on the day of admission and persists for months to years, involving a coordinated multidisciplinary team including physical therapists, occupational therapists, psychologists, and physicians to address physical, functional, and psychosocial sequelae.[158] This approach aims to restore range of motion (ROM), prevent contractures, and promote functional independence, with early intervention critical to mitigating complications like muscle atrophy and joint stiffness.[159] No formal consensus guidelines exist for burn rehabilitation overall, though specific evidence-based practices guide components such as early mobilization.[159] In the acute phase, particularly for critically ill patients in intensive care units, the American Burn Association conditionally recommends early mobilization and rehabilitation, including in-bed exercises, resistance training, and progressive activities like sitting or standing, to reduce ICU-acquired weakness and delirium, based on available evidence from observational and randomized studies.[160] Anti-contracture positioning—such as maintaining the neck in extension and shoulders at 90-degree abduction—begins immediately, alongside custom splinting with thermoplastic materials to preserve joint alignment, typically worn continuously initially and tapered to nighttime use as ROM stabilizes.[158][159] Active and passive ROM exercises, conducted multiple times daily under analgesia, form the core of physical therapy to counteract tissue shortening, with supervised programs extending to strengthening and cardiovascular fitness post-discharge.[158] Evidence from 20 studies, including randomized controlled trials, supports prescribing resistance exercises at 50–85% of three-repetition maximum (3–12 reps, 3 sets, 3 days/week) and aerobic training (30 minutes at 70–85% VO2 peak, 3–5 days/week) for adults, yielding improvements in lean body mass, endurance, and quality of life over 6–12 weeks.[161] Occupational therapy focuses on activities of daily living (ADLs), hand function restoration—where full recovery occurs in 90% of superficial burns but only 10% of full-thickness hand injuries—and adaptive equipment to support independence.[159][162] Scar management employs pressure garments worn 23 hours daily for 1–1.5 years to reduce hypertrophic scarring (prevalent in 32–72% of survivors), combined with massage, moisturization, and silicone sheets, though efficacy varies and lacks uniform high-level evidence.[159][162] Pharmacologic aids like propranolol or oxandrolone may enhance outcomes in select cases, alongside gabapentin for pruritus or neuropathic pain.[159] Psychological rehabilitation addresses prevalent issues such as posttraumatic stress disorder and depression (affecting 23–61% long-term), through screening tools like the PHQ-9 and interventions including cognitive-behavioral therapy and peer support to facilitate social reintegration.[159][162] Long-term care emphasizes ongoing monitoring for chronic impairments, including joint contractures (38–54%), chronic pain (25–36%), and heat intolerance, with sun protection (SPF 30+) and vocational rehabilitation to counter reduced health-related quality of life persisting up to 20 years post-injury.[162] Approximately 28–33% of survivors never return to work, with median return times around 17 weeks influenced by burn severity, pre-injury employment, and barriers like pain or neurological deficits; functional independence in ADLs improves to supervision levels by discharge but often plateaus below baseline.[162] Multidisciplinary follow-up, including nutrition optimization and family involvement, supports sustained recovery, though protracted impairments remain common in major burns.[159][162]Controversies in Burn Care
Debates on Fluid Resuscitation Protocols
Fluid resuscitation in severe burns aims to restore intravascular volume lost due to capillary leak, but protocols remain contentious due to risks of under-resuscitation causing hypovolemic shock and organ failure versus over-resuscitation leading to edema, pulmonary complications, and abdominal compartment syndrome.[110] The Parkland formula, prescribing 4 mL/kg/% total body surface area (TBSA) burned of crystalloid (typically Ringer's lactate) over 24 hours with half in the first 8 hours post-injury, serves as the most common starting guideline since its proposal in 1968, yet it is not rigidly prescriptive and requires titration to endpoints like urine output of 0.5–1 mL/kg/hour.[110] [113] Critics argue it often underestimates needs in cases with inhalation injury or electrical burns, while empirical data show frequent over-administration, termed "fluid creep," exceeding formula predictions by 40–100% due to unaccounted insensible losses or protocol deviations.[113] [116] A central debate concerns fluid composition: exclusive crystalloid use versus adjunctive colloids like albumin. Proponents of colloids, citing burns-specific studies, contend they reduce total volumes needed by maintaining oncotic pressure and mitigating edema, with one review advocating hybrid regimens to curb fluid creep and improve outcomes like ventilator days.