Recent from talks
Nothing was collected or created yet.
Biogeochemical cycle
View on Wikipedia| Part of a series on |
| Biogeochemical cycles |
|---|
 |
A biogeochemical cycle, or more generally a cycle of matter,[1] is the movement and transformation of chemical elements and compounds between living organisms, the atmosphere, and the Earth's crust. Major biogeochemical cycles include the carbon cycle, the nitrogen cycle and the water cycle. In each cycle, the chemical element or molecule is transformed and cycled by living organisms and through various geological forms and reservoirs, including the atmosphere, the soil and the oceans. It can be thought of as the pathway by which a chemical substance cycles (is turned over or moves through) the biotic compartment and the abiotic compartments of Earth. The biotic compartment is the biosphere and the abiotic compartments are the atmosphere, lithosphere and hydrosphere.
For example, in the carbon cycle, atmospheric carbon dioxide is absorbed by plants through photosynthesis, which converts it into organic compounds that are used by organisms for energy and growth. Carbon is then released back into the atmosphere through respiration and decomposition. Additionally, carbon is stored in fossil fuels and is released into the atmosphere through human activities such as burning fossil fuels. In the nitrogen cycle, atmospheric nitrogen gas is converted by plants into usable forms such as ammonia and nitrates through the process of nitrogen fixation. These compounds can be used by other organisms, and nitrogen is returned to the atmosphere through denitrification and other processes. In the water cycle, the universal solvent water evaporates from land and oceans to form clouds in the atmosphere, and then precipitates back to different parts of the planet. Precipitation can seep into the ground and become part of groundwater systems used by plants and other organisms, or can runoff the surface to form lakes and rivers. Subterranean water can then seep into the ocean along with river discharges, rich with dissolved and particulate organic matter and other nutrients.
There are biogeochemical cycles for many other elements, such as for oxygen, hydrogen, phosphorus, calcium, iron, sulfur, mercury and selenium. There are also cycles for molecules, such as water and silica. In addition there are macroscopic cycles such as the rock cycle, and human-induced cycles for synthetic compounds such as for polychlorinated biphenyls (PCBs). In some cycles there are geological reservoirs where substances can remain or be sequestered for long periods of time.
Biogeochemical cycles involve the interaction of biological, geological, and chemical processes. Biological processes include the influence of microorganisms, which are critical drivers of biogeochemical cycling. Microorganisms have the ability to carry out wide ranges of metabolic processes essential for the cycling of nutrients (macronutrients and micronutrients) and chemicals throughout global ecosystems. Without microorganisms many of these processes would not occur, with significant impact on the functioning of land and ocean ecosystems and the planet's biogeochemical cycles as a whole. Changes to cycles can impact human health. The cycles are interconnected and play important roles regulating climate, supporting the growth of plants, phytoplankton and other organisms, and maintaining the health of ecosystems generally. Human activities such as burning fossil fuels and using large amounts of fertilizer can disrupt cycles, contributing to climate change, pollution, and other environmental problems.
Overview
[edit]

Energy flows directionally through ecosystems, entering as sunlight (or inorganic molecules for chemoautotrophs) and leaving as heat during the many transfers between trophic levels. However, the matter that makes up living organisms is conserved and recycled. The six most common elements associated with organic molecules — carbon, nitrogen, hydrogen, oxygen, phosphorus, and sulfur — take a variety of chemical forms and may exist for long periods in the atmosphere, on land, in water, or beneath the Earth's surface. Geologic processes, such as weathering, erosion, water drainage, and the subduction of the continental plates, all play a role in this recycling of materials. Because geology and chemistry have major roles in the study of this process, the recycling of inorganic matter between living organisms and their environment is called a biogeochemical cycle.[3]
The six aforementioned elements are used by organisms in a variety of ways. Hydrogen and oxygen are found in water and organic molecules, both of which are essential to life. Carbon is found in all organic molecules, whereas nitrogen is an important component of nucleic acids and proteins. Phosphorus is used to make nucleic acids and the phospholipids that comprise biological membranes. Sulfur is critical to the three-dimensional shape of proteins. The cycling of these elements is interconnected. For example, the movement of water is critical for leaching sulfur and phosphorus into rivers which can then flow into oceans. Minerals cycle through the biosphere between the biotic and abiotic components and from one organism to another.[4]
Ecological systems (ecosystems) have many biogeochemical cycles operating as a part of the system, for example, the water cycle, the carbon cycle, the nitrogen cycle, etc. All chemical elements occurring in organisms are part of biogeochemical cycles. In addition to being a part of living organisms, these chemical elements also cycle through abiotic factors of ecosystems such as water (hydrosphere), land (lithosphere), and/or the air (atmosphere).[5]
The living factors of the planet can be referred to collectively as the biosphere. All the nutrients — such as carbon, nitrogen, oxygen, phosphorus, and sulfur — used in ecosystems by living organisms are a part of a closed system; therefore, these chemicals are recycled instead of being lost and replenished constantly such as in an open system.[5]
The major parts of the biosphere are connected by the flow of chemical elements and compounds in biogeochemical cycles. In many of these cycles, the biota plays an important role. Matter from the Earth's interior is released by volcanoes. The atmosphere exchanges some compounds and elements rapidly with the biota and oceans. Exchanges of materials between rocks, soils, and the oceans are generally slower by comparison.[2]
The flow of energy in an ecosystem is an open system; the Sun constantly gives the planet energy in the form of light while it is eventually used and lost in the form of heat throughout the trophic levels of a food web. Carbon is used to make carbohydrates, fats, and proteins, the major sources of food energy. These compounds are oxidized to release carbon dioxide, which can be captured by plants to make organic compounds. The chemical reaction is powered by the light energy of sunshine.
Sunlight is required to combine carbon with hydrogen and oxygen into an energy source, but ecosystems in the deep sea, where no sunlight can penetrate, obtain energy from sulfur. Hydrogen sulfide near hydrothermal vents can be utilized by organisms such as the giant tube worm. In the sulfur cycle, sulfur can be forever recycled as a source of energy. Energy can be released through the oxidation and reduction of sulfur compounds (e.g., oxidizing elemental sulfur to sulfite and then to sulfate).
-
Examples of major biogeochemical processes
-
The oceanic whale pump showing how whales cycle nutrients through the ocean water column
-
The implications of shifts in the global carbon cycle due to human activity are concerning scientists.[6]
Although the Earth constantly receives energy from the Sun, its chemical composition is essentially fixed, as the additional matter is only occasionally added by meteorites. Because this chemical composition is not replenished like energy, all processes that depend on these chemicals must be recycled. These cycles include both the living biosphere and the nonliving lithosphere, atmosphere, and hydrosphere.
Biogeochemical cycles can be contrasted with geochemical cycles. The latter deals only with crustal and subcrustal reservoirs even though some process from both overlap.
Compartments
[edit]Biogeochemical cycles operate by moving substances, which may also undergo chemical rearrangements, through pathways in the biotic compartment and the abiotic compartments of Earth. The biotic compartment is the biosphere and the abiotic compartments are the atmosphere, lithosphere and hydrosphere.
Biotic compartment
[edit]Biosphere
[edit]Microorganisms drive much of the biogeochemical cycling in the earth system.[7][8]
Abiotic compartments
[edit]
Atmosphere
[edit]Hydrosphere
[edit]
The global ocean covers more than 70% of the Earth's surface and is remarkably heterogeneous. Marine productive areas, and coastal ecosystems comprise a minor fraction of the ocean in terms of surface area, yet have an enormous impact on global biogeochemical cycles carried out by microbial communities, which represent 90% of the ocean's biomass.[10] Work in recent years has largely focused on cycling of carbon and macronutrients such as nitrogen, phosphorus, and silicate: other important elements such as sulfur or trace elements have been less studied, reflecting associated technical and logistical issues.[11] Increasingly, these marine areas, and the taxa that form their ecosystems, are subject to significant anthropogenic pressure, impacting marine life and recycling of energy and nutrients.[12][13][14] A key example is that of cultural eutrophication, where agricultural runoff leads to nitrogen and phosphorus enrichment of coastal ecosystems, greatly increasing productivity resulting in algal blooms, deoxygenation of the water column and seabed, and increased greenhouse gas emissions,[15] with direct local and global impacts on nitrogen and carbon cycles. However, the runoff of organic matter from the mainland to coastal ecosystems is just one of a series of pressing threats stressing microbial communities due to global change. Climate change has also resulted in changes in the cryosphere, as glaciers and permafrost melt, resulting in intensified marine stratification, while shifts of the redox-state in different biomes are rapidly reshaping microbial assemblages at an unprecedented rate.[16][17][18][19][11]
Global change is, therefore, affecting key processes including primary productivity, CO2 and N2 fixation, organic matter respiration/remineralization, and the sinking and burial deposition of fixed CO2.[19] In addition to this, oceans are experiencing an acidification process, with a change of ~0.1 pH units between the pre-industrial period and today, affecting carbonate/bicarbonate buffer chemistry. In turn, acidification has been reported to impact planktonic communities, principally through effects on calcifying taxa.[20] There is also evidence for shifts in the production of key intermediary volatile products, some of which have marked greenhouse effects (e.g., N2O and CH4, reviewed by Breitburg in 2018,[17] due to the increase in global temperature, ocean stratification and deoxygenation, driving as much as 25 to 50% of nitrogen loss from the ocean to the atmosphere in the so-called oxygen minimum zones[21] or anoxic marine zones,[22] driven by microbial processes. Other products, that are typically toxic for the marine nekton, including reduced sulfur species such as H2S, have a negative impact for marine resources like fisheries and coastal aquaculture. While global change has accelerated, there has been a parallel increase in awareness of the complexity of marine ecosystems, and especially the fundamental role of microbes as drivers of ecosystem functioning.[18][11]
Lithosphere
[edit]Reservoirs
[edit]The chemicals are sometimes held for long periods of time in one place. This place is called a reservoir, which, for example, includes such things as coal deposits that are storing carbon for a long period of time.[23] When chemicals are held for only short periods of time, they are being held in exchange pools. Examples of exchange pools include plants and animals.[23]
Plants and animals utilize carbon to produce carbohydrates, fats, and proteins, which can then be used to build their internal structures or to obtain energy. Plants and animals temporarily use carbon in their systems and then release it back into the air or surrounding medium. Generally, reservoirs are abiotic factors whereas exchange pools are biotic factors. Carbon is held for a relatively short time in plants and animals in comparison to coal deposits. The amount of time that a chemical is held in one place is called its residence time or turnover time (also called the renewal time or exit age).[23]
Box models
[edit]
Box models are widely used to model biogeochemical systems.[24][25] Box models are simplified versions of complex systems, reducing them to boxes (or storage reservoirs) for chemical materials, linked by material fluxes (flows). Simple box models have a small number of boxes with properties, such as volume, that do not change with time. The boxes are assumed to behave as if they were mixed homogeneously.[25] These models are often used to derive analytical formulas describing the dynamics and steady-state abundance of the chemical species involved.
