Recent from talks
Nothing was collected or created yet.
Pregnancy
View on Wikipedia
| Pregnancy | |
|---|---|
| Other names | Gestation |
 | |
| A woman in the third trimester of pregnancy | |
| Specialty | Obstetrics, midwifery |
| Symptoms | Missed periods, tender breasts, nausea and vomiting, hunger, frequent urination[1] |
| Complications | Miscarriage, high blood pressure of pregnancy, gestational diabetes, iron-deficiency anemia, severe nausea and vomiting[2][3] |
| Duration | ~40 weeks from the last menstrual period (38 weeks after conception)[4][5] |
| Diagnostic method | Pregnancy test[6] |
| Prevention | Birth control (including emergency contraception)[7] |
| Frequency | 213 million (2012)[8] |
| Deaths | |
Pregnancy is the time during which one or more offspring gestates inside a woman's uterus.[4][10] A multiple pregnancy involves more than one offspring, such as with twins.[11]
Conception usually occurs following vaginal intercourse, but can also occur through assisted reproductive technology procedures.[12] A pregnancy may end in a live birth, a miscarriage, an induced abortion, or a stillbirth. Childbirth typically occurs around 40 weeks from the start of the last menstrual period (LMP), a span known as the gestational age;[4][5] this is just over nine months. Counting by fertilization age, the length is about 38 weeks.[5][10] Implantation occurs on average 8–9 days after fertilization.[13] An embryo is the term for the developing offspring during the first seven weeks following implantation (i.e. ten weeks' gestational age), after which the term fetus is used until the birth of a baby.[5]
Signs and symptoms of early pregnancy may include missed periods, tender breasts, morning sickness (nausea and vomiting), hunger, implantation bleeding, and frequent urination.[1] Pregnancy may be confirmed with a pregnancy test.[6] Methods of "birth control"—or, more accurately, contraception—are used to avoid pregnancy.
Pregnancy is divided into three trimesters of approximately three months each. The first trimester includes conception, which is when the sperm fertilizes the egg. The fertilized egg then travels down the fallopian tube and attaches to the inside of the uterus, where it begins to form the embryo and placenta. During the first trimester, the possibility of miscarriage (natural death of embryo or fetus) is at its highest. Around the middle of the second trimester, movement of the fetus may be felt. At 28 weeks, more than 90% of babies can survive outside of the uterus if provided with high-quality medical care, though babies born at this time will likely experience serious health complications such as heart and respiratory problems and long-term intellectual and developmental disabilities.
Prenatal care improves pregnancy outcomes.[14] Nutrition during pregnancy is important to ensure healthy growth of the fetus.[15] Prenatal care also include avoiding recreational drugs (including tobacco and alcohol), taking regular exercise, having blood tests, and regular physical examinations.[14] Complications of pregnancy may include disorders of high blood pressure, gestational diabetes, iron-deficiency anemia, and severe nausea and vomiting.[3] In the ideal childbirth, labour begins on its own "at term".[16] Babies born before 37 weeks are "preterm" and at higher risk of health problems such as cerebral palsy.[4] Babies born between weeks 37 and 39 are considered "early term" while those born between weeks 39 and 41 are considered "full term".[4] Babies born between weeks 41 and 42 weeks are considered "late-term" while after 42 weeks they are considered "post-term".[4] Delivery before 39 weeks by labour induction or caesarean section is not recommended unless required for other medical reasons.[17]
Terminology
[edit]
Associated terms for pregnancy are gravid and parous. Gravidus and gravid come from the Latin word meaning "heavy" and a pregnant female is sometimes referred to as a gravida.[18] Gravidity refers to the number of times that a female has been pregnant. Similarly, the term parity is used for the number of times that a female carries a pregnancy to a viable stage.[19] Twins and other multiple births are counted as one pregnancy and birth.
A woman who has never been pregnant is referred to as a nulligravida. A woman who is (or has been only) pregnant for the first time is referred to as a primigravida,[20] and a woman in subsequent pregnancies as a multigravida or as multiparous.[18][21] Therefore, during a second pregnancy a woman would be described as gravida 2, para 1 and upon live delivery as gravida 2, para 2. In-progress pregnancies, abortions, miscarriages and/or stillbirths account for parity values being less than the gravida number. Women who have never carried a pregnancy more than 20 weeks are referred to as nulliparous.[22]
A pregnancy is considered term at 37 weeks of gestation. It is preterm if less than 37 weeks and post-term at or beyond 42 weeks of gestation. The American College of Obstetricians and Gynecologists have recommended further division with early term 37 weeks up to 39 weeks, full term 39 weeks up to 41 weeks, and late term 41 weeks up to 42 weeks.[23] The terms preterm and post-term have largely replaced earlier terms of premature and postmature. Preterm and postterm are defined above, whereas premature and postmature have historical meaning and relate more to the infant's size and state of development rather than to the stage of pregnancy.[24][25]
Demographics
[edit]About 213 million pregnancies occurred in 2012, of which, 190 million (89%) were in the developing world and 23 million (11%) were in the developed world.[8] The number of pregnancies in women aged between 15 and 44 is 133 per 1,000 women.[8] Pregnancy rates are 140 per 1000 women of childbearing age in the developing world and 94 per 1000 in the developed world.[8] The rate of pregnancy, as well as the ages at which it occurs, differ by country and region. It is influenced by a number of factors, such as cultural, social and religious norms; access to contraception; and rates of education. The total fertility rate (TFR) in 2024 was estimated to be highest in Niger (6.64 children/woman) and lowest in South Korea (1.12 children/woman).[26]
About 10% to 15% of recognized pregnancies end in miscarriage.[2] In 2016, complications of pregnancy resulted in 230,600 maternal deaths, down from 377,000 deaths in 1990.[9] Common causes include bleeding, infections, hypertensive diseases of pregnancy, obstructed labour, miscarriage, abortion, or ectopic pregnancy.[9] Globally, 44% of pregnancies are unplanned.[27] Over half (56%) of unplanned pregnancies are aborted.[27] In countries where abortion is prohibited, or only carried out in circumstances where the mother's life is at risk, 48% of unplanned pregnancies are aborted illegally. Compared to the rate in countries where abortion is legal, at 69%.[27] Among unintended pregnancies in the United States, 60% of the women used birth control to some extent during the month pregnancy began.[28][needs update]
In the United States, a woman's educational attainment and her marital status are historically correlated with childbearing: the percentage of women unmarried at the time of first birth drops with increasing educational level. Three studies conducted between 2015 and 2018 indicate a large fraction (~80%) of women without a high school diploma or local equivalent in the US are unmarried at the time of their first birth. By contrast, the same studies indicated fewer women with a bachelor's degree or higher (~24%) have their first child while unmarried. However, this phenomenon also has a strong generational component: a 1996 study found 48.2% of US women without a bachelor's degree had their first child whilst unmarried, and only 4% of women with a bachelor's degree had their first child whilst unmarried. These studies indicate a rising trend for US women of all educational levels to be unmarried at the time of their first birth.[29]
Teenage pregnancy
[edit]Teenage pregnancy is also known as adolescent pregnancy.[30] The WHO defines adolescence as the period between the ages of 10 and 19 years.[31] Adolescents face higher health risks than women who give birth at age 20 to 24 and their infants are at a higher risk for preterm birth, low birth weight, and other severe neonatal conditions. Their children continue to face greater challenges, both behavioral and physical, throughout their lives. Teenage pregnancies are also related to social issues, including social stigma, lower educational levels, and poverty.[32][30] Female adolescents are often in abusive relationships at the time of their conceiving.[33]
Diagnosis
[edit]The beginning of pregnancy may be detected either based on symptoms by the woman herself, or by using pregnancy tests. However, an important condition with serious health implications that is quite common is the denial of pregnancy by the pregnant woman. About 1 in 475 denials will last until around the 20th week of pregnancy. The proportion of cases of denial, persisting until delivery is about 1 in 2500.[34] Conversely, some non-pregnant women have a very strong belief that they are pregnant along with some of the physical changes. This condition is known as a false pregnancy.[35]
Symptoms and signs
[edit]
Most pregnant women experience a number of symptoms which can signify pregnancy[36] such as breast tenderness[11] or morning sickness. A number of early medical signs are associated with pregnancy.[37][38] Physical signs of pregnancy include:
- the presence of human chorionic gonadotropin (hCG) in the blood and urine
- missed menstrual period
- implantation bleeding that occurs at implantation of the embryo in the uterus during the third or fourth week after last menstrual period[39]
- increased basal body temperature sustained for over two weeks after ovulation
- Chadwick's sign (bluish discolouration of the cervix, vagina, and vulva)
- Goodell's sign (softening of the vaginal portion of the cervix)
- Hegar's sign (softening of the uterine isthmus)
- Pigmentation of the linea alba, called linea nigra (darkening of the skin in a midline of the abdomen, resulting from hormonal changes, usually appearing around the middle of pregnancy).[37][38]
- Darkening of the nipples and areolas due to an increase in hormones[40]
- Stretch marks
- Varicose veins
- Peripheral edema (swelling of legs, feet, and ankles)[41]
- Gum bleeding[42]
- Melasma
Other common symptoms include constipation, back pain, pelvic girdle pain, headaches,[42] and food cravings or food aversions.[39] Pregnant women may also experience urinary tract infections,[43] increased urinary frequency,[44] worsened sleep quality, increased dream recall, and nightmares.[45] In later pregnancy, hemorrhoids are more common.[46] Each person's pregnancy can be different and many women do not experience all of the common signs and symptoms.[47] The usual signs and symptoms of pregnancy do not significantly interfere with activities of daily living or pose a health-threat to the mother or fetus.[47] Complications during pregnancy can cause other more severe symptoms, such as those associated with anemia.[48]
Biomarkers
[edit]Pregnancy detection can be accomplished using one or more various pregnancy tests,[49] which detect hormones generated by the newly formed placenta, serving as biomarkers of pregnancy.[50] Blood and urine tests can detect pregnancy by 11 and 14 days, respectively, after fertilization.[51][52] Blood pregnancy tests are more sensitive than urine tests (giving fewer false negatives).[53] Home pregnancy tests are urine tests, and normally detect a pregnancy 12 to 15 days after fertilization.[54] A quantitative blood test can determine approximately the date the embryo was fertilized because hCG levels double every 36 to 72 hours before 8 weeks' gestation.[55][52] A single test of progesterone levels can also help determine how likely a fetus will survive in those with a threatened miscarriage (bleeding in early pregnancy), but only if the ultrasound result was inconclusive.[56]
Ultrasound
[edit]Obstetric ultrasonography can detect fetal abnormalities, detect multiple pregnancies, and improve gestational dating at 24 weeks.[57] The resultant estimated gestational age and due date of the fetus are slightly more accurate than methods based on last menstrual period.[58] Ultrasound is used to measure the nuchal fold in order to screen for Down syndrome.[59]
Timeline
[edit]| Event | Gestational age
(from the start of the last menstrual period) |
Fertilization age | Implantation age |
|---|---|---|---|
| Menstrual period begins | Day 1 of pregnancy | Not pregnant | Not pregnant |
| Has sex and ovulates | 2 weeks pregnant | Not pregnant | Not pregnant |
| Fertilization; cleavage stage begins[60] | Day 15[60] | Day 1[60][61] | Not pregnant |
| Implantation of blastocyst begins | Day 20 | Day 6[60][61] | Day 0 |
| Implantation finished | Day 26 | Day 12[60][61] | Day 6 (or Day 0) |
| Embryo stage begins; also, first missed period | 4 weeks | Day 15[60] | Day 9 |
| Primitive heart function can be detected | 5 weeks, 5 days[60] | Day 26[60] | Day 20 |
| Fetal stage begins | 10 weeks, 1 day[60] | 8 weeks, 1 day[60] | 7 weeks, 2 days |
| First trimester ends | 13 weeks | 11 weeks | 10 weeks |
| Second trimester ends | 26 weeks | 24 weeks | 23 weeks |
| Childbirth | 39–40 weeks | 37–38 weeks[61]: 108 | 36–37 weeks |
The chronology of pregnancy is, unless otherwise specified, generally given as gestational age, where the starting point is the beginning of the woman's last menstrual period (LMP), or the corresponding age of the gestation as estimated by a more accurate method if available. This model means that the woman is counted as being "pregnant" two weeks before conception and three weeks before implantation. Sometimes, timing may also use the fertilization age, which is the age of the embryo since conception.
