Recent from talks
Nothing was collected or created yet.
Human evolution
View on Wikipedia
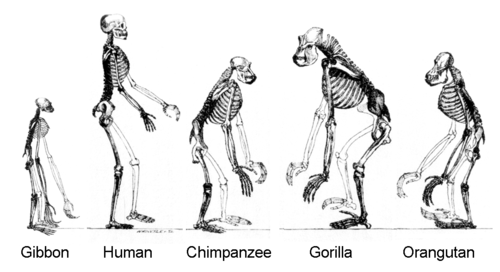
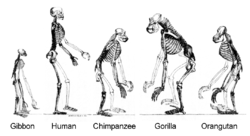
Homo sapiens is a distinct species of the hominid family of primates, which also includes all the great apes.[1] Over their evolutionary history, humans gradually developed traits such as bipedalism, dexterity, and complex language,[2] as well as interbreeding with other hominins (a tribe of the African hominid subfamily),[3] indicating that human evolution was not linear but weblike.[4][5][6][7] The study of the origins of humans involves several scientific disciplines, including physical and evolutionary anthropology, paleontology, and genetics; the field is also known by the terms anthropogeny, anthropogenesis, and anthropogony[8][9]—with the latter two sometimes used to refer to the related subject of hominization.
Primates diverged from other mammals about 85 million years ago (mya), in the Late Cretaceous period, with their earliest fossils appearing over 55 mya, during the Paleocene.[10] Primates produced successive clades leading to the ape superfamily, which gave rise to the hominid and the gibbon families; these diverged some 15–20 mya. African and Asian hominids (including orangutans) diverged about 14 mya. Hominins (including the Australopithecine and Panina subtribes) parted from the Gorillini tribe between 8 and 9 mya; Australopithecine (including the extinct biped ancestors of humans) separated from the Pan genus (containing chimpanzees and bonobos) 4–7 mya.[11] The Homo genus is evidenced by the appearance of H. habilis over 2 mya,[a] while anatomically modern humans emerged in Africa approximately 300,000 years ago.
Before Homo
[edit]Early evolution of primates
[edit]The evolutionary history of primates can be traced back 65 million years.[12][13][14][15][16] The details of the origins and early evolution of primates are however still unknown to a large extent due to scarcity of fossil evidence. One of the oldest known primate-like mammal species, the Plesiadapis, came from North America;[17][18][19][20][21][22] another, Archicebus, came from China.[23] Other such early primates include Altiatlasius and Algeripithecus, which were found in Northern Africa.[24][25] Other similar basal primates were widespread in Eurasia and Africa during the tropical conditions of the Paleocene and Eocene.

David R. Begun theorized that early primates flourished first in Eurasia and that a lineage leading to the African apes and humans, including Dryopithecus, migrated south from Europe or Western Asia into Africa.[26] However, earlier potential ancestors of primates such as Altiatlasius are known to have lived on the African continent much earlier than Dryopithecus.[24]
The surviving tropical population of primates—which is seen most completely in the Upper Eocene and lowermost Oligocene fossil beds of the Faiyum depression southwest of Cairo—gave rise to all extant primate species, including the lemurs of Madagascar, lorises of Southeast Asia, galagos or "bush babies" of Africa, and to the anthropoids, which are the Platyrrhines or New World monkeys, the Catarrhines or Old World monkeys, and the great apes, including humans and other hominids.
The earliest known catarrhine is Kamoyapithecus from the uppermost Oligocene at Eragaleit in the northern Great Rift Valley in Kenya, dated to 24 million years ago.[27] Its ancestry is thought to be species related to Aegyptopithecus, Propliopithecus, and Parapithecus from the Faiyum, at around 35 mya.[28] In 2010, Saadanius was described as a close relative of the last common ancestor of the crown catarrhines, and tentatively dated to 29–28 mya, helping to fill an 11-million-year gap in the fossil record.[29]

In the Early Miocene, about 22 million years ago, the many kinds of arboreally-adapted (tree-dwelling) primitive catarrhines from East Africa suggest a long history of prior diversification. Fossils at 20 million years ago include fragments attributed to Victoriapithecus, the earliest Old World monkey. Among the genera thought to be in the ape lineage leading up to 13 million years ago are Proconsul, Rangwapithecus, Dendropithecus, Limnopithecus, Nacholapithecus, Equatorius, Nyanzapithecus, Afropithecus, Heliopithecus, and Kenyapithecus, all from East Africa.
The presence of other generalized non-cercopithecids of Middle Miocene from sites far distant, such as Otavipithecus from cave deposits in Namibia, and Pierolapithecus and Dryopithecus from France, Spain and Austria, is evidence of a wide diversity of forms across Africa and the Mediterranean basin during the relatively warm and equable climatic regimes of the Early and Middle Miocene. The youngest of the Miocene hominoids, Oreopithecus, is from coal beds in Italy that have been dated to 9 million years ago.
Molecular evidence indicates that the lineage of gibbons diverged from the line of great apes some 18–12 mya, and that of orangutans (subfamily Ponginae)[b] diverged from the other great apes at about 12 million years; there are no fossils that clearly document the ancestry of gibbons, which may have originated in a so-far-unknown Southeast Asian hominoid population, but fossil proto-orangutans may be represented by Sivapithecus from India and Griphopithecus from Turkey, dated to around 10 mya.[30]
Hominidae subfamily Homininae (African hominids) diverged from Ponginae (orangutans) about 14 mya. Hominins (including humans and the Australopithecine and Panina subtribes) parted from the Gorillini tribe (gorillas) between 8 and 9 mya; Australopithecine (including the extinct biped ancestors of humans) separated from the Pan genus (containing chimpanzees and bonobos) 4–7 mya.[11] The Homo genus is evidenced by the appearance of H. habilis over 2 mya,[a] while anatomically modern humans emerged in Africa approximately 300,000 years ago.
Divergence of the human clade from other great apes
[edit]
Species close to the last common ancestor of gorillas, chimpanzees and humans may be represented by Nakalipithecus fossils found in Kenya. Molecular evidence suggests that between 8 and 4 million years ago, first the gorillas, and then the chimpanzees (genus Pan) split off from the line leading to the humans. Human DNA is approximately 98.4% identical to that of chimpanzees when comparing single nucleotide polymorphisms (see human evolutionary genetics). The fossil record, however, of gorillas and chimpanzees is limited; both poor preservation – rain forest soils tend to be acidic and dissolve bone – and sampling bias probably contribute to this problem.
Other hominins probably adapted to the drier environments outside the equatorial belt; and there they encountered antelope, hyenas, dogs, pigs, elephants, horses, and others. The equatorial belt contracted after about 8 million years ago, and there is very little fossil evidence for the split—thought to have occurred around that time—of the hominin lineage from the lineages of gorillas and chimpanzees. The earliest fossils argued by some to belong to the human lineage are Sahelanthropus tchadensis (7 Ma) and Orrorin tugenensis (6 Ma), followed by Ardipithecus (5.5–4.4 Ma), with species Ar. kadabba and Ar. ramidus.
It has been argued in a study of the life history of Ar. ramidus that the species provides evidence for a suite of anatomical and behavioral adaptations in very early hominins unlike any species of extant great ape.[32] This study demonstrated affinities between the skull morphology of Ar. ramidus and that of infant and juvenile chimpanzees, suggesting the species evolved a juvenalised or paedomorphic craniofacial morphology via heterochronic dissociation of growth trajectories. It was also argued that the species provides support for the notion that very early hominins, akin to bonobos (Pan paniscus) the less aggressive species of the genus Pan, may have evolved via the process of self-domestication. Consequently, arguing against the so-called "chimpanzee referential model"[33] the authors suggest it is no longer tenable to use chimpanzee (Pan troglodytes) social and mating behaviors in models of early hominin social evolution. When commenting on the absence of aggressive canine morphology in Ar. ramidus and the implications this has for the evolution of hominin social psychology, they wrote:
Of course Ar. ramidus differs significantly from bonobos, bonobos having retained a functional canine honing complex. However, the fact that Ar. ramidus shares with bonobos reduced sexual dimorphism, and a more paedomorphic form relative to chimpanzees, suggests that the developmental and social adaptations evident in bonobos may be of assistance in future reconstructions of early hominin social and sexual psychology. In fact the trend towards increased maternal care, female mate selection and self-domestication may have been stronger and more refined in Ar. ramidus than what we see in bonobos.[32]: 128
The authors argue that many of the basic human adaptations evolved in the ancient forest and woodland ecosystems of late Miocene and early Pliocene Africa. Consequently, they argue that humans may not represent evolution from a chimpanzee-like ancestor as has traditionally been supposed. This suggests many modern human adaptations represent phylogenetically deep traits and that the behavior and morphology of chimpanzees may have evolved subsequent to the split with the common ancestor they share with humans.
Genus Australopithecus
[edit]
The genus Australopithecus evolved in eastern Africa around 4 million years ago before spreading throughout the continent and eventually becoming extinct 2 million years ago. During this time period various forms of australopiths existed, including Australopithecus anamensis, A. afarensis, A. sediba, and A. africanus. There is still some debate among academics whether certain African hominid species of this time, such as A. robustus and A. boisei, constitute members of the same genus; if so, they would be considered to be "robust australopiths" while the others would be considered "gracile australopiths". However, if these species do indeed constitute their own genus, then they may be given their own name, Paranthropus.
- Australopithecus (4–1.8 Ma), with species A. anamensis, A. afarensis, A. africanus, A. bahrelghazali, A. garhi, and A. sediba;
- Kenyanthropus (3–2.7 Ma), with species K. platyops;
- Paranthropus (3–1.2 Ma), with species P. aethiopicus, P. boisei, and P. robustus
A new proposed species Australopithecus deyiremeda is claimed to have been discovered living at the same time period of A. afarensis. There is debate whether A. deyiremeda is a new species or is A. afarensis.[34] Australopithecus prometheus, otherwise known as Little Foot, has recently been dated at 3.67 million years old through a new dating technique, making the genus Australopithecus as old as afarensis.[35] Given the opposable big toe found on Little Foot, it seems that the specimen was a good climber. It is thought given the night predators of the region that he built a nesting platform at night in the trees in a similar fashion to chimpanzees and gorillas.

Evolution of genus Homo
[edit]−10 — – −9 — – −8 — – −7 — – −6 — – −5 — – −4 — – −3 — – −2 — – −1 — – 0 — |
| |||||||||||||||||||||||||||
The earliest documented representative of the genus Homo is the Ledi jaw, which is dated 2.75 - [36][37] 2.8 million years ago (mya), [38][39] and is arguably the earliest species for which there is positive evidence of the use of stone tools. The brains of these early hominins were about the same size as that of a chimpanzee, although it has been suggested that this was the time in which the human SRGAP2 gene doubled, producing a more rapid wiring of the frontal cortex. During the next million years a process of rapid encephalization occurred, and with the arrival of Homo erectus and Homo ergaster in the fossil record, cranial capacity had doubled to 850 cm3.[40] (Such an increase in human brain size is equivalent to each generation having 125,000 more neurons than their parents.) It is believed that H. erectus and H. ergaster were the first to use fire and complex tools, and they were the first of the hominin line to leave Africa, spreading throughout Africa, Asia, and Europe between 1.3 to 1.8 mya.
According to the recent African origin theory, modern humans evolved in Africa possibly from H. heidelbergensis, H. rhodesiensis or H. antecessor and migrated out of the continent some 50,000 to 100,000 years ago, gradually replacing local populations of H. erectus, Denisova hominins, H. floresiensis, H. luzonensis and H. neanderthalensis, whose ancestors had left Africa in earlier migrations.[41][42][43][44][45] Archaic Homo sapiens, the forerunner of anatomically modern humans, evolved in the Middle Paleolithic between 400,000 and 250,000 years ago.[46][47][48] Recent DNA evidence suggests that several haplotypes of Neanderthal origin are present among all non-African populations, and Neanderthals and other hominins, such as Denisovans, may have contributed up to 6% of their genome to present-day humans, suggestive of a limited interbreeding between these species.[49][50][51] According to some anthropologists, the transition to behavioral modernity with the development of symbolic culture, language, and specialized lithic technology happened around 50,000 years ago (beginning of the Upper Paleolithic),[52] although others point to evidence of a gradual change over a longer time span during the Middle Paleolithic.[53]

Homo sapiens is the only extant species of its genus, Homo. While some (extinct) Homo species might have been ancestors of Homo sapiens, many, perhaps most, were likely "cousins", having speciated away from the ancestral hominin line.[55][56] There is yet no consensus as to which of these groups should be considered a separate species and which should be subspecies; this may be due to the dearth of fossils or to the slight differences used to classify species in the genus Homo.[56] The Sahara pump theory (describing an occasionally passable "wet" Sahara desert) provides one possible explanation of the intermittent migration and speciation in the genus Homo.
Based on archaeological and paleontological evidence, it has been possible to infer, to some extent, the ancient dietary practices[57] of various Homo species and to study the role of diet in physical and behavioral evolution within Homo.[58][59][60][61][62]
Some anthropologists and archaeologists subscribe to the Toba catastrophe theory, which posits that the supereruption of Lake Toba on Sumatra in Indonesia some 70,000 years ago caused global starvation,[63] killing the majority of humans and creating a population bottleneck that affected the genetic inheritance of all humans today.[64] The genetic and archaeological evidence for this remains in question however.[65] A 2023 genetic study suggests that a similar human population bottleneck of between 1,000 and 100,000 survivors occurred "around 930,000 and 813,000 years ago ... lasted for about 117,000 years and brought human ancestors close to extinction."[66][67]
H. habilis and H. gautengensis
[edit]Homo habilis lived from about 2.8[39] to 1.4 Ma. The species evolved in South and East Africa in the Late Pliocene or Early Pleistocene, 2.5–2 Ma, when it diverged from the australopithecines with the development of smaller molars and larger brains. One of the first known hominins, it made tools from stone and perhaps animal bones, leading to its name homo habilis (Latin 'handy man') bestowed by discoverer Louis Leakey. Some scientists have proposed moving this species from Homo into Australopithecus due to the morphology of its skeleton being more adapted to living in trees rather than walking on two legs like later hominins.[68]
In May 2010, a new species, Homo gautengensis, was discovered in South Africa.[69]
H. rudolfensis and H. georgicus
[edit]These are proposed species names for fossils from about 1.9–1.6 Ma, whose relation to Homo habilis is not yet clear.
- Homo rudolfensis refers to a single, incomplete skull from Kenya. Scientists have suggested that this was a specimen of Homo habilis, but this has not been confirmed.[70]
- Homo georgicus, from Georgia, may be an intermediate form between Homo habilis and Homo erectus,[71] or a subspecies of Homo erectus.[72]
H. ergaster and H. erectus
[edit]
The first fossils of Homo erectus were discovered by Dutch physician Eugene Dubois in 1891 on the Indonesian island of Java. He originally named the material Anthropopithecus erectus (1892–1893, considered at this point as a chimpanzee-like fossil primate) and Pithecanthropus erectus (1893–1894, changing his mind as of based on its morphology, which he considered to be intermediate between that of humans and apes).[73] Years later, in the 20th century, the German physician and paleoanthropologist Franz Weidenreich (1873–1948) compared in detail the characters of Dubois' Java Man, then named Pithecanthropus erectus, with the characters of the Peking Man, then named Sinanthropus pekinensis. Weidenreich concluded in 1940 that because of their anatomical similarity with modern humans it was necessary to gather all these specimens of Java and China in a single species of the genus Homo, the species H. erectus.[74][75]
Homo erectus lived from about 1.8 Ma to about 108,000 years ago.[76] This population appears to have died out when the savannah corridors closed, and tropical jungle took over.;[77] however, nearby H. floresiensis survived it. The early phase of H. erectus, from 1.8 to 1.25 Ma, is considered by some to be a separate species, H. ergaster, or as H. erectus ergaster, a subspecies of H. erectus. Many paleoanthropologists now use the term Homo ergaster for the non-Asian forms of this group, and reserve H. erectus only for those fossils that are found in Asia and meet certain skeletal and dental requirements which differ slightly from H. ergaster.
In Africa in the Early Pleistocene, 1.5–1 Ma, some populations of Homo habilis are thought to have evolved larger brains and to have made more elaborate stone tools; these differences and others are sufficient for anthropologists to classify them as a new species, Homo erectus—in Africa.[78] This species also may have used fire to cook meat. Richard Wrangham notes that Homo seems to have been ground dwelling, with reduced intestinal length, smaller dentition, and "brains [swollen] to their current, horrendously fuel-inefficient size",[79] and hypothesizes that control of fire and cooking, which released increased nutritional value, was the key adaptation that separated Homo from tree-sleeping Australopithecines.[80]
H. cepranensis and H. antecessor
[edit]These are proposed as species intermediate between H. erectus and H. heidelbergensis.
- H. antecessor is known from fossils from Spain and England that are dated 1.2 Ma–500 ka.[81][82]
- H. cepranensis refers to a single skull cap from Italy, estimated to be about 800,000 years old.[83]
H. heidelbergensis
[edit]H. heidelbergensis ("Heidelberg Man") lived from about 800,000 to about 300,000 years ago. It is also proposed as Homo sapiens heidelbergensis or Homo sapiens paleohungaricus.[84]
H. rhodesiensis, and the Gawis cranium
[edit]- H. rhodesiensis, estimated to be 300,000–125,000 years old. Most current researchers place Rhodesian Man within the group of Homo heidelbergensis, though other designations such as archaic Homo sapiens and Homo sapiens rhodesiensis have been proposed.
- In February 2006 a fossil, the Gawis cranium, was found which might possibly be a species intermediate between H. erectus and H. sapiens or one of many evolutionary dead ends. The skull from Gawis, Ethiopia, is believed to be 500,000–250,000 years old. Only summary details are known, and the finders have not yet released a peer-reviewed study. Gawis man's facial features suggest that it is either an intermediate species or an example of a "Bodo man" female.[85]
Neanderthal and Denisovan
[edit]
Homo neanderthalensis, alternatively designated as Homo sapiens neanderthalensis,[86] lived in Europe and Asia from 400,000[87] to about 28,000 years ago.[88] There are a number of clear anatomical differences between anatomically modern humans (AMH) and Neanderthal specimens, many relating to the superior Neanderthal adaptation to cold environments. Neanderthal surface to volume ratio was even lower than that among modern Inuit populations, indicating superior retention of body heat.
Neanderthals also had significantly larger brains, as shown from brain endocasts, casting doubt on their intellectual inferiority to modern humans. However, the higher body mass of Neanderthals may have required larger brain mass for body control.[89] Also, recent research by Pearce, Stringer, and Dunbar has shown important differences in brain architecture. The larger size of the Neanderthal orbital chamber and occipital lobe suggests that they had a better visual acuity than modern humans, useful in the dimmer light of glacial Europe.
Neanderthals may have had less brain capacity available for social functions. Inferring social group size from endocranial volume (minus occipital lobe size) suggests that Neanderthal groups may have been limited to 120 individuals, compared to 144[citation needed][90] possible relationships for modern humans. Larger social groups could imply that modern humans had less risk of inbreeding within their clan, trade over larger areas (confirmed in the distribution of stone tools), and faster spread of social and technological innovations. All these may have all contributed to modern Homo sapiens replacing Neanderthal populations by 28,000 BP.[89]
Earlier evidence from sequencing mitochondrial DNA suggested that no significant gene flow occurred between H. neanderthalensis and H. sapiens, and that the two were separate species that shared a common ancestor about 660,000 years ago.[91][92][93] However, a sequencing of the Neanderthal genome in 2010 indicated that Neanderthals did indeed interbreed with anatomically modern humans c. 45,000-80,000 years ago, around the time modern humans migrated out from Africa, but before they dispersed throughout Europe, Asia and elsewhere.[94] The genetic sequencing of a 40,000-year-old human skeleton from Romania showed that 11% of its genome was Neanderthal, implying the individual had a Neanderthal ancestor 4–6 generations previously,[95] in addition to a contribution from earlier interbreeding in the Middle East. Though this interbred Romanian population seems not to have been ancestral to modern humans, the finding indicates that interbreeding happened repeatedly.[96]
All modern non-African humans have about 1% to 4% (or 1.5% to 2.6% by more recent data) of their DNA derived from Neanderthals.[97][94][98] This finding is consistent with recent studies indicating that the divergence of some human alleles dates to one Ma, although this interpretation has been questioned.[99][100] Neanderthals and AMH Homo sapiens could have co-existed in Europe for as long as 10,000 years, during which AMH populations exploded, vastly outnumbering Neanderthals, possibly outcompeting them by sheer numbers.[101]
In 2008, archaeologists working at the site of Denisova Cave in the Altai Mountains of Siberia uncovered a small bone fragment from the fifth finger of a juvenile member of another human species, the Denisovans.[102] Artifacts, including a bracelet, excavated in the cave at the same level were carbon dated to around 40,000 BP. As DNA had survived in the fossil fragment due to the cool climate of the Denisova Cave, both mtDNA and nuclear DNA were sequenced.[49][103]
While the divergence point of the mtDNA was unexpectedly deep in time,[104] the full genomic sequence suggested the Denisovans belonged to the same lineage as Neanderthals, with the two diverging shortly after their line split from the lineage that gave rise to modern humans.[49] Modern humans are known to have overlapped with Neanderthals in Europe and the Near East for possibly more than 40,000 years,[105] and the discovery raises the possibility that Neanderthals, Denisovans, and modern humans may have co-existed and interbred. The existence of this distant branch creates a much more complex picture of humankind during the Late Pleistocene than previously thought.[103][106] Evidence has also been found that as much as 6% of the DNA of some modern Melanesians derive from Denisovans, indicating limited interbreeding in Southeast Asia.[107][108]
Alleles thought to have originated in Neanderthals and Denisovans have been identified at several genetic loci in the genomes of modern humans outside Africa. Human leukocyte antigen (HLA) haplotypes from Denisovans and Neanderthal represent more than half the HLA alleles of modern Eurasians,[51] indicating strong positive selection for these introgressed alleles. Corinne Simoneti at Vanderbilt University, in Nashville and her team have found from medical records of 28,000 people of European descent that the presence of Neanderthal DNA segments may be associated with a higher rate of depression.[109]
The flow of genes from Neanderthal populations to modern humans was not all one way. Sergi Castellano of the Max Planck Institute for Evolutionary Anthropology reported in 2016 that while Denisovan and Neanderthal genomes are more related to each other than they are to us, Siberian Neanderthal genomes show more similarity to modern human genes than do European Neanderthal populations. This suggests Neanderthal populations interbred with modern humans around 100,000 years ago, probably somewhere in the Near East.[110]
Studies of a Neanderthal child at Gibraltar show from brain development and tooth eruption that Neanderthal children may have matured more rapidly than Homo sapiens.[111]
H. floresiensis
[edit]
H. floresiensis, which lived from approximately 190,000 to 50,000 years before present (BP), has been nicknamed the hobbit for its small size, possibly a result of insular dwarfism.[112] H. floresiensis is intriguing both for its size and its age, being an example of a recent species of the genus Homo that exhibits derived traits not shared with modern humans. In other words, H. floresiensis shares a common ancestor with modern humans, but split from the modern human lineage and followed a distinct evolutionary path. The main find was a skeleton believed to be a woman of about 30 years of age. Found in 2003, it has been dated to approximately 18,000 years old. The living woman was estimated to be one meter in height, with a brain volume of just 380 cm3 (considered small for a chimpanzee and less than a third of the H. sapiens average of 1400 cm3).[112]
However, there is an ongoing debate over whether H. floresiensis is indeed a separate species.[113] Some scientists hold that H. floresiensis was a modern H. sapiens with pathological dwarfism.[114] This hypothesis is supported in part, because some modern humans who live on Flores, the Indonesian island where the skeleton was found, are pygmies. This, coupled with pathological dwarfism, could have resulted in a significantly diminutive human. The other major attack on H. floresiensis as a separate species is that it was found with tools only associated with H. sapiens.[114]
The hypothesis of pathological dwarfism, however, fails to explain additional anatomical features that are unlike those of modern humans (diseased or not) but much like those of ancient members of our genus. Aside from cranial features, these features include the form of bones in the wrist, forearm, shoulder, knees, and feet. Additionally, this hypothesis fails to explain the find of multiple examples of individuals with these same characteristics, indicating they were common to a large population, and not limited to one individual.[113]
In 2016, fossil teeth and a partial jaw from hominins assumed to be ancestral to H. floresiensis were discovered[115] at Mata Menge, about 74 km (46 mi) from Liang Bua. They date to about 700,000 years ago[116] and are noted by Australian archaeologist Gerrit van den Bergh for being even smaller than the later fossils.[117]
H. luzonensis
[edit]A small number of specimens from the island of Luzon, dated 50,000 to 67,000 years ago, have recently been assigned by their discoverers, based on dental characteristics, to a novel human species, H. luzonensis.[118]
H. sapiens
[edit]
H. sapiens (the adjective sapiens is Latin for "wise" or "intelligent") emerged in Africa around 300,000 years ago, likely derived from H. heidelbergensis or a related lineage.[119][120] In September 2019, scientists reported the computerized determination, based on 260 CT scans, of a virtual skull shape of the last common human ancestor to modern humans (H. sapiens), representative of the earliest modern humans, and suggested that modern humans arose between 260,000 and 350,000 years ago through a merging of populations in East and South Africa.[121][122]
Between 400,000 years ago and the second interglacial period in the Middle Pleistocene, around 250,000 years ago, the trend in intra-cranial volume expansion and the elaboration of stone tool technologies developed, providing evidence for a transition from H. erectus to H. sapiens. The direct evidence suggests there was a migration of H. erectus out of Africa, then a further speciation of H. sapiens from H. erectus in Africa. A subsequent migration (both within and out of Africa) eventually replaced the earlier dispersed H. erectus. This migration and origin theory is usually referred to as the "recent single-origin hypothesis" or "out of Africa" theory. H. sapiens interbred with archaic humans both in Africa and in Eurasia, in Eurasia notably with Neanderthals and Denisovans.[49][107]
The Toba catastrophe theory, which postulates a population bottleneck for H. sapiens about 70,000 years ago,[123] was controversial from its first proposal in the 1990s and by the 2010s had very little support.[124] Distinctive human genetic variability has arisen as the result of the founder effect, by archaic admixture and by recent evolutionary pressures.
Anatomical changes
[edit]Since Homo sapiens separated from its last common ancestor shared with chimpanzees, human evolution is characterized by a number of morphological, developmental, physiological, behavioral, and environmental changes.[9] Environmental (cultural) evolution discovered much later during the Pleistocene played a significant role in human evolution observed via human transitions between subsistence systems.[125][9] The most significant of these adaptations are bipedalism, increased brain size, lengthened ontogeny (gestation and infancy), and decreased sexual dimorphism. The relationship between these changes is the subject of ongoing debate.[126] Other significant morphological changes included the evolution of a power and precision grip, a change first occurring in H. erectus.[127]
Bipedalism
[edit]
Bipedalism (walking on two legs) is the basic adaptation of the hominid and is considered the main cause behind a suite of skeletal changes shared by all bipedal hominids. The earliest hominin, of presumably primitive bipedalism, is considered to be either Sahelanthropus[128] or Orrorin, both of which arose some 6 to 7 million years ago. The non-bipedal knuckle-walkers, the gorillas and chimpanzees, diverged from the hominin line over a period covering the same time, so either Sahelanthropus or Orrorin may be our last shared ancestor. Ardipithecus, a full biped, arose approximately 5.6 million years ago.[129]
The early bipeds eventually evolved into the australopithecines and still later into the genus Homo. There are several theories of the adaptation value of bipedalism. It is possible that bipedalism was favored because it freed the hands for reaching and carrying food, saved energy during locomotion,[130] enabled long-distance running and hunting, provided an enhanced field of vision, and helped avoid hyperthermia by reducing the surface area exposed to direct sun; features all advantageous for thriving in the new savanna and woodland environment created as a result of the East African Rift Valley uplift versus the previous closed forest habitat.[130][131][132] A 2007 study provides support for the hypothesis that bipedalism evolved because it used less energy than quadrupedal knuckle-walking.[133][134] However, recent studies suggest that bipedality without the ability to use fire would not have allowed global dispersal.[135] This change in gait saw a lengthening of the legs proportionately when compared to the length of the arms, which were shortened through the removal of the need for brachiation. Another change is the shape of the big toe. Recent studies suggest that australopithecines still lived part of the time in trees as a result of maintaining a grasping big toe. This was progressively lost in habilines.
Anatomically, the evolution of bipedalism has been accompanied by a large number of skeletal changes, not just to the legs and pelvis, but also to the vertebral column, feet and ankles, and skull.[136] The femur evolved into a slightly more angular position to move the center of gravity toward the geometric center of the body. The knee and ankle joints became increasingly robust to better support increased weight. To support the increased weight on each vertebra in the upright position, the human vertebral column became S-shaped and the lumbar vertebrae became shorter and wider. In the feet the big toe moved into alignment with the other toes to help in forward locomotion. The arms and forearms shortened relative to the legs making it easier to run. The foramen magnum migrated under the skull and more anterior.[137]
The most significant changes occurred in the pelvic region, where the long downward facing iliac blade was shortened and widened as a requirement for keeping the center of gravity stable while walking;[30] bipedal hominids have a shorter but broader, bowl-like pelvis due to this. A drawback is that the birth canal of bipedal apes is smaller than in knuckle-walking apes, though there has been a widening of it in comparison to that of australopithecine and modern humans, thus permitting the passage of newborns due to the increase in cranial size. This is limited to the upper portion, since further increase can hinder normal bipedal movement.[138]
The shortening of the pelvis and smaller birth canal evolved as a requirement for bipedalism and had significant effects on the process of human birth, which is much more difficult in modern humans than in other primates. During human birth, because of the variation in size of the pelvic region, the fetal head must be in a transverse position (compared to the mother) during entry into the birth canal and rotate about 90 degrees upon exit.[139] The smaller birth canal became a limiting factor to brain size increases in early humans and prompted a shorter gestation period leading to the relative immaturity of human offspring, who are unable to walk much before 12 months and have greater neoteny, compared to other primates, who are mobile at a much earlier age.[132] The increased brain growth after birth and the increased dependency of children on mothers had a major effect upon the female reproductive cycle,[140] and the more frequent appearance of alloparenting in humans when compared with other hominids.[141] Delayed human sexual maturity also led to the evolution of menopause with one explanation, the grandmother hypothesis, providing that elderly women could better pass on their genes by taking care of their daughter's offspring, as compared to having more children of their own.[142][143]
Encephalization
[edit]
The human species eventually developed a much larger brain than that of other primates—typically 1,330 cm3 (81 cu in) in modern humans, nearly three times the size of a chimpanzee or gorilla brain.[146] After a period of stasis with Australopithecus anamensis and Ardipithecus, species which had smaller brains as a result of their bipedal locomotion,[147] the pattern of encephalization started with Homo habilis, whose 600 cm3 (37 cu in) brain was slightly larger than that of chimpanzees. This evolution continued in Homo erectus with 800–1,100 cm3 (49–67 cu in), and reached a maximum in Neanderthals with 1,200–1,900 cm3 (73–116 cu in), larger even than modern Homo sapiens. This brain increase manifested during postnatal brain growth, far exceeding that of other apes (heterochrony). It also allowed for extended periods of social learning and language acquisition in juvenile humans, beginning as much as 2 million years ago. Encephalization may be due to a dependency on calorie-dense, difficult-to-acquire food.[148]
Furthermore, the changes in the structure of human brains may be even more significant than the increase in size.[149][150][151][58] Fossilized skulls shows the brain size in early humans fell within the range of modern humans 300,000 years ago, but only got its present-day brain shape between 100,000 and 35,000 years ago.[152]

The temporal lobes, which contain centers for language processing, have increased disproportionately, as has the prefrontal cortex, which has been related to complex decision-making and moderating social behavior.[146] Encephalization has been tied to increased starches[57] and meat[153][154] in the diet, however a 2022 meta study called into question the role of meat.[155] Other factors are the development of cooking,[156] and it has been proposed that intelligence increased as a response to an increased necessity for solving social problems as human society became more complex.[157] Changes in skull morphology, such as smaller mandibles and mandible muscle attachments, allowed more room for the brain to grow.[158]
The increase in volume of the neocortex also included a rapid increase in size of the cerebellum. Its function has traditionally been associated with balance and fine motor control, but more recently with speech and cognition. The great apes, including hominids, had a more pronounced cerebellum relative to the neocortex than other primates. It has been suggested that because of its function of sensory-motor control and learning complex muscular actions, the cerebellum may have underpinned human technological adaptations, including the preconditions of speech.[159][160][161][162]
The immediate survival advantage of encephalization is difficult to discern, as the major brain changes from Homo erectus to Homo heidelbergensis were not accompanied by major changes in technology. It has been suggested that the changes were mainly social and behavioural, including increased empathic abilities,[163][164] increases in size of social groups,[157][165][166] and increased behavioral plasticity.[167] Humans are unique in the ability to acquire information through social transmission and adapt that information.[168] The emerging field of cultural evolution studies human sociocultural change from an evolutionary perspective.[169]
Sexual dimorphism
[edit]The reduced degree of sexual dimorphism in humans is visible primarily in the reduction of the male canine tooth relative to other ape species (except gibbons) and reduced brow ridges and general robustness of males. Another important physiological change related to sexuality in humans was the evolution of hidden estrus. Humans are the only hominoids in which the female is fertile year round and in which no special signals of fertility are produced by the body (such as genital swelling or overt changes in proceptivity during estrus).[182]
Nonetheless, humans retain a degree of sexual dimorphism in the distribution of body hair and subcutaneous fat, and in the overall size, males being around 15% larger than females.[183] These changes taken together have been interpreted as a result of an increased emphasis on pair bonding as a possible solution to the requirement for increased parental investment due to the prolonged infancy of offspring.[184]
Ulnar opposition
[edit]
The ulnar opposition—the contact between the thumb and the tip of the little finger of the same hand—is unique to the genus Homo,[185] including Neanderthals, the Sima de los Huesos hominins and anatomically modern humans.[186][187] In other primates, the thumb is short and unable to touch the little finger.[186] The ulnar opposition facilitates the precision grip and power grip of the human hand, underlying all the skilled manipulations.
