Recent from talks
Nothing was collected or created yet.
Mercury (element)
View on Wikipedia
 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mercury | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Appearance | shiny, silvery liquid | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Standard atomic weight Ar°(Hg) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mercury in the periodic table | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic number (Z) | 80 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group | group 12 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Period | period 6 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Block | d-block | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Xe] 4f14 5d10 6s2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2, 8, 18, 32, 18, 2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase at STP | liquid | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | 234.3210 K (−38.8290 °C, −37.8922 °F) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | 629.88 K (356.73 °C, 674.11 °F) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density (near r.t.) | 13.546 g/cm3 [3] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Triple point | 234.3156 K, 1.65 × 10−7 kPa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Critical point | 1750 K, 172.00 MPa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of fusion | 2.29 kJ/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of vaporization | 59.11 kJ/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar heat capacity | 27.983 J/(mol·K) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Vapor pressure[4]
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Oxidation states | common: +1, +2 −2[5] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electronegativity | Pauling scale: 2.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ionization energies |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic radius | empirical: 151 pm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Covalent radius | 132±5 pm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Van der Waals radius | 155 pm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Natural occurrence | primordial | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal structure | rhombohedral (hR1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lattice constants | ar = 301.06 pm α = 70.529° ah = 347.64 pm ch = 673.20 pm (at triple point)[6] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal expansion | 60.4 µm/(m⋅K) (at 25 °C) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal conductivity | 8.30 W/(m⋅K) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrical resistivity | 961 nΩ⋅m (at 25 °C) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Magnetic ordering | diamagnetic[7] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar magnetic susceptibility | −33.44×10−6 cm3/mol (293 K)[8] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Speed of sound | liquid: 1451.4 m/s (at 20 °C) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Number | 7439-97-6 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Naming | from the planet Mercury, with which it was associated in medieval alchemy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Discovery | Ancient Egyptians (before 1500 BCE) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symbol | "Hg": from its Latin name hydrargyrum, itself from Greek hydrárgyros, 'water-silver' | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes of mercury | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Mercury is a chemical element; it has symbol Hg and atomic number 80. It is commonly known as quicksilver. A heavy, silvery d-block element, mercury is the only metallic element that is known to be liquid at standard temperature and pressure;[a] the only other element that is liquid under these conditions is bromine, one of the halogens, though metals such as caesium, gallium, and rubidium melt just above room temperature.[b]
Mercury occurs in deposits throughout the world mostly as cinnabar (mercuric sulfide). The red pigment vermilion is obtained by grinding natural cinnabar or synthetic mercuric sulfide. Exposure to mercury and mercury-containing organic compounds is toxic to the nervous system, immune system and kidneys of humans and other animals; mercury poisoning can result from exposure to water-soluble forms of mercury (such as mercuric chloride or methylmercury) either directly or through mechanisms of biomagnification.
Mercury is used in thermometers, barometers, manometers, sphygmomanometers, float valves, mercury switches, mercury relays, fluorescent lamps and other devices, although concerns about the element's toxicity have led to the phasing out of—or reduction in the amount of mercury used in—these instruments.[11] It remains in use in scientific research applications and in amalgam for dental restoration in some locales. It is also still used in fluorescent lighting, although the quantity of mercury used is now smaller. Electricity passed through mercury vapor in a fluorescent lamp produces short-wave ultraviolet light, which then causes the phosphor in the tube to fluoresce, making visible light.
Properties
[edit]Physical properties
[edit]
Mercury is a heavy, silvery-white metal that is liquid at room temperature. Compared to other metals, it is a poor conductor of heat, but a fair conductor of electricity.[13]
It has a melting point of −38.83 °C[c] and a boiling point of 356.73 °C,[d][14][15][16] both the lowest of any stable metal, although preliminary experiments on copernicium and flerovium have indicated that they have even lower boiling points.[17] This effect is due to lanthanide contraction and relativistic contraction reducing the orbit radius of the outermost electrons, and thus weakening the metallic bonding in mercury.[14] Upon freezing, the volume of mercury decreases by 3.59% and its density changes from 13.69 g/cm3 when liquid to 14.184 g/cm3 when solid. The coefficient of volume expansion is 181.59 × 10−6 at 0 °C, 181.71 × 10−6 at 20 °C and 182.50 × 10−6 at 100 °C (per °C). Solid mercury is malleable and ductile, and can be cut with a knife.[18]
Chemical properties
[edit]Mercury does not react with most acids, such as dilute sulfuric acid, although oxidizing acids such as concentrated sulfuric acid and nitric acid or aqua regia dissolve it to give sulfate, nitrate, and chloride. Like silver, mercury reacts with atmospheric hydrogen sulfide. Mercury reacts with solid sulfur flakes, which are used in mercury spill kits to absorb mercury (spill kits also use activated carbon and powdered zinc).[19]
Amalgams
[edit]
Mercury dissolves many metals such as gold and silver to form amalgams. Iron is an exception, and iron flasks have traditionally been used to transport the material.[20] Several other first row transition metals (with the exception of manganese, copper and zinc) are also resistant in forming amalgams. Other elements that do not readily form amalgams with mercury include platinum.[21][22] Sodium amalgam is a common reducing agent in organic synthesis, and is also used in high-pressure sodium lamps.
Mercury readily combines with aluminium to form a mercury-aluminium amalgam when the two pure metals come into contact.[citation needed] Since the amalgam destroys the aluminium oxide layer which protects metallic aluminium from oxidizing in-depth (as in iron rusting), even small amounts of mercury can seriously corrode aluminium. For this reason, mercury is not allowed aboard an aircraft under most circumstances because of the risk of it forming an amalgam with exposed aluminium parts in the aircraft.[23]
Mercury embrittlement is the most common type of liquid metal embrittlement, as mercury is a natural component of some hydrocarbon reservoirs and will come into contact with petroleum processing equipment under normal conditions.[24]
Isotopes
[edit]There are seven stable isotopes of mercury, with 202
Hg being the most abundant (29.86%). The longest-lived radioisotopes are 194
Hg with a half-life of 444 years, and 203
Hg with a half-life of 46.612 days. Most of the remaining radioisotopes have half-lives that are less than a day. 206
Hg occurs naturally in tiny traces as an intermediate decay product of 238
U. 199
Hg and 201
Hg are the most often studied NMR-active nuclei, having spins of 1⁄2 and 3⁄2 respectively.[13]
Table of properties
[edit]| Temperature (°C) |
Density (kg/m3) |
Specific heat (kJ/(kg·K)) |
Kinematic viscosity (m2/s) |
Conductivity (W/(m·K)) |
Thermal diffusivity (m2/s) |
Prandtl number | Bulk modulus (K−1) |
|---|---|---|---|---|---|---|---|
| 0 | 13628.22 | 0.1403 | 1.24 × 10−7 | 8.20 | 4.30 × 10−6 | 0.0288 | 0.000181 |
| 20 | 13579.04 | 0.1394 | 1.14 × 10−7 | 8.69 | 4.61 × 10−6 | 0.0249 | 0.000181 |
| 50 | 13505.84 | 0.1386 | 1.04 × 10−7 | 9.40 | 5.02 × 10−6 | 0.0207 | 0.000181 |
| 100 | 13384.58 | 0.1373 | 9.28 × 10−8 | 10.51 | 5.72 × 10−6 | 0.0162 | 0.000181 |
| 150 | 13264.28 | 0.1365 | 8.53 × 10−8 | 11.49 | 6.35 × 10−6 | 0.0134 | 0.000181 |
| 200 | 13144.94 | 0.1570 | 8.02 × 10−8 | 12.34 | 6.91 × 10−6 | 0.0116 | 0.000181 |
| 250 | 13025.60 | 0.1357 | 7.65 × 10−8 | 13.07 | 7.41 × 10−6 | 0.0103 | 0.000183 |
| 315.5 | 12847.00 | 0.1340 | 6.73 × 10−8 | 14.02 | 8.15 × 10−6 | 0.0083 | 0.000186 |
Etymology
[edit]
Hg is the modern chemical symbol for mercury.[27] It is an abbreviation of hydrargyrum, a romanized form of the ancient Greek name for mercury, ὑδράργυρος (hydrargyros). Hydrargyrum (/haɪˈdrɑːrdʒərəm/ hy-DRAR-jər-əm)[28] has also been used in English, though the term is now dated. Hydrargyros is a Greek compound word meaning 'water-silver', from ὑδρ- (hydr-), the root of ὕδωρ (hydor) 'water', and ἄργυρος (argyros) 'silver'.[28] Like the English name quicksilver ('living-silver'), this name was due to mercury's liquid and shiny properties.[29]
The modern English name mercury comes from the planet Mercury. In medieval alchemy, the seven known metals—quicksilver, gold, silver, copper, iron, tin, and lead—were associated with the seven classical planets (Mercury, the sun, moon, Venus, Mars, Jupiter, and Saturn respectively). Quicksilver was associated with the fastest planet, which had been named after the Roman god Mercury, who was associated with speed and mobility. The astrological symbol for the planet became one of the alchemical symbols for the metal, and Mercury became an alternative name for the metal. Mercury is the only metal for which the alchemical planetary name survives, as it was decided it was preferable to quicksilver as a chemical name.[30][31]
History
[edit]Mercury was found in Egyptian tombs that date from 1500 BC;[32] cinnabar, the most common natural source of mercury, has been in use since the Neolithic Age.[33]
In China and Tibet, mercury use was thought to prolong life, heal fractures, and maintain generally good health, although it is now known that exposure to mercury vapor leads to serious adverse health effects.[34] The first emperor of a unified China, Qín Shǐ Huáng Dì—allegedly buried in a tomb that contained rivers of flowing mercury on a model of the land he ruled, representative of the rivers of China—was reportedly killed by drinking a mercury and powdered jade mixture formulated by Qin alchemists intended as an elixir of immortality.[35][36] Khumarawayh ibn Ahmad ibn Tulun, the second Tulunid ruler of Egypt (r. 884–896), known for his extravagance and profligacy, reportedly built a basin filled with mercury, on which he would lie on top of air-filled cushions and be rocked to sleep.[37]
In November 2014 "large quantities" of mercury were discovered in a chamber 60 feet below the 1800-year-old pyramid known as the Temple of the Feathered Serpent, the third-largest pyramid of Teotihuacan, Mexico, along with "jade statues, jaguar remains, a box filled with carved shells and rubber balls".[38] In Lamanai, once a major city of the Maya civilization, a pool of mercury was found under a marker in a Mesoamerican ballcourt.[39][40]
Aristotle recounts that Daedalus made a wooden statue of Aphrodite move by pouring quicksilver in its interior.[41] In Greek mythology Daedalus gave the appearance of voice in his statues using quicksilver. The ancient Greeks used cinnabar (mercury sulfide) in ointments; the ancient Egyptians and the Romans used it in cosmetics. By 500 BC mercury was used to make amalgams (Medieval Latin amalgama, "alloy of mercury") with other metals.[42]
Alchemists thought of mercury as the First Matter from which all metals were formed. They believed that different metals could be produced by varying the quality and quantity of sulfur contained within the mercury. The purest of these was gold, and mercury was called for in attempts at the transmutation of base (or impure) metals into gold, which was the goal of many alchemists.[30]
The mines in Almadén (Spain), Monte Amiata (Italy), and Idrija (now Slovenia) dominated mercury production from the opening of the mine in Almadén 2500 years ago, until new deposits were found at the end of the 19th century.[43]
Beginning in 1558, with the invention of the patio process to extract silver from ore using mercury, mercury became an essential resource in the economy of Spain and its American colonies. Mercury was used to extract silver from the lucrative mines in New Spain and Peru. Initially, the Spanish Crown's mines in Almadén in Southern Spain supplied all the mercury for the colonies.[44] Mercury deposits were discovered in the New World, and more than 100,000 tons of mercury were mined from the region of Huancavelica, Peru, over the course of three centuries following the discovery of deposits there in 1563.[45] In 1786 the main mine at Huancavelica suffered a sudden collapse that killed over 100 persons and greatly reduced the mine's output.[46] Through the legalization of scavenging known as pallaqueo mercury production rose again peaking in 1794–1796.[47] The French Revolutionary Wars disrupted European mercury supply to Spanish America leading to an increasing reliance for the mines in present-day Peru and Bolivia on mercury from Huancavelica but this mines production was clearly by 1799 not enough to supply the demand in the Andean mines.[47] Spain abolished the royal mercury monopoly in 1813.[47]
Mercury poisoning in the mines left many people disabled through the early modern period but mercury itself was not the chief cause of deaths in the mines.[48]
The patio process and later pan amalgamation process continued to create great demand for mercury to treat silver ores until the late 19th century.[45]
Occurrence
[edit]Mercury is an extremely rare element in Earth's crust; it has an average crustal abundance by mass of only 0.08 parts per million (ppm)[49] and is the 66th most abundant element in the Earth's crust.[50] Because it does not blend geochemically with those elements that constitute the majority of the crustal mass, mercury ores can be extraordinarily concentrated considering the element's abundance in ordinary rock. The richest mercury ores contain up to 2.5% mercury by mass, and even the leanest concentrated deposits are at least 0.1% mercury (12,000 times average crustal abundance). It is found either as a native metal (rare) or in cinnabar, metacinnabar, sphalerite, corderoite, livingstonite and other minerals, with cinnabar (HgS) being the most common ore.[51][52] Mercury ores often occur in hot springs or other volcanic regions.[53]

Former mines in Italy, the United States and Mexico, which once produced a large proportion of the world supply, have now been completely mined out or, in the case of Slovenia (Idrija) and Spain (Almadén), shut down due to the fall of the price of mercury. Nevada's McDermitt Mine, the last mercury mine in the United States, closed in 1992. The price of mercury has been highly volatile over the years and in 2006 was $650 per 76-pound (34.46 kg) flask.[54]
Mercury is extracted by heating cinnabar in a current of air and condensing the vapor. The equation for this extraction is:
- HgS + O2 → Hg + SO2

In 2020, China was the top producer of mercury, providing 88% of the world output (2200 out of 2500 tonnes), followed by Tajikistan (178 t), Russia (50 t) and Mexico (32 t).[55]
| Country | Production |
|---|---|
| World | 1,200 |
| 1,000 | |
| 100 | |
| 30 | |
| 30 | |
| 20 | |
| 15 | |
| 10 | |
| 6 | |
| 2 |
Because of the high toxicity of mercury, both the mining of cinnabar and refining for mercury are hazardous and historic causes of mercury poisoning.[58] In China, prison labor was used by a private mining company as recently as the 1950s to develop new cinnabar mines. Thousands of prisoners were used by the Luo Xi mining company to establish new tunnels.[59] Worker health in functioning mines is at high risk.
