Recent from talks
Nothing was collected or created yet.
Lunar resources
View on Wikipedia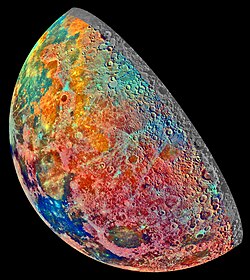

The Moon bears substantial natural resources which could be exploited in the future.[1][2] Potential lunar resources may encompass processable materials such as volatiles and minerals, along with geologic structures such as lava tubes that, together, might enable lunar habitation. The in-situ use (ISRU) of resources on the Moon may provide a means of reducing the cost and risk of lunar exploration and beyond.[3][4]
Resource mapping and sample-return missions have enhanced the understanding of the potential for lunar ISRU. An assessment in 2019 concluded that knowledge was not yet sufficient to justify the commitment of large financial resources to implement an ISRU-based campaign.[5] The determination of resource availability will drive the selection of sites for human settlement.[6][7]
Overview
[edit]Lunar materials could facilitate continued exploration of the Moon, facilitate scientific and economic activity in the vicinity of both Earth and Moon (so-called cislunar space), or they could be imported to the Earth's surface where they would contribute directly to the global economy.[1] Regolith (lunar soil) is the easiest product to obtain; it can provide radiation and micrometeoroid protection as well as construction and paving material by melting.[8] Oxygen from lunar regolith oxides can be a source for metabolic oxygen and rocket propellant oxidizer. Water ice can provide water for radiation shielding, life-support, oxygen and rocket propellant feedstock. Volatiles from permanently shadowed craters may provide methane (CH
4), ammonia (NH
3), carbon dioxide (CO
2) and carbon monoxide (CO).[9] Metals and other elements for local industry may be obtained from the various minerals found in regolith.
The Moon is known to be poor in carbon and nitrogen, and rich in metals and in atomic oxygen, but their distribution and concentrations are still unknown. Further lunar exploration will reveal additional concentrations of economically useful materials, and whether or not these will be economically exploitable will depend on the value placed on them and on the energy and infrastructure available to support their extraction.[10] For in situ resource utilization (ISRU) to be applied successfully on the Moon, landing site selection is imperative, as well as identifying suitable surface operations and technologies.
Scouting from lunar orbit by a few space agencies is ongoing, and landers and rovers are scouting resources and concentrations in situ (see: List of missions to the Moon).
Resources
[edit]| Compound | Formula | Composition | |
|---|---|---|---|
| Maria | Highlands | ||
| silica | SiO2 | 45.4% | 45.5% |
| alumina | Al2O3 | 14.9% | 24.0% |
| lime | CaO | 11.8% | 15.9% |
| iron(II) oxide | FeO | 14.1% | 5.9% |
| magnesia | MgO | 9.2% | 7.5% |
| titanium dioxide | TiO2 | 3.9% | 0.6% |
| sodium oxide | Na2O | 0.6% | 0.61% |
| 99.9% | 100.0% | ||
Solar power, oxygen, and metals are abundant resources on the Moon.[12] Elements known to be present on the lunar surface include, among others, hydrogen (H),[1][13] oxygen (O), silicon (Si), iron (Fe), magnesium (Mg), calcium (Ca), aluminium (Al), manganese (Mn) and titanium (Ti). Among the more abundant are oxygen, iron and silicon. The atomic oxygen content in the regolith is estimated at 45% by weight.[14][15]
Studies from Apollo 17's Lunar Atmospheric Composition Experiment (LACE) show that the lunar exosphere contains trace amounts of hydrogen (H2), helium (He), argon (Ar), and possibly ammonia (NH3), carbon dioxide (CO2), and methane (CH4). Several processes can explain the presence of trace gases on the Moon: high energy photons or solar winds reacting with materials on the lunar surface, evaporation of lunar regolith, material deposits from comets and meteoroids, and out-gassing from inside the Moon. However, these are trace gases in very low concentration.[16] The total mass of the Moon's exosphere is roughly 25,000 kilograms (55,000 lb) with a surface pressure of 3×10−15 bar (2×10−12 torr).[17] Trace gas amounts are unlikely to be useful for in situ resource utilization.
Solar power
[edit]Daylight on the Moon lasts approximately two weeks, followed by approximately two weeks of night, while both lunar poles are illuminated almost constantly.[18][19][20] The lunar south pole features a region with crater rims exposed to near constant solar illumination, yet the interior of the craters are permanently shaded from sunlight.
Solar cells could be fabricated directly on the lunar soil by a medium-size (~200 kg) rover with the capabilities for heating the regolith, evaporation of the appropriate semiconductor materials for the solar cell structure directly on the regolith substrate, and deposition of metallic contacts and interconnects to finish off a complete solar cell array directly on the ground.[21] This process however requires the importation of potassium fluoride from Earth to purify the necessary materials from regolith.[22]
Nuclear power
[edit]The Kilopower nuclear fission system is being developed for reliable electric power generation that could enable long-duration crewed bases on the Moon, Mars and destinations beyond.[23][24] This system is ideal for locations on the Moon and Mars where power generation from sunlight is intermittent.[24][25] Uranium and thorium are both present on the Moon, but due to the high energy density of nuclear fuels, it could be more economical to import suitable fuels from Earth rather than producing them in situ.
Radioisotope thermoelectric generators (RTGs) are another form of nuclear power which use the natural decay of radioisotopes rather than their induced fission. They have been used in space—including on the Moon—for decades. The usual process is to source the suitable substances from Earth, but plutonium-238 or strontium-90 could be produced on the Moon if feedstocks such as spent nuclear fuel are present (either delivered from Earth for processing or produced by local fission reactors). RTGs could be used to deliver power independent of available sunlight, for both lunar and non-lunar applications. RTGs do contain harmful toxic and radioactive materials, which leads to concerns of unintentional distribution of those materials in the event of an accident. Protests by the general public therefore often focus on the phaseout of RTGs (instead recommending alternative power sources), due to an overestimation of the dangers of radiation.
A more theoretical lunar resource are potential fuels for nuclear fusion. Helium-3 has received particular media attention as its abundance in lunar regolith is higher than on Earth. However, thus far nuclear fusion has not been employed by humans in a controlled fashion releasing net usable energy (devices like the fusor are net energy consumers while the hydrogen bomb is not a controlled fusion reaction). Furthermore, while helium-3 is required for one possible pathway of nuclear fusion, others instead rely on nuclides which are more easily obtained on Earth, such as tritium, lithium or deuterium.
Oxygen
[edit]The elemental oxygen content in the regolith is estimated at 45% by weight.[15][14] Oxygen is often found in iron-rich lunar minerals and glasses as iron oxide. Such lunar minerals and glass include ilmenite, olivine, pyroxene, impact glass, and volcanic glass.[26] Various isotopes of oxygen are present on the Moon in the form of 16O, 17O, and 18O.[27]
At least twenty different possible processes for extracting oxygen from lunar regolith have been described,[28][29] and all require high energy input: between 2–4 megawatt-years of energy (i.e. (6–12)×1013 J) to produce 1,000 tons of oxygen.[1] While oxygen extraction from metal oxides also produces useful metals, using water as a feedstock does not.[1] One possible method of producing oxygen from lunar soil requires two steps. The first step involves the reduction of iron oxide with hydrogen gas (H2) to form elemental iron (Fe) and water (H2O).[26] Water can then be electrolyzed to produce oxygen which can be liquified at low temperatures and stored. The amount of oxygen released depends on the iron oxide abundance in lunar minerals and glass. Oxygen production from lunar soil is a relatively fast process, occurring in a few tens of minutes. In contrast, oxygen extraction from lunar glass requires several hours.[26]
Human oxygen consumption depends on physical activity and is affected by diet and also gravity. A commonly assumed round number for CO2-production of humans of low to moderate physical activity assumes 2.2 kilograms (4.9 lb) CO2 being exhaled per person per day. In the microgravity-environment of the International Space Station this value can be as low as 1 kilogram (2.2 lb) per person per day.[30] If one conservatively assumes that one mole of oxygen is consumed per mole of carbon dioxide produced (this ratio holds true for glucose but less oxygen is consumed per unit of carbon dioxide produced if fat or protein are the source of metabolic energy) 2.2 kg of carbon dioxide produced are equivalent to 1.6 kilograms (3.5 lb) of oxygen consumed. The yearly oxygen need of a human would thus be roughly 584 kilograms (1,287 lb) and per the above-mentioned energy requirements about 1.3-2.6 kilowatts would be constantly required per person to produce this amount of oxygen from lunar rocks. For comparison the average per person electricity consumption in the US in 2022 was 12,809 kWh (46,110 MJ) or about 1,462 Watts.
Water
[edit]
Cumulative evidence from several orbiters strongly indicate that water ice is present on the surface at the Moon poles, but mostly on the south pole region.[31][32] However, results from these datasets are not always correlated.[33][34] It has been determined that the cumulative area of permanently shadowed lunar surface is 13,361 km2 in the northern hemisphere and 17,698 km2 in the southern hemisphere, giving a total area of 31,059 km2.[1] The extent to which any or all of these permanently shadowed areas contain water ice and other volatiles is not currently known, so more data is needed about lunar ice deposits, its distribution, concentration, quantity, disposition, depth, geotechnical properties and any other characteristics necessary to design and develop extraction and processing systems.[34][35] The intentional impact of the LCROSS orbiter into the crater Cabeus was monitored to analyze the resulting debris plume, and it was concluded that the water ice must be in the form of small (< ~10 cm), discrete pieces of ice distributed throughout the regolith, or as a thin coating on ice grains.[36] This, coupled with monostatic radar observations, suggest that the water ice present in the permanently shadowed regions of lunar polar craters is unlikely to be present in the form of thick, pure ice deposits.[36]
Water may have been delivered to the Moon over geological timescales by the regular bombardment of water-bearing comets, asteroids and meteoroids[37] or continuously produced in situ by the hydrogen ions (protons) of the solar wind impacting oxygen-bearing minerals.[1][38]
The lunar south pole features a region with crater rims exposed to near constant solar illumination, where the craters' interior are permanently shaded from sunlight, allowing for natural trapping and collection of water ice that could be mined in the future.
Water molecules (H
2O) can be broken down to form molecular hydrogen (H
2) and molecular oxygen (O
2) to be used as rocket bi-propellant or produce compounds for metallurgic and chemical production processes.[3] Just the production of propellant, was estimated by a joint panel of industry, government and academic experts, identified a near-term annual demand of 450 metric tons of lunar-derived propellant equating to 2,450 metric tons of processed lunar water, generating US$2.4 billion of revenue annually.[25]
Hydrogen
[edit]Slopes on the lunar surface that face the Moon's poles show a higher concentration of hydrogen. This is because pole facing slopes have less exposure to sunlight that will cause vaporization of hydrogen. Additionally, slopes closer to the Moon's poles show a higher concentration of hydrogen of about 45 ppmw. There are various theories to explain the presence of hydrogen on the Moon. Water, which contains hydrogen, could have been deposited on the Moon by comets and asteroids. Additionally, solar winds interacting with compounds on the lunar surface may have led to the formation of hydrogen-bearing compounds such as hydroxyl and water.[39] The solar wind implants protons on the regolith, forming a protonated atom, which is a chemical compound of hydrogen (H). Although bound hydrogen is plentiful, questions remain about how much of it diffuses into the subsurface, escapes into space or diffuses into cold traps.[40] Hydrogen would be needed for propellant production, and it has a multitude of industrial uses. For example, hydrogen can be used for the production of oxygen by hydrogen reduction of ilmenite.[41][42][43]
Metals
[edit]Iron
[edit]| Mineral | Elements | Lunar rock appearance |
|---|---|---|
| Plagioclase feldspar | Calcium (Ca) Aluminium (Al) Silicon (Si) Oxygen (O) |
White to transparent gray; usually as elongated grains. |
| Pyroxene | Iron (Fe), Magnesium (Mg) Calcium (Ca) Silicon (Si) Oxygen (O) |
Maroon to black; the grains appear more elongated in the maria and more square in the highlands. |
| Olivine | Iron (Fe) Magnesium (Mg) Silicon (Si) Oxygen (O) |
Greenish color; generally, it appears in a rounded shape. |
| Ilmenite | Iron (Fe), Titanium (Ti) Oxygen (O) |
Black, elongated square crystals. |
Iron (Fe) is abundant in all mare basalts (~14–17% per weight) but is mostly locked into silicate minerals (i.e. pyroxene and olivine) and into the oxide mineral ilmenite in the lowlands.[1][45] Extraction would be quite energy-demanding, but some prominent lunar magnetic anomalies are suspected as being due to surviving Fe-rich meteoritic debris. Only further exploration in situ will determine whether or not this interpretation is correct, and how exploitable such meteoritic debris may be.[1] Hematite, a mineral composed of ferric oxide (Fe2O3), has been found on the Moon. This mineral is a product of a reaction between iron, oxygen, and liquid water. Oxygen from the Earth's atmosphere may cause this reaction as indicated by there being more hematite on the side of the Moon facing the Earth.[46]
Free iron also exists in the regolith (0.5% by weight) naturally alloyed with nickel and cobalt and it can easily be extracted by simple magnets after grinding.[45] This iron dust can be processed to make parts using powder metallurgy techniques,[45] such as additive manufacturing, 3D printing, selective laser sintering (SLS), selective laser melting (SLM), and electron beam melting (EBM).
Titanium
[edit]Titanium (Ti) can be alloyed with iron, aluminium, vanadium, and molybdenum, among other elements, to produce strong, lightweight alloys for aerospace use. It exists almost exclusively in the mineral ilmenite (FeTiO3) in the range of 5–8% by weight.[1] Ilmenite is the main source of titanium on earth and processes like the chloride process to extract titanium from ilmenite are well established at industrial scale. Ilmenite minerals on the moon also trap hydrogen (protons) from the solar wind, so that processing of ilmenite will also produce hydrogen, a valuable element on the Moon.[45] The vast flood basalts on the northwest nearside (Mare Tranquillitatis) possess some of the highest titanium contents on the Moon,[34] with 10 times as much titanium as rocks on Earth.[47]
Aluminum
[edit]Aluminum (Al) is found with a concentration in the range of 10–18% by weight, present in the mineral anorthite (CaAl
2Si
2O
8),[45] the calcium endmember of the plagioclase feldspar mineral series.[1] Aluminum is a good electrical conductor, and atomized aluminum powder also makes a good solid rocket fuel when burned with oxygen.[45] Extraction of aluminum would also require breaking down plagioclase (CaAl2Si2O8).[1] Extraction of aluminum would require large amounts of electricity, even more than on earth, because the aluminum mineral bauxite which is the basis of most aluminum production on earth does not exist on the moon as it is formed by chemical weathering involving liquid water.
Silicon
[edit]
Silicon (Si) is an abundant metalloid in all lunar material, with a concentration of about 20% by weight. It is of enormous importance to produce solar panel arrays for the conversion of sunlight into electricity, as well as glass, fiber glass, and a variety of useful ceramics. Achieving a very high purity for use as semi-conductor would be challenging, especially in the lunar environment.[1] Converting silica into silicon is an energy-intensive process. On earth this is usually done via carbothermic reduction, a process that requires carbon, an element in comparatively short supply on the moon.
