Recent from talks
Nothing was collected or created yet.
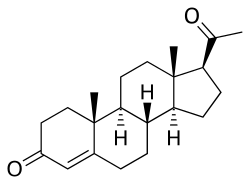
Progesterone
View on Wikipedia
 | |
 | |
| Names | |
|---|---|
| IUPAC name | |
| Systematic IUPAC name
(1S,3aS,3bS,9aR,9bS,11aS)-1-Acetyl-9a,11a-dimethyl-1,2,3,3a,3b,4,5,8,9,9a,9b,10,11,11a-tetradecahydro-7H-cyclopenta[a]phenanthren-7-one | |
| Other names
P4;[1] Pregnenedione
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.000.318 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C21H30O2 | |
| Molar mass | 314.469 g/mol |
| Density | 1.171 |
| Melting point | 126 |
| log P | 4.04[4] |
| Pharmacology | |
| G03DA04 (WHO) | |
| By mouth, topical/transdermal, vaginal, intramuscular injection, subcutaneous injection, subcutaneous implant | |
| Pharmacokinetics: | |
| OMP: <10%[5][6] | |
| • Albumin: 80% • CBG: 18% • SHBG: <1% • Free: 1–2%[7][8] | |
| Hepatic (CYP2C19, CYP3A4, CYP2C9, 5α-reductase, 3α-HSD, 17α-hydroxylase, 21-hydroxylase, 20α-HSD)[9][10] | |
| OMP: 16–18 hours[5][6][11] IM: 22–26 hours[6][12] SC: 13–18 hours[12] | |
| Renal | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Progesterone (/proʊˈdʒɛstəroʊn/ ⓘ; P4) is an endogenous steroid and progestogen sex hormone involved in the menstrual cycle, pregnancy, and embryogenesis of humans and other species.[1][13] It belongs to a group of steroid hormones called the progestogens[13] and is the major progestogen in the body. Progesterone has a variety of important functions in the body. It is also a crucial metabolic intermediate in the production of other endogenous steroids, including the sex hormones and the corticosteroids, and plays an important role in brain function as a neurosteroid.[14]
In addition to its role as a natural hormone, progesterone is also used as a medication, such as in combination with estrogen for contraception, to reduce the risk of uterine or cervical cancer, in hormone replacement therapy, and in feminizing hormone therapy.[15] It was first prescribed in 1934.[16]
Biological activity
[edit]Progesterone is the most important progestogen in the body. As a potent agonist of the nuclear progesterone receptor (nPR) (with an affinity of KD = 1 nM), the resulting effects on ribosomal transcription play a major role in regulation of female reproduction.[13][17] In addition, progesterone is an agonist of the more recently discovered membrane progesterone receptors (mPRs),[18] of which the expression has regulation effects in reproduction function (oocyte maturation, labor, and sperm motility) and cancer, although additional research is required to further define the roles.[19] It also functions as a ligand of the PGRMC1 (progesterone receptor membrane component 1) which impacts tumor progression, metabolic regulation, and viability control of nerve cells.[20][21][22] Moreover, progesterone is also known to be an antagonist of the sigma σ1 receptor,[23][24] a negative allosteric modulator of nicotinic acetylcholine receptors,[14] and a potent antagonist of the mineralocorticoid receptor (MR).[25] Progesterone prevents MR activation by binding to this receptor with an affinity exceeding even those of aldosterone, and glucocorticoids such as cortisol and corticosterone,[25] and it produces antimineralocorticoid effects, such as natriuresis, at physiological concentrations.[26] Progesterone also binds to, and behaves as a partial agonist of, the glucocorticoid receptor (GR), albeit with very low potency (EC50 >100-fold less relative to cortisol).[27][28]
Through its neurosteroid active metabolites, such as 5α-dihydroprogesterone and allopregnanolone, progesterone acts indirectly as a positive allosteric modulator of the GABAA receptor.[29]
Progesterone and some of its metabolites, such as 5β-dihydroprogesterone, are agonists of the pregnane X receptor (PXR),[30] albeit weakly so (EC50 >10 μM).[31] In accordance, progesterone induces several hepatic cytochrome P450 enzymes,[32] such as CYP3A4,[33][34] especially during pregnancy when concentrations are much higher than usual.[35] Perimenopausal women have been found to have greater CYP3A4 activity relative to men and postmenopausal women, and it has been inferred that this may be due to the higher progesterone levels present in perimenopausal women.[33]
Progesterone modulates the activity of CatSper (cation channels of sperm) voltage-gated Ca2+ channels. Since eggs release progesterone, sperm may use progesterone as a homing signal to swim toward eggs (chemotaxis). As a result, it has been suggested that substances that block the progesterone binding site on CatSper channels could potentially be used in male contraception.[36][37]
Biological function
[edit]
Hormonal interactions
[edit]Progesterone has a number of physiological effects that are amplified in the presence of estrogens. Estrogens through estrogen receptors (ERs) induce or upregulate the expression of the PR.[39] One example of this is in breast tissue, where estrogens allow progesterone to mediate lobuloalveolar development.[40][41][42]
Elevated levels of progesterone potently reduce the sodium-retaining activity of aldosterone, resulting in natriuresis and a reduction in extracellular fluid volume. Progesterone withdrawal, on the other hand, is associated with a temporary increase in sodium retention (reduced natriuresis, with an increase in extracellular fluid volume) due to the compensatory increase in aldosterone production, which combats the blockade of the mineralocorticoid receptor by the previously elevated level of progesterone.[43]
Early sexual differentiation
[edit]Placental progesterone can be converted into 5α-dihydrotestosterone (DHT), a potent androgen that is responsible for the development of male genitalia.[44] This can be done both by conversion into testosterone, which is then converted to DHT, and via the androgen backdoor pathway, which is particularly important for fetal development.[45] Progesterone is the precursor for both pathways and therefore plays a key role in sexual differentiation.[46][47]
Reproductive system
[edit]
Progesterone has key effects via non-genomic signalling on human sperm as they migrate through the female reproductive tract before fertilization occurs, though the receptor(s) as yet remain unidentified.[48] Detailed characterisation of the events occurring in sperm in response to progesterone has elucidated certain events including intracellular calcium transients and maintained changes,[49] slow calcium oscillations,[50] now thought to possibly regulate motility.[51] It is produced by the ovaries.[52] Progesterone has also been shown to demonstrate effects on octopus spermatozoa.[53]
Progesterone is sometimes called the "hormone of pregnancy",[54] and it has many roles relating to the development of the fetus:
- Progesterone converts the endometrium to its secretory stage to prepare the uterus for implantation. At the same time progesterone affects the vaginal epithelium and cervical mucus, making it thick and impenetrable to sperm. Progesterone is anti-mitogenic in endometrial epithelial cells, and as such, mitigates the tropic effects of estrogen.[55] If pregnancy does not occur, progesterone levels will decrease, leading to menstruation. Normal menstrual bleeding is progesterone-withdrawal bleeding. If ovulation does not occur, and the corpus luteum does not develop, levels of progesterone may be low, leading to anovulatory dysfunctional uterine bleeding.
- During implantation and gestation, progesterone appears to decrease the maternal immune response to allow for the acceptance of the pregnancy.[56]
- Progesterone decreases contractility of the uterine smooth muscle.[54] This effect contributes to prevention of preterm labor.[56] Studies have shown that in individuals who are pregnant with a single fetus, asymptomatic in the prenatal stage, and at a high risk of giving pre-term birth spontaneously, vaginal progesterone medication has been found to be effective in preventing spontaneous pre-term birth. Individuals who are at a high risk of giving pre-term birth spontaneously are those who have a short cervix of less than 25 mm or have previously given pre-term birth spontaneously. Although pre-term births are generally considered to be less than 37 weeks, these studies found that vaginal progesterone is associated with fewer pre-term births of less than 34 weeks.[57]
- A drop in progesterone levels is possibly one step that facilitates the onset of labor.[citation needed]
- In addition, progesterone inhibits lactation during pregnancy. The fall in progesterone levels following delivery is one of the triggers for milk production.[citation needed]
The fetus metabolizes placental progesterone in the production of adrenal steroids.[45]
Breasts
[edit]Lobuloalveolar development
[edit]Progesterone plays an important role in breast development. In conjunction with prolactin, it mediates lobuloalveolar maturation of the mammary glands during pregnancy to allow for milk production, and thus lactation and breastfeeding of offspring following parturition (childbirth).[58] Estrogen induces expression of the progesterone receptors (PR) in breast tissue, and hence progesterone is dependent on estrogen to mediate lobuloalveolar development.[40][41][42] It has been found that RANKL is a critical downstream mediator of progesterone-induced lobuloalveolar maturation.[59] RANKL knockout mice show an almost identical mammary phenotype to PR knockout mice, including normal mammary ductal development, but complete failure of the development of lobuloalveolar structures.[59]
Ductal development
[edit]Though to a far lesser extent than estrogen, which is the major mediator of mammary ductal development (via the ERα),[60][61] progesterone may also be involved in ductal development of the mammary glands to some extent.[62] PR knockout mice or mice treated with the PR antagonist mifepristone show delayed although otherwise normal mammary ductal development at puberty.[62] In addition, mice modified to have overexpression of PRA display ductal hyperplasia,[59] and progesterone induces ductal growth in the mouse mammary gland.[62] Progesterone mediates ductal development mainly via induction of the expression of amphiregulin, the same growth factor that estrogen primarily induces the expression of to mediate ductal development.[62] These animal findings suggest that, while not essential for full mammary ductal development, progesterone seems to play a potentiating or accelerating role in estrogen-mediated mammary ductal development.[62]
Breast cancer risk
[edit]Progesterone also appears to be involved in the pathophysiology of breast cancer, though its role, and whether it is a promoter or inhibitor of breast cancer risk, has not been fully elucidated.[63][64] Most progestins, or synthetic progestogens, like medroxyprogesterone acetate, have been found to increase the risk of breast cancer in postmenopausal people in combination with estrogen as a component of menopausal hormone therapy.[65][64] The combination of natural oral progesterone or the atypical progestin dydrogesterone with estrogen has been associated with less risk of breast cancer than progestins plus estrogen.[66][67][68] However, this may simply be an artifact of the low progesterone levels produced with oral progesterone.[63][69] More research is needed on the role of progesterone in breast cancer.[64]
Skin health
[edit]The estrogen receptor, as well as the progesterone receptor, have been detected in the skin, including in keratinocytes and fibroblasts.[70][71] At menopause and thereafter, decreased levels of female sex hormones result in atrophy, thinning, and increased wrinkling of the skin, and a reduction in skin elasticity, firmness, and strength.[70][71] These skin changes constitute an acceleration in skin aging and are the result of decreased collagen content, irregularities in the morphology of epidermal skin cells, decreased ground substance between skin fibers, and reduced capillaries and blood flow.[70][71] The skin also becomes more dry during menopause, as a result of reduced skin hydration and surface lipids (sebum production).[70] Along with chronological aging and photoaging, estrogen deficiency in menopause is one of the three main factors that predominantly influences skin aging.[70]
Hormone replacement therapy, consisting of systemic treatment with estrogen alone or in combination with a progestogen, has well-documented and considerable beneficial effects on the skin of postmenopausal people.[70][71] These benefits include increased skin collagen content, skin thickness and elasticity, and skin hydration and surface lipids.[70][71] Topical estrogen has been found to have similar beneficial effects on the skin.[70] In addition, a study has found that topical 2% progesterone cream significantly increases skin elasticity and firmness and observably decreases wrinkles in peri- and postmenopausal people.[71] Skin hydration and surface lipids, on the other hand, did not significantly change with topical progesterone.[71]
These findings suggest that progesterone, like estrogen, also has beneficial effects on the skin and may be independently protective against skin aging.[71]
Sexuality
[edit]Libido
[edit]Progesterone and its neurosteroid active metabolite, allopregnanolone, appear to be importantly involved in libido in females.[72]
Homosexuality
[edit]Dr. Diana Fleischman, of the University of Portsmouth, and colleagues looked for a relationship between progesterone and sexual attitudes in 92 women. Their research, published in the Archives of Sexual Behavior found that women who had higher levels of progesterone scored higher on a questionnaire measuring homoerotic motivation. They also found that men who had high levels of progesterone were more likely to have higher homoerotic motivation scores after affiliative priming compared to men with low levels of progesterone.[73][74][75][76]
Nervous system
[edit]Progesterone, like pregnenolone and dehydroepiandrosterone (DHEA), belongs to an important group of endogenous steroids called neurosteroids. It can be metabolized within all parts of the central nervous system.[77]
Neurosteroids are neuromodulators and are neuroprotective, neurogenic, and regulate neurotransmission and myelination.[78] The effects of progesterone as a neurosteroid are mediated predominantly through its interactions with non-nuclear PRs, namely the mPRs and PGRMC1, as well as certain other receptors, such as the σ1 and nACh receptors.[79]
Brain damage
[edit]Previous studies have shown that progesterone supports the normal development of neurons in the brain, and that the hormone has a protective effect on damaged brain tissue. It has been observed in animal models that females have reduced susceptibility to traumatic brain injury, and this protective effect has been hypothesized to be caused by increased circulating levels of estrogen and progesterone in females.[80]
Proposed mechanism
[edit]The mechanism of progesterone protective effects may be the reduction of inflammation that follows brain trauma and hemorrhage.[81][82]
Damage incurred by traumatic brain injury is believed to be caused in part by mass depolarization leading to excitotoxicity. One way in which progesterone helps to alleviate some of this excitotoxicity is by blocking the voltage-dependent calcium channels that trigger neurotransmitter release.[83] It does so by manipulating the signaling pathways of transcription factors involved in this release. Another method for reducing the excitotoxicity is by up-regulating the GABAA, a widespread inhibitory neurotransmitter receptor.[84]
Progesterone has also been shown to prevent apoptosis in neurons, a common consequence of brain injury. It does so by inhibiting enzymes involved in the apoptosis pathway specifically concerning the mitochondria, such as activated caspase-3 and cytochrome c.[85]
Not only does progesterone help prevent further damage, it has also been shown to aid in neuroregeneration.[86] One of the serious effects of traumatic brain injury includes edema. Animal studies show that progesterone treatment leads to a decrease in edema levels by increasing the concentration of macrophages and microglia sent to the injured tissue.[83][87] This was observed in the form of reduced leakage from the blood brain barrier in secondary recovery in progesterone treated rats. In addition, progesterone was observed to have antioxidant properties, reducing the concentration of oxygen free radicals faster than without.[84] There is also evidence that the addition of progesterone can also help remyelinate damaged axons due to trauma, restoring some lost neural signal conduction.[84] Another way progesterone aids in regeneration includes increasing the circulation of endothelial progenitor cells in the brain. This aids the growth of new vasculature around scar tissue, helping to repair the area of insult.[88]
Addiction
[edit]Progesterone enhances the function of serotonin receptors in the brain, so an excess or deficit of progesterone has the potential to result in significant neurochemical issues. This provides an explanation for why some people resort to substances that enhance serotonin activity such as nicotine, alcohol, and cannabis when their progesterone levels fall below optimal levels.[89]
- Sex differences in hormone levels may induce women to respond differently than men to nicotine. When women undergo cyclic changes or different hormonal transition phases (menopause, pregnancy, adolescence), there are changes in their progesterone levels.[90] Therefore, females have an increased biological vulnerability to nicotine's reinforcing effects compared to males, and progesterone may be used to counter this enhanced vulnerability. This information supports the idea that progesterone can affect behavior.[89]
- Similar to nicotine, cocaine also increases the release of dopamine in the brain. The neurotransmitter is involved in the reward center and is one of the main neurotransmitters involved with substance abuse and reliance. In a study of cocaine users, it was reported that progesterone reduced craving and the feeling of being stimulated by cocaine. Thus, progesterone was suggested as an agent that decreases cocaine craving by reducing the dopaminergic properties of the drug.[91]
Societal
[edit]In a 2012 University of Amsterdam study of 120 women, the women's luteal phase (higher levels of progesterone, and increasing levels of estrogen) was correlated with a lower level of competitive behavior in gambling and math contest scenarios, while their premenstrual phase (sharply-decreasing levels of progesterone, and decreasing levels of estrogen) was correlated with a higher level of competitive behavior.[92]
Other effects
[edit]- Progesterone also has a role in skin elasticity and bone strength, in respiration, in nerve tissue and in female sexuality, and the presence of progesterone receptors in certain muscle and fat tissue may hint at a role in sexually dimorphic proportions of those.[93]
- During pregnancy, progesterone is said to decrease uterine irritability.[94]
- During pregnancy, progesterone helps to suppress immune responses of the mother to fetal antigens, thus preventing rejection of the fetus.[94]
- Progesterone raises epidermal growth factor-1 (EGF-1) levels, a factor often used to induce proliferation of stem cells, and used to sustain stem cell cultures.[95]
- Progesterone increases core temperature (thermogenic function) during ovulation.[96][97]
- Progesterone reduces spasm and relaxes smooth muscle. Bronchi are widened and mucus regulated. (PRs are widely present in submucosal tissue.)[98]
- Progesterone acts as an anti-inflammatory agent and regulates the immune response.[99]
- Progesterone reduces gallbladder activity.[100]
- Progesterone normalizes blood clotting and vascular tone, zinc and copper levels, cell oxygen levels, and use of fat stores for energy.[citation needed]
- Progesterone may affect gum health, increasing risk of gingivitis (gum inflammation).[101]
- Progesterone appears to prevent endometrial cancer (involving the uterine lining) by regulating the effects of estrogen.
