Recent from talks
Nothing was collected or created yet.
Ozone depletion
View on Wikipedia
| Part of a series on |
| Pollution |
|---|
 |
Ozone depletion consists of two related events observed since the late 1970s: a lowered total amount of ozone in Earth's upper atmosphere, and a much larger springtime decrease in stratospheric ozone (the ozone layer) around Earth's polar regions.[1] The latter phenomenon is referred to as the ozone hole. There are also springtime polar tropospheric ozone depletion events in addition to these stratospheric events.
The main causes of ozone depletion and the ozone hole are manufactured chemicals, especially manufactured halocarbon refrigerants, solvents, propellants, and foam-blowing agents (chlorofluorocarbons (CFCs), HCFCs, halons), referred to as ozone-depleting substances (ODS).[2] These compounds are transported into the stratosphere by turbulent mixing after being emitted from the surface, mixing much faster than the molecules can settle.[3] Once in the stratosphere, they release atoms from the halogen group through photodissociation, which catalyze the breakdown of ozone (O3) into oxygen (O2).[4] Both types of ozone depletion were observed to increase as emissions of halocarbons increased.
Ozone depletion and the ozone hole have generated worldwide concern over increased cancer risks and other negative effects. The ozone layer prevents harmful wavelengths of ultraviolet (UVB) light from passing through the Earth's atmosphere. These wavelengths cause skin cancer, sunburn, permanent blindness, and cataracts,[5] which were projected to increase dramatically as a result of thinning ozone, as well as harming plants and animals. These concerns led to the adoption of the Montreal Protocol in 1987, which bans the production of CFCs, halons, and other ozone-depleting chemicals.[6] Over time, scientists have developed new refrigerants with lower global warming potential (GWP) to replace older ones. For example, in new automobiles, R-1234yf systems are now common, being chosen over refrigerants with much higher GWP such as R-134a and R-12.
The ban came into effect in 1989. Ozone levels stabilized by the mid-1990s and began to recover in the 2000s, as the shifting of the jet stream in the Southern Hemisphere towards the South Pole has stopped and might even be reversing.[7] Recovery was projected to continue over the next century, with the ozone hole expected to reach pre-1980 levels by around 2075.[8] In 2019, NASA reported that the ozone hole was the smallest ever since it was first discovered in 1982.[9][10] The UN now projects that under the current regulations the ozone layer will completely regenerate by 2045.[11][12] The Montreal Protocol is considered the most successful international environmental agreement to date.[13][14]
Ozone cycle overview
[edit]
Three forms (or allotropes) of oxygen are involved in the ozone-oxygen cycle: oxygen atoms (O or atomic oxygen), oxygen gas (O
2 or diatomic oxygen), and ozone gas (O
3 or triatomic oxygen).[15] Ozone is formed in the stratosphere when oxygen gas molecules photodissociate after absorbing UVC photons. This converts a single O
2 into two atomic oxygen radicals. The atomic oxygen radicals then combine with separate O
2 molecules to create two O
3 molecules. These ozone molecules absorb UVB light, following which ozone splits into a molecule of O
2 and an oxygen atom. The oxygen atom then joins up with an oxygen molecule to regenerate ozone. This is a continuing process that terminates when an oxygen atom recombines with an ozone molecule to make two O
2 molecules. It is worth noting that ozone is the only atmospheric gas that absorbs UVB light.
- O + O
3 → 2 O
2


The total amount of ozone in the stratosphere is determined by a balance between photochemical production and recombination.
Ozone can be destroyed by a number of free radical catalysts; the most important are the hydroxyl radical (OH·), nitric oxide radical (NO·), chlorine radical (Cl·) and bromine radical (Br·). The dot is a notation to indicate that each species has an unpaired electron and is thus extremely reactive. The effectiveness of different halogens and pseudohalogens as catalysts for ozone destruction varies, in part due to differing routes to regenerate the original radical after reacting with ozone or dioxygen.[16]
While all of the relevant radicals have both natural and man-made sources, human activity has impacted some more than others. As of 2020, most of the OH· and NO· in the stratosphere is naturally occurring, but human activity has drastically increased the levels of chlorine and bromine.[17] These elements are found in stable organic compounds, especially chlorofluorocarbons, which can travel to the stratosphere without being destroyed in the troposphere due to their low reactivity. Once in the stratosphere, the Cl and Br atoms are released from the parent compounds by the action of ultraviolet light, e.g.
- CFCl
3 + electromagnetic radiation → Cl· + ·CFCl
2
Ozone is a highly reactive molecule that easily reduces to the more stable oxygen form with the assistance of a catalyst. Cl and Br atoms destroy ozone molecules through a variety of catalytic cycles. In the simplest example of such a cycle,[18] a chlorine atom reacts with an ozone molecule (O
3), taking an oxygen atom to form chlorine monoxide (ClO) and leaving an oxygen molecule (O
2). The ClO can react with a second molecule of ozone, releasing the chlorine atom and yielding two molecules of oxygen. The chemical shorthand for these gas-phase reactions is:
- Cl· + O
3 → ClO + O
2
A chlorine atom removes an oxygen atom from an ozone molecule to make a ClO molecule - ClO + O
3 → Cl· + 2 O
2
This ClO can also remove an oxygen atom from another ozone molecule; the chlorine is free to repeat this two-step cycle
The overall effect is a decrease in the amount of ozone, though the rate of these processes can be decreased by the effects of null cycles. More complicated mechanisms have also been discovered that lead to ozone destruction in the lower stratosphere.
A single chlorine atom would continuously destroy ozone (thus a catalyst) for up to two years (the time scale for transport back down to the troposphere) except for reactions that remove it from this cycle by forming reservoir species such as hydrogen chloride (HCl) and chlorine nitrate (ClONO
2). Bromine is even more efficient than chlorine at destroying ozone on a per-atom basis, but there is much less bromine in the atmosphere at present. Both chlorine and bromine contribute significantly to overall ozone depletion. Laboratory studies have also shown that fluorine and iodine atoms participate in analogous catalytic cycles. However, fluorine atoms react rapidly with water vapour, methane and hydrogen to form strongly bound hydrogen fluoride (HF) in the Earth's stratosphere,[19] while organic molecules containing iodine react so rapidly in the lower atmosphere that they do not reach the stratosphere in significant quantities.[20]
A single chlorine atom is able to react with an average of 100,000 ozone molecules before it is removed from the catalytic cycle. This fact plus the amount of chlorine released into the atmosphere yearly by chlorofluorocarbons (CFCs) and hydrochlorofluorocarbons (HCFCs) demonstrates the danger of CFCs and HCFCs to the environment.[21][22]
Observations on ozone layer depletion
[edit]
The ozone hole is usually measured by reduction in the total column ozone above a point on the Earth's surface. This is normally expressed in Dobson units; abbreviated as "DU". The most prominent decrease in ozone has been in the lower stratosphere. Marked decreases in column ozone in the Antarctic spring and early summer compared to the early 1970s and before have been observed using instruments such as the Total Ozone Mapping Spectrometer (TOMS).[23]
Reductions of up to 70 percent in the ozone column observed in the austral (Southern Hemispheric) spring over Antarctica and first reported in 1985 (Farman et al.) are continuing. As of 2010[update], Antarctic total column ozone in September and October continued to be 40–50 percent lower than pre-ozone-hole values since the 1990s.[1] A gradual trend toward "healing" was reported in 2016.[24] In 2017, NASA announced that the ozone hole was the weakest since 1988 because of warm stratospheric conditions. It is expected to recover around 2070.[25]
The amount lost is more variable year-to-year in the Arctic than in the Antarctic. The greatest Arctic declines are in the winter and spring, reaching up to 30 percent when the stratosphere is coldest.[26]
Reactions that take place on polar stratospheric clouds (PSCs) play an important role in enhancing ozone depletion.[27] PSCs form more readily in the extreme cold of the Arctic and Antarctic stratosphere. This is why ozone holes first formed, and are deeper, over Antarctica. Early models failed to take PSCs into account and predicted a gradual global depletion, which is why the sudden Antarctic ozone hole was such a surprise to many scientists.[28][29][30]
It is more accurate to speak of ozone depletion in middle latitudes rather than holes. Total column ozone declined below pre-1980 values between 1980 and 1996 for mid-latitudes. In the northern mid-latitudes, it then increased from the minimum value by about two percent from 1996 to 2009 as regulations took effect and the amount of chlorine in the stratosphere decreased. In the Southern Hemisphere's mid-latitudes, total ozone remained constant over that time period. There are no significant trends in the tropics, largely because halogen-containing compounds have not had time to break down and release chlorine and bromine atoms at tropical latitudes.[1][31]
Large volcanic eruptions have been shown to have substantial albeit uneven ozone-depleting effects, as observed with the 1991 eruption of Mt. Pinatubo in the Philippines.[32]
Ozone depletion also explains much of the observed reduction in stratospheric and upper tropospheric temperatures.[33][34] The source of the warmth of the stratosphere is the absorption of UV radiation by ozone, hence reduced ozone leads to cooling. Some stratospheric cooling is also predicted from increases in greenhouse gases such as CO
2 and CFCs themselves; however, the ozone-induced cooling appears to be dominant.[35]
Predictions of ozone levels remain difficult, but the precision of models' predictions of observed values and the agreement among different modeling techniques have increased steadily.[1] The World Meteorological Organization Global Ozone Research and Monitoring Project—Report No. 44 is strongly in favor of the Montreal Protocol, but notes that a UNEP 1994 Assessment overestimated ozone loss for the 1994–1997 period.[36]
Compounds in the atmosphere
[edit]CFCs and related compounds
[edit]Chlorofluorocarbons (CFCs) and other halogenated ozone-depleting substances (ODS) are mainly responsible for man-made chemical ozone depletion. The total amount of effective halogens (chlorine and bromine) in the stratosphere can be calculated and are known as the equivalent effective stratospheric chlorine (EESC).[37]
CFCs as refrigerants were invented by Thomas Midgley Jr. in the 1930s.[38] They were used in air conditioning and cooling units, as aerosol spray propellants prior to the 1970s, and in the cleaning processes of delicate electronic equipment. They also occur as by-products of some chemical processes. No significant natural sources have ever been identified for these compounds—their presence in the atmosphere is due almost entirely to human manufacture. As mentioned above, when such ozone-depleting chemicals reach the stratosphere, they are dissociated by ultraviolet light to release chlorine atoms. The chlorine atoms act as a catalyst, and each can break down tens of thousands of ozone molecules before being removed from the stratosphere. Given the longevity of CFC molecules, recovery times are measured in decades. It is calculated that a CFC molecule takes an average of about five to seven years to go from the ground level up to the upper atmosphere, and it can stay there for about a century,[39] destroying up to one hundred thousand ozone molecules during that time.[40][41]
1,1,1-Trichloro-2,2,2-trifluoroethane, also known as CFC-113a, is one of four man-made chemicals newly discovered in the atmosphere by a team at the University of East Anglia. CFC-113a is the only known CFC whose abundance in the atmosphere is still growing. Its source remains a mystery, but illegal manufacturing is suspected by some. CFC-113a seems to have been accumulating unabated since 1960. Between 2012 and 2017, concentrations of the gas jumped by 40 percent.[42]
A study by an international team of researchers published in Nature found that since 2013 emissions that are predominately from north-eastern China have released large quantities of the banned chemical Chlorofluorocarbon-11 (CFC-11) into the atmosphere. Scientists estimate that without action, these CFC-11 emissions will delay the recovery of the planet's ozone hole by a decade.[43][44][45]
Aluminum oxide
[edit]Satellites burning up upon re-entry into Earth's atmosphere produce aluminum oxide (Al2O3) nanoparticles that endure in the atmosphere for decades.[46] Estimates for 2022 alone were ~17 metric tons (~30 kg of nanoparticles per ~250 kg satellite).[46] Increasing populations of satellite constellations can eventually lead to significant ozone depletion.[46]
Very short-lived substances (VSLS)
[edit]"Very short-lived substances" are a class of ozone-depleting chemicals, allowed by the Montreal Protocol, that degrade in under 6 months.[47] 90% are naturally produced, for example bromine-based chemicals generated by seaweed and phytoplankton, but 10% are manmade, for example dichloromethane.[47]
Computer modeling
[edit]Scientists have attributed ozone depletion to the increase of man-made (anthropogenic) halogen compounds from CFCs by combining observational data with computer models. These complex chemistry transport models (e.g. SLIMCAT, CLaMS—Chemical Lagrangian Model of the Stratosphere) work by combining measurements of chemicals and meteorological fields with chemical reaction rate constants. They identify key chemical reactions and transport processes that bring CFC photolysis products into contact with ozone.
Ozone hole and its causes
[edit]
The Antarctic ozone hole is an area of the Antarctic stratosphere in which the recent ozone levels have dropped to as low as 33 percent of their pre-1975 values.[49] The ozone hole occurs during the Antarctic spring, from September to early December, as strong westerly winds start to circulate around the continent and create an atmospheric container. Within this polar vortex, over 50 percent of the lower stratospheric ozone is destroyed during the Antarctic spring.[50]
As explained above, the primary cause of ozone depletion is the presence of chlorine-containing source gases (primarily CFCs and related halocarbons). In the presence of UV light, these gases dissociate, releasing chlorine atoms, which then go on to catalyze ozone destruction. The Cl-catalyzed ozone depletion can take place in the gas phase, but it is substantially enhanced in the presence of polar stratospheric clouds (PSCs).[51]
These polar stratospheric clouds form during winter, in the extreme cold. Polar winters are dark, consisting of three months without solar radiation (sunlight). The lack of sunlight contributes to a decrease in temperature and the polar vortex traps and chills the air. Temperatures are around or below −80 °C. These low temperatures form cloud particles. There are three types of PSC clouds—nitric acid trihydrate clouds, slowly cooling water-ice clouds, and rapid cooling water-ice (nacreous) clouds—that provide surfaces for chemical reactions whose products will, in the spring lead to ozone destruction.[52]
The photochemical processes involved are complex but well understood. The key observation is that, ordinarily, most of the chlorine in the stratosphere resides in "reservoir" compounds, primarily chlorine nitrate (ClONO
2) as well as stable end products such as HCl. The formation of end products essentially removes Cl from the ozone depletion process. Reservoir compounds sequester Cl, which can later be made available via absorption of light at wavelengths shorter than 400 nm.[53] During the Antarctic winter and spring, reactions on the surface of the polar stratospheric cloud particles convert these "reservoir" compounds into reactive free radicals (Cl and ClO). Denitrification is the process by which the clouds remove NO
2 from the stratosphere by converting it to nitric acid in PSC particles, which then are lost by sedimentation. This prevents newly formed ClO from being converted back into ClONO
2.
The role of sunlight in ozone depletion is the reason why the Antarctic ozone depletion is greatest during spring. During winter, even though PSCs are at their most abundant, there is no light over the pole to drive chemical reactions. During the spring, however, sunlight returns and provides energy to drive photochemical reactions and melt the polar stratospheric clouds, releasing considerable ClO, which drives the hole mechanism. Further warming temperatures near the end of spring break up the vortex around mid-December. As warm, ozone and NO
2-rich air flows in from lower latitudes, the PSCs are destroyed, the enhanced ozone depletion process shuts down, and the ozone hole closes.[54]
Most of the ozone that is destroyed is in the lower stratosphere, in contrast to the much smaller ozone depletion through homogeneous gas-phase reactions, which occurs primarily in the upper stratosphere.[55]
Effects
[edit]Since the ozone layer absorbs UVB ultraviolet light from the sun, ozone layer depletion increases surface UVB levels (all else equal), which could lead to damage, including an increase in skin cancer. This was the reason for the Montreal Protocol. Although decreases in stratospheric ozone are well-tied to CFCs and increases in surface UVB, there is no direct observational evidence linking ozone depletion to higher incidence of skin cancer and eye damage in human beings. This is partly because UVA, which has also been implicated in some forms of skin cancer, is not absorbed by ozone, and because it is nearly impossible to control statistics for lifestyle changes over time. Ozone depletion may also influence wind patterns.[56]
Increased UV
[edit]Ozone, while a minority constituent in Earth's atmosphere, is responsible for most of the absorption of UVB radiation. The amount of UVB radiation that penetrates through the ozone layer decreases exponentially with the slant-path thickness and density of the layer.[57] When stratospheric ozone levels decrease, higher levels of UVB reach the Earth's surface.[1][58] UV-driven phenolic formation in tree rings has dated the start of ozone depletion in northern latitudes to the late 1700s.[59]
In October 2008, the Ecuadorian Space Agency published a report called HIPERION. The study used ground instruments in Ecuador and the last 28 years' data from 12 satellites of several countries, and found that the UV radiation reaching equatorial latitudes was far greater than expected, with the ultraviolet index climbing as high as 24 in Quito; the WHO considers 11 as an extreme index and a great risk to health. The report concluded that depleted ozone levels around the mid-latitudes of the planet are already endangering large populations in these areas.[60] Later, the CONIDA, the Peruvian Space Agency, published its own study, which yielded almost the same findings as the Ecuadorian study.
Biological effects
[edit]The main public concern regarding the ozone hole has been the effects of increased surface UV radiation on human health. So far, ozone depletion in most locations has been typically a few percent and, as noted above, no direct evidence of health damage is available in most latitudes. If the high levels of depletion seen in the ozone hole were to be common across the globe, the effects could be substantially more dramatic. As the ozone hole over Antarctica has in some instances grown so large as to affect parts of Australia, New Zealand, Chile, Argentina, and South Africa, environmentalists have been concerned that the increase in surface UV could be significant.[61] Excessive ultraviolet radiation (UVR) has reducing effects on the rates of photosynthesis and growth of benthic diatom communities (microalgae species that increase water quality and are pollution resistant) that are present in shallow freshwater.[62] Ozone depletion not only affects human health but also has a profound impact on biodiversity. It damages plants and trees at the cellular level, affecting their growth, vitality, photosynthesis, water balance, and defense mechanisms against pests and diseases. This sets off a cascade of ecological impacts, harming soil microbes, insects, wildlife, and entire ecosystems.[63]
Ozone depletion would magnify all of the effects of UV on human health, both positive (including production of vitamin D) and negative (including sunburn, skin cancer, and cataracts). In addition, increased surface UV leads to increased tropospheric ozone, which is a health risk to humans.[64]
Basal and squamous cell carcinomas
[edit]The most common forms of skin cancer in humans, basal and squamous cell carcinomas, have been strongly linked to UV-B exposure. The mechanism by which UVB induces these cancers is well understood—absorption of UV-B radiation causes the pyrimidine bases in the DNA molecule to form dimers, resulting in transcription errors when the DNA replicates. These cancers are relatively mild and rarely fatal, although the treatment of squamous cell carcinoma sometimes requires extensive reconstructive surgery. By combining epidemiological data with results of animal studies, scientists have estimated that every one percent decrease in long-term stratospheric ozone would increase the incidence of these cancers by 2%.[65]
Melanoma
[edit]Another form of skin cancer, melanoma, is much less common but far more dangerous, being lethal in about 15–20 percent of the cases diagnosed. The relationship between melanoma and ultraviolet exposure is not yet fully understood, but it appears that both UV-B and UV-A are involved. Because of this uncertainty, it is difficult to estimate the effect of ozone depletion on melanoma incidence. One study showed that a 10 percent increase in UV-B radiation was associated with a 19 percent increase in melanomas for men and 16 percent for women.[66] A study of people in Punta Arenas, at the southern tip of Chile, showed a 56 percent increase in melanoma and a 46 percent increase in non-melanoma skin cancer over a period of seven years, along with decreased ozone and increased UVB levels.[67]
Cortical cataracts
[edit]Epidemiological studies suggest an association between ocular cortical cataracts and UV-B exposure, using crude approximations of exposure and various cataract assessment techniques. A detailed assessment of ocular exposure to UV-B was carried out in a study on Chesapeake Bay Watermen, where increases in average annual ocular exposure were associated with increasing risk of cortical opacity.[68] In this highly exposed group of predominantly white males, the evidence linking cortical opacities to sunlight exposure was the strongest to date. Based on these results, ozone depletion is predicted to cause hundreds of thousands of additional cataracts by 2050.[69]
Increased tropospheric ozone
[edit]Increased surface UV leads to increased tropospheric ozone. Ground-level ozone is generally recognized to be a health risk, as ozone is toxic due to its strong oxidant properties. The risks are particularly high for young children, the elderly, and those with asthma or other respiratory difficulties. At this time, ozone at ground level is produced mainly by the action of UV radiation on combustion gases from vehicle exhausts.[70]
Increased production of vitamin D
[edit]Vitamin D is produced in the skin by ultraviolet light. Thus, higher UVB exposure raises human vitamin D in those deficient in it.[71] Recent research (primarily since the Montreal Protocol) shows that many humans have less than optimal vitamin D levels. In particular, in the U.S. population, the lowest quarter of vitamin D (<17.8 ng/ml) were found using information from the National Health and Nutrition Examination Survey to be associated with an increase in all-cause mortality in the general population.[72] While blood level of vitamin D in excess of 100 ng/ml appear to raise blood calcium excessively and to be associated with higher mortality, the body has mechanisms that prevent sunlight from producing vitamin D in excess of the body's requirements.[73]
Effects on animals
[edit]A November 2011 report by scientists at the Institute of Zoology in London, England found that whales off the coast of California have shown a sharp rise in sun damage, and these scientists "fear that the thinning ozone layer is to blame".[74] The study photographed and took skin biopsies from over 150 whales in the Gulf of California and found "widespread evidence of epidermal damage commonly associated with acute and severe sunburn", having cells that form when the DNA is damaged by UV radiation. The findings suggest "rising UV levels as a result of ozone depletion are to blame for the observed skin damage, in the same way that human skin cancer rates have been on the increase in recent decades."[75] Apart from whales many other animals such as dogs, cats, sheep and terrestrial ecosystems also suffer the negative effects of increased UV-B radiations.[76]
Effects on crops
[edit]An increase of UV radiation would be expected to affect crops. A number of economically important species of plants, such as rice, depend on cyanobacteria residing on their roots for the retention of nitrogen. Cyanobacteria are sensitive to UV radiation and would be affected by its increase.[77] "Despite mechanisms to reduce or repair the effects of increased ultraviolet radiation, plants have a limited ability to adapt to increased levels of UVB, therefore plant growth can be directly affected by UVB radiation."[78]
Effects on plant life
[edit]Over the years, the Arctic ozone layer has depleted severely. As a consequence species that live above the snow cover or in areas where snow has melted abundantly, due to hot temperatures, are negatively impacted due to UV radiation that reaches the ground.[79] Depletion of the ozone layer and allowing excess UVB radiation would initially be assumed to increase damage to plant DNA. Reports have found that when plants are exposed to UVB radiation similar to stratospheric ozone depletion, there was no significant change in plant height or leaf mass, but showed a response in shoot biomass and leaf area with a small decrease.[80] However, UVB radiation has been shown to decrease quantum yield of photosystem II.[81] UVB damage only occurs under extreme exposure, and most plants also have UVB absorbing flavonoids which allow them to acclimatize to the radiation present. Plants experience different levels of UV radiation throughout the day. It is known that they are able to shift the levels and types of UV sunscreens (i.e. flavonoids), that they contain, throughout the day. This allows them to increase their protection against UV radiation.[82] Plants that have been affected by radiation throughout development are more affected by the inability to intercept light with a larger leaf area than having photosynthetic systems compromised.[83] Damage from UVB radiation is more likely to be significant on species interactions than on plants themselves.[84]
Another significant impact of ozone depletion on plant life is the stress experienced by plants when exposed to UV radiation. This can cause a decrease in plant growth and an increase in oxidative stress, due to the production of nitric oxide and hydrogen peroxide.[85] In areas where substantial ozone depletion has occurred, increased UV-B radiation reduces terrestrial plant productivity (and likewise carbon sequestration) by about 6%.[86][87]
Moreover, if plants are exposed to high levels of UV radiation, it can elicit the production of harmful volatile organic compounds, like isoprenes. The emission of isoprenes into the air, by plants, can severely impact the environment by adding to air pollution and increasing the amount of carbon in the atmosphere, ultimately contributing to climate change.[88]
Public policy
[edit]
The full extent of the damage that CFCs have caused to the ozone layer is not known and will not be known for decades; however, marked decreases in column ozone have already been observed. The Montreal and Vienna conventions were installed long before a scientific consensus was established or important uncertainties in the science field were being resolved.[89] The ozone case was understood comparably well by lay persons as e.g. Ozone shield or ozone hole were useful "easy-to-understand bridging metaphors".[90] Americans voluntarily switched away from aerosol sprays, resulting in a 50 percent sales loss even before legislation was enforced.[90]
After a 1976 report by the United States National Academy of Sciences concluded that credible scientific evidence supported the ozone depletion hypothesis[91] a few countries, including the United States, Canada, Sweden, Denmark, and Norway, moved to eliminate the use of CFCs in aerosol spray cans.[92] At the time this was widely regarded as a first step towards a more comprehensive regulation policy, but progress in this direction slowed in subsequent years, due to a combination of political factors (continued resistance from the halocarbon industry and a general change in attitude towards environmental regulation during the first two years of the Reagan administration) and scientific developments (subsequent National Academy assessments that indicated that the first estimates of the magnitude of ozone depletion had been overly large).