Calcium
[edit]
Calcium (Ca) is the fourth most abundant element in the lunar highlands, present in anorthite minerals (formula CaAl
2Si
2O
8).[45][48] Calcium oxides and calcium silicates are not only useful for ceramics, but pure calcium metal is flexible and an excellent electrical conductor in the absence of oxygen.[45] Anorthite is rare on the Earth[49] but abundant on the Moon.[45]
Calcium can also be used to fabricate silicon-based solar cells, requiring lunar silicon, iron, titanium oxide, calcium and aluminum.[50]
When combined with water, lime (calcium oxide) produces significant amounts of heat. Hydrated lime (calcium hydroxide) meanwhile absorbs carbon dioxide which can be used as a (non-replenishing) filter. The resulting material, calcium carbonate is commonly used as a building material on earth.
Magnesium
[edit]Magnesium (Mg) is present in magmas and in the lunar minerals pyroxene and olivine,[51] so it is suspected that magnesium is more abundant in the lower lunar crust.[52] Magnesium has multiple uses as alloys for aerospace, automotive and electronics. On earth aluminum alloys are used particularly where low weight and high strength are needed, examples include airplane fuselages, high speed train carriages and the likes. Magnesium extraction from lunar rocks likewise requires large amounts of energy.
Thorium
[edit]The Compton–Belkovich Thorium Anomaly is a volcanic complex on the far side of the Moon.[53] It was found by a gamma-ray spectrometer in 1998 and is an area of concentrated thorium, a 'fertile' element.[53][54] Likewise KREEP has areas with several ppm of Thorium in the rock.
Rare-earth elements
[edit]Rare-earth elements are used to manufacture everything from electric or hybrid vehicles, wind turbines, electronic devices and clean energy technologies.[55][56] Despite their name, rare-earth elements are – with the exception of promethium – relatively plentiful in Earth's crust. However, because of their geochemical properties, rare-earth elements are typically dispersed and not often found concentrated in rare-earth minerals; as a result, economically exploitable ore deposits are less common.[57] Major reserves exist in China, California, India, Brazil, Australia, South Africa, and Malaysia,[58] but China accounts for over 95% of the world's production of rare-earths.[59] (See: Rare earth industry in China.)
Although current evidence suggests rare-earth elements are less abundant on the Moon than on Earth,[60] NASA views the mining of rare-earth minerals as a viable lunar resource[61] because they exhibit a wide range of industrially important optical, electrical, magnetic and catalytic properties.[1] KREEP are parts of the lunar surface richer in potassium (the "K" stands for the element symbol) rare earth elements and Phosphorus. Potassium and phosphorus are two of the three essential plant nutrients, the third being fixed nitrogen (hence NPK fertilizer) any agricultural activity on the moon would need a supply of those elements — whether sourced in situ or brought from elsewhere e.g. earth.
Helium-3
[edit]The solar wind has deposited more than 1 million tons of helium-3 (3He) on the Moon's surface.[62] Materials on the Moon's surface contain helium-3 at concentrations estimated between 1.4 and 15 parts per billion (ppb) in sunlit areas,[1][63][64] and may contain concentrations as much as 50 ppb in permanently shadowed regions.[65] For comparison, helium-3 in the Earth's atmosphere occurs at 7.2 parts per trillion (ppt).
Since 1986[66] proposals to exploit the lunar regolith and use the helium-3 for nuclear fusion have been presented.[61] Although as of 2020, functioning experimental nuclear fusion reactors have existed for decades[67][68] – none of them has yet provided electricity commercially.[69][70] Because of the low concentrations of helium-3, any mining equipment would need to process large amounts of regolith. Over 150 tons of regolith must be processed to obtain 1 gram (0.035 oz) of helium 3.[71] China has begun the Chinese Lunar Exploration Program for exploring the Moon and is investigating the prospect of lunar mining, specifically looking for the isotope helium-3 for use as an energy source on Earth.[72] Not all authors think the extraterrestrial extraction of helium-3 is feasible,[69] and even if it was possible to extract helium-3 from the Moon, no useful fusion power reactor has produced more energy output than the electrical energy input.[69][70] However, on 13 December 2022, the United States Department of Energy announced that the National Ignition Facility "conducted the first controlled fusion experiment in history to reach this milestone, also known as scientific energy breakeven, meaning it produced more energy from fusion than the laser energy used to drive it."[73] The downside remains that Helium-3 is a limited lunar resource that can be exhausted once mined.[10]
Carbon and nitrogen
[edit]Carbon (C) would be required for the production of lunar steel, but it is present in lunar regolith in trace amounts (82 ppm[74]), contributed by the solar wind and micrometeorite impacts.[75] Due to extremely low temperatures, permanently shadowed regions of the Moon's poles have cold traps which possibly contain solid carbon dioxide.[76] The presence of carbon is mostly due to solar wind carbon implanted in bulk regolith. Carbon is present in carbon-bearing ices at the lunar poles in concentrations as high as 20% by weight. However, most carbon-bearing ices have a 0–3% by weight carbon concentration. Carbon-bearing compounds that could exist include carbon monoxide (CO), ethylene (C2H4), carbon dioxide (CO2), methanol (CH3OH), methane (CH4), carbonyl sulfide (OCS), hydrogen cyanide (HCN), and toluene (C7H8). These compounds form roughly 5000 ppm of elemental carbon in soil samples brought back from the Moon. These polar regions contain C, H, and O which can serve as propellant sources for methalox spacecraft.[77]
Nitrogen (N) was measured from soil samples brought back to Earth, and it exists as trace amounts at less than 5 ppm.[78] It was found as isotopes 14N, 15N, and 16N.[78][79] As much as 87% of nitrogen found in lunar regolith may come from non-solar sources (not from the Sun) or from other planets. Comets and meteorites contribute less than ~10% of nitrogen from non-solar sources.[80] Carbon and fixed nitrogen would be required for farming activities within a sealed biosphere. Air on earth is about 78.08% nitrogen by volume.
Changesite–(Y)
[edit]Regolith for construction
[edit]Developing a lunar economy will require a significant amount of infrastructure on the lunar surface, which will rely heavily on In situ resource utilization (ISRU) technologies to develop. One of the primary requirements will be to provide construction materials to build habitats, storage bins, landing pads, roads and other infrastructure.[81][82] Unprocessed lunar soil, also called regolith, may be turned into usable structural components,[83][84] through techniques such as sintering, hot-pressing, liquification, the cast basalt method,[20][85] and 3D printing.[81] Glass and glass fiber are straightforward to process on the Moon, and it was found regolith material strengths can be improved by using glass fiber, such as 70% basalt glass fiber and 30% PETG mixture.[81] Successful tests have been performed on Earth using some lunar regolith simulants,[86] including MLS-1 and MLS-2.[87]
The lunar soil, although it poses a problem for any mechanical moving parts, can be mixed with carbon nanotubes and epoxies in the construction of telescope mirrors up to 50 meters in diameter.[88][89][90] Several craters near the poles are permanently dark and cold, a favorable environment for infrared telescopes.[91]
Some proposals suggest to build a lunar base on the surface using modules brought from Earth, and covering them with lunar soil. The lunar soil is composed of a blend of silica and iron-containing compounds that may be fused into a glass-like solid using microwave radiation.[92][93]
The European Space Agency working in 2013 with an independent architectural firm, tested a 3D-printed structure that could be constructed of lunar regolith for use as a Moon base.[94][95][96] 3D-printed lunar soil would provide both "radiation and temperature insulation. Inside, a lightweight pressurized inflatable with the same dome shape would be the living environment for the first human Moon settlers."[96]
In early 2014, NASA funded a small study at the University of Southern California to further develop the Contour Crafting 3D printing technique. Potential applications of this technology include constructing lunar structures of a material that could consist of up to 90-percent lunar material with only ten percent of the material requiring transport from Earth.[97] NASA is also looking at a different technique that would involve the sintering of lunar dust using low-power (1500 watt) microwave radiation. The lunar material would be bound by heating to 1,200 to 1,500 °C (2,190 to 2,730 °F), somewhat below the melting point, in order to fuse the nanoparticle dust into a solid block that is ceramic-like, and would not require the transport of a binder material from Earth.[98]
Mining
[edit]There are several models and proposals on how to exploit lunar resources, yet few of them consider sustainability.[99] Long-term planning is required to achieve sustainability and ensure that future generations are not faced with a barren lunar wasteland by wanton practices.[99][100][101] To be truly sustainable, lunar mining would have to adopt processes that do not use nor yield toxic material, and would minimize waste through recycling loops.[99][82]
Scouting
[edit]Numerous orbiters have mapped the lunar surface composition, including Clementine, LRO, LCROSS, the Artemis orbiter, SELENE, Lunar Prospector, Chandrayaan-1, and Chang'e 1. The Soviet Luna programme and Apollo Program brought lunar samples back to Earth for analysis. As of 2019, a new "Moon race" is ongoing that features prospecting for lunar resources to support crewed bases.
In the 21st century, China's Chinese Lunar Exploration Program,[102][103] is executing a step-wise approach to incremental technology development and scouting for resources for a crewed base, projected for the 2030s, according to Chinese state media Xinhua News Agency.[104] India's Chandrayaan programme is focused in understanding the lunar water cycle first, and on mapping mineral location and concentrations from orbit and in situ. Russia's Luna-Glob programme is planning and developing a series of landers, rovers and orbiters for prospecting and science exploration, and to eventually employ in situ resource utilization (ISRU) methods with the intent to construct and operate their own crewed lunar base in the 2030s.[105][106]
The US has been studying the Moon for decades and in 2019 it started to implement the Commercial Lunar Payload Services (CLPS) program to support the crewed Artemis program, both aimed at scouting and exploiting lunar resources to facilitate a long-term crewed base on the Moon, and depending on the lessons learned, then move on to a crewed mission to Mars.[107] NASA's lunar Resource Prospector rover was planned to prospect for resources on a polar region of the Moon, and it was to be launched in 2022.[108][109] The mission concept was in its pre-formulation stage, and a prototype rover was being tested when it was cancelled in April 2018.[110][108][109] Its science instruments will be flown instead on several commercial lander missions contracted by NASA's CLPS program, that aims to focus on testing various lunar ISRU processes by landing several payloads on multiple commercial robotic landers and rovers. The first payload contracts were awarded on February 21, 2019,[111][112] and will fly on separate missions. The CLPS will inform and support NASA's Artemis program, leading to a crewed lunar outpost for extended stays.[107]
A European non-profit organization has called for a global synergistic collaboration between all space agencies and nations instead of a "Moon race"; this proposed collaborative concept is called the Moon Village.[113] Moon Village seeks to create a vision where both international cooperation and the commercialization of space can thrive.[114][115][116]
Some early private companies like Shackleton Energy Company,[117] Deep Space Industries, Planetoid Mines, Golden Spike Company, Planetary Resources, Astrobotic Technology, and Moon Express are planning private commercial scouting and mining ventures on the Moon.[1][118]
In 2024, an American startup called Interlune announced plans to mine Helium-3 on the Moon for export back on Earth. The first mission plans to use NASA's Commercial Lunar Payload Services program to arrive on the Moon.[119] In August 2025 Interlune indicated the Astrobotic FLIP rover will carry a multispectral camera they designed to search for Helium-3.[120]
Extraction methods
[edit]The extensive lunar maria are composed of basaltic lava flows. Their mineralogy is dominated by a combination of five minerals: anorthites (CaAl2Si2O8), orthopyroxenes ((Mg,Fe)SiO3), clinopyroxenes (Ca(Fe,Mg)Si2O6), olivines ((Mg,Fe)2SiO4), and ilmenite (FeTiO3),[1][49] all abundant on the Moon.[121] It has been proposed that smelters could process the basaltic lava to break it down into pure calcium, aluminium, oxygen, iron, titanium, magnesium, and silica glass.[122] The European Space Agency has awarded funding to Metalysis in 2020 to further develop the FFC Cambridge process to extract titanium from regolith while generating oxygen as a byproduct.[123] Raw lunar anorthite could also be used for making fiberglass and other ceramic products.[122][45] Another proposal envisions the use of fluorine brought from Earth as potassium fluoride to separate the raw materials from the lunar rocks.[124]
Legal status of mining
[edit]Although Luna landers scattered pennants of the Soviet Union on the Moon, and United States flags were symbolically planted at their landing sites by the Apollo astronauts, no nation claims ownership of any part of the Moon's surface as of 2025,[125] and the international legal status of mining space resources is unclear and controversial.[126][127]
The five treaties and agreements[128] of international space law cover "non-appropriation of outer space by any one country, arms control, the freedom of exploration, liability for damage caused by space objects, the safety and rescue of spacecraft and astronauts, the prevention of harmful interference with space activities and the environment, the notification and registration of space activities, scientific investigation and the exploitation of natural resources in outer space and the settlement of disputes."[129]
Russia, China, and the United States are party to the 1967 Outer Space Treaty (OST),[130] which is the most widely adopted treaty, with 104 parties.[131] The OST treaty offers imprecise guidelines to newer space activities such as lunar and asteroid mining,[132] and it therefore remains under contention whether the extraction of resources falls within the prohibitive language of appropriation or whether the use encompasses the commercial use and exploitation. Although its applicability on exploiting natural resources remains in contention, leading experts generally agree with the position issued in 2015 by the International Institute of Space Law (ISSL) stating that, "in view of the absence of a clear prohibition of the taking of resources in the Outer Space Treaty, one can conclude that the use of space resources is permitted."[133]
The 1979 Moon Treaty is a proposed framework of laws to develop a regime of detailed rules and procedures for orderly resource exploitation.[134][135] This treaty would regulate exploitation of resources if it is "governed by an international regime" of rules (Article 11.5),[136] but there has been no consensus and the precise rules for commercial mining have not been established.[137] The Moon Treaty was ratified by very few nations, and thus suggested to have little to no relevancy in international law.[138][139] The last attempt to define acceptable detailed rules for exploitation, ended in June 2018, after S. Neil Hosenball, who was the NASA General Counsel and chief US negotiator for the Moon Treaty, decided that negotiation of the mining rules in the Moon Treaty should be delayed until the feasibility of exploitation of lunar resources had been established.[140]
Seeking clearer regulatory guidelines, private companies in the US prompted the US government, and legalized space mining in 2015 by introducing the US Commercial Space Launch Competitiveness Act of 2015.[141] Similar national legislations legalizing extraterrestrial appropriation of resources are now being replicated by other nations, including Luxembourg, Japan, China, India and Russia.[132][142][143][144] This has created an international legal controversy on mining rights for profit.[142][139] A legal expert stated in 2011 that the international issues "would probably be settled during the normal course of space exploration."[139] In April 2020, U.S. President Donald Trump signed an executive order to support moon mining.[145]
See also
[edit]- Colonization of the Moon – Settlement on the Moon
- Exploration of the Moon – Missions to the Moon
- Geology of the Moon – Structure and composition of the Moon
- In situ resource utilization
References
[edit]- ^ a b c d e f g h i j k l m n o p q Crawford, Ian (2015). "Lunar Resources: A Review". Progress in Physical Geography. 39 (2): 137–167. arXiv:1410.6865. Bibcode:2015PrPG...39..137C. doi:10.1177/0309133314567585. S2CID 54904229.