- Progesterone plays an important role in the signaling of insulin release and pancreatic function, and it may affect the susceptibility to diabetes or gestational diabetes.[102][103]
- Progesterone levels in the blood were found to be lower in those who had higher weight and higher BMI among those who became pregnant through in vitro fertilization.[104]
- Current data shows that micronized progesterone, which is chemically identical to the progesterone produced in the human body, in combination with estrogen in menopausal hormone therapy does not seem to have significant effects on venous thromboembolism (blood clots in veins) and ischemic stroke (lack of blood flow to the brain due to blockage of a blood vessel that supplies the brain). However, more studies need to be conducted to see whether or not micronized progesterone alone or in combined menopausal hormone therapy changes the risk of myocardial infarctions (heart attacks).[105]
- There have not been any studies done yet on the effects of micronized progesterone on hair loss due to menopause.[106]
- Despite suggestions for using hormone therapy to prevent loss of muscle mass in post-menopausal individuals (age 50+), menopausal hormone therapy involving either estrogen alone, or estrogen and progesterone combined, has not been found to preserve muscle mass.[107] Menopausal hormone therapy also does not result in body weight reduction, BMI reduction, or change in glucose metabolism.[108]
Biochemistry
[edit]Biosynthesis
[edit]
In mammals, progesterone, like all other steroid hormones, is synthesized from pregnenolone, which itself is derived from cholesterol.[citation needed]
Cholesterol undergoes double oxidation to produce 22R-hydroxycholesterol and then 20α,22R-dihydroxycholesterol. This vicinal diol is then further oxidized with loss of the side chain starting at position C22 to produce pregnenolone. This reaction is catalyzed by cytochrome P450scc.[citation needed]
The conversion of pregnenolone to progesterone takes place in two steps. First, the 3β-hydroxyl group is oxidized to a keto group and second, the double bond is moved to C4, from C5 through a keto/enol tautomerization reaction.[110] This reaction is catalyzed by 3β-hydroxysteroid dehydrogenase/δ5-4-isomerase.[citation needed]
Progesterone in turn is the precursor of the mineralocorticoid aldosterone, and after conversion to 17α-hydroxyprogesterone, of cortisol and androstenedione. Androstenedione can be converted to testosterone, estrone, and estradiol, highlighting the critical role of progesterone in testosterone synthesis.[citation needed]
Pregnenolone and progesterone can also be synthesized by yeast.[111]
Approximately 30 mg of progesterone is secreted from the ovaries per day in reproductive-age women, while the adrenal glands produce about 1 mg of progesterone per day.[112]
| Sex | Sex hormone | Reproductive phase |
Blood production rate |
Gonadal secretion rate |
Metabolic clearance rate |
Reference range (serum levels) | |
|---|---|---|---|---|---|---|---|
| SI units | Non-SI units | ||||||
| Men | Androstenedione | –
|
2.8 mg/day | 1.6 mg/day | 2200 L/day | 2.8–7.3 nmol/L | 80–210 ng/dL |
| Testosterone | –
|
6.5 mg/day | 6.2 mg/day | 950 L/day | 6.9–34.7 nmol/L | 200–1000 ng/dL | |
| Estrone | –
|
150 μg/day | 110 μg/day | 2050 L/day | 37–250 pmol/L | 10–70 pg/mL | |
| Estradiol | –
|
60 μg/day | 50 μg/day | 1600 L/day | <37–210 pmol/L | 10–57 pg/mL | |
| Estrone sulfate | –
|
80 μg/day | Insignificant | 167 L/day | 600–2500 pmol/L | 200–900 pg/mL | |
| Women | Androstenedione | –
|
3.2 mg/day | 2.8 mg/day | 2000 L/day | 3.1–12.2 nmol/L | 89–350 ng/dL |
| Testosterone | –
|
190 μg/day | 60 μg/day | 500 L/day | 0.7–2.8 nmol/L | 20–81 ng/dL | |
| Estrone | Follicular phase | 110 μg/day | 80 μg/day | 2200 L/day | 110–400 pmol/L | 30–110 pg/mL | |
| Luteal phase | 260 μg/day | 150 μg/day | 2200 L/day | 310–660 pmol/L | 80–180 pg/mL | ||
| Postmenopause | 40 μg/day | Insignificant | 1610 L/day | 22–230 pmol/L | 6–60 pg/mL | ||
| Estradiol | Follicular phase | 90 μg/day | 80 μg/day | 1200 L/day | <37–360 pmol/L | 10–98 pg/mL | |
| Luteal phase | 250 μg/day | 240 μg/day | 1200 L/day | 699–1250 pmol/L | 190–341 pg/mL | ||
| Postmenopause | 6 μg/day | Insignificant | 910 L/day | <37–140 pmol/L | 10–38 pg/mL | ||
| Estrone sulfate | Follicular phase | 100 μg/day | Insignificant | 146 L/day | 700–3600 pmol/L | 250–1300 pg/mL | |
| Luteal phase | 180 μg/day | Insignificant | 146 L/day | 1100–7300 pmol/L | 400–2600 pg/mL | ||
| Progesterone | Follicular phase | 2 mg/day | 1.7 mg/day | 2100 L/day | 0.3–3 nmol/L | 0.1–0.9 ng/mL | |
| Luteal phase | 25 mg/day | 24 mg/day | 2100 L/day | 19–45 nmol/L | 6–14 ng/mL | ||
Notes and sources
Notes: "The concentration of a steroid in the circulation is determined by the rate at which it is secreted from glands, the rate of metabolism of precursor or prehormones into the steroid, and the rate at which it is extracted by tissues and metabolized. The secretion rate of a steroid refers to the total secretion of the compound from a gland per unit time. Secretion rates have been assessed by sampling the venous effluent from a gland over time and subtracting out the arterial and peripheral venous hormone concentration. The metabolic clearance rate of a steroid is defined as the volume of blood that has been completely cleared of the hormone per unit time. The production rate of a steroid hormone refers to entry into the blood of the compound from all possible sources, including secretion from glands and conversion of prohormones into the steroid of interest. At steady state, the amount of hormone entering the blood from all sources will be equal to the rate at which it is being cleared (metabolic clearance rate) multiplied by blood concentration (production rate = metabolic clearance rate × concentration). If there is little contribution of prohormone metabolism to the circulating pool of steroid, then the production rate will approximate the secretion rate." Sources: See template. | |||||||
Distribution
[edit]Progesterone binds extensively to plasma proteins, including albumin (50–54%) and transcortin (43–48%).[113] It has similar affinity for albumin relative to the PR.[17]
Metabolism
[edit]The metabolism of progesterone is rapid and extensive, and it occurs mainly in the liver,[114][115][116] though enzymes that metabolize progesterone are also expressed widely in the brain, skin, and various other extrahepatic tissues.[77][117] Progesterone has an elimination half-life of only approximately five minutes in circulation.[114] The metabolism of progesterone is complex, and it may form as many as 35 different unconjugated metabolites when it is ingested orally.[116][118] Progesterone is highly susceptible to enzymatic reduction via reductases and hydroxysteroid dehydrogenases because of its double bond (between the C4 and C5 positions) and its two ketones (at the C3 and C20 positions).[116]
The major metabolic pathway of progesterone is reduction by 5α-reductase[77] and 5β-reductase, into the dihydrogenated 5α-dihydroprogesterone and 5β-dihydroprogesterone, respectively.[115][116][119][120] This is followed by the further reduction of these metabolites via 3α-hydroxysteroid dehydrogenase and 3β-hydroxysteroid dehydrogenase into the tetrahydrogenated allopregnanolone, pregnanolone, isopregnanolone, and epipregnanolone.[121][115][116][119] Subsequently, 20α-hydroxysteroid dehydrogenase and 20β-hydroxysteroid dehydrogenase reduce these metabolites to form the corresponding hexahydrogenated pregnanediols (eight different isomers in total),[115][120] which are then conjugated via glucuronidation and/or sulfation, released from the liver into circulation, and excreted by the kidneys into the urine.[114][116] The major metabolite of progesterone in the urine is the 3α,5β,20α isomer of pregnanediol glucuronide, which has been found to constitute 15–30% of an injection of progesterone.[17][122] Other metabolites of progesterone formed by the enzymes in this pathway include 3α-dihydroprogesterone, 3β-dihydroprogesterone, 20α-dihydroprogesterone, and 20β-dihydroprogesterone, as well as various combination products of the enzymes aside from those already mentioned.[17][116][122][123] Progesterone can also first be hydroxylated (see below) and then reduced.[116] Endogenous progesterone is metabolized approximately 50% into 5α-dihydroprogesterone in the corpus luteum, 35% into 3β-dihydroprogesterone in the liver, and 10% into 20α-dihydroprogesterone.[124]
Relatively small portions of progesterone are hydroxylated via 17α-hydroxylase (CYP17A1) and 21-hydroxylase (CYP21A2), into 17α-hydroxyprogesterone and 11-deoxycorticosterone (21-hydroxyprogesterone), respectively,[118] and pregnanetriols are formed secondarily to 17α-hydroxylation.[125][126] Even smaller amounts of progesterone may also be hydroxylated via 11β-hydroxylase (CYP11B1) and, to a lesser extent, via aldosterone synthase (CYP11B2) into 11β-hydroxyprogesterone.[127][128][44] In addition, progesterone can be hydroxylated in the liver by other cytochrome P450 enzymes that are not steroid-specific.[129] Catalyzed mainly by CYP3A4, 6β-Hydroxylation is the major transformation and is responsible for approximately 70% of cytochrome P450-mediated progesterone metabolism.[129] Other routes include 6α-, 16α-, and 16β-hydroxylation.[116] However, treatment of women with ketoconazole (a strong CYP3A4 inhibitor) had minimal effects on progesterone levels, producing only a slight and non-significant increase, suggesting that cytochrome P450 enzymes play only a small role in progesterone metabolism.[130]
Metabolism of progesterone in humans[131]
|
Levels
[edit]
Relatively low during the preovulatory phase of the menstrual cycle, progesterone levels rise after ovulation and are elevated during the luteal phase, as shown in the diagram. Progesterone levels tend to be less than 2 ng/mL prior to ovulation and greater than 5 ng/mL after ovulation. If pregnancy occurs, human chorionic gonadotropin is released, maintaining the corpus luteum and allowing it to maintain levels of progesterone. Between seven and nine weeks gestation, the placenta begins to produce progesterone in place of the corpus luteum in a process called the luteal-placental shift.[133]
After the luteal-placental shift, progesterone levels start to rise further and may reach 100 to 200 ng/mL at term. Whether a decrease in progesterone levels is critical for the initiation of labor has been argued and may be species-specific. After delivery of the placenta and during lactation, progesterone levels are very low.[citation needed]
Progesterone levels are low in children and postmenopausal people.[134] Adult males have levels similar to those in women during the follicular phase of the menstrual cycle.
| Group | P4 production | P4 levels | ||
|---|---|---|---|---|
| Prepubertal children | ND | 0.06–0.5 ng/mL | ||
| Pubertal girls Tanner stage I (childhood) Tanner stage II (ages 8–12) Tanner stage III (ages 10–13) Tanner stage IV (ages 11–14) Tanner stage V (ages 12–15) Follicular phase (days 1–14) Luteal phase (days 15–28) |
ND ND ND ND ND ND |
0.22 (<0.10–0.32) ng/mL 0.30 (0.10–0.51) ng/mL 0.36 (0.10–0.75) ng/mL 1.75 (<0.10–25.0) ng/mL 0.35 (0.13–0.75) ng/mL 2.0–25.0 ng/mL | ||
| Premenopausal women Follicular phase (days 1–14) Luteal phase (days 15–28) Oral contraceptive (anovulatory) |
0.75–5.4 mg/day 15–50 mg/day ND |
0.02–1.2 ng/mL 4–30 ng/mL 0.1–0.3 ng/mL | ||
| Postmenopausal women Oophorectomized women Oophorectomized and adrenalectomized women |
ND 1.2 mg/day <0.3 mg/day |
0.03–0.3 ng/mL 0.39 ng/mL ND | ||
| Pregnant women First trimester (weeks 1–12) Second trimester (weeks 13–26) Third trimester (weeks 27–40) Postpartum (at 24 hours) |
55 mg/day 92–100 mg/day 190–563 mg/day ND |
9–75 ng/mL 17–146 ng/mL 55–255 ng/mL 19 ng/mL | ||
| Men | 0.75–3 mg/day | 0.1–0.3 ng/mL | ||
| Notes: Mean levels are given as a single value and ranges are given after in parentheses. Sources: [131][135][136][137][138][139][140][141][142] | ||||
Ranges
[edit]Blood test results should always be interpreted using the reference ranges provided by the laboratory that performed the results. Example reference ranges are listed below.
| Person type | Reference range for blood test | ||
|---|---|---|---|
| Lower limit | Upper limit | Unit | |
| Female - menstrual cycle | (see diagram below) | ||
| Female - postmenopausal | <0.2[143] | 1[143] | ng/mL |
| <0.6[144] | 3[144] | nmol/L | |
| Female on oral contraceptives | 0.34[143] | 0.92[143] | ng/mL |
| 1.1[144] | 2.9[144] | nmol/L | |
| Males ≥16 years | 0.27[143] | 0.9[143] | ng/mL |
| 0.86[144] | 2.9[144] | nmol/L | |
| Female or male 1–9 years | 0.1[143] | 4.1[143] or 4.5[143] | ng/mL |
| 0.3[144] | 13[144] | nmol/L | |

• The ranges denoted By biological stage may be used in closely monitored menstrual cycles in regard to other markers of its biological progression, with the time scale being compressed or stretched to how much faster or slower, respectively, the cycle progresses compared to an average cycle.