Start of gestational age
[edit]The American Congress of Obstetricians and Gynecologists recommends the following methods to calculate gestational age:[62]
- Directly calculating the days since the beginning of the last menstrual period.
- Early obstetric ultrasound, comparing the size of an embryo or fetus to that of a reference group of pregnancies of known gestational age (such as calculated from last menstrual periods), and using the mean gestational age of other embryos or fetuses of the same size. If the gestational age as calculated from an early ultrasound is contradictory to the one calculated directly from the last menstrual period, it is still the one from the early ultrasound that is used for the rest of the pregnancy.[62]
- In case of in vitro fertilization, calculating days since oocyte retrieval or co-incubation and adding 14 days.[63]
Trimesters
[edit]Pregnancy is divided into three trimesters, each lasting for approximately three months.[4] The exact length of each trimester can vary between sources.
- The first trimester begins with the start of gestational age as described above, that is, the beginning of week 1, or 0 weeks + 0 days of gestational age (GA). It ends at week 12 (11 weeks + 6 days of GA)[4] or end of week 14 (13 weeks + 6 days of GA).[64]
- The second trimester is defined as starting, between the beginning of week 13 (12 weeks +0 days of GA)[4] and beginning of week 15 (14 weeks + 0 days of GA).[64] It ends at the end of week 27 (26 weeks + 6 days of GA)[64] or end of week 28 (27 weeks + 6 days of GA).[4]
- The third trimester is defined as starting, between the beginning of week 28 (27 weeks + 0 days of GA)[64] or beginning of week 29 (28 weeks + 0 days of GA).[4] It lasts until childbirth.

Estimation of due date
[edit]
Due date estimation basically follows two steps:
- Determination of which time point is to be used as origin for gestational age, as described in the section above.
- Adding the estimated gestational age at childbirth to the above time point. Childbirth on average occurs at a gestational age of 280 days (40 weeks), which is therefore often used as a standard estimation for individual pregnancies.[66] However, alternative durations as well as more individualized methods have also been suggested.
The American College of Obstetricians and Gynecologists divides full term into three divisions:[67]
- Early-term: 37 weeks and 0 days through 38 weeks and 6 days
- Full-term: 39 weeks and 0 days through 40 weeks and 6 days
- Late-term: 41 weeks and 0 days through 41 weeks and 6 days
- Post-term: greater than or equal to 42 weeks and 0 days
Naegele's rule is a standard way of calculating the due date for a pregnancy when assuming a gestational age of 280 days at childbirth. The rule estimates the expected date of delivery (EDD) by adding a year, subtracting three months, and adding seven days to the origin of gestational age. Alternatively there are mobile apps, which essentially always give consistent estimations compared to each other and correct for leap year, while pregnancy wheels made of paper can differ from each other by 7 days and generally do not correct for leap year.[68]
Furthermore, actual childbirth has only a certain probability of occurring within the limits of the estimated due date. A study of singleton live births came to the result that childbirth has a standard deviation of 14 days when gestational age is estimated by first trimester ultrasound, and 16 days when estimated directly by last menstrual period.[65]
Physiology
[edit]Capacity
[edit]Fertility and fecundity are the respective capacities to fertilize and establish a clinical pregnancy and have a live birth. Infertility is an impaired ability to establish a clinical pregnancy and sterility is the permanent inability to establish a clinical pregnancy.[69]
The capacity for pregnancy depends on the reproductive system, its development and its variation, as well as on the condition of a person. Anyone who has a functioning female reproductive system, regardless of intersex or transgender identity, is capable of becoming pregnant.
Some people are not capable of becoming pregnant, even with advanced assisted reproductive technology. In some cases, someone might produce viable eggs, but might not have a womb or none that can sufficiently gestate, in which case they will not be able to become pregnant or sustain the pregnancy. Surrogacy is their only option for having genetic children.[70]
Initiation
[edit]
Through an interplay of hormones that includes follicle stimulating hormone that stimulates folliculogenesis and oogenesis creates a mature egg cell, the female gamete. Fertilization is the event where the egg cell fuses with the male gamete, spermatozoon. After the point of fertilization, the fused product of the female and male gamete is referred to as a zygote or fertilized egg. The fusion of female and male gametes usually occurs following the act of sexual intercourse. Pregnancy rates for sexual intercourse are highest during the menstrual cycle time from some 5 days before until 1 to 2 days after ovulation.[71] Fertilization can also occur by assisted reproductive technology such as artificial insemination and in vitro fertilisation.
Fertilization (conception) is sometimes used as the initiation of pregnancy, with the derived age being termed fertilization age. Fertilization usually occurs about two weeks before the next expected menstrual period.
A third point in time is also considered by some people to be the true beginning of a pregnancy: This is time of implantation, when the future fetus attaches to the lining of the uterus. This is about a week to ten days after fertilization.[72]
Development of embryo and fetus
[edit]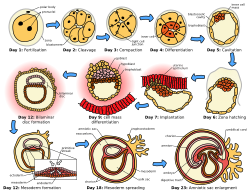
The sperm and the egg cell, which has been released from one of the female's two ovaries, unite in one of the two fallopian tubes. The fertilized egg, known as a zygote, then moves toward the uterus, a journey that can take up to a week to complete. Cell division begins approximately 24 to 36 hours after the female and male cells unite. Cell division continues at a rapid rate and the cells then develop into what is known as a blastocyst. The blastocyst arrives at the uterus and attaches to the uterine wall, a process known as implantation.
The development of the mass of cells that will become the infant is called embryogenesis during the first approximately ten weeks of gestation. During this time, cells begin to differentiate into the various body systems. The basic outlines of the organ, body, and nervous systems are established. By the end of the embryonic stage, the beginnings of features such as fingers, eyes, mouth, and ears become visible. Also during this time, there is development of structures important to the support of the embryo, including the placenta and umbilical cord. The placenta connects the developing embryo to the uterine wall to allow nutrient uptake, waste elimination, and gas exchange via the mother's blood supply. The umbilical cord is the connecting cord from the embryo or fetus to the placenta.
After about ten weeks of gestational age—which is the same as eight weeks after conception—the embryo becomes known as a fetus.[73] At the beginning of the fetal stage, the risk of miscarriage decreases sharply.[74] At this stage, a fetus is about 30 mm (1.2 inches) in length, the heartbeat is seen via ultrasound, and the fetus makes involuntary motions.[75] During continued fetal development, the early body systems, and structures that were established in the embryonic stage continue to develop. Sex organs begin to appear during the third month of gestation. The fetus continues to grow in both weight and length, although the majority of the physical growth occurs in the last weeks of pregnancy.
Electrical brain activity is first detected at the end of week 5 of gestation, but as in brain-dead patients, it is primitive neural activity rather than the beginning of conscious brain activity. Synapses do not begin to form until week 17.[76] Neural connections between the sensory cortex and thalamus develop as early as 24 weeks' gestational age, but the first evidence of their function does not occur until around 30 weeks, when minimal consciousness, dreaming, and the ability to feel pain emerges.[77]
Although the fetus begins to move during the first trimester, it is not until the second trimester that movement, known as quickening, can be felt. This typically happens in the fourth month, more specifically in the 20th to 21st week, or by the 19th week if the woman has been pregnant before. It is common for some women not to feel the fetus move until much later. During the second trimester, when the body size changes, maternity clothes may be worn.
-
Embryo at 4 weeks after fertilization (gestational age of 6 weeks)
-
Fetus at 8 weeks after fertilization (gestational age of 10 weeks)
-
Fetus at 18 weeks after fertilization (gestational age of 20 weeks)
-
Fetus at 38 weeks after fertilization (gestational age of 40 weeks)
-
Relative size in 1st month (simplified illustration)
-
Relative size in 3rd month (simplified illustration)
-
Relative size in 5th month (simplified illustration)
-
Relative size in 9th month (simplified illustration)
Maternal changes
[edit]

During pregnancy, a woman undergoes many normal physiological changes, including behavioral, cardiovascular, hematologic, metabolic, renal, and respiratory changes. Increases in blood sugar, breathing, and cardiac output are all required. Levels of progesterone and estrogens rise continually throughout pregnancy, suppressing the hypothalamic axis and therefore the menstrual cycle. A full-term pregnancy at an early age (less than 25 years) reduces the risk of breast, ovarian, and endometrial cancer, and the risk declines further with each additional full-term pregnancy.[78][79]

The fetus is genetically different from its mother and can therefore be viewed as an unusually successful allograft.[80] The main reason for this success is increased immune tolerance during pregnancy,[81] which prevents the mother's body from mounting an immune system response against certain triggers.[80] A Rho(D) immune globulin shot is recommended for women with RhD negative blood carrying a RhD positive fetus as a preventative measure against Rhesus disease.[82]
During the first trimester, minute ventilation increases by 40 percent.[83] The womb will grow to the size of a lemon by eight weeks. Many symptoms and discomforts of pregnancy, such as nausea and tender breasts, appear in the first trimester.[84] Braxton Hicks contractions are sporadic uterine contractions that may start around six weeks into a pregnancy but are usually not felt until the second or third trimester.[85]
Pregnant women have higher total blood volume that increases throughout the duration of the pregnancy.[86] It is during the third trimester that maternal activity and sleep positions may affect fetal development due to restricted blood flow. For instance, the enlarged uterus may impede blood flow by compressing the vena cava when lying flat, a condition that can be relieved by lying on the left side.[87]
Most weight gain takes place during the third trimester. A pregnant woman's navel may become convex ("popping" out) during this time. Her abdomen will expand and change in shape as the fetus turns in a downward position nearing childbirth.[88] Head engagement, also called "lightening" or "dropping", occurs as the fetal head descends into a cephalic presentation. While it relieves pressure on the upper abdomen and gives a renewed ease in breathing, it also severely reduces bladder capacity, resulting in a need to void more frequently, and increases pressure on the pelvic floor and the rectum. It is not possible to predict when lightening will occur. In a first pregnancy it may happen a few weeks before the due date, though it may happen later or even not until labour begins, as is typical with subsequent pregnancies.[89]
Childbirth
[edit]Childbirth, referred to as labour and delivery in the medical field, is the process whereby an infant is born.[55]
A woman is considered to be in labour when she begins experiencing regular uterine contractions, accompanied by changes of her cervix—primarily effacement and dilation. While childbirth is widely experienced as painful, some women do report painless labours, while others find that concentrating on the birth helps to quicken labour and lessen the sensations. Most births are successful vaginal births, but sometimes complications arise and a woman may undergo a cesarean section.