- ^ Yuhao Lu and Ramaa G. Reddy. Extraction of Metals and Oxygen from Lunar Soil. Archived 2021-11-23 at the Wayback Machine Department of Metallurgical and Materials Engineering; The University of Alabama, Tuscaloosa, Alabama. USA. 9 January 2009.
- ^ a b M. Anand, I. A. Crawford, M. Balat-Pichelin, S. Abanades, W. van Westrenen, G. Péraudeau, R. Jaumann, W. Seboldt. "Moon and likely initial in situ resource utilization (ISRU) applications." Planetary and Space Science; volume 74; issue 1; December 2012, pp: 42—48. doi:10.1016/j.pss.2012.08.012.
- ^ Gerald B. Sanders, Micael Dule. NASA In-Situ Resource Utilization (ISRU) Capability Roadmap Final Report. Archived 2020-09-05 at the Wayback Machine. May 19, 2005.
- ^ S. A. Bailey. "Lunar Resource Prospecting". Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019, Columbia, Maryland.
- ^ D. C. Barker. "Lunar Resources: From Finding to Making Demand." Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019, Columbia, Maryland.
- ^ J. L. Heldmann, A. C. Colaprete, R. C. Elphic, and D. R. Andrews. "Landing Site Selection And Effects On Robotic Resource Prospecting Mission Operations." Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019, Columbia, Maryland.
- ^ Alex Ignatiev and Elliot Carol. "The Use of a Lunar Vacuum Deposition Paver/Rover to Eliminate Hazardous Dust Plumes on the Lunar Surface." Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019, Columbia, Maryland.
- ^ B. R. Blair. "Emarging Markets for Lunar Resources." Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019, Columbia, Maryland.
- ^ a b David, Leonard (7 January 2015). "Is Moon Mining Economically Feasible?". Space.com.
- ^ Taylor, Stuart R. (1975). Lunar Science: a Post-Apollo View. Oxford: Pergamon Press. p. 64. ISBN 978-0-08-018274-2.
- ^ Hugo, Adam (2020-06-24) [April 2019]. "Why the Lunar South Pole?". The Space Resource. Retrieved 2024-05-16.
- ^ S. Maurice. "Distribution of hydrogen at the surface of the moon" (PDF).
- ^ a b Laurent Sibille, William Larson. Oxygen from Regolith. Archived 2020-09-05 at the Wayback Machine. NASA. 3 July 2012.
- ^ a b Gregory Bennett. The Artemis Project – How to Get Oxygen from the Moon Archived 2020-09-05 at the Wayback Machine. Artemis Society International. June 17, 2001.
- ^ Administrator, NASA (2013-06-07). "Is There an Atmosphere on the Moon?". NASA. Retrieved 2022-04-27.
- ^ "Moon Fact Sheet". nssdc.gsfc.nasa.gov. Retrieved 2022-04-27.
- ^ Speyerer, Emerson J.; Robinson, Mark S. (2013). "Persistently illuminated regions at the lunar poles: Ideal sites for future exploration". Icarus. 222 (1): 122–136. Bibcode:2013Icar..222..122S. doi:10.1016/j.icarus.2012.10.010. ISSN 0019-1035.
- ^ Gläser, P., Oberst, J., Neumann, G. A., Mazarico, E., Speyerer, E. J., Robinson, M. S. (2017). "Illumination conditions at the lunar poles: Implications for future exploration. Planetary and Space Science, vol. 162, p. 170–178. doi:10.1016/j.pss.2017.07.006
- ^ a b Spudis, Paul D. (2011). "Lunar Resources: Unlocking the Space Frontier". Ad Astra. National Space Society. Retrieved 30 April 2023.
- ^ Alex Ignatiev, Peter Curreri, Donald Sadoway, and Elliot Carol. "The Use of Lunar Resources for Energy Generation on the Moon." Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019, Columbia, Maryland.
- ^ Landis, Geoffrey A. (2005-12-01). Materials Refining for Solar Array Production on the Moon (Report).
- ^ Skocii, Collin (18 June 2019). "NASA concept for generating power in deep space a little KRUSTY". Spaceflight Insider.
- ^ a b Anderson, Gina; Wittry, Jan (May 2, 2018). "Demonstration Proves Nuclear Fission System Can Provide Space Exploration Power". NASA press release. Retrieved 2024-05-16.
- ^ a b David, Leonard (2019-03-15). "Moon Mining Could Actually Work, with the Right Approach". Space.com. Retrieved 2024-05-16.
- ^ a b c Allen, Carlton C.; McKay, David S. (1995). "Oxygen Production From Lunar Soil". SAE Transactions. 104: 1285–1290. ISSN 0096-736X. JSTOR 44612041.
- ^ Wiechert, U.; Halliday, A. N.; Lee, D.-C.; Snyder, G. A.; Taylor, L. A.; Rumble, D. (2001). "Oxygen Isotopes and the Moon-Forming Giant Impact". Science. 294 (5541): 345–348. Bibcode:2001Sci...294..345W. doi:10.1126/science.1063037. ISSN 0036-8075. JSTOR 3084837. PMID 11598294. S2CID 29835446.
- ^ Hepp, Aloysius F.; Linne, Diane L.; Groth, Mary F.; Landis, Geoffrey A.; Colvin, James E. (1994). "Production and use of metals and oxygen for lunar propulsion". Journal of Propulsion and Power. 10 (16): 834–840. doi:10.2514/3.51397. hdl:2060/19910019908. S2CID 120318455.
- ^ Larry Friesen. Processes for Getting Oxygen on the Moon. Archived 2022-01-18 at the Wayback Machine. Artemis Society International. 10 May 1998.
- ^ Pleil, J. D.; Wallace, M. A.; Davis, M. D.; Matty, C. M. (2021). "The physics of human breathing: Flow, timing, volume, and pressure parameters for normal, on-demand, and ventilator respiration". Journal of Breath Research. 15 (4): 10.1088/1752–7163/ac2589. doi:10.1088/1752-7163/ac2589. PMC 8672270. PMID 34507310.
- ^ "Ice Confirmed at the Moon's Poles". NASA Jet Propulsion Laboratory (JPL). 20 August 2018. Retrieved 2024-05-16.
- ^ "Water on the Moon: Direct evidence from Chandrayaan-1's Moon Impact…". The Planetary Society. April 7, 2010. Retrieved 2024-05-16.
- ^ H. M. Brown. "Identifying Resource-rich Lunar Permanently Shadowed Regions." Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019, Columbia, Maryland.
- ^ a b c J. E. Gruener. "The Lunar Northwest Nearside: The Price Is Right Before Your Eyes." Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019, Columbia, Maryland.
- ^ David, Leonard (13 July 2018). "Mining Moon Ice: Prospecting Plans Starting to Take Shape". Space.com.
- ^ a b L. M. Jozwiak, G. W. Patterson, R. Perkins. "Mini-RF Monostatic Radar Observations of Permanently Shadowed Crater Floors." Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019, Columbia, Maryland.
- ^ Elston, D. P. (1968) "Character and Geologic Habitat of Potential Deposits of Water, Carbon and Rare Gases on the Moon", Geological Problems in Lunar and Planetary Research, Proceedings of AAS/IAP Symposium, AAS Science and Technology Series, Supplement to Advances in the Astronautical Sciences., p. 441.
- ^ "NASA – Lunar Prospector". lunar.arc.nasa.gov. Archived from the original on 2016-09-14. Retrieved 2015-05-25.
- ^ Steigerwald, Bill (2015-02-27). "LRO Discovers Hydrogen More Abundant on Moon's Pole-Facing Slopes". NASA. Retrieved 2022-04-27.
- ^ H. L. Hanks. "Prospective Study for Harvesting Solar Wind Particles via Lunar Regolith Capture." Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019, Columbia, Maryland.
- ^ P. Reiss, F. Kerscher and L. Grill. "Thermogravimetric Analysis of the Reduction of ilmenite and NU-LHT-2M With Hydrogen and Methane." Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019, Columbia, Maryland.
- ^ H. M. Sargeant, F. Abernethy, M. Anand1, S. J. Barber, S. Sheridan, I. Wright, and A. Morse. "Experimental Development And Testing Of The Reduction Of Ilmenite For A Lunar ISRU Demonstration With PRO SPA." Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019, Columbia, Maryland.
- ^ J. W. Quinn. "Electrostatic Beneficiation of Lunar Regolith; A review of the Previous Testing As Starting Point For Future Work." Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019, Columbia, Maryland.
- ^ "Exploring the Moon – A Teacher's Guide with Activities, NASA EG-1997-10-116 - Rock ABCs Fact Sheet" (PDF). NASA. November 1997. Retrieved 19 January 2014.
- ^ a b c d e f g h i j Mark Prado. Major Lunar Minerals. Archived 2019-08-01 at the Wayback Machine. Projects to Employ Resources of the Moon and Asteroids Near Earth in the Near Term (PERMANENT). Accessed on 1 August 2019.
- ^ "The Moon Is Rusting, and Researchers Want to Know Why". NASA Jet Propulsion Laboratory (JPL). Retrieved 2022-04-27.
- ^ Space com Staff (2011-10-11). "Moon Packed with Precious Titanium, NASA Probe Finds". Space.com. Retrieved 2024-05-16.
- ^ "SMART-1 detects calcium on the Moon". www.esa.int. 8 June 2005. Retrieved 2024-05-16.
- ^ a b Deer, W. A.; Howie, R. A.; Zussman, J. (1966). An Introduction to the Rock Forming Minerals. London, England: Longman. p. 336. ISBN 0-582-44210-9.
- ^ A. Ignatiev and A. Freundlich. New Architecture for Space Solar Power Systems: Fabrication of Silicon Solar Cells Using In-Situ Resources. Archived 2019-01-01 at the Wayback Machine. NIAC 2nd Annual Meeting, June 6–7, 2000.
- ^ Rao, D. B.; Choudary, U. V.; Erstfeld, T. E.; Williams, R. J.; Chang, Y. A. (1979-01-01). "Extraction processes for the production of aluminum, titanium, iron, magnesium, and oxygen and nonterrestrial sources". NASA. Ames Research Center, Space Resources and Space Settlements. 428: 257. Bibcode:1979NASSP.428..257B.
- ^ Cordierite-Spinel Troctolite, a New Magnesium-Rich Lithology from the Lunar Highlands. Science. Vol 243, Issue 4893. 17 February 1989 {{doi}10.1126/science.243.4893.925}}.
- ^ a b Jolliff, B. L.; Tran, T. N.; Lawrence, S. J.; Robinson, M. S.; et al. (2011). Compton-Belkovich: Nonmare, Silicic Volcanism on the Moon's Far Side (PDF). 42nd Lunar and Planetary Science Conference. Retrieved May 14, 2012.
- ^ Lawrence, D. J.; Elphic, R. C.; Feldman, W. C.; Gasnault, O.; Genetay, I.; Maurice, S.; Prettyman, T. H. (March 2002). Small-Area Thorium Enhancements on the Lunar Surface. 33rd Annual Lunar and Planetary Science Conference. Harvard University. Bibcode:2002LPI....33.1970L.
- ^ "China may not issue new 2011 rare earths export quota: report". Reuters. 31 December 2010.
- ^ Medeiros, Carlos Aguiar De; Trebat, Nicholas M.; Medeiros, Carlos Aguiar De; Trebat, Nicholas M. (July 2017). "Transforming natural resources into industrial advantage: the case of China's rare earths industry". Brazilian Journal of Political Economy. 37 (3): 504–526. doi:10.1590/0101-31572017v37n03a03. ISSN 0101-3157.
- ^ Haxel, G.; Hedrick, J.; Orris, J. (2002). Stauffer, Peter H.; Hendley II, James W. (eds.). "Rare Earth Elements—Critical Resources for High Technology" (PDF). United States Geological Survey. USGS Fact Sheet: 087-02. Retrieved 2012-03-13.
However, in contrast to ordinary base and precious metals, REE have very little tendency to become concentrated in exploitable ore deposits. Consequently, most of the world's supply of REE comes from only a handful of sources.
- ^ Goldman, Joanne Abel (April 2014). "The U.S. Rare Earth Industry: Its Growth and Decline". Journal of Policy History. 26 (2): 139–166. doi:10.1017/s0898030614000013. ISSN 0898-0306. S2CID 154319330.
- ^ Tse, Pui-Kwan. "USGS Report Series 2011–1042: China's Rare-Earth Industry". pubs.usgs.gov. Retrieved 2018-04-04.
- ^ A. A. Mardon, G. Zhou, R. Witiw. "Lunar Rare-Earth Minerals For Commercialization." Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019, Columbia, Maryland.
- ^ a b "The Lunar Gold Rush: How Moon Mining Could Work". NASA Jet Propulsion Laboratory (JPL). May 28, 2019. Retrieved 2024-05-16.
- ^ L. J. Wittenberg, E. N. Cameron, G. L. Kulcinski, S. H. Ott, J. F. Santarius, G. I. Sviatoslavsky, I. N. SViatoslavsky & H. E. Thompson. A Review of 3He Resources and Acquisition for Use as Fusion Fuel. Archived 2020-05-14 at the Wayback Machine. Fusion Technology, volume 21, 1992; issue 4; pp: 2230–2253; 9 May 2017. doi:10.13182/FST92-A29718.
- ^ FTI Research Projects: 3He Lunar Mining Archived 2006-09-04 at the Wayback Machine. Fti.neep.wisc.edu. Retrieved on 2011-11-08.
- ^ E. N. Slyuta; A. M. Abdrakhimov; E. M. Galimov (2007). "The estimation of helium-3 probable reserves in lunar regolith" (PDF). Lunar and Planetary Science XXXVIII (1338): 2175. Bibcode:2007LPI....38.2175S.
- ^ Cocks, F. H. (2010). "3He in permanently shadowed lunar polar surfaces". Icarus. 206 (2): 778–779. Bibcode:2010Icar..206..778C. doi:10.1016/j.icarus.2009.12.032.
- ^ Hedman, Eric R. (January 16, 2006). "A fascinating hour with Gerald Kulcinski". The Space Review.
- ^ "Korean fusion reactor achieves record plasma – World Nuclear News". www.world-nuclear-news.org. Retrieved 2020-05-30.
- ^ "Fusion reactor – Principles of magnetic confinement". Encyclopedia Britannica. Retrieved 2020-05-30.
- ^ a b c Day, Dwayne (September 28, 2015). "The helium-3 incantation". The Space Review. Retrieved 11 January 2019.
- ^ a b "Nuclear Fusion: WNA". world-nuclear.org. November 2015. Archived from the original on 2015-07-19. Retrieved 2019-07-22.
- ^ Sviatoslavsky, I. N. (November 1993). "The challenge of mining He-3 on the lunar surface: how all the parts fit together" (PDF). Archived from the original (PDF) on 2019-01-20. Retrieved 2019-07-22. Wisconsin Center for Space Automation and Robotics Technical Report WCSAR-TR-AR3-9311-2.
- ^ David, Leonard (4 March 2003). "China Outlines its Lunar Ambitions". Space.com. Archived from the original on March 16, 2006. Retrieved 2006-03-20.
- ^ "DOE National Laboratory Makes History by Achieving Fusion Ignition". Energy.gov. 13 December 2022. Retrieved 2023-01-02.