• The ranges denoted Inter-cycle variability are more appropriate to use in non-monitored cycles with only the beginning of menstruation known, but where the woman accurately knows her average cycle lengths and time of ovulation, and that they are somewhat averagely regular, with the time scale being compressed or stretched to how much a woman's average cycle length is shorter or longer, respectively, than the average of the population.
• The ranges denoted Inter-woman variability are more appropriate to use when the average cycle lengths and time of ovulation are unknown, but only the beginning of menstruation is given.
Sources
[edit]Animal
[edit]Progesterone is produced in high amounts in the ovaries (by the corpus luteum) from the onset of puberty to menopause. It is produced in smaller amounts by the adrenal glands after the onset of adrenarche in both males and females. To a lesser extent, progesterone is produced in nervous tissue, especially in the brain, and also in adipose tissue (fat).[citation needed]
During human pregnancy, progesterone is produced in increasingly high amounts by the ovaries and placenta. At first, the source is the corpus luteum that has been "rescued" by the presence of human chorionic gonadotropin (hCG) from the conceptus. However, after the eighth week, production of progesterone shifts to the placenta which utilizes maternal cholesterol as the initial substrate, and most of the produced progesterone enters the maternal circulation, but some is picked up by the fetal circulation and used as substrate for fetal corticosteroids. At term, the placenta produces about 250 mg progesterone per day.[citation needed]
An additional animal source of progesterone is milk products. After consumption of milk products the level of bioavailable progesterone goes up.[146]
Plants
[edit]Progesterone has been positively identified in the plant Juglans regia, a species of walnut.[147] In addition, progesterone-like steroids are found in the plant Dioscorea mexicana, part of the yam family native to Mexico.[148] Dioscorea mexicana contains a steroid called diosgenin which is taken from the plant and converted into progesterone.[149] Diosgenin and progesterone are also found in other Dioscorea species, as well as in other plants that are not closely related, such as fenugreek.
Another plant that contains substances readily convertible to progesterone is Dioscorea pseudojaponica, native to Taiwan. Research has shown that the Taiwanese yam contains saponins—steroids that can be converted to diosgenin and thence to progesterone.[150]
Many other Dioscorea species of the yam family contain steroidal substances from which progesterone can be produced. Among the more notable of these are Dioscorea villosa and Dioscorea polygonoides. One study showed that the Dioscorea villosa contains 3.5% diosgenin.[151] Dioscorea polygonoides has been found to contain 2.64% diosgenin, as shown by gas chromatography-mass spectrometry.[152] Many of the Dioscorea species that originate from the yam family grow in countries with tropical and subtropical climates.[153]
Medical use
[edit]Progesterone is used as a medication. It is used in combination with estrogens mainly in hormone therapy for menopausal symptoms and low sex hormone levels.[118][154] It may also be used alone to treat menopausal symptoms. Studies have shown that transdermal progesterone (skin patch) and oral micronized progesterone are effective treatments for certain symptoms of menopause such as hot flashes and night sweats, otherwise referred to as vasomotor symptoms or VMS.[155]
It is also used to support pregnancy and fertility and to treat gynecological disorders.[156][157][158][159] Progesterone has been shown to prevent miscarriage in those with vaginal bleeding early in their current pregnancy and having a previous history of miscarriage.[160] Progesterone can be taken by mouth, through the vagina, and by injection into muscle or fat, among other routes.[118]
Chemistry
[edit]
Progesterone is a naturally occurring pregnane steroid and is also known as pregn-4-ene-3,20-dione.[161][162] It has a double bond (4-ene) between the C4 and C5 positions, and two ketone groups (3,20-dione), one at the C3 position and the other at the C20 position.[161][162]
Synthesis
[edit]Progesterone is commercially produced by semisynthesis. Two main routes are used: one from yam diosgenin first pioneered by Marker in 1940, and one based on soy phytosterols scaled up in the 1970s. Additional (not necessarily economical) semisyntheses of progesterone have also been reported starting from a variety of steroids. For the example, cortisone can be simultaneously deoxygenated at the C-17 and C-21 position by treatment with iodotrimethylsilane in chloroform to produce 11-keto-progesterone (ketogestin), which in turn can be reduced at position-11 to yield progesterone.[163]
Marker semisynthesis
[edit]An economical semisynthesis of progesterone from the plant steroid diosgenin isolated from yams was developed by Russell Marker in 1940 for the Parke-Davis pharmaceutical company.[164] This synthesis is known as the Marker degradation.

The 16-DPA intermediate is important to the synthesis of many other medically important steroids. A very similar approach can produce 16-DPA from solanine.[165]
Soy semisynthesis
[edit]Progesterone can also be made from the stigmasterol found in soybean oil also. c.f. Percy Julian.

Total synthesis
[edit]
A total synthesis of progesterone was reported in 1971 by William S. Johnson.[171] The synthesis begins with reacting the phosphonium salt 7 with phenyl lithium to produce the phosphonium ylide 8. The ylide 8 is reacted with an aldehyde to produce the alkene 9. The ketal protecting groups of 9 are hydrolyzed to produce the diketone 10, which in turn is cyclized to form the cyclopentenone 11. The ketone of 11 is reacted with methyl lithium to yield the tertiary alcohol 12, which in turn is treated with acid to produce the tertiary cation 13. The key step of the synthesis is the π-cation cyclization of 13 in which the B-, C-, and D-rings of the steroid are simultaneously formed to produce 14. This step resembles the cationic cyclization reaction used in the biosynthesis of steroids and hence is referred to as biomimetic. In the next step the enol orthoester is hydrolyzed to produce the ketone 15. The cyclopentene A-ring is then opened by oxidizing with ozone to produce 16. Finally, the diketone 17 undergoes an intramolecular aldol condensation by treating with aqueous potassium hydroxide to produce progesterone.[171]
History
[edit]George W. Corner and Willard M. Allen discovered the hormonal action of progesterone in 1929.[17][172][173][174] By 1931–1932, nearly pure crystalline material of high progestational activity had been isolated from the corpus luteum of animals; by 1934, pure crystalline progesterone had been refined and obtained, and the chemical structure of progesterone was determined.[17][173] This was achieved by Adolf Butenandt at the Chemisches Institut of Gdańsk Technical University in Danzig, who extracted this new compound from several thousand liters of urine.[175]
Chemical synthesis of progesterone from stigmasterol and pregnanediol was accomplished later that year.[173][176] Up to this point, progesterone, known generically as corpus luteum hormone, had been being referred to by several groups by different names, including corporin, lutein, luteosterone, and progestin.[17][177] In 1935, at the time of the Second International Conference on the Standardization of Sex Hormones in London, England, a compromise was reached between the groups, and the name 'progesterone' (progestational steroidal ketone) was created.[17][178]
Veterinary use
[edit]The use of progesterone tests in dog breeding to pinpoint ovulation is becoming more widely used. There are several tests available, the most reliable being a blood test with the blood sample drawn by a veterinarian and sent to a lab for processing. Results can usually be obtained within 24 to 72 hours. The rationale for using progesterone tests is that increased numbers begin in close proximity to preovulatory surge in gonadotrophins and continue through ovulation and estrus. When progesterone levels reach certain levels they can signal the stage of estrus the female is. Prediction of birth date of the pending litter can be very accurate if ovulation date is known. Puppies deliver within a day or two of nine weeks gestation in most cases. It is not possible to determine pregnancy using progesterone tests once a breeding has taken place, however. This is due to the fact that, in dogs, progesterone levels remain elevated throughout the estrus period.[179]
Pricing
[edit]Pricing for progesterone can vary depending location, insurance coverage, discount coupons, quantity, shortages, manufacturers, brand or generic versions, different pharmacies, and so on. As of 2023, 30 capsules of 100 mg of the generic version, Progesterone, from CVS Pharmacy is around $40 without any discounts or insurance applied. The brand version, Prometrium, is around $450 for 30 capsules without any discounts or insurance applied.[180] In comparison, Walgreens offers 30 capsules of 100 mg in the generic version for $51 without insurance or coupons applied. The brand name costs around $431 for 30 capsules of 100 mg.[181]
References
[edit] This article incorporates text available under the CC BY-SA 3.0 license.
This article incorporates text available under the CC BY-SA 3.0 license.
- ^ a b Jameson JL, De Groot LJ (25 February 2015). Endocrinology: Adult and Pediatric E-Book. Elsevier Health Sciences. p. 2179. ISBN 978-0-323-32195-2. Archived from the original on 14 January 2023. Retrieved 29 October 2017.
- ^ Adler N, Pfaff D, Goy RW (6 December 2012). Handbook of Behavioral Neurobiology Volume 7 Reproduction (1st ed.). New York: Plenum Press. p. 189. ISBN 978-1-4684-4834-4. Archived from the original on 14 January 2023. Retrieved 4 July 2015.
- ^ "progesterone (CHEBI:17026)". ChEBI. European Molecular Biology Laboratory-EBI. Archived from the original on 20 March 2016. Retrieved 4 July 2015.
- ^ "Progesterone_msds". Archived from the original on 12 February 2021. Retrieved 19 April 2018.
- ^ a b Stanczyk FZ (September 2002). "Pharmacokinetics and potency of progestins used for hormone replacement therapy and contraception". Reviews in Endocrine & Metabolic Disorders. 3 (3): 211–224. doi:10.1023/A:1020072325818. PMID 12215716. S2CID 27018468.
- ^ a b c Simon JA, Robinson DE, Andrews MC, Hildebrand JR, Rocci ML, Blake RE, Hodgen GD (July 1993). "The absorption of oral micronized progesterone: the effect of food, dose proportionality, and comparison with intramuscular progesterone". Fertility and Sterility. 60 (1): 26–33. doi:10.1016/S0015-0282(16)56031-2. PMID 8513955.
- ^ Fritz MA, Speroff L (28 March 2012). Clinical Gynecologic Endocrinology and Infertility. Lippincott Williams & Wilkins. pp. 44–. ISBN 978-1-4511-4847-3.
- ^ Marshall WJ, Bangert SK (2008). Clinical Chemistry. Elsevier Health Sciences. pp. 192–. ISBN 978-0-7234-3455-9. Archived from the original on 14 January 2023. Retrieved 5 October 2016.
- ^ Yamazaki H, Shimada T (October 1997). "Progesterone and testosterone hydroxylation by cytochromes P450 2C19, 2C9, and 3A4 in human liver microsomes". Archives of Biochemistry and Biophysics. 346 (1): 161–169. doi:10.1006/abbi.1997.0302. PMID 9328296.
- ^ McKay GA, Walters MR (6 February 2013). Lecture Notes: Clinical Pharmacology and Therapeutics. John Wiley & Sons. p. 33. ISBN 978-1-118-34489-7. Archived from the original on 14 January 2023. Retrieved 27 June 2015.
- ^ Zutshi V, Rathore AM, Sharma K (1 January 2005). Hormones in Obstetrics and Gynaecology. Jaypee Brothers Publishers. p. 74. ISBN 978-81-8061-427-9.[permanent dead link]
- ^ a b Cometti B (November 2015). "Pharmaceutical and clinical development of a novel progesterone formulation". Acta Obstetricia et Gynecologica Scandinavica. 94 (Suppl 161): 28–37. doi:10.1111/aogs.12765. PMID 26342177. S2CID 31974637.
- ^ a b c King TL, Brucker MC (25 October 2010). Pharmacology for Women's Health. Jones & Bartlett Publishers. pp. 372–373. ISBN 978-1-4496-5800-7.
- ^ a b Baulieu E, Schumacher M (2000). "Progesterone as a neuroactive neurosteroid, with special reference to the effect of progesterone on myelination". Steroids. 65 (10–11): 605–612. doi:10.1016/s0039-128x(00)00173-2. PMID 11108866. S2CID 14952168.
- ^ Prior JC (April 2019). "Progesterone Is Important for Transgender Women's Therapy-Applying Evidence for the Benefits of Progesterone in Ciswomen". The Journal of Clinical Endocrinology and Metabolism. 104 (4): 1181–1186. doi:10.1210/jc.2018-01777. PMID 30608551. S2CID 58620122.
Evidence has accrued that normal progesterone (and ovulation), as well as physiological estradiol levels, is necessary during ciswomen's premenopausal menstrual cycles for current fertility and long-term health; transgender women may require progesterone therapy and similar potential physiological benefits
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 47X. ISBN 978-3-527-60749-5.
- ^ a b c d e f g h Josimovich JB (11 November 2013). Gynecologic Endocrinology. Springer Science & Business Media. pp. 9, 25–29. ISBN 978-1-4613-2157-6. Archived from the original on 14 January 2023. Retrieved 1 February 2016.
- ^ Thomas P, Pang Y (2012). "Membrane progesterone receptors: evidence for neuroprotective, neurosteroid signaling and neuroendocrine functions in neuronal cells". Neuroendocrinology. 96 (2): 162–171. doi:10.1159/000339822. PMC 3489003. PMID 22687885.
- ^ Valadez-Cosmes P, Vázquez-Martínez ER, Cerbón M, Camacho-Arroyo I (October 2016). "Membrane progesterone receptors in reproduction and cancer". Molecular and Cellular Endocrinology. 434: 166–175. doi:10.1016/j.mce.2016.06.027. PMID 27368976. S2CID 3826650.
- ^ Meyer C, Schmid R, Schmieding K, Falkenstein E, Wehling M (February 1998). "Characterization of high affinity progesterone-binding membrane proteins by anti-peptide antiserum". Steroids. 63 (2): 111–116. doi:10.1016/s0039-128x(97)00143-8. PMID 9516722. S2CID 40096058.
- ^ Kabe Y, Handa H, Suematsu M (July 2018). "Function and structural regulation of the carbon monoxide (CO)-responsive membrane protein PGRMC1". Journal of Clinical Biochemistry and Nutrition. 63 (1): 12–17. doi:10.3164/jcbn.17-132. PMC 6064819. PMID 30087538.