Other changes
[edit]A number of other changes have also characterized the evolution of humans, among them an increased reliance on vision rather than smell (highly reduced olfactory bulb); a longer juvenile developmental period and higher infant dependency;[188] a smaller gut and small, misaligned teeth; faster basal metabolism;[189] loss of body hair;[190] an increase in eccrine sweat gland density that is ten times higher than any other catarrhinian primates,[191] yet humans use 30% to 50% less water per day compared to chimps and gorillas;[192] more REM sleep but less sleep in total;[193] a change in the shape of the dental arcade from u-shaped to parabolic; development of a chin (found in Homo sapiens alone); styloid processes; and a descended larynx. As the human hand and arms adapted to the making of tools and were used less for climbing, the shoulder blades changed too. As a side effect, it allowed human ancestors to throw objects with greater force, speed and accuracy.[194]
Use of tools
[edit]



The use of tools has been interpreted as a sign of intelligence, and it has been theorized that tool use may have stimulated certain aspects of human evolution, especially the continued expansion of the human brain.[196] Paleontology has yet to explain the expansion of this organ over millions of years despite being extremely demanding in terms of energy consumption. The brain of a modern human consumes, on average, about 13 watts (260 kilocalories per day), a fifth of the body's resting power consumption.[197] Increased tool use would allow hunting for energy-rich meat products, and would enable processing more energy-rich plant products. Researchers have suggested that early hominins were thus under evolutionary pressure to increase their capacity to create and use tools.[198]
Precisely when early humans started to use tools is difficult to determine, because the more primitive these tools are (for example, sharp-edged stones) the more difficult it is to decide whether they are natural objects or human artifacts.[196] There is some evidence that the australopithecines (4 Ma) may have used broken bones as tools, but this is debated.[199]
Many species make and use tools, but it is the human genus that dominates the areas of making and using more complex tools. The oldest known tools are flakes from West Turkana, Kenya, which date to 3.3 million years ago.[200] The next oldest stone tools are from Gona, Ethiopia, and are considered the beginning of the Oldowan technology. These tools date to about 2.6 million years ago.[201] A Homo fossil was found near some Oldowan tools, and its age was noted at 2.3 million years old, suggesting that maybe the Homo species did indeed create and use these tools. It is a possibility but does not yet represent solid evidence.[202] The third metacarpal styloid process enables the hand bone to lock into the wrist bones, allowing for greater amounts of pressure to be applied to the wrist and hand from a grasping thumb and fingers. It allows humans the dexterity and strength to make and use complex tools. This unique anatomical feature separates humans from other apes and other nonhuman primates, and is not seen in human fossils older than 1.8 million years.[203]
Bernard Wood noted that Paranthropus co-existed with the early Homo species in the area of the "Oldowan Industrial Complex" over roughly the same span of time. Although there is no direct evidence which identifies Paranthropus as the tool makers, their anatomy lends to indirect evidence of their capabilities in this area. Most paleoanthropologists agree that the early Homo species were indeed responsible for most of the Oldowan tools found. They argue that when most of the Oldowan tools were found in association with human fossils, Homo was always present, but Paranthropus was not.[202]
In 1994, Randall Susman used the anatomy of opposable thumbs as the basis for his argument that both the Homo and Paranthropus species were toolmakers. He compared bones and muscles of human and chimpanzee thumbs, finding that humans have 3 muscles which are lacking in chimpanzees. Humans also have thicker metacarpals with broader heads, allowing more precise grasping than the chimpanzee hand can perform. Susman posited that modern anatomy of the human opposable thumb is an evolutionary response to the requirements associated with making and handling tools and that both species were indeed toolmakers.[202]
Transition to behavioral modernity
[edit]Anthropologists describe modern human behavior to include cultural and behavioral traits such as specialization of tools, use of jewellery and images (such as cave drawings), organization of living space, rituals (such as grave gifts), specialized hunting techniques, exploration of less hospitable geographical areas, and barter trade networks, as well as more general traits such as language and complex symbolic thinking. Debate continues as to whether a "revolution" led to modern humans ("big bang of human consciousness"), or whether the evolution was more gradual.[53]
Until about 50,000–40,000 years ago, the use of stone tools seems to have progressed stepwise. Each phase (H. habilis, H. ergaster, H. neanderthalensis) marked a new technology, followed by very slow development until the next phase. Currently paleoanthropologists are debating whether these Homo species possessed some or many modern human behaviors. They seem to have been culturally conservative, maintaining the same technologies and foraging patterns over very long periods.
Around 50,000 BP, human culture started to evolve more rapidly. The transition to behavioral modernity has been characterized by some as a "Great Leap Forward",[204] or as the "Upper Palaeolithic Revolution",[205] due to the sudden appearance in the archaeological record of distinctive signs of modern behavior and big game hunting.[206] Evidence of behavioral modernity significantly earlier also exists from Africa, with older evidence of abstract imagery, widened subsistence strategies, more sophisticated tools and weapons, and other "modern" behaviors, and many scholars have recently argued that the transition to modernity occurred sooner than previously believed.[53][207][208][209]
Other scholars consider the transition to have been more gradual, noting that some features had already appeared among archaic African Homo sapiens 300,000–200,000 years ago.[210][211][212][213][214] Recent evidence suggests that the Australian Aboriginal population separated from the African population 75,000 years ago, and that they made a 160 km (99 mi) sea journey 60,000 years ago, which may diminish the significance of the Upper Paleolithic Revolution.[215]
Modern humans started burying their dead, making clothing from animal hides, hunting with more sophisticated techniques (such as using pit traps or driving animals off cliffs), and cave painting.[216] As human culture advanced, different populations innovated existing technologies: artifacts such as fish hooks, buttons, and bone needles show signs of cultural variation, which had not been seen prior to 50,000 BP. Typically, the older H. neanderthalensis populations did not vary in their technologies, although the Chatelperronian assemblages have been found to be Neanderthal imitations of H. sapiens Aurignacian technologies.[217]
Recent and ongoing human evolution
[edit]Anatomically modern human populations continue to evolve, as they are affected by both natural selection and genetic drift. Although selection pressure on some traits, such as resistance to smallpox, has decreased in the modern age, humans are still undergoing natural selection for many other traits. Some of these are due to specific environmental pressures, while others are related to lifestyle changes since the development of agriculture (10,000 years ago), urbanization (5,000), and industrialization (250 years ago). It has been argued that human evolution has accelerated since the development of agriculture 10,000 years ago and civilization some 5,000 years ago, resulting, it is claimed, in substantial genetic differences between different current human populations,[218] and more recent research indicates that for some traits, the developments and innovations of human culture have driven a new form of selection that coexists with, and in some cases has largely replaced, natural selection.[219]

Particularly conspicuous is variation in superficial characteristics, such as Afro-textured hair, or the recent evolution of light skin and blond hair in some populations, which are attributed to differences in climate. Particularly strong selective pressures have resulted in high-altitude adaptation in humans, with different ones in different isolated populations. Studies of the genetic basis show that some developed very recently, with Tibetans evolving over 3,000 years to have high proportions of an allele of EPAS1 that is adaptive to high altitudes.
Other evolution is related to endemic diseases: the presence of malaria selects for sickle cell trait (the heterozygous form of sickle cell gene), while in the absence of malaria, the health effects of sickle-cell anemia select against this trait. For another example, the population at risk of the severe debilitating disease kuru has significant over-representation of an immune variant of the prion protein gene G127V versus non-immune alleles. The frequency of this genetic variant is due to the survival of immune persons.[221][222] Some reported trends remain unexplained and the subject of ongoing research in the novel field of evolutionary medicine: polycystic ovary syndrome (PCOS) reduces fertility and thus is expected to be subject to extremely strong negative selection, but its relative commonality in human populations suggests a counteracting selection pressure. The identity of that pressure remains the subject of some debate.[223]
Recent human evolution related to agriculture includes genetic resistance to infectious disease that has appeared in human populations by crossing the species barrier from domesticated animals,[224] as well as changes in metabolism due to changes in diet, such as lactase persistence.
Culturally-driven evolution can defy the expectations of natural selection: while human populations experience some pressure that drives a selection for producing children at younger ages, the advent of effective contraception, higher education, and changing social norms have driven the observed selection in the opposite direction.[225] However, culturally-driven selection need not necessarily work counter or in opposition to natural selection: some proposals to explain the high rate of recent human brain expansion indicate a kind of feedback whereupon the brain's increased social learning efficiency encourages cultural developments that in turn encourage more efficiency, which drive more complex cultural developments that demand still-greater efficiency, and so forth.[226] Culturally-driven evolution has an advantage in that in addition to the genetic effects, it can be observed also in the archaeological record: the development of stone tools across the Palaeolithic period connects to culturally-driven cognitive development in the form of skill acquisition supported by the culture and the development of increasingly complex technologies and the cognitive ability to elaborate them.[227]
In contemporary times, since industrialization, some trends have been observed: for instance, menopause is evolving to occur later.[228] Other reported trends appear to include lengthening of the human reproductive period and reduction in cholesterol levels, blood glucose and blood pressure in some populations.[228]
History of study
[edit]Before Darwin
[edit]The name Homo of the biological genus to which humans belong is Latin for 'human'.[e] It was chosen originally by Carl Linnaeus in his classification system.[f] The English word human is from the Latin humanus, the adjectival form of homo. The Latin homo derives from the Indo-European root *dhghem, or 'earth'.[229] Linnaeus and other scientists of his time also considered the great apes to be the closest relatives of humans based on morphological and anatomical similarities.[230]
Darwin
[edit]The possibility of linking humans with earlier apes by descent became clear only after 1859 with the publication of Charles Darwin's On the Origin of Species, in which he argued for the idea of the evolution of new species from earlier ones. Darwin's book did not address the question of human evolution, saying only that "Light will be thrown on the origin of man and his history."[231]
The first debates about the nature of human evolution arose between Thomas Henry Huxley and Richard Owen. Huxley argued for human evolution from apes by illustrating many of the similarities and differences between humans and other apes, and did so particularly in his 1863 book Evidence as to Man's Place in Nature. Many of Darwin's early supporters (such as Alfred Russel Wallace and Charles Lyell) did not initially agree that the origin of the mental capacities and the moral sensibilities of humans could be explained by natural selection, though this later changed. Darwin applied the theory of evolution and sexual selection to humans in his 1871 book The Descent of Man, and Selection in Relation to Sex.[232]
First fossils
[edit]A major problem in the 19th century was the lack of fossil intermediaries. Neanderthal remains were discovered in a limestone quarry in 1856, three years before the publication of On the Origin of Species, and Neanderthal fossils had been discovered in Gibraltar even earlier, but it was originally claimed that these were the remains of a modern human who had suffered some kind of illness.[233] Despite the 1891 discovery by Eugène Dubois of what is now called Homo erectus at Trinil, Java, it was only in the 1920s when such fossils were discovered in Africa, that intermediate species began to accumulate.[234] In 1925, Raymond Dart described Australopithecus africanus.[235] The type specimen was the Taung Child, an australopithecine infant which was discovered in a cave. The child's remains were a remarkably well-preserved tiny skull and an endocast of the brain.
Although the brain was small (410 cm3), its shape was rounded, unlike that of chimpanzees and gorillas, and more like a modern human brain. Also, the specimen showed short canine teeth, and the position of the foramen magnum (the hole in the skull where the spine enters) was evidence of bipedal locomotion. All of these traits convinced Dart that the Taung Child was a bipedal human ancestor, a transitional form between apes and humans.
The East African fossils
[edit]
During the 1960s and 1970s, hundreds of fossils were found in East Africa in the regions of the Olduvai Gorge and Lake Turkana. These searches were carried out by the Leakey family, with Louis Leakey and his wife Mary Leakey, and later their son Richard and daughter-in-law Meave, fossil hunters and paleoanthropologists. From the fossil beds of Olduvai and Lake Turkana they amassed specimens of the early hominins: the australopithecines and Homo species, and even H. erectus.
These finds cemented Africa as the cradle of humankind. In the late 1970s and the 1980s, Ethiopia emerged as the new hot spot of paleoanthropology after "Lucy", the most complete fossil member of the species Australopithecus afarensis, was found in 1974 by Donald Johanson near Hadar in the desertic Afar Triangle region of northern Ethiopia. Although the specimen had a small brain, the pelvis and leg bones were almost identical in function to those of modern humans, showing with certainty that these hominins had walked erect.[236] Lucy was classified as a new species, Australopithecus afarensis, which is thought to be more closely related to the genus Homo as a direct ancestor, or as a close relative of an unknown ancestor, than any other known hominid or hominin from this early time range.[237] (The specimen was nicknamed "Lucy" after the Beatles' song "Lucy in the Sky with Diamonds", which was played loudly and repeatedly in the camp during the excavations.)[238] The Afar Triangle area would later yield discovery of many more hominin fossils, particularly those uncovered or described by teams headed by Tim D. White in the 1990s, including Ardipithecus ramidus and A. kadabba.[239]
In 2013, fossil skeletons of Homo naledi, an extinct species of hominin assigned (provisionally) to the genus Homo, were found in the Rising Star Cave system, a site in South Africa's Cradle of Humankind region in Gauteng province near Johannesburg.[240][241] As of September 2015[update], fossils of at least fifteen individuals, amounting to 1,550 specimens, have been excavated from the cave.[241] The species is characterized by a body mass and stature similar to small-bodied human populations, a smaller endocranial volume similar to Australopithecus, and a cranial morphology (skull shape) similar to early Homo species. The skeletal anatomy combines primitive features known from australopithecines with features known from early hominins. The individuals show signs of having been deliberately disposed of within the cave near the time of death. The fossils were dated close to 250,000 years ago,[242] and thus are not ancestral but contemporary with the first appearance of larger-brained anatomically modern humans.[243]
The genetic revolution
[edit]The genetic revolution in studies of human evolution started when Vincent Sarich and Allan Wilson measured the strength of immunological cross-reactions of blood serum albumin between pairs of creatures, including humans and African apes (chimpanzees and gorillas).[244] The strength of the reaction could be expressed numerically as an immunological distance, which was in turn proportional to the number of amino acid differences between homologous proteins in different species. By constructing a calibration curve of the ID of species' pairs with known divergence times in the fossil record, the data could be used as a molecular clock to estimate the times of divergence of pairs with poorer or unknown fossil records.
In their seminal 1967 paper in Science, Sarich and Wilson estimated the divergence time of humans and apes as four to five million years ago,[244] at a time when standard interpretations of the fossil record gave this divergence as at least 10 to as much as 30 million years. Subsequent fossil discoveries, notably "Lucy", and reinterpretation of older fossil materials, notably Ramapithecus, showed the younger estimates to be correct and validated the albumin method.
Progress in DNA sequencing, specifically mitochondrial DNA (mtDNA) and then Y-chromosome DNA (Y-DNA) advanced the understanding of human origins.[131][245][246] Application of the molecular clock principle revolutionized the study of molecular evolution.
On the basis of a separation from the orangutan between 10 and 20 million years ago, earlier studies of the molecular clock suggested that there were about 76 mutations per generation that were not inherited by human children from their parents; this evidence supported the divergence time between hominins and chimpanzees noted above. However, a 2012 study in Iceland of 78 children and their parents suggests a mutation rate of only 36 mutations per generation; this datum extends the separation between humans and chimpanzees to an earlier period greater than 7 million years ago (Ma). Additional research with 226 offspring of wild chimpanzee populations in eight locations suggests that chimpanzees reproduce at age 26.5 years on average; which suggests the human divergence from chimpanzees occurred between 7 and 13 mya. And these data suggest that Ardipithecus (4.5 Ma), Orrorin (6 Ma) and Sahelanthropus (7 Ma) all may be on the hominid lineage, and even that the separation may have occurred outside the East African Rift region.
Furthermore, analysis of the two species' genes in 2006 provides evidence that after human ancestors had started to diverge from chimpanzees, interspecies mating between "proto-human" and "proto-chimpanzees" nonetheless occurred regularly enough to change certain genes in the new gene pool:
- A new comparison of the human and chimpanzee genomes suggests that after the two lineages separated, they may have begun interbreeding... A principal finding is that the X chromosomes of humans and chimpanzees appear to have diverged about 1.2 million years more recently than the other chromosomes.
The research suggests:
- There were in fact two splits between the human and chimpanzee lineages, with the first being followed by interbreeding between the two populations and then a second split. The suggestion of a hybridization has startled paleoanthropologists, who nonetheless are treating the new genetic data seriously.[247]
The quest for the earliest hominin
[edit]In the 1990s, several teams of paleoanthropologists were working throughout Africa looking for evidence of the earliest divergence of the hominin lineage from the great apes. In 1994, Meave Leakey discovered Australopithecus anamensis. The find was overshadowed by Tim D. White's 1995 discovery of Ardipithecus ramidus, which pushed back the fossil record to 4.2 million years ago.
In 2000, Martin Pickford and Brigitte Senut discovered, in the Tugen Hills of Kenya, a 6-million-year-old bipedal hominin which they named Orrorin tugenensis. And in 2001, a team led by Michel Brunet discovered the skull of Sahelanthropus tchadensis which was dated as 7.2 million years ago, and which Brunet argued was a bipedal, and therefore a hominid—that is, a hominin (cf Hominidae; terms "hominids" and hominins).
Human dispersal
[edit]Anthropologists in the 1980s were divided regarding some details of reproductive barriers and migratory dispersals of the genus Homo. Subsequently, genetics has been used to investigate and resolve these issues. According to the Sahara pump theory evidence suggests that the genus Homo have migrated out of Africa at least three and possibly four times (e.g. Homo erectus, Homo heidelbergensis and two or three times for Homo sapiens). Recent evidence suggests these dispersals are closely related to fluctuating periods of climate change.[252]
Recent evidence suggests that humans may have left Africa half a million years earlier than previously thought. A joint Franco-Indian team has found human artifacts in the Siwalk Hills north of New Delhi dating back at least 2.6 million years. This is earlier than the previous earliest finding of genus Homo at Dmanisi, in Georgia, dating to 1.85 million years. Although controversial, tools found at a Chinese cave strengthen the case that humans used tools as far back as 2.48 million years ago.[253] This suggests that the Asian "Chopper" tool tradition, found in Java and northern China may have left Africa before the appearance of the Acheulian hand axe.
Dispersal of modern Homo sapiens
[edit]Up until the genetic evidence became available, there were two dominant models for the dispersal of modern humans. The multiregional hypothesis proposed that the genus Homo contained only a single interconnected population as it does today (not separate species), and that its evolution took place worldwide continuously over the last couple of million years. This model was proposed in 1988 by Milford H. Wolpoff.[254][255] In contrast, the "out of Africa" model proposed that modern H. sapiens speciated in Africa recently (that is, approximately 200,000 years ago) and the subsequent migration through Eurasia resulted in the nearly complete replacement of other Homo species. This model has been developed by Chris Stringer and Peter Andrews.[256][257]
Sequencing mtDNA and Y-DNA sampled from a wide range of indigenous populations revealed ancestral information relating to both male and female genetic heritage, and strengthened the "out of Africa" theory and weakened the views of multiregional evolutionism.[258] Aligned in genetic tree differences were interpreted as supportive of a recent single origin.[259]
"Out of Africa" has thus gained much support from research using female mitochondrial DNA and the male Y chromosome. After analysing genealogy trees constructed using 133 types of mtDNA, researchers concluded that all were descended from a female African progenitor, dubbed Mitochondrial Eve. "Out of Africa" is also supported by the fact that mitochondrial genetic diversity is highest among African populations.[260]
A broad study of African genetic diversity, headed by Sarah Tishkoff, found the San people had the greatest genetic diversity among the 113 distinct populations sampled, making them one of 14 "ancestral population clusters". The research also located a possible origin of modern human migration in southwestern Africa, near the coastal border of Namibia and Angola.[261] The fossil evidence was insufficient for archaeologist Richard Leakey to resolve the debate about exactly where in Africa modern humans first appeared.[262] Studies of haplogroups in Y-chromosomal DNA and mitochondrial DNA have largely supported a recent African origin.[263] All the evidence from autosomal DNA also predominantly supports a Recent African origin. However, evidence for archaic admixture in modern humans, both in Africa and later, throughout Eurasia has recently been suggested by a number of studies.[264]
Recent sequencing of Neanderthal[97] and Denisovan[49] genomes shows that some admixture with these populations has occurred. All modern human groups outside Africa have 1–4% or (according to more recent research) about 1.5–2.6% Neanderthal alleles in their genome,[98] and some Melanesians have an additional 4–6% of Denisovan alleles. These new results do not contradict the "out of Africa" model, except in its strictest interpretation, although they make the situation more complex. After recovery from a genetic bottleneck that some researchers speculate might be linked to the Toba supervolcano catastrophe, a fairly small group left Africa and interbred with Neanderthals, probably in the Middle East, on the Eurasian steppe or even in North Africa before their departure. Their still predominantly African descendants spread to populate the world. A fraction in turn interbred with Denisovans, probably in southeastern Asia, before populating Melanesia.[107] HLA haplotypes of Neanderthal and Denisova origin have been identified in modern Eurasian and Oceanian populations.[51] The Denisovan EPAS1 gene has also been found in Tibetan populations.[265] Studies of the human genome using machine learning have identified additional genetic contributions in Eurasians from an "unknown" ancestral population potentially related to the Neanderthal-Denisovan lineage.[266]

There are still differing theories on whether there was a single exodus from Africa or several. A multiple dispersal model involves the Southern Dispersal theory,[267][268][269] which has gained support in recent years from genetic, linguistic and archaeological evidence. In this theory, there was a coastal dispersal of modern humans from the Horn of Africa crossing the Bab el Mandib to Yemen at a lower sea level around 70,000 years ago. This group helped to populate Southeast Asia and Oceania, explaining the discovery of early human sites in these areas much earlier than those in the Levant.[267] This group seems to have been dependent upon marine resources for their survival.
Stephen Oppenheimer has proposed a second wave of humans may have later dispersed through the Persian Gulf oases, and the Zagros mountains into the Middle East. Alternatively it may have come across the Sinai Peninsula into Asia, from shortly after 50,000 yrs BP, resulting in the bulk of the human populations of Eurasia. It has been suggested that this second group possibly possessed a more sophisticated "big game hunting" tool technology and was less dependent on coastal food sources than the original group. Much of the evidence for the first group's expansion would have been destroyed by the rising sea levels at the end of each glacial maximum.[267] The multiple dispersal model is contradicted by studies indicating that the populations of Eurasia and the populations of Southeast Asia and Oceania are all descended from the same mitochondrial DNA L3 lineages, which support a single migration out of Africa that gave rise to all non-African populations.[270]
On the basis of the early date of Badoshan Iranian Aurignacian, Oppenheimer suggests that this second dispersal may have occurred with a pluvial period about 50,000 years before the present, with modern human big-game hunting cultures spreading up the Zagros Mountains, carrying modern human genomes from Oman, throughout the Persian Gulf, northward into Armenia and Anatolia, with a variant travelling south into Israel and to Cyrenicia.[206]
Recent genetic evidence suggests that all modern non-African populations, including those of Eurasia and Oceania, are descended from a single wave that left Africa between 65,000 and 50,000 years ago.[271][272][273]
Evidence
[edit]The evidence on which scientific accounts of human evolution are based comes from many fields of natural science. The main source of knowledge about the evolutionary process has traditionally been the fossil record, but since the development of genetics beginning in the 1970s, DNA analysis has come to occupy a place of comparable importance. The studies of ontogeny, phylogeny and especially evolutionary developmental biology of both vertebrates and invertebrates offer considerable insight into the evolution of all life, including how humans evolved. The specific study of the origin and life of humans is anthropology, particularly paleoanthropology which focuses on the study of human prehistory.[274]
Evidence from genetics
[edit]
The closest living relatives of humans are bonobos and chimpanzees (both genus Pan) and gorillas (genus Gorilla).[275] With the sequencing of both the human and chimpanzee genome, as of 2012[update] estimates of the similarity between their DNA sequences range between 95% and 99%.[275][276][33] It is also noteworthy that mice share around 97.5% of their working DNA with humans.[277] By using the technique called the molecular clock which estimates the time required for the number of divergent mutations to accumulate between two lineages, the approximate date for the split between lineages can be calculated.
The gibbons (family Hylobatidae) and then the orangutans (genus Pongo) were the first groups to split from the line leading to the hominins, including humans—followed by gorillas (genus Gorilla), and, ultimately, by the chimpanzees (genus Pan). The splitting date between hominin and chimpanzee lineages is placed by some between 4 to 8 million years ago, that is, during the Late Miocene.[278][279][280][281] Speciation, however, appears to have been unusually drawn out. Initial divergence occurred sometime between 7 to 13 million years ago, but ongoing hybridization blurred the separation and delayed complete separation during several millions of years. Patterson (2006) dated the final divergence at 5 to 6 million years ago.[282]
Genetic evidence has also been employed to compare species within the genus Homo, investigating gene flow between early modern humans and Neanderthals, and to enhance the understanding of the early human migration patterns and splitting dates. By comparing the parts of the genome that are not under natural selection and which therefore accumulate mutations at a fairly steady rate, it is possible to reconstruct a genetic tree incorporating the entire human species since the last shared ancestor.
Each time a certain mutation (single-nucleotide polymorphism) appears in an individual and is passed on to his or her descendants, a haplogroup is formed including all of the descendants of the individual who will also carry that mutation. By comparing mitochondrial DNA which is inherited only from the mother, geneticists have concluded that the last female common ancestor whose genetic marker is found in all modern humans, the so-called mitochondrial Eve, must have lived around 200,000 years ago.
Human evolutionary genetics studies how human genomes differ among individuals, the evolutionary past that gave rise to them, and their current effects. Differences between genomes have anthropological, medical and forensic implications and applications. Genetic data can provide important insight into human evolution.
In May 2023, scientists reported a more complicated pathway of human evolution than previously understood. According to the studies, humans evolved from different places and times in Africa, instead of from a single location and period of time.[283][284]
Evidence from the fossil record
[edit]

There is little fossil evidence for the divergence of the gorilla, chimpanzee and hominin lineages.[285] The earliest fossils that have been proposed as members of the hominin lineage are Sahelanthropus tchadensis dating from 7 million years ago, Orrorin tugenensis dating from 5.7 million years ago, and Ardipithecus kadabba dating to 5.6 million years ago. Each of these have been argued to be a bipedal ancestor of later hominins but, in each case, the claims have been contested. It is also possible that one or more of these species are ancestors of another branch of African apes, or that they represent a shared ancestor between hominins and other apes.
The question then of the relationship between these early fossil species and the hominin lineage is still to be resolved. From these early species, the australopithecines arose around 4 million years ago and diverged into robust (also called Paranthropus) and gracile branches, one of which (possibly A. garhi) probably went on to become ancestors of the genus Homo. The australopithecine species that is best represented in the fossil record is Australopithecus afarensis with more than 100 fossil individuals represented, found from Northern Ethiopia (such as the famous "Lucy"), to Kenya, and South Africa. Fossils of robust australopithecines such as A. robustus (or alternatively Paranthropus robustus) and A./P. boisei are particularly abundant in South Africa at sites such as Kromdraai and Swartkrans, and around Lake Turkana in Kenya.
The earliest member of the genus Homo is Homo habilis which evolved around 2.8 million years ago.[39] H. habilis is the first species for which we have positive evidence of the use of stone tools. They developed the Oldowan lithic technology, named after the Olduvai Gorge in which the first specimens were found. Some scientists consider Homo rudolfensis, a larger bodied group of fossils with similar morphology to the original H. habilis fossils, to be a separate species, while others consider them to be part of H. habilis—simply representing intraspecies variation, or perhaps even sexual dimorphism. The brains of these early hominins were about the same size as that of a chimpanzee, and their main adaptation was bipedalism as an adaptation to terrestrial living.
During the next million years, a process of encephalization began and, by the arrival (about 1.9 million years ago) of H. erectus in the fossil record, cranial capacity had doubled. H. erectus were the first of the hominins to emigrate from Africa, and, from 1.8 to 1.3 million years ago, this species spread through Africa, Asia, and Europe. One population of H. erectus, also sometimes classified as separate species H. ergaster, remained in Africa and evolved into H. sapiens. It is believed that H. erectus and H. ergaster were the first to use fire and complex tools. In Eurasia, H. erectus evolved into species such as H. antecessor, H. heidelbergensis and H. neanderthalensis. The earliest fossils of anatomically modern humans are from the Middle Paleolithic, about 300–200,000 years ago such as the Herto and Omo remains of Ethiopia, Jebel Irhoud remains of Morocco, and Florisbad remains of South Africa; later fossils from the Skhul Cave in Israel and Southern Europe begin around 90,000 years ago (0.09 million years ago).
As modern humans spread out from Africa, they encountered other hominins such as H. neanderthalensis and the Denisovans, who may have evolved from populations of H. erectus that had left Africa around 2 million years ago. The nature of interaction between early humans and these sister species has been a long-standing source of controversy, the question being whether humans replaced these earlier species or whether they were in fact similar enough to interbreed, in which case these earlier populations may have contributed genetic material to modern humans.[286][287]
This migration out of Africa is estimated to have begun about 70–50,000 years BP and modern humans subsequently spread globally, replacing earlier hominins either through competition or hybridization. They inhabited Eurasia and Oceania by 40,000 years BP, and the Americas by at least 14,500 years BP.[288]
Inter-species breeding
[edit]
The hypothesis of interbreeding, also known as hybridization, admixture or hybrid-origin theory, has been discussed ever since the discovery of Neanderthal remains in the 19th century.[289] The linear view of human evolution began to be abandoned in the 1970s as different species of humans were discovered that made the linear concept increasingly unlikely. In the 21st century with the advent of molecular biology techniques and computerization, whole-genome sequencing of Neanderthal and human genome were performed, confirming recent admixture between different human species.[97] In 2010, evidence based on molecular biology was published, revealing unambiguous examples of interbreeding between archaic and modern humans during the Middle Paleolithic and early Upper Paleolithic. It has been demonstrated that interbreeding happened in several independent events that included Neanderthals and Denisovans, as well as several unidentified hominins.[290] Today, approximately 2% of DNA from all non-African populations (including Europeans, Asians, and Oceanians) is Neanderthal,[97] with traces of Denisovan heritage.[291] Also, 4–6% of modern Melanesian genetics are Denisovan.[291] Comparisons of the human genome to the genomes of Neandertals, Denisovans and apes can help identify features that set modern humans apart from other hominin species. In a 2016 comparative genomics study, a Harvard Medical School/UCLA research team made a world map on the distribution and made some predictions about where Denisovan and Neanderthal genes may be impacting modern human biology.[292][293]
For example, comparative studies in the mid-2010s found several traits related to neurological, immunological,[294] developmental, and metabolic phenotypes, that were developed by archaic humans to European and Asian environments and inherited to modern humans through admixture with local hominins.[295][296]
Although the narratives of human evolution are often contentious, several discoveries since 2010 show that human evolution should not be seen as a simple linear or branched progression, but a mix of related species.[49][5][6][7] In fact, genomic research has shown that hybridization between substantially diverged lineages is the rule, not the exception, in human evolution.[4] Furthermore, it is argued that hybridization was an essential creative force in the emergence of modern humans.[4]
Stone tools
[edit]Stone tools are first attested around 2.6 million years ago, when hominins in Eastern Africa used so-called core tools, choppers made out of round cores that had been split by simple strikes.[297] This marks the beginning of the Paleolithic, or Old Stone Age; its end is taken to be the end of the last Ice Age, around 10,000 years ago. The Paleolithic is subdivided into the Lower Paleolithic (Early Stone Age), ending around 350,000–300,000 years ago, the Middle Paleolithic (Middle Stone Age), until 50,000–30,000 years ago, and the Upper Paleolithic, (Late Stone Age), 50,000–10,000 years ago.
