Uranium
View on Wikipedia
 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Uranium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pronunciation | /jʊˈreɪniəm/ ⓘ | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | silvery gray metallic; corrodes to a spalling black oxide coat in air | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Standard atomic weight Ar°(U) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Uranium in the periodic table | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic number (Z) | 92 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group | f-block groups (no number) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Period | period 7 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Block | f-block | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Rn] 5f3 6d1 7s2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2, 8, 18, 32, 21, 9, 2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase at STP | solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | 1405.3 K (1132.2 °C, 2070 °F) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | 4404 K (4131 °C, 7468 °F) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density (at 20° C) | 19.050 g/cm3 [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| when liquid (at m.p.) | 17.3 g/cm3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of fusion | 9.14 kJ/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of vaporization | 417.1 kJ/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar heat capacity | 27.665 J/(mol·K) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
Vapor pressure
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Oxidation states | common: +6 −1,[4] +1,? +2,? +3,[5] +4,[6] +5[6] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electronegativity | Pauling scale: 1.38 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ionization energies |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic radius | empirical: 156 pm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Covalent radius | 196±7 pm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Van der Waals radius | 186 pm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Natural occurrence | primordial | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal structure | orthorhombic (oS4) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lattice constants | a = 285.35 pm b = 586.97 pm c = 495.52 pm (at 20 °C)[3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal expansion | 15.46×10−6/K (at 20 °C)[a] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal conductivity | 27.5 W/(m⋅K) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrical resistivity | 0.280 µΩ⋅m (at 0 °C) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Magnetic ordering | paramagnetic | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Young's modulus | 208 GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Shear modulus | 111 GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bulk modulus | 100 GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Speed of sound thin rod | 3155 m/s (at 20 °C) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Poisson ratio | 0.23 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vickers hardness | 1960–2500 MPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Brinell hardness | 2350–3850 MPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Number | 7440-61-1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Naming | after planet Uranus, itself named after Greek god of the sky Uranus | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Discovery | Martin Heinrich Klaproth (1789) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| First isolation | Eugène-Melchior Péligot (1841) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes of uranium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||
Uranium is a chemical element; it has symbol U and atomic number 92. It is a silvery-grey metal in the actinide series of the periodic table. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons. Uranium radioactively decays, usually by emitting an alpha particle. The half-life of this decay varies between 159,200 and 4.5 billion years for different isotopes, making them useful for dating the age of the Earth. The most common isotopes in natural uranium are uranium-238 (which has 146 neutrons and accounts for over 99% of uranium on Earth) and uranium-235 (which has 143 neutrons). Uranium has the highest atomic weight of the primordially occurring elements. Its density is about 70% higher than that of lead and slightly lower than that of gold or tungsten. It occurs naturally in low concentrations of a few parts per million in soil, rock and water, and is commercially extracted from uranium-bearing minerals such as uraninite.[8]
Many contemporary uses of uranium exploit its unique nuclear properties. Uranium is used in nuclear power plants and nuclear weapons because it is the only naturally occurring element with a fissile isotope – uranium-235 – present in non-trace amounts. However, because of the low abundance of uranium-235 in natural uranium (which is overwhelmingly uranium-238), uranium needs to undergo enrichment so that enough uranium-235 is present. Uranium-238 is fissionable by fast neutrons and is fertile, meaning it can be transmuted to fissile plutonium-239 in a nuclear reactor. Another fissile isotope, uranium-233, can be produced from natural thorium and is studied for future industrial use in nuclear technology. Uranium-238 has a small probability for spontaneous fission or even induced fission with fast neutrons; uranium-235, and to a lesser degree uranium-233, have a much higher fission cross-section for slow neutrons. In sufficient concentration, these isotopes maintain a sustained nuclear chain reaction. This generates the heat in nuclear power reactors and produces the fissile material for nuclear weapons. The primary civilian use for uranium harnesses the heat energy to produce electricity. Depleted uranium (238U) is used in kinetic energy penetrators and armor plating.[9]
The 1789 discovery of uranium in the mineral pitchblende is credited to Martin Heinrich Klaproth, who named the new element after the recently discovered planet Uranus. Eugène-Melchior Péligot was the first person to isolate the metal, and its radioactive properties were discovered in 1896 by Henri Becquerel. Research by Otto Hahn, Lise Meitner, Enrico Fermi and others, such as J. Robert Oppenheimer starting in 1934 led to its use as a fuel in the nuclear power industry and in Little Boy, the first nuclear weapon used in war. An ensuing arms race during the Cold War between the United States and the Soviet Union produced tens of thousands of nuclear weapons that used uranium metal and uranium-derived plutonium-239. Dismantling of these weapons and related nuclear facilities is carried out within various nuclear disarmament programs and costs billions of dollars. Weapon-grade uranium obtained from nuclear weapons is diluted with uranium-238 and reused as fuel for nuclear reactors. Spent nuclear fuel forms radioactive waste, which mostly consists of uranium-238 and poses a significant health threat and environmental impact.
Characteristics
[edit]
Uranium is a silvery white, weakly radioactive metal. It has a Mohs hardness of 6, sufficient to scratch glass and roughly equal to that of titanium, rhodium, manganese and niobium. It is malleable, ductile, slightly paramagnetic, strongly electropositive and a poor electrical conductor.[10][11] Uranium metal has a very high density of 19.1 g/cm3,[12] denser than lead (11.3 g/cm3),[13] but slightly less dense than tungsten and gold (19.3 g/cm3).[14][15]
Uranium metal reacts with almost all non-metallic elements (except noble gases) and their compounds, with reactivity increasing with temperature.[16] Hydrochloric and nitric acids dissolve uranium, but non-oxidizing acids other than hydrochloric acid attack the element very slowly.[10] When finely divided, it can react with cold water; in air, uranium metal becomes coated with a dark layer of uranium dioxide.[11] Uranium in ores is extracted chemically and converted into uranium dioxide or other chemical forms usable in industry.
Uranium-235 was the first isotope that was found to be fissile. Other naturally occurring isotopes are fissionable, but not fissile.[citation needed] On bombardment with slow neutrons, uranium-235 most of the time splits into two smaller nuclei, releasing nuclear binding energy and more neutrons. If too many of these neutrons are absorbed by other uranium-235 nuclei, a nuclear chain reaction occurs that results in a burst of heat or (in some circumstances) an explosion. In a nuclear reactor, such a chain reaction is slowed and controlled by a neutron poison, absorbing some of the free neutrons. Such neutron absorbent materials are often part of reactor control rods (see nuclear reactor physics for a description of this process of reactor control).
As little as 15 lb (6.8 kg) of uranium-235 can be used to make an atomic bomb.[17] The nuclear weapon detonated over Hiroshima, called Little Boy, relied on uranium fission. However, the first nuclear bomb (the Gadget used at Trinity) and the bomb that was detonated over Nagasaki (Fat Man) were both plutonium bombs.
Uranium metal has three allotropic forms:[18]
- α (orthorhombic) stable up to 668 °C (1,234 °F). Orthorhombic, space group No. 63, Cmcm, lattice parameters a = 285.4 pm, b = 587 pm, c = 495.5 pm.[19]
- β (tetragonal) stable from 668 to 775 °C (1,234 to 1,427 °F). Tetragonal, space group P42/mnm, P42nm, or P4n2, lattice parameters a = 565.6 pm, b = c = 1075.9 pm.[19]
- γ (body-centered cubic) from 775 °C (1,427 °F) to melting point—this is the most malleable and ductile state. Body-centered cubic, lattice parameter a = 352.4 pm.[19]
Applications
[edit]Military
[edit]
The major application of uranium in the military sector is in high-density penetrating projectiles. This ammunition consists of depleted uranium (DU) alloyed with 1–2% other elements, such as titanium or molybdenum.[20] At high impact speed, the density, hardness, and pyrophoricity of the projectile enable the destruction of heavily armored targets. Tank armor and other removable vehicle armor can also be hardened with depleted uranium plates. The use of depleted uranium became politically and environmentally contentious after the use of such munitions by the US, UK and other countries during wars in the Persian Gulf and the Balkans raised health questions concerning uranium compounds left in the soil (see Gulf War syndrome).[17]
Depleted uranium is also used as a shielding material in some containers used to store and transport radioactive materials. While the metal itself is radioactive, its high density makes it more effective than lead in halting radiation from strong sources such as radium.[10] Other uses of depleted uranium include counterweights for aircraft control surfaces, as ballast for missile re-entry vehicles and as a shielding material.[11] Due to its high density, this material is found in inertial guidance systems and in gyroscopic compasses.[11] Depleted uranium is preferred over similarly dense metals due to its ability to be easily machined and cast as well as its relatively low cost.[21] The main risk of exposure to depleted uranium is chemical poisoning by uranium oxide rather than radioactivity (uranium being only a weak alpha emitter).
During the later stages of World War II, the entire Cold War, and to a lesser extent afterwards, uranium-235 has been used as the fissile explosive material to produce nuclear weapons. Initially, two major types of fission bombs were built: a relatively simple device that uses uranium-235 and a more complicated mechanism that uses plutonium-239 derived from uranium-238. Later, a much more complicated and far more powerful type of fission/fusion bomb (thermonuclear weapon) was built, that uses a plutonium-based device to cause a mixture of tritium and deuterium to undergo nuclear fusion. Such bombs are jacketed in a non-fissile (unenriched) uranium case, and they derive more than half their power from the fission of this material by fast neutrons from the nuclear fusion process.[22]
Civilian
[edit]The main use of uranium in the civilian sector is to fuel nuclear power plants. One kilogram of uranium-235 can theoretically produce about 20 terajoules of energy (2×1013 joules), assuming complete fission; as much energy as 1.5 million kilograms (1,500 tonnes) of coal.[9]
Commercial nuclear power plants use fuel that is typically enriched to around 3% uranium-235.[9] The CANDU and Magnox designs are the only commercial reactors capable of using unenriched uranium fuel. Fuel used for United States Navy reactors is typically highly enriched in uranium-235 (the exact values are classified). In a breeder reactor, uranium-238 can also be converted into plutonium-239 through the following reaction:[11]

Before (and, occasionally, after) the discovery of radioactivity, uranium was primarily used in small amounts for yellow glass and pottery glazes, such as uranium glass and in Fiestaware.[23]
The discovery and isolation of radium in uranium ore (pitchblende) by Marie Curie sparked the development of uranium mining to extract the radium, which was used to make glow-in-the-dark paints for clock and aircraft dials.[24][25] This left a prodigious quantity of uranium as a waste product, since it takes three tonnes of uranium to extract one gram of radium. This waste product was diverted to the glazing industry, making uranium glazes very inexpensive and abundant. Besides the pottery glazes, uranium tile glazes accounted for the bulk of the use, including common bathroom and kitchen tiles which can be produced in green, yellow, mauve, black, blue, red and other colors.


Uranium was also used in photographic chemicals (especially uranium nitrate as a toner),[11] in lamp filaments for stage lighting bulbs,[26] to improve the appearance of dentures,[27] and in the leather and wood industries for stains and dyes. Uranium salts are mordants of silk or wool. Uranyl acetate and uranyl formate are used as electron-dense "stains" in transmission electron microscopy, to increase the contrast of biological specimens in ultrathin sections and in negative staining of viruses, isolated cell organelles and macromolecules.
The discovery of the radioactivity of uranium ushered in additional scientific and practical uses of the element. The long half-life of uranium-238 (4.47×109 years) makes it well-suited for use in estimating the age of the earliest igneous rocks and for other types of radiometric dating, including uranium–thorium dating, uranium–lead dating and uranium–uranium dating. Uranium metal is used for X-ray targets in the making of high-energy X-rays.[11]
History
[edit]Pre-discovery use
[edit]The use of pitchblende, uranium in its natural oxide form, dates back to at least the year 79 AD, when it was used in the Roman Empire to add a yellow color to ceramic glazes.[11] Yellow glass with 1% uranium oxide was found in a Roman villa on Cape Posillipo in the Gulf of Naples, Italy, by R. T. Gunther of the University of Oxford in 1912.[28] Starting in the late Middle Ages, pitchblende was extracted from the Habsburg silver mines in Joachimsthal, Bohemia (now Jáchymov in the Czech Republic) in the Ore Mountains, and was used as a coloring agent in the local glassmaking industry.[29] In the early 19th century, the world's only known sources of uranium ore were these mines.
Discovery
[edit]
The discovery of the element is credited to the German chemist Martin Heinrich Klaproth. While he was working in his experimental laboratory in Berlin in 1789, Klaproth was able to precipitate a yellow compound (likely sodium diuranate) by dissolving pitchblende in nitric acid and neutralizing the solution with sodium hydroxide.[29] Klaproth assumed the yellow substance was the oxide of a yet-undiscovered element and heated it with charcoal to obtain a black powder, which he thought was the newly discovered metal itself (in fact, that powder was an oxide of uranium).[29][30] He named the newly discovered element "Uranit" after the planet Uranus (named after the primordial Greek god of the sky), which had been discovered eight years earlier by William Herschel.[31] He later renamed it "Uranium" to conform to the naming standard.[32]
In 1841, Eugène-Melchior Péligot, Professor of Analytical Chemistry at the Conservatoire National des Arts et Métiers (Central School of Arts and Manufactures) in Paris, isolated the first sample of uranium metal by heating uranium tetrachloride with potassium.[29][33]

Henri Becquerel discovered radioactivity by using uranium in 1896.[16] Becquerel made the discovery in Paris by leaving a sample of a uranium salt, K2UO2(SO4)2 (potassium uranyl sulfate), on top of an unexposed photographic plate in a drawer and noting that the plate had become "fogged".[34] He determined that a form of invisible light or rays emitted by uranium had exposed the plate.
During World War I when the Central Powers suffered a shortage of molybdenum to make artillery gun barrels and high speed tool steels, they routinely used ferrouranium alloy as a substitute, as it presents many of the same physical characteristics as molybdenum. When this practice became known in 1916 the US government requested several prominent universities to research the use of uranium in manufacturing and metalwork. Tools made with these formulas remained in use for several decades,[35][36] until the Manhattan Project and the Cold War placed a large demand on uranium for fission research and weapon development.
Fission research
[edit]
A team led by Enrico Fermi in 1934 found that bombarding uranium with neutrons produces beta rays (electrons or positrons from the elements produced; see beta particle).[37] The fission products were at first mistaken for new elements with atomic numbers 93 and 94, which the Dean of the Sapienza University of Rome, Orso Mario Corbino, named ausenium and hesperium, respectively.[38][39][40][41] The experiments leading to the discovery of uranium's ability to fission (break apart) into lighter elements and release binding energy were conducted by Otto Hahn and Fritz Strassmann[37] in Hahn's laboratory in Berlin. Lise Meitner and her nephew, physicist Otto Robert Frisch, published the physical explanation in February 1939 and named the process "nuclear fission".[42] Soon after, Fermi hypothesized that fission of uranium might release enough neutrons to sustain a fission reaction. Confirmation of this hypothesis came in 1939, and later work found that on average about 2.5 neutrons are released by each fission of uranium-235.[37] Fermi urged Alfred O. C. Nier to separate uranium isotopes for determination of the fissile component, and on 29 February 1940, Nier used an instrument he built at the University of Minnesota to separate the world's first uranium-235 sample in the Tate Laboratory. Using Columbia University's cyclotron, John Dunning confirmed the sample to be the isolated fissile material on 1 March.[43] Further work found that the far more common uranium-238 isotope can be transmuted into plutonium, which, like uranium-235, is also fissile by thermal neutrons. These discoveries led numerous countries to begin working on the development of nuclear weapons and nuclear power. Despite fission having been discovered in Germany, the Uranverein ("uranium club") Germany's wartime project to research nuclear power and/or weapons was hampered by limited resources, infighting, the exile or non-involvement of several prominent scientists in the field and several crucial mistakes such as failing to account for impurities in available graphite samples which made it appear less suitable as a neutron moderator than it is in reality. Germany's attempts to build a natural uranium / heavy water reactor had not come close to reaching criticality by the time the Americans reached Haigerloch, the site of the last German wartime reactor experiment.[44]
On 2 December 1942, as part of the Manhattan Project, another team led by Enrico Fermi was able to initiate the first artificial self-sustained nuclear chain reaction, Chicago Pile-1. An initial plan using enriched uranium-235 was abandoned as it was as yet unavailable in sufficient quantities.[45] Working in a lab below the stands of Stagg Field at the University of Chicago, the team created the conditions needed for such a reaction by piling together 360 tonnes of graphite, 53 tonnes of uranium oxide, and 5.5 tonnes of uranium metal, most of which was supplied by Westinghouse Lamp Plant in a makeshift production process.[37][46]
Nuclear weaponry
[edit]
Two types of atomic bomb were developed by the United States during World War II: a uranium-based device (codenamed "Little Boy") whose fissile material was highly enriched uranium, and a plutonium-based device (see Trinity test and "Fat Man") whose plutonium was derived from uranium-238. Little Boy became the first nuclear weapon used in war when it was detonated over Hiroshima, Japan, on 6 August 1945. Exploding with a yield equivalent to 12,500 tonnes of TNT, the blast and thermal wave of the bomb destroyed nearly 50,000 buildings and killed about 75,000 people (see Atomic bombings of Hiroshima and Nagasaki).[34]
In 1943 the Manhattan Project contracted two private companies, Union Carbide and Chevron, to quietly compile a survey of uranium deposits around the world. As the survey results came in, two geology professors studied the results and suggested general guidelines for new sources, including uranium associated with gold mines in the Rand area in South Africa.[47]
Initially it was believed that uranium was relatively rare, and that nuclear proliferation could be avoided by simply buying up all known uranium stocks, but within a decade large deposits of it were discovered in many places around the world.[48]
Reactors
[edit]
The X-10 Graphite Reactor at Oak Ridge National Laboratory (ORNL) in Oak Ridge, Tennessee, formerly known as the Clinton Pile and X-10 Pile, was the world's second artificial nuclear reactor (after Enrico Fermi's Chicago Pile) and was the first reactor designed and built for continuous operation. Argonne National Laboratory's Experimental Breeder Reactor I, located at the Atomic Energy Commission's National Reactor Testing Station near Arco, Idaho, became the first nuclear reactor to create electricity on 20 December 1951.[49] Initially, four 150-watt light bulbs were lit by the reactor, but improvements eventually enabled it to power the whole facility (later, the town of Arco became the first in the world to have all its electricity come from nuclear power generated by BORAX-III, another reactor designed and operated by Argonne National Laboratory).[50][51] The world's first commercial scale nuclear power station, Obninsk in the Soviet Union, began generation with its reactor AM-1 on 27 June 1954. Other early nuclear power plants were Calder Hall in England, which began generation on 17 October 1956,[52] and the Shippingport Atomic Power Station in Pennsylvania, which began on 26 May 1958. Nuclear power was used for the first time for propulsion by a submarine, the USS Nautilus, in 1954.[37][53]
Prehistoric naturally occurring fission
[edit]In 1972, French physicist Francis Perrin discovered fifteen ancient and no longer active natural nuclear fission reactors in three separate ore deposits at the Oklo mine in Gabon, Africa, collectively known as the Oklo Fossil Reactors. The ore deposit is 1.7 billion years old; then, uranium-235 constituted about 3% of uranium on Earth.[54] This is high enough to permit a sustained chain reaction, if other supporting conditions exist. The capacity of the surrounding sediment to contain the health-threatening nuclear waste products has been cited by the U.S. federal government as supporting evidence for the feasibility to store spent nuclear fuel at the Yucca Mountain nuclear waste repository.[54]
Contamination and the Cold War legacy
[edit]
Above-ground nuclear tests by the Soviet Union and the United States in the 1950s and early 1960s and by France into the 1970s and 1980s[21] spread a significant amount of fallout from uranium daughter isotopes around the world.[55] Additional fallout and pollution occurred from several nuclear accidents.[56]
Uranium miners have a higher incidence of cancer. An excess risk of lung cancer among Navajo uranium miners, for example, has been documented and linked to their occupation.[57] The Radiation Exposure Compensation Act, a 1990 law in the US, required $100,000 in "compassion payments" to uranium miners diagnosed with cancer or other respiratory ailments.[58]
During the Cold War between the Soviet Union and the United States, huge stockpiles of uranium were amassed and tens of thousands of nuclear weapons were created using enriched uranium and plutonium made from uranium. After the break-up of the Soviet Union in 1991, an estimated 600 short tons (540 metric tons) of highly enriched weapons grade uranium (enough to make 40,000 nuclear warheads) had been stored in often inadequately guarded facilities in the Russian Federation and several other former Soviet states.[17] Police in Asia, Europe, and South America on at least 16 occasions from 1993 to 2005 have intercepted shipments of smuggled bomb-grade uranium or plutonium, most of which was from ex-Soviet sources.[17] From 1993 to 2005 the Material Protection, Control, and Accounting Program, operated by the federal government of the United States, spent about US$550 million to help safeguard uranium and plutonium stockpiles in Russia. This money was used for improvements and security enhancements at research and storage facilities.[17]
Safety of nuclear facilities in Russia has been significantly improved since the stabilization of political and economical turmoil of the early 1990s. For example, in 1993 there were 29 incidents ranking above level 1 on the International Nuclear Event Scale, and this number dropped under four per year in 1995–2003. The number of employees receiving annual radiation doses above 20 mSv, which is equivalent to a single full-body CT scan,[59] saw a strong decline around 2000. In November 2015, the Russian government approved a federal program for nuclear and radiation safety for 2016 to 2030 with a budget of 562 billion rubles (ca. 8 billion USD). Its key issue is "the deferred liabilities accumulated during the 70 years of the nuclear industry, particularly during the time of the Soviet Union". About 73% of the budget will be spent on decommissioning aged and obsolete nuclear reactors and nuclear facilities, especially those involved in state defense programs; 20% will go in processing and disposal of nuclear fuel and radioactive waste, and 5% into monitoring and ensuring of nuclear and radiation safety.[60]
Occurrence
[edit]Uranium is a naturally occurring element found in low levels in all rock, soil, and water. It is the highest-numbered element found naturally in significant quantities on Earth and is almost always found combined with other elements.[11] Uranium is the 48th most abundant element in the Earth's crust.[61] The decay of uranium, thorium, and potassium-40 in Earth's mantle is thought to be the main source of heat[62][63] that keeps the Earth's outer core in the liquid state and drives mantle convection, which in turn drives plate tectonics.