[163] [164] However, large trials such as the SAFE study (2006) and Cochrane meta-analysis (2018) in critically ill patients, including burns subsets, found no mortality benefit from colloids over crystalloids and potential harm from starches due to renal injury, leading guidelines to recommend delaying colloids until after 24 hours unless crystalloid volumes exceed Parkland predictions.[165] [166] In burns, a 2016 perspective notes unresolved cost-effectiveness and infection risks with colloids, though recent evidence supports early albumin in larger burns (>30% TBSA) to limit edema without increased adverse events.[163] [167] Restrictive strategies, aiming for lower volumes (e.g., 2–3 mL/kg/%TBSA) guided by advanced monitoring like lactate clearance or stroke volume variation, have gained traction to avoid over-resuscitation harms observed in up to 80% of cases, with retrospective data showing reduced complications like fasciotomy rates.[168] [169] Yet, a 2016 analysis warns that aggressive restriction risks under-resuscitation, as seen in cohorts with higher acute kidney injury when deviating below Parkland, emphasizing individualized titration over formulaic caps.[169] For inhalation injury, debates persist on supplemental fluids; a 2024 study found no consistent need for increased volumes beyond standard formulas, challenging older assumptions of higher requirements.[170] Emerging approaches, such as supplementing crystalloids with fresh frozen plasma, show promise in retrospective reviews for further volume reduction and endothelial stabilization, though prospective trials are lacking.[168] Overall, no consensus formula outperforms others universally, with evidence underscoring protocol flexibility, early goal-directed adjustments, and avoidance of both extremes to optimize survival, which exceeds 95% for burns <40% TBSA in modern centers when managed judiciously.[110] [171]Controversies in Topical Agents like Silver Sulfadiazine
Silver sulfadiazine (SSD), a sulfonamide-based cream with silver ions for broad-spectrum antibacterial activity, has been a staple in burn wound management since its introduction in the 1960s, yet multiple studies have highlighted its drawbacks, particularly in delaying epithelialization and promoting cytotoxicity to regenerating tissues. A 2008 Cochrane review concluded that SSD prolongs healing time in partial-thickness burns and necessitates more frequent dressing changes due to its pseudes char formation and poor eschar penetration, increasing patient discomfort and care burden. In a 2015 murine model of partial-thickness burns, topical SSD significantly retarded wound closure compared to untreated controls, with treated wounds showing expanded size by day 3 and reduced collagen deposition by day 7, attributed to disrupted cytokine signaling and suppressed macrophage recruitment.01210-4/fulltext) Human retrospective analyses corroborate this, reporting mean healing times extended by up to 9 days in SSD-treated partial-thickness burns versus alternatives like bacitracin.[172] Adverse effects further fuel debate, including transient leukopenia in up to 5% of patients, linked to the sulfadiazine component's bone marrow suppression, and rare but severe reactions such as methemoglobinemia or hemolytic anemia in susceptible individuals.[173] SSD's hyperosmolar nature can exacerbate pain during application and contribute to tissue desiccation, while silver deposition may cause argyria-like staining in healed skin, though systemic argyria remains uncommon with topical use.[174] Critics argue these toxicities stem from SSD's interference with fibroblast proliferation and keratinocyte migration, as evidenced by in vitro studies showing dose-dependent cytotoxicity to human keratinocytes at concentrations used clinically.[175] A 2019 comprehensive review recommended against SSD for burns, favoring nanocrystalline silver dressings or non-silver antimicrobials like octenidine, which exhibit less healing impairment while maintaining infection control.[176] Efficacy controversies center on SSD's limited superiority over simpler regimens; randomized trials demonstrate no significant reduction in infection rates compared to moist dressings alone in low-risk burns, yet it promotes bacterial resistance, particularly to Pseudomonas aeruginosa, due to subtherapeutic silver release and sulfadiazine selection pressure.[177] In pediatric first-degree burns, honey outperformed SSD in healing speed and reduced scarring, with SSD linked to higher pain scores and dressing adherence issues.[178] Despite persistent use in severe burns for its prophylactic role, guidelines from bodies like the American Burn Association increasingly advocate selective application, reserving SSD for colonized wounds while prioritizing early excision and grafting to mitigate healing delays.[123] This shift reflects growing evidence that SSD's antimicrobial benefits do not outweigh its inhibitory effects on innate repair mechanisms, prompting calls to reevaluate it as outdated standard care.01210-4/fulltext)Blister Debridement Practices