Archaeologists working in the Great Rift Valley in Kenya have discovered the oldest known stone tools in the world. Dated to around 3.3 million years ago, the implements are some 700,000 years older than stone tools from Ethiopia that previously held this distinction.[200][298][299][300]
The period from 700,000 to 300,000 years ago is also known as the Acheulean, when H. ergaster (or erectus) made large stone hand axes out of flint and quartzite, at first quite rough (Early Acheulian), later "retouched" by additional, more-subtle strikes at the sides of the flakes. After 350,000 BP the more refined so-called Levallois technique was developed, a series of consecutive strikes, by which scrapers, slicers ("racloirs"), needles, and flattened needles were made.[297] Finally, after about 50,000 BP, ever more refined and specialized flint tools were made by the Neanderthals and the immigrant Cro-Magnons (knives, blades, skimmers). Bone tools were also made by H. sapiens in Africa by 90,000–70,000 years ago[207][301] and are also known from early H. sapiens sites in Eurasia by about 50,000 years ago.
Species list
[edit]This list is in chronological order across the table by genus. Some species/subspecies names are well-established, and some are less established – especially in genus Homo. Please see articles for more information.
| Sahelanthropus | Homo (humans) |
|---|---|
| S. tchadensis | H. gautengensis |
| Orrorin | H. habilis |
| O. tugenensis | H. rudolfensis |
| Ardipithecus | H. floresiensis |
| A. kadabba | H. ergaster |
| A. ramidus | H. erectus |
| Australopithecus | • H. e. georgicus |
| A. anamensis | H. cepranensis |
| A. afarensis | H. antecessor |
| A. bahrelghazali | H. heidelbergensis |
| A. africanus | H. rhodesiensis |
| A. garhi | H. naledi |
| A. sediba | H. helmei |
| Kenyanthropus | H. neanderthalensis |
| K. platyops | H. sapiens |
| Paranthropus | • H. s. idaltu |
| P. aethiopicus | • H. s. sapiens (early) |
| P. boisei | • H. s. sapiens (modern) |
| P. robustus |
See also
[edit]- Adaptive evolution in the human genome
- Amity–enmity complex
- Archaeogenetics
- Dual inheritance theory
- Evolution of human intelligence
- Evolution of morality
- Evolutionary medicine
- Evolutionary neuroscience
- Evolutionary origin of religion
- Evolutionary psychology
- Human behavioral ecology
- Human origins
- Human vestigiality
- List of human evolution fossils
- Molecular paleontology
- Obstetrical dilemma
- Origin of language
- Origin of speech
- Prehistory of nakedness and clothing
- Sexual selection in humans
- Transgenerational trauma
- Timeline of human evolution
Notes
[edit]- ^ a b c The conventional estimate on the age of H. habilis is at roughly 2.1 to 2.3 million years.[41][172] Suggestions for pushing back the age to 2.8 Mya were made in 2015 based on the discovery of a jawbone.[173]
- ^ Not to be confused with Pongidae, an obsolete family which grouped together orangutans, gorillas and chimpanzees to separate them from humans
- ^ There is no general agreement on the line of special descent of H. sapiens from H. erectus. Some of the species depicted in the image may not actually represent a direct evolutionary ancestor to H. sapiens, and may not directly derive from one another, namely:
- H. heidelbergensis likely did not descend from H. antecessor.[144]
- H. heidelbergensis is likely not an ancestor to H. sapiens, nor is H. antecessor.[144]
- H. ergaster is often considered the next evolutionary ancestor to H. sapiens following H. erectus, however, there is considerable uncertainty as to the accuracy of classifying it as a separate species from H. erectus at all.[145]
- ^ H. erectus in the narrow sense (the Asian species) was extinct by 140,000 years ago, Homo erectus soloensis, found in Java, is considered the latest known survival of H. erectus. Formerly dated to as late as 50,000 to 40,000 years ago, a 2011 study pushed back the date of its extinction of H. e. soloensis to 143,000 years ago at the latest, more likely before 550,000 years ago.[175]
- ^ The Latin word which refers to adult males only is vir
- ^ See the Binomial nomenclature and Systema Naturae articles.
References
[edit]- ^ "Human evolution". Encyclopædia Britannica. June 8, 2024.
- ^ Hall, Brian K.; Hallgrímsson, Benedikt (2011). Strickberger's Evolution. Jones & Bartlett. p. 488. ISBN 978-1-4496-6390-2.
- ^ a b Mondal, M.; Bertranpetit, J.; Lao, O. (January 2019). "Approximate Bayesian computation with deep learning supports a third archaic introgression in Asia and Oceania". Nature Communications. 10 (1): 246. Bibcode:2019NatCo..10..246M. doi:10.1038/s41467-018-08089-7. PMC 6335398. PMID 30651539.
- ^ a b c Rogers Ackermann, Rebecca; Mackay, Alex; Arnold, Michael L. (October 2015). "The Hybrid Origin of "Modern" Humans". Evolutionary Biology. 43 (1): 1–11. doi:10.1007/s11692-015-9348-1. S2CID 14329491.
- ^ a b Antrosio, Jason (August 23, 2018). "Denisovans and Neandertals: Rethinking Species Boundaries". Living Anthropologically. Archived from the original on August 1, 2020. Retrieved August 25, 2018.
- ^ a b Hammer, Michael F. (May 2013). "Human Hybrids" (PDF). Scientific American. Archived from the original (PDF) on August 24, 2018.
- ^ a b Yong, Ed (July 2011). "Mosaic humans, the hybrid species". New Scientist. Vol. 211, no. 2823. pp. 34–38. Bibcode:2011NewSc.211...34Y. doi:10.1016/S0262-4079(11)61839-3. ISSN 0262-4079.
- ^ Heng, Henry H. Q. (May 2009). "The genome-centric concept: Resynthesis of evolutionary theory". BioEssays. 31 (5): 512–525. doi:10.1002/bies.200800182. ISSN 0265-9247. PMID 19334004. S2CID 1336952.
- ^ a b c Marlowe, Frank W. (April 13, 2005). "Hunter-gatherers and human evolution". Evolutionary Anthropology: Issues, News, and Reviews. 14 (2): 54–67. doi:10.1002/evan.20046. S2CID 53489209.
- ^ Tyson, Peter (July 1, 2008). "Meet Your Ancestors". Nova ScienceNow. WGBH Educational Foundation. Archived from the original on March 8, 2021. Retrieved April 18, 2015.
- ^ a b Gibbons, Ann (June 13, 2012). "Bonobos Join Chimps as Closest Human Relatives". TimeTree. Archived from the original on September 13, 2021. Retrieved May 19, 2018.
- ^ Maxwell 1984, p. 296.
- ^ Zhang, Rui; Wang, Yin-Qiu; Su, Bing (July 2008). "Molecular Evolution of a Primate-Specific microRNA Family". Molecular Biology and Evolution. 25 (7): 1493–1502. doi:10.1093/molbev/msn094. ISSN 0737-4038. PMID 18417486.
- ^ Willoughby, Pamela R. (2005). "Palaeoanthropology and the Evolutionary Place of Humans in Nature". International Journal of Comparative Psychology. 18 (1): 60–91. doi:10.46867/IJCP.2005.18.01.02. ISSN 0889-3667. Archived from the original on January 17, 2012. Retrieved April 27, 2015.
- ^ Martin 2001, pp. 12032–12038.
- ^ Tavaré, Simon; Marshall, Charles R.; Will, Oliver; et al. (April 18, 2002). "Using the fossil record to estimate the age of the last common ancestor of extant primates". Nature. 416 (6882): 726–729. Bibcode:2002Natur.416..726T. doi:10.1038/416726a. ISSN 0028-0836. PMID 11961552. S2CID 4368374.
- ^ Rose, Kenneth D. (1994). "The earliest primates". Evolutionary Anthropology: Issues, News, and Reviews. 3 (5): 159–173. doi:10.1002/evan.1360030505. ISSN 1060-1538. S2CID 85035753.
- ^ Fleagle, John; Gilbert, Chris (2011). Rowe, Noel; Myers, Marc (eds.). "Primate Evolution". All The World's Primates. Charlestown, Rhode Island: Primate Conservation. Archived from the original on May 12, 2015. Retrieved April 27, 2015.
- ^ Roach, John (March 3, 2008). "Oldest Primate Fossil in North America Discovered". National Geographic News. Washington, DC: National Geographic Society. Archived from the original on October 16, 2012. Retrieved April 27, 2015.
- ^ McMains, Vanessa (December 5, 2011). "Found in Wyoming: New fossils of oldest American primate". The Gazette. Baltimore: Johns Hopkins University. Archived from the original on January 16, 2019. Retrieved April 27, 2015.
- ^ Caldwell, Sara B. (May 19, 2009). "Missing link found, early primate fossil 47 million years old". Digital Journal. Toronto. Archived from the original on July 22, 2015. Retrieved April 27, 2015.
- ^ Watts, Alex (May 20, 2009). "Scientists Unveil Missing Link In Evolution". Sky News Online. London: BSkyB. Archived from the original on July 28, 2011. Retrieved April 27, 2015.
- ^ Wilford, J. N. (June 5, 2013). "Palm-size fossil resets primates' clock, scientists say". The New York Times. Archived from the original on January 1, 2022. Retrieved June 5, 2013.
- ^ a b Williams, B. A.; Kay, R. F.; Kirk, E. C. (2010). "New perspectives on anthropoid origins". Proceedings of the National Academy of Sciences. 107 (11): 4797–4804. Bibcode:2010PNAS..107.4797W. doi:10.1073/pnas.0908320107. PMC 2841917. PMID 20212104.
- ^ Tabuce, R.; Marivaux, L.; Lebrun, R.; Adaci, M.; Bensalah, M.; Fabre, P. -H.; Fara, E.; Gomes Rodrigues, H.; Hautier, L.; Jaeger, J. -J.; Lazzari, V.; Mebrouk, F.; Peigne, S.; Sudre, J.; Tafforeau, P.; Valentin, X.; Mahboubi, M. (2009). "Anthropoid versus strepsirhine status of the African Eocene primates Algeripithecus and Azibius: Craniodental evidence". Proceedings of the Royal Society B: Biological Sciences. 276 (1676): 4087–4094. doi:10.1098/rspb.2009.1339. PMC 2821352. PMID 19740889.
- Lay summary in: "African Origin Of Anthropoid Primates Called Into Question With New Fossil Discovery". Science Daily. September 15, 2009.
- ^ Kordos, László; Begun, David R. (January 2001). "Primates from Rudabánya: allocation of specimens to individuals, sex and age categories". Journal of Human Evolution. 40 (1). Amsterdam, the Netherlands: Elsevier: 17–39. Bibcode:2001JHumE..40...17K. doi:10.1006/jhev.2000.0437. PMID 11139358.
- ^ Cameron 2004, p. 76.
- ^ Wallace 2004, p. 240.
- ^ Zalmout, Iyad S.; Sanders, William J.; MacLatchy, Laura M.; et al. (July 15, 2010). "New Oligocene primate from Saudi Arabia and the divergence of apes and Old World monkeys". Nature. 466 (7304): 360–364. Bibcode:2010Natur.466..360Z. doi:10.1038/nature09094. ISSN 0028-0836. PMID 20631798. S2CID 205220837.
- ^ a b Srivastava 2009, p. 87.
- ^ "Scientists observe chimpanzees using human-like warfare tactic". Reuters. November 3, 2023.
- ^ a b Clark, G.; Henneberg, M. (June 2015). "The life history of Ardipithecus ramidus: A heterochronic model of sexual and social maturation". Anthropological Review. 78 (2): 109–132. doi:10.1515/anre-2015-0009. S2CID 54900467.
- ^ a b Sayers, Ken; Raghanti, Mary Ann; Lovejoy, C. Owen (October 2012). "Human Evolution and the Chimpanzee Referential Doctrine". Annual Review of Anthropology. 41: 119–138. doi:10.1146/annurev-anthro-092611-145815. ISSN 0084-6570.
- ^ Zimmer, Carl (May 27, 2015). "The Human Family Tree Bristles With New Branches". The New York Times. Archived from the original on January 1, 2022. Retrieved May 30, 2015.
- ^ Gardner., Elizabeth K.; Purdue University (April 1, 2015). "New instrument dates old skeleton before 'Lucy'; 'Little Foot' 3.67 million years old". Science Daily. Retrieved April 3, 2015.
- ^ Andrea Elyse Messer. "Earliest known fossil of the genus Homo dates to 2.8 to 2.75 million years ago". Penn State.
- ^ Smithsonian Institute. "The oldest fossil of our genus". humanorigins.si.edu.
- ^ Brandon Bryn. "Science: Oldest Fossil of Homo Genus Found in Ethiopia". AAAS.
- ^ a b c Ghosh, Pallab (March 4, 2015). "'First human' discovered in Ethiopia". BBC News. London. Archived from the original on April 18, 2015. Retrieved April 19, 2015.
- ^ Swisher, Curtis & Lewin 2001.
- ^ a b Stringer 1994, p. 242.
- ^ McHenry 2009, p. 265.
- ^ "Out of Africa Revisited". Science (This Week in Science). 308 (5724): 921. May 13, 2005. doi:10.1126/science.2005.308.5724.twis. ISSN 0036-8075. S2CID 220100436.
- ^ Stringer, Chris (June 12, 2003). "Human evolution: Out of Ethiopia". Nature. 423 (6941): 692–695. Bibcode:2003Natur.423..692S. doi:10.1038/423692a. ISSN 0028-0836. PMID 12802315. S2CID 26693109.
- ^ Johanson, Donald (May 2001). "Origins of Modern Humans: Multiregional or Out of Africa?". actionbioscience. Washington, DC: American Institute of Biological Sciences. Archived from the original on November 14, 2010. Retrieved November 23, 2009.
- ^ Mixon, Bobbie; Ehardt, Carolyn; Hammer, Michael (September 6, 2011). "Evolution's Past Is Modern Human's Present" (Press release). National Science Foundation. Press Release 11-181. Archived from the original on December 17, 2014. Retrieved April 20, 2015.
- ^ O'Neil, Dennis. "Early Modern Homo sapiens". Evolution of Modern Humans: A Survey of the Biological and Cultural Evolution of Archaic and Modern Homo sapiens (Tutorial). San Marcos, California: Palomar College. Archived from the original on April 30, 2015. Retrieved April 20, 2015.
- ^ "Fossil Reanalysis Pushes Back Origin of Homo sapiens". Scientific American. February 17, 2005. ISSN 0036-8733. Archived from the original on January 15, 2016. Retrieved April 20, 2015.
- ^ a b c d e f Reich, David; Green, Richard E.; Kircher, Martin; et al. (December 23, 2010). "Genetic history of an archaic hominin group from Denisova Cave in Siberia". Nature. 468 (7327): 1053–1060. Bibcode:2010Natur.468.1053R. doi:10.1038/nature09710. hdl:10230/25596. ISSN 0028-0836. PMC 4306417. PMID 21179161.
- ^ Noonan, James P. (May 2010). "Neanderthal genomics and the evolution of modern humans". Genome Research. 20 (5): 547–553. doi:10.1101/gr.076000.108. ISSN 1088-9051. PMC 2860157. PMID 20439435.
- ^ a b c Abi-Rached, Laurent; Jobin, Matthew J.; Kulkarni, Subhash; et al. (October 7, 2011). "The Shaping of Modern Human Immune Systems by Multiregional Admixture with Archaic Humans". Science. 334 (6052): 89–94. Bibcode:2011Sci...334...89A. doi:10.1126/science.1209202. ISSN 0036-8075. PMC 3677943. PMID 21868630.
- ^ Mellars, Paul (June 20, 2006). "Why did modern human populations disperse from Africa ca. 60,000 years ago? A new model". Proceedings of the National Academy of Sciences of the United States of America. 103 (25): 9381–9386. Bibcode:2006PNAS..103.9381M. doi:10.1073/pnas.0510792103. ISSN 0027-8424. PMC 1480416. PMID 16772383.
- ^ a b c McBrearty, Sally; Brooks, Alison S. (November 2000). "The revolution that wasn't: A new interpretation of the origin of modern human behavior". Journal of Human Evolution. 39 (5): 453–563. Bibcode:2000JHumE..39..453M. doi:10.1006/jhev.2000.0435. ISSN 0047-2484. PMID 11102266. S2CID 42968840.
- ^ based on Schlebusch, C. M.; Malmström, H.; Günther, T.; Sjödin, P.; Coutinho, A.; Edlund, H.; Munters, A. R.; Vicente, M.; Steyn, M.; Soodyall, H.; Lombard, M.; Jakobsson, M. (2017). "Southern African ancient genomes estimate modern human divergence to 350,000 to 260,000 years ago". Science. 358 (6363): 652–655. Bibcode:2017Sci...358..652S. doi:10.1126/science.aao6266. PMID 28971970. S2CID 206663925., Fig. 3 Archived January 14, 2018, at the Wayback Machine (H. sapiens divergence times) and Stringer, C. (2012). "What makes a modern human". Nature. 485 (7396): 33–35. Bibcode:2012Natur.485...33S. doi:10.1038/485033a. PMID 22552077. S2CID 4420496. (archaic admixture).
- ^ Strait, David S.; Grine, Frederick E.; Moniz, Marc A. (January 1997). "A reappraisal of early hominid phylogeny". Journal of Human Evolution. 32 (1): 17–82. Bibcode:1997JHumE..32...17S. doi:10.1006/jhev.1996.0097. ISSN 0047-2484. PMID 9034954. S2CID 37754799.
- ^ a b Bryson 2004, pp. 522–543.
- ^ a b Zimmer, Carl (August 13, 2015). "For Evolving Brains, a 'Paleo' Diet Full of Carbs". The New York Times. Archived from the original on January 1, 2022. Retrieved August 14, 2015.
- ^ a b Leonard, William R.; Snodgrass, J. Josh; Robertson, Marcia L. (August 2007). "Effects of brain evolution on human nutrition and metabolism". Annual Review of Nutrition. 27: 311–327. doi:10.1146/annurev.nutr.27.061406.093659. ISSN 0199-9885. PMID 17439362. S2CID 18869516.
- ^ Walker 2007, pp. 3–10.
- ^ Ungar & Teaford 2002.
- ^ Bogin 1997, pp. 96–142.
- ^ Barnicot, Nigel A. (April–June 2005). "Human nutrition: Evolutionary perspectives". Integrative Physiological & Behavioral Science. 40 (2): 114–117. doi:10.1007/BF02734246. ISSN 1932-4502. PMID 17393680. S2CID 39549910.
- ^ "The new batch – 150,000 years ago". The evolution of man. London: BBC Science & Nature. Archived from the original on January 18, 2006. Retrieved April 28, 2015.
- ^ Whitehouse, David (June 9, 2003). "When humans faced extinction". BBC News. London: BBC. Archived from the original on September 4, 2010. Retrieved January 5, 2007.
- ^ "Modern humans flourished through ancient supervolcano eruption 74,000 years ago: Modern humans flourished through ancient supervolcano eruption". ScienceDaily. Archived from the original on January 24, 2019. Retrieved January 24, 2019.
- ^ Zimmer, Carl (August 31, 2023). "Humanity's Ancestors Nearly Died Out, Genetic Study Suggests - The population crashed following climate change about 930,000 years ago, scientists concluded. Other experts aren't convinced by the analysis". the New York Times. Archived from the original on August 31, 2023. Retrieved September 2, 2023.
- ^ Hu, Wangjie; et al. (August 31, 2023). "Genomic inference of a severe human bottleneck during the Early to Middle Pleistocene transition". Science. 381 (6661): 979–984. Bibcode:2023Sci...381..979H. doi:10.1126/science.abq7487. PMID 37651513. S2CID 261396309. Archived from the original on September 1, 2023. Retrieved September 2, 2023.
- ^ Wood, Bernard; Collard, Mark (1999). "The changing face of Genus Homo". Evolutionary Anthropology: Issues, News, and Reviews. 8 (6): 195–207. doi:10.1002/(SICI)1520-6505(1999)8:6<195::AID-EVAN1>3.0.CO;2-2. ISSN 1060-1538. S2CID 86768101.
- ^ Viegas, Jennifer (May 21, 2010). "Toothy Tree-Swinger May Be Earliest Human". Discovery News. Silver Spring, Maryland: Discovery Communications. Archived from the original on May 9, 2015. Retrieved April 28, 2015.
- ^ Wood, Bernard A. (January 1999). "Homo rudolfensis Alexeev, 1986 – fact or phantom?". Journal of Human Evolution. 36 (1): 115–118. Bibcode:1999JHumE..36..115W. doi:10.1006/jhev.1998.0246. ISSN 0047-2484. PMID 9924136.
- ^ Gabounia, Léo; de Lumley, Marie-Antoinette; Vekua, Abesalom; et al. (September 2002). "Découverte d'un nouvel hominidé à Dmanissi (Transcaucasie, Géorgie)" [Discovery of a new hominid at Dmanisi (Transcaucasia, Georgia)]. Comptes Rendus Palevol (in French). 1 (4): 243–253. Bibcode:2002CRPal...1..243G. doi:10.1016/S1631-0683(02)00032-5. ISSN 1631-0683.
- ^ Lordkipanidze, David; Vekua, Abesalom; Ferring, Reid; et al. (November 2006). "A fourth hominin skull from Dmanisi, Georgia". The Anatomical Record Part A: Discoveries in Molecular, Cellular, and Evolutionary Biology. 288A (11): 1146–1157. doi:10.1002/ar.a.20379. ISSN 1552-4884. PMID 17031841.
- ^ Turner, William (April 1895). "On M. Dubois' Description of Remains recently found in Java, named by him Pithecanthropus erectus. With Remarks on so-called Transitional Forms between Apes and Man". Journal of Anatomy and Physiology. 29 (Pt 3): 424–445. PMC 1328414. PMID 17232143.
- ^ Weidenreich, Franz (July 1940). "Some Problems Dealing with Ancient Man". American Anthropologist. 42 (3): 375–383. doi:10.1525/aa.1940.42.3.02a00010. ISSN 0002-7294.
- ^ Grine, Frederick E.; Fleagle, John G. (2009), "The First Humans: A Summary Perspective on the Origin and Early Evolution of the Genus Homo", The First Humans – Origin and Early Evolution of the Genus Homo, "Vertebrate Paleobiology and Paleoanthropology" series, Springer Netherlands, pp. 197–207, doi:10.1007/978-1-4020-9980-9_17, ISBN 978-1-4020-9979-3
- ^ Rizal, Y.; Westaway, K. E.; Zaim, Y.; van den Bergh; Bettis III, E. A.; Morwood, M. J.; Huffman, O. F.; Grün, R.; Joannes-Boyau, R.; Bailey, R. M.; Sidarto; Westaway, M. C.; Kurniawan, I.; Moore, M. W.; Storey, M.; Aziz, F.; Suminto; Zhao, J.; Aswan; Sipola, M. E.; Larick, R.; Zonneveld, J.-P.; Scott, R.; Putt, S.; Ciochon, R. L. (2020). "Last appearance of Homo erectus at Ngandong, Java, 117,000–108,000 years ago". Nature. 577 (7790): 381–385. doi:10.1038/s41586-019-1863-2. hdl:10072/397335. ISSN 0028-0836. PMID 31853068. S2CID 209410644.
- ^ Louys, J.; Roberts, P. (2020). "Environmental drivers of megafauna and hominin extinction in Southeast Asia". Nature. 586 (7829): 402–406. Bibcode:2020Natur.586..402L. doi:10.1038/s41586-020-2810-y. hdl:10072/402368. PMID 33029012. S2CID 222217295.
- ^ Spoor, Fred; Wood, Bernard A.; Zonneveld, Frans (June 23, 1994). "Implications of early hominid labyrinthine morphology for evolution of human bipedal locomotion". Nature. 369 (6482): 645–648. Bibcode:1994Natur.369..645S. doi:10.1038/369645a0. ISSN 0028-0836. PMID 8208290. S2CID 4344784.
- ^ Ings, Simon (October 4, 2009). "Catching Fire: How Cooking Made Us Human by Richard Wrangham: Review". Archived from the original on January 11, 2022. Retrieved February 23, 2016.
- ^ Wrangham, Richard (2011). Catching Fire: How cooking made us human.[page needed]
- ^ Bermúdez de Castro, José María; Arsuaga, Juan Luis; Carbonell, Eudald; et al. (May 30, 1997). "A Hominid from the Lower Pleistocene of Atapuerca, Spain: Possible Ancestor to Neandertals and Modern Humans". Science. 276 (5317): 1392–1395. doi:10.1126/science.276.5317.1392. ISSN 0036-8075. PMID 9162001.
- ^ Carbonell, Eudald; Bermúdez de Castro, José María; Parés, Josep M.; et al. (March 27, 2008). "The first hominin of Europe". Nature. 452 (7186): 465–469. Bibcode:2008Natur.452..465C. doi:10.1038/nature06815. hdl:2027.42/62855. ISSN 0028-0836. PMID 18368116. S2CID 4401629.
- ^ Manzi, Giorgio; Mallegni, Francesco; Ascenzi, Antonio (August 14, 2001). "A cranium for the earliest Europeans: Phylogenetic position of the hominid from Ceprano, Italy". Proceedings of the National Academy of Sciences of the United States of America. 98 (17): 10011–10016. Bibcode:2001PNAS...9810011M. doi:10.1073/pnas.151259998. ISSN 0027-8424. PMC 55569. PMID 11504953.
- ^ Czarnetzki, Alfred; Jakob, Tina; Pusch, Carsten M. (April 2003). "Palaeopathological and variant conditions of the Homo heidelbergensis type specimen (Mauer, Germany)". Journal of Human Evolution. 44 (4): 479–495. Bibcode:2003JHumE..44..479C. doi:10.1016/S0047-2484(03)00029-0. ISSN 0047-2484. PMID 12727464.
- ^ Semaw, Sileshi; Toth, Nicholas; Schick, Kathy; et al. (March 27, 2006). "Scientists discover hominid cranium in Ethiopia" (Press release). Bloomington: Indiana University. Archived from the original on November 15, 2006. Retrieved November 26, 2006.
- ^ Harvati, Katerina (January 2003). "The Neanderthal taxonomic position: Models of intra- and inter-specific craniofacial variation". Journal of Human Evolution. 44 (1): 107–132. Bibcode:2003JHumE..44..107H. doi:10.1016/S0047-2484(02)00208-7. ISSN 0047-2484. PMID 12604307.
- ^ Herrera, K. J.; Somarelli, J. A.; Lowery, R. K.; Herrera, R. J. (2009). "To what extent did Neanderthals and modern humans interact?". Biological Reviews. 84 (2): 245–257. doi:10.1111/j.1469-185X.2008.00071.x. PMID 19391204. S2CID 25787484.
- ^ Finlayson, C.; Giles Pacheco, F.; Rodríguez-Vidal, J.; et al. (2006). "Late survival of Neanderthals at the southernmost extreme of Europe". Nature. 443 (7113): 850–853. Bibcode:2006Natur.443..850F. doi:10.1038/nature05195. hdl:10261/18685. PMID 16971951. S2CID 4411186.Full list of authors
- Clive Finlayson
- Francisco Giles Pacheco
- Joaquín Rodríguez-Vidal
- Darren A. Fa
- José María Gutierrez López
- Antonio Santiago Pérez
- Geraldine Finlayson
- Ethel Allue
- Javier Baena Preysler
- Isabel Cáceres
- José S. Carrión
- Yolanda Fernández-Jalvo
- Christopher P. Gleed-Owen
- Francisco J. Jimenez-Espejo
- Pilar López Martínez
- José Antonio López Sáez
- José Antonio Riquelme Cantal
- Antonio Sánchez Marco
- Francisco Giles Guzman
- Kimberly Brown
- Noemí Fuentes
- Claire A. Valarino
- Antonio Villalpando
- Christopher B. Stringer
- Francisca Martinez Ruiz
- Tatsuhiko Sakamoto
- ^ a b Pearce, Eiluned; Stringer, Chris; Dunbar, R. I. M. (2013). "New insights into differences in brain organization between Neanderthals and anatomically modern humans". Proceedings of the Royal Society of London B: Biological Sciences. 280 (1758) 20130168. doi:10.1098/rspb.2013.0168. PMC 3619466. PMID 23486442.
- ^ Bocquet-Appel, Jean-Pierre; Degioanni, Anna (December 2013). "Neanderthal Demographic Estimates". The University of Chicago Press Journals. 54 (S8): S202 – S213. doi:10.1086/673725.
- ^ Krings, Matthias; Stone, Anne; Schmitz, Ralf W.; et al. (July 11, 1997). "Neandertal DNA sequences and the origin of modern humans". Cell. 90 (1): 19–30. doi:10.1016/S0092-8674(00)80310-4. hdl:11858/00-001M-0000-0025-0960-8. ISSN 0092-8674. PMID 9230299. S2CID 13581775.
- ^ Green, Richard E.; Malaspinas, Anna-Sapfo; Krause, Johannes; et al. (August 8, 2008). "A Complete Neandertal Mitochondrial Genome Sequence Determined by High-Throughput Sequencing". Cell. 134 (3): 416–426. doi:10.1016/j.cell.2008.06.021. ISSN 0092-8674. PMC 2602844. PMID 18692465.
- ^ Serre, David; Langaney, André; Chech, Mario; et al. (March 2004). "No Evidence of Neandertal mtDNA Contribution to Early Modern Humans". PLOS Biology. 2 (3) e57. doi:10.1371/journal.pbio.0020057. ISSN 1545-7885. PMC 368159. PMID 15024415.
- ^ a b Viegas, Jennifer (May 6, 2010). "Neanderthals, Humans Interbred, DNA Proves". Discovery News. Silver Spring, Maryland: Discovery Communications. Archived from the original on May 8, 2015. Retrieved April 30, 2015.
- ^ Calloway, Ewan (May 13, 2015). "Early European may have had Neanderthal great-great-grandparent". Nature. doi:10.1038/nature.2015.17534. S2CID 181973496. Archived from the original on January 15, 2019. Retrieved January 23, 2019.
- ^ Sample, Ian (June 22, 2015). "My Neanderthal sex secret: Modern European's great-great grandparent link". The Guardian. Archived from the original on September 23, 2016. Retrieved July 27, 2018.