During the time immediately after birth, both the mother and the baby are hormonally cued to bond, the mother through the release of oxytocin, a hormone also released during breastfeeding. Studies show that skin-to-skin contact between a mother and her newborn immediately after birth is beneficial for both the mother and baby. A review done by the World Health Organization found that skin-to-skin contact between mothers and babies after birth reduces crying, improves mother–infant interaction, and helps mothers to breastfeed successfully. They recommend that neonates be allowed to bond with the mother during their first two hours after birth, the period that they tend to be more alert than in the following hours of early life.[90]
Childbirth maturity stages
[edit]
| stage | starts | ends |
|---|---|---|
| Preterm[91] | - | at 37 weeks |
| Early term[92] | 37 weeks | 39 weeks |
| Full term[92] | 39 weeks | 41 weeks |
| Late term[92] | 41 weeks | 42 weeks |
| Postterm[92] | 42 weeks | - |
In the ideal childbirth, labour begins on its own when a woman is "at term".[16] Events before completion of 37 weeks are considered preterm.[91] Preterm birth is associated with a range of complications and should be avoided if possible.[93]
Sometimes if a woman's water breaks or she has contractions before 39 weeks, birth is unavoidable.[92] However, spontaneous birth after 37 weeks is considered term and is not associated with the same risks of a preterm birth.[55] Planned birth before 39 weeks by caesarean section or labour induction, although "at term", results in an increased risk of complications.[94] This is from factors including underdeveloped lungs of newborns, infection due to underdeveloped immune system, feeding problems due to underdeveloped brain, and jaundice from underdeveloped liver.[95]
Babies born between 39 and 41 weeks' gestation have better outcomes than babies born either before or after this range.[92] This special time period is called "full term".[92] Whenever possible, waiting for labour to begin on its own in this time period is best for the health of the mother and baby.[16] The decision to perform an induction must be made after weighing the risks and benefits, but is safer after 39 weeks.[16]
Events after 42 weeks are considered postterm.[92] When a pregnancy exceeds 42 weeks, the risk of complications for both the woman and the fetus increases significantly.[96][97] Therefore, in an otherwise uncomplicated pregnancy, obstetricians usually prefer to induce labour at some stage between 41 and 42 weeks.[98]
Postnatal period
[edit]The postpartum period also referred to as the puerperium, is the postnatal period that begins immediately after delivery and extends for about six weeks.[55] During this period, the mother's body begins the return to pre-pregnancy conditions that includes changes in hormone levels and uterus size.[55]
Management
[edit]
Prenatal care
[edit]Pre-conception counseling is care that is provided to a woman or couple to discuss conception, pregnancy, current health issues and recommendations for the period before pregnancy.[101]
Prenatal medical care is the medical and nursing care recommended for women during pregnancy, time intervals and exact goals of each visit differ by country.[102] Women who are high risk have better outcomes if they are seen regularly and frequently by a medical professional than women who are low risk.[103] A woman can be labeled as high risk for different reasons including previous complications in pregnancy, complications in the current pregnancy, current medical diseases, or social issues.[104][105]
The aim of good prenatal care is prevention, early identification, and treatment of any medical complications.[106] A basic prenatal visit consists of measurement of blood pressure, fundal height, weight and fetal heart rate, checking for symptoms of labour, and guidance for what to expect next.[101] Healthcare providers may screen for domestic violence during pregnancy, particularly in regards to reproductive coercion.[107]
Nutrition
[edit]Nutrition during pregnancy is important to ensure healthy growth of the fetus.[15] Nutrition during pregnancy is different from the non-pregnant state.[15] There are increased energy requirements and specific micronutrient requirements.[15] Women benefit from education to encourage a balanced energy and protein intake during pregnancy.[108] Some women may need professional medical advice if their diet is affected by medical conditions, food allergies, or specific religious or ethical beliefs.[109] Further studies are needed to access the effect of dietary advice to prevent gestational diabetes, although low quality evidence suggests some benefit.[110] Adequate periconceptional (time before and right after conception) folic acid (also called folate or Vitamin B9) intake has been shown to decrease the risk of fetal neural tube defects, such as spina bifida.[111] L-methylfolate, the bioavailable form of folate is also considered acceptable to take. L-methylfolate is best used by the 40% to 60% of the population with genetic polymorphisms that reduce or impair conversion of folic acid into its active form.[112] The neural tube develops during the first 28 days of pregnancy, a urine pregnancy test is not usually positive until 14 days post-conception, explaining the necessity to guarantee adequate folate intake before conception.[54][113] Folate is abundant in green leafy vegetables, legumes, and citrus.[114] In the United States and Canada, most wheat products (flour, noodles) are fortified with folic acid.[115]
Weight gain
[edit]
The amount of healthy weight gain during a pregnancy varies.[116] Weight gain is related to the weight of the baby, the placenta, extra circulatory fluid, larger tissues, and fat and protein stores.[15] Most needed weight gain occurs later in pregnancy.[117]
The Institute of Medicine recommends an overall pregnancy weight gain for those of normal weight (body mass index of 18.5–24.9), of 11.3–15.9 kg (25–35 pounds) having a singleton pregnancy.[118] Women who are underweight (BMI of less than 18.5), should gain between 12.7 and 18 kg (28–40 lb), while those who are overweight (BMI of 25–29.9) are advised to gain between 6.8 and 11.3 kg (15–25 lb) and those who are obese (BMI ≥ 30) should gain between 5–9 kg (11–20 lb).[119] These values reference the expectations for a term pregnancy.
During pregnancy, insufficient or excessive weight gain can compromise the health of the mother and fetus.[117] The most effective intervention for weight gain in underweight women is not clear.[117] Being or becoming overweight in pregnancy increases the risk of complications for mother and fetus, including cesarean section, gestational hypertension, pre-eclampsia, macrosomia and shoulder dystocia.[116] Excessive weight gain can make losing weight after the pregnancy difficult.[116][120] Some of these complications are risk factors for stroke.[121]
Around 50% of women of childbearing age in developed countries like the United Kingdom are overweight or obese before pregnancy.[120] Diet modification is the most effective way to reduce weight gain and associated risks in pregnancy.[120]
Medication
[edit]Drugs used during pregnancy can have temporary or permanent effects on the fetus.[122] Anything (including drugs) that can cause permanent deformities in the fetus are labeled as teratogens.[123] In the U.S., drugs were classified into categories A, B, C, D and X based on the Food and Drug Administration (FDA) rating system to provide therapeutic guidance based on potential benefits and fetal risks.[124] Drugs, including some multivitamins, that have demonstrated no fetal risks after controlled studies in humans are classified as Category A.[122] On the other hand, drugs like thalidomide with proven fetal risks that outweigh all benefits are classified as Category X.[122]
Recreational drugs
[edit]The use of recreational drugs in pregnancy can cause various pregnancy complications.[55]
- Alcoholic drinks consumed during pregnancy can cause one or more fetal alcohol spectrum disorders.[55] According to the CDC, there is no known safe amount of alcohol during pregnancy and no safe time to drink during pregnancy, including before a woman knows that she is pregnant.[125]
- Tobacco smoking during pregnancy can cause a wide range of behavioral, neurological, and physical difficulties.[126] Smoking during pregnancy causes twice the risk of premature rupture of membranes, placental abruption and placenta previa.[127] Smoking is associated with 30% higher odds of preterm birth.[128]
- Prenatal cocaine exposure is associated with premature birth, birth defects and attention deficit disorder.[55]
- Prenatal methamphetamine exposure can cause premature birth and congenital abnormalities.[129] Short-term neonatal outcomes in methamphetamine babies show small deficits in infant neurobehavioral function and growth restriction.[130] Long-term effects in terms of impaired brain development may also be caused by methamphetamine use.[129]
- Cannabis in pregnancy has been shown to be teratogenic in large doses in animals, but has not shown any teratogenic effects in humans.[55]
Exposure to toxins
[edit]Intrauterine exposure to environmental toxins in pregnancy has the potential to cause adverse effects on prenatal development, and to cause pregnancy complications.[55] Air pollution has been associated with low birth weight infants.[131] Conditions of particular severity in pregnancy include mercury poisoning and lead poisoning.[55] To minimize exposure to environmental toxins, the American College of Nurse-Midwives recommends: checking whether the home has lead paint, washing all fresh fruits and vegetables thoroughly and buying organic produce, and avoiding cleaning products labeled "toxic" or any product with a warning on the label.[132]
Pregnant women can also be exposed to toxins in the workplace, including airborne particles. The effects of wearing an N95 filtering facepiece respirator are similar for pregnant women as for non-pregnant women, and wearing a respirator for one hour does not affect the fetal heart rate.[133]
Death by violence
[edit]Pregnant women or those who have recently given birth in the U.S. are more likely to be murdered than to die from obstetric causes. These homicides are a combination of intimate partner violence and firearms. Health authorities have called the violence "a health emergency for pregnant women", but say that pregnancy-related homicides are preventable if healthcare providers identify those women at risk and offer assistance to them.[134][135][136]
Sexual activity
[edit]Most women can continue to engage in sexual activity, including sexual intercourse, throughout pregnancy.[137] Research suggests that during pregnancy both sexual desire and frequency of sexual relations decrease during the first and third trimester, with a rise during the second trimester.[138][139][140][141] Sex during pregnancy is low-risk except when the healthcare provider advises that sexual intercourse be avoided for particular medical reasons.[137] For a healthy pregnant woman, there is no single safe or right way to have sex during pregnancy.[137]
Exercise
[edit]
Regular aerobic exercise during pregnancy appears to improve (or maintain) physical fitness.[142] Physical exercise during pregnancy appears to decrease the need for C-section[143] and reduce time in labour,[144] and even vigorous exercise carries no significant risks to babies[145] while providing significant health benefits to the mother. Studies show that performing light moderate intensity and strength exercises while pregnant does not harm the mother's cardiovascular system and may limit excessive weight gain.[146][additional citation(s) needed]
The American College of Sports and Medicine recommends pregnant women should participate in at least 150 minutes/week of moderate exercise.[147] These forms of exercise should avoid heavy lifting, hot temperatures, and high impact sports. The Clinical Practice Obstetrics Committee of Canada recommends that "All women without contraindications should be encouraged to participate in aerobic and strength-conditioning exercises as part of a healthy lifestyle during their pregnancy".[148] Although an upper level of safe exercise intensity has not been established, women who were regular exercisers before pregnancy and who have uncomplicated pregnancies should be able to engage in high intensity exercise programs without a higher risk of prematurity, lower birth weight, or gestational weight gain.[145] In general, participation in a wide range of recreational activities appears to be safe, with the avoidance of those with a high risk of falling such as horseback riding or skiing or those that carry a risk of abdominal trauma, such as soccer or hockey.[149]
Bed rest, outside of research studies, is not recommended as there is potential harm and no evidence of benefit.[150]
High intensity exercise
[edit]During pregnancy, women can experience a loss of postural stability, pelvic incontinence, back pain, and fatigue, among other symptoms.[citation needed] Resistance training has been found to reduce pregnancy symptoms and reduce postpartum complications.[citation needed] Provided that women also regularly participate in low-impact training, strength training can improve pelvic girdle pain severity postpartum.[151] When incorporating exercises that focus on pelvic muscle strength, they can help reduce pain and stress urinary incontinence.[151]
Engaging in regular exercise and physical activity has been shown to be beneficial during pregnancy. Acute bouts of high intensity interval training can help decrease the risks of health complications associated with pregnancy, maintain a healthy body fat percentage during pregnancy, as well as improve overall well-being.[152] Pregnant women who participated in high intensity interval training have been shown to undergo physical improvements in body composition after intervention as well as show general improvement in cardiorespiratory fitness and exercise tolerance.[153] Taking part in this style of exercise, similarly to moderate intensity continuous training, has also been shown to improve glycemic response and insulin sensitivity.[154] There are specific concerns to be avoided with exercise during pregnancy such as overheating, fall-risk, and remaining in a supine position for an extended period of time. Inexperienced individuals new to high-intensity interval training could potentially increase their risk for negative conditions associated with hypertension, such as pre-eclampsia.[155]
Sleep
[edit]It has been suggested that shift work and exposure to bright light at night should be avoided at least during the last trimester of pregnancy to decrease the risk of psychological and behavioral problems in the newborn.[156]
Stress
[edit]The children of women who had high stress levels during pregnancy are slightly more likely to have externalizing behavioral problems such as impulsivity.[153] The behavioral effect was most pronounced during early childhood.[153]
Dental care
[edit]The increased levels of progesterone and estrogen during pregnancy make gingivitis more likely; the gums become edematous, red in colour, and tend to bleed.[157] Also a pyogenic granuloma or "pregnancy tumor", is commonly seen on the labial surface of the papilla. Lesions can be treated by local debridement or deep incision depending on their size, and by following adequate oral hygiene measures.[158] There have been suggestions that severe periodontitis may increase the risk of having preterm birth and low birth weight; however, a Cochrane review found insufficient evidence to determine if periodontitis can develop adverse birth outcomes.[159]
Flying
[edit]In low risk pregnancies, most health care providers approve flying until about 36 weeks of gestational age.[160] Most airlines allow pregnant women to fly short distances at less than 36 weeks, and long distances at less than 32 weeks.[161] Many airlines require a doctor's note that approves flying, especially at over 28 weeks.[161] During flights, the risk of deep vein thrombosis is decreased by getting up and walking occasionally, as well as by avoiding dehydration. The exposure to cosmic radiation is negligible for most travelers. For pregnant women, even the longest intercontinental fight would expose them less than 15% of both the NCRPM and ICRP limit.[162][161] Full body scanners use non-ionizing radiation that does not penetrate the body for more than 1 mm, and are believed not to pose a risk in pregnancy.[citation needed]
Pregnancy classes and birth plan
[edit]To prepare for the birth of the baby, health care providers recommend that parents attend antenatal classes during the third trimester of pregnancy. Classes include information about the process of labour and birth and the various kinds of births, including both vaginal and caesarean delivery, the use of forceps, and other interventions that may be needed to safely deliver the infant. Types of pain relief, including relaxation techniques, are discussed. Partners or others who may plan to support a woman during her labour and delivery learn how to assist in the birth.[citation needed]
It is also suggested that a birth plan be written at this time. A birth plan is a written statement that outlines the desires of the mother during labour and delivery of the baby. Discussing the birth plan with the midwife or other care provider gives parents a chance to ask questions and learn more about the process of labour.[163]
In 1991 the WHO launched the Baby-Friendly Hospital Initiative, a global program that recognizes birthing centers and hospitals that offer optimal levels of care for giving birth. Facilities that have been certified as "Baby Friendly" accept visits from expecting parents to familiarize them with the facility and the staff.[164]
Complications
[edit]Each year, ill health as a result of pregnancy is experienced (sometimes permanently) by more than 20 million women around the world.[165] In 2016, complications of pregnancy resulted in 230,600 deaths down from 377,000 deaths in 1990.[9] Common causes include bleeding (72,000), infections (20,000), hypertensive diseases of pregnancy (32,000), obstructed labour (10,000), and pregnancy with abortive outcome (20,000), which includes miscarriage, abortion, and ectopic pregnancy.[9]
The following are some examples of pregnancy complications:
- Pregnancy induced hypertension
- Anemia[166]
- Perinatal depression[167]
- Postpartum depression, a common but solvable complication following childbirth that may result from decreased hormonal levels.[168]
- Postpartum psychosis
- Thromboembolic disorders, with an increased risk due to hypercoagulability in pregnancy. These are the leading cause of death in pregnant women in the US.[169][170]
- Pruritic urticarial papules and plaques of pregnancy (PUPPP), a skin disease that develops around the 32nd week. Signs are red plaques, papules, and itchiness around the belly button that then spreads all over the body except for the inside of hands and face.
