Recent from talks
Nothing was collected or created yet.
 Small balls of white tin (left) and gray tin (right) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tin | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Allotropes | silvery-white, β (beta); gray, α (alpha) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Standard atomic weight Ar°(Sn) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tin in the periodic table | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic number (Z) | 50 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group | group 14 (carbon group) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Period | period 5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Block | p-block | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Kr] 4d10 5s2 5p2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2, 8, 18, 18, 4 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase at STP | solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | 505.08 K (231.93 °C, 449.47 °F) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | 2875 K (2602 °C, 4716 °F) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density (at 20° C) | white (β): 7.289 g/cm3 gray (α): 5.770 g/cm3[3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| when liquid (at m.p.) | 6.99 g/cm3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of fusion | white (β): 7.03 kJ/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of vaporization | white (β): 296.1 kJ/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar heat capacity | white (β): 27.112 J/(mol·K) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Vapor pressure
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Oxidation states | common: −4, +2, +4 −3,[4] −2,[5] −1,[6] 0,[7] +1,[8] +3[9] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electronegativity | Pauling scale: 1.96 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ionization energies |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic radius | empirical: 140 pm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Covalent radius | 139±4 pm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Van der Waals radius | 217 pm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Natural occurrence | primordial | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
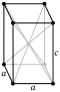
| Crystal structure | white (β): body-centered tetragonal (tI4) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lattice constants | white (β): a = 583.13 pm c = 318.11 pm (at 20 °C)[3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal structure | gray (α): face-centered diamond-cubic (cF8) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lattice constant | gray (α): a = 648.96 pm (at 20 °C)[3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal expansion | white (β): 21.76×10−6/K (at 20 °C)[a] gray (α): 5.20×10−6/K (at 20 °C)[3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal conductivity | 66.8 W/(m⋅K) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrical resistivity | 115 nΩ⋅m (at 0 °C) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Magnetic ordering | white (β): paramagnetic gray (α): diamagnetic[10] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar magnetic susceptibility | white (β): +3.1×10−6 cm3/mol (298 K)[11] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Young's modulus | 50 GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Shear modulus | 18 GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bulk modulus | 58 GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Speed of sound thin rod | 2730 m/s (at r.t.) (rolled) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Poisson ratio | 0.36 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mohs hardness | 1.5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Brinell hardness | 50–440 MPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Number | 7440-31-5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Naming | a Proto-Germanic word | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Discovery | protohistoric, around 35th century BC | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symbol | "Sn": from Latin stannum | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes of tin | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Tin is a chemical element; it has the symbol Sn (from Latin stannum) and atomic number 50. A metallic-gray metal, tin is soft enough to be cut with little force,[13] and a bar of tin can be bent by hand with little effort. When bent, a bar of tin makes a sound, the so-called "tin cry", as a result of twinning in tin crystals.[14]
Tin is a post-transition metal in group 14 of the periodic table of elements. It is obtained chiefly from the mineral cassiterite, which contains stannic oxide, SnO
2. Tin shows a chemical similarity to both of its neighbors in group 14, germanium and lead, and has two main oxidation states, +2 and the slightly more stable +4. Tin is the 49th most abundant element on Earth, making up 0.00022% of its crust, and with 10 stable isotopes, it has the largest number of stable isotopes in the periodic table, due to its magic number of protons.
It has two main allotropes: at room temperature, the stable allotrope is β-tin, a silvery-white, malleable metal; at low temperatures it is less dense grey α-tin, which has the diamond cubic structure. Metallic tin does not easily oxidize in air and water.
The first tin alloy used on a large scale was bronze, made of 1⁄8 tin and 7⁄8 copper (12.5% and 87.5% respectively), from as early as 3000 BC. After 600 BC, pure metallic tin was produced. Pewter, which is an alloy of 85–90% tin with the remainder commonly consisting of copper, antimony, bismuth, and sometimes lead and silver, has been used for flatware since the Bronze Age. In modern times, tin is used in many alloys, most notably tin-lead soft solders, which are typically 60% or more tin, and in the manufacture of transparent, electrically conducting films of indium tin oxide in optoelectronic applications. Another large application is corrosion-resistant tin plating of steel. Because of the low toxicity of inorganic tin, tin-plated steel is widely used for food packaging as "tin cans". Some organotin compounds can be extremely toxic.
Characteristics
[edit]Physical
[edit]
Tin is a soft, malleable, ductile and highly crystalline silvery-white metal. When a bar of tin is bent a crackling sound known as the "tin cry" can be heard from the twinning of the crystals.[14] This trait is shared by indium, cadmium, zinc, and mercury in its solid state. Tin melts at about 232 °C (450 °F), the lowest in group 14, and boils at 2,602 °C (4,716 °F), the second lowest (ahead of lead) in its group. The melting point is further lowered to 177.3 °C (351.1 °F) for 11 nm particles.[15][16]
| External videos | |
|---|---|
β-tin, also called white tin, is the allotrope (structural form) of elemental tin that is stable at and above room temperature. It is metallic and malleable, and has body-centered tetragonal crystal structure. α-tin, or gray tin, is the nonmetallic form. It is stable below 13.2 °C (55.8 °F) and is brittle. α-tin has a diamond cubic crystal structure, as do diamond and silicon. α-tin does not have metallic properties because its atoms form a covalent structure in which electrons cannot move freely. α-tin is a dull-gray powdery material with no common uses other than specialized semiconductor applications.[14] γ-tin and σ-tin exist at temperatures above 161 °C (322 °F) and pressures above several GPa.[17]
In cold conditions β-tin tends to transform spontaneously into α-tin, a phenomenon known as "tin pest" or "tin disease".[18] Some unverifiable sources also say that, during Napoleon's Russian campaign of 1812, the temperatures became so cold that the tin buttons on the soldiers' uniforms disintegrated over time, contributing to the defeat of the Grande Armée,[19] a persistent legend.[20][21][22]
The α-β transformation temperature is 13.2 °C (55.8 °F), but impurities (e.g. Al, Zn, etc.) lower it well below 0 °C (32 °F). With the addition of antimony or bismuth the transformation might not occur at all, increasing durability.[23]
Commercial grades of tin (99.8% tin content) resist transformation because of the inhibiting effect of small amounts of bismuth, antimony, lead, and silver present as impurities. Alloying elements such as copper, antimony, bismuth, cadmium, and silver increase the hardness of tin.[24] Tin easily forms hard, brittle intermetallic phases that are typically undesirable. It does not mix into a solution with most metals and elements so tin does not have much solid solubility. Tin mixes well with bismuth, gallium, lead, thallium and zinc, forming simple eutectic systems.[23]
Tin becomes a superconductor below 3.72 K[25] and was one of the first superconductors to be studied.[26] The Meissner effect, one of the characteristic features of superconductors, was first discovered in superconducting tin crystals.[26]
Chemical
[edit]Tin resists corrosion from water, but can be corroded by acids and alkalis. Tin can be highly polished and is used as a protective coat for other metals.[14] When heated in air it oxidizes slowly to form a thin passivation layer of stannic oxide (SnO2) that inhibits further oxidation.[27][28]
Isotopes
[edit]Tin has ten stable isotopes, the greatest number of any element. Their mass numbers are 112, 114, 115, 116, 117, 118, 119, 120, 122, and 124. Tin-120 makes up almost a third of all tin. Tin-118 and tin-116 are also common. Tin-115 is the least common stable isotope.[29] The isotopes with even mass numbers have no nuclear spin, while those with odd mass numbers have a nuclear spin of 1/2. It is thought that tin has such a great multitude of stable isotopes because of tin's atomic number being 50, which is a "magic number" in nuclear physics.[30][31]
Tin is one of the easiest elements to detect and analyze by NMR spectroscopy, which relies on molecular weight and its chemical shifts are referenced against tetramethyltin (SnMe
4).[b][32]
Of the stable isotopes, tin-115 has a high neutron capture cross section for thermal neutrons, at 30 barns. Tin-117 has a cross section of 2.3 barns, one order of magnitude smaller, while tin-119 has a slightly smaller cross section of 2.2 barns.[33] Before these cross sections were well known, it was proposed to use tin-lead solder as a coolant for fast reactors because of its low melting point. Current studies are for lead or lead-bismuth reactor coolants because both heavy metals are nearly transparent to fast neutrons, with very low capture cross sections.[34] In order to use a tin or tin-lead coolant, the tin would first have to go through isotopic separation to remove the isotopes with odd mass number. Combined, these three isotopes make up about 17% of natural tin but represent nearly all of the capture cross section. Of the remaining seven isotopes tin-112 has a capture cross section of 1 barn. The other six isotopes forming 82.7% of natural tin have capture cross sections of 0.3 barns or less, making them effectively transparent to neutrons.[33]
Tin has 33 unstable isotopes, ranging in mass number from 98 to 140. The unstable tin isotopes have half-lives of less than a year except for tin-126, which has a half-life of about 230,000 years. Tin-100 and tin-132 are two of the very few nuclides with a "doubly magic" nucleus; though they are unstable, being far off the valley of stability in neutron–proton ratio, the tin isotopes lighter than tin-100 and heavier than tin-132 are even less stable.[35] Another 55 metastable isomers have been identified for tin isotopes between 111 and 131, the most stable being tin-121m, with a half-life of 43.9 years.[36]
Etymology
[edit]The word tin is shared among Germanic languages and can be traced back to reconstructed Proto-Germanic *tin-om; cognates include German Zinn, Swedish tenn and Dutch tin. It is not found in other branches of Indo-European, except by borrowing from Germanic (e.g., Irish tinne from English).[37][38]
The Latin name for tin, stannum, originally meant an alloy of silver and lead, and came to mean 'tin' in the fourth century[39]—the earlier Latin word for it was plumbum candidum, or "white lead". Stannum apparently came from an earlier stāgnum (meaning the same substance),[37] the origin of the Romance and Celtic terms for tin, such as French étain, Spanish estaño, Italian stagno, and Irish stán.[37][40] The origin of stannum/stāgnum is unknown; it may be pre-Indo-European.[41]
The Meyers Konversations-Lexikon suggests instead that stannum came from Cornish stean, and is evidence that Cornwall in the first centuries AD was the main source of tin.[citation needed]
History
[edit]
Tin extraction and use can be dated to the beginnings of the Bronze Age around 3000 BC, when it was observed that copper objects formed of polymetallic ores with different metal contents had different physical properties.[42] The earliest bronze objects had a tin or arsenic content of less than 2% and are believed to be the result of unintentional alloying due to trace metal content in the copper ore.[43] The addition of a second metal to copper increases its hardness, lowers the melting temperature, and improves the casting process by producing a more fluid melt that cools to a denser, less spongy metal.[43] This was an important innovation that allowed for the much more complex shapes cast in closed molds of the Bronze Age. Arsenical bronze objects appear first in the Near East where arsenic is commonly found with copper ore, but the health risks were quickly realized and the quest for sources of the much less hazardous tin ores began early in the Bronze Age.[44] This created the demand for rare tin metal and formed a trade network that linked the distant sources of tin to the markets of Bronze Age cultures.[45]
Cassiterite (SnO
2), the oxide form of tin, was most likely the original source of tin. Other tin ores are less common sulfides such as stannite that require a more involved smelting process. Cassiterite often accumulates in alluvial channels as placer deposits because it is harder, heavier, and more chemically resistant than the accompanying granite.[43] Cassiterite is usually black or dark in color, and these deposits can be easily seen in river banks. Alluvial (placer) deposits may incidentally have been collected and separated by methods similar to gold panning.[46]
Compounds and chemistry
[edit]In the great majority of its compounds, tin has the oxidation state II or IV. Compounds containing bivalent tin are called stannous while those containing tetravalent tin are termed stannic.
Inorganic compounds
[edit]Halide compounds are known for both oxidation states. For Sn(IV), all four halides are well known: SnF4, SnCl4, SnBr4, and SnI4. The three heavier members are volatile molecular compounds, whereas the tetrafluoride is polymeric. All four halides are known for Sn(II) also: SnF2, SnCl
2, SnBr2, and SnI2. All are polymeric solids. Of these eight compounds, only the iodides are colored.[47]
Tin(II) chloride (also known as stannous chloride) is the most important commercial tin halide. Illustrating the routes to such compounds, chlorine reacts with tin metal to give SnCl4 whereas the reaction of hydrochloric acid and tin produces SnCl
2 and hydrogen gas. Alternatively SnCl4 and Sn combine to stannous chloride by a process called comproportionation:[48]
- SnCl4 + Sn → 2 SnCl
2
Tin can form many oxides, sulfides, and other chalcogenide derivatives. The dioxide SnO
2 (cassiterite) forms when tin is heated in the presence of air.[47] SnO
2 is amphoteric, which means that it dissolves in both acidic and basic solutions.[49] Stannates with the structure [Sn(OH)
6]2−, like K
2[Sn(OH)
6], are also known, though the free stannic acid H
2[Sn(OH)
6] is unknown.[citation needed]
Sulfides of tin exist in both the +2 and +4 oxidation states: tin(II) sulfide and tin(IV) sulfide (mosaic gold).