- ^ a b c d Green, R. E.; Krause, J.; Briggs, A. W.; Maricic, T.; et al. (May 7, 2010). "A Draft Sequence of the Neandertal Genome". Science. 328 (5979): 710–722. Bibcode:2010Sci...328..710G. doi:10.1126/science.1188021. ISSN 0036-8075. PMC 5100745. PMID 20448178.Full list of authors
- Richard E. Green
- Johannes Krause
- Adrian W. Briggs
- Tomislav Maricic
- Udo Stenzel
- Martin Kircher
- Nick Patterson
- Heng Li
- Weiwei Zhai
- Markus Hsi-Yang Fritz
- Nancy F. Hansen
- Eric Y. Durand
- Anna-Sapfo Malaspinas
- Jeffrey D. Jensen
- Tomas Marques-Bonet
- Can Alkan
- Kay Prüfer
- Matthias Meyer
- Hernán A. Burbano
- Jeffrey M. Good
- Rigo Schultz
- Ayinuer Aximu-Petri
- Anne Butthof
- Barbara Höber
- Barbara Höffner
- Madlen Siegemund
- Antje Weihmann
- Chad Nusbaum
- Eric S. Lander
- Carsten Russ
- Nathaniel Novod
- Jason Affourtit
- Michael Egholm
- Christine Verna
- Pavao Rudan
- Dejana Brajkovic
- Željko Kucan
- Ivan Gušic
- Vladimir B. Doronichev
- Liubov V. Golovanova
- Carles Lalueza-Fox
- Marco de la Rasilla
- Javier Fortea
- Antonio Rosas
- Ralf W. Schmitz
- Philip L. F. Johnson
- Evan E. Eichler
- Daniel Falush
- Ewan Birney
- James C. Mullikin
- Montgomery Slatkin
- Rasmus Nielsen
- Janet Kelso
- Michael Lachmann
- David Reich
- Svante Pääbo
- ^ a b Prüfer, K.; de Filippo, C.; Grote, S.; Mafessoni, F.; Korlević, P.; Hajdinjak, M.; et al. (2017). "A high-coverage Neandertal genome from Vindija Cave in Croatia". Science. 358 (6363): 655–658. Bibcode:2017Sci...358..655P. doi:10.1126/science.aao1887. PMC 6185897. PMID 28982794.
- ^ Gutiérrez, Gabriel; Sánchez, Diego; Marín, Antonio (August 2002). "A Reanalysis of the Ancient Mitochondrial DNA Sequences Recovered from Neandertal Bones". Molecular Biology and Evolution. 19 (8): 1359–1366. doi:10.1093/oxfordjournals.molbev.a004197. ISSN 0737-4038. PMID 12140248.
- ^ Hebsgaard, M. B.; Wiuf, C.; Gilbert, M. T.; Glenner, H.; Willerslev, E. (January 2007). "Evaluating Neanderthal Genetics and Phylogeny" (PDF). Journal of Molecular Evolution. 64 (1): 50–60. Bibcode:2007JMolE..64...50H. CiteSeerX 10.1.1.174.8969. doi:10.1007/s00239-006-0017-y. ISSN 0022-2844. PMID 17146600. S2CID 2746487. Archived from the original (PDF) on April 1, 2011. Retrieved October 24, 2017.
- ^ Mellars, Paul; French, Jennifer C. (July 29, 2011). "Tenfold Population Increase in Western Europe at the Neandertal–to–Modern Human Transition Paul". Science. 333 (6042): 623–627. Bibcode:2011Sci...333..623M. doi:10.1126/science.1206930. ISSN 0036-8075. PMID 21798948. S2CID 28256970.
- ^ Brown, Terence A. (April 8, 2010). "Human evolution: Stranger from Siberia". Nature. 464 (7290): 838–839. Bibcode:2010Natur.464..838B. doi:10.1038/464838a. ISSN 0028-0836. PMID 20376137. S2CID 4320406.
- ^ a b Krause, Johannes; Fu, Qiaomei; Good, Jeffrey M.; et al. (April 8, 2010). "The complete mitochondrial DNA genome of an unknown hominin from southern Siberia". Nature. 464 (7290): 894–897. Bibcode:2010Natur.464..894K. doi:10.1038/nature08976. ISSN 0028-0836. PMC 10152974. PMID 20336068. S2CID 4415601.
- ^ Katsnelson, Alla (March 24, 2010). "New hominin found via mtDNA". The Nutshell (Blog). Philadelphia: The Scientist. ISSN 0890-3670. Archived from the original on July 2, 2015. Retrieved May 1, 2015.
- ^ "Kaufman, Danial (2002), "Comparisons and the Case for Interaction among Neanderthals and Early Modern Humans in the Levant" (Oxford Journal of Anthropology)
- ^ Bokma, Folmer; van den Brink, Valentijn; Stadler, Tanja (September 2012). "Unexpectedly many extinct hominins". Evolution. 66 (9): 2969–2974. doi:10.1111/j.1558-5646.2012.01660.x. ISSN 0014-3820. PMID 22946817. S2CID 13145359.
- ^ a b c Reich, David; Patterson, Nick; Kircher, Martin; et al. (October 7, 2011). "Denisova Admixture and the First Modern Human Dispersals into Southeast Asia and Oceania". American Journal of Human Genetics. 89 (4): 516–528. doi:10.1016/j.ajhg.2011.09.005. ISSN 0002-9297. PMC 3188841. PMID 21944045.
- ^ Martinón-Torres, María; Dennell, Robin; Bermúdez de Castro, José María (February 2011). "The Denisova hominin need not be an out of Africa story". Journal of Human Evolution. 60 (2): 251–255. Bibcode:2011JHumE..60..251M. doi:10.1016/j.jhevol.2010.10.005. ISSN 0047-2484. PMID 21129766.
- ^ Simonti, C. N.; Vernot, B.; Bastarache, L.; Bottinger, E.; Carrell, D. S.; Chisholm, R. L.; Crosslin, D. R.; Hebbring, S. J.; Jarvik, G. P.; Kullo, I. J.; Li, R.; Pathak, J.; Ritchie, M. D.; Roden, D. M.; Verma, S. S.; Tromp, G.; Prato, J. D.; Bush, W. S.; Akey, J. M.; Denny, J. C.; Capra, J. A. (2016). "The phenotypic legacy of admixture between modern humans and Neandertals". Science. 351 (6274): 737–741. Bibcode:2016Sci...351..737S. doi:10.1126/science.aad2149. PMC 4849557. PMID 26912863.
- ^ Kuhlwilm, M.; Gronau, I.; Hubisz, M. J.; de Filippo, C.; Prado-Martinez, J.; Kircher, M.; Fu, Q.; Burbano, H. A.; Lalueza-Fox, C.; de la Rasilla, M.; Rosas, A.; Rudan, P.; Brajkovic, D.; Kucan, Ž.; Gušic, I.; Marques-Bonet, T.; Andrés, A. M.; Viola, B.; Pääbo, S.; Meyer, M.; Siepel, A.; Castellano, S. (2016). "Ancient gene flow from early modern humans into Eastern Neanderthals". Nature. 530 (7591): 429–433. Bibcode:2016Natur.530..429K. doi:10.1038/nature16544. PMC 4933530. PMID 26886800.
- ^ Dean, MC, Stringer, CB et al, (1986) "Age at death of the Neanderthal child from Devil's Tower, Gibraltar and the implications for studies of general growth and development in Neanderthals" (American Journal of Physical Anthropology, Vol 70 Issue 3, July 1986)
- ^ a b Brown, P.; Sutikna, T.; Morwood, M. J.; Soejono, R. P.; Jatmiko, A.; Wayhu, S. E.; Awe Due, R. (October 28, 2004). "A new small-bodied hominin from the Late Pleistocene of Flores, Indonesia" (PDF). Nature. 431 (7012): 1055–1061. Bibcode:2004Natur.431.1055B. doi:10.1038/nature02999. ISSN 0028-0836. PMID 15514638. S2CID 26441. Archived (PDF) from the original on January 3, 2023. Retrieved January 3, 2023.
- ^ a b c Argue, Debbie; Donlon, Denise; Groves, Colin; Wright, Richard (October 2006). "Homo floresiensis: Microcephalic, pygmoid, Australopithecus, or Homo?". Journal of Human Evolution. 51 (4): 360–374. Bibcode:2006JHumE..51..360A. doi:10.1016/j.jhevol.2006.04.013. ISSN 0047-2484. PMID 16919706.
- ^ a b Martin, Robert D.; Maclarnon, Ann M.; Phillips, James L.; Dobyns, William B. (November 2006). "Flores hominid: New species or microcephalic dwarf?". The Anatomical Record Part A: Discoveries in Molecular, Cellular, and Evolutionary Biology. 288A (11): 1123–1145. doi:10.1002/ar.a.20389. ISSN 1552-4884. PMID 17031806.
- ^ Callaway, E. (June 8, 2016). "'Hobbit' relatives found after ten-year hunt". Nature. 534 (7606): 164–165. Bibcode:2016Natur.534Q.164C. doi:10.1038/534164a. PMID 27279191.
- ^ Brumm, A.; van den Bergh, G. D.; Storey, M.; Kurniawan, I.; et al. (June 8, 2016). "Age and context of the oldest known hominin fossils from Flores" (PDF). Nature. 534 (7606): 249–253. Bibcode:2016Natur.534..249B. doi:10.1038/nature17663. PMID 27279222. S2CID 28608179. Archived (PDF) from the original on March 6, 2020. Retrieved November 11, 2021.Full list of authors
- Adam Brumm
- Gerrit D. van den Bergh
- Michael Storey
- Iwan Kurniawan
- Brent V. Alloway
- Ruly Setiawan
- Erick Setiyabudi
- Rainer Grün
- Mark W. Moore
- Dida Yurnaldi
- Mika R. Puspaningrum
- Unggul P. Wibowo
- Halmi Insani
- Indra Sutisna
- John A. Westgate
- Nick J. G. Pearce
- Mathieu Duval
- Hanneke J. M. Meijer
- Fachroel Aziz
- Thomas Sutikna
- Sander van der Kaars
- Stephanie Flude
- Michael J. Morwood
- ^ van den Bergh, G. D.; Kaifu, Y.; Kurniawan, I.; Kono, R. T.; Brumm, A.; Setiyabudi, E.; Aziz, F.; Morwood, M. J. (June 8, 2016). "Homo floresiensis-like fossils from the early Middle Pleistocene of Flores". Nature. 534 (7606): 245–248. Bibcode:2016Natur.534..245V. doi:10.1038/nature17999. PMID 27279221. S2CID 205249218.
- ^ Détroit, F.; Mijares, A. S.; Corny, J.; Daver, G.; Zanolli, C.; Dizon, E.; Robles, E.; Grün, R. & Piper, P. J. (2019). "A new species of Homo from the Late Pleistocene of the Philippines" (PDF). Nature. 568 (7751): 181–186. Bibcode:2019Natur.568..181D. doi:10.1038/s41586-019-1067-9. hdl:10072/386785. PMID 30971845. S2CID 106411053. Archived (PDF) from the original on October 13, 2022. Retrieved May 11, 2021.
- ^ Schlebusch, Carina M.; Malmström, Helena; Günther, Torsten; Sjödin, Per; Coutinho, Alexandra; Edlund, Hanna; Munters, Arielle R.; Vicente, Mário; Steyn, Maryna; Soodyall, Himla; Lombard, Marlize; Jakobsson, Mattias (2017). "Southern African ancient genomes estimate modern human divergence to 350,000 to 260,000 years ago". Science. 358 (6363): 652–655. Bibcode:2017Sci...358..652S. doi:10.1126/science.aao6266. PMID 28971970. S2CID 206663925.
- ^ Sample, Ian (June 7, 2017). "Oldest Homo sapiens bones ever found shake foundations of the human story". The Guardian. Archived from the original on October 31, 2019. Retrieved June 7, 2017.
- ^ Zimmer, Carl (September 10, 2019). "Scientists Find the Skull of Humanity's Ancestor — on a Computer: By comparing fossils and CT scans, researchers say they have reconstructed the skull of the last common forebear of modern humans". The New York Times. Archived from the original on January 1, 2022. Retrieved September 10, 2019.
- ^ Mounier, Aurélien; Lahr, Marta (2019). "Deciphering African late middle Pleistocene hominin diversity and the origin of our species". Nature Communications. 10 (1): 3406. Bibcode:2019NatCo..10.3406M. doi:10.1038/s41467-019-11213-w. PMC 6736881. PMID 31506422.
- ^ Ambrose, Stanley H. (June 1998). "Late Pleistocene human population bottlenecks, volcanic winter, and differentiation of modern humans". Journal of Human Evolution. 34 (6): 623–651. Bibcode:1998JHumE..34..623A. doi:10.1006/jhev.1998.0219. ISSN 0047-2484. PMID 9650103. S2CID 33122717.
- ^ Huff, Chad D.; Xing, Jinchuan; Rogers, Alan R.; Witherspoon, David; Jorde, Lynn B. (January 19, 2010). "Mobile Elements Reveal Small Population Size in the Ancient Ancestors of Homo sapiens". Proceedings of the National Academy of Sciences of the United States of America. 107 (5): 2147–2152. Bibcode:2010PNAS..107.2147H. doi:10.1073/pnas.0909000107. PMC 2836654. PMID 20133859.
- ^ Clark, Jamie L. (September 2011). "The evolution of human culture during the later Pleistocene: Using fauna to test models on the emergence and nature of "modern" human behavior". Journal of Anthropological Archaeology. 30 (3): 273–291. doi:10.1016/j.jaa.2011.04.002. Archived from the original on May 25, 2021. Retrieved October 27, 2021.
- ^ Boyd & Silk 2003, p. [page needed].
- ^ Brues & Snow 1965, pp. 1–39.
- ^ Brunet, M.; Guy, F.; Pilbeam, D.; Mackaye, H.; Likius, A.; Ahounta, D.; et al. (July 11, 2002). "A new hominid from the Upper Miocene of Chad, Central Africa" (PDF). Nature. 418 (6894): 145–151. Bibcode:2002Natur.418..145B. doi:10.1038/nature00879. ISSN 0028-0836. PMID 12110880. S2CID 1316969. Archived (PDF) from the original on February 25, 2023. Retrieved February 20, 2023.Full list of authors
- Michel Brunet
- Franck Guy
- David Pilbeam
- Hassane Taisso Mackaye
- Andossa Likius
- Djimdoumalbaye Ahounta
- Alain Beauvilain
- Cécile Blondel
- Hervé Bocherens
- Jean-Renaud Boisserie
- Louis De Bonis
- Yves Coppens
- Jean Dejax
- Christiane Denys
- Philippe Duringer
- Véra Eisenmann
- Gongdibé Fanone
- Pierre Fronty
- Denis Geraads
- Thomas Lehmann
- Fabrice Lihoreau
- Antoine Louchart
- Adoum Mahamat
- Gildas Merceron
- Guy Mouchelin
- Olga Otero
- Pablo Pelaez Campomanes
- Marcia Ponce De Leon
- Jean-Claude Rage
- Michel Sapanet
- Mathieu Schuster
- Jean Sudre
- Pascal Tassy
- Xavier Valentin
- Patrick Vignaud
- Laurent Viriot
- Antoine Zazzo
- Christoph Zollikofer
- ^ White, T. D.; Asfaw, B.; Beyene, Y.; Haile-Selassie, Y.; Lovejoy, C. O.; Suwa, G.; Woldegabriel, G. (2009). "Ardipithecus ramidus and the Paleobiology of Early Hominids". Science. 326 (5949): 75–86. Bibcode:2009Sci...326...75W. doi:10.1126/science.1175802. PMID 19810190. S2CID 20189444.
- ^ a b Kwang Hyun, Ko (2015). "Origins of Bipedalism". Brazilian Archives of Biology and Technology. 58 (6): 929–934. arXiv:1508.02739. Bibcode:2015arXiv150802739K. doi:10.1590/S1516-89132015060399. S2CID 761213.
- ^ a b DeSalle & Tattersall 2008, p. 146.
- ^ a b Curry 2008, pp. 106–109.
- ^ "Study Identifies Energy Efficiency as Reason for Evolution of Upright Walking". ScienceDaily. July 17, 2007. Archived from the original on May 4, 2015. Retrieved April 9, 2015.
"Study identifies energy efficiency as reason for evolution of upright walking". UANews. Tucson: University of Arizona. July 16, 2007. Archived from the original on July 3, 2022. Retrieved April 23, 2015. - ^ Sockol, Michael D.; Raichlen, David A.; Pontzer, Herman (July 24, 2007). "Chimpanzee locomotor energetics and the origin of human bipedalism". Proceedings of the National Academy of Sciences of the United States of America. 104 (30): 12265–12269. Bibcode:2007PNAS..10412265S. doi:10.1073/pnas.0703267104. ISSN 0027-8424. PMC 1941460. PMID 17636134.
- ^ David-Barrett, T.; Dunbar, R. I. M. (2016). "Bipedality and Hair-loss Revisited: The Impact of Altitude and Activity Scheduling". Journal of Human Evolution. 94: 72–82. doi:10.1016/j.jhevol.2016.02.006. PMC 4874949. PMID 27178459.
- ^ Aiello & Dean 1990.
- ^ Kondo 1985.
- ^ Strickberger 2000, pp. 475–476.
- ^ Trevathan 2011, p. 20.
- ^ Zuk, Marlene (2014). Paleofantasy: What Evolution Really Tells Us About Sex, Diet, and How We Live. W.W. Norton & Company. ISBN 978-0-393-34792-0. OCLC 846889455.[page needed]
- ^ Hrdy, Sarah Blaffer (2011). Mothers and Others: The Evolutionary Origins of Mutual Understanding. Harvard University Press. ISBN 978-0-674-06032-6. OCLC 940575388.[page needed]
- ^ Wayman, Erin (August 19, 2013). "Killer whales, grandmas and what men want: Evolutionary biologists consider menopause". Science News. ISSN 0036-8423. Archived from the original on November 6, 2014. Retrieved April 24, 2015.
- ^ Blell, Mwenza (September 29, 2017). "Grandmother Hypothesis, Grandmother Effect, and Residence Patterns". The International Encyclopedia of Anthropology. pp. 1–5. doi:10.1002/9781118924396.wbiea2162. ISBN 978-1-118-92439-6.
- ^ a b Welker, F.; Ramos-Madrigal, J.; Gutenbrunner, P.; et al. (April 1, 2020). "The dental proteome of Homo antecessor". Nature. 580 (7802): 235–238. Bibcode:2020Natur.580..235W. doi:10.1038/s41586-020-2153-8. ISSN 1476-4687. PMC 7582224. PMID 32269345. S2CID 214736611. Archived from the original on November 10, 2021. Retrieved November 10, 2021.Full list of authors
- Frido Welker
- Jazmín Ramos-Madrigal
- Petra Gutenbrunner
- Meaghan Mackie
- Shivani Tiwary
- Rosa Rakownikow Jersie-Christensen
- Cristina Chiva
- Marc R. Dickinson
- Martin Kuhlwilm
- Marc de Manuel
- Pere Gelabert
- María Martinón-Torres
- Ann Margvelashvili
- Juan Luis Arsuaga
- Eudald Carbonell
- Tomas Marques-Bonet
- Kirsty Penkman
- Eduard Sabidó
- Jürgen Cox
- Jesper V. Olsen
- David Lordkipanidze
- Fernando Racimo
- Carles Lalueza-Fox
- José María Bermúdez de Castro
- Eske Willerslev
- Enrico Cappellini
- ^ Dennell, Robin; Roebroeks, Wil (2005). "An Asian perspective on early human dispersal from Africa". Nature. 438 (7071): 1099–1104. Bibcode:2005Natur.438.1099D. doi:10.1038/nature04259. PMID 16371999. S2CID 4405913. Archived from the original on October 31, 2021. Retrieved November 10, 2021.
- ^ a b Schoenemann, P. Thomas (October 2006). "Evolution of the Size and Functional Areas of the Human Brain". Annual Review of Anthropology. 35: 379–406. doi:10.1146/annurev.anthro.35.081705.123210. ISSN 0084-6570. S2CID 7611321.
- ^ Brown, Graham; Fairfax, Stephanie; Sarao, Nidhi. "Tree of Life Web Project: Human Evolution". www.tolweb.org. Archived from the original on June 6, 2020. Retrieved August 24, 2015.
- ^ Kaplan, Hillard; Hill, Kim; Lancaster, Jane; Hurtado, Magdelena (August 16, 2000). "A Theory of Human Life History Evolution: Diet, Intelligence, and Longevity" (PDF). Evolutionary Anthropology. 9 (4): 156–185. doi:10.1002/1520-6505(2000)9:4<156::AID-EVAN5>3.0.CO;2-7. S2CID 2363289. Archived (PDF) from the original on February 15, 2020. Retrieved September 20, 2019.
- ^ Park, M. S.; Nguyen, A. D.; Aryan, H. E.; U, H. S.; Levy, M. L.; Semendeferi, K. (March 2007). "Evolution of the human brain: Changing brain size and the fossil record". Neurosurgery. 60 (3): 555–562. doi:10.1227/01.NEU.0000249284.54137.32. ISSN 0148-396X. PMID 17327801. S2CID 19610624.
- ^ Bruner, Emiliano (December 2007). "Cranial shape and size variation in human evolution: Structural and functional perspectives". Child's Nervous System. 23 (12): 1357–1365. CiteSeerX 10.1.1.391.288. doi:10.1007/s00381-007-0434-2. ISSN 0256-7040. PMID 17680251. S2CID 16163137.
- ^ Potts, Richard (October 2012). "Evolution and Environmental Change in Early Human Prehistory". Annual Review of Anthropology. 41: 151–167. doi:10.1146/annurev-anthro-092611-145754. ISSN 0084-6570.
- ^ Neubauer, Simon; Hublin, Jean-Jacques; Gunz, Philipp (January 5, 2018). "The evolution of modern human brain shape - Science". Science Advances. 4 (1) eaao5961. Bibcode:2018SciA....4.5961N. doi:10.1126/sciadv.aao5961. PMC 5783678. PMID 29376123.
- ^ Mann, Neil (September 2007). "Meat in the human diet: An anthropological perspective". Nutrition & Dietetics. 64 (Supplement 4): S102 – S107. doi:10.1111/j.1747-0080.2007.00194.x. ISSN 1747-0080.
- ^ McBroom, Patricia (June 14, 1999). "Meat-eating was essential for human evolution, says UC Berkeley anthropologist specializing in diet" (Press release). Berkeley: University of California Press. Archived from the original on April 20, 2015. Retrieved April 25, 2015.
- ^ Barr, W. Andrew; Pobiner, Briana; Rowan, John; Du, Andrew; Faith, J. Tyler (February 1, 2022). "No sustained increase in zooarchaeological evidence for carnivory after the appearance of Homo erectus". Proceedings of the National Academy of Sciences of the United States of America. 119 (5) e2115540119. Bibcode:2022PNAS..11915540B. doi:10.1073/pnas.2115540119. ISSN 0027-8424. PMC 8812535. PMID 35074877.
- ^ Organ, Chris; Nunn, Charles L.; Machanda, Zarin; Wrangham, Richard W. (August 30, 2011). "Phylogenetic rate shifts in feeding time during the evolution of Homo". Proceedings of the National Academy of Sciences of the United States of America. 108 (35): 14555–14559. Bibcode:2011PNAS..10814555O. doi:10.1073/pnas.1107806108. ISSN 0027-8424. PMC 3167533. PMID 21873223.
- ^ a b David-Barrett, T.; Dunbar, R. I. M. (2013). "Processing Power Limits Social Group Size: Computational Evidence for the Cognitive Costs of Sociality". Proceedings of the Royal Society of London B: Biological Sciences. 280 (1765) 20131151. doi:10.1098/rspb.2013.1151. PMC 3712454. PMID 23804623.
- ^ Bown & Rose 1987.
- ^ Barton, Robert A.; Venditti, Chris (October 20, 2014). "Rapid Evolution of the Cerebellum in Humans and Other Great Apes". Current Biology. 24 (20): 2440–2444. Bibcode:2014CBio...24.2440B. doi:10.1016/j.cub.2014.08.056. ISSN 0960-9822. PMID 25283776. S2CID 5041106.
- ^ Starowicz-Filip, Anna; Milczarek, Olga; Kwiatkowski, Stanisław; Bętkowska-Korpała, Barbara; Prochwicz, Katarzyna (2013). "Cerebellar cognitive affective syndrome CCAS – a case report". Archives of Psychiatry and Psychotherapy. 15 (3): 57–64. doi:10.12740/APP/18666.
- ^ Yu, Feng; Jiang, Qing-jun; Sun, Xi-yan; Zhang, Rong-wei (August 22, 2014). "A new case of complete primary cerebellar agenesis: Clinical and imaging findings in a living patient". Brain. 138 (Pt 6): e353. doi:10.1093/brain/awu239. ISSN 1460-2156. PMC 4614135. PMID 25149410.
- ^ Weaver, Anne H. (March 8, 2005). "Reciprocal evolution of the cerebellum and neocortex in fossil humans". Proceedings of the National Academy of Sciences of the United States of America. 102 (10): 3576–3580. Bibcode:2005PNAS..102.3576W. doi:10.1073/pnas.0500692102. ISSN 0027-8424. PMC 553338. PMID 15731345.
- ^ Klein, Stefan (2014). Survival of the Nicest. The Experiment. ISBN 978-1-61519-090-4.
- ^ Sallet, J.; Mars, R. B.; Noonan, M. P.; Andersson, J. L.; O'Reilly, J. K.; Jbabdi, S.; Croxson, P. L.; Jenkinson, M.; Miller, K. L. (November 4, 2011). "Social Network Size Affects Neural Circuits in Macaques". Science. 334 (6056): 697–700. Bibcode:2011Sci...334..697S. doi:10.1126/science.1210027. ISSN 0036-8075. PMID 22053054. S2CID 206536017.
- ^ Dunbar, R. I. M. (1992). "Neocortex size as a constraint on group size in primates". Journal of Human Evolution. 22 (6): 469–493. Bibcode:1992JHumE..22..469D. doi:10.1016/0047-2484(92)90081-j.
- ^ Shultz, Susanne; Dunbar, Robin (December 14, 2010). "Encephalization is not a universal macroevolutionary phenomenon in mammals but is associated with sociality". Proceedings of the National Academy of Sciences of the United States of America. 107 (50): 21582–21586. Bibcode:2010PNAS..10721582S. doi:10.1073/pnas.1005246107. ISSN 0027-8424. PMC 3003036. PMID 21098277.
- ^ Richard, Potts (1998). "Environmental Hypotheses of Hominin Evolution". American Journal of Physical Anthropology. 107 (S27): 93–136. doi:10.1002/(sici)1096-8644(1998)107:27+<93::aid-ajpa5>3.0.co;2-x. PMID 9881524.
- ^ Richerson, Peter J. (2006). Not by Genes Alone: How Culture Transformed Human Evolution. University of Chicago Press. ISBN 0-226-71212-5. OCLC 642503808.
- ^ Mesoudi, Alex; Whiten, Andrew; Laland, Kevin N. (August 2006). "Towards a unified science of cultural evolution". Behavioral and Brain Sciences. 29 (4): 329–347. doi:10.1017/s0140525x06009083. ISSN 0140-525X. PMID 17094820.
- ^ Berger, L. R.; Hawks, J.; Dirks, P. H.; Elliott, M.; Roberts, E. M. (May 9, 2017). "Homo naledi and Pleistocene hominin evolution in subequatorial Africa". eLife. 6 e24234. doi:10.7554/elife.24234. PMC 5423770. PMID 28483041.
- ^ Strait D, Grine F, Fleagle J (2015). Analyzing Hominin Hominin Phylogeny: Cladistic Approach. Springer. pp. 1989–2014 (cladogram p. 2006). ISBN 978-3-642-39978-7..
- ^ Schrenk, F.; Kullmer, O.; Bromage, T. (2007). "Chapter 9: The Earliest Putative Homo Fossils". In Henke, W.; Tattersall, I. (eds.). Handbook of Paleoanthropology. pp. 1611–1631. doi:10.1007/978-3-540-33761-4_52.
- ^ Spoor, F.; Gunz, P.; Neubauer, S.; Stelzer, S.; Scott, N.; Kwekason, A.; Dean, M. C. (March 2015). "Reconstructed Homo habilis type OH 7 suggests deep-rooted species diversity in early Homo". Nature. 519 (7541): 83–6. Bibcode:2015Natur.519...83S. doi:10.1038/nature14224. PMID 25739632. S2CID 4470282.
- ^ Schuster, A. M. (1997). "Earliest Remains of Genus Homo". Archaeology. 50 (1). Archived from the original on March 17, 2015. Retrieved March 5, 2015. The line to the earliest members of Homo was derived from Australopithecus, a genus which had separated from the Chimpanzee–human last common ancestor by late Miocene or early Pliocene times.
- ^ Indriati, E.; Swisher, C. C.; Lepre, C.; Quinn, R. L.; Suriyanto, R. A.; et al. (2011). "The age of the 20 meter Solo River terrace, Java, Indonesia and the survival of Homo erectus in Asia". PLoS One. 6 (6) e21562. Bibcode:2011PLoSO...621562I. doi:10.1371/journal.pone.0021562. PMC 3126814. PMID 21738710.Full list of authors
- Etty Indriati
- Carl C. Swisher III
- Christopher Lepre
- Rhonda L. Quinn
- Rusyad A. Suriyanto
- Agus T. Hascaryo
- Rainer Grün
- Craig S. Feibel
- Briana L. Pobiner
- Maxime Aubert
- Wendy Lees
- Susan C. Antón
- ^ Zeitoun, V. (September 2003). "High occurrence of a basicranial feature in Homo erectus: Anatomical description of the preglenoid tubercle". The Anatomical Record Part B: The New Anatomist. 274 (1): 148–56. doi:10.1002/ar.b.10028. PMID 12964205.
- ^ Dembo, M.; Matzke, N. J.; Mooers, A. Ø.; Collard, M. (August 2015). "Bayesian analysis of a morphological supermatrix sheds light on controversial fossil hominin relationships". Proceedings. Biological Sciences. 282 (1812) 20150943. doi:10.1098/rspb.2015.0943. PMC 4528516. PMID 26202999.
- ^ Dembo, M.; Radovčić, D.; et al. (August 2016). "The evolutionary relationships and age of Homo naledi: An assessment using dated Bayesian phylogenetic methods". Journal of Human Evolution. 97: 17–26. Bibcode:2016JHumE..97...17D. doi:10.1016/j.jhevol.2016.04.008. hdl:2164/8796. PMID 27457542.Full list of authors
- Mana Dembo
- Davorka Radovčić
- Heather M. Garvinc
- Myra F. Laird
- Lauren Schroeder
- Jill E. Scott
- Juliet Brophy
- Rebecca R. Ackermann
- Chares M. Musiba
- Darryl J. de Ruiter
- Arne Ø. Mooers
- Mark Collard
- ^ Mounier, A.; Caparros, M. (2015). "The phylogenetic status of Homo heidelbergensis – a cladistic study of Middle Pleistocene hominins". BMSAP (in French). 27 (3–4): 110–134. doi:10.1007/s13219-015-0127-4. ISSN 0037-8984. S2CID 17449909.
- ^ Harrison, N. (May 1, 2019). The Origins of Europeans and Their Pre-Historic Innovations from 6 Million to 10,000 BCE: From 6 Million to 10,000 BCE. Algora Publishing. ISBN 978-1-62894-379-5.
- ^ Rogers, A. R.; Harris, N. S.; Achenbach, A. A. (February 2020). "Neanderthal-Denisovan ancestors interbred with a distantly related hominin". Science Advances. 6 (8) eaay5483. Bibcode:2020SciA....6.5483R. doi:10.1126/sciadv.aay5483. PMC 7032934. PMID 32128408.
- ^ Tanner, Nancy Makepeace (1981). On Becoming Human. CUP Archive. p. 202. ISBN 978-0-521-28028-0.
- ^ Reno, Philip L.; Lovejoy, C. Owen (April 28, 2015). "From Lucy to Kadanuumuu: Balanced analyses of Australopithecus afarensis assemblages confirm only moderate skeletal dimorphism". PeerJ. 3 e925. doi:10.7717/peerj.925. ISSN 2167-8359. PMC 4419524. PMID 25945314.
- ^ Lovejoy, C. Owen (October 2, 2009). "Reexamining Human Origins in Light of Ardipithecus ramidus" (PDF). Science. 326 (5949): 74–74e8. Bibcode:2009Sci...326...74L. doi:10.1126/science.1175834. ISSN 0036-8075. PMID 19810200. S2CID 42790876. Archived (PDF) from the original on February 24, 2021. Retrieved December 6, 2019.
- ^ Young, Richard W. (January 2003). "Evolution of the human hand: The role of throwing and clubbing". Journal of Anatomy. 202 (1): 165–174. doi:10.1046/j.1469-7580.2003.00144.x. ISSN 0021-8782. PMC 1571064. PMID 12587931.