A newspaper claimed that an unidentified European Union directive calling for energy-efficient lightbulbs to be made mandatory by 2012 encouraged China to re-open cinnabar mines to obtain the mercury required for CFL bulb manufacture. Environmental dangers have been a concern, particularly in the southern cities of Foshan and Guangzhou, and in Guizhou province in the southwest.[59]
Abandoned mercury mine processing sites often contain very hazardous waste piles of roasted cinnabar calcines. Water run-off from such sites is a recognized source of ecological damage. Former mercury mines may be suited for constructive re-use; for example, in 1976 Santa Clara County, California purchased the historic Almaden Quicksilver Mine and created a county park on the site, after conducting extensive safety and environmental analysis of the property.[60][61]
Chemistry
[edit]All known mercury compounds exhibit one of two positive oxidation states: I and II. Experiments have failed to unequivocally demonstrate any higher oxidation states: both the claimed 1976 electrosynthesis of an unstable Hg(III) species and 2007 cryogenic isolation of HgF4 have disputed interpretations and remain difficult (if not impossible) to reproduce.[62]
Compounds of mercury(I)
[edit]Unlike its lighter neighbors, cadmium and zinc, mercury usually forms simple stable compounds with metal-metal bonds. Most mercury(I) compounds are diamagnetic and feature the dimeric cation, Hg2+
2. Stable derivatives include the chloride and nitrate. In aqueous solution of a mercury(I) salt, slight disproportion of Hg2+
2 into Hg and Hg2+
results in >0.5% of dissolved mercury existing as Hg2+
. In these solutions, complexation of the Hg2+
with addition of ligands such as cyanide causes disproportionation to go to completion, with all Hg2+
2 precipitating as elemental mercury and insoluble mercury(II) compounds (e.g. mercury(II) cyanide if cyanide is used as the ligand).[63] Mercury(I) chloride, a colorless solid also known as calomel, is really the compound with the formula Hg2Cl2, with the connectivity Cl-Hg-Hg-Cl. It reacts with chlorine to give mercury(II) chloride, which resists further oxidation. Mercury(I) hydride, a colorless gas, has the formula HgH, containing no Hg-Hg bond; however, the gas has only ever been observed as isolated molecules.[64]
Indicative of its tendency to bond to itself, mercury forms mercury polycations, which consist of linear chains of mercury centers, capped with a positive charge. One example is Hg
3(AsF
6)
2 containing the Hg2+
3 cation.[65]
Compounds of mercury(II)
[edit]Mercury(II) is the most common oxidation state and is the main one in nature as well. All four mercuric halides are known and have been demonstrated to form linear coordination geometry, despite mercury's tendency to form tetrahedral molecular geometry with other ligands. This behavior is similar to the Ag+ ion. The best known mercury halide is mercury(II) chloride, an easily sublimating white solid.[66]
Mercury(II) oxide, the main oxide of mercury, arises when the metal is exposed to air for long periods at elevated temperatures. It reverts to the elements upon heating near 400 °C, as was demonstrated by Joseph Priestley in an early synthesis of pure oxygen.[19] Hydroxides of mercury are poorly characterized, as attempted isolation studies of mercury(II) hydroxide have yielded mercury oxide instead.[67]
Being a soft metal, mercury forms very stable derivatives with the heavier chalcogens. Preeminent is mercury(II) sulfide, HgS, which occurs in nature as the ore cinnabar and is the brilliant pigment vermilion. Like ZnS, HgS crystallizes in two forms, the reddish cubic form and the black zinc blende form.[13] The latter sometimes occurs naturally as metacinnabar.[52] Mercury(II) selenide (HgSe) and mercury(II) telluride (HgTe) are known, these as well as various derivatives, e.g. mercury cadmium telluride and mercury zinc telluride being semiconductors useful as infrared detector materials.[68]
Mercury(II) salts form a variety of complex derivatives with ammonia. These include Millon's base (Hg2N+), the one-dimensional polymer (salts of HgNH+
2)
n), and "fusible white precipitate" or [Hg(NH3)2]Cl2. Known as Nessler's reagent, potassium tetraiodomercurate(II) (K
2HgI
4) is still occasionally used to test for ammonia owing to its tendency to form the deeply colored iodide salt of Millon's base.[69]
Mercury fulminate is a detonator widely used in explosives.[13]
Organomercury compounds
[edit]Organic mercury compounds are historically important but are of little industrial value in the western world. Mercury(II) salts are a rare example of simple metal complexes that react directly with aromatic rings. Organomercury compounds are always divalent and usually two-coordinate and linear geometry. Unlike organocadmium and organozinc compounds, organomercury compounds do not react with water. They usually have the formula HgR2, which are often volatile, or HgRX, which are often solids, where R is aryl or alkyl and X is usually halide or acetate. Methylmercury, a generic term for compounds with the formula CH3HgX, is a dangerous family of compounds that are often found in polluted water.[70] They arise by a process known as biomethylation.
Applications
[edit]
Mercury is used primarily for the manufacture of industrial chemicals or for electrical and electronic applications. It is used in some liquid-in-glass thermometers, especially those used to measure high temperatures. A still increasing amount is used as gaseous mercury in fluorescent lamps, while most of the other applications are slowly being phased out due to health and safety regulations. In some applications, mercury is replaced with less toxic but considerably more expensive Galinstan alloy.[71]
Medicine
[edit]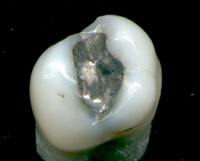
Historical and folk
[edit]Mercury and its compounds have been used in medicine, although they are much less common today than they once were, now that the toxic effects of mercury and its compounds are more widely understood. An example of the early therapeutic application of mercury was published in 1787 by James Lind.[72]
The first edition of The Merck Manuals (1899) featured many then-medically relevant mercuric compounds, such as mercury-ammonium chloride, yellow mercury proto-iodide, calomel, and mercuric chloride, among others.[73]
Mercury in the form of one of its common ores, cinnabar, is used in various traditional medicines, especially in traditional Chinese medicine. Review of its safety has found that cinnabar can lead to significant mercury intoxication when heated, consumed in overdose, or taken long term, and can have adverse effects at therapeutic doses, though effects from therapeutic doses are typically reversible. Although this form of mercury appears to be less toxic than other forms, its use in traditional Chinese medicine has not yet been justified, as the therapeutic basis for the use of cinnabar is not clear.[74]
Mercury(I) chloride (also known as calomel or mercurous chloride) has been used in traditional medicine as a diuretic, topical disinfectant, and laxative. Mercury(II) chloride (also known as mercuric chloride or corrosive sublimate) was once used to treat syphilis (along with other mercury compounds), although it is so toxic that sometimes the symptoms of its toxicity were confused with those of the syphilis it was believed to treat.[75] It is also used as a disinfectant. Blue mass, a pill or syrup in which mercury is the main ingredient, was prescribed throughout the 19th century for numerous conditions including constipation, depression, child-bearing and toothaches.[76] In the early 20th century, mercury was administered to children yearly as a laxative and dewormer, and it was used in teething powders for infants. The mercury-containing organohalide merbromin (sometimes sold as Mercurochrome) is still widely used but has been banned in some countries, such as the U.S.[77]
Contemporary
[edit]Mercury is an ingredient in dental amalgams.[78]
Thiomersal (called Thimerosal in the United States) is an organic compound used as a preservative in vaccines, although this use is in decline.[79] Although it was widely speculated that this mercury-based preservative could cause or trigger autism in children, no evidence supports any such link.[80] Nevertheless, thiomersal has been removed from, or reduced to trace amounts in, all U.S. vaccines recommended for children 6 years of age and under, with the exception of the inactivated influenza vaccine.[79] Merbromin (Mercurochrome), another mercury compound, is a topical antiseptic used for minor cuts and scrapes in some countries. Today, the use of mercury in medicine has greatly declined in all respects, especially in developed countries.[81]
Mercury is still used in some diuretics, although substitutes such as thiazides now exist for most therapeutic uses.[82] In 2003, mercury compounds were found in some over-the-counter drugs, including topical antiseptics, stimulant laxatives, diaper-rash ointment, eye drops, and nasal sprays. The FDA has "inadequate data to establish general recognition of the safety and effectiveness" of the mercury ingredients in these products.[83]
Production of chlorine and caustic soda
[edit]Chlorine is produced from sodium chloride (common salt, NaCl) using electrolysis to separate metallic sodium from chlorine gas. Usually salt is dissolved in water to produce a brine. By-products of any such chloralkali process are hydrogen (H2) and sodium hydroxide (NaOH), which is commonly called caustic soda or lye. By far the largest use of mercury[84][85] in the late 20th century was in the mercury cell process (also called the Castner-Kellner process) where metallic sodium is formed as an amalgam at a cathode made from mercury; this sodium is then reacted with water to produce sodium hydroxide.[86] Many of the industrial mercury releases of the 20th century came from this process, although modern plants claim to be safe in this regard.[85] From the 1960s onward, the majority of industrial plants moved away from mercury cell processes towards diaphragm cell technologies to produce chlorine, though 11% of the chlorine made in the United States was still produced with the mercury cell method as of 2005.[87]
Laboratory uses
[edit]Thermometers
[edit]Thermometers containing mercury were invented in the early 18th century by Daniel Gabriel Fahrenheit, though earlier attempts at making temperature-measuring instruments filled with quicksilver had been described in the 1650s.[88]: 23 Fahrenheit's mercury thermometer was based on an earlier design that used alcohol rather than mercury; the mercury thermometer was significantly more accurate than those using alcohol.[89] From the early 21st century onwards, the use of mercury thermometers has been declining, and mercury-containing instruments have been banned in many jurisdictions following the 1998 Protocol on Heavy Metals.[90][91] Modern alternatives to mercury thermometers include resistance thermometers, thermocouples, and thermistor sensors that output to a digital display.[92]
Mirrors
[edit]Some transit telescopes use a basin of mercury to form a flat and absolutely horizontal mirror, useful in determining an absolute vertical or perpendicular reference. Concave horizontal parabolic mirrors may be formed by rotating liquid mercury on a disk, the parabolic form of the liquid thus formed reflecting and focusing incident light. Such liquid-mirror telescopes are cheaper than conventional large mirror telescopes by up to a factor of 100, but the mirror cannot be tilted and always points straight up.[93][94][95]
Electrochemistry
[edit]Liquid mercury is part of a popular secondary reference electrode (called the calomel electrode) in electrochemistry as an alternative to the standard hydrogen electrode. The calomel electrode is used to work out the electrode potential of half cells.[96] The triple point of mercury, −38.8344 °C, is a fixed point used as a temperature standard for the International Temperature Scale (ITS-90).[13]
Polarography and crystallography
[edit]In polarography, both the dropping mercury electrode[97] and the hanging mercury drop electrode[98] use elemental mercury. This use allows a new uncontaminated electrode to be available for each measurement or each new experiment.