- Ectopic pregnancy, including abdominal pregnancy, implantation of the embryo outside the uterus
- Hyperemesis gravidarum, excessive nausea and vomiting that is more severe than normal morning sickness.
- Pulmonary embolism, a blood clot that forms in the legs and migrates to the lungs.[170]
- Acute fatty liver of pregnancy is a rare complication thought to be brought about by a disruption in the metabolism of fatty acids by mitochondria.
There is also an increased susceptibility and severity of certain infections in pregnancy.
Miscarriage and stillbirth
[edit]Miscarriage is the most common complication of early pregnancy. It is defined as the loss of an embryo or fetus before it is able to survive independently. The most common symptom of miscarriage is vaginal bleeding with or without pain. The miscarriage may be evidenced by a clot-like material passing through and out of the vagina.[171] About 80% of miscarriages occur in the first 12 weeks of pregnancy. The underlying cause in about half of cases involves chromosomal abnormalities.[172]
Stillbirth is defined as fetal death after 20 or 28 weeks of pregnancy, depending on the source. It results in a baby born without signs of life. Each year about 21,000 babies are stillborn in the U.S.[173] Sadness, anxiety, and guilt may occur after a miscarriage or a stillbirth. Emotional support may help with processing the loss.[174] Fathers may experience grief over the loss as well. A large study found that there is a need to increase the accessibility of support services available for fathers.[175]
Diseases in pregnancy
[edit]A pregnant woman may have a pre-existing disease, which is not directly caused by the pregnancy, but may cause complications to develop that include a potential risk to the pregnancy; or a disease may develop during pregnancy.
- Diabetes mellitus and pregnancy deals with the interactions of diabetes mellitus (not restricted to gestational diabetes) and pregnancy. Risks for the child include miscarriage, growth restriction, growth acceleration, large for gestational age (macrosomia), polyhydramnios (too much amniotic fluid), and birth defects.
- Thyroid disease in pregnancy can, if uncorrected, cause adverse effects on fetal and maternal well-being. The deleterious effects of thyroid dysfunction can also extend beyond pregnancy and delivery to affect neurointellectual development in the early life of the child. Demand for thyroid hormones is increased during pregnancy, which may cause a previously unnoticed thyroid disorder to worsen.
- Untreated celiac disease can cause a miscarriage, intrauterine growth restriction, small for gestational age, low birthweight and preterm birth. Often reproductive disorders are the only manifestation of undiagnosed celiac disease and most cases are not recognized. Complications or failures of pregnancy cannot be explained simply by malabsorption, but by the autoimmune response elicited by the exposure to gluten, which causes damage to the placenta. A gluten-free diet avoids or reduces the risk of developing reproductive disorders in pregnant women with celiac disease.[176][177] Also, pregnancy can be a trigger for the development of celiac disease in genetically susceptible women who are consuming gluten.[178]
- Lupus in pregnancy confers an increased rate of fetal death in utero, miscarriage, and of neonatal lupus.
- Hypercoagulability in pregnancy is the propensity of pregnant women to develop thrombosis (blood clots). Pregnancy itself is a factor of hypercoagulability (pregnancy-induced hypercoagulability), as a physiologically adaptive mechanism to prevent postpartum bleeding.[179] However, in combination with an underlying hypercoagulable state, the risk of thrombosis or embolism may become substantial.[179]
Abortion
[edit]An abortion is the termination of an embryo or fetus via medical method. It is usually done within the first trimester, sometimes in the second, and rarely in the third. Reasons for pregnancies being undesired are broad.[180] Many jurisdictions restrict or prohibit abortion, with rape being the most legally permissible exception.[181]
Birth control and education
[edit]Family planning, as well as the availability and use of contraception, along with increased comprehensive sex education, has enabled many to prevent pregnancies when they are not desired. Schemes and funding to support education and the means to prevent pregnancies when they are not intended have been instrumental and are part of the third of the Sustainable Development Goals (SDGs) advanced by the United Nations.[182]
Technologies and science
[edit]Assisted reproductive technology
[edit]Modern reproductive medicine offers many forms of assisted reproductive technology for couples who stay childless against their will, such as fertility medication, artificial insemination, in vitro fertilization and surrogacy.
Medical imaging
[edit]

Medical imaging may be indicated in pregnancy because of pregnancy complications, disease, or routine prenatal care. Medical ultrasonography including obstetric ultrasonography, and magnetic resonance imaging (MRI) without contrast agents are not associated with any risk for the mother or the fetus, and are the imaging techniques of choice for pregnant women.[183] Projectional radiography, CT scan and nuclear medicine imaging result in some degree of ionizing radiation exposure, but in most cases the absorbed doses are not associated with harm to the baby.[183] At higher dosages or frequency, effects can include miscarriage, birth defects and intellectual disability.[183]
Legal and social aspects
[edit]Legal protections
[edit]Many countries have various legal regulations in place to protect pregnant women and their children. Many countries have laws against pregnancy discrimination.[184]
The Maternity Protection Convention ensures that pregnant women are exempt from activities such as night shifts or carrying heavy stocks. Maternity leave typically provides paid leave from work during roughly the last trimester of pregnancy and for some time after birth. Notable extreme cases include Norway (8 months with full pay) and the United States (no paid leave at all except in some states).
In the United States, some actions that result in miscarriage or stillbirth, such as beating a pregnant woman, are considered crimes. One law that does so is the federal Unborn Victims of Violence Act. In 2014, the American state of Tennessee passed a law which allows prosecutors to charge a woman with criminal assault if she uses illegal drugs during her pregnancy and her fetus or newborn is harmed as a result.[185]
However, protections are not universal. In Singapore, the Employment of Foreign Manpower Act forbids current and former work permit holders from becoming pregnant or giving birth in Singapore without prior permission.[186][187] Violation of the Act is punishable by a fine of up to S$10,000 (US$7300) and deportation,[186][188] and until 2010, their employers would lose their $5,000 security bond.[189]
Racial disparities
[edit]There are significant racial imbalances in pregnancy and neonatal care systems.[190] Midwifery guidance, treatment, and care have been related to better birth outcomes. Diminishing racial inequities in health is an increasingly large public health challenge in the United States. Despite the fact that average rates have decreased, data on neonatal mortality demonstrates that racial disparities have persisted and grown. The death rate for African American babies is nearly double that of white neonates. According to studies, congenital defects, SIDS, preterm birth, and low birth weight are all more common among African American babies.[191]
Transgender people
[edit]Transgender people have experienced significant advances in societal acceptance in recent years[when?] leaving many health professionals unprepared to provide quality care. A 2015 report suggests that "numbers of transgender individuals who are seeking family planning, fertility, and pregnancy services could certainly be quite large". Regardless of prior hormone replacement therapy treatments, the progression of pregnancy and birthing procedures for transgender people who carry pregnancies are typically the same as those of cisgender women.[192] However, transgender people may be subjected to discrimination, which can include a variety of negative social, emotional, and medical experiences, as pregnancy is regarded as an exclusively female activity. According to a study by the American College of Obstetricians and Gynecologists, there is a lack of awareness, services, and medical assistance available to pregnant trans men.[193]
Culture
[edit]
In most cultures, pregnant women have a special status in society and receive particularly gentle care.[194] At the same time, they are subject to expectations that may exert great psychological pressure, such as having to produce a son and heir. In many traditional societies, pregnancy must be preceded by marriage, on pain of ostracism of mother and (illegitimate) child.
Overall, pregnancy is accompanied by numerous customs that are often subject to ethnological research, often rooted in traditional medicine or religion. The baby shower is an example of a modern custom. Contrary to common misconception, women historically in the United States were not expected to seclude themselves during pregnancy, as was popularized by Gone With the Wind.[195][196]
Pregnancy is an important topic in sociology of the family. The prospective child may preliminarily be placed into numerous social roles. The parents' relationship and the relation between parents and their surroundings are also affected.
A belly cast may be made during pregnancy as a keepsake.
Arts
[edit]Images of pregnant women, especially small figurines, were made in traditional cultures in many places and periods, though it is rarely one of the most common types of image. These include ceramic figures from some Pre-Columbian cultures, and a few figures from most of the ancient Mediterranean cultures. Many of these seem to be connected with fertility. Identifying whether such figures are actually meant to show pregnancy is often a problem, as well as understanding their role in the culture concerned.
Among the oldest surviving examples of the depiction of pregnancy are prehistoric figurines found across much of Eurasia and collectively known as Venus figurines. Some of these appear to be pregnant.
Due to the important role of the Mother of God in Christianity, the Western visual arts have a long tradition of depictions of pregnancy, especially in the biblical scene of the Visitation, and devotional images called a Madonna del Parto.[197]
The unhappy scene usually called Diana and Callisto, showing the moment of discovery of Callisto's forbidden pregnancy, is sometimes painted from the Renaissance onwards. Gradually, portraits of pregnant women began to appear, with a particular fashion for "pregnancy portraits" in elite portraiture of the years around 1600.