The diagram at the right shows a basic one-box model. The reservoir contains the amount of material M under consideration, as defined by chemical, physical or biological properties. The source Q is the flux of material into the reservoir, and the sink S is the flux of material out of the reservoir. The budget is the check and balance of the sources and sinks affecting material turnover in a reservoir. The reservoir is in a steady state if Q = S, that is, if the sources balance the sinks and there is no change over time.[25]
The residence or turnover time is the average time material spends resident in the reservoir. If the reservoir is in a steady state, this is the same as the time it takes to fill or drain the reservoir. Thus, if τ is the turnover time, then τ = M/S.[25] The equation describing the rate of change of content in a reservoir is
When two or more reservoirs are connected, the material can be regarded as cycling between the reservoirs, and there can be predictable patterns to the cyclic flow.[25] More complex multibox models are usually solved using numerical techniques.

Global biogeochemical box models usually measure:
- reservoir masses in petagrams (Pg)
- flow fluxes in petagrams per year (Pg yr−1)
The diagram on the left shows a simplified budget of ocean carbon flows. It is composed of three simple interconnected box models, one for the euphotic zone, one for the ocean interior or dark ocean, and one for ocean sediments. In the euphotic zone, net phytoplankton production is about 50 Pg C each year. About 10 Pg is exported to the ocean interior while the other 40 Pg is respired. Organic carbon degradation occurs as particles (marine snow) settle through the ocean interior. Only 2 Pg eventually arrives at the seafloor, while the other 8 Pg is respired in the dark ocean. In sediments, the time scale available for degradation increases by orders of magnitude with the result that 90% of the organic carbon delivered is degraded and only 0.2 Pg C yr−1 is eventually buried and transferred from the biosphere to the geosphere.[26]

The diagram on the right shows a more complex model with many interacting boxes. Reservoir masses here represents carbon stocks, measured in Pg C. Carbon exchange fluxes, measured in Pg C yr−1, occur between the atmosphere and its two major sinks, the land and the ocean. The black numbers and arrows indicate the reservoir mass and exchange fluxes estimated for the year 1750, just before the Industrial Revolution. The red arrows (and associated numbers) indicate the annual flux changes due to anthropogenic activities, averaged over the 2000–2009 time period. They represent how the carbon cycle has changed since 1750. Red numbers in the reservoirs represent the cumulative changes in anthropogenic carbon since the start of the Industrial Period, 1750–2011.[28][29][27]
Fast and slow cycles
[edit]

There are fast and slow biogeochemical cycles. Fast cycle operate in the biosphere and slow cycles operate in the lithosphere in rocks. Fast or biological cycles can complete within years, moving substances from atmosphere to biosphere, then back to the atmosphere. Slow or geological cycles can take millions of years to complete, moving substances through the Earth's crust between rocks, soil, ocean and atmosphere.[31]
As an example, the fast carbon cycle is illustrated in the diagram on the right. This cycle involves relatively short-term biogeochemical processes between the environment and living organisms in the biosphere. It includes movements of carbon between the atmosphere and terrestrial and marine ecosystems, as well as soils and seafloor sediments. The fast cycle includes annual cycles involving photosynthesis and decadal cycles involving vegetative growth and decomposition. The reactions of the fast carbon cycle to human activities will determine many of the more immediate impacts of climate change.[32][33][34][35]
The slow cycle is illustrated in the other diagram. It involves medium to long-term geochemical processes belonging to the rock cycle. The exchange between the ocean and atmosphere can take centuries, and the weathering of rocks can take millions of years. Carbon in the ocean precipitates to the ocean floor where it can form sedimentary rock and be subducted into the Earth's mantle. Mountain building processes result in the return of this geologic carbon to the Earth's surface. There the rocks are weathered and carbon is returned to the atmosphere by degassing and to the ocean by rivers. Other geologic carbon returns to the ocean through the hydrothermal emission of calcium ions. In a given year between 10 and 100 million tonnes of carbon moves around this slow cycle. This includes volcanoes returning geologic carbon directly to the atmosphere in the form of carbon dioxide. However, this is less than one percent of the carbon dioxide put into the atmosphere by burning fossil fuels.[31][32]
Deep cycles
[edit]The terrestrial subsurface is the largest reservoir of carbon on earth, containing 14–135 Pg of carbon[36] and 2–19% of all biomass.[37] Microorganisms drive organic and inorganic compound transformations in this environment and thereby control biogeochemical cycles. Current knowledge of the microbial ecology of the subsurface is primarily based on 16S ribosomal RNA (rRNA) gene sequences. Recent estimates show that <8% of 16S rRNA sequences in public databases derive from subsurface organisms[38] and only a small fraction of those are represented by genomes or isolates. Thus, there is remarkably little reliable information about microbial metabolism in the subsurface. Further, little is known about how organisms in subsurface ecosystems are metabolically interconnected. Some cultivation-based studies of syntrophic consortia[39][40][41] and small-scale metagenomic analyses of natural communities[42][43][44] suggest that organisms are linked via metabolic handoffs: the transfer of redox reaction products of one organism to another. However, no complex environments have been dissected completely enough to resolve the metabolic interaction networks that underpin them. This restricts the ability of biogeochemical models to capture key aspects of the carbon and other nutrient cycles.[45] New approaches such as genome-resolved metagenomics, an approach that can yield a comprehensive set of draft and even complete genomes for organisms without the requirement for laboratory isolation[42][46][47] have the potential to provide this critical level of understanding of biogeochemical processes.[48]
Some examples
[edit]Some of the more well-known biogeochemical cycles are shown below:
Many biogeochemical cycles are currently being studied for the first time. Climate change and human impacts are drastically changing the speed, intensity, and balance of these relatively unknown cycles, which include:
- the mercury cycle,[49] and
- the human-caused cycle of PCBs.[50]
-
Coal is a reservoir of carbon
Biogeochemical cycles always involve active equilibrium states: a balance in the cycling of the element between compartments. However, overall balance may involve compartments distributed on a global scale.
As biogeochemical cycles describe the movements of substances on the entire globe, the study of these is inherently multidisciplinary. The carbon cycle may be related to research in ecology and atmospheric sciences.[53] Biochemical dynamics would also be related to the fields of geology and pedology.[54]
See also
[edit]- Carbonate–silicate cycle – Geochemical transformation of silicate rocks
- Ecological recycling – Set of processes exchanging nutrients between parts of a system
- Great Acceleration – Surge in human activity and impact upon the Earth
- Hydrogen cycle – Hydrogen exchange between the living and non-living world
- Redox gradient – Variation of the redox potential with distance (or depth)
References
[edit]- ^ "CK12-Foundation". flexbooks.ck12.org. Retrieved 2022-03-21.
- ^ a b Moses, M. (2012) Biogeochemical cycles Archived 2021-11-22 at the Wayback Machine. Encyclopedia of Earth.
- ^ Biogeochemical Cycles Archived 2021-09-27 at the Wayback Machine, OpenStax, 9 May 2019.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
- ^ Fisher M. R. (Ed.) (2019) Environmental Biology, 3.2 Biogeochemical Cycles Archived 2021-09-27 at the Wayback Machine, OpenStax.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
- ^ a b "Biogeochemical Cycles". The Environmental Literacy Council. Archived from the original on 30 April 2015. Retrieved 20 November 2017.
- ^ Avelar, S., van der Voort, T.S. and Eglinton, T.I. (2017) "Relevance of carbon stocks of marine sediments for national greenhouse gas inventories of maritime nations". Carbon balance and management, 12(1): 10.doi:10.1186/s13021-017-0077-x.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License. Archived 2017-10-16 at the Wayback Machine.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License. Archived 2017-10-16 at the Wayback Machine.
- ^ Falkowski, P. G.; Fenchel, T.; Delong, E. F. (2008). "The Microbial Engines That Drive Earth's Biogeochemical Cycles". Science. 320 (5879): 1034–1039. Bibcode:2008Sci...320.1034F. doi:10.1126/science.1153213. PMID 18497287. S2CID 2844984.
- ^ Zakem, Emily J.; Polz, Martin F.; Follows, Michael J. (2020). "Redox-informed models of global biogeochemical cycles". Nature Communications. 11 (1): 5680. Bibcode:2020NatCo..11.5680Z. doi:10.1038/s41467-020-19454-w. PMC 7656242. PMID 33173062.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
- ^ Henley, Sian F.; Cavan, Emma L.; Fawcett, Sarah E.; Kerr, Rodrigo; Monteiro, Thiago; Sherrell, Robert M.; Bowie, Andrew R.; Boyd, Philip W.; Barnes, David K. A.; Schloss, Irene R.; Marshall, Tanya; Flynn, Raquel; Smith, Shantelle (2020). "Changing Biogeochemistry of the Southern Ocean and Its Ecosystem Implications". Frontiers in Marine Science. 7: 581. Bibcode:2020FrMaS...7..581H. doi:10.3389/fmars.2020.00581. hdl:11336/128446.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
- ^ Alexander, Vera; Miloslavich, Patricia; Yarincik, Kristen (2011). "The Census of Marine Life—evolution of worldwide marine biodiversity research". Marine Biodiversity. 41 (4): 545–554. Bibcode:2011MarBd..41..545A. doi:10.1007/s12526-011-0084-1. S2CID 25888475.
- ^ a b c Murillo, Alejandro A.; Molina, Verónica; Salcedo-Castro, Julio; Harrod, Chris (2019). "Editorial: Marine Microbiome and Biogeochemical Cycles in Marine Productive Areas". Frontiers in Marine Science. 6: 657. Bibcode:2019FrMaS...6..657M. doi:10.3389/fmars.2019.00657.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
- ^ Galton, D. (1884) 10th Meeting: report of the royal commission on metropolitan sewage Archived 2021-09-24 at the Wayback Machine. J. Soc. Arts, 33: 290.