Mercury-containing compounds are also of use in the field of structural biology. Mercuric compounds such as mercury(II) chloride or potassium tetraiodomercurate(II) can be added to protein crystals in an effort to create heavy atom derivatives that can be used to solve the phase problem in X-ray crystallography via isomorphous replacement or anomalous scattering methods.[99]
Niche uses
[edit]Gaseous mercury is used in mercury-vapor lamps, fluorescent lamps, and some neon signs. Those low-pressure lamps emit very spectrally narrow lines, which are traditionally used in optical spectroscopy for calibration of spectral position. Commercial calibration lamps are sold for this purpose; reflecting a fluorescent ceiling light into a spectrometer is a common calibration practice.[100] Gaseous mercury is also found in some electron tubes, including ignitrons, thyratrons, and mercury arc rectifiers.[101] It is also used in specialist medical care lamps for skin tanning and disinfection.[102] Gaseous mercury is added to cold cathode argon-filled lamps to increase the ionization and electrical conductivity. An argon-filled lamp without mercury will have dull spots and will fail to light correctly. Lighting containing mercury can be bombarded/oven pumped only once. When added to neon filled tubes, inconsistent red and blue spots are produced in the light emissions until the initial burning-in process is completed; eventually it will light a consistent dull off-blue color.[103]
-
The deep violet glow of a mercury vapor discharge in a germicidal lamp, whose spectrum is rich in invisible ultraviolet radiation.
-
Skin tanner containing a low-pressure mercury vapor lamp and two infrared lamps, which act both as light source and electrical ballast
-
Assorted types of fluorescent lamps.
-
The miniaturized Deep Space Atomic Clock is a linear ion-trap-based mercury ion clock, designed for precise and real-time radio navigation in deep space.
The Deep Space Atomic Clock (DSAC) under development by the Jet Propulsion Laboratory utilises mercury in a linear ion-trap-based clock. The novel use of mercury permits the creation of compact atomic clocks with low energy requirements ideal for space probes and Mars missions.[104]
Skin whitening
[edit]Mercury is effective as an active ingredient in skin whitening compounds used to depigment skin.[105] The Minamata Convention on Mercury limits the concentration of mercury in such whiteners to 1 part per million. However, as of 2022, many commercially sold whitener products continue to exceed that limit, and are considered toxic.[106]
Firearms
[edit]Mercury(II) fulminate is a primary explosive, which has mainly been used as a primer of a cartridge in firearms throughout the 19th and 20th centuries.[107]
Mining
[edit]Mercury is used in illegal gold mining to help separate gold particles from a mixture of sand or gravel and water.[108] Small gold particles may form mercury-gold amalgam and therefore increase the gold recovery rates.[13] The use of mercury causes a severe pollution problem in places such as Ghana.[108]
Historic uses
[edit]

Many historic applications made use of the peculiar physical properties of mercury, especially as a dense liquid and a liquid metal:
- Quantities of liquid mercury ranging from 90 to 600 grams (3.2 to 21.2 oz) have been recovered from elite Maya tombs (100–700 AD)[38] or ritual caches at six sites. This mercury may have been used in bowls as mirrors for divinatory purposes. Five of these date to the Classic Period of Maya civilization (c. 250–900) but one example predated this.[109]
- In Islamic Spain, it was used for filling decorative pools. Later, the American artist Alexander Calder built a mercury fountain for the Spanish Pavilion at the 1937 World Exhibition in Paris. The fountain is now on display at the Fundació Joan Miró in Barcelona.[110]
- The Fresnel lenses of old lighthouses used to float and rotate in a bath of mercury which acted like a bearing.[111]
- Mercury sphygmomanometers, barometers, diffusion pumps, coulometers, and many other laboratory instruments took advantage of mercury's properties as a very dense, opaque liquid with a nearly linear thermal expansion.[112]
- As an electrically conductive liquid, it was used in mercury switches (including home mercury light switches installed prior to 1970), tilt switches used in old fire detectors and in some home thermostats.[113]
- Owing to its acoustic properties, mercury was used as the propagation medium in delay-line memory devices used in early digital computers of the mid-20th century, such as the SEAC computer.[114]
- In 1911, Heike Kamerlingh Onnes discovered superconductivity through the cooling of mercury below 4 kelvin shortly after the discovery and production of liquid helium.[115] Its superconductive properties were later determined to be unusual compared to other later-discovered superconductors, such as the more popular niobium alloys.[116][117]
- Experimental mercury vapor turbines were installed to increase the efficiency of fossil-fuel electrical power plants.[118] The South Meadow power plant in Hartford, CT employed mercury as its working fluid, in a binary configuration with a secondary water circuit, for a number of years starting in the late 1920s in a drive to improve plant efficiency. Several other plants were built, including the Schiller Station in Portsmouth, NH, which went online in 1950. The idea did not catch on industry-wide due to the weight and toxicity of mercury, as well as the advent of supercritical steam plants in later years.[119][120]
- Similarly, liquid mercury was used as a coolant for some nuclear reactors; however, sodium is proposed for reactors cooled with liquid metal, because the high density of mercury requires much more energy to circulate as coolant.[121]
- Mercury was a propellant for early ion engines in electric space propulsion systems. Advantages were mercury's high molecular weight, low ionization energy, low dual-ionization energy, high liquid density and liquid storability at room temperature. Disadvantages were concerns regarding environmental impact associated with ground testing and concerns about eventual cooling and condensation of some of the propellant on the spacecraft in long-duration operations. The first spaceflight to use electric propulsion was a mercury-fueled ion thruster developed at NASA Glenn Research Center and flown on the Space Electric Rocket Test "SERT-1" spacecraft launched by NASA at its Wallops Flight Facility in 1964. The SERT-1 flight was followed up by the SERT-2 flight in 1970. Mercury and caesium were preferred propellants for ion engines until Hughes Research Laboratory performed studies finding xenon gas to be a suitable replacement. Xenon is now the preferred propellant for ion engines, as it has a high molecular weight, little or no reactivity due to its noble gas nature, and high liquid density under mild cryogenic storage.[122][123]
Other applications made use of the chemical properties of mercury:
- The mercury battery is a non-rechargeable electrochemical battery, a primary cell, that was common in the middle of the 20th century. It was used in a wide variety of applications and was available in various sizes, particularly button sizes. Its constant voltage output and long shelf life gave it a niche use for camera light meters and hearing aids. The mercury cell was effectively banned in most countries in the 1990s due to concerns about the mercury contaminating landfills.[124]
- Mercury was used for preserving wood, developing daguerreotypes, silvering mirrors,[125] anti-fouling paints,[126] herbicides,[127] interior latex paint, handheld maze games, cleaning, and road leveling devices in cars. Mercury compounds have been used in antiseptics, laxatives, antidepressants, and in antisyphilitics.[76] Mercury has been replaced with safer compounds in most, if not all, of these applications.
- It was allegedly used by allied spies to sabotage Luftwaffe planes: a mercury paste was applied to bare aluminium, causing the metal to rapidly corrode; this would cause structural failures.[128]
- Mercury was once used as a gun barrel bore cleaner.[129][130]
- From the mid-18th to the mid-19th centuries, a process called "carroting" was used in the making of felt hats. Animal skins were rinsed in an orange solution (the term "carroting" arose from this color) of the mercury compound mercuric nitrate, Hg(NO3)2.[131] This process separated the fur from the pelt and matted it together. This solution and the vapors it produced were highly toxic. The United States Public Health Service banned the use of mercury in the felt industry in December 1941. The psychological symptoms associated with mercury poisoning inspired the phrase "mad as a hatter".[132] Lewis Carroll's "Mad Hatter" in his book Alice's Adventures in Wonderland was a play on words based on the older phrase, but the character himself does not exhibit symptoms of mercury poisoning.[133]
- Historically, mercury was used extensively in hydraulic gold mining (see #Mining. Large-scale use of mercury stopped in the 1960s. However, mercury is still used in small scale, often clandestine, gold prospecting. It is estimated that 45,000 metric tons of mercury used in California for placer mining have not been recovered.[134] Mercury was also used in silver mining to extract the metal from ore through the patio process.[135]
Toxicity and safety
[edit]| Hazards | |
|---|---|
| GHS labelling: | |
  
| |
| Danger | |
| H330, H360D, H372, H410 | |
| P201, P233, P260, P273, P280, P304, P308, P310, P313, P340, P391, P403[136] | |
| NFPA 704 (fire diamond) | |
| Part of a series on |
| Pollution |
|---|
 |
Due to its physical properties and relative chemical inertness, liquid mercury is absorbed very poorly through intact skin and the gastrointestinal tract.[137] Mercury vapor is the primary hazard of elemental mercury. As a result, containers of mercury are securely sealed to avoid spills and evaporation. Heating of mercury, or of compounds of mercury that may decompose when heated, should be carried out with adequate ventilation in order to minimize exposure to mercury vapor. The most toxic forms of mercury are its organic compounds, such as dimethylmercury and methylmercury. Mercury can cause both chronic and acute poisoning.[138][139]
Releases in the environment
[edit]
Preindustrial deposition rates of mercury from the atmosphere may be about 4 ng per 1 L of ice deposited. Volcanic eruptions and related natural sources are responsible for approximately half of atmospheric mercury emissions.[140]
Atmospheric mercury contamination in outdoor urban air at the start of the 21st century was measured at 0.01–0.02 μg/m3. A 2001 study measured mercury levels in 12 indoor sites chosen to represent a cross-section of building types, locations and ages in the New York area. This study found mercury concentrations significantly elevated over outdoor concentrations, at a range of 0.0065 – 0.523 μg/m3. The average was 0.069 μg/m3.[141]
Half of mercury emissions are attributed to mankind. The sources can be divided into the following estimated percentages:[142]
- 65% from stationary combustion, of which coal-fired power plants are the largest aggregate source (40% of U.S. mercury emissions in 1999). This includes power plants fueled with gas where the mercury has not been removed. Emissions from coal combustion are between one and two orders of magnitude higher than emissions from oil combustion, depending on the country.[142]
- 11% from gold production. The three largest point sources for mercury emissions in the U.S. are the three largest gold mines. Hydrogeochemical release of mercury from gold-mine tailings has been accounted as a significant source of atmospheric mercury in eastern Canada.[143]
- 6.8% from non-ferrous metal production, typically smelters.
- 6.4% from cement production.
- 3.0% from waste disposal, including municipal and hazardous waste, crematoria, and sewage sludge incineration.
- 3.0% from caustic soda production.
- 1.4% from pig iron and steel production.
- 1.1% from mercury production, mainly for batteries.
- 2.0% from other sources.