Uranium's concentration in the Earth's crust is (depending on the reference) 2 to 4 parts per million,[10][21] or about 40 times as abundant as silver.[16] The Earth's crust from the surface to 25 km (15 mi) down is calculated to contain 1017 kg (2×1017 lb) of uranium while the oceans may contain 1013 kg (2×1013 lb).[10] The concentration of uranium in soil ranges from 0.7 to 11 parts per million (up to 15 parts per million in farmland soil due to use of phosphate fertilizers containing uranium impurities),[64] and its concentration in sea water is 3 parts per billion.[21]
Uranium is more plentiful than antimony, tin, cadmium, mercury, or silver, and it is about as abundant as arsenic or molybdenum.[11][21] Uranium is found in hundreds of minerals, including uraninite (the most common uranium ore), carnotite, autunite, uranophane, torbernite, and coffinite.[11] Significant concentrations of uranium occur in some substances such as phosphate rock deposits, and minerals such as lignite, and monazite sands in uranium-rich ores[11] (it is recovered commercially from sources with as little as 0.1% uranium[16]).
Origin
[edit]Like all elements with atomic weights higher than that of iron, uranium is only naturally formed by the r-process (rapid neutron capture) in supernovae and neutron star mergers.[65] Primordial thorium and uranium are only produced in the r-process, because the s-process (slow neutron capture) is too slow and cannot pass the gap of instability after bismuth.[66][67] Besides the two extant primordial uranium isotopes, 235U and 238U, the r-process also produced significant quantities of 236U, which has a shorter half-life and so is an extinct radionuclide, having long since decayed completely to 232Th. Further uranium-236 was produced by the decay of 244Pu, accounting for the observed higher-than-expected abundance of thorium and lower-than-expected abundance of uranium.[68] While the natural abundance of uranium has been supplemented by the decay of extinct 242Pu (half-life 375,000 years) and 247Cm (half-life 16 million years), producing 238U and 235U respectively, this occurred to an almost negligible extent due to the shorter half-lives of these parents and their lower production than 236U and 244Pu, the parents of thorium: the 247Cm/235U ratio at the formation of the Solar System was (7.0±1.6)×10−5.[69]
Biotic and abiotic
[edit]

Some bacteria, such as Shewanella putrefaciens, Geobacter metallireducens and some strains of Burkholderia fungorum, can use uranium for their growth and convert U(VI) to U(IV).[70][71] Recent research suggests that this pathway includes reduction of the soluble U(VI) via an intermediate U(V) pentavalent state.[72][73] Other organisms, such as the lichen Trapelia involuta or microorganisms such as the bacterium Citrobacter, can absorb concentrations of uranium that are up to 300 times the level of their environment.[74] Citrobacter species absorb uranyl ions when given glycerol phosphate (or other similar organic phosphates). After one day, one gram of bacteria can encrust themselves with nine grams of uranyl phosphate crystals; this creates the possibility that these organisms could be used in bioremediation to decontaminate uranium-polluted water.[29][75] The proteobacterium Geobacter has also been shown to bioremediate uranium in ground water.[76] The mycorrhizal fungus Glomus intraradices increases uranium content in the roots of its symbiotic plant.[77]
In nature, uranium(VI) forms highly soluble carbonate complexes at alkaline pH. This leads to an increase in mobility and availability of uranium to groundwater and soil from nuclear wastes which leads to health hazards. However, it is difficult to precipitate uranium as phosphate in the presence of excess carbonate at alkaline pH. A Sphingomonas sp. strain BSAR-1 has been found to express a high activity alkaline phosphatase (PhoK) that has been applied for bioprecipitation of uranium as uranyl phosphate species from alkaline solutions. The precipitation ability was enhanced by overexpressing PhoK protein in E. coli.[78]
Plants absorb some uranium from soil. Dry weight concentrations of uranium in plants range from 5 to 60 parts per billion, and ash from burnt wood can have concentrations up to 4 parts per million.[29] Dry weight concentrations of uranium in food plants are typically lower with one to two micrograms per day ingested through the food people eat.[29]
Production and mining
[edit]Worldwide production of uranium in 2021 was 48,332 tonnes, of which 21,819 t (45%) was mined in Kazakhstan. Other important uranium mining countries are Namibia (5,753 t), Canada (4,693 t), Australia (4,192 t), Uzbekistan (3,500 t), and Russia (2,635 t).[79]
Uranium ore is mined in several ways: open pit, underground, in-situ leaching, and borehole mining.[9] Low-grade uranium ore mined typically contains 0.01 to 0.25% uranium oxides. Extensive measures must be employed to extract the metal from its ore.[80] High-grade ores found in Athabasca Basin deposits in Saskatchewan, Canada can contain up to 23% uranium oxides on average.[81] Uranium ore is crushed and rendered into a fine powder and then leached with either an acid or alkali. The leachate is subjected to one of several sequences of precipitation, solvent extraction, and ion exchange. The resulting mixture, called yellowcake, contains at least 75% uranium oxides U3O8. Yellowcake is then calcined to remove impurities from the milling process before refining and conversion.[82]
Commercial-grade uranium can be produced through the reduction of uranium halides with alkali or alkaline earth metals.[11] Uranium metal can also be prepared through electrolysis of KUF
5 or
UF
4, dissolved in molten calcium chloride (CaCl
2) and sodium chloride (NaCl) solution.[11] Very pure uranium is produced through the thermal decomposition of uranium halides on a hot filament.[11]
-
World uranium production (mines) and demand[79]
-
Yellowcake is a concentrated mixture of uranium oxides that is further refined to extract pure uranium.
-
Uranium production 2015, in tonnes[83]
Resources and reserves
[edit]
It is estimated that 6.1 million tonnes of uranium exists in ores that are economically viable at US$130 per kg of uranium,[84] while 35 million tonnes are classed as mineral resources (reasonable prospects for eventual economic extraction).[85]
Australia has 28% of the world's known uranium ore reserves[84] and the world's largest single uranium deposit is located at the Olympic Dam Mine in South Australia.[86] There is a significant reserve of uranium in Bakouma, a sub-prefecture in the prefecture of Mbomou in the Central African Republic.[87]
Some uranium also originates from dismantled nuclear weapons.[88] For example, in 1993–2013 Russia supplied the United States with 15,000 tonnes of low-enriched uranium within the Megatons to Megawatts Program.[89]
An additional 4.6 billion tonnes of uranium are estimated to be dissolved in sea water (Japanese scientists in the 1980s showed that extraction of uranium from sea water using ion exchangers was technically feasible).[90][91] There have been experiments to extract uranium from sea water,[92] but the yield has been low due to the carbonate present in the water. In 2012, ORNL researchers announced the successful development of a new absorbent material dubbed HiCap which performs surface retention of solid or gas molecules, atoms or ions and also effectively removes toxic metals from water, according to results verified by researchers at Pacific Northwest National Laboratory.[93][94]
Supplies
[edit]
In 2005, ten countries accounted for the majority of the world's concentrated uranium oxides: Canada (27.9%), Australia (22.8%), Kazakhstan (10.5%), Russia (8.0%), Namibia (7.5%), Niger (7.4%), Uzbekistan (5.5%), the United States (2.5%), Argentina (2.1%) and Ukraine (1.9%).[96] In 2008, Kazakhstan was forecast to increase production and become the world's largest supplier of uranium by 2009;[97][98] Kazakhstan has dominated the world's uranium market since 2010. In 2021, its share was 45.1%, followed by Namibia (11.9%), Canada (9.7%), Australia (8.7%), Uzbekistan (7.2%), Niger (4.7%), Russia (5.5%), China (3.9%), India (1.3%), Ukraine (0.9%), and South Africa (0.8%), with a world total production of 48,332 tonnes.[79] Most uranium was produced not by conventional underground mining of ores (29% of production), but by in situ leaching (66%).[79][99]
In the late 1960s, UN geologists discovered major uranium deposits and other rare mineral reserves in Somalia. The find was the largest of its kind, with industry experts estimating the deposits at over 25% of the world's then known uranium reserves of 800,000 tons.[100]
The ultimate available supply is believed to be sufficient for at least the next 85 years,[85] though some studies indicate underinvestment in the late twentieth century may produce supply problems in the 21st century.[101] Uranium deposits seem to be log-normal distributed. There is a 300-fold increase in the amount of uranium recoverable for each tenfold decrease in ore grade.[102] In other words, there is little high grade ore and proportionately much more low grade ore available.
Compounds
[edit]
Oxidation states and oxides
[edit]Oxides
[edit]Calcined uranium yellowcake, as produced in many large mills, contains a distribution of uranium oxidation species in various forms ranging from most oxidized to least oxidized. Particles with short residence times in a calciner will generally be less oxidized than those with long retention times or particles recovered in the stack scrubber. Uranium content is usually referenced to U
3O
8, which dates to the days of the Manhattan Project when U
3O
8 was used as an analytical chemistry reporting standard.[103]
Phase relationships in the uranium-oxygen system are complex. The most important oxidation states of uranium are uranium(IV) and uranium(VI), and their two corresponding oxides are, respectively, uranium dioxide (UO
2) and uranium trioxide (UO
3).[104] Other uranium oxides such as uranium monoxide (UO), diuranium pentoxide (U
2O
5), and uranium peroxide (UO
4·2H
2O) also exist.
The most common forms of uranium oxide are triuranium octoxide (U
3O
8) and UO
2.[105] Both oxide forms are solids that have low solubility in water and are relatively stable over a wide range of environmental conditions. Triuranium octoxide is (depending on conditions) the most stable compound of uranium and is the form most commonly found in nature. Uranium dioxide is the form in which uranium is most commonly used as a nuclear reactor fuel.[105] At ambient temperatures, UO
2 will gradually convert to U
3O
8. Because of their stability, uranium oxides are generally considered the preferred chemical form for storage or disposal.[105]
Aqueous chemistry
[edit]
Salts of many oxidation states of uranium are water-soluble and may be studied in aqueous solutions. The most common ionic forms are U3+
(brown-red), U4+
(green), UO+
2 (unstable), and UO2+
2 (yellow), for U(III), U(IV), U(V), and U(VI), respectively.[106] A few solid and semi-metallic compounds such as UO and US exist for the formal oxidation state uranium(II), but no simple ions are known to exist in solution for that state. Ions of U3+
liberate hydrogen from water and are therefore considered to be highly unstable. The UO2+
2 ion represents the uranium(VI) state and is known to form compounds such as uranyl carbonate, uranyl chloride and uranyl sulfate. UO2+
2 also forms complexes with various organic chelating agents, the most commonly encountered of which is uranyl acetate.[106]
Unlike the uranyl salts of uranium and polyatomic ion uranium-oxide cationic forms, the uranates, salts containing a polyatomic uranium-oxide anion, are generally not water-soluble.
Carbonates
[edit]The interactions of carbonate anions with uranium(VI) cause the Pourbaix diagram to change greatly when the medium is changed from water to a carbonate containing solution. While the vast majority of carbonates are insoluble in water (students are often taught that all carbonates other than those of alkali metals are insoluble in water), uranium carbonates are often soluble in water. This is because a U(VI) cation is able to bind two terminal oxides and three or more carbonates to form anionic complexes.
 |
 |
| Uranium in a non-complexing aqueous medium (e.g. perchloric acid/sodium hydroxide).[107] |
Uranium in carbonate solution |
 |
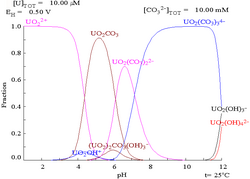 |
| Relative concentrations of the different chemical forms of uranium in a non-complexing aqueous medium (e.g. perchloric acid/sodium hydroxide).[107] |
Relative concentrations of the different chemical forms of uranium in an aqueous carbonate solution.[107] |
Effects of pH
[edit]The uranium fraction diagrams in the presence of carbonate illustrate this further: when the pH of a uranium(VI) solution increases, the uranium is converted to a hydrated uranium oxide hydroxide and at high pHs it becomes an anionic hydroxide complex.
When carbonate is added, uranium is converted to a series of carbonate complexes if the pH is increased. One effect of these reactions is increased solubility of uranium in the pH range 6 to 8, a fact that has a direct bearing on the long term stability of spent uranium dioxide nuclear fuels.
Hydrides, carbides and nitrides
[edit]Uranium metal heated to 250 to 300 °C (482 to 572 °F) reacts with hydrogen to form uranium hydride. Even higher temperatures will reversibly remove the hydrogen. This property makes uranium hydrides convenient starting materials to create reactive uranium powder along with various uranium carbide, nitride, and halide compounds.[108] Two crystal modifications of uranium hydride exist: an α form that is obtained at low temperatures and a β form that is created when the formation temperature is above 250 °C.[108]
Uranium carbides and uranium nitrides are both relatively inert semimetallic compounds that are minimally soluble in acids, react with water, and can ignite in air to form U
3O
8.[108] Carbides of uranium include uranium monocarbide (UC), uranium dicarbide (UC
2), and diuranium tricarbide (U
2C
3). Both UC and UC
2 are formed by adding carbon to molten uranium or by exposing the metal to carbon monoxide at high temperatures. Stable below 1800 °C, U
2C
3 is prepared by subjecting a heated mixture of UC and UC
2 to mechanical stress.[109] Uranium nitrides obtained by direct exposure of the metal to nitrogen include uranium mononitride (UN), uranium dinitride (UN
2), and diuranium trinitride (U
2N
3).[109]
Halides
[edit]
All uranium fluorides are created using uranium tetrafluoride (UF
4); UF
4 itself is prepared by hydrofluorination of uranium dioxide.[108] Reduction of UF
4 with hydrogen at 1000 °C produces uranium trifluoride (UF
3). Under the right conditions of temperature and pressure, the reaction of solid UF
4 with gaseous uranium hexafluoride (UF
6) can form the intermediate fluorides of U
2F
9, U
4F
17, and UF
5.[108]
At room temperatures, UF
6 has a high vapor pressure, making it useful in the gaseous diffusion process to separate the rare uranium-235 from the common uranium-238 isotope. This compound can be prepared from uranium dioxide and uranium hydride by the following process:[108]
- UO
2 + 4 HF → UF
4 + 2 H
2O (500 °C, endothermic) - UF
4 + F
2 → UF
6 (350 °C, endothermic)
The resulting UF
6, a white solid, is highly reactive (by fluorination), easily sublimes (emitting a vapor that behaves as a nearly ideal gas), and is the most volatile compound of uranium known to exist.[108]
One method of preparing uranium tetrachloride (UCl
4) is to directly combine chlorine with either uranium metal or uranium hydride. The reduction of UCl
4 by hydrogen produces uranium trichloride (UCl
3) while the higher chlorides of uranium are prepared by reaction with additional chlorine.[108] All uranium chlorides react with water and air.
Bromides and iodides of uranium are formed by direct reaction of, respectively, bromine and iodine with uranium or by adding UH
3 to those element's acids.[108] Known examples include: UBr
3, UBr
4, UI
3, and UI
4. UI
5 has never been prepared. Uranium oxyhalides are water-soluble and include UO
2F
2, UOCl
2, UO
2Cl
2, and UO
2Br
2. Stability of the oxyhalides decrease as the atomic weight of the component halide increases.[108]
Isotopes
[edit]Uranium, like all elements with an atomic number greater than 82, has no stable isotopes. All isotopes of uranium are radioactive because the strong nuclear force does not prevail over electromagnetic repulsion in nuclides containing more than 82 protons.[110] Nevertheless, the two most stable isotopes, 238U and 235U, have half-lives long enough to occur in nature as primordial radionuclides, with measurable quantities having survived since the formation of the Earth.[111] These two nuclides, along with thorium-232, are the only confirmed primordial nuclides heavier than nearly-stable bismuth-209.[7][112]
Natural uranium consists of three major isotopes: uranium-238 (99.28% natural abundance), uranium-235 (0.71%), and uranium-234 (0.0054%). There are also five other trace isotopes: uranium-240, a decay product of plutonium-244;[112] uranium-239, which is formed when 238U undergoes spontaneous fission, releasing neutrons that are captured by another 238U atom; uranium-237, which is formed when 238U captures a neutron but emits two more, which then decays to neptunium-237; uranium-236, which occurs in trace quantities due to neutron capture on 235U and as a decay product of plutonium-244;[112] and finally, uranium-233, which is formed in the decay chain of neptunium-237. Additionally, uranium-232 would be produced by the double beta decay of natural thorium-232, though this energetically possible process has never been observed.[115]
Uranium-238 is the most stable isotope of uranium, with a half-life of about 4.463×109 years,[7] roughly the age of the Earth. Uranium-238 is predominantly an alpha emitter, decaying to thorium-234. It ultimately decays through the uranium series, which has 18 members, into lead-206.[16] Uranium-238 is not fissile, but is a fertile isotope, because after neutron activation it can be converted to plutonium-239, another fissile isotope. Indeed, the 238U nucleus can absorb one neutron to produce the radioactive isotope uranium-239. 239U decays by beta emission to neptunium-239, also a beta-emitter, that decays in its turn, within a few days into plutonium-239. 239Pu was used as fissile material in the first atomic bomb detonated in the "Trinity test" on 16 July 1945 in New Mexico.[37]
Uranium-235 has a half-life of about 7.04×108 years; it is the next most stable uranium isotope after 238U and is also predominantly an alpha emitter, decaying to thorium-231.[7] Uranium-235 is important for both nuclear reactors and nuclear weapons, because it is the only uranium isotope existing in nature on Earth in significant amounts that is fissile. This means that it can be split into two or three fragments (fission products) by thermal neutrons.[16] The decay chain of 235U, which is called the actinium series, has 15 members and eventually decays into lead-207.[16] The constant rates of decay in these decay series makes the comparison of the ratios of parent to daughter elements useful in radiometric dating.
Uranium-236 has a half-life of 2.342×107 years[7] and is not found in significant quantities in nature. The half-life of uranium-236 is too short for it to be primordial, though it has been identified as an extinct progenitor of its alpha decay daughter, thorium-232.[68] Uranium-236 occurs in spent nuclear fuel when neutron capture on 235U does not induce fission, or as a decay product of plutonium-240. Uranium-236 is not fertile, as three more neutron captures are required to produce fissile 239Pu, and is not itself fissile; as such, it is considered long-lived radioactive waste.[116]
Uranium-234 is a member of the uranium series and occurs in equilibrium with its progenitor, 238U; it undergoes alpha decay with a half-life of 245,500 years[7] and decays to lead-206 through a series of relatively short-lived isotopes.
Uranium-233 undergoes alpha decay with a half-life of 160,000 years and, like 235U, is fissile.[11] It can be bred from thorium-232 via neutron bombardment, usually in a nuclear reactor; this process is known as the thorium fuel cycle. Owing to the fissility of 233U and the greater natural abundance of thorium (three times that of uranium),[117] 233U has been investigated for use as nuclear fuel as a possible alternative to 235U and 239Pu,[118] though is not in widespread use as of 2022[update].[117] The decay chain of uranium-233 forms part of the neptunium series and ends at nearly-stable bismuth-209 (half-life 2.01×1019 years)[7] and stable thallium-205.