Pregnancy, and especially pregnancy of unmarried women, is also an important motif in literature. Notable examples include Thomas Hardy's 1891 novel Tess of the d'Urbervilles and Goethe's 1808 play Faust.
See also
[edit]References
[edit]- ^ a b "What are some common signs of pregnancy?". Eunice Kennedy Shriver National Institute of Child Health and Human Development. 12 July 2013. Archived from the original on 19 March 2015. Retrieved 14 March 2015.
- ^ a b The Johns Hopkins Manual of Gynecology and Obstetrics (4 ed.). Lippincott Williams & Wilkins. 2012. p. 438. ISBN 978-1-4511-4801-5. Archived from the original on 10 September 2017.
- ^ a b "What are some common complications of pregnancy?". Eunice Kennedy Shriver National Institute of Child Health and Human Development. 12 July 2013. Archived from the original on 26 February 2015. Retrieved 14 March 2015.
- ^ a b c d e f g h i j k "Pregnancy: Condition Information". Eunice Kennedy Shriver National Institute of Child Health and Human Development. 19 December 2013. Archived from the original on 19 March 2015. Retrieved 14 March 2015.
- ^ a b c d Abman SH (2011). Fetal and neonatal physiology (4th ed.). Philadelphia: Elsevier/Saunders. pp. 46–47. ISBN 978-1-4160-3479-7.
- ^ a b "How do I know if I'm pregnant?". Eunice Kennedy Shriver National Institute of Child Health and Human Development. 30 November 2012. Archived from the original on 2 April 2015. Retrieved 14 March 2015.
- ^ Taylor D, James EA (2011). "An evidence-based guideline for unintended pregnancy prevention". Journal of Obstetric, Gynecologic, and Neonatal Nursing. 40 (6): 782–793. doi:10.1111/j.1552-6909.2011.01296.x. ISSN 0090-0311. PMC 3266470. PMID 22092349.
- ^ a b c d Sedgh G, Singh S, Hussain R (September 2014). "Intended and unintended pregnancies worldwide in 2012 and recent trends". Studies in Family Planning. 45 (3): 301–314. doi:10.1111/j.1728-4465.2014.00393.x. PMC 4727534. PMID 25207494.
- ^ a b c d e Naghavi M, Abajobir AA, Abbafati C, Abbas KM, Abd-Allah F, Abera SF, et al. (GBD 2016 Causes of Death Collaborators) (September 2017). "Global, regional, and national age-sex specific mortality for 264 causes of death, 1980-2016: a systematic analysis for the Global Burden of Disease Study 2016". Lancet. 390 (10100): 1151–1210. doi:10.1016/S0140-6736(17)32152-9. PMC 5605883. PMID 28919116.
- ^ a b Mosby (2009). Mosby's Pocket Dictionary of Medicine, Nursing & Health Professions - E-Book. Elsevier Health Sciences. p. 1078. ISBN 978-0-323-06604-4.
- ^ a b Wylie L (2005). Essential anatomy and physiology in maternity care (Second ed.). Edinburgh: Churchill Livingstone. p. 172. ISBN 978-0-443-10041-3. Archived from the original on 10 September 2017.
- ^ Shehan CL (2016). The Wiley Blackwell Encyclopedia of Family Studies, 4 Volume Set. John Wiley & Sons. p. 406. ISBN 978-0-470-65845-1. Archived from the original on 10 September 2017.
- ^ Massachusetts General Laws c.112 § 12K: Definitions applicable to Secs. 12L to 12U, Commonwealth of Massachusetts, 2022
- ^ a b "What is prenatal care and why is it important?". Eunice Kennedy Shriver National Institute of Child Health and Human Development. 12 July 2013. Archived from the original on 2 April 2015. Retrieved 14 March 2015.
- ^ a b c d e Lammi-Keefe CJ, Couch SC, Philipson EH, eds. (2008). Handbook of Nutrition and Pregnancy. Nutrition and health. Totowa, NJ: Humana Press. p. 28. doi:10.1007/978-1-59745-112-3. ISBN 978-1-59745-112-3.
- ^ a b c d American Congress of Obstetricians and Gynecologists (February 2013), "Five Things Physicians and Patients Should Question", Choosing Wisely: an initiative of the ABIM Foundation, American Congress of Obstetricians and Gynecologists, archived from the original on 1 September 2013, retrieved 1 August 2013
- ^ World Health Organization (November 2014). "Preterm birth Fact sheet N°363". who.int. Archived from the original on 7 March 2015. Retrieved 6 March 2015.
- ^ a b "definition of gravida". The Free Dictionary. Retrieved 17 January 2008.
- ^ "Gravidity and Parity Definitions (Implications in Risk Assessment)". patient.info. Archived from the original on 12 December 2016.
- ^ Robinson, Victor, ed. (1939). "Primipara". The Modern Home Physician, A New Encyclopedia of Medical Knowledge. WM. H. Wise & Company (New York)., page 596.
- ^ "Definition of nulligravida". Merriam-Webster, Incorporated. Archived from the original on 8 September 2008. Retrieved 9 March 2012.
- ^ "Nulliparous definition". Medterms. MedicineNet, Inc. 18 November 2000. Archived from the original on 9 July 2009.
- ^ "Definition of Term Pregnancy – ACOG". www.acog.org. Retrieved 27 September 2019.
- ^ "Definition of Premature birth". Medterms. Medicine.net. Archived from the original on 9 July 2009. Retrieved 16 January 2008.
- ^ Lama Rimawi, MD (22 September 2006). "Premature Infant". Disease & Conditions Encyclopedia. Discovery Communications, LLC. Archived from the original on 19 January 2008. Retrieved 16 January 2008.
- ^ "Total Fertility Rate 2025". World Population Review. Retrieved 29 August 2025.
- ^ a b c Bearak J, Popinchalk A, Alkema L, Sedgh G (April 2018). "Global, regional, and subregional trends in unintended pregnancy and its outcomes from 1990 to 2014: estimates from a Bayesian hierarchical model". The Lancet. Global Health. 6 (4): e380 – e389. doi:10.1016/S2214-109X(18)30029-9. PMC 6055480. PMID 29519649.
- ^ Hurt KJ, Guile MW, Bienstock JL, Fox HE, Wallach EE (28 March 2012). The Johns Hopkins manual of gynecology and obstetrics (4th ed.). Philadelphia: Wolters Kluwer Health / Lippincott Williams & Wilkins. p. 382. ISBN 978-1-60547-433-5.
- ^ Cherlin AJ (September 2021). "Rising nonmarital first childbearing among college-educated women: Evidence from three national studies". Proceedings of the National Academy of Sciences of the United States of America. 118 (37) e2109016118. Bibcode:2021PNAS..11809016C. doi:10.1073/pnas.2109016118. PMC 8449381. PMID 34493673.
- ^ a b "Adolescent pregnancy". World Health Organization. Retrieved 26 October 2022.
- ^ "Adolescent health". www.who.int.
- ^ "The Adverse Effects of Teen Pregnancy". youth.gov. Retrieved 26 October 2022.
- ^ Bekaert S, SmithBattle L (2016). "Teen Mothers' Experience of Intimate Partner Violence: A Metasynthesis". ANS. Advances in Nursing Science. 39 (3): 272–290. doi:10.1097/ANS.0000000000000129. PMID 27490882. S2CID 10471475.
- ^ Jenkins A, Millar S, Robins J (July 2011). "Denial of pregnancy: a literature review and discussion of ethical and legal issues". Journal of the Royal Society of Medicine. 104 (7): 286–291. doi:10.1258/jrsm.2011.100376. PMC 3128877. PMID 21725094.
- ^ Gabbe S (1 January 2012). Obstetrics: normal and problem pregnancies (6th ed.). Philadelphia: Elsevier/Saunders. p. 1184. ISBN 978-1-4377-1935-2.
- ^ "Pregnancy Symptoms". National Health Service (NHS). 11 March 2010. Archived from the original on 28 February 2010. Retrieved 11 March 2010.
- ^ a b "Early symptoms of pregnancy: What happens right away". Mayo Clinic. 22 February 2007. Archived from the original on 14 September 2007. Retrieved 22 August 2007.
- ^ a b "Pregnancy Symptoms – Early Signs of Pregnancy: American Pregnancy Association". Archived from the original on 15 January 2008. Retrieved 16 January 2008.
- ^ a b "Early Signs of Pregnancy". American Pregnancy Association. 2022. Retrieved 23 January 2025.
- ^ Alex A, Bhandary E, McGuire K (2020). Anatomy and Physiology of the Breast during Pregnancy and Lactation. Advances in Experimental Medicine and Biology. Vol. 1252. pp. 3–7. PMID 32816256.
- ^ "Swollen ankles, feet and fingers in pregnancy". NHS. 3 December 2020. Retrieved 4 May 2025.
- ^ a b "Common health problems in pregnancy". NHS. 22 April 2024. Retrieved 2 July 2025.
- ^ Habak P, Carlson K, Griggs R. "Urinary Tract Infection in Pregnancy". National Library of Medicine. Retrieved 26 June 2025.
- ^ Tucker Blackburn S (2007). Maternal, Fetal, & Neonatal Physiology: A Clinical Perspective. Saunders Elsevier. p. 384. ISBN 978-1-4160-2944-1.
- ^ Scarpelli S, Alfonsi V, De Gennaro L, Gorgoni M (August 2024). "Dreaming for two: A systematic review of mental sleep activity during pregnancy". Neuroscience & Biobehavioral Reviews. 163 105763. doi:10.1016/j.neubiorev.2024.105763. PMID 38852848.
- ^ Vazquez JC (August 2010). "Constipation, haemorrhoids, and heartburn in pregnancy". BMJ Clinical Evidence. 2010: 1411. PMC 3217736. PMID 21418682.
- ^ a b "Signs and symptoms of pregnancy". nhs.uk. 2 December 2020. Retrieved 23 January 2025.
- ^ Sifakis S, Pharmakides G (2006). "Anemia in pregnancy". Annals of the New York Academy of Sciences. 900 (1): 125–136. doi:10.1111/j.1749-6632.2000.tb06223.x. PMID 10818399.
- ^ "NHS Pregnancy Planner". National Health Service (NHS). 19 March 2010. Archived from the original on 29 August 2021. Retrieved 19 March 2010.
- ^ Cole LA, Butler SA, eds. (2015). Human chorionic gonadotropin (hCG) (2nd ed.). Amsterdam: Elsevier. ISBN 978-0-12-800821-8. Archived from the original on 26 January 2021. Retrieved 10 November 2015.
- ^ Qasim SM, Callan C, Choe JK (October 1996). "The predictive value of an initial serum beta human chorionic gonadotropin level for pregnancy outcome following in vitro fertilization". Journal of Assisted Reproduction and Genetics. 13 (9): 705–708. doi:10.1007/BF02066422. PMID 8947817. S2CID 36218409.
- ^ a b "What is HCG?". American Pregnancy Association. 18 October 2021. Retrieved 23 July 2023.
- ^ "BestBets: Serum or Urine beta-hCG?". Archived from the original on 31 December 2008.
- ^ a b Cole LA, Khanlian SA, Sutton JM, Davies S, Rayburn WF (January 2004). "Accuracy of home pregnancy tests at the time of missed menses". American Journal of Obstetrics and Gynecology. 190 (1): 100–105. doi:10.1016/j.ajog.2003.08.043. PMID 14749643.
- ^ a b c d e f g h i j k Cunningham FG, Leveno KJ, Bloom SL, Spong CY, Dashe JS, Hoffman BL, Casey BM, Sheffield JS, eds. (2014). "Chapter 12. Teratology, Teratogens, and Fetotoxic Agents". William's Obstetrics. McGraw-Hill Education. ISBN 978-0-07-179893-8. Archived from the original on 31 December 2018. Retrieved 9 November 2015.