2)[50]
Hydrides
[edit]Stannane (SnH
4), with tin in the +4 oxidation state, is unstable. Organotin hydrides are however well known, e.g. tributyltin hydride (Sn(C4H9)3H).[14] These compounds release transient tributyl tin radicals, which are rare examples of compounds of tin(III).[51]
Organotin compounds
[edit]Organotin compounds, sometimes called stannanes, are chemical compounds with tin–carbon bonds.[52] Of the tin compounds, the organic derivatives are commercially the most useful.[53] Some organotin compounds are highly toxic and have been used as biocides. The first organotin compound to be reported was diethyltin diiodide ((C2H5)2SnI2), reported by Edward Frankland in 1849.[54]
Most organotin compounds are colorless liquids or solids that are stable to air and water. They adopt tetrahedral geometry. Tetraalkyl- and tetraaryltin compounds can be prepared using Grignard reagents:[53]
- SnCl
4 + 4 RMgBr → R
4Sn + 4 MgBrCl
The mixed halide-alkyls, which are more common and more important commercially than the tetraorgano derivatives, are prepared by redistribution reactions:
- SnCl
4 + R
4Sn → 2 SnCl
2R2
Divalent organotin compounds are uncommon, although more common than related divalent organogermanium and organosilicon compounds. The greater stabilization enjoyed by Sn(II) is attributed to the "inert pair effect". Organotin(II) compounds include both stannylenes (formula: R2Sn, as seen for singlet carbenes) and distannylenes (R4Sn2), which are roughly equivalent to alkenes. Both classes exhibit unusual reactions.[55]
Occurrence
[edit]
Tin is generated via the long s-process in low-to-medium mass stars (with masses of 0.6 to 10 times that of the Sun), and finally by beta decay of the heavy isotopes of indium.[56]
Tin is the 49th most abundant element in Earth's crust, representing 2 ppm compared with 75 ppm for zinc, 50 ppm for copper, and 14 ppm for lead.[57]
Tin does not occur as the native element but must be extracted from various ores. Cassiterite (SnO
2) is the only commercially important source of tin, although small quantities of tin are recovered from complex sulfides such as stannite, cylindrite, franckeite, canfieldite, and teallite. Minerals with tin are almost always associated with granite rock, usually at a level of 1% tin oxide content.[58]
Because of the higher specific gravity of tin dioxide, about 80% of mined tin is from secondary deposits found downstream from the primary lodes. Tin is often recovered from granules washed downstream in the past and deposited in valleys or the sea. The most economical ways of mining tin are by dredging, hydraulicking, or open pits. Most of the world's tin is produced from placer deposits, which can contain as little as 0.015% tin.[59]
| Country | Reserves |
|---|---|
| 1,500,000 | |
| 250,000 | |
| 310,000 | |
| 800,000 | |
| 590,000 | |
| 400,000 | |
| 350,000 | |
| 180,000 | |
| 170,000 | |
| Other | 180,000 |
| Total | 4,800,000 |
| Year | Million tonnes |
|---|---|
| 1965 | 4,265 |
| 1970 | 3,930 |
| 1975 | 9,060 |
| 1980 | 9,100 |
| 1985 | 3,060 |
| 1990 | 7,100 |
| 2000 | 7,100[60] |
| 2010 | 5,200[60] |
About 253,000 tonnes of tin were mined in 2011, mostly in China (110,000 t), Indonesia (51,000 t), Peru (34,600 t), Bolivia (20,700 t) and Brazil (12,000 t).[60] Estimates of tin production have historically varied with the market and mining technology. It is estimated that, at current consumption rates and technologies, the Earth will run out of mine-able tin in 40 years.[61] In 2006 Lester Brown suggested tin could run out within 20 years based on conservative estimates of 2% annual growth.[62]
Scrap tin is an important source of the metal. Recovery of tin through recycling is increasing rapidly as of 2019.[63] Whereas the United States has neither mined (since 1993) nor smelted (since 1989) tin, it was the largest secondary producer, recycling nearly 14,000 tonnes in 2006.[60]
New deposits are reported in Mongolia,[64] and in 2009, new deposits of tin were discovered in Colombia.[65]
Production
[edit]Tin is produced by carbothermic reduction of the oxide ore with carbon or coke. Both reverberatory furnace and electric furnace can be used:[66][67][68]
- SnO2 + C Sn + CO2↑
Mining and smelting
[edit]Industry
[edit]The ten largest tin-producing companies produced most of the world's tin in 2007.[citation needed]
Most of the world's tin is traded on LME, from 8 countries, under 17 brands.[69]
| Company | Polity | 2006 | 2007 | 2017[71] | 2006–2017 % change |
|---|---|---|---|---|---|
| Yunnan Tin | China | 52,339 | 61,129 | 74,500 | 42.3 |
| PT Timah | Indonesia | 44,689 | 58,325 | 30,200 | −32.4 |
| Malaysia Smelting Corp | Malaysia | 22,850 | 25,471 | 27,200 | 19.0 |
| Yunnan Chengfeng | China | 21,765 | 18,000 | 26,800 | 23.1 |
| Minsur | Peru | 40,977 | 35,940 | 18,000 | −56.1 |
| EM Vinto | Bolivia | 11,804 | 9,448 | 12,600 | 6.7 |
| Guangxi China Tin | China | / | / | 11,500 | / |
| Thaisarco | Thailand | 27,828 | 19,826 | 10,600 | −61.9 |
| Metallo-Chimique | Belgium | 8,049 | 8,372 | 9,700 | 20.5 |
| Gejiu Zi Li | China | / | / | 8,700 | / |
The International Tin Council was established in 1947 to control the price of tin. It collapsed in 1985. In 1984, the Association of Tin Producing Countries was created, with Australia, Bolivia, Indonesia, Malaysia, Nigeria, Thailand, and Zaire as members.[72]
Price and exchanges
[edit]
Tin is unique among mineral commodities because of the complex agreements between producer countries and consumer countries dating back to 1921. Earlier agreements tended to be somewhat informal and led to the "First International Tin Agreement" in 1956, the first of a series that effectively collapsed in 1985. Through these agreements, the International Tin Council (ITC) had a considerable effect on tin prices. ITC supported the price of tin during periods of low prices by buying tin for its buffer stockpile and was able to restrain the price during periods of high prices by selling from the stockpile. This was an anti-free-market approach, designed to assure a sufficient flow of tin to consumer countries and a profit for producer countries. However, the buffer stockpile was not sufficiently large, and during most of those 29 years tin prices rose, sometimes sharply, especially from 1973 through 1980 when rampant inflation plagued many world economies.[73]
During the late 1970s and early 1980s, the U.S. reduced its strategic tin stockpile, partly to take advantage of historically high tin prices. The 1981–82 recession damaged the tin industry. Tin consumption declined dramatically. ITC was able to avoid truly steep declines through accelerated buying for its buffer stockpile; this activity required extensive borrowing. ITC continued to borrow until late 1985 when it reached its credit limit. Immediately, a major "tin crisis" ensued—tin was delisted from trading on the London Metal Exchange for about three years. ITC dissolved soon afterward, and the price of tin, now in a free-market environment, fell to $4 per pound and remained around that level through the 1990s.[73] The price increased again by 2010 with a rebound in consumption following the 2008 financial crisis and the Great Recession, accompanying restocking and continued growth in consumption.[60]

London Metal Exchange (LME) is tin's principal trading site.[60] Other tin contract markets are Kuala Lumpur Tin Market (KLTM) and Indonesia Tin Exchange (INATIN).[74]
Due to factors involved in the 2021 global supply chain crisis, tin prices almost doubled during 2020–21 and have had their largest annual rise in over 30 years. Global refined tin consumption dropped 1.6 percent in 2020 as the COVID-19 pandemic disrupted global manufacturing industries.[75]
Applications
[edit]
In 2018, just under half of all tin produced was used in solder. The rest was divided between tin plating, tin chemicals, brass and bronze alloys, and niche uses.[76]
Pigments
[edit]Pigment Yellow 38, tin(IV) sulfide, is known as mosaic gold.[77]
Purple of Cassius, Pigment Red 109, a hydrous double stannate of gold, was mainly, in terms of painting, restricted to miniatures due to its high cost. It was widely used to make cranberry glass. It has also been used in the arts to stain porcelain.[78]
Lead-tin yellow (which occurs in two yellow forms — a stannate and a silicate) was a pigment that was historically highly important for oil painting and which had some use in fresco in its silicate form.[79] Lead stannate is also known in orange form but has not seen wide use in the fine arts. It is available for purchase in pigment form from specialist artists' suppliers. There is another minor form, in terms of artistic usage and availability, of lead-tin yellow known as Lead-tin Antimony Yellow.[citation needed]
Cerulean blue, a somewhat dull cyan chemically known as cobalt stannate, continues to be an important artists' pigment. Its hue is similar to that of Manganese blue, Pigment Blue 33, although it lacks that pigment's colorfulness and is more opaque.[80] Artists typically must choose between cobalt stannate and manganese blue imitations made with phthalocyanine blue green shade (Pigment Blue 15:3), as industrial production of manganese blue pigment ceased in the 1970s.[81] Cerulean blue made with cobalt stannate, however, was popular with artists prior to the production of Manganese blue.[82][83]
Pigment Red 233, commonly known as Pinkcolor or Potter's Pink and more precisely known as Chrome Tin Pink Sphene, is a historically important pigment in watercolor.[84] However, it has enjoyed a large resurgence in popularity due to Internet-based word-of-mouth. It is fully lightfast and chemically stable in both oil paints and watercolors. Other inorganic mixed metal complex pigments, produced via calcination, often feature tin as a constituent. These pigments are known for their lightfastness, weatherfastness, chemical stability, lack of toxicity, and opacity. Many are rather dull in terms of colorfulness. However, some possess enough colorfulness to be competitive for use cases that require more than a moderate amount of it. Some are prized for other qualities. For instance, Pinkcolor is chosen by many watercolorists for its strong granulation, even though its chroma is low. Recently, NTP Yellow (a pyrochlore) has been brought to market as a non-toxic replacement for lead(II) chromate with greater opacity, lightfastness, and weathering resistance than proposed organic lead chromate replacement pigments possess.[85] NTP Yellow possesses the highest level of color saturation of these contemporary inorganic mixed metal complex pigments. More examples of this group include Pigment Yellow 158 (Tin Vanadium Yellow Cassiterite),[86] Pigment Yellow 216 (Solaplex Yellow),[87] Pigment Yellow 219 (Titanium Zinc Antimony Stannate),[88] Pigment Orange 82 (Tin Titanium Zinc oxide, also known as Sicopal Orange),[89] Pigment Red 121 (also known as Tin Violet and Chromium stannate),[90] Pigment Red 230 (Chrome Alumina Pink Corundum),[91] Pigment Red 236 (Chrome Tin Orchid Cassiterite),[92] and Pigment Black 23 (Tin Antimony Grey Cassiterite).[93] Another blue pigment with tin and cobalt is Pigment Blue 81, Cobalt Tin Alumina Blue Spinel.[citation needed]
Pigment White 15, tin(IV) oxide, is used for its iridescence, most commonly as a ceramic glaze.[94] There are no green pigments that have been used by artists that have tin as a constituent and purplish pigments with tin are classified as red, according to the Colour Index International.[citation needed]
Solder
[edit]
Tin has long been used in alloys with lead as solder, in amounts of 5 to 70% w/w. Tin with lead forms a eutectic mixture at the weight proportion of 61.9% tin and 38.1% lead (the atomic proportion: 73.9% tin and 26.1% lead), with melting temperature of 183 °C (361.4 °F). Such solders are primarily used for joining pipes or electric circuits. Since the European Union Waste Electrical and Electronic Equipment Directive (WEEE Directive) and Restriction of Hazardous Substances Directive came into effect on 1 July 2006, the lead content in such alloys has decreased. While lead exposure is associated with serious health problems, lead-free solder is not without its challenges, including a higher melting point, and the formation of tin whiskers that cause electrical problems. Tin pest can occur in lead-free solders, leading to loss of the soldered joint. Replacement alloys are being found, but the problems of joint integrity remain.[95] A common lead-free alloy is 99% tin, 0.7% copper, and 0.3% silver, with melting temperature of 217 °C (422.6 °F).[96]
Tin plating
[edit]
Tin bonds readily to iron and is used for coating lead, zinc, and steel to prevent corrosion. Tin-plated (or tinned) steel containers are widely used for food preservation, and this forms a large part of the market for metallic tin. A tinplate canister for preserving food was first manufactured in London in 1812.[97] Speakers of British English call such containers "tins", while speakers of U.S. English call them "cans" or "tin cans". One derivation of such use is the slang term "tinnie" or "tinny", meaning "can of beer" in Australia. The tin whistle is so called because it was mass-produced first in tin-plated steel.[98][99]
Copper cooking vessels such as saucepans and frying pans are frequently lined with a thin plating of tin, by electroplating or by traditional chemical methods, since use of copper cookware with acidic foods can be toxic.[100][101]
Specialized alloys
[edit]

Tin in combination with other elements forms a wide variety of useful alloys. Tin is most commonly alloyed with copper. Pewter is 85–99% tin,[102] and bearing metal has a high percentage of tin as well.[103][104] Bronze is mostly copper with 12% tin, while the addition of phosphorus yields phosphor bronze. Bell metal is also a copper–tin alloy, containing 22% tin. Tin has sometimes been used in coinage; it once formed a single-digit percentage (usually five percent or less) of American[105] and Canadian[106] pennies.