- ^ Hasler, Arthur D. (1969). "Cultural Eutrophication is Reversible". BioScience. 19 (5): 425–431. doi:10.2307/1294478. JSTOR 1294478.
- ^ Jickells, T. D.; Buitenhuis, E.; Altieri, K.; Baker, A. R.; Capone, D.; Duce, R. A.; Dentener, F.; Fennel, K.; Kanakidou, M.; Laroche, J.; Lee, K.; Liss, P.; Middelburg, J. J.; Moore, J. K.; Okin, G.; Oschlies, A.; Sarin, M.; Seitzinger, S.; Sharples, J.; Singh, A.; Suntharalingam, P.; Uematsu, M.; Zamora, L. M. (2017). "A reevaluation of the magnitude and impacts of anthropogenic atmospheric nitrogen inputs on the ocean". Global Biogeochemical Cycles. 31 (2): 289. Bibcode:2017GBioC..31..289J. doi:10.1002/2016GB005586. hdl:1874/348077. S2CID 5158406.
- ^ Bouwman, A. F.; Van Drecht, G.; Knoop, J. M.; Beusen, A. H. W.; Meinardi, C. R. (2005). "Exploring changes in river nitrogen export to the world's oceans". Global Biogeochemical Cycles. 19 (1). Bibcode:2005GBioC..19.1002B. doi:10.1029/2004GB002314. S2CID 131163837.
- ^ Altieri, Andrew H.; Gedan, Keryn B. (2015). "Climate change and dead zones". Global Change Biology. 21 (4): 1395–1406. Bibcode:2015GCBio..21.1395A. doi:10.1111/gcb.12754. PMID 25385668. S2CID 24002134.
- ^ a b Breitburg, Denise; Levin, Lisa A.; Oschlies, Andreas; Grégoire, Marilaure; Chavez, Francisco P.; Conley, Daniel J.; Garçon, Véronique; Gilbert, Denis; Gutiérrez, Dimitri; Isensee, Kirsten; Jacinto, Gil S.; Limburg, Karin E.; Montes, Ivonne; Naqvi, S. W. A.; Pitcher, Grant C.; Rabalais, Nancy N.; Roman, Michael R.; Rose, Kenneth A.; Seibel, Brad A.; Telszewski, Maciej; Yasuhara, Moriaki; Zhang, Jing (2018). "Declining oxygen in the global ocean and coastal waters". Science. 359 (6371) eaam7240. Bibcode:2018Sci...359M7240B. doi:10.1126/science.aam7240. PMID 29301986. S2CID 206657115.
- ^ a b Cavicchioli, Ricardo; et al. (2019). "Scientists' warning to humanity: Microorganisms and climate change". Nature Reviews Microbiology. 17 (9): 569–586. doi:10.1038/s41579-019-0222-5. PMC 7136171. PMID 31213707.
- ^ a b Hutchins, David A.; Jansson, Janet K.; Remais, Justin V.; Rich, Virginia I.; Singh, Brajesh K.; Trivedi, Pankaj (2019). "Climate change microbiology — problems and perspectives". Nature Reviews Microbiology. 17 (6): 391–396. doi:10.1038/s41579-019-0178-5. PMID 31092905. S2CID 155102440.
- ^ Stillman, Jonathon H.; Paganini, Adam W. (2015). "Biochemical adaptation to ocean acidification". Journal of Experimental Biology. 218 (12): 1946–1955. Bibcode:2015JExpB.218.1946S. doi:10.1242/jeb.115584. PMID 26085671. S2CID 13071345.
- ^ Bertagnolli, Anthony D.; Stewart, Frank J. (2018). "Microbial niches in marine oxygen minimum zones". Nature Reviews Microbiology. 16 (12): 723–729. doi:10.1038/s41579-018-0087-z. PMID 30250271. S2CID 52811177.
- ^ Ulloa, O.; Canfield, D. E.; Delong, E. F.; Letelier, R. M.; Stewart, F. J. (2012). "Microbial oceanography of anoxic oxygen minimum zones". Proceedings of the National Academy of Sciences. 109 (40): 15996–16003. Bibcode:2012PNAS..10915996U. doi:10.1073/pnas.1205009109. PMC 3479542. PMID 22967509. S2CID 6630698.
- ^ a b c Baedke, Steve J.; Fichter, Lynn S. "Biogeochemical Cycles: Carbon Cycle". Supplemental Lecture Notes for Geol 398. James Madison University. Archived from the original on 1 December 2017. Retrieved 20 November 2017.
- ^ Sarmiento, J.L.; Toggweiler, J.R. (1984). "A new model for the role of the oceans in determining atmospheric P CO 2". Nature. 308 (5960): 621–24. Bibcode:1984Natur.308..621S. doi:10.1038/308621a0. S2CID 4312683.
- ^ a b c d e Bianchi, Thomas (2007) Biogeochemistry of Estuaries Archived 2021-09-25 at the Wayback Machine page 9, Oxford University Press. ISBN 9780195160826.
- ^ a b Middelburg, J.J.(2019) Marine carbon biogeochemistry: a primer for earth system scientists, page 5, Springer Nature. ISBN 9783030108229. doi:10.1007/978-3-030-10822-9.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
- ^ a b Kandasamy, Selvaraj; Nagender Nath, Bejugam (2016). "Perspectives on the Terrestrial Organic Matter Transport and Burial along the Land-Deep Sea Continuum: Caveats in Our Understanding of Biogeochemical Processes and Future Needs". Frontiers in Marine Science. 3: 259. Bibcode:2016FrMaS...3..259K. doi:10.3389/fmars.2016.00259. S2CID 30408500.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
- ^ Sarmiento, Jorge L.; Gruber, Nicolas (2002). "Sinks for Anthropogenic Carbon". Physics Today. 55 (8): 30–36. Bibcode:2002PhT....55h..30S. doi:10.1063/1.1510279. S2CID 128553441.
- ^ Chhabra, Abha (2013). "Carbon and Other Biogeochemical Cycles". Intergovernmental Panel on Climate Change. doi:10.13140/2.1.1081.8883.
- ^ Riebeek, Holli (16 June 2011). "The Carbon Cycle". Earth Observatory. NASA. Archived from the original on 5 March 2016. Retrieved 5 April 2018.
- ^ a b Libes, Susan M. (2015). Blue planet: The role of the oceans in nutrient cycling, maintain the atmosphere system, and modulating climate change Archived 2021-01-20 at the Wayback Machine In: Routledge Handbook of Ocean Resources and Management, Routledge, pages 89–107. ISBN 9781136294822.
- ^ a b Bush, Martin J. (2020). Climate Change and Renewable Energy. pp. 109–141. doi:10.1007/978-3-030-15424-0_3. ISBN 978-3-030-15423-3. S2CID 210305910. Archived from the original on 2021-09-27. Retrieved 2021-09-27.
- ^ Rothman, D. H. (2002). "Atmospheric carbon dioxide levels for the last 500 million years". Proceedings of the National Academy of Sciences. 99 (7): 4167–4171. Bibcode:2002PNAS...99.4167R. doi:10.1073/pnas.022055499. PMC 123620. PMID 11904360.
- ^ Carpinteri, Alberto; Niccolini, Gianni (2019). "Correlation between the Fluctuations in Worldwide Seismicity and Atmospheric Carbon Pollution". Sci. 1: 17. doi:10.3390/sci1010017.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
- ^ Rothman, Daniel (January 2015). "Earth's carbon cycle: A mathematical perspective". Bulletin of the American Mathematical Society. 52 (1): 47–64. doi:10.1090/S0273-0979-2014-01471-5. hdl:1721.1/97900. ISSN 0273-0979. Archived from the original on 2021-11-22. Retrieved 2021-09-27.
- ^ McMahon, Sean; Parnell, John (2014). "Weighing the deep continental biosphere". FEMS Microbiology Ecology. 87 (1): 113–120. Bibcode:2014FEMME..87..113M. doi:10.1111/1574-6941.12196. PMID 23991863. S2CID 9491320.
- ^ Kallmeyer, J.; Pockalny, R.; Adhikari, R. R.; Smith, D. C.; d'Hondt, S. (2012). "Global distribution of microbial abundance and biomass in subseafloor sediment". Proceedings of the National Academy of Sciences. 109 (40): 16213–16216. doi:10.1073/pnas.1203849109. PMC 3479597. PMID 22927371.
- ^ Schloss, Patrick D.; Girard, Rene A.; Martin, Thomas; Edwards, Joshua; Thrash, J. Cameron (2016). "Status of the Archaeal and Bacterial Census: An Update". mBio. 7 (3). doi:10.1128/mBio.00201-16. PMC 4895100. PMID 27190214.
- ^ Abreu, Nicole A.; Taga, Michiko E. (2016). "Decoding molecular interactions in microbial communities". FEMS Microbiology Reviews. 40 (5): 648–663. doi:10.1093/femsre/fuw019. PMC 5007284. PMID 27417261.
- ^ Bosse, Magnus; Heuwieser, Alexander; Heinzel, Andreas; Nancucheo, Ivan; Melo Barbosa Dall'Agnol, Hivana; Lukas, Arno; Tzotzos, George; Mayer, Bernd (2015). "Interaction networks for identifying coupled molecular processes in microbial communities". BioData Mining. 8: 21. doi:10.1186/s13040-015-0054-4. PMC 4502522. PMID 26180552.
- ^ Braker, Gesche; Dörsch, Peter; Bakken, Lars R. (2012). "Genetic characterization of denitrifier communities with contrasting intrinsic functional traits". FEMS Microbiology Ecology. 79 (2): 542–554. Bibcode:2012FEMME..79..542B. doi:10.1111/j.1574-6941.2011.01237.x. PMID 22092293.
- ^ a b Hug, Laura A.; Thomas, Brian C.; Sharon, Itai; Brown, Christopher T.; Sharma, Ritin; Hettich, Robert L.; Wilkins, Michael J.; Williams, Kenneth H.; Singh, Andrea; Banfield, Jillian F. (2016). "Critical biogeochemical functions in the subsurface are associated with bacteria from new phyla and little studied lineages". Environmental Microbiology. 18 (1): 159–173. Bibcode:2016EnvMi..18..159H. doi:10.1111/1462-2920.12930. PMID 26033198. S2CID 43160538. Archived from the original on 2021-09-27. Retrieved 2021-09-27.