- ^ Carbon on the Moon. Archived 2010-06-13 at the Wayback Machine Artemis Society International. 8 August 1999.
- ^ Colin Trevor Pillinger and Geoffrey Eglinton. "The chemistry of carbon in the lunar regolith." Philosophical Transactions of the Royal Society. 1 January 1997. doi:10.1098/rsta.1977.0076.
- ^ American Geophysical Union. "Carbon dioxide cold traps on the moon are confirmed for the first time". phys.org. Retrieved 2022-04-27.
- ^ Cannon, Kevin M. (2021-04-27). "Accessible Carbon on the Moon". arXiv:2104.13521 [astro-ph.EP].
- ^ a b Richard H. Becker and Robert N. Clayton. Nitrogen abundances and isotopic compositions in lunar samples Archived 2019-07-23 at the Wayback Machine. Proceedings Lunar Science Conference, 6th (1975); pp: 2131–2149. Bibcode:1975LPSC....6.2131B.
- ^ Füri, Evelyn; Barry, Peter H.; Taylor, Lawrence A.; Marty, Bernard (2015). "Indigenous nitrogen in the Moon: Constraints from coupled nitrogen–noble gas analyses of mare basalts". Earth and Planetary Science Letters. 431: 195–205. Bibcode:2015E&PSL.431..195F. doi:10.1016/j.epsl.2015.09.022. ISSN 0012-821X.
- ^ Mortimer, J.; Verchovsky, A. B.; Anand, M. (2016-11-15). "Predominantly non-solar origin of nitrogen in lunar soils". Geochimica et Cosmochimica Acta. 193: 36–53. Bibcode:2016GeCoA.193...36M. doi:10.1016/j.gca.2016.08.006. ISSN 0016-7037. S2CID 99355135.
- ^ a b c Brad Buckles, Robert P. Mueller, and Nathan Gelino. "Additive Construction Technology For Lunar Infrastructure." Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019.
- ^ a b A. K. Hayes, P. Ye, D. A. Loy, K. Muralidharan, B. G. Potter, and J. J. Barnes. "Additive Manufacturing of Lunar Mineral-Based Composites." Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019.
- ^ "Indigenous lunar construction materials". AIAA PAPER 91-3481. Retrieved 2007-01-14.
- ^ "In-Situ Resource Utilization (ISRU) – NASA". Retrieved 2024-05-16.
- ^ "Cast Basalt" (PDF). Ultratech. Archived from the original (PDF) on 2006-08-28. Retrieved 2007-01-14.
- ^ Gerald B. Sanders, William E. Larson. Title: Integration of In-Situ Resource Utilization Into Lunar/Mars Exploration Through Field Analogs. Archived 2019-07-23 at the Wayback Machine. NASA Johnson Space Center. 2010.
- ^ Tucker, Dennis S.; Ethridge, Edwin C. (May 11, 1998). Processing Glass Fiber from Moon/Mars Resources (PDF). Proceedings of American Society of Civil Engineers Conference, 26–30 April 1998. Albuquerque, New Mexico, United States. 19990104338. Archived from the original (PDF) on 2000-09-18.
- ^ Naeye, Robert (6 April 2008). "NASA Scientists Pioneer Method for Making Giant Lunar Telescopes". Goddard Space Flight Center. Retrieved 27 March 2011.
- ^ Lowman, Paul D.; Lester, Daniel F. (November 2006). "Build astronomical observatories on the Moon?". Physics Today. Vol. 59, no. 11. p. 50. Archived from the original on 7 November 2007. Retrieved 16 February 2008.
- ^ Bell, Trudy (9 October 2008). "Liquid Mirror Telescopes on the Moon". Science News. NASA. Retrieved 27 March 2011.
- ^ Chandler, David (15 February 2008). "MIT to lead development of new telescopes on moon". MIT News. Retrieved 27 March 2011.
- ^ "Lunar Dirt Factories? A look at how regolith could be the key to permanent outposts on the moon". The Space Monitor. 2007-06-18. Retrieved 30 April 2023.
- ^ Blacic, James D. (1985). "Mechanical Properties of Lunar Materials Under Anhydrous, Hard Vacuum Conditions: Applications of Lunar Glass Structural Components". Lunar Bases and Space Activities of the 21st Century: 487–495. Bibcode:1985lbsa.conf..487B.
- ^ "Building a lunar base with 3D printing / Technology / Our Activities / ESA". Esa.int. 2013-01-31. Retrieved 2014-03-13.
- ^ "Foster + Partners works with European Space Agency to 3D print structures on the moon". Foster + Partners. 31 January 2013. Archived from the original on 3 February 2013. Retrieved 3 February 2013.
- ^ a b Diaz, Jesus (2013-01-31). "This Is What the First Lunar Base Could Really Look Like". Gizmodo. Retrieved 2013-02-01.
- ^ "NASA's plan to build homes on the Moon: Space agency backs 3D print technology which could build base". TechFlesh. 2014-01-15. Archived from the original on 2014-01-16. Retrieved 2014-01-16.
- ^ Steadman, Ian (1 March 2013). "Giant Nasa spider robots could 3D print lunar base using microwaves (Wired UK)". Wired UK. Retrieved 2014-03-13.
- ^ a b c A. A. Ellery. "Sustainable Lunar In-Situ Resource Utilization = Long-Term Planning." Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019, Columbia, Maryland.
- ^ G. Harmer. "Integrating ISRU Projects to Create A Sustainable In-Space Economy." Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019, Columbia, Maryland.
- ^ A. A. Mardon, G. Zhou, R. Witiw. "Ethical Conduct in Lunar Commercialization." Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019, Columbia, Maryland.
- ^ Devlin, Hannah (21 January 2019). "Battlefield moon: how China plans to win the lunar space race". The Guardian.
- ^ Bender, Bryan (13 June 2019). "A new moon race is on. Is China already ahead?". Politico.
- ^ "China has no timetable for manned moon landing: chief scientist". Xinhua. 19 September 2012. Archived from the original on October 5, 2012.
- ^ "Russia Plans to Colonize Moon by 2030, Newspaper Reports". The Moscow Times. 8 May 2014. Archived from the original on 19 July 2017. Retrieved 8 May 2014.
- ^ Litvak, Maxim (2016). "The vision of the Russian Space Agency on the robotic settlements in the Moon" (PDF). IKI/Roscosmos.
- ^ a b Moon to Mars. Archived 2019-07-25 at the Wayback Machine NASA. Accessed on 23 July 2019.
- ^ a b Grush, Loren (April 27, 2018). "NASA scraps a lunar surface mission – just as it's supposed to focus on a Moon return". The Verge.
- ^ a b Berger, Eric (27 April 2018). "New NASA leader faces an early test on his commitment to Moon landings". ARS Technica.
- ^ Resource Prospector Archived 2019-03-08 at the Wayback Machine. Advanced Exploration Systems, NASA. 2017.
- ^ Richardson, Derek (February 26, 2019). "NASA selects experiments to fly aboard commercial lunar landers". Spaceflight Insider.
- ^ Szondy, David (21 February 2019). "NASA picks 12 lunar experiments that could fly this year". New Atlas.
- ^ Foust, Jeff (26 December 2018). "Urban planning for the Moon Village". Space News.
- ^ Jan Wörner, ESA Director General. Moon Village: A vision for global cooperation and Space 4.0 Archived 2019-10-16 at the Wayback Machine. April 2016.
- ^ David, Leonard (2016-04-26). "Europe Aiming for International 'Moon Village'". Space.com. Retrieved 2024-05-16.
- ^ Moon Village: humans and robots together on the Moon Archived 2019-06-04 at the Wayback Machine. ESA. 1 March 2016.
- ^ Wall, Mike (14 January 2011). "Mining the Moon's Water: Q&A with Shackleton Energy's Bill Stone". space.com. Retrieved April 30, 2023.
- ^ Hennigan, W. J. (2011-08-20). "MoonEx aims to scour Moon for rare materials". Los Angeles Times. Retrieved 2011-04-10.
MoonEx's machines are designed to look for materials that are scarce on Earth but found in everything from a Toyota Prius car battery to guidance systems on cruise missiles.
- ^ Eaton, Kit (Mar 14, 2024). "Space Startup Interlune Emerges From Stealth Mode to Start Moon Mining Effort". Inc.
- ^ "Interlune will team up with Astrolab to send a camera to the moon for helium-3 survey". 5 August 2025.
- ^ "Significant Lunar Minerals" (PDF). In Situ Resource Utilization (ISRU). NASA. Archived from the original (PDF) on 27 December 2016. Retrieved 23 August 2018.
- ^ a b "Mining and Manufacturing on the Moon". NASA. Archived from the original on 2006-12-06. Retrieved 2007-01-14.
- ^ "Metalysis gets ESA development contract for FFC process". Institute of Materials, Minerals & Mining.
- ^ Landis, Geoffrey. "Refining Lunar Materials for Solar Array Production on the Moon" (PDF). NASA. Archived from the original (PDF) on 2006-10-09. Retrieved 2007-03-26.
- ^ "Can any State claim a part of outer space as its own?". United Nations Office for Outer Space Affairs. Archived from the original on 21 April 2010. Retrieved 28 March 2010.
- ^ David, Leonard (25 July 2014). "Mining the Moon? Space Property Rights Still Unclear, Experts Say". Space.com.
- ^ Wall, Mike (14 January 2011). "Moon Mining Idea Digs Up Lunar Legal Issues". Space.com.
- ^
- The 1967 Treaty on Principles Governing the Activities of States in the Exploration and Use of Outer Space, including the Moon and Other Celestial Bodies (the "Outer Space Treaty").
- The 1968 Agreement on the Rescue of Astronauts, the Return of Astronauts and the Return of Objects Launched into Outer Space (the "Rescue Agreement").
- The 1972 Convention on International Liability for Damage Caused by Space Objects (the "Liability Convention").
- The 1975 Convention on Registration of Objects Launched into Outer Space (the "Registration Convention").
- The 1979 Agreement Governing the Activities of States on the Moon and Other Celestial Bodies (the "Moon Treaty").
- ^ United Nations Office for Outer Space Affairs. "United Nations Treaties and Principles on Space Law". unoosa.org. Retrieved 23 February 2019.
- ^ "How many States have signed and ratified the five international treaties governing outer space?". United Nations Office for Outer Space Affairs. 1 January 2006. Archived from the original on 21 April 2010. Retrieved 28 March 2010.
- ^ Committee on the Peaceful Uses of Outer Space Legal Subcommittee: Fifty-fifth session. Archived 2019-01-19 at the Wayback Machine Vienna, Austria, 4–15 April 2016. Item 6 of the provisional agenda: Status and application of the five United Nations treaties on outer space.
- ^ a b Senjuti Mallick and Rajeswari Pillai Rajagopalan. If space is 'the province of mankind', who owns its resources? Archived 2020-05-10 at the Wayback Machine.The Observer Research Foundation. 24 January 2019. Quote 1: "The Outer Space Treaty (OST) of 1967, considered the global foundation of the outer space legal regime, […] has been insufficient and ambiguous in providing clear regulations to newer space activities such as asteroid mining." *Quote2: "Although the OST does not explicitly mention "mining" activities, under Article II, outer space including the Moon and other celestial bodies are "not subject to national appropriation by claim of sovereignty" through use, occupation or any other means."
- ^ "Institutional Framework for the Province of all Mankind: Lessons from the International Seabed Authority for the Governance of Commercial Space Mining." Jonathan Sydney Koch. "Institutional Framework for the Province of all Mankind: Lessons from the International Seabed Authority for the Governance of Commercial Space Mining." Astropolitics, 16:1, 1–27, 2008. doi:10.1080/14777622.2017.1381824
- ^ Louis de Gouyon Matignon. The 1979 Moon Agreement. Archived 2019-11-06 at the Wayback Machine. Space Legal Issues. 17 July 2019.
- ^ J. K. Schingler and A. Kapoglou. "Common Pool Lunar Resources." Archived 2020-07-25 at the Wayback Machine. Lunar ISRU 2019: Developing a New Space Economy Through Lunar Resources and Their Utilization. July 15–17, 2019, Columbia, Maryland.
- ^ United Nations (5 December 1979). "Moon Agreement". www.unoosa.org. Retrieved 2024-05-16.
Resolution 34/68 Adopted by the United Nations General Assembly. 89th plenary meeting; 5 December 1979.
- ^ Fabio Tronchetti. Current International Legal Framework Applicability to Space Resource Activities. Archived 2020-10-20 at the Wayback Machine. IISL/ECSL Space Law Symposium 2017, Vienna, Austria. 27 March 2017.
- ^ Listner, Michael (24 October 2011). "The Moon Treaty: failed international law or waiting in the shadows?". The Space Review.
- ^ a b c James R. Wilson. Regulation of the Outer Space Environment Through International Accord: The 1979 Moon Treaty. Archived 2020-08-03 at the Wayback Machine. Fordham Environmental Law Review, Volume 2, Number 2, Article 1, 2011.
- ^ Beldavs, Vidvuds (15 January 2018). "Simply fix the Moon Treaty". The Space Review.
- ^ H.R. 2262 – U.S. Commercial Space Launch Competitiveness Act. 114th Congress (2015–2016) Archived 2015-11-19 at the Wayback Machine. Sponsor: Representative McCarthy, Kevin. 5 December 2015.
- ^ a b Davies, Rob (6 February 2016). "Asteroid mining could be space's new frontier: the problem is doing it legally". The Guardian.
- ^ Ridderhof, R. (18 December 2015). "Space Mining and (U.S.) Space Law". Peace Palace Library. Archived from the original on 27 February 2019. Retrieved 26 February 2019.
- ^ "Law Provides New Regulatory Framework for Space Commerce | RegBlog". www.regblog.org. 31 December 2015. Retrieved 2016-03-28.
- ^ Wall, Mike (6 April 2020). "Trump signs executive order to support Moon mining, tap asteroid resources". Space.com.