- ^ Verhaegen J, Gallos ID, van Mello NM, Abdel-Aziz M, Takwoingi Y, Harb H, et al. (September 2012). "Accuracy of single progesterone test to predict early pregnancy outcome in women with pain or bleeding: meta-analysis of cohort studies". BMJ. 345 e6077. doi:10.1136/bmj.e6077. PMC 3460254. PMID 23045257.
- ^ Whitworth M, Bricker L, Mullan C (July 2015). "Ultrasound for fetal assessment in early pregnancy". The Cochrane Database of Systematic Reviews. 2015 (7) CD007058. doi:10.1002/14651858.CD007058.pub3. PMC 4084925. PMID 26171896.
- ^ Nguyen TH, Larsen T, Engholm G, Møller H (July 1999). "Evaluation of ultrasound-estimated date of delivery in 17,450 spontaneous singleton births: do we need to modify Naegele's rule?". Ultrasound in Obstetrics & Gynecology. 14 (1): 23–28. doi:10.1046/j.1469-0705.1999.14010023.x. PMID 10461334. S2CID 30749264.
- ^ Pyeritz RE (2014). Current Medical Diagnosis & Treatment 2015. McGraw-Hill.
- ^ a b c d e f g h i j Nair M, Kumar B (7 April 2016). "Embryology for fetal medicine". In Kumar B, Alfirevic Z (eds.). Fetal Medicine. Cambridge University Press. pp. 54–59. ISBN 978-1-107-06434-8.
- ^ a b c d Mishra S, ed. (7 August 2019). Langman's Medical Embryology. Wolters kluwer india Pvt Ltd. p. 48. ISBN 978-93-88696-53-1.
- ^ a b Obstetric Data Definitions Issues and Rationale for Change – Gestational Age & Term Archived 6 November 2013 at the Wayback Machine from Patient Safety and Quality Improvement at American Congress of Obstetricians and Gynecologists. Created November 2012.
- ^ Tunón K, Eik-Nes SH, Grøttum P, Von Düring V, Kahn JA (January 2000). "Gestational age in pregnancies conceived after in vitro fertilization: a comparison between age assessed from oocyte retrieval, crown-rump length and biparietal diameter". Ultrasound in Obstetrics & Gynecology. 15 (1): 41–46. doi:10.1046/j.1469-0705.2000.00004.x. PMID 10776011. S2CID 20029116.
- ^ a b c d "Pregnancy – the three trimesters". University of California San Francisco. Retrieved 30 November 2019.
- ^ a b Hoffman CS, Messer LC, Mendola P, Savitz DA, Herring AH, Hartmann KE (November 2008). "Comparison of gestational age at birth based on last menstrual period and ultrasound during the first trimester". Paediatric and Perinatal Epidemiology. 22 (6): 587–596. doi:10.1111/j.1365-3016.2008.00965.x. PMID 19000297.
- ^ "Pregnancy week by week". Mayo Clinic. 3 June 2022. Retrieved 8 July 2023.
- ^ "ement Health IT and Clinical Informatics reVITALize: Obstetrics Data Definitions reVITALize: Obstetrics Data Definitions". ACOG. Retrieved 27 November 2022.
- ^ Chambliss LR, Clark SL (February 2014). "Paper gestational age wheels are generally inaccurate". American Journal of Obstetrics and Gynecology. 210 (2): 145.e1–145.e4. doi:10.1016/j.ajog.2013.09.013. PMID 24036402.
- ^ Zegers-Hochschild F, Adamson GD, Dyer S, Racowsky C, de Mouzon J, Sokol R, et al. (September 2017). "The International Glossary on Infertility and Fertility Care, 2017". Fertility and Sterility. 108 (3). Elsevier BV: 393–406. doi:10.1016/j.fertnstert.2017.06.005. PMID 28760517. S2CID 3640374.
- ^ "Differences in sex development". nhs.uk. 18 November 2021. Retrieved 29 June 2022.
- ^ Weschler T (2002). Taking Charge of Your Fertility (Revised ed.). New York: HarperCollins. pp. 242, 374. ISBN 978-0-06-093764-5.
- ^ Berger KS (2011). The Developing Person Through the Life Span. Macmillan. p. 90. ISBN 978-1-4292-3205-0. Archived from the original on 25 April 2016.
- ^ "Stages of Development of the Fetus – Women's Health Issues". MSD Manual Consumer Version. Retrieved 10 July 2020.
- ^
- Lennart Nilsson, A Child is Born 91 (1990): at eight weeks, "the danger of a miscarriage ... diminishes sharply."
- "Women's Health Information Archived 30 April 2007 at the Wayback Machine", Hearthstone Communications Limited: "The risk of miscarriage decreases dramatically after the 8th week as the weeks go by." Retrieved 2007-04-22.
- ^ Kalverboer AF, Gramsbergen AA (1 January 2001). Handbook of Brain and Behaviour in Human Development. Springer. p. 1. ISBN 978-0-7923-6943-1. Archived from the original on 19 September 2015.
- ^ Illes J, ed. (2008). Neuroethics: defining the issues in theory, practice, and policy (Repr. ed.). Oxford: Oxford University Press. p. 142. ISBN 978-0-19-856721-9. Archived from the original on 19 September 2015.
- ^
- Harley TA (2021). The Science of Consciousness: Waking, Sleeping and Dreaming. Cambridge, United Kingdom: Cambridge University Press. p. 245. ISBN 978-1-107-12528-5. Retrieved 3 May 2022.
- Cleeremans A, Wilken P, Bayne T, eds. (2009). The Oxford Companion to Consciousness. New York, NY: Oxford University Press. p. 229. ISBN 978-0-19-856951-0. Retrieved 3 May 2022.
- Thompson E, Moscovitch M, Zelazo PD, eds. (2007). The Cambridge Handbook of Consciousness. Cambridge, United Kingdom: Cambridge University Press. pp. 415–417. ISBN 978-1-139-46406-2. Retrieved 3 May 2022.
- ^ "Abortion & Pregnancy Risks". Louisiana Department of Health. Retrieved 22 August 2019.
- ^ "Reproductive History and Cancer Risk". National Cancer Institute. 30 November 2016. Retrieved 23 August 2019.
- ^ a b Mor G, ed. (2006). Immunology of pregnancy. Medical intelligence unit. Georgetown, Tex. : New York: Landes Bioscience/Eurekah.com; Springer Science+Business Media. pp. 1–4. doi:10.1007/0-387-34944-8. ISBN 978-0-387-34944-2.
- ^ Williams Z (September 2012). "Inducing tolerance to pregnancy". The New England Journal of Medicine. 367 (12): 1159–1161. doi:10.1056/NEJMcibr1207279. PMC 3644969. PMID 22992082.
- ^ "Rhesus disease". NHS. 16 November 2021. Retrieved 26 June 2025.
- ^ Campbell LA, Klocke RA (April 2001). "Implications for the pregnant patient". American Journal of Respiratory and Critical Care Medicine. 163 (5): 1051–1054. doi:10.1164/ajrccm.163.5.16353. PMID 11316633.
- ^ "Your baby at 0–8 weeks pregnancy – Pregnancy and baby guide – NHS Choices". nhs.uk. 20 December 2017. Archived from the original on 20 November 2013.
- ^ Hennen L, Murray L, Scott J (2005). The BabyCenter Essential Guide to Pregnancy and Birth: Expert Advice and Real-World Wisdom from THE tip Top Pregnancy and Parenting Resource. Emmaus, Penn.: Rodale Books. ISBN 1-59486-211-7.
- ^ Soma-Pillay P, Nelson-Piercy C, Tolppanen H, Mebazaa A (2016). "Physiological changes in pregnancy". Cardiovascular Journal of Africa. 27 (2): 89–94. PMC 4928162.
- ^ Stacey T, Thompson JM, Mitchell EA, Ekeroma AJ, Zuccollo JM, McCowan LM (June 2011). "Association between maternal sleep practices and risk of late stillbirth: a case-control study". BMJ. 342 d3403. doi:10.1136/bmj.d3403. PMC 3114953. PMID 21673002.
- ^ Human Anatomy, Physiology and Health Education. S. Chand Publishing. 2010. p. 239. ISBN 978-81-219-3357-5.
- ^ "Pregnancy: Dropping (Lightening)". University of Michigan. Retrieved 9 June 2021.
- ^ "RHL". apps.who.int. Archived from the original on 27 December 2011.
- ^ a b World Health Organization (November 2013). "Preterm birth". who.int. Archived from the original on 7 September 2014. Retrieved 19 September 2014.
- ^ a b c d e f g h American Congress of Obstetricians and Gynecologists, Society for Maternal-Fetal Medicine (22 October 2013). "Ob-Gyns Redefine Meaning of 'Term Pregnancy'". acog.org. Archived from the original on 15 September 2014. Retrieved 19 September 2014.
- ^ Saigal S, Doyle LW (January 2008). "An overview of mortality and sequelae of preterm birth from infancy to adulthood". Lancet. 371 (9608): 261–269. doi:10.1016/S0140-6736(08)60136-1. PMID 18207020. S2CID 17256481.
- ^ American Congress of Obstetricians and Gynecologists (February 2013), "Five Things Physicians and Patients Should Question", Choosing Wisely: an initiative of the ABIM Foundation, American Congress of Obstetricians and Gynecologists, archived from the original on 1 September 2013, retrieved 1 August 2013, which cites
- Main E, Oshiro B, Chagolla B, Bingham D, Dang-Kilduff L, Kowalewski L, Elimination of Non-medically Indicated (Elective) Deliveries Before 39 Weeks Gestational Age (PDF), March of Dimes; California Maternal Quality Care Collaborative; Maternal, Child and Adolescent Health Division; Center for Family Health; California Department of Public Health, archived from the original (PDF) on 10 November 2012, retrieved 1 August 2013
- ^ Michele Norris (18 July 2011). "Doctors To Pregnant Women: Wait at Least 39 Weeks". All Things Considered. Archived from the original on 23 July 2011. Retrieved 20 August 2011.
- ^ Norwitz ER. "Postterm Pregnancy (Beyond the Basics)". UpToDate, Inc. Archived from the original on 7 October 2012. Retrieved 24 August 2012.
- ^ The American College of Obstetricians and Gynecologists (April 2006). "What To Expect After Your Due Date". Medem. Medem, Inc. Archived from the original on 29 April 2003. Retrieved 16 January 2008.
- ^ "Induction of labour – Evidence-based Clinical Guideline Number 9" (PDF). Royal College of Obstetricians and Gynaecologists. 2001. Archived from the original (PDF) on 30 December 2006. Retrieved 18 January 2008.
- ^ Waters TR, MacDonald LA, Hudock SD, Goddard DE (February 2014). "Provisional recommended weight limits for manual lifting during pregnancy". Human Factors. 56 (1): 203–214. doi:10.1177/0018720813502223. PMC 4606868. PMID 24669554. Archived from the original on 1 April 2017.
- ^ MacDonald LA, Waters TR, Napolitano PG, Goddard DE, Ryan MA, Nielsen P, et al. (August 2013). "Clinical guidelines for occupational lifting in pregnancy: evidence summary and provisional recommendations". American Journal of Obstetrics and Gynecology. 209 (2): 80–88. doi:10.1016/j.ajog.2013.02.047. PMC 4552317. PMID 23467051.
- ^ a b Lyons P (2015). Obstetrics in family medicine: a practical guide. Current clinical practice (2nd ed.). Cham, Switzerland: Humana Press. pp. 19–28. ISBN 978-3-319-20077-4. Archived from the original on 26 January 2021. Retrieved 11 November 2015.
- ^ "WHO | Antenatal care". www.who.int. Archived from the original on 20 November 2015. Retrieved 10 November 2015.