- ^ Ryu CS, Klein K, Zanger UM (27 March 2017). "Membrane Associated Progesterone Receptors: Promiscuous Proteins with Pleiotropic Functions - Focus on Interactions with Cytochromes P450". Frontiers in Pharmacology. 8: 159. doi:10.3389/fphar.2017.00159. PMC 5366339. PMID 28396637.
- ^ Maurice T, Urani A, Phan VL, Romieu P (November 2001). "The interaction between neuroactive steroids and the sigma1 receptor function: behavioral consequences and therapeutic opportunities". Brain Research. Brain Research Reviews. 37 (1–3): 116–132. doi:10.1016/s0165-0173(01)00112-6. PMID 11744080. S2CID 44931783.
- ^ Johannessen M, Fontanilla D, Mavlyutov T, Ruoho AE, Jackson MB (February 2011). "Antagonist action of progesterone at σ-receptors in the modulation of voltage-gated sodium channels". American Journal of Physiology. Cell Physiology. 300 (2): C328 – C337. doi:10.1152/ajpcell.00383.2010. PMC 3043630. PMID 21084640.
- ^ a b Rupprecht R, Reul JM, van Steensel B, Spengler D, Söder M, Berning B, et al. (October 1993). "Pharmacological and functional characterization of human mineralocorticoid and glucocorticoid receptor ligands". European Journal of Pharmacology. 247 (2): 145–154. doi:10.1016/0922-4106(93)90072-H. PMID 8282004.
- ^ Elger W, Beier S, Pollow K, Garfield R, Shi SQ, Hillisch A (November 2003). "Conception and pharmacodynamic profile of drospirenone". Steroids. 68 (10–13): 891–905. doi:10.1016/j.steroids.2003.08.008. PMID 14667981. S2CID 41756726.
- ^ Attardi BJ, Zeleznik A, Simhan H, Chiao JP, Mattison DR, Caritis SN (December 2007). "Comparison of progesterone and glucocorticoid receptor binding and stimulation of gene expression by progesterone, 17-alpha hydroxyprogesterone caproate, and related progestins". American Journal of Obstetrics and Gynecology. 197 (6): 599.e1–599.e7. doi:10.1016/j.ajog.2007.05.024. PMC 2278032. PMID 18060946.
- ^ Lei K, Chen L, Georgiou EX, Sooranna SR, Khanjani S, Brosens JJ, et al. (2012). "Progesterone acts via the nuclear glucocorticoid receptor to suppress IL-1β-induced COX-2 expression in human term myometrial cells". PLOS ONE. 7 (11) e50167. Bibcode:2012PLoSO...750167L. doi:10.1371/journal.pone.0050167. PMC 3509141. PMID 23209664.
- ^ Paul SM, Purdy RH (March 1992). "Neuroactive steroids". FASEB Journal. 6 (6): 2311–2322. doi:10.1096/fasebj.6.6.1347506. PMID 1347506. S2CID 221753076.
- ^ Kliewer SA, Goodwin B, Willson TM (October 2002). "The nuclear pregnane X receptor: a key regulator of xenobiotic metabolism". Endocrine Reviews. 23 (5): 687–702. doi:10.1210/er.2001-0038. PMID 12372848.
- ^ Lehmann JM, McKee DD, Watson MA, Willson TM, Moore JT, Kliewer SA (September 1998). "The human orphan nuclear receptor PXR is activated by compounds that regulate CYP3A4 gene expression and cause drug interactions". The Journal of Clinical Investigation. 102 (5): 1016–1023. doi:10.1172/JCI3703. PMC 508967. PMID 9727070.
- ^ Meanwell NA (8 December 2014). Tactics in Contemporary Drug Design. Springer. pp. 161–. ISBN 978-3-642-55041-6. Archived from the original on 14 January 2023. Retrieved 1 February 2016.
- ^ a b Legato MJ, Bilezikian JP (2004). Principles of Gender-specific Medicine. Gulf Professional Publishing. pp. 146–. ISBN 978-0-12-440906-4. Archived from the original on 14 January 2023. Retrieved 1 February 2016.
- ^ Williams DA (24 January 2012). "Drug Metabolism". In Lemke TL, Williams DA (eds.). Foye's Principles of Medicinal Chemistry. Lippincott Williams & Wilkins. p. 164. ISBN 978-1-60913-345-0.
- ^ Estrogens—Advances in Research and Application: 2013 Edition: ScholarlyBrief. ScholarlyEditions. 21 June 2013. pp. 4–. ISBN 978-1-4816-7550-5. Archived from the original on 14 January 2023. Retrieved 1 February 2016.
- ^ Strünker T, Goodwin N, Brenker C, Kashikar ND, Weyand I, Seifert R, Kaupp UB (March 2011). "The CatSper channel mediates progesterone-induced Ca2+ influx in human sperm". Nature. 471 (7338): 382–386. Bibcode:2011Natur.471..382S. doi:10.1038/nature09769. PMID 21412338. S2CID 4431334.
- ^ Lishko PV, Botchkina IL, Kirichok Y (March 2011). "Progesterone activates the principal Ca2+ channel of human sperm". Nature. 471 (7338): 387–391. Bibcode:2011Natur.471..387L. doi:10.1038/nature09767. PMID 21412339. S2CID 4340309.
- ^ Prior JC (2020). "Women's reproductive system as balanced estradiol and progesterone actions—A revolutionary, paradigm-shifting concept in women's health". Drug Discovery Today: Disease Models. 32 (Part B): 31–40. doi:10.1016/j.ddmod.2020.11.005.
- ^ Kastner P, Krust A, Turcotte B, Stropp U, Tora L, Gronemeyer H, Chambon P (May 1990). "Two distinct estrogen-regulated promoters generate transcripts encoding the two functionally different human progesterone receptor forms A and B". The EMBO Journal. 9 (5): 1603–1614. doi:10.1002/j.1460-2075.1990.tb08280.x. PMC 551856. PMID 2328727.
- ^ a b Cline JM, Wood CE (1 January 2006). Hallam SZ, Osuch JR (eds.). "Hormonal effects on the mammary gland of postmenopausal nonhuman primates". Breast Disease. 24. IOS Press: 59–70. doi:10.3233/bd-2006-24105. ISBN 978-1-58603-653-9. PMID 16917139. Archived from the original on 27 November 2023. Retrieved 2 August 2023.
- ^ a b Johnson LR (2003). Essential Medical Physiology. Academic Press. p. 770. ISBN 978-0-12-387584-6. Archived from the original on 14 January 2023. Retrieved 1 February 2016.
- ^ a b Coad J, Dunstall M (2011). Anatomy and Physiology for Midwives, with Pageburst online access,3: Anatomy and Physiology for Midwives. Elsevier Health Sciences. p. 413. ISBN 978-0-7020-3489-3.
- ^ Landau RL, Bergenstal DM, Lugibihl K, Kascht ME (October 1955). "The metabolic effects of progesterone in man". The Journal of Clinical Endocrinology and Metabolism. 15 (10): 1194–1215. doi:10.1210/jcem-15-10-1194. PMID 13263410.
- ^ a b Masiutin M, Yadav M (2023). "Alternative androgen pathways". WikiJournal of Medicine. 10: X. doi:10.15347/WJM/2023.003. S2CID 257943362.
- ^ a b O'Shaughnessy PJ, Antignac JP, Le Bizec B, Morvan ML, Svechnikov K, Söder O, et al. (February 2019). "Alternative (backdoor) androgen production and masculinization in the human fetus". PLOS Biology. 17 (2) e3000002. doi:10.1371/journal.pbio.3000002. PMC 6375548. PMID 30763313.
- ^ Flück CE, Pandey AV (May 2014). "Steroidogenesis of the testis -- new genes and pathways". Annales d'Endocrinologie. 75 (2): 40–47. doi:10.1016/j.ando.2014.03.002. PMID 24793988.
- ^ Zachmann M (February 1996). "Prismatic cases: 17,20-desmolase (17,20-lyase) deficiency". The Journal of Clinical Endocrinology and Metabolism. 81 (2): 457–459. doi:10.1210/jcem.81.2.8636249. PMID 8636249.
- ^ Correia JN, Conner SJ, Kirkman-Brown JC (May 2007). "Non-genomic steroid actions in human spermatozoa. "Persistent tickling from a laden environment"". Seminars in Reproductive Medicine. 25 (3): 208–219. doi:10.1055/s-2007-973433. PMID 17447210. S2CID 260318879.
- ^ Kirkman-Brown JC, Bray C, Stewart PM, Barratt CL, Publicover SJ (June 2000). "Biphasic elevation of [Ca(2+)](i) in individual human spermatozoa exposed to progesterone". Developmental Biology. 222 (2): 326–335. doi:10.1006/dbio.2000.9729. PMID 10837122.
- ^ Kirkman-Brown JC, Barratt CL, Publicover SJ (March 2004). "Slow calcium oscillations in human spermatozoa". The Biochemical Journal. 378 (Pt 3): 827–832. doi:10.1042/BJ20031368. PMC 1223996. PMID 14606954.
- ^ Harper CV, Barratt CL, Publicover SJ (October 2004). "Stimulation of human spermatozoa with progesterone gradients to simulate approach to the oocyte. Induction of [Ca(2+)](i) oscillations and cyclical transitions in flagellar beating". The Journal of Biological Chemistry. 279 (44): 46315–46325. doi:10.1074/jbc.M401194200. PMID 15322137.
- ^ Marieb E (2013). Anatomy & physiology. Benjamin-Cummings. p. 903. ISBN 978-0-321-88760-3.
- ^ Tosti E, Di Cosmo A, Cuomo A, Di Cristo C, Gragnaniello G (May 2001). "Progesterone induces activation in Octopus vulgaris spermatozoa". Molecular Reproduction and Development. 59 (1): 97–105. doi:10.1002/mrd.1011. PMID 11335951. S2CID 28390608.
- ^ a b Bowen R (6 August 2000). "Placental Hormones". Archived from the original on 17 May 2007. Retrieved 12 March 2008.
- ^ Patel B, Elguero S, Thakore S, Dahoud W, Bedaiwy M, Mesiano S (2014). "Role of nuclear progesterone receptor isoforms in uterine pathophysiology". Human Reproduction Update. 21 (2): 155–173. doi:10.1093/humupd/dmu056. PMC 4366574. PMID 25406186.
- ^ a b Di Renzo GC, Giardina I, Clerici G, Brillo E, Gerli S (July 2016). "Progesterone in normal and pathological pregnancy". Hormone Molecular Biology and Clinical Investigation. 27 (1): 35–48. doi:10.1515/hmbci-2016-0038. PMID 27662646. S2CID 32239449.
- ^ Care A, Nevitt SJ, Medley N, Donegan S, Good L, Hampson L, et al. (February 2022). "Interventions to prevent spontaneous preterm birth in women with singleton pregnancy who are at high risk: systematic review and network meta-analysis". BMJ. 376 e064547. doi:10.1136/bmj-2021-064547. PMC 8845039. PMID 35168930.
- ^ Macias H, Hinck L (2012). "Mammary gland development". Wiley Interdisciplinary Reviews. Developmental Biology. 1 (4): 533–557. doi:10.1002/wdev.35. PMC 3404495. PMID 22844349.
- ^ a b c Hilton HN, Graham JD, Clarke CL (September 2015). "Minireview: Progesterone Regulation of Proliferation in the Normal Human Breast and in Breast Cancer: A Tale of Two Scenarios?". Molecular Endocrinology. 29 (9): 1230–1242. doi:10.1210/me.2015-1152. PMC 5414684. PMID 26266959.
- ^ Barbieri RL (13 September 2013). "The Breast". In Strauss JF, Barbieri RL (eds.). Yen and Jaffe's Reproductive Endocrinology. Elsevier Health Sciences. pp. 236–. ISBN 978-1-4557-2758-2. Archived from the original on 14 January 2023. Retrieved 1 February 2016.
- ^ Scaling AL, Prossnitz ER, Hathaway HJ (June 2014). "GPER mediates estrogen-induced signaling and proliferation in human breast epithelial cells and normal and malignant breast". Hormones & Cancer. 5 (3): 146–160. doi:10.1007/s12672-014-0174-1. PMC 4091989. PMID 24718936.
- ^ a b c d e Aupperlee MD, Leipprandt JR, Bennett JM, Schwartz RC, Haslam SZ (May 2013). "Amphiregulin mediates progesterone-induced mammary ductal development during puberty". Breast Cancer Research. 15 (3): R44. doi:10.1186/bcr3431. PMC 3738150. PMID 23705924.
- ^ a b Kuhl H, Schneider HP (August 2013). "Progesterone--promoter or inhibitor of breast cancer". Climacteric. 16 (Suppl 1): 54–68. doi:10.3109/13697137.2013.768806. PMID 23336704. S2CID 20808536.
- ^ a b c Trabert B, Sherman ME, Kannan N, Stanczyk FZ (April 2020). "Progesterone and Breast Cancer". Endocrine Reviews. 41 (2): 320–344. doi:10.1210/endrev/bnz001. PMC 7156851. PMID 31512725.
- ^ Collaborative Group on Hormonal Factors in Breast Cancer (September 2019). "Type and timing of menopausal hormone therapy and breast cancer risk: individual participant meta-analysis of the worldwide epidemiological evidence". Lancet. 394 (10204): 1159–1168. doi:10.1016/S0140-6736(19)31709-X. PMC 6891893. PMID 31474332.
- ^ Stute P, Wildt L, Neulen J (April 2018). "The impact of micronized progesterone on breast cancer risk: a systematic review" (PDF). Climacteric. 21 (2): 111–122. doi:10.1080/13697137.2017.1421925. PMID 29384406. S2CID 3642971. Archived (PDF) from the original on 2 February 2024. Retrieved 2 February 2024.
- ^ Asi N, Mohammed K, Haydour Q, Gionfriddo MR, Vargas OL, Prokop LJ, et al. (July 2016). "Progesterone vs. synthetic progestins and the risk of breast cancer: a systematic review and meta-analysis". Systematic Reviews. 5 (1): 121. doi:10.1186/s13643-016-0294-5. PMC 4960754. PMID 27456847.
- ^ Gompel A, Plu-Bureau G (August 2018). "Progesterone, progestins and the breast in menopause treatment". Climacteric. 21 (4): 326–332. doi:10.1080/13697137.2018.1476483. PMID 29852797. S2CID 46922084.
- ^ Davey DA (October 2018). "Menopausal hormone therapy: a better and safer future". Climacteric. 21 (5): 454–461. doi:10.1080/13697137.2018.1439915. PMID 29526116. S2CID 3850275.
- ^ a b c d e f g h Raine-Fenning NJ, Brincat MP, Muscat-Baron Y (2003). "Skin aging and menopause: implications for treatment". American Journal of Clinical Dermatology. 4 (6): 371–378. doi:10.2165/00128071-200304060-00001. PMID 12762829. S2CID 20392538.