A critical DuPont manufacturing patent for Freon was set to expire in 1979. The United States banned the use of CFCs in aerosol cans in 1978.[92] The European Community rejected proposals to ban CFCs in aerosol sprays, and in the U.S., CFCs continued to be used as refrigerants and for cleaning circuit boards. Worldwide CFC production fell sharply after the U.S. aerosol ban, but by 1986 had returned nearly to its 1976 level.[92] In 1993, DuPont Canada closed its CFC facility.[93]
The U.S. government's attitude began to change again in 1983, when William Ruckelshaus replaced Anne M. Burford as Administrator of the United States Environmental Protection Agency (EPA). Under Ruckelshaus and his successor, Lee Thomas, the EPA pushed for an international approach to halocarbon regulations. In 1985 twenty nations, including most of the major CFC producers, signed the Vienna Convention for the Protection of the Ozone Layer, which established a framework for negotiating international regulations on ozone-depleting substances. That same year, the discovery of the Antarctic ozone hole was announced, causing a revival in public attention to the issue.
In 1987, representatives from 43 nations signed the Montreal Protocol. Meanwhile, the halocarbon industry shifted its position and started supporting a protocol to limit CFC production. However, this shift was uneven with DuPont acting more quickly than its European counterparts. DuPont may have feared court action related to increased skin cancer, especially as the EPA had published a study in 1986 claiming that an additional 40 million cases and 800,000 cancer deaths were to be expected in the U.S. in the next 88 years.[94] The EU shifted its position as well after Germany gave up its defence of the CFC industry and started supporting moves towards regulation. Government and industry in France and the UK tried to defend their CFC producing industries even after the Montreal Protocol had been signed.[95]
At Montreal, the participants agreed to freeze production of CFCs at 1986 levels and to reduce production by 50 percent by 1999.[92] After a series of scientific expeditions to the Antarctic produced convincing evidence that the ozone hole was indeed caused by chlorine and bromine from manmade organohalogens, the Montreal Protocol was strengthened at a 1990 meeting in London. The participants agreed to phase out CFCs and halons entirely (aside from a very small amount marked for certain "essential" uses, such as asthma inhalers) by 2000 in non-Article 5 countries and by 2010 in Article 5 (less developed) signatories.[96] At a 1992 meeting in Copenhagen, Denmark, the phase-out date was moved up to 1996.[96] At the same meeting, methyl bromide (MeBr), a fumigant used primarily in agricultural production, was added to the list of controlled substances. For all substances controlled under the protocol, phaseout schedules were delayed for less developed ('Article 5(1)') countries, and phaseout in these countries was supported by transfers of expertise, technology, and money from non-Article 5(1) Parties to the Protocol. Additionally, exemptions from the agreed schedules could be applied for under the Essential Use Exemption (EUE) process for substances other than methyl bromide and under the Critical Use Exemption (CUE) process for methyl bromide.[97][98]
Civil society, including especially non-governmental organizations (NGOs), played critical roles at all stages of policy development leading to the Vienna Conference, the Montreal Protocol, and in assessing compliance afterwards.[99][100][101][102] The major companies claimed that no alternatives to HFC existed.[103] An ozone-safe hydrocarbon refrigerant was developed at a technological institute in Hamburg, Germany, consisting of a mixture of the hydrocarbon gases propane and butane, and in 1992 came to the attention of the NGO Greenpeace. Greenpeace called it "Greenfreeze".[104][105] The NGO then worked successfully first with a small and struggling company to market an appliance beginning in Europe, then Asia and later Latin America, receiving a 1997 UNEP award.[106][107] By 1995, Germany had made CFC refrigerators illegal.[107] Since 2004, corporations like Coca-Cola, Carlsberg, and IKEA formed a coalition to promote the ozone-safe Greenfreeze units. Production spread to companies like Electrolux, Bosch, and LG, with sales reaching some 300 million refrigerators by 2008.[106][108] In Latin America, a domestic Argentinian company began Greenfreeze production in 2003, while the giant Bosch in Brazil began a year later.[109][110] By 2013 it was being used by some 700 million refrigerators, making up about 40 percent of the market.[103]
In the U.S., however, change has been much slower. To some extent, CFCs were being replaced by the less damaging hydrochlorofluorocarbons (HCFCs), although concerns remain regarding HCFCs also. In some applications, hydrofluorocarbons (HFCs) were being used to replace CFCs. HFCs, which contain no chlorine or bromine, do not contribute to ozone depletion although they are potent greenhouse gases. The best known of these compounds is probably HFC-134a (R-134a), which in the United States has largely replaced CFC-12 (R-12) in automobile air conditioners. In laboratory analytics (a former "essential" use) the ozone depleting substances can be replaced with other solvents.[111] Chemical companies like Du Pont, whose representatives disparaged Greenfreeze as "that German technology," maneuvered the EPA to block the technology in the U.S. until 2011.[112][113][114][115] Ben & Jerry's of Unilever and General Electric, spurred by Greenpeace, had expressed formal interest in 2008 which figured in the EPA's final approval.[106][116]
The EU recast its Ozone Regulation in 2009. The law bans ozone-depleting substances with the goal of protecting the ozone layer.[117] The list of ODS that are subject to the regulation is the same as those under the Montreal Protocol, with some additions.
More recently, policy experts have advocated for efforts to link ozone protection efforts to climate protection efforts.[118][119] Many ODS are also greenhouse gases, some thousands of times more powerful agents of radiative forcing than carbon dioxide over the short and medium term. Thus policies protecting the ozone layer have had benefits in mitigating climate change. The reduction of the radiative forcing due to ODS probably masked the true level of climate change effects of other greenhouse gases, and was responsible for the "slow down" of global warming from the mid-90s.[120][additional citation(s) needed] Policy decisions in one arena affect the costs and effectiveness of environmental improvements in the other.
ODS requirements in the marine industry
[edit]The IMO has amended MARPOL Annex VI Regulation 12 regarding ozone depleting substances. As from 1 July 2010, all vessels where MARPOL Annex VI is applicable should have a list of equipment using ozone depleting substances. The list should include the name of ODS, type and location of equipment, quantity in kilograms and date. All changes since that date should be recorded in an ODS Record book on board recording all intended or unintended releases to the atmosphere. Furthermore, new ODS supply or landing to shore facilities should be recorded as well.
Prospects of ozone depletion
[edit]

Since the adoption and strengthening of the Montreal Protocol has led to reductions in the emissions of CFCs, atmospheric concentrations of the most-significant compounds have been declining. These substances are being gradually removed from the atmosphere; since peaking in 1994, the Effective Equivalent Chlorine (EECl) level in the atmosphere had dropped about 10 percent by 2008. The decrease in ozone-depleting chemicals has also been significantly affected by a decrease in bromine-containing chemicals. The data suggest that substantial natural sources exist for atmospheric methyl bromide (CH
3Br).[1] The phase-out of CFCs means that nitrous oxide (N
2O), which is not covered by the Montreal Protocol, has become the most highly emitted ozone-depleting substance and is expected to remain so throughout the 21st century.[121]
According to the IPCC Sixth Assessment Report, global stratospheric ozone levels experienced rapid decline in the 1970s and 1980s and have since been increasing, but have not reached preindustrial levels. Although considerable variability is expected from year to year, including in polar regions where depletion is largest, the ozone layer is expected to continue recovering in coming decades due to declining ozone-depleting substance concentrations, assuming full compliance with the Montreal Protocol.[122]
The Antarctic ozone hole is expected to continue for decades. Ozone concentrations in the lower stratosphere over Antarctica increased by 5–10 percent by 2020 and will return to pre-1980 levels by about 2060–2075. This is 10–25 years later than predicted in earlier assessments, because of revised estimates of atmospheric concentrations of ozone-depleting substances, including a larger predicted future usage in developing countries. Another factor that may prolong ozone depletion is the drawdown of nitrogen oxides from above the stratosphere due to changing wind patterns.[123] A gradual trend toward "healing" was reported in 2016.[24] In 2019, the ozone hole was at its smallest in the previous thirty years, due to the warmer polar stratosphere weakening the polar vortex.[124] In September 2023, the Antarctic ozone hole was one of the largest on record, at 26 million square kilometers. The anomalously large ozone loss may have been a result of the 2022 Tonga volcanic eruption.[125] According to a 2023 United Nations assessment, the ozone layer is on track to recover to 1980 levels by around 2066 over Antarctica, by 2045 over the Arctic, and by 2040 for the rest of the world, assuming current regulations remain in place.[126]
Research history
[edit]The basic physical and chemical processes that lead to the formation of an ozone layer in the Earth's stratosphere were discovered by Sydney Chapman in 1930. Short-wavelength UV radiation splits an oxygen (O
2) molecule into two oxygen (O) atoms, which then combine with other oxygen molecules to form ozone. Ozone is removed when an oxygen atom and an ozone molecule "recombine" to form two oxygen molecules, i.e. O + O
3 → 2O
2. In the 1950s, David Bates and Marcel Nicolet presented evidence that various free radicals, in particular hydroxyl (OH) and nitric oxide (NO), could catalyze this recombination reaction, reducing the overall amount of ozone. These free radicals were known to be present in the stratosphere, and so were regarded as part of the natural balance—it was estimated that in their absence, the ozone layer would be about twice as thick as it currently is.
In 1970 Paul Crutzen pointed out that emissions of nitrous oxide (N
2O), a stable, long-lived gas produced by soil bacteria, from the Earth's surface could affect the amount of nitric oxide (NO) in the stratosphere. Crutzen showed that nitrous oxide lives long enough to reach the stratosphere, where it is converted into NO. Crutzen then noted that increasing use of fertilizers might have led to an increase in nitrous oxide emissions over the natural background, which would in turn result in an increase in the amount of NO in the stratosphere. Thus human activity could affect the stratospheric ozone layer. In the following year, Crutzen and (independently) Harold Johnston suggested that NO emissions from supersonic passenger aircraft, which would fly in the lower stratosphere, could also deplete the ozone layer. However, more recent analysis in 1995 by David W. Fahey, an atmospheric scientist at the National Oceanic and Atmospheric Administration, found that the drop in ozone would be from 1–2 percent if a fleet of 500 supersonic passenger aircraft were operated.[127] This, Fahey expressed, would not be a showstopper for advanced supersonic passenger aircraft development.[128]
Rowland–Molina hypothesis
[edit]In 1974 Frank Sherwood Rowland, Chemistry Professor at the University of California at Irvine, and his postdoctoral associate Mario J. Molina suggested that long-lived organic halogen compounds, such as CFCs, might behave in a similar fashion as Crutzen had proposed for nitrous oxide. James Lovelock had recently discovered, during a cruise in the South Atlantic in 1971, that almost all of the CFC compounds manufactured since their invention in 1930 were still present in the atmosphere. Molina and Rowland concluded that, like N
2O, the CFCs would reach the stratosphere where they would be dissociated by UV light, releasing chlorine atoms. A year earlier, Richard Stolarski and Ralph Cicerone at the University of Michigan had shown that Cl is even more efficient than NO at catalyzing the destruction of ozone. Similar conclusions were reached by Michael McElroy and Steven Wofsy at Harvard University. Neither group, however, had realized that CFCs were a potentially large source of stratospheric chlorine—instead, they had been investigating the possible effects of HCl emissions from the Space Shuttle, which are very much smaller.
The Rowland–Molina hypothesis was strongly disputed by representatives of the aerosol and halocarbon industries. The Chair of the Board of DuPont was quoted as saying that ozone depletion theory is "a science fiction tale ... a load of rubbish ... utter nonsense".[129] Robert Abplanalp, the President of Precision Valve Corporation (and inventor of the first practical aerosol spray can valve), wrote to the Chancellor of UC Irvine to complain about Rowland's public statements.[130] Nevertheless, within three years most of the basic assumptions made by Rowland and Molina were confirmed by laboratory measurements and by direct observation in the stratosphere. The concentrations of the source gases (CFCs and related compounds) and the chlorine reservoir species (HCl and ClONO
2) were measured throughout the stratosphere, and demonstrated that CFCs were indeed the major source of stratospheric chlorine, and that nearly all of the CFCs emitted would eventually reach the stratosphere. Even more convincing was the measurement, by James G. Anderson and collaborators, of chlorine monoxide (ClO) in the stratosphere. ClO is produced by the reaction of Cl with ozone—its observation thus demonstrated that Cl radicals not only were present in the stratosphere but also were actually involved in destroying ozone. McElroy and Wofsy extended the work of Rowland and Molina by showing that bromine atoms were even more effective catalysts for ozone loss than chlorine atoms and argued that the brominated organic compounds known as halons, widely used in fire extinguishers, were a potentially large source of stratospheric bromine. In 1976 the United States National Academy of Sciences released a report concluding that the ozone depletion hypothesis was strongly supported by the scientific evidence. In response the United States, Canada and Norway banned the use of CFCs in aerosol spray cans in 1978. Early estimates were that, if CFC production continued at 1977 levels, the total atmospheric ozone would after a century or so reach a steady state, 15 to 18 percent below normal levels. By 1984, when better evidence on the speed of critical reactions was available, this estimate was changed to 5 to 9 percent steady-state depletion.[131]
Crutzen, Molina, and Rowland were awarded the 1995 Nobel Prize in Chemistry for their work on stratospheric ozone.
Antarctic ozone hole
[edit]The discovery of the Antarctic "ozone hole" by British Antarctic Survey scientists Farman, Gardiner and Shanklin (first reported in a paper in Nature in May 1985[132]) came as a shock to the scientific community, because the observed decline in polar ozone was far larger than had been anticipated.[133] Satellite measurements (TOMS onboard Nimbus 7) showing massive depletion of ozone around the south pole were becoming available at the same time.[134] However, these were initially rejected as unreasonable by data quality control algorithms (they were filtered out as errors since the values were unexpectedly low); the ozone hole was detected only in satellite data when the raw data was reprocessed following evidence of ozone depletion in in situ observations.[95] When the software was rerun without the flags, the ozone hole was seen as far back as 1976.[135]
Susan Solomon, an atmospheric chemist at the National Oceanic and Atmospheric Administration (NOAA), proposed that chemical reactions on polar stratospheric clouds (PSCs) in the cold Antarctic stratosphere caused a massive, though localized and seasonal, increase in the amount of chlorine present in active, ozone-destroying forms. The polar stratospheric clouds in Antarctica are only formed at very low temperatures, as low as −80 °C, and early spring conditions. In such conditions the ice crystals of the cloud provide a suitable surface for conversion of unreactive chlorine compounds into reactive chlorine compounds, which can easily deplete ozone.
Moreover, the polar vortex formed over Antarctica is very tight and the reaction occurring on the surface of the cloud crystals is far different from when it occurs in atmosphere. These conditions have led to ozone hole formation in Antarctica. This hypothesis was decisively confirmed, first by laboratory measurements and subsequently by direct measurements, from the ground and from high-altitude airplanes, of very high concentrations of chlorine monoxide (ClO) in the Antarctic stratosphere.[136]
Alternative hypotheses, which had attributed the ozone hole to variations in solar UV radiation or to changes in atmospheric circulation patterns, were also tested and shown to be untenable.[137]
Meanwhile, analysis of ozone measurements from the worldwide network of ground-based Dobson spectrophotometers led an international panel to conclude that the ozone layer was in fact being depleted, at all latitudes outside of the tropics.[31] These trends were confirmed by satellite measurements. As a consequence, the major halocarbon-producing nations agreed to phase out production of CFCs, halons, and related compounds, a process that was completed in 1996.
Since 1981 the United Nations Environment Programme, under the auspices of the World Meteorological Organization, has sponsored a series of technical reports on the Scientific Assessment of Ozone Depletion, based on satellite measurements. The 2007 report showed that the hole in the ozone layer was recovering and the smallest it had been for about a decade.[138]
A 2010 report found, "Over the past decade, global ozone and ozone in the Arctic and Antarctic regions is no longer decreasing but is not yet increasing. The ozone layer outside the Polar regions is projected to recover to its pre-1980 levels some time before the middle of this century. In contrast, the springtime ozone hole over the Antarctic is expected to recover much later."[139]
In 2012, NOAA and NASA reported "Warmer air temperatures high above the Antarctic led to the second smallest season ozone hole in 20 years averaging 17.9 million square kilometres. The hole reached its maximum size for the season on Sept 22, stretching to 21.2 million square kilometres."[140] A gradual trend toward "healing" was reported in 2016[24] and then in 2017.[141] It is reported that the recovery signal is evident even in the ozone loss saturation altitudes.[142]
The hole in the Earth's ozone layer over the South Pole has affected atmospheric circulation in the Southern Hemisphere all the way to the equator.[143] The ozone hole has influenced atmospheric circulation all the way to the tropics and increased rainfall at low, subtropical latitudes in the Southern Hemisphere.[144]
Arctic ozone "mini-hole"
[edit]On March 3, 2005, the journal Nature[145] published an article linking 2004's unusually large Arctic ozone hole to solar wind activity.