Uranium-232 is an alpha emitter with a half-life of 68.9 years.[7] This isotope is produced as a byproduct in production of 233U and is considered a nuisance, as it is not fissile and decays through short-lived alpha and gamma emitters such as 208Tl.[118] It is also expected that thorium-232 should be able to undergo double beta decay, which would produce uranium-232, but this has not yet been observed experimentally.[7]
All isotopes from 232U to 236U inclusive have minor cluster decay branches (less than 10−10%), and all these bar 233U, in addition to 238U, have minor spontaneous fission branches;[7] the greatest branching ratio for spontaneous fission is about 5×10−5% for 238U, or about one in every two million decays.[119] The shorter-lived trace isotopes 237U and 239U exclusively undergo beta decay, with respective half-lives of 6.752 days and 23.45 minutes.[7]
In total, 28 isotopes of uranium have been identified, ranging in mass number from 214[120] to 242, with the exception of 220.[7][121] Among the uranium isotopes not found in natural samples or nuclear fuel, the longest-lived is 230U, an alpha emitter with a half-life of 20.23 days.[7] This isotope has been considered for use in targeted alpha-particle therapy (TAT).[122] All other isotopes have half-lives shorter than one hour, except for 231U (half-life 4.2 days) and 240U (half-life 14.1 hours).[7] The shortest-lived known isotope is 221U, with a half-life of 660 nanoseconds, and it is expected that the hitherto unknown 220U has an even shorter half-life.[123] The proton-rich isotopes lighter than 232U primarily undergo alpha decay, except for 229U and 231U, which decay to protactinium isotopes via positron emission and electron capture, respectively; the neutron-rich 240U, 241U, and 242U undergo beta decay to form neptunium isotopes.[7][121]
Enrichment
[edit]
In nature, uranium is found as uranium-238 (99.2742%) and uranium-235 (0.7204%). Isotope separation concentrates (enriches) the fissile uranium-235 for nuclear weapons and most nuclear power plants, except for gas cooled reactors and pressurized heavy water reactors. Most neutrons released by a fissioning atom of uranium-235 must impact other uranium-235 atoms to sustain the nuclear chain reaction. The concentration and amount of uranium-235 needed to achieve this is called a 'critical mass'.
To be considered 'enriched', the uranium-235 fraction should be between 3% and 5%.[124] This process produces huge quantities of uranium that is depleted of uranium-235 and with a correspondingly increased fraction of uranium-238, called depleted uranium or 'DU'. To be considered 'depleted', the 235U concentration should be no more than 0.3%.[125] The price of uranium has risen since 2001, so enrichment tailings containing more than 0.35% uranium-235 are being considered for re-enrichment, driving the price of depleted uranium hexafluoride above $130 per kilogram in July 2007 from $5 in 2001.[125]
The gas centrifuge process, where gaseous uranium hexafluoride (UF
6) is separated by the difference in molecular weight between 235UF6 and 238UF6 using high-speed centrifuges, is the cheapest and leading enrichment process.[34] The gaseous diffusion process had been the leading method for enrichment and was used in the Manhattan Project. In this process, uranium hexafluoride is repeatedly diffused through a silver-zinc membrane, and the different isotopes of uranium are separated by diffusion rate (since uranium-238 is heavier it diffuses slightly slower than uranium-235).[34] The molecular laser isotope separation method employs a laser beam of precise energy to sever the bond between uranium-235 and fluorine. This leaves uranium-238 bonded to fluorine and allows uranium-235 metal to precipitate from the solution.[9] An alternative laser method of enrichment is known as atomic vapor laser isotope separation (AVLIS) and employs visible tunable lasers such as dye lasers.[126] Another method used is liquid thermal diffusion.[10]
The only significant deviation from the 235U to 238U ratio in any known natural samples occurs in Oklo, Gabon, where natural nuclear fission reactors consumed some of the 235U some two billion years ago when the ratio of 235U to 238U was more akin to that of low enriched uranium allowing regular ("light") water to act as a neutron moderator akin to the process in humanmade light water reactors. The existence of such natural fission reactors which had been theoretically predicted beforehand was proven as the slight deviation of 235U concentration from the expected values were discovered during uranium enrichment in France. Subsequent investigations to rule out any nefarious human action (such as stealing of 235U) confirmed the theory by finding isotope ratios of common fission products (or rather their stable daughter nuclides) in line with the values expected for fission but deviating from the values expected for non-fission derived samples of those elements.
Human exposure
[edit]A person can be exposed to uranium (or its radioactive daughters, such as radon) by inhaling dust in air or by ingesting contaminated water and food. The amount of uranium in air is usually very small; however, people who work in factories that process phosphate fertilizers containing uranium impurities, live near government facilities that made or tested nuclear weapons, live or work near a modern battlefield where depleted uranium weapons have been used, or live or work near a coal-fired power plant, facilities that mine or process uranium ore, or enrich uranium for reactor fuel, may have increased exposure to uranium.[127][128] Houses or structures that are over uranium deposits (either natural or man-made slag deposits) may have an increased incidence of exposure to radon gas.
The health impacts of natural and of deleted uranium are chemical rather than due to radiation.[128] The Occupational Safety and Health Administration (OSHA) has set the permissible exposure limit for uranium exposure in the workplace as 0.25 mg/m3 over an 8-hour workday. The National Institute for Occupational Safety and Health (NIOSH) has set a recommended exposure limit (REL) of 0.2 mg/m3 over an 8-hour workday and a short-term limit of 0.6 mg/m3. At 10 mg/m3, uranium is immediately dangerous to life and health.[129]
Most ingested uranium is excreted during digestion. Only 0.5% is absorbed when insoluble forms of uranium, such as its oxide, are ingested, whereas absorption of the more soluble uranyl ion can be up to 5%.[29] However, soluble uranium compounds tend to quickly pass through the body, whereas insoluble uranium compounds, especially when inhaled by way of dust into the lungs, pose a more serious exposure hazard. After entering the bloodstream, the absorbed uranium tends to bioaccumulate and stay for many years in bone tissue because of uranium's affinity for phosphates.[29] Incorporated uranium becomes uranyl ions, which accumulate in bone, liver, kidney, and reproductive tissues.[130]
Elements of high atomic number like uranium exhibit phantom or secondary radiotoxicity through absorption of natural background gamma and X-rays and re-emission of photoelectrons, which in combination with the high affinity of uranium to the phosphate moiety of DNA cause increased single and double strand DNA breaks.[131]
Uranium is not absorbed through the skin, and alpha particles released by uranium cannot penetrate the skin.[26]
Uranium can be decontaminated from steel surfaces[132] and aquifers.[133][134]
Effects and precautions
[edit]Normal functioning of the kidney, brain, liver, heart, and other systems can be affected by uranium exposure, because, besides being weakly radioactive, uranium is a toxic metal.[29][135][136] Uranium is also a reproductive toxicant.[137][138] Radiological effects are generally local because alpha radiation, the primary form of 238U decay, has a very short range, and will not penetrate skin. Alpha radiation from inhaled uranium has been demonstrated to cause lung cancer in exposed nuclear workers.[139] The Centers for Disease Control have published one study stating that neither natural nor depleted uranium have been classified with respect to carcinogenicity.[140] Exposure to its decay products, especially radon, is a significant health threat, and uranium processing produces wastes contaminated with radium which in turn produces radon gas.[141] Because of its long half-life, purified uranium will not produce significant amounts of daughter nuclides for millions of years. Exposure to strontium-90, iodine-131, and other fission products is unrelated to uranium exposure, but may result from medical procedures or exposure to spent reactor fuel or fallout from nuclear weapons.[142]
Although accidental inhalation exposure to a high concentration of uranium hexafluoride has resulted in human fatalities, those deaths were associated with the generation of highly toxic hydrofluoric acid and uranyl fluoride rather than with uranium itself.[143] Finely divided uranium metal presents a fire hazard because uranium is pyrophoric; small grains will ignite spontaneously in air at room temperature.[11]
Uranium metal is commonly handled with gloves as a sufficient precaution.[144] Uranium concentrate is handled and contained so as to ensure that people do not inhale or ingest it.[144]
See also
[edit]- K-65 residues
- List of countries by uranium production
- List of countries by uranium reserves
- List of uranium projects
- Lists of nuclear disasters and radioactive incidents
- Nuclear and radiation accidents and incidents
- Nuclear engineering
- Nuclear fuel cycle
- Nuclear physics
- Quintuple bond (earlier thought to be a phi bond), in the molecule U2
- Thorium fuel cycle
- World Uranium Hearing
Notes
[edit]- ^ The thermal expansion is anisotropic: the coefficients for each crystal axis (at 20 °C) are αa = 25.27×10−6/K, αb = 0.76×10−6/K, αc = 20.35×10−6/K, and αaverage = αvolume/3 = 15.46×10−6/K.
References
[edit]- ^ "Standard Atomic Weights: Uranium". CIAAW. 1999.
- ^ Prohaska, Thomas; Irrgeher, Johanna; Benefield, Jacqueline; Böhlke, John K.; Chesson, Lesley A.; Coplen, Tyler B.; Ding, Tiping; Dunn, Philip J. H.; Gröning, Manfred; Holden, Norman E.; Meijer, Harro A. J. (4 May 2022). "Standard atomic weights of the elements 2021 (IUPAC Technical Report)". Pure and Applied Chemistry. doi:10.1515/pac-2019-0603. ISSN 1365-3075.
- ^ a b Arblaster, John W. (2018). Selected Values of the Crystallographic Properties of Elements. Materials Park, Ohio: ASM International. ISBN 978-1-62708-155-9.
- ^ Th(-I) and U(-I) have been detected in the gas phase as octacarbonyl anions; see Chaoxian, Chi; Sudip, Pan; Jiaye, Jin; Luyan, Meng; Mingbiao, Luo; Lili, Zhao; Mingfei, Zhou; Gernot, Frenking (2019). "Octacarbonyl Ion Complexes of Actinides [An(CO)8]+/− (An=Th, U) and the Role of f Orbitals in Metal–Ligand Bonding". Chemistry (Weinheim an der Bergstrasse, Germany). 25 (50): 11772–11784. 25 (50): 11772–11784. doi:10.1002/chem.201902625. ISSN 0947-6539. PMC 6772027. PMID 31276242.
- ^ Morss, L.R.; Edelstein, N.M.; Fuger, J., eds. (2006). The Chemistry of the Actinide and Transactinide Elements (3rd ed.). Netherlands: Springer. ISBN 978-90-481-3146-4.
- ^ a b Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 28. doi:10.1016/C2009-0-30414-6. ISBN 978-0-08-037941-8.
- ^ a b c d e f g h i j k l m n o Kondev, F. G.; Wang, M.; Huang, W. J.; Naimi, S.; Audi, G. (2021). "The NUBASE2020 evaluation of nuclear properties" (PDF). Chinese Physics C. 45 (3) 030001. doi:10.1088/1674-1137/abddae.
- ^ "Uranium". Encyclopaedia Britannica. Retrieved 22 April 2017.
- ^ a b c d e Emsley 2001, p. 479.
- ^ a b c d e f "Uranium". The McGraw-Hill Science and Technology Encyclopedia (5th ed.). The McGraw-Hill Companies, Inc. 2005. ISBN 978-0-07-142957-3.
- ^ a b c d e f g h i j k l m n o p q Hammond, C. R. (2000). The Elements, in Handbook of Chemistry and Physics (PDF) (81st ed.). CRC press. ISBN 978-0-8493-0481-1.
- ^ "Uranium". Royal Society of Chemistry.
- ^ "Lead". Royal Society of Chemistry.
- ^ "Tungsten". Royal Society of Chemistry.
- ^ "Gold". Royal Society of Chemistry.
- ^ a b c d e f g "uranium". Columbia Electronic Encyclopedia (6th ed.). Columbia University Press. Archived from the original on 27 July 2011. Retrieved 27 September 2008.
- ^ a b c d e "uranium". Encyclopedia of Espionage, Intelligence, and Security. The Gale Group, Inc. Archived from the original on 27 July 2011. Retrieved 27 September 2008.
- ^ Rollett, A. D. (2008). Applications of Texture Analysis. John Wiley and Sons. p. 108. ISBN 978-0-470-40835-3.
- ^ a b c Grenthe, Ingmar; Drożdżyński, Janusz; Fujino, Takeo; Buck, Edgar C.; Albrecht-Schmitt, Thomas E.; Wolf, Stephen F. (2006). "Uranium". In Morss, Lester R.; Edelstein, Norman M.; Fuger, Jean (eds.). The Chemistry of the Actinide and Transactinide Elements (PDF). Vol. 5 (3rd ed.). Dordrecht, the Netherlands: Springer. pp. 52–160. doi:10.1007/1-4020-3598-5_5. ISBN 978-1-4020-3555-5. Archived from the original (PDF) on 7 March 2016.
- ^ "Development of DU Munitions". Depleted Uranium in the Gulf (II). Gulflink, official website of Force Health Protection & Readiness. 2000.
- ^ a b c d e Emsley 2001, p. 480.
- ^ "Nuclear Weapon Design". Federation of American Scientists. 1998. Archived from the original on 26 December 2008. Retrieved 19 February 2007.
- ^ "Statement regarding the Good Morning America broadcast," The Homer Laughlin China Co. Archived 1 April 2012 at the Wayback Machine, 16 March 2011, accessed 25 March 2012.
- ^ "Dial R for radioactive – 12 July 1997 – New Scientist". Newscientist.com. Retrieved 12 September 2008.
- ^ "Uranium Mining". Atomic Heritage Foundation. Retrieved 23 December 2020.
- ^ a b "EPA Facts about Uranium" (PDF). U.S. Environmental Protection Agency. Archived from the original (PDF) on 29 November 2014. Retrieved 20 September 2014.
- ^ "Uranium Containing Dentures (ca. 1960s, 1970s)". ORAU Museum of Radiation and Radioactivity. Oak Ridge Associated Universities. 1999. Retrieved 11 October 2021.
- ^ Emsley 2001, p. 482.
- ^ a b c d e f g h i j Emsley 2001, p. 477.
- ^ Klaproth, M. H. (1789). "Chemische Untersuchung des Uranits, einer neuentdeckten metallischen Substanz" [Chemical Investigation of Uranite, a Newly Discovered Metallic Substance]. Chemische Annalen für die Freunde der Naturlehre, Arzneygelahrtheit, Haushaltungskunst und Manufacturen (in German). 2: 387–403.
- ^ "Uranium". The American Heritage Dictionary of the English Language (4th ed.). Houghton Mifflin Company. Archived from the original on 27 July 2011. Retrieved 15 January 2007.
- ^ Miśkowiec, Paweł (1 April 2023). "Name game: the naming history of the chemical elements—part 1—from antiquity till the end of 18th century". Foundations of Chemistry. 25 (1): 29–51. doi:10.1007/s10698-022-09448-5. ISSN 1572-8463.
- ^ Péligot, E.-M. (1842). "Recherches Sur L'Uranium". Annales de chimie et de physique. 5 (5): 5–47.
- ^ a b c d Emsley 2001, p. 478.
- ^ "The Electric Journal". Westinghouse Club. 10 April 1920 – via Google Books.
- ^ Gillett, Horace Wadsworth; Mack, Edward Lawrence (10 April 1917). Preparation of ferro-uranium. Technical Paper 177 – U.S. Bureau of Mines. U.S. Govt. print. off. – via Google Books.
- ^ a b c d e f Seaborg 1968, p. 773.
- ^ Fermi, Enrico (12 December 1938). "Artificial radioactivity produced by neutron bombardment: Nobel Lecture" (PDF). Royal Swedish Academy of Sciences. Archived from the original (PDF) on 9 August 2018. Retrieved 14 June 2017.
- ^ De Gregorio, A. (2003). "A Historical Note About How the Property was Discovered that Hydrogenated Substances Increase the Radioactivity Induced by Neutrons". Nuovo Saggiatore. 19: 41–47. arXiv:physics/0309046. Bibcode:2003physics...9046D.
- ^ Nigro, M. (2004). "Hahn, Meitner e la teoria della fissione" (PDF). Archived from the original (PDF) on 25 March 2009. Retrieved 5 May 2009.
- ^ van der Krogt, Peter. "Elementymology & Elements Multidict". Retrieved 5 May 2009.
- ^ Meitner, L.; Frisch, O. (1939). "Disintegration of Uranium by Neutrons: a New Type of Nuclear Reaction". Nature. 143 (5218): 239–240. Bibcode:1969Natur.224..466M. doi:10.1038/224466a0. S2CID 4188874.
- ^ "Alfred O. C. Nier". www.aps.org. Archived from the original on 19 July 2018. Retrieved 4 December 2016.
- ^ Manfred Popp (21 September 2016). "Wissenschaftsgeschichte: Hitlers Atombombe – warum es sie nicht gab – Spektrum der Wissenschaft". Spektrum.de. Retrieved 25 February 2022.
- ^ "Chicago Pile One". large.stanford.edu. Retrieved 4 December 2016.
- ^ Walsh, John (19 June 1981). "A Manhattan Project Postscript" (PDF). Science. 212 (4501). AAAS: 1369–1371. Bibcode:1981Sci...212.1369W. doi:10.1126/science.212.4501.1369. PMID 17746246. Retrieved 23 March 2013.
- ^ Groves 1962, pp. 180–184.
- ^ Helmreich, J.E. Gathering Rare Ores: The Diplomacy of Uranium Acquisition, 1943–1954, Princeton UP, 1986: ch. 10 ISBN 0-7837-9349-9
- ^ "Reactors Designed by Argonne National Laboratory: Fast Reactor Technology". U.S. Department of Energy, Argonne National Laboratory. 2012. Retrieved 25 July 2012.
- ^ "History and Success of Argonne National Laboratory: Part 1". U.S. Department of Energy, Argonne National Laboratory. 1998. Archived from the original on 26 September 2006. Retrieved 28 January 2007.
- ^ "Reactors Designed by Argonne National Laboratory: Light Water Reactor Technology Development". U.S. Department of Energy, Argonne National Laboratory. 2012. Retrieved 25 July 2012.
- ^ "1956: Queen switches on nuclear power". BBC News. 17 October 1956. Retrieved 28 June 2006.
- ^ "STR (Submarine Thermal Reactor) in "Reactors Designed by Argonne National Laboratory: Light Water Reactor Technology Development"". U.S. Department of Energy, Argonne National Laboratory. 2012. Retrieved 25 July 2012.
- ^ a b "Oklo: Natural Nuclear Reactors". Office of Civilian Radioactive Waste Management. Archived from the original on 3 June 2004. Retrieved 28 June 2006.
- ^ Warneke, T.; Croudace, I. W.; Warwick, P. E. & Taylor, R. N. (2002). "A new ground-level fallout record of uranium and plutonium isotopes for northern temperate latitudes". Earth and Planetary Science Letters. 203 (3–4): 1047–1057. Bibcode:2002E&PSL.203.1047W. doi:10.1016/S0012-821X(02)00930-5.
- ^ "The Worst Nuclear Disasters". Time. 25 March 2009. Archived from the original on 28 March 2009. Retrieved 24 May 2010.
- ^ Gilliland, Frank D.; Hunt, William C.; Pardilla, Marla; Key, Charles R. (March 2000). "Uranium Mining and Lung Cancer Among Navajo Men in New Mexico and Arizona, 1969 to 1993". Journal of Occupational and Environmental Medicine. 42 (3): 278–283. doi:10.1097/00043764-200003000-00008. PMID 10738707.
- ^ Brugge, Doug; Goble, Rob (2002). "The History of Uranium Mining and the Navajo People". American Journal of Public Health. 92 (9). Ajph.org: 1410–1419. doi:10.2105/AJPH.92.9.1410. PMC 3222290. PMID 12197966.
- ^ Van Unnik, J. G.; Broerse, J. J.; Geleijns, J.; Jansen, J. T.; Zoetelief, J.; Zweers, D. (1997). "Survey of CT techniques and absorbed dose in various Dutch hospitals". The British Journal of Radiology. 70 (832): 367–371. doi:10.1259/bjr.70.832.9166072. PMID 9166072. (3000 examinations from 18 hospitals)
- ^ Russia's Nuclear Fuel Cycle. World Nuclear Association. Updated December 2021.
- ^ Emsley, John (2003). Nature's Building Blocks: An A-Z Guide to the Elements. Oxford University Press. ISBN 978-0-19-850340-8.
- ^ Biever, Celeste (27 July 2005). "First measurements of Earth's core radioactivity". New Scientist. Retrieved 7 July 2022.
- ^ "Potassium-40 heats up Earth's core". physicsworld.com. 7 May 2003. Retrieved 14 January 2007.
- ^ Schnug, E., Sun, Y., Zhang, L., Windmann, H., Lottermoser, B.G., Ulrich, A. E., Bol, R., Makeawa, M., and Haneklaus, S.H. (2023) "Elemental loads with phosphate fertilizers – a constraint for soil productivity?" In: Bolan, N.S. and Kirkham, M.B. (eds.) Managing Soil Constraints for Sustaining Productivity. CRC Press.