- ^ McCarren, J.; Becker, J. W.; Repeta, D. J.; Shi, Y.; Young, C. R.; Malmstrom, R. R.; Chisholm, S. W.; Delong, E. F. (2010). "Microbial community transcriptomes reveal microbes and metabolic pathways associated with dissolved organic matter turnover in the sea". Proceedings of the National Academy of Sciences. 107 (38): 16420–16427. doi:10.1073/pnas.1010732107. PMC 2944720. PMID 20807744.
- ^ Embree, Mallory; Liu, Joanne K.; Al-Bassam, Mahmoud M.; Zengler, Karsten (2015). "Networks of energetic and metabolic interactions define dynamics in microbial communities". Proceedings of the National Academy of Sciences. 112 (50): 15450–15455. Bibcode:2015PNAS..11215450E. doi:10.1073/pnas.1506034112. PMC 4687543. PMID 26621749.
- ^ Long, Philip E.; Williams, Kenneth H.; Hubbard, Susan S.; Banfield, Jillian F. (2016). "Microbial Metagenomics Reveals Climate-Relevant Subsurface Biogeochemical Processes". Trends in Microbiology. 24 (8): 600–610. doi:10.1016/j.tim.2016.04.006. PMID 27156744. S2CID 3983278.
- ^ Eren, A. Murat; Esen, Özcan C.; Quince, Christopher; Vineis, Joseph H.; Morrison, Hilary G.; Sogin, Mitchell L.; Delmont, Tom O. (2015). "Anvi'o: An advanced analysis and visualization platform for 'omics data". PeerJ. 3 e1319. doi:10.7717/peerj.1319. PMC 4614810. PMID 26500826.
- ^ Alneberg, Johannes; Bjarnason, Brynjar Smári; De Bruijn, Ino; Schirmer, Melanie; Quick, Joshua; Ijaz, Umer Z.; Lahti, Leo; Loman, Nicholas J.; Andersson, Anders F.; Quince, Christopher (2014). "Binning metagenomic contigs by coverage and composition". Nature Methods. 11 (11): 1144–1146. doi:10.1038/nmeth.3103. PMID 25218180. S2CID 24696869.
- ^ Anantharaman, Karthik; Brown, Christopher T.; Hug, Laura A.; Sharon, Itai; Castelle, Cindy J.; Probst, Alexander J.; Thomas, Brian C.; Singh, Andrea; Wilkins, Michael J.; Karaoz, Ulas; Brodie, Eoin L.; Williams, Kenneth H.; Hubbard, Susan S.; Banfield, Jillian F. (2016). "Thousands of microbial genomes shed light on interconnected biogeochemical processes in an aquifer system". Nature Communications. 7 13219. Bibcode:2016NatCo...713219A. doi:10.1038/ncomms13219. PMC 5079060. PMID 27774985.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
- ^ "Mercury Cycling in the Environment". Wisconsin Water Science Center. United States Geological Survey. 10 January 2013. Archived from the original on 11 April 2021. Retrieved 20 November 2017.
- ^ Organic contaminants that leave traces: sources, transport and fate. Ifremer. 2006. pp. 22–23. ISBN 978-2-7592-0013-9.
- ^ Galy, Valier; Peucker-Ehrenbrink, Bernhard; Eglinton, Timothy (2015). "Global carbon export from the terrestrial biosphere controlled by erosion". Nature. 521 (7551): 204–207. Bibcode:2015Natur.521..204G. doi:10.1038/nature14400. PMID 25971513. S2CID 205243485.
- ^ Hedges, J.I; Oades, J.M (1997). "Comparative organic geochemistries of soils and marine sediments". Organic Geochemistry. 27 (7–8): 319–361. Bibcode:1997OrGeo..27..319H. doi:10.1016/S0146-6380(97)00056-9.
- ^ McGuire, 1A. D.; Lukina, N. V. (2007). "Biogeochemical cycles" (PDF). In Groisman, P.; Bartalev, S. A.; NEESPI Science Plan Development Team (eds.). Northern Eurasia earth science partnership initiative (NEESPI), Science plan overview. Global Planetary Change. Vol. 56. pp. 215–234. Archived (PDF) from the original on 5 March 2016. Retrieved 20 November 2017.
{{cite book}}: CS1 maint: numeric names: authors list (link) - ^ "Distributed Active Archive Center for Biogeochemical Dynamics". daac.ornl.gov. Oak Ridge National Laboratory. Archived from the original on 11 February 2011. Retrieved 20 November 2017.
Further reading
[edit]- Schink, Bernhard; "Microbes: Masters of the Global Element Cycles" pp 33–58. "Metals, Microbes and Minerals: The Biogeochemical Side of Life", pp xiv + 341. Walter de Gruyter, Berlin. DOI 10.1515/9783110589771-002
- Butcher, Samuel S., ed. (1993). Global biogeochemical cycles. London: Academic Press. ISBN 978-0-08-095470-7.
- Exley, C (15 September 2003). "A biogeochemical cycle for aluminium?". Journal of Inorganic Biochemistry. 97 (1): 1–7. doi:10.1016/S0162-0134(03)00274-5. PMID 14507454.
- Jacobson, Michael C.; Charlson, Robert J.; Rodhe, Henning; Orians, Gordon H. (2000). Earth system science from biogeochemical cycles to global change (2nd ed.). San Diego, Calif.: Academic Press. ISBN 978-0-08-053064-2.
- Palmeri, Luca; Barausse, Alberto; Jorgensen, Sven Erik (2013). "12. Biogeochemical cycles". Ecological processes handbook. Boca Raton: Taylor & Francis. ISBN 978-1-4665-5848-9.
Biogeochemical cycle
View on GrokipediaFundamentals
Definition and Core Principles
Biogeochemical cycles refer to the pathways through which chemical elements and compounds, such as carbon, nitrogen, phosphorus, and sulfur, move and transform across the Earth's atmosphere, hydrosphere, biosphere, and lithosphere.[1] These cycles integrate biological processes—like photosynthesis and decomposition—with geological mechanisms, such as weathering and sedimentation, and chemical reactions, including oxidation and reduction, to recycle essential nutrients and maintain ecosystem productivity.[3] The term "biogeochemical" derives from "bio" (referring to the biosphere), "geo" (the geosphere), and "chemical" (elemental transformations), emphasizing the interplay among living organisms, Earth's physical compartments, and molecular changes.[7] At their core, these cycles operate on the principle of matter conservation within a relatively closed planetary system, where elements are neither created nor destroyed but redistributed through fluxes between reservoirs.[8] Biological organisms play a pivotal role in accelerating transformations, such as nitrogen fixation by bacteria or carbon sequestration via plant uptake, which abiotic processes alone would conduct far more slowly.[9] Feedback mechanisms, including positive ones like enhanced weathering from increased CO2 levels or negative ones like nutrient limitation curbing primary production, regulate cycle dynamics and contribute to Earth's long-term habitability by modulating atmospheric composition and climate.[10] These principles underscore the interdependence of Earth's spheres: disruptions in one cycle, such as anthropogenic nitrogen additions exceeding natural fluxes by factors of 2-3 globally since the mid-20th century, can cascade through food webs and alter other cycles like carbon storage.[11] Empirical measurements, including isotopic tracing and flux budgeting, reveal that biotic mediation distinguishes biogeochemical cycles from purely geochemical ones, enabling efficient nutrient recycling that supports biodiversity and planetary homeostasis.[5]Historical Development
Early investigations into processes resembling biogeochemical cycles emerged in the 17th and 18th centuries through studies of photosynthesis, decomposition, and elemental transformations. In 1699, John Woodward conducted experiments on plant growth using water from different sources, highlighting the role of minerals in vegetation. Joseph Priestley demonstrated the restoration of air by plants in 1772, while Jan Ingenhousz elucidated photosynthesis in 1779, linking biological activity to atmospheric gases. Antoine Lavoisier advanced chemical understanding of respiration and combustion in the late 18th century, establishing foundational principles for element cycling.[12][13] Geological and chemical perspectives contributed in the 19th century, with James Hutton's 1785 uniformitarianism emphasizing continuous Earth processes, and George Bischof's 1826 work on rock weathering. Justus Liebig's 1840 agricultural chemistry and 1862 soil fertility studies stressed nutrient recycling, while Robert Warington's 1851 analyses connected organic and inorganic realms. Sergei Winogradsky's discoveries of chemosynthesis (1887) and nitrification (1891–1893) revealed microbial mediation of elemental transformations, bridging biology and geochemistry. Svante Arrhenius's 1896 calculations on CO2's climatic effects linked geochemical cycles to atmospheric dynamics.[12][13] The formal discipline of biogeochemistry crystallized in the early 20th century through Vladimir Ivanovich Vernadsky's integrative framework. In 1924, Vernadsky published La Géochimie, laying groundwork for geochemical analysis of living systems. His 1926 La Biosphère defined the biosphere as a geochemical force driven by living matter and coined "biogeochemistry" to describe the interplay of biological, geological, and chemical processes in element cycling. By 1945, in "The Biosphere and the Noösphere," Vernadsky extended this to human influences on planetary systems. These works synthesized prior elemental studies into a holistic view, establishing biogeochemistry as a distinct field.[12] Post-1920s advancements incorporated microbial and ecological insights, with Cornelis B. van Niel's 1929–1949 research on bacterial photosynthesis refining cycle mechanisms, and A.P. Vinogradov's 1953 analyses of marine organism composition quantifying biotic fluxes. G.E. Hutchinson's 1940s–1950s treatises, including his 1947 phosphorus cycle model, formalized quantitative cycling in ecosystems. The field expanded globally from the 1960s, integrating long-term experiments like Hubbard Brook (1967) and addressing human perturbations, culminating in syntheses such as Bert Bolin et al.'s 1983 The Major Biogeochemical Cycles. This evolution shifted focus from local processes to planetary-scale modeling and climate interactions.[12][13]System Components
Reservoirs and Pools
In biogeochemical cycles, reservoirs denote the large, predominantly abiotic compartments that store chemical elements and compounds for extended periods, often spanning geological timescales, while pools encompass both these reservoirs and smaller, more dynamic exchange pools that facilitate biotic interactions and rapid turnover. Reservoirs typically exhibit low exchange rates due to physical or chemical stability, serving as long-term sinks that regulate elemental availability across Earth's systems, whereas exchange pools—such as surface ocean layers or soil organic matter—enable fluxes between biotic and abiotic realms. This compartmentalization reflects the interplay of residence times, with reservoirs maintaining vast inventories against depletion.[14][3] Major reservoirs vary by element, dictated by chemical reactivity and geological history. In the carbon cycle, the lithosphere holds approximately 70,000,000 gigatons (Gt), chiefly in sedimentary carbonates and organic sediments, the ocean contains 38,000 Gt partitioned between dissolved inorganic and organic forms, the atmosphere stores 750 Gt as CO₂ and minor gases, and the biosphere maintains 600 Gt in terrestrial vegetation, detritus, and soils.[15] These sizes underscore carbon's dual gaseous-sedimentary character, with oceanic and lithospheric pools dominating storage.[16] For nitrogen, the atmospheric reservoir overwhelms at 3,900,000 teragrams (Tg) of N₂, dwarfing oceanic dissolved nitrogen at 20,000 Tg and biospheric pools at 200 Tg in organisms and soils, which limits bioavailability despite abundance.[15] Phosphorus, conversely, features sedimentary dominance, with lithospheric reserves of 1,000,000 Tg in apatite minerals, oceanic pools of 90,000 Tg in sediments and water column, and scant biospheric storage of 3 Tg, reflecting its low mobility and reliance on rock weathering.[15] Such reservoirs ensure elemental persistence amid fluxes, with human perturbations—like fossil fuel extraction altering carbon pools—potentially disrupting equilibria by mobilizing lithospheric stores at accelerated rates.[17]Fluxes, Transformations, and Rates