- ^ a b c d e f g h Holzer G, Riegler E, Hönigsmann H, Farokhnia S, Schmidt JB (September 2005). "Effects and side-effects of 2% progesterone cream on the skin of peri- and postmenopausal women: results from a double-blind, vehicle-controlled, randomized study". The British Journal of Dermatology. 153 (3): 626–634. doi:10.1111/j.1365-2133.2005.06685.x. PMID 16120154. S2CID 6077829.
- ^ King SR (9 November 2012). Neurosteroids and the Nervous System. Springer Science & Business Media. pp. 44–46. ISBN 978-1-4614-5559-2.
- ^ Fleischman DS, Fessler DM, Cholakians AE (July 2015). "Testing the Affiliation Hypothesis of Homoerotic Motivation in Humans: The Effects of Progesterone and Priming". Archives of Sexual Behavior. 44 (5): 1395–1404. doi:10.1007/s10508-014-0436-6. PMID 25420899. S2CID 9864224. Archived from the original on 23 September 2020. Retrieved 2 August 2023.
- ^ "Homosexuality may help us bond". UoP News. Archived from the original on 2 July 2019. Retrieved 2 July 2019.
- ^ "Having homosexual thoughts 'is an essential part of human evolution' study suggests". The Telegraph. 25 November 2014. Archived from the original on 15 February 2018. Retrieved 4 April 2018.
- ^ "New Study Identifies Evolutionary Basis Of Homosexuality". HuffPost. 26 November 2014. Archived from the original on 27 November 2023. Retrieved 21 October 2023.
- ^ a b c Hanukoglu I, Karavolas HJ, Goy RW (April 1977). "Progesterone metabolism in the pineal, brain stem, thalamus and corpus callosum of the female rat". Brain Research. 125 (2): 313–324. doi:10.1016/0006-8993(77)90624-2. PMID 558037. S2CID 35814845. Archived from the original on 5 March 2021. Retrieved 28 June 2019.
- ^ Schumacher M, Guennoun R, Robert F, Carelli C, Gago N, Ghoumari A, et al. (June 2004). "Local synthesis and dual actions of progesterone in the nervous system: neuroprotection and myelination". Growth Hormone & IGF Research. 14 (Suppl A): S18 – S33. doi:10.1016/j.ghir.2004.03.007. PMID 15135772.
- ^ Singh M, Su C, Ng S (September 2013). "Non-genomic mechanisms of progesterone action in the brain". Frontiers in Neuroscience. 7: 159. doi:10.3389/fnins.2013.00159. PMC 3776940. PMID 24065876.
- ^ Roof RL, Hall ED (May 2000). "Gender differences in acute CNS trauma and stroke: neuroprotective effects of estrogen and progesterone". Journal of Neurotrauma. 17 (5): 367–388. doi:10.1089/neu.2000.17.367. PMID 10833057.
- ^ Pan DS, Liu WG, Yang XF, Cao F (October 2007). "Inhibitory effect of progesterone on inflammatory factors after experimental traumatic brain injury". Biomedical and Environmental Sciences. 20 (5): 432–438. PMID 18188998.
- ^ Jiang C, Zuo F, Wang Y, Wan J, Yang Z, Lu H, et al. (June 2016). "Progesterone exerts neuroprotective effects and improves long-term neurologic outcome after intracerebral hemorrhage in middle-aged mice". Neurobiology of Aging. 42: 13–24. doi:10.1016/j.neurobiolaging.2016.02.029. PMC 4857017. PMID 27143417.
- ^ a b Luoma JI, Stern CM, Mermelstein PG (August 2012). "Progesterone inhibition of neuronal calcium signaling underlies aspects of progesterone-mediated neuroprotection". The Journal of Steroid Biochemistry and Molecular Biology. 131 (1–2): 30–36. doi:10.1016/j.jsbmb.2011.11.002. PMC 3303940. PMID 22101209.
- ^ a b c Stein DG (March 2008). "Progesterone exerts neuroprotective effects after brain injury". Brain Research Reviews. 57 (2): 386–397. doi:10.1016/j.brainresrev.2007.06.012. PMC 2699575. PMID 17826842.
- ^ Espinoza TR, Wright DW (2011). "The role of progesterone in traumatic brain injury". The Journal of Head Trauma Rehabilitation. 26 (6): 497–499. doi:10.1097/HTR.0b013e31823088fa. PMC 6025750. PMID 22088981.
- ^ Jiang C, Zuo F, Wang Y, Lu H, Yang Q, Wang J (January 2016). "Progesterone Changes VEGF and BDNF Expression and Promotes Neurogenesis After Ischemic Stroke". Molecular Neurobiology. 54 (1): 571–581. doi:10.1007/s12035-015-9651-y. PMC 4938789. PMID 26746666.
- ^ Herson PS, Koerner IP, Hurn PD (May 2009). "Sex, sex steroids, and brain injury". Seminars in Reproductive Medicine. 27 (3): 229–239. doi:10.1055/s-0029-1216276. PMC 2675922. PMID 19401954.
- ^ Li Z, Wang B, Kan Z, Zhang B, Yang Z, Chen J, et al. (January 2012). "Progesterone increases circulating endothelial progenitor cells and induces neural regeneration after traumatic brain injury in aged rats". Journal of Neurotrauma. 29 (2): 343–353. doi:10.1089/neu.2011.1807. PMC 3261789. PMID 21534727.
- ^ a b Lynch WJ, Sofuoglu M (December 2010). "Role of progesterone in nicotine addiction: evidence from initiation to relapse". Experimental and Clinical Psychopharmacology. 18 (6): 451–461. doi:10.1037/a0021265. PMC 3638762. PMID 21186920.
- ^ Cosgrove KP, Esterlis I, McKee SA, Bois F, Seibyl JP, Mazure CM, et al. (April 2012). "Sex differences in availability of β2*-nicotinic acetylcholine receptors in recently abstinent tobacco smokers". Archives of General Psychiatry. 69 (4): 418–427. doi:10.1001/archgenpsychiatry.2011.1465. PMC 3508698. PMID 22474108.
- ^ Mello NK, Knudson IM, Kelly M, Fivel PA, Mendelson JH (October 2011). "Effects of progesterone and testosterone on cocaine self-administration and cocaine discrimination by female rhesus monkeys". Neuropsychopharmacology. 36 (11): 2187–2199. doi:10.1038/npp.2011.130. PMC 3176575. PMID 21796112.
- ^ Buser T (1 June 2012). "The impact of the menstrual cycle and hormonal contraceptives on competitiveness" (PDF). Journal of Economic Behavior & Organization. Gender Differences in Risk Aversion and Competition. 83 (1): 1–10. doi:10.1016/j.jebo.2011.06.006. ISSN 0167-2681. Archived (PDF) from the original on 2 February 2024. Retrieved 2 February 2024.
- ^ Sriram D (2007). Medicinal Chemistry. New Delhi: Dorling Kindersley India Pvt. Ltd. p. 432. ISBN 978-81-317-0031-0.
- ^ a b Blackburn S (14 April 2014). Maternal, Fetal, & Neonatal Physiology. Elsevier Health Sciences. pp. 92–. ISBN 978-0-323-29296-2.
- ^ Faivre EJ, Lange CA (January 2007). "Progesterone receptors upregulate Wnt-1 to induce epidermal growth factor receptor transactivation and c-Src-dependent sustained activation of Erk1/2 mitogen-activated protein kinase in breast cancer cells". Molecular and Cellular Biology. 27 (2): 466–480. doi:10.1128/MCB.01539-06. PMC 1800800. PMID 17074804.
- ^ Nosek TM. "Section 5/5ch9/s5ch9_13". Essentials of Human Physiology. Archived from the original on 24 March 2016.
- ^ Rothchild I (1969), Salhanick HA, Kipnis DM, Wiele RL (eds.), "The Physiologic Basis for the Temperature Raising Effect of Progesterone", Metabolic Effects of Gonadal Hormones and Contraceptive Steroids, Boston, MA: Springer US, pp. 668–675, doi:10.1007/978-1-4684-1782-1_49, ISBN 978-1-4684-1782-1
- ^ Lam M, Lamanna E, Bourke JE (2019), Hashitani H, Lang RJ (eds.), "Regulation of Airway Smooth Muscle Contraction in Health and Disease", Smooth Muscle Spontaneous Activity: Physiological and Pathological Modulation, Singapore: Springer, pp. 381–422, doi:10.1007/978-981-13-5895-1_16, ISBN 978-981-13-5895-1, retrieved 19 October 2025
- ^ Straub RH (1 August 2007). "The Complex Role of Estrogens in Inflammation". Endocrine Reviews. 28 (5): 521–574. doi:10.1210/er.2007-0001. ISSN 0163-769X.
- ^ Hould FS, Fried GM, Fazekas AG, Tremblay S, Mersereau WA (December 1988). "Progesterone receptors regulate gallbladder motility". The Journal of Surgical Research. 45 (6): 505–512. doi:10.1016/0022-4804(88)90137-0. PMID 3184927.
- ^ "Hormones and Oral Health". WebMD. Archived from the original on 18 June 2016. Retrieved 22 July 2013.
- ^ Picard F, Wanatabe M, Schoonjans K, Lydon J, O'Malley BW, Auwerx J (November 2002). "Progesterone receptor knockout mice have an improved glucose homeostasis secondary to beta -cell proliferation". Proceedings of the National Academy of Sciences of the United States of America. 99 (24): 15644–15648. doi:10.1073/pnas.202612199. PMC 137770. PMID 12438645.
- ^ Brănişteanu DD, Mathieu C (March 2003). "Progesterone in gestational diabetes mellitus: guilty or not guilty?". Trends in Endocrinology and Metabolism. 14 (2): 54–56. doi:10.1016/S1043-2760(03)00003-1. PMID 12591170. S2CID 38209977.
- ^ Whynott RM, Summers KM, Jakubiak M, Van Voorhis BJ, Mejia RB (June 2021). "The effect of weight and body mass index on serum progesterone values and live birth rate in cryopreserved in vitro fertilization cycles". F&S Reports. 2 (2): 195–200. doi:10.1016/j.xfre.2021.02.005. PMC 8267385. PMID 34278354.
- ^ Kaemmle LM, Stadler A, Janka H, von Wolff M, Stute P (August 2022). "The impact of micronized progesterone on cardiovascular events - a systematic review". Climacteric. 25 (4): 327–336. doi:10.1080/13697137.2021.2022644. PMID 35112635. S2CID 246487187.
- ^ Gasser S, Heidemeyer K, von Wolff M, Stute P (June 2021). "Impact of progesterone on skin and hair in menopause - a comprehensive review". Climacteric. 24 (3): 229–235. doi:10.1080/13697137.2020.1838476. PMID 33527841. S2CID 231757325.
- ^ Javed AA, Mayhew AJ, Shea AK, Raina P (August 2019). "Association Between Hormone Therapy and Muscle Mass in Postmenopausal Women: A Systematic Review and Meta-analysis". JAMA Network Open. 2 (8): e1910154. doi:10.1001/jamanetworkopen.2019.10154. PMC 6716293. PMID 31461147.
- ^ Coquoz A, Gruetter C, Stute P (April 2019). "Impact of micronized progesterone on body weight, body mass index, and glucose metabolism: a systematic review". Climacteric. 22 (2): 148–161. doi:10.1080/13697137.2018.1514003. PMID 30477366. S2CID 53782622.
- ^ Häggström M, Richfield D (2014). "Diagram of the pathways of human steroidogenesis". WikiJournal of Medicine. 1 (1). doi:10.15347/wjm/2014.005. ISSN 2002-4436.
- ^ Bewick PM (2002). Medicinal natural products: a biosynthetic approach. New York: Wiley. p. 244. ISBN 0-471-49641-3.
- ^ Duport C, Spagnoli R, Degryse E, Pompon D (February 1998). "Self-sufficient biosynthesis of pregnenolone and progesterone in engineered yeast". Nature Biotechnology. 16 (2): 186–189. doi:10.1038/nbt0298-186. PMID 9487528. S2CID 852617.
- ^ Zavod RM (24 January 2012). "Women's Health". In Lemke TL, Williams DA (eds.). Foye's Principles of Medicinal Chemistry. Lippincott Williams & Wilkins. pp. 1397–. ISBN 978-1-60913-345-0. Archived from the original on 14 January 2023. Retrieved 19 July 2018.
- ^ Progesterone - Drugs.com, archived from the original on 27 March 2019, retrieved 23 August 2015
- ^ a b c Falcone T, Hurd WW (2007). Clinical Reproductive Medicine and Surgery. Elsevier Health Sciences. pp. 22–. ISBN 978-0-323-03309-1. Archived from the original on 10 January 2023. Retrieved 6 November 2016.
- ^ a b c d Cupps PT (20 February 1991). Reproduction in Domestic Animals. Elsevier. pp. 101–. ISBN 978-0-08-057109-6.
- ^ a b c d e f g h i Stanczyk FZ (November 2003). "All progestins are not created equal". Steroids. 68 (10–13): 879–890. doi:10.1016/j.steroids.2003.08.003. PMID 14667980. S2CID 44601264.
- ^ Dowd FJ, Johnson B, Mariotti A (3 September 2016). Pharmacology and Therapeutics for Dentistry. Elsevier Health Sciences. pp. 448–. ISBN 978-0-323-44595-5.
- ^ a b c d Kuhl H (August 2005). "Pharmacology of estrogens and progestogens: influence of different routes of administration". Climacteric. 8 (Suppl 1): 3–63. doi:10.1080/13697130500148875. PMID 16112947. S2CID 24616324.
- ^ a b Plant TM, Zeleznik AJ (15 November 2014). Knobil and Neill's Physiology of Reproduction. Academic Press. pp. 304–. ISBN 978-0-12-397769-4.
- ^ a b Santoro NF, Neal-Perry G (11 September 2010). Amenorrhea: A Case-Based, Clinical Guide. Springer Science & Business Media. pp. 13–. ISBN 978-1-60327-864-5. Archived from the original on 14 January 2023. Retrieved 6 November 2016.
- ^ Reddy DS (2010). "Neurosteroids". Sex Differences in the Human Brain, their Underpinnings and Implications. Progress in Brain Research. Vol. 186. Elsevier. pp. 113–137. doi:10.1016/B978-0-444-53630-3.00008-7. ISBN 978-0-444-53630-3. PMC 3139029. PMID 21094889.
- ^ a b Baulieu E, Kelly PA (30 November 1990). Hormones: From Molecules to Disease. Springer Science & Business Media. pp. 401–. ISBN 978-0-412-02791-8.
- ^ Beranič N, Gobec S, Rižner TL (May 2011). "Progestins as inhibitors of the human 20-ketosteroid reductases, AKR1C1 and AKR1C3". Chemico-Biological Interactions. 191 (1–3): 227–233. Bibcode:2011CBI...191..227B. doi:10.1016/j.cbi.2010.12.012. PMID 21182831.
- ^ Anderson GD, Odegard PS (October 2004). "Pharmacokinetics of estrogen and progesterone in chronic kidney disease". Advances in Chronic Kidney Disease. 11 (4): 357–360. doi:10.1053/j.ackd.2004.07.001. PMID 15492972.