Lunar resources
View on GrokipediaGeological and Compositional Overview
Lunar Formation and Bulk Composition
, with earth-like abundances of refractory lithophile elements like aluminum, calcium, and titanium, but it exhibits significant depletion in volatile elements such as potassium, sodium, rubidium, and cesium by a factor of approximately 4 relative to BSE.[15] This volatile depletion, evident in Apollo samples and lunar meteorites, likely resulted from high-temperature processes during the impact and subsequent magma ocean crystallization, which partitioned volatiles into the early atmosphere or lost them to space.[16] The lunar mantle is dominantly composed of olivine, pyroxene, and plagioclase, with a small core estimated at 1-2% of the Moon's mass, leading to an overall FeO content of about 12-13 wt% in the bulk silicate Moon, higher than Earth's mantle but lower than chondritic meteorites.[15] These compositional traits underpin the Moon's resource potential, emphasizing abundant silicates and oxides suitable for extraction.[17]Surface Regolith Characteristics
The lunar regolith consists of unconsolidated, fragmental debris overlying the bedrock, formed predominantly through meteoroid impacts that comminute and mix surface materials over billions of years. This layer, lacking any weathering processes seen on Earth due to the absence of atmosphere and water, incorporates rock fragments, mineral grains, impact glasses, and agglutinates derived from local bedrock and deeper ejecta. Its thickness varies regionally, averaging 5 to 10 meters in the basaltic maria but reaching 10 to 15 meters or more in the anorthositic highlands, as determined from seismic data and sample analyses. Physically, regolith particles range from submicrometer dust to multicentimeter fragments, with a mean grain size of 60 to 80 micrometers in mature soils, though finer fractions dominate due to repeated impact grinding. Bulk density is approximately 1.5 g/cm³, with low cohesion (0.1 to 1.0 kN/m²) and internal friction angles of 30° to 50°, rendering it cohesionless and prone to flow under mechanical disturbance, as observed in Apollo excavation tests. Agglutinates—complex glassy aggregates formed by micrometeorite-induced melting and rapid quenching—comprise 20% to 50% by volume in mature regolith, increasing with exposure age and contributing to its cohesive yet friable texture; these structures preserve solar wind-implanted volatiles and exhibit vesicularity from degassing.[21][22][23] Mineralogically, the regolith reflects underlying geology: highland samples are dominated by plagioclase feldspar (up to 70-80% anorthite-rich), with lesser pyroxene, olivine, and minor troilite, while mare regolith features higher proportions of clinopyroxene, ilmenite (up to 10% in titanium-rich basalts), and olivine from volcanic sources. Chemical composition is primarily oxides—oxygen (42-45%), silicon (20-21%), aluminum (13-15% in highlands), calcium (12-16%), iron (5-15%), magnesium (5-10%), and titanium (up to 6% in maria)—with trace meteoritic additions (<2%) enriching siderophile elements. Apollo mission analyses confirm these variations, with solar wind implantation adding hydrogen, helium, and other gases at parts-per-million levels, though bulk regolith remains anhydrous.[24][21][25]Polar Volatiles and Permanently Shadowed Regions
Permanently shadowed regions (PSRs) on the Moon are topographic depressions, primarily impact craters near the lunar poles, that remain in perpetual darkness due to the Moon's low axial tilt of approximately 1.5 degrees relative to its orbit. These areas maintain extremely low temperatures, often below 100 K, enabling them to act as cold traps that capture and preserve volatile compounds delivered by solar wind implantation, micrometeorite impacts, or outgassing. The south polar region hosts a larger total area of PSRs, estimated at over 13,000 square kilometers, compared to about 2,500 square kilometers at the north pole, with Cabeus crater being a prominent example at the south pole.[26][27] Remote sensing data from orbital instruments have provided indirect evidence of elevated hydrogen concentrations in PSRs, interpreted as signatures of water ice or hydrated minerals. The Lunar Prospector Neutron Spectrometer, operating from 1998 to 1999, detected suppressed epithermal neutron fluxes at both poles, indicating hydrogen abundances up to 150 parts per million or higher in some areas, consistent with water ice deposits. Subsequent observations by the Lunar Reconnaissance Orbiter's Lunar Exploration Neutron Detector (LEND) confirmed higher hydrogen signals within PSRs, particularly in Cabeus crater, where concentrations exceed those in surrounding illuminated terrain.[28][29] Direct confirmation of water ice came from the LCROSS mission on October 9, 2009, which impacted a Centaur rocket stage into Cabeus crater, ejecting material analyzed by accompanying spectrometers. The impact plume revealed water vapor and absorption features corresponding to 5.6 ± 2.9% water by mass in the regolith, alongside other volatiles such as carbon monoxide, carbon dioxide, and ammonia. Near-infrared spectroscopy from the Chandrayaan-1 Moon Mineralogy Mapper in 2009 and LRO's Diviner instrument further identified diagnostic 3-micron absorption bands indicative of surface-exposed water ice in select PSRs, comprising up to 30% of the surface in some small craters.[30][31] Beyond water, PSRs may harbor additional volatiles including molecular nitrogen, methane, and sulfur compounds, potentially from cometary or asteroidal delivery, though their abundances remain uncertain without in-situ sampling. Recent modeling suggests volatile migration via ballistic transport and re-deposition influences distribution, with subsurface ice possibly extending meters deep in stable traps. These deposits' persistence relies on minimal solar heating, but micro-scale cold traps may be ephemeral, lasting only thousands of years under current flux rates.[32][33][27]Primary Resources for In-Situ Utilization
Water Ice and Volatiles
Water ice exists on the Moon predominantly within permanently shadowed regions (PSRs) at the polar craters, where low temperatures prevent sublimation and preserve volatiles delivered by comets or solar wind implantation.[26] These deposits are crucial for in-situ resource utilization, potentially providing hydrogen and oxygen for propellant, life support, and radiation shielding.[34] The presence of water was first inferred in 1998 by NASA's Lunar Prospector orbiter, which detected elevated hydrogen concentrations via neutron spectroscopy in polar regions, suggesting possible ice mixed with regolith.[26] Definitive confirmation came from the 2009 LCROSS mission, where a kinetic impactor struck Cabeus crater at the south pole, ejecting material analyzed by a shepherding spacecraft; this revealed water vapor and ice amounting to approximately 155 kilograms in the plume, with regolith water content estimated at 5.6% ± 2.9% by mass.[35] [36] The mission also identified other volatiles, including molecular hydrogen, carbon monoxide, and nitrogen- and sulfur-bearing compounds, indicating a diverse mix preserved in the cold trap.[37] In 2018, analysis of data from the Moon Mineralogy Mapper (M3) instrument aboard India's Chandrayaan-1 orbiter provided direct spectroscopic evidence of surface-exposed water ice in multiple PSRs, particularly at the south pole, distinguishing it from hydrated minerals through distinct absorption features at 1.25, 1.38, and 2.20 micrometers.[38] [39] These findings mapped patchy ice distributions, with higher abundances in the Cabeus and Shoemaker craters. Estimates suggest polar PSRs could hold over 600 billion kilograms of water ice, equivalent to filling hundreds of thousands of Olympic-sized swimming pools, though accessibility varies due to depth and mixing with dry regolith.[34] Recent missions have refined understanding of volatile accessibility. India's Chandrayaan-3 lander, which touched down near the south pole in August 2023, used its ChaSTE thermal probe to measure subsurface temperatures, revealing that ice may exist just a few centimeters below the surface in sunlit areas adjacent to PSRs, potentially easing extraction compared to deep-buried deposits in shadowed craters.[40] [41] NASA's PRIME-1 mission, launched in February 2025, aims to drill and analyze ice in a south polar PSR to assess usability quantities.[26] Other volatiles, such as hydroxyl (OH) in sunlit regions from solar wind interactions, complement polar ice but are less concentrated for resource purposes.[26]Oxygen and Metals from Regolith
Lunar regolith consists primarily of fine-grained, unconsolidated material derived from meteorite impacts and volcanic activity, with an average oxygen content of 41-45% by weight, locked in minerals such as silicates, oxides, and glasses.[42][24] The remaining composition includes silicon (around 20%), aluminum (10-15%), calcium (10-15%), iron (5-15% as FeO, varying by location), magnesium (5-10%), and titanium (up to 6% in ilmenite-rich highlands).[42][43] These elements occur mainly in minerals like plagioclase feldspars (e.g., anorthite for aluminum and calcium), pyroxenes (for magnesium and iron), and ilmenite (FeTiO3 for iron and titanium), making regolith a viable feedstock for in-situ resource utilization (ISRU) to produce oxygen for life support and propulsion, as well as metals for construction and manufacturing.[24][44] Oxygen extraction from regolith targets the reduction of metal oxides, with processes operating under lunar vacuum conditions to minimize energy needs. Hydrogen reduction involves reacting regolith with hydrogen gas at 700-1000°C to form water vapor, which is then electrolyzed into oxygen and hydrogen; NASA has tested this method using lunar simulants, achieving yields dependent on ilmenite content (up to 5-10% of regolith mass).[45] Molten salt electrolysis dissolves regolith in a salt bath (e.g., calcium chloride) at 900-1000°C, applying electricity to liberate oxygen at the anode while depositing metals at the cathode; in April 2023, NASA engineers at Johnson Space Center successfully demonstrated this on JSC-1A simulant, extracting oxygen with efficiencies approaching 96% under optimized conditions.[46][47] Carbothermal reduction heats regolith with carbon to 1500-2000°C, producing carbon monoxide and metals, followed by gas separation for oxygen recovery via CO/CO2 electrolysis; this solar-thermal variant has been prototyped for scalability but requires higher temperatures.[48][49] These methods could yield 100-200 kg of oxygen per ton of regolith, depending on mineralogy and process efficiency, enabling propellant production for return missions.[50] Metals are co-extracted as byproducts, providing raw materials for habitats, tools, and infrastructure without Earth resupply. Iron, often reduced to metallic form or alloys, constitutes 5-15% of regolith oxides and can be magnetically separated from native micrometeorite-derived grains (0.1-0.5% free iron by weight).[51] Aluminum from anorthositic feldspars and titanium from ilmenite enable alloy production via electrolysis or smelting, with processes like the Airbus ROXY system targeting integrated output of oxygen alongside ferrotitanium and aluminosilicates at rates suitable for 1-10 tons per year in early lunar bases.[52][43] Challenges include energy intensity (e.g., 10-20 kWh/kg oxygen) and dust abrasion on equipment, but vacuum sintering of regolith fines can preprocess material for higher purity feeds.[53] Overall, regolith-derived metals reduce launch mass from Earth by factors of 10-100 for structural elements, supporting sustainable lunar operations.[54] ![Lunar ferroan anorthosite sample 60025][float-right]Construction and Propellant Materials
Lunar regolith, the unconsolidated surficial layer covering the Moon's surface with thicknesses ranging from meters to tens of meters, provides abundant feedstock for in-situ construction materials through processes such as sintering and molten regolith electrolysis (MRE).[55] Sintering involves heating regolith to form solid blocks or structures with compressive strengths suitable for habitats and infrastructure, leveraging its high content of silicates and oxides like SiO2, Al2O3, and FeO. MRE electrolyzes molten regolith to yield oxygen gas and metal alloys, with the resulting glassy slag serving as a potential binder or aggregate in construction composites, reducing reliance on Earth-sourced materials.[56] Highlands regions, dominated by ferroan anorthosite rich in plagioclase feldspar (primarily anorthite), offer aluminum- and calcium-enriched regolith ideal for producing high-strength ceramics or cement analogs via thermal processing.[57] For propellant materials, regolith-derived oxygen extracted via carbothermal reduction of ilmenite (FeTiO3, comprising up to 10% of mare regolith) or MRE enables production of liquid oxygen (LOX) for use in bipropellant systems.[58] These processes can yield oxygen at rates sufficient for refueling landers, with MRE additionally producing iron and silicon alloys usable in propellant tankage or engine components.[56] Metals such as aluminum (from anorthositic regolith) and iron support fabrication of lightweight structures for propellant storage depots, enhancing ISRU efficiency by minimizing launch mass from Earth.[3] Additive manufacturing techniques, including 3D printing with regolith-polymer composites optimized for lunar vacuum and radiation, further enable on-site production of propellant handling infrastructure.[59] Demonstrations using regolith simulants have validated these approaches, confirming material properties like tensile strength exceeding 10 MPa for sintered products under simulated lunar conditions.[60]Strategic and Energy Resources
Helium-3 Deposits
Helium-3 (^3He), a rare isotope on Earth, accumulates in the lunar regolith primarily through implantation by solar wind particles, as the Moon lacks a global magnetic field or atmosphere to deflect them.[61] [9] This process has deposited ^3He over billions of years, with concentrations correlating to regolith exposure age, solar wind fluence, and mineral composition, particularly higher trapping in ilmenite (FeTiO_3).[9] [62] Interest in lunar ^3He stems from its potential as a fuel for aneutronic fusion reactions, such as deuterium-^3He, which produce minimal neutrons and could enable cleaner energy production compared to deuterium-tritium fusion.[63] Direct measurements from Apollo mission samples reveal ^3He abundances ranging from 0.4 to 15 parts per billion (ppb) by mass in lunar soils, with total helium contents varying from 1 to 63 parts per million (ppm).[62] Apollo 11 regolith from sample 10084 averaged 11.8 ppb ^3He, with individual measurements between 9.22 and 17.9 ppb.[64] These values are higher in mature, solar wind-exposed soils and increase with titanium oxide (TiO_2) content, as ^3He preferentially adheres to iron and titanium-bearing phases during heating extraction experiments, where up to 75% releases by 600°C.[65] [62] Global estimates place total lunar ^3He reserves at approximately 6.5 × 10^8 kg (650,000 metric tons), with about 57% (3.72 × 10^8 kg) on the nearside and 43% (2.78 × 10^8 kg) on the farside, derived from remote sensing data on regolith composition and maturity.[66] Alternative models yield similar inventories of 6.6 × 10^8 kg, emphasizing equatorial maria regions where ilmenite-rich basalts enhance concentrations up to 20-30 ppb in some projections.[67] [68] Distribution is uneven, favoring areas of prolonged surface exposure and low gardening by micrometeorites, though polar regions show lower abundances due to reduced solar wind access in shadowed craters.[9] Extraction feasibility hinges on processing vast regolith volumes, as average concentrations of ~10 ppb imply handling 100-150 million tons of soil to yield 1 ton of ^3He, with beneficiation targeting ilmenite separation via magnetic or electrostatic methods.[64] [63] While remote sensing from missions like Lunar Prospector has mapped proxies like iron and titanium to infer ^3He hotspots, in-situ validation remains limited beyond Apollo sites, underscoring uncertainties in scalable mining economics.[69]Rare-Earth Elements and Phosphates
The KREEP geochemical component, characterized by enrichments in potassium (K), rare-earth elements (REE), and phosphorus (P), represents a key reservoir of REEs and phosphates on the Moon, originating from residual melts of the lunar magma ocean that were later incorporated into impact breccias and late-stage volcanics.[70] This component is unevenly distributed, with highest abundances in the Procellarum KREEP Terrain (PKT) on the nearside and the South Pole-Aitken (SPA) basin on the farside, as mapped by orbital gamma-ray spectrometers using thorium as a proxy for KREEP due to its geochemical association.[71] REE patterns in KREEP-rich samples, such as Apollo 14 breccias and lunar meteorites, typically show light REE enrichment and negative europium anomalies, reflecting fractional crystallization processes in the lunar interior.[72] Bulk REE concentrations in lunar KREEP materials range from tens to several hundred ppm total REE oxides, significantly lower than the 150–220 ppm average in Earth's continental crust but elevated relative to the depleted lunar highlands anorthosites (often <10 ppm).[73] Accessory minerals hosting REEs include monazite and yttrium-rich phases like yittrobetafite (up to 94,500 ppm yttrium) and tranquillityite (up to 0.25 wt% REE), though these constitute minor fractions of bulk regolith.[74] Remote sensing data from missions like Lunar Prospector indicate peak KREEP signatures in PKT basalts, where REE abundances correlate with incompatible element proxies, but surface regolith dilution by impacts reduces extractable yields.[71] Phosphates in lunar samples primarily occur as merrillite (a REE-bearing whitlockite-group mineral) and subordinate apatite, comprising up to 1–2 wt% P₂O₅ in KREEP-rich breccias from Apollo 12 and 14 sites.[75] These minerals formed through late-stage magmatic differentiation and are preserved in impact-metamorphosed rocks, with U-Pb dating of phosphates in Apollo 14 melts yielding ages around 3.9–4.0 Ga, linking them to early lunar bombardment events.[76] Phosphorus contents exceed 0.5 wt% P₂O₅ in many basalts and soils, often exceeding predictions from simple magma ocean models due to volatile partitioning or metasomatism.[77] Unlike REEs, phosphates show less regional concentration but are ubiquitous in fertile regolith for potential in-situ fertilizer or alloy production, though extraction challenges include their fine-grained, disseminated nature in breccias.[75]Nuclear Fuels: Thorium and Uranium