Fluxes denote the rates at which chemical elements transfer between reservoirs in biogeochemical cycles, quantified as mass flow per unit time, such as gigatons per year (Gt yr⁻¹). These transfers, including processes like evaporation, sedimentation, and biological uptake, sustain elemental distribution across Earth's atmosphere, oceans, land, and biosphere. In the global carbon cycle, for example, photosynthetic fixation by terrestrial vegetation and marine phytoplankton drives a flux of approximately 120 GtC yr⁻¹ from the atmosphere to biomass, while ocean outgassing and dissolution contribute to bidirectional air-sea exchanges of about 90 GtC yr⁻¹.[18] Similarly, nitrogen fluxes involve atmospheric deposition and biological fixation totaling around 140 TgN yr⁻¹, with riverine transport to oceans at roughly 50 TgN yr⁻¹.[19] Transformations refer to the chemical alterations of elements during their cycling, encompassing reactions such as oxidation-reduction, precipitation-dissolution, and assimilation-mineralization, frequently catalyzed by microbial enzymes under specific redox potentials. These processes convert elements between inorganic and organic forms or alter their valence states, enabling bioavailability; for instance, nitrogen fixation transforms atmospheric N₂ into ammonium via nitrogenase enzymes in diazotrophic bacteria. Phosphorus transformations include weathering of apatite minerals to soluble phosphates, with global fluxes from rock weathering estimated at 0.026 GtP yr⁻¹ entering soil and aquatic systems.[20][21] Rates of fluxes and transformations are governed by environmental controls including temperature, moisture, pH, oxygen availability, and nutrient concentrations, which modulate kinetic parameters like microbial respiration and enzymatic activity. Elevated temperatures can accelerate decomposition rates, increasing carbon flux from soil organic matter by factors of 1.5 to 2 per 10°C rise under Q₁₀ assumptions, while anaerobic conditions favor denitrification over nitrification in nitrogen cycles, with gross transformation rates varying from 0.1 to 10 mgN kg⁻¹ soil d⁻¹ across ecosystems. Measurement of these rates often employs stable isotope tracing or flux chamber techniques to quantify in situ dynamics, revealing spatial heterogeneity; for example, oceanic nitrogen fixation rates range from 100 to 200 TgN yr⁻¹, concentrated in oligotrophic gyres.[22][23][18]Biotic and Abiotic Compartments
Biotic compartments comprise living organisms across the biosphere, including autotrophs, heterotrophs, and microorganisms, which facilitate the uptake, assimilation, and release of chemical elements. Primary producers, such as plants and algae, convert inorganic nutrients like atmospheric CO₂ and dissolved nitrates into organic biomass via photosynthesis and nitrogen fixation, while consumers and decomposers transfer and mineralize these elements through ingestion, respiration, and decay. Microbes dominate biotic transformations, recycling 350–550 gigatons of carbon and 85–130 gigatons of nitrogen annually through processes like ammonification and methanogenesis.[24] Abiotic compartments include non-living reservoirs in the atmosphere, hydrosphere, lithosphere, and pedosphere, where elements exist in gaseous, aqueous, or solid phases subject to physical and chemical alterations. The atmosphere stores volatile compounds like N₂ (78% of total volume) and CO₂ (approximately 0.04%), the oceans hold vast dissolved inorganic carbon (about 40,000 gigatons), and geological formations contain massive sedimentary pools, such as 81 million gigatons of carbon in limestone. Abiotic processes, including diffusion, precipitation, weathering, and volcanic outgassing, drive element mobilization and speciation without biological involvement.[24][25] Interactions between biotic and abiotic compartments generate dynamic fluxes essential to cycle continuity, with biological activities accelerating transformations in abiotic media and abiotic vectors supplying substrates to biota. For instance, microbial denitrification converts soil nitrates to atmospheric N₂, while runoff transports biotic residues like dissolved organic carbon from soils to aquatic systems, influencing redox conditions and bioavailability. These feedbacks, modulated by environmental factors such as temperature and pH, maintain elemental balances but can amplify perturbations like eutrophication when biotic demands exceed abiotic replenishment rates.[25][26]Dynamics and Timescales
Fast and Intermediate Cycles
Fast biogeochemical cycles operate on timescales from hours to several years, facilitating rapid nutrient and element turnover primarily through physical and biological processes in the atmosphere, hydrosphere, and biosphere. These cycles ensure short-term availability of essential compounds for ecosystems, with fluxes dominated by evaporation, precipitation, photosynthesis, respiration, and microbial activity. The water cycle represents a prototypical fast cycle, where atmospheric water vapor has a global mean residence time of about 9 days, enabling continuous redistribution via evaporation from oceans (contributing ~86% of atmospheric moisture) and precipitation returning ~78% to oceans annually. Similarly, the biological components of the oxygen cycle, involving photosynthetic production and respiratory consumption, cycle on scales of months to years, maintaining atmospheric O2 levels through net primary production estimated at 100-120 GtC yr⁻¹ globally.[16] Intermediate biogeochemical cycles encompass timescales from decades to several millennia, involving slower exchanges with larger reservoirs such as soils, surface oceans, and long-lived biomass. These cycles bridge fast biological dynamics and slow geological processes, often featuring accumulation and gradual release modulated by environmental factors like temperature and ocean circulation. In the carbon cycle, the fast biological component moves ~120 GtC yr⁻¹ through vegetation and soils over years to centuries, while oceanic uptake and mixing occur over centuries to millennia, with surface waters turning over in ~10 years but deep ocean ventilation requiring ~1,000 years.[16] The nitrogen cycle's intermediate aspects, including soil organic matter mineralization and denitrification, operate over decades to centuries, with global soil N turnover times averaging 50-100 years, influencing long-term fertility and greenhouse gas emissions like N2O. These timescales determine cycle responsiveness to perturbations; fast cycles quickly buffer short-term variability, whereas intermediate cycles integrate over longer periods, amplifying effects from land use or climate shifts. For instance, enhanced biological carbon uptake in fast phases can temporarily mitigate atmospheric CO2 rises, but intermediate soil and ocean reservoirs may release stored carbon under warming, as evidenced by observations of thawing permafrost contributing ~1.7 GtC over recent decades. Empirical models confirm that neglecting intermediate timescales underestimates feedbacks, with residence times in soils ranging 20-500 years depending on organic matter quality.Slow and Deep Geological Cycles
Slow and deep geological cycles involve the long-term transfer of elements between Earth's surface environments and its deep interior, primarily through tectonic, magmatic, and metamorphic processes operating on timescales of millions to billions of years. These cycles contrast with faster biotic and surficial exchanges by engaging vast reservoirs in the lithosphere, mantle, and crust, where elements like carbon and phosphorus are sequestered in rocks and sediments before potential recycling via subduction or exhumation. Plate tectonics serves as the primary driver, facilitating subduction of oceanic lithosphere laden with carbon- and nutrient-rich sediments into the mantle, while mid-ocean ridges and arc volcanism return mantle-derived volatiles to the surface.[27][28][4] In the geological carbon cycle, organic matter and carbonates buried in sediments over hundreds of thousands of years undergo diagenesis and deeper burial, locking away carbon for tens to hundreds of millions of years until tectonic uplift or subduction alters their fate. Subduction zones recycle an estimated 40–100 teragrams of carbon annually into the mantle, with 45–65% potentially released back through arc volcanism, influencing long-term atmospheric CO₂ concentrations and global climate stability over Phanerozoic timescales exceeding 500 million years. Variations in plate speeds modulate these fluxes; faster tectonics enhance degassing and weathering, correlating with elevated CO₂ and greenhouse conditions, as evidenced in models linking supercontinent assembly to climatic shifts.[29][30][31][32][28] The phosphorus cycle exemplifies slowness due to phosphorus's lack of a significant atmospheric phase, relying on rock weathering as the primary input, with continental uplift exposing fresh apatite deposits over hundreds of millions of years. Global phosphorus dynamics have evolved over 3.5 billion years, tied to tectonic exposure of continental crust and sedimentary recycling, limiting marine productivity on geological timescales and linking to carbon burial via organic matter co-precipitation. Subduction incorporates phosphorus into mantle phases, with minimal return flux compared to inputs, sustaining low oceanic concentrations over 20,000–100,000 years in the water column but extending full crustal recycling to eons.[33][34][35][36] These deep cycles maintain elemental balance against surficial perturbations, with tectonic rates—averaging 2–10 cm/year globally—dictating flux magnitudes and influencing habitability by buffering against extreme atmospheric compositions. Disruptions, such as during supercontinent cycles every 300–500 million years, alter weathering and volcanism, driving oscillations in nutrient availability and climate.[37][28]Modeling and Analysis
Box Models and Their Assumptions