- ^ Greenblatt JM, Brogan K (27 April 2016). Integrative Therapies for Depression: Redefining Models for Assessment, Treatment and Prevention. CRC Press. pp. 201–. ISBN 978-1-4987-0230-0.
- ^ Graham C (2 December 2012). Reproductive Biology of the Great Apes: Comparative and Biomedical Perspectives. Elsevier. pp. 179–. ISBN 978-0-323-14971-6. Archived from the original on 14 January 2023. Retrieved 6 November 2016.
- ^ Strushkevich N, Gilep AA, Shen L, Arrowsmith CH, Edwards AM, Usanov SA, Park HW (February 2013). "Structural insights into aldosterone synthase substrate specificity and targeted inhibition". Molecular Endocrinology. 27 (2): 315–324. doi:10.1210/me.2012-1287. PMC 5417327. PMID 23322723.
- ^ van Rooyen D, Gent R, Barnard L, Swart AC (April 2018). "The in vitro metabolism of 11β-hydroxyprogesterone and 11-ketoprogesterone to 11-ketodihydrotestosterone in the backdoor pathway". The Journal of Steroid Biochemistry and Molecular Biology. 178: 203–212. doi:10.1016/j.jsbmb.2017.12.014. PMID 29277707. S2CID 3700135.
- ^ a b de Azevedo Piccinato C (2008). Regulation of Steroid Metabolism and the Hepatic Transcriptome by Estradiol and Progesterone. pp. 24–25. ISBN 978-1-109-04632-8.[permanent dead link]
- ^ Akalin S (January 1991). "Effects of ketoconazole in hirsute women". Acta Endocrinologica. 124 (1): 19–22. doi:10.1530/acta.0.1240019. PMID 1825737. S2CID 9831739.
- ^ a b Aufrère MB, Benson H (June 1976). "Progesterone: an overview and recent advances". Journal of Pharmaceutical Sciences. 65 (6): 783–800. doi:10.1002/jps.2600650602. PMID 945344.
- ^ Stricker R, Eberhart R, Chevailler MC, Quinn FA, Bischof P, Stricker R (2006). "Establishment of detailed reference values for luteinizing hormone, follicle stimulating hormone, estradiol, and progesterone during different phases of the menstrual cycle on the Abbott ARCHITECT analyzer". Clinical Chemistry and Laboratory Medicine. 44 (7): 883–887. doi:10.1515/CCLM.2006.160. PMID 16776638. S2CID 524952.
- ^ Csapo AI, Pulkkinen MO, Wiest WG (March 1973). "Effects of luteectomy and progesterone replacement therapy in early pregnant patients". American Journal of Obstetrics and Gynecology. 115 (6): 759–765. doi:10.1016/0002-9378(73)90517-6. PMID 4688578.
- ^ NIH Clinical Center (16 August 2004). "Progesterone Historical Reference Ranges". United States National Institutes of Health. Archived from the original on 9 January 2009. Retrieved 12 March 2008.
- ^ Chernecky CC, Berger BJ (31 October 2012). Laboratory Tests and Diagnostic Procedures - E-Book. Elsevier Health Sciences. pp. 908–. ISBN 978-1-4557-4502-9. Archived from the original on 27 February 2024. Retrieved 23 August 2023.
- ^ Becker KL (2001). Principles and Practice of Endocrinology and Metabolism. Lippincott Williams & Wilkins. pp. 889, 940. ISBN 978-0-7817-1750-2. Archived from the original on 27 February 2024. Retrieved 23 August 2023.
- ^ Josimovich JB (11 November 2013). Gynecologic Endocrinology. Springer Science & Business Media. pp. 9, 25–29, 139. ISBN 978-1-4613-2157-6. Archived from the original on 14 January 2023. Retrieved 1 February 2016.
- ^ van Keep P, Utian W (6 December 2012). The Premenstrual Syndrome: Proceedings of a workshop held during the Sixth International Congress of Psychosomatic Obstetrics and Gynecology, Berlin, September 1980. Springer Science & Business Media. pp. 51–52. ISBN 978-94-011-6255-5. Archived from the original on 14 January 2023. Retrieved 1 February 2016.
- ^ Strauss JF, Barbieri RL (2009). Yen and Jaffe's Reproductive Endocrinology: Physiology, Pathophysiology, and Clinical Management. Elsevier Health Sciences. pp. 807–. ISBN 978-1-4160-4907-4. Archived from the original on 10 January 2023. Retrieved 23 August 2023.
- ^ Bajaj L, Berman S (1 January 2011). Berman's Pediatric Decision Making. Elsevier Health Sciences. pp. 160–. ISBN 978-0-323-05405-8. Archived from the original on 11 January 2023. Retrieved 23 August 2023.
- ^ Lauritzen C (1988). "Natürliche und Synthetische Sexualhormone – Biologische Grundlagen und Behandlungsprinzipien" [Natural and Synthetic Sexual Hormones – Biological Basis and Medical Treatment Principles]. In Hermann P. G. Schneider, Christian Lauritzen, Eberhard Nieschlag (eds.). Grundlagen und Klinik der Menschlichen Fortpflanzung [Foundations and Clinic of Human Reproduction] (in German). Walter de Gruyter. pp. 229–306. ISBN 978-3-11-010968-9. OCLC 35483492. Archived from the original on 1 October 2023. Retrieved 23 August 2023.
- ^ Little, A. B., & Billiar, R. B. (1983). Progestagens. In Endocrinology of Pregnancy, 3rd Edition (pp. 92–111). Harper and Row Philadelphia. https://scholar.google.com/scholar?cluster=2512291948467467634 Archived 22 February 2022 at the Wayback Machine
- ^ a b c d e f g h i Progesterone Reference Ranges, Performed at the Clinical Center at the National Institutes of Health, Bethesda MD, 03Feb09
- ^ a b c d e f g h Converted from mass values using molar mass of 314.46 g/mol
- ^ Häggström M (2014). "Reference ranges for estradiol, progesterone, luteinizing hormone and follicle-stimulating hormone during the menstrual cycle". WikiJournal of Medicine. 1 (1). doi:10.15347/wjm/2014.001. ISSN 2002-4436. S2CID 88035135.
- ^ Goodson III WH, Handagama P, Moore II DH, Dairkee S (13 December 2007). "Milk products are a source of dietary progesterone". 30th Annual San Antonio Breast Cancer Symposium. pp. abstract # 2028. Archived from the original on 20 September 2008. Retrieved 12 March 2008.
- ^ Pauli GF, Friesen JB, Gödecke T, Farnsworth NR, Glodny B (March 2010). "Occurrence of progesterone and related animal steroids in two higher plants". Journal of Natural Products. 73 (3): 338–345. Bibcode:2010JNAtP..73..338P. doi:10.1021/np9007415. PMID 20108949. S2CID 26467578.
- ^ Applezweig N (May 1969). "Steroids". Chemical Week. 104: 57–72. PMID 12255132.
- ^ Noguchi E, Fujiwara Y, Matsushita S, Ikeda T, Ono M, Nohara T (September 2006). "Metabolism of tomato steroidal glycosides in humans". Chemical & Pharmaceutical Bulletin. 54 (9): 1312–1314. doi:10.1248/cpb.54.1312. PMID 16946542.
- ^ Yang DJ, Lu TJ, Hwang LS (October 2003). "Isolation and identification of steroidal saponins in Taiwanese yam cultivar (Dioscorea pseudojaponica Yamamoto)" (PDF). Journal of Agricultural and Food Chemistry. 51 (22): 6438–6444. Bibcode:2003JAFC...51.6438Y. doi:10.1021/jf030390j. PMID 14558759. Archived (PDF) from the original on 3 August 2022. Retrieved 2 April 2022.
- ^ "Final report of the amended safety assessment of Dioscorea Villosa (Wild Yam) root extract". International Journal of Toxicology. 23 (Suppl 2): 49–54. 2004. doi:10.1080/10915810490499055. PMID 15513824. S2CID 962216.
- ^ Niño J, Jiménez DA, Mosquera OM, Correa YM (2007). "Diosgenin quantification by HPLC in a Dioscorea polygonoides tuber collection from colombian flora". Journal of the Brazilian Chemical Society. 18 (5): 1073–1076. doi:10.1590/S0103-50532007000500030. S2CID 95193700.
- ^ Myoda T, Nagai T, Nagashima T (2005). "Properties of starches in yam (Dioscorea spp.) tuber". Current Topics in Food Science and Technology: 105–114. ISBN 81-308-0003-9.
- ^ Wesp LM, Deutsch MB (March 2017). "Hormonal and Surgical Treatment Options for Transgender Women and Transfeminine Spectrum Persons". The Psychiatric Clinics of North America. 40 (1): 99–111. doi:10.1016/j.psc.2016.10.006. PMID 28159148.
- ^ Dolitsky SN, Cordeiro Mitchell CN, Stadler SS, Segars JH (November 2020). "Efficacy of progestin-only treatment for the management of menopausal symptoms: a systematic review". Menopause. 28 (2): 217–224. doi:10.1097/GME.0000000000001676. PMID 33109992. S2CID 225100434.
- ^ Ruan X, Mueck AO (November 2014). "Systemic progesterone therapy--oral, vaginal, injections and even transdermal?". Maturitas. 79 (3): 248–255. doi:10.1016/j.maturitas.2014.07.009. PMID 25113944.
- ^ Filicori M (November 2015). "Clinical roles and applications of progesterone in reproductive medicine: an overview". Acta Obstetricia et Gynecologica Scandinavica. 94 (Suppl 161): 3–7. doi:10.1111/aogs.12791. PMID 26443945.
- ^ Ciampaglia W, Cognigni GE (November 2015). "Clinical use of progesterone in infertility and assisted reproduction". Acta Obstetricia et Gynecologica Scandinavica. 94 (Suppl 161): 17–27. doi:10.1111/aogs.12770. PMID 26345161. S2CID 40753277.
- ^ Choi SJ (September 2017). "Use of progesterone supplement therapy for prevention of preterm birth: review of literatures". Obstetrics & Gynecology Science. 60 (5): 405–420. doi:10.5468/ogs.2017.60.5.405. PMC 5621069. PMID 28989916.
- ^ Coomarasamy A, Harb HM, Devall AJ, Cheed V, Roberts TE, Goranitis I, et al. (June 2020). "Progesterone to prevent miscarriage in those with early pregnancy bleeding: the PRISM RCT". Health Technology Assessment. 24 (33): 1–70. doi:10.3310/hta24330. PMC 7355406. PMID 32609084.
- ^ a b Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 1024–. ISBN 978-1-4757-2085-3.
- ^ a b Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. pp. 880–. ISBN 978-3-88763-075-1.
- ^ Numazawa M, Nagaoka M, Kunitama Y (September 1986). "Regiospecific deoxygenation of the dihydroxyacetone moiety at C-17 of corticoid steroids with iodotrimethylsilane". Chemical & Pharmaceutical Bulletin. 34 (9): 3722–3726. doi:10.1248/cpb.34.3722. PMID 3815593.
- ^ a b Marker RE, Krueger J (1940). "Sterols. CXII. Sapogenins. XLI. The Preparation of Trillin and its Conversion to Progesterone". J. Am. Chem. Soc. 62 (12): 3349–3350. Bibcode:1940JAChS..62.3349M. doi:10.1021/ja01869a023.
- ^ Goswami A, Kotoky R, Rastogi RC, Ghosh AC (1 May 2003). "A One-Pot Efficient Process for 16-Dehydropregnenolone Acetate". Organic Process Research & Development. 7 (3): 306–308. doi:10.1021/op0200625.
- ^ Heyl FW (1950). "Progesterone from 3-Acetoxybisnor-5-cholenaldehyde and 3-Ketobisnor-4-cholenaldehyde". Journal of the American Chemical Society. 72 (6): 2617–2619. Bibcode:1950JAChS..72.2617H. doi:10.1021/ja01162a076.
- ^ Slomp G (1958). "Ozonolysis. II. 1 The Effect of Pyridine on the Ozonolysis of 4,22-Stigmastadien-3-one 2". Journal of the American Chemical Society. 80 (4): 915–921. Bibcode:1958JAChS..80..915S. doi:10.1021/ja01537a041.
- ^ Sundararaman P, Djerassi C (October 1977). "A convenient synthesis of progesterone from stigmasterol". The Journal of Organic Chemistry. 42 (22): 3633–3634. doi:10.1021/jo00442a044. PMID 915584.
- ^ "Nova Transcripts: Forgotten Genius". PBS.org. 6 February 2007. Archived from the original on 11 October 2018. Retrieved 8 September 2017.
- ^ "Giants of the Past". lipidlibrary.aocs.org. Archived from the original on 15 April 2012.
- ^ a b c Johnson WS, Gravestock MB, McCarry BE (August 1971). "Acetylenic bond participation in biogenetic-like olefinic cyclizations. II. Synthesis of dl-progesterone". Journal of the American Chemical Society. 93 (17): 4332–4334. Bibcode:1971JAChS..93.4332J. doi:10.1021/ja00746a062. PMID 5131151.
- ^ Corner GW, Allen WM (1 March 1929). "Physiology of the corpus luteum". American Journal of Physiology. Legacy Content. 88 (2): 326–339. doi:10.1152/ajplegacy.1929.88.2.326. ISSN 0002-9513. Archived from the original on 12 August 2021. Retrieved 12 August 2021.
- ^ a b c Coutinho EM, Segal SJ (1999). Is Menstruation Obsolete?. Oxford University Press. pp. 31–. ISBN 978-0-19-513021-8. Archived from the original on 14 January 2023. Retrieved 5 October 2016.
- ^ Walker A (7 March 2008). The Menstrual Cycle. Routledge. pp. 49–. ISBN 978-1-134-71411-7.
- ^ Piosik R (2003). "Adolf Butenandt und sein Wirken an der Technischen Hochschule Danzig". CHEMKON. 10 (3): 135–138. doi:10.1002/ckon.200390038.
- ^ Ginsburg B (6 December 2012). Premenstrual Syndrome: Ethical and Legal Implications in a Biomedical Perspective. Springer Science & Business Media. pp. 274–. ISBN 978-1-4684-5275-4. Archived from the original on 14 January 2023. Retrieved 5 October 2016.
- ^ Rolleston HD (1936). The Endocrine Organs in Health and Disease: With an Historical Review. Oxford University Press, H. Milford. p. 406. Archived from the original on 14 January 2023. Retrieved 5 October 2016.
- ^ Allen WM (October 1970). "Progesterone: how did the name originate?". Southern Medical Journal. 63 (10): 1151–1155. doi:10.1097/00007611-197010000-00012. PMID 4922128. S2CID 35867375.
- ^ Refsal K (February 2009). "Interpretation of Serum Progesterone Results for Management of Breeding in Dogs" (PDF). Webcd.endo.ref. Archived from the original on 29 August 2021. Retrieved 26 February 2018.
- ^ "Progesterone Prices, Coupons & Savings Tips - GoodRx". goodrx.com. Archived from the original on 30 March 2023. Retrieved 1 August 2023.