Blister debridement in burn care refers to the removal of blister roofs or aspiration of fluid from intact blisters in partial-thickness (second-degree) burns, aimed at facilitating wound assessment and reducing infection risk.[122] Practices vary, with ruptured blisters universally recommended for debridement to eliminate necrotic tissue that may harbor bacteria and to enable evaluation of the underlying wound bed, including capillary refill.[179] For intact blisters, controversy persists: deroofing involves sterile incision and removal of the epidermal roof, while aspiration uses a needle to drain fluid without disrupting the roof, preserving a potential barrier.[180] Debridement proponents argue it allows accurate burn depth assessment, application of topical antimicrobials, and pressure relief, potentially lowering infection rates by excising dead tissue early.[181] Evidence from clinical studies shows no significant difference in healing times between aspiration and deroofing, with mean complete wound healing at approximately 12 days for aspiration and 12.55 days for deroofing in one systematic review of partial-thickness burns.[180] Infection rates appear comparable across methods, though limited randomized controlled trials hinder definitive conclusions; smaller blisters often resolve spontaneously within 12 days without intervention, as epithelial regeneration outpaces fluid accumulation.[182] Leaving intact blisters intact provides pain relief and a natural eschar-like barrier, reducing exogenous contamination risks, particularly in superficial partial-thickness burns expected to heal in 7-21 days.[94] However, blister fluid may contain inflammatory mediators and bacteria from the burn injury itself, potentially promoting deeper infection if unaddressed in larger blisters exceeding the size of a patient's little fingernail.[183] Guidelines reflect this divide: the 2024 International Consensus on Second-Degree Burn Wounds recommends preserving intact blisters during prehospital care for their protective role, transitioning to debridement in clinical settings for monitoring.[184] The British Burn Association advocates debridement to enable wound bed visualization and depth reassessment after 48 hours under silver dressings.[181] In austere or resource-limited environments, breaking blisters for cleansing is advised to mitigate infection.[185] Overall, decisions hinge on blister size, location, and provider expertise, with small, intact blisters on non-critical areas often managed conservatively to avoid unnecessary exposure, while larger or ruptured ones warrant intervention to optimize outcomes.[186] High-quality evidence remains scarce, with calls for prospective trials to resolve whether routine debridement alters infection or scarring rates beyond anecdotal expert opinion.[187]Prevention Strategies
Individual and Home-Based Prevention
Lowering the temperature of residential water heaters to 120°F (49°C) significantly reduces the risk of scald burns from hot tap water, which account for approximately 17% of all burns globally and predominantly affect children under 5 years old.[1] At 140°F (60°C), water can cause third-degree burns in as little as 3 seconds, whereas at 120°F, it requires about 5 minutes of exposure, providing critical time for reaction.[188] Installing anti-scald devices on faucets and showerheads further mitigates this hazard by limiting output temperature, a measure endorsed for households with young children or elderly residents.[189] In kitchen settings, where scalds from hot liquids and fats comprise a leading cause of home burns—particularly among children aged 1 to 4—establishing a 3-foot "kid-free zone" around stoves and hot surfaces during cooking prevents unsupervised access.[190] Turning pot and pan handles inward and out of reach avoids pulls by children, while never leaving cooking unattended reduces ignition risks from unattended flames or heating elements.[191] Wearing short or fitted sleeves when cooking minimizes contact burns from splatters or steam, and keeping flammable materials like towels or curtains at least 3 feet from heat sources prevents flame spread.[192] Fire prevention in homes relies on functional smoke alarms installed on every level and outside sleeping areas, tested monthly, as they provide early warning for the 50% of fatal home fires occurring at night when occupants are asleep.[193] Extinguishing candles before leaving the room or sleeping, and maintaining a 12-inch clearance from combustible materials, addresses risks from open flames, which contribute to thousands of annual residential fires.[194] Having accessible, maintained fire extinguishers in kitchens and garages enables rapid response to small fires. Electrical burns, often from outlet tampering by toddlers or frayed cords, can be curtailed by using childproof outlet covers and securing cords out of reach, alongside avoiding extension cord overloads that lead to overheating.[195] For chemical burns, storing household cleaners, acids, and alkalis in locked cabinets above toddler height prevents ingestion or spills, as these substances cause about 10% of pediatric burns requiring medical attention.[1] Basic first-aid knowledge, such as cooling minor burns immediately with running cool water for 10-20 minutes without applying home remedies like butter or ice, empowers individuals to limit injury severity until professional care is sought if needed.[193] Parental education on these measures has demonstrated reductions in burn incidence, with community programs showing up to 30% decreases in targeted households through consistent reinforcement.[196]Occupational and Regulatory Measures