Thorium and uranium occur in the lunar regolith as incompatible elements concentrated during magmatic differentiation, primarily within the Procellarum KREEP Terrane (PKT) on the nearside, where KREEP (potassium-rare earth element-phosphorus) materials are enriched.[78] Global mapping from the Lunar Prospector Gamma-Ray Spectrometer (1998–1999) revealed thorium concentrations averaging approximately 1.2 ppm, with peaks exceeding 10 ppm in PKT highlands and Oceanus Procellarum basalts, while uranium averages ~0.3 ppm and reaches up to 2 ppm in similar regions.[79] These elements correlate spatially due to a consistent U/Th ratio of ~0.25–0.27, reflecting fractional crystallization processes that partitioned them into late-stage melts.[79] Detection relies on natural gamma-ray emissions from radioactive decay, with Lunar Prospector's spectrometer providing the first global thorium map, showing depletions in highlands (<1 ppm) and enrichments tied to mare volcanism and impact excavation of deeper KREEP.[80] Subsequent missions, including Chang'E-2 (2010), refined thorium distributions, confirming hotspots in western nearside maria and the South Pole-Aitken basin rim at levels up to ~6–8 ppm, with lower resolutions limiting uranium mapping.[81] Apollo samples from highlands yielded thorium at 0.3–1.5 ppm and uranium at ~0.1–0.4 ppm, validating remote data but indicating bulk regolith dilution requires processing ~10^5–10^6 tons for reactor-grade fuel yields.[79] For in-situ utilization, thorium's abundance (global ~5–10 times Earth's crustal average) supports thorium-based reactors, which offer higher fuel efficiency and reduced long-lived waste compared to uranium cycles, though extraction involves regolith beneficiation via electrostatic separation or acid leaching to concentrate ores from ppm levels.[82] Uranium's lower prevalence limits its viability, but co-extraction with thorium could enable hybrid fission systems for lunar bases, powering habitats or propellant production at ~1–10 MW scales with feasible regolith throughput.[79] Challenges include radiation shielding needs during mining and isotopic enrichment for fissile U-235 or Th-232 breeding to U-233, with no confirmed lunar deposits exceeding terrestrial high-grade ores (e.g., >100 ppm).[82] Ongoing analyses from Chang'E-5 samples (2020) suggest minor thorium enrichments in young volcanics, potentially indicating untapped nearside resources.[83]Historical Discovery and Mapping
Apollo-Era Sample Returns
![Lunar ferroan anorthosite 60025 from Apollo 16][float-right]The Apollo program conducted six successful crewed lunar landings between 1969 and 1972, returning a total of 381.7 kilograms of lunar material, including rocks, soil, and core samples from the Moon's surface.[84] These samples, collected primarily from the lunar maria and highlands near the equator, provided the first direct evidence of the Moon's bulk composition and regolith properties, forming the foundational dataset for assessing potential in-situ resources.[85] Analysis revealed that the regolith consists predominantly of fine-grained silicate minerals, with oxygen comprising approximately 45% by weight bound in oxides, alongside metals such as silicon, iron, magnesium, aluminum, and titanium.[86] Key resource-relevant findings included the identification of ilmenite (FeTiO3) in mare basalts, which is enriched in titanium and serves as a potential source for oxygen and metal extraction through processes like hydrogen reduction or molten salt electrolysis.[87] Highland anorthosites, dominated by plagioclase feldspar (calcium aluminum silicate), indicated abundant aluminum oxides suitable for producing aluminum metal and oxygen for propulsion or life support.[87] Core samples from Apollo 15 and 17 demonstrated regolith layering and maturity, with solar wind-implanted volatiles like helium-3 adsorbed on grain surfaces, though concentrations were low (5-30 parts per billion) and limited to the uppermost layers disturbed by micrometeorite impacts.[88] The samples confirmed negligible indigenous water or hydrated minerals in equatorial regions, attributing any trace hydrogen to solar wind implantation rather than polar ice deposits.[88] These analyses established the feasibility of regolith as a primary resource for oxygen production, estimated at yields of 1-2 tons per ton of processed regolith, and metals for construction or manufacturing, influencing subsequent in-situ resource utilization concepts by highlighting the need for beneficiation to separate oxides like ilmenite from bulk soil.[86] Breccias and volcanic glasses in the samples, such as the orange soil from Apollo 17, revealed localized enrichments in iron and titanium, underscoring site-specific variability that would require targeted prospecting for optimal resource extraction.[85] Overall, Apollo-era returns demonstrated the Moon's surface materials as viable feedstock for self-sustaining lunar operations, though extraction efficiencies depend on energy-intensive processing to overcome the regolith's refractory nature.[89]
Remote Sensing from Orbiters
Remote sensing from lunar orbiters utilizes spectroscopic and nuclear techniques to infer resource compositions without direct sampling. Multispectral and hyperspectral imaging in ultraviolet, visible, and near-infrared wavelengths identifies mineral assemblages by their unique reflectance signatures, while gamma-ray and neutron spectrometers detect elemental abundances through interactions with cosmic rays and solar particles producing secondary radiation. These methods probe surface and shallow subsurface layers, typically to depths of meters for neutrons, enabling global mapping of volatiles like water ice via hydrogen proxies and silicates such as pyroxenes and olivines.[90][91] The Clementine mission (1994) pioneered systematic resource mapping with its ultraviolet-visible (UVVIS) and near-infrared (NIR) cameras, acquiring global coverage in 11 spectral bands from February 19 to May 3. Data revealed distributions of clinopyroxene, orthopyroxene, olivine, ilmenite, and plagioclase, highlighting mafic-rich highlands and titanium concentrations in maria basalts essential for oxygen and metal extraction. These maps, processed via Hapke radiative transfer models, provided foundational compositional insights despite calibration challenges from the mission's short duration.[92][93][94] Lunar Prospector (1998–1999) advanced volatile detection using a neutron spectrometer sensitive to hydrogen moderation of epithermal neutrons, mapping polar enhancements suggesting 300 million metric tons of water ice in permanently shadowed craters. The instrument, orbiting at 30–100 km altitude, resolved hydrogen to ~10% concentration levels over 30-km footprints, corroborated by gamma-ray data on major elements like Fe, Ti, and Al. Initial announcements in March 1998 indicated ice at both poles, though subsequent reanalyses noted ambiguities in distinguishing ice from other hydrogen sources like solar wind-implanted protons.[95][91][96] Later missions refined these findings; Chandrayaan-1's Moon Mineralogy Mapper (2008), a hyperspectral imager spanning 0.4–3 μm at 140-m resolution, detected hydroxyl and water absorption features globally, with 2018 reanalysis confirming exposed water ice in south polar craters like Shackleton via distinct 1.8-, 1.9-, and 3-μm bands. NASA's Lunar Reconnaissance Orbiter (2009–present), equipped with the Lunar Exploration Neutron Detector (LEND), mapped hydrogen distributions at 5–10 km resolution, validating polar cold traps as ice reservoirs while infrared instruments like Diviner assessed regolith thermophysical properties for resource accessibility. These orbital datasets, cross-validated across missions, underscore heterogeneous resource patches, informing in-situ utilization prospects despite resolution limits and illumination biases.[38][31][97]Recent Missions and In-Situ Confirmations
China's Chang'e-5 mission, which landed in the Oceanus Procellarum region on December 1, 2020, conducted the first in-situ spectroscopic observations confirming the presence of water molecules on the lunar surface using the Solid-state Imager and Lunar Mineral Spectrometer aboard the lander. These measurements, taken under Earth's magnetospheric shielding and at surface temperatures around 200–300 K, detected OH/H₂O absorption features at 3 μm, indicating water content influenced by solar wind implantation rather than high-latitude cold traps.[98] Analysis of the returned regolith samples, totaling 1,731 grams, further quantified water in minerals such as apatite and amphibole, with concentrations exceeding 170 ppm in some fractions—higher than previously estimated for equatorial regions—and revealed multiple water sources including solar wind-derived volatiles preserved in impact glasses.[99][100] The Chang'e-6 mission, launched in May 2024 and returning 1,935 grams of samples from the far side's Apollo Basin on June 25, 2024, provided in-situ data from a higher-latitude site (around 42°S), extending insights into volatile distribution. Preliminary analyses of these samples identified water molecules at concentrations comparable to Chang'e-5, with evidence suggesting forms stable at mid-latitudes, potentially from solar wind or endogenous processes, though detailed isotopic studies are ongoing to distinguish origins.[101] These missions corroborated remote sensing data by demonstrating that water-bearing phases persist in surficial regolith, albeit at low abundances (tens to hundreds of ppm), challenging assumptions of water exclusivity to polar shadowed craters.[102] India's Chandrayaan-3 lander, touching down at 69.37°S, 32.35°E near the lunar south pole on August 23, 2023, performed the first in-situ elemental abundance measurements in a high-latitude region using the Alpha Particle X-ray Spectrometer (APXS) on the Pragyan rover. Over six days of operations covering 100 meters, APXS detected major elements including silicon (45 wt%), iron (15–20 wt%), aluminum (10–15 wt%), and unexpectedly high sulfur (up to 0.4 wt% in fine regolith), consistent with immature, volatile-enriched soils derived from primitive mantle material and supporting the lunar magma ocean hypothesis.[103][104] Complementary thermal probing by the Chandra's Surface Thermophysical Experiment (ChaSTE) inserted a probe 10 cm into the regolith, measuring vertical temperature gradients (from 0.5°C increase at depth) and conductivity profiles indicative of porous, low-density subsurface layers suitable for resource extraction assessments.[105] These findings affirm the south polar region's regolith as a viable source for metals, oxygen, and potential volatiles, though direct water ice detection remains pending further polar lander deployments.[106] NASA's Polar Resources Ice Mining Experiment-1 (PRIME-1), manifested on Intuitive Machines' IM-1 Odysseus lander (February 2024) and IM-2 Athena lander (February 2025), aimed to drill 1 meter into permanently shadowed regions for in-situ volatile analysis via mass spectrometry, but IM-1's partial deployment failure limited initial results, while IM-2 operations confirmed regolith properties without definitive ice quantification as of mid-2025. These efforts underscore ongoing technical challenges in accessing polar volatiles, with regolith drilling and gas extraction demos validating ISRU precursors despite mission anomalies.[107]Extraction and Processing Technologies
Resource Prospecting Techniques
Remote sensing from lunar orbiters constitutes the primary initial phase of resource prospecting, enabling broad-scale mapping of potential deposits before targeted surface operations. Instruments such as neutron spectrometers detect hydrogen concentrations—indicative of water ice or hydrated minerals—by measuring the moderation of neutrons produced from cosmic ray interactions with the regolith, with sensing depths up to approximately 1 meter.[108] The Neutron Spectrometer System (NSS), developed by NASA's Ames Research Center, quantifies total subsurface hydrogen volume through energy changes in scattered neutrons.[108] Complementary near-infrared spectrometers, like the Near-Infrared Volatiles Imaging Spectrometer System (NIRVSS), identify surface volatiles including water molecules, hydroxyl groups, and compounds such as ammonia or methane by analyzing absorption and emission spectra in the 1.6–3.0 micrometer range, often paired with multispectral imaging for contextual mapping.[108] Radar techniques, including synthetic aperture radar, probe subsurface structures and dielectric properties to infer ice presence in permanently shadowed regions, as demonstrated by the Mini-RF instrument on NASA's Lunar Reconnaissance Orbiter.[109] These orbital methods provide global coverage but are limited by resolution (typically kilometers) and require ground truthing due to ambiguities in spectral interpretations from regolith mixing or temperature effects.[110] In-situ prospecting refines remote data through direct sampling and analysis via landers or rovers, targeting high-priority sites like polar craters for volatiles or highland anorthosites for metals. Mobile platforms traverse terrains while deploying neutron and infrared spectrometers for localized hydrogen mapping, supplemented by visible/near-infrared cameras for geological context and hazard avoidance.[110] Drilling systems access subsurface regolith, typically to 1–2 meters, to retrieve intact samples for volatile extraction via heating ovens that vaporize and analyze released gases using mass or infrared spectrometers, distinguishing ice forms, concentrations, and isotopic compositions.[110] NASA's Volatiles Investigating Polar Exploration Rover (VIPER), revived in September 2025 for deployment via Blue Origin's lander, integrates a 1-meter regolith and ice drill (TRIDENT), NSS for hydrogen profiling, NIRVSS for spectral identification, and a mass spectrometer (MSolo) for gas composition in shadowed environments.[111] [112] Similarly, the European Space Agency's PROSPECT package, intended for Russia's Luna-27 mission, employs the ProSEED drill to acquire samples up to 2 meters deep and the ProSPA analytical suite—featuring modular ovens for thermal extraction and spectrometers for elemental/isotopic assays—to evaluate regolith volatiles and demonstrate in-situ resource utilization precursors.[113] These techniques prioritize efficiency in low-gravity, vacuum conditions, with drills designed for minimal power (under 200 watts) and rovers for autonomous navigation over rough terrain spanning kilometers.[113][110] Prospecting campaigns integrate these approaches hierarchically: orbital surveys identify candidates, followed by in-situ validation to assess extractability, grain size, and purity, informing economic viability for resources like water ice (estimated at 100–400 ppm hydrogen equivalents in polar deposits) or helium-3 in regolith.[114] Challenges include instrument calibration for lunar extremes (temperatures from 40 K to 400 K) and data fusion across methods to resolve discrepancies, such as between radar-inferred ice blocks and spectroscopic signals.[109] Ongoing developments emphasize miniaturized, radiation-hardened payloads for commercial landers under NASA's Commercial Lunar Payload Services program.[108]Mining Methods and Equipment
Lunar regolith excavation presents unique challenges due to its fine-grained, abrasive nature, high cohesion in vacuum conditions, electrostatic charging, and the Moon's low gravity, which reduces traction and alters tool dynamics compared to terrestrial operations.[115] Primary methods focus on mechanical disruption and transport for in-situ resource utilization (ISRU), targeting resources like water ice, oxygen, and metals embedded in the upper 1-2 meters of surface material.[116] These techniques emphasize minimal energy use and dust mitigation to prevent equipment abrasion and habitat contamination.[117] Surface-level methods dominate initial proposals, employing scoops, rakes, and bucket-wheel excavators to loosen and collect loose regolith. For instance, rake systems drag tines across the surface to shear material, achieving excavation rates of up to 0.5-1 cubic meter per hour in simulations, while bucket wheels rotate to continuously load regolith onto conveyors for transport. Auger drills, suited for vertical extraction, bore into soil and elevate it via helical screws, with prototypes demonstrating efficiencies in low-gravity analogs by minimizing horizontal forces that could cause rover slippage. Dozer blades, as in NASA's ISRU Pilot Excavator (IPEx), push regolith into windrows or trenches, capable of handling approximately 10,000 kg per lunar day (about 29 Earth days) in operational tests using simulant soils. For permanently shadowed regions (PSRs) harboring water ice, subsurface methods incorporate thermal or pneumatic techniques to volatilize and capture ices without direct mechanical digging, which risks contaminating or sublimating the resource in vacuum.[3] Drilling rigs with coring bits access depths up to 1 meter, followed by heating elements to extract volatiles, as prototyped in NASA's Resource Prospector mission concepts.[44] Robotic platforms, such as six-wheeled rovers or legged systems, integrate these tools with mobility in uneven terrain, prioritizing autonomy via AI for path planning and fault tolerance, as human teleoperation faces communication delays of 2.5 seconds one-way.[115] Equipment development centers on rugged, radiation-hardened robots to operate without constant supervision. NASA's IPEx, a modular excavator-dozer hybrid, uses electric actuators and regolith-traction tracks tested in Hawaii's volcanic simulants, emphasizing scalability for propellant production.[122] Complementary systems include magnetic separators for ilmenite beneficiation post-excavation and pneumatic conveyors to transport fines without mechanical wear.[44] Emerging international efforts, like Japan's "Lunar Mushi" autonomous miners targeting metals by 2030, employ multi-limb grippers for precise digging, though scalability remains unproven beyond lab demos.[123] Overall, methods prioritize dry, non-water-intensive processes to avoid altering regolith properties, with power budgets typically under 10 kW for early robotic units.[115]In-Situ Resource Utilization Processes