On March 15, 2011, a record ozone layer loss was observed, with about half of the ozone present over the Arctic having been destroyed.[146][147][148] The change was attributed to increasingly cold winters in the Arctic stratosphere at an altitude of approximately 20 km (12 mi), a change associated with global warming in a relationship that is still under investigation.[147] By March 25, the ozone loss had become the largest compared to that observed in all previous winters with the possibility that it would become an ozone hole.[149] This would require that the quantities of ozone to fall below 200 Dobson units, from the 250 recorded over central Siberia.[149] It is predicted that the thinning layer would affect parts of Scandinavia and Eastern Europe on March 30–31.[149]
On October 2, 2011, a study was published in the journal Nature, which said that between December 2010 and March 2011 up to 80 percent of the ozone in the atmosphere at about 20 kilometres (12 mi) above the surface was destroyed.[150] The level of ozone depletion was severe enough that scientists said it could be compared to the ozone hole that forms over Antarctica every winter.[150] According to the study, "for the first time, sufficient loss occurred to reasonably be described as an Arctic ozone hole."[150] The study analyzed data from the Aura and CALIPSO satellites, and determined that the larger-than-normal ozone loss was due to an unusually long period of cold weather in the Arctic, some 30 days more than typical, which allowed for more ozone-destroying chlorine compounds to be created.[151] According to Lamont Poole, a co-author of the study, cloud and aerosol particles on which the chlorine compounds are found "were abundant in the Arctic until mid March 2011—much later than usual—with average amounts at some altitudes similar to those observed in the Antarctic, and dramatically larger than the near-zero values seen in March in most Arctic winters".[151]
In 2013, researchers analyzed the data and found the 2010–2011 Arctic event did not reach the ozone depletion levels to classify as a true hole. A hole in the ozone is generally classified as 220 Dobson units or lower;[152] the Arctic hole did not approach that low level.[153][154] It has since been classified as a "mini-hole."[155]
Following the ozone depletion in 1997 and 2011, a 90% drop in ozone was measured by weather balloons over the Arctic in March 2020, as they normally recorded 3.5 parts per million of ozone, compared to only around 0.3 parts per million lastly, due to the coldest temperatures ever recorded since 1979, and a strong polar vortex which allowed chemicals, including chlorine and bromine, to reduce ozone.[156]
A rare hole, the result of unusually low temperatures in the atmosphere above the North Pole, was studied in 2020.[157][158]
Tibet ozone hole
[edit]As winters that are colder are more affected, at times there is an ozone hole over Tibet. In 2006, a 2.5 million square kilometer ozone hole was detected over Tibet.[159] Again in 2011, an ozone hole appeared over mountainous regions of Tibet, Xinjiang, Qinghai and the Hindu Kush, along with an unprecedented hole over the Arctic, though the Tibet one was far less intense than the ones over the Arctic or Antarctic.[160]
Potential depletion by storm clouds
[edit]Research in 2012 showed that the same process that produces the ozone hole over Antarctica, occurs over summer storm clouds in the United States, and thus may be destroying ozone there as well.[161][162]
Ozone hole over tropics
[edit]Physicist Qing-Bin Lu, of the University of Waterloo, claimed to have discovered a large, all-season ozone hole in the lower stratosphere over the tropics in July 2022.[163] However, other researchers in the field refuted this claim, stating that the research was riddled with "serious errors and unsubstantiated assertions."[164] According to Dr Paul Young, a lead author of the 2022 WMO/UNEP Scientific Assessment of Ozone Depletion, "The author's identification of a 'tropical ozone hole' is down to him looking at percentage changes in ozone, rather than absolute changes, with the latter being much more relevant for damaging UV reaching the surface." Specifically, Lu's work defines "ozone hole" as "an area with O3 loss in percent larger than 25%, with respect to the undisturbed O3 value when there were no significant CFCs in the stratosphere (~ in the 1960s)"[165] instead of the general definition of 220 Dobson units or lower. Dr Marta Abalos Alvarez has added "Ozone depletion in the tropics is nothing new and is mainly due to the acceleration of the Brewer-Dobson circulation."
Depletion caused by wildfire smoke
[edit]Analyzing the atmospheric impacts of the 2019–2020 Australian bushfire season, scientists led by MIT researcher Susan Solomon found the smoke destroyed 3–5% of ozone in affected areas of the Southern Hemisphere. Smoke particles absorb hydrogen chloride and act as a catalyst to create chlorine radicals that destroy ozone.[166][167][168][169]
Ozone depletion and global warming
[edit]Among others, Robert Watson had a role in the science assessment and in the regulation efforts of ozone depletion and global warming.[89] Prior to the 1980s, the EU, NASA, NAS, UNEP, WMO and the British government had dissenting scientific reports and Watson played a role in the process of unified assessments. Based on the experience with the ozone case, the IPCC started to work on a unified reporting and science assessment[89] to reach a consensus to provide the IPCC Summary for Policymakers.
There are various areas of linkage between ozone depletion and global warming science:
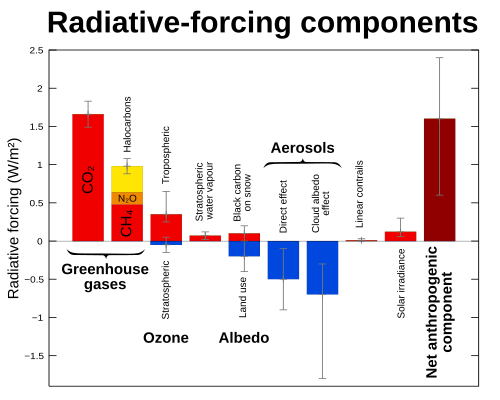
- The same CO
2 radiative forcing that produces global warming is expected to cool the stratosphere.[170] This cooling, in turn, is expected to produce a relative increase in ozone (O
3) depletion in polar areas and the frequency of ozone holes.[171] - Conversely, ozone depletion represents a radiative forcing of the climate system. There are two opposing effects: Reduced ozone causes the stratosphere to absorb less solar radiation, thus cooling the stratosphere while warming the troposphere; the resulting colder stratosphere emits less long-wave radiation downward, thus cooling the troposphere. Overall, the cooling dominates; the IPCC concludes "observed stratospheric O
3 losses over the past two decades have caused a negative forcing of the surface-troposphere system"[33] of about −0.15 ± 0.10 watts per square meter (W/m2).[122] - One of the strongest predictions of the greenhouse effect is that the stratosphere will cool.[170] Although this cooling has been observed, it is not trivial to separate the effects of changes in the concentration of greenhouse gases and ozone depletion since both will lead to cooling. However, this can be done by numerical stratospheric modeling. Results from the National Oceanic and Atmospheric Administration's Geophysical Fluid Dynamics Laboratory show that above 20 km (12 mi), the greenhouse gases dominate the cooling.[172]
- Ozone depleting chemicals are also often greenhouse gases. The increases in concentrations of these chemicals have produced 0.34 ± 0.03 W/m2 of radiative forcing, corresponding to about 14 percent of the total radiative forcing from increases in the concentrations of well-mixed greenhouse gases.[122]
- The long-term modeling of the process, its measurement, study, design of theories and testing take decades to document, gain wide acceptance, and ultimately become the dominant paradigm. Several theories about the destruction of ozone were hypothesized in the 1980s, published in the late 1990s, and are now[when?] being investigated. Drew Schindell and Paul Newman of Goddard Space Flight Center proposed a theory in the late 1990s, using computational modeling methods to model ozone destruction, which accounted for 78 percent of the ozone destroyed. Further refinement of that model accounted for 89 percent of the ozone destroyed, but pushed back the estimated recovery of the ozone hole from 75 years to 150 years. (The model includes the lack of stratospheric flight due to depletion of fossil fuels.)[citation needed]
In 2019, NASA reported that there was no significant relation between size of the ozone hole and climate change.[9]
Misconceptions
[edit]CFC weight
[edit]Since CFC molecules are heavier than air (nitrogen or oxygen), it is commonly believed that the CFC molecules cannot reach the stratosphere in significant amounts.[173] However, atmospheric gases are not sorted by weight at these altitudes; the forces of wind can fully mix the gases in the atmosphere. Some of the heavier CFCs are not evenly distributed.[174]
Percentage of human-made chlorine
[edit]
Another misconception is that natural sources of chlorine are several times larger than human-made ones. While this statement is true for tropospheric chlorine, that is irrelevant to ozone depletion, which is only affected by stratospheric chlorine. Chlorine from ocean spray is soluble and thus is washed by rainfall before it reaches the stratosphere. CFCs, in contrast, are insoluble and long-lived, allowing them to reach the stratosphere. In the lower atmosphere, there is much more chlorine from CFCs and related haloalkanes than there is in HCl from salt spray, and in the stratosphere halocarbons are dominant.[175] Only methyl chloride, which is one of these halocarbons, has a mainly natural source,[176] and it is responsible for about 20 percent of the chlorine in the stratosphere; the remaining 80 percent comes from human-made sources.
Very violent volcanic eruptions can inject HCl into the stratosphere, but researchers have shown that the contribution is not significant compared to that from CFCs.[177] A similar erroneous assertion is that soluble halogen compounds from the volcanic plume of Mount Erebus on Ross Island, Antarctica are a major contributor to the Antarctic ozone hole.[177]
Nevertheless, a 2015 study showed that the role of Mount Erebus volcano in the Antarctic ozone depletion was probably underestimated. Based on the NCEP/NCAR reanalysis data over the last 35 years and by using the NOAA HYSPLIT trajectory model, researchers showed that gas emissions from the volcano (including hydrogen chloride (HCl)) can reach the Antarctic stratosphere via high-latitude cyclones and then the polar vortex. Depending on the level of its volcanic activity, the additional annual HCl mass entering the stratosphere from Erebus varies from 1.0 to 14.3 kt.[178]
First observation
[edit]G.M.B. Dobson mentioned that when springtime ozone levels in the Antarctic over Halley Bay were first measured in 1956, he was surprised to find that they were only about 320 DU, about 150 DU below typical spring Arctic levels of around 450 DU. What Dobson observed was not an ozone hole but in fact a typical annual maximum Antarctic ozone concentration: actual ozone hole values are in the 150–100 DU range.[179] While Arctic ozone concentrations vary on a smooth annual cycle from around 300 to 450 DU, peaking in the northern hemisphere spring, Antarctic concentrations drop sharply in the southern hemisphere spring from highs of around 300 DU to much lower values. Peak values are not reached again until December.[180]
Location of hole
[edit]Some people thought that the ozone hole should be above the sources of CFCs. However, CFCs are well mixed globally in the troposphere and stratosphere. The reason for occurrence of the ozone hole above Antarctica is not because there are more CFCs concentrated but because the low temperatures help form polar stratospheric clouds.[181] In fact, there are findings of significant and localized "ozone holes" above other parts of the Earth, such as above Central Asia.[182]
Awareness campaigns
[edit]Public misconceptions and misunderstandings of complex issues like ozone depletion are common. The limited scientific knowledge of the public led to confusion about global warming[183] or the perception of global warming as a subset of the "ozone hole".[184] In the beginning, classical green NGOs refrained from using CFC depletion for campaigning, as they assumed the topic was too complicated.[89] They became active much later, e.g. in Greenpeace's support for a CFC-free refrigerator produced by the former East German company VEB dkk Scharfenstein.[89][185]
The metaphors used in the CFC discussion (ozone shield, ozone hole) are not "exact" in the scientific sense. The "ozone hole" is more of a depression, less "a hole in the windshield". The ozone does not disappear through the layer, nor is there a uniform "thinning" of the ozone layer. However, they resonated better with non-scientists and their concerns.[90] The ozone hole was seen as a "hot issue" and imminent risk[186] as laypeople feared severe personal consequences such as skin cancer, cataracts, damage to plants, and reduction of plankton populations in the ocean's photic zone. Not only on the policy level, ozone regulation compared to climate change fared much better in public opinion. Americans voluntarily switched away from aerosol sprays before legislation was enforced, while climate change failed to achieve comparable concern and public action.[90] The sudden identification in 1985 that there was a substantial "hole" was widely reported in the press. The especially rapid ozone depletion in Antarctica had previously been dismissed as a measurement error.[133] Scientific consensus was established after regulation.[89]
While the Antarctic ozone hole has a relatively small effect on global ozone, the hole has generated a great deal of public interest because:
- Many have worried that ozone holes might start appearing over other areas of the globe, though to date the only other large-scale depletion is a smaller ozone "dimple" observed during the Arctic spring around the North Pole. Ozone at middle latitudes has declined, but by a much smaller extent (a decrease of about 4–5 percent).
- If stratospheric conditions become more severe (cooler temperatures, more clouds, more active chlorine), global ozone may decrease at a greater pace. Standard global warming theory predicts that the stratosphere will cool.[187]
- When the Antarctic ozone hole breaks up each year, the ozone-depleted air drifts into nearby regions. Decreases in the ozone level of up to 10 percent have been reported in New Zealand in the month following the breakup of the Antarctic ozone hole,[188] with ultraviolet-B radiation intensities increasing by more than 15 percent since the 1970s.[189][190]
World Ozone Day
[edit]In 1994, the United Nations General Assembly voted to designate 16 September as the International Day for the Preservation of the Ozone Layer, or "World Ozone Day".[191] The designation commemorates the signing of the Montreal Protocol[192] on that date in 1987.[193]
See also
[edit]References
[edit]- ^ a b c d e f "Twenty Questions and Answers About the Ozone Layer" (PDF). Scientific Assessment of Ozone Depletion: 2010. World Meteorological Organization. 2011. Archived (PDF) from the original on 2013-03-05. Retrieved March 13, 2015.
- ^ Gruijl, Frank de; Leun, Jan (October 3, 2000). "Environment and health: 3. Ozone depletion and ultraviolet radiation". CMAJ. 163 (7): 851–855. PMC 80511. PMID 11033716 – via www.cmaj.ca.
- ^ Andino, Jean M. (October 21, 1999). "Chlorofluorocarbons (CFCs) are heavier than air, so how do scientists suppose that these chemicals reach the altitude of the ozone layer to adversely affect it ?". Scientific American. 264: 68.
- ^ "Part III. The Science of the Ozone Hole". Retrieved March 5, 2007.
- ^ "Ultraviolet (UV) Radiation". www.cancer.org. Retrieved 2022-04-06.
- ^ "The Montreal Protocol on Substances That Deplete the Ozone Layer". United States Department of State. Retrieved 2022-04-06.
- ^ Banerjee, Antara; et al. (2020). "A pause in Southern Hemisphere circulation trends due to the Montreal Protocol". Vol. 579. Nature. pp. 544–548. doi:10.1038/s41586-020-2120-4.
- ^ a b "The Antarctic Ozone Hole Will Recover". NASA. June 4, 2015. Retrieved 2017-08-05.
- ^ a b Bowden, John (2019-10-21). "Ozone hole shrinks to lowest size since 1982, unrelated to climate change: NASA". The Hill. Retrieved 2019-10-22.
- ^ Ansari, Talal (October 23, 2019). "Ozone Hole Above Antarctica Shrinks to Smallest Size on Record". The Wall Street Journal – via www.wsj.com.
- ^ "The Week". No. 1418. Future PLC. 14 January 2023. p. 2.
- ^ Laboratory (CSL), NOAA Chemical Sciences. "NOAA CSL: Scientific Assessment of Ozone Depletion: 2022". www.csl.noaa.gov. Retrieved 2024-03-24.
- ^ Waserman, Lynda (16 September 1987). "The Ozone Hole – The Montreal Protocol on Substances that Deplete the Ozone Layer". The Ozone Hole. Theozonehole.com. Archived from the original on 2012-09-12. Retrieved 2019-05-15.
- ^ "Background for International Day for the Preservation of the Ozone Layer – 16 September". www.un.org. Retrieved 2019-05-15.
- ^ "Ozone". earthobservatory.nasa.gov. 1999-07-30. Retrieved 2022-04-06.
- ^ Lary, D. J. (2004). "Atmospheric pseudohalogen chemistry". Atmospheric Chemistry & Physics Discussions. 4 (5): 5381. Bibcode:2004ACPD....4.5381L. doi:10.5194/acpd-4-5381-2004.
- ^ "World of Change: Antarctic Ozone Hole". earthobservatory.nasa.gov. 2009-06-01. Retrieved 2020-06-26.
- ^ Newman, Paul A. "Chapter 5: Stratospheric Photochemistry Section 4.2.8 ClX catalytic reactions". In Todaro, Richard M. (ed.). Stratospheric ozone: an electronic textbook. NASA Goddard Space Flight Center Atmospheric Chemistry and Dynamics Branch. Retrieved May 28, 2016.
- ^ Ricaud, P.; Lefèvre, F. (2006). "Fluorine in the Atmosphere". Advances in Fluorine Science. 1: 1–32 See 12–13. doi:10.1016/S1872-0358(06)01001-3. hal-00256296.
Thus, fluorine chemistry does not represent a significant sink for stratospheric ozone. All fluorine released from the source gases ends up in the form of HF, which accumulates in the stratosphere (Fig. 8). ... The high stability of HF makes it an effective tracer of fluorine input in the stratosphere arising from fluorinated anthropogenic gases
- ^ "Q7 What emissions from human activities lead to ozone depletion?" (PDF). 20 Questions: 2010 Update: Section II The Ozone Depletion Process. Chemical Sciences Laboratory, National Oceanic and Atmospheric Administration (NOAA). pp. 3–4. Archived (PDF) from the original on 2021-02-26. Retrieved 22 October 2022.
Iodine is a component of several gases that are naturally emitted from the oceans. Although iodine can participate in ozone destruction reactions, these iodine-containing source gases generally have very short lifetimes and, as a result, only a very small fraction reaches the stratosphere. There are large uncertainties in how these emissions vary with season and geographical region.
- ^ "Stratospheric Ozone Depletion by Chlorofluorocarbons (Nobel Lecture)—Encyclopedia of Earth". Eoearth.org. Archived from the original on September 9, 2011.
- ^ Laboratory (CSL), NOAA Chemical Sciences. "NOAA CSL: Scientific Assessment of Ozone Depletion: 2010". csl.noaa.gov. Retrieved 2024-04-01.
- ^ "The Ozone Hole Tour: Part II. Recent Ozone Depletion". University of Cambridge. Retrieved March 28, 2011.
- ^ a b c Solomon, S.; Ivy, D. J.; Kinnison, D.; Mills, M. J.; Neely Rr, 3rd; Schmidt, A. (June 30, 2016). "Emergence of healing in the Antarctic ozone layer". Science. 353 (6296): 269–274. Bibcode:2016Sci...353..269S. doi:10.1126/science.aae0061. PMID 27365314.
{{cite journal}}: CS1 maint: numeric names: authors list (link) - ^ Mersmann, Katy; Stein, Theo (November 2, 2017). "Warm Air Helped Make 2017 Ozone Hole Smallest Since 1988". nasa.gov. Retrieved December 31, 2017.
- ^ "Spring 2020 brings rare ozone "hole" to the Arctic". www.climate.gov. Archived from the original on April 29, 2020. Retrieved 2022-04-06.
- ^ "U.S. EPA: Ozone Depletion". 2006-09-30. Archived from the original on 2006-09-30. Retrieved 2024-04-01.
- ^ Zafar, A. Mannan; Müller, Rolf; Grooss, Jens-Uwe; Robrecht, Sabine; Vogel, Bärbel; Lehmann, Ralph (January 2018). "The relevance of reactions of the methyl peroxy radical (CH3O2) and methylhypochlorite (CH3OCl) for Antarctic chlorine activation and ozone loss" (PDF). Tellus B: Chemical and Physical Meteorology. 70 (1) 1507391. Bibcode:2018TellB..7007391Z. doi:10.1080/16000889.2018.1507391. ISSN 1600-0889. S2CID 106298119.
- ^ Son, Seok-Woo; Han, Bo-Reum; Garfinkel, Chaim I.; Kim, Seo-Yeon; Park, Rokjin; Abraham, N. Luke; Hideharu Akiyoshi; Archibald, Alexander T.; Butchart, N. (2018). "Tropospheric jet response to Antarctic ozone depletion: An update with Chemistry-Climate Model Initiative (CCMI) models". Environmental Research Letters. 13 (5): 054024. Bibcode:2018ERL....13e4024S. doi:10.1088/1748-9326/aabf21. hdl:20.500.11850/265235. ISSN 1748-9326.
- ^ "Largest-ever Ozone Hole over Antarctica". earthobservatory.nasa.gov. 2000-09-11. Retrieved 2018-11-26.
- ^ a b "Myth: Ozone Depletion Occurs Only In Antarctica". EPA. June 28, 2006. Archived from the original on December 13, 2012. Retrieved March 28, 2011.
- ^ Self, Stephen, et al. (1996). "The Atmospheric Impact of the 1991 Mount Pinatubo Eruption". USGS. Retrieved May 28, 2016.
- ^ a b "Climate Change 2001: Working Group I: The Scientific Basis". Intergovernmental Panel on Climate Change Work Group I. 2001. pp. Chapter 6.4 Stratospheric Ozone. Archived from the original on June 3, 2016. Retrieved May 28, 2016.
- ^ 2008 News, Briefs, and Features. NASA
- ^ "Climate Change 2013: The Physical Science Basis". UNEP. Retrieved May 28, 2016.
- ^ "Scientific Assessment of Ozone Depletion 1998 – Preface". US National Oceanic & Atmospheric Administration. Retrieved 21 December 2012.
- ^ Newman, P. A.; Daniel, J. S.; Waugh, D. W.; Nash, E. R. (2007). "A new formulation of equivalent effective stratospheric chlorine (EESC)" (PDF). Atmos. Chem. Phys. 7 (17): 4537–52. Bibcode:2007ACP.....7.4537N. doi:10.5194/acp-7-4537-2007. S2CID 1934089. Archived (PDF) from the original on 2011-05-11.
- ^ Kauffman, G. B. (2005). "CFCs: On the 75th Anniversary of Their Introduction as Commercial Refrigerants by Thomas Midgley, Jr. (1889–1944)". The Chemical Educator. 10 (3): 217–226. doi:10.1333/s00897050916a.
- ^ The Future of Energy Gases. U.S. Government Printing Office. 1993.
- ^ "chlorofluorocarbons". Encyclopedia.com. Retrieved March 28, 2011.
- ^ Ponting, Clive (15 April 1999). The Twentieth Century: A World History. Macmillan. ISBN 978-0-8050-6088-1.
- ^ Adcock, Karina; Reeves, Claire; Gooch, Lauren; Leedham Elvidge, Emma; Ashfold, Matthew; Brenninkmeijer, Carl; Chou, Charles; Fraser, Paul; Langenfelds, Ray; Mohd Hanif, Norfazrin; O'Doherty, Simon; Oram, David; Ou-Yang, Chang-Feng; Phang, Siew Moi; Samah, Azizan Abu; Röckmann, Thomas; Sturges, William; Laube, Johannes (9 April 2018). "Continued increase of CFC-113a (CCl3CF3) mixing ratios in the global atmosphere: emissions, occurrence and potential sources". Atmospheric Chemistry and Physics. 18 (7): 4737–4751. Bibcode:2018ACP....18.4737A. doi:10.5194/acp-18-4737-2018.
- ^ McGrath, Matt (2019-05-22). "China confirmed as source of rise in CFCs". BBC News. Retrieved 2020-04-08.
- ^ "China factories releasing thousands of tonnes of illegal CFC gases, study finds". The Guardian. 2019-05-23. Retrieved 2020-04-08.