The niobium–tin compound Nb3Sn is commercially used in coils of superconducting magnets for its high critical temperature (18 K) and critical magnetic field (25 T). A superconducting magnet weighing as little as two kilograms is capable of producing the magnetic field of a conventional electromagnet weighing tons.[107]
A small percentage of tin is added to zirconium alloys for the cladding of nuclear fuel.[108]
Most metal pipes in a pipe organ are of a tin/lead alloy, with 50/50 as the most common composition. The proportion of tin in the pipe defines the pipe's tone, since tin has a desirable tonal resonance. When a tin/lead alloy cools, the lead phase solidifies first, then when the eutectic temperature is reached, the remaining liquid forms the layered tin/lead eutectic structure, which is shiny; contrast with the lead phase produces a mottled or spotted effect. This metal alloy is referred to as spotted metal. Major advantages of using tin for pipes include its appearance, workability, and resistance to corrosion.[109][110]
Manufacturing of chemicals
[edit]Tin compounds are used in the production of various chemicals, including stabilizers for PVC and catalysts for industrial processes. Tin in form of ingots provide the raw material necessary for these chemical reactions, ensuring consistent quality and performance.[citation needed]
Optoelectronics
[edit]The oxides of indium and tin are electrically conductive and transparent, and are used to make transparent electrically conducting films with applications in optoelectronics devices such as liquid crystal displays.[111]
Other applications
[edit]
Punched tin-plated steel, also called pierced tin, is an artisan technique originating in central Europe for creating functional and decorative housewares. Decorative piercing designs exist in a wide variety, based on local tradition and the artisan. Punched tin lanterns are the most common application of this artisan technique. The light of a candle shining through the pierced design creates a decorative light pattern in the room where it sits. Lanterns and other punched tin articles were created in the New World from the earliest European settlement. A well-known example is the Revere lantern, named after Paul Revere.[112]
In America, pie safes and food safes were in use in the days before refrigeration. These were wooden cupboards of various styles and sizes – either floor standing or hanging cupboards meant to discourage vermin and insects and to keep dust from perishable foodstuffs. These cabinets had tinplate inserts in the doors and sometimes in the sides, punched out by the homeowner, cabinetmaker, or a tinsmith in varying designs to allow for air circulation while excluding flies. Modern reproductions of these articles remain popular in North America.[113]
Window glass is most often made by floating molten glass on molten tin (float glass), resulting in a flat and flawless surface. This is also called the "Pilkington process".[114]
Tin is used as a negative electrode in advanced Li-ion batteries. Its application is somewhat limited by the fact that some tin surfaces[which?] catalyze decomposition of carbonate-based electrolytes used in Li-ion batteries.[115]
Tin(II) fluoride is added to some dental care products[116] as stannous fluoride (SnF2). Tin(II) fluoride can be mixed with calcium abrasives while the more common sodium fluoride gradually becomes biologically inactive in the presence of calcium compounds.[117] It has also been shown to be more effective than sodium fluoride in controlling gingivitis.[118]
Tin is used as a target to create laser-induced plasmas that act as the light source for extreme ultraviolet lithography.[119]
Organotin compounds
[edit]Organotin compounds are organometallic compounds containing tin–carbon bonds. Worldwide industrial production of organotin compounds likely exceeds 50,000 tonnes.[120]
PVC stabilizers
[edit]The major commercial application of organotin compounds is in the stabilization of PVC plastics. In the absence of such stabilizers, PVC would rapidly degrade under heat, light, and atmospheric oxygen, resulting in discolored, brittle products. Tin scavenges labile chloride ions (Cl−), which would otherwise strip HCl from the plastic material.[121] Typical tin compounds are carboxylic acid derivatives of dibutyltin dichloride, such as dibutyltin dilaurate.[122]
Biocides
[edit]Some organotin compounds are relatively toxic, with both advantages and problems. They are used for biocidal properties as fungicides, pesticides, algaecides, wood preservatives, and antifouling agents.[121] Tributyltin oxide is used as a wood preservative.[123] Tributyltin is used for various industrial purposes such as slime control in paper mills and disinfection of circulating industrial cooling waters.[124] Tributyltin was used as additive for ship paint to prevent growth of fouling organisms on ships, with use declining after organotin compounds were recognized as persistent organic pollutants with high toxicity for some marine organisms (the dog whelk, for example).[125] The EU banned the use of organotin compounds in 2003,[126] while concerns over the toxicity of these compounds to marine life and damage to the reproduction and growth of some marine species[121] (some reports describe biological effects to marine life at a concentration of 1 nanogram per liter) have led to a worldwide ban by the International Maritime Organization.[127] Many nations now restrict the use of organotin compounds to vessels greater than 25 m (82 ft) long.[121] The persistence of tributyltin in the aquatic environment is dependent upon the nature of the ecosystem.[128] Because of this persistence and its use as an additive in ship paint, high concentrations of tributyltin have been found in marine sediments located near naval docks.[129] Tributyltin has been used as a biomarker for imposex in neogastropods, with at least 82 known species.[130] With the high levels of TBT in the local inshore areas, due to shipping activities, the shellfish had an adverse effect.[128] Imposex is the imposition of male sexual characteristics on female specimens where they grow a penis and a pallial vas deferens.[130][131] A high level of TBT can damage mammalian endocrine glands, reproductive and central nervous systems, bone structure and gastrointestinal tract.[131] Tributyltin also affect mammals, Including sea otters, whales, dolphins, and humans.[131]
Organic chemistry
[edit]Some tin reagents are useful in organic chemistry. In the largest application, stannous chloride is a common reducing agent for the conversion of nitro and oxime groups to amines. The Stille reaction couples organotin compounds with organic halides or pseudohalides.[132]
Li-ion batteries
[edit]Tin forms several inter-metallic phases with lithium metal, making it a potentially attractive material for battery applications. Large volumetric expansion of tin upon alloying with lithium and instability of the tin-organic electrolyte interface at low electrochemical potentials are the greatest challenges to employment in commercial cells.[133] Tin inter-metallic compound with cobalt and carbon was implemented by Sony in its Nexelion cells released in the late 2000s. The composition of the active material is approximately Sn0.3Co0.4C0.3. Research showed that only some crystalline facets of tetragonal (beta) Sn are responsible for undesirable electrochemical activity.[134]
Precautions
[edit]Cases of poisoning from tin metal, its oxides, and its salts are almost unknown. On the other hand, certain organotin compounds are almost as toxic as cyanide.[53]
Exposure to tin in the workplace can occur by inhalation, skin contact, and eye contact. The US Occupational Safety and Health Administration (OSHA) set the permissible exposure limit for tin exposure in the workplace as 2 mg/m3 over an 8-hour workday. The National Institute for Occupational Safety and Health (NIOSH) determined a recommended exposure limit (REL) of 2 mg/m3 over an 8-hour workday. At levels of 100 mg/m3, tin is immediately dangerous to life and health.[135]
See also
[edit]- Cassiterides (the mythical Tin Islands)
- Stannary
- Terne
- Tin pest
- Tin mining in Britain
- Tinning
- Whisker (metallurgy) (tin whiskers)
Notes
[edit]- ^ The thermal expansion of β-Sn is anisotropic: the parameters (at 20 °C) for each crystal axis are αa = 16.19×10−6/K, αc = 32.89×10−6/K, and αaverage = αV/3 = 21.76×10−6/K.[3]
- ^ Only hydrogen, fluorine, phosphorus, thallium and xenon are easier to use NMR analysis with for samples containing isotopes at their natural abundance.
References
[edit]- ^ "Standard Atomic Weights: Tin". CIAAW. 1983.
- ^ Prohaska, Thomas; Irrgeher, Johanna; Benefield, Jacqueline; Böhlke, John K.; Chesson, Lesley A.; Coplen, Tyler B.; Ding, Tiping; Dunn, Philip J. H.; Gröning, Manfred; Holden, Norman E.; Meijer, Harro A. J. (4 May 2022). "Standard atomic weights of the elements 2021 (IUPAC Technical Report)". Pure and Applied Chemistry. doi:10.1515/pac-2019-0603. ISSN 1365-3075.
- ^ a b c d e Arblaster, John W. (2018). Selected Values of the Crystallographic Properties of Elements. Materials Park, Ohio: ASM International. ISBN 978-1-62708-155-9.
- ^ Sn(−3) has been observed in [Sn2]6−, e.g. in (Ba2)4+(Mg4)8+Sn4−(Sn2)6−Sn2− (with square (Sn2−)n sheets), see Papoian, Garegin A.; Hoffmann, Roald (2000). "Hypervalent Bonding in One, Two, and Three Dimensions: Extending the Zintl–Klemm Concept to Nonclassical Electron-Rich Networks". Angew. Chem. Int. Ed. 2000 (39): 2408–2448. doi:10.1002/1521-3773(20000717)39:14<2408::aid-anie2408>3.0.co;2-u. PMID 10941096. Retrieved 2015-02-23.
- ^ Sn(−2) has been observed in SrSn; see Holleman, Arnold F.; Wiberg, Egon; Wiberg, Nils (2008). Lehrbuch der Anorganischen Chemie (in German) (102 ed.). Walter de Gruyter. p. 1007. ISBN 978-3-11-020684-5.
- ^ Sn(−1) has been observed in CsSn; see Holleman, Arnold F.; Wiberg, Egon; Wiberg, Nils (2008). Lehrbuch der Anorganischen Chemie (in German) (102 ed.). Walter de Gruyter. p. 1007. ISBN 978-3-11-020684-5.
- ^ "New Type of Zero-Valent Tin Compound". Chemistry Europe. 27 August 2016.
- ^ "HSn". NIST Chemistry WebBook. National Institute of Standards and Technology. Retrieved 2013-01-23.
- ^ "SnH3". NIST Chemistry WebBook. National Institure of Standards and Technology. Retrieved 2013-01-23.
- ^ Lide, D. R., ed. (2005). "Magnetic susceptibility of the elements and inorganic compounds". CRC Handbook of Chemistry and Physics (PDF) (86th ed.). Boca Raton (FL): CRC Press. ISBN 0-8493-0486-5.
- ^ Weast, Robert (1984). CRC, Handbook of Chemistry and Physics. Boca Raton, Florida: Chemical Rubber Company Publishing. pp. E110. ISBN 0-8493-0464-4.
- ^ Kondev, F. G.; Wang, M.; Huang, W. J.; Naimi, S.; Audi, G. (2021). "The NUBASE2020 evaluation of nuclear properties" (PDF). Chinese Physics C. 45 (3) 030001. doi:10.1088/1674-1137/abddae.
- ^ Gray, Theodore (2007). "Tin Images". The Elements. Black Dog & Leventhal.
- ^ a b c d e Holleman, Arnold F.; Wiberg, Egon; Wiberg, Nils (1985). "Tin". Lehrbuch der Anorganischen Chemie (in German) (91–100 ed.). Walter de Gruyter. pp. 793–800. ISBN 978-3-11-007511-3.
- ^ "Ink with tin nanoparticles could print future circuit boards". Phys.org. 12 April 2011. Archived from the original on 2011-09-16.
- ^ Jo, Yun Hwan; Jung, Inyu; Choi, Chung Seok; Kim, Inyoung; Lee, Hyuck Mo (2011). "Synthesis and characterization of low temperature Sn nanoparticles for the fabrication of highly conductive ink". Nanotechnology. 22 (22) 225701. Bibcode:2011Nanot..22v5701J. doi:10.1088/0957-4484/22/22/225701. PMID 21454937. S2CID 25202674.
- ^ Molodets, A.M.; Nabatov, S.S. (2000). "Thermodynamic potentials, diagram of state, and phase transitions of tin on shock compression". High Temperature. 38 (5): 715–721. Bibcode:2000HTemp..38..715M. doi:10.1007/BF02755923. S2CID 120417927.
- ^ "Tin Pests | Center for Advanced Life Cycle Engineering". calce.umd.edu. Retrieved 2022-11-04.
- ^ Le Coureur, Penny; Burreson, Jay (2004). Napoleon's Buttons: 17 molecules that changed history. New York: Penguin Group, USA.
- ^ Öhrström, Lars (2013). The Last Alchemist in Paris. Oxford: Oxford University Press. ISBN 978-0-19-966109-1.
- ^ Cotton, Simon (29 April 2014). "Book review: The last alchemist in Pari". Chemistry World. Royal Society of Chemistry. Archived from the original on 2014-08-10. Retrieved 2019-11-22.
- ^ Emsley, John (1 October 2011) [2001]. Nature's Building Blocks: an A-Z Guide to the Elements (New ed.). New York, United States: Oxford University Press. p. 552. ISBN 978-0-19-960563-7.
Only officers had metal buttons, and those were made of brass.
- ^ a b Schwartz, Mel (2002). "Tin and alloys, properties". Encyclopedia of Materials, Parts and Finishes (2nd ed.). CRC Press. ISBN 978-1-56676-661-6.
- ^ "Tin Alloys – Characteristics and Uses". Nuclear Power. Retrieved 2022-11-04.