The above percentages are estimates of the global human-caused mercury emissions in 2000, excluding biomass burning, an important source in some regions.[142]
A serious industrial disaster was the dumping of waste mercury compounds into Minamata Bay, Japan, between 1932 and 1968. It is estimated that over 3,000 people suffered various deformities, severe mercury poisoning symptoms or death from what became known as Minamata disease.[144][145]
China is estimated to produce 50% of mercury emissions, most of which result from production of vinyl chloride.[146]
Mercury also enters into the environment through the improper disposal of mercury-containing products.[147] Due to health concerns, toxics use reduction efforts are cutting back or eliminating mercury in such products. For example, the amount of mercury sold in thermostats in the United States decreased from 14.5 tons in 2004 to 3.9 tons in 2007.[148]
The tobacco plant readily absorbs and accumulates heavy metals such as mercury from the surrounding soil into its leaves. These are subsequently inhaled during tobacco smoking.[149] While mercury is a constituent of tobacco smoke,[150] studies have largely failed to discover a significant correlation between smoking and mercury uptake by humans compared to sources such as occupational exposure, fish consumption, and amalgam tooth fillings.[151]
A less well-known source of mercury is the burning of joss paper,[152] which is a common tradition practiced in Asia, including China,[153] Vietnam, Hong Kong, Thailand, Taiwan and Malaysia.[154]
Spill cleanup
[edit]Mercury spills pose an immediate threat to people handling the material, in addition to being an environmental hazard if the material is not contained properly. This is of particular concern for visible mercury, or mercury in liquid state, as its unusual appearance and behavior for a metal makes it an attractive nuisance to the uninformed.[155] Procedures have been developed to contain mercury spills, as well as recommendations on appropriate responses based on the conditions of a spill.[156][157] Tracking liquid mercury away from the site of a spill is a major concern in liquid mercury spills; regulations emphasize containment of the visible mercury as the first course of action, followed by monitoring of mercury vapors and vapor cleanup. Several products are sold as mercury spill adsorbents, ranging from metal salts to polymers and zeolites.[158]
Sediment contamination
[edit]Sediments within large urban-industrial estuaries act as an important sink for point source and diffuse mercury pollution within catchments.[159] A 2015 study of foreshore sediments from the Thames estuary measured total mercury at 0.01 to 12.07 mg/kg with mean of 2.10 mg/kg and median of 0.85 mg/kg (n = 351).[159] The highest mercury concentrations were shown to occur in and around the city of London in association with fine grain muds and high total organic carbon content.[159] The strong affinity of mercury for carbon rich sediments has also been observed in salt marsh sediments of the River Mersey, with a mean concentration of 2 mg/kg, up to 5 mg/kg.[160] These concentrations are far higher than those in the salt marsh river creek sediments of New Jersey and mangroves of Southern China, which exhibit low mercury concentrations of about 0.2 mg/kg.[161][162]
Occupational exposure
[edit]
Due to the health effects of mercury exposure, industrial and commercial uses are regulated in many countries. The World Health Organization,[163] OSHA, and NIOSH all treat mercury as an occupational hazard; both OSHA and NIOSH, among other regulatory agencies, have established specific occupational exposure limits on the element and its derivative compounds in liquid and vapor form.[164][165] Environmental releases and disposal of mercury are regulated in the U.S. primarily by the United States Environmental Protection Agency.
Fish
[edit]Fish and shellfish have a natural tendency to concentrate mercury in their bodies, often in the form of methylmercury, a highly toxic organic compound of mercury. Species of fish that are high on the food chain, such as shark, swordfish, king mackerel, bluefin tuna, albacore tuna, and tilefish contain higher concentrations of mercury than others. Because mercury and methylmercury are fat soluble, they primarily accumulate in the viscera, although they are also found throughout the muscle tissue.[166] Mercury presence in fish muscles can be studied using non-lethal muscle biopsies.[167] Mercury present in prey fish accumulates in the predator that consumes them. Since fish are less efficient at depurating than accumulating methylmercury, methylmercury concentrations in the fish tissue increase over time. Thus species that are high on the food chain amass body burdens of mercury that can be ten times higher than the species they consume. This process is called biomagnification. Mercury poisoning happened this way in Minamata, Japan, now called Minamata disease.[144][145]
In the Lower Amazon, mercury contamination in fish is driven by anthropogenic activities such as gold mining and deforestation, which release mercury into aquatic ecosystems.[168][169] Studies report mercury concentrations in fish muscle tissue ranging from 0.01 to 0.67 μg/g, with carnivorous species like Plagioscion squamosissimus showing higher levels due to biomagnification, sometimes exceeding the World Health Organization's safety threshold of 0.5 μg/g.[168] Local communities relying on fish as a dietary staple face potential health risks from mercury exposure. Mercury levels in aquatic species, including fish and shrimp (Macrobrachium amazonicum), indicate broader environmental contamination, particularly near mining areas.[170]
Cosmetics
[edit]Some facial creams contain dangerous levels of mercury. Most contain comparatively non-toxic inorganic mercury, but products containing highly toxic organic mercury have been encountered.[171][172] New York City residents have been found to be exposed to significant levels of inorganic mercury compounds through the use of skin care products.[173]
Effects and symptoms of mercury poisoning
[edit]Toxic effects include damage to the brain, kidneys and lungs. Mercury poisoning can result in several diseases, including acrodynia (pink disease), Hunter-Russell syndrome, and Minamata disease. Symptoms typically include sensory impairment (vision, hearing, speech), disturbed sensation and a lack of coordination. The type and degree of symptoms exhibited depend upon the individual toxin, the dose, and the method and duration of exposure. Case–control studies have shown effects such as tremors, impaired cognitive skills, and sleep disturbance in workers with chronic exposure to mercury vapor even at low concentrations in the range 0.7–42 μg/m3.[174][175]
A study has shown that acute exposure (4–8 hours) to calculated elemental mercury levels of 1.1 to 44 mg/m3 resulted in chest pain, dyspnea, cough, hemoptysis, impairment of pulmonary function, and evidence of interstitial pneumonitis.[138] Acute exposure to mercury vapor has been shown to result in profound central nervous system effects, including psychotic reactions characterized by delirium, hallucinations, and suicidal tendency. Occupational exposure has resulted in broad-ranging functional disturbance, including erethism, irritability, excitability, excessive shyness, and insomnia. With continuing exposure, a fine tremor develops and may escalate to violent muscular spasms. Tremor initially involves the hands and later spreads to the eyelids, lips, and tongue. Long-term, low-level exposure has been associated with more subtle symptoms of erethism, including fatigue, irritability, loss of memory, vivid dreams and depression.[139][176]
Treatment
[edit]Research on the treatment of mercury poisoning is limited. Currently available drugs for acute mercurial poisoning include chelators N-acetyl-D,L-penicillamine (NAP), British Anti-Lewisite (BAL), 2,3-dimercapto-1-propanesulfonic acid (DMPS), and dimercaptosuccinic acid (DMSA). In one small study including 11 construction workers exposed to elemental mercury, patients were treated with DMSA and NAP.[177] Chelation therapy with both drugs resulted in the mobilization of a small fraction of the total estimated body mercury. DMSA was able to increase the excretion of mercury to a greater extent than NAP.[177]
Regulations
[edit]International
[edit]140 countries agreed in the Minamata Convention on Mercury by the United Nations Environment Programme (UNEP) to prevent mercury vapor emissions.[178] The convention was signed on 10 October 2013.[179]
United States
[edit]In the United States, the Environmental Protection Agency is charged with regulating and managing mercury contamination. Several laws give the EPA this authority, including the Clean Air Act, the Clean Water Act, the Resource Conservation and Recovery Act, and the Safe Drinking Water Act. Additionally, the Mercury-Containing and Rechargeable Battery Management Act, passed in 1996, phases out the use of mercury in batteries, and provides for the efficient and cost-effective disposal of many types of used batteries.[180] North America contributed approximately 11% of the total global anthropogenic mercury emissions in 1995.[181]
The United States Clean Air Act, passed in 1990, put mercury on a list of toxic pollutants that need to be controlled to the greatest possible extent. Thus, industries that release high concentrations of mercury into the environment agreed to install maximum achievable control technologies (MACT). In March 2005, the EPA promulgated a regulation[182] that added power plants to the list of sources that should be controlled and instituted a national cap and trade system. States were given until November 2006 to impose stricter controls, but after a legal challenge from several states, the regulations were struck down by a federal appeals court on 8 February 2008. The rule was deemed not sufficient to protect the health of persons living near coal-fired power plants, given the negative effects documented in the EPA Study Report to Congress of 1998.[183] However newer data published in 2015 showed that after introduction of the stricter controls mercury declined sharply, indicating that the Clean Air Act had its intended impact.[184]
The EPA announced new rules for coal-fired power plants on 22 December 2011.[185] Cement kilns that burn hazardous waste are held to a looser standard than are standard hazardous waste incinerators in the United States, and as a result are a disproportionate source of mercury pollution.[186]
European Union
[edit]In the European Union, the directive on the Restriction of the Use of Certain Hazardous Substances in Electrical and Electronic Equipment (see RoHS) bans mercury from certain electrical and electronic products, and limits the amount of mercury in other products to less than 1000 ppm.[187] There are restrictions for mercury concentration in packaging (the limit is 100 ppm for sum of mercury, lead, hexavalent chromium and cadmium) and batteries (the limit is 5 ppm).[188] In July 2007, the European Union also banned mercury in non-electrical measuring devices, such as thermometers and barometers. The ban applies to new devices only, and contains exemptions for the health care sector and a two-year grace period for manufacturers of barometers.[189]
Scandinavia
[edit]Norway enacted a total ban on the use of mercury in the manufacturing and import/export of mercury products, effective 1 January 2008.[190] In 2002, several lakes in Norway were found to have a poor state of mercury pollution, with an excess of 1 μg/g of mercury in their sediment.[191] In 2008, Norway's Minister of Environment Development Erik Solheim said: "Mercury is among the most dangerous environmental toxins. Satisfactory alternatives to Hg in products are available, and it is therefore fitting to induce a ban."[192] Products containing mercury were banned in Sweden in 2009,[193][194] while elemental mercury has been banned from manufacture and use in all but a few applications (such as certain energy-saving light sources and amalgam dental fillings) in Denmark since 2008.[195]
See also
[edit]- COLEX process (isotopic separation)
- Mercury pollution in the ocean
- Red mercury
Notes
[edit]- ^ Theoretical calculations indicate that copernicium, which lies directly beneath mercury on the periodic table, is likely a liquid at standard pressure and temperature.[10]
- ^ A room can easily reach 29 °C (84 °F) to melt caesium, and 30 °C (86 °F) to melt gallium.
- ^ −37.89 °F; 234.32 °K
- ^ 674.11 °F; 629.88 °K
References
[edit]- ^ "Standard Atomic Weights: Mercury". CIAAW. 2011.
- ^ Prohaska, Thomas; Irrgeher, Johanna; Benefield, Jacqueline; Böhlke, John K.; Chesson, Lesley A.; Coplen, Tyler B.; Ding, Tiping; Dunn, Philip J. H.; Gröning, Manfred; Holden, Norman E.; Meijer, Harro A. J. (4 May 2022). "Standard atomic weights of the elements 2021 (IUPAC Technical Report)". Pure and Applied Chemistry. doi:10.1515/pac-2019-0603. ISSN 1365-3075.
- ^ Fehlauer, H.; Bettin, H. (2004). "Density of mercury—measurements and reference values". Metrologia. 41 (2): S16 – S22. doi:10.1088/0026-1394/41/2/S02. Retrieved 8 July 2023.
- ^ Supplementary table for vapor pressures of the elements § 80 Hg
- ^ Brauer, G.; Haucke, W. (1 June 1936). "Kristallstruktur der intermetallischen Phasen MgAu und MgHg". Zeitschrift für Physikalische Chemie. 33B (1): 304–310. doi:10.1515/zpch-1936-3327. ISSN 2196-7156.
MgHg then lends itself to an oxidation state of +2 for Mg and -2 for Hg because it consists entirely of these polar bonds with no evidence of electron unpairing. (translated)
- ^ Arblaster, John W. (2018). Selected Values of the Crystallographic Properties of Elements. Materials Park, Ohio: ASM International. ISBN 978-1-62708-155-9.
- ^ "Magnetic Susceptibility of the Elements And Inorganic Compounds" (PDF). www-d0.fnal.gov. Fermi National Accelerator Laboratory: DØ Experiment (lagacy document). Archived from the original (PDF) on 24 March 2004. Retrieved 18 February 2015.
- ^ Weast, Robert (1984). CRC, Handbook of Chemistry and Physics. Boca Raton, Florida: Chemical Rubber Company Publishing. pp. E110. ISBN 0-8493-0464-4.
- ^ Kondev, F. G.; Wang, M.; Huang, W. J.; Naimi, S.; Audi, G. (2021). "The NUBASE2020 evaluation of nuclear properties" (PDF). Chinese Physics C. 45 (3) 030001. doi:10.1088/1674-1137/abddae.
- ^ Mewes, J.-M.; Smits, O. R.; Kresse, G.; Schwerdtfeger, P. (2019). "Copernicium is a Relativistic Noble Liquid". Angewandte Chemie International Edition. 58 (50): 17964–17968. Bibcode:2019ACIE...5817964M. doi:10.1002/anie.201906966. PMC 6916354. PMID 31596013.