- ^ Dowswell T, Carroli G, Duley L, Gates S, Gülmezoglu AM, Khan-Neelofur D, et al. (American College of Obstetricians Gynecologists Committee on Health Care for Undeserved Women) (July 2015). "Alternative versus standard packages of antenatal care for low-risk pregnancy". The Cochrane Database of Systematic Reviews. 2015 (7) CD000934. doi:10.1002/14651858.cd000934.pub3. PMC 7061257. PMID 26184394.
- ^ American College of Obstetricians Gynecologists Committee on Health Care for Undeserved Women (August 2006). "ACOG Committee Opinion No. 343: psychosocial risk factors: perinatal screening and intervention". Obstetrics and Gynecology. 108 (2): 469–477. doi:10.1097/00006250-200608000-00046. PMID 16880322.
- ^ Hurt JK, ed. (2011). The Johns Hopkins manual of gynecology and obstetrics (4th ed.). Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins. ISBN 978-1-4511-0913-9.
- ^ McCormick MC, Siegel JE, eds. (1999). Prenatal care: effectiveness and implementation. Cambridge, UK; New York: Cambridge University Press. ISBN 978-0-521-66196-6. Archived from the original on 6 November 2018. Retrieved 10 November 2015.
- ^ Deshpande N, Lewis-O'Connor A (2013). "Screening for Intimate Partner Violence During Pregnancy". Rev Obstet Gynecol. 6 (3–4): 141–148. PMC 4002190. PMID 24920977.
- ^ Ota E, Hori H, Mori R, Tobe-Gai R, Farrar D (June 2015). "Antenatal dietary education and supplementation to increase energy and protein intake". The Cochrane Database of Systematic Reviews. 2015 (6) CD000032. doi:10.1002/14651858.CD000032.pub3. PMID 26031211.
- ^ "| Choose MyPlate". Choose MyPlate. 29 April 2015. Archived from the original on 17 November 2015. Retrieved 15 November 2015.
- ^ Tieu J, Shepherd E, Middleton P, Crowther CA (January 2017). "Dietary advice interventions in pregnancy for preventing gestational diabetes mellitus". The Cochrane Database of Systematic Reviews. 1 (1) CD006674. doi:10.1002/14651858.CD006674.pub3. PMC 6464792. PMID 28046205.
- ^ Klusmann A, Heinrich B, Stöpler H, Gärtner J, Mayatepek E, Von Kries R (November 2005). "A decreasing rate of neural tube defects following the recommendations for periconceptional folic acid supplementation". Acta Paediatrica. 94 (11): 1538–1542. doi:10.1080/08035250500340396. PMID 16303691. S2CID 13506877.
- ^ Greenberg JA (2011). "Multivitamin Supplementation During Pregnancy: Emphasis on Folic Acid and l-Methylfolate". Reviews in Obstetrics and Gynecology. 4 (3–4): 126–127. PMC 3250974. PMID 22229066.
- ^ Stevenson RE, Allen WP, Pai GS, Best R, Seaver LH, Dean J, et al. (October 2000). "Decline in prevalence of neural tube defects in a high-risk region of the United States". Pediatrics. 106 (4): 677–683. doi:10.1542/peds.106.4.677. PMID 11015508. S2CID 39696556.
- ^ "Folic acid in diet: MedlinePlus Medical Encyclopedia". www.nlm.nih.gov. Archived from the original on 17 November 2015. Retrieved 15 November 2015.
- ^ Centers for Disease Control Prevention (CDC) (January 2008). "Use of supplements containing folic acid among women of childbearing age--United States, 2007". MMWR. Morbidity and Mortality Weekly Report. 57 (1): 5–8. PMID 18185493.
- ^ a b c Viswanathan M, Siega-Riz AM, Moos MK, et al. (May 2008). "Outcomes of Maternal Weight Gain". Evidence Report/Technology Assessment. Evidence Reports/Technology Assessments, No. 168 (168). Agency for Healthcare Research and Quality: 1–223. PMC 4781425. PMID 18620471. Archived from the original on 28 May 2013. Retrieved 23 June 2013.
- ^ a b c Institute for Quality and Efficiency in Health Care (22 January 2013). "Weight gain in pregnancy". Fact sheet. Institute for Quality and Efficiency in Health Care. Archived from the original on 14 December 2013. Retrieved 23 June 2013.
- ^ "Weight Gain During Pregnancy: Reexaminging the Guidelines, Report Brief". Institute of Medicine. Archived from the original on 10 August 2010. Retrieved 29 July 2010.
- ^ American College of Obstetricians Gynecologists (January 2013). "ACOG Committee opinion no. 548: weight gain during pregnancy". Obstetrics and Gynecology. 121 (1): 210–212. doi:10.1097/01.AOG.0000425668.87506.4c. PMID 23262962.
- ^ a b c Thangaratinam S, Rogozińska E, Jolly K, Glinkowski S, Duda W, Borowiack E, et al. (July 2012). "Interventions to reduce or prevent obesity in pregnant women: a systematic review". Health Technology Assessment. 16 (31). National Institute for Health and Care Research: iii–iv, 1–191. doi:10.3310/hta16310. PMC 4781281. PMID 22814301.
- ^ Bushnell C, McCullough LD, Awad IA, Chireau MV, Fedder WN, Furie KL, et al. (May 2014). "Guidelines for the prevention of stroke in women: a statement for healthcare professionals from the American Heart Association/American Stroke Association". Stroke. 45 (5): 1545–1588. doi:10.1161/01.str.0000442009.06663.48. PMC 10152977. PMID 24503673. S2CID 6297484.
- ^ a b c Briggs GG, Freeman RK (2015). Drugs in pregnancy and lactation: A Reference Guide to Fetal and Neonatal Risk (Tenth ed.). Philadelphia: Wolters Kluwer/Lippincott Williams & Wilkins Health. p. Appendix. ISBN 978-1-4511-9082-3. Archived from the original on 25 February 2021. Retrieved 16 November 2015.
- ^ Genetic Alliance, The New England Public Health Genetics Education Collaborative (17 February 2010). "Appendix A: Teratogens/Prenatal Substance Abuse". Understanding Genetics: A New England Guide for Patients and Health Professionals. Genetic Alliance.
- ^ "Press Announcements – FDA issues final rule on changes to pregnancy and lactation labeling information for prescription drug and biological products". www.fda.gov. Archived from the original on 17 November 2015. Retrieved 16 November 2015.
- ^ "Basics about FASDs". CDC. Retrieved 25 July 2018.
- ^ Hackshaw A, Rodeck C, Boniface S (September–October 2011). "Maternal smoking in pregnancy and birth defects: a systematic review based on 173 687 malformed cases and 11.7 million controls". Human Reproduction Update. 17 (5): 589–604. doi:10.1093/humupd/dmr022. PMC 3156888. PMID 21747128.
- ^ Centers for Disease Control and Prevention. 2007. Preventing Smoking and Exposure to Secondhand Smoke Before, During, and After Pregnancy Archived 11 September 2011 at the Wayback Machine.
- ^ "Tobacco Use and Pregnancy – Reproductive Health". www.cdc.gov. 16 January 2019. Archived from the original on 29 July 2017.
- ^ a b "New Mother Fact Sheet: Methamphetamine Use During Pregnancy". North Dakota Department of Health. Archived from the original on 10 September 2011. Retrieved 7 October 2011.
- ^ Della Grotta S, LaGasse LL, Arria AM, Derauf C, Grant P, Smith LM, et al. (July 2010). "Patterns of methamphetamine use during pregnancy: results from the Infant Development, Environment, and Lifestyle (IDEAL) Study". Maternal and Child Health Journal. 14 (4): 519–527. doi:10.1007/s10995-009-0491-0. PMC 2895902. PMID 19565330.
- ^ Martin R, Dombrowski SC (2008). "12. Air and Water Pollution". Prenatal exposures: psychological and educational consequences for children. New York: Springer. doi:10.1007/978-0-387-74398-1. ISBN 978-0-387-74398-1.
- ^ Byrne CC (2006). "Environmental hazards during pregnancy". Journal of Midwifery and Women's Health. 1 (51): 57–58. doi:10.1016/j.jmwh.2005.09.008. PMID 16402445.
- ^ "N95 Respirator Use During Pregnancy – Findings from Recent NIOSH Research | NIOSH Science Blog | Blogs | CDC". blogs.cdc.gov. 18 June 2015. Archived from the original on 16 November 2016. Retrieved 16 November 2016.
- ^ "Homicide leading cause of death for pregnant women in U.S." News. Harvard T.H. Chan School of Public Health. 21 October 2022. Retrieved 8 November 2022.
- ^ "With homicide a leading cause of maternal death, doctors urged to screen pregnant women for domestic violence". CNN Health. 20 October 2022. Retrieved 8 November 2022.
- ^ Wallace ME (September 2022). "Trends in Pregnancy-Associated Homicide, United States, 2020". American Journal of Public Health. 112 (9): 1333–1336. doi:10.2105/AJPH.2022.306937. PMC 9382166. PMID 35797500.
- ^ a b c Cunningham FG, Leveno KJ, Bloom SL, Spong CY, Dashe JS, Hoffman BL, Casey BM, Sheffield JS, eds. (2014). "Chapter 9: Prenatal Care". Williams Obstetrics (24th ed.). New York: McGraw-Hill Education. ISBN 978-0-07-179893-8. Archived from the original on 31 December 2018. Retrieved 9 November 2015.
- ^ Bermudez MP, Sanchez AI, Buela-Casal G (2001). "Influence of the Gestation Period on Sexual Desire". Psychology in Spain. 5 (1): 14–16. Archived from the original on 9 February 2012.
- ^ Fok WY, Chan LY, Yuen PM (October 2005). "Sexual behavior and activity in Chinese pregnant women". Acta Obstetricia et Gynecologica Scandinavica. 84 (10): 934–938. doi:10.1111/j.0001-6349.2005.00743.x. PMID 16167907. S2CID 23075166.
- ^ Reamy K, White SE, Daniell WC, Le Vine ES (June 1982). "Sexuality and pregnancy. A prospective study". The Journal of Reproductive Medicine. 27 (6): 321–327. PMID 7120209.
- ^ Malarewicz A, Szymkiewicz J, Rogala J (September 2006). "[Sexuality of pregnant women]". Ginekologia Polska (in Polish). 77 (9): 733–739. PMID 17219804.
- ^ Kramer MS, McDonald SW (July 2006). Kramer MS (ed.). "Aerobic exercise for women during pregnancy". The Cochrane Database of Systematic Reviews. 3 (3) CD000180. doi:10.1002/14651858.CD000180.pub2. PMC 7043271. PMID 16855953.
- ^ Domenjoz I, Kayser B, Boulvain M (October 2014). "Effect of physical activity during pregnancy on mode of delivery". American Journal of Obstetrics and Gynecology. 211 (4): 401.e1–401.11. doi:10.1016/j.ajog.2014.03.030. PMID 24631706.
- ^ Barakat R, Franco E, Perales M, López C, Mottola MF (May 2018). "Exercise during pregnancy is associated with a shorter duration of labor. A randomized clinical trial". European Journal of Obstetrics & Gynecology and Reproductive Biology. 224: 33–40. doi:10.1016/j.ejogrb.2018.03.009. PMID 29529475.
- ^ a b Beetham KS, Giles C, Noetel M, Clifton V, Jones JC, Naughton G (August 2019). "The effects of vigorous intensity exercise in the third trimester of pregnancy: a systematic review and meta-analysis". BMC Pregnancy and Childbirth. 19 (1) 281. doi:10.1186/s12884-019-2441-1. PMC 6686535. PMID 31391016.
- ^ Perales M, Santos-Lozano A, Sanchis-Gomar F, Luaces M, Pareja-Galeano H, Garatachea N, et al. (May 2016). "Maternal Cardiac Adaptations to a Physical Exercise Program during Pregnancy". Medicine & Science in Sports & Exercise. 48 (5): 896–906. doi:10.1249/MSS.0000000000000837. hdl:11268/4797. ISSN 0195-9131. PMID 26694848.