In the United States, the Occupational Safety and Health Administration (OSHA) mandates employers to develop and implement fire prevention plans under 29 CFR 1910.39, which include identifying fire hazards, establishing control procedures such as housekeeping to minimize ignition sources, and designating personnel responsible for maintenance of equipment like portable fire extinguishers.[197] For construction sites, OSHA's 29 CFR 1926.150 requires a comprehensive fire protection program covering temporary water supplies, fire extinguishers, and on-site fire watches to mitigate risks from welding, cutting, and open flames.[198] These measures address thermal, chemical, and electrical burns, which account for an estimated 150,000 annual emergency department visits for work-related burns, with 30-40% of hospitalized burn cases linked to occupational incidents.[199] The National Fire Protection Association (NFPA) complements OSHA through consensus standards like NFPA 70E, which specifies arc flash hazard assessments, personal protective equipment (PPE) such as flame-resistant clothing, and safe work practices to prevent electrical burns from shock, arc flash, and arc blast in industrial settings.[200] NFPA 30 regulates storage and handling of flammable liquids to reduce ignition risks, requiring spill control, ventilation, and bonding/grounding to prevent static-induced fires leading to burns.[201] Compliance with these standards has contributed to a decline in workplace fire-related injuries, though burns remain a significant cost, exceeding $2 billion annually in the U.S. due to medical treatment, lost productivity, and rehabilitation.[202] Internationally, the International Labour Organization (ILO) Convention No. 155 on Occupational Safety and Health requires employers to provide adequate PPE and training to prevent workplace hazards, including burns from heat, chemicals, or electricity, emphasizing risk assessments and worker consultation.[203] ISO 45001:2018 establishes a global framework for occupational health and safety management systems, mandating hazard identification, legal compliance, and continual improvement to minimize burn risks through engineering controls like machine guarding and emergency shutdowns.[204] In high-risk sectors such as manufacturing and construction, where thermal burns predominate, these regulatory measures prioritize causal factors like unguarded hot surfaces or inadequate ventilation, with enforcement varying by jurisdiction but supported by data showing reduced incidence where implemented rigorously.[205]Public Health Campaigns
Public health campaigns targeting burn prevention emphasize education on common hazards such as scalds, flames, and contact burns, often focusing on vulnerable populations like children and the elderly. In the United States, the American Burn Association's National Burn Awareness Week, observed annually during the first full week of February—such as February 2–8, 2025—promotes localized prevention strategies under themes like "Burn Prevention Starts Where You Live," advocating actions including setting water heaters below 120°F (49°C), maintaining a 3-foot clearance around heat sources, and storing flammables away from ignition points.[206] [207] These efforts collaborate with organizations like the National Fire Protection Association to distribute resources on smoke alarms, which have contributed to declining burn-related fatalities in high-income countries by providing early warnings in residential fires.[208] [1] Globally, the World Health Organization advocates multisectoral approaches in its 2008 "Plan for Burn Prevention and Care," which calls for awareness-raising, policy enforcement on safe hot water temperatures (below 48.9°C), and community programs to reduce incidence, particularly in low- and middle-income countries where burns cause an estimated 180,000 deaths yearly, mostly from flames and scalds.[209] [1] Partnering with the International Society for Burn Injuries, WHO initiatives prioritize evidence-based interventions like hot water tap regulations and safe cooking practices, which have lowered burn death rates in adopting regions through combined prevention and treatment improvements.[210] Evaluations of such campaigns show variable but positive impacts on injury rates. A nationwide multimedia effort in Mongolia reduced non-severe scald burns among young children by enhancing caregiver knowledge of tap water testing and supervision, with post-campaign hospital data indicating fewer admissions for minor injuries.[211] Community-based multi-strategy programs, including education and environmental modifications, have been associated with population-level reductions in pediatric burns and scalds, though randomized trials highlight greater gains in self-efficacy for prevention behaviors rather than uniform incidence drops.[196] [212] Earlier U.S. projects, like Project Burn Prevention, increased hazard awareness but yielded only moderate, temporary declines in burn severity, underscoring the need for sustained, multi-component efforts beyond awareness alone.[213]Prognosis and Complications