- ^ a b Ittyerah, Miriam (2013). Hand Preference and Hand Ability: Evidence from studies in Haptic Cognition. John Benjamins Publishing. pp. 37–38. ISBN 978-90-272-7164-8.
- ^ Wilson, Frank R. "The Hand How Its Use Shapes the Brain, Language, and Human Culture". The New York Times. Archived from the original on August 28, 2017. Retrieved July 2, 2017.
- ^ Kuzawa, Christopher W.; Chugani, Harry T.; Grossman, Lawrence I.; Lipovich, Leonard; Muzik, Otto; Hof, Patrick R.; Wildman, Derek E.; Sherwood, Chet C.; Leonard, William R.; Lange, Nicholas (August 25, 2014). "Metabolic costs and evolutionary implications of human brain development". Proceedings of the National Academy of Sciences of the United States of America. 111 (36): 13010–13015. Bibcode:2014PNAS..11113010K. doi:10.1073/pnas.1323099111. PMC 4246958. PMID 25157149.
- ^ Pontzer, Herman; Brown, Mary H.; Raichlen, David A.; Dunsworth, Holly; Hare, Brian; Walker, Kara; Luke, Amy; Dugas, Lara R.; Durazo-Arvizu, Ramon; Schoeller, Dale; Plange-Rhule, Jacob; Bovet, Pascal; Forrester, Terrence E.; Lambert, Estelle V.; Thompson, Melissa Emery; Shumaker, Robert W.; Ross, Stephen P. (May 4, 2016). "Metabolic acceleration and the evolution of human brain size and life history". Nature. 533 (7603): 390–392. Bibcode:2016Natur.533..390P. doi:10.1038/nature17654. PMC 4942851. PMID 27144364.
- ^ Rantala, Markus J. (August 20, 2007). "Evolution of nakedness in Homo sapiens". Journal of Zoology. 273 (1): 1–7. doi:10.1111/j.1469-7998.2007.00295.x. Archived from the original on November 5, 2022. Retrieved November 5, 2022.
- ^ Differential modularity of the mammalian Engrailed 1 enhancer network directs sweat gland development
- ^ Humans Evolved to Be the Water-Saving Ape | Duke Today
- ^ Why Humans Sleep Less Than Their Primate Relatives
- ^ Potter, Lisa Marie (September 8, 2015). "Shouldering the Burden of Evolution". UCSF News Center. University of California San Francisco. Archived from the original on May 8, 2023. Retrieved May 8, 2023.
- ^ Weber, G.W.; Lukeneder, A.; Harzhauser, M. (February 28, 2022). "The microstructure and origin of the Venus of Willendorf". Scientific Reports. 12 (2926). Nature: 2926. doi:10.1038/s41598-022-06799-z. PMC 8885675. PMID 35228605.
- ^ a b Ko, Kwang Hyun (2016). "Origins of human intelligence: The chain of tool-making and brain evolution" (PDF). Anthropological Notebooks. 22 (1): 5–22. Archived (PDF) from the original on August 17, 2016. Retrieved August 8, 2016.
- ^ Jabr, Ferris (July 18, 2012). "Does Thinking Really Hard Burn More Calories?". Scientific American. ISSN 0036-8733. Archived from the original on May 3, 2015. Retrieved May 3, 2015.
- ^ Gibbons, Ann (May 29, 1998). "Solving the Brain's Energy Crisis". Science. 280 (5368): 1345–1347. doi:10.1126/science.280.5368.1345. ISSN 0036-8075. PMID 9634409. S2CID 28464775.
- ^ Robinson 2008, p. 398.
- ^ a b Harmand, Sonia; Lewis, Jason E.; Feibel, Craig S.; Lepre, Christopher J.; Prat, Sandrine; Lenoble, Arnaud; Boës, Xavier; Quinn, Rhonda L.; Brenet, Michel (May 20, 2015). "3.3-million-year-old stone tools from Lomekwi 3, West Turkana, Kenya". Nature. 521 (7552): 310–315. Bibcode:2015Natur.521..310H. doi:10.1038/nature14464. ISSN 0028-0836. PMID 25993961. S2CID 1207285.
- ^ Semaw, Sileshi; Rogers, Michael J.; Quade, Jay; Renne, Paul R.; Butler, Robert F.; Dominguez-Rodrigo, Manuel; Stout, Dietrich; Hart, William S.; Pickering, Travis; Simpson, Scott W. (August 1, 2003). "2.6-Million-year-old stone tools and associated bones from OGS-6 and OGS-7, Gona, Afar, Ethiopia". Journal of Human Evolution. 45 (2): 169–177. Bibcode:2003JHumE..45..169S. doi:10.1016/S0047-2484(03)00093-9. ISSN 0047-2484. PMID 14529651.
- ^ a b c Freeman & Herron 2007, pp. 786–788.
- ^ Ward, Carol V.; Tocheri, Matthew W.; Plavcan, J. Michael; et al. (January 7, 2014). "Early Pleistocene third metacarpal from Kenya and the evolution of modern human-like hand morphology". Proceedings of the National Academy of Sciences of the United States of America. 111 (1): 121–124. Bibcode:2014PNAS..111..121W. doi:10.1073/pnas.1316014110. ISSN 0027-8424. PMC 3890866. PMID 24344276.
- ^ Diamond 1999, p. 39.
- ^ Bar-Yosef, Ofer (October 2002). "The Upper Paleolithic Revolution". Annual Review of Anthropology. 31: 363–393. doi:10.1146/annurev.anthro.31.040402.085416. ISSN 0084-6570.
- ^ a b Oppenheimer, Stephen (2012). Out of Eden: The Peopling of the World (New ed.). Robinson.
- ^ a b Henshilwood, C. S.; d'Errico, F.; Yates, R.; et al. (2002). "Emergence of Modern Human Behavior: Middle Stone Age Engravings from South Africa". Science. 295 (5558): 1278–1280. Bibcode:2002Sci...295.1278H. doi:10.1126/science.1067575. PMID 11786608. S2CID 31169551.
- ^ Henshilwood, Christopher; Marean, Curtis (2003). "The Origin of Modern Human Behavior: Critique of the Models and Their Test Implications". Current Anthropology. 44 (5): 627–651. doi:10.1086/377665. PMID 14971366. S2CID 11081605.
- ^ Backwell, L.; d'Errico, F.; Wadley, L. (2008). "Middle Stone Age bone tools from the Howiesons Poort layers, Sibudu Cave, South Africa". Journal of Archaeological Science. 35 (6): 1566–1580. Bibcode:2008JArSc..35.1566B. doi:10.1016/j.jas.2007.11.006.
- ^ Nowell, April (October 2010). "Defining Behavioral Modernity in the Context of Neandertal and Anatomically Modern Human Populations". Annual Review of Anthropology. 39: 437–452. doi:10.1146/annurev.anthro.012809.105113. ISSN 0084-6570.
- ^ d'Errico, Francesco; Stringer, Chris B. (April 12, 2011). "Evolution, revolution or saltation scenario for the emergence of modern cultures?". Philosophical Transactions of the Royal Society B. 366 (1567): 1060–1069. doi:10.1098/rstb.2010.0340. ISSN 0962-8436. PMC 3049097. PMID 21357228.
- ^ Chatterjee, Rhitu (March 15, 2018). "Scientists Are Amazed By Stone Age Tools They Dug Up In Kenya". NPR. Archived from the original on March 15, 2018. Retrieved March 15, 2018.
- ^ Yong, Ed (March 15, 2018). "A Cultural Leap at the Dawn of Humanity – New finds from Kenya suggest that humans used long-distance trade networks, sophisticated tools, and symbolic pigments right from the dawn of our species". The Atlantic. Archived from the original on November 17, 2020. Retrieved March 15, 2018.
- ^ Brooks, A. S.; Yellen, J. E.; Potts, R.; Behrensmeyer, A. K.; Deino, A. L.; Leslie, D. E.; Ambrose, S. H.; Ferguson, J. R.; d'Errico, F.; Zipkin, A. M.; Whittaker, S.; Post, J.; Veatch, E. G.; Foecke, K.; Clark, J. B. (2018). "Long-distance stone transport and pigment use in the earliest Middle Stone Age". Science. 360 (6384): 90–94. Bibcode:2018Sci...360...90B. doi:10.1126/science.aao2646. PMID 29545508. S2CID 14051717.
- ^ Rasmussen, Morten; Guo, Xiaosen; Wang, Yong; Lohmueller, Kirk E.; Rasmussen, Simon; Albrechtsen, Anders; et al. (2011). "An Aboriginal Australian Genome Reveals Separate Human Dispersals into Asia". Science. 334 (6052): 94–98. Bibcode:2011Sci...334...94R. doi:10.1126/science.1211177. PMC 3991479. PMID 21940856.
- ^ Ambrose, Stanley H. (March 2, 2001). "Paleolithic Technology and Human Evolution". Science. 291 (5509): 1748–1753. Bibcode:2001Sci...291.1748A. doi:10.1126/science.1059487. ISSN 0036-8075. PMID 11249821. S2CID 6170692.
- ^ Mellars, P. (2010). "Neanderthal symbolism and ornament manufacture: The bursting of a bubble?". Proc Natl Acad Sci USA. 107 (47): 20147–20148. Bibcode:2010PNAS..10720147M. doi:10.1073/pnas.1014588107. PMC 2996706. PMID 21078972.
- ^ Cochran & Harpending 2009.
- ^ Bender, Andrea (August 8, 2019). "The Role of Culture and Evolution for Human Cognition". Topics in Cognitive Science. 12 (4): 1403–1420. doi:10.1111/tops.12449. hdl:1956/22031. ISSN 1756-8757. PMID 31392845.
- ^ Fu, Q.; Hajdinjak, M.; Moldovan, O. T.; Constantin, S.; Mallick, S.; Skoglund, Pontus; Patterson, N.; Rohland, N.; Lazaridis, I.; Nickel, B.; Viola, B.; Prüfer, Kay; Meyer, M.; Kelso, J.; Reich, D.; Pääbo, S. (2015). "An early modern human from Romania with a recent Neanderthal ancestor". Nature. 524 (7564): 216–219. Bibcode:2015Natur.524..216F. doi:10.1038/nature14558. PMC 4537386. PMID 26098372.
- ^ Medical Research Council [UK] (November 21, 2009). "Brain Disease 'Resistance Gene' evolves in Papua New Guinea community; could offer insights into CJD". ScienceDaily. Archived from the original on June 8, 2019. Retrieved November 22, 2009.
- ^ Mead, S.; Whitfield, J.; Poulter, M.; et al. (2009). "A Novel Protective Prion Protein Variant that Colocalizes with Kuru Exposure" (PDF). The New England Journal of Medicine. 361 (21): 2056–2065. doi:10.1056/NEJMoa0809716. PMID 19923577. Archived (PDF) from the original on July 20, 2018. Retrieved August 20, 2019.Full list of authors
- Simon Mead
- Jerome Whitfield
- Mark Poulter
- Paresh Shah
- James Uphill
- Tracy Campbell
- Huda Al-Dujaily
- Holger Hummerich
- Jon Beck
- Charles A. Mein
- Claudio Verzilli
- John Whittaker
- Michael P. Alpers
- John Collinge
- ^ Corbett, Stephen; Morin-Papunen, Laure (July 2013). "The Polycystic Ovary Syndrome and recent human evolution". Molecular and Cellular Endocrinology. 373 (1–2): 39–50. doi:10.1016/j.mce.2013.01.001. PMID 23352610. S2CID 13078403. Archived from the original on May 25, 2021. Retrieved October 8, 2020.
- ^ Diamond 1999.
- ^ Courtiol, Alexandre; Tropf, Felix C.; Mills, Melinda C. (July 12, 2016). "When genes and environment disagree: Making sense of trends in recent human evolution". Proceedings of the National Academy of Sciences of the United States of America. 113 (28): 7693–7695. Bibcode:2016PNAS..113.7693C. doi:10.1073/pnas.1608532113. ISSN 0027-8424. PMC 4948334. PMID 27402758.
- ^ Markov, Alexander V.; Markov, Mikhail A. (June 2020). "Runaway brain-culture coevolution as a reason for larger brains: Exploring the "cultural drive" hypothesis by computer modeling". Ecology and Evolution. 10 (12): 6059–6077. Bibcode:2020EcoEv..10.6059M. doi:10.1002/ece3.6350. ISSN 2045-7758. PMC 7319167. PMID 32607213.
- ^ Stout, Dietrich (April 12, 2011). "Stone toolmaking and the evolution of human culture and cognition". Philosophical Transactions of the Royal Society B: Biological Sciences. 366 (1567): 1050–1059. doi:10.1098/rstb.2010.0369. ISSN 0962-8436. PMC 3049103. PMID 21357227.
- ^ a b Byars, S. G.; Ewbank, D.; Govindaraju, D. R.; Stearns, S. C. (2009). "Natural selection in a contemporary human population". Proceedings of the National Academy of Sciences of the United States of America. 107 (suppl_1): 1787–1792. Bibcode:2010PNAS..107.1787B. doi:10.1073/pnas.0906199106. PMC 2868295. PMID 19858476.
- ^ Editors of the American Heritage Dictionaries, ed. (2006). More Word Histories and Mysteries: From Aardvark to Zombie. Boston: Houghton Mifflin. ISBN 978-0-618-71681-4. LCCN 2006020835. OCLC 70199867.[page needed]
- ^ "Nested Hierarchies, the Order of Nature: Carolus Linnaeus". Understanding Evolution: The History of Evolutionary Thought. The University of California at Berkeley. Archived from the original on August 2, 2019. Retrieved August 2, 2019.
- ^ Darwin 1859, pp. 367–404.
- ^ Darwin 1981.
- ^ Montgomery 1988, pp. 95–96.
- ^ Shipman, Pat; Storm, Paul (2002). "Missing links: Eugène Dubois and the origins of paleoanthropology". Evolutionary Anthropology: Issues, News, and Reviews. 11 (3): 108–116. doi:10.1002/evan.10021. ISSN 1520-6505. S2CID 84889438.
- ^ Dart, Raymond (February 7, 1925). "Australopithecus africanus: The Man-Ape of South Africa". Nature. 115 (2884): 195–199. Bibcode:1925Natur.115..195D. doi:10.1038/115195a0. ISSN 0028-0836. S2CID 4142569.
- ^ Johanson & Edey 1981, pp. 20–22, 184–185.
- ^ Cartmill, Matt; Smith, Fred H.; Brown, Kaye B. (2009). The Human Lineage. Wiley-Blackwell. p. 151. ISBN 978-0-471-21491-5.
- ^ Johanson & Edey 1981, p. 22.
- ^ Shreeve, Jamie (July 2010). "The Evolutionary Road". National Geographic. ISSN 0027-9358. Archived from the original on January 26, 2016. Retrieved May 28, 2015.
- ^ Berger, L. R.; Hawks, J.; de Ruiter, D. J.; Churchill, S. E.; et al. (September 10, 2015). "Homo naledi, a new species of the genus Homo from the Dinaledi Chamber, South Africa". eLife. 4 e09560. doi:10.7554/eLife.09560. PMC 4559886. PMID 26354291.Full list of authors
- Lee R Berger
- John Hawks
- Darryl J de Ruiter
- Steven E Churchill
- Peter Schmid
- Lucas K Delezene
- Tracy L Kivell
- Heather M Garvin
- Scott A Williams
- Jeremy M DeSilva
- Matthew M Skinner
- Charles M Musiba
- Noel Cameron
- Trenton W Holliday
- William Harcourt-Smith
- Rebecca R Ackermann
- Markus Bastir
- Barry Bogin
- Debra Bolter
- Juliet Brophy
- Zachary D Cofran
- Kimberly A Congdon
- Andrew S Deane
- Mana Dembo
- Michelle Drapeau
- Marina C Elliott
- Elen M Feuerriegel
- Daniel Garcia-Martinez
- David J Green
- Alia Gurtov
- Joel D Irish
- Ashley Kruger
- Myra F Laird
- Damiano Marchi
- Marc R Meyer
- Shahed Nalla
- Enquye W Negash
- Caley M Orr
- Davorka Radovcic
- Lauren Schroeder
- Jill E Scott
- Zachary Throckmorton
- Matthew W Tocheri
- Caroline VanSickle
- Christopher S Walker
- Pianpian Wei
- Bernhard Zipfel
- ^ a b Shreeve, Jamie (September 10, 2015). "This Face Changes the Human Story. But How?". National Geographic News. Archived from the original on April 14, 2018. Retrieved September 10, 2015.
- ^ Dirks et al. (2017): between 335 and 236 ka. The lower limit of 236 ka is due to optically stimulated luminescence dating of sediments with U-Th and palaeomagnetic analyses of flowstones; the upper limit of 335 ka is due to U-series and electron spin resonance (US-ESR) dating of two H. naledi teeth, to 253+82
−70 ka, for an estimated age of the fossils of 253+82
−17 ka. - ^ Dirks PH, Roberts EM, Hilbert-Wolf H, Kramers JD, et al. (May 9, 2017). "The age of Homo naledi and associated sediments in the Rising Star Cave, South Africa". eLife. 6 e24231. doi:10.7554/eLife.24231. PMC 5423772. PMID 28483040.Full list of authors
- Paul H.G.M. Dirks
- Eric M. Roberts
- Hannah Hilbert-Wolf
- Jan D. Kramers
- John Hawks
- Anthony Dosseto
- Mathieu Duval
- Marina Elliott
- Mary Evans
- Rainer Grün
- John Hellstrom
- Andy I.R. Herries
- Renaud Joannes-Boyau
- Tebogo V. Makhubela
- Christa J. Placzek
- Jessie Robbins
- Carl Spandler
- Jelle Wiersma
- Jon Woodhead
- Lee R. Berger
- ^ a b Sarich, V. M.; Wilson, A. C. (1967). "Immunological time scale for hominid evolution". Science. 158 (3805): 1200–1204. Bibcode:1967Sci...158.1200S. doi:10.1126/science.158.3805.1200. PMID 4964406. S2CID 7349579.
- ^ M'charek 2005, p. 96.
- ^ Trent 2005, p. 6.
- ^ Wade, Nicholas (May 18, 2006). "Two Splits Between Human and Chimp Lines Suggested". The New York Times. Archived from the original on May 18, 2022. Retrieved April 20, 2015.
- ^ Behar, D. M.; Villems, R.; Soodyall, H.; et al. (May 9, 2008). "The Dawn of Human Matrilineal Diversity". American Journal of Human Genetics. 82 (5): 1130–1140. doi:10.1016/j.ajhg.2008.04.002. ISSN 0002-9297. PMC 2427203. PMID 18439549.Full list of authors
- Doron M. Behar
- Richard Villems
- Himla Soodyall
- Jason Blue-Smith
- Luisa Pereira
- Ene Metspalu
- Rosaria Scozzari
- Heeran Makkan
- Shay Tzur
- David Comas
- Jaume Bertranpetit
- Lluis Quintana-Murci
- Chris Tyler-Smith
- R. Spencer Wells
- Saharon Rosset
- The Genographic Consortium
- ^ Gonder, M. K.; Mortensen, H. M.; Reed, F. A.; de Sousa, A.; Tishkoff, S. A. (March 2007). "Whole-mtDNA Genome Sequence Analysis of Ancient African Lineages". Molecular Biology and Evolution. 24 (3): 757–768. doi:10.1093/molbev/msl209. ISSN 0737-4038. PMID 17194802.
- ^ Tishkoff, S. A.; Reed, F. A.; Friedlaender, F. R.; et al. (2009). "The Genetic Structure and History of Africans and African Americans". Science. 324 (5930) (published May 22, 2009): 1035–1044. Bibcode:2009Sci...324.1035T. doi:10.1126/science.1172257. PMC 2947357. PMID 19407144.Full list of authors
- Sarah A. Tishkoff
- Floyd A. Reed
- Françoise R. Friedlaender
- Christopher Ehret
- Alessia Ranciaro
- Alain Froment
- Jibril B. Hirbo
- Agnes A. Awomoyi
- Jean-Marie Bodo
- Ogobara Doumbo
- Muntaser Ibrahim
- Abdalla T. Juma
- Maritha J. Kotze
- Godfrey Lema
- Jason H. Moore
- Holly Mortensen
- Thomas B. Nyambo
- Sabah A. Omar
- Kweli Powell
- Gideon S. Pretorius
- Michael W. Smith
- Mahamadou A. Thera
- Charles Wambebe
- James L. Weber
- Scott M. Williams
- ^ Templeton, Alan R. (2005). "Haplotype Trees and Modern Human Origins". American Journal of Physical Anthropology. 128 (Supplement 41: Yearbook of Physical Anthropology): 33–59. Bibcode:2005AJPA..128S..33T. doi:10.1002/ajpa.20351. ISSN 0002-9483. PMID 16369961.
- ^ deMenocal, Peter B. (2016). "Climate Shocks". Scientific American. Vol. 25, no. 4.
- ^ Barras, Colin (February 6, 2016). "Stone tools hint humans reached Asia much earlier". New Scientist.
- ^ Wolpoff, Milford H.; Hawks, John; Caspari, Rachel (May 2000). "Multiregional, Not Multiple Origins" (PDF). American Journal of Physical Anthropology. 112 (1): 129–136. doi:10.1002/(SICI)1096-8644(200005)112:1<129::AID-AJPA11>3.0.CO;2-K. hdl:2027.42/34270. ISSN 0002-9483. PMID 10766948. Archived (PDF) from the original on January 25, 2021. Retrieved May 12, 2013.
- ^ Wolpoff, M. H.; Spuhler, J. N.; Smith, F. H.; Radovcic, J.; Pope, G.; Frayer, D. W.; Eckhardt, R.; Clark, G. (August 12, 1988). "Modern Human Origins". Science. 241 (4867): 772–774. Bibcode:1988Sci...241..772W. doi:10.1126/science.3136545. ISSN 0036-8075. PMID 3136545. S2CID 5223638.
- ^ Owen, James (July 18, 2007). "Modern Humans Came Out of Africa, 'Definitive' Study Says". National Geographic News. Washington, DC: National Geographic Society. Archived from the original on June 27, 2018. Retrieved May 14, 2011.
- ^ Stringer, Chris B.; Andrews, Peter (March 11, 1988). "Genetic and fossil evidence for the origin of modern humans". Science. 239 (4845): 1263–1268. Bibcode:1988Sci...239.1263S. doi:10.1126/science.3125610. ISSN 0036-8075. PMID 3125610. S2CID 39200956.
- ^ Webster 2010, p. 53.
- ^ Ramachandran et al. 2010, p. 606.
- ^ Cann, Rebecca L.; Stoneking, Mark; Wilson, Allan C. (January 1, 1987). "Mitochondrial DNA and human evolution". Nature. 325 (6099): 31–36. Bibcode:1987Natur.325...31C. doi:10.1038/325031a0. ISSN 0028-0836. PMID 3025745. S2CID 4285418. Archived from the original on August 13, 2010. Retrieved April 21, 2015.
- ^ Gill, Victoria (May 1, 2009). "Africa's genetic secrets unlocked". BBC News. London: BBC. Archived from the original on May 14, 2020. Retrieved June 8, 2011.
- ^ Leakey 1994, pp. 87–89.
- ^ Jorde, Lynn B.; Bamshad, Michael; Rogers, Alan R. (February 1998). "Using mitochondrial and nuclear DNA markers to reconstruct human evolution". BioEssays. 20 (2): 126–136. doi:10.1002/(SICI)1521-1878(199802)20:2<126::AID-BIES5>3.0.CO;2-R. ISSN 0265-9247. PMID 9631658. S2CID 17203268.
- ^ Wall, Jeffrey D.; Lohmueller, Kirk E.; Plagnol, Vincent (August 2009). "Detecting Ancient Admixture and Estimating Demographic Parameters in Multiple Human Populations". Molecular Biology and Evolution. 26 (8): 1823–1827. doi:10.1093/molbev/msp096. ISSN 0737-4038. PMC 2734152. PMID 19420049.
- ^ Huertha Sanchez, Emilia et al. (2014), "Altitude adaptation in Tibetans caused by introgression of Denisovan-like DNA" (Nature Vol 512, August 14, 2014)
- ^ "Artificial intelligence applied to the genome identifies an unknown human ancestor". ScienceDaily. Archived from the original on January 16, 2019. Retrieved January 17, 2019.
- ^ a b c Lahr, Marta Mirazón; Petraglia, Mike; Stokes, Stephen; et al. "Searching for traces of the Southern Dispersal". Leverhulme Centre for Human Evolutionary Studies. University of Cambridge. Archived from the original on May 10, 2012. Retrieved April 21, 2015.
- ^ Simpson, Colin. DNA evidence suggests the whole world is a little bit Arab. The National. 16, June 2020. Archived at The Wayback Machine
- ^ Fernandes, V.; Alshamali, F.; Alves, M.; et al. (2012). "The Arabian cradle: Mitochondrial relicts of the first steps along the southern route out of Africa". American Journal of Human Genetics. 90 (2): 347–355. doi:10.1016/j.ajhg.2011.12.010. PMC 3276663. PMID 22284828.
- ^ Macaulay, Vincent; Hill, Catherine; Achilli, Alessandro; et al. (May 13, 2005). "Single, Rapid Coastal Settlement of Asia Revealed by Analysis of Complete Mitochondrial Genomes" (PDF). Science. 308 (5724): 1034–1036. Bibcode:2005Sci...308.1034M. doi:10.1126/science.1109792. ISSN 0036-8075. PMID 15890885. S2CID 31243109. Archived (PDF) from the original on August 28, 2022. Retrieved August 20, 2019.
- ^ Posth, C.; Renaud, G.; Mittnik, M.; Drucker, D. G.; Rougier, H.; Cupillard, C.; et al. (2016). "Pleistocene Mitochondrial Genomes Suggest a Single Major Dispersal of Non-Africans and a Late Glacial Population Turnover in Europe". Current Biology. 26 (6): 827–833. Bibcode:2016CBio...26..827P. doi:10.1016/j.cub.2016.01.037. hdl:2440/114930. PMID 26853362. S2CID 140098861.
- ^ Karmin, M.; Saag, L.; Vicente, M.; Wilson Sayres, M. A.; Järve, M.; Talas, U. G.; et al. (April 2015). "A recent bottleneck of Y chromosome diversity coincides with a global change in culture". Genome Research. 25 (4): 459–66. doi:10.1101/gr.186684.114. PMC 4381518. PMID 25770088.
- ^ Haber, M.; Jones, A. L.; Connell, B. A.; Arciero, E.; Yang, H.; Thomas, M. G.; et al. (August 2019). "A Rare Deep-Rooting D0 African Y-Chromosomal Haplogroup and Its Implications for the Expansion of Modern Humans Out of Africa". Genetics. 212 (4): 1421–1428. doi:10.1534/genetics.119.302368. PMC 6707464. PMID 31196864.
- ^ Stanford, Allen & Antón 2009.
- ^ a b Wood, Bernard A.; Richmond, Brian G. (July 2000). "Human evolution: Taxonomy and paleobiology". Journal of Anatomy. 197 (1): 19–60. doi:10.1046/j.1469-7580.2000.19710019.x. ISSN 1469-7580. PMC 1468107. PMID 10999270.
- ^ Ajit, Varki; Nelson, David L. (October 2007). "Genomic Comparisons of Humans and Chimpanzees" (PDF). Annual Review of Anthropology. 36: 191–209. doi:10.1146/annurev.anthro.36.081406.094339. ISSN 0084-6570. Archived (PDF) from the original on September 23, 2015. Retrieved April 26, 2015.
Sequence differences from the human genome were confirmed to be ~1% in areas that can be precisely aligned, representing ~35 million single base-pair differences. Some 45 million nucleotides of insertions and deletions unique to each lineage were also discovered, making the actual difference between the two genomes ~4%.
- ^ #author. Coghlan, Andy (May 30, 2002). "Just 2.5% of DNA turns mice into men". New Scientist. Retrieved August 18, 2024.
{{cite web}}:|last=has generic name (help) - ^ Dawkins 2004.
- ^ "Find Time of Divergence: Hominidae versus Hylobatidae". TimeTree. Archived from the original on April 18, 2015. Retrieved April 18, 2015.
- ^ Ruvolo, Maryellen (October 1997). "Genetic Diversity in Hominoid Primates". Annual Review of Anthropology. 26: 515–540. doi:10.1146/annurev.anthro.26.1.515. ISSN 0084-6570.
- ^ Ruvolo, Maryellen (March 1997). "Molecular Phylogeny of the Hominoids: Inferences from Multiple Independent DNA Sequence Data Sets". Molecular Biology and Evolution. 14 (3): 248–265. doi:10.1093/oxfordjournals.molbev.a025761. ISSN 0737-4038. PMID 9066793.
- ^ Patterson, N.; Richter, D. J.; Gnerre, S.; Lander, E. S.; Reich, D. (2006). "Genetic evidence for complex speciation of humans and chimpanzees". Nature. 441 (7097): 1103–1108. Bibcode:2006Natur.441.1103P. doi:10.1038/nature04789. PMID 16710306. S2CID 2325560.
- ^ Zimmer, Carl (May 17, 2023). "Study Offers New Twist in How the First Humans Evolved - A new genetic analysis of 290 people suggests that humans emerged at various times and places in Africa". The New York Times. Archived from the original on May 17, 2023. Retrieved May 18, 2023.
- ^ Ragsdale, Aaron P.; et al. (May 17, 2023). "A weakly structured stem for human origins in Africa". Nature. 167 (7962): 755–763. Bibcode:2023Natur.617..755R. doi:10.1038/s41586-023-06055-y. PMC 10208968. PMID 37198480.
- ^ Begun, David R. (October 2010). "Miocene Hominids and the Origins of the African Apes and Humans". Annual Review of Anthropology. 39: 67–84. doi:10.1146/annurev.anthro.012809.105047. ISSN 0084-6570.
- ^ Wood 2009, pp. 17–27.
- ^ Mitchell, Alanna (January 30, 2012). "DNA Turning Human Story Into a Tell-All". The New York Times. Archived from the original on January 1, 2022. Retrieved February 13, 2012.
- ^ Wood, Bernard A. (December 1996). "Human evolution". BioEssays. 18 (12): 945–954. doi:10.1002/bies.950181204. ISSN 0265-9247. PMID 8976151. S2CID 221464189.
- ^ Huxley, T. (1890). "The Aryan Question and Pre-Historic Man". Collected Essays: Volume VII, Man's Place in Nature. Archived from the original on July 26, 2011. Retrieved August 25, 2018.
- ^ Coghlan, Andy (March 15, 2018). "Our ancestors mated with the mystery 'Denisovan' people – twice". New Scientist. Archived from the original on August 5, 2018. Retrieved August 25, 2018.
- ^ a b Wei-Haas, Maya (August 22, 2018). "Ancient Girl's Parents Were Two Different Human Species – Born 90,000 years ago, the child is the first direct evidence of interbreeding among Neanderthals and their cousins the Denisovans". National Geographic. Archived from the original on June 14, 2019. Retrieved August 22, 2018.
- ^ Cell Press (March 28, 2016). "A world map of Neanderthal and Denisovan ancestry in modern humans". ScienceDaily. Archived from the original on August 26, 2018. Retrieved August 25, 2018.
- ^ Sankararaman, S.; Mallick, S.; Patterson, N.; Reich, D. (March 28, 2016). "The Combined Landscape of Denisovan and Neanderthal Ancestry in Present-Day Humans". Current Biology. 26 (9): 1241–1247. Bibcode:2016CBio...26.1241S. doi:10.1016/j.cub.2016.03.037. PMC 4864120. PMID 27032491.
- ^ "Human-Neandertal Comparisons". the Neandertal Genome. American Association for the Advancement of Science. 2010. Archived from the original on May 28, 2020.
- ^ Dannemann, M.; Andrés, A. M.; Kelso, J. (2016). "Introgression of Neandertal- and Denisovan-like Haplotypes Contributes to Adaptive Variation in Human Toll-like Receptors". American Journal of Human Genetics. 98 (1): 22–33. doi:10.1016/j.ajhg.2015.11.015. PMC 4716682. PMID 26748514.