- ^ "History/Origin of Chemicals". NASA. Retrieved 1 January 2013.
- ^ Burbidge, E. M.; Burbidge, G. R.; Fowler, W. A.; Hoyle, F. (1957). "Synthesis of the Elements in Stars". Reviews of Modern Physics. 29 (4): 547. Bibcode:1957RvMP...29..547B. doi:10.1103/RevModPhys.29.547.
- ^ Clayton, Donald D. (1968). Principles of Stellar Evolution and Nucleosynthesis. New York: Mc-Graw-Hill. pp. 577–91. ISBN 978-0-226-10953-4.
- ^ a b Trenn, Thaddeus J. (1978). "Thoruranium (U-236) as the extinct natural parent of thorium: The premature falsification of an essentially correct theory". Annals of Science. 35 (6): 581–97. doi:10.1080/00033797800200441.
- ^ Tissot, François L. H.; Dauphas, Nicolas; Grossmann, Lawrence (4 March 2016). "Origin of uranium isotope variations in early solar nebula condensates". Science Advances. 2 (3) e1501400. arXiv:1603.01780. Bibcode:2016SciA....2E1400T. doi:10.1126/sciadv.1501400. PMC 4783122. PMID 26973874.
- ^ Min, M.; Xu, H.; Chen, J.; Fayek, M. (2005). "Evidence of uranium biomineralization in sandstone-hosted roll-front uranium deposits, northwestern China". Ore Geology Reviews. 26 (3–4): 198. Bibcode:2005OGRv...26..198M. doi:10.1016/j.oregeorev.2004.10.003.
- ^ Koribanics, N. M.; Tuorto, S. J.; Lopez-Chiaffarelli, N.; McGuinness, L. R.; Häggblom, M. M.; Williams, K. H.; Long, P. E.; Kerkhof, L. J. (2015). "Spatial Distribution of an Uranium-Respiring Betaproteobacterium at the Rifle, CO Field Research Site". PLOS ONE. 10 (4) e0123378. Bibcode:2015PLoSO..1023378K. doi:10.1371/journal.pone.0123378. PMC 4395306. PMID 25874721.
- ^ Renshaw, J. C.; Butchins, L. J. C.; Livens, F. R.; et al. (June 2005). "Bioreduction of uranium: environmental implications of a pentavalent intermediate". Environmental Science & Technology. 39 (15): 5657–5660. Bibcode:2005EnST...39.5657R. doi:10.1021/es048232b. PMID 16124300.
- ^ Vitesse, GF; Morris, K; Natrajan, LS; Shaw, S (January 2020). "Multiple Lines of Evidence Identify U(V) as a Key Intermediate during U(VI) Reduction by Shewanella oneidensis MR1". Environmental Science & Technology. 54 (4): 2268–2276. Bibcode:2020EnST...54.2268V. doi:10.1021/acs.est.9b05285. PMID 31934763.
- ^ Emsley 2001, pp. 476 and 482.
- ^ Macaskie, L. E.; Empson, R. M.; Cheetham, A. K.; Grey, C. P. & Skarnulis, A. J. (1992). "Uranium bioaccumulation by a Citrobacter sp. as a result of enzymically mediated growth of polycrystalline HUO
2PO
4". Science. 257 (5071): 782–784. Bibcode:1992Sci...257..782M. doi:10.1126/science.1496397. PMID 1496397. - ^ Anderson, R. T.; Vrionis, H. A.; Ortiz-Bernad, I.; Resch, C. T.; Long, P. E.; Dayvault, R.; Karp, K.; Marutzky, S.; Metzler, D. R.; Peacock, A.; White, D. C.; Lowe, M.; Lovley, D. R. (2003). "Stimulating the in situ activity of Geobacter species to remove uranium from the groundwater of a uranium-contaminated aquifer". Applied and Environmental Microbiology. 69 (10): 5884–5891. Bibcode:2003ApEnM..69.5884A. doi:10.1128/AEM.69.10.5884-5891.2003. PMC 201226. PMID 14532040.
- ^ Gadd, G. M. (March 2010). "Metals, minerals and microbes: geomicrobiology and bioremediation". Microbiology. 156 (Pt 3): 609–643. doi:10.1099/mic.0.037143-0. PMID 20019082.
- ^ Nilgiriwala, K.S.; Alahari, A.; Rao, A. S. & Apte, S.K. (2008). "Cloning and Overexpression of Alkaline Phosphatase PhoK from Sphingomonas sp. Strain BSAR-1 for Bioprecipitation of Uranium from Alkaline Solutions". Applied and Environmental Microbiology. 74 (17): 5516–5523. Bibcode:2008ApEnM..74.5516N. doi:10.1128/AEM.00107-08. PMC 2546639. PMID 18641147.
- ^ a b c d "World Uranium Mining". World Nuclear Association. Retrieved 31 January 2023.
- ^ Seaborg 1968, p. 774.
- ^ "Athabasca Basin, Saskatchewan". Retrieved 4 September 2009.
- ^ Gupta, C. K. & Mukherjee, T. K. (1990). Hydrometallurgy in extraction processes. Vol. 1. CRC Press. pp. 74–75. ISBN 978-0-8493-6804-2.
- ^ "Uranium production". Our World in Data. Retrieved 6 March 2020.
- ^ a b "Uranium Supplies: Supply of Uranium – World Nuclear Association". www.world-nuclear.org.
- ^ a b "Global Uranium Resources to Meet Projected Demand". International Atomic Energy Agency. 2006. Retrieved 29 March 2007.
- ^ "Uranium Mining and Processing in South Australia". South Australian Chamber of Mines and Energy. 2002. Archived from the original on 6 January 2012. Retrieved 14 January 2007.
- ^ Ngoupana, P.-M.; Felix, B. (2011). Barker, A. (ed.). "Areva suspends CAR uranium mine project". Central African Republic News. Retrieved 7 March 2020.
- ^ "Military Warheads as a Source of Nuclear Fuel". World-nuclear.org. Retrieved 24 May 2010.
- ^ "Megatons to Megawatts". U.S. Enrichment Corp. Archived from the original on 16 July 2008.
- ^ "Uranium recovery from Seawater". Japan Atomic Energy Research Institute. 23 August 1999. Archived from the original on 17 October 2009. Retrieved 3 September 2008.
- ^ "How long will nuclear energy last?". 12 February 1996. Archived from the original on 10 April 2007. Retrieved 29 March 2007.
- ^ Tsezos, M.; Noh, S. H. (1984). "Extraction of uranium from sea water using biological origin adsorbents". The Canadian Journal of Chemical Engineering. 62 (4): 559–561. Bibcode:1984CJChE..62..559T. doi:10.1002/cjce.5450620416.
- ^ "ORNL technology moves scientists closer to extracting uranium from seawater". Oak Ridge National Laboratory, United States. 21 August 2012. Archived from the original on 25 August 2012. Retrieved 22 February 2013.
- ^ "Fueling nuclear power with seawater". Pnnl.gov. 21 August 2012. Archived from the original on 25 August 2012. Retrieved 22 February 2013.
- ^ "NUEXCO Exchange Value (Monthly Uranium Spot)". Archived from the original on 12 December 2007.
- ^ "World Uranium Production". UxC Consulting Company, LLC. Archived from the original on 27 February 2007. Retrieved 11 February 2007.
- ^ Mithridates (24 July 2008). "Page F30: Kazakhstan to surpass Canada as the world's largest producer of uranium by last year (2009)". Mithridates.blogspot.com. Archived from the original on 4 March 2010. Retrieved 12 September 2008.
- ^ "Kazakistan uranyum üretimini artıracak". Zaman.com.tr (in Turkish). Zaman Gazetesi. 28 July 2008. Archived from the original on 13 January 2009. Retrieved 12 September 2008.
- ^ "In Situ Leach Mining (ISL) of Uranium – World Nuclear Association". www.world-nuclear.org. Retrieved 6 May 2021.
- ^ "Big Uranium Find Announced in Somalia". The New York Times. 16 March 1968. Retrieved 16 May 2014.
- ^ "Lack of fuel may limit U.S. nuclear power expansion". Massachusetts Institute of Technology. 21 March 2007. Retrieved 29 March 2007.
- ^ Deffeyes, Kenneth S. & MacGregor, Ian D. (January 1980). "World Uranium Resources". Scientific American. 242 (1): 66. Bibcode:1980SciAm.242a..66D. doi:10.1038/scientificamerican0180-66. OSTI 6665051.
- ^ Kloprogge, J. Theo; Ponce, Concepcion P.; Loomis, Tom A. (2021). The periodic table : nature's building blocks: an introduction to the naturally occurring elements, their origins and their uses. Amsterdam: Elsevier. pp. 861–862. ISBN 978-0-12-821538-8. OCLC 1223058470.
- ^ Seaborg 1968, p. 779.
- ^ a b c "Chemical Forms of Uranium". Argonne National Laboratory. Archived from the original on 22 September 2006. Retrieved 18 February 2007.
- ^ a b Seaborg 1968, p. 778.
- ^ a b c d Puigdomenech, Ignasi (2004) Hydra/Medusa Chemical Equilibrium Database and Plotting Software. KTH Royal Institute of Technology
- ^ a b c d e f g h i j Seaborg 1968, p. 782.
- ^ a b Seaborg 1968, p. 780.
- ^ Beiser, A. (2003). "Nuclear Transformations" (PDF). Concepts of Modern Physics (6th ed.). McGraw-Hill Education. pp. 432–434. ISBN 978-0-07-244848-1. Archived from the original (PDF) on 4 October 2016. Retrieved 4 July 2016.
- ^ Roederer, I. U.; Kratz, K.; Frebel, A.; Christlieb, N.; Pfeiffer, B.; Cowan, J. J.; Sneden, C. (2009). "The end of nucleosynthesis: Production of lead and thorium in the early galaxy". The Astrophysical Journal. 698 (2): 1963–1980. arXiv:0904.3105. Bibcode:2009ApJ...698.1963R. doi:10.1088/0004-637X/698/2/1963. hdl:2152/35050. S2CID 14814446.
- ^ a b c The occurrence of plutonium-244 as a primordial nuclide is disputed, though some reports of its detection have also been attributed to infall from the interstellar medium.[113][114]
- ^ Lachner, J.; et al. (2012). "Attempt to detect primordial 244Pu on Earth". Physical Review C. 85 (1) 015801. Bibcode:2012PhRvC..85a5801L. doi:10.1103/PhysRevC.85.015801.
- ^ Wallner, A.; Faestermann, T.; Feige, J.; Feldstein, C.; Knie, K.; Korschinek, G.; Kutschera, W.; Ofan, A.; Paul, M.; Quinto, F.; Rugel, G.; Steier, P. (2015). "Abundance of live 244Pu in deep-sea reservoirs on Earth points to rarity of actinide nucleosynthesis". Nature Communications. 6 5956. arXiv:1509.08054. Bibcode:2015NatCo...6.5956W. doi:10.1038/ncomms6956. ISSN 2041-1723. PMC 4309418. PMID 25601158.
- ^ Tretyak, V.I.; Zdesenko, Yu.G. (2002). "Tables of Double Beta Decay Data — An Update". At. Data Nucl. Data Tables. 80 (1): 83–116. Bibcode:2002ADNDT..80...83T. doi:10.1006/adnd.2001.0873.
- ^ Marouli, M.; Pommé, S.; Jobbágy, V.; Van Ammel, R.; Paepen, J.; Stroh, H.; Benedik, L. (2014). "Alpha-particle emission probabilities of 236U obtained by alpha spectrometry". Applied Radiation and Isotopes. 87: 292–296. doi:10.1016/j.apradiso.2013.11.020. ISSN 0969-8043. PMID 24309010.
- ^ a b Near Term and Promising Long Term Options for the Deployment of Thorium Based Nuclear Energy (PDF) (Report). Vienna: International Atomic Energy Agency. 2022.
- ^ a b Forsburg, C. W.; Lewis, L. C. (24 September 1999). "Uses For Uranium-233: What Should Be Kept for Future Needs?" (PDF). Ornl-6952. Oak Ridge National Laboratory.
- ^ Goffer, Zvi (2006). Archaeological Chemistry (2nd ed.). Wiley. p. 106. ISBN 978-0-471-91515-7.
- ^ Zhang, Z. Y.; et al. (2021). "New α -Emitting Isotope 214 U and Abnormal Enhancement of α -Particle Clustering in Lightest Uranium Isotopes". Physical Review Letters. 126 (15) 152502. arXiv:2101.06023. Bibcode:2021PhRvL.126o2502Z. doi:10.1103/PhysRevLett.126.152502. PMID 33929212. S2CID 231627674. Retrieved 15 May 2021.
- ^ a b Niwase, T.; Watanabe, Y. X.; Hirayama, Y.; et al. (2023). "Discovery of New Isotope 241U and Systematic High-Precision Atomic Mass Measurements of Neutron-Rich Pa-Pu Nuclei Produced via Multinucleon Transfer Reactions" (PDF). Physical Review Letters. 130 (13): 132502-1 – 132502-6. Bibcode:2023PhRvL.130m2502N. doi:10.1103/PhysRevLett.130.132502. PMID 37067317. S2CID 257976576.
- ^ Mastren, T.; Stein, B. W.; Parker, T. G.; Radchenko, V.; Copping, R.; Owens, A.; Wyant, L. E.; Brugh, M.; Kozimor, S. A.; Noriter, F. M.; Birnbaum, E. R.; John, K. D.; Fassbender, M. E. (2018). "Separation of protactinium employing sulfur-based extraction chromatographic resins". Analytical Chemistry. 90 (11): 7012–7017. doi:10.1021/acs.analchem.8b01380. ISSN 0003-2700. OSTI 1440455. PMID 29757620.
- ^ Khuyagbaatar, J.; et al. (11 December 2015). "New Short-Lived Isotope 221U and the Mass Surface Near N = 126". Physical Review Letters. 115 (24) 242502. Bibcode:2015PhRvL.115x2502K. doi:10.1103/PhysRevLett.115.242502. PMID 26705628. S2CID 12184696.
- ^ "Uranium Enrichment". Argonne National Laboratory. Archived from the original on 24 January 2007. Retrieved 11 February 2007.
- ^ a b Diehl, Peter. "Depleted Uranium: a by-product of the Nuclear Chain". Laka Foundation. Archived from the original on 13 January 2013. Retrieved 31 July 2009.
- ^ Duarte, F. J.; Hillman, L. W., eds. (1990). Dye Laser Principles. Academic. p. 413. ISBN 978-0-12-222700-4. Archived from the original on 17 September 2010.
- ^ "Radionuclide Basics: Uranium". U.S. Environmental Protection Agency. 16 February 2023. Retrieved 19 April 2023.
- ^ a b "ToxFAQ for Uranium". Agency for Toxic Substances and Disease Registry. 18 March 2014. Retrieved 19 April 2023.
- ^ "CDC – NIOSH Pocket Guide to Chemical Hazards – Uranium (insoluble compounds, as U)". National Institute for Occupational Safety and Health. 30 October 2019. Retrieved 19 April 2023.
- ^ Permyakov, Eugene (2009). Metalloproteomics. Hoboken: John Wiley & Sons. p. 564. ISBN 978-0-470-44774-1. OCLC 609844907.
- ^ Busby, C. and Schnug, E. (2008). "Advanced biochemical and biophysical aspects of uranium contamination". In: De Kok, L.J. and Schnug, E. (Eds) Loads and Fate of Fertilizer Derived Uranium. Backhuys Publishers, Leiden, The Netherlands. ISBN 978-90-5782-193-6
- ^ Francis, A. J.; Dodge, C. J.; McDonald, J. A.; Halada, G. P. (2005). "Decontamination of uranium-contaminated steel surfaces by hydroxycarboxylic acid with uranium recovery". Environmental Science & Technology. 39 (13): 5015–21. Bibcode:2005EnST...39.5015F. doi:10.1021/es048887c. PMID 16053105.
- ^ Gandhi, T. Pushparaj; Sampath, Prasanna Venkatesh; Maliyekkal, Shihabudheen M. (15 June 2022). "A critical review of uranium contamination in groundwater: Treatment and sludge disposal". The Science of the Total Environment. 825 153947. Bibcode:2022ScTEn.82553947G. doi:10.1016/j.scitotenv.2022.153947. ISSN 1879-1026. PMID 35189244. S2CID 246988421.
- ^ Prusty, Sourav; Somu, Prathap; Sahoo, Jitendra Kumar; Panda, Debasish; Sahoo, Sunil Kumar; Sahoo, Shraban Kumar; Lee, Yong Rok; Jarin, T.; Sundar, L. Syam; Rao, Koppula Srinivas (December 2022). "Adsorptive sequestration of noxious uranium (VI) from water resources: A comprehensive review". Chemosphere. 308 (Pt 1) 136278. Bibcode:2022Chmsp.30836278P. doi:10.1016/j.chemosphere.2022.136278. ISSN 1879-1298. PMID 36057349. S2CID 251999162.
- ^ Craft, E. S.; Abu-Qare, A. W.; Flaherty, M. M.; Garofolo, M. C.; Rincavage, H. L. & Abou-Donia, M. B. (2004). "Depleted and natural uranium: chemistry and toxicological effects" (PDF). Journal of Toxicology and Environmental Health Part B: Critical Reviews. 7 (4): 297–317. Bibcode:2004JTEHB...7..297C. CiteSeerX 10.1.1.535.5247. doi:10.1080/10937400490452714. PMID 15205046. S2CID 9357795.
- ^ Agency for Toxic Substances and Disease Registry (ATSDR) (February 2013). "2. Relevance to Public Health" (PDF). Toxicological Profile for Uranium (Report). Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service. pp. 11–38. CAS# 7440-61-1.
- ^ Hindin, Rita; Brugge, D.; Panikkar, B. (2005). "Teratogenicity of depleted uranium aerosols: A review from an epidemiological perspective". Environ Health. 4 (1): 17. Bibcode:2005EnvHe...4...17H. doi:10.1186/1476-069X-4-17. PMC 1242351. PMID 16124873.
- ^ Arfsten, D. P.; Still, K. R.; Ritchie, G. D. (2001). "A review of the effects of uranium and depleted uranium exposure on reproduction and fetal development". Toxicology and Industrial Health. 17 (5–10): 180–91. Bibcode:2001ToxIH..17..180A. doi:10.1191/0748233701th111oa. PMID 12539863. S2CID 25310165.
- ^ Grellier, James; Atkinson, Will; Bérard, Philippe; Bingham, Derek; Birchall, Alan; Blanchardon, Eric; Bull, Richard; Guseva Canu, Irina; Challeton-de Vathaire, Cécile; Cockerill, Rupert; Do, Minh T; Engels, Hilde; Figuerola, Jordi; Foster, Adrian; Holmstock, Luc; Hurtgen, Christian; Laurier, Dominique; Puncher, Matthew; Riddell, Tony; Samson, Eric; Thierry-Chef, Isabelle; Tirmarche, Margot; Vrijheid, Martine; Cardis, Elisabeth (2017). "Risk of lung cancer mortality in nuclear workers from internal exposure to alpha particle-emitting radionuclides". Epidemiology. 28 (5): 675–684. doi:10.1097/EDE.0000000000000684. PMC 5540354. PMID 28520643.
- ^ "Public Health Statement for Uranium" (PDF). CDC. Retrieved 5 May 2023.
- ^ Radon Exposures to Workers at the Fernald Feed Materials Production Center. Page reviewed: April 8, 2020. U.S. National Institute for Occupational Safety and Health (NIOSH)
- ^ Chart of the Nuclides, US Atomic Energy Commission 1968
- ^ Dart, Richard C. (2004). Medical Toxicology. Lippincott Williams & Wilkins. p. 1468. ISBN 978-0-7817-2845-4.
- ^ a b "Radiation Fact Sheets #27, Uranium (U)". Washington State Department of Health, Office of Radiation Protection. 2010. Archived from the original on 28 September 2011. Retrieved 23 August 2011.
Sources cited
[edit]- Emsley, John (2001). "Uranium". Nature's Building Blocks: An A to Z Guide to the Elements. Oxford: Oxford University Press. pp. 476–482. ISBN 978-0-19-850340-8.
- Seaborg, Glenn T. (1968). "Uranium". The Encyclopedia of the Chemical Elements. Skokie, Illinois: Reinhold Book Corporation. pp. 773–786. LCCN 68029938.
- Groves, Leslie R. (1962). Now It Can Be Told: The Story of the Manhattan Project. New York: Harper and Co. ISBN 0-306-80189-2.
{{cite book}}: ISBN / Date incompatibility (help)
External links
[edit]- Nuclear fuel data and analysis from the U.S. Energy Information Administration
- World Uranium deposit maps
- Dittmar, William (1888). . Encyclopædia Britannica. Vol. XXIV (9th ed.). p. 7.