- ^ "What is EPA doing about mercury air emissions?". United States Environmental Protection Agency (EPA). Archived from the original on 8 February 2007. Retrieved 1 May 2007.
- ^ "New 12-sided pound coin to enter circulation in March". BBC News. 1 January 2017. Archived from the original on 1 August 2024. Retrieved 2 January 2017.
- ^ a b c d e f Hammond, C. R. "The Elements" (PDF). Archived from the original (PDF) on 26 June 2008. in Lide, D. R., ed. (2005). CRC Handbook of Chemistry and Physics (86th ed.). Boca Raton, Florida: CRC Press. ISBN 0-8493-0486-5.
- ^ a b Norrby, L.J. (1991). "Why is mercury liquid? Or, why do relativistic effects not get into chemistry textbooks?". Journal of Chemical Education. 68 (2): 110. Bibcode:1991JChEd..68..110N. doi:10.1021/ed068p110. S2CID 96003717.
- ^ Senese, F. "Why is mercury a liquid at STP?". General Chemistry Online at Frostburg State University. Archived from the original on 4 April 2007. Retrieved 1 May 2007.
- ^ Lide, D. R., ed. (2005). CRC Handbook of Chemistry and Physics (86th ed.). Boca Raton, Florida: CRC Press. pp. 4.125 – 4.126. ISBN 0-8493-0486-5.
- ^ "Dynamic Periodic Table". www.ptable.com. Archived from the original on 20 November 2016. Retrieved 22 November 2016.
- ^ Simons, E. N. (1968). Guide to Uncommon Metals. Frederick Muller. p. 111.
- ^ a b Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. doi:10.1016/C2009-0-30414-6. ISBN 978-0-08-037941-8.
- ^ Swackhamer, Barry (26 November 2011). "Mercury Storage Vault". The Historic Marker Database. Retrieved 11 December 2023.
- ^ Gmelin, Leopold (1852). Hand book of chemistry. Cavendish Society. pp. 103 (Na), 110 (W), 122 (Zn), 128 (Fe), 247 (Au), 338 (Pt). Archived from the original on 9 May 2013. Retrieved 30 December 2012.
- ^ Soratur, S. H. (2002). Essentials of Dental Materials. Jaypee Brothers Publishers. p. 14. ISBN 978-81-7179-989-3.
- ^ Vargel, C.; Jacques, M.; Schmidt, M. P. (2004). Corrosion of Aluminium. Elsevier. p. 158. ISBN 978-0-08-044495-6.
- ^ Case, Raymundo; McIntyre, Dale R. (14 March 2010). Mercury Liquid Metal Embrittlement Of Alloys For Oil And Gas Production And Processing.
- ^ Holman, Jack P. (2002). Heat Transfer (9th ed.). New York, NY: cGraw-Hill Companies, Inc. pp. 600–606. ISBN 978-0-07-240655-9.
- ^ Incropera, Frank P. (2007). Fundamentals of Heat and Mass Transfer (6th ed.). Hoboken, NJ: John Wiley and Sons, Inc. pp. 941–950. ISBN 978-0-471-45728-2.
- ^ Blum, Joel D. (December 2013). "Mesmerized by mercury". Nature Chemistry. 5 (12): 1066. Bibcode:2013NatCh...5.1066B. doi:10.1038/nchem.1803. ISSN 1755-4349. PMID 24256872.
- ^ a b "Definition of hydrargyrum | Dictionary.com". Archived from the original on 12 August 2014. Retrieved 22 December 2022. Random House Webster's Unabridged Dictionary.
- ^ Webster's Revised Unabridged Dictionary. Springfield, Mass.: G. & C. Merriam. 1913. OCLC 800618302. Retrieved 27 December 2023.
- ^ a b Stillman, J. M. (2003). Story of Alchemy and Early Chemistry. Kessinger Publishing. pp. 7–9. ISBN 978-0-7661-3230-6. OCLC 233637688.
- ^ Maurice Crosland (2004) Historical Studies in the Language of Chemistry
- ^ "Mercury and the environment — Basic facts". Environment Canada, Federal Government of Canada. 2004. Archived from the original on 16 September 2011. Retrieved 27 March 2008.
- ^ Martín Gil, J.; Martín Gil, F. J.; Delibes de Castro, G.; Zapatero Magdaleno, P.; Sarabia Herrero, F. J. (1995). "The first known use of vermillion". Experientia. 51 (8): 759–761. doi:10.1007/BF01922425. ISSN 0014-4754. PMID 7649232. S2CID 21900879.
- ^ "Mercury — Element of the ancients". Center for Environmental Health Sciences, Dartmouth College. Archived from the original on 2 December 2012. Retrieved 9 April 2012.
- ^ "Qin Shihuang". Ministry of Culture, People's Republic of China. 2003. Archived from the original on 4 July 2008. Retrieved 27 March 2008.
- ^ Wright, David Curtis (2001). The History of China. Greenwood Publishing Group. p. 49. ISBN 978-0-313-30940-3.
- ^ Sobernheim, Moritz (1987). "Khumārawaih". In Houtsma, Martijn Theodoor (ed.). E.J. Brill's first encyclopaedia of Islam, 1913–1936, Volume IV: 'Itk–Kwaṭṭa. Leiden: BRILL. p. 973. ISBN 978-90-04-08265-6. Archived from the original on 3 June 2016.
- ^ a b Yuhas, Alan (24 April 2015). "Liquid mercury found under Mexican pyramid could lead to king's tomb". The Guardian. ISSN 0261-3077. Archived from the original on 1 December 2016. Retrieved 22 November 2016.
- ^ Pendergast, David M. (6 August 1982). "Ancient maya mercury". Science. 217 (4559): 533–535. Bibcode:1982Sci...217..533P. doi:10.1126/science.217.4559.533. PMID 17820542. S2CID 39473822.
- ^ "Lamanai". Archived from the original on 11 June 2011. Retrieved 17 June 2011.
- ^ Hicks, R. D. (1907). "Chapter 3". Aristotle De Anima. Cambridge: Cambridge University Press. Text
- ^ Hesse, R W (2007). Jewelrymaking through history. Greenwood Publishing Group. p. 120. ISBN 978-0-313-33507-5.
- ^ Eisler, R. (2006). Mercury hazards to living organisms. CRC Press. ISBN 978-0-8493-9212-2.
- ^ Burkholder, M. & Johnson, L. (2008). Colonial Latin America. Oxford University Press. pp. 157–159. ISBN 978-0-19-504542-0.
- ^ a b Jamieson, R W (2000). Domestic Architecture and Power. Springer. p. 33. ISBN 978-0-306-46176-7.
- ^ Lang, Mervyn (1986). "El derrumbe de Huancavelica en 1786: Fracaso de una reforma borbónica". Histórica (in Spanish). X (2): 213–226. doi:10.18800/historica.198602.003.
- ^ a b c Povea Moreno, Isabel María (2012). "Los busconesde metal. El sistema de pallaqueoen Huancavelica (1793-1820)" [The buscones of metal. The pallaqueo system in Huancavelica (1793-1820)]. Anuario de Estudios Americanos (in Spanish). 69 (1): 109–138. doi:10.3989/aeamer.2012.1.04.
- ^ Laris Pardo, Jorge Alejandro (2022). "La cadena del mercurio en la Monarquía Hispánica a partir de su historiografía" [Historiographical essay on the Commodity Chain of mercury in the Hispanic Monarchy]. Relaciones. Estudios de historia y sociedad (in Spanish). 43 (170).
- ^ Ehrlich, H. L.; Newman, D. K. (2008). Geomicrobiology. CRC Press. p. 265. ISBN 978-0-8493-7906-2.
- ^ Meyer, Lorraine; Guyot, Stéphane; Chalot, Michel; Capelli, Nicolas (1 September 2023). "The potential of microorganisms as biomonitoring and bioremediation tools for mercury-contaminated soils". Ecotoxicology and Environmental Safety. 262 115185. Bibcode:2023EcoES.26215185M. doi:10.1016/j.ecoenv.2023.115185. ISSN 0147-6513. PMID 37385017.
- ^ Rytuba, James J (2003). "Mercury from mineral deposits and potential environmental impact". Environmental Geology. 43 (3): 326–338. Bibcode:2003EnGeo..43..326R. doi:10.1007/s00254-002-0629-5. S2CID 127179672.
- ^ a b "Metacinnabar". Mindat.org. Retrieved 16 November 2023.
- ^ "Mercury Recycling in the United States in 2000" (PDF). USGS. Archived (PDF) from the original on 26 March 2009. Retrieved 7 July 2009.
- ^ Brooks, W. E. (2007). "Mercury" (PDF). U.S. Geological Survey. Archived (PDF) from the original on 27 May 2008. Retrieved 30 May 2008.
- ^ "World Mineral Production" (PDF). p. 48. Retrieved 22 November 2023.
- ^ "Mineral Commodity Summaries 2025 - Mercury" (PDF). Retrieved 17 September 2025.
- ^ "World mineral statistics data". MineralsUK. Retrieved 17 September 2025.
- ^ "Thank President Obama and Administrator Jackson for protecting us from toxic mercury". Act.credoaction.com. 21 December 2011. Archived from the original on 1 May 2012. Retrieved 30 December 2012.
- ^ a b Sheridan, M. (3 May 2009). "'Green' Lightbulbs Poison Workers: hundreds of factory staff are being made ill by mercury used in bulbs destined for the West". The Sunday Times (of London, UK). Archived from the original on 17 May 2009.
- ^ "Almaden Quicksilver Park, then and now in pictures". The Mercury News. 12 March 2015. Retrieved 2 January 2025.
- ^ Boulland M (2006). New Almaden. Arcadia Publishing. p. 8. ISBN 978-0-7385-3131-1.
- ^ For a general overview, see Riedel, S.; Kaupp, M. (2009). "The Highest Oxidation States of the Transition Metal Elements". Coordination Chemistry Reviews. 253 (5–6): 606–624. doi:10.1016/j.ccr.2008.07.014. The claimed 1976 synthesis is Deming, Richard L.; Allred, A. L.; Dahl, Alan R.; Herlinger, Albert W.; Kestner, Mark O. (July 1976). "Tripositive mercury. Low temperature electrochemical oxidation of 1,4,8,11-tetraazacyclotetradecanemercury(II) tetrafluoroborate". Journal of the American Chemical Society. 98 (14): 4132–4137. Bibcode:1976JAChS..98.4132D. doi:10.1021/ja00430a020; but note that Reidel & Kaupp cite more recent work arguing that the cyclam ligand is instead oxidized. The claimed 2007 isolation is Xuefang Wang; Andrews, Lester; Riedel, Sebastian; Kaupp, Martin (2007). "Mercury Is a Transition Metal: The First Experimental Evidence for HgF4". Angew. Chem. Int. Ed. 46 (44): 8371–8375. Bibcode:2007ACIE...46.8371W. doi:10.1002/anie.200703710. PMID 17899620, but the spectral identifications are disputed in Rooms, J. F.; Wilson, A. V.; Harvey, I.; Bridgeman, A. J.; Young, N. A. (2008). "Mercury-fluorine interactions: a matrix isolation investigation of Hg⋯F2, HgF2 and HgF4 in argon matrices". Phys Chem Chem Phys. 10 (31): 4594–605. Bibcode:2008PCCP...10.4594R. doi:10.1039/b805608k. PMID 18665309.
- ^ Henderson, W. (2000). Main group chemistry. Great Britain: Royal Society of Chemistry. p. 162. ISBN 978-0-85404-617-1. Archived from the original on 13 May 2016.
- ^ Knight, Lon B. (1971). "Hyperfine Interaction, Chemical Bonding, and Isotope Effect in ZnH, CdH, and HgH Molecules". The Journal of Chemical Physics. 55 (5): 2061–2070. Bibcode:1971JChPh..55.2061K. doi:10.1063/1.1676373.
- ^ Brown, I. D.; Gillespie, R. J.; Morgan, K. R.; Tun, Z.; Ummat, P. K. (1984). "Preparation and crystal structure of mercury hexafluoroniobate (Hg
3NbF
6) and mercury hexafluorotantalate (Hg
3TaF
6): mercury layer compounds". Inorganic Chemistry. 23 (26): 4506–4508. doi:10.1021/ic00194a020. - ^ Chisholm, Hugh, ed. (1911). . Encyclopædia Britannica. Vol. 7 (11th ed.). Cambridge University Press. p. 197.