- ^ Dehaas, W.; Deboer, J.; Vandenberg, G. (1935). "The electrical resistance of cadmium, thallium and tin at low temperatures". Physica. 2 (1–12): 453. Bibcode:1935Phy.....2..453D. doi:10.1016/S0031-8914(35)90114-8.
- ^ a b Meissner, W.; R. Ochsenfeld (1933). "Ein neuer effekt bei eintritt der Supraleitfähigkeit". Naturwissenschaften. 21 (44): 787–788. Bibcode:1933NW.....21..787M. doi:10.1007/BF01504252. S2CID 37842752.
- ^ Craig, Bruce D.; Anderson, David S.; International, A. S. M. (January 1995). Handbook of corrosion data. ASM International. p. 126. ISBN 978-0-87170-518-1. Archived from the original on 2016-05-11.
- ^ Crutchlow, Charlotte (25 June 2021). "Tin-Formation About the Element Tin | Periodic Table". ChemTalk. Retrieved 2022-11-04.
- ^ "Tin | NIDC: National Isotope Development Center". www.isotopes.gov. Retrieved 2025-04-13.
- ^ "Testing the Possible Doubly Magic Nature of Tin-100, Researchers Study the Electromagnetic Properties of Indium Isotopes". Energy.gov. Retrieved 2025-04-13.
- ^ Yang, X. F.; Wang, S. J.; Wilkins, S. G.; Ruiz, R. F. Garcia (1 March 2023). "Laser spectroscopy for the study of exotic nuclei". Progress in Particle and Nuclear Physics. 129 104005. arXiv:2209.15228. Bibcode:2023PrPNP.12904005Y. doi:10.1016/j.ppnp.2022.104005. ISSN 0146-6410.
- ^ "Interactive NMR Frequency Map". Archived from the original on 2011-06-04. Retrieved 2009-05-05.
- ^ a b Sears, Varley F. (January 1992). "Neutron scattering lengths and cross sections". Neutron News. 3 (3): 26–37. doi:10.1080/10448639208218770. ISSN 1044-8632. Table of cross sections available at NIST: Neutron Scattering Lengths and cross sections.
- ^ "Westinghouse Nuclear > Energy Systems > Lead-cooled Fast Reactor".
- ^ Walker, Phil (1994). "Doubly Magic Discovery of Tin-100". Physics World. 7 (June): 28. doi:10.1088/2058-7058/7/6/24.
- ^ Kondev, F. G.; Wang, M.; Huang, W. J.; Naimi, S.; Audi, G. (2021). "The NUBASE2020 evaluation of nuclear properties" (PDF). Chinese Physics C. 45 (3) 030001. doi:10.1088/1674-1137/abddae.
- ^ a b c "tin". Oxford English Dictionary (Online ed.). Oxford University Press. (Subscription or participating institution membership required.)
- ^ Harper, Douglas. "tin". Online Etymology Dictionary.
- ^ Encyclopædia Britannica, 11th Edition, 1911, s.v. 'tin', citing H. Kopp
- ^ "The Ancient Mining of Tin". oxleigh.freeserve.co.uk. Archived from the original on 2009-04-03. Retrieved 2009-07-07.
- ^ American Heritage Dictionary
- ^ Cierny, J.; Weisgerber, G. (2003). "The "Bronze Age tin mines in Central Asia". In Giumlia-Mair, A.; Lo Schiavo, F. (eds.). The Problem of Early Tin. Oxford: Archaeopress. pp. 23–31. ISBN 978-1-84171-564-3.
- ^ a b c Penhallurick, R. D. (1986). Tin in Antiquity: its Mining and Trade Throughout the Ancient World with Particular Reference to Cornwall. London: The Institute of Metals. ISBN 978-0-904357-81-3.
- ^ Lamberg-Karlovsky, C. C.; Franklin, Alan D.; Olin, Jacqueline S.; Wertime, Theodore A., eds. (July 1980). "The development of the usage of tin and tin-bronze: some problems". The Search for Ancient Tin. Vol. 21. Washington D.C.: A seminar organized by Theodore A. Wertime and held at the Smithsonian Institution and the National Bureau of Standards, Washington D.C. March 14–15, 1977. p. 474. doi:10.2307/3103162. JSTOR 3103162.
{{cite book}}:|journal=ignored (help) - ^ "Project Ancient Tin".
- ^ Dube, RK (September 2006). "Interrelation between gold and tin: A historical perspective". Gold Bulletin. 39 (3): 103–113. doi:10.1007/BF03215537.
- ^ a b Holleman, Arnold Frederik; Wiberg, Egon (2001), Wiberg, Nils (ed.), Inorganic Chemistry, translated by Eagleson, Mary; Brewer, William, San Diego/Berlin: Academic Press/De Gruyter, ISBN 0-12-352651-5
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. doi:10.1016/C2009-0-30414-6. ISBN 978-0-08-037941-8.[page needed]
- ^ Taylor, F. Sherwood (1942). Inorganic & Theoretical Chemistry (6th ed.). Heineman.
- ^ Leger, J. M.; Haines, J.; Atouf, A. (1996). "The high pressure behaviour of the cotunnite and post-cotunnite phases of PbCl2 and SnCl
2". J. Phys. Chem. Solids. 57 (1): 7–16. Bibcode:1996JPCS...57....7L. doi:10.1016/0022-3697(95)00060-7. - ^ Gaur, D. P.; Srivastava, G.; Mehrotra, R. C. (1973). "Organic Derivatives of Tin. III. Reactions of Trialkyltin Ethoxide with Alkanolamines". Zeitschrift für Anorganische und Allgemeine Chemie. 398 (1): 72. Bibcode:1973ZAACh.398...72G. doi:10.1002/zaac.19733980109.
- ^ Elschenbroich, Christoph (2006). Organometallics (3rd, completely rev. and extended ed.). Weinheim: Wiley-VCH. ISBN 3-527-29390-6. OCLC 64305455.
- ^ a b c Graf, Günter G. (15 June 2000). "Tin, Tin Alloys, and Tin Compounds". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a27_049. ISBN 978-3-527-30385-4.
- ^ Thoonen, Sander H. L.; Deelman, Berth-Jan; van Koten, Gerard (2004). "Synthetic aspects of tetraorganotins and organotin(IV) halides" (PDF). Journal of Organometallic Chemistry. 689 (13): 2145–2157. doi:10.1016/j.jorganchem.2004.03.027. hdl:1874/6594. S2CID 100214292. Archived from the original (PDF) on 2011-08-07. Retrieved 2009-07-31.
- ^ Peng, Yang; Ellis, Bobby D.; Wang, Xinping; Fettinger, James C.; Power, P. P. (2009). "Reversible Reactions of Ethylene with Distannynes Under Ambient Conditions". Science. 325 (5948): 1668–1670. Bibcode:2009Sci...325.1668P. doi:10.1126/science.1176443. PMID 19779193. S2CID 3011002.
- ^ Shu, Frank H. (1982). The physical universe: An introduction to astronomy. University Science Books. pp. 119–121. ISBN 978-0-935702-05-7.
- ^ Emsley 2001, pp. 124, 231, 449 and 503.
- ^ a b "Tin: From Ore to Ingot". International Tin Research Institute. 1991. Archived from the original on 2009-03-22. Retrieved 2009-03-21.
- ^ Sutphin, David M.; Sabin, Andrew E.; Reed, Bruce L. (1 June 1992). Tin – International Strategic Minerals Inventory Summary Report. DIANE. p. 9. ISBN 978-0-941375-62-7. Archived from the original on 2016-04-28.
- ^ a b c d e f g Carlin, James F. Jr. "Tin: Statistics and Information" (PDF). United States Geological Survey. Archived from the original on 2008-12-06. Retrieved 2008-11-23.
- ^ Reilly, Michael (26 May 2007). "How Long Will it Last?". New Scientist. 194 (2605): 38–39. Bibcode:2007NewSc.194...38R. doi:10.1016/S0262-4079(07)61508-5.
- ^ Brown, Lester (2006). Plan B 2.0. New York: W.W. Norton. p. 109. ISBN 978-0-393-32831-8.
- ^ Alves, Bruna (15 February 2024). "U.S. annual tin recycling 2023". Statista. Retrieved 2024-06-23.
- ^ Kovalenko, V. I.; Yarmolyuk, V. V. (1995). "Endogenous rare metal ore formations and rare metal metallogeny of Mongolia". Economic Geology. 90 (3): 520. Bibcode:1995EcGeo..90..520K. doi:10.2113/gsecongeo.90.3.520.
- ^ "Seminole Group Colombia Discovers High Grade Tin Ore in the Amazon Jungle". FreePR101 PressRelease. Archived from the original on 2014-08-26.
- ^ Schrader, George F.; Elshennawy, Ahmad K.; Doyle, Lawrence E. (July 2000). Manufacturing processes and materials. Society of Manufacturing Engineers. ISBN 978-0-87263-517-3. Archived from the original on 2016-05-11.
- ^ Louis, Henry (1911). Metallurgy of tin. McGraw-Hill book Company.
- ^ Knorr, Klaus (1945). Tin Under Control. Stanford University Press. ISBN 978-0-8047-2136-3. Archived from the original on 2016-05-19.
{{cite book}}: ISBN / Date incompatibility (help) - ^ "LME Tin Brands". ITRI.co.uk. International Tin Research Institute. Archived from the original on 2008-12-07. Retrieved 2009-05-05.
- ^ "Top Ten Tin Producing Companies". itri.co.uk. International Tin Research Institute. Archived from the original on 2008-12-07. Retrieved 2009-05-05.
- ^ "The World's Biggest Tin Producers". The Balance. 13 January 2019.
- ^ "Agreement establishing the Association of Tin Producing Countries [1984] ATS 10". Australasian Legal Information Institute, Australian Treaties Library.
- ^ a b Carlin, James F. Jr. (1998). "Significant events affecting tin prices since 1958" (PDF). USGS. Archived from the original (PDF) on 2011-10-28.
- ^ "12 Januari Pemasaran Perdana INATIN". 15 December 2011. Archived from the original on 2012-04-26.
- ^ Daly, Tom (5 December 2021). "Tin surge worsens supply chain woes for electronics, solar and auto firms". Reuters. Retrieved 2021-12-07.
- ^ "Tin demand to decline – International Tin Association". Mining.com. 18 October 2019. Retrieved 2021-07-03.
- ^ "The Color of Art Pigment Database – Pigment Yellow – PY". artiscreation. 2024. Retrieved 2024-08-17.
- ^ "The Color of Art Pigment Database – Pigment Red – PR". artiscreation. 2024. Retrieved 2024-08-17.
- ^ "The Color of Art Pigment Database – Pigment Yellow – PY". artiscreation. 2024. Retrieved 2024-08-17.
- ^ "The Color of Art Pigment Database – Pigment Blue – PB". artiscreation. 2024. Retrieved 2024-08-17.
- ^ "The Color of Art Pigment Database – Pigment Blue – PB". artiscreation. 2024. Retrieved 2024-08-17.
- ^ "Blue pigments". Professional development courses, distance learning and consultancy. 30 August 2021. Retrieved 2025-04-13.
- ^ Hatch, Evie (15 October 2021). "Pigment Colour Index: Blue Pigments - Jackson's Art Blog". Jackson's Art Blog. Archived from the original on 2025-01-08. Retrieved 2025-04-13.
- ^ "The Color of Art Pigment Database – Pigment Red – PR". artiscreation. 2024. Retrieved 2024-08-17.
- ^ "The Color of Art Pigment Database – Pigment Yellow – PY". artiscreation. 2024. Retrieved 2024-08-17.
- ^ "The Color of Art Pigment Database – Pigment Yellow – PY". artiscreation. 2024. Retrieved 2024-08-17.
- ^ "The Color of Art Pigment Database – Pigment Yellow – PY". artiscreation. 2024. Retrieved 2024-08-17.
- ^ "The Color of Art Pigment Database – Pigment Yellow – PY". artiscreation. 2024. Retrieved 2024-08-17.
- ^ "The Color of Art Pigment Database – Pigment Orange – PO". artiscreation. 2024. Retrieved 2024-08-17.
- ^ "The Color of Art Pigment Database – Pigment Red – PR". artiscreation. 2024. Retrieved 2024-08-17.
- ^ "The Color of Art Pigment Database – Pigment Red – PR". artiscreation. 2024. Retrieved 2024-08-17.
- ^ "The Color of Art Pigment Database – Pigment Red – PR". artiscreation. 2024. Retrieved 2024-08-17.
- ^ "The Color of Art Pigment Database – Pigment Black – PBk". artiscreation. 2024. Retrieved 2024-08-17.
- ^ "The Color of Art Pigment Database – Pigment White – PW". artiscreation. 2024. Retrieved 2024-08-17.
- ^ Black, Harvey (2005). "Getting the Lead Out of Electronics". Environmental Health Perspectives. 113 (10): A682–5. doi:10.1289/ehp.113-a682. PMC 1281311. PMID 16203230.
- ^ "Technical data Sheet - Lead free alloy" (PDF). RS Online. Retrieved 2023-06-18.
- ^ Childs, Peter (July 1995). "The tin-man's tale". Education in Chemistry. Vol. 32, no. 4. Royal Society of Chemistry. p. 92. Retrieved 2018-06-19.
- ^ Control, Tin Under (1945). Tin Under Control. Stanford University Press. pp. 10–15. ISBN 978-0-8047-2136-3. Archived from the original on 2016-05-31.