- Annotated bibliography for uranium from the Alsos Digital Library
- NLM Hazardous Substances Databank – Uranium, Radioactive
- CDC – NIOSH Pocket Guide to Chemical Hazards
- ATSDR Case Studies in Environmental Medicine: Uranium Toxicity Archived 4 February 2016 at the Wayback Machine U.S. Department of Health and Human Services
- Uranium at The Periodic Table of Videos (University of Nottingham)
Uranium
View on GrokipediaPhysical and Chemical Properties
Atomic Structure and Physical Characteristics
Uranium (U) possesses atomic number 92, positioning it as the third actinide in the periodic table after thorium and protactinium.[10] Its ground-state electron configuration is [Rn] 5f³ 6d¹ 7s², featuring valence electrons in f, d, and s orbitals that contribute to its chemical reactivity and multiple oxidation states ranging from +3 to +6.[11] This configuration arises from the filling of the 5f subshell, characteristic of actinides, leading to complex electronic interactions and relativistic effects that contract the orbitals and influence bonding, with an electronegativity of 1.38 (Pauling scale), atomic radius of 156 pm (empirical, corresponding to a diameter of approximately 312 pm or 3.12 Å; values vary by definition such as empirical, covalent, or van der Waals, with other sources giving an empirical radius of 175 pm and diameter of ~350 pm), first ionization energy of 6.19 eV, and typical ionic radii of 89 pm for U⁴⁺ and 73 pm for U⁶⁺ (effective, coordination number 6).[10][12][1] As a dense, silvery-white radioactive metal, uranium exhibits a standard density of 19.05 g/cm³ at 20°C for its alpha phase, making it one of the heaviest naturally occurring elements.[13] It melts at 1135°C and boils at 4131°C under standard pressure, reflecting strong metallic bonding tempered by its large atomic mass.[12] Uranium is malleable and ductile at room temperature, capable of being rolled into wires or sheets, though it tarnishes in air due to oxide formation.[14] Its thermal conductivity measures 27.6 W/(m·K), and electrical resistivity is approximately 2.8 × 10⁻⁷ Ω·m at room temperature, indicating moderate conductivity for a transition metal influenced by f-electron scattering.[14][13] Uranium displays allotropy with three distinct phases dependent on temperature: the low-temperature α-phase (orthorhombic, stable below 668°C), the intermediate β-phase (body-centered tetragonal, 668–776°C), and the high-temperature γ-phase (body-centered cubic, above 776°C).[15] The α-phase, prevalent at ambient conditions, adopts an orthorhombic crystal structure in space group Cmcm with lattice parameters a ≈ 285.4 pm, b ≈ 587.0 pm, and c ≈ 495.5 pm, consisting of layered uranium atoms that contribute to anisotropic thermal expansion and mechanical properties.[16] These phase transitions involve volume changes of up to 1.5% at the α-β boundary, impacting applications requiring dimensional stability, such as nuclear fuel fabrication.[15]| Phase | Crystal System | Stability Range (°C) | Key Feature |
|---|---|---|---|
| α | Orthorhombic (Cmcm) | < 668 | Room-temperature form, dense packing |
| β | Body-centered tetragonal | 668–776 | Intermediate density |
| γ | Body-centered cubic | > 776 | High symmetry, lowest density |
Chemical Reactivity and Compounds Overview
Uranium metal, a dense actinide, exhibits significant chemical reactivity, tarnishing rapidly in air to form a protective layer of uranium dioxide (UO₂) that limits further oxidation under ambient conditions.[17] Finely divided uranium reacts with cold water to produce hydrogen gas and uranium oxides or hydrides, while bulk metal corrodes more slowly but reacts vigorously with steam or hot water, generating flammable hydrogen via the reaction U + 2H₂O → UO₂ + 2H₂.[17][18] Clean uranium turnings or chips oxidize readily in stagnant air and can ignite spontaneously if confined without ventilation.[19] Uranium dissolves in acids, such as hydrochloric (U + 2HCl → UCl₄ + H₂) or nitric acid, often yielding uranyl ions (UO₂²⁺) and hydrogen gas, with reactivity enhanced by the metal's ability to reduce protons.[7][18] Uranium reacts with nearly all nonmetallic elements and their compounds, including halogens, oxygen, nitrogen, and carbon, with reaction rates increasing markedly with temperature; for instance, it burns in air to form U₃O₈ and ignites pyrophorically when powdered.[7][1] It remains largely inert to alkalis and hydroxide ions under standard conditions, though the uranyl ion can precipitate as hydroxides in basic media.[18] In aqueous environments, corrosion kinetics depend on factors like pH, temperature, and surface oxide layers, producing hydrogen and uranium(IV) or (VI) species, as evidenced by studies showing parabolic growth of oxide films initially followed by linear kinetics in humid conditions.[20] Uranium displays oxidation states ranging from +3 to +6, with +4 and +6 predominating in stable compounds; the +6 state, as the yellow uranyl ion (UO₂²⁺), is most persistent in aqueous solutions due to its thermodynamic stability.[7][1] Over 3,000 uranium compounds are documented, primarily oxides, halides, and organometallics, many featuring coordination with oxygen or fluorine ligands.[21] Key oxides include uranium dioxide (UO₂, black, used in nuclear fuel), uranium trioxide (UO₃, orange, acidic), and triuranium octoxide (U₃O₈, olive-green, the principal form in ores and oxidation products).[22] Halides such as uranium tetrafluoride (UF₄, green, intermediate in processing) and uranium hexafluoride (UF₆, volatile white solid critical for isotope enrichment) highlight uranium's utility in gaseous and solid-state chemistry.[22] Other notable classes encompass carbides (e.g., UC for reactor applications), nitrides, and sulfides, often synthesized under controlled atmospheres to mitigate reactivity.[21]Isotopes
Natural Abundance and Stability
Natural uranium comprises three primary isotopes: uranium-238 (U-238), which constitutes approximately 99.274% by mass; uranium-235 (U-235), at about 0.72% by mass; and uranium-234 (U-234), at roughly 0.0055% by mass.[23] These proportions reflect atomic abundances in terrestrial uranium deposits, with minor variations arising from geological processes and decay chain dynamics.[24] U-238 and U-235 are primordial isotopes, surviving from the solar system's formation due to their extended half-lives, while U-234 is primarily generated through the alpha decay of thorium-234, a daughter of U-238, maintaining its low but steady abundance in secular equilibrium within the uranium-238 decay series.[25] All three isotopes are radioactive and unstable, undergoing primarily alpha decay to form shorter-lived progeny, though their long half-lives confer relative stability on geological timescales. U-238 has a half-life of 4.468 billion years, decaying to thorium-234; U-235 has a half-life of 703.8 million years, decaying to thorium-231; and U-234 has a half-life of 244,500 years, decaying to thorium-230.[26] These decay modes release alpha particles (helium nuclei) with energies of approximately 4.2 MeV for U-238, 4.4 MeV for U-235, and 4.8 MeV for U-234, alongside minor gamma emissions from daughter products.[23] The extended half-lives of U-238 and U-235—comparable to or exceeding Earth's age of 4.54 billion years—ensure their persistence in the Earth's crust, whereas U-234's shorter half-life results in its abundance being dynamically balanced by ongoing production rather than primordial retention.[27] This isotopic composition influences uranium's radiological properties, with natural uranium exhibiting specific activities dominated by U-234 and its daughters due to their higher decay rates per unit mass, despite U-238's overwhelming mass fraction.[28] Secular equilibrium in ore bodies sustains chain reactions, but isolation or chemical separation can disrupt these balances, altering effective stability profiles over time.[29] Experimental measurements confirm these abundances and half-lives with high precision, derived from mass spectrometry and radiometric dating techniques applied to uranium standards.[24]Fissile Isotopes and Enrichment
Uranium-235 (U-235) is the only naturally occurring fissile isotope of uranium, meaning it can undergo fission induced by low-energy thermal neutrons and sustain a self-propagating nuclear chain reaction.[8][2] In contrast, the dominant isotope uranium-238 (U-238) is fertile but not fissile with thermal neutrons, requiring fast neutrons or conversion to fissile plutonium-239 for energy release.[8] Natural uranium ore contains approximately 0.7% U-235 by weight, with U-238 comprising 99.3% and trace amounts of uranium-234 (U-234).[30][2] This low fissile content necessitates isotopic enrichment to concentrate U-235 for practical applications in nuclear reactors and weapons, as unenriched natural uranium cannot sustain efficient chain reactions in most designs without moderators like heavy water.[31] Enrichment exploits the slight mass difference between U-235 and U-238 atoms (three neutrons heavier in U-238) through methods that separate uranium hexafluoride (UF6) gas isotopes.[30] The first large-scale process, gaseous diffusion, was developed during the Manhattan Project and became operational at the K-25 plant in Oak Ridge, Tennessee, in 1945, forcing UF6 vapor through porous barriers to preferentially permeate lighter U-235.[32][33] This energy-intensive method dominated mid-20th-century production but was largely supplanted by gas centrifugation starting in the 1970s, which spins UF6 at high speeds to separate isotopes by centrifugal force, achieving separative work units with far lower electricity use—about 50 times less than diffusion.[30][34] Emerging techniques like laser isotope separation, which selectively excites U-235 using tuned lasers, remain developmental but promise even greater efficiency.[30] Experimental biological methods utilize metal-reducing microorganisms, such as Shewanella oneidensis or Geobacter species and their enzymes, to preferentially reduce U-235 via kinetic isotope effects under anaerobic conditions, forming precipitates enriched in the lighter isotope; genetically engineered strains overexpressing reduction enzymes like cytochrome c enhance selectivity, though these approaches remain developmental as described in patents including US7452703B1.[35] Low-enriched uranium (LEU), defined as less than 20% U-235, powers light-water reactors at typical levels of 3-5% U-235 to balance neutron economy and criticality.[30] High-assay LEU (HALEU), enriched to 5-20% U-235, enables compact advanced reactor designs requiring higher burnup.[36] Highly enriched uranium (HEU), above 20% U-235 and often exceeding 90% for weapons-grade material, supports naval propulsion and nuclear explosives but poses proliferation risks due to its direct usability in bombs.[37] Enrichment to HEU levels demands thousands of separative work units per kilogram, underscoring the technological barriers and safeguards needed to prevent diversion.[30]Artificial Isotopes and Production
Artificial isotopes of uranium are those nuclides not occurring significantly in nature and synthesized primarily through neutron capture reactions in nuclear reactors or, less commonly, particle accelerators. The most notable include uranium-233 and uranium-236, which arise as products or byproducts in nuclear fuel cycles, while others such as uranium-232 and uranium-237 are produced in trace amounts during specific irradiation processes. These isotopes exhibit varying half-lives and nuclear properties, influencing their roles in reactor operations, waste management, and potential fuel applications. Production typically involves bombarding precursor materials with neutrons, leveraging the high neutron fluxes available in fission reactors. Uranium-233, with a half-life of 159,200 years, is generated via the thorium-uranium fuel cycle by irradiating thorium-232 targets in nuclear reactors. The process begins with thorium-232 capturing a thermal neutron to form thorium-233, which undergoes beta decay to protactinium-233 (half-life 27 days), followed by another beta decay to uranium-233.[38] This method has been demonstrated in light water reactors using separated thorium targets to yield high-purity uranium-233, as patented in designs separating irradiated thorium from fissile fuel to minimize contamination.[39] Production occurs in breeder reactors or experimental setups, where thorium's abundance—three times that of uranium—supports potential scalability, though challenges include protactinium-233 extraction to prevent neutron absorption losses.[40] Uranium-236, possessing a half-life of 23.42 million years, forms predominantly in uranium-fueled reactors when uranium-235 captures a neutron without fissioning, yielding uranium-236 directly.[41] This isotope accumulates in spent nuclear fuel, acting as a neutron poison that reduces reactor efficiency by competing for neutrons needed for fission.[42] Trace natural occurrences exist from cosmic ray interactions or ancient reactor sites, but anthropogenic production dominates, with concentrations up to parts per million in irradiated fuel depending on burnup levels.[43] Other synthetic uranium isotopes, such as uranium-232 (half-life 68.9 years), emerge as contaminants in thorium-based cycles through side reactions like neutron capture on thorium-230 or pa-231 decay chains, complicating handling due to intense gamma emissions from decay daughters.[38] Uranium-237, with a short half-life of 6.75 days, results from neutron capture on uranium-236 or other pathways in high-flux environments. These lesser isotopes are typically produced in milligram quantities for research via cyclotron acceleration or reactor irradiation, lacking the industrial scale of uranium-233 or -236.[44] Overall yields depend on neutron spectrum, flux (often 10^14 neutrons/cm²/s in reactors), and irradiation duration, with purification via chemical reprocessing to isolate targets from fission products.[45]Natural Occurrence
Stellar and Geological Formation
Uranium, one of the heaviest naturally occurring elements with atomic number 92, forms primarily through the rapid neutron-capture process (r-process) during explosive astrophysical events. This nucleosynthesis pathway involves the bombardment of lighter "seed" nuclei, such as iron-group elements, with a flux of neutrons at rates exceeding one per millisecond, producing highly neutron-rich isotopes that subsequently undergo beta decay to form stable heavy nuclei up to uranium and beyond.[46] The r-process accounts for approximately half of the heavy elements beyond iron in the universe, with uranium yields depending on the neutron density and duration of the event.[47] Core-collapse supernovae of massive stars (greater than 8 solar masses) and mergers of compact objects like neutron stars provide the requisite extreme conditions, including high temperatures above 10^9 K and neutron fluxes from dissociated material.[48] Observations of kilonova events, such as GW170817 detected in 2017, have confirmed r-process signatures in neutron star mergers through spectral lines of heavy elements and gravitational wave data correlating with electromagnetic counterparts rich in neutron-capture products.[48] These events eject synthesized uranium into the interstellar medium, where it mixes with gas and dust to form subsequent generations of stars and planetary systems. In the Solar System, uranium originated from multiple r-process events in the interstellar medium prior to the collapse of the molecular cloud that birthed the Sun around 4.6 billion years ago.[49] Earth's primordial uranium inventory derives from this accreted material, with the planet's bulk composition reflecting chondritic abundances adjusted for volatile loss during formation. During Hadean and Archean differentiation, uranium's geochemical incompatibility—due to its large ionic radius and high charge—prevented incorporation into mantle minerals like olivine and pyroxene, leading to enrichment in the felsic continental crust at concentrations up to 2-5 ppm in granitic rocks, compared to 0.01-0.02 ppm in the bulk silicate Earth.[50] Radiogenic heat from uranium decay (primarily U-238, half-life 4.468 billion years) has contributed significantly to Earth's thermal budget, with models estimating it accounts for about 50% of present-day radiogenic power alongside thorium and potassium.[49] Uranium ore deposits form through subsequent geological mobilization and concentration, spanning igneous, hydrothermal, and sedimentary processes across Earth's history. Primary magmatic deposits occur in peralkaline granites and pegmatites via late-stage crystal fractionation, as seen in Archean complexes like those in Greenland, where uranium minerals such as uraninite crystallize directly from melt.[51] Hydrothermal systems, driven by convective fluids in volcanic or metamorphic settings, transport uranium as uranyl complexes (UO2^2+) and precipitate it in veins or breccias, often associated with molybdenum or fluorite, as in the 2.5 billion-year-old deposits of the Great Bear Magmatic Zone in Canada.[50] Sedimentary deposits, dominant for economic ores, result from supergene leaching and redistribution under oxidizing conditions, forming roll-front accumulations in permeable sandstones (e.g., Wyoming basins, sourced from weathered Precambrian granites) or tabular strata-bound ores in reduced paleochannels, with the oldest known examples in 2.7 billion-year-old Witwatersrand conglomerates via placer mechanisms under anoxic atmospheres.[52] Unconformity-related deposits, like those at Athabasca Basin (Canada), involve basinal brines interacting with graphitic faults at depths of 1-2 km, precipitating pitchblende (UO2) through redox fronts, with formation ages clustered around 1.8-1.3 billion years ago.[50] These processes reflect uranium's solubility in oxidized, acidic fluids (as U^6+) and reduction to insoluble U^4+ phases, enabling cyclic enrichment over billions of years without significant biotic mediation in primary stages.[51]Terrestrial Distribution and Biotic Accumulation
Uranium occurs naturally throughout the Earth's crust at an average concentration of 2.7 parts per million (ppm) by mass, comparable to elements like tin and molybdenum.[53] This abundance places it among the more common heavy metals, though economic deposits require concentrations exceeding 100-1,000 ppm in host rocks.[52] Concentrations vary by rock type: granitic rocks typically hold 3-5 ppm, sedimentary rocks 2-3 ppm, and basalts around 1 ppm, with higher levels in phosphate-rich sediments and black shales due to adsorptive affinity for organic matter and phosphates.[54][55] In soils, uranium levels average about 3 ppm globally, influenced by parent rock weathering and leaching, though values range from 0.5-20 ppm depending on local geology and anthropogenic inputs.[56] Surface and groundwater contain uranium at trace levels, typically 0.1-10 micrograms per liter (μg/L), derived from rock dissolution and atmospheric deposition, with elevated concentrations in arid regions or near mineralized zones exceeding 30 μg/L.[57] Uranium ore deposits, which represent localized enrichments, form in diverse terrestrial settings including sandstone-hosted (e.g., roll-front deposits in permeable aquifers), unconformity-related (basement-sediment interfaces), and quartz-pebble conglomerates (paleoplacers), often associated with reducing conditions that precipitate uraninite (UO₂) or coffinite.[52] Biotic accumulation begins with plant uptake from soil pore water, where uranium exists primarily as uranyl ions (UO₂²⁺) complexed with carbonates or phosphates; transfer factors (plant/soil concentration ratios) range from 0.1-10 for most species, higher in hyperaccumulators like sunflowers or ferns under acidic conditions (pH <6).[58][59] In terrestrial food chains, herbivores ingest uranium via contaminated forage, achieving bioconcentration factors of 0.01-0.1 in muscle tissue but higher in bones (up to 1-10) due to chemical similarity to calcium; carnivores and humans further bioaccumulate through diet, with human kidney burdens averaging 0.1-1 μg/g wet weight from chronic low-level exposure.[60][61] Soil-to-plant-to-animal transfer is limited by uranium's low bioavailability in neutral-alkaline soils, but acidification or phosphate fertilization enhances mobility, potentially elevating risks in agricultural systems near deposits.[62] In humans, dietary intake (e.g., from root vegetables or grains) contributes 1-5 μg/day, primarily excreted via urine, though chronic exposure correlates with renal proximal tubule accumulation and potential nephrotoxicity at levels above 50 μg/g kidney tissue.[63]Global Resources and Exploration
Global identified recoverable uranium resources totaled approximately 7.93 million tonnes of uranium (tU) as of January 1, 2023, according to the joint OECD Nuclear Energy Agency (NEA) and International Atomic Energy Agency (IAEA) "Red Book," encompassing reasonably assured recoverable resources (RAR) and inferred resources recoverable at extraction costs below $130 per kgU.[64] These figures represent resources sufficient to fuel projected nuclear energy expansion through 2050 under high-growth scenarios, assuming timely investments in mining and exploration, though undiscovered resources and speculative potential could extend supplies further.[65] Resource estimates are price-sensitive, with higher costs unlocking additional tonnes from lower-grade deposits or unconventional sources like seawater or phosphates, which currently exceed 20 billion tU in total but remain economically marginal.[53] The distribution of known resources is highly concentrated, with Australia holding the largest share at around 28% (approximately 1.7-3.6 million tU, varying by source classification), followed by Kazakhstan (13-15%, ~900,000-2.9 million tU), Canada (9-15%, ~600,000-1.7 million tU), Russia (8%, ~480,000-1.2 million tU), and Namibia (6-7%, ~400,000-1 million tU).[66] [53] Other significant holders include South Africa, Niger, Brazil, China, and Uzbekistan, while regions like the United States, Greenland, and Mongolia host deposits with redevelopment potential.[53] These estimates derive from geological surveys and drilling data, but discrepancies arise due to differing national reporting standards and political factors, such as restricted access in Russia or environmental moratoriums in Australia.[65] Uranium exploration worldwide has historically been underfunded relative to demand forecasts, with global expenditures peaking at $250-300 million annually in the early 2000s before declining post-Fukushima to under $100 million by 2015, though recent nuclear revival has spurred increases to around $200 million per year by 2023.[65] [67] Activity focuses on sandstone-hosted deposits in Kazakhstan and Central Asia, unconformity-related ores in Canada’s Athabasca Basin, and paleoplacer formations in South Africa, employing geophysical surveys, airborne radiometrics, and targeted drilling to delineate inferred resources.[68] Emerging frontiers include Greenland’s Kvanefjeld project, Mongolia’s Dornod deposit, and Tanzania’s Mkuju River, where joint ventures by companies like Orano and Kazatomprom aim to convert exploration results into viable reserves amid regulatory hurdles and commodity price volatility.[53] Challenges persist from legacy environmental concerns, supply chain dependencies on state-owned enterprises in producer nations, and the need for $20-30 billion in cumulative investment to avert mid-century shortfalls if reactor builds accelerate.[64]Production and Economics