- ^ Anderegg, G.; Schwarzenbach, G.; Padmoyo, M.; Borg, Ö. F. (1958). "Monomolekular gelöstes Quecksilberhydroxyd und seine Basizität". Helvetica Chimica Acta. 41 (4): 988–996. Bibcode:1958HChAc..41..988A. doi:10.1002/hlca.19580410411.
- ^ Rogalski, A (2000). Infrared detectors. CRC Press. p. 507. ISBN 978-90-5699-203-3.
- ^ Vogel, Arthur I.; Svehla, G. (1979), Vogel's Textbook of Macro and Semimicro Qualitative Inorganic Analysis (5th ed.), London: Longman, p. 319, ISBN 0-582-44367-9 – via the Internet Archive
- ^ Committee on the Toxicological Effects of Methylmercury; Board on Environmental Studies and Toxicology; Commission on Life Sciences; National Research Council (2000). Toxicological effects of methylmercury. National Academies Press. ISBN 978-0-309-07140-6.
- ^ Surmann, P; Zeyat, H (November 2005). "Voltammetric analysis using a self-renewable non-mercury electrode". Analytical and Bioanalytical Chemistry. 383 (6): 1009–13. doi:10.1007/s00216-005-0069-7. PMID 16228199. S2CID 22732411.
- ^ Lind, J (1787). "An Account of the Efficacy of Mercury in the Cure of Inflammatory Diseases, and the Dysentery". The London Medical Journal. 8 (Pt 1): 43–56. ISSN 0952-4177. PMC 5545546. PMID 29139904.
- ^ Merck's Manual 1899 (1st ed.). Archived from the original on 24 August 2013. Retrieved 16 June 2013.
- ^ Liu J; Shi JZ; Yu LM; Goyer RA; Waalkes MP (2008). "Mercury in traditional medicines: is cinnabar toxicologically similar to common mercurials?". Exp. Biol. Med. (Maywood). 233 (7): 810–7. doi:10.3181/0712-MR-336. PMC 2755212. PMID 18445765.
- ^ Pimple KD, Pedroni JA, Berdon V (9 July 2002). "Syphilis in history". Poynter Center for the Study of Ethics and American Institutions at Indiana University-Bloomington. Archived from the original on 16 February 2005. Retrieved 17 April 2005.
- ^ a b Mayell, H. (17 July 2007). "Did Mercury in "Little Blue Pills" Make Abraham Lincoln Erratic?". National Geographic News. Archived from the original on 22 May 2008. Retrieved 15 June 2008.
- ^ "What happened to Mercurochrome?". 23 July 2004. Archived from the original on 11 April 2009. Retrieved 7 July 2009.
- ^ "Dental Amalgam Fillings". Silver Spring, MD: U.S. Food and Drug Administration (FDA). 29 September 2020. Archived from the original on 30 September 2020.
- ^ a b "Thimerosal in Vaccines". Food and Drug Administration / Center for Biologics Evaluation and Research. 6 September 2007. Archived from the original on 29 September 2007. Retrieved 1 October 2007.
- ^ Parker SK, Schwartz B, Todd J, Pickering LK (2004). "Thimerosal-containing vaccines and autistic spectrum disorder: a critical review of published original data". Pediatrics. 114 (3): 793–804. CiteSeerX 10.1.1.327.363. doi:10.1542/peds.2004-0434. PMID 15342856. S2CID 1752023.
Erratum: Parker SK, Todd J, Schwartz B, Pickering LK (January 2005). "Thimerosal-containing vaccines and autistic spectrum disorder: a critical review of published original data". Pediatrics. 115 (1): 200. doi:10.1542/peds.2004-2402. PMID 15630018. S2CID 26700143. - ^ "Quantitative and Qualitative Analysis of Mercury Compounds in the List". Federal Food, Drug, and Cosmetic Act (FD&C Act). U.S. Food and Drug Administration. 30 April 2009. Archived from the original on 5 April 2017.
- ^ Beyer KH (September 1993). "Chlorothiazide. How the thiazides evolved as antihypertensive therapy". Hypertension. 22 (3): 388–91. doi:10.1161/01.hyp.22.3.388. PMID 8349332.
- ^ "Title 21—Food and Drugs Chapter I—Food and Drug Administration Department of Health and Human Services Subchapter D—Drugs for Human Use Code of federal regulations". United States Food and Drug Administration. Archived from the original on 13 March 2007. Retrieved 1 May 2007.
- ^ "The CRB Commodity Yearbook (annual)". The CRB Commodity Yearbook: 173. 2000. ISSN 1076-2906.
- ^ a b Leopold, B. R. (2002). "Chapter 3: Manufacturing Processes Involving Mercury. Use and Release of Mercury in the United States" (PDF). National Risk Management Research Laboratory, Office of Research and Development, U.S. Environmental Protection Agency, Cincinnati, Ohio. Archived from the original (PDF) on 21 June 2007. Retrieved 1 May 2007.
- ^ "Chlorine Online Diagram of mercury cell process". Euro Chlor. Archived from the original on 2 September 2006. Retrieved 15 September 2006.
- ^ O'Brien, Thomas F.; Bommaraju, Tilak V.; Hine, Fumio, eds. (2005). "History of the Chlor-Alkali Industry". Handbook of Chlor-Alkali Technology. Boston, MA: Springer. pp. 17–36. doi:10.1007/0-306-48624-5_2. ISBN 978-0-306-48624-1. Retrieved 5 October 2020.
- ^ Middleton, W. E. K. (1966). A history of the thermometer and its use in meteorology. Johns Hopkins Press. ISBN 9780801871535.
- ^ Grigull, Ulrich (1966). Fahrenheit, a Pioneer of Exact Thermometry. (The Proceedings of the 8th International Heat Transfer Conference, San Francisco, 1966, Vol. 1, pp. 9–18.)
- ^ "Protocol on Heavy Metals". UNECE. Retrieved 10 August 2014.
- ^ "Mercury Reduction Act of 2003". United States. Congress. Senate. Committee on Environment and Public Works. Retrieved 6 June 2009.
- ^ "Mercury Thermometer Alternatives: Hg Alternatives". nist.gov. National Institute of Standards and Technology. 29 November 2021. Retrieved 22 December 2023.
- ^ "Liquid-mirror telescope set to give stargazing a new spin". Govert Schilling. 14 March 2003. Archived from the original on 18 August 2003. Retrieved 11 October 2008.
- ^ Gibson, B. K. (1991). "Liquid Mirror Telescopes: History". Journal of the Royal Astronomical Society of Canada. 85: 158. Bibcode:1991JRASC..85..158G.
- ^ "Laval University Liquid mirrors and adaptive optics group". Archived from the original on 18 September 2011. Retrieved 24 June 2011.
- ^ Brans, Y W; Hay W W (1995). Physiological monitoring and instrument diagnosis in perinatal and neonatal medicine. CUP Archive. p. 175. ISBN 978-0-521-41951-2.
- ^ Zoski, Cynthia G. (7 February 2007). Handbook of Electrochemistry. Elsevier Science. ISBN 978-0-444-51958-0.
- ^ Kissinger, Peter; Heineman, William R. (23 January 1996). Laboratory Techniques in Electroanalytical Chemistry, Second Edition, Revised and Expanded (2nd ed.). CRC. ISBN 978-0-8247-9445-3.
- ^ Pike, Ashley C. W.; Garman, Elspeth F.; Krojer, Tobias; von Delft, Frank; Carpenter, Elisabeth P. (1 March 2016). "An overview of heavy-atom derivatization of protein crystals". Acta Crystallographica Section D. 72 (Pt 3): 303–318. Bibcode:2016AcCrD..72..303P. doi:10.1107/S2059798316000401. ISSN 2059-7983. PMC 4784662. PMID 26960118.
- ^ Hopkinson, G.R.; Goodman, T.M.; Prince, S.R. (2004). A guide to the use and calibration of detector array equipment. SPIE Press. p. 125. Bibcode:2004gucd.book.....H. ISBN 978-0-8194-5532-1.
- ^ Howatson A H (1965). "Chapter 8". An Introduction to Gas Discharges. Oxford: Pergamon Press. ISBN 978-0-08-020575-5.
- ^ Milo G E; Casto B C (1990). Transformation of human diploid fibroblasts. CRC Press. p. 104. ISBN 978-0-8493-4956-0.
- ^ Shionoya, S. (1999). Phosphor handbook. CRC Press. p. 363. ISBN 978-0-8493-7560-6.
- ^ Robert L. Tjoelker; et al. (2016). "Mercury Ion Clock for a NASA Technology Demonstration Mission". IEEE Transactions on Ultrasonics, Ferroelectrics, and Frequency Control. 63 (7): 1034–1043. Bibcode:2016ITUFF..63.1034T. doi:10.1109/TUFFC.2016.2543738. PMID 27019481. S2CID 3245467.
- ^ Mohammed, Terry; Mohammed, Elisabeth; Bascombe, Shermel (9 October 2017). "The evaluation of total mercury and arsenic in skin bleaching creams commonly used in Trinidad and Tobago and their potential risk to the people of the Caribbean". Journal of Public Health Research. 6 (3): 1097. doi:10.4081/jphr.2017.1097. PMC 5736993. PMID 29291194.
- ^ Meera Senthilingam, "Skin whitening creams containing high levels of mercury continue to be sold on the world's biggest e-commerce sites, new report finds", 9 March 2022, CNN https://www.cnn.com/2022/03/09/world/zmwg-skin-whitening-creams-mercury-ecommerce-sites-intl-cmd/index.html
- ^ Wisniak, Jaime (2012). "Edward Charles Howard. Explosives, meteorites, and sugar". Educación Química. 23 (2). Universidad Nacional Autonoma de Mexico: 230–239. doi:10.1016/s0187-893x(17)30114-3. ISSN 0187-893X.
- ^ a b "Business Daily: Ghana's illegal gold mines". BBC. 3 December 2024.
- ^ Healy, Paul F.; Blainey, Marc G. (2011). "Ancient Maya Mosaic Mirrors: Function, Symbolism, And Meaning". Ancient Mesoamerica. 22 (2): 229–244 (241). doi:10.1017/S0956536111000241. S2CID 162282151.
- ^ Lew K. (2008). Mercury. The Rosen Publishing Group. p. 10. ISBN 978-1-4042-1780-5.
- ^ Pearson L. F. (2003). Lighthouses. Osprey Publishing. p. 29. ISBN 978-0-7478-0556-4.
- ^ Ramanathan E. AIEEE Chemistry. Sura Books. p. 251. ISBN 978-81-7254-293-1.
- ^ Shelton, C. (2004). Electrical Installations. Nelson Thornes. p. 260. ISBN 978-0-7487-7979-6.
- ^ Eckert, J. P. (October 1953). "A Survey of Digital Computer Memory Systems". Proceedings of the IRE. 41 (10): 1393–1406. doi:10.1109/JRPROC.1953.274316.
- ^ van Delft, Dirk; Kes, Peter (1 September 2010). "The discovery of superconductivity". Physics Today. 63 (9): 38–43. Bibcode:2010PhT....63i..38V. doi:10.1063/1.3490499. Archived from the original on 14 November 2023. Retrieved 6 December 2023.
- ^ Tresca, Cesare; Profeta, Gianni; Marini, Giovanni; Bachelet, Giovanni B.; Sanna, Antonio; Calandra, Matteo; Boeri, Lilia (3 November 2022). "Why mercury is a superconductor". Physical Review B. 106 (18) L180501. arXiv:2111.13867. Bibcode:2022PhRvB.106r0501T. doi:10.1103/PhysRevB.106.L180501. hdl:11573/1659661. ISSN 2469-9950. S2CID 244715089.
- ^ Berlincourt, T. G. & Hake, R. R. (1962). "Pulsed-Magnetic-Field Studies of Superconducting Transition Metal Alloys at High and Low Current Densities". Bulletin of the American Physical Society. II-7: 408.
- ^ "Popular Science". The Popular Science Monthly. 118 (3). Bonnier Corporation: 40. 1931. ISSN 0161-7370.
- ^ Mueller, Grover C. (September 1929). "Cheaper Power from Quicksilver". Popular Science.
- ^ "Mercury as a Working Fluid". Museum of Retro Technology. 13 November 2008. Archived from the original on 21 February 2011.
- ^ James Collier; Geoffrey F. Hewitt (1987). Introduction to Nuclear Power. Taylor & Francis. p. 64. ISBN 978-1-56032-682-3.