{{cite book}}: ISBN / Date incompatibility (help) - ^ Panel On Tin, National Research Council (U.S.). Committee on Technical Aspects of Critical and Strategic Materials (1970). Trends in the use of tin. pp. 10–22. Archived from the original on 2016-05-22.
- ^ "Cooking utensils and nutrition Information | Mount Sinai - New York". Mount Sinai Health System. Retrieved 2025-04-13.
- ^ Ali Sultan, Saif Ali; Ahmed Khan, Fawad; Wahab, Abdul; Fatima, Batool; Khalid, Hira; Bahader, Ali; Safi, Sher Zaman; Selvaraj, Chandrabose; Ali, Abid; Alomar, Suliman Yousef; Imran, Muhammad (24 July 2023). "Assessing Leaching of Potentially Hazardous Elements from Cookware during Cooking: A Serious Public Health Concern". Toxics. 11 (7): 640. Bibcode:2023Toxic..11..640A. doi:10.3390/toxics11070640. ISSN 2305-6304. PMC 10386729. PMID 37505605.
- ^ Hull, Charles (1992). Pewter. Osprey Publishing. pp. 1–5. ISBN 978-0-7478-0152-8.
- ^ Brakes, James (2009). "Introduction". Analysis of Babbit. BiblioBazaar, LLC. pp. 1–2. ISBN 978-1-110-11092-6.
- ^ Williams, Robert S. (2007). Principles of Metallography. Read books. pp. 46–47. ISBN 978-1-4067-4671-6.
- ^ "The Composition of the Cent". US Mint. Archived from the original on 2011-10-25. Retrieved 2011-10-28.
- ^ "Composition of canadian coins" (PDF). Canadian Mint. Archived from the original (PDF) on 2012-01-13. Retrieved 2011-10-28.
- ^ Geballe, Theodore H. (October 1993). "Superconductivity: From Physics to Technology". Physics Today. 46 (10): 52–56. Bibcode:1993PhT....46j..52G. doi:10.1063/1.881384.
- ^ Campbell, Flake C. (2008). "Zirconium". Elements of Metallurgy and Engineering Alloys. ASM International. p. 597. ISBN 978-0-87170-867-0. Archived from the original on 2016-05-28.
- ^ Robert Palmieri, ed. (2006). "Pipe Metal". Encyclopedia of keyboard instruments. New York: Garland. p. 411. ISBN 978-0-415-94174-7.
- ^ George Ashdown Audsley (1988). "Metal Pipes: And the Materials used in their Construction". The Art of Organ Building Audsley, George Ashdown. Courier Dover Publications. p. 501. ISBN 978-0-486-21315-6.
- ^ Kim, H.; Gilmore, C.; Pique, A.; Horwitz, J.; Mattoussi, H.; Murata, H.; Kafafi, Z.; Chrisey, D. (1999). "Electrical, optical, and structural properties of indium tin oxide thin films for organic light-emitting devices". Journal of Applied Physics. 86 (11): 6451. Bibcode:1999JAP....86.6451K. doi:10.1063/1.371708.
- ^ Bridge, Janet (September 1996). Making & decorating picture frames. North Light Books. ISBN 978-0-89134-739-2.
- ^ "Tin punching". Archived from the original on 2011-08-11. Retrieved 2011-08-15.
- ^ Pilkington, L. A. B. (1969). "Review Lecture. The Float Glass Process". Proceedings of the Royal Society of London. Series A, Mathematical and Physical Sciences. 314 (1516): 1–25. Bibcode:1969RSPSA.314....1P. doi:10.1098/rspa.1969.0212. JSTOR 2416528. S2CID 109981215.
- ^ Lucas, Ivan T.; Syzdek, Jarosław; Kostecki, Robert (2011). "Interfacial processes at single-crystal β-Sn electrodes in organic carbonate electrolytes". Electrochemistry Communications. 13 (11): 1271–1275. doi:10.1016/j.elecom.2011.08.026.
- ^ "Colgate Gel-Kam". Archived from the original on 2009-04-27. Retrieved 2009-05-05.
- ^ Hattab, F. (April 1989). "The State of Fluorides in Toothpastes". Journal of Dentistry. 17 (2): 47–54. doi:10.1016/0300-5712(89)90129-2. PMID 2732364.
- ^ Perlich, M. A.; Bacca, L. A.; Bollmer, B. W.; Lanzalaco, A. C.; McClanahan, S. F.; Sewak, L. K.; Beiswanger, B. B.; Eichold, W. A.; Hull, J. R.; et al. (1995). "The clinical effect of a stabilized stannous fluoride dentifrice on plaque formation, gingivitis and gingival bleeding: a six-month study". The Journal of Clinical Dentistry. 6 (Special Issue): 54–58. PMID 8593194.
- ^ Versolato, Oscar O. (2019). "Physics of laser-driven tin plasma sources of EUV radiation for nanolithography". Plasma Sources Science and Technology. 28 (8). Bibcode:2019PSST...28h3001V. doi:10.1088/1361-6595/ab3302.
- ^ Ebdon, L.; Britain), Royal Society of Chemistry (Great (2001). "Organotin in Industrial and Domestic Products". Trace element speciation for environment, food and health. Royal Society of Chemistry. p. 144. ISBN 978-0-85404-459-7. Archived from the original on 2016-05-21.
- ^ a b c d Atkins, Peter; Shriver, Duward F.; Overton, Tina & Rourke, Jonathan (2006). Inorganic chemistry (4th ed.). W.H. Freeman. pp. 343, 345. ISBN 978-0-7167-4878-6.
- ^ Wilkes, Charles E.; Summers, James W.; Daniels, Charles Anthony; Berard, Mark T. (August 2005). PVC handbook. Hanser. p. 108. ISBN 978-1-56990-379-7. Archived from the original on 2016-05-09.
- ^ David N.-S. Hon; Nobuo Shiraishi, eds. (2001). "Preservation of Wood". Wood and cellulosic chemistry. New York, NY: Dekker. p. 799. ISBN 978-0-8247-0024-9.
- ^ Antizar-Ladislao, Blanca (1 February 2008). "Environmental levels, toxicity and human exposure to tributyltin (TBT)-contaminated marine environment. A review". Environment International. 34 (2): 292–308. Bibcode:2008EnInt..34..292A. doi:10.1016/j.envint.2007.09.005. PMID 17959247.
- ^ Eisler, Ronald. "Tin Hazards To Fish, Wildlife, and Invertebrates: A Synoptic Review" (PDF). U.S. Fish and Wildlife Service Patuxent Wildlife Research Center. Archived (PDF) from the original on 2012-01-18.
- ^ "Regulation (EC) No 782/2003 of the European Parliament and of the Council of 14 April 2003 on the prohibition of organotin compounds on ships". europa.eu. Retrieved 2009-05-05.
- ^ Dürr, Simone; Thomason, Jeremy, eds. (2008). "Fouling on Shipping". Biofouling. Oxford: Blackwell. p. 227. ISBN 978-1-4051-6926-4.
- ^ a b Maguire, R. James (1987). "Environmental aspects of tributyltin". Applied Organometallic Chemistry. 1 (6): 475–498. doi:10.1002/aoc.590010602.
- ^ de Mora, S. J.; Stewart, C.; Phillips, D. (1 January 1995). "Sources and rate of degradation of tri(n-butyl)tin in marine sediments near Auckland, New Zealand". Marine Pollution Bulletin. 30 (1): 50–57. Bibcode:1995MarPB..30...50D. doi:10.1016/0025-326X(94)00178-C.
- ^ a b Axiak, Victor; Micallef, Diane; Muscat, Joanne; Vella, Alfred; Mintoff, Bernardette (1 March 2003). "Imposex as a biomonitoring tool for marine pollution by tributyltin: some further observations". Environment International. Secotox S.I. 28 (8): 743–749. Bibcode:2003EnInt..28..743A. doi:10.1016/S0160-4120(02)00119-8. PMID 12605923.
- ^ a b c "The Effects of Tributyltin on the Marine Environment". ScienceBuzz. 17 November 2018. Archived from the original on 2021-01-25. Retrieved 2020-11-17.
- ^ Farina, Vittorio; Krishnamurthy, Venkat; Scott, William J. (1997). "The Stille Reaction". Organic Reactions. pp. 1–652. doi:10.1002/0471264180.or050.01. ISBN 0-471-26418-0.
- ^ Mou, Haoyi; Xiao, Wei; Miao, Chang; Li, Rui; Yu, Liming (2020). "Tin and Tin Compound Materials as Anodes in Lithium-Ion and Sodium-Ion Batteries: A Review". Frontiers in Chemistry. 8 141. Bibcode:2020FrCh....8..141M. doi:10.3389/fchem.2020.00141. PMC 7096543. PMID 32266205.
- ^ Lucas, Ivan; Syzdek, Jaroslaw (2011). "Interfacial processes at single-crystal β-Sn electrodes in organic carbonate electrolytes". Electrochemistry Communications. 13 (11): 1271. doi:10.1016/j.elecom.2011.08.026.
- ^ "NIOSH Pocket Guide to Chemical Hazards – Tin". CDC. Archived from the original on 2015-11-25. Retrieved 2015-11-24.
Bibliography
[edit] This article incorporates text from this source, which is in the public domain: Carlin, James F., Jr. (1998). "Significant events affecting tin prices since 1958". U.S. National Geodetic Survey
This article incorporates text from this source, which is in the public domain: Carlin, James F., Jr. (1998). "Significant events affecting tin prices since 1958". U.S. National Geodetic Survey- Lide, David R., ed. (2006). Handbook of Chemistry and Physics (87th ed.). Boca Raton, Florida: CRC Press, Taylor & Francis Group. ISBN 978-0-8493-0487-3.
- Emsley, John (2001). "Tin". Nature's Building Blocks: An A–Z Guide to the Elements. Oxford, England, UK: Oxford University Press. pp. 445–450. ISBN 978-0-19-850340-8.
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. doi:10.1016/C2009-0-30414-6. ISBN 978-0-08-037941-8.
- Heiserman, David L. (1992). "Element 50: Tin". Exploring Chemical Elements and their Compounds. New York: TAB Books. ISBN 978-0-8306-3018-9.
- MacIntosh, Robert M. (1968). "Tin". In Clifford A. Hampel (ed.). The Encyclopedia of the Chemical Elements. New York: Reinhold Book Corporation. pp. 722–732. LCCN 68-29938.
- Stwertka, Albert (1998). "Tin". Guide to the Elements (Revised ed.). Oxford University Press. ISBN 978-0-19-508083-4.
External links
[edit]- Tin at The Periodic Table of Videos (University of Nottingham)
- Theodore Gray's Wooden Periodic Table Table: Tin samples and castings
- Base Metals: Tin
- CDC – NIOSH Pocket Guide to Chemical Hazards
- Tin (USD cents per kg)
Properties
Physical properties
Tin is a post-transition metal with atomic number 50 and atomic mass of 118.71 u.[6] Its electron configuration is [Kr] 4d¹⁰ 5s² 5p².[6] Elemental tin appears as a soft, malleable, and ductile silvery-white metal with a slight blue tint.[2] It has a density of 7.287 g/cm³ at 20°C.[2] The metal's low hardness, rated at 1.5–1.8 on the Mohs scale, contributes to its ease of deformation. Tensile strength varies by form but typically ranges from 11 to 18 MPa for pure tin.[7] Tin exhibits notable thermal properties, including a melting point of 231.93°C and a boiling point of 2602°C.[2] Its specific heat capacity is 0.227 J/g·K.[8] Tin exists in two primary allotropes: white β-tin, which is stable at room temperature and features a tetragonal crystal structure, and gray α-tin, stable below 13.2°C with a diamond cubic structure.[9] The allotropic transformation from β-tin to α-tin, known as tin pest, causes the metal to become brittle and crumble due to the lower density of the gray form (5.77 g/cm³).[9] Optically, tin displays a bright, silvery luster and high reflectivity, particularly in the infrared spectrum, which enhances its use in coatings.[2]Chemical properties
Tin primarily exhibits two oxidation states in its compounds: +2, known as the stannous state, and +4, known as the stannic state. The +2 state is more reducing in nature, while the +4 state is generally more stable, reflecting the influence of the inert pair effect that becomes more pronounced down group 14 but is still relatively weak for tin compared to lead.[10] Tin's reactivity is characterized by its resistance to corrosion in air and water at ambient temperatures, attributed to the formation of a thin, adherent oxide layer (SnO₂) that passivates the surface and prevents further oxidation. This protective layer contributes to tin's use in coatings for other metals. However, tin displays amphoteric behavior, reacting with both acids and bases. With acids, it dissolves slowly in concentrated hydrochloric acid to form tin(II) chloride and hydrogen gas:while it is inert toward dilute sulfuric acid but reacts with hot concentrated sulfuric acid or nitric acid to yield corresponding tin salts. In alkaline conditions, tin reacts with hot solutions of sodium hydroxide to form sodium stannate and hydrogen:
or, under more forcing conditions, hexahydroxostannate(IV):
This dual reactivity underscores tin's amphoteric nature, allowing it to form both cationic and anionic species depending on the pH environment.[11]/Qualitative_Analysis/Characteristic_Reactions_of_Select_Metal_Ions/Characteristic_Reactions_of_Tin_Ions_(Sn_Sn)) In electrochemical contexts, tin's behavior is defined by its standard reduction potentials, which indicate moderate oxidizing and reducing capabilities. The potential for the Sn⁴⁺/Sn²⁺ couple is +0.15 V, showing that Sn⁴⁺ is a moderate oxidant relative to Sn²⁺, while the Sn²⁺/Sn couple at -0.14 V positions Sn²⁺ as a reducing agent compared to hydrogen. These values facilitate tin's role in redox processes, such as in electroplating or battery applications, where the +2 state can disproportionate under certain conditions.[12] Tin ions in aqueous solutions tend to hydrolyze, but they form coordination complexes with ligands like chloride, influenced by their geometry preferences. Sn(IV) commonly adopts octahedral coordination in complexes such as [SnCl₆]²⁻, while neutral SnCl₄ is tetrahedral; Sn(II) favors more irregular geometries but can form octahedral structures in solvated forms. In chloride media, Sn(II) forms stepwise chloro complexes, with the tetrachloro complex [SnCl₄]²⁻ having a stability constant log β₄ ≈ 1.3 at 25°C and zero ionic strength, indicating weak but measurable association that stabilizes Sn(II) in high-chloride environments. These constants vary with ionic strength and temperature, with endothermic formation enthalpies leading to increased stability at higher temperatures.[13][14][15] The gray α-tin allotrope, stable below 13.2°C, exhibits higher chemical reactivity than the metallic β-tin due to its non-metallic, diamond-like structure, which facilitates easier oxidation and dissolution.