Mining and Extraction Methods
Uranium mining employs three principal methods: open-pit mining, underground mining, and in-situ recovery (ISR), also known as in-situ leaching (ISL). Selection of method depends on ore depth, grade, and geological conditions, with ISR dominating modern production due to its lower operational costs and reduced surface disturbance compared to conventional techniques.[68] In 2023, global uranium production reached approximately 49,355 metric tons, with ISR accounting for over 55% of output, particularly in permeable sandstone-hosted deposits.[69][70][71] Open-pit mining is utilized for shallow deposits where ore lies near the surface, involving the stripping of overburden and extraction of ore via large-scale excavation. This method suits low- to medium-grade ores in unconformity or sandstone formations, as seen in operations in Australia and Canada, but generates significant waste rock and tailings.[72][73] Underground mining targets deeper deposits inaccessible by open-pit means, employing shafts, drifts, and stoping to access high-grade veins or tabular ores, such as uraninite in Precambrian shields; it requires substantial ventilation and support systems to manage radon and dust hazards.[73][74] ISR involves injecting a leaching solution, typically alkaline bicarbonate or acidic, into groundwater aquifers containing uranium-bearing sands, dissolving the mineral for pumping to the surface as pregnant liquor. Developed independently in the United States and Soviet Union during the late 1950s to early 1960s, ISR avoids physical ore removal and has expanded rapidly, comprising 13% of production in 1997 and rising to 46% by 2011.[71][75] Kazakhstan, producing 21,227 metric tons in 2022 (43% of global total), relies almost exclusively on ISR in roll-front deposits, followed by operations in the United States and Uzbekistan.[69][71] The process demands hydrogeological containment to prevent excursion of lixiviant beyond the ore zone, with restoration involving groundwater flushing post-extraction.[76] Following extraction, whether from conventional mining or ISR, uranium undergoes milling to concentrate it into yellowcake (U₃O₈). Ore is crushed and ground, then leached with sulfuric acid or carbonate solutions to solubilize uranium, followed by solid-liquid separation, purification via solvent extraction or ion exchange, and precipitation as ammonium or sodium diuranate, which is calcined to yellowcake containing 70-90% U₃O₈.[77][78] For ISR liquors, processing mirrors this, yielding yellowcake directly from the recovered solution without initial crushing.[71] Heap leaching, a variant for low-grade ores, stacks crushed material and percolates acid, but remains less prevalent for uranium than ISR.[78] Experimental methods under investigation include uranium extraction from seawater, which holds an estimated 4.5 billion tonnes of the element, using adsorption techniques with amidoxime-functionalized materials or electrochemical processes to selectively capture and recover uranium amid competing ions. These approaches, while currently uneconomic due to high costs exceeding $200/kg, benefit from ongoing research aimed at scalability and cost reduction for accessing dilute oceanic resources.[79][80] Biological leaching (bioleaching) using acidophilic bacteria such as Acidithiobacillus ferrooxidans has also been studied for solubilizing uranium from low-grade ores or waste dumps, offering environmental advantages through reduced chemical inputs but limited by slower kinetics and challenges in large-scale implementation compared to abiotic leaching.[81][82] Furthermore, research on engineered bacteria, such as Bacillus velezensis strains optimized for ultrafast uranium recovery from seawater or Escherichia coli with enhanced uranyl-binding proteins, seeks to improve selectivity and efficiency in dilute environments or contaminated sites.[83][84]Processing and Refining
Uranium processing from ore to yellowcake involves milling to crush and grind the ore, followed by chemical leaching to dissolve uranium minerals. In conventional agitation leaching, the ground ore is mixed with sulfuric acid, which reacts with uranium oxides to form soluble uranyl sulfate, achieving extraction efficiencies of 80-95% for low-grade ores containing 0.1-0.2% uranium.[77][85] Alkaline leaching with sodium carbonate-bicarbonate is used for ores high in carbonate content to avoid acid consumption by limestone, though it yields lower recovery rates of 70-85%.[77][86] The resulting pregnant leach solution is separated from solids via thickening and filtration, then purified to concentrate uranium and remove impurities like iron, vanadium, and molybdenum. Solvent extraction, the predominant method, employs organic extractants such as tertiary amines in kerosene to selectively transfer uranyl ions into an organic phase, followed by stripping with ammonium sulfate to produce a high-purity uranium liquor.[85][87] Ion exchange resins serve as an alternative for smaller operations, adsorbing uranium complexes before elution.[85] Precipitation occurs by adding ammonia or magnesia to form ammonium or magnesium diuranate, which is filtered, dried, and calcined at 500-600°C to produce yellowcake, a coarse powder of triuranium octoxide (U₃O₈) assaying 70-90% uranium oxide with impurities limited to 0.1% for nuclear applications.[87][88] Refining yellowcake to nuclear-grade materials entails dissolution in concentrated nitric acid to yield uranyl nitrate, followed by further solvent extraction purification to achieve impurity levels below 10 ppm for elements like boron and cadmium that affect neutron economy.[89] The purified solution undergoes thermal denitration at 300-500°C to form uranium trioxide (UO₃), which is reduced with hydrogen at 600-700°C to uranium dioxide (UO₂), suitable for direct use in some reactor fuels.[90] For gaseous diffusion or centrifugation enrichment, UO₂ is hydrofluorinated with hydrogen fluoride to uranium tetrafluoride (UF₄), then fluorinated with fluorine gas at 300-400°C to produce uranium hexafluoride (UF₆), a volatile compound sublimate at 56°C essential for isotope separation due to the slight mass difference between ²³⁵UF₆ and ²³⁸UF₆.[89][90] Alternative enrichment methods under development include laser isotope separation, which selectively excites and ionizes ²³⁵U atoms for extraction, offering potential advantages in efficiency and reduced energy use over traditional centrifugation.[30] These steps, conducted in specialized conversion facilities, ensure the uranium meets stringent specifications for fuel fabrication or weapons-grade material, with global capacity exceeding 60,000 tonnes UF₆ equivalent annually as of 2024.[89] Heap leaching and in-situ leaching (ISL) adapt processing for lower-grade deposits, spraying acid onto ore piles or injecting it underground to percolate and dissolve uranium, followed by similar purification circuits, recovering 60-80% of uranium while minimizing waste rock handling.[78][91] Tailings from processing, containing residual radioactivity, are managed in engineered impoundments to prevent environmental release, though historical sites have posed groundwater contamination risks mitigated by modern liners and reclamation.[72]Reserves, Supply Chains, and Market Dynamics
Global identified recoverable uranium resources totaled 7,934,500 tonnes as of January 1, 2023, according to reasonably assured and inferred categories reported to the OECD Nuclear Energy Agency (NEA) and International Atomic Energy Agency (IAEA); these amounts are considered sufficient to meet projected demand through 2050 under high nuclear growth scenarios, though sustained exploration and development investments are required to avoid future shortfalls.[92][93] Australia possesses the largest share of these resources, followed by Kazakhstan, Canada, Russia, and Namibia, with concentrations often in sandstone-hosted deposits amenable to in-situ leaching.[94]| Country | Approximate Share of Identified Resources (%) |
|---|---|
| Australia | 28 |
| Kazakhstan | 13 |
| Canada | 9 |
| Russia | 8 |
| Namibia | 7 |
History
Ancient and Pre-Modern Uses
Uranium-bearing minerals, such as those containing uranium oxides, were employed as pigments during the Roman era to impart yellow coloration to glass and ceramic glazes.[105] Analysis of a glass fragment from a Roman villa has revealed uranium content, indicating its use for vibrant hues as early as the 1st century AD.[106] These applications relied on the natural coloring properties of the ores without awareness of the specific element involved or its radioactive emissions.[2] This practice extended through the medieval and early modern periods, where uranium-rich minerals continued to serve as colorants in European glassmaking and pottery production.[105] Ores like pitchblende, mined in regions such as Bohemia from the 16th century onward primarily for silver extraction, yielded byproducts suitable for pigmentation.[107] Artisans valued the resulting yellow, orange, and green tones for decorative ceramics and mosaics, though quantities remained limited due to incidental sourcing rather than targeted mining for colorants.[108] No evidence exists of uranium ores being used for structural, medicinal, or other non-pigment purposes in antiquity or the pre-modern era, as their chemical and physical properties beyond coloration were unrecognized.[2] The isolation of uranium as a distinct element in 1789 by Martin Heinrich Klaproth marked the transition to more systematic applications, but pre-modern uses were confined to aesthetic enhancements in artisanal crafts.[105]19th-Century Discovery and Isolation
Uranium was discovered in 1789 by German chemist Martin Heinrich Klaproth, who isolated an oxide of the element from pitchblende ore sourced from the silver mines of Joachimsthal in Bohemia.[2] Klaproth named the new substance uranium in honor of the recently discovered planet Uranus, detected by William Herschel in 1781.[109] Although Klaproth initially believed he had obtained the pure metal by reducing the oxide with charcoal, subsequent analysis revealed that his product remained an oxide, likely UO₂, rather than elemental uranium.[109] In the early 19th century, uranium compounds found limited applications, particularly in ceramics for producing yellow glazes, but scientific interest focused on isolating the pure element to study its chemical properties. Efforts to refine isolation techniques culminated in 1841, when French chemist Eugène-Melchior Péligot successfully produced metallic uranium by heating anhydrous uranium tetrachloride (UCl₄) with potassium metal in a sealed vessel.[109] [7] Péligot's method yielded a ductile, silver-white metal that tarnished in air, and he conducted detailed studies on its atomic weight, establishing it at approximately 240 (later refined), as well as its reactivity with oxygen and acids.[110] Péligot's isolation confirmed uranium as a heavy metal with distinct chemical behavior, including the formation of multiple oxidation states, which laid groundwork for further investigations into its compounds. This achievement marked the first verifiable production of elemental uranium, distinguishing it from earlier oxide preparations and enabling precise characterization.[109] By the mid-19th century, small-scale production of uranium metal supported emerging industrial uses, though its radioactivity remained undiscovered until Henri Becquerel's work in 1896.[2]20th-Century Fission Research and Manhattan Project
In December 1938, German chemists Otto Hahn and Fritz Strassmann conducted experiments bombarding uranium with neutrons at the Kaiser Wilhelm Institute for Chemistry in Berlin, observing the production of lighter elements including barium isotopes, which indicated the splitting of the uranium nucleus.[111][112] This experimental result puzzled Hahn and Strassmann, as they expected transuranic elements rather than fission products.[113] In early 1939, Lise Meitner, who had fled Nazi Germany, and her nephew Otto Robert Frisch provided the theoretical interpretation, proposing that neutron capture by uranium-235 led to an unstable uranium-236 nucleus that deformed and split into two fragments, releasing approximately 200 million electron volts of energy per fission event, comparable to the binding energy of medium-mass nuclei.[111][114] They termed the process "nuclear fission" by analogy to biological fission and verified the energy release through calculations based on the liquid drop model of the nucleus.[115] This explanation, published in Nature on February 11, 1939, highlighted the potential for a self-sustaining chain reaction if emitted neutrons could induce further fissions.[115] Physicists Leo Szilard, Eugene Wigner, and Edward Teller, concerned about Nazi Germany exploiting fission for weaponry, drafted a letter signed by Albert Einstein on August 2, 1939, warning President Franklin D. Roosevelt of the possibility of "extremely powerful bombs of a new type" from uranium chain reactions and urging accelerated U.S. research, including securing uranium supplies from Belgium's Congo mines.[116][117] The letter prompted the formation of the Advisory Committee on Uranium, which evolved into the Manhattan Project under the U.S. Army Corps of Engineers, directed by Brigadier General Leslie Groves from September 1942, with J. Robert Oppenheimer as scientific director of the Los Alamos Laboratory for bomb design.[118][119] Early Manhattan Project efforts focused on uranium-235, the fissile isotope comprising only 0.7% of natural uranium, necessitating large-scale isotope separation to achieve weapons-grade enrichment above 90%.[119] At Oak Ridge, Tennessee, the Y-12 plant employed electromagnetic separation via calutrons, ionizing uranium tetrachloride and accelerating ions in magnetic fields to separate U-235 from U-238 based on mass differences, while the K-25 plant used gaseous diffusion of uranium hexafluoride through porous barriers, exploiting the slight velocity difference of lighter U-235 molecules.[120][121] These methods produced sufficient highly enriched uranium, though inefficiently; the project consumed vast resources, including over 14,000 pounds of silver for calutron coils.[120] A critical milestone occurred on December 2, 1942, when Enrico Fermi's team at the University of Chicago achieved the first controlled, self-sustaining nuclear chain reaction in Chicago Pile-1 (CP-1), a graphite-moderated lattice of uranium metal and oxide lumps under the west stands of Stagg Field, demonstrating neutron multiplication with a reactivity exceeding criticality by withdrawing cadmium control rods.[122][123] This experiment validated plutonium production pathways but also informed uranium-fueled reactor designs, powering subsequent pilot plants for isotope separation feed material.[124] The uranium bomb, code-named Little Boy, adopted a gun-type design assembling two subcritical masses of highly enriched uranium-235 by firing one into the other via conventional explosives, achieving supercriticality without implosion complexities.[125] On August 6, 1945, the B-29 Enola Gay dropped Little Boy over Hiroshima, Japan, detonating at 1,900 feet altitude with a yield of about 15 kilotons from the fission of roughly 0.7 kilograms of its 64-kilogram uranium core, as the design's simplicity prioritized reliability over efficiency.[126][127]Post-WWII Weapons and Reactor Development
The United States Atomic Energy Commission (AEC), established by the Atomic Energy Act of 1946, assumed control of uranium enrichment and weapons production from wartime efforts, prioritizing expansion of highly enriched uranium (HEU) output for fission primaries and depleted uranium tampers in emerging thermonuclear designs. Gaseous diffusion plants at Oak Ridge, Tennessee, were scaled up immediately post-war, with additional facilities commissioned at Paducah, Kentucky, in 1952 and Portsmouth, Ohio, in 1954, enabling annual HEU production in the tons by the mid-1950s to support growing stockpiles of gun-type and implosion devices. These advances facilitated Operation Sandstone tests in 1948, which validated composite uranium-plutonium cores, and the 1952 Ivy Mike thermonuclear test, where uranium-238 jackets enhanced neutron reflection and yield efficiency.[128] Parallel developments in the Soviet Union involved constructing the Verkh-Niz gaseous diffusion plant by late 1945 for HEU, supporting RDS-1 (a plutonium device tested in 1949) and subsequent uranium-fueled graphite-moderated reactors at Chelyabinsk-65 for fissile material production starting in 1948. The United Kingdom, leveraging Tube Alloys collaboration, initiated low-enrichment facilities at Capenhurst in the early 1950s to fuel Magnox reactors, though initial weapons-grade HEU was acquired via U.S. exchanges under the 1958 Mutual Defence Agreement. These efforts underscored uranium's centrality in early post-war arsenals, despite plutonium's rising dominance for compact implosion designs, as HEU's simplicity enabled rapid stockpiling amid escalating U.S.-Soviet tensions.[129] Shifting toward civilian applications under Eisenhower's 1953 Atoms for Peace initiative, uranium-fueled reactors emphasized enriched uranium to achieve criticality in light-water moderated systems unsuitable for natural uranium. The U.S. Materials Testing Reactor (MTR) at Idaho, operational in 1946, pioneered HEU-aluminum dispersion fuels for research, informing subsequent power prototypes. In 1951, Experimental Breeder Reactor-I (EBR-I) generated the world's first nuclear-derived electricity using enriched uranium metal fuel, demonstrating fast-spectrum fission viability.[129] The Soviet Union's Obninsk AM-1 reactor, connected to the grid in 1954, marked the first uranium-graphite power plant with low-enriched uranium, producing 5 MWe and validating controlled fission for baseload electricity. U.S. naval propulsion advanced with the USS Nautilus submarine reactor in 1954, employing compact HEU cores for high-density power, while the 1957 Shippingport Atomic Power Station debuted pressurized water reactor (PWR) technology with 93% enriched uranium oxide pins, generating 60 MWe and paving the way for commercial low-enriched uranium (LEU) fuels by the 1960s as enrichment efficiency improved. Internationally, France's 1950s Zoé reactor and Canada's NRX (1952) tested enriched uranium configurations, fostering global adoption despite proliferation risks from dual-use HEU pathways.[129]Cold War Expansion and Proliferation Challenges
Following the Soviet Union's first nuclear test in 1949, the United States and USSR initiated a massive expansion of uranium enrichment facilities to fuel their burgeoning nuclear arsenals. The US, which had produced about 2 tons of highly enriched uranium (HEU) by 1945 for the Manhattan Project, scaled up operations at Oak Ridge with gaseous diffusion plants like K-25, achieving annual production of over 10 tons of HEU by the mid-1950s to support thousands of warheads.[130] Similarly, the USSR constructed four large gaseous diffusion plants between 1949 and 1963, transitioning to gas centrifuges for efficiency, enabling rapid stockpile growth from a few bombs in 1949 to over 20,000 by the 1980s.[131] This arms race demanded enormous uranium supplies; the US alone purchased approximately 250,000 metric tons of uranium concentrate from 1942 to 1971, sourced from domestic mines and imports from Canada, the Belgian Congo, and South Africa.[132] The dual-use nature of uranium enrichment technology posed significant proliferation risks, as the same processes for weapons-grade HEU (over 90% U-235) could produce lower-enriched fuel for reactors, blurring lines between civilian and military programs. Western intelligence assessments in the 1960s warned that building covert enrichment plants for HEU was feasible for technically capable nations, heightening fears of horizontal proliferation beyond the superpowers.[133] The United Kingdom, leveraging shared US technology, conducted its first test in 1952; France developed independent capabilities, testing in 1960; and China, initially aided by Soviet designs before a 1959 rift, detonated its device in 1964, each requiring dedicated uranium mining and enrichment efforts.[134] International efforts to curb proliferation intensified with the 1953 Atoms for Peace initiative, establishing the International Atomic Energy Agency (IAEA) in 1957 for safeguards on civilian uranium use, culminating in the 1968 Nuclear Non-Proliferation Treaty (NPT), which aimed to prevent spread while allowing peaceful nuclear energy.[135] However, challenges persisted: espionage facilitated early Soviet acquisition of US enrichment secrets, and resource competition in uranium-rich regions like Africa fueled geopolitical tensions, with non-signatories and covert programs evading controls.[134] By the 1970s, the NPT's five recognized nuclear states (US, USSR, UK, France, China) held the bulk of global HEU stocks, but verification gaps and technology diffusion underscored ongoing vulnerabilities in securing fissile materials.[135]21st-Century Renaissance and Geopolitical Shifts
The early 21st century witnessed a stagnation in global nuclear expansion following the 2011 Fukushima Daiichi accident, which prompted shutdowns in Japan and Germany and heightened regulatory scrutiny worldwide, yet this gave way to a revival driven by imperatives for low-carbon energy, energy security amid fossil fuel volatility, and surging electricity demand from electrification and data centers.[136] By 2024, approximately 440 commercial nuclear reactors operated globally with a total capacity of about 400 gigawatts electrical (GWe), while 65 more were under construction, representing over 70 GWe, marking one of the highest construction pipelines since the 1990s.[136] China led this resurgence, constructing half of the world's new reactors and expanding its nuclear capacity faster than any other nation since 2000, with plans to surpass U.S. capacity by 2030 through dozens of advanced pressurized water reactors.[137] [138] This renaissance correlated with uranium market recovery: spot prices, which languished below $30 per pound from 2011 to 2020 after crashing from a 2007 peak of $136, surged past $100 per pound in 2024 for the first time in 17 years before moderating to around $60 per pound by mid-2025, fueled by supply tightness and projected demand growth from 66,000 tons annually in 2024 to 180,000 tons by mid-century.[139] [140] Geopolitically, uranium supply chains revealed vulnerabilities, with production concentrated in Kazakhstan (over 40% of global output via in-situ leaching), Canada, Australia, and Namibia, while Russia dominated enrichment at roughly 40% of world capacity through state-owned Rosatom, creating dependencies exacerbated by the 2022 Russian invasion of Ukraine.[141] [142] In response, the United States enacted the Prohibiting Russian Uranium Imports Act in May 2024, banning enriched uranium imports from Russia by August 2028 (with waivers until 2027 for non-proliferation reasons), prompting restarts of domestic mines in Wyoming, Texas, Arizona, and Utah—dormant since pre-Fukushima levels—and investments in new enrichment facilities like the planned Centrus operation in Ohio.[143] [144] European nations, facing similar constraints, accelerated diversification via Australian and Canadian contracts, though Russia's integrated fuel cycle (mining to fuel fabrication) and alliances in Central Asia, including joint ventures in Kazakhstan, sustained its leverage despite Western sanctions.[104] [142] These shifts intertwined with proliferation risks: Iran's uranium enrichment program advanced to near-weapons-grade levels by 2023 despite the 2015 Joint Comprehensive Plan of Action's partial revival, while North Korea expanded its fissile material stockpile, underscoring uranium's dual-use tensions amid civilian demand.[144] U.S. policy under President Trump in 2025 emphasized accelerating advanced reactors like small modular designs to bolster domestic fuel independence, contrasting China's state-driven buildout and Russia's export-oriented model, which together reshaped uranium as a strategic asset in great-power competition over clean energy transitions.[145] [146]Applications