- ^ Gittelman, Rachel M.; Schraiber, Joshua G.; Vernot, Benjamin; Mikacenic, Carmen; Wurfel, Mark M.; Akey, Joshua M. (2016). "Archaic Hominin Admixture Facilitated Adaptation to Out-of-Africa Environments". Current Biology. 26 (24): 3375–3382. Bibcode:2016CBio...26.3375G. doi:10.1016/j.cub.2016.10.041. PMC 6764441. PMID 27839976.
- ^ a b Plummer, Thomas (2004). "Flaked stones and old bones: Biological and cultural evolution at the dawn of technology". American Journal of Physical Anthropology. 125 (Supplement 39: Yearbook of Physical Anthropology): 118–164. Bibcode:2004AJPA..125S.118P. doi:10.1002/ajpa.20157. ISSN 0002-9483. PMID 15605391.
- ^ Wong, Kate (April 15, 2015). "Archaeologists Take Wrong Turn, Find World's Oldest Stone Tools". Scientific American (Blog). ISSN 0036-8733. Archived from the original on May 8, 2015. Retrieved May 3, 2015.
- ^ Balter, Michael (April 14, 2015). "World's oldest stone tools discovered in Kenya". Science (News). doi:10.1126/science.aab2487. ISSN 0036-8075. Archived from the original on October 20, 2021. Retrieved May 3, 2015.
- ^ Drake, Nadia (April 16, 2015). "Oldest Stone Tools Discovered in Kenya". National Geographic News. Washington, DC: National Geographic Society. Archived from the original on April 23, 2015. Retrieved May 3, 2015.
- ^ Yellen, J. E.; Brooks, A. S.; Cornelissen, E.; Mehlman, M. J.; Stewart, K. (April 28, 1995). "A middle stone age worked bone industry from Katanda, Upper Semliki Valley, Zaire". Science. 268 (5210): 553–556. Bibcode:1995Sci...268..553Y. doi:10.1126/science.7725100. PMID 7725100.
Sources
[edit]- Aiello, Leslie; Dean, Christopher (1990). An Introduction to Human Evolutionary Anatomy. London / San Diego: Elsevier Academic Press. ISBN 978-0-12-045591-1. LCCN 95185095. OCLC 33408268.
- Bogin, Barry (1997). "The Evolution of Human Nutrition". In Romanucci-Ross, Lola; Moerman, Daniel E.; Tancredi, Laurence R. (eds.). The Anthropology of Medicine: From Culture to Method (3rd ed.). Westport, Connecticut: Bergin & Garvey. ISBN 978-0-89789-516-3. LCCN 96053993. OCLC 36165190.
- Bown, Thomas M.; Rose, Kenneth D. (1987). Patterns of Dental Evolution in Early Eocene Anaptomorphine Primates (Omomyidae) From the Bighorn Basin, Wyoming (PDF). Memoir (Paleontological Society). Vol. 23. Tulsa, Oklahoma: Paleontological Society. OCLC 16997265. Archived from the original on November 11, 2016. Retrieved April 25, 2015.
- Boyd, Robert; Silk, Joan B. (2003). How Humans Evolved (3rd ed.). New York: W. W. Norton & Co. ISBN 978-0-393-97854-4. LCCN 2002075336. OCLC 49959461.
- Boyd, Robert; Silk, Joan B. (2021). How Humans Evolved (9th ed.). New York: W. W. Norton & Co. ISBN 978-0-393-53315-6. OCLC 1282924892.
- Brues, Alice M.; Snow, Clyde C. (1965). "Physical Anthropology". In Siegel, Bernard J. (ed.). Biennial Review of Anthropology 1965. Biennial Review of Anthropology. Vol. 4. Stanford University Press. ISBN 978-0-8047-1746-5. ISSN 0067-8503. LCCN 59012726. OCLC 01532912.
- Bryson, Bill (2004) [Originally published 2003]. "The Mysterious Biped". A Short History of Nearly Everything. Toronto: Anchor Canada. ISBN 978-0-385-66004-4. LCCN 2003046006. OCLC 55016591.
- Cameron, David W. (2004). Hominid Adaptations and Extinctions. Sydney: University of New South Wales Press. ISBN 978-0-86840-716-6. LCCN 2004353026. OCLC 57077633.
- Cochran, Gregory; Harpending, Henry (2009). The 10,000 Year Explosion: How Civilization Accelerated Human Evolution. New York: Basic Books. ISBN 978-0-465-00221-4. LCCN 2008036672. OCLC 191926088.
- Curry, James R. (2008). Children of God: Children of Earth. Bloomington, Indiana: AuthorHouse. ISBN 978-1-4389-1846-4. OCLC 421466369.
- Darwin, Charles (1859). On the Origin of Species by Means of Natural Selection, or the Preservation of Favoured Races in the Struggle for Life (PDF). Archived (PDF) from the original on March 4, 2016. Retrieved May 13, 2017.
- Darwin, Charles (1981) [Originally published 1871; London: John Murray]. The Descent of Man, and Selection in Relation to Sex. Introduction by John Tyler Bonner and Robert M. May (Reprint ed.). Princeton University Press. ISBN 978-0-691-02369-4. LCCN 80008679. OCLC 7197127.
- Dawkins, Richard (2004). The Ancestor's Tale: A Pilgrimage to the Dawn of Evolution. Boston: Houghton Mifflin. ISBN 978-0-618-00583-3. LCCN 2004059864. OCLC 56617123.
- DeSalle, Rob; Tattersall, Ian (2008). Human Origins: What Bones and Genomes Tell Us About Ourselves. Texas A&M University Anthropology Series. Vol. 13. College Station: Texas A&M University Press. ISBN 978-1-58544-567-7. OCLC 144520427.
- Diamond, Jared (1999). Guns, Germs, and Steel: The Fates of Human Societies. New York: W. W. Norton & Co. ISBN 978-0-393-31755-8. LCCN 2005284124. OCLC 35792200.
- Freeman, Scott; Herron, Jon C. (2007). Evolutionary Analysis (4th ed.). Upper Saddle River, New Jersey: Pearson Prentice Hall. ISBN 978-0-13-227584-2. LCCN 2006034384. OCLC 73502978.
- Johanson, Donald; Edey, Maitland (1981). Lucy, the Beginnings of Humankind. St Albans: Granada. ISBN 978-0-586-08437-3.
- Montgomery, William M. (1988) [Originally published 1974]. "Germany". In Glick, Thomas F. (ed.). The Comparative Reception of Darwinisms. University of Chicago Press. ISBN 978-0-226-29977-8. LCCN 87035814. OCLC 17328115. "The Conference on the Comparative Reception of Darwinism was held in Austin, Texas, on April 22 and 23, 1972, under the joint sponsorship of the American Council of Learned Societies and the University of Texas at Austin"
- Kondo, Shiro, ed. (1985). "Primate Morphophysiology, Locomotor Analyses, and Human Bipedalism". American Journal of Physical Anthropology. 70 (2). University of Tokyo Press: 278–279. doi:10.1002/ajpa.1330700214. ISBN 978-4-13-066093-8. LCCN 85173489. OCLC 12352830.
- Leakey, Richard E. (1994). The Origin of Humankind. Science Masters Series. New York: Basic Books. ISBN 978-0-465-03135-1. LCCN 94003617. OCLC 30739453.
- M'charek, Amade (2005). The Human Genome Diversity Project: An Ethnography of Scientific Practice. Cambridge Studies in Society and the Life Sciences. Cambridge University Press. ISBN 978-0-521-83222-9. LCCN 2004052648. OCLC 55600894.
- Martin, Robert D. (2001). "Primates, Evolution of". In Smelser, Neil J.; Baltes, Paul B. (eds.). International Encyclopedia of the Social & Behavioral Sciences. Amsterdam / New York: Elsevier. pp. 12032–12038. doi:10.1016/B0-08-043076-7/03083-7. ISBN 978-0-08-043076-8. LCCN 2001044791. OCLC 47869490.
- Maxwell, Mary (1984). Human Evolution: A Philosophical Anthropology. New York: Columbia University Press. ISBN 978-0-231-05946-6. LCCN 83024005. OCLC 10163036.
- McHenry, Henry M. (2009). "Human Evolution". In Ruse, Michael; Travis, Joseph (eds.). Evolution: The First Four Billion Years. Foreword by Edward O. Wilson. Cambridge, Massachusetts: Belknap Press of Harvard University. ISBN 978-0-674-03175-3. LCCN 2008030270. OCLC 225874308.
- Ramachandran, Sohini; Tang, Hua; Gutenkunst, Ryan N.; Bustamante, Carlos D. (2010). "Genetics and Genomics of Human Population Structure". In Speicher, Michael R.; Antonarakis, Stylianos E.; Motulsky, Arno G. (eds.). Vogel and Motulsky's Human Genetics: Problems and Approaches (4th ed.). Heidelberg / London / New York: Springer. doi:10.1007/978-3-540-37654-5. ISBN 978-3-540-37653-8. LCCN 2009931325. OCLC 549541244.
- Robinson, J. T. (2008) [Originally published 1963; Chicago, Illinois: Aldine Transaction]. "Adaptive Radiation in the Australopithecines and the Origin of Man". In Howell, F. Clark; Bourlière, François (eds.). African Ecology and Human Evolution. New Brunswick, New Jersey: Transaction Publishers. ISBN 978-0-202-36136-9. LCCN 2007024716. OCLC 144770218.
- Srivastava, R. P. (2009). Morphology of the Primates and Human Evolution. New Delhi: PHI Learning. ISBN 978-81-203-3656-8. OCLC 423293609.
- Stanford, Craig; Allen, John S.; Antón, Susan C. (2009). Biological Anthropology: The Natural History of Humankind (2nd ed.). Upper Saddle River, New Jersey: Pearson Prentice Hall. ISBN 978-0-13-601160-6. LCCN 2007052429. OCLC 187548835.
- Strickberger, Monroe W. (2000). Evolution (3rd ed.). Sudbury, Massachusetts: Jones & Bartlett Publishers. ISBN 978-0-7637-1066-8. LCCN 99032072. OCLC 41431683.
- Stringer, Chris B. (1994) [1992]. "Evolution of Early Humans". In Jones, Steve; Martin, Robert D.; Pilbeam, David (eds.). The Cambridge Encyclopedia of Human Evolution. Cambridge University Press. ISBN 978-0-521-32370-3. LCCN 92018037. OCLC 444512451.
- Swisher, Carl C. III; Curtis, Garniss H.; Lewin, Roger (2001) [Originally published 2000]. Java Man: How Two Geologists Changed Our Understanding of Human Evolution. University of Chicago Press. ISBN 978-0-226-78734-3. LCCN 2001037337. OCLC 48066180.
- Trent, Ronald J. (2005). Molecular Medicine: An Introductory Text (3rd ed.). Burlington, Massachusetts: Elsevier Academic Press. ISBN 978-0-12-699057-7. LCCN 2004028087. OCLC 162577235.
- Trevathan, Wenda R. (2011) [Originally published 1987; New York: Aldine De Gruyter]. Human Birth: An Evolutionary Perspective. New Brunswick, New Jersey: Transaction Publishers. ISBN 978-1-4128-1502-4. LCCN 2010038249. OCLC 669122326.
- Ungar, Peter S.; Teaford, Mark F., eds. (2002). Human Diet: Its Origin and Evolution. Westport, Connecticut: Bergin & Garvey. ISBN 978-0-89789-736-5. LCCN 2001043790. OCLC 537239907.
- Walker, Alan (2007). "Early Hominin Diets: Overview and Historical Perspectives". In Ungar, Peter (ed.). Evolution of the Human Diet: The Known, the Unknown, and the Unknowable. Human Evolution Series. Oxford University Press. ISBN 978-0-19-518346-7. LCCN 2005036120. OCLC 132816551.
- Wallace, David Rains (2004). Beasts of Eden: Walking Whales, Dawn Horses, and Other Enigmas of Mammal Evolution. Berkeley: University of California Press. ISBN 978-0-520-24684-3. LCCN 2003022857. OCLC 53254011.
- Webster, Donovan (2010). Meeting the Family: One Man's Journey Through His Human Ancestry. Foreword by Spencer Wells. Washington, DC: National Geographic Society. ISBN 978-1-4262-0573-6. LCCN 2009050471. OCLC 429022321.
- Wood, Bernard A. (2009). "Where Does the Genus Homo Begin, and How Would We Know?". In Grine, Frederick E.; Fleagle, John G.; Leakey, Richard E. (eds.). The First Humans – Origin and Early Evolution of the Genus Homo. Vertebrate Paleobiology and Paleoanthropology. Dordrecht: Springer Netherlands. pp. 17–28. doi:10.1007/978-1-4020-9980-9_3. ISBN 978-1-4020-9979-3. ISSN 1877-9077. LCCN 2009927083. OCLC 310400980. "Contributions from the Third Stony Brook Human Evolution Symposium and Workshop October 3–7, 2006."
Further reading
[edit]- Alexander, Richard D. (1990). How Did Humans Evolve? Reflections on the Uniquely Unique Species (PDF). Special Publication. Ann Arbor: Museum of Zoology, University of Michigan. pp. 1–38. LCCN 90623893. OCLC 22860997. Archived from the original (PDF) on March 4, 2016. Retrieved May 6, 2015.
- Barton, Nicholas H.; Briggs, Derek E. G.; Eisen, Jonathan A.; et al. (2007). Evolution. Cold Spring Harbor Laboratory Press. ISBN 978-0-87969-684-9. LCCN 2007010767. OCLC 86090399.
- Bellwood, Peter (2022). The Five-Million-Year Odyssey: The Human Journey from Ape to Agriculture. Princeton: Princeton University Press. ISBN 978-0-691-19757-9.
- Enard, Wolfgang; Przeworski, Molly; Fisher, Simon E.; et al. (August 22, 2002). "Molecular evolution of FOXP2, a gene involved in speech and language". Nature. 418 (6900): 869–872. Bibcode:2002Natur.418..869E. doi:10.1038/nature01025. hdl:11858/00-001M-0000-0012-CB89-A. ISSN 0028-0836. PMID 12192408. S2CID 4416233.
- Flinn, Mark V.; Geary, David C.; Ward, Carol V. (2005). "Ecological dominance, social competition, and coalitionary arms races: Why humans evolved extraordinary intelligence" (PDF). Evolution and Human Behavior. 26 (1): 10–46. Bibcode:2005EHumB..26...10F. doi:10.1016/j.evolhumbehav.2004.08.005. ISSN 1090-5138. Archived (PDF) from the original on June 4, 2007. Retrieved May 5, 2015.
- Galinon-Melenec, Béatrice (2015). "From "Traces" and "Human Trace" to "Human–Trace Paradigm"". In Parrend, Pierre; Bourgine, Paul; Collet, Pierre (eds.). First Complex systems Digital Campus World E-Conference. Tempe, Arizona: Springer.
- Gibbons, Ann (2006). The First Human: The Race to Discover our Earliest Ancestors. New York: Doubleday. ISBN 978-0-385-51226-8. LCCN 2005053780. OCLC 61652817.
- Hartwig, Walter C., ed. (2002). The Primate Fossil Record. Cambridge Studies in Biological and Evolutionary Anthropology. Vol. 33. Cambridge University Press. ISBN 978-0-521-66315-1. LCCN 2001037847. OCLC 47254191.
- Heizmann, Elmar P. J.; Begun, David R. (November 2001). "The oldest Eurasian hominoid" (PDF). Journal of Human Evolution. 41 (5): 463–481. Bibcode:2001JHumE..41..463H. doi:10.1006/jhev.2001.0495. ISSN 0047-2484. PMID 11681862. S2CID 21230992.
- Hill, Andrew; Ward, Steven (1988). "Origin of the hominidae: The record of African large hominoid evolution between 14 my and 4 my". American Journal of Physical Anthropology. 31 (59): 49–83. Bibcode:1988AJPA...31S..49H. doi:10.1002/ajpa.1330310505. ISSN 0002-9483.
- Hoagland, Hudson (1964). "Science and the New Humanism". Science. 143 (3602): 111–114. Bibcode:1964Sci...143..111H. doi:10.1126/science.143.3602.111. PMID 17781189.
- Ijdo, Jacob W.; Baldini, Antonio; Ward, David C.; et al. (October 15, 1991). "Origin of human chromosome 2: An ancestral telomere-telomere fusion". Proceedings of the National Academy of Sciences of the United States of America. 88 (20): 9051–9055. Bibcode:1991PNAS...88.9051I. doi:10.1073/pnas.88.20.9051. ISSN 0027-8424. PMC 52649. PMID 1924367. – two ancestral ape chromosomes fused to give rise to human chromosome 2
- Johanson, Donald; Wong, Kate (2010). Lucy's Legacy: The Quest for Human Origins. New York: Three Rivers Press. ISBN 978-0-307-39640-2. LCCN 2010483830. OCLC 419801728.
- Jones, Steve; Martin, Robert D.; Pilbeam, David, eds. (1994) [1992]. The Cambridge Encyclopedia of Human Evolution. Foreword by Richard Dawkins. Cambridge University Press. ISBN 978-0-521-32370-3. LCCN 92018037. OCLC 444512451. (This book contains very useful, information-dense chapters on primate evolution in general, and human evolution in particular, including fossil history.)
- Kawabata, Hiroshi; Kaifu, Yousuke (2020). Lost in Evolution: Exploring Humanity's Path in Asia. Tokyo: Japan Publishing Industry Foundation for Culture. ISBN 978-4-86658-133-0. Archived from the original on October 20, 2022. Retrieved October 20, 2022.
- Leakey, Richard E.; Lewin, Roger (1992). Origins Reconsidered: In Search of What Makes us Human. New York: Doubleday. ISBN 978-0-385-41264-3. LCCN 92006661. OCLC 25373161.
- Lewin, Roger (1997). Bones of Contention: Controversies in the Search for Human Origins (2nd ed.). University of Chicago Press. ISBN 978-0-226-47651-3. LCCN 97000972. OCLC 36181117.
- Morwood, Mike; van Oosterzee, Penny (2007). A New Human: The Startling Discovery and Strange Story of the 'Hobbits' of Flores, Indonesia. New York: Smithsonian Books / HarperCollins. ISBN 978-0-06-089908-0. LCCN 2006052267. OCLC 76481584.
- Oppenheimer, Stephen (2003). Out of Eden: The Peopling of the World. London: Constable & Robinson. ISBN 978-1-84119-697-8. LCCN 2005482222. OCLC 52195607.
- Ovchinnikov, Igor V.; Götherström, Anders; Romanova, Galina P.; et al. (March 30, 2000). "Molecular analysis of Neanderthal DNA from the Northern Caucasus". Nature. 404 (6777): 490–493. Bibcode:2000Natur.404..490O. doi:10.1038/35006625. ISSN 0028-0836. PMID 10761915. S2CID 3101375.
- Roberts, Alice M. (2009). The Incredible Human Journey: The Story of How We Colonised the Planet. London: Bloomsbury Publishing. ISBN 978-0-7475-9839-8. OCLC 310156315.
- Shreeve, James (1995). The Neandertal Enigma: Solving the Mystery of Modern Human Origins. New York: William Morrow & Co. ISBN 978-0-688-09407-2. LCCN 95006337. OCLC 32088673.
- Stringer, Chris B. (2011). The Origin of Our Species. London: Allen Lane. ISBN 978-1-84614-140-9. LCCN 2011489742. OCLC 689522193.
- Stringer, Chris B.; Andrews, Peter (2005). The Complete World of Human Evolution. London / New York: Thames & Hudson. ISBN 978-0-500-05132-0. LCCN 2004110563. OCLC 224377190.
- Stringer, Christopher; McKie, Robin (1997). African Exodus: The Origins of Modern Humanity. New York: Henry Holt & Co. ISBN 978-0-8050-2759-4. LCCN 96037718. OCLC 36001167.
- Sykes, Rebecca Wragg (2020). Kindred: Neanderthal Life, Love, Death and Art. London: Bloomsbury Sigma. ISBN 978-1-4729-3749-0.
- Tattersall, Ian (2008). The Fossil Trail: How We Know What We Think We Know About Human Evolution (2nd ed.). Oxford University Press. ISBN 978-0-19-536766-9. LCCN 2008013654. OCLC 218188644.
- van Oosterzee, Penny (1999). Dragon Bones: The Story of Peking Man. St Leonards, New South Wales: Allen & Unwin Australia. ISBN 978-1-86508-123-6. LCCN 00300421. OCLC 45853997.
- Wade, Nicholas (2006). Before the Dawn: Recovering the Lost History of Our Ancestors. New York: Penguin. ISBN 978-1-59420-079-3. LCCN 2005055293. OCLC 62282400.
- Walker, Alan; Shipman, Pat (1996). The Wisdom of the Bones: In Search of Human Origins. London: Weidenfeld & Nicolson. ISBN 978-0-297-81670-6. OCLC 35202130.
- Weiss, Mark L.; Mann, Alan E. (1985). Human Biology and Behavior: An Anthropological Perspective (4th ed.). Boston: Little, Brown & Co. ISBN 978-0-316-92894-6. LCCN 85000158. OCLC 11726796. (This book contains very accessible descriptions of human and non-human primates, their evolution, and fossil history.)
- Wells, Spencer (2003) [Originally published 2002; Princeton, NJ: Princeton University Press]. The Journey of Man: A Genetic Odyssey (Random House trade paperback ed.). New York: Random House. ISBN 978-0-8129-7146-0. LCCN 2003066679. OCLC 53287806.
External links
[edit]- "Race, Evolution and the Science of Human Origins" by Allison Hopper, Scientific American (July 5, 2021).
- "The evolution of man". BBC Science & Nature. Retrieved May 6, 2015.
- "Becoming Human". Arizona State University's Institute of Human Origins. Retrieved May 6, 2015.
- "Bones, Stones and Genes: The Origin of Modern Humans" (Video lecture series). Howard Hughes Medical Institute. Archived from the original on April 24, 2015. Retrieved May 6, 2015.
- "Evolution Figures: Chapter 25". Cold Spring Harbor Laboratory Press. Retrieved May 6, 2015. – Illustrations from the book Evolution (2007)
- "Human Evolution". Smithsonian Institution's Human Origins Program. Retrieved June 24, 2013.
- "Human Evolution Timeline". ArchaeologyInfo.com. Archived from the original on June 18, 2013. Retrieved June 24, 2013.
- "Human Trace" video (2015) Normandy University UNIHAVRE, CNRS, IDEES, E.Laboratory on Human Trace Unitwin Complex System Digital Campus UNESCO.
- Lambert, Tim (Producer) (June 24, 2015). First Peoples. London: Wall to Wall Television. OCLC 910115743. Retrieved July 18, 2015.
- Shaping Humanity Video 2013 Yale University
- Human Timeline (Interactive) – Smithsonian, National Museum of Natural History (August 2016).
- Human Evolution, BBC Radio 4 discussion with Steve Jones, Fred Spoor & Margaret Clegg (In Our Time, February 16, 2006)
- Evolutionary Timeline of Home Sapiens − Smithsonian (February 2021)
- History of Human Evolution in the United States – Salon (August 24, 2021)
Human evolution
View on GrokipediaOrigins of Primates and Hominins
Early Primate Evolution
Primates are a diverse order of mammals characterized by several key adaptations that facilitated an arboreal lifestyle, including forward-facing eyes providing stereoscopic vision for depth perception, grasping hands and feet with opposable thumbs and toes, and nails instead of claws on digits for enhanced manipulation of objects and branches.[4] These traits, combined with a relatively large brain size compared to body mass, enabled early primates to navigate complex forest environments and exploit varied food resources such as fruits and insects.[5] Unlike many other mammals, primates exhibit reduced reliance on olfaction, with a shift toward enhanced visual acuity, which supported precise foraging and predator avoidance in treetop habitats.[6] The order Primates emerged in the fossil record shortly after the Cretaceous-Paleogene extinction event around 66 million years ago (Mya), with the earliest potential representatives appearing between 65 and 55 Mya during the Paleocene epoch.[6] These initial forms, known as Plesiadapiformes, were small, squirrel-like mammals that displayed proto-primate features such as elongated fingers for grasping and forward-oriented orbits, though they lacked some defining traits like fully opposable thumbs.[4] By the Eocene epoch (approximately 56-34 Mya), true primates (Euprimates) had diversified in response to warming climates and expanding forests, marking a period of adaptive radiation that set the stage for later evolutionary branches.[7] Early primates diverged into two major suborders: Strepsirrhini, encompassing lemurs, lorises, and galagos, which retain more primitive traits like a wet nose and grooming claws; and Haplorhini, including tarsiers, monkeys, and apes, characterized by dry noses and more advanced visual systems.[4] The split between these groups is estimated to have occurred around 74 Mya based on molecular clock analyses, though the fossil record primarily documents it in the early Eocene.[8] Within Haplorhini, the anthropoids (monkeys and apes) evolved around 40 Mya during the late Eocene to early Oligocene, featuring further refinements in brain size and social complexity.[9] Social behaviors, such as group living and cooperative grooming, began to emerge in these early primates, promoting kin recognition and resource sharing in arboreal settings.[5] Fossil evidence from the Eocene and Oligocene epochs provides critical insights into these developments, with sites in North America, Europe, and Africa yielding specimens of early euprimates. For instance, omomyids and adapids from the Eocene represent proto-haplorhines and proto-strepsirrhines, respectively, showcasing small body sizes and insectivorous diets.[7] A pivotal Oligocene fossil is Aegyptopithecus zeuxis from Egypt's Fayum Depression, dated to about 30 Mya, which is considered an early catarrhine (ancestor to Old World monkeys and apes) with a monkey-like dentition and arboreal adaptations.[10] These fossils illustrate the gradual refinement of primate traits, including opposable thumbs that improved locomotor efficiency and dexterity.[11]Divergence of Hominins from Great Apes
The divergence of the hominin lineage from other great apes represents a pivotal event in primate evolution, marking the separation of the human ancestral line from the common ancestor shared with chimpanzees and bonobos (Pan troglodytes and Pan paniscus) as well as gorillas (Gorilla spp.). Molecular clock analyses, which estimate divergence times based on genetic mutation rates, indicate that the split between the human and chimpanzee lineages occurred approximately 6 to 7 million years ago (Mya), while the gorilla lineage diverged slightly earlier, around 8 to 10 Mya.[12] These estimates are derived from genomic comparisons across great apes, incorporating fossil-calibrated rates to account for generation times and substitution patterns observed in nuclear DNA.[13] The broader great ape divergence, including orangutans, extends back further to about 12 to 16 Mya, but the key hominin-specific split aligns with late Miocene transitions in African ecosystems.[14] Fossil evidence from the late Miocene provides the earliest potential glimpses of this divergence, though the scarcity of remains complicates precise attribution. Sahelanthropus tchadensis, dated to about 7 Mya from sites in Chad, exhibits reduced canine size—a trait more aligned with hominins than extant great apes—along with a relatively small braincase and possible indicators of upright posture, suggesting it may represent the earliest known member of the human lineage post-split.[15] Similarly, Orrorin tugenensis fossils from Kenya, approximately 6 Mya, include femoral fragments showing morphological features consistent with partial bipedalism, such as a thickened cortex and shortened neck, hinting at locomotor adaptations distinct from those of arboreal apes.[16] By around 4.4 Mya, Ardipithecus ramidus from Ethiopia demonstrates woodland adaptations, with dentition and postcranial elements indicating a mix of tree-climbing and ground-dwelling behaviors in forested environments, further supporting the transition away from a fully arboreal great ape lifestyle.[17] This divergence coincided with significant environmental changes during the Miocene, including global cooling and drying trends that promoted the expansion of open savannas across East Africa around 10 to 15 Mya. These shifts, driven by tectonic uplift and altered monsoon patterns, reduced dense forest cover and encouraged a transition from arboreal to more terrestrial habits among early hominins, contrasting with the persistent woodland preferences of great ape ancestors.[18] Genetic evidence reinforces this split, notably the end-to-end fusion of two ancestral chromosomes in the human lineage, resulting in human chromosome 2, which retains vestigial telomere sequences and a centromere from the original ape chromosomes—a marker absent in chimpanzees and gorillas.[19] Additionally, the loss of function in genes such as MYH16, which encodes a myosin heavy chain protein essential for powerful jaw muscles, occurred in the early hominin lineage, leading to reduced masticatory strength compared to great apes and facilitating subsequent cranial expansions.[20] Initial adaptations following the divergence emphasized flexibility in locomotion and diet rather than full commitment to open habitats. Partial bipedalism likely emerged in the late Miocene as an energy-efficient means for traversing mixed woodland-savanna mosaics, though retained arboreal capabilities are evident in early fossils. Dietary shifts toward C4 resources, such as grasses and sedges, are indicated by isotopic signatures in late Miocene and early Pliocene hominin teeth, suggesting opportunistic consumption of these plants or their herbivores, which provided a nutritional edge in expanding grasslands without requiring specialized grazing dentition.[21] These changes underscore the hominin lineage's adaptive versatility during the immediate post-divergence period.Genus Australopithecus and Early Hominins
The genus Australopithecus represents a pivotal group of early hominins that lived between approximately 4.2 and 2 million years ago (Mya), bridging the gap from Miocene apes to the genus Homo through the establishment of obligate bipedalism and adaptations to diverse African environments.[22] These species, primarily known from East and South African fossil sites, exhibited a mix of primitive and derived traits, including small brain sizes and dental adaptations for processing tough plant foods, while demonstrating fully terrestrial locomotion.[22] Fossils of Australopithecus have been crucial in reconstructing the mosaic nature of hominin evolution, where locomotor changes preceded significant cognitive advancements.[23] The earliest recognized species, Australopithecus anamensis, dates to 4.2–3.9 Mya and is known from sites in Kenya and Ethiopia, featuring a mix of ape-like and hominin dental traits such as thick enamel and a forward-positioned jaw.[22] This was followed by A. afarensis (3.9–2.9 Mya), best exemplified by the partial skeleton "Lucy" (AL 288-1) discovered in Hadar, Ethiopia, in 1974, which preserves about 40% of the skeleton and confirms bipedal posture through pelvic and lower limb morphology.[24] A. africanus (3–2 Mya) from South African caves like Taung and Sterkfontein shows similar bipedal adaptations but with more gracile cranial features.[22] Later species include A. sediba (~2 Mya) from Malapa, South Africa, which displays a unique combination of Australopithecus and early Homo traits in its hands and feet.[22] The "robust" forms, often classified under Paranthropus—such as P. robustus (2–1.2 Mya) in South Africa and P. boisei (2.3–1.2 Mya) in East Africa—existed contemporaneously with gracile australopiths but specialized in heavy chewing.[25] Key anatomical traits of Australopithecus include obligate bipedalism, evidenced by a valgus knee angle, arched foot structure, and S-shaped spinal curvature that positioned the foramen magnum centrally for upright posture.[22] Brain volumes ranged from 400–500 cubic centimeters (cc), comparable to those of chimpanzees, indicating limited encephalization at this stage.[22] In Paranthropus, megadontia—large molars and robust mandibles—facilitated grinding of abrasive foods, with sagittal crests providing attachment for massive temporalis muscles.[25] Sexual dimorphism was pronounced, with males larger and more robust than females, as seen in canine size differences and body mass estimates.[23] These hominins inhabited mixed woodland-savanna environments in the East and South African rift valleys and cave systems, adapting to fluctuating climates during the Pliocene-Pleistocene transition.[22] Dietary evidence from dental microwear, stable carbon isotopes (δ¹³C), and plant microfossils in tartar reveals a reliance on C₃ plants (trees, shrubs) supplemented by C₄ grasses and sedges, indicating opportunistic foraging for fruits, leaves, and underground storage organs.[26] Paranthropus species show higher C₄ consumption, suggesting specialization on tougher, low-quality vegetation amid increasing aridity.[25] Direct evidence for bipedal locomotion comes from the Laetoli footprints in Tanzania, dated to 3.66 Mya, where three trails of 72 prints demonstrate a human-like striding gait with heel-strike and toe-off, likely made by A. afarensis.[27] These tracks, preserved in volcanic ash, show a compliant gait with shorter strides than modern humans, reflecting a transitional locomotor style that retained some arboreal capabilities.[28] Debates persist on the classification of "robust" forms: some paleoanthropologists treat Paranthropus as a distinct genus due to their specialized craniodental adaptations, while others view them as an extreme variant within Australopithecus, emphasizing shared postcranial bipedal features.[25] Their extinction around 1.2–1 Mya is attributed to environmental shifts toward more open grasslands and intensified competition, as C₄-dominated landscapes reduced access to preferred foods.[29]Evolution of the Genus Homo
Early Homo Species
The early Homo species represent the initial diversification of the genus Homo in Africa, emerging around 2.8 million years ago (Mya) from Australopithecus ancestors and characterized by modest increases in brain size and the onset of systematic stone tool manufacture.[30][31] These species include Homo habilis, dated to approximately 2.3–1.4 Mya, known from fossils at sites like Olduvai Gorge in Tanzania and Koobi Fora in Kenya; Homo rudolfensis, spanning 2.4–1.8 Mya, primarily represented by the KNM-ER 1470 cranium from Koobi Fora; Homo gautengensis, around 2 Mya, identified from cranial and dental remains in South African cave sites such as Sterkfontein and Swartkrans; and Homo georgicus, dated to about 1.8 Mya, based on skulls from Dmanisi in Georgia. This period marks a transitional phase, with these taxa coexisting alongside robust australopiths like Paranthropus boisei in eastern Africa, suggesting competitive or complementary ecological niches.[32] Key anatomical traits of early Homo species include cranial capacities ranging from 500 to 800 cubic centimeters (cc), a reduction in jaw and tooth size compared to Australopithecus, and more rounded braincases, though postcranial remains indicate body sizes similar to those of earlier hominins (around 30–40 kg).[30][33] These features are evident in H. habilis specimens like OH 7 from Olduvai Gorge, which show a brain size of about 650 cc and smaller molars, and in H. rudolfensis, with its larger face and teeth but similar overall encephalization. H. gautengensis fossils, such as StW 53, exhibit a braincase up to 775 cc and a broader face, while H. georgicus crania from Dmanisi (e.g., D2700) display primitive traits like prominent browridges alongside reduced cheek teeth. Associated archaeological evidence points to the Oldowan tool industry, consisting of simple choppers, flakes, and cores made from basalt or quartzite, first appearing around 2.6 Mya but firmly linked to early Homo by 2.3 Mya at sites like Gona and Olduvai. Faunal remains at these locations, including cut-marked bones of small mammals and scavenged large herbivores, suggest opportunistic scavenging and possibly hunting of small game, indicating dietary flexibility beyond plant-based foraging.[32] Significant debates surround the classification and evolutionary coherence of these species, particularly whether H. habilis truly belongs in the genus Homo or should be reclassified as a late Australopithecus due to its mosaic of primitive (e.g., long arms, curved fingers) and derived (e.g., tool-associated) traits.[34] Some researchers argue that H. habilis and H. rudolfensis represent a single variable species or sexual dimorphs rather than distinct taxa, based on overlapping cranial metrics from Koobi Fora, while others maintain separation due to consistent morphological differences.[32] H. gautengensis remains controversial, with critics viewing it as a junior synonym of H. habilis, though its South African context highlights regional variation in early Homo. The H. georgicus fossils, while sometimes subsumed under Homo erectus, underscore early Homo's morphological diversity and potential for dispersal. These early Homo species played a pivotal role as a bridge to later Homo lineages, facilitating adaptations like group living inferred from clustered tool scatters and bone accumulations at Olduvai and Koobi Fora, which imply social foraging and resource sharing among small bands.[32] Their tool use and meat consumption likely contributed to selective pressures for encephalization, setting the stage for the more advanced traits seen in subsequent species, though they remained largely confined to African savanna-woodland environments.[31]Homo erectus and Its Descendants