Prognostic Factors
Prognostic assessment in burn patients primarily relies on factors influencing mortality and long-term outcomes, with total body surface area (TBSA) burned, patient age, and presence of inhalation injury emerging as the most consistent predictors across studies.[214] [215] Larger TBSA percentages correlate directly with increased mortality risk, as extensive burns overwhelm physiological reserves, leading to higher rates of multiorgan failure; for instance, burns exceeding 40% TBSA are associated with mortality rates approaching 50% in adults without inhalation injury.[216] Advanced age exacerbates this, with patients over 60 years facing odds ratios for death up to 10-fold higher than younger cohorts due to reduced skin elasticity, diminished immune response, and comorbidities, independent of burn extent.[217] Inhalation injury, often from smoke exposure, independently elevates mortality by 20-30% through airway edema, carbon monoxide poisoning, and systemic inflammation, compounding TBSA effects.[216][217] Validated scoring systems integrate these factors for precise risk stratification. The revised Baux score, calculated as age plus TBSA percentage plus 17 points for confirmed inhalation injury, predicts mortality with area under the receiver operating characteristic curve (AUC) values exceeding 0.90 in multiple cohorts, outperforming the original Baux by accounting for inhalational effects; scores above 100 indicate over 50% mortality risk in adults.[218][219] Similarly, the Abbreviated Burn Severity Index (ABSI) incorporates age group, TBSA, inhalation injury, full-thickness burn presence, and sex, yielding AUCs of 0.85-0.95 for in-hospital mortality prediction, with higher scores (e.g., >6) signaling poor survival in severe cases.[220][221] These tools facilitate early triage and resource allocation, though their accuracy diminishes in pediatric or very elderly populations without adjustments.[218] Additional modifiers include burn depth, with full-thickness (third-degree) burns worsening outcomes via deeper tissue necrosis and infection risk, and early systemic inflammatory response syndrome (SIRS) on admission, which independently predicts death through unchecked cytokine storms.[216][222] Comorbidities such as diabetes or cardiovascular disease further impair recovery by hindering wound healing and increasing sepsis incidence, while prompt fluid resuscitation and mechanical ventilation needs reflect injury acuity and influence survival.[215] Gender shows minor effects, with males sometimes exhibiting higher mortality due to occupational exposure patterns, though not consistently across datasets.[215] Overall, these factors underscore causal links between initial injury burden and physiological decompensation, guiding evidence-based prognostication without overreliance on unverified variables.[214]Acute Complications
Burn shock represents a primary acute circulatory complication, characterized by hypovolemia from increased vascular permeability and fluid sequestration into interstitial spaces, compounded by inflammatory mediator release that exacerbates distributive shock elements. This leads to reduced cardiac output, tissue hypoperfusion, and potential progression to multiorgan dysfunction if resuscitation is inadequate, with risks heightened in burns involving greater than 20% total body surface area (TBSA).[15][78] Fluid resuscitation protocols, such as the Parkland formula, aim to mitigate this by replacing estimated losses, but over-resuscitation can induce edema-related issues like abdominal compartment syndrome.[110] Respiratory complications arise acutely, especially with inhalation injury from smoke or hot gases, causing upper airway edema, bronchospasm, and carbon monoxide or cyanide poisoning that impairs oxygenation. In severe cases, this evolves into acute respiratory distress syndrome (ARDS), marked by hypoxemia and bilateral infiltrates, with inhalation injury independently increasing ARDS risk by up to fourfold in burn patients requiring intensive care.[78][223] Early intubation may be necessary to secure airways before swelling compromises ventilation, particularly in burns near the face or in enclosed fires.[12] Infection emerges as a critical acute threat due to the loss of skin barrier function, with partial-thickness burns becoming colonized by bacteria within hours and progressing to sepsis if not managed with topical antimicrobials and debridement. Common pathogens include Pseudomonas aeruginosa and Staphylococcus aureus, contributing to systemic inflammatory response syndrome (SIRS) and high mortality rates in the first week post-injury, especially without prophylactic measures.