- ^ "Glenn Contributions to Deep Space 1". NASA. 21 May 2008. Archived from the original on 1 October 2009. Retrieved 7 July 2009.
- ^ "Electric space propulsion". The Internet Encyclopedia of Science. David Darling. Archived from the original on 30 May 2009. Retrieved 7 July 2009.
- ^ "IMERC Fact Sheet: Mercury Use in Batteries". Northeast Waste Management Officials' Association. January 2010. Archived from the original on 29 November 2012. Retrieved 20 June 2013.
- ^ Mercury Silvering, archived from the original on 4 March 2005, retrieved 12 February 2010.
- ^ "Organotin Compounds in the Environment". Open Chemist. Archived from the original on 10 March 2007.
- ^ Smart, N. A. (1968). "Use and residues of mercury compounds in agriculture". In Francis A. Gunther (ed.). Residue Reviews: Residues of Pesticides and Other Foreign Chemicals in Foods and Feeds. Residue Reviews / Rückstands-Berichte. Vol. 23. Springer. pp. 1–36. doi:10.1007/978-1-4615-8437-7_1. ISBN 978-1-4615-8439-1. PMID 4875698.
- ^ Gray, T. (22 September 2004). "The Amazing Rusting Aluminum". Popular Science. Archived from the original on 20 July 2009. Retrieved 7 July 2009.
- ^ Francis, G. W. (1849). Chemical Experiments. D. Francis. p. 62.
- ^ Castles, W. T.; Kimball, V. F. (2005). Firearms and Their Use. Kessinger Publishing. p. 104. ISBN 978-1-4179-8957-7.
- ^ Lee, J. D. (1999). Concise Inorganic Chemistry. Wiley-Blackwell. ISBN 978-0-632-05293-6.
- ^ Crean, J. F. (1962). "Hats and the Fur Trade". The Canadian Journal of Economics and Political Science. 28 (3): 380. doi:10.2307/139669. ISSN 0315-4890. JSTOR 139669.
- ^ Waldron, H. A. (1983). "Did the Mad Hatter have mercury poisoning?". Br. Med. J. (Clin. Res. Ed.). 287 (6409): 1961. doi:10.1136/bmj.287.6409.1961. PMC 1550196. PMID 6418283.
- ^ Alpers, C. N.; Hunerlach, M. P.; May, J. Y.; Hothem, R. L. "Mercury Contamination from Historical Gold Mining in California". U.S. Geological Survey. Archived from the original on 22 February 2008. Retrieved 26 February 2008.
- ^ "Mercury amalgamation". Corrosion Doctors. Archived from the original on 19 May 2009. Retrieved 7 July 2009.
- ^ "Mercury 294594". Sigma-Aldrich.
- ^ "Toxicological Profile for Mercury" (PDF). Atlanta, GA: Agency for Toxic Substances and Disease Registry. 1999. Archived (PDF) from the original on 21 July 2011. Retrieved 22 February 2011.
- ^ a b McFarland, RB & Reigel, H (1978). "Chronic Mercury Poisoning from a Single Brief Exposure". J. Occup. Med. 20 (8): 532–4. doi:10.1097/00043764-197808000-00003. PMID 690736.
- ^ a b Mercury, Environmental Health Criteria monograph No. 001, Geneva: World Health Organization, 1976, ISBN 92-4-154061-3
- ^ "Glacial Ice Cores Reveal A Record of Natural and Anthropogenic Atmospheric Mercury Deposition for the Last 270 Years". United States Geological Survey (USGS). Archived from the original on 4 July 2007. Retrieved 1 May 2007.
- ^ "Indoor Air Mercury" (PDF). Northeast Waste Management Officials' Association. May 2003. Archived from the original (PDF) on 25 March 2009. Retrieved 7 July 2009.
- ^ a b c Pacyna E G; Pacyna J M; Steenhuisen F; Wilson S (2006). "Global anthropogenic mercury emission inventory for 2000". Atmos Environ. 40 (22): 4048. Bibcode:2006AtmEn..40.4048P. doi:10.1016/j.atmosenv.2006.03.041.
- ^ Maprani, Antu C.; Al, Tom A.; MacQuarrie, Kerry T.; Dalziel, John A.; Shaw, Sean A.; Yeats, Phillip A. (2005). "Determination of Mercury Evasion in a Contaminated Headwater Stream". Environmental Science & Technology. 39 (6): 1679–87. Bibcode:2005EnST...39.1679M. doi:10.1021/es048962j. PMID 15819225.
- ^ a b "Minamata Disease The History and Measures". Ministry of the Environment, Government of Japan. Archived from the original on 24 June 2009. Retrieved 7 July 2009.
- ^ a b Dennis Normile (27 September 2013). "In Minamata, Mercury Still Divides". Science. 341 (6153): 1446–7. Bibcode:2013Sci...341.1446N. doi:10.1126/science.341.6153.1446. PMID 24072902.
- ^ Ciriminna, Rosaria; Falletta, Ermelinda; Della Pina, Cristina; Teles, Joaquim Henrique; Pagliaro, Mario (2016). "Industrial Applications of Gold Catalysis". Angewandte Chemie International Edition. 55 (46): 1433–7851. Bibcode:2016ACIE...5514210C. doi:10.1002/anie.201604656. hdl:2434/463818. PMID 27624999. S2CID 28730917.
- ^ "Mercury-containing Products". United States Environmental Protection Agency (EPA). Archived from the original on 12 February 2007. Retrieved 1 May 2007.
- ^ "IMERC Fact Sheet: Mercury Use in Thermostats" (PDF). Northeast Waste Management Officials' Association. January 2010. Archived from the original (PDF) on 17 June 2012.
- ^ Pourkhabbaz, A.; Pourkhabbaz, H. (2012). "Investigation of Toxic Metals in the Tobacco of Different Iranian Cigarette Brands and Related Health Issues". Iranian Journal of Basic Medical Sciences. 15 (1): 636–644. PMC 3586865. PMID 23493960.
- ^ Talhout, Reinskje; Schulz, Thomas; Florek, Ewa; Van Benthem, Jan; Wester, Piet; Opperhuizen, Antoon (2011). "Hazardous Compounds in Tobacco Smoke". International Journal of Environmental Research and Public Health. 8 (12): 613–628. doi:10.3390/ijerph8020613. ISSN 1660-4601. PMC 3084482. PMID 21556207.
- ^ Bernhard D, Rossmann A, Wick G (2005). "Metals in Cigarette Smoke". IUBMB Life. 57 (12): 805–809. doi:10.1080/15216540500459667. PMID 16393783. S2CID 35694266.
- ^ Shen, Huazhen; Tsai, Cheng-Mou; Yuan, Chung-Shin; Jen, Yi-Hsiu; Ie, Iau-Ren (2017). "How incense and joss paper burning during the worship activities influences ambient mercury concentrations in indoor and outdoor environments of an Asian temple?". Chemosphere. 167: 530–540. Bibcode:2017Chmsp.167..530S. doi:10.1016/j.chemosphere.2016.09.159. PMID 27764746.
- ^ Lin, Chunshui; Huang, Ru-Jin; Duan, Jing; Zhong, Haobin; Xu, Wei; Wu, Yunfei; Zhang, Renjian (2022). "Large contribution from worship activities to the atmospheric soot particles in northwest China". Environmental Pollution. 299 118907. Bibcode:2022EPoll.29918907L. doi:10.1016/j.envpol.2022.118907. PMID 35091017. S2CID 246355499.
- ^ "Parkinson's disease in occupational exposure to joss paper, a report of two cases".
- ^ Azziz-Baumgartner, E; Luber, G; Schurz-Rogers, H; Backer, L; Belson, M; Kieszak, S; Caldwell, K; Lee, B; Jones, R (2007). "Exposure assessment of a mercury spill in a Nevada school -- 2004". Clin Toxicol. 45 (4): 391–395. doi:10.1080/15563650601031569. PMID 17486480. S2CID 33770481.
- ^ "Mercury: Spills, Disposal and Site Cleanup". Environmental Protection Agency. Archived from the original on 13 May 2008. Retrieved 11 August 2007.
- ^ "Action Levels for Elemental Mercury Spills" (PDF). www.atsdr.cdc.gov. 22 March 2012.
- ^ Yu, Jin-Gang; Yue, Bao-Yu; Wu, Xiong-Wei; Liu, Qi; Jiao, Fei-Peng; Jiang, Xin-Yu; Chen, Xiao-Qing (1 December 2015). "Removal of mercury by adsorption: a review". Environmental Science and Pollution Research. 23 (6): 5056–5076. doi:10.1007/s11356-015-5880-x. PMID 26620868. S2CID 28365564.
- ^ a b c Vane, C.H.; Beriro, D.J.; Turner, G.H. (2015). "Rise and fall of mercury (Hg) pollution in sediment cores of the Thames Estuary, London, UK". Earth and Environmental Science Transactions of the Royal Society of Edinburgh. 105 (4): 285–296. doi:10.1017/S1755691015000158. ISSN 1755-6910.
- ^ Vane, C.H.; Jones, D.G.; Lister, T.R. (2009). "Mercury contamination in surface sediments and sediment cores of the Mersey Estuary, UK" (PDF). Marine Pollution Bulletin. 58 (6): 940–946. Bibcode:2009MarPB..58..940V. doi:10.1016/j.marpolbul.2009.03.006. ISSN 0025-326X. PMID 19356771.
- ^ Vane, C.H.; Harrison, I.; Kim, A.W.; Moss-Hayes, V.; Vickers, B.P.; Horton, B.P. (2008). "Status of organic pollutants in surface sediments of Barnegat Bay-Little Egg Harbor Estuary, New Jersey, USA" (PDF). Marine Pollution Bulletin. 56 (10): 1802–1808. Bibcode:2008MarPB..56.1802V. doi:10.1016/j.marpolbul.2008.07.004. ISSN 0025-326X. PMID 18715597.
- ^ Vane, C.H.; Harrison, I.; Kim, A.W.; Moss-Hayes, V.; Vickers, B.P.; Hong, K. (2009). "Organic and metal contamination in surface mangrove sediments of South China" (PDF). Marine Pollution Bulletin. 58 (1): 134–144. Bibcode:2009MarPB..58..134V. doi:10.1016/j.marpolbul.2008.09.024. ISSN 0025-326X. PMID 18990413.
- ^ "Mercury and health". World Health Organization. 31 March 2017. Retrieved 22 December 2023.
- ^ "1910.1000 TABLE Z-2". Occupational Safety and Health Administration. 23 June 2006. Retrieved 22 December 2023.
- ^ "Mercury compounds [except (organo) alkyls] (as Hg)". Centers for Disease Control and Prevention. The National Institute for Occupational Safety and Health. 30 October 2019. Retrieved 22 December 2023.
- ^ Cocoros, Glenn; Cahn, Phyllis H.; Siler, William (November 1973). "Mercury concentrations in fish, plankton and water from three Western Atlantic estuaries". Journal of Fish Biology. 5 (6): 641–647. Bibcode:1973JFBio...5..641C. doi:10.1111/j.1095-8649.1973.tb04500.x.
- ^ "How We Do Things at IISD-ELA: Collecting a fish muscle biopsy". IISD. 30 September 2015. Retrieved 7 July 2020.
- ^ a b Albuquerque, Fabio Edir Amaral; Minervino, Antonio Humberto Hamad; Miranda, Marta; Herrero-Latorre, Carlos; Barrêto Júnior, Raimundo Alves; Oliveira, Francisco Leonardo Costa; Sucupira, Maria Claudia Araripe; Ortolani, Enrico Lippi; López-Alonso, Marta (25 August 2020). "Toxic and essential trace element concentrations in fish species in the Lower Amazon, Brazil". Science of the Total Environment. 732 138983. Bibcode:2020ScTEn.73238983A. doi:10.1016/j.scitotenv.2020.138983. ISSN 0048-9697. PMID 32417551.
- ^ Albuquerque, Fabio Edir Amaral; Herrero-Latorre, Carlos; Miranda, Marta; Barrêto Júnior, Raimundo Alves; Oliveira, Francisco Leonardo Costa; Sucupira, Maria Cláudia Araripe; Ortolani, Enrico Lippi; Minervino, Antonio Humberto Hamad; López-Alonso, Marta (15 August 2021). "Fish tissues for biomonitoring toxic and essential trace elements in the Lower Amazon". Environmental Pollution. 283 117024. Bibcode:2021EPoll.28317024A. doi:10.1016/j.envpol.2021.117024. ISSN 0269-7491. PMID 33857879.