- ^ Stoye, Emma (May 22, 2019). "China identified as source of unexpected rise in CFC emissions". Chemistry World. Retrieved 2020-04-08.
- ^ a b c Ferreira, Jose P.; Huang, Ziyu; Nomura, Ken-ichi; Wang, Joseph (11 June 2024). "Potential Ozone Depletion From Satellite Demise During Atmospheric Reentry in the Era of Mega-Constellations". Geophysical Research Letters. 51 (11) e2024GL109280. Bibcode:2024GeoRL..5109280F. doi:10.1029/2024GL109280.
- ^ a b Puiu, Tibi (2015-02-18). "Short-lived chemicals that burn a hole in the ozone layer are on the rise". ZME Science. Retrieved 2025-02-09.
- ^ Nash, Eric; Newman, Paul (September 19, 2001). "NASA Confirms Arctic Ozone Depletion Trigger". Image of the Day. NASA. Retrieved April 16, 2011.
- ^ "Emissions of a banned ozone-depleting gas are back on the decline". NOAA Research News. 11 February 2021.
- ^ Sparling, Brien (June 26, 2001). "Antarctic Ozone Hole". NASA Advanced Supercomputing Department. Archived from the original on March 12, 2005.
- ^ Parson, Robert (December 16, 1997). "Antarctic ozone-depletion FAQ, section 7". Faqs.org. Retrieved April 16, 2011.
- ^ Toon, Owen B.; Turco, Richard P. (June 1991). "Polar Stratospheric Clouds and Ozone Depletion" (PDF). Scientific American. 264 (6): 68–74. Bibcode:1991SciAm.264f..68T. doi:10.1038/scientificamerican0691-68. Archived from the original (PDF) on February 25, 2011. Retrieved April 16, 2011.
- ^ Sumi´nska-Ebersoldt; Lehmann, R.; Wegner, T.; Grooß, J.-U.; Hösen, E.; Weigel, R.; Frey, W.; Griessbach, S.; Mitev, V.; Emde, C.; Volk, C. M.; Borrmann, S.; Rex, M.; Stroh, F.; von Hobe, M. (July 2011). "ClOOCl photolysis at high solar zenith angles: analysis of the RECONCILE self-match flight". Atmos. Chem. Phys. 12 (3): 1353–1365. Bibcode:2012ACP....12.1353S. doi:10.5194/acp-12-1353-2012.
- ^ "Ozone Facts: What is the Ozone Hole?". Ozone Hole Watch. NASA. November 18, 2009. Archived from the original on November 20, 2010. Retrieved April 16, 2011.
- ^ Rowland, Frank Sherwood (May 29, 2006). "Stratospheric ozone depletion". Phil. Trans. R. Soc. B. 361 (1469): 769–790. doi:10.1098/rstb.2005.1783. PMC 1609402. PMID 16627294.
Free radical reactions for ozone removal: Reaction 4.1
- ^ Banerjee, Antara (25 March 2020). "A pause in Southern Hemisphere circulation trends due to the Montreal Protocol". Nature. 579 (7800): 544–548. Bibcode:2020Natur.579..544B. doi:10.1038/s41586-020-2120-4. PMID 32214266. S2CID 214648481. Retrieved 31 March 2020.
- ^ "Ozone and You | Ozone Secretariat". ozone.unep.org. Retrieved 2022-04-06.
- ^ "Health and Environmental Effects of Ozone Layer Depletion". EPA. 2013-02-15. Archived from the original on August 30, 2009. Retrieved September 26, 2013.
- ^ "Reconstruction of Paleobehavior of Ozonosphere Based on Response to UV-B Radiation Effect in Dendrochronologic Signal" (PDF). Atmospheric Radiation Measurement, USA. Archived (PDF) from the original on 2004-10-29. Retrieved May 28, 2016.
- ^ The HIPERION Report (PDF) (Report). Ecuadorian Civilian Space Agency. 2008. Archived (PDF) from the original on 2017-12-31.
- ^ Lilley, Ray (October 5, 2000). "Ozone Hole Over City for First Time". Associated Press. Retrieved March 13, 2015.
- ^ Bothwell, Max L.; Sherbot, Darren M. J.; Pollock, Colleen M. (July 6, 1994). "Ecosystem Response to Solar Ultraviolet-B Radiation: Influence of Trophic-Level Interactions". Science. 265 (5168): 97–100. Bibcode:1994Sci...265...97B. doi:10.1126/science.265.5168.97. PMID 17774696. S2CID 43683982.
- ^ "Ozone Pollution: An Insidious and Growing Threat to Biodiversity". Yale E360. Retrieved 2024-04-12.
- ^ Bais, F.; Luca, R. M.; Bornman, J. F.; Williamson, C. E.; Sulzberger, B.; Austin, A. T.; Wilson, S. R.; Andrady, A. L.; Bernhard, G.; McKenzie, R. L.; Aucamp, P. J. (2018-02-14). "Environmental effects of ozone depletion, UV radiation and interactions with climate change: UNEP Environmental Effects Assessment Panel, update 2017". Photochemical & Photobiological Sciences. 17 (2): 127–179. Bibcode:2018PhPhS..17..127B. doi:10.1039/c7pp90043k. ISSN 1474-905X. PMC 6155474. PMID 29404558.
- ^ de Gruijl, Frank R. (Summer 1995). "Impacts of a Projected Depletion of the Ozone Layer". Consequences. 1 (2).
- ^ Fears, T. R.; Bird, C. C.; Guerry d, 4th; Sagebiel, R. W.; Gail, M. H.; Elder, D. E.; Halpern, A.; Holly, E. A.; Hartge, P.; Tucker, M. A. (2002). "Average midrange ultraviolet radiation flux and time outdoors predict melanoma risk". Cancer Res. 62 (14): 3992–6. PMID 12124332.
{{cite journal}}: CS1 maint: numeric names: authors list (link) - ^ Abarca, J. F.; Casiccia, C. C. (December 2002). "Skin cancer and ultraviolet-B radiation under the Antarctic ozone hole: southern Chile, 1987–2000". Photodermatol Photoimmunol Photomed. 18 (6): 294–302. doi:10.1034/j.1600-0781.2002.02782.x. PMID 12535025. S2CID 25748826.
- ^ West, S. K.; Duncan, D. D.; Muñoz, B.; Rubin, G. S.; Fried, L. P.; Bandeen-Roche, K.; Schein, O. D. (1998). "Sunlight exposure and risk of lens opacities in a population-based study: the Salisbury Eye Evaluation project". JAMA. 280 (8): 714–8. doi:10.1001/jama.280.8.714. PMID 9728643.
- ^ Dobson, R. (2005). "Ozone depletion will bring big rise in number of cataracts". BMJ. 331 (7528): 1292–1295. doi:10.1136/bmj.331.7528.1292-d. PMC 1298891.
- ^ "Ozone: Good Up High, Bad Nearby" (PDF). EPA. Archived from the original on June 2, 2013. Retrieved March 13, 2015.
- ^ Webb, Ann R.; Engelsen, Ola (2006). "Calculated Ultraviolet Exposure Levels for a Healthy Vitamin D Status". Photochemistry and Photobiology. 82 (6): 1697–1703. doi:10.1111/j.1751-1097.2006.tb09833.x. ISSN 1751-1097. PMID 16958558. S2CID 222102318.
- ^ Melamed, M. L.; Michos, E. D.; Post, W.; Astor, B. (2008). "25-hydroxyl Vitamin D Levels and the Risk of Mortality in the General Population". Arch. Intern. Med. 168 (15): 1629–37. doi:10.1001/archinte.168.15.1629. PMC 2677029. PMID 18695076.
- ^ Vieth, R. (1999). "Vitamin D supplementation, 25-hydroxyvitamin D concentrations, and safety". American Journal of Clinical Nutrition. 69 (5): 842–56. doi:10.1093/ajcn/69.5.842. PMID 10232622.
- ^ "Sunburned whales: Troubling environment news of the week". The Washington Post. BlogPost (blog). November 11, 2010. Archived from the original on January 7, 2012. Retrieved March 28, 2011.
- ^ Thomas, Abbie (November 10, 2010). "Whales showing more sun damage". Abc.net.au. Retrieved March 28, 2011.
- ^ Mayer, S. J. (1992-08-08). "Stratospheric ozone depletion and animal health". Veterinary Record. 131 (6): 120–122. doi:10.1136/vr.131.6.120 (inactive 12 July 2025). ISSN 0042-4900. PMID 1529513. S2CID 22177257.
{{cite journal}}: CS1 maint: DOI inactive as of July 2025 (link) - ^ Sinha, R. P.; Singh, S. C.; Häder, D. P. (1999). "Photoecophysiology of cyanobacteria". Recent Research Developments in Photochemistry and Photobiology. 3: 91–101.
- ^ "Health and Environmental Effects of Ozone Layer In Plants". U.S Environmental Protection Agency. 2013-02-15. Archived from the original on August 30, 2009. Retrieved November 12, 2013.
- ^ Barnes, P. W.; Robson, T. M.; Neale, P. J.; Williamson, C. E.; Zepp, R. G.; Madronich, S.; Wilson, S. R.; Andrady, A. L.; Heikkilä, A. M.; Bernhard, G. H.; Bais, A. F. (2022-03-01). "Environmental effects of stratospheric ozone depletion, UV radiation, and interactions with climate change: UNEP Environmental Effects Assessment Panel, Update 2021". Photochemical & Photobiological Sciences. 21 (3): 275–301. Bibcode:2022PhPhS..21..275B. doi:10.1007/s43630-022-00176-5. ISSN 1474-9092. PMC 8860140. PMID 35191005.
- ^ Searles, Peter S.; Flint, Stephan D.; Caldwell, Martyn M. (2001-03-01). "A meta-analysis of plant field studies simulating stratospheric ozone depletion". Oecologia. 127 (1): 1–10. Bibcode:2001Oecol.127....1S. doi:10.1007/s004420000592. ISSN 1432-1939. PMID 28547159. S2CID 7049908.
- ^ Xiong, Fusheng S.; Day, Thomas A. (2001-02-01). "Effect of Solar Ultraviolet-B Radiation during Springtime Ozone Depletion on Photosynthesis and Biomass Production of Antarctic Vascular Plants". Plant Physiology. 125 (2): 738–751. doi:10.1104/pp.125.2.738. ISSN 0032-0889. PMC 64875. PMID 11161031.
- ^ United Nations Environment Programme, Environmental Effects Assessment Panel (2017). "Environmental effects of ozone depletion and its interactions with climate change: Progress report, 2016". Photochemical & Photobiological Sciences. 16 (2): 107–145. Bibcode:2017PhPhS..16..107U. doi:10.1039/C7PP90001E. hdl:11336/183828. ISSN 1474-905X. PMC 6400464. PMID 28124708.
- ^ Allen, Damian J.; Nogués, Salvador; Baker, Neil R. (1998-11-01). "Ozone depletion and increased UV-B radiation: is there a real threat to photosynthesis?". Journal of Experimental Botany. 49 (328): 1775–1788. doi:10.1093/jxb/49.328.1775. ISSN 0022-0957.
- ^ Björn, Lars Olof (1996-12-01). "Effects of ozone depletion and increased UV-B on terrestrial ecosystems". International Journal of Environmental Studies. 51 (3): 217–243. Bibcode:1996IJEnS..51..217B. doi:10.1080/00207239608711082. ISSN 0020-7233.
- ^ Bornman, J. F.; Barnes, P. W.; Robinson, S. A.; Ballaré, C. L.; Flint, S. D.; Caldwell, M. M. (2015). "Solar ultraviolet radiation and ozone depletion-driven climate change: effects on terrestrial ecosystems". Photochemical & Photobiological Sciences. 14 (1): 88–107. Bibcode:2015PhPhS..14...88B. doi:10.1039/C4PP90034K. hdl:20.500.11937/28562. ISSN 1474-905X. PMID 25435216. S2CID 10176384.
- ^ "Environmental effects of ozone depletion and its interactions with climate change: 2010 assessment: Executive summary". Photochemical & Photobiological Sciences. 10 (2): 178–181. 2011. Bibcode:2011PhPhS..10..178.. doi:10.1039/c0pp90043e. ISSN 1474-905X. PMID 21253669. S2CID 40238255.
- ^ Björn, L. O.; Callaghan, T. V; Gehrke, C.; Johanson, U.; Sonesson, M. (November 1999). "Ozone depletion, ultraviolet radiation and plant life". Chemosphere – Global Change Science. 1 (4): 449–454. Bibcode:1999ChGCS...1..449B. doi:10.1016/s1465-9972(99)00038-0. ISSN 1465-9972.
- ^ Bornman, Janet F.; Barnes, Paul W.; Robson, T. Matthew; Robinson, Sharon A.; Jansen, Marcel A. K.; Ballaré, Carlos L.; Flint, Stephan D. (2019). "Linkages between stratospheric ozone, UV radiation and climate change and their implications for terrestrial ecosystems". Photochemical & Photobiological Sciences. 18 (3): 681–716. Bibcode:2019PhPhS..18..681B. doi:10.1039/C8PP90061B. hdl:10138/307029. ISSN 1474-905X. PMID 30810560. S2CID 73506953.
- ^ a b c d e f Reiner Grundmann Technische Problemlösung, Verhandeln und umfassende Problemlösung, generic problem solving capability) Archived 2016-03-03 at the Wayback Machine in Gesellschaftliche Komplexität und kollektive Handlungsfähigkeit (Societys complexity and collective ability to act), ed. Schimank, U. (2000). Frankfurt/Main, Germany: Campus, pp. 154–182, book summary at the Max Planck Gesellschaft Archived 2014-10-12 at the Wayback Machine.
- ^ a b c d Ungar, Sheldon (1 July 2000). "Knowledge, ignorance and the popular culture: climate change versus the ozone hole". Public Understanding of Science. 9 (3): 297–312. doi:10.1088/0963-6625/9/3/306. S2CID 7089937.
- ^ National Academy of Sciences (1976). Halocarbons, effects on stratospheric ozone. Washington, DC. ISBN 978-0-309-02532-4. Retrieved May 28, 2016.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ a b c d Morrisette, Peter M. (1989). "The Evolution of Policy Responses to Stratospheric Ozone Depletion". Natural Resources Journal. 29: 793–820. Archived from the original on June 8, 2009. Retrieved April 20, 2010.
- ^ Sawchuk, Arthur R. (December 19, 1994). "Voluntary Initiatives to Reduce Greenhouse Gas Emissions" (PDF). Archived from the original (PDF) on July 6, 2011. Retrieved 2010-06-03. DuPont Canada Incorporated.
- ^ Shabecoff, Philip (November 5, 1986). "U.S. Report Predicts Rise in Skin Cancer with Loss of Ozone". The New York Times. p. A1. Retrieved January 10, 2013.
- ^ a b Grundmann, Reiner (2001). Transnational Environmental Policy: the ozone layer. New York: Routledge. ISBN 978-0-415-22423-9.
- ^ a b "Amendments to the Montreal Protocol | Ozone Layer Protection | US EPA". Epa.gov. June 28, 2006. Archived from the original on September 25, 2008. Retrieved March 28, 2011.
- ^ Gareau, Brian J. (2010). "A critical review of the successful CFC phase-out versus the delayed methyl bromide phase-out in the Montreal Protocol". International Environmental Agreements: Politics, Law and Economics. 10 (3): 209–231. Bibcode:2010IEAPL..10..209G. doi:10.1007/s10784-010-9120-z. S2CID 153692785.
- ^ Decanio, Stephen J.; Norman, Catherine S. (July 2005). "Economics of the 'Critical use' of Methyl Bromide under the Montreal Protocol". Contemporary Economic Policy. 23 (3): 376–393. doi:10.1093/cep/byi028.
- ^ Sarma, K. Madhava, "Compliance with the Multilateral Environmental Agreements to Protect the Ozone Layer" in Ulrich Beyerlin et al. Ensuring Compliance with Multilateral Environmental Agreements. Leiden: Martinus Nijhoff 2006.
- ^ Mate, John (2001). "Making a Difference: A Case Study of the Greenpeace Ozone Campaign". Review of European Community and International Environmental Law. 10 (2): 190–198. doi:10.1111/1467-9388.00275.
- ^ Currie, Duncan E. J. (2005) "The Experience of Greenpeace International" in Tullio Treves et al. (eds.) Civil Society, International Courts, and Compliance Bodies, The Hague, The Netherlands: TMC Asser.
- ^ Benedick, Richard Elliot (1991) Ozone Diplomacy. Cambridge, Massachusetts: Harvard University.
- ^ a b "Happy birthday, Greenfreeze!". Greenpeace International. Retrieved May 28, 2016.
- ^ Stafford, Edwin R.; Hartman, Cathy L.; Liang, Ying (2016-10-10). "Forces driving environmental innovation diffusion in China: The case of Greenfreeze" (PDF). Business Horizons. 46 (2): 47–56. doi:10.1016/S0007-6813(03)00009-0. Archived from the original (PDF) on 2016-10-10.
- ^ "Climate-Friendly Greenfreezers Come to the United States". NBC New York. 2 October 2008. Retrieved May 28, 2016.
- ^ a b c "Greenpeace USA". Greenpeace.org. September 23, 2015. Retrieved September 27, 2015.
- ^ a b "Greenfreeze: A Revolution In Domestic Refrigeration". Ecomall.com. January 1, 1995. Retrieved May 28, 2016.
- ^ "Natural Refrigerants – Businesses". Greenpeace International. Retrieved May 28, 2016.
- ^ "La Historia del "Greenfreeze"". Ilustrados!. Archived from the original on September 12, 2015. Retrieved September 27, 2015.
- ^ "Lanzan la primera de las "Propuestas Greenpeace": la heladera "Greenfreeze" | Greenpeace Argentina". Greenpeace.org. Retrieved September 27, 2015.
- ^ "Use of Ozone Depleting Substances in Laboratories. TemaNord 516/2003" (PDF). Norden.org. January 1, 2003. Archived from the original on February 27, 2008. Retrieved March 28, 2011.
- ^ "Der Greenfreeze – endlich in den USA angekommen". Greenpeace (in German). 14 November 2014. Retrieved May 28, 2016.
- ^ "Discurso de Frank Guggenheim no lançamento do Greenfreeze". Brasil. Archived from the original on September 24, 2015. Retrieved May 28, 2016.
- ^ "SNAP Program Chronology | Alternatives / SNAP | US EPA". Epa.gov. 2014-10-15. Archived from the original on June 11, 2002. Retrieved September 27, 2015.
- ^ "Greenfreeze F-Gas Victory! Greener Refrigerators Finally Legal in the U.S." Greenpeace USA. December 14, 2011. Archived from the original on January 29, 2012. Retrieved January 1, 2018.
- ^ "GE Opening a Door to a Future of Cleaner Home Refrigeration" (Press release). Archived from the original on June 5, 2011. Retrieved August 24, 2014.
- ^ "EUR-Lex – 32009R1005 – EN – EUR-Lex". eur-lex.europa.eu. Retrieved 2022-12-07.
- ^ Molina, M.; Zaelke, D.; Sarma, K. M.; Andersen, S. O.; Ramanathan, V.; Kaniaru, D. (2009). "Reducing abrupt climate change risk using the Montreal Protocol and other regulatory actions to complement cuts in CO2 emissions". Proceedings of the National Academy of Sciences. 106 (49): 20616–20621. Bibcode:2009PNAS..10620616M. doi:10.1073/pnas.0902568106. PMC 2791591. PMID 19822751.
- ^ Norman, Catherine; Decanio, Stephen; Fan, Lin (2008). "The Montreal Protocol at 20: Ongoing opportunities for integration with climate protection". Global Environmental Change. 18 (2): 330–340. Bibcode:2008GEC....18..330N. doi:10.1016/j.gloenvcha.2008.03.003.
- ^ Estrada, Francisco (2013). "Statistically derived contributions of diverse human influences to twentieth-century temperature changes". Nature Geoscience. 6 (12): 1050–1055. Bibcode:2013NatGe...6.1050E. doi:10.1038/ngeo1999. hdl:2144/27169. S2CID 130224979.
- ^ "NOAA Study Shows Nitrous Oxide Now Top Ozone-Depleting Emission". Noaanews.noaa.gov. August 27, 2009. Retrieved April 6, 2011.
- ^ a b c Naik, Vaishali; Szopa, Sophie; Adhikary, Bhupesh; Artaxo Netto, Paulo Eduardo; et al. (2021). "Chapter 6: Short-lived climate forcers" (PDF). IPCC AR6 WG1 2021.
- ^ "CNW Group | CANADIAN SPACE AGENCY | Canada's SCISAT satellite explains 2006 ozone-layer depletion". 2007-12-09. Archived from the original on 2007-12-09. Retrieved 2024-04-01.
- ^ "Ozone hole set to close". Space Daily. Space Media Network. 12 November 2019. Retrieved 8 December 2019.
- ^ Baker, Harry (7 October 2023). "'One of the biggest on record': Ozone hole bigger than North America opens above Antarctica". livescience.com. Retrieved 10 October 2023.
- ^ "Ozone layer recovery is on track, helping avoid global warming by 0.5°C". UN Environment Programme (UNEP). 9 January 2023. Retrieved 19 April 2025.
- ^ Lipkin, Richard (October 7, 1995). SST emissions cut stratospheric ozone. (The introduction of 500 new supersonic transport planes by 2015 could deplete the ozone layer by as much as 1%) Archived 2023-01-07 at the Wayback Machine. Science News.
- ^ "Increase in supersonic jets could be threat to ozone U-2 plane trails Concorde, studies exhaust particles". The Baltimore Sun. Newsday. October 8, 1995. Archived from the original on September 1, 2016. Retrieved December 21, 2012.
- ^ "Du Pont: A case study in the 3D corporate strategy". Greenpeace. 1997. Archived from the original on April 6, 2012.