[15][224] Acute kidney injury can follow from hypovolemia, myoglobin release in deep burns, or nephrotoxic agents, occurring immediately or within days and necessitating renal replacement therapy in up to 30% of major burn cases.[225] Other acute issues include compartment syndrome from circumferential eschar restricting blood flow, requiring urgent escharotomy, and hypermetabolic states triggering early catabolism, though these interface with subacute phases.[14] Prompt recognition and intervention in specialized burn units reduce these risks, as delays amplify mortality from combined hypovolemic, septic, and respiratory insults.[75]Long-term Complications
Severe burns often result in hypertrophic scarring and contractures, which can cause significant functional limitations and require ongoing surgical interventions such as scar release or grafting.[224] These scars form due to excessive collagen deposition during wound healing, leading to joint stiffness and reduced range of motion, particularly in areas like the neck, axillae, and hands, affecting up to 60-80% of survivors with burns exceeding 20% total body surface area (TBSA).[226] Contractures develop when scar tissue shortens, pulling skin and underlying structures, and prevalence increases with burn depth and delayed treatment, necessitating physical therapy or reconstructive surgery in many cases.[227] Chronic pain persists in approximately 30-50% of burn survivors beyond one year post-injury, stemming from neuropathic damage, ongoing inflammation, and scar maturation processes that sensitize nociceptors.[228] This pain manifests as burning, itching (pruritus), or hypersensitivity, impairing daily activities and sleep, with itch affecting up to 80% initially but resolving slower in deeper burns.[228] Sensory neuropathy and loss of protective sensation in healed areas heighten reinjury risk, while muscle weakness and fatigue contribute to long-term physical debility, reported in cohort studies of major burn patients followed for over a decade.[227] Psychological sequelae include post-traumatic stress disorder (PTSD) in up to 45% of survivors, depression in up to 54% with mild symptoms, and anxiety disorders, exacerbated by visible scarring, pain, and social reintegration challenges.[229] These conditions arise from the trauma of injury, prolonged hospitalization, and body image alterations, with risk factors including pre-existing mental health issues and burn visibility; longitudinal data show elevated suicide ideation rates persisting years later.[230] Quality of life metrics, such as SF-36 scores, remain below population norms, though some stabilization occurs after initial recovery phases.[227] Systemically, burn survivors exhibit elevated long-term mortality, with life expectancy reduced by factors linked to burn size, inhalation injury, and age; Danish registry data indicate 1.6- to 3-fold higher all-cause mortality up to 10 years post-burn compared to matched controls.[231] Increased risks of cardiovascular disease, diabetes, musculoskeletal disorders, and certain cancers (e.g., skin malignancies in chronic wounds) stem from hypermetabolic responses, immune dysregulation, and scarring-induced immobility, as evidenced in population-based studies.[232] Sepsis and infection susceptibility persist for at least five years, driven by impaired skin barrier function.[233]Epidemiology
Global Incidence and Mortality
Burns affect an estimated 8 to 11 million people globally each year, with incident cases reported at approximately 6.19 million thermal burns in 2021 according to Global Burden of Disease (GBD) analyses.[234] [235] These figures encompass injuries from fire, heat, and hot substances, though exact numbers vary due to underreporting in low-resource settings and differences in classification between sources like the World Health Organization (WHO) and GBD studies.[1] Incidence rates are highest in low- and middle-income countries (LMICs), where inadequate infrastructure and household practices contribute to elevated risks, contrasting with declining rates in high-income regions.[1] Mortality from burns stands at around 120,000 to 180,000 deaths annually, with WHO estimates at the higher end reflecting broader inclusion of non-hospitalized fatalities prevalent in LMICs.[1] [236] GBD data for 2021 indicate about 111,000 deaths, a decrease from prior decades, driven by improved prevention and care in select areas but offset by persistent challenges in regions like sub-Saharan Africa and Southeast Asia.[235] Children under five years and adult females bear disproportionate mortality burdens, often linked to open fires for cooking and scalds in domestic settings without safety measures.[1]| Metric | Global Estimate (Recent Data) | Primary Source |
|---|---|---|
| Annual Incident Cases | 6.19 million (2021) | GBD via Frontiers[234] |
| Annual Deaths | 111,000–180,000 | GBD/WHO[235] [1] |