Box models simplify biogeochemical cycles by partitioning the Earth system into discrete compartments, or "boxes," each representing a reservoir with a defined inventory of the element in question, connected by fluxes that quantify transfers between them.[38] These models apply mass balance principles, where the rate of change in mass within a box equals net inputs minus outputs, often expressed as , with as input flux, as output flux, and as residence time.[38] Early applications, such as three-box representations of the carbon cycle, treated the atmosphere as a single well-mixed reservoir exchanging with surface and deep ocean layers.[39] Core assumptions include internal homogeneity within each box, implying perfect mixing and negligible spatial gradients, which facilitates analytical solutions but overlooks heterogeneities like latitudinal or vertical variations in ocean biogeochemistry.[40] Fluxes are typically modeled as linear functions of reservoir masses, assuming constant transfer coefficients independent of external forcings, as in box-diffusion schemes for ocean carbon uptake where fractional partitioning to upper and lower layers remains fixed.[41] Steady-state conditions are often presumed for long-term inventories, equating inputs to outputs, though perturbations like anthropogenic emissions require transient formulations.[38] These simplifications enable tractable global-scale analyses, such as estimating nitrogen inventories across atmosphere, biosphere, and soils, but introduce limitations by aggregating processes that may exhibit nonlinear feedbacks or spatial dependencies.[38] For instance, two-box ocean models assume homogeneous surface and deep layers, ignoring upwelling or biological patchiness that spatially explicit models capture better.[40] Validation against observations, like tracer distributions, tests these assumptions, revealing overestimations in mixing times for complex cycles. Despite limitations, box models remain foundational for hypothesis testing and informing more detailed simulations.[43]Advanced Approaches and Recent Frameworks
Coupled physical-biogeochemical models represent a key advancement, integrating ocean circulation dynamics with nutrient and carbon transformations to simulate mesoscale and large-scale influences on cycle fluxes, as demonstrated in applications to the Indian Ocean where such models quantified upwelling-driven productivity enhancements.[44] These approaches address limitations of decoupled systems by resolving feedbacks, such as how physical advection modulates biological uptake rates, with resolutions down to eddy-permitting scales in recent implementations.[45] Data assimilation and model-data fusion frameworks have gained prominence for constraining uncertainties in biogeochemical simulations using sparse observations. The CARDAMOM (CARbon DAta MOdel fraMework) system, refined through multi-institutional efforts since the early 2000s, employs Bayesian optimization to fuse satellite, flux tower, and inventory data, yielding improved posterior estimates of terrestrial carbon stocks and fluxes with quantified error bounds across global ecosystems.[46] Similarly, variational inference methods applied to soil biogeochemical models enable probabilistic updates of differential equations governing carbon-nitrogen transfers, enhancing predictive skill for decomposition rates under varying moisture and temperature regimes.[47] In marine contexts, along-track assimilation frameworks link Biogeochemical Argo float profiles to one-dimensional models, providing near-real-time hindcasts of oxygen and nutrient anomalies with reduced biases relative to standalone simulations.[48] Machine learning integration and omics-informed parameterizations mark recent frontiers, particularly for resolving microbial-scale processes. Learning-based calibration techniques adjust ocean carbon model parameters against imperfect physical forcings and biogeochemical observations, achieving convergence in export production estimates that traditional least-squares methods fail to attain under data scarcity.[49] Genome-scale metabolic models embedded within Earth system models predict phytoplankton functional diversity and biogeochemical rates by linking gene expression to elemental stoichiometries, as shown in simulations revealing acclimation responses to iron limitation that alter global primary production by up to 15%.[50] These frameworks prioritize causal linkages from molecular traits to ecosystem fluxes, bypassing empirical parameterizations prone to extrapolation errors in altered climates.[51]Major Cycles
Carbon Cycle
The carbon cycle encompasses the continuous exchange of carbon between Earth's atmosphere, oceans, terrestrial biosphere, and geological reservoirs through physical, chemical, and biological processes. This cycle regulates atmospheric carbon dioxide (CO₂) concentrations, which influence global temperatures via the greenhouse effect, and sustains life by facilitating photosynthesis and respiration. Natural fluxes in the fast carbon cycle, involving biological and surface ocean processes, move approximately 120 gigatons of carbon (GtC) per year from the atmosphere to land and oceans via photosynthesis and solubility, balanced by equivalent returns through respiration, decomposition, and outgassing.[16][52] Major reservoirs include the atmosphere, holding about 900 GtC equivalent to roughly 426 parts per million (ppm) CO₂ as of 2025; the oceans, storing around 38,000 GtC primarily as dissolved bicarbonate and carbonate ions; the terrestrial biosphere, containing approximately 2,000–2,500 GtC in vegetation, soils, and detritus; and vast geological pools exceeding 60 million GtC in sedimentary rocks, fossil fuels, and mantle sources.[53][54] The slow geological cycle operates over millions of years, with fluxes of about 0.1 GtC per year from rock weathering removing CO₂ and volcanic degassing returning it, maintaining long-term equilibrium disrupted only minimally by biological activity.[16] Key transformations involve the biological pump in oceans, where phytoplankton fix CO₂ into organic matter that sinks as particulate flux, sequestering carbon in deep waters for centuries to millennia, and the solubility pump, driven by CO₂'s higher solubility in colder waters, leading to poleward transport and storage. On land, net primary productivity absorbs around 120 GtC annually, with roughly half respired back quickly, while soils and peatlands act as long-term sinks. Human activities, primarily fossil fuel combustion emitting about 10 GtC per year and land-use changes adding 1–2 GtC, have increased atmospheric CO₂ by over 140 ppm since pre-industrial levels, with oceans absorbing 25–31% and land sinks another 25–30% of anthropogenic emissions, though saturation risks emerge from acidification and warming.[54][52]Nitrogen Cycle
The nitrogen cycle comprises the microbial and abiotic transformations that convert nitrogen among its gaseous, organic, and inorganic forms across atmospheric, terrestrial, aquatic, and lithospheric reservoirs. Atmospheric dinitrogen gas (N₂), which constitutes 78% of the atmosphere and totals approximately 3.9 × 10¹⁵ teragrams of nitrogen, serves as the dominant reservoir but remains biologically inert due to its strong triple bond.[55] The cycle's core processes—nitrogen fixation, ammonification, nitrification, assimilation, denitrification, and anaerobic ammonium oxidation (anammox)—facilitate the continuous flux of nitrogen to support primary production while maintaining balance through returns to the atmosphere.[55] Nitrogen fixation initiates the cycle by reducing N₂ to ammonia (NH₃) or ammonium (NH₄⁺), enabling uptake by organisms. Biological nitrogen fixation (BNF), mediated by diazotrophic prokaryotes such as symbiotic Rhizobium bacteria in legume root nodules and free-living cyanobacteria, dominates natural inputs, with global rates estimated at 198 Tg N yr⁻¹ pre-industrially (terrestrial BNF at 58 Tg N yr⁻¹ and marine at 140 Tg N yr⁻¹). Abiotic fixation through lightning contributes an additional 5 Tg N yr⁻¹. Ammonification follows, as heterotrophic bacteria and fungi decompose organic nitrogen compounds (e.g., proteins, nucleic acids) from dead biomass into NH₄⁺, recycling nitrogen in soils and sediments.[56][55] Nitrification oxidizes NH₄⁺ to nitrite (NO₂⁻) via ammonia-oxidizing bacteria like Nitrosomonas, followed by nitrite-oxidizing bacteria such as Nitrobacter converting NO₂⁻ to nitrate (NO₃⁻), primarily in aerobic soils and waters. Plants and microbes then assimilate NO₃⁻ or NH₄⁺ into amino acids and other biomolecules. Closure occurs via denitrification, where facultative anaerobes (e.g., Pseudomonas) reduce NO₃⁻ stepwise to N₂ under oxygen-limited conditions, and anammox, in which anaerobic bacteria oxidize NH₄⁺ with NO₂⁻ to yield N₂, particularly in marine and wastewater environments. These reductive processes prevent indefinite accumulation of fixed nitrogen.[55] Pre-industrial total reactive nitrogen creation stood at about 203 Tg N yr⁻¹, but anthropogenic fixation—primarily the Haber-Bosch process synthesizing ~120 Tg N yr⁻¹ for fertilizers and enhanced crop BNF adding ~60 Tg N yr⁻¹—has elevated global totals to 413 Tg N yr⁻¹ by 2010, doubling fixed nitrogen availability. This amplification increases downstream fluxes, including ~100 Tg N yr⁻¹ of atmospheric NH₃ and NOₓ emissions and 40–70 Tg N yr⁻¹ of riverine delivery to oceans, altering cycle dynamics and contributing to issues like eutrophication and stratospheric ozone depletion via N₂O.[56]Phosphorus Cycle
The phosphorus cycle governs the transformation and transport of phosphorus, an essential element for nucleic acids, phospholipids, and energy transfer in organisms, primarily through sedimentary and biological processes without a significant gaseous phase.[57] Unlike carbon or nitrogen cycles, phosphorus mobility is limited to solid and aqueous forms, resulting in slower global turnover dominated by rock weathering and sediment burial.[58] Major reservoirs include continental rocks (~25,000,000 Tg P), marine sediments (~100,000,000 Tg P), soils (~27,000 Tg P), and ocean water (~3,000 Tg P), with fluxes measured in teragrams per year (Tg P/yr).[59] Primary inputs occur via tectonic uplift exposing phosphate-rich apatite minerals, followed by chemical weathering that solubilizes orthophosphate at global rates of 6-15 Tg P/yr under natural conditions.[60] In terrestrial ecosystems, plants assimilate soil phosphate through roots, incorporating it into biomass; herbivores and decomposers recycle it via consumption and mineralization, with soil retention influenced by adsorption to iron and aluminum oxides.[61] Erosion and runoff deliver 10-20 Tg P/yr to rivers, where much binds to particles or is taken up by aquatic biota before reaching oceans.[62] In marine environments, dissolved reactive phosphorus (DRP) concentrations average 2-3 μmol/L in deep waters, supporting phytoplankton growth; unused phosphorus sinks as organic particles, with burial in anoxic sediments exceeding inputs by 1-3 Tg P/yr pre-industrially, maintaining steady-state through geological uplift.[63] Atmospheric dust contributes minor fluxes of 0.6-1.3 Tg P/yr via aerosols, primarily from arid regions, but this is negligible compared to weathering.[64] Anthropogenic activities, including fertilizer application from mined rock phosphate (~20 Tg P/yr globally), have doubled riverine phosphorus exports to oceans (now ~10-15 Tg P/yr), disrupting balances and promoting eutrophication in freshwater systems via excess algal blooms and hypoxia.[60] [65] Long-term, non-renewable rock phosphate depletion projects peak extraction around 2030-2040, potentially limiting agricultural productivity absent recycling advancements.[66] Sedimentary recycling over millions of years underscores phosphorus scarcity relative to demand, with biological enhancements via mycorrhizal fungi optimizing uptake in P-limited soils.[61]Sulfur and Other Elemental Cycles