In-situ resource utilization (ISRU) processes on the Moon focus on extracting and converting indigenous materials, primarily water ice and regolith, into mission-critical products such as oxygen, hydrogen, water, and propellants, thereby minimizing mass launched from Earth.[116] These techniques leverage the Moon's polar permanently shadowed regions (PSRs) for volatiles and the oxygen-rich silicate regolith, which comprises about 40-45% oxygen by weight, to support habitat life support, propulsion, and construction. Development efforts, led by agencies like NASA and ESA, emphasize scalable, energy-efficient methods tested with simulants and aimed at demonstration in the 2020s via Artemis missions.[3] Water extraction from icy regolith in PSRs begins with thermal desorption techniques to sublimate volatiles under vacuum conditions. Drilling-based thermal methods involve auger-like systems that heat regolith to 150-200°C, capturing released water vapor for condensation and purification, with pilot-scale tests achieving up to 90% recovery efficiency in cryogenic simulants.[124] Microwave heating offers a non-contact alternative, penetrating regolith to selectively volatilize ice while minimizing energy loss, as demonstrated in experiments extracting 5-10% water by mass from icy samples without significant dust generation.[125] NASA's Lunar Auger Dryer ISRU (LADI) prototype uses a screw conveyor within PSRs to process regolith continuously, producing water for electrolysis into breathable oxygen and propellant-grade hydrogen.[126] Extracted water is then electrolyzed via proton exchange membrane systems, yielding H2 and O2 at efficiencies exceeding 70% in lunar vacuum tests.[127] Oxygen production from anhydrous regolith employs reduction or electrolytic processes to liberate bound oxygen from minerals like ilmenite (FeTiO3) and anorthite. Hydrogen reduction, the most mature technique with a Technology Readiness Level (TRL) of 5-6, reacts regolith with H2 at 700-1000°C to form water, which is subsequently electrolyzed: FeTiO3 + H2 → Fe + TiO2 + H2O.[128] Lab-scale tests have yielded 1-2% oxygen per regolith mass per cycle.[129] Carbothermal reduction, demonstrated by NASA in April 2023, heats regolith with carbon to produce carbon monoxide and oxygen, extracting up to 96% of available oxygen from simulants in a reactor operating at 1000-1500°C.[46] Molten oxide electrolysis (MOE) melts regolith at ~1600°C and applies voltage to separate oxygen gas at the anode, with NASA prototypes producing 5-10 g O2 per kWh while yielding ferrous byproducts for metallurgy.[130] ESA's electrochemical setup, tested in 2020, extracted 30-50% oxygen from regolith simulants over 100 hours using a molten salt electrolyte.[131] Propellant production integrates water-derived LOX/LH2 or regolith-derived LOX with imported methane for storable hybrids. NASA's Lunar Propellant Production Plant (LP3) targets polar ice for electrolysis, aiming to produce 10-100 kg/day of propellants by separating H2 and O2 via cryogenic distillation, with ground prototypes validating storage in lunar temperatures as low as -200°C.[132] Hybrid architectures combine carbothermal oxygen from dry regolith with water extraction, potentially reducing energy needs by 20-30% compared to standalone systems, as modeled for scalable plants processing 1-10 tons of regolith daily.[133] Energy requirements for these processes range from 10-50 kWh/kg O2, primarily from solar concentrators, with vacuum pyrolysis variants using focused sunlight to achieve 80-90% efficiency in oxygen yield.[134] Challenges include dust mitigation and power scaling, addressed in ongoing TRL 4-6 demonstrations.[44]Economic Feasibility and Challenges
Cost-Benefit Analyses
Cost-benefit analyses of lunar resource extraction emphasize in-situ resource utilization (ISRU) for producing propellants from water ice, as returning bulk materials to Earth remains uneconomical given current launch costs exceeding $1,000 per kg to the lunar surface. These assessments typically model upfront capital expenditures for mining infrastructure, operational costs including power generation, and benefits from reduced payload mass for subsequent missions, such as propellant depots enabling cis-lunar transport at lower effective costs.[135] Analyses often project breakeven points contingent on sustained demand from government or commercial entities, with private-sector models assuming public-private partnerships to offset risks. For water ice extraction at the lunar poles, mechanical beneficiation methods—such as crushing, sieving, and electrostatic separation—offer lower energy demands (e.g., 118 W for 100 kg/day output) compared to thermal mining, which requires hundreds of kW due to inefficiencies in heating permanently shadowed craters.[136] Infrastructure costs for a scalable ice mining operation are estimated at $2.5 billion in nonrecurring expenses, including development, production, and launches, with annual operations at $78 million for systems producing 1,100 metric tons yearly.[135] Benefits include electrolyzing ice into hydrogen and oxygen, reducing mission costs by factors of up to 70 for lunar surface-to-orbit transfers and 2-3 for Earth-to-Mars trajectories by avoiding Earth-launched propellants.[135] Revenue projections at $500 per kg for lunar-sourced propellant yield internal rates of return of 9-16% over a 10-year lifespan, assuming reliable extraction from deposits with 10-30% ice concentration.[135] Regolith-derived resources, such as oxygen from ilmenite reduction, face higher barriers, with models indicating limited cost savings unless integrated with large-scale habitats requiring minimal Earth resupply. Helium-3 extraction for potential fusion applications is dismissed in rigorous analyses as non-viable, given dilute concentrations (parts per billion), energy-intensive separation, and the absence of commercial fusion reactors as of 2025. Breakeven for ISRU propellant plants demands operational lifetimes exceeding five years and annual demand above 30 metric tons, achievable in extended lunar campaigns but not in short-duration or Mars-centric missions where Earth-sourced alternatives remain cheaper by up to 97%.| Resource | Extraction Cost Driver | Projected Benefit | Key Assumption for Viability |
|---|---|---|---|
| Water Ice (Propellant) | $2.5B CapEx; low-energy mechanical methods | 70x reduction in surface-to-orbit costs | >5-year ops; NASA/commercial demand >1,100 t/yr[135] |
| Regolith Oxygen | High power for processing | Enables habitat air supply | Scaled infrastructure; minimal Earth imports |
| Helium-3 | Energy for isotope separation | Hypothetical fusion fuel | Commercial fusion tech (not current) |
Market Demands and Scalability Issues
The primary market demand for lunar resources centers on water ice, particularly for conversion into propellants such as liquid hydrogen and oxygen via electrolysis, which could enable in-situ refueling and reduce the mass of material launched from Earth for deep-space missions.[58] Economic models indicate that lunar-derived propellants could lower overall space transportation costs, with analyses suggesting commercial viability once production scales to support routine operations, potentially capturing a share of the projected lunar economy valued at up to €142 billion by 2040, including resource utilization segments.[137] [138] Secondary demands include regolith-derived oxygen for life support and construction materials like sintered bricks, driven by needs for permanent habitats, though these remain contingent on establishing initial outposts.[139] Helium-3 extraction from regolith has been proposed as a fusion fuel source, with estimated lunar reserves potentially yielding energy equivalents of terrestrial oil supplies, but practical demand is limited by the absence of scalable fusion reactors as of 2025, rendering it economically speculative despite ongoing research into extraction via thermal desorption.[64] [140] Scalability challenges arise from the high energy and infrastructural requirements for large-volume extraction, where initial systems must process thousands of tons of regolith annually to yield meaningful outputs like 10-100 tons of propellant, necessitating solar or nuclear power plants capable of megawatt-scale operations in the lunar vacuum and temperature extremes.[141] Terrestrial mining analogies highlight difficulties in adapting excavation techniques—such as bucket-wheel or swarm robotics—to lunar regolith's abrasive, electrostatic properties, which complicate equipment durability and throughput scaling without excessive downtime.[115] [142] Integration issues further impede progress, including physical interfaces between ISRU processors, habitats, and landers, compounded by the need for autonomous, long-duration reliability to avoid Earth-dependent resupplies.[141] Economically, the "scale relevance" of ISRU creates a bootstrapping dilemma: viable markets emerge only after demonstrating production at levels supporting multiple users, yet upfront investments exceed billions, with risk amplified by unproven fault-tolerant systems and the 3-6 month communication delays for oversight.[143] Projections for the lunar ISRU market, valued at USD 1.04 billion in 2024, underscore that growth to support broader space economies hinges on overcoming these hurdles through iterative demonstrations, such as NASA's planned polar ice mining tests.[144]Technical and Logistical Hurdles
Lunar resource extraction and in-situ utilization (ISRU) operations must contend with the Moon's extreme environmental conditions, including temperature swings from approximately -173°C during the 14-day lunar night to 127°C in sunlight, near-vacuum pressures that complicate heat dissipation and material outgassing, and constant exposure to solar radiation and micrometeorites, all of which demand robust, radiation-hardened equipment capable of autonomous operation over extended uncrewed periods.[145] Low gravity, at one-sixth of Earth's, further challenges mechanical systems by altering material flow in excavators and processors, potentially leading to uneven settling or fluid dynamics issues in metallurgical or chemical reactions.[44] These factors necessitate specialized designs, such as thermal insulation layers and vibration-isolated components, tested in analog environments like vacuum chambers, yet full-scale demonstrations remain limited as of 2023.[146] A primary technical obstacle is lunar regolith, the loose surface layer averaging 5-10 meters deep in maria regions, consisting of fine, jagged particles (often <20 micrometers) that exhibit electrostatic levitation in vacuum and adhere persistently to surfaces, causing abrasion, seal failures, and reduced solar panel efficiency by up to 10-20% over time.[147] This dust, lacking atmospheric weathering, infiltrates mechanisms during excavation—estimated to produce 10-100 kg of respirable particles per cubic meter of dug material—exacerbating wear on drills, conveyors, and filters while posing health risks to crews via inhalation or habitat contamination.[148] Mitigation strategies include electrostatic repulsion technologies; for instance, NASA's Electrodynamic Dust Shield (EDS), using transparent electrodes to generate traveling waves, removed over 90% of simulant dust in ground tests and demonstrated efficacy against actual regolith during a March 2025 lunar deployment.[149] Passive approaches, like sintered regolith coatings or brushless seals, show promise but require integration with active systems for sustained operations, as terrestrial dust control methods fail in vacuum due to absent sedimentation.[150] Power generation and storage represent another bottleneck, as solar arrays— the baseline for initial ISRU—yield intermittent output due to the Moon's 28-day synodic cycle, with polar sites offering near-constant insolation but shadowed craters (rich in water ice) demanding mobile or nuclear alternatives.[151] ISRU processes like hydrogen reduction of ilmenite for oxygen production require 10-20 kWh per kg of output, straining kilowatt-scale systems; fission reactors, capable of 10-40 kWe continuously, face deployment challenges including thermal management in regolith burial for shielding and regulatory hurdles for radioisotope alternatives.[152] Energy storage via regenerative fuel cells or flywheels must bridge 14-day nights, adding mass penalties of 20-50% for round-trip efficiency, while grid transmission over kilometers demands high-voltage DC lines resilient to dust accumulation and seismic events (moonquakes up to magnitude 5).[153] Logistically, transporting personnel, equipment, and extracted materials incurs high delta-v costs—approximately 5.9 km/s for Earth-to-lunar orbit insertion and another 2-3 km/s for descent/ascent—necessitating reusable landers and prepositioned caches, yet current architectures project cargo delivery rates below 100 tons annually until 2030s infrastructure scales.[154] Supply chain vulnerabilities include dependency on Earth for spares during 3-6 month resupply cycles, compounded by regolith-induced failures shortening hardware life from years to months, and the need for autonomous robotics to prospect and mine in advance of crews, as human oversight is limited by communication delays of 2.5 seconds one-way.[155] Returning refined resources, such as metals or volatiles, amplifies costs, with launch masses exceeding 10 tons per mission for viable payloads, underscoring the imperative for closed-loop ISRU to minimize Earth dependency before economic viability emerges.[156]Legal and Geopolitical Dimensions
Outer Space Treaty Interpretations
The Outer Space Treaty (OST), formally the Treaty on Principles Governing the Activities of States in the Exploration and Use of Outer Space, including the Moon and Other Celestial Bodies, entered into force on October 10, 1967, and has been ratified by over 110 states, including major spacefaring nations. Article II explicitly prohibits "national appropriation by claim of sovereignty, by means of use or occupation, or by any other means," while Article I affirms the freedom of exploration and use of outer space, including the Moon, "for the benefit and in the interests of all countries" and declares celestial bodies the "province of all mankind." These provisions create ambiguity regarding the extraction and ownership of lunar resources, as the treaty does not directly address commercial mining or private property rights in extracted materials, a scenario unforeseen during its drafting amid Cold War-era priorities focused on preventing territorial claims rather than resource exploitation. Interpretations favoring resource utilization, particularly by the United States and its partners, hold that the OST permits the removal and ownership of lunar resources by private entities or states, provided no sovereignty is asserted over the celestial body itself. This view analogizes resource extraction to activities like deep-sea fishing, where removal from a commons area confers ownership without appropriating the underlying territory. The U.S. codified this in Title IV of the Commercial Space Launch Competitiveness Act of 2015, enacted November 25, 2015, which grants U.S. citizens rights to "possess, own, transport, use, and sell" asteroid or space resources obtained via commercial recovery, explicitly stating it does not authorize territorial claims in violation of the OST. Similarly, Luxembourg's 2017 space resources law mirrors this approach. Legal scholars supporting this position argue that Article I's endorsement of "use" encompasses extraction, and the treaty's silence on post-extraction ownership implies permissibility, as evidenced by historical practices like Apollo sample returns, which were retained by the U.S. without international objection.[157][158][159] Opposing interpretations contend that such national laws indirectly undermine the OST's non-appropriation principle and "benefit of all" mandate by enabling de facto control through private proxies, potentially leading to monopolization of scarce lunar resources like helium-3 or water ice. Critics, including some international lawyers, assert that allowing ownership of extracted resources contravenes the treaty's intent to treat celestial bodies as a global commons, drawing parallels to the Antarctic Treaty System's resource moratorium. A 2019 analysis labeled the U.S. 2015 Act "illegal" under the OST, arguing it obliges the U.S. government to protect private extractions, effectively extending state authority over lunar sites. These views often invoke the 1979 Moon Agreement, which declares lunar resources the "common heritage of mankind" and requires an international regime for exploitation—though the agreement has only 18 ratifications and lacks endorsement from the U.S., Russia, or China, rendering it non-binding on major actors.[160][161] The Artemis Accords, signed by the U.S. and 48 partner nations as of October 2024, reinforce the permissive interpretation by affirming "the ability to extract and utilize space resources" as consistent with the OST, emphasizing transparency, interoperability, and safety zones around operations to mitigate interference without claiming territory. Russia and China have criticized the Accords as a U.S.-led attempt to bypass multilateral consensus, proposing instead the International Lunar Research Station without explicit resource rights. This divergence highlights ongoing tensions, with proponents of utilization arguing that restrictive readings stifle innovation absent technological feasibility in 1967, while skeptics warn of geopolitical risks from unilateralism, though empirical evidence of violations remains absent given no large-scale extraction to date.[162][163]National Legislation and Accords