- ^ "Progesterone Prices, Coupons & Savings Tips - GoodRx". goodrx.com. Archived from the original on 30 March 2023. Retrieved 1 August 2023.
External links
[edit]- Progesterone MS Spectrum
- Progesterone at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Kimball JW (27 May 2007). "Progesterone". Kimball's Biology Pages. Archived from the original on 18 June 2008. Retrieved 18 June 2008.
Progesterone
View on GrokipediaBiological functions
Hormonal interactions
Progesterone plays a central role in the hypothalamic-pituitary-ovarian axis during the menstrual cycle, primarily exerting negative feedback on gonadotropin-releasing hormone (GnRH), luteinizing hormone (LH), and follicle-stimulating hormone (FSH) secretion, in contrast to the biphasic effects of estrogen. In the follicular phase, low progesterone levels allow rising estrogen to initially provide negative feedback, suppressing GnRH and gonadotropin pulses to maintain early follicular development. As estrogen peaks mid-cycle, it switches to positive feedback, triggering a surge in GnRH and LH that induces ovulation. Post-ovulation, progesterone from the corpus luteum dominates, reinforcing negative feedback on the hypothalamus and pituitary to inhibit GnRH pulsatility and reduce LH and FSH secretion, thereby preventing further follicular maturation during the luteal phase.[9][10][11] In uterine preparation for implantation, progesterone synergizes with estrogen to promote endometrial proliferation and differentiation while also antagonizing certain estrogen-driven effects to establish receptivity. Estrogen initially stimulates endometrial growth and vascularization during the proliferative phase, but rising progesterone in the secretory phase induces secretory transformation, decidualization, and immune modulation necessary for embryo attachment. This synergy is evident in the coordinated expression of progesterone receptors (PR) and estrogen receptors (ER), where progesterone enhances ER activity for stromal remodeling but antagonizes estrogen's mitogenic effects on epithelial cells to create a narrow window of implantation (days 20-24 of the cycle). Disruptions in this balance, such as progesterone resistance, can impair receptivity and lead to implantation failure.[12][13][14] Progesterone modulates glucocorticoid and mineralocorticoid activity through its affinity for the mineralocorticoid receptor (MR), acting as a competitive antagonist to cortisol and aldosterone. With binding affinity similar to aldosterone, progesterone inhibits MR activation in target tissues like the kidney and vasculature, counteracting sodium retention and potassium excretion promoted by aldosterone during high-progesterone states such as pregnancy. This antagonism helps maintain fluid balance, though elevated progesterone can also influence cortisol feedback indirectly by altering hypothalamic-pituitary-adrenal axis sensitivity without directly binding glucocorticoid receptors.[15][16][17] The progesterone-to-estrogen ratio varies markedly across the menstrual cycle and pregnancy, reflecting their dynamic interplay. In the follicular phase, progesterone levels remain low (<1 ng/mL) relative to rising estradiol (20-400 pg/mL), yielding a low ratio that favors estrogen dominance. During the luteal phase, progesterone surges to 5-20 ng/mL while estradiol stabilizes at 50-250 pg/mL, increasing the ratio to approximately 20-100:1, which sustains endometrial support. In pregnancy, both hormones rise dramatically—progesterone to 100-200 ng/mL and estradiol to >10 ng/mL by term—but the ratio shifts to around 10-20:1, ensuring progesterone's primacy for maintaining gestation.[18][9][19]Early sexual differentiation
In males, the regression of the Müllerian ducts, which would otherwise form the female reproductive tract, is primarily driven by anti-Müllerian hormone (AMH) secreted by Sertoli cells in the developing testes starting around gestational week 8. Progesterone enhances AMH's regressive effects on the Müllerian ducts through direct interactions, as demonstrated in organ culture studies where progesterone at concentrations of 10^{-6} M potentiated AMH activity, leading to more complete ductal degeneration.[20] This enhancement occurs in conjunction with rising androgen levels, which stabilize the male phenotype, during the critical differentiation window of weeks 8 to 12 in human gestation when internal genitalia form.[21] In females, the absence of androgens prevents stabilization of the Wolffian ducts, while the lack of AMH allows the Müllerian ducts to persist and differentiate into the fallopian tubes, uterus, and upper vagina during the same gestational weeks 8 to 12. Although this development proceeds largely as a default pathway without active hormonal promotion, progesterone levels from the corpus luteum contribute to the early embryonic environment that supports Müllerian duct maintenance in the absence of regressive signals from androgens or AMH.[21] Animal models provide evidence for progesterone's influence on sexual differentiation, particularly through disruptions observed in exposure and genetic studies. In progesterone receptor knockout (PRKO) mice, while gross genital tract anatomy forms normally, altered progesterone signaling leads to impaired reproductive tract maturation and function, highlighting its role in fine-tuning differentiation processes.[22] These findings from rodent and fish models underscore progesterone's modulatory effects on early sexual characteristics, often via interactions with androgen pathways.[23]Reproductive system
Progesterone plays a central role in preparing the female reproductive system for pregnancy by inducing decidualization of the endometrial stromal cells, a process essential for embryo implantation. During the luteal phase of the menstrual cycle, rising progesterone levels from the corpus luteum transform the proliferative endometrium into a secretory state, promoting vascular remodeling, immune modulation, and nutrient provision to support the implanted blastocyst. This decidual reaction involves the expression of progesterone receptors in stromal cells, which trigger morphological changes such as cellular enlargement and the secretion of prolactin and other factors that create a receptive environment for implantation.[24][25][26] In maintaining pregnancy, progesterone inhibits uterine contractions by relaxing the myometrium and suppressing inflammatory pathways that could lead to preterm labor or miscarriage. It achieves this through negative regulation of contraction-associated proteins like connexin-43 and oxytocin receptors, while enhancing the expression of relaxin and other quiescence-promoting factors in the uterine smooth muscle. This inhibitory effect is particularly critical in the first trimester, where progesterone maintains low vascular tone and prevents spontaneous contractions until the placenta assumes hormone production. The balance between progesterone and estrogen further ensures uterine quiescence, with progesterone dominating to override estrogen's potential stimulatory effects on contractility.[3][27][28] Progesterone also regulates ovulation timing by providing negative feedback on the hypothalamic-pituitary axis post-ovulation, suppressing further luteinizing hormone (LH) surges to prevent multiple ovulations in a single cycle. Secreted by the corpus luteum, it reduces GnRH pulse frequency and inhibits LH release from the pituitary, thereby supporting the luteal phase and sustaining endometrial receptivity for up to 14 days if implantation occurs. This feedback mechanism is vital for corpus luteum maintenance in early pregnancy, where human chorionic gonadotropin (hCG) from the embryo prolongs its function until placental progesterone production takes over.[11][3][29] Low levels of progesterone in women commonly cause irregular menstrual periods, difficulty conceiving, and abnormal uterine bleeding, reflecting disruptions in cycle regulation, endometrial preparation, and implantation support.[30][4] In males, progesterone modulates prostate function and spermatogenesis through local synthesis in testicular and prostatic tissues. Within the prostate and male reproductive tract, it activates progesterone receptors to oppose estrogen effects, influencing epithelial and stromal cell proliferation and potentially exerting a protective role against hyperplasia by counteracting androgen-driven growth. Progesterone also affects spermatogenesis by regulating sperm capacitation, acrosome reaction, and motility, with intratesticular levels promoting germ cell maturation while high exogenous levels can suppress gonadotropin-driven sperm production. These effects highlight progesterone's broader involvement in male reproductive physiology, including modulation of Leydig cell steroidogenesis.[31][32][33][34][35]Breasts
Progesterone, in conjunction with estrogen, plays a key role in promoting ductal morphogenesis during puberty and early reproductive life, facilitating the elongation and branching of mammary ducts to establish the foundational architecture of breast tissue. Estrogen primarily drives the initial ductal elongation, while progesterone induces side-branching and further morphogenesis through paracrine signaling mechanisms involving factors such as amphiregulin and Wnt4, which stimulate epithelial cell proliferation and invasion into the surrounding stroma.[36][37] During pregnancy, progesterone is essential for the stimulation of lobuloalveolar development, transforming the ductal network into a structure capable of milk production by promoting the proliferation and differentiation of alveolar epithelial cells. This process involves progesterone receptor-mediated signaling that coordinates with prolactin and other hormones to induce alveolar budding and secretory differentiation, ensuring the mammary gland's readiness for lactation.[38][39][40] Prolonged exposure to progesterone, particularly in combined estrogen-progestin hormone replacement therapy (HRT), is associated with an increased risk of breast cancer, primarily through mechanisms that enhance cell proliferation and disrupt normal mammary gland homeostasis. Synthetic progestins in HRT can activate progesterone receptors to promote the expansion of stem and progenitor cells in breast tissue, leading to higher incidence rates compared to estrogen-only therapy, with risks escalating with duration of use beyond five years.[41][42][43][44] In contrast, endogenous progesterone exposure through full-term pregnancies (parity) exerts protective effects against breast cancer by inducing terminal differentiation of mammary epithelial cells, which reduces the proliferative potential of stem and progenitor populations. This differentiation, mediated by progesterone signaling during pregnancy, alters gene expression profiles to favor a more mature, less susceptible epithelial state, thereby lowering lifetime risk, especially for estrogen receptor-positive tumors.[45][46][47]Skin health
Progesterone modulates sebum production in the skin, with mixed effects stemming from its partial anti-androgenic properties, including inhibition of 5α-reductase, which reduces the conversion of testosterone to the more potent dihydrotestosterone (DHT), a key stimulator of sebaceous gland activity.[48] However, elevated progesterone levels overall stimulate sebaceous glands, increasing sebum secretion and contributing to conditions like acne vulgaris, particularly during the luteal phase of the menstrual cycle when hyperseborrhea can exacerbate inflammation and comedone formation.[49] Regarding collagen synthesis and wound healing, progesterone influences dermal fibroblasts, often in synergy with estrogen, to promote extracellular matrix remodeling. In ovariectomized models, sequential administration of estradiol followed by progesterone enhances collagen biosynthesis and increases fibroblast estrogen receptor expression in abdominal skin, supporting tissue repair processes.[50] Progesterone also stimulates keratinocyte migration in vitro, facilitating epithelialization during wound closure and aiding in the restoration of skin integrity without excessive scarring.[51] However, in postmenopausal contexts, combined estrogen-progestin therapy does not significantly alter skin collagen content or synthesis rates compared to estrogen alone, suggesting a modulatory rather than stimulatory role for progesterone.[52] Progesterone plays a notable role in melasma and hyperpigmentation, particularly during pregnancy where it is termed chloasma gravidarum. Elevated progesterone levels in the third trimester, alongside estrogen and melanocyte-stimulating hormone, heighten melanocyte activity and tyrosinase expression, leading to symmetric hyperpigmented patches on sun-exposed areas like the face.[53] Postmenopausal women receiving progesterone supplementation develop melasma more frequently than those on estrogen alone, underscoring progesterone's direct stimulatory effect on melanin production independent of pregnancy.[54] Progesterone exerts anti-inflammatory effects that bolster skin barrier function by binding to progesterone receptors on keratinocytes and immune cells, thereby suppressing pro-inflammatory cytokine release such as TNF-α and modulating immune responses.[55] This activity helps maintain epidermal integrity, reduces transepidermal water loss, and prevents barrier disruption in inflammatory dermatoses, contributing to overall skin homeostasis during hormonal fluctuations.[56]Sexuality
Progesterone plays a significant role in modulating sexual desire and behavior across the menstrual cycle in women. During the follicular phase, rising estradiol levels are associated with increased sexual motivation, while the subsequent elevation in progesterone during the luteal phase correlates with a decline in subjective sexual desire.[57] This negative effect of progesterone on sexual motivation is thought to reflect an adaptive mechanism to reduce mating efforts when conception is unlikely, as supported by evolutionary models of hormonal influences on female sexuality.[58] Human studies tracking daily hormone levels and self-reported desire over multiple cycles confirm that progesterone mediates the post-ovulatory drop in libido, with no significant between-women differences attributable to baseline hormone profiles.[57] Additionally, peri-ovulatory increases in sexual activity align with low progesterone levels, further highlighting its inhibitory role during the luteal phase.[59] In pregnancy, elevated progesterone levels contribute to suppressed sexual desire, particularly as concentrations rise progressively across trimesters. This suppression is evident in reduced libido reported by many women, potentially due to progesterone's counteraction of estrogen's pro-sexual effects and its promotion of offspring-focused attention over sexual stimuli.[60][61] Central nervous system effects underlie this modulation, as progesterone influences neural circuits involved in reward and motivation, shifting behavioral priorities toward gestation maintenance.[61] Postpartum progesterone withdrawal can paradoxically delay libido recovery despite its overall inhibitory influence during pregnancy.[61] Progesterone modulates sexual receptivity primarily through its receptors in the hypothalamus, particularly in the ventromedial nucleus (VMH), as demonstrated in rodent models. In estradiol-primed female rats, progesterone binding to hypothalamic receptors facilitates lordosis and other receptive behaviors via both genomic (nuclear receptor-mediated gene transcription) and nongenomic (membrane-initiated rapid signaling) pathways.[62] Activation of progesterone receptor-expressing neurons in the VMH is required for the expression of female sexual behaviors, with periodic remodeling of these circuits timing receptivity to the estrous cycle.[63] These mechanisms highlight the hypothalamus as a key site for progesterone's central regulation of sexual responsiveness. Animal models, such as domestic sheep, reveal correlations between prenatal hormonal influences and sexual orientation. Approximately 8% of rams exhibit exclusive male-oriented preferences, associated with female-like features in the ovine sexually dimorphic nucleus (oSDN) of the hypothalamus, which develops under prenatal hormonal influences, primarily androgens.[64] These findings from sheep models provide insights into potential hormonal contributions to sexual orientation across species.[65]Nervous system