The biogeochemical sulfur cycle encompasses the transformations and transport of sulfur among atmospheric, oceanic, lithospheric, and biospheric reservoirs, primarily through microbial dissimilatory processes that shift sulfur between oxidized sulfate (SO₄²⁻, +6 oxidation state) and reduced sulfide (HS⁻ or H₂S, -2 oxidation state).[67] Key microbial pathways include dissimilatory sulfate reduction (DSR) by anaerobic bacteria in anoxic sediments and waters, producing sulfide as a metabolic end product, and subsequent sulfide oxidation by chemolithoautotrophic microbes under oxic or microoxic conditions.[68] These biological transformations dominate the cycle, integrating with abiotic inputs such as volcanic degassing (estimated at 10-20 teragrams of sulfur per year globally) and weathering of sulfide minerals, alongside oceanic emissions of dimethyl sulfide (DMS) from phytoplankton, contributing 15-33 Tg S yr⁻¹ to the atmosphere.[69] The cycle regulates Earth's redox balance, influences climate via sulfate aerosol formation that scatters sunlight and seeds clouds, and affects ocean acidity through sulfide oxidation products.[70] In marine environments, which host the largest sulfur reservoir in dissolved sulfate (approximately 2.7 x 10¹⁹ moles in seawater), the cycle is tightly coupled to organic matter remineralization in sediments where DSR consumes up to 90% of sulfate reduction in continental margins.[68] Sulfide produced diffuses upward and oxidizes, often reforming sulfate or intermediate species like elemental sulfur (S⁰) via disproportionation reactions facilitated by microbes such as Thiomargarita spp. Empirical measurements from coastal sediments indicate sulfate reduction rates of 0.1-10 mmol m⁻² day⁻¹, with isotopic fractionation (δ³⁴S up to 70‰) providing evidence of microbial control over these fluxes.[71] On land, sulfur inputs derive from atmospheric deposition (wet and dry, ~20-50 kg S ha⁻¹ yr⁻¹ in industrialized regions) and rock weathering, cycling through soils where plants assimilate sulfate for cysteine synthesis, followed by microbial immobilization and mineralization. Long-term sinks include pyrite burial in anoxic sediments, removing ~100-300 Tg S yr⁻¹ globally and stabilizing oxidized sulfur in the ocean.[72][73] Other elemental cycles, such as those of iron and silicon, operate on similar principles but with distinct reservoirs and biological dependencies. The iron cycle involves terrestrial weathering releasing Fe(II), atmospheric dust deposition to oceans (5-20 Tg Fe yr⁻¹, primarily from arid regions), and microbial mediation of oxidation-reduction, limiting phytoplankton productivity in high-nutrient low-chlorophyll (HNLC) regions where dissolved iron concentrations are <0.1 nM.[1] Silicon cycling centers on terrestrial silicate weathering and biogenic opal formation by diatoms, with oceanic export of biogenic silica (0.2-0.4 Gt Si yr⁻¹) driving burial in sediments and influencing carbon export via the biological pump.[4] These cycles interconnect with major nutrient loops; for instance, iron fertilization enhances diatom blooms, amplifying silicon and carbon sequestration, as demonstrated in Southern Ocean experiments where iron addition increased chlorophyll by 2-10 fold. Less prominent cycles for elements like calcium and manganese follow sedimentary pathways, with calcium cycling via carbonate dissolution-precipitation and manganese redox shuttling in sediments analogous to sulfur.[3] Empirical data underscore microbial primacy across these cycles, with genomic surveys revealing widespread sulfur-, iron-, and silicon-metabolizing genes in marine microbiomes.[74]Human Perturbations
Natural Variability Versus Anthropogenic Changes
Biogeochemical cycles exhibit natural variability driven by astronomical, geological, and biological processes, including Milankovitch orbital cycles that modulate insolation and trigger glacial-interglacial shifts in carbon sequestration, resulting in atmospheric CO₂ fluctuations of 80–100 ppm over 10,000–100,000-year timescales. Shorter-term variations, such as interannual perturbations from ENSO or volcanic eruptions, alter fluxes temporarily; for example, large eruptions like Pinatubo in 1991 reduced global net primary productivity and ocean CO₂ uptake, leading to transient atmospheric CO₂ increases of 0.5–1 ppm. In the nitrogen cycle, natural fixation by diazotrophs and lightning maintains a pre-industrial budget of approximately 90–140 Tg N yr⁻¹, with minimal long-term trends absent major perturbations like asteroid impacts.[75][76][77] Anthropogenic activities have imposed changes exceeding natural variability in both magnitude and rate across major cycles. Since 1750, fossil fuel combustion and deforestation have elevated atmospheric CO₂ from 280 ppm to 421 ppm by 2023, at a mean rate of ~2.5 ppm yr⁻¹—100 times faster than peak Holocene variations of ~0.01–0.02 ppm yr⁻¹ derived from ice-core records spanning 800,000 years. This rise bears a distinct isotopic fingerprint, with δ¹³C declining from -6.4‰ to -8.5‰ due to the depleted ¹³C in fossil-derived CO₂, ruling out dominant natural sources like volcanism or ocean outgassing. In the nitrogen cycle, human processes—chiefly Haber-Bosch synthesis for fertilizers (adding ~100–150 Tg N yr⁻¹) and fossil fuel NOx emissions (~30 Tg N yr⁻¹)—have doubled global reactive nitrogen creation to ~190–250 Tg N yr⁻¹, far surpassing natural terrestrial and marine fixation rates and causing widespread eutrophication and N₂O accumulation.[78][79][77][80] Detection and attribution analyses confirm these shifts cannot be replicated by natural forcings alone in Earth system models. For oceanic carbon uptake, anthropogenic signals emerged distinctly by the 1960s–2000s, with pCO₂ trends 20–50% higher than expected from internal variability like PDO or AMO oscillations, as validated against shipboard and float observations. Similarly, elevated N₂O concentrations (now ~335 ppb, up 20% since pre-industrial) trace primarily to agricultural soils and manure, with isotopic and isotopocule data distinguishing them from stratospheric or oceanic natural sources; human contributions account for ~40–50% of the emissions budget. While natural variability modulates short-term trends—e.g., amplifying or dampening regional uptake—long-term disequilibria, such as reduced soil carbon residence times from excess nitrogen deposition, require anthropogenic drivers for empirical consistency.[81][82][80]Empirical Evidence of Alterations
Atmospheric carbon dioxide concentrations have risen from approximately 280 parts per million (ppm) in pre-industrial times to over 420 ppm as of 2023, a ~50% increase primarily driven by fossil fuel combustion and land-use changes, as evidenced by continuous measurements at the Mauna Loa Observatory since 1958.[83] Isotopic analysis confirms this anthropogenic origin: the decline in the ¹³C/¹²C ratio (δ¹³C) from -6.5‰ pre-industrial to -8.5‰ currently, and the near absence of radiocarbon (¹⁴C) in recent CO₂, align with signatures of ancient fossil carbon depleted in these isotopes due to radioactive decay and fractionation during biosynthesis.[84] These shifts exceed natural variability observed in ice-core records spanning 800,000 years, where CO₂ never surpassed 300 ppm during glacial-interglacial cycles.[83] In the nitrogen cycle, human activities have approximately doubled global reactive nitrogen (Nr) creation rates since pre-industrial levels, from ~100 Tg N yr⁻¹ to ~190 Tg N yr⁻¹ by the early 2000s, mainly through fertilizer production via the Haber-Bosch process and fossil fuel combustion.[85] This has led to elevated nitrogen deposition, with measurements showing coastal eutrophication in over 400 systems worldwide, including algal blooms and hypoxia; for instance, riverine Nr exports to oceans have increased 20-50% in regions like the North Atlantic due to agricultural intensification.[86] Atmospheric nitrous oxide (N₂O) concentrations have risen ~20% since 1750 to 335 ppb by 2020, corroborated by ice-core data and flask measurements, contributing to stratospheric ozone depletion and radiative forcing.[87] Phosphorus perturbations are evident in accelerated fluvial transport from agricultural runoff, with global river phosphorus loads increasing 2-3 fold over the 20th century due to fertilizer application exceeding 20 million metric tons annually.[88] Empirical data link this to hypoxic "dead zones," such as the Gulf of Mexico's seasonal area exceeding 15,000 km² since the 1980s, where dissolved oxygen drops below 2 mg L⁻¹, driven by Mississippi River discharges of ~100,000 metric tons of phosphorus yearly from Midwest farmlands.[89] Lake sediment cores worldwide document phosphorus accumulation rates 2-10 times pre-industrial baselines, confirming eutrophication causality over natural weathering inputs.[90] Sulfur cycle alterations peaked mid-20th century with anthropogenic SO₂ emissions reaching ~100 Tg S yr⁻¹ globally by the 1970s from coal burning, causing acid deposition rates 10-50 times natural levels in eastern North America and Europe, as measured in precipitation pH dropping to <4.0 and sulfate concentrations in lakes rising correspondingly.[91] Regulatory interventions, such as the U.S. Acid Rain Program since 1995, have reduced U.S. power plant SO₂ emissions by over 90% to ~2 million tons annually by 2020, reflected in declining wet sulfate deposition from 20-30 kg ha⁻¹ yr⁻¹ in the 1980s to <5 kg ha⁻¹ yr⁻¹ recently, with ecosystem recovery in soil and water chemistry.[92] Atmospheric sulfur burdens have similarly declined, with global models and satellite observations (e.g., from TOMS) showing reduced aerosol optical depth over industrialized regions.[93]Controversies and Uncertainties
Debates on Cycle Imbalances