- ^ Albuquerque, Fabio Edir Amaral; Minervino, Antonio Humberto Hamad; Miranda, Marta; Herrero-Latorre, Carlos; Barrêto Júnior, Raimundo Alves; Oliveira, Francisco Leonardo Costa; Dias, Salatiel Ribeiro; Ortolani, Enrico Lippi; López-Alonso, Marta (1 March 2020). "Toxic and essential trace element concentrations in the freshwater shrimp Macrobrachium amazonicum in the Lower Amazon, Brazil". Journal of Food Composition and Analysis. 86 103361. doi:10.1016/j.jfca.2019.103361. ISSN 0889-1575.
- ^ Mole, Beth (20 December 2019). "Woman had 524x the normal level of mercury in her blood from skin cream use". ArsTechnica. Retrieved 20 July 2021.
- ^ Mudan, Anita, Copan L, Wang R, et al. (20 December 2019). "Notes from the Field: Methylmercury Toxicity from a Skin Lightening Cream Obtained from Mexico — California, 2019". Morbidity and Mortality Weekly Report. 68 (50): 1166–1167. doi:10.15585/mmwr.mm6850a4. PMC 6936160. PMID 31856147.
- ^ McKelvey W, Jeffery N, Clark N, Kass D, Parsons PJ (2010). "Population-Based Inorganic Mercury Biomonitoring and the Identification of Skin Care Products as a Source of Exposure in New York City". Environ Health Perspect. 119 (2) (published 2011): 203–9. doi:10.1289/ehp.1002396. PMC 3040607. PMID 20923743.
- ^ Ngim, CH; Foo, SC; Boey, KW; Keyaratnam, J (1992). "Chronic neurobehavioral effects of elemental mercury in dentists". British Journal of Industrial Medicine. 49 (11): 782–90. doi:10.1136/oem.49.11.782. PMC 1039326. PMID 1463679.
- ^ Liang, Y. X.; Sun, R. K.; Sun, Y.; Chen, Z. Q.; Li, L. H. (1993). "Psychological effects of low exposure to mercury vapor: Application of computer-administered neurobehavioral evaluation system". Environmental Research. 60 (2): 320–7. Bibcode:1993ER.....60..320L. doi:10.1006/enrs.1993.1040. PMID 8472661.
- ^ Inorganic mercury, Environmental Health Criteria monograph No. 118, Geneva: World Health Organization, 1991, ISBN 92-4-157118-7
- ^ a b Bluhm, RE; Bobbitt, RG; Welch, LW; Wood, AJJ; Bonfiglio, JF; Sarzen, C; Heath, AJ; Branch, RA (1992). "Elemental Mercury Vapour Toxicity, Treatment, and Prognosis After Acute, Intensive Exposure in Chloralkali Plant Workers. Part I: History, Neuropsychological Findings and Chelator effects". Hum Exp Toxicol. 11 (3): 201–10. Bibcode:1992HETox..11..201B. doi:10.1177/096032719201100308. PMID 1352115. S2CID 43524794.
- ^ "Minamata Convention Agreed by Nations". United Nations Environment Program. Archived from the original on 30 January 2013. Retrieved 19 January 2013.
- ^ Section, United Nations News Service (19 January 2013). "UN News — Governments at UN forum agree on legally-binding treaty to curb mercury pollution". UN News Service Section. Archived from the original on 16 October 2016. Retrieved 22 November 2016.
- ^ "Mercury: Laws and regulations". United States Environmental Protection Agency. 16 April 2008. Archived from the original on 13 May 2008. Retrieved 30 May 2008.
- ^ "Reductions in Mercury Emissions". International Joint Commission on the Great Lakes. Archived from the original on 28 August 2008. Retrieved 21 July 2008.
- ^ "Clean Air Mercury Rule". United States Environmental Protection Agency (EPA). Archived from the original on 30 June 2007. Retrieved 1 May 2007.
- ^ "State of New Jersey et al., Petitioners vs. Environmental Protection Agency (Case No. 05-1097)" (PDF). United States Court of Appeals for the District of Columbia Circuit. Argued 6 December 2007, Decided 8 February 2008. Archived (PDF) from the original on 3 February 2011. Retrieved 30 May 2008.
- ^ Castro MS, Sherwell J (2015). "Effectiveness of Emission Controls to Reduce the Atmospheric Concentrations of Mercury". Environmental Science & Technology. 49 (24): 14000–14007. Bibcode:2015EnST...4914000C. doi:10.1021/acs.est.5b03576. PMID 26606506.
- ^ "Oldest, dirtiest power plants told to clean up". Boston Globe. 22 December 2011. Archived from the original on 14 July 2014. Retrieved 2 January 2012.
- ^ Howard Berkes (10 November 2011). "EPA Regulations Give Kilns Permission To Pollute". NPR. Archived from the original on 17 November 2011. Retrieved 2 January 2012.
- ^ "Directive 2002/95/EC on the Restriction of the Use of Certain Hazardous Substances in Electrical and Electronic Equipment". 27 January 2003. Article 4 Paragraph 1. e.g. "Member States shall ensure that, from July 1, 2006, new electrical and electronic equipment put on the market does not contain lead, mercury, cadmium, hexavalent chromium, polybrominated biphenyls (PBB) or polybrominated diphenyl ethers (PBDE)."
- ^ "Mercury compounds in European Union". EIA Track. 2007. Archived from the original on 28 April 2008. Retrieved 30 May 2008.
- ^ Jones H. (10 July 2007). "EU bans mercury in barometers, thermometers". Reuters. Archived from the original on 3 January 2009. Retrieved 12 September 2017.
- ^ "Norway to ban mercury". EU Business. 21 December 2007. Archived from the original on 21 January 2008. Retrieved 30 May 2008.
- ^ Berg, T; Fjeld, E; Steinnes, E (2006). "Atmospheric mercury in Norway: contributions from different sources". The Science of the Total Environment. 368 (1): 3–9. Bibcode:2006ScTEn.368....3B. doi:10.1016/j.scitotenv.2005.09.059. PMID 16310836.
- ^ Edlich, Richard F; Rhoads, Samantha K.; Cantrell, Holly S.; Azavedo, Sabrina M.; Newkirk, Anthony T. Banning Mercury Amalgam in the United States (PDF) (Report). USA: Food and Drug Administration. Archived from the original (PDF) on 1 November 2013.
- ^ "Sweden to ban mercury". The Local. 14 January 2009. Archived from the original on 28 August 2016. Retrieved 22 November 2016.
- ^ "Sweden may be forced to lift ban on mercury". The Local. 21 April 2012. Archived from the original on 28 August 2016. Retrieved 22 November 2016.
- ^ "Survey of mercury and mercury compounds" (PDF). Miljøstyrelsen. 2014. Retrieved 21 December 2023.
Further reading
[edit]- Johnston, Andrew Scott (15 September 2013). Mercury and the Making of California: Mining, Landscape, and Race, 1840–1890. TotalBoox, TBX. University Press of Colorado. ISBN 978-1-4571-8399-7. OCLC 969039240.
External links
[edit]- Chemistry in its element podcast (MP3) from the Royal Society of Chemistry's Chemistry World: Mercury
- Mercury at The Periodic Table of Videos (University of Nottingham)
- Centers for Disease Control and Prevention – Mercury Topic
- EPA fish consumption guidelines
- Hg 80 Mercury
- Material Safety Data Sheet – Mercury ICSC 0056
- Stopping Pollution: Mercury – Oceana
- Natural Resources Defense Council (NRDC): Mercury Contamination in Fish guide – NRDC
- NLM Hazardous Substances Databank – Mercury
- BBC – Earth News – Mercury "turns" wetland birds such as ibises homosexual
- Changing Patterns in the Use, Recycling, and Material Substitution of Mercury in the United States United States Geological Survey
- Thermodynamical data on liquid mercury.
- . Encyclopædia Britannica (11th ed.). 1911.
- usgs.gov: Mercury (Mineral Commodity Summaries 2025)
Mercury (element)
View on GrokipediaProperties
Physical properties
Mercury exists as a silvery-white liquid at standard temperature and pressure, unique among metallic elements, with a melting point of −38.87 °C and a boiling point of 356.73 °C.[1] Its high density of 13.534 g/cm³ at 25 °C contributes to its ability to support objects denser than water but less dense than itself, such as a British pound coin.[1] The element exhibits high surface tension of 470 dynes/cm at 20 °C, leading to the formation of coherent droplets and beads rather than spreading.[1] Vapor pressure is 0.002 mmHg at 25 °C, higher than most metals, facilitating evaporation at ambient conditions.[1] Optically, mercury displays a metallic luster with reflectivity around 73% and a refractive index ranging from 1.6 to 1.9 at 20 °C, properties that historically enabled its use in mirrors despite chemical limitations. [1] The coefficient of volume thermal expansion is approximately 182 × 10^{-6} /°C near room temperature, a value exploited in thermometers for precise volume-based temperature measurement due to its uniformity. In solid form, below its melting point, mercury adopts a rhombohedral crystal structure in the α phase at atmospheric pressure.[1] Under high pressure, it undergoes phase transitions to denser allotropes: β phase above about 1 GPa, γ phase (monoclinic) around 2-6 GPa, and δ phase at higher pressures exceeding 10 GPa, reflecting compression-induced changes in atomic packing.[6] [7]Chemical properties
Mercury exhibits low reactivity with most common reagents at ambient conditions, resisting oxidation by air or water and showing no reaction with dilute acids such as hydrochloric or sulfuric acid. However, it dissolves in oxidizing acids like concentrated nitric acid or sulfuric acid, and particularly in aqua regia—a 3:1 mixture of concentrated hydrochloric and nitric acids—yielding mercury(II) ions and chloride complexes.[8]/22%3A_d-Block_Metal_Chemistry_-The_Heavier_Metals/22.13%3A_Group_12-_Cadmium_and_Mercury/22.13A%3A_The_Metals) This solubility in aqua regia stems from the generation of nascent chlorine and nitrosyl chloride, which facilitate oxidation to Hg(II).[9] The element predominantly adopts +1 and +2 oxidation states, with the +1 state occurring as the dimeric Hg₂²⁺ ion rather than monomeric Hg⁺. This Hg(I) species is thermodynamically unstable, undergoing disproportionation via the reaction 2Hg₂²⁺ → 2Hg²⁺ + Hg(l), driven by the favorable formation of metallic mercury and the stability of the Hg²⁺ aquo complex./Qualitative_Analysis/Characteristic_Reactions_of_Select_Metal_Ions/Characteristic_Reactions_of_Mercury_Ions_(Hg_and_Hg))[1] Mercury's reluctance to form stable oxides contributes to its anomalous nobility among metals; elemental mercury does not tarnish in air and requires heating above 300°C in oxygen to produce mercury(II) oxide (HgO), which itself decomposes at around 500°C.[10] Mercury readily forms amalgams—alloys with other metals such as gold, silver, sodium, and aluminum—owing to its low melting point and ability to dissolve these elements into its liquid matrix without requiring elevated temperatures.[11] In electrochemical contexts, mercury displays reversible redox behavior, with the Hg²⁺/Hg couple exhibiting a standard reduction potential of +0.854 V versus the standard hydrogen electrode, reflecting its moderate oxidizing power compared to other heavy metals.[12] Its catalytic properties arise from surface adsorption capabilities, enabling facilitation of reactions like acetylene hydrochlorination in supported systems, though elemental mercury's inertness limits broad catalytic utility.[13]Isotopes
Mercury has seven stable isotopes, ranging from 196Hg to 204Hg, with 202Hg being the most abundant at approximately 29.65%. These isotopes contribute to the element's standard atomic weight of 200.592(3) u, determined from weighted averages of their masses and natural abundances. The stable isotopes exhibit no observed radioactive decay, though theoretical predictions suggest potential instability for some over extremely long timescales.[14][15]| Isotope | Atomic Mass (u) | Natural Abundance (%) |
|---|---|---|
| 196Hg | 195.96583(2) | 0.15(1) |
| 198Hg | 197.966752(3) | 9.97(20) |
| 199Hg | 198.968280(3) | 16.87(16) |
| 200Hg | 199.968326(3) | 23.10(9) |
| 201Hg | 200.970302(3) | 13.18(8) |
| 202Hg | 201.970643(3) | 29.65(30) |
| 204Hg | 203.973493(5) | 6.85(11) |