The United States enacted the Commercial Space Launch Competitiveness Act of 2015, which includes the Space Resource Exploration and Utilization Act provisions granting U.S. citizens and companies the right to own, transport, and sell resources extracted from asteroids, the Moon, and other celestial bodies, provided activities comply with the Outer Space Treaty (OST).[164] This legislation interprets Article I of the OST—affirming the freedom of exploration and use of outer space—as permitting the extraction and ownership of non-territorial resources without constituting national appropriation under Article II.[164] In 2020, President Trump issued Executive Order 13914, directing U.S. agencies to promote international support for space resource recovery and use, emphasizing commercial opportunities while rejecting the Moon Agreement's common heritage framework due to its limited ratification and restrictions on private activity.[165] Luxembourg passed the Law of July 20, 2017, on the Exploration and Use of Space Resources, authorizing private entities to explore and extract space resources, including those on the Moon, and granting ownership rights over obtained materials upon issuance of a mission authorization by the relevant minister.[166] The law explicitly aligns with OST interpretations allowing resource utilization without claiming sovereignty over celestial bodies, positioning Luxembourg as a hub for space mining firms by offering legal certainty and investment incentives.[166] The United Arab Emirates issued Cabinet Resolution No. (19) of 2023 Concerning Space Resources Regulation, which recognizes property rights—including ownership, sale, trade, and disposal—over space resources extracted during authorized activities, applicable to lunar and other extraterrestrial materials.[167] This framework requires licensing for resource operations and prohibits activities risking UAE compliance with international obligations, reflecting a pro-commercial stance amid the nation's expanding space program.[167] The Artemis Accords, initiated by the United States in 2020 and signed by 56 nations as of October 2025, outline non-binding principles for lunar and deep-space cooperation, explicitly endorsing the extraction and utilization of space resources as compatible with the OST to support sustainable exploration.[168] Signatories commit to transparency, interoperability, and safety zones around operations, including resource sites, while preserving outer space heritage; the accords reject Moon Agreement constraints and facilitate commercial involvement in NASA's Artemis program.[169] In contrast, China has not enacted enabling legislation for private space resource ownership, asserting in UN submissions that the OST's non-appropriation principle precludes national claims to lunar resources and requires an international regime for equitable benefit-sharing prior to exploitation.[170] Russia similarly lacks specific domestic laws permitting extraterrestrial resource extraction, viewing unilateral national authorizations as inconsistent with OST prohibitions on appropriation and advocating multilateral governance to prevent commercialization without global consensus.[171] These positions underscore geopolitical tensions, with Russia and China proposing alternative frameworks emphasizing state-led activities and collective oversight.[172]Competition Between Spacefaring Powers
The primary competition for lunar resources centers on the United States' Artemis program and China's International Lunar Research Station (ILRS), with the latter partnering with Russia. Artemis, initiated in 2017, seeks to establish a sustainable human presence at the Moon's south pole, targeting water ice deposits for in-situ resource utilization (ISRU) to produce oxygen, water, and propellant, thereby enabling extended missions and potential commercial mining operations. The program has garnered commitments from over 40 nations through the Artemis Accords, which affirm resource extraction rights under interpretations of the Outer Space Treaty allowing non-appropriative use. In contrast, the ILRS, announced in 2021, aims for a similar base by 2030, focusing on polar resource prospecting including helium-3 from regolith and water ice, with Russia providing nuclear power technologies amid strained U.S. relations post-Ukraine invasion. This rivalry has intensified since China's Chang'e-6 mission returned south pole samples in June 2024, demonstrating capabilities for targeted resource sampling. Geopolitical tensions arise from overlapping claims to resource-rich sites, particularly the south pole's permanently shadowed craters holding an estimated 600 million metric tons of water ice, critical for reducing Earth-launch dependencies. U.S. officials have expressed concerns that Chinese precedence could establish de facto control zones, complicating Artemis landings and fostering parallel infrastructure ecosystems that fragment global standards for ISRU.[173] China's advancements, including planned taikonaut landings by 2030, contrast with Artemis delays—Artemis III, targeting crewed landing, slipped from 2025 to at least 2026 due to SpaceX Starship development setbacks—prompting warnings from former NASA administrators that the U.S. risks ceding strategic high ground.[174] Russia's pivot to ILRS, formalized in a 2021 memorandum, reflects sanctions-driven decoupling from the International Space Station, positioning the alliance to challenge U.S. dominance in cislunar space.[175] Private sector involvement amplifies U.S. competitiveness, with companies like SpaceX and Blue Origin pursuing reusable landers for resource extraction demos, potentially lowering costs to $1,000 per kg to orbit versus China's state-centric model. However, this introduces risks of uneven progress, as China's centralized approach has yielded consistent robotic successes, including three south pole orbiters by 2024. Other powers, such as India via Chandrayaan-3's 2023 south pole landing and Japan through Artemis partnerships, align with the U.S. to counterbalance China, but resource-specific rivalries remain nascent, with helium-3 mining—touted for fusion energy—facing technical hurdles rendering it speculative amid water ice's nearer-term viability.[176] Overall, the contest prioritizes technological precedence over immediate extraction, with prestige and dual-use technologies (e.g., ISRU for propulsion) carrying broader implications for Earth-Moon hegemony.[177]Controversies and Debates
Overstated Resource Hype vs. Empirical Limits
Proponents of lunar resource utilization often portray the Moon's regolith and polar deposits as transformative assets, capable of enabling a self-sustaining space economy through extraction of water for propellant, metals for construction, and isotopes like helium-3 for fusion energy.[68] Such claims, advanced by figures including former Apollo astronaut Harrison Schmitt, emphasize helium-3 concentrations estimated at 10 to 20 parts per billion in regolith, suggesting potential yields of millions of tons across the lunar surface to fuel terrestrial power needs.[68] However, these projections overlook the causal chain of extraction: processing regolith at scale would require heating billions of tons to release microgram quantities per ton, demanding energy inputs far exceeding current solar or nuclear capabilities on the Moon without massive infrastructure investment.[64] Empirical data underscores the limits of helium-3 viability, as commercial fusion reactors remain decades away from practical deployment, with no operational aneutronic fusion systems demonstrating net energy gain as of 2025.[58] Economic analyses indicate that even optimistic mining scenarios yield costs of $1-3 billion per kilogram returned to Earth, dwarfing terrestrial helium-3 prices under $20,000 per liter due to abundant substitutes like deuterium-tritium fusion pathways.[68] Peer-reviewed feasibility studies highlight that end-to-end missions, including excavation, separation, and return, face insurmountable scaling issues without prior lunar industrialization, rendering near-term profitability implausible.[178] Lunar water ice, hyped as a propellant precursor via electrolysis into hydrogen and oxygen, is confined primarily to permanently shadowed craters at the south pole, with radar and spectroscopic surveys estimating totals between 100 million and 600 million metric tons globally but confirming accessible deposits in only select sites like those near Shackleton Crater.[179] Accessibility challenges include excavation in frigid, low-gravity environments where ice-saturated regolith resists mechanical processing, as laboratory tests with simulants show increased cohesion reducing excavator efficiency by up to 50% at 5-10% ice content.[180] In-situ extraction trials, such as NASA's Light WAVE system, have demonstrated small-scale vaporization but require sustained power inputs of kilowatts per kilogram, with losses from sublimation and incomplete recovery limiting yields to under 50% efficiency.[181] Regolith, comprising 40-45% oxygen by weight and silicates amenable to reduction, offers in-situ potential for life support and building materials, yet beneficiation processes like carbothermal or hydrogen reduction demand temperatures above 900°C and consume 10-20 kWh per kilogram of oxygen, straining nascent lunar power grids.[182] Benefit-cost models from industry assessments reveal that while local use could offset 20-30% of mission mass via propellant production, full-scale operations necessitate upfront investments exceeding $100 billion for robotics and habitats, with no demonstrated closure of the economic loop absent orbital demand or reduced Earth-launch costs.[183] These limits stem from fundamental physics: the Moon's 6 km/s escape velocity imposes perpetual transport penalties, ensuring that extracted resources retain marginal value until spacefaring infrastructure matures beyond current prototypes.[44]Claims of Lunar "Environmental" Harm
Critics of lunar resource extraction have raised concerns about potential disruptions to the Moon's surface integrity, primarily framed as threats to scientific research and preservation of geological features rather than harm to a biological environment, given the Moon's barren, airless nature.[184] A key claim involves the risk of contaminating water ice deposits in permanently shadowed regions (PSRs) at the lunar poles, where surface ice concentrations have been mapped by instruments like NASA's Lunar Reconnaissance Orbiter.[185] Astronomers warned NASA in January 2024 that the planned influx of dozens of commercial and governmental landers could deposit dust or propellants, potentially altering or obscuring these volatile resources essential for future in-situ utilization.[185] Such activities might jeopardize astrobiological studies or isotopic analyses by introducing Earth-sourced contaminants, though the Moon's vacuum and low temperatures limit widespread dispersion.[82] Another set of claims targets the generation of lunar dust during mining operations, which could electrostatically levitate and abrade equipment, obscure views for astronomical observatories, or blanket nearby sites, thereby degrading opportunities for pristine surface studies.[186] For helium-3 extraction proposals, a 1992 NASA assessment identified three potential effects: visual alterations from open mining pits covering small surface areas (estimated at less than 0.1% for large-scale operations), temporary dust injection into the exosphere that settles rapidly due to lack of atmosphere, and accumulation of solid waste from processing, potentially requiring burial to avoid interference.[187] Proponents of these concerns, often from academic and advocacy circles, argue for regulatory safeguards akin to terrestrial environmental impact assessments, citing risks to "lunar heritage" sites like Apollo landing areas.[188] These claims, however, rest on anthropocentric valuations of scientific access rather than evidence of intrinsic ecological damage, as the Moon hosts no known life forms, hydrological cycles, or stable atmosphere susceptible to pollution in the Earthly sense. Empirical data from past missions, such as the Apollo program's regolith disturbances, show negligible long-term propagation of effects beyond localized areas, with dust settling within hours to days under lunar gravity and solar wind influences.[146] Sources advancing strong "harm" narratives, including media reports amplifying astronomer petitions, may reflect institutional preferences for conservation over commercialization, potentially overlooking scalable mitigation technologies like enclosed mining or dust suppression systems already under NASA development.[185][184] In practice, resource activities could enhance lunar knowledge through generated data on regolith dynamics, outweighing preservational losses when weighed against first-order causal realities of the Moon's geophysical stability.[187]Equity Arguments and Property Rights Disputes
The Outer Space Treaty of 1967 prohibits national appropriation of celestial bodies by claim of sovereignty, use, or occupation, yet permits their exploration and use for peaceful purposes, creating ambiguity regarding property rights over extracted lunar resources. This framework interprets resource extraction as allowable without granting ownership of the lunar surface itself, akin to non-appropriative use of high seas fisheries, where harvested resources become private property post-extraction. The 2015 U.S. Commercial Space Launch Competitiveness Act explicitly authorizes U.S. citizens to possess, own, transport, and sell extracted space resources, a position echoed in national laws of Luxembourg (2017) and the United Arab Emirates (2021), prioritizing commercial incentives over international redistribution.[189] Equity arguments emphasize that lunar resources, as part of the "common heritage of mankind" under the 1979 Moon Agreement, should yield benefits shared internationally, particularly with non-spacefaring developing nations to avoid replicating terrestrial extractive inequalities. Ratified by only 18 states—none of which are major space powers like the U.S., Russia, or China—the Agreement mandates an international regime for resource exploitation to ensure equitable distribution, reflecting concerns from Global South advocates that advanced nations' dominance in mining helium-3 or water ice would exacerbate global disparities without technology transfer or profit-sharing.[190][191] Proponents argue this principle aligns with first-mover disadvantages for poorer states lacking launch capabilities, potentially requiring mechanisms like a Space Resources Fund for monetary benefit-sharing from commercial operations.[192] Critics, including U.S. policymakers, counter that mandatory sharing would deter investment, as evidenced by the Moon Agreement's failure to attract participants and subsequent stagnation in regulated resource regimes.[193] Property rights disputes intensify under competing frameworks like the 2020 Artemis Accords, signed by over 40 nations including the U.S., which endorse "safety zones" around operations to prevent interference and affirm resource extraction rights consistent with the Outer Space Treaty, but without endorsing the Moon Agreement's heritage clause. Non-signatories such as China and Russia, pursuing the International Lunar Research Station, view Artemis as enabling de facto exclusivity, prompting calls for UN-led governance to enforce transparency and non-appropriation.[194] Australia's dual adherence to both the Moon Agreement and Artemis Accords highlights tensions, as the former demands benefit-sharing while the latter prioritizes operational norms, underscoring risks of fragmented claims leading to conflicts over prime sites like lunar south pole water deposits.[195] Empirical precedents from Antarctic resource moratoriums suggest that absent clear rights, "who dares, wins" dynamics favor technologically advanced actors, potentially sidelining equity without binding multilateral enforcement.[196]References
- https://www.jpl.[nasa](/page/NASA).gov/news/ice-confirmed-at-the-moons-poles/
- https://www.pnas.[org](/page/.org)/doi/10.1073/pnas.1802345115
- https://www.[nasa](/page/NASA).gov/overview-in-situ-resource-utilization/
- https://science.[nasa](/page/NASA).gov/moon/composition/
- https://www.lpi.usra.edu/publications/[books](/page/The_Books)/lunar_sourcebook/pdf/Chapter07.pdf
- https://www.[nasa](/page/NASA).gov/wp-content/uploads/2019/04/05_1_snoble_thelunarregolith.pdf
- https://www.[sciencedirect](/page/ScienceDirect).com/science/article/pii/S003206331930162X
- https://ntrs.[nasa](/page/NASA).gov/api/citations/20110016233/downloads/20110016233.pdf
- https://www.[nasa](/page/NASA).gov/isru-pilot-excavator/
- https://interestingengineering.[com](/page/.com)/innovation/nasas-powerful-moon-mining-robot-could-dig-10000-kg-of-soil-in-a-single-lunar-day