- ^ Roan, Sharon (1989) Ozone crisis: The 15-year evolution of a sudden global emergency, New York: Wiley, p. 56, ISBN 0-471-52823-4.
- ^ Causes and Effects of Stratospheric Ozone Reduction: An Update. National Research Council. 1982. p. Summary, 3. doi:10.17226/319. ISBN 978-0-309-03248-3.
- ^ Farman, J. C.; Gardiner, B. G.; Shanklin, J. D. (1985). "Large losses of total ozone in Antarctica reveal seasonal ClOx/NOx interaction". Nature. 315 (6016): 207–210. Bibcode:1985Natur.315..207F. doi:10.1038/315207a0. S2CID 4346468.
- ^ a b Zehr, Stephen C. (1994). "Accounting for the Ozone Hole: Scientific Representations of an Anomaly and Prior Incorrect Claims in Public Settings". The Sociological Quarterly. 35 (4): 603–619. doi:10.1111/j.1533-8525.1994.tb00419.x. JSTOR 4121521.
- ^ Bhartia, Pawan Kumar; McPeters, Richard D. (2018). "The discovery of the Antarctic Ozone Hole". Comptes Rendus Geoscience. 350 (7). Elsevier BV: 335–340. Bibcode:2018CRGeo.350..335B. doi:10.1016/j.crte.2018.04.006. ISSN 1631-0713.
- ^ History and politics Archived 2016-10-05 at the Wayback Machine accessed September 30, 2016.
- ^ Solomon, P. M.; Connor, B.; De Zafra, R. L.; Parrish, A.; Barrett, J.; Jaramillo, M. (1987). "High concentrations of chlorine monoxide at low altitudes in the Antarctic spring stratosphere: Secular variation". Nature. 328 (6129): 411–413. Bibcode:1987Natur.328..411S. doi:10.1038/328411a0. S2CID 4335797.
- ^ Reddy, Jeevananda (4 November 2008). Climate Change Myths and Realities. p. 32. Retrieved 20 December 2018.
- ^ "Ozone hole closing up, research shows". ABC News. Australian Broadcasting Commission. November 16, 2007. Archived from the original on July 15, 2012.
- ^ "New report highlights two-way link between ozone layer and climate change". UNEP News Center. November 16, 2010. Archived from the original on December 5, 2010. Retrieved September 18, 2010.
- ^ "NOAA, NASA: Antarctic ozone hole second smallest in 20 years". October 24, 2012.
- ^ Kuttippurath, Jayanarayanan; Nair, Prijitha J. (2017-04-03). "The signs of Antarctic ozone hole recovery". Scientific Reports. 7 (1): 585. Bibcode:2017NatSR...7..585K. doi:10.1038/s41598-017-00722-7. ISSN 2045-2322. PMC 5429648. PMID 28373709.
- ^ Kuttippurath, J.; Kumar, P.; Nair, P. J.; Pandey, P. C. (2018-11-21). "Emergence of ozone recovery evidenced by reduction in the occurrence of Antarctic ozone loss saturation". npj Climate and Atmospheric Science. 1 (1): 42. Bibcode:2018npCAS...1...42K. doi:10.1038/s41612-018-0052-6. ISSN 2397-3722.
- ^ "Study Links Ozone Hole to Weather Shifts". The Earth Institute – Columbia University. April 22, 2011. Retrieved December 21, 2012.
- ^ "Study Links Ozone Hole to Weather Shifts – The Earth Institute – Columbia University". www.earth.columbia.edu. Retrieved 2022-07-13.
- ^ Schiermeier, Quirin (2005). "Solar wind hammers the ozone layer". Nature: news050228–12. doi:10.1038/news050228-12. Retrieved May 28, 2016.
- ^ Dell'Amore, Christine (March 22, 2011). "First North Pole Ozone Hole Forming?". National Geographic. Archived from the original on March 24, 2011. Retrieved April 6, 2011.
- ^ a b Helmholtz Association of German Research Centres (March 14, 2011). "Arctic on the verge of record ozone loss". Science Daily. Retrieved April 6, 2011.
- ^ "The Arctic Ozone Sieve: More Global Weirding?". Scienceblogs.com. March 25, 2011. Archived from the original on April 4, 2011. Retrieved April 6, 2011.
- ^ a b c "Developing ozone hole approaches Europe". EurActiv. Archived from the original on April 4, 2011. Retrieved April 6, 2011.
- ^ a b c "Arctic ozone loss at record level". BBC News Online. October 2, 2011. Archived from the original on October 2, 2011. Retrieved October 3, 2011.
- ^ a b "Unprecedented Arctic Ozone Loss in 2011, Says NASA-Led Study" (Press release). NASA. October 2, 2011. Archived from the original on July 9, 2023. Retrieved July 1, 2016.
- ^ Millan, Luis; Manney, Gloria (2017-05-02). "An assessment of Ozone Mini-holes Representation in Reanalyses Over the Northern Hemisphere". Atmospheric Chemistry and Physics Discussions. 17 (15): 9277. Bibcode:2017ACP....17.9277M. doi:10.5194/acp-2017-341.
- ^ Strahan, S. E.; Douglass, A. R.; Newman, P. A. (2013). "The contributions of chemistry and transport to low arctic ozone in March 2011 derived from Aura MLS observations". Journal of Geophysical Research: Atmospheres. 118 (3): 1563–1576. Bibcode:2013JGRD..118.1563S. doi:10.1002/jgrd.50181. hdl:2060/20120011691. ISSN 2169-8996. S2CID 128447261.
- ^ Zell, Holly (2013-06-07). "NASA Pinpoints Causes of 2011 Arctic Ozone Hole". NASA. Archived from the original on 2019-09-07. Retrieved 2019-10-03.
- ^ Earth, Stephanie Pappas 2013-03-11T23:38:39Z Planet (11 March 2013). "Cause of Odd Arctic Ozone 'Hole' Found". livescience.com. Retrieved 2019-10-03.
{{cite web}}: CS1 maint: numeric names: authors list (link) - ^ Witze, Alexandra (27 March 2020). "Rare ozone hole opens over Arctic — and it's big". Nature. 580 (7801): 18–19. Bibcode:2020Natur.580...18W. doi:10.1038/d41586-020-00904-w. PMID 32221510. S2CID 214694393.
- ^ Harvey, Fiona (2020-04-07). "Record-size hole opens in ozone layer above the Arctic". The Guardian. ISSN 0261-3077. Retrieved 2020-04-08.
- ^ Lubben, Alex (8 April 2020). "Now There's Another Hole in the Ozone Layer. Great". Vice.
- ^ "Earth news: Chinese Scientists Find New Ozone Hole Over Tibet". Elainemeinelsupkis.typepad.com. May 4, 2006. Retrieved April 6, 2011.
- ^ Schiermeier, Quirin (February 22, 1999). "The Great Beyond: Arctic ozone hole causes concern". Blogs.nature.com. Retrieved April 6, 2011.
- ^ Oskin, Becky (July 26, 2012). "Storm Clouds May Punch Holes in Ozone". LiveScience. Retrieved March 13, 2015.
- ^ Fountain, Henry (July 27, 2012). "Storms Threaten Ozone Layer Over U.S., Study Says". The New York Times. p. A1. Retrieved March 13, 2015.
- ^ American Institute of Physics (2022-07-05). "Discovery reveals large, year-round ozone hole over tropics: 'New' ozone hole much larger than Antarctic ozone hole". ScienceDaily. Retrieved 2022-07-06.
- ^ "Expert reaction to research claiming ozone hole over tropics | Science Media Centre".
- ^ Lu, Qing-Bin (2022), "Observation of large and all-season ozone losses over the tropics", AIP Advances, 12 (7) 075006, arXiv:2112.14977, Bibcode:2022AIPA...12g5006L, doi:10.1063/5.0094629, S2CID 251643894.
- ^ Gramling, Carolyn (March 8, 2023). "How wildfires deplete the Earth's ozone layer". ScienceNews.
- ^ Chu, Jennifer (February 28, 2022). "Study reveals chemical link between wildfire smoke and ozone depletion".
- ^ Solomon, Susan; Stone, Kane; Yu, Pengfei; Murphy, D. M.; Kinnison, Doug; Ravishankara, A. R.; Wang, Peidong (March 8, 2023). "Chlorine activation and enhanced ozone depletion induced by wildfire aerosol". Nature. 615 (7951): 259–264. Bibcode:2023Natur.615..259S. doi:10.1038/s41586-022-05683-0. PMID 36890371.
- ^ Solomon, Susan; Dube, Kimberlee; Stone, Kane; Yu, Pengfei; Kinnison, Doug; Toon, Owen B.; Strahan, Susan E.; Rosenlof, Karen H.; Portmann, Robert; Davis, Sean; Randel, William; Bernath, Peter; Boone, Chris; Bardeen, Charles G.; Bourassa, Adam; Daniel Zawada; Doug Degenstein (March 1, 2022). "On the stratospheric chemistry of midlatitude wildfire smoke". PNAS. 119 (10) e2117325119. Bibcode:2022PNAS..11917325S. doi:10.1073/pnas.2117325119. PMC 8915979. PMID 35238658.
- ^ a b Hegerl, Gabriele C.; et al. "Understanding and Attributing Climate Change" (PDF). Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Intergovernmental Panel on Climate Change. p. 675. Archived from the original (PDF) on May 8, 2018. Retrieved February 1, 2008.
- ^ "Ozone Depletion". UNEP/DEWA/Earthwatch. 16 January 2010. Archived from the original on 16 January 2010.
- ^ "The Relative Roles of Ozone and Other Greenhouse Gases in Climate Change in the Stratosphere". Geophysical Fluid Dynamics Laboratory. February 29, 2004. Archived from the original on January 20, 2009. Retrieved March 13, 2015.
- ^ Silverman, Amy (May 4, 1995). "Freon Easy". Phoenix News. Archived from the original on October 11, 2007. Retrieved April 6, 2011.
- ^ Fabian, P.; Borchers, R.; Krüger, B. C.; Lal, S. (1985). "The vertical distribution of CFC-114 (CClF2-CClF2) in the atmosphere". Journal of Geophysical Research. 90 (D7) 13091. Bibcode:1985JGR....9013091F. doi:10.1029/JD090iD07p13091.
- ^ ozone-depletion FAQ, Part II Archived 2009-02-03 at the Wayback Machine, section 4.3
- ^ Yokouchi, Y.; Noijiri, Y.; Barrie, L. A.; Toom-Sauntry, D.; Machida, T.; Inuzuka, Y.; Akimoto, H.; Li, H. -J.; Fujinuma, Y.; Aoki, S. (2000). "A strong source of methyl chloride to the atmosphere from tropical coastal land". Nature. 403 (6767): 295–298. Bibcode:2000Natur.403..295Y. doi:10.1038/35002049. PMID 10659845. S2CID 4318352.
- ^ a b ozone-depletion FAQ, Part II Archived 2009-02-03 at the Wayback Machine, section 4.4
- ^ Zuev, V. V.; Zueva, N. E.; Savelieva, E. S.; Gerasimov, V. V. (2015). "The Antarctic ozone depletion caused by Erebus volcano gas emissions". Atmospheric Environment. 122: 393–399. Bibcode:2015AtmEn.122..393Z. doi:10.1016/j.atmosenv.2015.10.005.
- ^ Dobson, G.M.B. (1968) Exploring the Atmosphere, 2nd Edition, Oxford University Press.
- ^ ozone-depletion FAQ, Part III Archived 2009-02-24 at the Wayback Machine, section 6. faqs.org
- ^ "ozone-depletion FAQ, Antarctic". Faqs.org. Retrieved April 6, 2011.
- ^ Chen, Sheng Bo; Zhao, Liang; Tao, Yu Long (2017), "Stratospheric ozone change over the Tibetan Plateau", Atmospheric Pollution Research, 8 (3): 528–534, Bibcode:2017AtmPR...8..528C, doi:10.1016/j.apr.2016.11.007
- ^ Boyesa, Edward; Stanisstreeta, Martin (1992). "Students' perceptions of global warming". International Journal of Environmental Studies. 42 (4): 287–300. Bibcode:1992IJEnS..42..287B. doi:10.1080/00207239208710804.
- ^ Compare Sheldon Ungar, 2000 and various web sites such as Gavin Schmidt's realclimate complaint in Ozone depletion and global warming 2005 Archived 2014-10-10 at the Wayback Machine or the UCS FAQ on the topic
- ^ Gunkel, Christoph (September 13, 2013). "Öko-Coup aus Ostdeutschland". Der Spiegel (in German). Retrieved 4 September 2015.
- ^ Grundmann, Reiner (May 14, 2007). "Climate Change and Knowledge Politics" (PDF). Environmental Politics. 16 (3): 414–432. Bibcode:2007EnvPo..16..414G. CiteSeerX 10.1.1.535.4984. doi:10.1080/09644010701251656. S2CID 153866225. Archived from the original (PDF) on August 26, 2014.
- ^ "Climate Change 2001: Working Group I: The Scientific Basis". Intergovernmental Panel on Climate Change Work Group I. 2001. pp. Chapter 9.3.2 Patterns of Future Climate Change. Archived from the original on June 3, 2016. Retrieved May 28, 2016.
- ^ Muir, Patricia (March 6, 2008). "Stratospheric Ozone Depletion". Oregon State University. Retrieved April 16, 2011.
- ^ "Long-term increase in summer UV radiation". NIWA. 1999-09-09. Retrieved December 4, 2013.
- ^ McKenzie, Richard; Conner, Brian; Bodeker, Greg (September 10, 1999). "Increased Summertime UV Radiation in New Zealand in Response to Ozone Loss". Science. 285 (5434): 1709–1711. doi:10.1126/science.285.5434.1709. PMID 10481002.
- ^ "International Day for the Preservation of the Ozone Layer, 16 September". www.un.org. Retrieved 2020-04-22.
- ^ Canada, Environment and Climate Change (2015-02-20). "Ozone layer depletion: Montreal Protocol". aem. Retrieved 2020-04-22.
- ^ Andersen, Stephen O.; Sarma, K. Madhava (2002). Protecting the Ozone Layer: The United Nations History. Earthscan. p. 272. ISBN 978-1-84977-226-6.
Sources
[edit]- IPCC (2021). Masson-Delmotte, V.; Zhai, P.; Pirani, A.; Connors, S. L.; et al. (eds.). Climate Change 2021: The Physical Science Basis (PDF). Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge, United Kingdom and New York, NY, US: Cambridge University Press (In Press).
Further reading
[edit]- Andersen, S. O. and K. M. Sarma. (2002). Protecting the Ozone Layer: The United Nations History, Earthscan Press. London, England.[ISBN missing]
- Benedick, Richard Elliot; World Wildlife Fund (U.S.); Institute for the Study of Diplomacy. Georgetown University. (1998). Ozone Diplomacy: New Directions in Safeguarding the Planet (2nd ed.). Harvard University Press. ISBN 978-0-674-65003-9. Retrieved May 28, 2016. (Ambassador Benedick was the Chief U.S. Negotiator at the meetings that resulted in the Montreal Protocol.)
- Chasek, Pamela S., David L. Downie, and Janet Welsh Brown (2013). Global Environmental Politics, 6th ed., Boulder, Colorado: Westview Press.[ISBN missing]
- Gareau, Brian (2013). From Precaution to Profit: Contemporary Challenges to Environmental Protection in the Montreal Protocol. Yale University Press. ISBN 978-0-300-17526-4. Archived from the original on 2013-03-30.
- Grundmann, Reiner (2001). Transnational Environmental Policy: Reconstructing Ozone. Psychology Press. ISBN 978-0-415-22423-9. Retrieved May 28, 2016.
- Haas, P. (1992). Banning chlorofluorocarbons: Epistemic community efforts to protect stratospheric ozone. International Organization, 46(1), 187–224.
- Parson, Edward (2004). Protecting the Ozone Layer: Science and Strategy. Oxford, England: Oxford University Press.[ISBN missing]
External links
[edit]- "WMO/UNEP Scientific Assessments of Ozone Depletion (Latest Report 2022)". Chemical Sciences Laboratory, National Oceanic and Atmospheric Administration (NOAA). NOAA/ESRL Ozone Depletion
- NOAA/ESRL Ozone Depleting Gas Index
- MACC stratospheric ozone service Archived 2014-03-08 at the Wayback Machine delivers maps, datasets and validation reports about the past and current state of the ozone layer.
- Green Cooling Initiative on alternative natural refrigerants cooling technologies
- "Ozone Hole: How We Saved the Planet" premiered April 10, 2019 PBS
- "Whatever happened to the Ozone Hole?", Distillations Podcast Episode 230, April 17, 2018, Science History Institute
Ozone depletion
View on GrokipediaStratospheric Ozone Fundamentals
Natural Ozone Formation and Destruction Cycle
The natural formation and destruction of stratospheric ozone occur through the Chapman cycle, a set of photochemical reactions proposed by Sydney Chapman in 1930 that maintains a dynamic equilibrium in the ozone layer.[14] This cycle is driven by ultraviolet (UV) radiation from the Sun and predominates in the stratosphere between approximately 15 and 35 kilometers altitude, where molecular oxygen (O₂) is abundant.[15] Ozone formation begins with the photodissociation of O₂ by high-energy UV radiation with wavelengths shorter than 240 nanometers:O₂ + hν (λ < 240 nm) → O + O.
The resulting atomic oxygen (O) atoms then react with O₂ molecules in the presence of a third-body collision partner (M, such as N₂ or O₂) to form ozone (O₃):
O + O₂ + M → O₃ + M.
These processes convert solar UV energy into chemical potential, with ozone production occurring primarily during daylight hours and peaking at around 25-30 km altitude.[14] Destruction of ozone proceeds via two main pathways within the cycle. First, ozone absorbs lower-energy UV radiation (wavelengths shorter than 320 nm), dissociating into O₂ and an oxygen atom:
O₃ + hν (λ < 320 nm) → O₂ + O(¹D).
Second, the recombination reaction between atomic oxygen and ozone molecules yields two O₂ molecules:
O + O₃ → 2O₂.
The net result of these reactions is the conversion of UV radiation into heat through subsequent collisional deactivation, shielding Earth's surface from harmful UV wavelengths.[15][14] In steady-state equilibrium, the rates of ozone production and destruction balance, sustaining a global average total ozone column of approximately 300 Dobson units, with local variations due to solar zenith angle, season, and latitude.[14] Although the Chapman mechanism accurately predicts the vertical profile's general shape, it overestimates observed ozone concentrations by a factor of about two, as additional natural catalytic destruction by trace species such as hydroxyl (HOₓ), nitric oxide (NOₓ), and hydrogen radicals contributes to the actual loss rates.[14] This natural cycle operates independently of human influences, regulating ozone levels over geological timescales through variations in solar output and atmospheric dynamics.