Progesterone exerts neuromodulatory effects in the nervous system primarily through its metabolite allopregnanolone, which acts as a positive allosteric modulator of GABA_A receptors. This interaction enhances inhibitory neurotransmission, contributing to anxiolytic effects by reducing neuronal excitability and promoting a calming influence on brain activity.[66] Clinical and preclinical studies have demonstrated that elevated allopregnanolone levels, derived from progesterone, correlate with decreased anxiety behaviors in various models.[67] Progesterone also supports myelination and neuronal survival, essential processes for maintaining neural integrity. In the peripheral nervous system, progesterone stimulates Schwann cells to form myelin sheaths around axons, enhancing the rate of myelin formation in cocultures of neurons and glial cells.[68] Furthermore, progesterone and its derivatives promote neuronal viability in the central and peripheral nervous systems, protecting against degeneration by modulating anti-apoptotic pathways and reducing oxidative stress.[69] Recent research highlights the synergistic neuroprotective roles of estrogen and progesterone in aging and Alzheimer's disease. A 2024 study in animal models of Alzheimer's showed that progesterone administration improved cognitive performance and reduced neuroinflammation, with interactions with estrogen's neuroprotection being mixed—some studies showing enhancement and others indicating potential antagonism. In males, progesterone activates progesterone receptors in the brain to oppose estrogen's regulatory actions, such as by down-regulating estrogen receptors in regions like the periventricular preoptic area. This suggests potential therapeutic applications for hormone combinations in mitigating age-related cognitive decline. In human studies, associations between endogenous progesterone levels and cognitive performance are inconsistent; positive associations with verbal memory and global cognition have been observed in early postmenopausal women (within 6 years of menopause), but findings are null in older postmenopausal women, with no consistent evidence of clinically important effects overall. No direct associations between progesterone and intelligence quotient (IQ) in adults have been established.[70][71] Progesterone influences sleep-wake cycles and mood stabilization via its sedative properties mediated by allopregnanolone's GABA_A modulation. Administration of progesterone reduces wakefulness during sleep EEG recordings in postmenopausal women, increasing total sleep time without impairing cognitive function.[72] These effects contribute to mood stabilization by alleviating symptoms of anxiety and irritability, particularly during hormonal fluctuations in the menstrual cycle.[73] Conversely, low progesterone levels in women are associated with mood changes such as anxiety or depression, as well as headaches.[30]Brain damage
Progesterone exhibits neuroprotective properties in models of traumatic brain injury (TBI), primarily through the reduction of cerebral edema and inflammation. In rodent studies, administration of progesterone following TBI significantly decreases brain swelling by modulating aquaporin expression and fluid dynamics, thereby limiting secondary tissue damage.[74] Similarly, progesterone attenuates neuroinflammatory responses by suppressing pro-inflammatory cytokine production, such as interleukin-1β and tumor necrosis factor-α, in both male and female subjects.[75] These effects have been consistently observed across multiple experimental paradigms, including fluid percussion and controlled cortical impact models, highlighting progesterone's potential to mitigate the acute consequences of brain trauma.[76] The proposed mechanisms underlying these protective actions include antioxidant activity, inhibition of apoptosis, and stabilization of the blood-brain barrier (BBB). Progesterone's antioxidant effects involve scavenging reactive oxygen species and upregulating endogenous antioxidants like superoxide dismutase, which counteract oxidative stress post-injury.[77] It also inhibits apoptotic pathways by modulating Bcl-2 family proteins and caspase-3 activation, preserving neuronal viability.[78] Additionally, progesterone reinforces BBB integrity by enhancing tight junction proteins such as occludin and claudin-5, preventing leakage and further edema formation.[74] Clinical trials evaluating progesterone for TBI and stroke have yielded mixed results up to 2023, with preclinical promise not fully translating to human outcomes. Phase II trials, such as the Progesterone for Traumatic Brain Injury (ProTECT) studies, suggested improved functional recovery and reduced mortality, but larger phase III trials like ProTECT III and SYNAPSE reported no significant benefits over placebo in reducing mortality or improving neurological outcomes.[79] [80] Ongoing preclinical and early-phase studies as of 2025 continue to investigate progesterone's role in ischemic neuroprotection, focusing on optimized dosing and timing to enhance efficacy.[81] Gender differences in progesterone's efficacy may arise from variations in endogenous hormone levels, with females often showing greater neuroprotection due to higher baseline progesterone concentrations during reproductive cycles. In animal models, exogenous progesterone provides more pronounced benefits in males, who have lower endogenous levels, compared to females where it may interact with fluctuating ovarian hormones.[82] These disparities underscore the need for sex-specific considerations in therapeutic applications.[83]Addiction
Progesterone modulates dopamine release in the nucleus accumbens through its receptors, thereby influencing reward processing and potentially reducing the reinforcing effects of addictive substances.[84] In animal models, progesterone and its metabolites have been shown to attenuate self-administration of nicotine, alcohol, and cocaine. For instance, progesterone administration reduces nicotine intake and cocaine-seeking behavior in female rats, while its metabolite allopregnanolone decreases ethanol self-administration in male rats by enhancing GABAergic inhibition in reward pathways.[85][86][87] In women, vulnerability to addiction relapse exhibits cycle-dependent patterns, with increased risk during the late luteal phase when progesterone levels are declining. This phase is associated with heightened craving and withdrawal symptoms for substances like cocaine and nicotine, potentially due to reduced progesterone-mediated dampening of dopaminergic activity.[88][89] Progesterone interacts with opioid systems to modulate pain perception and reward, often through its neuroactive metabolite allopregnanolone, which enhances mu-opioid receptor signaling and reduces opioid-induced reward in preclinical studies. These interactions may contribute to progesterone's protective effects against substance dependence by altering pain-reward balance.[90][91]Other effects
Progesterone exerts significant immunomodulatory effects, particularly in promoting maternal-fetal tolerance during pregnancy by suppressing proinflammatory T-cell activity. It inhibits the proliferation and cytokine production of Th1 and Th17 cells while enhancing regulatory T cells (Tregs), which dampen immune responses at the maternal-fetal interface to prevent rejection of the fetus.[92] This suppression of T-cell activation is mediated through progesterone receptors on immune cells, leading to reduced interferon-gamma and interleukin-17 secretion, thereby fostering an anti-inflammatory environment essential for successful gestation.[93] Additionally, progesterone promotes the expansion of decidual Tregs, further contributing to immune tolerance by limiting cytotoxic T-cell responses.[94] In skeletal health, progesterone supports bone density maintenance by stimulating osteoblast activity and differentiation. It binds to progesterone receptors on osteoblasts, promoting their proliferation and enhancing bone formation through increased expression of osteogenic factors such as alkaline phosphatase and osteocalcin.[95] This action counters bone resorption, particularly in premenopausal women, and has been shown to increase bone mineral density in trabecular sites like the spine when progesterone levels are adequate.[96] Studies indicate that progesterone's osteoanabolic effects help mitigate the risk of osteoporosis by favoring osteoblast-mediated matrix deposition over adipogenesis in mesenchymal stem cells. Progesterone influences cardiovascular function through vasodilatory and anti-atherogenic mechanisms. It stimulates endothelial nitric oxide synthase (eNOS) expression, leading to nitric oxide production that induces vasodilation and improves vascular relaxation, particularly in coronary and peripheral arteries.[97] Furthermore, progesterone exhibits anti-atherogenic properties by reducing low-density lipoprotein (LDL) oxidation, decreasing lipid accumulation in arterial walls, and modulating inflammatory responses in the endothelium to inhibit plaque formation.[97] These effects contribute to overall vascular protection, though they may vary in combination with estrogen. Recent research has highlighted progesterone's role in facilitating cancer immune evasion within tumor microenvironments, drawing parallels to its immunosuppressive functions in pregnancy. In breast cancer models, progesterone receptor signaling downregulates major histocompatibility complex class I (MHC-I) expression on tumor cells, impairing CD8+ T-cell recognition and promoting immune escape.[98] Similarly, progestogens upregulate B7-H4, an immune checkpoint ligand, in ovarian and endometrial tumors, which suppresses antitumor T-cell activity and enhances tumor progression by mimicking pregnancy-associated tolerance.00652-4) A 2024 study further demonstrated that progesterone reprograms the tumor microenvironment to share immunosuppressive features with the fetoplacental unit, including elevated Treg infiltration and reduced natural killer cell cytotoxicity, thereby fostering therapy resistance.[99]Biochemistry
Biosynthesis
Progesterone is synthesized endogenously through the steroidogenesis pathway, beginning with cholesterol as the precursor substrate. The initial and rate-limiting step involves the transport of cholesterol into the mitochondria, facilitated by the steroidogenic acute regulatory protein (StAR), followed by its conversion to pregnenolone by the enzyme cytochrome P450 side-chain cleavage enzyme (CYP11A1). This cleavage removes the side chain from cholesterol, yielding pregnenolone. Subsequently, pregnenolone is transformed into progesterone by the enzyme 3β-hydroxysteroid dehydrogenase (3β-HSD), which oxidizes the 3β-hydroxyl group to a keto group while reducing NAD⁺ to NADH.[3][100][101] The biochemical reaction catalyzed by 3β-HSD can be represented as: This step occurs primarily in the endoplasmic reticulum and is essential for progesterone production across steroidogenic tissues. Multiple isoforms of 3β-HSD exist, with 3β-HSD2 being predominant in the adrenals, ovaries, and placenta.[100][3] The primary sites of progesterone biosynthesis are the corpus luteum in the ovaries, the placenta during pregnancy, and the adrenal cortex. In non-pregnant individuals, the corpus luteum, formed post-ovulation, serves as the main source during the luteal phase of the menstrual cycle, producing up to 25 mg of progesterone daily. The adrenal cortex contributes smaller amounts, approximately 1-2 mg per day, serving as a precursor for other steroids. During pregnancy, the placenta becomes the dominant site after approximately 10 weeks, synthesizing large quantities to maintain gestation, with production reaching 250-500 mg per day by term.[102][3] Biosynthesis is tightly regulated by hormonal signals. In the corpus luteum, luteinizing hormone (LH) from the anterior pituitary stimulates progesterone production by activating the cAMP-protein kinase A pathway, which upregulates StAR and key enzymes like CYP11A1 and 3β-HSD, ensuring peak output during the mid-luteal phase. During pregnancy, placental progesterone synthesis shifts to largely autocrine regulation independent of maternal or fetal endocrine inputs, though initial support comes from human chorionic gonadotropin (hCG) and local placental factors such as corticotropin-releasing hormone (CRH) and estradiol, which modulate enzyme expression to sustain elevated levels.[103][104][3]Distribution
Progesterone circulates in the plasma bound to carrier proteins, which facilitate its transport while regulating its bioavailability. Approximately 80% of circulating progesterone is bound to serum albumin with low affinity, 15-20% binds to corticosteroid-binding globulin (CBG) with high affinity, less than 1% to sex hormone-binding globulin (SHBG), and the remaining 1–2% exists in the unbound, free form that is biologically active.[105][106] This binding distribution helps maintain stable hormone levels and prevents rapid clearance, with the free fraction available for diffusion into tissues. Due to its inherent lipophilicity as a steroid hormone, progesterone readily diffuses across lipid bilayers of cell membranes via passive transport, independent of specific carriers or energy-dependent mechanisms.[107] This property enables progesterone to penetrate various barriers, including the blood-brain barrier, where it can influence neuronal function directly.[70] Similarly, during pregnancy, progesterone crosses the placental barrier by diffusion, contributing to fetal exposure, though the extent of maternal-to-fetal transfer is limited to about 1% of circulating maternal levels as the placenta increasingly synthesizes its own progesterone.[108] Tissue-specific concentrations of progesterone fluctuate in response to physiological demands, particularly across the menstrual cycle. In the uterus, for instance, myometrial progesterone levels are markedly elevated during the luteal phase (ranging from 2.06 to 14.85 ng/g wet weight) compared to the follicular phase, reflecting targeted accumulation to support endometrial preparation for implantation.[109] These variations underscore progesterone's role in localized signaling at reproductive target sites.Metabolism
Progesterone undergoes rapid metabolism primarily in the liver, where it is transformed into various inactive and active metabolites to facilitate its elimination from the body.[5] The biological half-life of unbound progesterone in circulation is approximately 5 minutes, reflecting its swift hepatic clearance and limiting its persistence in blood.[105] This rapid turnover ensures that progesterone's physiological effects are tightly regulated, with over 90% of the hormone metabolized during the first pass through the liver.[6] A key step in progesterone's catabolism involves hepatic reduction of its Δ4-3-keto structure by 5α-reductases and 5β-reductases, leading to the formation of 5α-dihydroprogesterone and 5β-dihydroprogesterone, respectively.[110] These intermediates are further reduced, primarily via 3α-hydroxysteroid dehydrogenase activity, to yield pregnanediol (5β-pregnane-3α,20α-diol), a major urinary metabolite that accounts for about 12% of progesterone's metabolic products.[111] Among the notable metabolites are allopregnanolone (3α-hydroxy-5α-pregnan-20-one) and pregnanolone (3α-hydroxy-5β-pregnan-20-one), which are neuroactive neurosteroids derived from the respective dihydroprogesterones and exhibit potent modulatory effects on GABA_A receptors in the brain.[112][100] For excretion, progesterone metabolites such as pregnanediol are conjugated primarily with glucuronic acid in the liver, forming water-soluble glucuronides that are efficiently eliminated via the urine, accounting for roughly 80% of total steroid excretion.[113] This conjugation process enhances renal clearance and prevents reabsorption in the intestines, ensuring the complete removal of progesterone-derived compounds from the body.[114]Levels
Progesterone concentrations in the blood vary significantly across the menstrual cycle, pregnancy, and other physiological states, providing key insights into reproductive health. In non-pregnant women, serum progesterone levels are typically low during the follicular phase, ranging from 0.1 to 1.5 ng/mL, reflecting minimal ovarian production before ovulation.[3] Following ovulation, levels rise sharply in the luteal phase to 2 to 25 ng/mL, driven by the corpus luteum, and remain elevated for about 10 to 14 days before declining if pregnancy does not occur.[3] In men and postmenopausal women, baseline levels are consistently low, generally below 1 ng/mL.[115] During pregnancy, progesterone levels increase dramatically to support implantation and fetal development, starting at 10 to 44 ng/mL in the first trimester and progressively rising to 65 to 290 ng/mL by the third trimester, with peaks often observed around 32 weeks.[3] This escalation is essential for maintaining uterine quiescence and preventing miscarriage.[3] Progesterone exhibits diurnal variations, with concentrations showing a circadian rhythm: levels are lowest in the early morning (nadir around 8:00 a.m.) and peak toward midnight, particularly pronounced in the luteal phase and pregnancy.[116] Age-related changes include a gradual decline post-reproduction, with postmenopausal levels stabilizing at under 0.2 to 1 ng/mL due to ovarian senescence.[117] These variations can be influenced by factors such as plasma protein binding, as detailed in the distribution section. Serum progesterone is commonly measured using immunoassays, such as enzyme-linked immunosorbent assays (ELISA) or chemiluminescent immunoassays, which are rapid but may suffer from cross-reactivity and variability.[118] For higher accuracy and specificity, liquid chromatography-tandem mass spectrometry (LC-MS/MS) serves as the reference method, particularly useful in clinical research and when immunoassay results are ambiguous.[118] Clinically, progesterone levels hold diagnostic value: in the mid-luteal phase, concentrations above 5 ng/mL confirm ovulation, while levels below 3 ng/mL suggest anovulation or luteal phase deficiency, often indicating infertility risks or conditions like polycystic ovary syndrome.[119] Elevated levels beyond normal ranges may signal ectopic pregnancy or molar disease, whereas low levels in early pregnancy (<10 ng/mL) are associated with increased miscarriage risk.[3]| Physiological State | Typical Serum Progesterone Range (ng/mL) | Source |
|---|---|---|
| Follicular Phase | 0.1–1.5 | NCBI StatPearls |
| Luteal Phase | 2–25 | NCBI StatPearls |
| First Trimester Pregnancy | 10–44 | Healthline |
| Third Trimester Pregnancy | 65–290 | Healthline |
| Postmenopausal | <1 | Medscape |