Homo erectus represents a pivotal species in human evolution, characterized by its long duration and extensive geographic range. Emerging around 1.9 million years ago in Africa, this species persisted until approximately 100,000 years ago in some regions, marking one of the longest-lived hominin taxa.[35] African populations, often classified as Homo ergaster, date from about 1.8 to 1.3 million years ago and exhibit traits transitional between earlier Homo and later forms.[36] European variants include Homo antecessor, known from fossils dated to around 800,000 years ago at sites like Gran Dolina in Atapuerca, Spain, and Homo cepranensis, represented by a calvarium from Ceprano, Italy, dated to approximately 430,000–385,000 years ago.[37] These descendants highlight the species' morphological variability and adaptability across continents. Key anatomical features of H. erectus include a brain size ranging from 800 to 1,100 cubic centimeters, significantly larger than that of earlier hominins, supporting enhanced cognitive capabilities.[38] Body proportions approached those of modern humans, with elongated legs and narrower pelvises facilitating efficient locomotion.[39] Technologically, H. erectus is associated with the Acheulean tool industry, featuring symmetrical bifacial hand axes that indicate improved planning and dexterity compared to earlier Oldowan tools.[35] Evidence for fire control emerges around 1 million years ago at Wonderwerk Cave in South Africa, where microstratigraphic analysis reveals in situ ash and burned sediments in Acheulean layers, suggesting habitual use for cooking or warmth.[40] The dispersal of H. erectus out of Africa began around 1.8 million years ago, with the earliest evidence outside the continent at Dmanisi, Georgia, where fossils dated to 1.85–1.78 million years ago show a diverse population adapted to varied environments. In Asia, remains such as Java Man from Trinil, Indonesia (approximately 1.6 million years old), and Peking Man from Zhoukoudian, China (around 700,000 years old), demonstrate successful colonization of tropical and temperate zones.[41] European incursions are evidenced by fossils from Atapuerca, including H. antecessor specimens indicating repeated migrations into cooler climates by 800,000 years ago.[42] Adaptations in H. erectus supported its wide-ranging lifestyle, including physiological changes for endurance running, such as an arched foot, long Achilles tendon, and efficient thermoregulation through sweating, enabling persistence hunting over long distances—a capability derived around 2 million years ago. A meat-heavy diet, inferred from cut-marked bones and stable isotope analyses at sites like Olduvai Gorge, likely supplemented scavenging and contributed to larger body sizes and brain growth.[39] Social cooperation is suggested by the morphological diversity at Dmanisi, including individuals with disabilities who survived into adulthood, implying group care and shared resource strategies.[43] Recent discoveries at Ledi-Geraru, Ethiopia, reported in 2025, include Homo fossils dated to approximately 2.78 million years ago, such as a lower premolar (LD 302-23), alongside Australopithecus remains around 2.63 million years old. These findings indicate early Homo lineages coexisting with other hominins before 2.5 million years ago, potentially representing erectus-like forms predating the classic African emergence and challenging timelines for genus diversification.[44]Archaic Humans and Regional Variants
Archaic humans encompass a diverse array of late Middle to early Late Pleistocene Homo species that developed specialized adaptations to the challenging environments of Eurasia, particularly during glacial periods of the ice ages. These regional variants, evolving from earlier Homo erectus descendants, include Homo heidelbergensis, which lived from approximately 700,000 to 200,000 years ago across Europe, Africa, and possibly Asia, characterized by a robust skull with prominent brow ridges, a large face, and body proportions suited to colder climates. Fossils such as the Heidelberg jaw from Germany and the Boxgrove shinbone from England illustrate their tall stature, averaging 1.8 meters, and use of advanced Acheulean tools for hunting large game. In Africa, Homo rhodesiensis, represented by the Kabwe 1 skull dated to around 300,000 years ago, exhibits similar archaic features like a massive brow ridge and brain size exceeding 1,200 cubic centimeters, suggesting it as a potential African counterpart or variant of H. heidelbergensis. The Gawis cranium from Ethiopia, dated to approximately 500,000–200,000 years ago but retaining archaic traits such as a low vault and projecting face, underscores the persistence of primitive morphologies into later periods despite its earlier context.[45] Neanderthals (Homo neanderthalensis), emerging around 400,000 years ago and persisting until approximately 40,000 years ago, represent the most well-known archaic humans, inhabiting Europe and western Asia during repeated ice age cycles. Their robust build, with barrel-shaped chests, short limbs, and large noses for warming cold air, was an adaptation to frigid habitats, as evidenced by fossils from sites like the Sima de los Huesos in Spain, a pit containing over 28 individuals dated to 430,000 years ago and identified as early Neanderthal ancestors through nuclear DNA analysis showing close affinity to later Neanderthals. Neanderthals had average brain volumes of about 1,400 cubic centimeters, larger than modern humans relative to body size, and produced the Mousterian tool industry, featuring prepared-core Levallois techniques for flake tools used in hunting and processing hides. Evidence of intentional burials, such as those at La Chapelle-aux-Saints and Shanidar Cave, indicates ritualistic behavior, while symbolic expressions include perforated eagle talons from Krapina, Croatia, dated to around 130,000 years ago, suggesting jewelry or adornment. A well-preserved hyoid bone from Kebara Cave, Israel, dated to about 60,000 years ago, mirrors the modern human form in size and structure, implying the anatomical capacity for complex vocalizations and possibly language. Denisovans, another key archaic group known primarily from genetic evidence and sparse fossils, occupied Siberia and East Asia from roughly 200,000 to 50,000 years ago, adapting to diverse environments including high-altitude regions like the Tibetan Plateau. Their most notable trait is the contribution of the EPAS1 gene variant to modern Tibetans, enabling efficient oxygen use at elevations over 4,000 meters without excessive red blood cell production, as identified through genomic comparisons of Denisovan DNA from Denisova Cave. This adaptation highlights their resilience in harsh, low-oxygen terrains during Pleistocene climate fluctuations. The extinction of Neanderthals around 40,000 years ago coincided with the arrival of Homo sapiens in Europe and abrupt climate shifts, including the onset of the Last Glacial Maximum, which fragmented habitats and increased resource scarcity. Studies modeling population dynamics indicate that competition for food and territory with incoming modern humans, combined with nutritional stress from cooling temperatures, likely reduced Neanderthal viability without evidence of direct violence or complete replacement. Denisovans similarly vanished by about 50,000 years ago, possibly due to analogous environmental pressures and demographic isolation in Asia.Island and Peripheral Homo Species
Island and peripheral Homo species represent isolated populations of the genus Homo that evolved in geographically constrained environments, such as islands in Southeast Asia and peripheral regions of mainland Eurasia, leading to distinct morphological adaptations distinct from mainland archaic humans.[46] These groups, often exhibiting small body sizes and specialized traits, illustrate evolutionary experiments driven by insular conditions and resource limitations, persisting alongside or after the arrival of Homo sapiens in the region.[47] Homo floresiensis, discovered in Liang Bua cave on Flores Island, Indonesia, in 2004, is the most well-documented example of an island-adapted Homo species. Fossils, including the partial skeleton LB1, date to between approximately 100,000 and 50,000 years ago, indicating survival until relatively recently in human evolutionary history.[47] Key traits include extreme dwarfism, with estimated adult heights of about 1 meter and brain volumes around 400 cubic centimeters, alongside the production of simple stone tools comparable to those of early Homo species like H. habilis. Despite the small brain size, evidence suggests cognitive capabilities sufficient for tool manufacture and possibly fire use.[48] The adaptations of H. floresiensis are attributed to island biogeography, where limited resources and isolation promote insular dwarfism—a phenomenon observed in other large mammals on islands, reducing body size to match scarce food availability.[47] This evolutionary response likely began early in their lineage, with recent analyses indicating small body size originated before 700,000 years ago, possibly from an ancestor like Homo erectus that reached Flores via Wallacea.[46] Debates persist regarding their phylogenetic position: some evidence supports derivation from H. erectus through prolonged isolation and dwarfing, while primitive features in the wrist and feet suggest ancestry from an earlier Homo species predating H. erectus.[48] Their late survival until around 50,000 years ago highlights the role of geographic barriers in preserving isolated lineages. Another island species, Homo luzonensis, was identified from fossils unearthed in Callao Cave on Luzon Island, Philippines, with the third metatarsal initially discovered in 2007 and formally described in 2019.[49] Dated to at least 67,000 years ago and possibly older, these remains include small teeth, hand and foot bones from at least three individuals, indicating a small-bodied hominin adapted to the Philippine archipelago.[49] Notable traits include curved phalanges in the foot, resembling those of arboreal primates and suggesting enhanced climbing abilities for navigating forested island environments.[49] Like H. floresiensis, H. luzonensis exemplifies insular evolution, with potential dwarfism and isolation preventing interbreeding with incoming populations.[46] Potential peripheral populations on mainland Eurasia include the Red Deer Cave people, represented by fossils from Maludong and Longtanshan caves in southern China, dated to around 14,000 years ago.[50] These remains exhibit a mosaic of modern and archaic traits, such as robust brows and large molars alongside gracile features, initially prompting speculation of a late-surviving distinct Homo lineage.[51] However, ancient DNA analysis in 2022 confirmed they were anatomically modern humans (Homo sapiens) with genetic affinities to ancient East Asians and contributions to Native American ancestry, indicating a peripheral modern population retaining archaic morphologies due to isolation or admixture.[50] Recent genetic studies from 2024 and 2025 have highlighted the influence of peripheral archaic lineages on modern Asian populations, revealing "ghost" ancestries through admixture events. For instance, analyses of ancient genomes from Yunnan Province uncovered a basal East Asian lineage contributing to modern Tibetan genetics, suggesting undetected peripheral hominin introgression shaped regional diversity. These findings underscore how isolated Homo groups, beyond well-known species like H. floresiensis, left subtle genetic legacies in contemporary East Asians via interbreeding with early H. sapiens dispersals.[52]Emergence and Dispersal of Homo sapiens
Anatomically modern Homo sapiens first appeared in Africa around 300,000 years ago, with the earliest known fossils discovered at Jebel Irhoud in Morocco, dated to approximately 315,000 years ago through thermoluminescence dating of associated artifacts and sediments.[53] These remains, including a partial skull and jaw, exhibit a mix of modern and archaic features, such as a modern-like facial structure but a more elongated braincase, supporting a pan-African origin for the species rather than a single East African cradle.[53] Subsequent key fossils from eastern Africa, such as those from Omo Kibish in Ethiopia dated to over 233,000 years ago via volcanic ash correlation and uranium-series dating, and the Herto skulls from Ethiopia at 160,000–155,000 years ago, further illustrate the gradual emergence of fully modern traits across the continent.[54] Early dispersals of H. sapiens beyond Africa occurred in multiple waves, though most were unsuccessful or limited in scope. Fossils from Skhul and Qafzeh caves in the Levant, dated to 120,000–90,000 years ago using electron spin resonance and thermoluminescence on associated materials, represent an early exodus around 120,000 years ago that failed to establish lasting populations outside Africa, likely due to climatic barriers and competition with Neanderthals.[55] The successful major dispersal began around 70,000–50,000 years ago via a southern route through the Arabian Peninsula, facilitated by lower sea levels exposing coastal pathways, as evidenced by genetic and archaeological data from sites like Misliya Cave in Israel.[56] This migration wave led to rapid colonization: H. sapiens reached Australia by approximately 65,000 years ago, inferred from dated occupation sites like Madjedbebe rock shelter using optically stimulated luminescence, and the Americas between 20,000 and 15,000 years ago, supported by Clovis-era artifacts and pre-Clovis sites dated via radiocarbon and Bayesian modeling.[57] Distinguishing H. sapiens from archaic predecessors were key anatomical adaptations, including a high, rounded forehead, prominent chin on the mandible, and a gracile (slender) skeleton with reduced robusticity in the limbs and cranium, reflecting adaptations to diverse environments and possibly reduced masticatory stress from dietary shifts.[58] These traits, evident in fossils like those from Jebel Irhoud and Omo, combined with enhanced behavioral flexibility—such as advanced tool use and social cooperation—enabled the species' swift global expansion despite initial small population sizes.[53] A recent 2025 analysis of the ~1-million-year-old Yunxian 2 skull from China has provided new insights into archaic Homo evolution in Asia, suggesting it represents a distinct lineage (H. longi clade) potentially related to Denisovans, while upholding the African origin of H. sapiens.[59]Anatomical and Physiological Adaptations
Development of Bipedalism
Bipedalism, the ability to walk upright on two legs, emerged as a hallmark adaptation in the hominin lineage, marking a pivotal shift from the quadrupedal locomotion of earlier primates. Evidence suggests that facultative bipedalism—capable of both bipedal and arboreal movement—first appeared around 7 million years ago (Mya) in Sahelanthropus tchadensis, based on the anteriorly positioned foramen magnum in its cranium, which indicates a head balanced over the vertebral column for upright posture.[60] By approximately 3.7 Mya, obligate bipedalism—fully committed terrestrial walking—had evolved in Australopithecus afarensis, as evidenced by its fully bipedal lower limb morphology and lack of significant arboreal adaptations.[23] This progression likely responded to environmental pressures, including Miocene climate shifts that expanded open savannas and grasslands, reducing forest cover and favoring terrestrial foraging strategies over arboreal ones.[61] Several key anatomical modifications facilitated the transition to efficient bipedalism. The vertebral column developed an S-shaped curvature, with lumbar lordosis positioning the trunk's center of gravity over the hips for balance during upright gait.[62] The foramen magnum shifted inferiorly beneath the cranium, allowing the head to sit atop the spine and enabling forward-facing gaze without neck strain.[63] The pelvis underwent significant reconfiguration, broadening with flared iliac blades to support abdominal organs and provide leverage for gluteal muscles in hip extension, while the sacrum angled to align the spine with the lower limbs.[63] In the lower extremities, the femur adopted a valgus angle—angled inward from hip to knee—for stability and weight distribution, and the foot evolved a longitudinal arch with an elongated heel and shortened toes, anchored by the Achilles tendon to store elastic energy during strides.[64] These changes collectively transformed the hominin skeleton into a system optimized for sustained, upright locomotion. Bipedalism conferred adaptive advantages that likely drove its fixation in hominin populations. Biomechanical analyses indicate that human-like bipedal walking requires approximately 25% less energy than knuckle-walking quadrupedalism in comparably sized primates, enabling efficient long-distance travel across open terrains. Upright posture also freed the forelimbs for carrying food, tools, or infants during foraging expeditions, enhancing resource acquisition in dispersed savanna environments.[1] Additionally, elevated eye level improved vigilance against predators and for spotting distant resources in grassland habitats.[61] Fossil and experimental evidence robustly supports the antiquity and mechanics of hominin bipedalism. The 3.66-Mya Laetoli footprints in Tanzania preserve clear impressions of bipedal strides, showing heel-to-toe progression, extended hindlimbs, and a compliant gait distinct from chimpanzee locomotion but akin to modern humans.[65] Skeletal remains, such as the A. afarensis "Lucy" specimen, reveal femoral valgus angles of about 9–15 degrees, indicative of convergent knee alignment for bipedal stability, alongside pelvic metrics confirming upright weight-bearing.[23] Biomechanical models, integrating these fossils with musculoskeletal simulations, demonstrate enhanced gait stability and reduced lateral sway in early hominins compared to apes, underscoring the locomotor efficiency gained through these adaptations.[66]Encephalization and Brain Evolution
Encephalization, the evolutionary increase in brain size relative to body size, is a defining feature of hominin evolution, enabling enhanced cognitive capacities. In early hominins like Australopithecus, average endocranial volumes were approximately 400 cubic centimeters (cc), similar to those of great apes.[67] This expanded gradually in early Homo species to around 600 cc, with a more rapid increase during the emergence of Homo erectus around 1.8 million years ago, reaching about 950 cc on average.[67] Neanderthals (Homo neanderthalensis) exhibited even larger brains, averaging 1,415 cc, while modern Homo sapiens have an average of 1,350 cc, reflecting a fourfold increase over the past seven million years.[67] The encephalization quotient (EQ), a measure of relative brain mass, rose from about 2.5 in great apes to 7.5 in humans, underscoring this disproportionate growth.[68] However, the encephalization quotient has faced criticism in comparative primatology; research indicates that absolute brain size often predicts cognitive performance better than EQ in non-human primates,[69] and due to variability in allometric scaling slopes across taxonomic groups and potential biases (e.g., overestimating EQ in small animals and underestimating in large ones), some researchers advocate abandoning EQ in favor of alternative measures such as a cognitive equivalence line for primate cognition.[70] Anatomically, this encephalization involved not just overall size but reorganization, particularly the expansion of the prefrontal cortex, which supports executive functions like planning and decision-making.[71] In humans, the granular prefrontal cortex underwent major enlargement alongside other association areas, altering corticocortical connectivity and contributing to advanced neural processing.[71] These changes are evident in the relative growth of brain mass compared to body size, with hominin brains becoming metabolically costlier, demanding up to 20% of total energy expenditure in modern humans.[72] Several drivers propelled this brain evolution. Dietary shifts played a key role; the expensive tissue hypothesis posits that reductions in gut size, facilitated by higher-quality foods, freed metabolic energy for brain growth.[72] The control of fire and cooking around 1.8 million years ago increased caloric availability from food, supporting larger brains by making nutrients more digestible and reducing chewing time.[73] Access to omega-3 fatty acids, particularly docosahexaenoic acid (DHA) from aquatic or plant sources, provided essential building blocks for neural membranes, correlating with brain size increases in early hominins.[74] Social complexity also drove encephalization; the social brain hypothesis links neocortex expansion to managing larger group sizes, with Dunbar's number estimating stable human social groups at around 150 individuals, necessitating advanced theory-of-mind and alliance-tracking abilities. Recent studies also highlight that greater parental investment, enabling larger newborns, and higher, stable body temperatures synergistically promoted encephalization in endothermic lineages like mammals.[75] Evidence for these changes comes from multiple lines. Endocranial casts, natural molds of the braincase interior, reveal progressive increases in volume and shifts in shape, such as parietal lobe expansion in Homo erectus.[76] Comparative MRI studies of living primates and humans highlight structural homologies and divergences, confirming prefrontal reorganization.[77] Genetically, variants in genes like ASPM and MCPH1, associated with microcephaly when mutated, show signatures of positive selection in the hominin lineage, influencing progenitor cell proliferation and brain size.[78] The consequences of encephalization include the potential for complex cognition, such as abstract thought and symbolic language, arising from expanded neural networks in the prefrontal and temporal lobes.[79] This encephalization co-evolved with enhanced hand dexterity, facilitating complex tool use and cultural developments.[80] However, this came with trade-offs, including a prolonged infancy and extended developmental period to accommodate brain growth, increasing dependency on social caregiving and extending gestation and childhood phases compared to other primates.[81]Reduction in Sexual Dimorphism and Other Traits
One of the notable trends in human evolution is the progressive reduction in sexual dimorphism, particularly in body size, observed from early hominins to modern Homo sapiens. In Australopithecus species, such as A. afarensis around 3-4 million years ago, male-to-female body size ratios were relatively high, estimated at approximately 1.5:1, reflecting substantial differences likely tied to intense male-male competition for mates in a polygynous mating system.[82] This dimorphism decreased markedly over time, reaching about 1.2:1 in Homo sapiens, where males are only moderately larger than females in terms of body mass and stature.[83] The decline is attributed to a shift toward pair-bonding and monogamy, which reduced the selective pressure for large male body sizes driven by intrasexual competition, while promoting greater male investment in offspring care and cooperative behaviors.[84] Accompanying this reduction, several other anatomical traits refined in later hominins contributed to enhanced functionality and adaptation. The jaws and teeth underwent significant reduction, transitioning from the megadontia—large molars and premolars—seen in early hominins to smaller, more efficient dentition in Homo species, with canine size specifically declining around 4 million years ago as evidenced by fossils from Ardipithecus ramidus.[85] This change paralleled a decrease in canine sexual dimorphism, further indicating lessened aggression between males. Additionally, the evolution of ulnar opposition in the hand, where the thumb opposes the ring and little fingers, enabled a precision grip crucial for manipulating objects, emerging prominently in early Homo around 2 million years ago through modifications in thumb length and finger curvature. Similar precision grip capabilities, with human-like thumb-to-finger proportions, are also evident in Paranthropus boisei fossils from around 1.52 million years ago, suggesting broader distribution of advanced manual dexterity among early hominins.[86][87] Humans also developed near-complete hairlessness across the body, coupled with an increase in eccrine sweat glands, facilitating effective thermoregulation via evaporative cooling during prolonged physical activity in open environments.[88] Finally, the descent of the larynx in the vocal tract, unique to humans among primates, lowered its position to allow for a longer pharynx and greater phonetic versatility in speech production.[89] Fossil evidence supports these changes through direct measurements, such as pelvic and femoral dimensions indicating body size ratios in Australopithecus specimens, and comparative dental metrics showing canine reduction across hominin lineages when contrasted with the projecting, sexually dimorphic canines of extant apes like chimpanzees.[90] Hand bones from sites like Olduvai Gorge reveal progressive adaptations for opposition grips, while endocasts and hyoid fossils suggest laryngeal repositioning by at least 300,000 years ago in archaic humans.[91] These traits collectively played a key evolutionary role by fostering enhanced social cooperation, improved biparental infant care through reduced conflict, and greater manual dexterity that supported complex interactions, ultimately aiding the survival and proliferation of Homo sapiens.[92]Behavioral and Cultural Developments
Origins and Evolution of Tool Use
The earliest evidence of intentional tool use by hominins dates to approximately 3.3 million years ago at Lomekwi 3 in West Turkana, Kenya, where stone artifacts produced through percussion—such as flakes, cores, and anvils—suggest rudimentary knapping techniques predating systematic stone tool industries.[93] These tools, associated with Australopithecus afarensis or a related species, indicate early manipulation of stone for sharp edges, though they lack the standardization of later assemblages.[93] The Oldowan industry emerged around 2.6 million years ago, marking the onset of habitual stone tool production linked to early Homo species like Homo habilis.[94] Characterized by simple flaking methods to detach sharp-edged flakes from cobble cores, Oldowan tools served for cutting, scraping, and processing food, with assemblages found across East Africa.[95] At Ledi-Geraru in Ethiopia, artifacts dated to over 2.58 million years ago represent some of the earliest confirmed Oldowan examples, highlighting technological diversity and continuity in tool-making traditions.[94] Recent analyses from 2024 and 2025, including environmental context studies in the Turkana Basin, suggest Oldowan-like tool use may extend back to around 2.75 million years ago, reflecting adaptive responses to climatic shifts.[96] By approximately 1.7 million years ago, the Acheulean industry succeeded the Oldowan, primarily associated with Homo erectus and its descendants, and persisted until about 250,000 years ago.[97] This technological stage introduced bifacial handaxes and cleavers, requiring more sophisticated flaking techniques to shape both sides of a core into symmetrical forms, which demanded foresight and sequential planning.[97] The symmetry observed in many Acheulean bifaces implies advanced spatial cognition and motor control, as hominins imposed standardized shapes beyond functional necessity.[97] Key sites like Gesher Benot Ya'aqov in Israel, dated to 790,000 years ago, provide evidence of wooden artifacts alongside stone artifacts, such as a polished plank, demonstrating material diversity in Acheulean technology.[98] Fire-altered materials also appear in Acheulean contexts, such as thermally modified flints at Gesher Benot Ya'aqov, suggesting controlled heating to improve knappability or hafting adhesives.[99] These composite tools reflect enhanced planning and resource integration. The Mousterian industry, spanning roughly 300,000 to 40,000 years ago and primarily linked to Neanderthals, built on earlier traditions with the Levallois technique—a prepared-core method for producing predetermined flake shapes.[100] This allowed for versatile tools like scrapers and points, often hafted, and indicates refined flaking precision.[100] By around 100,000 years ago, evidence of pigments like red ochre in tool kits, such as at sites in the Levant, points to their use in processing hides or adhesives, extending functional applications.[101] Cognitive advancements underpinned these developments, with Acheulean biface production requiring mental templates for symmetry and multi-step sequences that fostered planning abilities.[95] Social transmission played a crucial role, as experimental studies show that imitation and emulation—rather than individual invention—sustained complex knapping skills across generations in early hominins.[95] The evolution of dexterous grips in hominin hands further supported these manipulative demands.[102]Transition to Behavioral Modernity
The transition to behavioral modernity in Homo sapiens, often termed the "Upper Paleolithic Revolution," represents a pivotal shift toward complex symbolic cognition, including abstract art, personal adornment, and ritual practices, emerging primarily between 100,000 and 50,000 years ago. This period marks the onset of behaviors that enabled advanced social organization and cultural transmission, distinguishing early modern humans from preceding hominins. While the most dramatic evidence appears in the European Upper Paleolithic around 50,000 years ago, foundational signs originated in Africa much earlier, suggesting a gradual rather than abrupt development. Archaeological findings from Blombos Cave in South Africa reveal some of the earliest indicators of symbolic thought, including engraved ochre plaques and abstract geometric patterns dated to approximately 100,000–75,000 years ago, interpreted as deliberate non-utilitarian markings possibly linked to identity or information encoding. Similarly, perforated shell beads from the same site and nearby locations, dated to about 75,000 years ago, indicate personal ornamentation and potential social signaling, as these Nassarius shells were sourced from distant coastal areas and show wear consistent with prolonged use as jewelry. Ochre processing, evidenced by grinding tools and pigment residues at multiple African sites from 100,000 years ago, further supports ritualistic or decorative applications, such as body painting, which may have reinforced group cohesion. By around 50,000–40,000 years ago, as Homo sapiens dispersed from Africa into Eurasia, behavioral modernity manifested in more elaborate forms. Cave art in Chauvet Cave, France, featuring vivid depictions of animals and hand stencils dated to approximately 36,000 years ago, demonstrates sophisticated aesthetic expression and possibly narrative or shamanistic intent. Portable art objects, like the ivory Venus of Hohle Fels figurine from southwestern Germany, dated to about 40,000 years ago, represent stylized human forms and highlight emerging symbolic representation of the body and fertility. Musical instruments, including bone flutes from sites like Geissenklösterle Cave in Germany (around 40,000 years ago), crafted from bird bones with precisely drilled finger holes, suggest aesthetic pursuits and communal activities that could have facilitated language and social bonding. Intentional burials with grave goods, such as those at Sungir, Russia, around 34,000 years ago—where a child and adult were interred with thousands of mammoth ivory beads and spears—imply beliefs in an afterlife, status differentiation, or ritual commemoration. Several factors likely drove this transition. Genetic adaptations, notably variants in the FOXP2 gene associated with orofacial motor control and vocal learning, may have enhanced articulatory skills crucial for complex language, with human-specific changes arising around 300,000–400,000 years ago.[103] Demographic pressures, including a severe population bottleneck approximately 70,000 years ago—potentially triggered by environmental events like the Toba supervolcano eruption—could have intensified selection for cognitive flexibility and innovation. Ecological challenges during the Out of Africa migrations, starting around 70,000–60,000 years ago, such as adapting to diverse climates and competing with other hominins, probably accelerated the adoption of symbolic behaviors for cooperation and knowledge sharing. While Homo sapiens led this cultural florescence, debates persist over Neanderthal capabilities, with limited evidence of overlapping symbolic acts like ochre use and possible art in Europe around 60,000–40,000 years ago, though lacking the diversity seen in sapiens assemblages. The global spread of behavioral modernity accompanied sapiens migrations, fostering region-specific traditions: for instance, rock art in Australia by 45,000 years ago and engraved plaques in Asia by 40,000 years ago, which diversified into varied symbolic systems worldwide.Recent and Ongoing Human Evolution
The Neolithic Revolution, commencing around 12,000 years ago, transitioned human societies from foraging to agriculture, fostering biological adaptations to altered diets, denser populations, and new pathogens, with evolutionary pressures extending through the Industrial era into the present. Genetic studies indicate that human adaptive evolution has accelerated significantly during the Holocene, particularly in the last 10,000 years following the advent of agriculture, with rates of beneficial genetic changes estimated to be more than 100 times higher than in earlier periods due to population growth and novel selective pressures.[104] Human evolution among modern populations has proceeded in parallel across groups since the dispersal of Homo sapiens from Africa, with no ethnic or racial group being evolutionarily "newer" than others; all contemporary humans trace descent from ancestors that migrated out of Africa approximately 60,000–70,000 years ago, rendering all groups equally recent evolutionarily.[105] This period has seen accelerated natural selection on traits enhancing survival in farming communities and migratory contexts, as populations expanded and encountered novel environments.[106] Key adaptations include lactase persistence, enabling adult digestion of milk sugars, which arose independently in European pastoralists around 7,500 years ago and in East African herders approximately 3,000–7,000 years ago, driven by the selective advantage of dairy consumption.[107] High-altitude tolerance among Tibetans involves variants in the EPAS1 gene, introgressed from Denisovans around 40,000 years ago but under recent positive selection to regulate hemoglobin levels and prevent maladaptive polycythemia in hypoxic conditions.[108] Disease resistance exemplifies balancing selection, as the sickle cell allele (HbS) in heterozygous form confers protection against severe malaria in sub-Saharan African populations, with its frequency rising post-agriculture due to increased mosquito exposure in settled communities.[106] Agriculture-induced genetic changes encompass mandibular reduction, where softer, processed foods led to smaller jaws and altered craniofacial morphology over the last 10,000 years, as evidenced by comparative analyses of prehistoric skeletons showing decreased robusticity and increased impaction risks.[109] Human stature initially declined by up to 10–13 cm following the Neolithic shift due to nutritional deficits from carbohydrate-heavy diets, but polygenic scores from ancient DNA indicate partial genetic recovery and selection favoring taller variants in post-medieval European populations amid improved nutrition and socioeconomic changes.[110] A 2025 study revealed recent positive selection on a Denisovan-derived MUC19 haplotype, featuring expanded repeat units that enhance mucin production for mucosal immunity, with elevated frequencies in ancient Siberian and modern Indigenous American genomes, suggesting adaptation to cold climates or novel pathogens during migrations.[111] Ancient DNA from over 5,000-year-old remains demonstrates dynamic evolution, including allele frequency sweeps for immune and metabolic traits in the last 5,000 years, as seen in Eurasian samples showing selection bursts tied to plagues and dietary shifts.[112] Genome-wide association studies (GWAS) corroborate this by identifying recent selection signals on height-related loci, integrating ancient and modern data to reveal accelerated change in polygenic traits since the Bronze Age.[113] Contemporary factors like urbanization and medicine modulate selection: dense cities amplify pathogen exposure, potentially favoring immune alleles, while interventions reduce mortality from genetic disorders, relaxing selection on some traits but not halting evolution entirely.[114] The COVID-19 pandemic underscored ongoing adaptation, with GWAS revealing human genetic variants influencing severity—such as those in interferon pathways—under potential selection in exposed populations, highlighting persistent evolutionary responses to infectious pressures.[115]Evidence Supporting Human Evolution