| Age-Standardized Death Rate | Declining ~1–2% annually since 1990 | GBD trends[234] |
Demographic Patterns
Burn injuries exhibit distinct demographic patterns influenced by age, sex, and socioeconomic factors. Globally, incidence rates are bimodal, peaking in children under 5 years—particularly in low- and middle-income countries where open fires and scalds predominate—and in working-age adults aged 20–40 years, often linked to occupational hazards or high-risk behaviors.[1][7] Mortality from burns is highest among children aged 1–4 years, accounting for a disproportionate share of the 111,292 deaths reported in 2019, with 95% uncertainty intervals of 88,188–132,392.[7] Elderly individuals over 65 face elevated vulnerability due to reduced mobility, comorbidities, and thinner skin, contributing to higher complication rates and fatalities; in the United States from 1999–2020, 96,498 older adults died from burns, with mortality rising since 2012.[238] Sex disparities show males comprising the majority of burn cases, with U.S. hospital admissions at 66% male versus 34% female, reflecting greater male exposure to occupational fires, explosions, and recreational risks.[33] However, females experience slightly higher global death rates from burns, potentially due to domestic cooking accidents in resource-poor settings and higher suicide-related burns in certain regions.[1] In severe burns exceeding 20% total body surface area, women demonstrate increased mortality risk despite lower overall incidence, attributed to physiological differences in immune response and hormone influences on healing.[239] Racial and ethnic patterns reveal inequities, particularly in high-income contexts like the U.S., where Black individuals over 65 endure the highest age-adjusted burn mortality rates among all groups, with Black children aged 1–9 facing rates 2.7 times higher than white children.[238][240] Among U.S. burn admissions, racial distribution includes 64% white, 23% Black or African American, and 9% multiple races, with 16% Hispanic ethnicity; these disparities persist after adjusting for injury severity, linked to socioeconomic barriers, housing conditions, and access to preventive measures.[33] Low socioeconomic status amplifies risks across demographics, as evidenced by correlations between poverty indicators from U.S. Census data and higher burn hospitalization rates.[241]Recent Trends and Disparities
Global incidence of burn injuries has declined in terms of age-standardized rates since 1990, with the Global Burden of Disease Study estimating 15.35 million total cases (95% UI 13.17–17.51 million) in 2021, including 8.55 million in males and 6.80 million in females.[242] Severe burn prevalence stood at 12.99 million cases (95% UI 11.96–14.29 million) that year, reflecting improvements in prevention and care in many regions, though absolute numbers remain substantial.[236] Mortality, however, persists at around 180,000 deaths annually, predominantly in low- and middle-income countries where over 90% of fatalities occur due to limited access to timely treatment.[1] Projections indicate a 6.42% rise in global burn cases by 2050, concentrated in Latin America and other developing regions amid urbanization and climate-related risks.[243] In the United States, burn-related hospital admissions totaled 29,165 in recent national data, with 795 inpatient deaths yielding a 2.7% mortality rate; of these, 52.4% involved inhalation injury.[33] Elderly patients (aged 65+) show rising mortality trends, with CDC data from 2000–2020 highlighting increased fire- and flame-related deaths in this group, exacerbated by comorbidities and reduced mobility.[238] Rural areas exhibit higher unintentional fire or flame death rates than urban ones—for instance, 2.3 per 100,000 in rural versus 1.1 in urban for females in 2020—linked to delayed emergency response and heating practices.[244] Demographic disparities amplify burn burdens. Globally, females sustain more severe burns than males, often from domestic sources like cooking fires, while males predominate in occupational and high-risk exposures.[245] [246] Racial and ethnic gaps in outcomes are evident: Black patients face elevated mortality risks independent of insurance, and both Black and Hispanic survivors experience inferior wound healing, community reintegration, and adjustment compared to whites.[247] [248] Socioeconomic marginalization correlates with overrepresentation in burn cases and poorer care quality, including longer lengths of stay and higher complication rates, though poverty alone does not uniformly elevate incidence risk.[249] [250]| Demographic Factor | Key Disparity Observation | Source |
|---|---|---|
| Gender | Females: Higher severity and home-based incidence; Males: Occupational predominance | [245] [246] |
| Race/Ethnicity | Black/Hispanic: Increased mortality, poorer healing vs. White | [247] [248] |
| Socioeconomic Status | Low SES: Worse outcomes, care access; Overrepresentation in injuries | [249] [250] |
| Geography (Rural/Urban) | Rural: 2x higher fire death rates | [244] |