Military and Defense Uses
Uranium-235, when highly enriched to levels exceeding 20% (and typically 90% or more for weapons-grade material), serves as the fissile core in nuclear fission weapons. The "Little Boy" bomb, detonated over Hiroshima on August 6, 1945, employed a gun-type design that propelled a subcritical mass of approximately 38 kilograms of uranium-235 into a stationary target of similar mass, achieving supercriticality and initiating a chain reaction.[147] This device contained about 64 kilograms of enriched uranium overall, though only around 0.7 kilograms underwent fission due to the design's inefficiency.[148] Such uranium-based fission primaries remain components in some modern thermonuclear weapons, often combined with plutonium for boosted yields, while highly enriched uranium also fuels compact reactors in naval propulsion systems for submarines and aircraft carriers.[149] Depleted uranium, consisting primarily of uranium-238 with less than 0.7% uranium-235, finds extensive military application in kinetic energy penetrators and armor due to its high density of 19.1 g/cm³, which enables superior penetration of armored targets.[150] In munitions like the 30 mm PGU-14/B rounds fired by the A-10 Thunderbolt II aircraft and the 120 mm M829 series for M1 Abrams tanks, depleted uranium projectiles self-sharpen upon impact via adiabatic shear banding, maintaining lethality against composite and reactive armors while their pyrophoric nature ignites post-penetration fires.[151] These rounds have been deployed in conflicts including the 1991 Gulf War and more recently supplied to Ukraine in 2023 for use against Russian armor.[152] Depleted uranium is also alloyed into reactive armor plating for tanks and vehicles, providing enhanced protection against shaped-charge warheads through its density and ability to disrupt incoming projectiles.[150] The U.S. military incorporates it in the M1 Abrams' armor composite, contributing to its resilience in combat scenarios.[153] While depleted uranium's radioactivity is minimal compared to its natural isotopic composition, its primary military value derives from physical properties rather than nuclear ones.[151]Civilian Energy Production
Uranium serves as the primary fuel for most civilian nuclear reactors, where enriched uranium-235 undergoes controlled fission to generate heat for electricity production. In light water reactors, which constitute the majority of operational units, natural uranium is enriched to 3-5% U-235 content to sustain a chain reaction.[154] The process begins with uranium ore mining and milling to produce uranium oxide concentrate, followed by conversion to uranium hexafluoride gas for enrichment via gaseous diffusion or centrifugation, and fabrication into ceramic pellets encased in zirconium alloy cladding for fuel assemblies.[155] Fission of U-235 nuclei releases neutrons and energy, moderated by water to slow neutrons and produce steam that drives turbines, yielding high thermal efficiency around 33% and capacity factors exceeding 90% in modern plants.[154] The first demonstration of uranium-fueled electricity generation occurred on December 20, 1951, at the Experimental Breeder Reactor-I (EBR-I) in Idaho, which powered four light bulbs using enriched uranium metal fuel.[124] The inaugural grid-connected civilian reactor, the 5 MW AM-1 Obninsk plant in the Soviet Union, began operation on June 27, 1954, marking the start of commercial nuclear power.[129] In the United States, the 60 MW Shippingport reactor achieved full-scale power production in 1957, utilizing pressurized light water technology that became dominant.[156] Subsequent decades saw rapid expansion, with boiling and pressurized water reactors proliferating due to their use of ordinary water as coolant and moderator, though alternative designs like gas-cooled and heavy-water reactors employ natural or differently enriched uranium. As of December 2023, 413 operational nuclear reactors in 31 countries provided 371.5 GW(e) capacity, generating approximately 2,650 TWh of electricity annually, equivalent to about 10% of global production.[157][158] France derives over 70% of its electricity from nuclear sources, while the U.S. fleet of 93 reactors contributes around 19% domestically.[159] A single kilogram of enriched uranium yields energy equivalent to 2,700 tons of coal, underscoring its density advantage.[136] Used fuel, containing unburned uranium and plutonium, is either stored or reprocessed in countries like France to recover fissile material, reducing waste volume by up to 96%; the remainder consists of high-level waste manageable in compact geologic repositories.[154] Empirical safety data indicate nuclear power's low mortality rate of 0.03-0.07 deaths per terawatt-hour, surpassing coal (24.6) and oil (18.4), and comparable to solar (0.02) when including lifecycle risks.[160] Major incidents like Chernobyl (1986, ~50 acute deaths, disputed long-term cancers) and Fukushima (2011, 1 direct radiation death) represent outliers, with no comparable fatalities in Western designs; overall, nuclear avoids millions of air pollution deaths annually versus fossil alternatives.[160][161] Proliferation risks from enrichment facilities persist, though IAEA safeguards mitigate dual-use concerns in civilian programs.[162] Future expansions, including small modular reactors, aim to leverage uranium's abundance—identified reserves suffice for 100+ years at current rates—while addressing supply chain dependencies on producers like Kazakhstan and Canada.[136]Depleted Uranium in Armor and Munitions
Depleted uranium (DU), consisting primarily of the isotope uranium-238 with the fissile uranium-235 content reduced to 0.2-0.3% through enrichment processes, possesses a density of 19.1 g/cm³, surpassing that of lead or tungsten alloys, which enables its application in high-performance military armor and kinetic energy penetrators.[150] DU's hardness, combined with its pyrophoric properties—igniting spontaneously upon impact due to rapid oxidation—enhances penetration effectiveness against armored targets by eroding and self-sharpening the projectile tip via adiabatic shear banding, outperforming alternatives like tungsten in real-world tests.[163][153] In munitions, DU is alloyed (typically with 0.75% titanium) to form long-rod penetrators in armor-piercing fin-stabilized discarding sabot (APFSDS) rounds, such as the U.S. 120 mm M829 series for M1 Abrams tanks and 30 mm rounds for A-10 Thunderbolt aircraft cannons.[163] These were first combat-deployed by U.S. forces during the 1991 Gulf War, with approximately 340 metric tons expended, primarily against Iraqi T-72 tanks, demonstrating superior armor defeat capabilities at ranges exceeding 2 km.[164] Subsequent uses include NATO operations in Kosovo in 1999 (about 10-15 tons) and the 2003 Iraq invasion (over 100 tons), where DU rounds provided decisive advantages in engaging Soviet-era armor.[165] Approximately 30% of a DU penetrator's mass fragments and aerosolizes into uranium oxide particles upon high-velocity impact, amplifying lethality through incendiary effects but generating respirable dust.[166] For armor, DU is incorporated as mesh layers within composite arrays in the M1A1 Heavy Armor (HA) and later Abrams variants, starting in the late 1980s, particularly in the turret frontal arc and sides, enhancing resistance to shaped-charge and kinetic threats by a factor of up to 1.5 compared to equivalent steel mass due to its ability to blunt or disrupt incoming projectiles.[167] This configuration, classified in detail but confirmed through declassified analyses, contributes to the Abrams' survivability in direct engagements, as evidenced by low penetration rates in Iraq combat data.[168] Health risks from DU in battlefield scenarios stem mainly from potential inhalation or ingestion of fine uranium particles, leading to chemical nephrotoxicity akin to soluble uranium compounds, with radiological effects secondary due to DU's low specific activity (40% of natural uranium).[150] Empirical studies, including follow-ups on Gulf War veterans with embedded DU fragments, report elevated urinary uranium levels but no clinically significant renal impairment or increased cancer incidence beyond baseline; for instance, a 20-year DoD cohort showed no excess malignancies attributable to DU.[153] Environmental assessments in Kosovo and Iraq by UNEP and WHO found soil and water contamination localized and below action levels, with no epidemiological evidence of population-level health spikes in leukemia or birth defects linked to DU sites, countering unsubstantiated claims from advocacy groups lacking causal data.[169][170] Battlefield exposures remain comparable to or lower than occupational limits for uranium workers, prioritizing kinetic advantages over marginal risks.[163]Medical, Research, and Industrial Uses
Depleted uranium, due to its high density of 19.1 g/cm³, has been employed in industrial applications such as counterweights for aircraft control surfaces, helicopter rotor blades, and ship keels, providing stability without significantly increasing volume.[63][171] It also serves as radiation shielding in containers for transporting radioactive materials and in equipment for industrial radiography, leveraging its ability to attenuate gamma rays despite limited neutron absorption.[172][171] Historically, uranium compounds like uranyl nitrate were used as colorants in ceramic glazes, producing vibrant orange-red hues in products such as Fiestaware dishes manufactured from the 1930s to the 1970s, and in glassware known as Vaseline glass, which fluoresces green under ultraviolet light due to uranyl ions.[2] In scientific research, uranium isotopes, particularly uranium-235, fuel research reactors that produce radioisotopes for applications including neutron activation analysis and materials testing, with facilities like those operated by Oak Ridge National Laboratory advancing actinide chemistry and nonproliferation studies through uranium-based experiments.[173] Highly enriched uranium (HEU) has been used in compact research reactors to generate short-lived isotopes for scientific instrumentation, though efforts since the 1990s have shifted toward low-enriched uranium (LEU) targets to minimize proliferation risks while maintaining flux levels above 10^14 neutrons per square centimeter per second.[174] Emerging research explores uranium-230, an alpha emitter with a 69.9-year half-life, for targeted alpha therapy in preclinical models, where cyclotron-produced U-230 decays to thorium-226, delivering localized radiation doses to cancer cells with energies up to 5.8 MeV.[175] Medically, depleted uranium shields gamma radiation in therapy machines, such as linear accelerators used for cancer treatment, where its density enables compact collimators to shape beams precisely and reduce exposure to healthy tissue.[176][177] HEU-powered reactors have supplied molybdenum-99 for technetium-99m generators, enabling over 40 million annual diagnostic scans worldwide via single-photon emission computed tomography, though global conversion to LEU by 2020 has reduced reliance on HEU stocks exceeding 20% U-235 enrichment.[174] Experimental protocols for U-230 in alpha therapy aim to conjugate decay products with biomolecules for prostate and neuroendocrine tumor targeting, potentially achieving tumor doses 1000 times higher than surrounding tissue due to alpha particles' 50-100 micrometer range in water.[175]Uranium Chemistry
Oxidation States and Oxides
Uranium displays oxidation states ranging from +3 to +6 in its compounds, with +4 and +6 predominating due to their relative thermodynamic stability under standard conditions.[178][29] The +6 state features the uranyl cation (UO₂²⁺), a linear dioxo species stable in aqueous solutions across a wide pH range, as evidenced by Pourbaix diagrams indicating its prevalence above approximately 0.1 V vs. standard hydrogen electrode in neutral to acidic media.[12] In contrast, the +4 state corresponds to U⁴⁺ ions, which are prone to hydrolysis and form insoluble oxides or hydroxides at neutral pH.[179] Lower states like +3 (U³⁺) are strongly reducing and oxidize rapidly in air or water, while +5 (U⁵⁺, often as UO₂⁺) is transient and disproportionates in solution.[178] Rare molecular complexes have demonstrated +2 and even +1 states under inert conditions, but these lack practical stability outside specialized organometallic environments.[180][181] The principal uranium oxides reflect these states: uranium(IV) oxide (UO₂), uranium trioxide (UO₃), and triuranium octoxide (U₃O₈). UO₂ adopts a fluorite crystal structure (face-centered cubic) with uranium coordinated to eight oxygen atoms, exhibiting a melting point of 2865 °C and semiconducting properties due to oxygen vacancies that enable mixed U(IV)/U(V) valence under oxidation.[182] This oxide occurs naturally in uraninite and serves as the primary fuel form in nuclear reactors, sintered into pellets with densities up to 10.96 g/cm³.[183] UO₃, in the +6 state, exists in multiple polymorphs (alpha, beta, gamma), with the alpha form featuring layered sheets of uranyl units linked by van der Waals forces; it decomposes above 600 °C and is hygroscopic, forming uranyl hydroxide in moist air.[184] U₃O₈, a mixed-valence oxide (two U⁴⁺ and one U⁶⁺ per formula unit), has an orthorhombic structure with layered uranium-oxygen polyhedra, appearing as a dark olive-green to black powder; it forms as an intermediate during UO₂ oxidation and is the primary component of "yellowcake" concentrate after calcination at 500–800 °C.[185] These oxides interconvert via controlled oxidation or reduction: for instance, UO₂ oxidizes stepwise to U₄O₉, U₃O₇, and ultimately U₃O₈ or UO₃ under increasing oxygen partial pressure at elevated temperatures, with phase transitions tracked by X-ray diffraction showing lattice expansion from fluorite to defect structures.[186] Stoichiometric deviations, such as hyperstoichiometric UO_{2+x}, introduce oxygen interstitials that enhance reactivity but lower sinterability in fuel fabrication.[187] Thermodynamic data indicate UO₂'s stability up to 10^{-20} atm O₂ at 1000 K, underscoring its resistance to autoignition compared to finely divided forms.[188] In aqueous environments, oxide solubility is pH-dependent, with UO₃-derived uranyl species dominating acidic leaching processes in ore processing, governed by equilibrium constants for hydrolysis (e.g., log K for UO₂²⁺ + H₂O ⇌ UO₂OH⁺ + H⁺ ≈ -5.0).[179]Inorganic Compounds and Reactions
Uranium forms a range of inorganic halides, primarily in oxidation states +3 to +6, with fluorides being the most stable and industrially significant due to their volatility and use in nuclear fuel processing.[189] Uranium hexafluoride (UF6), a colorless, reactive gas sublimate at 56.5 °C, serves as the key intermediate for uranium enrichment via gaseous diffusion or centrifugation, reacting exothermically with moisture to form uranyl fluoride (UO2F2) and hydrogen fluoride: UF6 + 2H2O → UO2F2 + 4HF.[190] Lower fluorides like uranium tetrafluoride (UF4), a green solid, are produced by reduction of UF6 with hydrogen at 600–800 °C and serve as precursors for metal production via the Kroll process: 2UF4 + Ca → 2UF3 + CaF2, followed by further reduction.[191] Chlorides such as uranium tetrachloride (UCl4), a dark green solid, and uranium trichloride (UCl3) exhibit reducing properties and undergo ammonolysis or metathesis reactions to form other compounds; for instance, UCl4 reacts with ethyl acetate to yield self-ionized complexes like [UCl3(EtOAc)3]+[UCl5(EtOAc)]-.[192] Mixed chloride fluorides, such as UClnF6-n (n=1–5), form via low-temperature reactions of UF6 with HCl, decomposing above –60 °C to release Cl2 and yield lower-valent uranium halides.[193] Bromides and iodides (e.g., UBr4, UI4) are less stable, prone to disproportionation, and synthesized from metal or oxides with HBr or HI, but they hydrolyze rapidly in aqueous media to form oxyhalides.[194] Other non-halide inorganic compounds include uranium carbide (UC and U2C3), produced by arc-melting uranium with carbon, which reacts with water to generate methane and hydrogen via hydrolysis: UC + 4H2O → UO2 + CH4 + 2H2.[189] Uranium phosphide (UP) and nitride (UN) form under high-temperature reactions with phosphorus or nitrogen, showing semiconductor properties but limited reactivity data beyond oxidation to oxides.[189] Cyanides like U(CN)64– emerge from halide-cyanide metathesis in liquid ammonia, stable under anhydrous conditions but hydrolyzing in protic solvents.[195] Aqueous reactions highlight uranium's hydrolysis propensity: U4+ hydrolyzes stepwise in acidic media (e.g., log β1 ≈ –0.5 for UOH3+ in perchlorate), forming polymers and precipitates like UO2·xH2O at pH >1, while UO22+ yields linear complexes like (UO2)3(OH)5+ dominating at pH 3–5.[196] In acidic brines, U4+ remains soluble under reducing conditions but precipitates as UO2 upon oxidation.[197] These behaviors underpin geochemical mobility and waste management, with reactions often controlled by pH and redox potential as depicted in Pourbaix diagrams showing UO22+ stability in oxidizing acidic waters.[189]Coordination and Organometallic Chemistry
Uranium displays versatile coordination chemistry across oxidation states +3 to +6, with +4 and +6 being most prevalent, enabling coordination numbers ranging from 4 to 14 due to its large ionic radii (e.g., 0.89 Å for U^{4+} and 0.83 Å for UO_2^{2+}) and involvement of 5f orbitals in bonding.[198] In U(VI) complexes, the uranyl ion (UO_2^{2+}) features a linear O=U=O core with equatorial coordination typically 4–6, yielding total coordination numbers of 6–8 and geometries such as hexagonal bipyramidal or pentagonal bipyramidal.[199] U(IV) supports higher coordination, exemplified by U(BH_4)_4 with 14 coordination sites in a distorted capped hexagonal antiprism geometry, where each bidentate BH_4^- ligand bridges via hydrogen atoms.[200] Lower oxidation states like U(III) favor arene coordination, forming stable bis(arene) complexes with elongated U–X bonds due to the ionic radius of 1.03 Å.[201] Common ligands include halides, oxides, nitrogen donors (e.g., bipy, phen), and carboxylates, with uranium adapting geometries like octahedral for U(IV) halides or distorted prismatic for multidentate N-ligands.[202] Thermodynamic stability of uranyl bio-coordination with oxygen/nitrogen donors follows log K trends increasing with denticity, as seen in speciation studies.[203] Vibrational spectroscopy (Raman/IR) identifies U–O stretches around 900–950 cm^{-1} for uranyl, aiding speciation in solids and solutions.[204] Organometallic uranium compounds feature σ-bonded alkyls (e.g., U(CH_2Ph)_4) and π-complexes, with U(IV) σ-organometallics showing reactivity toward CO_2 insertion and oxidative additions.[205] Uranocene (U(C_8H_8)_2), synthesized in 1968, represents the first actinide sandwich compound, adopting a parallel η^8 geometry akin to ferrocene but with larger U–C distances (2.64–2.71 Å) due to 5f involvement.[206] Low-valent species, such as U(III) alkyls or carbene complexes, enable catalysis in hydroelementation and small-molecule activation, leveraging redox flexibility absent in d-block metals.[207] Homoleptic polyalkyl U(IV) complexes exhibit β-hydride elimination, contrasting with thorium analogs, highlighting uranium's unique reductive potential.[208] Advances include multigram-scale synthesis of U(IV) organometallics for exploring U=C double bonds and carbynes.[209]Health Effects
Radiotoxicity from Alpha Emission
Uranium isotopes ^{238}U (half-life 4.468 billion years) and ^{235}U (half-life 704 million years) primarily decay by emitting alpha particles with energies of 4.27 MeV and 4.40 MeV, respectively, followed by lower-energy alphas from daughter nuclides like ^{234}U in secular equilibrium. These alpha particles, consisting of helium-4 nuclei, exhibit high linear energy transfer (LET) values of approximately 100 keV/μm in tissue, resulting in densely ionizing tracks that produce clustered DNA damage, including irreparable double-strand breaks and oxidative stress, far exceeding the effects of sparsely ionizing beta or gamma radiation.[210][211] Externally, alpha particles from uranium pose no significant hazard, as their range is limited to about 40-50 μm in soft tissue and they are fully stopped by the outer layer of dead skin cells or even a sheet of paper. Internal exposure, however, occurs via inhalation of respirable uranium particulates (e.g., oxides or metal dust <10 μm aerodynamic diameter) generated during mining, milling, or combustion of depleted uranium munitions, or less efficiently through ingestion with gastrointestinal absorption fractions of 0.2-2% for most compounds. Once internalized, localized alpha emissions irradiate proximate cells, inducing cytotoxicity, inflammation via pro-inflammatory cytokines, and potential mutagenesis, with risks concentrated in the lungs for insoluble forms that clear slowly (biological half-time >100 days).[212][213][214] The radiotoxicity is constrained by uranium's low specific activity—approximately 25 kBq/kg (0.0007 μCi/g) for natural uranium—yielding few decay events per atom over human timescales, such that even gram quantities internalized deliver committed radiation doses orders of magnitude below those from high-activity alpha emitters like ^{210}Po. International Commission on Radiological Protection (ICRP) dose coefficients for occupational inhalation of slowly dissolving uranium dioxide (Type S) estimate 2-5 × 10^{-5} Sv per Bq inhaled, primarily to lungs and bone surfaces, with effective doses from 1 mg chronic inhalation around 0.1-1 mSv/year, comparable to natural background but additive to chemical effects. Animal experiments demonstrate lung fibrosis and neoplasia at cumulative alpha doses >5 Gy, yet human studies of uranium workers show no consistent excess cancers attributable solely to uranium alphas, as opposed to radon progeny or chemical kidney damage.[215][216][213] In kidneys, where uranium concentrates (up to 50% of systemic burden), alpha emissions contribute to proximal tubule necrosis alongside chemical mechanisms, but modeling indicates radiation doses rarely exceed 1 Gy even in high-exposure scenarios, insufficient for deterministic effects and with stochastic risks <1% lifetime cancer increase per 100 mSv equivalent. For depleted uranium (^{235}U <0.3%), radiotoxicity is ~40% lower than natural uranium due to reduced activity, emphasizing that while alpha emission causally drives localized genotoxicity, empirical data from Gulf War veterans and mill workers reveal no elevated radiogenic disease incidence beyond baseline, underscoring the dominance of solubility-driven biokinetics over pure radiological burden.[217][215][213]Chemical Toxicity Independent of Radiation