Isotopes
Tin possesses ten stable isotopes, more than any other element, and over 40 known radioactive isotopes, with mass numbers ranging from ^{100}Sn to ^{138}Sn.[16][17] All stable tin isotopes are primordial nuclides, formed during the early history of the solar system, and contribute to the element's average atomic mass of 118.710.[18] The most abundant stable isotope is ^{120}Sn at 32.58%, followed by ^{118}Sn at 24.22%.[18] The natural isotopic abundances of tin's stable isotopes are as follows:| Isotope | Natural Abundance (%) | Relative Atomic Mass |
|---|---|---|
| ^{112}Sn | 0.97 | 111.904 823 87(61) |
| ^{114}Sn | 0.66 | 113.902 7827(10) |
| ^{115}Sn | 0.34 | 114.903 344 699(16) |
| ^{116}Sn | 14.54 | 115.901 742 80(10) |
| ^{117}Sn | 7.68 | 116.902 953 98(52) |
| ^{118}Sn | 24.22 | 117.901 606 57(54) |
| ^{119}Sn | 8.59 | 118.903 311 17(78) |
| ^{120}Sn | 32.58 | 119.902 201 63(97) |
| ^{122}Sn | 4.63 | 121.903 4438(26) |
| ^{124}Sn | 5.79 | 123.905 2766(11) |
Origin and history
Etymology
The English word "tin" derives from Old English tīn, which traces back to Proto-Germanic *tiną or *tinom, a term shared across Germanic languages such as Middle Dutch tin, Old High German zin, and Old Norse tin.[23] The ultimate origin of this Germanic root remains uncertain and is not attested outside Indo-European languages in a clear manner, though some scholars propose a possible pre-Indo-European substrate influence or a distant connection to Semitic terms for the metal.[23] In particular, the Akkadian word anaku (or annaku), meaning tin, appears in ancient Near Eastern texts from the third millennium BCE and may have influenced early trade-related nomenclature through Mesopotamian commerce, potentially linking to the Germanic form via ancient Mediterranean exchanges.[24] The chemical symbol Sn for tin originates from the Latin stannum, the Roman term for the metal, which first denoted an alloy of silver and lead before specifically referring to tin by the fourth century CE.[23] This Latin word likely derives from Celtic or pre-Indo-European roots, possibly related to words meaning "stagnant" or "dripping," reflecting tin's low melting point, as in Old Celtic stag- (compare Welsh ystain and Breton stean for tin).[25] In ancient Greek, tin was known as kassíteros, a term possibly borrowed from Phoenician cassiterid or kasdir, referring to tin ore or the mythical "Tin Islands" (Cassiterides) associated with distant sources like Britain or Iberia, highlighting early Phoenician trade networks in the metal.[25][26] Other ancient languages employed distinct terms for tin, often tied to its alloying properties or regional availability. In Sanskrit, trapu denoted tin in Vedic texts like the Atharvaveda (circa 1200–1000 BCE), possibly derived from the root trap, implying something "shining" or "easily worked" due to the metal's malleability.[27] In Chinese, the character 錫 (xī) has signified tin since ancient times, with its Old Chinese pronunciation reconstructed as /*s-lˁek/, evolving from pictographic representations of metal objects and integrated into early metallurgical terminology. Ancient texts frequently confused tin with lead, as evidenced by Roman naturalist Pliny the Elder's description of tin as plumbum album ("white lead"), distinguishing it from the darker plumbum nigrum (lead) based on appearance rather than composition.[23] This etymological overlap underscores the challenges in early mineral classification, where tin's silvery luster led to its association with refined or "white" forms of lead-like metals.[28]Historical development
The earliest evidence of tin's use appears in the Bronze Age, around 3000 BCE, with tin-bronze artifacts from sites in Mesopotamia, including an axehead from Ur dating to the mid-3rd millennium BCE, marking the advent of intentional alloying of tin with copper to create stronger tools and artifacts.[29][30] This innovation spread to Europe during the same period, where tin-bronze enabled advancements in metallurgy and weaponry across early civilizations.[31] In Southeast Asia, tin-bronze artifacts from sites like Ban Chiang in Thailand date to around 2000 BCE, demonstrating independent development of bronze technology in the region.[24][32] Ancient tin sources included the mines of Cornwall in the United Kingdom, exploited from around 2000 BCE during the Bronze Age, providing a vital resource for European bronze production.[33] Trade networks facilitated the distribution of Cornish tin to the Mediterranean, with the Phoenicians playing a key role in maritime commerce from the late second millennium BCE, transporting tin from the "Cassiterides" (likely Cornwall) to regions like the Levant for alloying into bronze.[34] During the medieval period, alchemical studies in both Arabic and European traditions examined tin's properties, classifying it as a base metal associated with Jupiter and exploring its transmutation potential alongside other elements like lead.[35] Advancements in alloying led to the refinement of bell metal, a high-tin bronze (typically 20–22% tin and 78–80% copper) that produced resonant tones for ecclesiastical bells, requiring precise separation and proportioning of tin from copper ores to achieve durability and acoustic quality.[36] In the 8th century, Chinese scholar Lu Yu referenced tin in the context of tea ware, advocating for its use in certain utensils within his comprehensive guide to tea culture, The Classic of Tea. In Europe, 16th-century physician and alchemist Paracelsus advanced medicinal applications of tin, introducing powdered tin as an anthelmintic to treat parasitic infections, integrating it into iatrochemistry as part of his chemical approach to pharmacology.[37] The 19th century marked tin's industrialization, with Peter Durand's 1810 patent for using tinplate to create sealed cans for food preservation, revolutionizing canning and enabling global trade in preserved goods.[38] This era also saw the adoption of electrolytic refining methods in the early 20th century for higher-purity tin production.Occurrence and extraction
Natural occurrence
Tin is a relatively rare element in the Earth's crust, with an average abundance of 2.2 parts per million (ppm), ranking it as the 49th most abundant element.[39] It occurs primarily in the form of the oxide mineral cassiterite (SnO₂), which is the principal source of tin worldwide, alongside secondary minerals such as stannite (Cu₂FeSnS₄).[40] These minerals form in various geological settings, including pegmatites, greisens, and skarns associated with late-stage granite intrusions. As a chalcophile element, tin preferentially binds with sulfur and other chalcophile elements during magmatic processes, often concentrating in hydrothermal systems linked to granitic magmatism.[41] Major tin ore deposits are concentrated in specific regions, notably Southeast Asia (including Indonesia and China), South America (particularly Bolivia), and Africa (such as Nigeria), where they are hosted in these igneous-related environments.[41] In seawater, tin concentrations are extremely low, typically around 0.3 parts per trillion (ppt), reflecting its limited solubility and mobility in aqueous systems. Globally, tin reserves are estimated at more than 4.2 million metric tons as of 2024, predominantly in the aforementioned regions.[3] Beyond Earth, tin is present in extraterrestrial materials, including meteorites such as iron meteorites where it occurs at concentrations of 0.8 to 20 ppm, and in lunar samples that contain fragments of iron meteorites.[42] Its cosmic abundance is approximately 1.4 atoms of tin per million silicon atoms, consistent with s-process nucleosynthesis in stellar environments.[43]Mining and concentration
Tin mining involves both primary and secondary deposit extraction methods, tailored to the geological context of cassiterite (SnO₂), the principal ore mineral. Primary deposits, often associated with granitic intrusions, are typically extracted using open-pit or underground techniques. Open-pit mining is employed for near-surface, low-grade vein or disseminated ores, such as those in pegmatites, where large volumes of overburden are removed to access the mineralized zones.[44] Underground mining predominates for deeper, higher-grade lode deposits, involving shaft sinking, tunneling, and stoping to follow ore veins, as seen in hard-rock operations.[45] Secondary deposits, formed by erosion and transport of primary ores, are primarily alluvial placers rich in cassiterite due to its high density (6.8–7.1 g/cm³). Placer mining targets these concentrations in river gravels, stream beds, and coastal sands, using methods like dredging, gravel pumping, and hydraulic monitoring to dislodge and collect heavy minerals. In Malaysia, a historical hub for tin production, placer deposits in alluvial gravels are exploited via open-pit gravel pump systems, where high-pressure water jets break down the ore-bearing sediments for sluicing and panning.[46] Following extraction, tin ores undergo concentration to upgrade the low-grade feed into a viable feedstock, typically achieving 60–70% Sn content in the final concentrate. Gravity separation is the cornerstone method, leveraging cassiterite's density to separate it from lighter gangue minerals like quartz and feldspar. This involves multi-stage processing with jigs for coarse particles (>200 μm), shaking tables for medium sizes (40–800 μm), and spirals for fines, often recovering 70–90% of the tin in primary operations.[47] For complex ores with sulfide associations, such as stannite, froth flotation is applied after grinding to liberate minerals, using collectors like xanthates to float tin sulfides into a concentrate.[48] Magnetic separation complements these steps by removing ferromagnetic impurities like magnetite and ilmenite, enhancing concentrate purity.[49] Ore grades in major deposits typically range from 0.5% to 2% Sn, with recovery rates of 70–90% depending on ore type and processing efficiency.[50] Prominent tin mines illustrate these practices and their challenges. The Renison Bell mine in Tasmania, Australia, operates as an underground facility targeting high-grade vein deposits in a slate host rock, producing over 680,000 tonnes of ore annually and accounting for about 67% of Australia's tin reserves.[45] In China, the Dachang orefield in Guangxi represents a major polymetallic tin province, with underground mining of stratabound and vein deposits yielding significant output amid karst terrain.[51] Environmental concerns, particularly tailings management, plague these operations; tailings from concentration often contain residual heavy metals like arsenic and lead, leading to soil and water contamination if not properly impounded. In Malaysian placer sites, unregulated tailings processing has elevated naturally occurring radioactive materials (NORM) in sediments, posing radiological hazards to nearby ecosystems and communities.[52] At Dachang, mining effluents have resulted in heavy metal pollution in farmland soils, with elevated levels of Cd, Pb, and As exceeding national standards and contributing to ecological risks.[53] Recent developments highlight operational improvements in tin mining. In 2025, Andrada Mining's Uis Mine in Namibia reported a 17% year-on-year increase in tin concentrate production to 453 tonnes for the quarter ended August 31, driven by enhanced throughput, better plant utilization, and recovery rates rising to 60%.[54]Production and refining
Smelting processes
The primary method for smelting tin involves the carbothermic reduction of cassiterite (SnO₂) concentrate in a reverberatory furnace using carbon as the reductant, following the reaction SnO₂ + 2C → Sn + 2CO at temperatures of 1200–1300°C.[55] This process occurs in two stages: the first reduces the oxide to produce crude tin metal and iron-rich slag, while the second refines the slag at around 1400°C with additional carbon and fluxes like lime to recover residual tin, resulting in secondary slag with 1-2% tin content.[56][55] The resulting crude tin achieves approximately 99% purity, with byproducts such as iron silicates managed through slag fuming or recycling.[55] Impurities like arsenic (As) and antimony (Sb) are removed prior to or during smelting via oxidative roasting, which volatilizes them as oxides, ensuring the crude metal meets initial quality standards.[56] Secondary processes, such as electrolytic refining, may follow to attain higher purity levels, though they are distinct from primary smelting.[55] Historically, tin smelting relied on charcoal as the carbon source in reverberatory furnaces, but modern operations have shifted to coke or electric furnaces for improved energy efficiency and reduced emissions.[57] This transition has enhanced overall process sustainability, with global smelting capacity supporting annual production of around 300,000 tonnes of tin metal as of 2024.[3] In 2024, global refined tin production declined by about 2.7% to 371,200 metric tons, largely due to supply disruptions in Indonesia from government crackdowns on illegal mining, which reduced the country's output by 30.7% to 49,900 metric tons.[58]Industrial refining