- ^ "ACSM Blog". ACSM_CMS. Retrieved 14 November 2024.
- ^ Davies GA, Wolfe LA, Mottola MF, MacKinnon C, Arsenault MY, Bartellas E, et al. (June 2003). "Exercise in pregnancy and the postpartum period". Journal of Obstetrics and Gynaecology Canada. 25 (6): 516–529. doi:10.1016/s1701-2163(16)30313-9. PMID 12806453.
- ^ Artal R, O'Toole M (February 2003). "Guidelines of the American College of Obstetricians and Gynecologists for exercise during pregnancy and the postpartum period". British Journal of Sports Medicine. 37 (1): 6–12, discussion 12. doi:10.1136/bjsm.37.1.6. PMC 1724598. PMID 12547738.
- ^ McCall CA, Grimes DA, Lyerly AD (June 2013). ""Therapeutic" bed rest in pregnancy: unethical and unsupported by data". Obstetrics and Gynecology. 121 (6): 1305–1308. doi:10.1097/aog.0b013e318293f12f. PMID 23812466.
- ^ a b Vesting S, Gutke A, Fagevik Olsén M, Rembeck G, Larsson ME (2024). "The Impact of Exercising on Pelvic Symptom Severity, Pelvic Floor Muscle Strength, and Diastasis Recti Abdominis After Pregnancy: A Longitudinal Prospective Cohort Study". Physical Therapy. 104 (4). doi:10.1093/ptj/pzad171. PMC 11021861. PMID 38109793.
- ^ Yu H, Santos-Rocha R, Radzimiński Ł, Jastrzębski Z, Bonisławska I, Szwarc A, et al. (11 December 2022). "Effects of 8-Week Online, Supervised High-Intensity Interval Training on the Parameters Related to the Anaerobic Threshold, Body Weight, and Body Composition during Pregnancy: A Randomized Controlled Trial". Nutrients. 14 (24): 5279. doi:10.3390/nu14245279. ISSN 2072-6643. PMC 9781372. PMID 36558438.
- ^ a b c Tung I, Hipwell AE, Grosse P, Battaglia L, Cannova E, English G, et al. (November 2023). "Prenatal stress and externalizing behaviors in childhood and adolescence: A systematic review and meta-analysis". Psychological Bulletin. 150 (2): 107–131. doi:10.1037/bul0000407. PMC 10932904. PMID 37971856. S2CID 265272043.
- ^ Wowdzia JB, Hazell TJ, Davenport MH (September 2022). "Glycemic response to acute high-intensity interval versus moderate-intensity continuous exercise during pregnancy". Physiological Reports. 10 (18) e15454. doi:10.14814/phy2.15454. ISSN 2051-817X. PMC 9483614. PMID 36117457.
- ^ Nagpal T, Everest C, Goudreau A, Manicks M, Adamo K (March 2021). "To HIIT or not to HIIT? The question pregnant women may be searching for online: a descriptive observational study". Perspectives in Public Health. 141 (2): 81–88. doi:10.1177/1757913920985898. ISSN 1757-9139. PMID 33579178.
- ^ Reiter RJ, Tan DX, Korkmaz A, Rosales-Corral SA (2014). "Melatonin and stable circadian rhythms optimize maternal, placental and fetal physiology". Human Reproduction Update. 20 (2): 293–307. doi:10.1093/humupd/dmt054. PMID 24132226.
- ^ "Oral health care during pregnancy: A strategies and considerations" (PDF). Archived from the original (PDF) on 22 August 2018. Retrieved 2 June 2018.
- ^ Jafarzadeh H, Sanatkhani M, Mohtasham N (December 2006). "Oral pyogenic granuloma: a review". Journal of Oral Science. 48 (4): 167–175. doi:10.2334/josnusd.48.167. PMID 17220613.
- ^ Iheozor-Ejiofor Z, Middleton P, Esposito M, Glenny AM (June 2017). "Treating periodontal disease for preventing adverse birth outcomes in pregnant women". The Cochrane Database of Systematic Reviews. 2017 (6) CD005297. doi:10.1002/14651858.CD005297.pub3. PMC 6481493. PMID 28605006.
- ^ Howland G (2017). The Mama Natural Week-by-Week Guide to Pregnancy and Childbirth. Simon and Schuster. p. 173. ISBN 978-1-5011-4668-8.
- ^ a b c Jarvis S, Stone J, Eddleman K, Duenwald M (2011). Pregnancy For Dummies. John Wiley & Sons. p. 157. ISBN 978-1-119-99706-1.
- ^ "Air Travel During Pregnancy". www.acog.org. Retrieved 22 December 2023.
- ^ "Antenatal classes". NHS. December 2020. Retrieved 16 November 2022.
- ^ "Promoting baby-friendly hospitals". World Health Organization. Retrieved 16 November 2022.
- ^ "Reproductive Health and Research Publications: Making Pregnancy Safer". World Health Organization Regional Office for South-East Asia. 2009. Archived from the original on 15 December 2009. Retrieved 7 December 2009.
- ^ "Pregnancy complicated by disease". Merck Manual, Home Health Handbook. Merck Sharp & Dohme.
- ^ Dagher R, Bruckheim H, Colpe L, Edwards E, White D (2021). "Perinatal Depression: Challenges and Opportunities". J Womens Health (Larchmt. 30 (2): 154–159. PMC 7891219. PMID 33156730.
- ^ Stewart DE, Vigod S (December 2016). "Postpartum Depression". The New England Journal of Medicine. 375 (22): 2177–2186. doi:10.1056/nejmcp1607649. PMID 27959754.
- ^ Lara A Friel. "Thromboembolic Disorders During Pregnancy". Merck Manuals Consumer Version. Merck Sharp & Dohme Corp.
- ^ a b Leveno K (2013). "52". Williams Manual of Pregnancy Complications. New York: McGraw-Hill Medical. pp. 323–334. ISBN 978-0-07-176562-6.
- ^ "What are the symptoms of pregnancy loss (before 20 weeks of pregnancy)?". NIH. September 2017. Retrieved 4 October 2022.
- ^ "Miscarriage Causes". WebMD. Retrieved 6 October 2022.
- ^ "What Is Stillborn". CDC. 29 September 2022. Retrieved 6 October 2022.
- ^ "Miscarriage". NHS. 9 March 2022.
- ^ Obst KL, Due C, Oxlad M, Middleton P (January 2020). "Men's grief following pregnancy loss and neonatal loss: a systematic review and emerging theoretical model". BMC Pregnancy and Childbirth. 20 (1) 11. doi:10.1186/s12884-019-2677-9. PMC 6953275. PMID 31918681.
- ^ Tersigni C, Castellani R, de Waure C, Fattorossi A, De Spirito M, Gasbarrini A, et al. (2014). "Celiac disease and reproductive disorders: meta-analysis of epidemiologic associations and potential pathogenic mechanisms". Human Reproduction Update. 20 (4): 582–593. doi:10.1093/humupd/dmu007. hdl:10807/56796. PMID 24619876.
- ^ Saccone G, Berghella V, Sarno L, Maruotti GM, Cetin I, Greco L, et al. (February 2016). "Celiac disease and obstetric complications: a systematic review and metaanalysis". American Journal of Obstetrics and Gynecology. 214 (2): 225–234. doi:10.1016/j.ajog.2015.09.080. hdl:11369/330101. PMID 26432464.
- ^ "The Gluten Connection". Health Canada. May 2009. Archived from the original on 5 July 2017. Retrieved 1 October 2013.
- ^ a b Page 264 in: Gresele, Paolo (2008). Platelets in hematologic and cardiovascular disorders: a clinical handbook. Cambridge, UK: Cambridge University Press. ISBN 978-0-521-88115-9.
- ^ Zdanowicz C (21 May 2019). "Women have abortions for many reasons aside from rape and incest. Here are some of them". CNN. Retrieved 2 July 2022.
- ^ "Law and Policy Guide: Rape and Incest Exceptions". Center for Reproductive Rights. 18 January 2022. Retrieved 2 July 2022.
- ^ "SDG Indicator 3.7.1 on Contraceptive Use". Population Division. Retrieved 3 July 2022.
- ^ a b c "Guidelines for Diagnostic Imaging During Pregnancy and Lactation". American Congress of Obstetricians and Gynecologists. Archived from the original on 30 July 2017. February 2016
- ^ "Maternity and paternity at work: Law and practice across the world". International Labour Organization. Retrieved 3 September 2022.
- ^ Katie Mcdonough (30 April 2014). "Tennessee just became the first state that will jail women for their pregnancy outcomes". Salon. Archived from the original on 5 May 2014. Retrieved 5 May 2014.
- ^ a b "NONFICTION: When Pregnancy is a Crime". 17 June 2021.
- ^ "Employment of Foreign Manpower (Work Passes) Regulations 2012 - Singapore Statutes Online".
- ^ "Employment of Foreign Manpower Act - Singapore Statutes Online".
- ^ "Employers will not lose security bond if FDW gets pregnant – MOM". twc2.org.sg. 1 July 2011. Retrieved 15 December 2021.
- ^ Pereira GM, Pimentel VM, Surita FG, Silva AD, Brito LG (2022). "Perceived racism or racial discrimination and the risk of adverse obstetric outcomes: a systematic review". Sao Paulo Med J. 140 (5): 705–718. doi:10.1590/1516-3180.2021.0505.R1.07042022. PMC 9514866. PMID 36043663.
- ^ Guerra-Reyes L, Hamilton LJ (February 2017). "Racial disparities in birth care: Exploring the perceived role of African-American women providing midwifery care and birth support in the United States". Women and Birth. 30 (1): e9 – e16. doi:10.1016/j.wombi.2016.06.004. PMID 27364419.
- ^ Obedin-Maliver J, Makadon HJ (March 2016). "Transgender men and pregnancy". Obstetric Medicine. 9 (1): 4–8. doi:10.1177/1753495X15612658. PMC 4790470. PMID 27030799.
- ^ Light AD, Obedin-Maliver J, Sevelius JM, Kerns JL (December 2014). "Transgender men who experienced pregnancy after female-to-male gender transitioning". Obstetrics and Gynecology. 124 (6): 1120–1127. doi:10.1097/AOG.0000000000000540. PMID 25415163. S2CID 36023275.
- ^ Womack M (2010). The anthropology of health and healing. Plymouth: AltaMira Press. p. 133. ISBN 978-0-7591-1044-1.
- ^ Baumgarten L (2002). What Clothes Reveal: The Language of Clothing in Colonial and Federal America. Williamsburg, Virginia: Colonial Williamsburg. p. 148. ISBN 0-87935-216-7.
- ^ Chrisman-Campbell K (12 July 2013). "Dressing for Two". Slate. Retrieved 26 June 2024.
- ^ Rossi TV (2005). Mary in western art. New York: In Association with Hudson Hills Press. p. 106. ISBN 978-0-9712981-9-4.
Further reading
[edit]- "Nutrition for the First Trimester of Pregnancy". IDEA Health & Fitness Association. Retrieved 9 December 2013.
- Bothwell TH (July 2000). "Iron requirements in pregnancy and strategies to meet them". The American Journal of Clinical Nutrition. 72 (1 Suppl): 257S – 264S. doi:10.1093/ajcn/72.1.257S. PMID 10871591.
- Stevens J (June 2005). "Pregnancy envy and the politics of compensatory masculinities". Politics & Gender. 1 (2): 265–296. CiteSeerX 10.1.1.485.5791. doi:10.1017/S1743923X05050087. S2CID 39231847.
External links
[edit]- Merck Manual Home Health Handbook – further details on the diseases, disorders, etc., which may complicate pregnancy.
- Pregnancy care – NHS guide to having a baby including preconception, pregnancy, labor, and birth.