Uranium acts as a heavy metal toxin, with its primary non-radiological effects manifesting as nephrotoxicity, damaging the proximal tubules of the kidneys through mechanisms including oxidative stress, mitochondrial dysfunction, and disruption of electron transport chains.[218][219] This toxicity arises from uranium's affinity for phosphate groups in cellular proteins and enzymes, inhibiting key renal functions such as reabsorption and secretion.[220] Acute exposure to soluble uranium compounds can induce tubular necrosis, proteinuria, and elevated blood urea nitrogen, with animal models demonstrating renal failure at doses as low as 0.1–1 mg/kg body weight via intravenous administration.[213][218] The degree of toxicity varies significantly by compound solubility: highly soluble forms like uranyl nitrate (UO₂(NO₃)₂) and uranyl fluoride (UO₂F₂) exhibit greater systemic absorption and renal targeting due to rapid entry into the bloodstream, whereas insoluble oxides (e.g., UO₂, U₃O₈) primarily cause localized pulmonary retention if inhaled, with slower dissolution leading to protracted but lower-intensity kidney exposure.[221][222] Ingested soluble uranium is absorbed at 0.1–6% efficiency in the gastrointestinal tract, concentrating in kidneys where it reaches levels up to 50% of the body burden, far exceeding other organs.[213] Human epidemiological data from occupational exposures, such as uranium processing workers, correlate urine uranium concentrations above 10–30 μg/g creatinine with subtle glomerular filtration rate declines, though overt failure requires higher acute doses.[217][223] Beyond kidneys, chemical effects include potential neurotoxicity and reproductive impacts at elevated exposures, but these are less pronounced and dose-dependent; for instance, rodent studies show testicular degeneration at chronic oral doses exceeding 20 mg/kg/day, attributed to heavy metal interference rather than radiation.[213] Occupational safety limits, such as the American Conference of Governmental Industrial Hygienists' threshold limit value of 0.2 mg/m³ for soluble uranium, reflect chemical risks dominating over radiological ones for depleted uranium scenarios.[215] Empirical evidence from depleted uranium handling indicates that chemical thresholds for kidney impairment occur at lower exposures than those for significant radiotoxicity, underscoring the metal's inherent toxicity profile.[215][217]Exposure Pathways and Dose Assessments
Uranium exposure in humans occurs primarily through three pathways: inhalation of airborne particles, ingestion via contaminated food or water, and dermal contact, though the latter contributes minimally due to low skin absorption rates of less than 1% for most compounds.[63] Inhalation predominates in occupational settings such as uranium mining and milling, where respirable dust or aerosols from processes like ore crushing can deposit insoluble uranium oxides in the lungs, leading to prolonged retention with absorption fractions ranging from 0.5% to 5% depending on particle solubility.[224] [222] Ingestion represents the main route for the general population, particularly in regions with elevated natural uranium in groundwater, where gastrointestinal absorption is typically low at 0.2-2% for adults but higher (up to 5%) in children or with soluble forms like uranyl nitrate.[63] [222] Dose assessments for uranium incorporate both radiological and chemical components, employing biokinetic models to estimate committed effective doses from intake quantities measured in becquerels (Bq) or milligrams. Internal dosimetry relies on monitoring techniques such as urinary uranium bioassay, which correlates excretion rates (typically 0.02-2% of intake daily for soluble compounds) with systemic uptake using International Commission on Radiological Protection (ICRP) models like those in Publication 78, accounting for age-specific deposition in target organs like the kidneys where uranium accumulates preferentially.[225] [226] For inhalation exposures, lung deposition models classify compounds by solubility (Types F, M, S for fast, medium, slow), with slow-dissolving particles yielding higher committed doses due to extended pulmonary retention half-times exceeding 100 days.[227] Empirical validation from worker studies, such as those involving depleted uranium, shows effective doses rarely exceeding 1-10 mSv per year for chronic low-level exposures, far below thresholds for observable radiogenic effects, with chemical nephrotoxicity emerging at urinary uranium levels above 30 μg/g creatinine.[213] [228] Bayesian methods enhance uncertainty quantification in dose reconstruction by integrating prior biokinetic data with measured bioassay results, particularly useful for historical exposures where variability in absorption (e.g., 1-20% for inhalation of mixed aerosols) complicates deterministic calculations.[226] Population-level assessments, such as those for communities near uranium facilities, use probabilistic modeling to derive lifetime cancer risks, estimating that natural background intakes yield doses of 0.01-0.1 mSv annually, comparable to or lower than radon exposure from soil gas.[229] These evaluations prioritize kidneys for dose-limiting due to uranium's affinity, with total effective doses summed across pathways per regulatory limits like 20 mSv/year for workers under 10 CFR 20.[230]Environmental and Safety Considerations
Mining Impacts and Remediation
Uranium mining generates large volumes of radioactive tailings and waste rock containing uranium decay products such as radium-226 and radon-222, which can contaminate soil, surface water, and groundwater if not properly managed.[72] These wastes pose risks through radon gas emanation, leading to elevated airborne radiation levels, and leaching of heavy metals and radionuclides into aquifers, as observed in historical operations in the U.S. Grants Mineral Belt where mill tailings affected shallow groundwater quality.[231] Open-pit and underground methods exacerbate dust dispersion and erosion, while in-situ leaching introduces acids that mobilize contaminants, though modern regulations require containment liners and monitoring to limit off-site migration.[232] Worker health impacts primarily stem from inhalation of radon progeny and uranium ore dust, with epidemiological studies of U.S. Colorado Plateau miners showing a linear dose-response for lung cancer mortality, where cumulative exposures exceeding 100 working level months (WLM) increased risk by factors of 5-10 compared to unexposed populations.[233] Historical data from 1950-1960s operations indicate excess lung cancer rates among Navajo and other miners due to inadequate ventilation, though post-1970s federal standards capping radon at 4 WLM/year have reduced incidences in regulated sites.[234] Non-radiological effects include chemical nephrotoxicity from soluble uranium species, but radiation dominates long-term morbidity, with no conclusive links to other cancers beyond lung in most cohorts.[235] Remediation focuses on stabilizing tailings through covers of clay, rock, or geomembranes to suppress radon flux by 80-95% and prevent erosion, as implemented in IAEA-guided long-term strategies emphasizing minimal maintenance designs.[236] In the U.S., the Uranium Mill Tailings Radiation Control Act of 1978 has overseen relocation or capping of over 5,000 acres of tailings at 24 sites, reducing public exposure doses by orders of magnitude.[237] For abandoned mines on the Navajo Nation, EPA-led cleanups address legacy contamination, such as a $600 million settlement in 2017 for remediating 94 sites involving waste removal and groundwater treatment to below 30 μg/L uranium standards.[238] Costs vary widely, with individual site interventions ranging from $13 million for small-scale features to over $60 million for comprehensive operations including adit sealing and water diversion.[239] Emerging techniques like bioremediation and phytoremediation show promise for uranium uptake but remain supplementary to physical containment due to scalability limits.[240]Fuel Cycle Waste Management
The nuclear fuel cycle generates radioactive and chemical wastes across its stages, from uranium extraction to spent fuel handling, but these are produced in relatively small volumes compared to the energy output and are managed through containment, treatment, and isolation strategies to minimize environmental release. Mining and milling produce the largest waste volumes in the form of tailings—finely ground ore residues containing low levels of uranium daughters like radium and thorium—which are impounded in engineered facilities, covered with soil or clay barriers, and monitored for radon emanation and groundwater infiltration. In the United States, under Nuclear Regulatory Commission oversight, tailings piles are stabilized in situ or relocated to designated sites, with remediation efforts at legacy sites like those under the Uranium Mill Tailings Remediation Action Project having addressed over 5,000 acres by 2020 through evaporation controls and vegetation covers to reduce erosion and radiation exposure.[241][72] Front-end processes, including conversion to uranium hexafluoride (UF6) and enrichment, yield depleted uranium tails (primarily U-238) stored as UF6 cylinders or converted to stable oxides, alongside smaller quantities of low-level waste (LLW) like contaminated solvents and filters, which are compacted, incinerated, or solidified for shallow land burial after verifying activity below regulatory limits. Reactor operations produce operational LLW (e.g., resins, tools) and intermediate-level waste (ILW) from decontamination, typically dewatered and grouted in steel drums for interim storage. The primary back-end concern is high-level waste (HLW), mainly spent nuclear fuel assemblies containing unburned uranium, plutonium, and fission products, discharged at rates of about 2,000 metric tons annually in the U.S. from reactors generating roughly 800 billion kWh yearly—equating to under 30 grams of HLW per capita if scaled to full national electricity supply.[242][243] Globally, cumulative spent fuel generation since 1954 totals around 400,000 tonnes, with one-third reprocessed to extract recyclable materials.[244] Spent fuel is initially cooled in on-site pools for 5–10 years to dissipate decay heat, then transferred to dry cask storage systems—sealed concrete or steel modules with inert gas atmospheres—certified by the U.S. Nuclear Regulatory Commission for at least 60 years of safe containment under seismic and environmental stresses, with no radiological releases recorded from licensed facilities. Long-term disposal targets deep geological repositories, such as Finland's Onkalo facility, operational since 2025 at 400–450 meters in crystalline bedrock, designed to isolate waste for millennia via multiple barriers including copper canisters and bentonite clay buffers. Reprocessing, practiced in France and Japan, separates uranium (96% of spent fuel mass) and plutonium for reuse, reducing HLW volume by a factor of five and long-term radiotoxicity by ten through vitrification of fission products into stable glass logs, though proliferation risks have limited its adoption in proliferation-sensitive contexts like the U.S.[245][246][247] Overall, fuel cycle waste management prioritizes engineered isolation over dilution, with empirical data showing containment effectiveness far exceeding unmanaged alternatives like fossil fuel combustion residues, where annual U.S. coal ash alone exceeds 100 million tons with dispersed radionuclides.[248]Comparative Risks to Alternative Energy Sources
Lifecycle analyses of energy sources typically measure risks in terms of fatalities per terawatt-hour (TWh) of electricity produced, encompassing occupational accidents, major disasters, and air pollution-related deaths attributable to emissions.[160] This metric accounts for the full fuel cycle, from extraction to waste management, providing a standardized basis for comparison. Empirical data indicate that nuclear power, reliant on uranium fuel, exhibits one of the lowest death rates among major sources, at approximately 0.03 deaths per TWh, comparable to modern renewables like wind (0.04 deaths per TWh) and solar (0.02 deaths per TWh for rooftop installations).[160] In contrast, fossil fuels impose far higher tolls, with coal at 24.6 deaths per TWh—primarily from particulate matter, sulfur dioxide, and nitrogen oxide emissions causing respiratory diseases and premature mortality—and oil at 18.4 deaths per TWh.[160] Natural gas fares better at 2.8 deaths per TWh but remains elevated due to methane leaks and combustion byproducts.[160]| Energy Source | Deaths per TWh (accidents + air pollution) |
|---|---|
| Coal | 24.6 |
| Oil | 18.4 |
| Natural Gas | 2.8 |
| Hydro | 1.3 |
| Wind | 0.04 |
| Solar (rooftop) | 0.02 |
| Nuclear | 0.03 |
Controversies and Empirical Realities
Proliferation Risks versus Deterrence Benefits
Uranium enrichment to produce highly enriched uranium (HEU) exceeding 90% U-235 serves as a primary pathway for nuclear weapons proliferation, enabling states to acquire fissile material for bombs under the guise of civilian nuclear programs.[250] This dual-use nature of enrichment technology, involving centrifuges or gaseous diffusion, has facilitated proliferation in cases such as Pakistan's covert program, which produced HEU for its arsenal by the 1980s, and North Korea's operations yielding weapons-grade material since the 2000s.[134] Risks include breakout scenarios where safeguarded facilities expel inspectors to rapidly produce bomb fuel, as assessed in proliferation models, and theft of material by non-state actors, given HEU's compact form suitable for improvised devices.[251] Such pathways heighten global instability, with empirical instances like Iran's undeclared enrichment activities prompting sanctions and near-threshold capabilities by 2023.[30] Counterbalancing these risks, nuclear arsenals derived from uranium-based fissile materials have underpinned deterrence strategies that empirically correlate with reduced interstate conflict among major powers since 1945. No full-scale war has occurred between nuclear-armed states, despite proxy conflicts and crises like the Cuban Missile Crisis in 1962, where mutual assured destruction (MAD) logic prevented escalation.[252] Stockpiles peaking at over 70,000 warheads globally by the 1980s enforced strategic stability, contributing to a "long peace" with battle deaths from state conflicts declining by over 90% compared to pre-1945 eras.[253] Deterrence extends beyond superpowers; India's 1974 test and subsequent arsenal deterred Pakistani invasions post-1998 tests, stabilizing South Asia despite conventional skirmishes.[254] Critics argue proliferation risks outweigh benefits, citing potential for accidental use or irrational actors eroding deterrence reliability, yet historical data shows no nuclear exchange amid nine states possessing weapons by 2025.[255] While enrichment safeguards under the NPT have limited overt spread since 1970, with only four additional nuclear states emerging, the causal link between nuclear possession and restraint holds in crises, as evidenced by restrained responses in Kargil (1999) and Russo-Ukrainian tensions.[256] Proliferation controls, including IAEA monitoring, mitigate uranium pathway threats without negating deterrence's pacifying effect, as non-proliferation efforts coexist with arsenal maintenance in stable powers.[134] Overall, the absence of great-power conflict substantiates deterrence's empirical efficacy over proliferation fears, though vigilance against dual-use abuses remains essential.[257]Accident Analyses and Safety Statistics
Nuclear power plants, which rely on uranium as fuel, have demonstrated one of the lowest rates of accidental fatalities among energy sources, with empirical estimates placing the death rate at approximately 0.04 deaths per terawatt-hour (TWh) when accounting for major accidents like Chernobyl.[160] This figure contrasts sharply with coal's 24.6 to 161 deaths per TWh (including air pollution and accidents) and oil's 18.4 deaths per TWh, highlighting uranium-fueled nuclear's superior safety profile based on lifecycle data spanning decades of operation.[160] [258] These statistics derive from comprehensive analyses incorporating direct fatalities, radiation-induced cancers, and occupational risks, underscoring that while rare severe events occur, their aggregated impact remains minimal compared to fossil fuels' routine emissions-related harms.[259] The 1979 Three Mile Island accident in Pennsylvania involved a partial core meltdown in a uranium-fueled pressurized water reactor due to equipment failure and operator errors, releasing minimal radioactive gases but causing no immediate deaths or detectable long-term health effects among the public or workers.[260] Epidemiological studies, including those tracking cancer incidence near the site, found no statistically significant increases attributable to radiation exposure, with off-site doses averaging under 1 millisievert—far below natural background levels. [261] This event prompted enhanced safety protocols, such as improved operator training and redundant cooling systems, without evidence of elevated fatalities. Chernobyl's 1986 explosion in a graphite-moderated uranium reactor resulted in 28 acute radiation syndrome deaths among firefighters and workers, plus 19 subsequent worker fatalities linked to the incident, with long-term cancer estimates varying widely but conservatively pegged at around 4,000 excess cases among exposed liquidators and residents by United Nations assessments.[262] Empirical data indicate no widespread spike in thyroid cancers beyond treated cases in children, and overall mortality patterns align more closely with socioeconomic factors than radiation alone, challenging higher projections from some models that assume linear no-threshold extrapolations from high-dose exposures.[263] The accident's severity stemmed from design flaws in Soviet RBMK reactors, absent in Western uranium-fueled light-water designs, which incorporate containment structures limiting releases. The 2011 Fukushima Daiichi meltdowns, triggered by a tsunami overwhelming uranium-fueled boiling water reactors, produced no direct radiation fatalities among workers or the public, with one 2018 case of worker lung cancer officially attributed but debated as causally linked given low doses.[264] [265] UNSCEAR evaluations confirm radiation exposures below thresholds for observable cancer increases, with over 2,000 evacuation-related deaths exceeding any potential radiation harm due to disrupted medical access and stress.[266] Post-accident fortifications, including sea walls and flexible grid designs, have mitigated similar risks globally. Uranium mining and processing accidents, historically tied to radon inhalation causing lung cancers in early 20th-century operations, show improved safety in modern regulated practices, with standardized mortality ratios near unity (SMR 1.05) for recent cohorts excluding legacy exposures.[267] Open-pit methods prevalent today minimize underground radon risks, and ventilation standards have reduced occupational hazards.[268] Criticality accidents during uranium enrichment or handling—unintended chain reactions from fissile mass accumulation—have occurred rarely, with 21 of 22 historical process incidents involving enriched uranium solutions, resulting in two fatalities at Tokaimura in 1999 from improper mixing.[269] These events, confined to facilities with geometric controls and neutron absorbers, underscore procedural safeguards' effectiveness, as no such accidents have impacted commercial power generation.[270]| Energy Source | Deaths per TWh (Accidents + Air Pollution) |
|---|---|
| Nuclear | 0.04 [160] |
| Coal | 24.6–161 [160] [258] |
| Oil | 18.4 [259] |
| Hydro | 1.3 [160] |
Waste Myths Debunked with Lifecycle Data
A prevalent misconception portrays nuclear waste as generating insurmountable volumes relative to energy produced, yet lifecycle assessments reveal that spent nuclear fuel constitutes a minuscule fraction of total waste from electricity generation. Worldwide, approximately 400,000 tonnes of used nuclear fuel have been discharged from reactors since commercial operations began, with about one-third reprocessed, equating to roughly 27 tonnes per TWh of electricity generated over decades of operation.[244] In contrast, coal-fired power plants produce orders of magnitude more solid waste; a typical 1 GW coal plant generates 100,000 to 200,000 tonnes of ash annually, much of which contains trace radioactive elements like uranium and thorium that are released into the environment without containment, exceeding nuclear waste volumes by factors of thousands per equivalent energy output.[272] High-level waste, often the focus of alarm, comprises only about 3% of total nuclear-generated radioactive waste volume, with the remaining 97% classified as low- or intermediate-level, which decays rapidly and requires far less stringent management.[244] Empirical radiotoxicity profiles demonstrate that the initial intense radioactivity from fission products like strontium-90 and cesium-137, with half-lives of approximately 30 years, diminishes by over 90% within 300-500 years, transitioning the primary long-term concern to actinides whose collective hazard integrates to equivalence with the original uranium ore radiotoxicity after about 10,000 years.[273][274] This finite decay trajectory, verifiable through isotopic analysis, underscores that nuclear waste's hazard is temporally bounded, unlike persistent chemical pollutants from fossil fuels that lack natural attenuation. Lifecycle comparisons further dispel notions of nuclear waste as uniquely burdensome by quantifying containment efficacy: all nuclear high-level waste is securely stored in engineered facilities, preventing environmental dispersal, whereas coal ash—more radioactive per unit mass due to concentrated natural radionuclides—often leaches into waterways, contributing to measurable population doses exceeding those from tightly regulated nuclear operations.[272][244] Data from operational histories, including over 70 years of global reactor experience, confirm no instances of public health impacts from properly managed spent fuel storage, affirming that waste volumes and risks are empirically low when contextualized against alternatives.[246]| Energy Source | Approx. Waste Volume per TWh | Key Characteristics |
|---|---|---|
| Nuclear (spent fuel) | ~27 tonnes | Fully contained; decays to low hazard in millennia[244] |
| Coal (ash) | ~40,000-80,000 tonnes | Partially dispersed; contains natural radionuclides, higher routine releases[272] |



![World uranium production (mines) and demand[79]](http://upload.wikimedia.org/wikipedia/commons/thumb/7/75/U_production-demand.png/500px-U_production-demand.png)

![Uranium production 2015, in tonnes[83]](http://upload.wikimedia.org/wikipedia/commons/thumb/9/9a/Uranium_production%2C_OWID.svg/500px-Uranium_production%2C_OWID.svg.png)