Industrial refining of tin involves purifying crude tin metal obtained from smelting processes to achieve commercial grades suitable for various applications. The primary methods include fire refining and electrolytic refining, each targeting specific impurities such as iron, arsenic, copper, and sulfur. Fire refining, the most common approach, utilizes liquation and oxidation techniques to separate and remove contaminants. In liquation, crude tin is heated to its melting point (around 232°C) while higher-melting impurities like iron and arsenic remain solid and are separated as slag, leveraging differences in melting points.[59] Oxidation follows, where air is bubbled through the molten tin to form oxides of impurities such as copper and sulfur, which float to the surface as dross and are skimmed off, yielding tin with up to 99.85% purity.[59] Electrolytic refining is employed for higher purity levels, particularly when fire refining cannot adequately remove certain impurities. In this process, impure tin anodes are dissolved in an acidic electrolyte, typically hydrochloric acid (HCl), under direct current, with pure tin depositing on starter cathodes made of high-purity tin sheets. Impurities like iron, copper, and arsenic either remain in solution or form an anode slime, achieving tin purity of 99.99%.[60] This method is especially useful for producing tin for electronics and specialty alloys. Following purification, alloying occurs on-site for applications like solders, where elements such as lead, silver, or bismuth are added to the molten refined tin to meet specific compositions, such as 63% tin-37% lead for electronics soldering. For ultra-pure tin required in semiconductors and advanced materials, vacuum distillation is applied, heating the metal in a vacuum to volatilize and separate residual impurities like lead and antimony based on vapor pressure differences, resulting in 99.999% purity or higher.[61] Byproduct recovery is integrated into refining to enhance efficiency and reduce waste. During fire and electrolytic refining, slags and anode slimes containing antimony and bismuth are collected and processed via leaching or smelting to recover these valuable metals, with antimony often extracted through sulfide leaching and bismuth via precipitation, contributing to overall economic viability.[59] Refined tin adheres to international standards for quality assurance. The ISO 9453 standard specifies sampling methods for spectrometric analysis of tin ingots, while commercial grades typically meet 99.85% purity for plating and general use, and 99.99% for high-performance applications, as outlined in ASTM B339 for Grade A tin. Recycling is increasingly integrated, with secondary production from scrap supporting primary refining; in the US, tin recovered from scrap reached 18,000 tons in 2024.[3][62] Major refining facilities are concentrated in leading producer nations. In China, Yunnan Tin Company and Yunnan Chengfeng Non-Ferrous Metals operate large-scale refineries with combined capacities exceeding 100,000 metric tons annually, while in Indonesia, PT Timah manages key plants; however, production fell below 50,000 metric tons in 2024 due to regulatory actions against illegal mining.[63][64] As of 2025, the global tin market remains in a tight balance or small deficit, with low stocks and elevated prices amid ongoing supply concerns.[65][66]Chemical compounds
Inorganic compounds
Tin forms several important inorganic compounds, primarily in the +2 and +4 oxidation states, with halides, oxides, and sulfides being the most studied. These compounds exhibit diverse structures and reactivities, influenced by tin's ability to adopt tetrahedral or octahedral coordination geometries.Halides
The tin halides include prominent chlorides such as tin(II) chloride (SnCl₂) and tin(IV) chloride (SnCl₄). SnCl₄ adopts a tetrahedral structure in the gas phase and is a volatile liquid with a boiling point of approximately 114°C, acting as a strong Lewis acid due to its ability to accept electron pairs and form complexes like the octahedral SnCl₆²⁻ (log₁₀β₆ = 9.83 ± 0.49 at zero ionic strength).[14] It is prepared by direct reaction of tin metal with chlorine gas. In contrast, SnCl₂ features a linear polymeric structure in the solid state or a crystalline form, appearing as a white solid that is highly soluble in water and serves as a reducing agent, forming soluble chlorido complexes such as SnCl₃⁻ and SnCl₄²⁻ (log₁₀β₃ ≈ 3.0, log₁₀β₄ ≈ 3.5).[14] Anhydrous SnCl₂ is synthesized by passing dry hydrogen chloride gas over tin metal, while the dihydrate forms similarly in moist conditions.[67] Both halides hydrolyze in aqueous media, with SnCl₄ reacting vigorously to form oxychlorides and SnCl₂ forming basic salts.[67]Oxides
Tin oxides exist as SnO and SnO₂, reflecting the +2 and +4 oxidation states, respectively. SnO possesses a tetragonal structure similar to litharge (PbO), with lattice parameters a = 3.8029 Å and c = 4.8328 Å, appearing as a blue-black or red solid that is unstable and disproportionates at elevated temperatures (4 SnO → Sn + Sn₃O₄ above 400°C).[14] It exhibits amphoteric behavior, dissolving in acids or bases. SnO₂, known as cassiterite in its mineral form, has a tetragonal rutile structure and is a white, insoluble solid with a high melting point of about 1630°C, widely recognized as an n-type semiconductor with a wide band gap of 3.6 eV and excellent thermal stability.[14][68] Its solubility is low in water (<0.5 μg/mL) but increases in alkaline solutions, forming stannate species.[14]Sulfides
The sulfides of tin include SnS and SnS₂. SnS, occurring naturally as the mineral herzenbergite, adopts an orthorhombic layered structure (Pnma space group) with Sn-S bond lengths of 2.633–2.671 Å and Sn-Sn interlayer distances of 3.48 Å, manifesting as a black solid with p-type semiconducting properties and a band gap of 1.16–1.79 eV.[69] It is insoluble in water but shows increased solubility in acidic media, such as 3.9 × 10⁻⁵ M in 0.1 M HCl at 20°C.[14] SnS₂, historically known as mosaic gold for its golden luster, features a hexagonal layered structure (P3m1 space group) composed of Sn(IV)S₆ octahedra with S-S van der Waals separations of 3.6 Å, appearing as a pale yellow solid that is an n-type semiconductor with a band gap of 2.04–3.30 eV.[69] Its solubility is minimal in neutral conditions (0.14 × 10⁻⁶ M at 18°C) but rises significantly in alkaline environments, forming thiostannate ions like SnS₃²⁻ (log₁₀K ≈ 9.2).[14]Other Compounds
Stannates, such as sodium stannate (Na₂SnO₃), derive from Sn(IV) and form in alkaline solutions where SnO₂ dissolves to yield species like Sn(OH)₆²⁻ (ΔfG° = -1312.625 ± 1.839 kJ/mol), exhibiting octahedral coordination.[14] Orthostannates involve similar tetrahedral or octahedral Sn centers in complex salts. Coordination polymers of tin, including those based on Sn(II) and Sn(IV), assemble via covalent Sn-O or Sn-N bonds with ligands like carboxylates or phosphonates, forming multidimensional networks influenced by ligand bulkiness and tin geometry; examples include Sn₂₁Cl₁₆(OH)₁₄O₆ with intricate hydroxide-bridged structures (ΔfG° = -7611.662 ± 20.29 kJ/mol).[14] Solubility trends among tin inorganic compounds vary with pH and anion presence: SnCl₂ is highly water-soluble, while oxides and sulfides like SnO₂ and SnS are generally insoluble in neutral water but dissolve in acids or bases due to amphoteric nature.[14][67] Sn⁴⁺ ions undergo extensive hydrolysis in aqueous solutions, forming colloidal species such as Sn(OH)₄ or Sn(OH)₃⁺ (log K ≈ -17 for Sn(OH)₃⁻), with colloid formation prominent at neutral pH and suppressed in high-acid or high-ionic-strength media.[14][70]Organotin compounds
Organotin compounds, characterized by at least one covalent carbon-tin bond, are classified according to the number of alkyl or aryl groups (R) bound to the tin atom, typically in the +4 oxidation state. These include tetraorganotins (R₄Sn), which are relatively inert; triorganotins (R₃SnX), known for their bioactivity; diorganotins (R₂SnX₂); and monoorganotins (RSnX₃), where X denotes a halogen or other substituent.[71] This classification reflects variations in stability and reactivity, with tetraorganotins exhibiting minimal reactivity under standard conditions due to the absence of labile ligands.[71] Inorganic tin halides, such as SnCl₄, serve as common precursors for these syntheses.[72] The primary synthesis route for tetraorganotins involves the reaction of tin tetrachloride with Grignard reagents, proceeding as SnCl₄ + 4RMgBr → R₄Sn + 4MgClBr, often conducted in ether solvents to yield high-purity products after hydrolysis and distillation.[72] For tri- and diorganotin halides, redistribution reactions are employed, exemplified by the Kocheshkov disproportionation, where a tetraorganotin reacts with a tin tetrahalide (e.g., R₄Sn + SnX₄ → 2R₂SnX₂) under heating or catalysis, allowing selective formation of mixed derivatives with controlled stoichiometry.[71] These methods enable scalable production, though yields depend on the steric bulk of R groups, with alkyl chains like butyl or methyl being most common.[72] Structurally, four-coordinate organotin(IV) compounds adopt a tetrahedral geometry around the tin atom, with Sn–C bond lengths averaging 2.15 Å, longer than typical C–C bonds (1.54 Å), contributing to enhanced reactivity in certain contexts.[72] In the presence of donor ligands, such as nitrogen or oxygen from chelating groups, tin can achieve hypervalency, expanding to five- or six-coordinate species with trigonal bipyramidal or octahedral arrangements, as seen in adducts like trimethyltin chloride with pyridine.[73] This coordination flexibility arises from the d-orbitals of tin, facilitating intramolecular interactions in chelated systems.[73] Reactivity patterns vary by class: tetraorganotins are stable to hydrolysis but can undergo transmetallation with transition metals, such as in palladium-catalyzed cross-coupling reactions where R groups transfer to organic electrophiles.[71] Triorganotins participate in radical processes, including homolytic cleavage of Sn–C bonds to generate stannyl radicals, useful for initiating polymerization or addition reactions.[71] Toxicity mechanisms, particularly for triorganotins, involve disruption of cellular processes; for instance, tributyltin (TBT, (C₄H₉)₃Sn⁺) inhibits ATPases by binding to the enzyme's active site, blocking proton translocation and impairing energy metabolism, as demonstrated in renal H⁺/K⁺-ATPase assays where inhibition exceeds 50% at micromolar concentrations.[74] This leads to oxidative stress via reactive oxygen species accumulation, exacerbating cellular damage.[74] Representative examples include tributyltin azide ((C₄H₉)₃SnN₃), a triorganotin derivative that exhibits persistent reactivity in azide transfer processes, and dimethyltin dichloride (Me₂SnCl₂), a diorganotin halide synthesized via direct methylation of tin, noted for its role in redistribution equilibria and moderate environmental persistence due to slow hydrolysis rates.[71] Organotin compounds, especially tri- and diorganotins like TBT, demonstrate high environmental persistence, with log Kₒw values around 3.8–4.8 promoting bioaccumulation in sediments and biota, where degradation half-lives can exceed years under anaerobic conditions.[74]Hydrides
Tin hydrides, primarily known in laboratory settings with no significant natural occurrence, encompass covalent compounds featuring tin-hydrogen bonds, the most prominent being stannane (). This colorless, gaseous compound serves as the tin analogue to methane and silane, exhibiting high volatility with a boiling point of -52°C and a melting point of -146°C, and a gas density of 5.4 kg m⁻³.[75] Stannane is notoriously unstable, decomposing spontaneously at room temperature into elemental tin and hydrogen gas (Sn + 2H₂), which limits its handling to cryogenic or inert conditions.[76] Higher homologues, such as oligostannanes and polystannanes with the general formula (e.g., disnane, ), represent catenated structures analogous to alkanes but are even less stable, prone to rapid thermal decomposition.[77] These compounds arise transiently during stannane decomposition or targeted syntheses, forming chains or clusters of Sn-Sn bonds terminated by Sn-H groups, though they lack the persistence seen in lighter group 14 catenates like disilane.[78] Synthesis of stannane typically involves the reduction of tin(IV) chloride () with lithium aluminum hydride () in anhydrous solvents like diglyme or dibutyl ether at low temperatures (-30°C to -70°C), yielding 70-89% with high purity confirmed by FTIR absence of hydrogen halides.[79][76] Oligostannanes can form via disproportionation pathways from stannane or related reductions, often as intermediates in gas-phase or matrix-isolated experiments.[77] First prepared in 1924 by Paneth via hydrolysis of magnesium stannide, modern methods prioritize hydride reductions for controlled production.[76] Key properties include the pyramidal geometry of the stannyl anion (), a reactive intermediate with a lone pair on tin, mirroring phosphine-like structures and enabling nucleophilic behavior in solution.[80] Stannane's volatility suits niche applications like chemical vapor deposition precursors, though its instability (decomposing above -10°C) restricts practical use to specialized contexts such as matrix isolation or plasma-generated species.[81] In contrast to inorganic tin hydrides, organotin hydrides exhibit enhanced thermal stability due to alkyl substituents.[80] Reactivity of stannane centers on facile hydrolysis to tin metal and hydrogen, alongside polymerization to oligostannanes under thermal or photolytic conditions, often via radical intermediates like stannylene ().[76] It is pyrophoric and can deflagrate explosively upon air exposure.[79] Spectroscopic characterization relies on infrared (IR) for Sn-H stretches around 1800-2200 cm⁻¹ and ¹¹⁹Sn NMR for chemical shifts in the -2000 to -2500 ppm range for and derivatives, with mass spectrometry revealing fragments like (68%) and (32%).[79][76]Applications
Alloys and solders