Ozone's Role in Absorbing Ultraviolet Radiation
Stratospheric ozone absorbs the majority of solar ultraviolet (UV) radiation in the UV-B (280–315 nm) and UV-C (<280 nm) wavelengths, preventing this biologically damaging energy from reaching Earth's surface.[17] This absorption occurs via photodissociation, wherein an ozone molecule (O₃) interacts with a UV photon, dissociating into an oxygen molecule (O₂) and a free oxygen atom (O), thereby converting the radiation's energy into thermal heat that warms the stratosphere.[18] [19] The process is most efficient in the ozone layer's peak concentration zone, approximately 15–35 km above the surface, where ozone densities reach up to 10 parts per million by volume.[20] This shielding effect is essential for life, as unabsorbed UV-B and UV-C radiation induces DNA damage, including thymine dimer formation, which can lead to mutations, skin cancers, cataracts, and immunosuppression in humans and animals.[21] In aquatic ecosystems, elevated UV penetration harms phytoplankton productivity, disrupting food chains and carbon sequestration.[22] Ozone absorbs over 97% of incoming UV-C and about 90–95% of UV-B, with absorption efficiency decreasing toward longer UV-B wavelengths near 315 nm, allowing minimal amounts to reach the troposphere.[23] The Hartley band, spanning 200–310 nm, dominates this absorption spectrum, underpinning ozone's role as Earth's natural UV filter.[17] The thermal byproduct of UV absorption establishes the stratosphere's temperature inversion, influencing atmospheric circulation patterns, while variations in ozone levels directly modulate surface UV irradiance, with depletions correlating to measurable increases in biologically effective UV doses.[24] Empirical measurements from ground-based spectrometers and satellites confirm that ozone column densities of around 300 Dobson units attenuate UV flux by factors of 10³ for UV-C and 10–10² for UV-B relative to extraterrestrial levels.[25]Causes of Ozone Depletion
Anthropogenic Contributions: Halocarbons and Ozone-Depleting Substances
![Total stratospheric chlorine from NOAA measurements]float-right Halocarbons, particularly chlorofluorocarbons (CFCs), hydrochlorofluorocarbons (HCFCs), and halons, constitute the primary anthropogenic ozone-depleting substances (ODS) responsible for stratospheric ozone loss.[26] These synthetic compounds, developed for uses in refrigeration, air conditioning, aerosol propellants, foam blowing agents, and fire suppression, are stable in the troposphere and ascend to the stratosphere intact.[27] Upon exposure to ultraviolet radiation, they undergo photolysis, releasing chlorine or bromine atoms that initiate catalytic cycles destroying ozone molecules.[28] The catalytic mechanism, first proposed by Mario Molina and F. Sherwood Rowland in 1974, involves chlorine atoms reacting with ozone to form chlorine monoxide and molecular oxygen, followed by chlorine monoxide reacting with atomic oxygen to regenerate chlorine, resulting in net ozone loss without consuming the catalyst.[28] One chlorine atom can destroy thousands of ozone molecules before being sequestered.[29] Atmospheric measurements confirm that anthropogenic halocarbons supply the majority of stratospheric chlorine, with natural sources like methyl chloride contributing less than 20% of total inorganic chlorine, and very short-lived substances adding only about 3%.[30][31] The Montreal Protocol, adopted in 1987 and strengthened by subsequent amendments, mandated the phaseout of ODS production and consumption, achieving over 99% reduction in emissions of key substances like CFCs by 2010 in developed nations.[32] This has led to declining atmospheric concentrations; for instance, HCFCs peaked in 2021 and are now decreasing, while CFCs have fallen by approximately 30% since peak levels.[33] Equivalent effective stratospheric chlorine (EESC), a metric of ozone-depleting potential from halocarbons, has decreased, correlating with observed ozone recovery trends.[34] Despite residual effects from long atmospheric lifetimes (50-100 years for CFCs), the protocol's enforcement has averted further severe depletion.[35]Natural Sources: Volcanic Eruptions, Solar Cycles, and Cosmic Rays
Volcanic eruptions inject sulfur dioxide (SO₂) and ash into the stratosphere, forming sulfate aerosols that catalyze ozone destruction through heterogeneous chemical reactions, particularly by activating chlorine species on aerosol surfaces, akin to processes in polar regions but on a more widespread, temporary scale.[36] The 1991 eruption of Mount Pinatubo in the Philippines released approximately 20 million tons of SO₂, leading to a global stratospheric aerosol loading increase by a factor of 10 and subsequent ozone reductions of 5-8% in mid-latitudes and up to 20% in the tropics by mid-1993, with effects persisting 2-3 years until aerosol clearance.[37][38] These depletions resulted from enhanced ClO production and reduced nitrogen oxides that would otherwise neutralize chlorine, confirming the aerosol-chlorine interaction mechanism via observations of increased NO₂ depletion alongside ozone loss.[39] Smaller eruptions, such as El Chichón in 1982, produced lesser but measurable ozone perturbations of 2-5%, underscoring that eruption magnitude and stratospheric injection height determine impact severity.[40] Solar cycles, with an approximately 11-year periodicity, modulate stratospheric ozone through variations in ultraviolet (UV) irradiance, which drives ozone photochemistry: higher solar activity increases O₂ photodissociation and thus ozone formation in the upper stratosphere, while dynamical transport effects influence lower levels.[41] Satellite and ground-based measurements since the 1960s reveal global total column ozone variations of 1-2% between solar maximum and minimum, with upper stratospheric ozone (above 30 km) exhibiting amplified responses up to 6% due to direct radiative and photochemical forcing.[42][43] During solar minima, reduced UV flux diminishes ozone production, contributing to slight depletions, though this is partially offset by decreased photodissociation of ozone itself; spectral solar irradiance reconstructions confirm that ozone responds primarily to changes in the 170-250 nm wavelength band.[44] These cycles introduce natural variability that must be accounted for in long-term ozone trend analyses, as unadjusted data can overestimate or mask anthropogenic signals.[41] Cosmic rays, high-energy particles from galactic and solar origins, ionize stratospheric molecules, generating nitrogen oxides (NOx) and hydrogen oxides (HOx) that catalytically destroy ozone via cycles such as NO + O → NO₂ + O and subsequent ClO-like radical chains.[45] Satellite data from 1980-2007, spanning two full 11-year cosmic ray modulation cycles modulated inversely by solar activity, show an anticorrelation with total ozone, with higher cosmic ray flux during solar minima linked to 0.2-0.5% global depletions through enhanced NOx production estimated at 10⁷-10⁸ molecules per ionization event.[46][47] Forbush decreases—abrupt cosmic ray reductions—have been observed to cause transient ozone increases of similar magnitude, supporting the ionization-NOx-ozone depletion pathway, though annual contributions remain minor (<<1% of total variability) compared to volcanic or anthropogenic forcings.[45] Peer-reviewed modeling indicates that cosmic ray-induced electron reactions provide a quantifiable mechanism for sporadic ozone loss, particularly in polar regions during high-flux periods, but empirical quantification requires disentangling from concurrent solar UV effects.[47]Secondary Factors: Very Short-Lived Substances and Particle Emissions
Very short-lived substances (VSLS) are halogenated organic compounds with atmospheric lifetimes shorter than 0.5 years, primarily originating from natural oceanic biogenic emissions such as bromoform (CHBr₃) and dibromomethane (CH₂Br₂), though anthropogenic sources like solvents contribute modestly.[48] These compounds are transported convectively into the tropical lower stratosphere, where rapid photolysis releases inorganic halogens that catalyze ozone destruction cycles, particularly bromine monoxide (BrO) cycles which are 40-60 times more efficient per atom than chlorine in depleting ozone.[49] VSLS account for approximately 25% of the total stratospheric bromine loading, supplying about 5 ± 2 parts per trillion by volume (pptv) of bromine, with natural sources dominating over anthropogenic ones.[50] In the tropical lower stratosphere, VSLS chemistry amplifies observed ozone depletion trends by roughly 25% over the 1998-2018 period, counteracting some recovery from phased-out long-lived ozone-depleting substances (ODS).[51] Chlorinated VSLS, such as dichloromethane (CH₂Cl₂), contribute a smaller but non-negligible fraction to stratospheric chlorine, partially offsetting the decline in inorganic chlorine from regulated ODS by 25-30% in recent decades, though their overall impact remains modest compared to bromine effects.[52] [53] Modeling studies indicate that without mitigation of growing anthropogenic VSLS emissions, such as from industrial solvents, future ozone recovery in low latitudes could be delayed, emphasizing the need for targeted controls despite their short lifetimes limiting global transport.[51] Observations and simulations confirm VSLS enhance polar and mid-latitude ozone loss through interactions with existing halogen reservoirs, but their episodic injection via deep convection introduces variability not captured in earlier assessments.[54] Stratospheric particle emissions, primarily sulfate aerosols from volcanic injections or background photochemical production of sulfur dioxide (SO₂), facilitate heterogeneous reactions on their surfaces that activate reservoir species like chlorine nitrate (ClONO₂) into reactive forms such as hypochlorous acid (HOCl), thereby accelerating catalytic ozone loss cycles.[55] Volcanic eruptions, such as the 1991 Mount Pinatubo event which injected ~20 million tons of SO₂, temporarily increased stratospheric aerosol optical depth by a factor of 10, enhancing global ozone depletion by 5-8% over 1-2 years through amplified chlorine activation, though effects wane as particles settle.[55] Background aerosols from tropospheric SO₂ uplift or cosmic ray-induced nucleation maintain a baseline influence, interacting synergistically with halogens to boost bromine-mediated depletion by providing additional reaction sites.[49] Emerging anthropogenic particle sources, including black carbon from increased rocket launches (projected to reach 1,000 launches annually by 2040-2050), could deposit soot in the stratosphere, potentially catalyzing ozone loss via direct radical production and surface reactions, with simulations estimating up to 5% additional depletion if unmitigated.[56] These secondary factors remain episodic and smaller in magnitude than primary ODS but introduce non-linear feedbacks, particularly in cold polar vortices where particle surfaces enhance halogen activation.[57]Observations and Monitoring
Historical and Global Ozone Trends
Ground-based measurements of total column ozone using Dobson spectrophotometers began in the 1930s, with a global network established during the International Geophysical Year of 1957, providing baseline data from the 1960s onward.[1] Satellite observations started in 1970 with the Backscatter Ultraviolet (BUV) instrument and expanded in 1979 via the Total Ozone Mapping Spectrometer (TOMS), enabling comprehensive monitoring of global and regional trends.[58] From the 1964–1980 baseline period, total column ozone (TCO) remained relatively stable until the late 1970s, after which declines became evident, averaging about 3% globally by the early 2000s, with depletions of 4% in Northern Hemisphere mid-latitudes and 5% in Southern Hemisphere mid-latitudes by 2017–2020.[1] Tropical regions (25°S–25°N) showed minimal change, around 1% below baseline.[1] The most dramatic manifestation appeared over Antarctica, where the seasonal ozone hole—defined as TCO below 220 Dobson Units (DU)—was first detected in 1985 through British Antarctic Survey ground measurements, confirming satellite data showing springtime minima dropping to 124 DU that year.[58] The hole deepened rapidly, reaching a record low of 73 DU in September 1994 and expanding to its largest areal extent in 2006, influenced by stratospheric dynamics and peak chlorofluorocarbon (CFC) levels.[58] Globally, TCO declined sharply from 1980 to 1996 amid rising ozone-depleting substances (ODS), with the near-global average (60°S–60°N) falling due to anthropogenic halocarbons.[1] Natural factors like the 11-year solar cycle, quasi-biennial oscillation, and volcanic eruptions (e.g., El Chichón in 1982, Pinatubo in 1991) superimposed short-term variability but did not drive the long-term trend.[1] Following the 1987 Montreal Protocol and its amendments, which phased out ODS production, ODS concentrations peaked in the late 1990s and began declining, leading to signs of recovery.[1] Near-global TCO increased by 0.3% per decade from 1996 to 2020, with stronger gains of 0.8% per decade in Southern Hemisphere mid-latitudes, while Northern Hemisphere mid-latitudes showed no significant trend and tropics remained stable amid rising tropospheric ozone.[1] The Antarctic ozone hole has diminished in size and depth since 2000, with reduced depletion of about 20% observed from 2005 to 2016; the 2024 hole ranked as the seventh-smallest since recovery trends began, though interannual variability persists due to meteorological conditions.[58][59] Arctic regions exhibit episodic spring depletions but no persistent hole, with high variability masking recovery signals through 2021.[1] Projections based on observed ODS declines and climate models indicate near-global TCO returning to 1980 levels around 2040, Antarctic recovery by 2066, and Arctic by 2045, assuming continued Montreal Protocol compliance and moderate greenhouse gas scenarios.[1] Upper stratospheric ozone layers show faster recovery, increasing 1.5–2.2% per decade at mid-latitudes from 2000 to 2020, driven by ODS reduction and stratospheric cooling from rising CO₂.[1] Despite progress, current TCO remains below pre-depletion baselines, and emerging factors like very short-lived substances could modestly delay full restoration.[1]Detection Methods: Satellite, Ground-Based, and Balloon Measurements
Satellite-based detection of stratospheric ozone utilizes instruments that measure ultraviolet radiation backscattered by the Earth's atmosphere to infer total column ozone amounts. The Total Ozone Mapping Spectrometer (TOMS), deployed on NASA's Nimbus-7 satellite from October 1978 to May 1993, provided daily global maps of total ozone with a resolution of approximately 50 km x 50 km by analyzing wavelengths at 308.6 nm, 317.5 nm, 331.2 nm, and 360 nm. [60] Successor instruments like the Ozone Monitoring Instrument (OMI) on the Aura satellite, launched in July 2004, offer enhanced spatial resolution of 13 km x 24 km at nadir and continue near-daily global coverage, distinguishing ozone profiles through spectral analysis in ultraviolet and visible bands. [61] These satellite methods enabled the first unambiguous detection of the Antarctic ozone hole in 1985, revealing seasonal total ozone minima below 220 Dobson Units (DU). [62] Ground-based measurements primarily employ spectrophotometers such as the Dobson instrument, operational since the 1930s, which quantify total ozone column by comparing ultraviolet intensities at paired wavelengths (e.g., 305.5 nm and 325.4 nm) where ozone absorption differs, using the Sun, Moon, or zenith sky as light sources. [63] The Brewer spectrophotometer, introduced in the 1980s, automates this process with direct Sun observations across five wavelengths (e.g., 306.3–320.1 nm) and provides simultaneous aerosol optical depth data, achieving accuracies of about 1% for total ozone under clear skies. [64] Networks like the World Ozone and Ultraviolet Radiation Data Centre maintain over 100 Dobson and Brewer stations worldwide, with routine measurements calibrated against standards traceable to the U.S. National Institute of Standards and Technology, contributing long-term records essential for validating satellite data and tracking depletion trends since the 1950s. [65] Balloon-borne ozonesondes deliver high-vertical-resolution profiles of ozone concentration from the troposphere to the lower stratosphere, typically up to 35 km altitude, using electrochemical cells that react ozone with potassium iodide to generate a measurable current proportional to ozone partial pressure. [66] Launched weekly at key sites like the South Pole since 1986, these instruments, often paired with radiosondes for temperature and pressure data, achieve resolutions of 10-100 meters and detect ozone layers with precisions of 5-10% in the stratosphere. [67] Historical profiles date back to the 1960s, with a pivotal 1967 launch from Laramie, Wyoming, confirming stratospheric ozone peaks around 25 km, while modern electrochemical concentration cells (ECC) ozonesondes, standardized since the 1980s, support detailed studies of polar depletion events by resolving vertical structure during satellite overpass limitations like polar night. [68] [69]
Data Reliability and Sources of Uncertainty
Stratospheric ozone measurements, derived from satellite instruments, ground-based spectrometers, and ozonesondes, exhibit high overall reliability when cross-validated across methods, with long-term records from NASA and NOAA instruments like SBUV/2 and TOMS demonstrating stability sufficient to detect multi-decadal trends on the order of 1-3% per decade.[70][71] However, systematic errors arise from instrument calibration drifts, as observed in TOMS data where slow degradation since 1978 launch affected retrieval accuracy by up to several percent without frequent post-launch adjustments.[72] Key sources of uncertainty include statistical variability from natural atmospheric fluctuations, such as solar cycles and quasi-biennial oscillations, which introduce noise in trend estimates, particularly in the tropical lower stratosphere where ozone changes are subtle.[73][74] Instrumental biases, including radiance-dependent errors in satellite backscatter ultraviolet sensors and imperfect regression models in merged datasets, contribute additional uncertainty, with SBUV Version 8.6 profiles showing errors from atmospheric variability and data merging on the order of 2-5% in total column ozone.[74][75] Ground-based observations, such as those from the Dobson network, face uncertainties from aerosol interference, sulfur dioxide absorption in pre-1976 data, and background current estimation below 25 km, potentially biasing upward trends by 1-2% in polluted regions.[76][77] Ozonesonde data, while stable to within ±5% against satellite profiles, encounter errors from chemical stoichiometry assumptions and pump efficiency variations during balloon ascents.[71][78] Merging multi-instrument records amplifies uncertainties due to differing vertical resolutions and sampling geometries, necessitating stability thresholds below 1% per decade for reliable detection of recovery signals under the Montreal Protocol.[79] Calibration efforts, including the World Dobson standard maintained by NOAA, mitigate these issues through periodic intercomparisons, ensuring global total ozone accuracy to within 1-2%.[80] Despite these challenges, ensemble analyses from independent platforms confirm robust depletion and incipient recovery trends, with uncertainties rarely exceeding the signal strength in polar regions.[81][73]Polar Ozone Depletion Phenomena
Antarctic Ozone Hole Formation and Characteristics
The Antarctic ozone hole refers to a seasonal region of severe ozone depletion over Antarctica where total column ozone falls below 220 Dobson units (DU), compared to typical values of around 300 DU in the region.[82] This phenomenon was first reported in 1985 by researchers from the British Antarctic Survey, who observed total ozone levels at Halley Station dropping to as low as 180 DU in October 1982 and 154 DU in 1983, with a trend of decline since the mid-1970s.[83] Satellite observations from the Total Ozone Mapping Spectrometer (TOMS) retroactively confirmed similar depletions as early as 1979, though initial data were initially dismissed as instrumental artifacts.[58] Formation begins during the Antarctic winter, when stratospheric temperatures drop below 195 K, enabling the formation of polar stratospheric clouds (PSCs) composed of nitric acid trihydrate or ice particles.[84] The strong polar vortex isolates cold air over the continent, preventing mixing with mid-latitude air. On PSC surfaces, heterogeneous chemical reactions convert inert chlorine reservoir species, such as chlorine nitrate (ClONO₂) and hydrogen chloride (HCl), into reactive forms like hypochlorous acid (HOCl) and chlorine (Cl₂).[84] In early spring (late August to September), returning sunlight photodissociates these compounds, releasing chlorine atoms (Cl) and chlorine monoxide (ClO) that participate in catalytic cycles destroying ozone: Cl + O₃ → ClO + O₂, followed by ClO + O → Cl + O₂, with net loss of odd oxygen.[85] Bromine species contribute similarly, enhancing depletion through ClO-BrO reactions.[82] These processes lead to near-complete removal of ozone below 20 km altitude in the lower stratosphere.[84] The ozone hole typically emerges in late August or early September, reaches maximum extent and depth in mid-October, and dissipates by December as the vortex weakens and sunlight warms the stratosphere, denitrifying PSCs and reforming reservoirs.[58] Peak area has varied, reaching up to 29 million km² in 2000 and 2006, while minimum total ozone over the South Pole has dropped as low as 92 DU in 2020.[59] [86] Depletion is most pronounced in the 12-20 km altitude range, where temperatures remain coldest and ClO levels peak above 1 part per billion by volume.[85] Annual variability arises from meteorological factors like vortex strength and final warming timing, with stronger, colder vortices correlating to deeper holes.[84]Arctic Ozone Depletion Events
Arctic ozone depletion events involve substantial chemical losses of stratospheric ozone over the northern polar region during winter and early spring, occurring irregularly when meteorological conditions favor the formation of polar stratospheric clouds (PSCs) within a stable, isolated polar vortex. These PSCs, forming at temperatures below approximately 195 K, enable the activation of chlorine and bromine from anthropogenic ozone-depleting substances (ODSs) like chlorofluorocarbons, initiating catalytic cycles that rapidly destroy ozone molecules. Unlike the Antarctic ozone hole, which forms annually due to consistently cold and stable conditions, Arctic events are less frequent and severe because the stratospheric vortex is typically warmer and more disrupted by upward-propagating planetary waves from mid-latitudes, which mix in ozone-rich air and warm the vortex. Ozone losses in the Arctic vortex core can reach 20-40% in typical cold winters but exceed 50% in exceptional cases, though total column ozone rarely drops below 220 Dobson units (DU) over extended areas.[87][88][89] The extent of depletion depends critically on vortex dynamics: a strong, cold, and persistent vortex isolates chemically processed air, minimizing dilution, while weak wave forcing—such as reduced upward propagation of quasi-stationary Rossby waves—prolongs these conditions. Quantified chemical ozone loss is estimated using passive tracer methods, comparing observed ozone to transport-only simulations, revealing vortex-wide depletions peaking in March at altitudes of 16-20 km. Persistent PSCs denitrify the stratosphere by sedimenting nitric acid particles, further enhancing chlorine activation and suppressing reformation of the chlorine reservoir via the NOx cycle. Natural variability, including sudden stratospheric warmings, often interrupts these processes in the Arctic, limiting losses compared to the Antarctic's more stable regime.[90][88][87] Notable events include the winters of 1997 and 2011, with chemical losses up to 35-40% in the lower stratosphere, and the record-breaking 2019/2020 season, where March 2020 ozone columns in the vortex fell to the lowest levels since 1979, with over 60% loss at 18 km altitude and areas exceeding the 220 DU threshold for an "ozone hole." The 2020 event stemmed from unusually weak wave activity from December 2019 through March, sustaining vortex temperatures 10-20 K below normal and PSC coverage for over 100 days. In contrast, warmer winters like 2024 saw record-high Arctic ozone due to enhanced wave-driven mixing. These episodes expose mid-to-high northern latitudes to elevated ultraviolet radiation temporarily, though recovery occurs by late spring as sunlight returns and the vortex breaks down.[91][92][90] Despite declining atmospheric ODS concentrations—down 10-20% since peak levels in the 1990s—cold winters still enable significant losses, underscoring the lingering impact of historical emissions. Satellite observations from instruments like the Aura MLS and ground-based lidars confirm these patterns, with no evidence of accelerating frequency tied to climate change, though projections suggest reduced severity as ODSs continue to decline under the Montreal Protocol. Arctic events highlight the interplay of anthropogenic chemicals and natural meteorology, with dynamical isolation as the key enabler rather than ODS increases.[93][94][95]Comparisons and Non-Polar Regional Depletions