Debates on the magnitude and attribution of biogeochemical cycle imbalances often center on distinguishing transient perturbations from long-term systemic disruptions, with contention over the relative roles of natural variability—such as volcanic emissions, solar forcing, and oceanic oscillations—and anthropogenic forcings like fossil fuel combustion and fertilizer use.[94] While empirical data, including isotopic signatures of atmospheric CO2, confirm human contributions to net fluxes, critics argue that models underemphasize feedbacks like enhanced terrestrial greening or soil carbon stabilization, potentially overstating imbalance severity.[95] These discussions highlight gaps in causal attribution, where academic sources sometimes prioritize alarmist narratives influenced by institutional incentives, though peer-reviewed analyses stress the need for integrated natural-human system modeling.[10] In the carbon cycle, a persistent controversy surrounds the "missing sink," where approximately 25-30% of cumulative anthropogenic CO2 emissions since the Industrial Revolution remain unaccounted for in quantified atmospheric, oceanic, and terrestrial reservoirs as of 2020 assessments.[96] Hypotheses attribute this to underestimated Northern Hemisphere forest regrowth or deep soil sequestration, but debates persist on whether saturation of these sinks—potentially exacerbated by warming-induced respiration—signals weakening resilience or merely observational incompleteness.[97] Proponents of stronger natural variability contend that decadal oscillations, like the Pacific Decadal Oscillation, explain flux anomalies better than purely anthropogenic models, challenging projections of accelerating imbalances.[98] For the nitrogen cycle, debates focus on the planetary boundary framework, which posits that human fixation of reactive nitrogen—reaching 190 teragrams per year by 2010, exceeding natural rates—has transgressed safe limits, driving eutrophication and N2O emissions.[99] Critics, including analyses of the boundary's formulation, argue it conflates local impacts with global thresholds, ignoring agricultural productivity gains from nitrogen inputs that have averted famines, and question whether denitrification feedbacks naturally mitigate excesses without invoking crisis.[87] Empirical evidence from regional studies shows variable exceedances, fueling contention over policy prescriptions like reduced fertilizer use versus precision application to balance benefits and harms.[100] The phosphorus cycle elicits vigorous debate over "peak phosphorus," with projections ranging from depletion of economically viable reserves by the 2030s to assertions that undiscovered deposits and recycling technologies avert scarcity.[101] While mining has accelerated flux from geological reservoirs—extracting 20 million tons annually by 2020, disrupting sedimentary recycling—skeptics highlight overestimated demand curves and new Moroccan discoveries extending supplies beyond 2100, dismissing imminent crisis as unsubstantiated fear.[102] This controversy underscores tensions between environmental concerns over runoff eutrophication and food security imperatives, with causal realism favoring enhanced recovery from wastewater over extraction limits.[103]Quantification Gaps and Model Limitations
Quantification of biogeochemical fluxes remains incomplete, particularly for subsurface and oceanic processes where direct measurements are sparse. For instance, soil carbon turnover rates exhibit uncertainties exceeding 50% in many regions due to heterogeneous microbial activity and environmental controls, complicating global estimates of terrestrial sinks.[104] Similarly, denitrification fluxes in aquatic systems are poorly constrained, with estimates varying by factors of 2-10 owing to episodic events and scale mismatches between lab-scale data and field observations.[105] These gaps arise from limited observational networks, such as the under-sampling of deep ocean layers and permafrost regions, hindering precise budgeting of elements like nitrogen and phosphorus.[106] Biogeochemical models, including those embedded in Earth system models (ESMs), suffer from structural and parametric limitations that amplify uncertainties. Parameterizations of sub-grid processes, such as microbial decomposition kinetics or ocean mixing, often rely on empirical fits with error propagation leading to simulated carbon storage variances of 20-40% across ensemble runs.[107] ESMs underestimate residence times for soil organic matter by up to 30% compared to observation-derived maps, reflecting inadequate representation of stabilization mechanisms like organo-mineral interactions.[108] Validation against independent data reveals biases, for example in the ocean biological carbon pump where export flux uncertainties dominate below 900 meters depth due to unresolved particle sinking dynamics.[109] Stochastic approaches to parameter uncertainty improve realism but cannot fully capture nonlinear feedbacks, such as those from calcifying plankton omitted in many models, which influence carbon sequestration efficiency.[110][111] Addressing these limitations requires integrating high-resolution observations with process-based refinements; however, persistent discrepancies between models and empirical budgets, such as for global land carbon sinks showing ESM spreads of 1-3 PgC yr⁻¹, underscore the need for better constraint on feedbacks like drought-induced trace gas emissions.[112] In nitrogen cycles, model uncertainties in N₂O flux predictions stem from incomplete microbial gene representations, with urban fragmentation altering functional gene abundances yet poorly simulated.[113] Overall, while advances in ensemble methods quantify parametric errors, fundamental gaps in causal process understanding limit predictive fidelity under perturbed conditions.Recent Advances
Observational and Modeling Innovations (2020-2025)
Between 2020 and 2025, observational advancements in biogeochemical cycles emphasized enhanced satellite remote sensing and in-situ networks, particularly for carbon and methane fluxes. The TROPOMI instrument on the Sentinel-5 Precursor satellite provided high-resolution column-averaged dry-air mole fractions of methane (XCH4), enabling quantification of seasonal variability driven by wetland emissions and atmospheric transport, with analyses revealing environmental drivers like temperature and precipitation influencing northern hemispheric peaks.[114] Similarly, multi-satellite fusion techniques produced decadal, spatially complete global surface chlorophyll-a datasets at 4 km resolution, improving estimates of phytoplankton productivity and its role in oceanic carbon drawdown by reconciling discrepancies across sensors like MODIS and VIIRS.[115] NOAA's Ocean Carbon Observing Science Plan, released in early 2025, integrated these satellite data with Argo biogeochemical floats to refine ocean carbon inventory observations, targeting uncertainties in air-sea CO2 fluxes through expanded autonomous profiling.[116] Modeling innovations during this period incorporated machine learning hybrids and process-based refinements to address scale mismatches in Earth system simulations. The FLaMe-v1.0 framework, introduced in 2025, coupled physical lake models with methane biogeochemistry, simulating ebullition and diffusion at regional scales with intermediate complexity to predict emissions under varying hydrology, validated against eddy covariance data.[117] GEOCLIM7, revised in 2025, extended long-term Earth system modeling to multi-million-year timescales by integrating revised weathering and volcanic outgassing parameterizations, enhancing simulations of Phanerozoic carbon and oxygen cycles against geological proxies.[118] Genome-scale metabolic models of phytoplankton were embedded into Earth system models, linking molecular physiology to ecosystem-scale biogeochemical rates, as demonstrated in 2025 simulations projecting altered marine carbon export under nutrient stress.[50] Hybrid approaches, blending physics-based parameterizations with data-driven machine learning, emerged in atmospheric components of models like those in JAMESS, reducing biases in terrestrial nitrogen and carbon feedbacks by assimilating satellite-derived fluxes.[119] These developments collectively narrowed quantification gaps, with observational datasets constraining model initial conditions and parameterizations, though persistent challenges include resolving sub-grid processes in heterogeneous landscapes. Peer-reviewed syntheses underscore that such integrations yield more robust projections of cycle perturbations, prioritizing empirical validation over unverified assumptions.[120]Implications for Earth System Understanding
Biogeochemical cycles underpin the interconnected dynamics of Earth's atmosphere, oceans, land, and biosphere, enabling a holistic comprehension of planetary processes through Earth system models (ESMs). These models integrate cycles such as carbon, nitrogen, and phosphorus to simulate fluxes, reservoirs, and feedbacks, revealing how elemental transformations regulate climate stability and ecosystem resilience. For instance, the carbon cycle's interaction with physical climate processes, including ocean uptake and terrestrial sequestration, constrains projections of atmospheric CO2 levels, with empirical data from ice cores and satellite observations validating model representations of historical variability.[18][121] Feedback mechanisms within biogeochemical cycles amplify or dampen Earth system responses to perturbations, as evidenced by nitrogen deposition's limited enhancement of CO2 sinks despite elevated fluxes, highlighting constraints on terrestrial carbon uptake. Causal linkages, such as phosphorus limitation in ocean productivity influencing dimethyl sulfide emissions and aerosol formation, demonstrate how nutrient cycles modulate radiative forcing and cloud albedo, thereby affecting global temperature equilibria. Empirical measurements from oceanographic surveys and atmospheric monitoring underscore these interactions, informing assessments of system stability against anthropogenic forcings like eutrophication and acidification.[122][75] Understanding these cycles addresses quantification gaps in ESMs, where inaccuracies in representing microbial processes and rock weathering lead to uncertainties in long-term carbon budgets, as quantified by discrepancies between modeled and observed ocean carbon inventories since the Industrial Revolution. Advances in observational networks, including eddy covariance towers and Argo floats, have refined cycle parametrizations, enhancing predictive fidelity for scenarios like permafrost thaw releasing methane, which could accelerate warming via positive feedbacks. This framework reveals Earth as a self-regulating yet vulnerable system, where cycle imbalances signal potential tipping points, such as altered ocean circulation impacting global nutrient distribution.[123][10]References
- https://ntrs.[nasa](/page/NASA).gov/api/citations/19940026113/downloads/19940026113.pdf




![The implications of shifts in the global carbon cycle due to human activity are concerning scientists.[6]](http://upload.wikimedia.org/wikipedia/commons/thumb/a/af/Global_carbon_cycle.webp/500px-Global_carbon_cycle.webp.png)







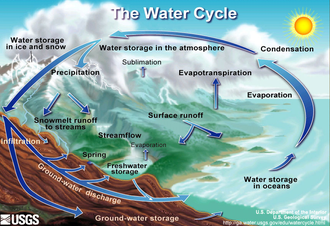

![Kerogen cycle[51][52]](http://upload.wikimedia.org/wikipedia/commons/thumb/2/21/Organic_carbon_cycle_including_the_flow_of_kerogen.png/500px-Organic_carbon_cycle_including_the_flow_of_kerogen.png)