Tin plays a crucial role in various metallic alloys, enhancing properties such as strength, ductility, and corrosion resistance when combined with metals like copper and antimony. One of the most historically significant tin alloys is bronze, typically containing 5–12% tin and the balance copper, which was first developed around 3000 BCE for tools and weapons due to its superior hardness over pure copper.[82] In modern applications, variations like phosphor bronze—incorporating 4–10% tin, 85–95% copper, and 0.01–0.35% phosphorus—offer excellent spring qualities and are widely used in electrical contacts, diaphragms, and bellows for their fatigue resistance and wear properties.[83][84] Solders, low-melting-point alloys essential for joining metals, frequently incorporate high tin content for improved wetting and flow characteristics. The traditional eutectic Sn-Pb solder, composed of 63% tin and 37% lead, melts sharply at 183°C, providing reliable bonds in electronics and plumbing due to its excellent flux compatibility and ability to spread evenly on surfaces by reducing surface tension through oxide removal.[85][86] Lead-free alternatives, driven by environmental regulations, include SAC305 (96.5% tin, 3% silver, 0.5% copper), which exhibits good wetting on copper substrates and mechanical reliability despite a higher melting range of 217–220°C.[87] These solders rely on flux to enhance wetting by preventing oxidation, ensuring strong intermetallic formation at the joint interface.[88] Pewter, a tin-based alloy with 85–99% tin alloyed with copper (up to 2%) and antimony (5–10%), is valued for its malleability and corrosion resistance, making it suitable for tableware, decorative items, and ornamental castings.[89] The high tin content imparts non-toxicity and resistance to acidic foods, while copper and antimony additions improve hardness without compromising luster.[90] The phase behavior of tin-copper alloys is governed by the Cu-Sn phase diagram, which features a eutectic point at approximately 97.5% tin and 2.5% copper (melting at 227°C) and key intermetallic compounds like Cu₆Sn₅ (η-phase), formed at the tin-rich end during solidification.[91] These intermetallics contribute to the alloy's microstructure, influencing joint integrity in solders by providing diffusion barriers and strengthening effects.[92] Tin alloys generally exhibit enhanced mechanical properties, including high ductility that allows deformation without fracture and superior fatigue resistance compared to pure metals, enabling applications in demanding cyclic loading environments like bearings and gears.[93][94] The 2006 EU RoHS Directive (2002/95/EC), effective from July 1, 2006, restricted lead in electrical and electronic equipment to 0.1% by weight, accelerating the shift to tin-rich lead-free solders to mitigate environmental and health risks.[95]Coatings and plating
Tin is primarily applied as a thin protective coating on steel through electrolytic deposition to produce tinplate, where tin layers typically ranging from 0.1 to 1.5 micrometers thick are electroplated onto both sides of cold-rolled low-carbon steel sheets.[96] This modern process, dominant since the 1930s, has superseded the historical hot-dipping method, which entailed immersing individual steel sheets in molten tin to achieve thicker coatings up to 0.1 millimeters but lacked the uniformity and efficiency of electrolytic plating.[96] Electrolytic tinning occurs in continuous lines using acidic electrolyte baths, such as those based on phenolsulfonic acid sulfate or methanesulfonic acid, with current densities of 5 to 65 amperes per square decimeter to deposit pure tin (over 99.9%) while controlling thickness and forming a thin tin-iron alloy intermetallic layer via flow-melting.[96][97][98] Post-plating, passivation is applied through cathodic electrolysis in a sodium dichromate solution (20 grams per liter at 50–85°C), creating a thin chromium oxide film (approximately 0.5 micrograms per square centimeter) that inhibits tin oxidation, enhances lacquer adhesion, and improves corrosion resistance.[96] Tin coatings provide key properties including high corrosion resistance in anaerobic conditions typical of packaged foods, where the tin acts sacrificially by dissolving preferentially to protect the underlying steel from perforation, particularly against acidic contents like fruit juices or tomatoes.[96][99] Additionally, the coatings exhibit excellent solderability owing to tin's low melting point of 232°C and ductility, enabling reliable joints during can fabrication.[96] The foremost application of tinplate is in food and beverage packaging, such as three-piece welded cans, drawn and ironed two-piece cans, and closures, where it combines steel's strength and formability with tin's non-toxicity and barrier properties; this sector accounts for the majority of tinplate production and approximately 11% of global refined tin consumption in 2023.[96][100] Chrome-plated steel, known as tin-free steel (TFS), serves as an alternative for similar packaging by applying a metallic chromium layer (about 100 milligrams per square meter) overlaid with chromium oxide via electrolytic chromic acid treatment, providing comparable corrosion resistance at lower cost without using tin.[101] Tin is also used in the float glass process, where a bath of molten tin supports the glass ribbon to produce flat glass sheets with uniform thickness and surface quality. This application accounts for a portion of global tin consumption in industrial processes, such as the 7% for tinning in U.S. data for 2023.[102] Alternatives including polymer-based lacquers, laminates, and tin-free options have reduced tin demand in plating by offering enhanced barrier properties and sustainability, though tinplate remains preferred for its recyclability and performance in demanding applications.[103][104]Chemical manufacturing
Tin compounds are primarily synthesized industrially from metallic tin through processes such as direct chlorination, where tin metal reacts with chlorine gas to produce tin(IV) chloride (SnCl₄) as an intermediate for further derivatization into other chlorides, oxides, and organotins.[105] This method allows for the production of a range of inorganic tin compounds that serve as feedstocks for chemical manufacturing. Globally, tin use in chemicals accounts for approximately 16% of refined tin consumption, or about 59,000 metric tons in 2023, with significant portions allocated to catalytic and pigment applications.[100] In chemical manufacturing, tin compounds function prominently as catalysts. Stannous chloride (SnCl₂) acts as a Lewis acid catalyst in esterification reactions, such as the conversion of oleic acid to biodiesel esters, where it facilitates protonation and enhances reaction rates under mild conditions.[106] Tin(IV) oxide (SnO₂) is employed in oxidation processes, including the catalytic activation of oxygen for organic transformations, owing to its defect-engineered surface properties that promote selective reactivity.[107] Organotin compounds, particularly dicarboxylates like dibutyltin dilaurate, catalyze urethane formation in polyurethane synthesis by coordinating with isocyanates and alcohols to accelerate nucleophilic addition while minimizing side reactions.[108] Tin compounds also play a key role in pigment production. Tin(IV) oxide serves as an opacifier in ceramic glazes, scattering light to create opaque, glossy white finishes when incorporated at 4-10% levels, a practice rooted in its high refractive index and thermal stability.[109] Historically, lead-tin yellow (Pb₂SnO₄), a lead stannate pigment, was widely used in European oil paintings from the 14th to 18th centuries for vibrant yellow hues in foliage and architectural elements, though its application declined due to lead toxicity concerns.[110] In glass manufacturing, tin(IV) oxide-based electrodes, often antimony-doped for conductivity, are utilized in electric melting furnaces for specialty glasses like lead crystal and optical types, providing corrosion resistance and preventing contamination during high-temperature processing.[111] Byproducts from tin refining, such as tin-bearing slags containing oxides and salts, are integrated into chemical production streams through recovery processes like carbothermic reduction, yielding additional tin compounds for reuse and minimizing waste.[112]Electronics and emerging technologies
Tin plays a pivotal role in electronics through its use in lead-free solders for printed circuit boards (PCBs), which became dominant following the European Union's Restriction of Hazardous Substances (RoHS) directive effective July 1, 2006, mandating the elimination of lead in electronic assemblies to reduce environmental impact.[113] These solders, primarily composed of tin with additions like silver and copper (e.g., SAC alloys), enable reliable interconnections in consumer devices, automotive electronics, and telecommunications equipment, offering improved thermal and mechanical stability over traditional leaded variants while complying with global regulations.[114] In optoelectronics, tin-doped indium oxide (Sn-doped In₂O₃), commonly known as indium tin oxide (ITO), serves as a transparent conductive oxide (TCO) alternative in applications like touchscreens, displays, and solar cells due to its high electrical conductivity and optical transparency exceeding 88% in the visible spectrum.[115] Additionally, tin disulfide (SnS₂) thin films are emerging as non-toxic buffer layers in photovoltaic devices, enhancing charge transport and stability in thin-film solar cells by leveraging their layered structure and suitable band gap for efficient light absorption and carrier collection.[116] For lithium-ion batteries, metallic tin anodes offer a theoretical capacity of 994 mAh/g—significantly higher than graphite's 372 mAh/g—but face challenges from over 250% volume expansion during lithiation, leading to pulverization and capacity fade, which researchers mitigate through nanostructuring and composite designs.[117] Emerging applications include tin halide perovskites, such as formamidinium tin iodide (FASnI₃), which have achieved power conversion efficiencies exceeding 14% in laboratory solar cells as of 2024, positioning them as lead-free alternatives with band gaps around 1.4 eV for tandem photovoltaic architectures.[118] Looking ahead, tin's integration in electronics and energy technologies is projected to drive market growth, with demand potentially increasing by up to 40% by 2030, fueled by advancements in batteries, photovoltaics, and next-generation devices amid the global energy transition.[119]Economy and trade
Market prices
The price of tin has exhibited significant volatility over the past two centuries, influenced by global supply dynamics, technological demand shifts, and geopolitical events. Nominal prices, measured in U.S. dollars per pound, ranged from lows of approximately $0.22 in 1932 during the Great Depression to highs exceeding $23 in March 2022 amid acute supply shortages.[120][121] From the early 1900s through the mid-20th century, prices generally hovered between $0.25 and $1.00 per pound, reflecting stable mining outputs from major producers like Malaysia and Bolivia, before rising sharply in the 1970s due to oil crises and inflation.[120] The 1980s saw a notable trough around $3.83 per pound in 1986, driven by oversupply from expanded production in Southeast Asia.[120] In recent years, tin prices have trended upward, with the London Metal Exchange (LME) serving as the primary benchmark for global pricing. The commodity reached $51,000 per metric ton (approximately $23.14 per pound) in March 2022, fueled by supply chain disruptions including export restrictions from Indonesia and logistical bottlenecks exacerbated by the COVID-19 pandemic.[121] This was surpassed in early 2026, when prices reached a record high of $56,800 per metric ton on January 26, 2026, driven by speculative demand linked to AI technologies and soldering for electronics and datacenters. Prices then moderated amid selloffs, market interventions, and currency effects, with values at $46,458 per metric ton on February 5, 2026 (down 4.26% from the prior day), and $45,845 per metric ton as of February 6, 2026 (down 1.83% from the previous close of $46,700). These latest prices are indicative spot prices influenced by LME data.[122][123] Prices subsequently moderated but remained elevated after the 2022 peak; in 2024, the annual average LME cash price was about $31,400 per metric ton (roughly $14.25 per pound), marking a 13.87% year-over-year increase from 2023.[124] Through the first half of 2025, averages climbed to $32,000–$35,000 per metric ton (about $14.50–$15.88 per pound), supported by persistent tightness.[122] For instance, the October 2024 LME price stood at $32,375 per metric ton, reflecting a 31% year-over-year rise at that point, though subsequent months showed moderation.[125] Key drivers of these trends include supply-side factors such as slow production resumption in key regions like Myanmar—where conflicts and mine suspensions have curtailed output, forcing global smelters to operate below 70% capacity—and global refined tin shortages, alongside robust demand from electronics, photovoltaics, and new energy sectors.[126][127] Demand for tin in soldering for semiconductors, AI hardware, solar panel interconnects, batteries, and electric vehicles has surged, with projections estimating a 40% global increase by 2030. LME inventory levels, which influence spot pricing, have remained low, amplifying volatility as traders respond to these imbalances.[128] As of early 2026, forecasts project tin prices to trade around $55,976 per metric ton by the end of Q1 2026 and approximately $60,730 per metric ton in 12 months, reflecting ongoing structural demand growth in electrification and technology sectors despite short-term volatility.[122] When adjusted for inflation using the U.S. Consumer Price Index, historical tin prices reveal moderated volatility; for example, the 2022 peak equates to about $15.50 per pound in 1992 dollars, while the 1986 low adjusts to roughly $8.00, underscoring a gradual upward trajectory in real terms driven by industrialization and green tech adoption.[129][120]| Period | Average Price (USD/MT) | Key Notes |
|---|---|---|
| 2022 Peak (March) | 51,000 | Supply chain bottlenecks [web:96] |
| 2024 Annual Avg. | 31,400 | 13.87% YoY rise [web:25] |
| 2025 H1 Avg. | 32,000–35,000 | Electronics demand surge [web:20] |
| Oct 2024 | 32,375 | +31% YoY [web:22] |
| January 26, 2026 | 56,800 | Record high [122] |
| February 5, 2026 | 46,458 | Down 4.26% [122] |
| February 6, 2026 | 45,845 | Down 1.83% from 46,700 [123] |
| 2025 Forecast (prior) | 30,000–33,000 | Varying surplus/deficit projections (outdated) [web:41] |