In polar regions, stratospheric ozone depletion is characterized by severe, seasonal losses exceeding 50% in the Antarctic ozone hole during September-October, driven by polar stratospheric clouds (PSCs) that facilitate heterogeneous chlorine and bromine activation within the isolated polar vortex.[1] Arctic events show shallower depletions, typically 20-40%, due to warmer temperatures, less stable vortices, and frequent disruptions from sudden stratospheric warmings, resulting in interannual variability rather than consistent "holes."[96] In contrast, non-polar regions experience milder, more uniform depletion through gas-phase catalytic cycles involving chlorine and bromine radicals, without PSC enhancement or vortex isolation, leading to historical total column ozone (TCO) declines of 3-6% in mid-latitudes from pre-1980 levels.[97] These differences arise from temperature thresholds below -78°C required for PSC formation, absent in mid-latitudes and tropics, where dynamical mixing dilutes depleted air masses.[98] Mid-latitude TCO (35°-60° latitude) declined by approximately 4-5% below 1964-1980 baselines by the late 1990s, with northern hemisphere trends showing negligible recent change (0.0 ± 0.7% per decade from 1998-2020) and southern hemisphere slight increases (0.8 ± 0.7% per decade).[98] Upper stratospheric ozone in these regions has increased by 1.5-2.2% per decade since 2000, attributed to declining ozone-depleting substances (ODSs) like chlorofluorocarbons, whose stratospheric chlorine peaked in 1993 and fell 11.5% by 2020.[99] Lower stratospheric trends remain uncertain, with small decreases (1-2% per decade) possibly linked to circulation changes rather than ODS.[98] Tropical regions (20°S-20°N) exhibit minimal historical depletion (about 1% below pre-1980), with no significant TCO trend (0.2 ± 0.3% per decade), as ozone production and transport balance losses; upper stratospheric increases of 1.1-1.6% per decade signal recovery, while lower levels decline slightly due to upwelling in the Brewer-Dobson circulation.[98] Non-polar regional depletions lack the acute, localized severity of polar holes, instead manifesting as gradual, widespread thinning influenced by meridional transport of ODS-activated air from higher latitudes, though this contributes less than 10% to mid-latitude losses.[97] Projections indicate mid-latitude TCO returning to 1980 levels around 2040 under moderate emissions scenarios, faster than polar recovery due to less dependence on vortex dynamics and PSC chemistry.[98] These trends underscore the efficacy of ODS controls under the Montreal Protocol, with non-polar recovery less vulnerable to dynamical variability than polar regions.[99]Impacts of Increased UV Radiation
Human Health Effects: Cancers, Eye Conditions, and Immune Suppression
Increased stratospheric ozone depletion allows greater penetration of ultraviolet B (UVB) radiation to Earth's surface, which is causally linked to elevated risks of non-melanoma skin cancers (NMSC), including basal cell carcinoma (BCC) and squamous cell carcinoma (SCC), as well as malignant melanoma.[100] UVB induces DNA damage in skin cells, primarily through pyrimidine dimer formation, leading to mutations that promote carcinogenesis in a dose-dependent manner.[101] Laboratory and epidemiological evidence confirms UVB as the primary cause of NMSC and a major contributor to melanoma, with fair-skinned populations at highest risk due to lower melanin protection.[100] Attribution studies estimate that ozone depletion has contributed to an additional global burden of approximately 33,000 melanoma and NMSC cases, though recovery under the Montreal Protocol has mitigated further increases.[102] Cortical cataracts, characterized by opacity in the lens cortex, are strongly associated with cumulative UVB exposure, which damages lens proteins via photochemical reactions and oxidative stress.[103] Ozone depletion exacerbates this by elevating surface UVB levels, with models projecting up to 830,000 additional U.S. cataract cases by 2050 under unchecked depletion scenarios, though actual incidence reflects partial recovery.[104] Other ocular effects include pterygium, a UVB-induced growth on the conjunctiva, and acute photokeratitis ("snow blindness"), both more prevalent in high-UV regions correlating with ozone minima.[105] Protective eyewear and latitude-dependent exposure gradients underscore the causal role of UVB in these conditions.[106] UVB radiation suppresses systemic and local immune function by depleting antigen-presenting cells like Langerhans cells in the skin and inducing regulatory T cells that inhibit T-cell responses.[107] This immunosuppression impairs contact hypersensitivity, increases susceptibility to skin infections, and may reduce vaccine efficacy, with human studies showing dose-dependent suppression of delayed-type hypersensitivity.[108] In the context of ozone depletion, elevated UVB has been linked to heightened reactivation of latent viruses, such as herpes, and broader infectious disease risks, particularly in immunocompromised individuals.[109] Empirical data from controlled exposures indicate that even moderate UVB increases can evoke neo-antigen formation, altering immune tolerance and exacerbating photocarcinogenesis.[21] Ozone depletion over Antarctica causes periodic high UV-B radiation in southern Argentina, particularly in Patagonia and Ushuaia, leading to increased risks of phototoxic effects such as sunburn, erythema, and skin damage, as well as systemic effects including immune suppression and elevated long-term risks of skin cancer and cataracts. These effects are most pronounced during austral spring when the ozone hole is present, although overall skin cancer rates in the region are influenced by multiple factors including behavioral and demographic variables.[100]Environmental and Agricultural Consequences
Increased ultraviolet-B (UV-B) radiation penetrating the Earth's surface due to stratospheric ozone depletion inhibits photosynthesis and growth in phytoplankton, the foundational primary producers of aquatic ecosystems. Experimental and field studies in regions like the Antarctic, where ozone thinning reached up to 50% during spring, have quantified UV-B-induced reductions in phytoplankton productivity, with inhibition rates varying by species and depth but often exceeding 10-20% under elevated exposure. These effects disrupt carbon fixation, diminish the ocean's capacity as a CO₂ sink, and propagate through food webs, potentially reducing populations of herbivorous zooplankton and fisheries yields.[110][111][112] In terrestrial ecosystems, elevated UV-B alters plant physiology by damaging DNA, proteins, and photosynthetic machinery, leading to stunted growth, reduced leaf area, and lower biomass accumulation in sensitive species such as herbaceous vegetation and crops. Ecosystem-level experiments simulating ozone depletion levels reveal subtle but consistent inhibitory impacts on primary productivity, shifts in species composition favoring UV-tolerant plants, and disruptions to nutrient cycling, including slowed decomposition by soil microbes. Woody plants and forests exhibit milder responses, but cumulative effects could amplify under combined stressors like drought.[113][114][100] Amphibian embryos and larvae, with their translucent eggs and aquatic early life stages, suffer elevated mortality and deformities from UV-B-induced DNA damage and oxidative stress, as evidenced by field enclosures and lab exposures mimicking ambient depletion-era radiation. Species-specific sensitivities contribute to population declines observed since the 1980s, with UV-B acting synergistically with pathogens and habitat loss, though direct causation remains debated due to confounding variables.[115][116][117] Agriculturally, UV-B increases from ozone depletion reduce yields in staple crops through impaired cell division, elongation, and photosynthetic efficiency. Soybean studies under enhanced UV-B report 10-25% drops in pod number and seed biomass, while rice and wheat show stunted growth and up to 15% biomass reductions in sensitive varieties. These impacts, documented in controlled field trials equivalent to 10-20% ozone loss, highlight varietal differences, with tropical crops often more resilient but overall productivity vulnerable to compounded climate effects.[118][119][120]Countervailing Effects: Vitamin D Synthesis and Ecosystem Adaptations
Increased stratospheric ozone depletion allows more ultraviolet B (UVB) radiation (280-315 nm) to penetrate to the Earth's surface, enhancing the photochemical conversion of 7-dehydrocholesterol in human skin to previtamin D3, the precursor to active vitamin D.[121] This process, which requires specific UVB doses of approximately 3-6 mJ/cm² for fair-skinned individuals to produce 10-20 µg of vitamin D, benefits populations at higher latitudes or during seasons with marginal solar exposure, where ozone loss of 5-10% globally (peaking at 50-60% over Antarctica in spring) correlates with 6-12% UVB increases.[122][103] Such elevations could mitigate vitamin D insufficiency, linked to risks of rickets, osteomalacia, and impaired immune function, particularly in mid-to-high latitude regions during mid-winter, as modeled in assessments of pre-recovery scenarios.[123] Peer-reviewed analyses indicate potential reductions in autoimmune disorders and metabolic issues from elevated vitamin D, though these gains are regionally variable and overshadowed by UV-induced harms like skin cancer.[124] Terrestrial and aquatic organisms possess innate biochemical, physiological, and behavioral mechanisms that counter elevated UVB from ozone depletion, preventing ecosystem destabilization. Plants activate DNA repair via nucleotide excision and photoreactivation pathways, while upregulating production of UV-absorbing flavonoids and antioxidants like ascorbate to limit reactive oxygen species damage; field studies in ozone-impacted regions show acclimation through leaf thickening and reduced stomatal conductance.[119] In marine systems, phytoplankton and zooplankton synthesize mycosporine-like amino acids (MAAs) for UV screening and employ vertical migration to deeper, less irradiated waters, sustaining productivity under Antarctic ozone hole conditions since the 1980s despite 10-20% UVB surges.[21] Animals, including fish larvae and amphibians, utilize pigmentation enhancements, behavioral avoidance (e.g., burrowing or shade-seeking), and efficient photorepair enzymes, with species-specific resilience documented in long-term polar monitoring; these adaptations, evolved under natural UV variability, dampen impacts without evidence of broad trophic collapse.[125][126] Overall, such responses highlight ecosystem robustness, though prolonged exposure exceeds repair capacities in UV-sensitive taxa.[127]Policy and Regulatory Responses
Development of the Montreal Protocol and Its Amendments
The Montreal Protocol on Substances that Deplete the Ozone Layer was adopted on September 16, 1987, during a diplomatic conference in Montreal, Canada, convened under the United Nations Environment Programme (UNEP) following the 1985 Vienna Convention for the Protection of the Ozone Layer.[128] The agreement established binding phase-down schedules for the production and consumption of specified ozone-depleting substances (ODS), initially targeting chlorofluorocarbons (CFCs) in Annex A (Groups I and II) and halons in Annex B, with developed countries required to achieve a 50% reduction in CFCs from 1986 baseline levels by 1998 and freeze halons at 1986 levels from 1992.[129] Developing countries, classified as Article 5 parties, received extended timelines of up to 10 years for compliance, reflecting differential responsibilities based on economic capacity.[128] The Protocol entered into force on January 1, 1989, after ratification by 20 instruments of ratification, acceptance, approval, or accession.[129] Subsequent amendments, adopted at meetings of the parties, expanded controlled substances, tightened phase-out timelines, and incorporated scientific assessments confirming the efficacy of reductions in halting further depletion. The London Amendment, adopted June 29, 1990, added controls on hydrobromofluorocarbons (HBFCs), methyl chloroform, and carbon tetrachloride to Annexes, committed developed countries to full phase-out of CFCs, halons, and other listed ODS by 2000 (or 2002 for methyl chloroform), and established the Multilateral Fund to assist developing countries with compliance costs; it entered into force August 10, 1992.[130] The Copenhagen Amendment, adopted November 25, 1992, accelerated phase-outs for developed countries (e.g., CFCs and carbon tetrachloride by 1996, halons by 1994) and introduced controls on bromochloromethane, entering into force January 14, 1994.[130] The 1997 Montreal Amendment controlled methyl bromide (with exemptions for critical uses like agriculture), further restricted hydrochlorofluorocarbons (HCFCs) in Annex C, and enhanced trade provisions to prevent leakage; it entered into force November 10, 1999.[131] The 1999 Beijing Amendment reinforced methyl bromide controls, added provisions on n-propyl bromide and bromomethane, and strengthened licensing for ODS trade, entering into force February 25, 2002.[131] These ozone-focused amendments progressively covered nearly 100 ODS categories, achieving 98% global phase-out of baseline ODS consumption by 2010 relative to 1990 levels.[128] The Kigali Amendment, adopted October 15, 2016, extended the Protocol's framework to phase down hydrofluorocarbons (HFCs)—ODS substitutes with high global warming potential—requiring developed countries to reduce HFC production and consumption by 85% from baselines by 2036, with developing countries freezing baselines in 2024 or 2028 and declining thereafter; it entered into force January 1, 2019, for ratifying parties, though primarily addressing climate co-benefits rather than direct ozone depletion.[132] By 2025, the Protocol and its amendments had achieved universal ratification by 198 parties, with adjustments (non-ratification-requiring updates to phase-out schedules based on quadrennial assessments) further refining controls, such as accelerating HCFC phase-outs to 2020 for developed countries and 2030 for developing ones.[128]Global Implementation, Compliance Issues, and Black Markets
The Montreal Protocol, adopted in 1987 and entering into force in 1989, achieved universal ratification by all 198 United Nations member states by September 2009, establishing binding production and consumption phase-out schedules for ozone-depleting substances (ODS) such as chlorofluorocarbons (CFCs) and halons.[128] Developed countries (non-Article 5 parties) completed CFC phase-outs by 1996, while developing countries (Article 5 parties) had until 2010, supported by the Multilateral Fund established in 1991, which has disbursed over $3.9 billion by 2024 to finance compliance projects like technology transfers and capacity building.[128] This framework has resulted in a global reduction of nearly 99% in ODS consumption since the 1980s, with controlled substances like CFCs dropping to trace atmospheric levels by 2024.[133] Compliance is monitored through mandatory data reporting under Article 7, with the Implementation Committee reviewing submissions and invoking the non-compliance procedure of Article 8 for identified shortfalls, such as excess production or import discrepancies.[134] By 2024, the Technology and Economic Assessment Panel (TEAP) reported broad adherence, with most parties meeting HCFC phase-out baselines ahead of the 2025 freeze for Article 5 countries, though isolated delays occurred in regions like sub-Saharan Africa due to enforcement gaps and reliance on legacy equipment.[135] The Multilateral Fund's executive committee has approved over 8,600 projects, aiding 197 countries, yet systemic challenges persist in verifying destruction of stockpiles and transitioning small-scale users in informal sectors.[136] Black markets for ODS emerged post-phase-out as legal supplies dwindled, driving smuggling and diversion from countries with extended grace periods, with estimates of illegal CFC trade peaking at 38,000 metric tons annually in the mid-1990s, equivalent to 10-20% of legal quotas in some markets.[137] Primary routes involve production in China and India—where HCFC manufacturing quotas allow diversion—smuggled via ports in Southeast Asia and the Middle East to Europe and North America for refrigeration servicing, often mislabeled as non-ODS or reclaimed stock.[138] By 2025, illegal HCFC trade volumes were estimated at 5,000-10,000 tons yearly, undermining phase-downs and contributing to excess stratospheric chlorine, as detected in atmospheric monitoring; U.S. authorities seized over 1,000 tons of smuggled refrigerants between 2018 and 2023, with penalties including fines up to $50,000 per violation under the Clean Air Act.[139][133] Efforts to curb this include UNEP's Green Customs Initiative, training over 10,000 border officials since 2004, and enhanced licensing under the Protocol's trade provisions, though online platforms have facilitated covert sales, as noted in 2025 investigations revealing unverified refrigerant offerings on e-commerce sites.[140][141]Economic Costs, Technological Shifts, and Industry Adaptations
The implementation of the Montreal Protocol imposed significant economic costs on industries reliant on ozone-depleting substances (ODS), primarily through the need to reformulate products, retrofit equipment, and invest in research and development for alternatives. Global compliance costs for phasing out ODS were estimated at approximately US$235 billion in 1997 prices, encompassing private sector expenditures on technological transitions across sectors like refrigeration, air conditioning, and foam manufacturing.[142] These costs were partially mitigated for developing countries via the Protocol's Multilateral Fund, which has disbursed over US$3.9 billion since 1991 to support ODS phaseout projects in Article 5 nations, covering incremental expenses such as technology transfer and capacity building.[143] In developed economies like the United States, compliance costs were relatively modest, representing less than 0.1% of annual GDP during peak phaseout periods, due to early adoption of substitutes and economies of scale in innovation.[144] Technological shifts driven by the Protocol accelerated the replacement of chlorofluorocarbons (CFCs) and hydrochlorofluorocarbons (HCFCs) with non-ozone-depleting alternatives, spurring innovations in chemical synthesis and engineering processes. In refrigeration and air conditioning, CFC-12 was largely supplanted by HFC-134a by the mid-1990s in developed countries, necessitating modifications to compressors, lubricants, and system designs to accommodate differing thermodynamic properties, while HCFCs served as interim bridges until their phaseout under subsequent amendments.[145] The foam sector transitioned from CFC-11 blowing agents to hydrofluorocarbons or hydrocarbons like cyclopentane, enabling closed-cell polyurethane production with comparable insulation efficiency but requiring adjustments in polymerization reactions to prevent defects.[146] These changes induced a surge in patent filings for ODS alternatives, with empirical analysis showing a 20-30% increase in relevant innovation post-1987, as regulatory certainty incentivized directed research toward low-ozone-depletion-potential (ODP) compounds.[147] Industries adapted through a combination of in-kind substitutions and process redesigns, often transforming initial compliance burdens into competitive advantages via enhanced efficiency and new markets. Chemical manufacturers, such as DuPont, voluntarily ceased CFC production ahead of deadlines in 1995, redirecting R&D toward HFC portfolios that generated billions in revenue from patented alternatives, while electronics firms shifted from CFC-113 solvents to aqueous or semi-aqueous cleaning methods, reducing solvent use by up to 90% in circuit board assembly.[148] The automotive sector redesigned mobile air-conditioning systems for HFC compatibility by 1994, incorporating variable displacement compressors that improved energy efficiency by 10-20% compared to legacy CFC setups.[149] Aerosol producers pivoted to hydrocarbon propellants or mechanical pumps, eliminating ODS entirely by 1995 in most markets and expanding into water-based formulations that lowered material costs over time. Despite short-term disruptions, such as equipment retrofits costing US$10-50 billion globally for existing refrigeration stock, these adaptations fostered long-term productivity gains, with studies indicating that Protocol-induced technologies reduced operational energy demands in affected sectors by 5-15%.[150][151]Recovery Trends and Projections
Evidence of Stratospheric Ozone Recovery
Satellite and ground-based observations indicate a statistically significant recovery in stratospheric ozone levels since the late 1990s, coinciding with the decline in atmospheric concentrations of ozone-depleting substances (ODS) following the Montreal Protocol's implementation. Upper stratospheric ozone, particularly above 30 km altitude, shows positive trends of up to 2% per decade in both hemispheres, attributed to reduced catalytic destruction by chlorine and bromine radicals from ODS.[152] Global total column ozone has increased by approximately 0.4% per decade since 1995, representing about one-third of the reversal of pre-1995 depletion rates.[153] In the Antarctic, the ozone hole—defined as regions with total column ozone below 220 Dobson units—has exhibited a long-term decrease in maximum size and depth since peaking around 2000. The 2024 Antarctic ozone hole reached a maximum extent of 22 million square kilometers in late September, ranking as the seventh smallest since recovery trends began in the 1990s, with minimum column ozone values around 100 Dobson units, higher than the record lows of the 1990s.[154] [155] A 2025 MIT-led analysis confirmed healing in the Antarctic ozone layer, linking it directly to a 99% reduction in ODS emissions, with springtime ozone losses decreasing by 20% from 2005 to recent years.[156] [58] Year-to-year variations, such as larger holes in 2023 due to meteorological factors like stable vortex conditions, overlay the recovery trend but do not alter the overall positive trajectory.[157] Arctic stratospheric ozone also shows recovery signatures, with statistically significant increases of up to 0.9% per decade in the upper stratosphere (5–1 hPa levels) detected through multi-decadal records.[158] Non-polar regions exhibit smaller depletions historically, and mid-latitude ozone columns have stabilized or slightly increased, with effective equivalent stratospheric chlorine (EESC) metrics declining post-1995, supporting causal linkage to ODS phase-out.[159] Projections from NASA and NOAA estimate return to 1980 levels globally by 2040–2066, contingent on continued ODS compliance, though recovery rates may be influenced by interactions with climate-driven circulation changes.[154]Factors Influencing Recovery Rates
The primary driver of stratospheric ozone recovery is the sustained decline in atmospheric abundances of ozone-depleting substances (ODS), particularly long-lived chlorofluorocarbons (CFCs) and halons, resulting from global phase-outs under the Montreal Protocol and its amendments. Equivalent effective stratospheric chlorine (EESC) levels, a metric of reactive halogen loading, peaked in the late 1990s and have since decreased by approximately 10-20% as of 2022, enabling chemical recovery in the upper stratosphere where ozone has increased by 1-3% per decade since 2000.[1] [98] Climate change modulates recovery rates through multiple pathways. Rising concentrations of carbon dioxide (CO₂) cool the stratosphere by 1-2°C per decade in the lower stratosphere, slowing temperature-dependent catalytic ozone destruction cycles involving chlorine and bromine radicals, which accelerates recovery by up to 5-10% in model projections. However, increased nitrous oxide (N₂O) from agricultural sources depletes ozone via conversion to nitric oxide (NOx), potentially offsetting gains by 5-15% in the middle stratosphere by 2100 under high-emission scenarios. Enhanced methane (CH₄) oxidizes to water vapor and hydroxyl radicals that can scavenge ozone-destroying species, providing a countervailing increase.[1] [160] Atmospheric dynamics and transport processes also influence spatial and temporal recovery patterns. A projected strengthening of the Brewer-Dobson circulation under greenhouse gas forcing could poleward shift ozone, delaying mid-latitude column ozone return to 1980 levels until after 2050 in some models, while accelerating polar recovery through increased descent of ozone-rich air. Year-to-year variability from the quasi-biennial oscillation (QBO), solar ultraviolet flux cycles (11-year periodicity), and sudden stratospheric warmings introduces fluctuations of 2-5% in total column ozone, complicating attribution of long-term trends.[1] [161] Emerging risks include emissions of unregulated very short-lived substances (VSLS), such as dichloromethane (CH₂Cl₂), which contribute 10-20% of stratospheric inorganic chlorine and have risen since 2010 due to industrial solvent use, potentially delaying Antarctic recovery by 5-10 years if unchecked. Episodic events like the 2022 Hunga Tonga-Hunga Ha'apai eruption injected massive stratospheric water vapor (up to 10% increase), enhancing polar stratospheric cloud formation and heterogeneous chlorine activation, leading to temporary ozone losses exceeding 10% in affected regions. Detected non-compliance, including a 2018-2019 slowdown in CFC-11 decline attributed to illicit production in eastern Asia, underscores vulnerabilities to black-market activities that could prolong elevated halogen levels by years. Wildfire smoke injections, increasingly frequent with climate change, loft organic halogens and aerosols that catalyze ozone depletion, as observed in Australian bushfires contributing to the 2020 Antarctic ozone hole persistence.[1] [57]Recent Developments and 2024-2025 Ozone Hole Status