Fossil and Archaeological Record
The fossil and archaeological record provides the primary tangible evidence for human evolution, consisting of preserved hominin bones, teeth, and associated artifacts that span from approximately 7 million years ago to the recent past. These remains, often fragmentary due to geological processes, offer insights into morphological changes, geographic dispersal, and behavioral adaptations of early hominins. Key discoveries have been concentrated in Africa, with later expansions documented in Eurasia and beyond, revealing a progression from arboreal apes to fully bipedal, tool-using species. Major fossil sites in the East African Rift Valley, such as Olduvai Gorge in Tanzania and Hadar in Ethiopia, have yielded some of the earliest hominin specimens and artifacts. Olduvai Gorge, a rift valley basin with layered volcanic sediments dating between 1.8 and 2.6 million years ago, contains fossils of Australopithecus boisei and early Homo species alongside Oldowan stone tools, indicating scavenging and basic processing of animal remains.[116] At Hadar, the 3.2-million-year-old skeleton of Australopithecus afarensis, known as "Lucy," demonstrates partial bipedalism through pelvic and limb proportions adapted for upright walking. Outside Africa, the Dmanisi site in Georgia preserves skulls and postcranial bones of early Homo erectus dated to about 1.8 million years ago, representing the earliest evidence of hominin migration from Africa with primitive brain sizes around 600 cubic centimeters.[117] Denisova Cave in Siberia has produced finger bones and teeth attributed to Denisovans, a sister group to Neanderthals, with layers indicating occupation from at least 200,000 years ago and stone tools suggesting repeated use over millennia.[118] More recently, the Rising Star Cave in South Africa yielded over 1,500 Homo naledi fossils in 2013, with uranium-thorium dating in 2023 confirming ages between 236,000 and 335,000 years ago, highlighting mosaic evolution in a small-brained species with modern-like hand bones.[119] Hominin fossils include diverse cranial and postcranial elements that illustrate evolutionary transitions. Cranial remains, such as the Taung Child—a juvenile Australopithecus africanus skull discovered in 1924 in South Africa and dated to around 2.8 million years ago—reveal a mix of ape-like face and human-like braincase positioning, supporting bipedal posture.[120] Postcranial fossils, like the partial cranium KNM-ER 1470 from Koobi Fora, Kenya, dated to approximately 1.9 million years ago, exhibit a large brain capacity of about 750 cubic centimeters and robust facial features associated with early Homo habilis or rudolfensis.[121] These specimens are dated using radiometric techniques, such as potassium-argon and argon-argon methods for volcanic layers, which provide absolute ages by measuring radioactive decay, alongside relative stratigraphy that sequences layers based on superposition.[122] Uranium-series dating, applied to cave deposits like those at Rising Star, complements these by analyzing thorium-uranium ratios in carbonates.[119] Archaeological evidence complements fossils through tool assemblages and activity sites that infer behavior. Early stone tools from Olduvai, including choppers and flakes from 2.6 million years ago, represent the Oldowan industry for cutting and pounding.[116] Later Acheulean hand axes, bifacially worked and symmetrical, appear around 1.7 million years ago at sites like Dmanisi. Hearths at Qesem Cave in Israel, dated to 300,000–400,000 years ago, show repeated burning of wood and bone, with charred remains indicating controlled fire use for cooking and social gathering among Middle Pleistocene hominins.[123] Upper Paleolithic sites like Lascaux Cave in France preserve parietal art, including over 600 painted animals from about 17,000 years ago, alongside tools and hearths that suggest symbolic thinking and communal rituals.[124] Despite these riches, the fossil record faces challenges from taphonomy—the processes of decay, burial, and preservation—that bias recovery toward durable bones in stable environments like caves or riverbeds, while soft tissues and open-plain remains rarely survive. Recent discoveries highlight ongoing revisions: a crushed cranium from central China, unearthed in 1990 and redated in 2025 to approximately 1 million years ago using digital reconstruction and stratigraphic analysis, which some researchers controversially suggest indicates earlier Homo sapiens emergence in Asia, challenging African-centric timelines, though this remains debated.[125] At Ledi-Geraru in Ethiopia, fossils dated to 2.5–2.8 million years ago include jaw fragments marking a transition from Australopithecus to early Homo, with a 2025 find of new Australopithecus teeth underscoring dietary shifts.[126] Gaps persist, particularly before 7 million years ago, where only sparse finds like Sahelanthropus tchadensis crania from Chad provide tentative evidence of the last common ancestor with chimpanzees, limited by poor preservation in forested tropics. Island contexts show scarcity of hominin fossils, with isolated examples like Homo floresiensis on Flores dated to 700,000 years ago, attributed to sea barriers and erosion, though 2025 evidence from Sulawesi pushes island occupation back over 800,000 years.[127]Genetic and Molecular Evidence
Genetic and molecular evidence provides a direct window into human evolutionary history by analyzing DNA sequences from modern and ancient samples, revealing timelines of divergence, population dynamics, and adaptive pressures. Key methods include the study of mitochondrial DNA (mtDNA), which is inherited solely from the mother and accumulates mutations at a relatively constant rate, allowing reconstruction of maternal lineages. Analysis of mtDNA from diverse global populations indicates that all modern humans descend from a common African ancestor, termed Mitochondrial Eve, who lived approximately 150,000 to 200,000 years ago.[128] Similarly, the non-recombining portion of the Y-chromosome traces paternal lineages to Y-chromosomal Adam, estimated to have lived between 120,000 and 200,000 years ago, also in Africa, highlighting the African origin of Homo sapiens.[129] These uniparental markers offer simplified views of ancestry but are complemented by whole-genome sequencing, which captures broader genetic variation; a landmark achievement was the 2010 sequencing of the Neanderthal genome, enabling comparisons of genetic similarities and differences with modern humans.[130] Major findings from genomic data pinpoint key evolutionary events, such as the divergence between human and chimpanzee lineages around 6 million years ago, based on comparisons of orthologous genes and mutation rates across primate genomes.[131] Within human history, evidence of population bottlenecks and expansions is evident in fluctuations of effective population size (Ne), which averaged about 10,000 individuals over the past million years but dipped dramatically during events like the ~70,000-year-ago bottleneck possibly linked to the Toba supervolcano eruption, reducing Ne to as low as 1,000-10,000 breeding individuals and leaving a signature of reduced genetic diversity.[132] These fluctuations are inferred from linkage disequilibrium patterns and coalescent models in whole-genome data, showing periods of contraction followed by expansions that shaped current human genetic variation.[133] Molecular evidence also illuminates adaptive evolution, particularly in genes under positive selection. For instance, the SLC24A5 gene variant associated with lighter skin pigmentation arose approximately 22,000–28,000 years ago, likely in West Eurasia, and swept to high frequencies in European and some Asian populations after migrations out of Africa around 40,000-60,000 years ago, likely as an adaptation to lower UV radiation for vitamin D synthesis.[134] Immune-related genes, such as those in the human leukocyte antigen (HLA) system, exhibit extraordinary diversity—over 7,000 alleles across HLA loci—maintained by balancing selection to recognize diverse pathogens, with higher heterozygosity in populations exposed to varied infectious pressures throughout human dispersal.[135] Recent studies underscore ongoing evolution in the Holocene and later periods. A 2025 analysis of ancient genomes revealed selection on height-related genes, such as those influencing skeletal traits, with polygenic shifts in European populations over the last 10,000 years reflecting adaptations to diet and environment post-agriculture.[136] Similarly, a 2025 study identified the MUC19 gene, carrying a Denisovan-derived haplotype in some modern humans (e.g., up to 30% frequency in Mexican populations), which enhances mucosal immunity against oral pathogens, aiding adaptation to new environments like the Americas through recurrent introgression events.[111] Despite these advances, genetic evidence has limitations. Ancient DNA (aDNA) degrades over time due to environmental factors like temperature and humidity, often yielding fragmented sequences that require sophisticated error-correction in sequencing, limiting recovery from older (>100,000-year-old) or warm-climate samples.[137] Additionally, sampling biases persist, with African genetic diversity underrepresented in databases because fewer aDNA samples have been retrieved from the continent due to poor preservation conditions, potentially skewing inferences about early human evolution.[138]Evidence of Interbreeding and Gene Flow
Genetic evidence reveals that modern humans interbred with Neanderthals and Denisovans after migrating out of Africa, introducing segments of archaic DNA into the Homo sapiens genome. Non-African populations carry approximately 1-2% Neanderthal ancestry, resulting from interbreeding events estimated between 50,000 and 60,000 years ago.[139] Similarly, Denisovan admixture occurred around 50,000 years ago, contributing up to 4-6% of the genome in populations from Oceania, such as Melanesians and Aboriginal Australians.[140] These introgressed sequences have been identified through comparative genomics, showing distinct haplotypes shared exclusively between archaic and modern human genomes. Methods like admixture mapping and analysis of linkage disequilibrium (LD) have been crucial in detecting and dating these gene flow events. Admixture mapping identifies regions of the genome with elevated archaic ancestry by comparing allele frequencies across populations, while LD patterns—where archaic segments show reduced recombination due to their recent introduction—help estimate the timing of interbreeding.[141] For instance, ancient DNA from the Oase 1 fossil, dated to about 40,000 years ago in Romania, reveals 6-9% Neanderthal ancestry, indicating a recent admixture event within four to six generations of the individual's life. A 2025 study further suggests additional archaic inputs from unidentified lineages, identifying adaptive introgression in reproductive genes that may have influenced modern human fertility and development.[142] Specific introgressed genes provide concrete examples of archaic contributions. The MC1R gene variant associated with red hair and pale skin in Europeans traces its origin to Neanderthals, where the Val92Met mutation likely aided adaptation to lower UV environments. From Denisovans, the EPAS1 haplotype in Tibetans regulates hypoxia response, enabling efficient oxygen use at high altitudes and conferring a survival advantage in low-oxygen conditions.[143] These beneficial effects extend to immunity and metabolism; for example, Neanderthal alleles in TLR genes enhance immune responses but may increase allergy risks, while others improve lipid metabolism.[144] Conversely, some Neanderthal variants have deleterious impacts, such as increased susceptibility to depression and nicotine addiction in modern populations. Debates persist regarding the extent and directionality of gene flow, particularly in Africa. Evidence points to "ghost" archaic admixture, where an unidentified hominin contributed about 2% of ancestry to West African populations, detected through divergent haplotype scans.[145] Recent analyses indicate bidirectional gene flow, with modern humans contributing ancestry back to Neanderthals, complicating models of unidirectional introgression.[139] Overall, while many archaic alleles were purged due to negative selection, surviving segments highlight how interbreeding enriched human genetic diversity and adaptability.[146]History of Research on Human Evolution
Pre-Darwinian and Early Ideas
In ancient traditions, explanations of human origins were predominantly rooted in religious and mythological narratives. The Hebrew Bible, particularly in the Book of Genesis, describes the creation of the first humans, Adam and Eve, by God from dust and rib, respectively, establishing a monotheistic account of divine origin without reference to prior forms or gradual development.[147] Similarly, Greek philosophers like Aristotle (384–322 BCE) conceptualized a hierarchical scala naturae, or great chain of being, positioning humans at the pinnacle of a fixed, continuous ladder of nature that extended from inanimate matter through plants and animals to rational souls, implying an eternal and unchanging order rather than historical transformation.[148] During the 18th century, Enlightenment thinkers built upon these ancient frameworks while advancing systematic classification. Carl Linnaeus, in the 10th edition of Systema Naturae (1758), classified humans as Homo sapiens within the order Primates, alongside apes and monkeys, marking a significant acknowledgment of anatomical similarities but still within a static hierarchy influenced by the great chain of being, where species were viewed as divinely fixed archetypes.[149] This period emphasized the fixity of species, with naturalists like Georges-Louis Leclerc, Comte de Buffon, initially rejecting notions of transformation and attributing variations to environmental influences on immutable forms, reflecting a broader cultural resistance to ideas of change that challenged theological and philosophical commitments to a stable natural order.[148] Into the early 19th century, debates intensified between monogenism, which posited a single human origin from Adam and Eve as supported by scripture, and polygenism, which argued for multiple independent creations of human races. Swiss-American naturalist Louis Agassiz became a prominent advocate for polygenism after 1846, proposing "zones of creation" where distinct races emerged separately in different geographic regions, using cranial measurements to justify racial hierarchies and influencing defenses of slavery by portraying non-European groups as inherently inferior species-like variants.[150] Meanwhile, Jean-Baptiste Lamarck's Philosophie Zoologique (1809) introduced the concept of inheritance of acquired characteristics, suggesting that environmental pressures could drive adaptive changes—such as upright posture in humans through habitual use—but framed these within a progressive, non-Darwinian transformism lacking natural selection or common descent.[151] Early fossil discoveries further highlighted the era's interpretive challenges, often leading to misattributions without an evolutionary context. In 1829, Belgian physician Philippe-Charles Schmerling unearthed the partial skull of a child (Engis 2) in a cave near Liège, Belgium, alongside modern human remains; while recognized as ancient, its robust features puzzled contemporaries, who cataloged it as evidence of prehistoric humans without linking it to ape-like ancestors or species change, simply storing it away amid a lack of theoretical framework.[152] These pre-Darwinian ideas, dominated by typological classifications and divine fixity, offered no mechanism for descent with modification, prioritizing hierarchical order and separate creations over shared ancestry or gradual evolution.[148]Darwin's Contributions and Initial Fossil Finds
Charles Darwin's On the Origin of Species (1859) laid the foundational principles of evolution by natural selection, with implications for human origins that he cautiously extended in later works, arguing that the same mechanisms shaping other species applied to humanity without providing direct evidence at the time. In this seminal text, Darwin emphasized descent with modification through natural selection, suggesting that humans, like all organisms, shared a common ancestry, though he deferred detailed discussion of human evolution to avoid controversy.[153] Darwin addressed human evolution explicitly in The Descent of Man, and Selection in Relation to Sex (1871), proposing that humans descended from ape-like ancestors in Africa, based on comparative anatomy, embryology, and behavioral similarities between humans and primates such as chimpanzees and gorillas.[154] He introduced sexual selection as a key mechanism alongside natural selection, explaining traits like human facial hair, beards, and differences in secondary sexual characteristics as products of mate choice rather than survival advantages.[154] This work positioned humans within the primate lineage, rejecting special creation and emphasizing continuity with the animal kingdom. Initial fossil discoveries provided empirical support for Darwin's ideas shortly before and after his publications. In 1856, workers in the Feldhofer Cave in Germany's Neander Valley unearthed a partial skeleton, including a skullcap, ribs, and limb bones, which anatomist Johann Carl Fuhlrott identified as an ancient human form distinct from modern Europeans, marking the first recognition of a prehistoric hominin.[155] Though predating Darwin's major works, this "Neanderthal" specimen fueled debates on human antiquity and was later classified as Homo neanderthalensis in 1864. In 1891–1892, Dutch anatomist Eugène Dubois excavated a skullcap, thighbone, and tooth at Trinil on Java, Indonesia, naming the find Pithecanthropus erectus (now Homo erectus), interpreted as a "missing link" between apes and humans due to its upright posture and brain size intermediate between apes and modern humans.[156] Darwin's theories elicited strong reactions from contemporaries. Thomas Henry Huxley, in Evidence as to Man's Place in Nature (1863), defended the human-ape connection using anatomical evidence from primates and fossils, arguing that structural similarities in skeletons and brains placed humans firmly in the natural order without invoking divine intervention.[157] Ernst Haeckel, in Generelle Morphologie der Organismen (1866), constructed the first comprehensive phylogenetic trees incorporating humans as a branch of primate evolution, popularizing Darwin's ideas through detailed illustrations of descent from a common ancestor.[158] Debates persisted into the early 20th century, exemplified by the Piltdown Man hoax, where fragments of a skull and jaw, "discovered" in 1912 in England and promoted as an early human ancestor (Eoanthropus dawsoni), combined a modern human cranium with an orangutan jaw stained to appear ancient, deceiving scientists for decades until fluorine dating and microscopic analysis exposed it as a forgery in 1953.[159] Despite such setbacks, Darwin's framework gained widespread scientific acceptance by the early 1900s, shifting paleoanthropology from theological interpretations toward evidence-based natural selection as the driver of human origins.[160] This transition marked a profound move from creationist views to a scientific consensus on human evolution, influencing subsequent fossil hunts and theoretical developments.[161]20th-Century Discoveries and Debates
The 20th century marked a transformative era in paleoanthropology, shifting the focus of human origins research from Eurocentric speculations to robust African evidence through groundbreaking fossil discoveries and methodological innovations. In 1924, anatomist Raymond Dart identified the Taung Child, a juvenile skull from a limestone quarry in South Africa, as a new species, Australopithecus africanus, arguing it represented an early bipedal human ancestor rather than an ape.[162] This find, dated later to approximately 2.8 million years ago, challenged prevailing views that human evolution occurred in Asia or Europe, but faced significant skepticism and rejection by the scientific community for over two decades due to its small brain size and perceived ape-like features.[163] Dart's persistence, supported by subsequent South African fossils like those from Sterkfontein in the 1930s and 1940s, gradually validated A. africanus as a key hominin, emphasizing Africa's central role in human ancestry.[164] The exposure of the Piltdown Man hoax in 1953 further cleared the path for genuine African evidence, as chemical analyses revealed the 1912 "fossil" from England—a composite of a modern human cranium and an orangutan jaw stained to appear ancient—had misled researchers for four decades by suggesting a large-brained European origin for humanity.[165] This scandal, confirmed through fluorine dating and microscopy by Joseph Weiner, Wilfrid Le Gros Clark, and Kenneth Oakley, discredited linear evolutionary models favoring Asian or European primacy and redirected attention to East Africa.[166] Concurrently, Sherwood Washburn's advocacy for a "new physical anthropology" in the 1950s integrated evolutionary biology, primatology, and functional anatomy, moving the field beyond racial typology toward studying adaptation and behavior in living primates to interpret fossils. Mid-century expeditions in East Africa revolutionized the timeline of human evolution. Louis and Mary Leakey's systematic excavations at Olduvai Gorge, Tanzania, beginning in the 1930s but yielding major results in the 1950s and 1960s, uncovered stone tools and fossils associated with early hominins. The introduction of potassium-argon (K-Ar) dating in the early 1960s, pioneered by geochemists like Jack Evernden and Garniss Curtis, allowed precise absolute dating of volcanic layers, establishing Olduvai's Bed I at about 1.8 million years old and confirming the association of tools with hominin remains. In 1964, the Leakeys, with Phillip Tobias and John Napier, described Homo habilis ("handy man") from Olduvai specimens, including a cranium (OH 7) and hand bones (OH 7 and OH 8), positioning it as an early tool-using member of the genus Homo dated to around 1.8–2.0 million years ago.[167] This discovery fueled debates on whether hominin evolution followed a linear progression from australopiths to Homo or a "bushy" pattern with multiple coexisting species, as H. habilis overlapped temporally with more robust australopiths like Paranthropus boisei (formerly Zinjanthropus, found in 1959 at Olduvai).[23] The 1970s brought further milestones with Donald Johanson's Hadar expeditions in Ethiopia, where in 1974 his team discovered "Lucy," a 3.2-million-year-old partial skeleton (AL 288-1) of Australopithecus afarensis, comprising over 40% of an adult female's bones and demonstrating bipedal adaptations like a curved toe and angled femur.[24] Published in 1976 and fully described in 1978, this find extended the bipedal record back to at least 3.9 million years and supported a bushier phylogeny, as A. afarensis predated and likely gave rise to later australopiths and early Homo. K-Ar dating of Hadar tuffs confirmed these ages, with refinements in the 1970s improving accuracy for volcanic contexts.[168] Theoretical debates intensified in the 1960s–1980s, reshaping models of Homo sapiens origins. Franz Weidenreich's multiregional hypothesis, building on his 1940s work with Homo erectus fossils from China (reclassified from Sinanthropus to H. erectus in the 1940s), proposed continuous evolution across Eurasia and Africa through gene flow, with regional continuity from archaic to modern forms.[169] Proponents like Milford Wolpoff argued in the 1980s that H. erectus populations dispersed widely around 1–2 million years ago, evolving locally into modern humans via reticulate evolution, countering isolationist views.[170] In contrast, William Howells and Chris Stringer advocated a "recent African origin" or Out of Africa model, citing fossil morphology and emerging genetic data to suggest modern H. sapiens replaced archaic forms outside Africa around 100,000–200,000 years ago with minimal interbreeding. These debates, exemplified in the 1989 volume The Human Revolution, highlighted tensions between fossil regionalism and African-centered replacement, setting the stage for later genetic resolutions while underscoring the shift from Piltdown-era myths to evidence-based paleoanthropology.Genetic Revolution and Contemporary Advances
The genetic revolution in human evolution studies began in the late 1980s with the pioneering analysis of mitochondrial DNA (mtDNA), which provided the first molecular evidence for a recent African origin of modern humans. In 1987, Rebecca Cann, Mark Stoneking, and Allan Wilson sequenced mtDNA from 147 individuals across diverse populations, constructing a phylogenetic tree that traced all contemporary human lineages to a common African ancestor approximately 200,000 years ago, often termed "Mitochondrial Eve."[171] This work shifted the field from reliance on fossils alone to integrating molecular clocks, resolving debates over multiregional versus recent African origins in favor of the latter.[171] A major milestone came in 2010 with the sequencing of the Neanderthal genome by Svante Pääbo's team at the Max Planck Institute for Evolutionary Anthropology, marking the first high-coverage archaic hominin genome. Using DNA extracted from three Neanderthal bones dated 38,000–80,000 years old, they generated over 4 billion nucleotides, enabling comparisons with modern human genomes. This effort revealed that non-African modern humans carry 1–4% Neanderthal ancestry, confirming interbreeding events between Homo sapiens and Neanderthals shortly after the former's migration out of Africa around 60,000–80,000 years ago. The discovery revolutionized understanding of gene flow, showing that archaic admixture contributed adaptive alleles, such as those enhancing immune responses and skin pigmentation. The 2010s witnessed an ancient DNA (aDNA) boom, driven by advancements in high-throughput sequencing, which allowed recovery of genetic material from thousands of prehistoric samples previously deemed unviable due to degradation. Techniques like shotgun sequencing and targeted capture enabled the analysis of over 5,000 ancient human genomes by 2020, illuminating population dynamics, migrations, and admixture events across Eurasia and the Americas.[172] This surge confirmed the Out of Africa model, with genetic diversity peaking in Africa and declining outward, consistent with serial founder effects during dispersals starting ~200,000 years ago.[173] It also uncovered severe population bottlenecks in Homo sapiens, including a drastic reduction to ~1,280 breeding individuals around 930,000 years ago, inferred from low genetic diversity in modern genomes, potentially linked to climate shifts rather than the debated Toba supervolcano eruption ~74,000 years ago.[174] Recent advances have further refined this narrative through novel genomic discoveries. In November 2024, researchers proposed Homo juluensis as a new eastern Asian hominin species encompassing Denisovans and fossils from sites in China, Tibet, Taiwan, and Laos, dated 300,000–50,000 years ago, based on shared morphological and genetic traits indicating a distinct lineage with sapiens interbreeding.[175] By March 2025, a University of Cambridge study using coalescent modeling of modern genomes revealed "lost" ancestral lineages, showing that Homo sapiens arose from the admixture of at least two deeply diverged African populations that separated ~900,000 years ago and reconnected ~300,000 years ago, contributing up to 20% of contemporary genetic variation and influencing cognitive traits.[176] In August 2025, analysis of the MUC19 gene demonstrated recurrent introgression from Denisovans via Neanderthals, with the variant persisting at high frequencies (up to 33%) in Indigenous American-descended populations, likely aiding adaptation to high-altitude and arid environments through enhanced mucus production for pathogen resistance.[111] In November 2025, additional studies advanced understanding of archaic influences and evolutionary mechanisms. Research published on November 16 indicated that ancient lead exposure may have provided neurological advantages to early humans over Neanderthals, subtly shaping cognitive evolution through environmental selection.[177] On November 18, a University of Michigan study challenged the 60-year-old neutral theory of molecular evolution (Kimura, 1968), demonstrating that non-neutral processes drive much of genetic variation in human lineages, with implications for interpreting adaptation rates.[178] Also on November 17, George Washington University researchers documented consistent stone tool use among early Homo populations spanning over 300,000 years in Africa, based on standardized artifact analyses, reinforcing gradual behavioral continuity rather than abrupt shifts.[179] Methodological innovations have been crucial to these insights, particularly next-generation sequencing (NGS), which handles fragmented aDNA by massively parallelizing short-read assembly, achieving >30× coverage even from sub-milligram samples.[180] For highly degraded remains where DNA yields are low, proteomics complements genomics by sequencing ancient proteins via mass spectrometry, preserving evolutionary signals up to 1.5 million years old and enabling species identification without genetic material, as in analyses of dental enamel from Middle Pleistocene hominins.[181] Integration of these approaches with fossils has advanced, exemplified by 2025 correlations at Ledi-Geraru, Ethiopia, where proteomic profiles from 2.8-million-year-old Homo teeth align with genomic models of early diversification, bridging molecular and morphological data. Despite these triumphs, challenges persist, including ethical concerns over sampling indigenous ancestors without consent, prompting guidelines for community engagement and data repatriation to respect cultural sovereignty.[182] Sampling biases, such as overrepresentation of European sites and undercoverage of tropical regions due to poor preservation, skew reconstructions of global diversity and admixture.[183] Future directions include AI-driven phylogenetics, where machine learning models like Phyloformer accelerate tree inference from vast datasets, potentially resolving complex admixture graphs and predicting undiscovered lineages by 2030.[184]Catalog of Human Species
List of Recognized Hominin Species
The hominin fossil record includes a series of recognized species that document the evolutionary lineage leading to modern humans, based on morphological, chronological, and genetic evidence evaluated under standards from paleoanthropological consensus, such as those outlined in recent taxonomic reviews using the unified species concept.[185] These species are accepted by most experts as distinct taxa, with dates calibrated via radiometric methods and traits assessed from key specimens. The following catalog presents them chronologically, focusing on estimated time ranges, average brain sizes (in cubic centimeters, cc), evidence of tool use, and primary geographic distributions.| Species | Time Range (Mya unless noted) | Brain Size (cc) | Key Traits and Tools | Primary Locations |
|---|---|---|---|---|
| Sahelanthropus tchadensis | 7.0–6.0 | ~350 | Possible bipedalism indicated by anteriorly positioned foramen magnum; no evidence of tool use.[60] | Central Africa (Chad) |
| Orrorin tugenensis | 6.0–5.8 | Unknown (small, chimp-like) | Bipedalism suggested by femur morphology with thickened cortical bone; no tool use.[60] | East Africa (Kenya) |
| Ardipithecus kadabba | 5.8–5.2 | Unknown (small) | Facultative bipedalism from toe bone curvature; no tool use.[60] | East Africa (Ethiopia) |
| Ardipithecus ramidus | 4.4–4.5 | 300–350 | Facultative bipedalism with arboreal adaptations; no advanced tools.[185] | East Africa (Ethiopia) |
| Australopithecus anamensis | 4.2–3.8 | ~400 | Fully bipedal with robust limbs; no direct tool association.[186] | East Africa (Kenya, Ethiopia) |
| Australopithecus afarensis | 3.9–2.9 | 400–500 | Bipedal locomotion with some arboreal features; possible simple tool use (debated).[185] | East Africa (Ethiopia, Tanzania) |
| Australopithecus africanus | 3.0–2.0 | 400–500 | Bipedal with gracile build; no confirmed tools.[185] | South Africa |
| Australopithecus sediba | ~2.0 | ~420 | Bipedal with mosaic traits (Homo-like pelvis, arboreal hands); no tools.[187] | South Africa |
| Paranthropus aethiopicus | 2.7–2.3 | ~410 | Robust skull with sagittal crest for chewing muscles; bipedal; no tools.[188] | East Africa (Kenya, Ethiopia) |
| Paranthropus boisei | 2.3–1.2 | 500–550 | Hyper-robust jaws and teeth for tough vegetation; bipedal; no advanced tools.[185] | East Africa (Tanzania, Kenya) |
| Paranthropus robustus | 2.0–1.2 | ~500 | Robust dentition and cresting; bipedal; possible simple tools nearby.[185] | South Africa |
| Homo habilis | 2.4–1.4 | 600–800 | Bipedal with larger brain; associated with Oldowan simple stone tools (choppers, flakes).[185] | East Africa (Tanzania, Kenya) |
| Homo rudolfensis | 2.4–1.8 | 700–900 | Larger face and brain than H. habilis; bipedal; Oldowan-like tools.[185] | East Africa (Kenya) |
| Homo erectus / H. ergaster | 1.9–0.1 (100 kya) | 800–1200 | Fully modern body proportions, endurance running; Acheulean hand axes, fire use.[39] | Africa, Asia, Europe |
| Homo antecessor | 1.2–0.8 | 1000–1150 | Modern-like face with archaic features; bipedal; Mode 1 stone tools.[189] | Europe (Spain) |
| Homo heidelbergensis | 0.7–0.2 (700–200 kya) | 1100–1400 | Large brow ridges, robust build; advanced Acheulean tools, possible spears.[185] | Africa, Europe |
| Homo naledi | 0.335–0.236 (335–236 kya) | 500–600 | Mosaic traits (small brain, curved fingers for climbing); bipedal; possible tool use.[185] | South Africa |
| Homo neanderthalensis | 0.4–0.04 (400–40 kya) | 1400–1600 | Stocky build adapted to cold; Mousterian tools, burials, symbolic behavior.[185] | Europe, Western Asia |
| Homo denisova | 0.2–0.05 (200–50 kya) | ~1400 (est.) | Known primarily from DNA; robust build; Middle Paleolithic tools. | Asia (Siberia, Tibet) |
| Homo sapiens | 0.3–present (300 kya) | 1300–1500 | High forehead, globular braincase; Upper Paleolithic tools, art, complex culture.[58] | Global (orig. Africa) |
| Homo floresiensis | 0.1–0.05 (100–50 kya) | ~400 | Dwarfed body (hobbit-like); bipedal; simple stone tools.[185] | Southeast Asia (Indonesia) |
| Homo luzonensis | ~0.067 (67 kya) | Unknown (small body suggests small brain) | Small-bodied with curved phalanges; bipedal/arboreal mix; stone tools nearby. | Southeast Asia (Philippines) |