In 2024, the Antarctic ozone hole attained a maximum extent of 22.4 million square kilometers on September 28, ranking seventh-smallest among records since regulatory recovery commenced in 1992.[59] Its average size during the core depletion period (September 7 to October 13) measured approximately 20 million square kilometers, while the minimum total ozone column reached 109 Dobson units on October 5.[59] These metrics reflect a contraction relative to holes in the early 2000s and prior decades, driven by sustained declines in stratospheric chlorine from phased-out ozone-depleting substances under the Montreal Protocol, augmented by mid-season influxes of ozone-rich air from lower latitudes.[59] [162] For the 2025 season, NASA's Ozone Watch satellite data document the hole's emergence in late August, escalation to peak dimensions around 23 million square kilometers by early September, and subsequent dissipation through October.[163] [164] As of late October 2025, residual depletion persists but at levels elevated above historical minima, aligning with the trajectory of diminished annual severity observed since the mid-2010s.[163] Advancements in 2024-2025 include a March 2025 MIT-led study attributing Antarctic ozone rebound primarily to anthropogenic curbs on chlorofluorocarbons, with statistical models isolating their causal role from natural forcings like volcanic aerosols or solar cycles.[156] The World Meteorological Organization's September 2025 bulletin affirms global stratospheric ozone stabilization, projecting complete restoration to 1980 baselines by 2066 contingent on unwavering Protocol adherence and minimal illegal emissions.[165] [59] Ongoing monitoring underscores vulnerabilities to transient events, such as stratospheric warming, yet reinforces empirical evidence of regulatory efficacy over alternative explanations emphasizing dominant natural oscillation.[166]Historical Research and Discovery
Early Theoretical Work: Chapman Cycle and Rowland-Molina Hypothesis
In 1930, British physicist Sydney Chapman proposed the first quantitative photochemical theory explaining the formation and steady-state concentration of stratospheric ozone, known as the Chapman cycle or mechanism.[167] This model posits that ultraviolet radiation photodissociates molecular oxygen (O₂) into atomic oxygen (O), which then reacts with O₂ to form ozone (O₃); ozone is subsequently photodissociated back into O and O₂, and atomic oxygen reacts with O₃ to regenerate O₂, establishing a dynamic equilibrium.[14] The key reactions are:- O₂ + hν (λ < 242 nm) → 2O
- O + O₂ + M → O₃ + M (M is a third body for energy transfer)
- O₃ + hν (λ < 320 nm) → O + O₂
- O + O₃ → 2O₂
- Cl + O₃ → ClO + O₂
- ClO + O → Cl + O₂
- Net: O₃ + O → 2O₂ (one Cl destroys ~100,000 O₃ molecules before being sequestered)
Empirical Discovery of the Ozone Hole
The Antarctic ozone hole was empirically identified through long-term ground-based measurements of atmospheric ozone concentrations at the Halley Research Station, operated by the British Antarctic Survey (BAS). Total column ozone had been monitored using a Dobson spectrophotometer since 1957, providing a continuous record of ozone levels in Dobson units (DU).[83] By the late 1970s, BAS scientists observed a marked decline in springtime (September–November) ozone readings, with concentrations falling below 220 DU in October 1984—about 40% lower than pre-1975 averages of around 300–320 DU.[173] [174] In a seminal paper published on May 16, 1985, in Nature, BAS researchers Joe Farman, Brian Gardiner, and Jonathan Shanklin documented these observations, reporting annual springtime depletions escalating from negligible levels before 1977 to over one-third of the total ozone column by 1984.[174] [175] The data indicated a localized phenomenon confined to the Antarctic region during the polar spring, characterized by rapid ozone loss rates exceeding 1% per day in some instances.[176] Farman et al. noted the measurements' reliability, as instrument calibration and atmospheric variability had been rigorously accounted for, ruling out systematic errors.[174] Satellite observations provided independent confirmation of the hole's scale shortly after the BAS publication. The Total Ozone Mapping Spectrometer (TOMS) aboard NASA's Nimbus-7 satellite, launched in October 1978, had recorded low ozone values over Antarctica since 1979, but initial data processing algorithms automatically discarded readings below 180 DU as implausible artifacts.[177] [178] Prompted by the BAS findings, NASA scientists recalibrated the algorithms in late 1985, validating the data and revealing the ozone hole as a continent-wide feature with 1985 minima reaching 109 DU on October 24—far below the global average of 300 DU.[177] [179] This reanalysis extended the record of significant depletion back to the satellite's inception, confirming the phenomenon's progression and spatial extent.[180] The discovery highlighted discrepancies between ground-based and early satellite data, underscoring the need for cross-validation in remote sensing. BAS measurements, limited to a single site, captured the core depletion but underestimated the hole's full areal coverage, which TOMS mapped as encompassing over 20 million square kilometers by the mid-1980s.[177] These empirical findings shifted scientific focus from gradual global thinning—predicted by earlier models—to acute, seasonal polar vulnerability, driven by unique Antarctic conditions like the polar vortex and stratospheric cooling.[174] Subsequent expeditions, including NASA's Airborne Antarctic Ozone Expeditions starting in 1987, further corroborated the observations through in-situ profiling, measuring ozone losses up to 95% at altitudes of 15–20 km.[181]Evolution of Computer Models and Validation Challenges
The initial computational models for stratospheric ozone emerged in the 1960s as one-dimensional (1D) representations, building on the Chapman photochemical theory of 1930, which described ozone formation and loss through oxygen photolysis and recombination but lacked transport dynamics.[170] These 1D models incorporated catalytic cycles involving hydrogen oxides (HOx) by the mid-1960s, yet they overestimated ozone concentrations due to neglect of meridional circulation and other loss mechanisms.[169] By the early 1970s, following the identification of nitrogen oxide (NOx) and chlorine (ClOx) catalytic destruction pathways—linked to supersonic aircraft emissions and chlorofluorocarbons (CFCs), respectively—1D models predicted modest global ozone reductions of 2-8% from CFC increases, though coverage of observations remained inadequate for distinguishing between competing formulations.[170][182] The 1970s and 1980s marked the shift to two-dimensional (2D) models, which integrated latitudinal transport and seasonal variations, enabling simulations of zonal-mean ozone distributions.[169] Post-1985 Antarctic ozone hole discovery, these models incorporated heterogeneous chemistry on polar stratospheric clouds (PSCs) and sulfate aerosols, explaining rapid springtime depletion via chlorine activation, which earlier homogeneous gas-phase models had failed to anticipate.[170] Validation against ground-based Dobson spectrophotometers and early satellite data (e.g., Nimbus 7 Total Ozone Mapping Spectrometer from 1979) confirmed upper stratospheric declines aligning with CFC-driven predictions, but mid-latitude and polar anomalies persisted due to unmodeled dynamical feedbacks and volcanic perturbations like El Chichón (1982) and Pinatubo (1991), which enhanced aerosol surface areas and temporary ozone losses of up to 5-10%.[183] The 1990s introduced three-dimensional (3D) chemistry-transport models (CTMs), evolving into fully coupled chemistry-climate models (CCMs) by the early 2000s, such as NASA's GEOSCCM (developed from the 1990s) and NCAR's MOZART (2000), which resolved global circulation, radiative transfer, and over 100 chemical species across altitudes.[184][169] These advancements allowed projections of ozone recovery under Montreal Protocol scenarios, with multimodel means forecasting Antarctic hole closure by 2060-2070, driven by declining ODS (ozone-depleting substances) concentrations.[185] CCM intercomparisons, like those in the SPARC Chemistry-Climate Model Initiative (CCMI) from 2010 onward, refined estimates by coupling ozone feedback to climate, revealing stratospheric cooling's amplification of polar depletion by 20-30%.[186] Validation challenges have persisted across model generations, stemming from sparse pre-1970s observations, which limited calibration of rate constants and transport parameters, leading to uncertainties in depletion attribution exceeding 10-20% in early 1D simulations.[182] In 3D CCMs, discrepancies arise from natural variability (e.g., quasi-biennial oscillation, solar cycles), requiring ensemble runs to isolate anthropogenic signals, as single realizations often mask trends below detection thresholds of 1-2% per decade.[187] Recent assessments highlight ongoing issues, including underrepresentation of very short-lived halogens and biosphere-chemistry interactions, with model-observation mismatches in the lower stratosphere persisting at 5-15% despite validations against modern datasets like Aura/OMI (2004-present) and ground networks.[57] Empirical adjustments, such as ridge regression for trend estimation, mitigate overfitting but underscore the reliance on assumed parameterizations over purely data-driven validation.[187]Linkages to Climate Change
Ozone-Depleting Substances as Greenhouse Gases
Many ozone-depleting substances (ODS), including chlorofluorocarbons (CFCs), hydrochlorofluorocarbons (HCFCs), halons, and certain brominated compounds, function as potent greenhouse gases due to their strong absorption of infrared radiation in atmospheric windows where CO₂ is less effective, leading to enhanced radiative forcing.[188] These long-lived compounds, with atmospheric lifetimes ranging from decades to over a century (e.g., CFC-12 persists for 100 years), accumulate in the troposphere and stratosphere, contributing to global warming independent of their ozone-depleting effects.[189] Their direct climate impact arises from molecular structures that trap heat efficiently, with collective emissions from 1930 to 2010 exerting a radiative forcing of approximately 0.33 W/m², comparable to about 15-20% of CO₂'s forcing over the same period.[190] The global warming potentials (GWPs) of major ODS, measured relative to CO₂ over a 100-year horizon per IPCC AR6 assessments, underscore their potency: CFC-11 at 4,660, CFC-12 at 10,200, CFC-113 at 4,640, HCFC-22 at 1,760, and halon-1301 at 7,210.[191]| Substance | Chemical Formula | 100-Year GWP | Atmospheric Lifetime (years) |
|---|---|---|---|
| CFC-11 | CFCl₃ | 4,660 | 52 |
| CFC-12 | CF₂Cl₂ | 10,200 | 100 |
| HCFC-22 | CHClF₂ | 1,760 | 11.9 |
| Halon-1211 | CBrClF₂ | 1,910 | 16 |
| Halon-1301 | CBrF₃ | 7,210 | 72 |
Stratospheric Ozone's Influence on Surface Climate
Stratospheric ozone modulates surface climate primarily through its role in absorbing ultraviolet radiation, which generates radiative heating in the stratosphere and influences meridional temperature gradients. This heating affects the strength and position of subtropical jets and planetary wave propagation, thereby altering tropospheric circulation patterns that extend to the surface. In the Southern Hemisphere, ozone depletion since the late 1970s has cooled the polar stratosphere by up to 2-3 K in austral spring, strengthening the westerly jet and contributing to a positive trend in the Southern Annular Mode (SAM), which shifts storm tracks poleward and expands the Hadley cell.[197][198] These circulation changes have led to drier conditions in mid-latitudes (e.g., reduced precipitation in southern Australia by 10-20% since 1980) and increased precipitation in high latitudes, influencing surface temperatures and sea ice extent. Ozone-driven cooling of the stratosphere has also modulated Southern Ocean surface temperatures, with models indicating a small cooling contribution (0.1-0.2°C) in the sub-Antarctic zone, though this is secondary to greenhouse gas forcings. In the Northern Hemisphere, boreal spring ozone variations correlate with surface temperature anomalies, such as warmer Eurasian winters linked to reduced stratospheric ozone, via downward propagation of anomalies affecting the Arctic Oscillation.[160][199][200] As stratospheric ozone recovers under the Montreal Protocol, these influences are projected to reverse, with ozone increases projected to weaken the SAM by 0.1-0.2 standard deviations per decade through 2100, shifting westerlies equatorward and potentially increasing mid-latitude precipitation while reducing Antarctic sea ice trends. Model simulations attribute about 20-30% of projected Southern Hemisphere circulation recovery to ozone, distinct from greenhouse gas-induced poleward shifts. However, uncertainties persist in attributing isolated surface temperature changes solely to ozone, as coupled ocean-atmosphere feedbacks amplify or dampen signals, and empirical data show ozone depletion's radiative forcing at the surface as a minor cooling (-0.05 W/m² globally since 1980).[201][199][202]Distinctions from Tropospheric Ozone and Warming Narratives
Stratospheric ozone, concentrated between approximately 15 and 35 kilometers altitude, serves a protective function by absorbing harmful ultraviolet-B (UV-B) radiation from the sun, thereby shielding terrestrial life from excessive UV exposure that can cause DNA damage, skin cancer, and ecosystem disruptions.[55] In contrast, tropospheric ozone, prevalent in the lower atmosphere up to about 10 kilometers, acts as a toxic air pollutant formed through photochemical reactions involving nitrogen oxides (NOx) and volatile organic compounds (VOCs) in the presence of sunlight, contributing to smog formation and respiratory health issues such as asthma exacerbations and premature mortality.[203] [204] The causes of stratospheric ozone depletion differ fundamentally from those generating tropospheric ozone; depletion primarily results from catalytic destruction by anthropogenic ozone-depleting substances (ODS) like chlorofluorocarbons (CFCs), which release chlorine and bromine atoms in the stratosphere, whereas tropospheric ozone arises from local pollution sources including vehicle emissions and industrial activities.[205] [206] While stratospheric ozone loss increases surface UV radiation, potentially elevating skin cancer rates by 2-4% per 10% depletion in some regions, tropospheric ozone exposure correlates with cardiovascular and pulmonary diseases, with global estimates attributing over 1 million premature deaths annually to it.[207] [208] Ozone depletion narratives must be distinguished from global warming accounts, as the former involves UV protection erosion primarily from ODS phased out under the 1987 Montreal Protocol, leading to observed Antarctic ozone recovery since the early 2000s, whereas warming stems predominantly from carbon dioxide (CO2) and other long-lived greenhouse gases trapping infrared radiation.[156] [209] Although ODS such as CFCs possess global warming potentials (e.g., CFC-11 at 4,660 times that of CO2 over 100 years), their atmospheric concentrations represent less than 10% of total radiative forcing, and depletion itself induces stratospheric cooling rather than surface warming.[29] [210] Tropospheric ozone, conversely, exerts a positive radiative forcing of about 0.4 W/m², augmenting warming, but its levels are regulated under air quality standards distinct from ODS controls.[211] Public and policy conflations, often amplified in media, overlook these mechanistic separations, with empirical data confirming ozone recovery decoupled from stalled or uncertain warming mitigation outcomes.[212]Controversies and Alternative Perspectives
Debates on Primary Causation: Anthropogenic vs. Natural Dominance
The primary debate on ozone depletion causation centers on whether anthropogenic emissions of ozone-depleting substances (ODS), particularly chlorofluorocarbons (CFCs), represent the dominant driver or if natural processes, such as solar variability and volcanic activity, exert greater influence on stratospheric ozone levels. Proponents of anthropogenic dominance cite the catalytic chlorine and bromine cycles initiated by ODS photolysis in the stratosphere, which amplify ozone destruction rates by factors of 10^4 to 10^6 per halogen atom under cold polar conditions.[1] This mechanism was empirically validated through aircraft campaigns like the 1987 Airborne Antarctic Ozone Experiment (AAOE), which measured elevated ClO radicals (up to 1 ppbv) and anticorrelated O3 and ClO in the Antarctic vortex, inconsistent with natural halogen sources alone.[213] Atmospheric chlorine loading peaked at approximately 3.7 ppb in the late 1990s, directly correlating with a 3-5% global ozone decline and over 50% loss in the Antarctic spring minimum from 1979 to 1995.[7] Isotopic analysis of chlorine further distinguishes anthropogenic contributions, revealing fractionation patterns (δ37Cl shifts of up to +1.5‰) during CFC photodissociation that match stratospheric observations but differ from natural reservoirs like sea salt or volcanic HCl, which photolyze differently and contribute negligibly to active stratospheric chlorine (<0.1 ppb).[214] [215] Post-1987 Montreal Protocol reductions in ODS emissions have driven a 20% decline in equivalent effective stratospheric chlorine (EESC) since 2000, coinciding with measurable ozone recovery: Antarctic column ozone increased by 1-3 Dobson units per decade, and the 2023 ozone hole was the fourth-smallest since 1992, supporting causal linkage over coincidence.[216] Natural factors alone fail to account for this reversal, as solar cycles and volcanism exhibit no comparable long-term upward trend.[1] Advocates for natural dominance argue that solar irradiance variations, including the 11-year cycle and grand solar minima, modulate ozone production via UV-driven O2 dissociation, with anti-correlated ozone minima during low solar activity (e.g., ~2% global decrease during the 1986 minimum).[217] Volcanic eruptions, such as El Chichón (1982) and Pinatubo (1991), injected sulfate aerosols that enhanced polar stratospheric cloud (PSC) formation, temporarily boosting heterogeneous Cl activation and causing 5-8% global ozone loss for 1-2 years, suggesting amplified natural halogen cycling.[55] Some researchers propose persistent natural sources, like HCl emissions from Mount Erebus, could sustain Antarctic chlorine levels (estimated 0.5-1 pptv flux), potentially explaining hole persistence without invoking CFCs.[218] Critics including physicist S. Fred Singer contended in the 1980s-1990s that depletion models overestimated CFC impacts, attributing trends to dynamical variability (e.g., quasi-biennial oscillation strengthening the polar vortex) and unverified pre-1979 balloon data indicating episodic low-ozone events.[219] These natural hypotheses, often advanced by industry-affiliated skeptics or contrarian outlets, struggle against empirical counterevidence: solar cycles explain <10% of observed trends, volcanic effects are short-lived and halogen-limited without ODS, and dynamical variability modulates hole size by 20-30% annually but not the multi-decadal decline.[1] [41] Peer-reviewed assessments, synthesizing satellite (TOMS/Nimbus-7 from 1979), ozonesonde, and lidar data, affirm anthropogenic ODS as the primary causal agent, with natural processes secondary amplifiers; dissenting views, while highlighting model uncertainties (e.g., PSC parameterization errors), lack quantitative replication of the observed spatiotemporal depletion signature.[220] Recovery trajectories, projected to near-1980 levels by 2066 barring violations, underscore this hierarchy, though ongoing monitoring addresses potential feedbacks like N2O increases from agriculture.[1]Critiques of Risk Assessments and Model Predictions
Critics have argued that early risk assessments overestimated the global impacts of ozone depletion by focusing on worst-case scenarios for ultraviolet-B (UV-B) radiation increases and associated health effects, such as a projected quadrupling of skin cancer incidence under unrestricted chlorofluorocarbon (CFC) emissions. These assessments, informed by models linking ozone loss to heightened UV exposure, anticipated millions of excess non-melanoma skin cancers annually in mid-latitudes, yet post-depletion epidemiological data from the 1980s onward show rising skin cancer rates primarily correlated with behavioral factors like increased tanning and outdoor recreation rather than stratospheric changes, with global UV-B enhancements limited to about 1-2% outside polar regions.[221][222] Atmospheric physicist S. Fred Singer, in congressional testimony and publications, contended that such projections ignored confounding variables and natural ozone variability, suggesting the emphasis on anthropogenic causes amplified perceived risks beyond empirical evidence at the time.[223] Model predictions have been critiqued for initial inaccuracies in forecasting the Antarctic ozone hole's extent and dynamics, as pre-1980s simulations based on gas-phase chemistry projected only modest global ozone reductions of 2-4% over centuries, underestimating the rapid, localized depletions triggered by heterogeneous reactions on polar stratospheric clouds (PSCs). This oversight stemmed from incomplete incorporation of seasonal vortex isolation and chlorine activation mechanisms, requiring post hoc adjustments after the hole's 1985 discovery, which some analysts viewed as evidence of premature policy reliance on unvalidated projections.[224][225] Furthermore, skeptics highlighted models' sensitivity to natural forcings, such as the quasi-biennial oscillation, solar cycles, and volcanic eruptions like Mount Pinatubo in 1991, which temporarily exacerbated depletion by 5-10% independently of CFCs, arguing that risk assessments downplayed these to emphasize human contributions.[10] Recovery projections under the Montreal Protocol have also drawn scrutiny for optimistic timelines amid persistent uncertainties in coupled ozone-climate interactions, with some studies indicating that greenhouse gas-driven stratospheric cooling could prolong PSC formation and delay Antarctic hole closure beyond mid-century estimates. Critics, including those referencing ensemble model discrepancies, note that while total column ozone has rebounded 20% in austral spring since 2000, interannual variability exceeds predicted trends, complicating attribution to CFC phaseout alone and raising questions about the robustness of long-term forecasts.[1][57]Policy Overreach Claims and Unintended Consequences
Critics of the Montreal Protocol, including atmospheric physicist S. Fred Singer, have argued that the 1987 agreement represented policy overreach by imposing a global phaseout of chlorofluorocarbons (CFCs) based on speculative models and insufficient peer-reviewed evidence linking CFCs directly to stratospheric ozone depletion.[10] Singer contended that natural factors, such as solar cycles and volcanic eruptions, better explained observed ozone variations, including the Antarctic ozone hole, and that the policy response prioritized alarmism over rigorous validation, leading to premature economic restrictions.[10] Industry representatives, including from DuPont and Pennwalt, warned Congress in the 1980s of potential "economic chaos," estimating compliance costs exceeding $135 billion globally, alongside significant job losses in refrigeration and manufacturing sectors.[10] Subsequent assessments quantified the phaseout's financial burden, with the United Nations Environment Programme (UNEP) reporting in 1994 that businesses incurred approximately $37 billion in conversion costs, while consumers faced an additional $3 billion, though proponents emphasized offsetting health benefits like averting 19.1 million skin cancer cases.[10] Critics maintained these figures understated long-term disruptions, including supply chain shifts and innovation stifling, arguing the treaty's trade sanctions on non-compliant nations pressured developing economies without adequate technological transfer, exacerbating inequities.[150] The protocol's allowance for hydrochlorofluorocarbons (HCFCs) as transitional substitutes, which themselves deplete ozone albeit less potently, further fueled claims of hasty implementation lacking first-order causal scrutiny.[194] Unintended consequences included the widespread adoption of hydrofluorocarbons (HFCs) as CFC replacements, which, while ozone-neutral, possess global warming potentials thousands of times greater than carbon dioxide, contributing an estimated 0.5°C of avoided warming if phased down but initially amplifying radiative forcing.[194] This substitution shifted environmental pressures from ozone to tropospheric warming, necessitating the 2016 Kigali Amendment to curb HFC production, yet critics like those at the Heritage Foundation highlighted additional U.S. economic costs from HFC restrictions, projecting billions in higher refrigeration and air-conditioning expenses without proportional climate gains.[226] The CFC ban also spurred black-market trade, with illegal production—such as CFC-11 emissions traced to China in 2019—driving up legitimate refrigerant prices and undermining compliance, as reduced legal supply increased scarcity-driven costs for maintenance in developing regions.[227][228] Furthermore, stratospheric ozone recovery has been linked to potential enhancements in planetary-scale warming dynamics, as diminished depletion reduces stratospheric cooling effects that previously moderated surface temperatures.[229]References
- https://earthobservatory.[nasa](/page/NASA).gov/features/Ozone/ozone_2.php

